Abstract
Aim:
We sought to elucidate whether stress, peers, and in vivo (i.e., direct) alcohol cues elicit alcohol craving in daily life among adolescents and to test whether individual variation in working memory function and biological sex alters these associations.
Methods:
We leveraged ecological momentary assessment (EMA) to examine momentary associations between stress, peers, and direct alcohol cues with craving, assessed as “urge to drink alcohol,” among 86 male (51.2%) and female (48.8%) frequent drinkers (i.e., two or more drinking days per week). Participants were ages 14 to 24 years (M = 20.7 years, SD = 2.1). Participants completed EMA throughout the day for about one week prior to randomization to a treatment condition for an AUD clinical trial. Pre-registered, secondary analyses focused on craving for assessments when adolescents were not drinking, and assessments occurring after drinking on drinking days were removed. Working memory performance was assessed in the laboratory via the Memory for Words subtest of the Woodcock-Johnson III Tests of Cognitive Abilities.
Results:
Craving was heightened at more stressful moments and when adolescents were with their peers and in the presence of direct alcohol cues. Working memory function was not related to craving but altered the relation of momentary stress, peers, and cues with craving once biological sex-related differences were considered. Females generally had lower craving than males, but working memory function served to buffer against stress-induced craving for males.
Conclusions:
Higher working memory function buffered the in-the-moment relation of stress with alcohol craving for males but not females.
Keywords: Stress, ecological momentary assessment, alcohol, adolescents, craving
1. Introduction
Vulnerability to alcohol use disorder (AUD) is greater for adolescents who drink heavily, potentially via neural adaptations promoting susceptibility to stress and cue-induced alcohol craving (Breese et al., 2011; Volkow et al., 2016). The relevance of craving, defined as the subjective experience of wanting alcohol or drugs (Tiffany & Wray, 2012), is underscored by its role as a diagnostic criterion for substance use disorder (DSM-5, 2013; ICD-10, 1992), primacy as a target for pharmacological and behavioral treatments (Haass-Koffler et al., 2014; Litten et al., 2016; Witkiewitz et al., 2019), and longstanding centrality to addiction theory and research (Brown et al., 2008; Drummond, 2001). A recent systematic review of 18 theoretical frameworks identified stress as an essential parameter for examining craving in real-world settings and working memory (WM) as a potentially supported parameter (van Lier et al., 2018). Alcohol craving in response to in vivo (i.e., direct) alcohol cues appears to be an important factor contributing to adolescent alcohol misuse and the development of AUD (Ramirez and Miranda, 2014; Thomas et al., 2005; Treloar Padovano and Miranda, 2020). Less research focuses on adolescents’ craving in other situations, such as when around their peers (Treloar Padovano et al., 2019) or under conditions of stress (Blumenthal et al., 2021). The present study assesses adolescent’s alcohol craving in real life situations including when in the presence of direct alcohol cues, when around peers, and at times of heightened stress. Potential moderating influences of individual differences in WM and biological sex are examined.
Adolescence is well-defined by social, behavioral, and cognitive changes that extend into the mid-twenties (Spear, 2013; Steinberg, 2008). Among these changes are proneness to stress response and increased substance-taking behavior, which is reflected in the rapid escalation of problem drinking patterns (Johnston et al., 2018). For over a century, theorists and researchers have sought to understand this period of “storm and stress” that is adolescence (Hall, 1916, p. 73). It is now generally agreed that while adolescents, on average, are more emotionally volatile and reckless than children or adults, there are important individual differences among adolescents that buffer against these tendencies (Arnett, 1999). Deficient WM, a core facet of neurocognitive function, is associated with stress reactivity and implicated in leading models of addiction as an individual difference that may promote substance misuse (Tiffany, 1999; Volkow et al., 2016). If stress and drinking are, as suggested, “a toxic combination for the teenage brain” (Goldstein et al., 2016), then understanding how WM deficits may exacerbate the link between stress and alcohol craving is a high priority.
Associations of stress, WM, and alcohol craving may differ for adolescent males and females. There is extensive evidence for behavioral differences in risk trajectories for alcohol misuse among adolescent males and females, and the emotional brain circuitry involved in stress processing may be a mechanism contributing to differences (Hardee et al., 2017). Research suggests that WM may develop earlier in females but is also more susceptible to alcohol-related damage (Verplaetse et al., 2020). At the same time, alcohol-cued craving is heightened with escalating drinking patterns and more so for males than females (Verplaetse et al., 2020). In a prospective study of neural markers of alcohol cue reactivity, males developed greater cue reactivity after drinking onset than females, even when both males and females were drinking at the same moderate-to-heavy rate (Nguyen-Louie et al., 2018). Adolescent males who drink heavily also show greater frontal brain activation during a WM task than same-sex controls, where females who drink heavily showed less activation (Squeglia et al., 2011). Other work shows that the frontal and limbic processing regions of adolescent female brains may be more reactive to negative emotional stimuli than those of their male counterparts (Hardee et al., 2017). Given that sex-related differences in drinking patterns and problems emerge during the transition from early to late adolescence, understanding differential vulnerabilities to alcohol misuse among adolescent male and female problem drinkers is of paramount importance (Schulte et al., 2009).
The present secondary analysis seeks to elucidate whether adolescents with diminished capacity for cognitive control, as demonstrated by poor performance on a laboratory task assessing WM, may be especially prone to exhibit alcohol craving under certain conditions in daily life. Current advances in ecological momentary assessment (EMA) technology allow for real-time monitoring of craving in natural environments via smartphones (van Lier et al., 2018). We hypothesize that stress will be positively associated with alcohol craving (assessed as “urge to drink alcohol”) reported on smartphones in adolescents’ daily lives, after accounting for other contextual influences (i.e., time of day, day of week), and this association will be more pronounced among youth with lower WM performance. Our hypotheses are pre-registered here: osf.io/c3n6s. Given biological sex-related differences in WM development and neural effects of alcohol (Verplaetse et al., 2020), we also explore whether WM deficits operate uniquely for adolescent males and females.
