Abstract
17β-estradiol (E2)-containing hormone therapy is a safe, effective way to alleviate unwanted menopause symptoms. Preclinical research has focused upon the role of E2 in learning and memory using a surgically menopausal rodent model whereby the ovaries are removed. Given that most women retain their reproductive tract and undergo a natural menopause transition, it is necessary to understand how exogenous E2 impacts a structurally intact, but follicle-deplete, system. In the current study, 8 month old female rats were administered the ovatoxin 4-vinylcyclohexene diepoxide (VCD), which accelerates ovarian follicular depletion, to model the human menopause transition. After follicular depletion, at 11 months old, rats were administered Vehicle or tonic E2 treatment for 12 days prior to behavioral evaluation on spatial working and reference memory tasks. Results demonstrated that E2 had both enhancing and impairing effects on taxed working memory depending upon the learning or retention phases of the water radial-arm maze, with no impact on reference memory. Relationships between memory scores and circulating estrogen levels were specific to follicle-depleted rats without E2 treatment. Collectively, findings demonstrate the complexity of E2 administration in a follicle-depleted background, with cognitive effects specific to working memory; furthermore, E2 administration altered circulating hormonal milieu and relationships between hormone profiles and memory. In sum, menopausal etiology impacts the parameters of E2 effects on cognition, complementing prior work with other estrogen compounds. Deciphering estrogenic actions in a system wherein the reproductive tract remains intact with follicle-depleted ovaries, thus modeling the majority or menopausal women, is critical for translational perspectives.
Keywords: VCD, Menopause, Estrogen, Learning, Memory, Hormone therapy
1. Introduction
A woman’s reproductive years span a significant proportion of her lifetime, from puberty until the fifth decade of life. Unless gynecological surgical intervention occurs prior, women then experience a natural transition to reproductive senescence, or menopause, over the course of several years and live up to a third of their lifetime in a post-reproductive state (Harlow et al., 2012; Hoffman et al., 2012; Koebele and Bimonte-Nelson, 2017; NAMS, 2014). Women are born with a finite pool of ovarian follicles; while up to 400 of these follicles reach full maturation and are eventually ovulated for potential pregnancy, the vast majority of this immature ovarian follicle reserve, consisting of tens to hundreds of thousands of resting-state and growing follicles, steadily declines across the lifespan via an apoptotic process called follicular atresia (Baker, 1963; Gougeon, 2010, 1998; Hsueh et al., 1994; Wallace and Kelsey, 2010). Growing ovarian follicles are the primary source of circulating sex steroid hormones in females. The eventual decline in the follicle pool coincides with a significant decrease in the production and availability of ovarian hormones, including estrogen and progesterone (Hoffman et al., 2012), as well as with the onset of menopause-associated symptoms. These symptoms include irregular bleeding patterns, vasomotor symptoms such as hot flashes and night sweats, urogenital symptoms, and changes in sleep quality, mood, metabolic processes, and often, memory (Al-Safi and Santoro, 2014; Mishra and Kuh, 2012; Mitchell and Woods, 2001; Weber et al., 2013; Weber and Mapstone, 2009), which can significantly disrupt quality of life depending on severity.
During the transition to menopause, many women opt to take estrogen-containing hormone therapy to alleviate associated unfavorable symptoms. Estrogen compound-containing hormone therapies are the only FDA-approved treatment that alleviates the broad spectrum of menopause-associated symptoms, from vaginal dryness to hot flashes (ACOG, 2014; Baber et al., 2016; Neves-E-Castro et al., 2015; Pinkerton et al., 2017; Stuenkel et al., 2015). Although estrogens are primarily considered reproductive hormones, the fluctuation and eventual decline in estrogen with menopause can impair optimal functioning in all major body systems; estrogen supplementation can mitigate some of these effects resulting from natural hormone decline with menopause. The primary and most potent circulating estrogen in humans is 17β-estradiol (E2), followed by its metabolites estrone (E1) and estriol (Kuhl, 2005). Although each of these belong to the class of estrogens, they have unique binding properties that result in distinct effects, particularly in the brain. There has been a multitude of research indicating that the loss of ovarian hormones —particularly estrogens following surgical ovary removal — is detrimental to not only various body systems, but also to memory in humans (Nappi et al., 1999; Rocca et al., 2007; Sherwin, 2003) as well as in animal models (for review, see: Daniel, 2013; Frick, 2015; Koebele and Bimonte-Nelson, 2015; Korol and Pisani, 2015; Luine, 2014). Furthermore, under certain administration conditions, E2 treatment following ovariectomy (Ovx) can improve learning and memory performance in a variety of animal models (e.g., Bimonte and Denenberg, 1999; Black et al., 2018, 2016; Bohacek et al., 2008; Fernandez et al., 2008; Harburger et al., 2009; Koebele et al., 2020; Lacreuse, 2006; Markowska and Savonenko, 2002; Prakapenka et al., 2018; Rapp et al., 2003; Rodgers et al., 2010; Savonenko and Markowska, 2003; Talboom et al., 2008; Wallace et al., 2006). However, not all estrogens behave in an identical fashion. For example, our laboratory has shown that continuous E1 administration in middle-aged Ovx rats resulted in significant impairments in spatial working memory (Engler-Chiurazzi et al., 2012). These divergent effects are of particular relevance to the use of estrogens in menopausal hormone therapy. Until the mid-2000’s, the most commonly prescribed hormone therapy for menopause symptoms was conjugated equine estrogens (CEE; tradename Premarin). CEE is derived from the urine of pregnant horses and contains at least ten types of estrogens, with the predominant estrogen being E1 sulfate, and many of the others uniquely equine and not naturally circulating in women (Bhavnani and Stanczyk, 2014). Initial findings from the Women’s Health Initiative (WHI) and the ancillary WHI Memory Study (WHIMS) indicated negative effects of CEE-including hormone therapy on cardiovascular and cognitive health (Coker et al., 2010; Espeland et al., 2004; Shumaker et al., 2004, 2003), resulting in a steep decline in prescription hormone therapy use, particularly those formulations that included CEE (Crawford et al., 2018; Hersh et al., 2004; Sprague et al., 2012). In the decades since, there has been a shift to utilizing hormone therapy formulations primarily containing E2 rather than CEE, which is structurally identical to endogenously-produced E2 and without additional compounds not found naturally in humans. There is substantial evidence in the preclinical literature that E2 administration following Ovx-induced hormone loss attenuates learning and memory deficits; however, less is known about the efficacy and longitudinal cognitive impacts of exogenous estrogen-containing menopausal hormone therapy in female rodents with intact reproductive tracts. The dose, type of estrogen, route of administration, and menopause etiology likely all factor into cognitive outcomes of hormone administration (for review, see: Koebele and Bimonte-Nelson, 2015). Indeed, our laboratory and others have demonstrated variable effects of CEE on cognitive and brain health depending on the model used (Acosta et al., 2010; Barha and Galea, 2013; Engler-Chiurazzi et al., 2011; Hiroi et al., 2016). For example, the same dose of CEE that enhanced spatial working memory in Ovx rats impaired spatial working memory in ovary-intact rats that had undergone follicle depletion (Acosta et al., 2010), underscoring menopause type as a particularly important variable of interest. Until now, the cognitive effects of E2 have only been methodically evaluated in a surgical menopause model. Given the popularity of E2-containing hormone therapy in transitionally menopausal women, a crucial next step is to investigate the effects of exogenous E2 in an ovary-intact, but follicle-deplete, rat model.
Despite comparable patterns of reproductive endocrinology during the reproductive life stage, rats do not undergo the transition to reproductive senescence in a manner physiologically similar to women (for reviews, see: Finch, 2014; Koebele and Bimonte-Nelson, 2016). The drug 4-vinylcyclohexene diepoxide (VCD) has been shown to selectively target non-growing and early-stage ovarian follicles and result in an accelerated follicular depletion that more closely models the ovarian and endocrine profiles of a transitionally menopausal woman (Frye et al., 2012; Hoyer et al., 2001; Hu et al., 2001; Mayer et al., 2002; Springer et al., 1996b, 1996a). As such, VCD can be used as a model of transitional menopause to help us better understand how menopausal therapies interact with an intact, but follicle-deplete, reproductive system. Given that the vast majority of women undergo a natural transition to menopause and retain their follicle-depleted ovarian tissue, this collective literature underscores the importance of methodically investigating the cognitive effects of various estrogenic hormone therapy formulations in rodent models that more closely mimic the menopause transition in women. It is especially pertinent to determine whether exogenous E2 administration has a favorable cognitive profile using a rodent model of transitional menopause, as E2 typically has more consistent beneficial effects on the brain and behavior compared to other clinically-available natural and synthetic estrogens in Ovx models (Engler-Chiurazzi et al., 2012, 2011; Hiroi et al., 2016; Mennenga et al., 2015b). The current experiment aimed to meet this need by systematically evaluating the effects of tonic E2 administration on spatial working and reference memory following VCD-induced follicular depletion, compared to VCD-induced follicle-deplete rats without subsequent hormone therapy treatment.
2. Methods
2.1. Subjects
Twenty-two female, virgin, Fischer-344 rats were acquired from the National Institute on Aging colony at Harlan Laboratories (Indianapolis, IN). Rats were approximately 8 months of age upon arrival to the vivarium facility at Arizona State University. Rats were pair- or single-housed, had free access to food and water, and were maintained on a 12-hour light/dark cycle (lights on at 7 am) for the duration of the experiment. An acclimation period of one week was provided prior to beginning experimental procedures. The Arizona State University Institutional Care and Use Committee approved all procedures, which adhered to National Institutes of Health standards. Behavioral assessments began when rats were 11–12 months of age, after follicular depletion was complete and hormone treatment was initiated. We targeted the 11–12 month old time point based on prior published research from our laboratory and others that operationally defines middle-age within this age range for various laboratory rat strains, including Fischer-344 rats (e.g. Bizon et al., 2009; Black et al., 2016; Daniel et al., 2006; Dubal and Wise, 2001; Foster et al., 2003; Gibbs et al., 2009; Maffucci et al., 2009; Rodgers et al., 2010; Savonenko and Markowska, 2003; Wang et al., 2009; Witty et al., 2012).
2.2. VCD injections
Rats were randomly assigned to either VCD-Vehicle or VCD-E2 treatment (n = 11/treatment group; 5 pair housed cages and one single-housed cage per group). All subjects were administered VCD (FYXX Foundation, Flagstaff, AZ) via intraperitoneal injection for 15 days (one injection per day, for a total of 15 injections) to safely induce accelerated follicular depletion with little to no subject attrition, based on prior established protocols (Acosta et al., 2010, 2009; Carolino et al., 2019; Frye et al., 2012; Kirshner and Gibbs, 2018; Koebele et al., 2017; Lohff et al., 2006, 2005; Mayer et al., 2004, 2002; Van Kempen et al., 2011; Zhang et al., 2016). Baseline weight (g) was obtained for each rat, and the daily injection volume was determined by the animal’s weight. VCD was administered at a dose of 160 mg/kg/day in 50% dimethyl sulfoxide (DMSO)/50% sterile saline vehicle (Sigma-Aldrich, St. Louis, MO) (Acosta et al., 2010, 2009; Frye et al., 2012; Golub et al., 2008; Koebele and Bimonte-Nelson, 2017; Wright et al., 2011). In accordance with IACUC standards and established protocols, if there was a 10% or more drop in baseline body weight, VCD injections were not administered until weight was regained. Injections were administered on Monday, Tuesday, Thursday, and Friday. Injections were not administered on Wednesday, Saturday, or Sunday to allow for weight recovery, as previously done in our laboratory (Acosta et al., 2010, 2009; Koebele et al., 2017). Due to weight loss and recovery periods, the 15 VCD injections were completed over approximately six weeks.
2.3. Hormone treatment administration
To ensure complete follicular depletion, a 95-day waiting period following the first VCD injection was implemented (Acosta et al., 2009; Lohff et al., 2005). When rats were approximately 11 months old, all subjects underwent Alzet osmotic pump insertion surgeries. The Alzet 2006 model was utilized to deliver either Vehicle (polyethylene glycol; PEG; Sigma Aldrich #202371, average Mn 300) or 3 μg E2/day (Sigma Aldrich #E8875) in PEG vehicle at a constant release rate of 0.15 μl of solution per hour for six weeks. All subjects were anesthetized using inhaled isoflurane anesthesia. A small skin incision was made in the scruff of the neck. Once a small subcutaneous pocket was made, osmotic pumps were inserted into each subject. The skin incision was then sutured with dissolvable Vicryl suture material. The E2 dose utilized in this experiment was based on prior data from our laboratory showing that a dose of 3 μg E2 administered daily to middle-aged Ovx rats produced beneficial spatial working memory effects in the water radial-arm maze (Hiroi et al., 2016; Prakapenka et al., 2018). Furthermore, this dose of E2 has been shown to produce circulating E2 levels within the moderate to high physiological range of ovary-intact and VCD-treated rats (Kirshner et al., 2020; Kirshner and Gibbs, 2018; Long et al., 2019, 2018). Rats were all single-housed for a 48-hour recovery period prior to re-housing with its cage mate of the same treatment group until the end of the experiment.1
2.4. Body weights
Body weights (g) were collected as a peripheral marker of general health, beginning at the onset of VCD injections and periodically throughout the experiment until euthanasia.
2.5. Vaginal cytology
Vaginal smears were obtained for two consecutive days after pump surgeries to assess estrous cycle status. Longer term daily handling associated with vaginal smears could have impacted the cognitive efficacy of estrogen treatment, as shown previously in rats tested on the eight arm radial maze (Bohacek and Daniel, 2007). The two-day vaginal smear assessment in conjunction with planned ovarian follicle counts and serum hormone data gave a thorough picture of follicle depletion status. A small cotton-tipped applicator soaked in sterile saline was gently inserted into the vaginal opening. Vaginal cells were viewed under a light microscope and classified as proestrus-, estrus-, metestrus-, or diestrus- like smears (Goldman et al., 2007; Koebele and Bimonte-Nelson, 2016; Mennenga and Bimonte-Nelson, 2015).
2.6. Behavioral testing
Twelve days after pump insertion, when rats were 11–12 months of age, all subjects were assessed on a battery of maze tasks measuring spatial working and reference memory, including the water radial-arm maze (WRAM) and Morris water maze (MM), as well as the control visible platform (VP) task. Procedures for each maze task are described in detail below.
