Abstract
The authors tested the hypothesis that a valsartan/cilnidipine combination would suppress the home morning blood pressure (BP) surge (HMBPS) more effectively than a valsartan/hydrochlorothiazide combination in patients with morning hypertension, defined as systolic BP (SBP) ≥135 mm Hg or diastolic BP ≥85 mm Hg assessed by a self‐measuring information and communication technology–based home BP monitoring device more than three times before either combination's administration. This was an 8‐week prospective, multicenter, randomized, open‐label clinical trial. The HMBPS, which is a new index, was defined as the mean morning SBP minus the mean nocturnal SBP, both measured on the same day. The authors randomly allocated 129 patients to the valsartan/cilnidipine (63 patients; mean 68.4 years) or valsartan/hydrochlorothiazide (66 patients; mean 67.3 years) combination groups, and the baseline HMBPS values were 17.4 mm Hg vs 16.9 mm Hg, respectively (P = .820). At the end of the treatment period, the changes in nocturnal SBP and morning SBP from baseline were significant in both the valsartan/cilnidipine and valsartan/hydrochlorothiazide groups (P < .001): −5.0 vs −10.0 mm Hg (P = .035) and −10.7 vs −13.6 mm Hg (P = .142), respectively. HMBPS was significantly decreased from baseline in both groups (P < .001), but there was no significant difference between the two groups: 14.4 mm Hg vs 14.0 mm Hg, respectively (P = .892). Valsartan/cilnidipine could not significantly suppress HMBPS compared with valsartan/hydrochlorothiazide. Large‐scale randomized controlled studies are needed to assess how reducing HMBPS will affect future cardiovascular outcomes. The information and communication technology–based home BP monitoring device may become an alternative to ambulatory BP monitoring, which has been a gold standard to measure nocturnal BP and the morning BP surge.
Keywords: automatic information and communication technology–based home blood pressure monitoring device, home morning blood pressure surge, morning blood pressure, nocturnal blood pressure, valsartan/cilnidipine combination
1. INTRODUCTION
Several studies have shown that measurements of out‐of‐office blood pressure (BP) such as ambulatory BP and home BP (HBP) are more strongly associated with cardiovascular disease than office BP.1, 2, 3, 4 Among several BP parameters that can be obtained by these monitoring methods, a higher morning surge was shown to be strongly associated with cardiovascular disease and target organ damage5, 6 independently of the mean 24‐hour ambulatory BP level and nocturnal BP dipping status in both patients with hypertension and community‐dwelling persons.7, 8, 9 Controlling the morning BP surge is an important strategy for reducing cardiovascular disease and target organ damage, but few studies have targeted the decrease of morning BP surge.
The use of home BP monitoring (HBPM), which is recommended by many hypertension guidelines,10, 11, 12, 13, 14 has spread widely because of its simplicity, convenience, and tolerability. A self‐measured information and communication technology (ICT)–based HBPM method was recently developed.15, 16, 17 In the past, nocturnal BP had been a “blind spot” of HBPM, and only ambulatory BP monitoring (ABPM) could measure nocturnal BP and morning BP surge. It was also not possible to measure the home morning BP surge (HMBPS). No study has investigated these indices.
It has been demonstrated that cilnidipine, which is a unique L/N‐type calcium channel blocker (CCB),18 significantly and markedly reduced morning systolic BP (SBP) in patients with higher baseline morning SBP19 and restored the abnormal nocturnal BP dipping status toward a normal dipping pattern.20 Previous studies also demonstrated that when morning hypertension was treated with diuretics, individuals who were nondippers shifted toward becoming dippers.21 It was also demonstrated that hydrochlorothiazide (HCTZ) reduced the nocturnal BP level more effectively than a CCB, while morning BP was preferentially reduced by a CCB rather than HCTZ.22 We thus hypothesized that a valsartan/cilnidipine combination would reduce HMBPS more effectively than a valsartan/HCTZ combination (Figure S1).
We tested our hypothesis in SUNLIGHT (Study on Uncontrolled Morning Surge for N‐type CCB and Low Dose of HCTZ, Using the Internet Through Blood Pressure Data Transmission System), a randomized controlled trial designed to compare HMBPS levels assessed by an ICT‐based HBPM device between groups of patients with morning hypertension treated with a valsartan/cilnidipine combination and patients treated with a valsartan/HCTZ combination groups.
2. METHODS
2.1. Study design
SUNLIGHT is an 8‐week, prospective, multicenter, randomized, open‐label, clinical trial investigating the effect of treatment with a valsartan/cilnidipine combination (80 mg/10 mg) or a valsartan/HCTZ combination (80 mg/12.5 mg) on HMBPS in patients with morning hypertension. Patients were enrolled between December 2015 and June 2016. This study was conducted in accord with the principles of the Declaration of Helsinki. The study protocol was approved by the ethics committee of the Jichi Medical University School of Medicine (Shimotsuke, Japan), and all of the patients gave written informed consent to participate. The protocol of the SUNLIGHT study was registered on the University Trials Registry (UMIN‐CTR) website (trial No. UMIN000019997). All data and procedures of this study were regularly monitored by Satt Co., Ltd., a contract research organization.
The study design is illustrated in Figure 1. During a run‐in period, all patients underwent ≥4 weeks of monotherapy with valsartan (80 mg). After the run‐in period, eligible patients were enrolled in the study and randomly allocated to either the valsartan/cilnidipine combination (cilnidipine group) or the valsartan/HCTZ combination (HCTZ group) on an electronic data capture system. The allocated patients underwent 8 weeks of this treatment. Physicians instructed the patients regarding adherence to the medication and confirmed the occurrence of adverse events.
Figure 1.