2. Materials and Methods
2.1. Participants
Participants were 14- to 24-year-old adolescents (n = 86) recruited from the community for a clinical trial testing the efficacy of a medication to reduce alcohol consumption (NCT01641445). The age criterion capitalized on the peak developmental window for AUD (Johnston et al., 2018) and allowed the parent trial to test the efficacy of a medication to treat AUD in adolescents. All data reported herein were from an initial EMA monitoring period lasting approximately 1 week and occurring prior to randomization for the parent trial. To be eligible for the parent trial, adolescents were required to drink alcohol at least two days per week in the past 30 days and read English. Because the parent trial tested a medication, females were excluded if they were pregnant, nursing, or unwilling to use a reliable method of birth control. Other exclusions included receiving alcohol treatment in the past 30 days, seeking formal alcohol treatment, and living with someone who was actively participating in the study. Active psychotic symptoms and suicidality were also exclusionary. The Brown University Institutional Review Board approved all study procedures.
2.2. Procedures
Interested volunteers completed a telephone screening; those who appeared eligible were scheduled for an in-person screening. Consent was obtained from those 18 years of age and older; parent permission and minor assent were obtained for those younger than 18 years. At the initial study visit, self-report and interview assessments were administered and the EMA protocol was explained to participants. EMA training included a “demo” feature that allowed participants to practice completing different report types with the help of study staff.
Participants were provided with Samsung smartphones operating custom EMA software. All other phone features were disabled. Aspects of our protocol were designed to maximize EMA compliance, including the ability to suspend device-initiated prompts for up to 7 hours to accommodate times when responding was restricted or unsafe (e.g., at sports practice, driving a car), an alarm function that suspended prompts while participants were sleeping and delivered an audible alarm in the morning at the participant’s chosen waking time, and a delay feature. To approximate a true “momentary” assessment of focal variables, however, responses could only be delayed twice, for 10 minutes as a time, resulting in a maximum delay of 20 minutes. Once a device-initiated prompt was delivered, participants had 2 minutes to either initiate or delay the assessment before it was marked as missed. The number of missed prompts each day was displayed on the main menu of the smartphone screen in bold, red text. Participants were compensated up to $5 per day for completing assessments. Compliance was tracked with a custom web application and checked by research staff near daily.
A combination of smartphone- and user-initiated prompts captured momentary feelings (e.g., stress, craving), situations (e.g., location, social context), and specific events (e.g., alcohol consumption). Smartphone-initiated audible prompts occurred throughout the day and notified participants to complete reports. These reports were delivered once randomly in each 3-hour block and are referred to herein as “random assessments.” Random assessments did not overlap with “sleep” mode or drinking episodes. Additionally, upon waking participants completed a morning report that assessed alcohol use the prior day (e.g., total standard drinks consumed). Participants were also instructed to self-initiate a drink report before and after each standard drink they consumed. Participants were trained to discern standard drinks using a graphic manual depicting common beverages and volumes. To simplify the instructional set, participants were instructed to self-initiate drink reports for every drink consumed, but the assessment battery only captured the first three standard drinks of a drinking episode.
2.3. Person-level Measures
2.3.1. Demographic and clinical characteristics.
Participants were asked to report demographic characteristics, including biological sex (male = 0; female = 1) and age. AUD symptom count was determined with the Kiddie Schedule for Affective Disorders for School-Age Children, a semi-structured interview based on Diagnostic and Statistical Manual of Mental Disorders IV, Text Revision (DSM-IV-TR) criteria (Kaufman et al., 1997). Interviewers received systematic training and achieved a high level of inter-rater reliability (kappa > 0.90). A symptom count combining abuse and dependence criteria provided a unidimensional measure of AUD symptomatology. The maximum possible AUD symptom count was 11.
2.3.2. Alcohol use.
Assessment of alcohol use prior to the study determined eligibilitly and described the participants’ typical drinking patterns. A Timeline Follow-Back (TLFB) interview (Sobell and Sobell, 1992) assessed the total number of standard drinks per day in the 90 days prior to this study. Percent drinking days and drinks per drinking day were calculated to describe drinking frequency and quantity, respecitvely.
2.3.3. Working memory performance.
The Memory for Words subtest of the Woodcock Johnson®-III Tests of Cognitive Abilities (Schrank, 2005) captured WM performance. WM most commonly describes processes involved in holding and manipulating information in immediate awareness for a short period of time. In the Memory for Words subtest, an examiner reads a list of sentences, and the examinee is asked to repeat the list of sentences in the same order as presented. According to the dominant Baddeley model of WM (Baddeley, 2003), the Memory for Words subtest measures storage and rehearsal components of the phonological loop, which is the most empirically supported of the WM components in this theoretical model (Leffard et al., 2006). Trained staff administered the test in accordance with standard administration instructions. Testing occurred in the afternoon whenever possible to control for any possible effects of diurnal variation on neuropsychological test performance, and all participants reported abstinence from drugs and alcohol on the day of testing, and a breathalyzer test indicated 0.00 g/210L breath alcohol content.
2.4. Momentary Measures
2.4.1. Alcohol craving.
Momentary craving was assessed with a single-item measure of “urge to drink” on an 11-point visual analog scale from “no urge” (0) to “strongest ever” (10). In EMA training, participants received a graphic manual with sample EMA items and a glossary of terms that defined “urge” as “how much you crave, want, or desire something like alcohol, pot, or a cigarette.”
2.4.2. Stress.
Momentary stress was assessed with two items asking participants to rate how “stressed” or “tense” they were on 11-point visual analogue scales from “not at all” (0) to “extremely” (10). A composite (average) of the two ratings indicated momentary stress (α = .84). Each person’s average of their momentary stress ratings over the duration of the EMA period quantified their general stress level, referred to herein as “average stress.”