2.6.1. Water radial-arm maze
The WRAM was a water-escape task that measured spatial working and reference memory (Bimonte-Nelson et al., 2015, 2004, 2003a; Bimonte et al., 2003, 2002, 2000; Bimonte and Denenberg, 1999). Fig. 1A illustrates a schematic of the WRAM. The apparatus was constructed with a circular center and eight arms protruding outward, which were equally sized (38.1 cm × 12.7 cm each) and spaced around the center arena. In addition to the room geometry, tables, and heat lamps situated in each testing room, prominent visual cues were hung around the room to aid in spatial navigation in the maze. The maze was filled with room temperature water made opaque with black non-toxic powdered paint and maintained at 18–20 °C, which served as a motivator for escape. Rats were each assigned a pre-determined set of platform location combinations, wherein four of the maze arms contained a hidden escape platform at the end of the arm, submerged 2–3 cm beneath the water’s surface; the other four arms (including the start arm) did not contain an escape platform. Platform location combinations assigned to each rat remained constant across all days of the task for a given rat, but platform location combinations varied among rats and were counterbalanced across treatment groups. The WRAM had a win-shift rule; that is, once a rat was rewarded in a specific place in space by being removed from the water maze (negative reinforcement) and placed back into its heated testing cage (positive reinforcement) as a consequence of locating a hidden escape platform (i.e., a “win”), the rat was required to shift to another place in space (i.e., a different platform location) in order to “win” on the next trial. Working memory load was progressively taxed across trials within each day as the number of locations to be recalled increased with each trial.
Fig. 1.
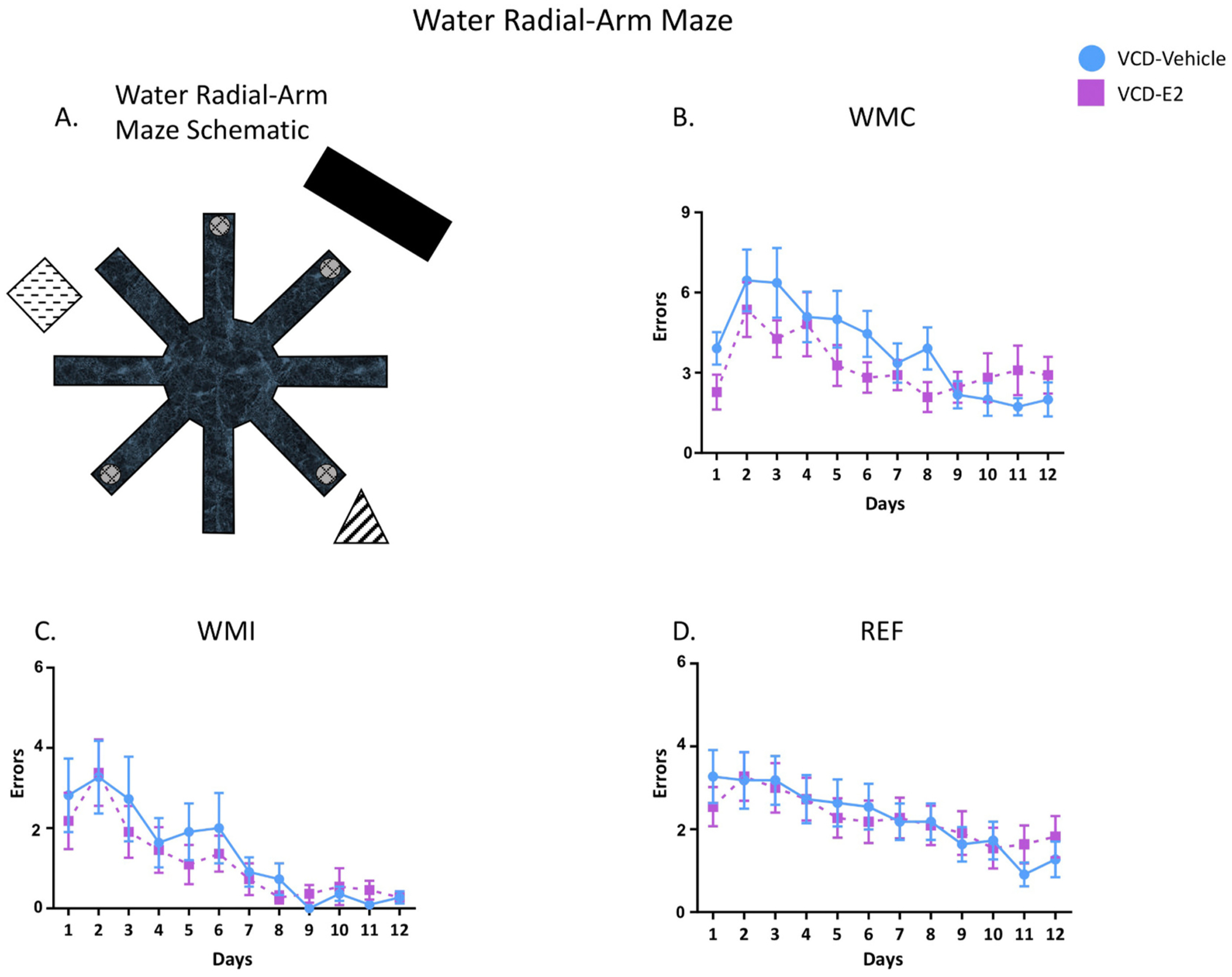
(A) An illustration of the WRAM apparatus. Learning curves for working memory correct errors (1B), working memory incorrect errors (1C), and reference memory errors (1D) across the 12 days of the WRAM task. Day 1 performance was considered training and was excluded from analyses. VCD-Vehicle: n = 11; VCD-E2: n = 11.
Each rat underwent four trials per day across 13 consecutive days. Day 1 was considered a training day, and the subsequent 11 days were considered normal testing days. At the beginning of each testing session, a rat was gently placed in the non-platformed start arm of the maze. Rats were given a 3 min time limit to locate a platform on each trial, which was monitored by the experimenter via a stopwatch. If the rat did not escape the maze from a hidden platform within the allotted 3 min, the experimenter used a lead stick to guide the rat to the nearest platform. When a platform was located, the rat was given 15 s of total platform time before being returned to its heated testing cage for a 30 s inter-trial-interval (ITI). The experimenter then removed the just-located platform from the WRAM (which was not replaced within a daily testing session), discarded any debris, and stirred the water to disperse potential olfactory cues during the ITI. Following twelve days of WRAM exposure, on Day 13, an 8 hour delay was employed between trials two and three to assess delayed working memory retention.
Across all days and trials, learning and memory performance was quantified by calculating the number of entries into non-platformed arms prior to locating a platform on each trial, which were considered errors. The experimenter manually recorded arm entry errors during each trial on a testing sheet, as well as trial time and any indicators of floating/freezing behavior to ensure all subjects were performing the task. An arm entry was defined as the tip of the rat’s snout crossing a marker delineating 11 cm into the arm (visible to the experimenter outside of the maze, but not visible to the rat). Errors were classified into three subcategories. Working memory correct (WMC) errors were defined by entries into an arm that previously contained a platform within a day (trials 2–4). Reference memory (RM) errors were the first entry into an arm within a day that never contained a platform. Working memory incorrect (WMI) errors were subsequent entries into an arm that never contained a platform within a day (Bimonte-Nelson et al., 2015).
2.6.2. Morris water maze
Following the WRAM delayed memory retention day, rats began testing on the MM, a water-escape task used to assess spatial reference memory (Morris, 2015; Morris et al., 1982). The MM is a large, circular tub (188 cm in diameter) filled with 18–20 °C room temperature water made opaque with non-toxic black paint (Fig. 5A). One platform (11 cm diameter) was placed 2–3 cm below the surface of the water in the northeast quadrant of the maze, where it remained across all days and trials. In this way, the MM was a “win-stay” task, such that once a subject located the platform and “won” on that trial by finding the hidden platform, the rat had to navigate back to the same place in space to be rewarded again with removal from the water maze and being placed back into the heated testing cage. The rats underwent four trials per day across five days. On each trial within a day, rats were dropped off in one of the four cardinal directions (north, south, east, or west). The order of drop-off locations changed across days, but was the same within a day for all rats. All trials for each rat were recorded by a video camera and Ethovision tracking software (Noldus Instruments; Wageningen, The Netherlands). The maximum trial time was 1 min. If the rat did not find the platform in the allotted trial time, the experimenter gently guided the rat to the platform using a lead stick. Once the rat found the platform, it was required to stay on it for 15 s of platform time before being returned to its heated testing cage for an ITI of about 10 min while the other subjects were tested on that trial. On the last day of testing, after the last regular trial, an additional 5th trial, called the probe trial, was completed. During the probe trial, the platform was completely removed from the maze and rats were dropped off and allowed to swim freely for 1 min. For baseline days and trials, the total swim distance (cm) from drop-off to the platform was recorded. For the probe trial, the percent of total swim distance in the previously platformed quadrant versus the opposite quadrant was calculated to assess whether the rats had spatially localized to the platform location during the probe trial. Swim distance/path length to the platform has been established as a valid and appropriate measure for evaluating performance on the MM, particularly in aging rat research, as it is less likely to be impacted by age-related swim speed and motor declines (Bimonte-Nelson et al., 2015; Morris, 2015).
Fig. 5.
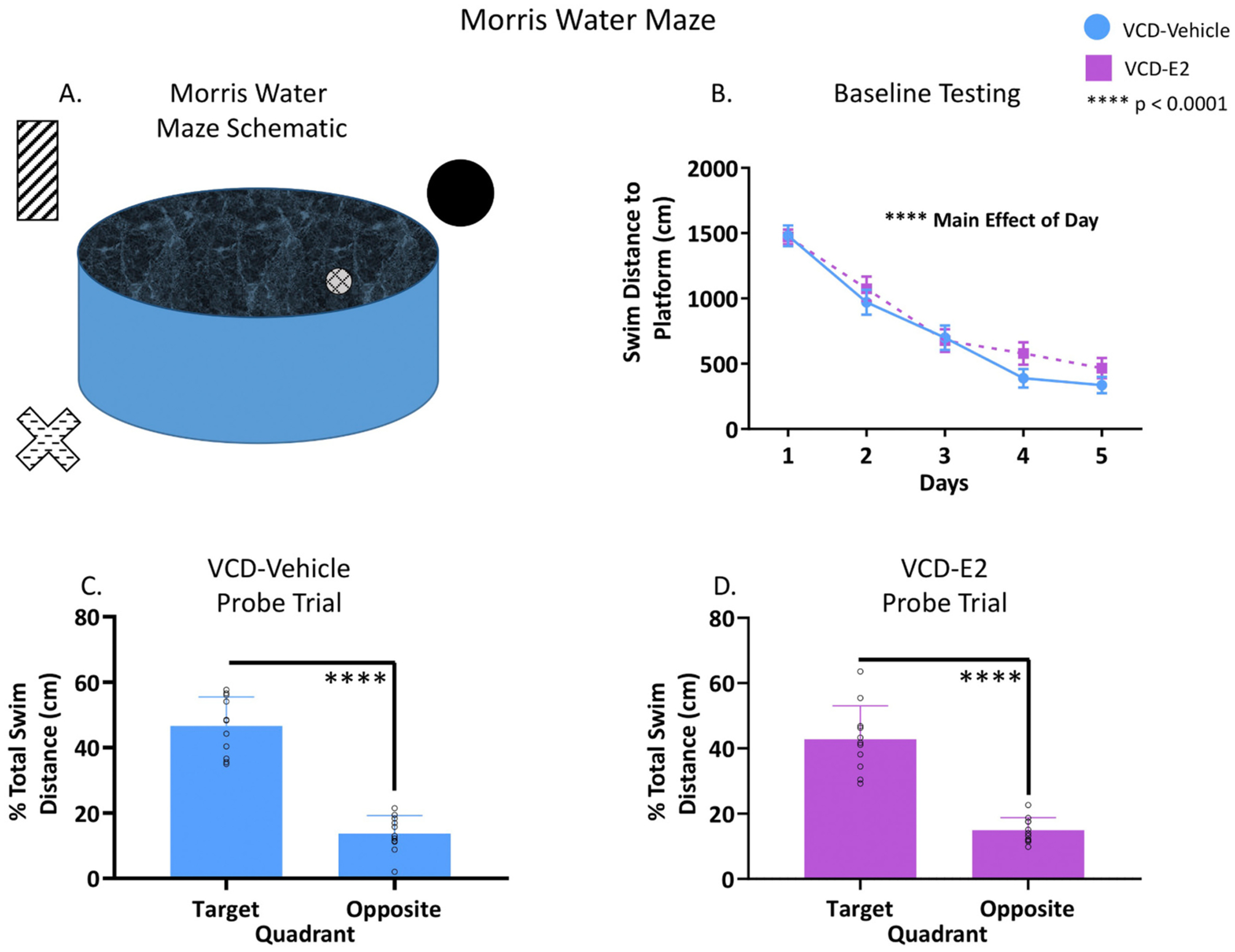
Morris water maze performance. VCD-Vehicle: n = 11; VCD-E2: n = 11. (A) A schematic of the MM apparatus. (B) Average swim distance to platform decreased across days and did not interact with Treatment, indicating that reference memory was intact and not differentially impacted by exogenous hormone treatment following follicular depletion. (C-D) Analysis of the probe trial indicated that both groups swam a greater percent of total distance in the maze quadrant that previously contained the hidden escape platform compared to swim distance in the opposite quadrant. Individual data points are overlaid on the mean ± SEM.
2.6.3. Visible platform
Following the last day of MM, immediately prior to euthanasia, rats were tested on the VP task to evaluate visual and swimming/motor competency. The VP was a simple task designed to be distinct from the other two mazes tested in the study. It included an apparatus that was unique in shape and size from the WRAM and MM so that it was generalizable to evaluate the procedural components of swim-based water escape tasks (Braden et al., 2017, 2015; Camp et al., 2012; Koebele et al., 2017; Mennenga et al., 2015a) (schematic illustrated in Fig. 6A). The apparatus was a rectangular tub (100 cm × 60 cm), smaller than the other two mazes used herein, filled with clear, room temperature (18–20 °C) water. A black platform (10 cm diameter) was placed approximately 4 cm above the surface of the water so that it was visible to the rats. Opaque curtains were hung in a circular fashion around the VP tub in order to block potential spatial or geometric cues. Rats received one day of VP testing, with six trials in that one day. Subjects were dropped off from a fixed location in the center of the south wall of the tub. The visible platform location varied across trials semi-randomly in three possible locations. A maximum of 90 s was allocated to reach the visible platform. The experimenter manually recorded latency with a stopwatch from rat drop off to when it reached the platform; because the apparatus is small and the rats typically escape the water via a straight path to the protruding visible platform within 10 s after just a few trial exposures, distance is not collected as a measurement for this task. After finding the platform, the rat remained on the platform for 15 s before being returned to its heated testing cage for an ITI of approximately 10 min while the other subjects were tested on that trial.
Fig. 6.