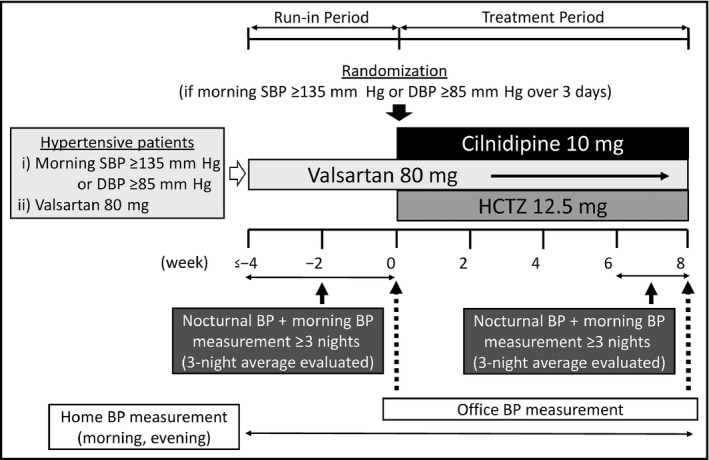
Study design. BP indicates blood pressure; DBP, diastolic blood pressure; HCTZ, hydrochlorothiazide; SBP, systolic blood pressure
2.2. Study patients
The participants who met all of the following inclusion criteria were eligible: (1) patients with morning hypertension, defined as morning SBP ≥135 mm Hg or diastolic BP (DBP) ≥85 mm Hg, as assessed by HBPM, more than three times during the run‐in period; (2) patients receiving valsartan (80 mg/d) over 4 weeks; and (3) patients older than 20 years.
The exclusion criteria are as follows: (1) pregnant or possibly pregnant patients; (2) patients taking a CCB; (3) patients taking a diuretic; (4) patients who were intolerant to angiotensin II receptor blocker (ARB)s, CCBs, or diuretics; (5) and patients considered inappropriate to participate in this study by an attending physician.
2.3. BP measurements
2.3.1. Home BP
The patients measured their own BP at home using an automatic ICT‐based device (HEM‐7252G‐HP, Omron Healthcare) according to the cuff‐oscillometric principle. All data obtained by the device were transmitted automatically to a cloud‐based remote monitoring system, the Medical LINK software program provided by Omron Healthcare,15 and the data were managed in an independent facility (Satt Co., Ltd).
Self‐measured HBP values were obtained according to the current Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014).10 The patients were instructed to measure their HBP in a sitting position in an appropriate environment after resting for 1 to 2 minutes with their legs not crossed. A 14.5‐cm wide and 46.6‐cm long (target arm girth: 22–32 cm) cuff was used. The arm cuff position was maintained at the heart level. The patients measured their HBP during the study period, and the HBPM protocol was as follows: two measurements in the morning and two measurements at bedtime. The patients measured their morning BP within 1 hour after waking, after urination, before breakfast, and before ingesting medications. The at‐bedtime BP was measured just before the patient went to bed.
The patients’ nocturnal HBP was measured during sleep periods. The nighttime BP ICT‐based device was preset to take three BP measurements at fixed times: 2 am, 3 am, and 4 am. The patients were instructed to wear the BP cuff and press the button to start the timer when they went to bed and to measure their nocturnal HBP. Nocturnal HBPM was conducted as follow periods: (1) over three nights (not necessarily consecutive) during the run‐in period; and (2) over three nights (not necessarily consecutive) at the end of the treatment period (during weeks 6 to 8).
HMBPS was defined as the average morning SBP minus the average nocturnal SBP, both measured on the same day. We evaluated the 3‐day average of HMBPS between the cilnidipine group and the HCTZ group both in the run‐in period and at the end of the treatment period. If the patients measured an HMBPS on more than 3 days, we selected maximum 5‐day HMBPS values close to the randomization date and the end of the treatment period without selection bias. We excluded HMBPS values that were not measured on the same day.
We calculated the average of morning BP and evening BP (MEave), both measured on the same day. We then subclassified the patients’ HBP dipping status according to the percentage of the nocturnal SBP reduction [(1−nocturnal SBP/MEave SBP) × 100] as follows: extreme dipper when the nocturnal SBP reduction was ≥20%; dipper when the reduction was ≥10% but <20%; nondipper when the reduction was ≥0% but <10%; and riser when the reduction was <0%. The nocturnal SBP used to calculate the dipping status was the value measured on the night of the day when the MEave was measured.
2.3.2. Office BP
Office BP was measured at each participating medical center using their own validated cuff oscillometric devices according to the JSH 2014 guideline.10 BP was measured after the patient rested for a few minutes in a seated position on a chair with their legs not crossed in a quiet environment. Two consecutive measurements were taken at 1‐ to 2‐minute intervals, and the average of the two measurements was used to define office BP.
2.4. Primary and secondary outcomes
The primary outcome was defined as HMBPS. We compared HMBPS between the cilnidipine and HCTZ groups at the end of the treatment period.
Secondary outcomes were as follows: the changes in HMBPS, nocturnal BP and heart rate (HR), morning BP and HR, evening BP and HR, MEave, and office BP and HR from baseline to the end of the treatment period. We compared these outcomes between the cilnidipine group and the HCTZ group.
2.5. Sample size
Assuming that the HCTZ group would show reduced nocturnal SBP levels that were 2 to 3 mm Hg greater than those of the cilnidipine group and that the cilnidipine group would show reduced morning SBP levels 2 to 3 mm Hg greater than those of the HCTZ group,19, 22 we set the HMBPS difference between the two groups at 5 mm Hg and the standard deviation of 10 mm Hg. Taking these differences into consideration, we calculated that the number of patients required to detect a difference with a statistical power of 80% and a two‐sided significance level of 0.05 was 65 patients in each group (65 patients × 2 groups). Adjusting by 20% for loss to follow‐up or dropout, a total of 160 patients was needed at randomization.