2.4.3. Alcohol cues.
Participants had options to identify whether alcohol cues were not present, indirectly present (e.g., on TV, in a magazine), or directly present (e.g., bottle or glass in real life) at random assessments. Only direct cues were considered to indicate the presence of alcohol cues (1). Each person’s average of 0’s (alcohol cues not directly present) and 1’s (alcohol cues directly present) over the duration of the EMA period quantified the proportion of time they spent in the presence of direct alcohol cues, referred to herein as “average alcohol cues.”
2.4.4. Contextual variables.
Date and time stamps recorded for each EMA entry were used to identify contextual covariates relating to diurnal and weekly patterns. Weekend status was defined as 6 p.m. on Friday through 6 p.m. on Sunday and dichotomized into weekend (1) or weekday (0). Time of day was represented by four, 6-hr blocks starting from midnight (6 a.m. to noon = reference category, 0).
2.5. Analytic Plan
Multilevel modeling (MLM) with an unstructured variance-covariance matrix and between-within degrees of freedom accounted for the nesting of random assessments (Level 1) within persons (Level 2). Reports occurring while or after drinking on a given day were excluded to avoid confounding the effects of drinking with craving. Level 1 predictors included momentary stress, momentary presence of environmental alcohol cues (i.e., presence of direct alcohol cues), time of day, and weekend status. Momentary stress and cues were centered at the person mean to disaggregate momentary fluctuations from person-level processes. Level 2 variables were age, sex (dichotomous, males as reference), person-level stress (i.e., the person average of momentary stress reports), person-level alcohol cues (i.e., the person average of momentary alcohol-cue reports), WM, and AUD symptom count. All continuous Level 2 variables were grand-mean centered. Models included random intercepts.
Initial models tested whether craving was associated with greater likelihood of drinking, accounting for covariates. These utilized a binary response distribution (0 = non-drinking day; 1 = drinking day) with a logit link, maximum likelihood estimation, Laplace approximation, and between-within degrees of freedom. Predicted probability of drinking was calculated from the odds, i.e., exponentiated sum of product estimates, as follows: odds ÷ (1 + odds). Focal models utilized a normal distribution with an unstructured covariance matrix, restricted maximum likelihood estimation, and between-within degrees of freedom. First, the intraclass correlation coefficient (ICC) was estimated from an intercept-only model to identify the relative variability in craving due to person vs. momentary influences. Next, cross-level interactive effects of WM × Momentary Stress, Cues, and Peers tested the primary hypotheses, accounting for covariates. Last, analyses tested whether interactive effects were dependent on the participant’s sex through including three-way interactive effects of Sex × WM × Stress, Cues, and Peers. In the case of a significant three-way interactive effect, these were probed through subsequent models testing interactive effects for WM × Stress, Cues, and Peers separately for males and females. Interactive effects were illustrated graphically through plotting simple slopes (Preacher et al., 2006).
3. Results
3.1. Person-level Descriptive Statistics.
“Person-level” refers to processes that account for individual differences between participants. Person-level variables are either measured only once (e.g., age, sex) or are averaged across time (e.g., person-level stress). In focal analyses, grand-mean centering was used to facilitate interpretation of the intercept and fixed-effect parameters. Descriptive statistics here refer to person-level variables in their original form (not centered).
On average, participants were 20.7 years old (SD = 2.1), and about half (48.8%) indicated female biological sex. The majority identified as White (73.3%), Black (14.0%), Asian (4.7%) or American Indian (3.5%); 20.9% indicated Hispanic ethnicity. Participants reported drinking, on average, 4.9 standard drinks per drinking day in the past month (SD = 3.1) and reported alcohol consumption on 37.6% of days (SD = 20.0). The number of AUD symptoms ranged from 0 to 9 (M = 3.4; SD = 2.1). The average Memory for Words number correct was 18.9 (SD = 1.9; min = 15; max = 24). Average stress was 3.0 (SD = 2.0), and average craving was 1.9 (SD = 1.8). On average, participants indicated direct alcohol cues were present for 19% of random assessments completed (M = 0.19, SD = 0.24), and peers were present for 31% of random assessments completed (M = 0.31, SD = 0.24).
3.2. Person-level and Momentary Craving Variability
“Momentary” refers to processes that account for a participant’s fluctuations in focal variables. Momentary variables are repeated measures over time (e.g., momentary stress, momentary presence of alcohol). In focal analyses, person-mean centering was used to disaggregate momentary effects from person-level effects. An initial, intercept-only mixed model explored the variability in craving due to person-level and momentary factors. The ICC was .42, indicating that 42% of the variability in craving was due to person-level influences. The remaining 58% of the variability in craving was due to momentary influences, which includes error variance.
3.3. Relation of Craving with Drinking
Momentary craving predicted greater likelihood of drinking that day, b = 0.09, SE = 0.04, OR = 1.09, p = .018, accounting for all covariates including the effect of person-level craving. In terms of predicted probability, when momentary craving was not endorsed, the percent chance of drinking that day was 10.1%. In contrast, when momentary craving was rated at the sample average, the percent chance of drinking that day was 19.6%, and when momentary craving was endorsed as “strongest ever” the percent chance of drinking that day was 35.1%. Person-level craving, aggregated across all timepoints, was marginally associated with greater likelihood of drinking, on average, b = 0.18, SE = 0.10, OR = 1.20, p = .080, accounting for all covariates including the effect of momentary craving.
3.4. Relation of Stress with Craving
Greater momentary stress was associated with heightened momentary craving, b = 0.16, SE = 0.03, p < .001, accounting for all covariates including the effect of person-level stress (Table 1). Likewise, greater person-level stress was associated with heighted craving, on average, b = 0.24, SE = 0.10, p = .022, accounting for all covariates including the effect of momentary stress. Being in the presence of peers at that moment was also associated with heightened momentary craving, b = 0.37, SE = 0.13, p = .003, where spending more time with peers, aggregated across all timepoints, was not related to craving, on average, p = .910. Being in the presence of alcohol at that moment was also associated with heightened momentary craving, b = 0.78, SE = 0.16, p < .001, where spending more time in the presence of alcohol, aggregated across all timepoints, was not related to craving, on average, p = .850. With regard to other person-level covariates, AUD symptom count and WM were not related to craving, ps = .220 and .196, respectively, where female sex was associated with lower craving, on average, b = − 1.00, SE = 0.40, p = .014.