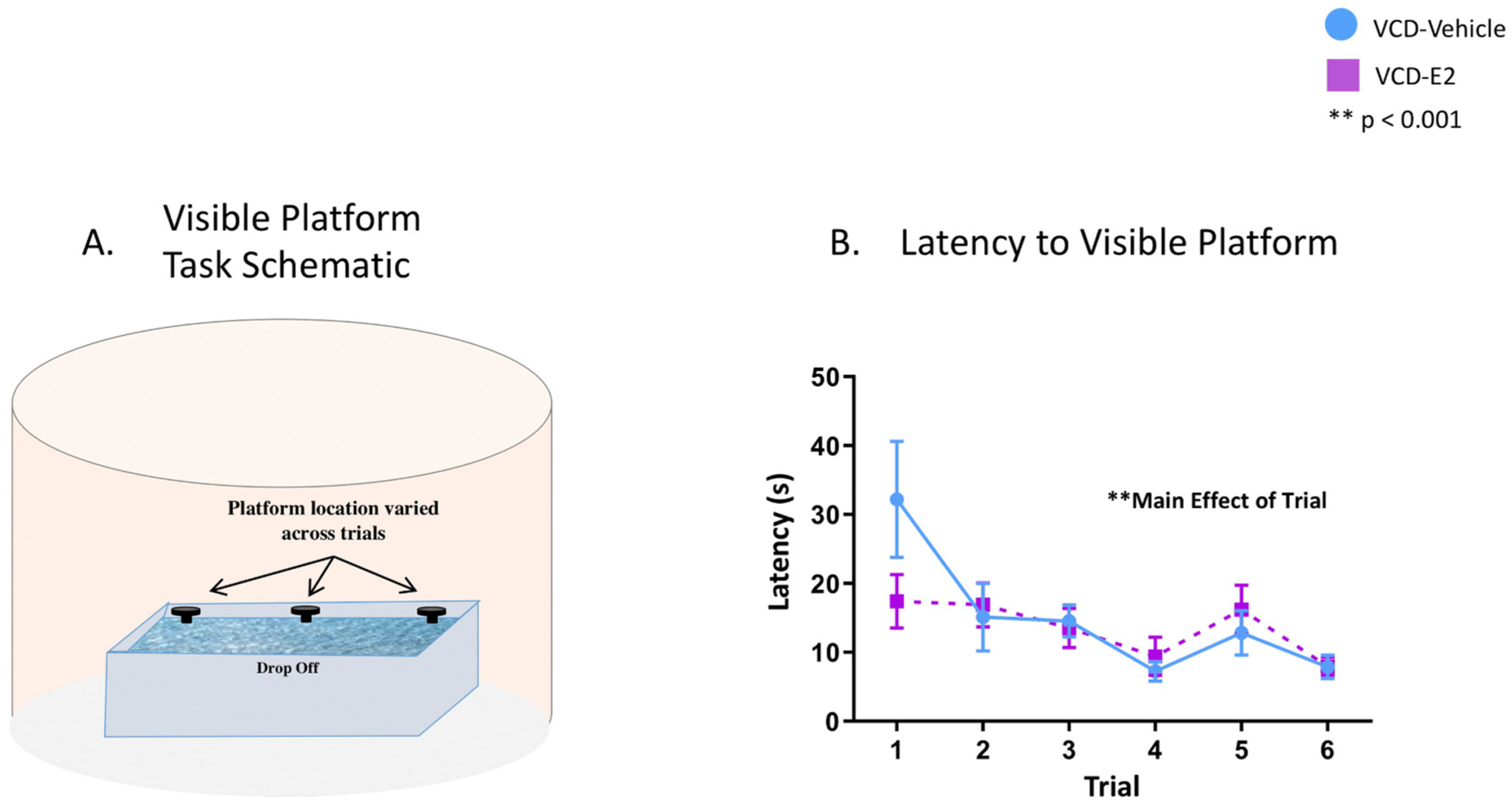
(A) A schematic of the apparatus used for the VP task. (B) Visible Platform Performance: VCD-Vehicle and VCD-E2 groups significantly decreased latency to the visible platform across trials of the control task. VCD-Vehicle: n = 11; VCD-E2: n = 11.
2.7. Euthanasia
Upon completion of the VP task, rats were euthanized. Rats were approximately 12 months of age at euthanasia. All subjects were deeply anesthetized with an overdose of inhaled isoflurane prior to cardio-centesis and decapitation. Blood was collected (Vacutainer 367986; Becton Dickinson and Company, Franklin Lakes, NJ) and allowed to clot at 4 °C for a minimum of 30 min. Then, blood samples were centrifuged at 2000 rpm at 4 °C for 20 min. Serum was collected and stored at −20 °C until analysis. Ovaries were separated from the uterine horns, trimmed of excess fat, and fixed in 4% paraformaldehyde for 48 h prior to being transferred to 0.1 M phosphate buffered solution until analysis. Uteri were dissected from the body cavity, trimmed of excess fat, and wet weight (g) was obtained.
2.8. Serum hormone measurements
Radioimmunoassay was utilized to detect serum hormone levels of E2, E1, and androstenedione as we have previously published (Acosta et al., 2010; Camp et al., 2012; Koebele et al., 2017; Mennenga et al., 2015b, 2015c). All radioimmunoassays were carried out at the Core Endocrine Laboratory at Pennsylvania State University under the supervision of Dr. Lawrence Demers. A double antibody liquid-phase radioimmunoassay purchased from Beckman Coulter (Brea, CA) was used to determine E2 levels. This kit employed estradiol-specific antibodies with an 125I-labeled estradiol as the tracer. Interassay coefficients of variation for the assay averaged 8% at a mean value of 6 pg/ml. Functional sensitivity of the E2 assay was 4 pg/ml. A double antibody liquid-phase radioimmunoassay purchased from Beckman Coulter (Brea, CA) was used to determine E1 levels. This kit employed estrone-specific antibodies with an 125I–labeled estrone as the tracer. Interassay coefficients of variation for the assay averaged 11% at a mean value of 90 pg/ml. Functional sensitivity of the E1 assay was 16 pg/ml. A solid-phase radioimmunoassay purchased from Siemens (Los Angeles, CA) was used to assess serum androstenedione levels, based on androstenedione-specific antibodies that were immobilized to the wall of poly-propylene tubes and an 125I –labeled androstenedione as the tracer. Interassay coefficients of variation for the assay averaged 3% at a mean value of 2.80 ng/ml. Functional sensitivity of the assay was 0.1 ng/ml.
2.9. Ovarian follicle counts
Following euthanasia, ovaries from rats were post-fixed, and one ovary from each rat was randomly selected for processing and quantification. All ovarian follicle processing and quantification was carried out by FYXX Foundation (Flagstaff, AZ). Ovarian follicle count estimates for primordial, primary, secondary, and antral follicles, as well as corpora lutea, were completed. Of particular interest were the primordial follicle counts, which are part of the non-growing, finite ovarian follicle pool and are the principal cells targeted by VCD administration. Ovarian tissues were paraffin embedded for processing, sectioned at 5 μm, mounted, and stained with hematoxylin and eosin Y-phloxine B. All follicle types were manually counted for every 20th section. Corpora lutea were counted at a magnification of 10× (Spencer compound microscope; American Optical, Buffalo, NY). Criteria from Haas et al., 2007 were used to classify follicle type, as we have previously published (Koebele et al., 2019, 2017). Briefly, a single layer of squamous granulosa cells surrounding an oocyte was considered a resting-state primordial ovarian follicle. Primary follicles included a single layer of cuboidal granulosa cells. Secondary follicles required several granulosa cell layers for classification. Antral follicles had two or more layers of granulosa cells in addition to a fluid-filled antral space within the follicle (Haas et al., 2007). Following classification and quantification of all follicle types via manual count, the total primordial follicle number estimate was calculated using the following formula: Nt = (N0 × St × ts)/(S0 × d0), where Nt = total follicle estimate, N0 = number of follicles observed in the ovary, St = total number of sections in the ovary, ts = thickness of the section (μm), S0 = total number of sections observed, and d0 = mean diameter of the nucleus (Gougeon and Chainy, 1987).
2.10. Statistical analyses
All data analyses were completed using Statview statistical software. The independent variable was Treatment (VCD-Vehicle or VCD-E2). A series of repeated measures ANOVAs were completed for WRAM data. The dependent variable was number of errors, and Trials nested within Days were the repeated measures. Day 1 of WRAM was considered training and excluded from the analysis. Data were separated into two blocks, as similarly published by our laboratory (Bimonte-Nelson et al., 2004, 2003b, 2003a; Bimonte and Denenberg, 1999; Braden et al., 2017; Koebele et al., 2017; Mennenga et al., 2015b). Days 2–8 were considered the Learning Phase, and Days 9–12 were considered the Asymptotic Phase. Separate repeated measures analyses were completed for each error type (WMC, WMI, and RM errors) within each block of testing. A priori planned comparisons for performance on the maximum working memory load trial only (Trial 4) were also analyzed separately for each block of testing based on prior age- and estrogen- associated findings in our laboratory (Acosta et al., 2010; Bimonte-Nelson et al., 2004, 2003a; Bimonte et al., 2003; Bimonte and Denenberg, 1999; Koebele et al., 2020, 2019, 2017; Mennenga et al., 2015c, 2015b; Prakapenka et al., 2018). MM data were analyzed using repeated measures ANOVA, with Swim Distance (cm) to the Platform as the dependent variable across repeated Days and Trials. The probe trial was analyzed as a repeated measures ANOVA with Percent of Total Swim Distance (cm) as the dependent variable and Target Quadrant versus Opposite Quadrant as repeated measures. VP data were analyzed using repeated measures ANOVA with Latency to Platform (s) as the dependent variable repeated across Trial. Body weight, uterine weight, serum hormone levels, and ovarian follicle count data were analyzed using ANOVA, with Treatment as the independent variable and body weight, uterine weight, serum hormone concentration, and follicle counts as the dependent variables, respectively. An additional ANOVA was completed for ovarian follicle counts including an ovary-intact, non-VCD treated reference group that received the complementary Vehicle injection for VCD injections (n = 10) from an independent data set in our laboratory to provide context for the VCD-induced follicular depletion in the current study. Pearson r correlations were also assessed among hormone values and WRAM errors during the Asymptotic Phase. The False Discovery Rate (FDR) correction (Q) for multiple comparisons (Benjamini and Hochberg, 1995; McDonald, 2014) with a FDR threshold of 0.10 was utilized, as we have previously done (Prakapenka et al., 2018). All statistical analyses were two-tailed with an α level set to 0.05. Results from the data analyses were considered marginal when the p value fell between 0.05 and 0.10. As we have previously published (Koebele et al., 2020, 2019, 2017), effect sizes for repeated measures ANOVA analyses were calculated as generalized eta squared (ηG2; Bakeman, 2005; Olejnik and Algina, 2003). Eta squared (η2) was calculated for all other ANOVAs that did not include repeated measures. Standard published guidelines were used to interpret effect size, where 0.02 is considered a small effect, 0.13 is considered a medium effect, and 0.26 is considered a large effect (Bakeman, 2005; Cohen, 1992, 1988). No subjects died or were excluded from analyses; unless otherwise noted, n = 11 for the VCD-Vehicle treatment group and n = 11 for the VCD-E2 treatment group.
3. Results
3.1. Water radial-arm maze
Fig. 1B–D demonstrates the learning curves for WMC, WMI, and RM errors across all days of testing.
3.1.1. Learning Phase (days 2–8)
For WMC errors, there was a main effect of Treatment [F(1,20) = 5.34, p < 0.05, ηG2 = 0.02], whereby the VCD-E2 group made fewer WMC errors compared to the VCD-Vehicle group. There was also a significant Treatment × Trial interaction for WMC errors during the Learning Phase [F(2,40) = 3.33, p < 0.05, ηG2 = 0.02; Fig. 2A]. When Trial 4, the highest working memory load trial, was probed individually for WMC errors, a main effect of Treatment was revealed [F(1,20) = 5.46, p < 0.05, ηG2 = 0.06], indicating that VCD-E2 rats made fewer WMC errors compared to VCD-Vehicle rats when working memory load was maximally taxed (Fig. 2B). For WMI errors during the Learning Phase, there was a significant Treatment × Trial interaction [F(3,60) = 5.69, p < 0.01, ηG2 = 0.03; Fig. 2C]. When Trial 4, the highest working memory load trial, was probed individually for WMI errors, a main effect of Treatment was observed [F(1,20) = 5.27, p < 0.05, ηG2 = 0.04], again indicating that the VCD-E2 group made fewer WMI errors compared to the VCD-Vehicle group when working memory load was maximally taxed (Fig. 2D). There were no main effects or interactions for RM errors during the Learning Phase. Thus, E2-induced effects for the Learning Phase were specific to working memory; indeed, for both working memory measures, VCD-E2 rats made fewer errors compared to VCD-Vehicle rats during the Learning Phase of the WRAM, and this effect was particularly evident when working memory load was highly taxed.
Fig. 2.
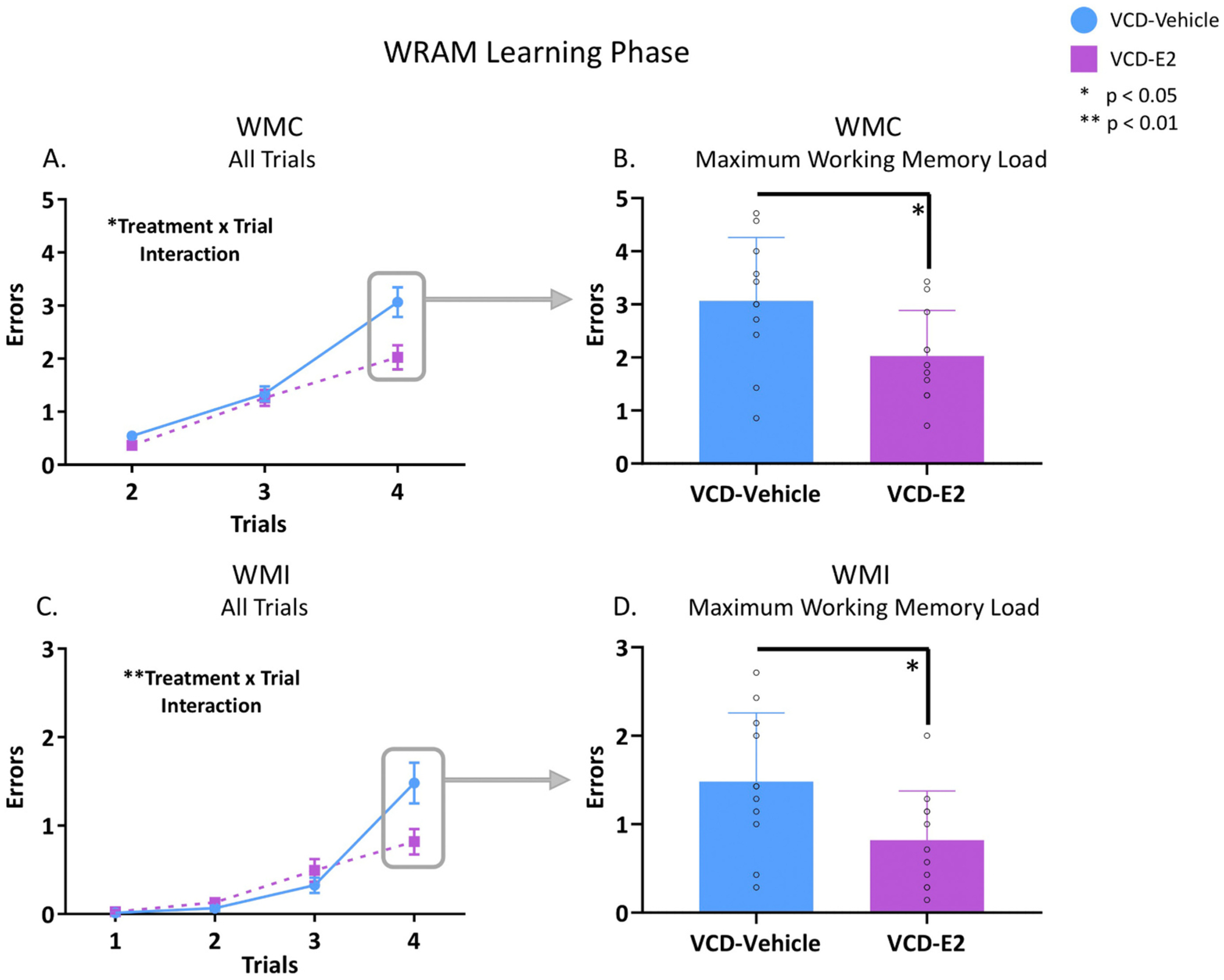
Working memory errors during the Learning Phase of WRAM. VCD-Vehicle: n = 11; VCD-E2: n = 11. (A) Across Trials 2–4, a Treatment × Trial interaction occurred for WMC errors (p < 0.05). (B) VCD-E2 rats made significantly fewer WMC errors compared to VCD-Vehicle rats when memory load was maximally burdened during learning (p < 0.05). Individual data points are overlaid on the mean ± SEM. (C) Across all trials, a Treatment × Trial interaction occurred for WMI errors (p < 0.01). (D) VCD-E2 rats made significantly fewer WMI errors compared to VCD-Vehicle rats when memory load was maximally burdened during learning (p < 0.05). Individual data points are overlaid on the mean ± SEM.
