Abstract
Background:
Accrual to cancer clinical trials is sub-optimal. Few data exist regarding whether financial reimbursement might increase accruals.
Objectives:
Assess perceptions about reimbursement to overcome barriers to trial accrual
Research Design:
Cross-sectional survey
Subjects:
Oncologists identified from the American Medical Association Physician Masterfile
Measures:
We report descriptive statistics, associations of physician characteristics with perceptions of reimbursement, domains and sub-themes of free-text comments.
Results:
Respondents (n=1,030) were mostly medical oncologists (59.4%), ages 35–54 (67%), and male (75%). 30% reported discussing trials with >25% of patients. Barriers perceived were administrative/regulatory, physician/staff time, and eligibility criteria. National Cancer Institute cooperative group participants and practice owners were more likely to endorse higher reimbursement. Respondents indicated targeted reimbursement would help improve infrastructure, but also noted potential ethical problems with reimbursement for discussion (40.7%) and accrual (85.9%). Free-text comments addressed reimbursement sources, recipients, and concerns about real and apparent conflict of interest.
Conclusions:
Though concerns about potential conflict of interest remain paramount and must be addressed in any new system of reimbursement, oncologists believe reimbursement to enhance infrastructure could help overcome barriers to trial accrual.
Keywords: clinical trial, reimbursement, oncology
Introduction
Accrual to cancer clinical trials is sub-optimal across the U.S., and there is lack of clarity about the extent to which potential strategies, such as financial reimbursement of oncologists or practices, may increase accrual and diminish disparities.1,2 Although national payment reform initiatives such as the Center for Medicare & Medicaid Services’ Oncology Care Model address episodes of oncology care, these efforts underestimate resources needed to foster best practices for clinical trial accrual, particularly in community settings.3–10 A recent American Society of Clinical Oncology (ASCO) survey reported that reimbursement for trials, especially National Cancer Institute (NCI)-funded National Clinical Trials Network (NCTN) trials, is low and has not increased in the past decade.11 To date, there has been little information about amounts of reimbursement oncologists would view as appropriate or sufficient to overcome barriers to trial discussion and accrual or about how they would use reimbursement if provided.12–15
To address this knowledge gap, we conducted a national survey of oncologists from academic and community practices to assess a range of factors influencing recruitment to clinical trials. Here, we describe level of reimbursement they perceive as necessary to overcome barriers, how they would use more available resources or reimbursement to enhance accrual, and whether they see enhanced reimbursement as a real or potential conflict of interest.
Methods
Formative research and survey methodology
We conducted semi-structured interviews with national stakeholders to understand the process of discussing trials with patients and accruing them to studies.16 We then developed a survey that included questions about characteristics of oncologists and their practices and barriers to trial accrual. Details of the survey sampling frame and response rate are reported elsewhere 17.
Stakeholder interviews showed that oncologists think of reimbursement in terms of patient visits. Therefore, we used “new patient visit” as a point of reference for response options to items asking what level of reimbursement (for discussing a trial or enrolling a patient) would make a difference. Response options were: less than, equivalent to, two times, or three times whatever reimbursement they received for a new patient visit, e.g. CMS Medicare Level N.18 We used a 5-point Likert scale to solicit level of agreement with five potential uses for those funds (Table 3), as well as whether reimbursement for discussing versus accruing a patient to a trial would present ethical problems or conflicts of interest. Finally, an open-end question invited respondents to voice any concerns regarding reimbursement for trial activities.
Table 3.