Table 1.
Effects of Momentary and Person-level Stress, Direct Alcohol Cues, Peers, and Working Memory on Craving, Accounting for Covariates.
| Parameters | Est. | 95 % Confidence Limits |
P | |
|---|---|---|---|---|
| LCL | UCL | |||
| Fixed intercept | 1.90 | 1.32 | 2.48 | < .001 |
| Location (Home = reference.) | ||||
| Vehicle | 0.42 | − 0.01 | 0.85 | .053 |
| Dormitory | 0.15 | − 0.42 | 0.71 | .608 |
| Friend’s home | 0.43 | − 0.03 | 0.90 | .069 |
| Other | 0.02 | − 0.59 | 0.63 | .955 |
| Party/club | 1.96 | 0.02 | 3.89 | .048 |
| Public place | 0.37 | 0.01 | 0.73 | .046 |
| School | 0.22 | − 0.32 | 0.75 | .427 |
| Work | 0.28 | − 0.15 | 0.71 | .200 |
| Weekend (Weekday = reference) | 0.55 | 0.35 | 0.75 | < .001 |
| Time of day (6 a.m. to Noon = reference) | ||||
| 6 p.m. to midnight | 0.11 | − 0.37 | 0.58 | .651 |
| Midnight to 6 a.m. | 0.91 | 0.67 | 1.15 | < .001 |
| Noon to 6 p.m. | − 0.45 | − 0.69 | − 0.20 | < .001 |
| Age | − 0.05 | − 0.25 | 0.15 | .612 |
| Alcohol use disorder severity | 0.12 | − 0.07 | 0.31 | .220 |
| Sex (Male = reference) | − 1.00 | − 1.79 | − 0.20 | .014 |
| Working memory | − 0.13 | − 0.34 | 0.07 | .196 |
| Person-Level peers | 0.10 | − 1.60 | 1.79 | .910 |
| Person-level alcohol cues | − 0.16 | − 1.86 | 1.54 | .850 |
| Person-level stress | 0.24 | 0.04 | 0.45 | .022 |
| Momentary peers | 0.37 | 0.12 | 0.62 | .003 |
| Momentary alcohol cues | 0.78 | 0.47 | 1.10 | < .001 |
| Momentary stress | 0.16 | 0.11 | 0.21 | < .001 |
| Momentary variance (error) | 3.62 | 3.37 | 3.88 | < .001 |
| Person-level variance (random intercept) Fit statistics | 2.87 | 1.90 | 3.35 | < .001 |
| − 2 residual log Likelihood (deviance) | 6984.4 | |||
| Akaike Information Criterion (AIC) | 6988.4 | |||
| Bayesian Information Criterion (BIC) | 6993.3 | |||
Note. Est. = unstandardized estimate; LCL = lower confidence limit; UCL = upper confidence limit. Continuous momentary variables are centered at the person mean. Continuous person-level variables are centered at the grand mean. Categorical covariates are not centered, with reference groups specified. Including random slopes for momentary peers, alcohol cues, and stress did not alter the significance of focal parameter estimates.
3.5. Moderation by Working Memory and Sex
On the whole, WM did not moderate the relation of momentary stress with craving, p = .738 (Table 2). Likewise, WM did not moderate the relation of momentary presence of peers or presence of direct alcohol cues with craving, ps = .947 and .972, respectively. Sex-differences analyses suggested, however, that the putative interaction of WM and stress on craving differed by biological sex, b = 0.09, SE = 0.03, p = .002. The three-way interactive effect was probed through examining interactive effects of WM and momentary stress separately for males and females. For males, WM buffered the association of momentary stress with craving, b = − 0.06, SE = 0.02, p = .008. For females, however, the relation of momentary stress and craving was not significant and in the opposite direction, b = 0.03, SE = 0.02, p = .138. The interactive effect is illustrated graphically in Figure 1 through plotting simple slopes for each biological sex at +/−1 standard deviation WM. Additional specificity analyses with other person-level moderators, i.e., AUD symptoms and age, were not significant.
Table 2.
Cross-level Interactive Effects of Working Memory with Momentary Stress, Direct Alcohol Cues, and Peers, Accounting for Person-level Effects and Covariates.
| Parameters | Est. | 95 % Confidence Limits |
P | |
|---|---|---|---|---|
| LCL | UCL | |||
| Fixed intercept | 1.90 | 1.32 | 2.48 | < .001 |
| Working memory | − 0.13 | − 0.34 | 0.07 | .196 |
| Person-level peers | 0.10 | − 1.60 | 1.79 | .910 |
| Person-Level alcohol cues | − 0.16 | − 1.86 | 1.54 | .851 |
| Person-level stress | 0.24 | 0.04 | 0.45 | .022 |
| Momentary peers | 0.37 | 0.12 | 0.62 | .004 |
| Momentary alcohol cues | 0.78 | 0.47 | 1.10 | < .001 |
| Momentary stress | 0.16 | 0.10 | 0.21 | < .001 |
| Working memory × momentary peers | 0.00 | − 0.13 | 0.12 | .947 |
| Working memory × momentary alcohol cues | 0.00 | − 0.15 | 0.14 | .972 |
| Working memory × momentary stress | 0.00 | − 0.03 | 0.02 | .738 |
| Momentary variance (error) | 2.87 | 1.90 | 3.85 | < .001 |
| Person-level variance (random intercept) | 3.63 | 3.37 | 3.89 | < .001 |
| Fit statistics | ||||
| − 2 residual log likelihood (deviance) | 6998.1 | |||
| Akaike Information Criterion (A1C) | 7002.1 | |||
| Save nan. Information Criterion (BIC) | 7007.0 | |||
Note. Est. = unstandardized estimate; LCL = lower confidence limit; UCL = upper confidence limit. Continuous momentary variables are centered at the person mean. Continuous person-level variables are centered at the grand mean. Categorical covariates are not centered, with reference groups specified. This model included all covariates listed in Table 1. Including random slopes for momentary peers, alcohol cues, and stress did not alter the significance of focal parameter estimates. Including interactive effects of working memory with person-level peers, alcohol cues, and stress did not alter the significance of focal parameter estimates.