2.6. Statistical analysis
We analyzed the data using a full‐analysis set. Fisher exact test was used for the allocated patients of the full‐analysis set to determine background differences and to compare proportions. Student t test was performed on the primary and secondary outcomes from baseline to the end of the treatment period to determine any difference between the cilnidipine group and the HCTZ group, and paired t test was used to determine intragroup differences. All statistical analyses were performed using SAS version 9.4 software (SAS Institute Inc) at the Jichi Medical University Center of Global Home and Ambulatory BP Analysis, Shimotsuke, Japan. A P value <.05 was considered significant.
3. RESULTS
3.1. Patient disposition and baseline characteristics
A total of 137 patients were randomly allocated to the cilnidipine group (68 patients) and HCTZ group (69 patients). In the cilnidipine and HCTZ groups, 63 and 66 patients completed the treatment, respectively. The reasons for withdrawal during the treatment period included withdrawn consent, lack of drug efficacy, and considered inappropriate by the attending physicians; no adverse events were identified (Figure 2). Table 1 provides the demographic variables and clinical characteristics of the study patients. There were no significant differences in demographic variables between the groups. However, among the clinical characteristics, the morning and office HR values were significantly higher in the cilnidipine group than the HCTZ group, and nocturnal HR also tended to be higher in the cilnidipine group compared with the HCTZ group.
Figure 2.
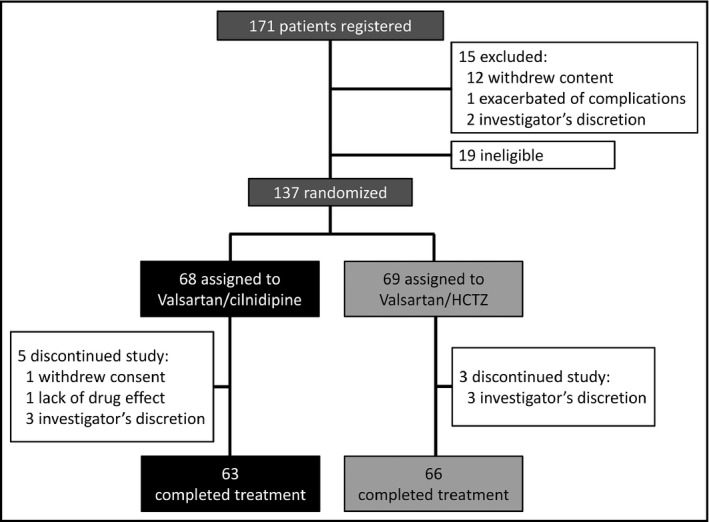
Patient disposition. HCTZ indicates hydrochlorothiazide
Table 1.
Clinical characteristics of the study patients
| Variable | Valsartan/cilnidipine (n = 63) | Valsartan/HCTZ (n = 66) | P value |
|---|---|---|---|
| Age, y | 68.4 ± 13.0 | 67.3 ± 11.7 | .611 |
| Men | 27 (42.9) | 29 (43.9) | 1.000 |
| Body mass index, kg/m2 | 23.8 ± 4.4 | 25.0 ± 4.6 | .155 |
| Current smoker | 5 (7.9) | 10 (15.2) | .274 |
| Daily drinker | 23 (36.5) | 27 (40.9) | .718 |
| Dyslipidemia | 31 (48.8) | 33 (50.0) | 1.000 |
| Diabetes mellitus | 14 (22.0) | 11 (16.7) | .588 |
| Prior stroke | 6 (9.8) | 6 (9.5) | 1.000 |
| Prior coronary heart disease | 3 (4.9) | 3 (4.8) | 1.000 |
| BP parameter | |||
| Nocturnal SBP, mm Hg | 124.3 ± 15.6 | 125.8 ± 15.2 | .597 |
| Nocturnal DBP, mm Hg | 76.4 ± 9.4 | 76.2 ± 10.2 | .918 |
| Nocturnal HR, bpm | 63.1 ± 8.4 | 60.3 ± 7.9 | .065 |
| Morning SBP, mm Hg | 142.4 ± 14.5 | 142.4 ± 15.7 | .988 |
| Morning DBP, mm Hg | 87.0 ± 10.3 | 87.7 ± 10.9 | .698 |
| Morning HR, bpm | 68.4 ± 8.2 | 65.5 ± 8.3 | .049 |
| Evening SBP, mm Hg | 133.3 ± 14.2 | 132.7 ± 14.0 | .806 |
| Evening DBP, mm Hg | 80.6 ± 10.8 | 79.7 ± 9.3 | .595 |
| Evening HR, bpm | 71.8 ± 10.5 | 70.6 ± 10.3 | .506 |
| MEave SBP, mm Hg | 137.9 ± 13.3 | 137.5 ± 13.8 | .891 |
| MEave DBP, mm Hg | 83.8 ± 9.8 | 83.7 ± 9.6 | .948 |
| Home morning SBP surge, mm Hg | 17.4 ± 13.4 | 16.9 ± 13.8 | .820 |
| Home morning DBP surge, mm Hg | 10.7 ± 9.1 | 11.3 ± 7.9 | .663 |
| Office SBP, mm Hg | 143.9 ± 17.7 | 144.2 ± 18.1 | .938 |
| Office DBP, mm Hg | 82.9 ± 13.3 | 85.2 ± 11.6 | .287 |
| Office HR, bpm | 75.9 ± 9.0 | 72.5 ± 10.2 | .046 |
| Dipping statusa | |||
| Extreme dipper | 8 (14.0) | 6 (10.0) | .501 |
| Dipper | 20 (35.1) | 22 (36.7) | .859 |
| Nondipper | 20 (35.1) | 22 (36.7) | .859 |
| Riser | 9 (15.8) | 10 (16.7) | .898 |
Abbreviations: BP, blood pressure; bpm, beats per minute; DBP, diastolic blood pressure; HR, heart rate; MEave, average of morning and evening blood pressure; SBP, systolic blood pressure.
