Abstract
Objectives
To investigate and synthesize systematically the evidence from animal studies pertaining to the effect of pharmacological agents on tooth movement relapse following cessation of orthodontic force application.
Materials and Methods
An electronic search was conducted in seven online databases (including gray sources) without restrictions until the third week of April 2019, followed by a hand search in the reference lists of eligible articles. Controlled animal studies investigating the effect of pharmacological agents on tooth movement relapse following orthodontic treatment were selected. Relevant data were extracted from eligible studies and the risk of bias assessment was done using SYRCLE's risk of bias tool. The quality of evidence was assessed using the Grading of Recommendations Assessment, Development, and Evaluation tool.
Results
The search identified 2354 records, of which 7 studies were deemed eligible for inclusion in the qualitative synthesis, with the majority presenting an unclear risk of bias. Orthodontic relapse was shown to decrease with the administration of pamidronate disodium, atorvastatin, aspirin, and chemically modified tetracycline-3. Inconsistent effects on relapse were observed after the use of simvastatin. The overall quality of retrieved evidence was assessed as low at best.
Conclusions
The available evidence shows that the investigated pharmacological agents may demonstrate variable effects on tooth movement relapse following cessation of orthodontic force. Additional evidence of higher quality is required to draw definitive conclusions on their effects and to make potential recommendations for clinical application.
Keywords: Systematic review, Orthodontic relapse, Drugs, Animals
INTRODUCTION
Rationale
Orthodontic relapse (OR) is an inevitable and unfavorable sequela of orthodontic treatment, which is believed to be multifactorial in etiology. The causes include those beyond the level of teeth and supporting structures, such as neuromuscular imbalance, continued craniofacial growth, and retained habits.1,2 Some authors emphasized recognizing OR as a distinct manifestation of physiological recovery in response to withdrawal of forces, different from developmental and age-related changes.3,4
Over the years, it has been generally accepted that the elasticity of overstretched supra-alveolar connective tissue fibers of the gingiva (primarily the transseptal fibers) exerted physiologic forces causing OR.5 This hypothesis was, however, refuted through recent studies, which concluded that periodontal tissue reorganization was rapid and therefore did not contribute to the etiology of relapse.4 Further histologic studies, conducted predominantly on animal models, demonstrated that the pattern of tissue and molecular level responses immediately after withdrawal of orthodontic force were similar to those seen during active orthodontic tooth movement (OTM).6–8 Continued activity of osteoclasts in the direction of relapse, osteoblasts in the direction opposite to that of relapse, gene expression of markers of alveolar bone and collagen remodeling were consistent with that observed during OTM, but in the opposite direction.6–8
Underpinned by this similarity in physiological patterns occurring during OTM and OR, chemical preparations determined to decrease the rate of OTM by targeting the cascades involved in alveolar bone remodeling can potentially modulate OR. Evidence from animal and human studies indicated that commonly prescribed drugs, such as nonsteroidal anti-inflammatory drugs, bisphosphonates, and statins, had an inhibitory effect on the rate of OTM.9,10 In this context, a systematic review of animal evidence on the effect of commonly prescribed pharmacological substances on OR would render an estimate of their effects in humans. This review is the first to aim at appraising the evidence from animal experiments on the effect of pharmacological agents on OR.
Objective
The objective of this systematic review was to investigate and evaluate the quality of evidence from animal experiments pertaining to the effect of pharmacological agents on tooth movement relapse following cessation of orthodontic force.
MATERIALS AND METHODS
Protocol and Registration
Based on the recommendations outlined in the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) protocol statement, a protocol was developed and registered with PROSPERO (CRD42019135536).11 The review was conducted in accordance with The Cochrane Handbook for Systematic Reviews of Interventions and reporting conforms to the PRISMA statement.12,13
Eligibility Criteria
Criteria for determining the eligibility of studies were defined according to participants, intervention, comparison, outcomes, and study design (Table 1). Experiments involving healthy animals subjected to relapse without retention following active tooth movement were assessed. The amount of OR was measured following local or systemic administration of pharmacological agents commonly prescribed to humans and compared with controls with placebo intervention or no intervention.14 Studies without a control group and reviews were excluded.
Table 1. .
Eligibility Criteria Considered for the Systematic Review
| Domain |
Inclusion Criteria |
Exclusion Criteria |
| Participants | ▪ Animal subjects subjected to relapse without any form of retention, immediately following orthodontic tooth movement | ▪ Animal subjects with any form of retention before or during the period of relapse |
| Interventions | ▪ Local or systemic administration of pharmacological agents during the period of relapse (excepting those substances that are administered once for a long period of time, such as the bisphosphonates) | ▪ Administration of substances that are experimental or not prescribed to humans ▪ Simultaneous administration of substances not considered in the present review ▪ Simultaneous use of other types of interventions intended to affect OR |
| Comparisons | ▪ Placebo intervention or no intervention | ▪ Studies with only baseline controls not subjected to OTM or control group with retention appliance |
| Outcomes | ▪ Quantitative data regarding tooth movement relapse following OTM (rate of relapse, percentage relapse, amount of relapse, etc.) | ▪ Qualitative assessments regarding tooth movement relapse ▪ Inadequate definition of outcomes |
| Study design | ▪ Experimental prospective controlled studies | ▪ Noncomparative studies (case reports and case series) ▪ Systematic reviews and meta-analyses ▪ In vitro or ex vivo studies ▪ Opinions and editorials |
Information Sources and Search
Seven online databases, including gray sources, were searched without restrictions on language or date until the third week of April 2019. A detailed search strategy developed for EMBASE (via OVID) by P.V. was later adapted appropriately for other databases (Table 2). Reference lists of all included and excluded articles were searched to ensure that no relevant studies were missed.