Figure 1.
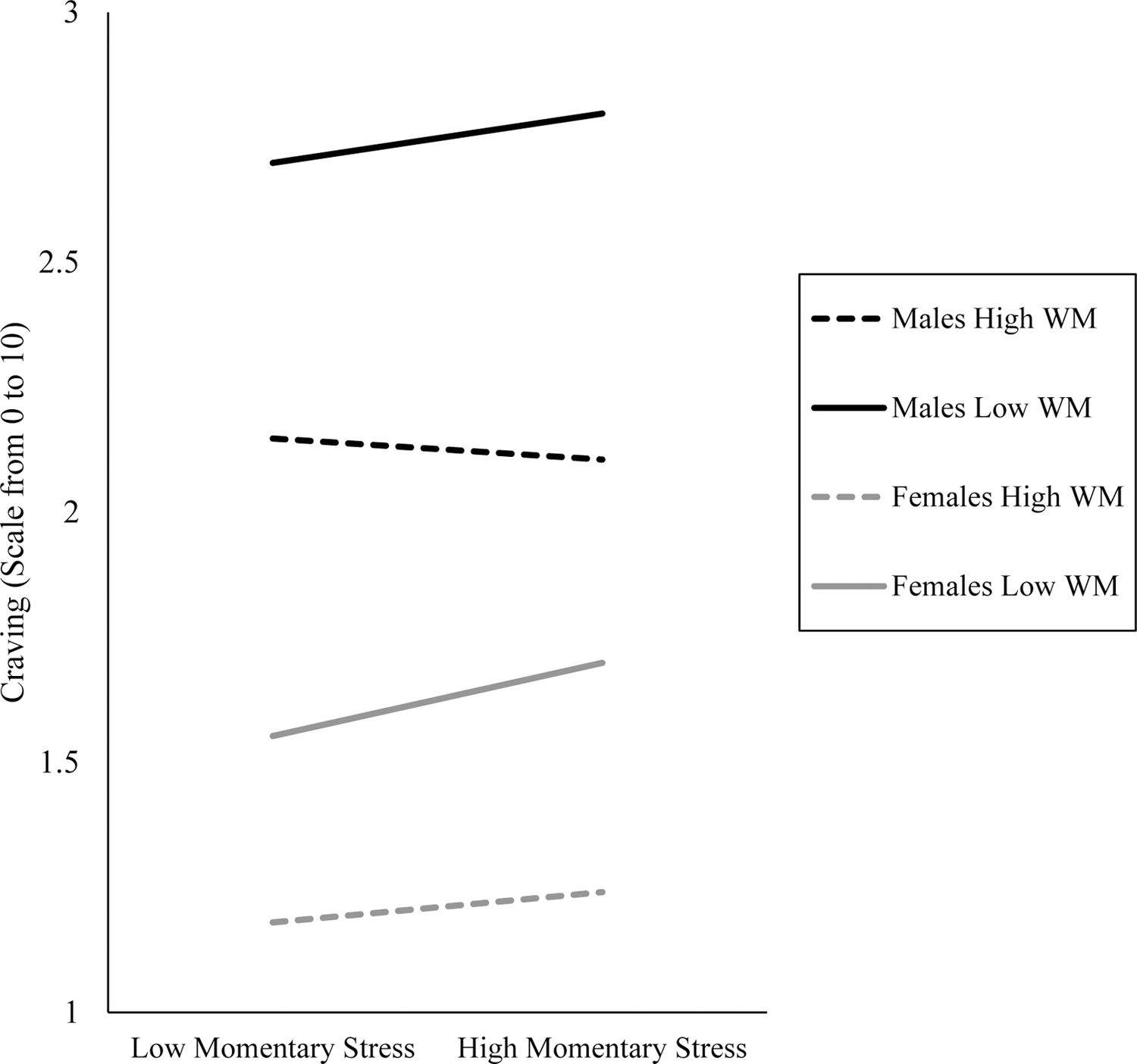
Simple slopes of craving (y axis) from times of low momentary stress to high momentary stress (x axis), plotted separately by biological sex and at high and low levels of working memory (WM). Black lines correspond to males, and gray lines correspond to females. Dashed lines correspond to +1 standard deviation working memory, and solid lines correspond to −1 standard deviation working memory. Momentary stress is a repeated measure over time and was person-mean centered to reflect a participant’s fluctuations in stress relative to their own average. Low and high values on the x axis correspond to +/−1 standard deviation for person-mean centered momentary stress.
Sex differences were also present with regard to WM moderating the momentary influences of being in the presence of peers and direct alcohol cues on craving (peers: b = − 0.45, SE = 0.13, p < .001; alcohol cues: b = 0.44, SE = 0.15, p = .004). For males, being in the presence of peers or direct alcohol cues was not related to craving, ps = .138 and .074, respectively, which contrasted results for momentary stress. For females, WM buffered the relation of being in the presence of peers with craving, b = − 0.32, SE = 0.12, p = .001, but exacerbated the relation of being in the presence of direct alcohol cues with craving, b = 0.29, SE = 0.12, p = .014, which contrasted results for momentary stress.