3.1.2. Asymptotic Phase (days 9–12)
For the Asymptotic Phase, when rats have learned the rules of the WRAM task and were approaching peak performance, there was a marginal main effect of Treatment [F(1,20) = 3.78, p = 0.07, ηG2 = 0.07], as well as a marginal Treatment × Trial interaction for WMC errors [F(2,40) = 3.12, p = 0.06, ηG2 = 0.02; Fig. 3A]. When Trial 4, the highest working memory load trial, was probed individually for WMC errors, a main effect of Treatment was revealed [F(1,20) = 4.86, p < 0.05, ηG2 = 0.05], indicating that VCD-E2 rats made more WMC errors on the maximum working memory load trial compared to VCD-Vehicle rats (Fig. 3B). Across all trials, there was a marginal Treatment × Trial interaction for WMI errors [F(3,60) = 2.20, p = 0.097, ηG2 = 0.02; Fig. 3A]. There was no main effect of Treatment when Trial 4 was assessed separately for WMI errors during the Asymptotic Phase (Fig. 3D). There were no main effects or interactions for RM errors during the Asymptotic Phase. Taken together, during the Asymptotic Phase of WRAM, the direction of the effect observed in the Learning Phase reversed in the Asymptotic Phase, such that VCD-E2 rats made more WMC errors compared to VCD-Vehicle rats when working memory load was highly taxed. This effect did not carry over into the WMI domain with statistical significance at this time point.
Fig. 3.
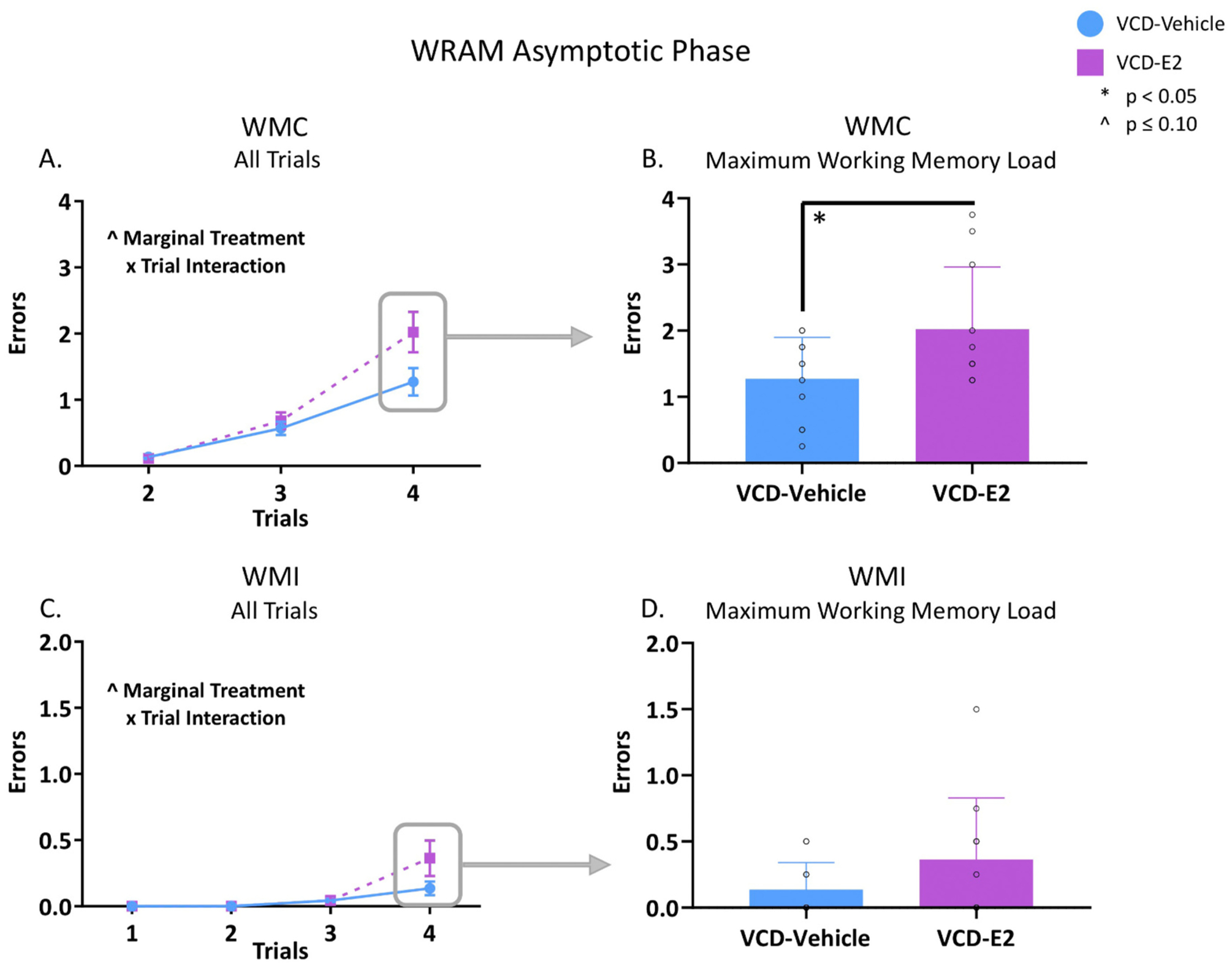
Working memory errors during the Asymptotic Phase of WRAM. VCD-Vehicle: n = 11; VCD-E2: n = 11. (A) Across Trials 2–4, a marginal Treatment × Trial interaction occurred for WMC errors. (B) VCD-E2 rats made significantly more WMC errors compared to VCD-Vehicle rats when memory load was maximally burdened during the latter block of testing (p < 0.05). Individual data points are overlaid on the mean ± SEM. (C) Across all trials, a marginal Treatment × Trial interaction occurred for WMI errors. (D) There was no statistically significant difference between VCD-E2 rats and VCD-Vehicle rats for WMI errors when memory load was maximally burdened during the Asymptotic Phase. Individual data points are overlaid on the mean ± SEM.
3.1.3. Eight-hour delayed memory retention
On Day 13 of WRAM testing, an 8 h delay was implemented between Trial 2 and Trial 3 to investigate delayed working memory retention. Each treatment group was assessed separately by comparing group performance for WMC errors on Trial 3 on the last day of baseline testing (Day 12) versus WMC errors on Trial 3 (the first post-delay trial) on Day 13 of WRAM testing, as done in prior publications (Braden et al., 2017, 2015; Hiroi et al., 2016; Koebele et al., 2020, 2019). For the VCD-Vehicle group, there was a main effect of Delay Day [F(1,10) = 13.75, p < 0.01, ηG2 = 0.74], where VCD-Vehicle rats made significantly more errors on the post-delay trial compared to the previous day’s performance (Fig. 4A). For the VCD-E2 group, there was no main effect of Delay Day [F(1,10) = 0.51, p = NS, ηG2 = 0.51], such that there was not an increase in WMC errors on Trial 3 following an 8 h delay compared to baseline performance the prior day (Fig. 4B).
Fig. 4.
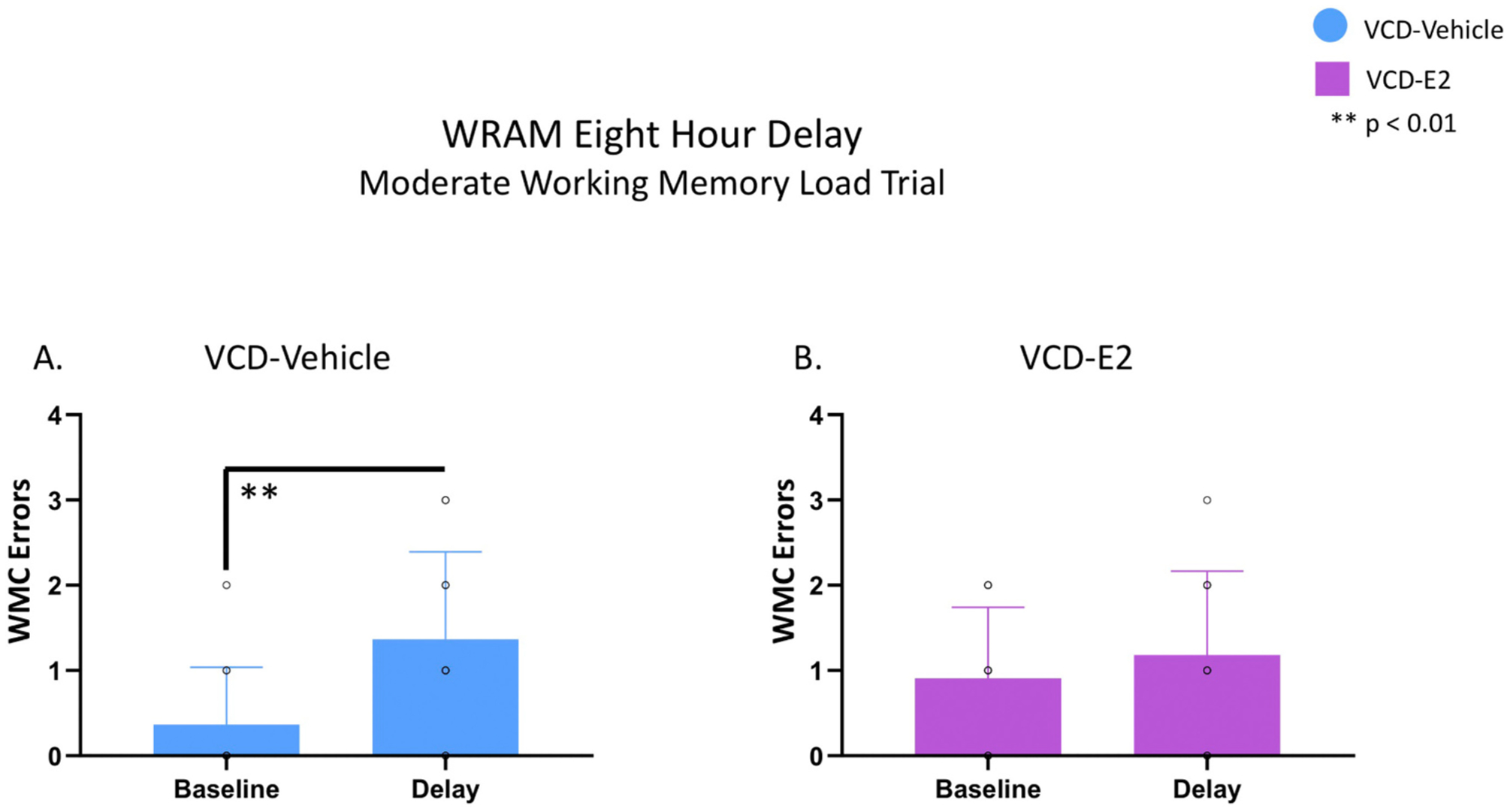
Eight-hour delayed memory retention WRAM test. VCD-Vehicle: n = 11; VCD-E2: n = 11. Individual data points are overlaid on the mean ± SEM (some data point icons are overlapping in the figure). (A) The VCD-Vehicle group exhibited a delay-induced impairment in WMC errors following an 8 hour delay interval compared to the previous day’s performance. (B) The VCD-E2 group working memory performance was not significantly affected by the 8 hour delay interval.
3.2. Morris water maze
There was a main effect of Day across all days of the MM [F(4,80) = 68.36, p < 0.0001, ηG2 = 0.38], and Day did not interact with Treatment (Fig. 5B). Analysis of the probe trial revealed a main effect of Quadrant for both the VCD-Vehicle group [F(1,10) = 64.01, p < 0.0001, ηG2 = 0.14; Fig. 5C] and the VCD-E2 group [F(1,10) = 49.80, p < 0.0001, ηG2 = 0.29; Fig. 5D], whereby rats in each group swam a significantly higher percent of total swim distance in the target, previously platformed quadrant compared to the opposite quadrant. Overall, these data indicate that both groups of rats were able to successfully localize to the spatial location of the platform by the end of MM testing.
3.3. Visible platform
There was a main effect of Trial [F(5,100) = 5.41, p < 0.001, ηG2 = 0.20] and no interaction with Treatment for Latency to Platform. (Fig. 6B). The average escape latency for the VCD-Vehicle group on Trial 1 was 32.19 ± 8.43 s, and 7.87 ± 1.68 s by Trial 6. The average escape latency for the VCD-E2 group on Trial 1 was 17.40 ± 3.86 s, and 7.95 ± 1.32 s by Trial 6. Thus, both groups decreased escape latency on the visible platform task by the last testing trial, with all rats reaching the visible platform in 18 s or less (average 7.91 ± 1.04 s) on Trial 6. Overall, this indicates that all subjects had sufficient intact visual and motoric capacity to perform the general procedural components of water maze tasks.
3.4. Serum hormone levels
Five subjects from the VCD-Vehicle group had E2 levels below the detection limit of the assay and were excluded from the analysis for E2, such that n = 6 for the VCD-Vehicle group and n = 11 for the VCD-E2 group for this analysis. There was a significant effect of Treatment for E2 levels [F(1,15) = 7.81, p < 0.05, η2 = 0.34], where VCD-E2 rats had higher circulating E2 levels compared to VCD-Vehicle rats (Fig. 7A).
Fig. 7.