Table 2. .
Detailed Search Strategy for All Electronic Databases, Until the Third Week of April 2019
| Database |
Search Strategy |
Hits |
| General sources | ||
| PubMed http://www.ncbi.nlm.nih.gov/pubmed | (orthodon*[All Fields] OR “fixed orthodontic"[All Fields] OR bracket*[All Fields] OR “orthodontic force"[All Fields] OR “mechanical force"[All Fields]) AND (“tooth movement"[All Fields] OR “orthodontic movement"[All Fields] OR “orthodontic anchorage"[All Fields]) AND (“recurrence"[MeSH Terms] OR “recurrence"[All Fields] OR “relapse"[All Fields]) | 290 |
| Cochrane Central Register of Controlled Trials https://www.cochranelibrary.com/advanced-search | Title Abstract Keyword: ((orthodon* OR “fixed orthodontic” OR bracket* OR “orthodontic force” OR “mechanical force”) AND (“tooth movement” OR “orthodontic movement” OR “orthodontic anchorage”) AND (relapse)) | 8 |
| Scopus https://www.scopus.com/search/form.uri?display=advanced | TITLE-ABS-KEY (orthodon* OR “fixed orthodontic” OR bracket* OR “orthodontic force” OR “mechanical force”) AND (“tooth movement” OR “orthodontic movement” OR “orthodontic anchorage”) AND (relapse) | 937 |
| Web of Science http://apps.webofknowledge.com/ | TS=((orthodon* OR “fixed orthodontic” OR bracket* OR “orthodontic force” OR “mechanical force”) AND (“tooth movement” OR “orthodontic movement” OR “orthodontic anchorage”) AND (relapse)) Timespan: All years Databases: All databases Search language: Auto | 293 |
| Embase (via OVID) http://ovidsp.tx.ovid.com/sp-3.33.0b/ovidweb.cgi | (((orthodon*.mp OR exp orthodontics/ or exp orthodontic device/) OR fixed orthodontic.mp OR (bracket*.mp OR exp orthodontic bracket/) OR orthodontic force.mp OR mechanical force.mp)) AND ((tooth movement.mp OR (orthodontic movement.mp OR exp orthodontic tooth movement/) OR (orthodontic anchorage.mp OR exp orthodontic anchorage/)) AND ((relapse OR exp relapse/))) | 820 |
| Gray literature sources | ||
| ClinicalTrials.gov http://clinicaltrials.gov/ | (orthodontic OR orthodontics) AND (tooth movement) AND (relapse) | 0 |
| ProQuest Dissertations and Theses Global http://search.proquest.com/dissertations | (orthodon* OR “fixed orthodontic” OR bracket* OR “orthodontic force” OR “mechanical force”) AND (“tooth movement” OR “orthodontic movement” OR “orthodontic anchorage”) AND (relapse) | 5 |
Study Selection
The assessment of articles obtained was done by two reviewers (P.V. and S.R.T.) independently and in duplicate. First, the titles and abstracts of articles were assessed for eligibility, following which full-text reports were obtained for articles that were deemed to have met the inclusion criteria.
Data Collection and Data Items
Relevant data from the included studies were extracted by two reviewers (P.V. and S.R.T.) in previously developed and piloted forms: author and year, study design, subject characteristics, mechanisms inducing OTM, OR characteristics, pharmacological interventions, outcome assessments, and results.
Risk of Bias in Individual Studies
Assessment for risk of bias (RoB) was done by P.V. and V.M. in duplicate using SYRCLE's tool15 following Makrygiannakis et al.10 The overall RoB was summarized for each study as specified in the Cochrane Handbook.12
Synthesis of Results
It was planned to use random effects meta-analysis to determine pooled estimates and relative effects of interventions on OR.16 However, the lack of homogeneity in the study design and interventions employed precluded quantitative data synthesis.12
RoB Across Studies and Additional Analyses
Although planned, analyses for small-study effects, publication bias, or exploratory subgroup analyses were not feasible because of the inadequate number of studies. The quality of evidence from the retrieved studies was assessed using Grading of Recommendation Assessment, Development, and Evaluation (GRADE) approach, which was graded as high, moderate, low, and very low.17 The grading represented the confidence in the effect estimate and was based on the following aspects: RoB, inconsistency, indirectness, imprecision, and others.