4. Discussion
Alcohol’s ability to relieve stress or tension is implicated in the most enduring etiological models of AUD development (Baker et al., 2004; Conger, 1956; Koob and Le Moal, 2001; Sher et al., 2005; Sher and Levenson, 1982; Sher and Walitzer, 1986; Wikler, 1948). The neurodevelopmental underpinnings of addiction suggest that neurocognitive deficits, as indicated by poor performance on tests of WM, may exacerbate alcohol misuse during adolescence, promoting AUD in adulthood (Squeglia et al., 2014a). The present work examined direct alcohol cues, peers, and stress as predictors of alcohol craving in adolescents’ daily lives, and, moreover, how these associations were modified by WM abilities. Adolescents were enrolled in a clinical trial for AUD treatment, and our analyses utilizing a baseline period prior to randomization to treatment condition. Contrary to hypotheses, WM did not alter the relation of momentary stress with craving for the sample as a whole. Instead, results suggested the influence of WM on stress-craving relations may be sex-linked. For males, having higher WM functioning buffered the in-the-moment relation of stress with alcohol craving. Females generally had lower craving than males, and the effect of stress on craving was not altered by WM function. Although preliminary, the protective effect of WM function for males appeared to be specific to momentary stress. While sex differences emerged for the interactive effects of WM with direct alcohol cues and with the presence of peers, results were specific to females rather than males. WM function served to protect against craving when females were with their peers and was associated with greater craving when females were in the presence of direct alcohol cues. Findings highlight the importance of considering biological sex when evaluating proneness to alcohol craving under the conditions of stress and when in the presence of peers and direct alcohol cues in daily life. Sex-related differences in prevalence rates for alcohol use and related problems emerge during later adolescence, and understanding factors underlying differences in drinking patterns may help to understand why more adult men develop AUD than adult women (Schulte et al., 2009).
Our work has important limitations. We report secondary analyses; although hypotheses were pre-registered, this study was not designed to address our specific research question. Our study applies intensive data collection in adolescents’ daily lives for a brief period, but it does not evaluate change over a longer period, such as months or years. The development of alcohol addiction is characterized by neurocognitive changes to the brain’s stress and reward systems leading to increased alcohol-seeking and craving (Volkow et al., 2016). Pre-existing neurocognitive abnormalities also increase risk for the heavier patterns of alcohol misuse, which, in turn, contribute to widespread alcohol-related deficits (Squeglia et al., 2014b; Squeglia and Gray, 2016). Only longitudinal research over time can distinguish whether sex-linked differences pre-date heavier drinking patterns, are a product of heavier drinking patterns, or both. Additionally, there is no single accepted measure or definition of craving (Sayette et al., 2000), and our EMA battery included a single item assessing “urge to drink.” Our measurement of WM is also a limitation. In our study, WM is measured as a static trait, rather than a dynamic state influenced by context. In prior research, WM load compromises decision making, amplifying facilitating effects of social contexts and reducing the inhibitory effects of disincentives (Ingram and Finn, 2019). EMA applications typically lay emphasis on the importance of contextual factors, and, as such, it is a limitation that a dynamic measure of WM was not used in addition to the gold-standard laboratory measure. These limitations considered, our work adds to the literature utilizing technological advances to monitor nuanced associations of individual differences and contextual influences on adolescents’ alcohol-related behaviors, and, moreover, emphasizes the importance of evaluating differential associations for adolescent males and females. Specifically, our findings suggest that sex-related differences in alcohol problems emerging in later adolescence could be influenced by the interplay of WM and in-the-moment contextual factors.
Table 3.
Moderating Influence of Sex on Cross-level Interactive Effects of Working Memory with Momentary Stress. Direct Alcohol Cues, and Peers, Accounting for Person-level Effects and Covariates.
| Parameters | Est. | 95 % Confidence Limits |
P | |
|---|---|---|---|---|
| LCL | UCL | |||
| Fixed intercept | 1.91 | 1.33 | 2.50 | < .001 |
| Sex (Male = reference) | − 1.00 | − 1.80 | − 0.20 | .164 |
| Working memory | −0.14 | −0.40 | 0.13 | .805 |
| Person−level peers | 0.11 | − 1.62 | 1.84 | .452 |
| Person−level alcohol cues | − 0.16 | − 1.93 | 1.61 | .915 |
| Person−level stress | 0.24 | 0.03 | 0.45 | .001 |
| Momentary peers | 0.58 | 0.26 | 0.91 | < .001 |
| Momentary alcohol cues | 0.84 | 0.38 | 1.31 | < .001 |
| Momentary stress | 0.09 | 0.01 | 0.17 | .037 |
| Working memory × momentary peers | 0.13 | − 0.03 | 0.29 | .113 |
| Working memory × momentary alcohol cues | −0.14 | − 0.33 | 0.04 | .122 |
| Working memory × momentary stress | −0.06 | −0.10 | − 0.02 | .008 |
| Working memory × sex | 0.00 | −0.45 | 0.45 | .588 |
| Momentary peers × sex | −0.59 | − 1.06 | − 0.12 | .016 |
| Momentary alcohol cues × sex | 0.00 | −0.61 | 0.62 | .968 |
| Momentary stress × sex | 0.09 | −0.02 | 0.20 | .088 |
| Working memory × momentary peers × sex | −0.45 | − 0.70 | − 0.19 | < .001 |
| Working memory × momentary alcohol cues × sex | 0.44 | 0.14 | 0.74 | .004 |
| Working memory × momentary stress × sex | 0.09 | 0.03 | 0.15 | .003 |
| Momentary variance (error) | 2.91 | 1.92 | 3.90 | < .001 |
| Person-level variance (random intercept) | 3.56 | 3.31 | 3.81 | < .001 |
| Fit statistics | ||||
| − 2 residual log likelihood (deviance) | 6977.2 | |||
| Akaike Information Criterion (AIC) | 6981.2 | |||
| Bayesian Information Criterion (BIC) | 6986.1 | |||
Note. Est. = unstandardized estimate; LCL = lower confidence limit; UCL = upper confidence limit. Continuous momentary variables are centered at the person mean. Continuous person-level variables are centered at the grand mean. Categorical covariates are not centered, with reference groups specified. This model included all covariates listed in Table 1. Including random slopes for momentary peers, alcohol cues, and stress reduced the significance of fixed-effect estimates for three-way interactive effects but did not alter the general directional pattern. Including interactive effects of sex and working memory with person-level peers, alcohol cues, and stress did not alter the significance of focal parameter estimates.