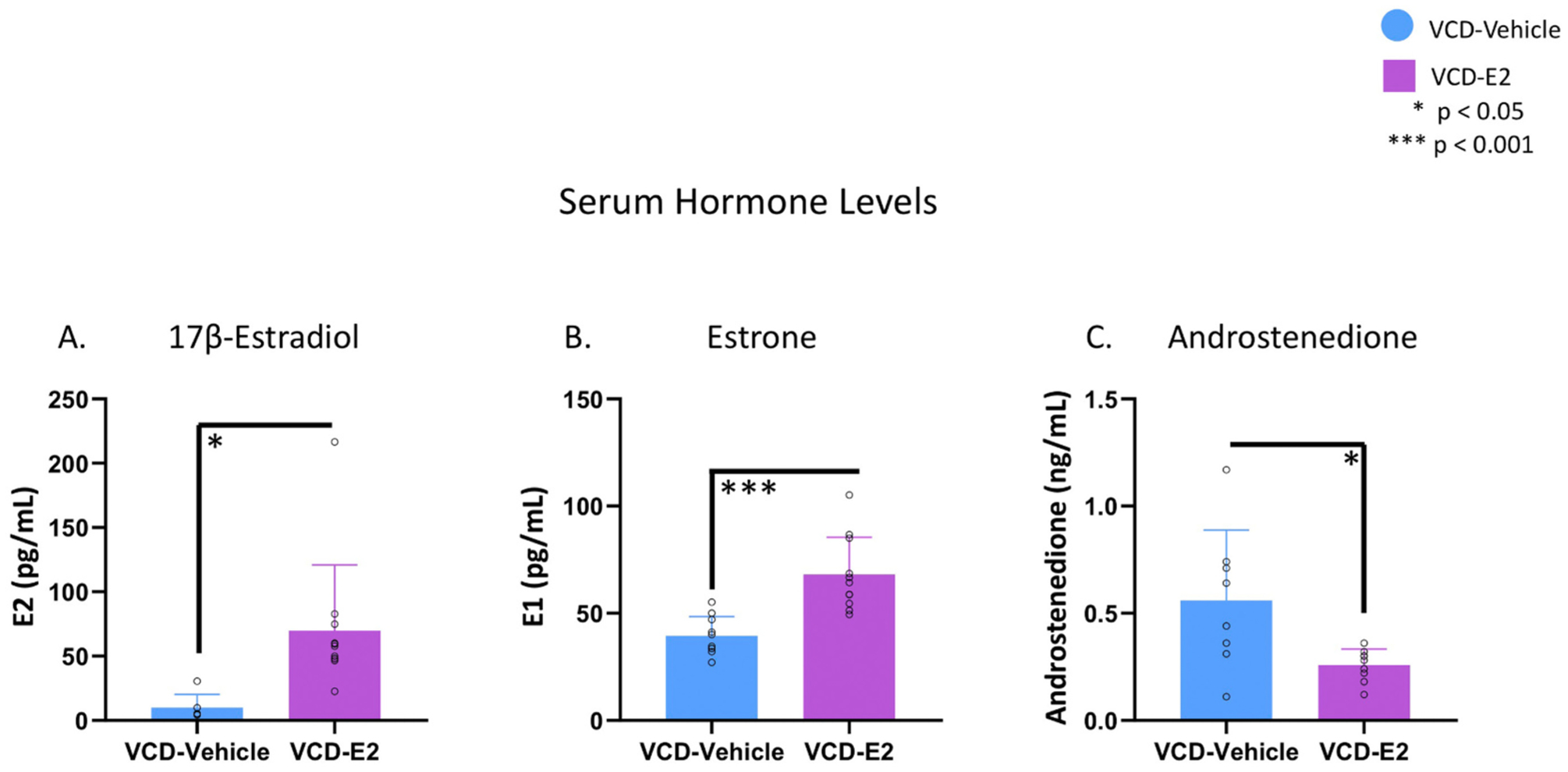
Serum ovarian hormone levels. Individual data points are overlaid on the mean ± SEM. (A) E2 levels were elevated in rats that received exogenous E2 treatment following follicular depletion (p < 0.05). VCD-Vehicle: n = 6; VCD-E2: n = 11. (B) E1 levels were elevated in rats that received exogenous E2 treatment following follicular depletion (p < 0.001). VCD-Vehicle: n = 10; VCD-E2: n = 11. (C) Androstenedione levels were decreased in rats that received exogenous E2 treatment following follicular depletion (p < 0.05). VCD-Vehicle: n = 8; VCD-E2: n = 9.
One subject from the VCD-Vehicle group did not have a sufficient quantity of serum to analyze E1 levels and was excluded from the analysis, such that n = 10 for the VCD-Vehicle group and n = 11 for the VCD-E2 group. There was a significant effect of Treatment for E1 levels [F(1,19) = 21.89, p < 0.001, η2 = 0.54], with VCD-E2 rats showing higher circulating E1 levels compared to VCD-Vehicle rats (Fig. 7B).
Two subjects from the VCD-Vehicle group did not have a sufficient quantity of serum to analyze androstenedione levels and were therefore excluded from the analysis. Furthermore, one subject from the VCD-Vehicle group and two subjects from the VCD-E2 group had androstenedione levels below the detection limit of the assay and were also excluded from the analysis. Therefore, for the androstenedione analysis, there was an n = 8 for the VCD-Vehicle group and n = 9 for the VCD-E2 group. There was a significant effect of Treatment for androstenedione levels [F(1,15) = 7.27, p < 0.05, η2 = 0.33], where VCD-E2 rats had lower circulating androstenedione levels compared to VCD-Vehicle rats (Fig. 7C).
3.5. Ovarian follicle counts
There were no Treatment differences between VCD-Vehicle and VCD-E2 groups for primordial follicles (Fig. 8A), secondary follicles (Fig. 8C), antral follicles (Fig. 8D), or corpora lutea (Fig. 8E). There was a significant effect of Treatment for primary follicles [F(1,20) = 6.72, p < 0.05, η2 = 0.25; Fig. 8B], where VCD-E2 rats had significantly fewer primary follicles present in the ovaries compared to VCD-Vehicle rats. Overall, both VCD-treated groups had significant depletion of ovarian follicles in contrast to follicle counts we have previously reported, wherein follicle counts are up to 10× higher in reproductive tract-intact rats that did not undergo VCD treatment (Frye et al., 2012; Koebele et al., 2019, 2017; Lohff et al., 2006; Mayer et al., 2004, 2002). To demonstrate this, we included follicle counts from an independent data set in our laboratory of ovary-intact, age-matched rats that received the complementary Vehicle injection for VCD administration as a comparison group. Results indicated a significant effect of Treatment for all follicle counts and corpora lutea counts (Fig. 8A–E; Primordials: [F(2,29) = 55.79, p < 0.0001, η2 = 0.79], Primaries: [F(2,29) = 10.35, p < 0.001, η2 = 0.42], Secondaries: [F(2,29) = 43.68, p < 0.0001, η2 = 0.75], Antrals: [F(2,29) = 15.37, p < 0.0001, η2 = 0.52], Corpora Lutea: [F(2,29) = 41.79, p < 0.0001, η2 = 0.74]). Fisher’s PLSD post hoc indicated that the ovary-intact, non-VCD treated group had significantly more follicles and corpora lutea than both the VCD-Vehicle group (Primordials: p < 0.0001; Primaries: p < 0.05; Secondaries: p < 0.0001; Antrals: p < 0.0001; Corpora Lutea: p < 0.0001) and the VCD-E2 group (Primordials: p < 0.0001; Primaries: p < 0.0001; Secondaries: p < 0.0001; Antrals: p < 0.0001; Corpora Lutea: p < 0.0001).
Fig. 8.
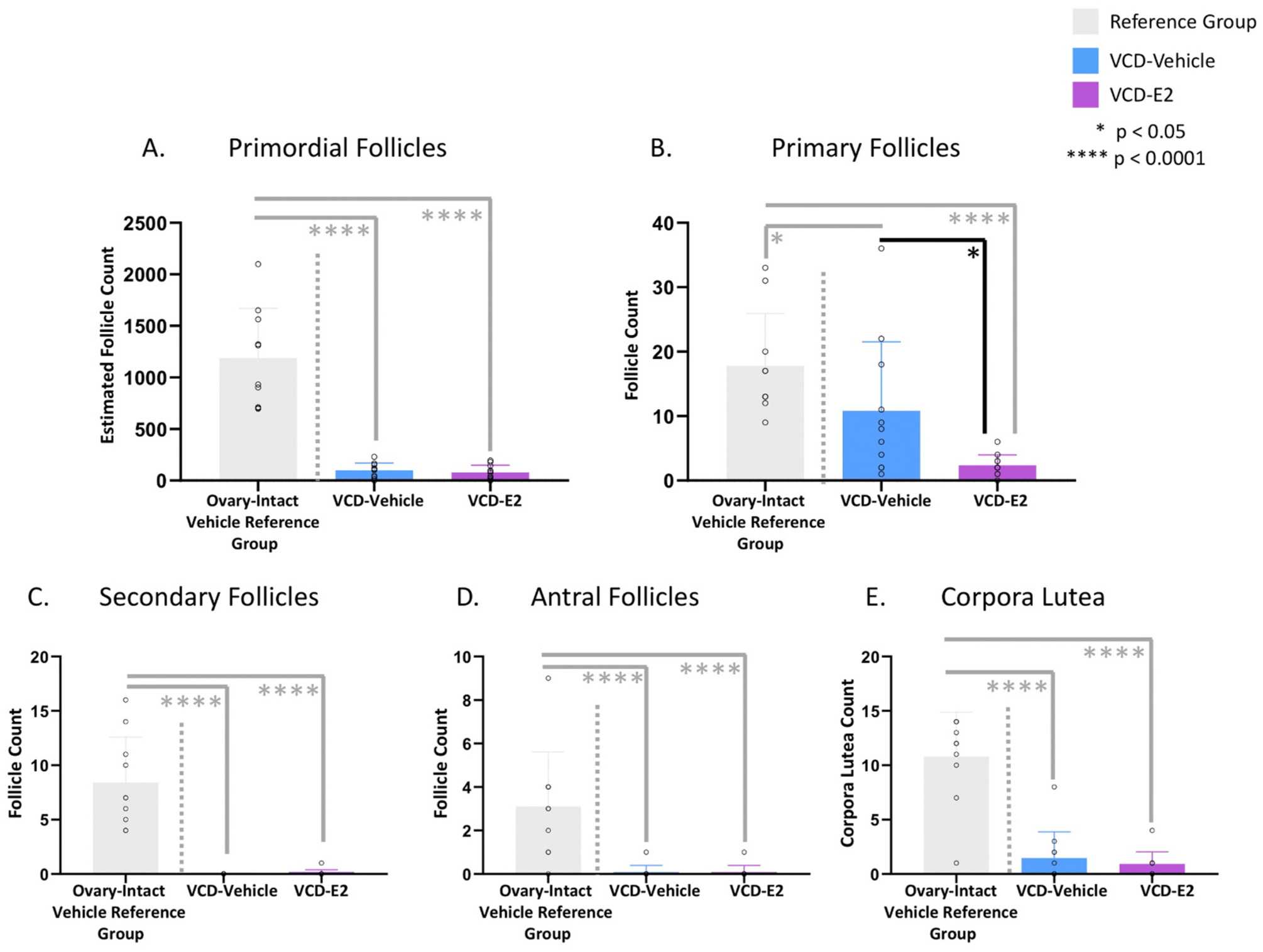
Markers of follicular depletion and hormone status. VCD-Vehicle: n = 11; VCD-E2: n = 11 for all follicle evaluations. An ovary-intact, age-matched, Vehicle control for VCD treatment Reference Group from an independent data set (n = 10) has been included to demonstrate follicular depletion. Individual data points are overlaid on the mean ± SEM (some data point icons are overlapping in the graphs) (A) Primordial follicles were reduced in both VCD-treated groups compared to the Reference Group. VCD-treated groups did not differ in primordial follicle counts. (B) Primary follicles were reduced in both VCD-treated groups compared to the Reference Group. Primary follicles were also decreased in VCD-E2 rats compared to VCD-Vehicle rats (p < 0.05). (C) Secondary follicles were reduced in both VCD-treated groups compared to the Reference Group. VCD-treated groups did not differ in secondary follicle counts. (D) Antral follicles were reduced in both VCD-treated groups compared to the Reference Group. VCD-treated groups did not differ in antral follicle counts. (E) Corpora lutea were reduced in both VCD-treated groups compared to the Reference Group. VCD-treated groups did not differ in corpora lutea counts.
3.6. Vaginal cytology
VCD-E2-treated rats exhibited estrus-like smears containing mostly cornified cells, indicating estrogen stimulation following initiation of daily exogenous hormone treatment. VCD-Vehicle-treated rats displayed either estrus or diestrus smears across the two-day evaluation period, which is consistent with prior observations in our laboratory in middle-aged VCD-treated rats after follicular depletion (Acosta et al., 2010, 2009).
3.7. Body weights
There were no Treatment differences in body weight at the euthanasia time point, indicating that following VCD treatment, the current E2 treatment regimen did not significantly alter body weight compared to Vehicle treatment in ovary-intact, follicle-depleted rats [F(1,20) = 1.85, p = NS, η2 = 0.09; Fig. 9A].
Fig. 9.