Multivariate analysis – Correlates of reimbursement (two or three times a new patient visit) for discussing a trial and accruing a patient
| Discussing a trial (n=940)* |
Accruing a patient (n=947)* |
|||||
|---|---|---|---|---|---|---|
| aOR | 95% CI | p-value | aOR | 95% CI | p-value | |
| Physician characteristics | ||||||
| Sex | ||||||
| Female | 1.00 | |||||
| Male | 1.45 | 1.05–2.01 | 0.02 | |||
| Employment status | ||||||
| Employed by hospital | 1.00 | 1.00 | ||||
| Employed by medical group | 0.96 | 0.63–1.46 | 0.53 | 0.90 | 0.62–1.32 | 0.93 |
| Practice owner or partner | 1.43 | 0.98–2.10 | 0.01 | 1.80 | 1.25–2.60 | <0.01 |
| Other | 0.40 | 0.18–0.89 | 0.01 | 0.38 | 0.19–0.78 | 0.01 |
| Currently in academic practice | 0.99 | 0.72–1.37 | 0.94 | |||
| Ever in academic practice (current or past 5 years) | ||||||
| Number of physicians in practice | 1.00 | 1.00 | ||||
| 1–10 | 1.18 | 0.83–1.68 | 0.09 | 1.04 | 0.75–1.44 | 0.36 |
| 11–50 | 0.79 | 0.50–1.33 | 0.12 | 0.82 | 0.55–1.21 | 0.22 |
| >50 | 1.05 | 0.76–1.46 | 0.76 | |||
| Study or site PI of trial (past 5 years) | 0.68 | 0.45–1.00 | 0.04 | 1.40 | 0.98–2.00 | 0.07 |
| Participate in NCI-NCTN | ||||||
| Physician experience with trials | ||||||
| Discussed risks and benefits of trial (past 5 years) | ||||||
| Less than 25% | 1.00 | |||||
| 25% or more | 1.15 | 0.73–1.79 | 0.55 | |||
| Recommend a trial (past 5 years) | ||||||
| Less than 25% | 1.00 | |||||
| 25% or more | 1.20 | 0.72–2.01 | 0.48 | |||
| Likely to recommend a trial | ||||||
| Very unlikely, unlikely or neither | 1.00 | |||||
| Likely | 1.18 | 0.74–1.90 | 0.97 | 1.18 | 0.77–1.79 | 0.79 |
| Very Likely | 1.42 | 0.88–2.29 | 0.15 | 1.51 | 0.99–2.31 | 0.05 |
| Consenting and enrolling patients | ||||||
| Oncologist | 1.00 | |||||
| All others | 0.80 | 0.59–1.09 | 0.16 | |||
| Physician and practice barriers to trial enrollment | ||||||
| Physician time and effort | 1.17 | 0.97–1.41 | 0.11 | 1.13 | 0.95–1.33 | 0.17 |
| Staff time and effort | 1.09 | 0.89–1.33 | 0.43 | 1.22 | 1.02–1.46 | 0.03 |
| Access to archival tissue or specialized testing determine eligibility | 1.05 | 0.87–1.26 | 0.60 | 1.07 | 0.91–1.27 | 0.40 |
| Patient history determine eligibility | 1.06 | 0.87–1.29 | 0.55 | 1.06 | 0.89–1.27 | 0.51 |
| Informed consent process | 1.26 | 1.05–1.51 | 0.01 | 1.03 | 0.87–1.21 | 0.75 |
| Number of study-related procedures | 0.94 | 0.77–1.16 | 0.57 | 0.97 | 0.81–1.16 | 0.73 |
| Lack of software or electronic tools | 0.95 | 0.79–1.13 | 0.54 | 1.04 | 0.89–1.22 | 0.63 |
| Administrative and regulatory burden | 1.20 | 0.98–1.45 | 0.08 | 1.12 | 0.95–1.34 | 0.19 |
Variables with p>0.2 in univariate model and not included in multivariable analysis have empty cells.
Analyses
We used logistic regression to assess associations of perceptions about reimbursement (≤1 vs. 2–3 times a new patient visit) with oncologist characteristics, practice characteristics, and perceived barriers to trial accrual. Variables with P<0.2 were then entered into a multivariable logistic regression model.
Using standard approaches to thematic coding, a qualitative methodologist (SCL) reviewed all free-text responses to generate a provisional codebook of four major domains and sub-themes. Four authors (SCL, RN, CCM, CSS) worked independently by theme to double-code responses, resolving discrepancies by consensus.
Results
Respondent demographics
Characteristics of the study population are shown in Table 1. About 75% represented practices participating in NCI-NCTN, and more than half reported having been a study or site principal investigators in the most recent 5 years). Most respondents reported having discussed and recommended trials for fewer than a quarter of their patients. Perceived barriers to trial enrollment are available, see Data (Supplemental Digital Content 1).
Table 1.