Highlights.
Adolescent craving heightened at stressful moments and with peers and alcohol cues
Females generally had lower craving than male adolescents
Working memory buffered against stress-induced craving for males not females
Role of Funding Source
The National Institutes of Health supported this research (AA024808; AA026326; AA007850).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of Competing Interest
The authors declare no competing interest.
Conflict of Interest
No conflict declared
References
- Arnett JJ, 1999. Adolescent Storm and Stress, Reconsidered. Am. Psychol 54, 317–326. 10.4324/9780203773642 [DOI] [PubMed] [Google Scholar]
- Baddeley A, 2003. Working memory: looking back and looking forward. Nat. Rev. Neurosci 4, 829–839. 10.1038/nrn1201 [DOI] [PubMed] [Google Scholar]
- Baker TB, Piper ME, McCarthy DE, Majeskie MR, Fiore MC, 2004. Addiction motivation reformulated: an affective processing model of negative reinforcement. Psychol. Rev 111, 33–51. 10.1037/0033-295X.111.1.33 [DOI] [PubMed] [Google Scholar]
- Blumenthal H, Cloutier RM, Douglas ME, Kearns NT, Carey CN, 2021. Desire to drink as a function of laboratory-induced social stress among adolescents. J. Behav. Ther. Exp. Psychiatry 70, 101617. 10.1016/j.jbtep.2020.101617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breese GR, Sinha R, Heilig M, 2011. Chronic alcohol neuroadaptation and stress contribute to susceptibility for alcohol craving and relapse. Pharmacol. Ther. 129, 149–171. 10.1016/j.pharmthera.2010.09.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown SA, McGue M, Maggs J, Schulenberg J, Hingson R, Swartzwelder S, Martin C, Chung T, Tapert SF, Sher K, Winters KC, Lowman C, Murphy S, 2008. A developmental perspective on alcohol and youths 16 to 20 years of age. Pediatrics 121 Suppl, S290–S310. 10.1542/peds.2007-2243D [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conger JJ, 1956. Reinforcement theory and the dynamics of alcoholism. Q. J. Stud. Alcohol 17, 296–305. [PubMed] [Google Scholar]
- Drummond DC, 2001. Theories of drug craving, ancient and modern. Addiction 96, 33–46. 10.1080/09652140020016941 [DOI] [PubMed] [Google Scholar]
- DSM-5, 2013. Diagnostic and Statistical Manual of Mental Disorders, 5th ed. American Psychiatric Association. [Google Scholar]
- Goldstein A, Déry N, Pilgrim M, Ioan M, Becker S, 2016. Stress and binge drinking: A toxic combination for the teenage brain. Neuropsychologia 90, 251–260. 10.1016/j.neuropsychologia.2016.07.035 [DOI] [PubMed] [Google Scholar]
- Haass-Koffler CL, Leggio L, Kenna GA, 2014. Pharmacological Approaches to Reducing Craving in Patients with Alcohol Use Disorders. CNS Drugs 28, 343–360. 10.1007/s40263-014-0149-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall GS, 1916. Adolescence: Its Psychology and Its Relations to Physiology, Anthropology, Sociology, Sex, Crime, Religion and Education, Volume II. ed. D. Appleton and Company, New York and London. [Google Scholar]
- Hardee JE, Cope LM, Munier EC, Welsh RC, Zucker RA, Heitzeg MM, 2017. Sex differences in the development of emotion circuitry in adolescents at risk for substance abuse: A longitudinal fMRI study. Soc. Cogn. Affect. Neurosci 12, 965–975. 10.1093/scan/nsx021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- ICD-10, 1992. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical descriptions and diagnostic guidelines, 1st ed. World Health Organization. [Google Scholar]
- Ingram PF, Finn PR, 2019. The effects of a working memory load on drinking-related decisions: The role of incentives, disincentives, and lifetime alcohol problems. Drug Alcohol Depend 204, 107567. 10.1016/j.drugalcdep.2019.107567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnston LD, Miech RA, O’Malley PM, Bachman JG, Schulenberg JE, Patrick ME, 2018. Monitoring the Future national survey results on drug use, 1975–2017: Overview, key findings on adolescent drug use
- Kaufman J, Birmaher B, Brent D, Rao U, Flynn C, Moreci P, Williamson D, Ryan N, 1997. Schedule for Affective Disorders and Schizophrenia for School-Age Children-Present and Lifetime Version (K-SADS-PL): initial reliability and validity data. J. Am. Acad. Child Adolesc. Psychiatry 36, 980–8. 10.1097/00004583-199707000-00021 [DOI] [PubMed] [Google Scholar]
- Koob GF, Le Moal M, 2001. Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24, 97–129. 10.1016/S0893-133X(00)00195-0 [DOI] [PubMed] [Google Scholar]
- Leffard SA, Miller JA, Bernstein J, DeMann JJ, Mangis HA, McCoy ELB, 2006. Substantive Validity of Working Memory Measures in Major Cognitive Functioning Test Batteries for Children. Appl. Neuropsychol 13, 230–241. 10.1207/s15324826an1304_4 [DOI] [PubMed] [Google Scholar]
- Litten RZ, Wilford BB, Falk DE, Ryan ML, Fertig JB, 2016. Potential medications for the treatment of alcohol use disorder: An evaluation of clinical efficacy and safety. Subst. Abus 37, 286–298. 10.1080/08897077.2015.1133472 [DOI] [PubMed] [Google Scholar]
- Nguyen-Louie TT, Courtney KE, Squeglia LM, Bagot K, Eberson S, Migliorini R, Alcaraz AR, Tapert SF, Pulido C, 2018. Prospective changes in neural alcohol cue reactivity in at-risk adolescents. Brain Imaging Behav 12, 931–941. 10.1007/s11682-017-9757-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Preacher KJ, Curran PJ, Bauer DJ, 2006. Computational Tools for Probing Interactions in Multiple Linear Regression, Multilevel Modeling, and Latent Curve Analysis. J. Educ. Behav. Stat 31, 437–448. 10.3102/10769986031004437 [DOI] [Google Scholar]
- Ramirez J, Miranda R, 2014. Alcohol craving in adolescents: Bridging the laboratory and natural environment. Psychopharmacology (Berl) 231, 1841–1851. 10.1007/s00213-013-3372-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayette MA, Shiffman S, Tiffany ST, Niaura RS, Martin CS, Schadel WG, 2000. The measurement of drug craving. Addiction 95, 189–210. 10.1080/09652140050111762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrank FA, 2005. Woodcock-Johnson III Tests of Cognitive Abilities, in: Flanagan DP, Harrison PL (Eds.), Contemporary Intellectual Assessment: Theories, Tests, and Issues New York: Guildford, pp. 371–401. [Google Scholar]
- Schulte MT, Ramo D, Brown SA, 2009. Gender differences in factors influencing alcohol use and drinking progression among adolescents. Clin. Psychol. Rev 29, 535–547. 10.1016/j.cpr.2009.06.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher KJ, Grekin ER, Williams NA, 2005. The development of alcohol use disorders. Annu. Rev. Clin. Psychol 1, 493–523. 10.1146/annurev.clinpsy.1.102803.144107 [DOI] [PubMed] [Google Scholar]
- Sher KJ, Levenson RW, 1982. Risk for alcoholism and individual differences in the stress-response-dampening effect of alcohol. J. Abnorm. Psychol 91, 350–67. [DOI] [PubMed] [Google Scholar]
- Sher KJ, Walitzer KS, 1986. Individual differences in the stress-response-dampening effect on alcohol: a dose-response study. J. Abnorm. Psychol 95, 159–67. [DOI] [PubMed] [Google Scholar]
- Sobell LC, Sobell MB, 1992. Measuring Alcohol Consumption: Psychosocial and biochemical methods Humana Press, Totowa, NJ. 10.1007/978-1-4612-0357-5 [DOI] [Google Scholar]
- Spear L, 2013. The Teenage Brain: Adolescents and Alcohol. 10.1177/0963721412472192 [DOI]
- Squeglia LM, Boissoneault J, Van Skike CE, Nixon SJ, Matthews DB, 2014a. Age-Related Effects of Alcohol from Adolescent, Adult, and Aged Populations Using Human and Animal Models. Alcohol. Clin. Exp. Res 38, 2509–2516. 10.1111/acer.12531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Squeglia LM, Gray KM, 2016. Alcohol and Drug Use and the Developing Brain. Curr. Psychiatry Rep 18, 46. 10.1007/s11920-016-0689-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Squeglia LM, Jacobus J, Tapert SF, 2014b. The effect of alcohol use on human adolescent brain structures and systems, in: Handbook of Clinical Neurology. pp. 501–510. 10.1016/B978-0-444-62619-6.00028-8 [DOI] [PMC free article] [PubMed]
- Squeglia LM, Schweinsburg AD, Pulido C, Tapert SF, 2011. Adolescent binge drinking linked to abnormal spatial working memory brain activation: Differential gender effects. Alcohol. Clin. Exp. Res 35, 1831–1841. 10.1111/j.1530-0277.2011.01527.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg L, 2008. A social neuroscience perspective on adolescent risk-taking. Dev. Rev 28, 78–106. 10.1016/j.dr.2007.08.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas SE, Drobes DJ, Deas D, 2005. Alcohol cue reactivity in alcohol-dependent adolescents. J. Stud. Alcohol 66, 354–360. [DOI] [PubMed] [Google Scholar]
- Tiffany ST, 1999. Cognitive concepts of craving. Alcohol Res. Heal 23, 215–24. [PMC free article] [PubMed] [Google Scholar]
- Tiffany ST, Wray JM, 2012. The clinical significance of drug craving. Ann. N. Y. Acad. Sci 1248, 1–17. 10.1111/j.1749-6632.2011.06298.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treloar Padovano H, Janssen T, Emery NN, Carpenter RW, Miranda R Jr, 2019. Risk-Taking Propensity, Affect, and Alcohol Craving in Adolescents’ Daily Lives. Subst. Use Misuse 0, 1–11. 10.1080/10826084.2019.1639753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treloar Padovano H, Miranda R, 2020. Incubation of alcohol craving as it naturally occurs in a developmentally diverse sample of dependent and nondependent drinkers. Addict. Biol 10.1111/adb.12934 [DOI] [PMC free article] [PubMed]
- van Lier HG, Pieterse ME, Schraagen JMC, Postel MG, Vollenbroek-Hutten MMR, de Haan HA, Noordzij ML, 2018. Identifying viable theoretical frameworks with essential parameters for real-time and real world alcohol craving research: a systematic review of craving models. Addict. Res. Theory 26, 35–51. 10.1080/16066359.2017.1309525 [DOI] [Google Scholar]
- Verplaetse TL, Cosgrove KP, Tanabe J, McKee SA, 2020. Sex/gender differences in brain function and structure in alcohol use: A narrative review of neuroimaging findings over the last 10 years. J. Neurosci. Res jnr.24625. 10.1002/jnr.24625 [DOI] [PMC free article] [PubMed]
- Volkow ND, Koob GF, McLellan AT, 2016. Neurobiologic Advances from the Brain Disease Model of Addiction. N. Engl. J. Med 374, 363–371. 10.1056/NEJMra1511480 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wikler A, 1948. Recent progress in research on the neurophysiologic basis of morphine addiction. Am. J. Psychiatry 105, 329–338. [DOI] [PubMed] [Google Scholar]
- Witkiewitz K, Litten RZ, Leggio L, 2019. Advances in the science and treatment of alcohol use disorder. Sci. Adv 5. 10.1126/sciadv.aax4043 [DOI] [PMC free article] [PubMed] [Google Scholar]