During the entire process, any disagreements were discussed and resolved by consensus and, when required, a third reviewer (S.G.) was consulted for arbitration.
RESULTS
Study Selection
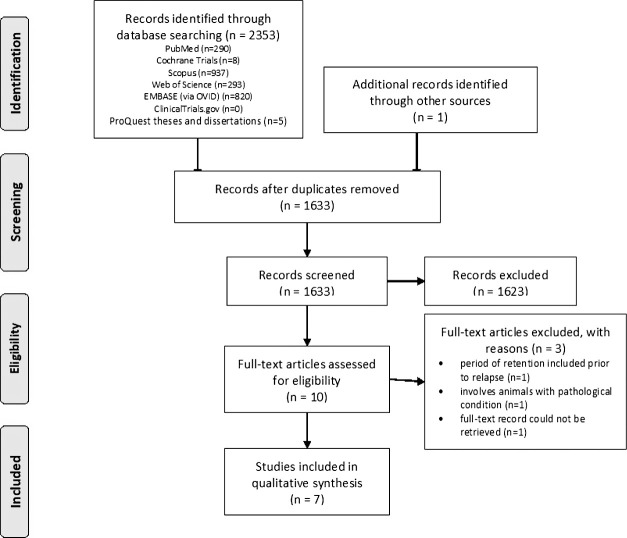
The process of screening of records is shown in the PRISMA flowchart in Figure 1. The search identified a total of 2354 studies, of which 1633 studies were screened for titles and abstracts after the removal of duplicates. From the remaining 10 records that were assessed for eligibility, 3 studies were excluded because one included a period of retention before experimental relapse,18 1 involved animals with pathological conditions,19 and the full-text record of 1 study could not be retrieved.20 Finally, seven studies were included in the qualitative synthesis.21–27
Figure 1.
PRISMA flowchart.
Study Characteristics
Table 3 presents the general characteristics of the studies included. Rats were employed as an animal model in all the studies except one26 that used a rabbit model. OTM was induced using springs placed between either incisors and molars, or incisors and premolars, delivering forces ranging from 50 to 100 cN for a period of 14 to 21 days. One study used elastic bands to effect OTM.21 The type of tooth movement achieved was tipping in most of the studies.
Table 3. .
General Characteristics of Studies Included in the Systematic Reviewa
| Author, Reference Year |
Pharmacological Agent (Therapeutic Category) |
Animals (Species, Age, Gender) |
OTM Model (Mechanism, Type of OTM, Amount of Force, Duration) |
Group Characteristics (Groups, Sample Size, Dose, Route, Administration) |
Timeline of Relapse Measurementb; Resultsc |
|
| Experimental Group |
Control Group |
|||||
| Kim et al.,21 1997 | Pamidronate disodium (bisphosphonates) | Wistar rats, 7 weeks, male | Elastic band: R&L Mx FM and SM; tipping; NS; 21 days | Pamidronate disodium; E- 7; 1.5 mg/mL/kg; IV; one time on the last day of OTM E1-0 days E2-5 days E3-10 days | C-7, PS; 1.0mL/kg; NS; one time C1-0 days C2-5 days C3-10 days | Days 5, 10; decrease |
| Han et al.,22 2010 | Simvastatin (antihyperlipidemic agents) | Wistar rats, 7–8 weeks, male | NiTi CCS: Mx Is to R&L FM; tipping; 50cN; 21 days | Simvastatin; E-16; 2.5 mg/kg; IP; daily for 4 weeks from last day of OTM | C-16; 0.9% NaCl NS; IP; daily for 4 weeks | Weeks 1 and 4; decrease |
| Vieira et al.,23 2015 | Simvastatin (antihyperlipidemic agents) | Wistar rats, 4 months, male | SS CCS: Mx I to L FM; tipping; 75 cN; 18 days | Simvastatin; E-15; 5 mg/kg, 400 μL; oral gavage; daily for 20 days | C- 10; 0.5% CMC; 400 μL; oral gavage; NS | Day 20; ND |
| Dolci et al.,24 2017 | Atorvastatin (antihyperlipidemic agents) | Wistar rats; 6 weeks; male | NiTi CCS: Mx Is to R FM; NS; 50 cN; 21 days | Atorvastatin; 15 mg/kg; oral gavage; E1: 6; daily for 7 days E2: 6; daily for 14 days E3: 6; daily for 21 days | C-18; PBS; 0.1 ml; oral gavage; daily for 7, 14 or 21 days BC-36; SMD-L Mx FMs of all animals | Days 7, 14, 21; decrease |
| Liu et al.,25 2017d | Aspirin (analgesics) | Sprague-Dawley rats; 6–8 weeks; male | NiTi CCS: Mx Is to R FM; NS; 50 gm; 14 days | Aspirin; E-3; 300 mg/kg; oral; daily for 10 days | UC-3 | Days 0.5,1,2,3, 5,7,10; decrease |
| AlSwafeeri et al.,26 2018 | Simvastatin (antihyperlipidemic agents) | New-Zealand rabbits; 16 weeks; male | NiTi CCS: Md Is and R&L PM; tipping; 100cN; 21 days | Simvastatin; RSMD; E-10 PMs; 0.5 mg/480 μL; IL (180 μL) and SMu (300 μL); days 21,28,35b | Pluronic F-127; C- 10 PMs; IL (180 μL) and SMu (300 μL); days 21,28,35e | Day 21; ND |
| Vieira et al.,27 2019f | CMT-3 (antibiotics) | Wistar rats; 4 months; male | SS CCS: Mx I to L FM; tipping; 75 cN; 18 days | CMT-3; E-15; 30 mg/kg; oral; daily for 20 days | C-15; 0.5% CMC; oral; daily for 20 days | Day 20; decrease |
BC indicates baseline control without any force application; C, control with placebo or vehicle intervention; CCS, closed-coil spring; CMC, carboxymethyl cellulose; CMT-3, chemically modified tetracycline-3; cN-centiNewton E, experimental; FM, first molar; I, incisors; IL, intraligamental; IP, intraperitoneal; IV, intravenous; L, left; NiTi, nickel titanium; Md, mandibular; Mx, maxillary; ND, no significant difference in relapse relative to control group/s; NS, Not specified; OTM, orthodontic tooth movement; PBS, phosphate buffer solution; PS, physiological saline; PM, premolar; R, right; RSMD, randomized split-mouth design; SM, second molar; SMD, split-mouth design; SMu, submucosal; SS, stainless steel; UC, untreated control force application without any intervention.