Data are expressed as the mean ± standard deviation or number (percentage).
Variables were obtained from 57 and 60 patients in the valsartan/cilnidipine combination and valsartan/hydrochlorothiazide (HCTZ) combination groups, respectively.
3.2. Changes in each BP parameter
3.2.1. Home BP and HR
Concerning the primary outcome, in the cilnidipine group, HMBPS at the end of the treatment period was 14.4 ± 13.0 mm Hg. In the HCTZ group, the corresponding HMBPS value was 14.0 ± 15.2 mm Hg. There was no significant difference in HMBPS between the two groups at the end of the treatment period (P = .892) (Figure 3).
Figure 3.
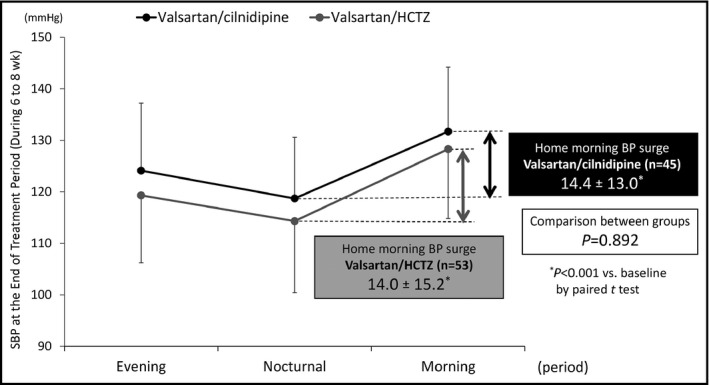
The comparison of morning systolic blood pressure (SBP) surge at the end of the treatment period between the valsartan/cilnidipine combination and valsartan/hydrochlorothiazide (HCTZ) combination. Data are presented as mean ± standard deviation. The P value refers to the comparison of home morning blood pressure (BP) surge between the valsartan/cilnidipine combination and the valsartan/HCTZ combination
Regarding the secondary outcomes, the changes in HMBPS from baseline to the end of the 8‐week treatment period were significant in both groups (both P < .001): −5.7 mm Hg in the cilnidipine group (n = 44) and −4.7 mm Hg in the HCTZ group (n = 50). There was no significant difference in the changes of HMBPS from baseline to the end of the 8‐week treatment period between the two groups (P = .665).
In the cilnidipine group, the changes in nocturnal SBP and DBP from baseline to the end of the 8‐week treatment period were −5.0 mm Hg (P < .001) and −2.3 mm Hg (P = .067), and in the HCTZ group, the corresponding changes were −10.0 mm Hg (P < .001) and −4.6 mm Hg (P < .001), respectively. In the HCTZ group, the nocturnal SBP was significantly decreased (P = .035) and the DBP also tended to be decreased but not significantly (P = .092) compared with the cilnidipine group (Table 2).
Table 2.
Changes in BP parameters
| Variable | Valsartan/cilnidipine (n = 63) | Valsartan/HCTZ (n = 66) | Comparison between groups | ||
|---|---|---|---|---|---|
| Change, mean ± standard deviation | P value | Change, mean ± standard deviation | P value | P value | |
| Nocturnal SBP, mm Hg | −5.0 ± 12.3 | <.001 | −10.0 ± 10.5 | <.001 | .035 |
| Nocturnal DBP, mm Hg | −2.3 ± 7.7 | .067 | −4.6 ± 5.5 | <.001 | .092 |
| Nocturnal HR, bpm | 0.2 ± 5.3 | .888 | −1.2 ± 3.9 | .022 | .137 |
| Morning SBP, mm Hg | −10.7 ± 11.7 | <.001 | −13.6 ± 10.7 | <.001 | .142 |
| Morning DBP, mm Hg | −4.5 ± 5.4 | <.001 | −6.5 ± 5.6 | <.001 | .042 |
| Morning HR, bpm | 1.1 ± 6.6 | .378 | 0.9 ± 4.3 | .115 | .844 |
| Evening SBP, mm Hg | −9.3 ± 9.8 | <.001 | −13.1 ± 10.7 | <.001 | .042 |
| Evening DBP, mm Hg | −4.8 ± 6.2 | <.001 | −5.6 ± 5.6 | <.001 | .411 |
| Evening HR, bpm | 0.3 ± 6.4 | .479 | 0.4 ± 5.5 | .549 | .913 |
| MEave SBP, mm Hg | −10.1 ± 9.7 | <.001 | −13.3 ± 9.7 | <.001 | .065 |
| MEave DBP, mm Hg | −4.7 ± 5.0 | <.001 | −5.9 ± 5.3 | <.001 | .203 |
| Office SBP, mm Hg | −11.8 ± 15.9 | <.001 | −13.8 ± 16.4 | <.001 | .490 |
| Office DBP, mm Hg | −5.6 ± 10.4 | <.001 | −8.3 ± 9.5 | <.001 | .124 |
| Office HR, mm Hg | −1.3 ± 6.9 | .085 | 0.5 ± 9.4 | .725 | .212 |
Abbreviations: bpm, beats per minute; BP, blood pressure; DBP, diastolic blood pressure; HCTZ, hydrochlorothiazide; HR, heart rate; MEave, the average of morning and evening blood pressure; SBP, systolic blood pressure.
In the cilnidipine group, the changes in morning SBP and DBP from baseline to the end of the 8‐week treatment were −10.7 mm Hg (P < .001) and −4.5 mm Hg (P < .001), and in the HCTZ group, the corresponding changes were −13.6 mm Hg (P < .001) and −6.5 mm Hg (P < .001), respectively. There was no significant difference in the changes of morning SBP from baseline to the end of the 8‐week treatment period between the two groups (P = .142). Morning DBP was significantly decreased in the HCTZ group compared with the cilnidipine group (P = .042) (Table 2).