Characteristics of oncologists by levels of reimbursement considered appropriate for discussing a trial and accruing a patient (n=1,030)1
| Discussing a trial | Accruing a patient | |||
|---|---|---|---|---|
| Reimbursement less than or equal to a new patient visit n = 744 n (%) |
Reimbursement two or three times a new patient visit n = 268 n (%) |
Reimbursement less than or equal to a new patient visit n = 615 n (%) |
Reimbursement two or three times a new patient visit n = 398 n (%) |
|
| Age at survey | ||||
| 25–34 | 22 (3.0) | 16 (6.0) | 18 (2.9) | 21 (5.2) |
| 35–44 | 315 (42.3) | 90 (33.6) | 261 (42.5) | 144 (36.2) |
| 45–54 | 185 (24.9) | 93 (34.7) | 157 (25.5) | 122 (30.7) |
| 55–64 | 152 (20.4) | 48 (17.9) | 118 (19.2) | 80 (20.1) |
| ≥65 | 70 (9.4) | 21 (7.8) | 61 (9.9) | 31 (7.8) |
| Sex | ||||
| Male | 545 (73.3) | 203 (75.8) | 442 (71.9) | 311 (78.1) |
| Female | 199 (26.7) | 65 (24.2) | 173 (28.1) | 87 (21.9) |
| Specialty | ||||
| Medical oncology | 435 (58.5) | 170 (63.4) | 371 (60.3) | 236 (59.3) |
| Radiation oncology | 271 (36.4) | 86 (32.1) | 212 (34.5) | 145 (36.4) |
| Surgical oncologyy | 31 (4.2) | 11 (4.1) | 25 (4.1) | 16 (4.0) |
| Other2 | 7 (0.9) | 1 (0.4) | 7 (1.1) | 1 (0.3) |
| Time in practice (years) | ||||
| ≤4 | 142 (19.1) | 47 (17.5) | 116 (18.9) | 73 (18.3) |
| 5–9 | 186 (25.0) | 49 (18.3) | 156 (25.3) | 80 (20.1) |
| 10–14 | 118 (15.9) | 53 (19.8) | 99 (16.1) | 72 (18.1) |
| ≥15 | 298 (40.0) | 119 (44.4) | 244 (39.7) | 173 (43.5) |
| Employment status | ||||
| Employed by hospital | 341 (45.8) | 124 (46.4) | 289 (47.0) | 179 (45.1) |
| Employed by medical group | 158 (21.3) | 51 (19.1) | 140 (22.8) | 70 (17.6) |
| Practice owner or partner | 192 (25.8) | 84 (31.5) | 139 (22.6) | 137 (34.5) |
| Other | 53 (7.1) | 8 (3.0) | 47 (7.6) | 11 (2.8) |
| Academic practice | ||||
| No | 423 (57.0) | 160 (59.9) | 343 (56.0) | 240 (60.5) |
| Yes | 319 (43.0) | 107 (40.1) | 270 (44.0) | 157 (39.5) |
| Number of physicians in practice | ||||
| 1–10 | 360 (48.4) | 120 (45.3) | 288 (46.8) | 190 (48.1) |
| 11–50 | 232 (31.2) | 102 (38.5) | 198 (32.2) | 140 (35.4) |
| >50 | 152 (20.4) | 43 (16.2) | 129 (21.0) | 65 (16.5) |
| Participate in regional or national network | ||||
| No | 607 (81.9) | 222 (83.5) | 500 (81.7) | 329 (83.1) |
| Yes | 134 (18.1) | 44 (16.5) | 112 (18.3) | 67 (16.9) |
| Participate in NCI-NCTN | ||||
| No | 219 (29.7) | 57 (21.6) | 181 (29.7) | 95 (24.2) |
| Yes | 518 (70.3) | 207 (78.4) | 428 (70.3) | 298 (75.8) |
| Number of patients on active therapy | ||||
| 1–50 | 40 (5.4) | 5 (1.9) | 35 (5.7) | 10 (2.5) |
| 51–100 | 86 (11.6) | 29 (10.9) | 73 (11.9) | 41 (10.4) |
| 101–200 | 218 (29.4) | 71 (26.6) | 171 (27.9) | 116 (29.3) |
| >200 | 397 (53.6) | 162 (60.7) | 334 (54.5) | 229 (57.8) |
| Study or site PI of trial (past 5 years) | ||||
| No | 315 (42.5) | 104 (39.0) | 266 (43.3) | 151 (38.2) |
| Yes | 426 (57.5) | 163 (61.0) | 348 (56.7) | 244 (61.8) |
| Discussed risks and benefits of trial (past 5 years) | ||||
| No patients | 32 (4.3) | 3 (1.1) | 24 (3.9) | 12 (3.0) |
| <10% of patients | 225 (30.2) | 84 (31.3) | 184 (29.9) | 125 (31.4) |
| 10–24% of patients | 273 (36.7) | 91 (34.0) | 224 (36.4) | 138 (34.7) |
| 25–50% of patients | 120 (16.1) | 51 (19.0) | 102 (16.6) | 71 (17.8) |
| 50% or more of patients | 94 (12.7) | 39 (14.6) | 81 (13.2) | 52 (13.1) |
| Recommend a trial (past 5 years) | ||||
| No patients | 32 (4.3) | 1 (0.4) | 24 (3.9) | 9 (2.2) |
| <10% of patients | 301 (40.6) | 111 (41.6) | 247 (40.3) | 165 (41.6) |
| 10–24% of patients | 288 (38.8) | 97 (36.3) | 236 (38.5) | 146 (36.8) |
| 25–50% of patients | 84 (11.3) | 39 (14.6) | 71 (11.6) | 56 (14.1) |
| 50% or more of patients | 37 (5.0) | 19 (7.1) | 35 (5.7) | 21 (5.3) |
Of the 1,072 respondents, 42 did not complete the survey. Analysis reflects 1,030 completers.