After cessation of tooth movement.
All results represent comparisons with control group/s.
Multiple experiments were conducted.
Days from the beginning of the experiment.
Results from simvastatin group not included in synthesis.
The pharmacological agents employed in the included studies were from the following therapeutic categories:14 antihyperlipidemic agents22–24,26,27; bisphosphonates21; analgesics25; and antibiotics.27 Administration was systemic in six studies,21–25,27 whereas local administration was employed in one.26 Systemic administration involved oral, intraperitoneal, or intravenous. Substances were administered either one time, daily, or weekly, for a maximum period of 4 weeks.
All of the included studies measured relapse as the distance moved by the experimental tooth following cessation of active orthodontic force. Timeline of tooth relapse measurements varied from 0.5 days to 4 weeks.
RoB Within Studies
The overview of the assessment of RoB for the included studies is presented in Table 4. All of the studies were assessed as presenting with an unclear RoB and only one study of low risk.26 Most of the studies presented an unclear RoB pertaining to random sequence generation, allocation concealment, blinding of caregivers/investigators, selection of animals for outcome assessment, and blinding of outcome assessor, as there was inadequate information to arrive at a definitive judgment on RoB. However, with respect to the random housing of the animals, one study presented a high RoB.21 The majority of the studies included groups that were comparable at baseline for age, gender, and weight and as a result were assessed as having a low RoB. Data from all the animals were analyzed in all of the studies receiving a low RoB rating for the corresponding domain. Finally, it was deemed unlikely that bias could have been introduced as a result of selectively reporting the outcomes and other potential sources.
Table 4. .
Summary of Risk of Bias Assessment According to SYRCLE RoB Tool15
| Study |
Signaling Questiona |
||||||||||
| 1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
Summary |
|
| Kim et al.,21 1997 | Unclear | Low | Unclear | High | Unclear | Unclear | Unclear | Low | Low | Unclear | Unclear |
| Han et al.,22 2010 | Unclear | Low | Unclear | Unclear | Unclear | Unclear | Unclear | Low | Low | Unclear | Unclear |
| Vieira et al.,23 2015 | Unclear | Low | Unclear | Unclear | Unclear | Unclear | Unclear | Low | Low | Unclear | Unclear |
| Dolci et al.,24 2017 | Unclear | Low | Unclear | Low | Unclear | Low | Low | Low | Low | Unclear | Unclear |
| Liu et al.,25 2017 | Unclear | Unclear | Unclear | Unclear | Unclear | Unclear | Unclear | Low | Low | Unclear | Unclear |
| AlSwafeeri et al.,26 2018 | Low | Low | Low | Low | Low | Low | Low | Low | Low | Unclear | Low |
| Vieira et al.,27 2019 | Unclear | Low | Unclear | Unclear | Unclear | Unclear | Unclear | Low | Low | Unclear | Unclear |
Signaling questions: (1) Was the allocation sequence adequately generated and applied? (2) Were the groups similar at baseline or were they adjusted for confounders in the analysis? (3) Was the allocation adequately concealed? (4) Were the animals randomly housed during the experiment? (5) Were the caregivers and investigators blinded to the intervention that each animal received? (6) Were animals selected at random for outcome assessment? (7) Was the outcome assessor blinded? (8) Were incomplete outcome data adequately addressed? (9) Are reports of the study free of selective outcome reporting? (10) Was the study apparently free of other problems that could result in high risk of bias?