In the cilnidipine group, the changes in evening SBP and DBP from baseline to the end of the 8‐week treatment were −9.3 mm Hg (P < .001) and −4.8 mm Hg (P < .001), and the corresponding changes in the HCTZ group were −13.1 mm Hg (P < .001) and −5.6 mm Hg (P < .001), respectively. Evening SBP was significantly decreased in the HCTZ group compared with the cilnidipine group (P = .042). There was no significant difference in the changes of evening DBP between the two groups (P = .411) (Table 2).
In the cilnidipine group, the changes in MEave SBP and DBP from baseline to the end of the 8‐week treatment period were −10.1 mm Hg (P < .001) and −4.7 mm Hg (P < .001), and the corresponding changes in the HCTZ group were −13.3 mm Hg (P < .001) and −5.9 mm Hg (P < .001), respectively. There were no significant differences in the changes of MEave SBP (P = .065) and MEave DBP (P = .203) between the two groups (Table 2). In addition, there were no significant differences in the HBP dipping status after the 8‐week treatment period between the two groups (Table S1).
The nocturnal HR in the HCTZ group was significantly decreased from baseline to the end of the 8‐week treatment (−1.2 beats per minute, P = .022), with no significant difference between the two groups (P = .137). Except for nocturnal HR in the HCTZ group, there were no significant reductions of HR from baseline in both groups. There was no significant difference in the reduction of HR from baseline to the end of the treatment period between the two groups (Table 2).
3.2.2. Office BP and HR
In the cilnidipine group, the changes in office SBP and DBP from baseline to the end of the 8‐week treatment period were −11.8 mm Hg (P < .001) and −5.6 mm Hg (P < .001), and in the HCTZ group, the corresponding changes were −13.8 mm Hg (P < .001) and −8.3 mm Hg (P < .001), respectively. There were no significant differences in the reduction of office SBP and DBP values between the two groups. Concerning HR, there were no significant reductions from baseline in both groups and no significant difference in the reduction of HR from baseline to the end of the treatment period (Table 2).
3.3. Safety
3.3.1. Adverse events
Neither serious adverse events nor drug‐related adverse events were reported in either group during the study period.
3.3.2. Adherence
The overall compliance during the study period was good in both groups. There was no patient with a compliance rate <75% in either group.
4. DISCUSSION
This study compared HMBPS measured by an ICT‐based HBPM device between valsartan/cilnidipine combination and valsartan/HCTZ combination groups of patients with morning hypertension, defined as SBP ≥135 mm Hg or DBP ≥85 mm Hg, as assessed by HBPM more than three times before either combination's administration. Although both the valsartan/cilnidipine combination and the valsartan/HCTZ combination significantly reduced the patients’ nocturnal, morning, evening, and MEave BP levels, including HMBPS levels from baseline, there were no significant differences in HMBPS between the two groups at the end of the treatment period.
These findings indicate that a valsartan/cilnidipine combination could control HMBPS level similar to a valsartan/HCTZ combination. A previous report indicated that an ARB/HCTZ combination reduced the nocturnal SBP level by 4.2 mm Hg more than an ARB/CCB combination,22 which supported our present finding that the HCTZ group reduced the nocturnal SBP level by 5.0 mm Hg more compared with the cilnidipine group. In addition, the HCTZ group also reduced the morning SBP level by 2.9 mm Hg more compared with the cilnidipine group, although the difference was not significant. This is why we could not show our hypothesis that the cilnidipine group would show a greater reduction in HMBPS compared with the HCTZ group.
The ACHIEVE‐ONE (Ambulatory Blood Pressure Control and Home Blood Pressure [Morning and Evening] Lowering by N‐Channel Blocker Cilnidipine) trial19 demonstrated that cilnidipine reduced morning SBP level more markedly in patients with higher morning SBP and HR (≥70 beats per minute) at baseline, which meant that cilnidipine could suppress increased morning sympathetic activity. Comparing the mean morning SBP levels at baseline between the cilnidipine group in the SUNLIGHT study and the ACHIEVE‐ONE trial (in which morning SBP was determined as the 3‐day average of morning BP assessed by HBPM), the inclusion criteria of these two clinical trials were almost the same, and the cilnidipine group of the SUNLIGHT study showed apparently lower morning SBP than those of the ACHIEVE‐ONE trial (142.4 mm Hg vs 152.9 mm Hg).19 The SUNLIGHT trial might have obtained different results if more patients with higher morning SBP levels at baseline compared with those of the present study were selected.
In the present study, the baseline HMBPS levels of the cilnidipine and HCTZ groups were 17.4 mm Hg and 16.9 mm Hg, respectively. These HMBPS levels were apparently lower than those in previous studies, even taking into consideration that the morning surge in the previous studies was assessed by ABPM. In the ACHIEVE‐ONE trial,20 the mean sleep‐trough morning SBP surge assessed by ABPM (which is defined as the average of SBPs during the first 2 hours after wake‐up time minus the average SBP of three readings centered on the lowest nocturnal reading7) was 30.4 mm Hg, and other clinical trials also demonstrated a greater number of higher sleep‐trough morning SBP surge levels than those of the present study: 34 mm Hg in the JMU‐ABPM (Jichi Medical University Ambulatory Blood Pressure Monitoring) study7 and 40.3 mm Hg in the ARTEMIS (International Ambulatory Blood Pressure Registry: Telemonitoring of Hypertension and Cardiovascular Risk Project) study of a Japanese database.23
Although a previous study demonstrated that there was an ethnic difference in the degree of morning BP surge and the sleep‐trough morning surge was lower in European than in Japanese patients with hypertension after adjusting for age and 24‐hour mean BP levels,23 the European database demonstrated higher sleep‐trough morning surge levels than those of the present study: 20.7 mm Hg in the population‐based IDACO (International Database of Ambulatory Blood Pressure in Relation to Cardiovascular Outcome) trial,9 28 mm Hg in the PIUMA (Progetto Ipertensione Umbria Monitoraggio Ambulatoriale) study,24 and 22.9 mm Hg in the ARTEMIS study of European database.23 We could not compare these results directly with those of the present study because of the difference of morning BP surge monitoring; however, the baseline data of the present study imply that our patients did not show higher HMBPS levels and that it was difficult to detect a significant difference in this study population.
