Clinical History
A 53‐year‐old woman presented with a 1‐month history of severe headache and intractable vomiting. Physical examination revealed left facial palsy and generalized weakness of the extremities (grade IV/V all extremities) without other localizing signs. She had no known underlying disease and there was no significant family history. MRI of the brain disclosed multiple ill‐defined high signal lesions in T2W at left lower pons, left thalamus, subcortical regions of temporal, parietal, and frontal lobes bilaterally, and periventricular white matter. Nodular enhancement was noted in the left frontal lobe lesion (Figure 1a), left thalamus (not shown) and left pons (Figure 1b). Irregular leptomeningeal enhancement was also noted diffusely. The largest mass present at the left frontal lobe was 2 cm in largest dimension and showed marked peritumoral vasogenic edema (Figure 1a). High resolution chest CT revealed an ill‐defined mass at the posterior basal segment of the right lower lobe, 3.2 cm in greatest dimension (not shown). There were also multiple small nodules involving both lungs, ranging from 0.4 to 0.5 cm. Stereotactic biopsy of the left frontal lobe lesion was performed.
Figure 1.
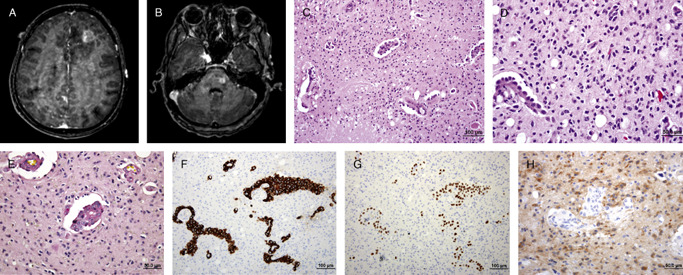
Microscopic Pathology
Sections showed papillary/perivascular structures present in variably necrotic and hypercellular backgrounds (Figure 1c). The former contained hyalinized vascular cores lined by single layers of rectangular to cuboidal cells. Some of these cells had intracytoplasmic vacuoles. These cells contained moderately pleomorphic hyperchromatic to vesicular nuclei with occasionally prominent nucleoli and frequent mitoses. The hypercellular parenchymal component contained moderately pleomorphic hyperchromatic nuclei with indistinct eosinophilic cytoplasm embedded within a fibrillar background (Figure 1d). Mitotic figure were exceptionally rare in this area. The intracytoplasmic vacuoles in the papillary structures were positive for mucicarmine (Figure 1e). Immunohistochemically, these cells strongly expressed CK7 (Figure 1f) and TTF‐1 (Figure 1g), but were CK 20 negative (not shown). The interspersed parenchymal cells were positive for GFAP (not shown), p53 protein (not shown), and the R132H mutant form of IDH‐1 protein (Figure 1h). The MIB‐1 labeling index in this area was <1%. FISH studies for chromosome 1p and 19q status were non‐informative despite multiple attempts. What is your diagnosis?
Diagnosis
Intracranial tumor‐to‐tumor metastasis: Metastatic pulmonary adenocarcinoma to a diffuse astrocytoma (WHO grade II).
Discussion
Two pathologic phenomenon, “collision tumor” and “tumor to tumor metastasis” both result in the intermingling of two separate primary neoplasms. While “collision tumor” results from growth of neighboring tumors within a shared space, “tumor to tumor metastasis” results from hematogenous spread of a primary donor usually extracranial tumor to a recipient/host tumor. The criteria used to defined “tumor to tumor metastasis” are as follows: (i) more than one primary tumor must exist; (ii) the recipient tumor must be a true neoplasm; (iii) the metastatic neoplasm should show established growth in the recipient tumor, not resulting from contiguous growth of an adjacent tumor or tumor embolism; and (iv) lymphatic metastasis to a site of pre‐existing lymphoid malignancy are excluded 1, 7. Tumor to tumor metastasis is an unusual phenomenon. The most common donor tumors are breast and lung carcinoma 7,while the most common benign and malignant recipient tumors are meningioma and renal cell carcinoma, respectively 7, 10. Metastasis to primary intracranial glioma is exceptionally rare and to our knowledge represents the first report of metastatic carcinoma to an IDH1mutant low‐grade diffuse glioma 9. To date, 14 cases of tumor metastasis to glioma, including our case, have been reported in the literature 5, 6, 7, 8. The three most common donors are lung carcinoma, melanoma, and breast carcinoma in descending order. The top three most common recipients are oligodendroglioma, anaplastic astrocytoma, and ependymoma. Nonetheless, some of the older reports are questionable and poorly substantiated given the lack of immunohistochemical and genetic tools of those times. For example, malignant epithelioid cells could represent epithelial‐like areas or frank epithelial metaplasia within an anaplastic astrocytoma or glioblastoma 2, 6. Similarly, without appropriate immunohistochemical confirmation, metastatic melanoma may occasionally mimic diffuse glioma 6. Although in general, the pathogenesis of cancer metastasis has been described using the “seed and soil” hypothesis 3, 4, there are currently no proven molecular explanations for “tumor‐to‐tumor metastasis” and why gliomas so rarely serve as recipients in this phenomenon.
References
- 1. Campbell LV Jr, Gilbert E, Chamberlain CR Jr, Watne AL (1968) Metastases of cancer to cancer. Cancer 22:635–643 [DOI] [PubMed] [Google Scholar]
- 2. Kleihues P, Burger PC, Aldape KD, Brat DJ, Biernat W, Bigner DD (2007) Glioblastoma. In: WHO Classification of tumours of the central nervous system, Louis DN, Ohgaki H, Wiestler OD, Cavenee WK (eds), pp 33–49, IARC: Lyon: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Langley RR, Fidler IJ (2011) The seed and soil hypothesis revisited—The role of tumor‐stroma interactions in metastasis to different organs. Int J Cancer 128:2527–2535 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mathot L, Stenninger J (2012) Behavior of seeds and soil in the mechanism of metastasis: a deeper understanding. Cancer Sci 103:626–631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Mizutani H, Suzuki H, Wakabayashi S, Mitake A, Mizutani M, Banno T, Oba S, Tada T, Kuhara H (1987) A case of metastasis from a colon carcinoma to an intracranial oligodendroglioma. Gan No Rinsho 33:1733–1739. [PubMed] [Google Scholar]
- 6. Mörk SJ, Rubinstein LJ (1988) Metastatic carcinoma to glioma: a report of three cases with a critical review of the literature. J Neurol Neurosurg Psychiatry 51:256–259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Takei H, Powell SZ (2009) Tumor‐to‐tumor metastasis to the central nervous system. Neuropathology 29:303–308. [DOI] [PubMed] [Google Scholar]
- 8. Tally PW, Laws ER Jr, Scheithauer BW (1988) Metastases of central nervous system neoplasms. Case report . J Neurosurg 68:811–816. [DOI] [PubMed] [Google Scholar]
- 9. Rivera‐Zengotita M, Yachnis AT (2012) Gliosis versus glioma?: don't grade until you know. Adv Anat Pathol 19:239–249. [DOI] [PubMed] [Google Scholar]
- 10. Zhou Q, Chang H, Gao Y, Cui L (2013) Tumor‐to‐tumor metastasis from pituitary carcinoma to radiation‐induced meningioma. Neuropathology 33:209–212 [DOI] [PubMed] [Google Scholar]