Results of Individual Studies
Table 3 presents the results from the included studies. There was a consistent decrease in the amount of relapse for the experimental period following the administration of pamidronate disodium,21 atorvastatin,24 aspirin,25 and chemically modified tetracycline (CMT)-3.27 Inconsistent effects were observed with administration of simvastatin.22,23,26 The results pertaining to the simvastatin group in the study by Vieira et al.27 was not included in the synthesis considering the significant overlap with a previous study.23
Additional Analysis
The quality of retrieved evidence on the effects of investigated substances on OR, as appraised using the GRADE approach, was at best low (Table 5).
Table 5. .
Quality of Available Evidence Using GRADE
| Quality Assessment Criteria |
Effect on Orthodontic Relapse |
Quality |
|||||
| Studies |
Risk of Bias |
Inconsistency |
Indirectness |
Imprecision |
Other Considerations |
||
| Bisphosphonates: pamidronate disodium | |||||||
| 1 | Not serious | Not serious | Seriousa | Seriousb | None | Decrease | ⊕⊕○○LOW |
| Analgesics: aspirin | |||||||
| 1 | Not serious | Not serious | Seriousa | Seriousb | None | Decrease | ⊕⊕○○LOW |
| Antibiotics: chemically modified tetracycline-3 | |||||||
| 1 | Not serious | Not serious | Seriousa | Seriousb | None | Decrease | ⊕⊕○○LOW |
| Antihyperlipidemic agents: atorvastatin | |||||||
| 1 | Not serious | Not serious | Seriousa | Seriousb | None | Decrease | ⊕⊕○○LOW |
| Antihyperlipidemic agents: simvastatin | |||||||
| 3 | Not serious | Seriousc | Seriousa | Seriousb | None | Inconsistent effects | ⊕○○○VERY LOW |
Results cannot be directly extrapolated to human clinical settings.
The number of animals analyzed were limited.
Inconsistent or conflicting effects were noted.
DISCUSSION
Summary of Evidence
In summary, based on the evidence retrieved from the eligible animal studies, pharmacological agents commonly prescribed to humans had a variable effect on tooth movement relapse following removal of active orthodontic force. Considering that the available evidence was assessed to have low quality at best, findings from the included studies require careful deliberation. Additional evidence from high-quality studies is required for making definitive recommendations for extrapolation of these findings to human settings.
Theoretically, any substance modulating the signaling pathways involved in periodontal tissue remodeling can affect tooth movement relapse.4,7,8 Accordingly, it was observed in the current review process that substantial research on the effects of various biomimetics, recombinant proteins and pharmacological substances on OR is being undertaken. However, contemplating the viability of translation of findings to human settings, the current review was restricted to studies investigating commonly prescribed medications.14 It was, however, surprising to find a scarcity of such research.
OR was shown to decrease with the systemic administration of pamidronate disodium,21 atorvastatin,24 aspirin,25 and CMT-3.27 The anti-osteoclast effect of bisphosphonates and consequent inhibition of OTM is well established in the literature.28–30 Bisphosphonates prevent osteoclastic bone resorption by interfering with cytoskeletal organization and the formation of ruffled borders, inducing apoptosis of osteoclasts, thereby modulating the remodeling of alveolar bone responsible for OR.31 Although systemic administration of bisphosphonates was shown to reduce OR, the associated deleterious effects on bone turnover in other tissues must be carefully regarded. Local administration of bisphosphonates was found to enhance the rate of bone regeneration comparable to that of systemic administration with minimal systemic effects.32
Statins such as atorvastatin have been shown to influence bone turnover by upregulating bone formation and inhibiting resorption.33,34 Although lipophilic simvastatin has been suggested to exert greater influence on bone turnover when compared with other statins belonging to same class such as atorvastatin,34 inconsistent effects on OR were noted with the administration of simvastatin.22,23,26,27 There was a dose-dependent effect of simvastatin on the magnitude of relapse; systemic administration of a low dose of simvastatin decreased OR,22 whereas systemic administration of a high dose and local administration of a low dose did not affect OR.23,26,27 Differences in sensitivity of osteoclasts and osteoblasts to different concentrations of simvastatin, experimental animal model, bioavailability associated with route of drug administration, duration of exposure, and experimental designs have been attributed to this varying response of bone remodeling.35 Further studies are warranted to investigate these assumptions.
CMTs have been shown to have beneficial effects in preventing periodontal tissue breakdown, owing to the inhibitory effects on proinflammatory cytokines and matrix metalloproteinase–mediated bone resorption.36–38 The elevated expression of matrix metalloproteinase in response to stresses in the periodontal ligament tissue during tooth movement alludes to the contributory role of this class of proteinases during relapse.39–41 Nonsteroidal anti-inflammatory drugs such as aspirin prevent prostaglandin E2–mediated osteoclastogenesis by inhibiting cyclooxygenase activity.42 Aspirin also reduces the release of proinflammatory cytokines such as TNF-α and IFN-γ responsible for promoting bone resorption.43
Strengths and Limitations
This review was the first to have appraised the evidence on the effects of pharmacological agents on OR. The methodology employed in the current review followed well-established guidelines. An exhaustive search strategy was used for retrieving data from electronic, manual, and gray sources until April 2019. It was comprehensive, without restrictions on date, language, or status of publication. The screening of records for eligibility, data abstraction, RoB assessment, and appraisal of the evidence were conducted in duplicate, and disagreements were discussed or arbitrated with another reviewer to eliminate any possible bias in the methodology.
The limitations of this review arose intrinsically from the study characteristics and retrieved data, resulting in the quality of evidence being assessed, at best, as low. The lack of adequate data precluded quantitative synthesis. Furthermore, most of the included studies were assessed to have an unclear RoB, owing primarily to the methodological characteristics. Imprecision because of the limited number of animals employed and inconsistent findings further increased the uncertainty of evidence.
In addition, it must be noted that the data included for analysis in this systematic review were drawn from animal experiments and cannot be directly extrapolated to humans. Also, the studies involved administration of pharmacological substances for periods of time different from those used commonly in clinical settings14 and dosages without rationale calculation for equivalence to humans.44 The generalizability of results presented in this synthesis may be limited by the methodology for assessment of relapse; most of the studies investigated relapse following tipping tooth movement for varying periods of time.
Recommendations for Future Research
OR continues to be difficult to manage for most clinicians, hence further research is warranted. In keeping with the existing knowledge on the biological basis of OR,6–8 pharmacological agents with antiosteoclast effect show promise in regulating OR. Future research must explore drugs belonging to this class. It is also imperative that future animal studies follow standardized protocols,45 with consideration to simulate human clinical scenarios with regard to the duration of drug administration, dose equivalence, and route as well as the attributes of mechanisms inducing tooth movement and methodology of relapse assessment. Emphasis must also be given to sample size calculation to enhance the power of the study and the significance of the results.46
CONCLUSIONS
Additional evidence of higher quality is required to draw definitive conclusions on the effects of the investigated pharmacological agents on tooth movement relapse following cessation of orthodontic force. Notwithstanding the fact that the overall certainty in evidence inhibits potential recommendations for human trials, the findings of this review provide a direction for future research.
REFERENCES
- 1.Blake M, Retention Bibby K. and stability: a review of the literature. Am J Orthod Dentofacial Orthop. 1998;114(3):299–306. doi: 10.1016/s0889-5406(98)70212-4. [DOI] [PubMed] [Google Scholar]
- 2.Littlewood SJ, Millett DT, Doubleday B, Bearn DR, Worthington HV. Retention procedures for stabilising tooth position after treatment with orthodontic braces. Cochrane Database Syst Rev. 2016 doi: 10.1002/14651858.CD002283.pub4. CD002283. [DOI] [PubMed]
- 3.Vaden JL, Harris EF, Gardner RLZ. Relapse revisited. Am J Orthod Dentofacial Orthop. 1997;111(5):543–553. doi: 10.1016/s0889-5406(97)70291-9. [DOI] [PubMed] [Google Scholar]
- 4.Maltha JC, Kuijpers-Jagtman AM, Von den Hoff JW, Ongkosuwito EM. Relapse revisited—animal studies and its translational application to the orthodontic office. Semin Orthod. 2017;23(4):390–398. [Google Scholar]
- 5.Reitan K. Clinical and histologic observations on tooth movement during and after orthodontic treatment. Am J Orthod. 1967;53(10):721–745. doi: 10.1016/0002-9416(67)90118-2. [DOI] [PubMed] [Google Scholar]
- 6.King GJ, Latta L, Rutenberg J, Ossi A, Keeling SD. Alveolar bone turnover and tooth movement in male rats after removal of orthodontic appliances. Am J Orthod Dentofacial Orthop. 1997;111(3):266–275. doi: 10.1016/s0889-5406(97)70184-7. [DOI] [PubMed] [Google Scholar]
- 7.Franzen TJ, Brudvik P, Vandevska-Radunovic V. Periodontal tissue reaction during orthodontic relapse in rat molars. Eur J Orthod. 2013;35(2):152–159. doi: 10.1093/ejo/cjr127. [DOI] [PubMed] [Google Scholar]
- 8.Franzen TJ, Monjo M, Rubert M, Vandevska-Radunovic V. Expression of bone markers and micro-CT analysis of alveolar bone during orthodontic relapse. Orthod Craniofac Res. 2014;17(4):249–258. doi: 10.1111/ocr.12050. [DOI] [PubMed] [Google Scholar]
- 9.Bartzela T, Türp JC, Motschall E, Maltha JC. Medication effects on the rate of orthodontic tooth movement: a systematic literature review. Am J Orthod Dentofacial Orthop. 2009;135(1):16–26. doi: 10.1016/j.ajodo.2008.08.016. [DOI] [PubMed] [Google Scholar]
- 10.Makrygiannakis MA, Kaklamanos EG, Athanasiou AE. Does common prescription medication affect the rate of orthodontic tooth movement? A systematic review. Eur J Orthod. 2018;40(6):649–659. doi: 10.1093/ejo/cjy001. [DOI] [PubMed] [Google Scholar]
- 11.Shamseer L, Moher D, Clarke M, et al. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: elaboration and explanation. BMJ. 2015;349 doi: 10.1136/bmj.g7647. g7647. [DOI] [PubMed] [Google Scholar]
- 12.Higgins JPT, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. 2019 Mar; Available at: http://www.cochrane-handbook.org. Accessed.