Our results also revealed that a valsartan/cilnidipine combination could significantly decrease morning, evening, MEave, and office BP levels from baseline without significant differences compared with the valsartan/HCTZ combination. An ARB is one of the first‐line antihypertensive drugs for most patients with hypertension; however, as in the present study, there are few patients who can achieve target BP levels with monotherapy.11 A meta‐analysis has also shown that combining two agents from any two classes of antihypertensive drugs increases BP reduction much more than dose increasing the dose of one agent.25
A large number of clinical trials have indicated that renin‐angiotensin‐aldosterone system inhibitor/CCB combination regimens decrease the number of cardiovascular disease events,26, 27 and the combination showed good tolerance.28 Patients with morning hypertension showed increased sympathetic activity6, 29 and increased renin‐angiotensin‐aldosterone system activity.30 In the present study, the valsartan/cilnidipine combination reduced morning SBP and SBP surge levels in patients who had already been administered valsartan. This additive BP‐lowering effect of cilnidipine may be based on the L‐type and N‐type CCB actions, which differ from renin‐angiotensin‐aldosterone system inhibitor actions.
A previous study also confirmed this different mechanism. Switching cilnidipine 10 mg monotherapy to a valsartan/cilnidipine combination (80 mg/10 mg) showed a significant additive BP‐lowering effect.31 A valsartan/cilnidipine combination would be an optimal combination for patients with morning hypertension.
This is the first study to investigate HMBPS using a self‐measured ICT‐based HBPM device. We successfully measured HMBPS (over three times per patient) of 98 patients (76.0% of the total group) using this device, and our findings demonstrate that it is possible for patients to self‐measure the morning BP surge at home. We previously demonstrated that the nocturnal BP level measured using an HBPM device was almost identical to the nocturnal BP level measured by ABPM, which has been a gold standard for measuring nocturnal BP, and nocturnal BPs measured by HBPM and ABPM were similarly correlated with hypertensive target organ damage.32 Another study also demonstrated that HBPM was well accepted for the assessment of nocturnal BP and the detection of nondippers.33 In addition, in the NOCTURNE study,17 we demonstrated for the first time that nocturnal HBPM with an ICT‐based device showed high feasibility and tolerability, even in patients who had no prior experience with the device. ICT‐based HBPM could thus have the potential to be an alternative to ABPM for the measurement of nocturnal BP and morning BP surge.
In the present study, we used the average of three nocturnal BPs taken at 2 am, 3 am, and 4 am. In the J‐HOP (Japan Morning Surge Home Blood Pressure) study,34 we observed that the same average of three nocturnal BPs at 2 am, 3 am, and 4 am was significantly correlated with hypertensive target organ damage. We used the definition of the average morning SBP measured twice within 1 hour after waking minus the average nocturnal SBP (both measured on the same day), which is a new index. Two main definitions of the morning BP surge measured by ABPM have been used: the sleep‐through morning surge and the prewaking morning surge, defined as the average of SBPs during the first 2 hours after waking up minus the average SBP during the 2 hours just before waking.7 There is no clinical study evaluating the differences between the morning surge measured by HBPM and these two definitions of morning surge measured by ABPM. Considering the large amount of evidence of nocturnal BP and morning BP both measured by HBPM,35, 36, 37, 38 this new index may have clinical significance. A future study could clarify the different impact of these morning BP surge parameters.
5. STUDY STRENGTHS AND LIMITATIONS
The strength of this study is that the BP levels were accurate and without selection bias because we used an ICT‐based HBPM device. This study also has some limitations. First, the sample size was small. Second, we did not set the criteria of the timing of antihypertensive administration. The differences in the administration timing might have affected the results. Third, we used a fixed‐dose valsartan/cilnidipine combination tablet (80 mg/10 mg per tablet) and a fixed‐dose valsartan/HCTZ combination tablet (80 mg/12.5 mg per tablet), which are the standard doses for Japanese patients. Further investigations are required to determine whether a dose‐dependent effect is observed in reducing HMBPS. Fourth, sodium sensitivity39 and the prevalence of the patients with chronic kidney disease17, 40 in this study population might have affected the changes of nocturnal BP levels. Fifth, the definition of morning hypertension in this study differed from that in the JSH 2014 guideline.10 Sixth, the true dipping status, usually assessed by ABPM, is unclear in this study. Seventh, among the baseline clinical characteristics, the morning and office HR values were significantly higher in the HCTZ group compared with the cilnidipine group. Finally, we cannot be certain how reducing HMBPS, a new index, will affect cardiovascular outcomes.
6. CONCLUSIONS
HMBPS was significantly decreased in both our valsartan/cilnidipine and valsartan/HCTZ combination groups compared with baseline, with no significant difference between the two groups. A large‐scale randomized controlled study is needed to determine whether a valsartan/cilnidipine combination can suppress morning BP surges compared with a valsartan/HCTZ combination in patients with morning hypertension and to assess how reducing HMBPS will affect future cardiovascular outcomes. The ICT‐based home BP monitoring device could have the potential to be an alternative to ABPM, which has been a gold standard for the measurement of nocturnal BP and morning BP surge.