Other reflects self-report write-in as: breast surgery, hematology, hospice/palliative care, hospitalist, internal medicine, single practice, infectious disease.
Perceptions about reimbursement
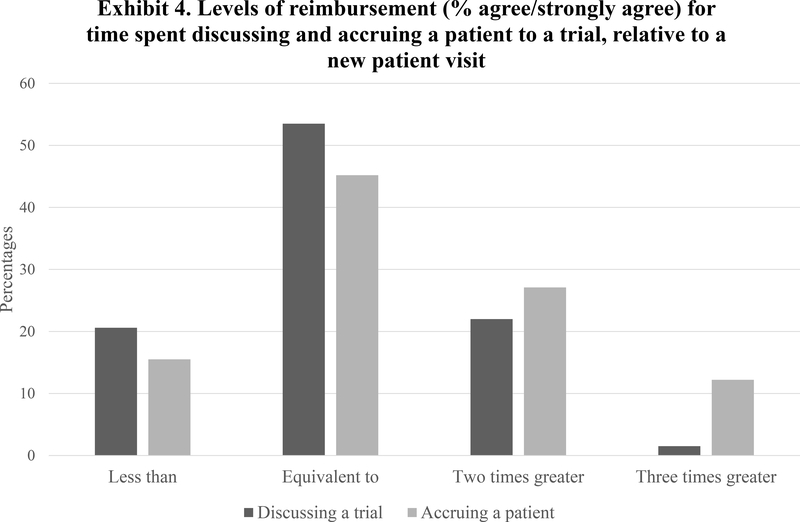
Figure 1 shows level of reimbursement perceived as adequate for discussing and accruing to clinical trials. Fewer than a quarter of respondents felt that reimbursement equivalent to less than a new patient visit would be adequate for discussing or enrolling to clinical trials. Approximately half of respondents considered reimbursement equivalent to a new patient visit would be adequate, while more than 10% felt that reimbursement for enrolling a patient to a clinical trial should be at least three times that of a new patient visit.
Figure 1.
shows the distribution of the levels of reimbursement (% reporting agree/strongly agree) for time spent discussing (in dark gray) and accruing (in light gray) a patient to a cancer clinical trial, relative to a new patient visit.
Most respondents agreed or strongly agreed that reimbursement could be used for purposes ranging from investment in electronic tools or software to hiring staff to manage trial activities (Table 2). Slightly fewer than half felt that reimbursement would increase capacity to monitor trial activity for accrual.
Table 2.
Activities supported by reimbursement for discussing (n=1,102) and accrual to a trial (n=1,013) (% agree/strongly agree)
| Discussing (%) | Accruing (%) | |
|---|---|---|
| Hire staff to manage trial activities | 88.9 | 87.8 |
| Enable staff to do tasks more efficiently | 82.2 | 81.4 |
| Increase capacity to monitor trial activity | 82.1 | 48 |
| Enable you to do tasks more efficiently | 80.5 | 76.7 |
| Allow investment in software or electronic tools to match patients to trials | 73.6 | 77.4 |
| Present ethical problems or conflicts of interest | 40.7 | 85.9 |
Correlates of perceptions about reimbursement
Multivariable logistic regression (Table 3) demonstrated that perceiving a need for greater reimbursement (2 or 3 times a new patient visit) for discussing a trial was associated with NCI-NCTN participation, working in a private practice, and barriers related to clinical trial consent process. For trial accrual, preference for greater reimbursement was more likely among men, those working in a private practice, and those reporting barriers related to staff time.
Of the 1,030 respondents, 431 (41.8%) submitted free-text responses to the question, Are there any other concerns regarding reimbursement for trial activities? Thematic coding of these responses generated the domains, sub-themes, and sample comments (see Data, Supplemental Digital Content 2, which lists free-text domains and themes). Respondents volunteered cogent rationales (or justifications) for trial-related reimbursement. They expressed opinions about how funds should be allocated, to whom, and for what use. However, few suggested potential sources of such funding. Some respondents equated reimbursement in terms of work Relative Value Units (wRVUs). Several raised pragmatic considerations about the effects of trial-related reimbursement, notably variations on selection bias, both with respect to which trials might be opened and which patients might be approached. Others raised questions about the burden of monitoring patients on trials, as opposed to discussing trial options or actually enrolling a patient on a protocol.
The majority of respondents who provided a free-text response explicitly addressed potential concerns regarding the ethics of trial-related reimbursement. Although some only noted reimbursement “would” or “might pose a conflict of interest”, without further explanation, many expounded on the nature of the potential conflict. Recognizing the survey’s distinction between reimbursement for discussing vs. enrolling to trials, they noted the conflict of interest would lie not with discussing, but with enrollment, by creating inappropriate incentives. Several expressed concerns about how patients or the public would perceive such reimbursement; some insisted on patient disclosures in advance.
Discussion
Substantive policy discussions have been underway in recent years on the financial implications of cancer care, 19 especially in the era of precision medicine 20 and other innovations 21. However, little attention has been directed to the operational costs to providers seeking to identify clinical trial options for their patients with cancer. Under the Affordable Care Act (ACA), while coverage for participants in approved clinical trials allowed for routine patient costs, these explicitly excluded data collection and analysis needs and, further, did not address initial provider activities to assess appropriateness of trials for individual patients.22 Importantly, policies to date restrict health plan discrimination in coverage but fall short of addressing public payer provisions that could support such related billable costs.
To our knowledge, this is the first study to assess in a national sample how oncologists perceive levels of reimbursement that might overcome barriers to clinical trial accrual. Our study assessed the amount or reimbursement considered adequate, how it would be used, and whether it would pose an actual or perceived conflict of interest. It is worth noting that three-quarters of respondents participated in clinical trial cooperative groups, and more than half had been a study or site principal investigator for a trial in the last five years. Thus, our sample represents the intended target population for national policies to increase accrual of patients in trials.
In terms of an amount that would make a difference for accrual, about half of respondents suggested the equivalent to reimbursement for a new patient clinic visit. However, a substantial minority indicated the need for even greater reimbursement for accrual, with one quarter indicating twice that of a new patient visit and more than 10% percent suggesting three times that amount. Notably, perceived need for greater reimbursement was associated with participation in NCI-NCTN, perhaps because current compensation for NCI-sponsored trials is generally lower than pharma-sponsored studies.11 Being a practice owner or partner was also associated with perceiving higher level of reimbursement for both discussing and accruing activities. Discussing a trial emphasizes individual oncologist time and effort whereas trial accrual may be perceived to impact other practice resources (e.g., staff time). This may explain differences in correlates of discussing a trial compared to accruing patients (Table 3).
Overall, respondents indicated that reimbursed funds would help to improve infrastructure such as electronic tools or software, or pay for additional staff to manage trial activities and overcome time and procedural barriers, including time required to complete the informed consent process after discussing trial options. Free-text responses expressed concerns about the costs of trial participant follow-up, which may exceed the up-front reimbursement provided at the time of accrual and thus create financial instability.23 But it is unclear, then, why fewer respondents thought funds for accrual to a trial would increase capacity to monitor trial activity. Should policy makers pursue reimbursement for trial discussion and accrual, future research will be needed to identify optimal mechanisms and deploy such changes.
Many respondents noted that reimbursement might present a real or perceived conflict of interest. Explanatory free-text comments (supplied by more than 40%) mentioned specific concerns about to whom the funds would flow, from what source they would come, for which activities they would be applied, and whether reimbursement would somehow affect selection of or influence over patients. Some suggested that tying reimbursement to trial discussion rather than accrual might mitigate such an apparent conflict of interest.
Limitations & Strengths
While the absolute number of participants in this national survey was large, we recognize the overall response rate was low – 8.1% compared to 18% from the 2018 practice-level survey conducted by ASCO.24
A study strength is the diversity of the sample in specialty, age, practice type, and previous experience with trials. In fact, we had higher response rates from surgical and radiation oncologists, who have been either under-represented 25,26 or not represented at all 27,28 in previous surveys. We were also able to assess specific barriers to trials and to explore perceptions about both level and potential use of funds if reimbursement for discussing and accruing to trials were increased.
Conclusion
Taken together, the open- and close-ended survey responses indicate strong desire for and expected benefit from funds to enhance infrastructure to support cancer clinical trials. Concerns about potential conflict of interest remain paramount among oncologists and must be addressed in any new system of reimbursement, particularly as value-based care initiatives move oncology toward bundled payment models. Issues to be addressed include how funds should be allocated, to whom, and for what purposes.
Supplementary Material
Acknowledgments
Grant support: Financial support for this work was provided, in part, by grants from the National Cancer Institute to Dr. Skinner and Lee (U54 CA163308-05S1); and Dr. Gerber (K24CA201543); with additional support to the Simmons Comprehensive Cancer Center (P30CA142543); and from the National Center for Advancing Translational Sciences via the UT Southwestern Center for Translational Medicine to Dr. Murphy (KL2TR001103).
Footnotes
Disclosures: The authors declare no conflicts of interest or financial disclosures.
References
- 1.Doroshow JH, Croyle RT, Niederhuber JE . Five Strategies for Accelerating the War on Cancer in an Era of Budget Deficits. Oncologist. 2009;14:110–116. doi: 10.1634/theoncologist.2009-0008 [DOI] [PubMed] [Google Scholar]
- 2.Schilsky RL. Publicly Funded Clinical Trials and the Future of Cancer Care. Oncologist. 2013;18:232–238. doi: 10.1634/theoncologist.2012-0423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mcclellan BMB, Thoumi AI. Special Series : Quality Care Symposium Original Contribution Oncology Payment Reform to Achieve Real Health Care Reform. J Oncol Pract. 2015;11(3):223–230. Walden library databases. [DOI] [PubMed] [Google Scholar]
- 4.Clough JD, Kamal AH. Oncology Care Model: Short- and Long-Term Considerations in the Context of Broader Payment Reform. J Oncol Pract. 2015;11(4):319–321. doi: 10.1200/jop.2015.005777 [DOI] [PubMed] [Google Scholar]
- 5.Apte SM, Patel K. Payment Reform: Unprecedented and Evolving Impact on Gynecologic Oncology. Front Oncol. 2016;6(April):1–10. doi: 10.3389/fonc.2016.00084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reinke T Cms Takes the Lead in Oncology Payment Reform. Manag Care. 2015;24:22–25. [PubMed] [Google Scholar]
- 7.Patel K, Thoumi A, Nadel J, O’Shea J, McClellan M. Transforming Oncology Care: Payment and Delivery Reform for Person-Centered Care. Am J Manag Care. 2015;21:388–393. [PubMed] [Google Scholar]
- 8.Centers for Medicare and Medicaid Services. Oncology Care Model (CMS, Baltimore MD, 2016). https://innovation.cms.gov/initiatives/oncology-care/. [Google Scholar]
- 9.Centers for Medicare and Medicaid Services. OCM Performance-Based Payment Methodology No. 1.1. Department of Health & Human Services; Baltimore MD. https://innovation.cms.gov/Files/x/ocm-methodology.pdf. [Google Scholar]
- 10.Abernethy A, Allen Lapointe N, Wheeler J, Irvine R, Patwardhan M, Matchar D. Ahrq Technology Assessments. In: Horizon Scan: To What Extent Do Changes in Third-Party Payment Affect Clinical Trials and the Evidence Base?. Agency for Healthcare Research and Quality (US); 2009. [PubMed] [Google Scholar]
- 11.Baer AR, Kelly CA, Bruinooge SS, Runowicz CD, Blayney DW. Challenges to National Cancer Institute–Supported Cooperative Group Clinical Trial Participation: An ASCO Survey of Cooperative Group Sites. J Oncol Pract. 2010;6(3):114–117. doi: 10.1200/jop.200028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Farrar WB. Clinical trials. Cancer. 1991;67:11779–11782. [DOI] [PubMed] [Google Scholar]
- 13.Dilts DM, Sandler AB. Invisible barriers to clinical trials: The impact of structural, infrastructural, and procedural barriers to opening oncology clinical trials. J Clin Oncol. 2006;24(28):4545–4552. doi: 10.1200/JCO.2005.05.0104 [DOI] [PubMed] [Google Scholar]
- 14.Minasian LM, O’Mara AM. Accrual to clinical trials: Let’s look at the physicians. J Natl Cancer Inst. 2011;103(5):357–358. doi: 10.1093/jnci/djr018 [DOI] [PubMed] [Google Scholar]
- 15.Palackdharry CS, Evans DL, McDonough RS, et al. Payer coverage for patients enrolled onto clinical trials: Making the process easy and transparent for everyone. Am J Manag Care. 2012;18(SPECIAL ISSUE):41–46. [PubMed] [Google Scholar]
- 16.Lee SJC, Murphy CC, Geiger AM, et al. Conceptual Model for Accrual to Cancer Clinical Trials. J Clin Oncol. 2019:JCO.19.00101. doi: 10.1200/JCO.19.00101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Murphy CC, Lee, Simon J. Craddock Geiger AM, Cox JV., et al. A randomized trial of mail vs. e-mail invitations for a physician survey on clinical trial accrual. BMC Med Res Methodol. 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Centers for Medicare and Medicaid Services. New and Established Patient Publication 100–04, Chapter 12, Section 30.6.7, Medicare Claims Processing Manual.; 2016. https://www.cms.gov/files/document/medicare-claims-processing-manual-chapter-12.
- 19.Callahan R, Darzi A. Analysis & commentary: Five policy levers to meet the value challenge in cancer care. Health Aff. 2015;34(9):1563–1568. doi: 10.1377/hlthaff.2015.0308 [DOI] [PubMed] [Google Scholar]
- 20.Pregelj L, Hwang TJ, Darrow JJ, et al. Precision medicines have faster approvals based on fewer and smaller trials than other medicines. Health Aff. 2018;37(5):724–731. doi: 10.1377/hlthaff.2017.1580 [DOI] [PubMed] [Google Scholar]
- 21.Miller KL, Lanthier M. Trends in orphan new molecular entities, 1983–2014: Half were first in class, and rare cancers were the most frequent target. Health Aff. 2016;35(3):464–470. doi: 10.1377/hlthaff.2015.0921 [DOI] [PubMed] [Google Scholar]
- 22.42 U.S.C. 300gg-8 - Coverage for individuals participating in approved clinical trials. https://www.govinfo.gov/app/details/USCODE-2010-title42/USCODE-2010-title42-chap6A-subchapXXV-partA-subpart1-sec300gg-8/summary. Published 2011.
- 23.Seow HY, Whelan P, Levine MN, et al. Funding oncology clinical trials: Are cooperative group trials sustainable? J Clin Oncol. 2012;30(13):1456–1461. doi: 10.1200/JCO.2011.37.2698 [DOI] [PubMed] [Google Scholar]
- 24.Kirkwood MK, Hanley A, Bruinooge SS, et al. The State of Oncology Practice in America, 2018: Results of the ASCO Practice Census Survey. J Oncol Pract. 2018;14(7):JOP.18.00149. doi: 10.1200/JOP.18.00149 [DOI] [PubMed] [Google Scholar]
- 25.Blanch-Hartigan D, Forsythe LP, Alfano CM, et al. Provision and discussion of survivorship care plans among cancer survivors: Results of a nationally representative survey of oncologists and primary care physicians. J Clin Oncol. 2014;32(15):1578–1585. doi: 10.1200/JCO.2013.51.7540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pirl WF, Muriel A, Hwang V, et al. Screening for psychosocial distress: ANational Survey of Oncologists. J Support Oncol. 2007;5:499–504. [PubMed] [Google Scholar]
- 27.Mori M, Shimizu C, Ogawa A, Okusaka T, Yoshida S, Morita T. A National Survey to Systematically Identify Factors Associated With Oncologists’ Attitudes Toward End-of-Life Discussions: What Determines Timing of End-of-Life Discussions? Oncologist. 2015;20(11):1304–1311. doi: 10.1634/theoncologist.2015-0147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Neumann PJ, Palmer JA, Nadler E, Fang C, Ubel P. Cancer therapy costs influence treatment: A national survey of oncologists. Health Aff. 2010;29(1):196–202. doi: 10.1377/hlthaff.2009.0077 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.