- 13.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ. 2009;339 doi: 10.1136/bmj.b2700. b2700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Joint Formulary Committee. British National Formulary 75. London: BMJ Publishing and the Royal Pharmaceutical Society; 2018. [Google Scholar]
- 15.Hooijmans CR, Rovers MM, de Vries RBM, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. 2014;14(1):43. doi: 10.1186/1471-2288-14-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Borenstein M, Hedges LV, Higgins JPT, Rothstein HR. Introduction to MetaAnalysis 2nd ed. Chichester, UK: Wiley; 2009. [Google Scholar]
- 17.Guyatt GH, Oxman AD, Schunemann HJ, Tugwell P, Knottnerus A. GRADE guidelines: a new series of articles in the Journal of Clinical Epidemiology. J Clin Epidemiol. 2011;64(4):380–382. doi: 10.1016/j.jclinepi.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 18.Lee SH, Kim KA, Anderson S, Kang YG, Kim SJ. Combined effect of photobiomodulation with a matrix metalloproteinase inhibitor on the rate of relapse in rats. Angle Orthod. 2016;86(2):206–213. doi: 10.2319/022515-118.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee HS, Heo HA, Park SH, Lee W, Pyo SW. Influence of human parathyroid hormone during orthodontic tooth movement and relapse in the osteoporotic rat model: a preliminary study. Orthod Craniofac Res. 2018;21(3):125–131. doi: 10.1111/ocr.12226. [DOI] [PubMed] [Google Scholar]
- 20.Chen YP, Han GH, Jin CW, Shi RX, Hou JH. Effect of simvastatin on bone morphogenetic protein-2 expression in the periodontal tissue after rat tooth movement [in Chinese] Chin J Stomatol. 2008;43(1):21–25. [PubMed] [Google Scholar]
- 21.Kim TW, Yoshida Y, Yokoya K, Sasaki T. An ultrastructural study of the effects of bisphosphonate administration on osteoclastic bone resorption during relapse of experimentally moved rat molars. Am J Orthod Dentofacial Orthop. 1999;115(6):645–653. doi: 10.1016/s0889-5406(99)70290-8. [DOI] [PubMed] [Google Scholar]
- 22.Han G, Chen Y, Hou J, et al. Effects of simvastatin on relapse and remodeling of periodontal tissues after tooth movement in rats. Am J Orthod Dentofacial Orthop 2010; 138(5):550.e551–557. doi: 10.1016/j.ajodo.2010.04.026. discussion 550–551. [DOI] [PubMed] [Google Scholar]
- 23.Vieira GM, Chaves SB, Moraes Ferreira VM, Salvatore de Freitas KM, Batista Amorim RF. The effect of simvastatin on relapse of tooth movement and bone mineral density in rats measured by a new method using microtomography. Acta Cir Bras. 2015;30(5):319–327. doi: 10.1590/S0102-865020150050000003. [DOI] [PubMed] [Google Scholar]
- 24.Dolci GS, Portela LV, Onofre de Souza D, Medeiros Fossati AC. Atorvastatin-induced osteoclast inhibition reduces orthodontic relapse. Am J Orthod Dentofacial Orthop. 2017;151(3):528–538. doi: 10.1016/j.ajodo.2016.08.026. [DOI] [PubMed] [Google Scholar]
- 25.Liu Y, Zhang T, Zhang C, et al. Aspirin blocks orthodontic relapse via inhibition of CD4(+) T lymphocytes. J Dent Res. 2017;96(5):586–594. doi: 10.1177/0022034516685527. [DOI] [PubMed] [Google Scholar]
- 26.AlSwafeeri H, ElKenany W, Mowafy M, Karam S. Effect of local administration of simvastatin on postorthodontic relapse in a rabbit model. Am J Orthod Dentofacial Orthop. 2018;153(6):861–871. doi: 10.1016/j.ajodo.2017.10.024. [DOI] [PubMed] [Google Scholar]
- 27.Vieira GM, Falcao DP, Fernandes de Queiroz SB, et al. A novel analysis via micro-CT imaging indicates that chemically modified tetracycline-3 (CMT-3) inhibits tooth relapse after orthodontic movement: a pilot experimental study. Int J Dent. 2019] doi: 10.1155/2019/3524207. [published online ahead of print April 1. [DOI] [PMC free article] [PubMed]
- 28.Fujimura Y, Kitaura H, Yoshimatsu M, et al. Influence of bisphosphonates on orthodontic tooth movement in mice. Eur J Orthod. 2009;31(6):572–577. doi: 10.1093/ejo/cjp068. [DOI] [PubMed] [Google Scholar]
- 29.Liu L, Igarashi K, Haruyama N, Saeki S, Shinoda H, Mitani H. Effects of local administration of clodronate on orthodontic tooth movement and root resorption in rats. Eur J Orthod. 2004;26(5):469–473. doi: 10.1093/ejo/26.5.469. [DOI] [PubMed] [Google Scholar]
- 30.Venkataramana V, Chidambaram S, Reddy BV, Goud EV, Arafath M, Krishnan S. Impact of bisphosphonate on orthodontic tooth movement and osteoclastic count: an animal study. J Int Oral Health. 2014;6(2):1–8. [PMC free article] [PubMed] [Google Scholar]
- 31.Michael JR. New Insights into the molecular mechanisms of action of bisphosphonates. Curr Pharm Des. 2003;9(32):2643–2658. doi: 10.2174/1381612033453640. [DOI] [PubMed] [Google Scholar]
- 32.Hong C, Quach A, Lin L, et al. Local vs. systemic administration of bisphosphonates in rat cleft bone graft: a comparative study. PloS ONE. 2018;13(1):e0190901. doi: 10.1371/journal.pone.0190901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mundy G, Garrett R, Harris S, et al. Stimulation of bone formation in vitro and in rodents by statins. Science. 1999;286(5446):1946–1949. doi: 10.1126/science.286.5446.1946. [DOI] [PubMed] [Google Scholar]
- 34.Stein EA, Farnier M, Waldstreicher J, Mercuri M. Effects of statins on biomarkers of bone metabolism: a randomised trial. Nutr Metab Cardiovasc Dis. 2001;11(2):84–87. [PubMed] [Google Scholar]
- 35.Maritz FJ, Conradie MM, Hulley PA, Gopal R, Hough S. Effect of statins on bone mineral density and bone histomorphometry in rodents. Arterioscler Thromb Vasc Biol. 2001;21(10):1636–1641. doi: 10.1161/hq1001.097781. [DOI] [PubMed] [Google Scholar]
- 36.Steinsvoll S. Periodontal disease, matrix metalloproteinases and chemically modified tetracyclines. Microb Ecol Health Dis. 2004;16(1):1–7. [Google Scholar]
- 37.Golub LM, Lee HM, Ryan ME, Giannobile WV, Payne J, Sorsa T. Tetracyclines inhibit connective tissue breakdown by multiple non-antimicrobial mechanisms. Adv Dent Res. 1998;12(2):12–26. doi: 10.1177/08959374980120010501. [DOI] [PubMed] [Google Scholar]
- 38.Ramamurthy NS, Rifkin BR, Greenwald RA, et al. Inhibition of matrix metalloproteinase-mediated periodontal bone loss in rats: a comparison of 6 chemically modified tetracyclines. J Periodontol. 2002;73(7):726–734. doi: 10.1902/jop.2002.73.7.726. [DOI] [PubMed] [Google Scholar]
- 39.Bildt MM, Bloemen M, Kuijpers-Jagtman AM, Von den Hoff JW. Matrix metalloproteinases and tissue inhibitors of metalloproteinases in gingival crevicular fluid during orthodontic tooth movement. Eur J Orthod. 2009;31(5):529–535. doi: 10.1093/ejo/cjn127. [DOI] [PubMed] [Google Scholar]
- 40.Takahashi I, Nishimura M, Onodera K, et al. Expression of MMP-8 and MMP-13 genes in the periodontal ligament during tooth movement in rats. J Dent Res. 2003;82:646–651. doi: 10.1177/154405910308200815. [DOI] [PubMed] [Google Scholar]
- 41.Xia L, Li H, Wang S, Al-Balaa M, Liu W, Hua X. The expression of extracellular matrix metalloproteinase inducer (EMMPRIN) in the compression area during orthodontic relapse. Eur J Orthod. 2019] doi: 10.1093/ejo/cjz046. [published online ahead of print July 12. [DOI] [PubMed]
- 42.Arias OR, Marquez-Orozco MC. Aspirin, acetaminophen, and ibuprofen: their effects on orthodontic tooth movement. Am J Orthod Dentofacial Orthop. 2006;130(3):364–370. doi: 10.1016/j.ajodo.2004.12.027. [DOI] [PubMed] [Google Scholar]
- 43.Liu Y, Wang L, Kikuiri T, et al. Mesenchymal stem cell-based tissue regeneration is governed by recipient T lymphocytes via IFN-gamma and TNF-alpha. Nat Med. 2011;17(12):1594–1601. doi: 10.1038/nm.2542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Reagan-Shaw S, Nihal M, Ahmad N. Dose translation from animal to human studies revisited. FASEB J. 2008;22(3):659–661. doi: 10.1096/fj.07-9574LSF. [DOI] [PubMed] [Google Scholar]
- 45.Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. 2010;8(6):e1000412. doi: 10.1371/journal.pbio.1000412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Charan J, Kantharia ND. How to calculate sample size in animal studies? J Pharmacol Pharmacother. 2013;4(4):303–306. doi: 10.4103/0976-500X.119726. [DOI] [PMC free article] [PubMed] [Google Scholar]