CONFLICTS OF INTEREST
The authors have no potential conflicts of interest to disclose.
Supporting information
Fujiwara T, Tomitani N, Kanegae H, Kario K. Comparative effects of valsartan plus either cilnidipine or hydrochlorothiazide on home morning blood pressure surge evaluated by information and communication technology–based nocturnal home blood pressure monitoring. J Clin Hypertens. 2018;20:159–167. 10.1111/jch.13154
Funding information
This study was financially supported by Mochida Pharmaceutical Co., Ltd., Tokyo and EA Pharma Co., Ltd., Tokyo.
REFERENCES
- 1. Ohkubo T, Imai Y, Tsuji I, et al. Home blood pressure measurement has a stronger predictive power for mortality than does screening blood pressure measurement: a population‐based observation in Ohasama, Japan. J Hypertens. 1998;16:971‐975. [DOI] [PubMed] [Google Scholar]
- 2. Sega R, Facchetti R, Bombelli M, et al. Prognostic value of ambulatory and home blood pressures compared with office blood pressure in the general population: follow‐up results from the Pressioni Arteriose Monitorate e Loro Associazioni (PAMELA) study. Circulation. 2005;111:1777‐1783. [DOI] [PubMed] [Google Scholar]
- 3. Stergiou GS, Baibas NM, Kalogeropoulos PG. Cardiovascular risk prediction based on home blood pressure measurement: the Didima study. J Hypertens. 2007;25:1590‐1596. [DOI] [PubMed] [Google Scholar]
- 4. Niiranen TJ, Hanninen MR, Johansson J, Reunanen A, Jula AM. Home‐measured blood pressure is a stronger predictor of cardiovascular risk than office blood pressure: the Finn‐Home study. Hypertension. 2010;55:1346‐1351. [DOI] [PubMed] [Google Scholar]
- 5. Gosse P, Ansoborio P, Lemetayer P, Clementy J. Left ventricular mass is better correlated with arising blood pressure than with office or occasional blood pressure. Am J Hypertens. 1997;10:505‐510. [DOI] [PubMed] [Google Scholar]
- 6. Marfella R, Gualdiero P, Siniscalchi M, et al. Morning blood pressure peak, QT intervals, and sympathetic activity in hypertensive patients. Hypertension. 2003;41:237‐243. [DOI] [PubMed] [Google Scholar]
- 7. Kario K, Pickering TG, Umeda Y, et al. Morning surge in blood pressure as a predictor of silent and clinical cerebrovascular disease in elderly hypertensives: a prospective study. Circulation. 2003;107:1401‐1406. [DOI] [PubMed] [Google Scholar]
- 8. Metoki H, Ohkubo T, Kikuya M, et al. Prognostic significance for stroke of a morning pressor surge and a nocturnal blood pressure decline: the Ohasama study. Hypertension. 2006;47:149‐154. [DOI] [PubMed] [Google Scholar]
- 9. Li Y, Thijs L, Hansen TW, et al. Prognostic value of the morning blood pressure surge in 5645 subjects from 8 populations. Hypertension. 2010;55:1040‐1048. [DOI] [PubMed] [Google Scholar]
- 10. Shimamoto K, Ando K, Fujita T, et al. The Japanese Society of Hypertension Guidelines for the Management of Hypertension (JSH 2014). Hypertens Res. 2014;37:253‐390. [DOI] [PubMed] [Google Scholar]
- 11. Mancia G, Fagard R, Narkiewicz K, et al, Task Force Members . 2013 ESH/ESC Guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J Hypertens. 2013;31:1281‐1357. [DOI] [PubMed] [Google Scholar]
- 12. Parati G, Stergiou GS, Asmar R, et al. European Society of Hypertension practice guidelines for home blood pressure monitoring. J Hum Hypertens. 2010;24:779‐785. [DOI] [PubMed] [Google Scholar]
- 13. Williams B, Williams H, Northedge J, et al. Hypertension: the clinical management of primary hypertension in adults. Update of clinical guidelines 18 and 34. NICE clinical guideline No. 127 2011. http://guidance.nice.org.uk/CG127. Accessed December 28, 2016.
- 14. Pickering TG, Miller NH, Ogedegbe G, et al. Call to action on use and reimbursement for home blood pressure monitoring: a joint scientific statement from the American Heart Association, American Society Of Hypertension, and Preventive Cardiovascular Nurses Association. Hypertension. 2008;52:10‐29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Takahashi H, Yoshika M, Yokoi T. Validation of two automatic devices: Omron HEM‐7252G‐HP and Omron HEM‐7251G for self‐measurement of blood pressure according to the European Society of Hypertension International Protocol revision 2010. Blood Press Monit. 2015;20:286‐290. [DOI] [PubMed] [Google Scholar]
- 16. Fujiwara T, Hoshide S, Nisizawa M, Matsuo T, Kario K. Difference in evening home blood pressure between before dinner and at bedtime in Japanese elderly hypertensive patients. J Clin Hypertens (Greenwich). 2017;19:731‐739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kario K, Tomitani N, Kanegae H, et al. Comparative effects of an angiotensin II receptor blocker (ARB)/diuretic vs. ARB/calcium‐channel blocker combination on uncontrolled nocturnal hypertension evaluated by information and communication technology‐based nocturnal home blood pressure monitoring––the NOCTURNE study. Circ J. 2017;81:948‐957. [DOI] [PubMed] [Google Scholar]
- 18. Takahara A. Cilnidipine: a new generation Ca channel blocker with inhibitory action on sympathetic neurotransmitter release. Cardiovasc Ther. 2009;27:124‐139. [DOI] [PubMed] [Google Scholar]
- 19. Kario K, Ando S, Kido H, et al. The effects of the L/N‐type calcium channel blocker (cilnidipine) on sympathetic hyperactive morning hypertension: results from ACHIEVE‐ONE. J Clin Hypertens (Greenwich). 2013;15:133‐142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kario K, Nariyama J, Kido H, et al. Effect of a novel calcium channel blocker on abnormal nocturnal blood pressure in hypertensive patients. J Clin Hypertens (Greenwich). 2013;15:465‐472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Uzu T, Kimura G. Diuretics shift circadian rhythm of blood pressure from nondipper to dipper in essential hypertension. Circulation. 1999;100:1635‐1638. [DOI] [PubMed] [Google Scholar]
- 22. Matsui Y, Eguchi K, O'Rourke MF, et al. Differential effects between a calcium channel blocker and a diuretic when used in combination with angiotensin II receptor blocker on central aortic pressure in hypertensive patients. Hypertension. 2009;54:716‐723. [DOI] [PubMed] [Google Scholar]
- 23. Hoshide S, Kario K, de la Sierra A, et al. Ethnic differences in the degree of morning blood pressure surge and in its determinants between Japanese and European hypertensive subjects: data From the ARTEMIS Study. Hypertension. 2015;66:750‐756. [DOI] [PubMed] [Google Scholar]
- 24. Verdecchia P, Angeli F, Mazzotta G, et al. Day‐night dip and early‐morning surge in blood pressure in hypertension: prognostic implications. Hypertension. 2012;60:34‐42. [DOI] [PubMed] [Google Scholar]
- 25. Wald DS, Law M, Morris JK, Bestwick JP, Wald NJ. Combination therapy versus monotherapy in reducing blood pressure: meta‐analysis on 11,000 participants from 42 trials. Am J Med. 2009;122:290‐300. [DOI] [PubMed] [Google Scholar]
- 26. Jamerson K, Weber MA, Bakris GL, et al. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high‐risk patients. N Engl J Med. 2008;359:2417‐2428. [DOI] [PubMed] [Google Scholar]
- 27. Dahlöf B, Sever PS, Poulter NR, et al. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo‐Scandinavian Cardiac Outcomes Trial‐Blood Pressure Lowering Arm (ASCOT‐BPLA): a multicentre randomised controlled trial. Lancet. 2005;366:895‐906. [DOI] [PubMed] [Google Scholar]
- 28. Dahlöf B. Management of cardiovascular risk with RAS inhibitor/CCB combination therapy. J Hum Hypertens. 2009;23:77‐85. [DOI] [PubMed] [Google Scholar]
- 29. Schöfl C, Becker C. Prank K, von zur Mühlen A, Brabant G. Twenty‐four‐hour rhythms of plasma catecholamines and their relation to cardiovascular parameters in healthy young men. Eur J Endocrinol. 1997;137:675‐683. [DOI] [PubMed] [Google Scholar]
- 30. White WB. Importance of blood pressure control over a 24‐hour period. J Manag Care Pharm. 2007;13:S34‐S39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ando T, Niijima Y. Retrospective study for the effects of switching to the valsartan‐cilnidioine combination tablet on office blood pressure and pulse rate in the real world. Jpn Pharmacol Ther. 2016;44:371‐379. [Google Scholar]
- 32. Ishikawa J, Hoshide S, Eguchi K, Ishikawa S, Shimada K, Kario K; Japan Morning Surge‐Home Blood Pressure Study Investigators Group. Nighttime home blood pressure and the risk of hypertensive target organ damage. Hypertension. 2012;60:921‐928. [DOI] [PubMed] [Google Scholar]
- 33. Stergiou GS, Nasothimiou EG, Destounis A, Poulidakis E, Evagelou I, Tzamouranis D. Assessment of the diurnal blood pressure profile and detection of non‐dippers based on home or ambulatory monitoring. Am J Hypertens. 2012;25:974‐978. [DOI] [PubMed] [Google Scholar]
- 34. Kario K, Hoshide S, Haimoto H, et al. Sleep blood pressure self‐measured at home as a novel determinant of organ damage: Japan Morning Surge Home Blood Pressure (J‐HOP) study. J Clin Hypertens (Greenwich). 2015;17:340‐348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Asayama K, Ohkubo T, Kikuya M, et al. Prediction of stroke by home “morning” versus “evening” blood pressure values: the Ohasama study. Hypertension. 2006;48:737‐743. [DOI] [PubMed] [Google Scholar]
- 36. Johansson JK, Niiranen TJ, Puukka PJ, Jula AM. Prognostic value of the variability in home‐measured blood pressure and heart rate: the Finn‐Home Study. Hypertension. 2012;59:212‐218. [DOI] [PubMed] [Google Scholar]
- 37. Hoshide S, Yano Y, Haimoto H, et al. Morning and evening home blood pressure and risks of incident stroke and coronary artery disease in the Japanese general practice population: the Japan Morning Surge‐Home Blood Pressure Study. Hypertension. 2016;68:54‐61. [DOI] [PubMed] [Google Scholar]
- 38. Kario K, Saito I, Kushiro T, et al. Morning home blood pressure is a strong predictor of coronary artery disease: the HONEST study. J Am Coll Cardiol. 2016;67:1519‐1527. [DOI] [PubMed] [Google Scholar]
- 39. Uzu T, Ishikawa K, Fujii T, Nakamura S, Inenaga T, Kimura G. Sodium restriction shifts circadian rhythm of blood pressure from nondipper to dipper in essential hypertension. Circulation. 1997;96:1859‐1862. [DOI] [PubMed] [Google Scholar]
- 40. Pogue V, Rahman M, Lipkowitz M, et al. African American Study of Kidney Disease and Hypertension Collaborative Research Group. Disparate estimates of hypertension control from ambulatory and clinic blood pressure measurements in hypertensive kidney disease. Hypertension. 2009;53:20‐27. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials