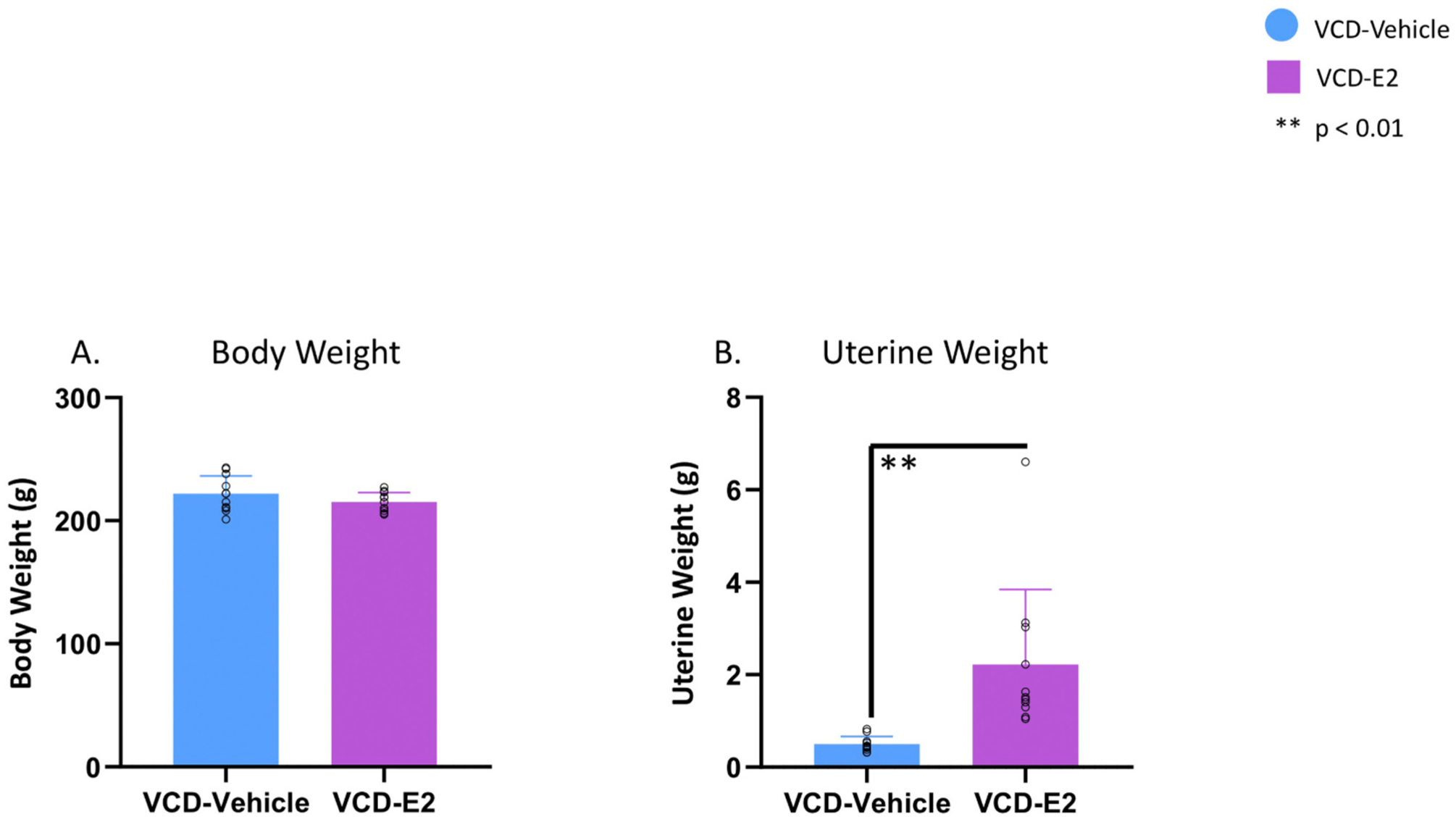
Body weight and uterine weight. Individual data points are overlaid on the mean ± SEM. (A) Overall body weight did not differ between groups at the end of the experiment, regardless of exogenous hormone treatment. (B) Follicle-deplete rats treated with exogenous E2 had significantly heavier uterine weights compared to the VCD-Vehicle group that did not receive exogenous hormone treatment, indicating a stimulatory effect of E2 on uterine tissue.
3.8. Uterine weights
A significant effect of Treatment was present for wet uterine weights [F(1,20) = 12.26, p < 0.01, η2 = 0.38; Fig. 9B], wherein the VCD-E2 group had higher uterine weights compared to VCD-Vehicle rats, indicating that tonic E2 treatment stimulated the uterus, as we and others have previously observed following exogenous E2 administration (Engler-Chiurazzi et al., 2012; Koebele et al., 2020; Prakapenka et al., 2018; Westerlind et al., 1998).
3.9. Correlations
For E2 correlations, as a result of exclusions based on available serum data, n = 6 for the VCD-Vehicle group and n = 11 for the VCD-E2 group. Following the FDR correction, there was a positive correlation between E2 levels and WMI errors made during the Asymptotic Phase of the WRAM [r(4) = 0.899, p < 0.05; R2 = 0.809; Q = 0.09] for the VCD-Vehicle group, such that higher E2 levels were associated with more errors for rats that had follicle-deplete ovaries but did not receive subsequent hormone treatment. This correlation was also present between E2 and WMI errors on Trial 4, the highest working memory load trial, during the Asymptotic Phase of WRAM for the VCD-Vehicle group only [r(4) = 0.954, p < 0.001; R2 = 0.909; Q = 0.04] (Fig. 10A–B). Exogenous E2 administration disrupted the relationship between endogenous E2 levels and maze performance, as E2 levels in the VCD-E2 group did not significantly correlate with the memory measures assessed. It is acknowledged that one subject had an outlying value for circulating E2 levels. When we excluded this subject from the correlation analysis, the correlation remained non-significant; therefore, all subjects are shown in Fig. 10A–B.
Fig. 10.
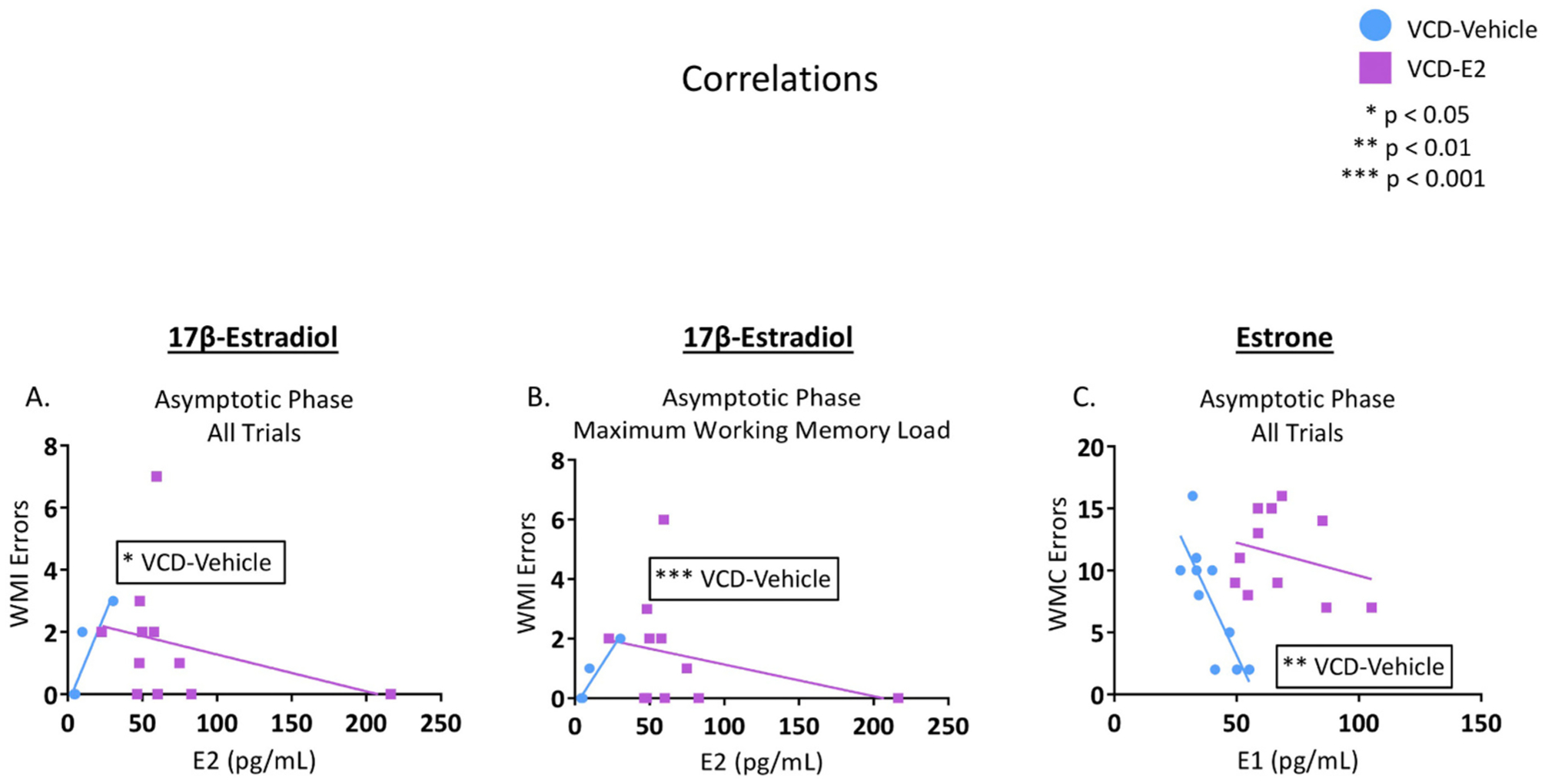
Correlations among hormone markers and cognitive scores (FDR correction set at 0.10). (A–B) Better working memory performance (fewer errors) at the end of WRAM testing was associated with lower E2 levels in the VCD-Vehicle group, but not the VCD-E2 group, regardless of whether the outlying value was included. VCD-Vehicle: n = 6 (some data point icons are overlapping in the figure); VCD-E2: n = 11. (C) Better working memory performance (fewer errors) at the end of WRAM testing was associated with higher E1 levels in the VCD-Vehicle group, but not the VCD-E2 group. VCD-Vehicle: n = 10; VCD-E2: n = 11.
For estrone correlations, as a result of exclusions based on available serum data, n = 10 for the VCD-Vehicle group and n = 11 for the VCD-E2 group. Following the FDR correction, there was a negative correlation between E1 levels and WMC errors during the Asymptotic Phase of WRAM [r(8) = −0.798, p < 0.01; R2 = 0.637; Q = 0.05] for the VCD-Vehicle group, where higher serum E1 levels were associated with fewer WMC errors across all trials of the Asymptotic Phase (Fig. 10C). Again, exogenous E2 administration disrupted the relationship seen in follicle-depleted rats that were not given exogenous E2. Estrone levels in the VCD-E2 group did not significantly correlate with the memory measures assessed.
4. Discussion
This experiment is the first systematic investigation testing the effects of tonic, exogenous E2 treatment on spatial working and reference memory in a transitionally menopausal, ovary-intact, follicle-deplete preclinical model. As previously noted, most of the seminal research on the cognitive impact of estrogens has been in the context of complete ovarian removal via Ovx followed by hormone treatment (e.g. Bimonte and Denenberg, 1999; Black et al., 2018, 2016; Bohacek et al., 2008; Fernandez et al., 2008; Gibbs, 2000; Gibbs et al., 2011a, 2011b, 2009; Harburger et al., 2009; Koebele et al., 2020; Lacreuse, 2006; Markowska and Savonenko, 2002; Prakapenka et al., 2018; Rapp et al., 2003; Rodgers et al., 2010; Savonenko and Markowska, 2003; Talboom et al., 2008; Wallace et al., 2006). While this work has been indisputably transformative in propelling the field of hormones, cognition, and aging forward, it remains essential to better understand the effects of distinct estrogen-containing hormone therapies in the context of an intact reproductive tract undergoing the transition to menopause. In the present experiment, rats in late adulthood (8 months old at the start of the experiment) were administered VCD to induce accelerated follicular depletion in order to model transitionally menopausal women. This age was specifically chosen as a translationally relevant time point with regard to reproductive capacity in females. Although we utilized virgin rats in this study, laboratory rodents, including the Fischer-344 strain used here, are typically retired as breeders by 9 months of age and experience estrous irregularity by 12 months of age. The longitudinal nature of the current experiment allowed us to capture a comparable life stage progression that reflects the human experience. (Koebele et al., 2017; Koebele and Bimonte-Nelson, 2016). Tonic E2 or Vehicle treatment began approximately three months after the first injection of VCD, such that rats were approaching middle-age and had undergone substantial VCD-induced follicular loss by the time hormone treatment was initiated. Rats were then tested on a series of complex behavioral tasks assessing spatial working and reference memory. E2 treatment enhanced the acquisition of a complex spatial working memory task in middle-aged transitionally menopausal rats, as the VCD-E2 group made fewer WMC and WMI errors compared to the VCD-Vehicle group during the Learning Phase of the WRAM. This beneficial effect of E2 was carried by the highest working memory load trial, when rats had the maximum number of items to retain in their short-term/working memory in order to effectively solve the maze task. Interestingly, at the end of testing, when rats had learned the rules of the task and were approaching asymptotic performance, VCD-E2 rats tended to make more working memory errors compared to the VCD-Vehicle group, although this effect did not reach statistical significance for either error measure across all trials. A priori planned analysis of the highest working memory load trial during the Asymptotic Phase revealed that the VCD-E2 group made more WMC errors compared to the VCD-Ve-hicle group; this effect was not present for WMI errors on this same high load trial. As such, at the end of testing, the influence of E2 on spatial working memory after transitional menopause is more ambiguous than the clear E2-induced benefit observed in the Learning Phase of WRAM, and should be further probed in future studies. With the challenge of an 8-hour delay to WRAM testing, follicle-depleted rats given Vehicle exhibited impaired memory retention, while follicle-depleted rats given E2 maintained their performance and did not show impaired memory retention following the delay challenge. This could be interpreted such that E2 treatment conferred a memory retention benefit following a delay interval, whereas Vehicle treatment did not. The E2-induced memory retention enhancements during transitional menopause that we show here are in accordance with rodent studies using surgical menopause models; specifically, we and others have demonstrated protective effects of E2 treatment on delayed memory retention on a working memory task (Harburger et al., 2007; Koebele et al., 2020; Luine et al., 1998; Prakapenka et al., 2018; Witty et al., 2012). However, it is acknowledged that the lack of impairment in the VCD-E2 group may have been the result of a ceiling effect due to elevated WMC errors in the VCD-E2 group on the last day of baseline testing.
In the transitionally menopausal rats tested here, there were neither Treatment differences for the reference memory component of the WRAM, nor were there Treatment differences in the reference memory-only MM task, and as such, E2 effects are likely specific to the spatial working memory domain, as we have previously observed (Koebele et al., 2020, 2019; Prakapenka et al., 2018), rather than a more global change under these parameters. Although sex differences and age effects have been reported in the spatial reference memory MM by our laboratory and others (Koebele et al., 2017; Markowska, 1999), the results here suggest that, at least within the context of a background of ovarian follicular depletion, E2 administration at the given parameters does not influence performance on reference memory. Of additional note, all rats successfully performed on the visible platform control task testing the procedural components of solving a water escape task, and the treatment groups did not differ in this regard.
We aimed to determine whether E2 administration in follicle-depleted rats would result in divergent, potentially beneficial, effects that would contrast with our previously reported findings with CEE, which produced a detrimental effect on spatial working memory in VCD-treated rats, as tested in the WRAM (Acosta et al., 2010). We focused on exogenous E2 treatment in the current study, as prior preclinical research has suggested that E2 administration following complete ovarian hormone loss via Ovx could be beneficial for cognition compared to other types of estrogens. For example, we have previously reported that administration of tonic E1, a metabolite of E2 and the primary estrogenic component of CEE, had impairing effects on spatial working memory in middle-aged Ovx rats (Engler-Chiurazzi et al., 2012), while exogenous E2 treatment has been associated with improved spatial memory in some paradigms with Ovx rats (Bimonte and Denenberg, 1999; Koebele et al., 2020; Prakapenka et al., 2018; Talboom et al., 2008). Further, in a study directly comparing CEE and E2 treatments in Ovx rats, we have shown that E2 benefitted working memory at the end of WRAM testing with a two-day-on, two-day-off E2 treatment schedule, while benefits of CEE treatment did not extend to the end of WRAM testing (Hiroi et al., 2016). In addition to the type of estrogen as a variable of interest, the role of menopause etiology is also a critical consideration for hormone response outcomes. Indeed, it was recently reported that key monoamine levels and metabolic enzymes differed in brain regions important for spatial working memory in response to E2 treatment or estrogen receptor alpha and beta agonists in young adult rats; importantly, the direction and strength of responses were dependent on Ovx or VCD menopause background (Kirshner et al., 2020; Long et al., 2019, 2018). Changes in monoamine and enzyme levels were both brain region- and time- dependent, and effects for monoamine levels were stronger in Ovx rats than VCD rats, potentially illuminating one reason why there have been variable cognitive results following hormone administration in animal models of surgical versus transitional menopause (Long et al., 2019). Considering that over 80% of women experience a natural transition to menopause (NAMS, 2014) and E2 is one of the most- prescribed estrogen-containing hormone therapies (Pinkerton et al., 2017), our findings herein that E2 treatment had some beneficial, and some detrimental, effects on spatial working memory after transitional menopause is of substantial interest. There are many possibilities that have yet to be explored to understand the divergent efficacy of E2 on learning and memory. The female reproductive system is dynamic and responsive to hypothalamic-pituitary-ovarian-axis feedback throughout life, and responsiveness can change with aging and ovarian senescence status (Koebele and Bimonte-Nelson, 2015). Some of the reproductive hormone profiles at play in the follicle-deplete ovary-intact system include low levels of progesterone, which is produced primarily by corpora lutea, as well as androstenedione, an androgen produced by the interstitial tissue of the ovaries that continues to be released even after follicular depletion in rats and women (Fogle et al., 2007; Mayer et al., 2004). We and others have shown that both progesterone and androstenedione impact spatial working memory in the aging Ovx rodents (Bimonte-Nelson et al., 2004, 2003a; Braden et al., 2015, 2010; Camp et al., 2012; Chesler and Juraska, 2000; Harburger et al., 2009, 2007; Mennenga et al., 2015c; Orr et al., 2009); exploring the role of these sex steroid hormones in an ovary-intact, follicle-deplete system will be key in future experiments.
The timing of exogenous hormone administration relative to ovarian follicular depletion was an important consideration and driver for the current study. In the current experiment, exogenous hormone treatment was initiated after substantial follicular depletion occurred. It is possible that the timeline in this experiment may have occurred outside of a critical window of opportunity for global cognitive benefit. For example, the negative cognitive effects observed in the WHIMS have been attributed, in part, to the participants’ age of 65 years or older at the time of study initiation and that women were likely post-menopausal for 10 or more years before receiving hormone therapy. Notably, WHIMS data assessing younger women (the WHIMS-Y arm) did not observe a cognitive detriment. Other large-scale clinical trials have since suggested that beginning hormone therapy within 10 years of the onset of the menopause transition does not have a negative cognitive impact, providing support for the critical window hypothesis (Espeland et al., 2017, 2013; Gleason et al., 2015; Kantarci et al., 2016; Maki et al., 2011; Vaughan et al., 2013; Wharton et al., 2013). As such, methodically investigating the initiation of exogenous E2 treatment prior to total follicular depletion in rodent models is an important future direction to elucidate the role of timing of hormone administration in learning and memory outcomes.
Various physiological and peripheral evaluations of VCD-induced follicular depletion and subsequent hormone treatments are routinely performed in our studies to not only confirm success of our experimental procedures, but also with the aim of yielding insight into a systems-approach understanding of outcomes. E2 treatment resulted in higher circulating E2 and E1 levels than Vehicle treatment in rats that had undergone VCD-induced follicular depletion, as expected. There was also a significant decrease in circulating androstenedione levels in E2-treated versus Vehicle-treated follicle-deplete rats. Given that follicle-depleted ovarian tissue continues to release androgens (Mayer et al., 2004), it is possible that tonic E2 treatment altered hormone feedback loops and biosynthesis pathways to inhibit excess androgen production in the presence of the exogenous E2 treatment. We have shown that the circulating E1:E2 ratio is correlated with cognitive performance in Ovx rats (Engler-Chiurazzi et al., 2011), and androgens can be aromatized to estrogens both peripherally and within the brain; as such, the ratio of circulating androgens to estrogens may prove to be an important factor in cognitive outcomes. Post-menopausal women have low, sometimes undetectable, circulating estrogen levels following complete follicular depletion of the ovaries, while more than half of the VCD-Vehicle rats still had detectable levels of E2. Therefore, E2 replacement prior to a final decline in E2 levels may interact with endogenous levels or attenuate possible beneficial cognitive effects in the rat. Our reported E2 levels in the rat VCD model are in line with other reports in the literature (Carolino et al., 2019; Frye et al., 2012; Kirshner et al., 2020; Kirshner and Gibbs, 2018; Koebele et al., 2017; Long et al., 2019, 2018). Interestingly, in the VCD-Vehicle group, higher E2 levels were associated with more WMC errors at the end of WRAM testing. Of note, this E2 correlation should be interpreted with caution, as 5/11 VCD-Vehicle rats had undetectable circulating E2 levels, and as a result, there was a low number of subjects included in this correlation. Higher E1 levels were associated with fewer WMC errors at the end of WRAM testing. Rats receiving exogenous E2 treatment did not show correlations between circulating ovarian hormone levels and cognitive performance. Thus, the collective correlation data suggest that exogenous E2 administration alters or disrupts relationships between endogenous circulating hormones and working memory in a follicle-deplete system without exogenous hormone treatment. It has recently been reported that different hormone therapy regimens uniquely alter natural associations among endogenously-derived circulating hormone levels in recently menopausal women (Kling et al., 2019); in the future, it will also be crucial to investigate relationships among reproductive hormones and cognition in humans.
Rats experience some natural follicular depletion with age, though not to the extent to which human females do (Finch, 2014). We and others have previously reported ovarian follicle counts for ovary-intact rats without VCD-induced follicular depletion (Frye et al., 2012; Koebele et al., 2019, 2017; Lohff et al., 2006; Mayer et al., 2002). In the current report, we included an independent reference group from another experiment in our laboratory where rats were given the Vehicle control regimen for VCD and retained their ovaries (Koebele et al., 2017). We quantified ovarian follicle count data for all groups and found that both VCD-Vehicle- and VCD-E2- treated groups had substantially depleted ovarian follicle and corpora lutea counts relative to the ovary-intact, age-matched, non-VCD-treated reference group. Overall, this comparison additionally confirmed that VCD induced the expected accelerated follicular depletion, regardless of whether Vehicle treatment or E2 treatment was administered. While both VCD-treated groups in the current report showed significant follicular depletion, the E2-treated rats had statistically fewer primary follicles compared to Vehicle-treated rats. The relationship between exogenous E2 administration and ovarian follicular atresia rate is not well established. In one study utilizing sexually immature, 28-day old rats, it was reported that E2 administration concomitantly administered with VCD treatment attenuated depletion of primary follicles 15 days later (Thompson et al., 2002). That simultaneous E2 and VCD administration was protective against VCD-induced follicular depletion in prepubertal rats in the Thompson study, but the current experiment demonstrates a decrease in primary follicles in rats treated with E2 in the post-follicular depletion time frame, highlights that the timing of E2 treatment, as related to age and concurrent VCD administration, could affect follicular depletion. Specifically, although follicle estimates are substantially reduced in VCD-induced follicle-depleted rats compared to ovary-intact rats, it is acknowledged that a small number of healthy immature follicles remain quantifiable in VCD-treated rats. These follicles could, in turn, be influenced by the presence or absence of exogenous E2 treatment. The ovarian follicle reserve is unresponsive to gonadotropin stimulation; rather, primordial recruitment to growing follicle status is likely mediated by other signaling pathways and mechanisms including Anti-Müllerian Hormone and steroid hormones, particularly estrogens (Chakravarthi et al., 2020; Gougeon, 2010, 1998; Gougeon and Chainy, 1987; Hsueh et al., 2015; La Marca et al., 2009). One report assessed perinatal rat ovaries and showed that E2 or progesterone treatment decreased the number of primary follicles while not affecting the estimated number of primordial follicles, and antagonizing ovarian estrogen receptors prevented this effect in vitro. However, results of E2 regulation of follicle recruitment in vivo were less clear (Kezele and Skinner, 2003). A more recent publication found estrogen receptor (ER) β-null (ESR2−/−) rats had increased activated-to-primordial follicle counts (Chakravarthi et al., 2020). Taken together, these data suggest a regulatory role for estrogen on active ovarian follicle recruitment driven by estrogen receptor binding. In the context of the current experiment, perhaps E2 administration in VCD-treated rats arrested remaining viable primordial follicles from moving into the primary phase of follicular development. In turn, because follicles can become atretic at any growth stage (Gougeon, 2010; Hsueh et al., 1994), remaining healthy follicles in the VCD-E2 treated rats were depleted at the primordial stage prior to active recruitment. On the other hand, VCD-Vehicle-treated rats may have experienced uninhibited follicle recruitment from remaining viable follicles due to insufficient endogenous E2 inhibition, leading to a larger number of quantifiable primary follicles. Primary follicles in the Vehicle-treated rats may become apoptotic at later growth stages (e.g. secondary, antral) resulting from disrupted HPO axis feedback following VCD-induced follicular depletion. Replication of this effect in VCD-treated rats receiving exogenous hormone treatment as well as further investigation into mechanisms regulating ovarian follicle activation and apoptosis during the transition to reproductive senescence is warranted.
The current report is the first to assess the cognitive impact of E2 treatment in a follicle-deplete, reproductive tract intact, middle-aged rat model. The current findings, collectively with prior literature, emphasize the complexity of menopausal hormone therapy administration in the context of estrogen type, menopause etiology, and cognitive outcomes. Indeed, estrogens can have beneficial, neutral, or even detrimental effects on learning and memory in women as well as animal models. Here, in rats that had undergone transitional menopause and maintained their follicle-deplete ovaries, E2 administration had both enhancing and impairing effects on burdened working memory that were dependent on the phase of learning and retention in the WRAM. Effects were specific to working memory, as reference memory performance within the WRAM as well as the MM was not impacted by E2 treatment. In contrast to our prediction, the dose and regimen of E2 used in the current experiment impaired working memory at the end of WRAM testing in a fashion similar to the cognitively impairing effects of CEE administration in middle aged VCD-treated rats our laboratory has previously demonstrated (Acosta et al., 2010). However, it is no-table that in the current experiment, the utilized dose and regimen of E2 did have a beneficial effect on working memory during learning in the WRAM, suggesting some, albeit transient, cognitive enhancement. Thus, the search continues for hormone therapy parameters that effectively provide an optimal benefit for cognitive function in the reproductive tract-intact, follicle-depleted background. Future investigations will build upon this foundation to systematically assess the role of individual and combined exogenous hormone therapy regimens, with particular notation of treatment timing and extent of follicular depletion at treatment initiation. Translationally, these investigations aim to improve current practices in hormone therapy treatments by deciphering which parameters are optimal for a given woman’s health history and menopause status in order to alleviate physical symptoms and enrich overall quality of life without compromising cognitive health. Such findings will allow women to make informed personalized health care decisions alongside their medical professionals to enhance overall quality of life and healthy brain aging in the post-reproductive life stage.
Funding
Dr. Heather Bimonte-Nelson: NIA (AG028084), state of Arizona, Arizona Department of Health Services (ADHS14-052688), NIH Alzheimer’s Disease Core Center (P30AG019610), Arizona State University Office of Knowledge Enterprise Development, College of Liberal Arts and Sciences, and Department of Psychology.
Footnotes
These subjects were part of a larger cohort of experimental animals, undergoing Sham surgery for Ovx 74 days after VCD injections were initiated. This procedure involved brief isoflurane anesthetization, small bilateral, dorsolateral skin and muscle incisions, skin sutures, and carprofen administration for pain relief. Additionally, beginning at hormone pump surgery (PEG Vehicle or E2 in PEG), all subjects in the VCD-Vehicle group and the VCD-E2 group received daily subcutaneous injections of 0.5 mL PEG until the end of the experiment.
References
- ACOG, 2014. Management of menopausal symptoms. Obstet. Gynecol 123, 202–216. 10.1097/01.AOG.0000441353.20693.78. [DOI] [PubMed] [Google Scholar]
- Acosta JI, Mayer L, Talboom JS, Tsang CWS, Smith CJ, Enders CK, Bimonte-Nelson HA, 2009. Transitional versus surgical menopause in a rodent model: etiology of ovarian hormone loss impacts memory and the acetylcholine system. Endocrinology 150, 4248–4259. 10.1210/en.2008-1802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acosta JI, Mayer LP, Braden BB, Nonnenmacher S, Mennenga SE, Bimonte-Nelson HA, 2010. The cognitive effects of conjugated equine estrogens depend on whether menopause etiology is transitional or surgical. Endocrinology 151, 3795–3804. 10.1210/en.2010-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Safi ZA, Santoro N, 2014. Menopausal hormone therapy and menopausal symptoms. Fertil. Steril 101, 905–915. 10.1016/j.fertnstert.2014.02.032. [DOI] [PubMed] [Google Scholar]
- Baber RJ, Panay N, Fenton A, 2016. 2016 IMS recommendations on women’s midlife health and menopause hormone therapy. Climacteric 19, 109–150. 10.3109/13697137.2015.1129166. [DOI] [PubMed] [Google Scholar]
- Bakeman R, 2005. Recommended effect size statistics for repeated measures designs. Behav. Res. Methods 37, 379–384. 10.3758/BF03192707. [DOI] [PubMed] [Google Scholar]
- Baker TG, 1963. A quantitative and cytological study of germ cells in human ovaries. Proc. R. Soc. London, Ser. B, Biol. Sci 158, 417–433. [DOI] [PubMed] [Google Scholar]
- Barha CK, Galea LAM, 2013. The hormone therapy, Premarin, impairs hippocampus-dependent spatial learning and memory and reduces activation of new granule neurons in response to memory in female rats. Neurobiol. Aging 34, 986–1004. 10.1016/j.neurobiolaging.2012.07.009. [DOI] [PubMed] [Google Scholar]
- Benjamini Y, Hochberg Y, 1995. Controlling the false discovery rate : a practical and powerful approach to multiple testing. R. Stat. Soc 57, 289–300. [Google Scholar]
- Bhavnani BR, Stanczyk FZ, 2014. Pharmacology of conjugated equine estrogens: efficacy, safety and mechanism of action. J. Steroid Biochem. Mol. Biol 142, 16–29. 10.1016/j.jsbmb.2013.10.011. [DOI] [PubMed] [Google Scholar]
- Bimonte HA, Denenberg VH, 1999. Estradiol facilitates performance as working memory load increases. Psychoneuroendocrinology 24, 161–173. 10.1016/S0306-4530(98)00068-7. [DOI] [PubMed] [Google Scholar]
- Bimonte HA, Hyde LA, Hoplight BJ, Denenberg VH, 2000. In two species, females exhibit superior working memory and inferior reference memory on the water radial-arm maze. Physiol. Behav 70, 311–317. [DOI] [PubMed] [Google Scholar]
- Bimonte HA, Granholm ACE, Seo H, Isacson O, 2002. Spatial memory testing decreases hippocampal amyloid precursor protein in young, but not aged, female rats. Neurosci. Lett 298, 50–54. [DOI] [PubMed] [Google Scholar]
- Bimonte HA, Nelson ME, Granholm ACE, 2003. Age-related deficits as working memory load increases: relationships with growth factors. Neurobiol. Aging 24, 37–48. 10.1016/S0197-4580(02)00015-5. [DOI] [PubMed] [Google Scholar]
- Bimonte-Nelson HA, Singleton RS, Hunter CL, Price KL, Moore AB, Granholm A-CE, 2003a. Ovarian hormones and cognition in the aged female rat: I. Long-term, but not short-term, ovariectomy enhances spatial performance. Behav. Neurosci 117, 1395–1406. 10.1037/h0087876. [DOI] [PubMed] [Google Scholar]
- Bimonte-Nelson HA, Singleton RS, Nelson ME, Eckman CB, Barber J, Scott TY, Granholm ACE, 2003b. Testosterone, but not nonaromatizable dihydrotestosterone, improves working memory and alters nerve growth factor levels in aged male rats. Exp. Neurol 181, 301–312. 10.1016/S0014-4886(03)00061-X. [DOI] [PubMed] [Google Scholar]
- Bimonte-Nelson HA, Singleton RS, Williams BJ, Granholm A-CE, 2004. Ovarian hormones and cognition in the aged female rat: II. Progesterone supplementation reverses the cognitive enhancing effects of ovariectomy. Behav. Neurosci 118, 707–714. 10.1037/0735-7044.118.4.707. [DOI] [PubMed] [Google Scholar]
- Bimonte-Nelson HA, Daniel JM, Koebele SV, 2015. The mazes. In: Bimonte-Nelson HA (Ed.), The Maze Book: Theories, Practice, and Protocols for Testing Rodent Cognition. Springer US, New York, pp. 37–72. 10.1007/978-1-4939-2159-1_2. [DOI] [Google Scholar]
- Bizon JL, LaSarge CL, Montgomery KS, McDermott AN, Setlow B, Griffith WH, 2009. Spatial reference and working memory across the lifespan of male Fischer 344 rats. Neurobiol. Aging 30, 646–655. 10.1016/j.neurobiolaging.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black KL, Witty CF, Daniel JM, 2016. Previous midlife oestradiol treatment results in Long-term maintenance of hippocampal oestrogen receptor α levels in ovar-iectomised rats: mechanisms and implications for memory. J. Neuroendocrinol 28. 10.1111/jne.12429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Black KL, Baumgartner NE, Daniel JM, 2018. Lasting impact on memory of midlife exposure to exogenous and endogenous estrogens. Behav. Neurosci 132, 547–551. 10.1037/bne0000270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohacek J, Daniel JM, 2007. Increased daily handling of ovariectomized rats enhances performance on a radial-maze task and obscures effects of estradiol replacement. Hormones and Behavior 52 (2), 237–243. 10.1016/j.yhbeh.2007.04.010. [DOI] [PubMed] [Google Scholar]
- Bohacek J, Bearl AM, Daniel JM, 2008. Long-term ovarian hormone deprivation alters the ability of subsequent oestradiol replacement to regulate choline acetyl-transferase protein levels in the hippocampus and prefrontal cortex of middle-aged rats. J. Neuroendocrinol 20, 1023–1027. 10.1111/j.1365-2826.2008.01752.x. [DOI] [PubMed] [Google Scholar]
- Braden BB, Talboom JS, Crain ID, Simard AR, Lukas RJ, Prokai L, Scheldrup MR, Bowman BL, Bimonte-Nelson HA, 2010. Medroxyprogesterone acetate impairs memory and alters the GABAergic system in aged surgically menopausal rats. Neurobiol. Learn. Mem 93, 444–453. 10.1016/j.nlm.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braden BB, Kingston ML, Whitton E, Lavery C, Tsang CWS, Bimonte-Nelson HA, 2015. The GABA-A antagonist bicuculline attenuates progesterone-induced memory impairments in middle-aged ovariectomized rats. Front. Aging Neurosci 7, 1–8. 10.3389/fnagi.2015.00149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braden BB, Andrews MG, Acosta JI, Mennenga SE, Lavery C, Bimonte-Nelson HA, 2017. A comparison of progestins within three classes: differential effects on learning and memory in the aging surgically menopausal rat. Behav. Brain Res 322, 258–268. 10.1016/j.bbr.2016.06.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camp BW, Gerson JE, Tsang CWS, Villa SR, Acosta JI, Blair Braden B, Hoffman AN, Conrad CD, Bimonte-Nelson HA, 2012. High serum androstenedione levels correlate with impaired memory in the surgically menopausal rat: a replication and new findings. Eur. J. Neurosci 36, 3086–3095. 10.1111/j.1460-9568.2012.08194.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carolino ROG, Barros PT, Kalil B, Anselmo-Franci J, 2019. Endocrine profile of the VCD-induced perimenopausal model rat. PLoS One 14, 1–19. 10.1371/journal.pone.0226874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakravarthi VP, Ghosh S, Roby KF, Wolfe MW, Rumi MAK, 2020. A gate-keeping role of ESR2 to maintain the primordial follicle reserve. Endocrinology 161, 1–15. 10.1210/endocr/bqaa037. [DOI] [PubMed] [Google Scholar]
- Chesler EJ, Juraska JM, 2000. Acute administration of estrogen and progesterone impairs the acquisition of the spatial morris water maze in ovariectomized rats. Horm. Behav 38, 234–242. 10.1006/hbeh.2000.1626. [DOI] [PubMed] [Google Scholar]
- Cohen J, 1988. Statistical Power Analysis for the Behavioral Sciences, 2nd ed. Erlbaum, Hisdale, NJ. [Google Scholar]
- Cohen J, 1992. A power primer. Psychol. Bull 112, 155–159. 10.1037/0033-2909.112.1.155. [DOI] [PubMed] [Google Scholar]
- Coker LH, Espeland MA, Rapp SR, Legault C, Resnick SM, Hogan P, Gaussoin S, Dailey M, Shumaker SA, 2010. Postmenopausal hormone therapy and cognitive outcomes: the Women’s Health Initiative memory study (WHIMS). J. Steroid Biochem. Mol. Biol 118, 304–310. 10.1016/j.jsbmb.2009.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crawford SL, Crandall CJ, Derby CA, El Khoudary SR, Waetjen LE, Fischer M, Joffe H, 2018. Menopausal hormone therapy trends before versus after 2002: impact of the Women’s Health Initiative study results. Menopause 26, 588–597. 10.1097/GME.0000000000001282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniel JM, 2013. Estrogens, estrogen receptors, and female cognitive aging: the impact of timing. Horm. Behav 63, 231–237. 10.1016/j.yhbeh.2012.05.003. [DOI] [PubMed] [Google Scholar]
- Daniel JM, Hulst JL, Berbling JL, 2006. Estradiol replacement enhances working memory in middle-aged rats when initiated immediately after ovariectomy but not after a long-term period of ovarian hormone deprivation. Endocrinology 147, 607–614. 10.1210/en.2005-0998. [DOI] [PubMed] [Google Scholar]
- Dubal DB, Wise PM, 2001. Neuroprotective effects of estradiol in middle-aged female rats. Endocrinology 142, 43–48. 10.1210/en.142.1.43. [DOI] [PubMed] [Google Scholar]
- Engler-Chiurazzi EB, Tsang C, Nonnenmacher S, Liang WS, Corneveaux JJ, Prokai L, Huentelman MJ, Bimonte-Nelson HA, 2011. Tonic Premarin dose-dependently enhances memory, affects neurotrophin protein levels and alters gene expression in middle-aged rats. Neurobiol. Aging 32, 680–697. 10.1016/j.neurobiolaging.2009.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engler-Chiurazzi EB, Talboom JS, Braden BB, Tsang CWS, Mennenga S, Andrews M, Demers LM, Bimonte-Nelson HA, 2012. Continuous estrone treatment impairs spatial memory and does not impact number of basal forebrain cholinergic neurons in the surgically menopausal middle-aged rat. Horm. Behav 62, 1–9. 10.1016/j.yhbeh.2012.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espeland MA, Rapp SR, Shumaker SA, Brunner R, Manson JE, Sherwin BB, Hsia J, Margolis KL, Hogan PE, Wallace R, Dailey M, Freeman R, Hays J, 2004. Conjugated equine estrogens and global cognitive function in postmenopausal women: Women’s Health Initiative memory study. JAMA 291, 2959–2968. 10.1001/jama.291.24.2959. [DOI] [PubMed] [Google Scholar]
- Espeland MA, Shumaker SA, Leng I, Manson JE, Brown CM, Leblanc ES, Vaughan L, Robinson J, Rapp SR, Goveas JS, Lane D, Wactawski-Wende J, Stefanick ML, Li W, Resnick SM, 2013. Long-term effects on cognitive function of postmenopausal hormone therapy prescribed to women aged 50 to 55 years. JAMA Intern. Med 173, 1429–1436. 10.1001/jamainternmed.2013.7727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espeland MA, Rapp SR, Manson JAE, Goveas JS, Shumaker SA, Hayden KM, Weitlauf JC, Gaussoin SA, Baker LD, Padula CB, Hou L, Resnick SM, 2017. Long-term effects on cognitive trajectories of postmenopausal hormone therapy in two age groups. J. Gerontol. A Biol. Sci. Med. Sci 72, 838–845. 10.1093/gerona/glw156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez SM, Lewis MC, Pechenino AS, Harburger LL, Orr PT, Gresack JE, Schafe GE, Frick KM, 2008. Estradiol-induced enhancement of object memory consolidation involves hippocampal extracellular signal-regulated kinase activation and membrane-bound estrogen receptors. J. Neurosci 28, 8660–8667. 10.1523/JNEUROSCI.1968-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finch CE, 2014. The menopause and aging, a comparative perspective. J. Steroid Biochem. Mol. Biol 142, 132–141. 10.1016/j.jsbmb.2013.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fogle RH, Stanczyk FZ, Zhang X, Paulson RJ, 2007. Ovarian androgen production in postmenopausal women. J. Clin. Endocrinol. Metab 92, 3040–3043. 10.1210/jc.2007-0581. [DOI] [PubMed] [Google Scholar]
- Foster TC, Sharrow KM, Kumar A, Masse J, 2003. Interaction of age and chronic estradiol replacement on memory and markers of brain aging. Neurobiol. Aging 24, 839–852. 10.1016/S0197-4580(03)00014-9. [DOI] [PubMed] [Google Scholar]
- Frick KM, 2015. Molecular mechanisms underlying the memory-enhancing effects of estradiol. Horm. Behav 74, 4–18. 10.1016/j.yhbeh.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frye JB, Lukefahr AL, Wright LE, Marion SL, Hoyer PB, Funk JL, 2012. Modeling perimenopause in sprague-dawley rats by chemical manipulation of the transition to ovarian failure. Comp. Med 62, 193–202. [PMC free article] [PubMed] [Google Scholar]
- Gibbs RB, 2000. Long-term treatment with estrogen and progesterone enhances acquisition of a spatial memory task by ovariectomized aged rats. Neurobiol. Aging 21, 107–116. 10.1016/S0197-4580(00)00103-2. [DOI] [PubMed] [Google Scholar]
- Gibbs RB, Mauk R, Nelson D, Johnson DA, 2009. Donepezil treatment restores the ability of estradiol to enhance cognitive performance in aged rats: evidence for the cholinergic basis of the critical period hypothesis. Horm. Behav 56, 73–83. 10.1016/j.yhbeh.2009.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbs RB, Chipman AM, Hammond R, Nelson D, 2011a. Galanthamine plus estradiol treatment enhances cognitive performance in aged ovariectomized rats. Horm. Behav 60, 607–616. 10.1016/j.yhbeh.2011.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibbs RB, Chipman AM, Nelson D, 2011b. Donepezil plus estradiol treatment enhances learning and delay-dependent memory performance by young ovariectomized rats with partial loss of septal cholinergic neurons. Horm. Behav 59, 503–511. 10.1016/j.yhbeh.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gleason CE, Dowling NM, Wharton W, Manson JAE, Miller VM, Atwood CS, Brinton EA, Cedars MI, Lobo RA, Merriam GR, Neal-Perry G, Santoro NF, Taylor HS, Black DM, Budoff MJ, Hodis HN, Naftolin F, Harman SM, Asthana S, 2015. Effects of hormone therapy on cognition and mood in recently postmenopausal women: findings from the randomized, controlled KEEPS–cognitive and affective study. PLoS Med. 12, 1–25. 10.1371/journal.pmed.1001833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldman J, Murr A, Cooper R, 2007. The rodent estrous cycle: characterization of vaginal cytology and its utility in toxicological studies. Birth defects Res. Part B 80, 84–97. [DOI] [PubMed] [Google Scholar]
- Golub MS, Germann SL, Mercer M, Gordon MN, Morgan DG, Mayer LP, Hoyer PB, 2008. Behavioral consequences of ovarian atrophy and estrogen replacement in the APPswe mouse. Neurobiol. Aging 29, 1512–1523. 10.1016/j.neurobiolaging.2007.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gougeon A, 1998. Ovarian follicular growth in humans: ovarian ageing and population of growing follicles. Maturitas 30, 137–142. 10.1016/S0378-5122(98)00069-3. [DOI] [PubMed] [Google Scholar]
- Gougeon A, 2010. Human ovarian follicular development: from activation of resting follicles to preovulatory maturation. Ann. Endocrinol. (Paris) 71, 132–143. 10.1016/j.ando.2010.02.021. [DOI] [PubMed] [Google Scholar]
- Gougeon A, Chainy GB, 1987. Morphometric studies of small follicles in ovaries of women at different ages. J. Reprod. Fertil 81, 433–442. [DOI] [PubMed] [Google Scholar]
- Haas JR, Christian PJ, Hoyer PB, 2007. Effects of impending ovarian failure induced by 4-vinylcyclohexene diepoxide on fertility in C57BL/6 female mice. Comp. Med 57, 443–449. [PubMed] [Google Scholar]
- Harburger LL, Bennett JC, Frick KM, 2007. Effects of estrogen and progesterone on spatial memory consolidation in aged females. Neurobiol. Aging 28, 602–610. 10.1016/j.neurobiolaging.2006.02.019. [DOI] [PubMed] [Google Scholar]
- Harburger LL, Saadi A, Frick KM, 2009. Dose-dependent effects of post-training estradiol plus progesterone treatment on object memory consolidation and hippocampal extracellular signal-regulated kinase activation in young ovariectomized mice. Neuroscience 160, 6–12. 10.1016/j.neuroscience.2009.02.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harlow SD, Gass M, Hall JE, Lobo R, Maki P, Rebar RW, Sherman S, Sluss PM, de Villiers TJ, FRCOG, FCOG(SA), 2012. Executive summary of the stages of reproductive aging workshop + 10: addressing the unfinished agenda of staging reproductive aging. Menopause 19, 387–395. 10.1097/gme.0b013e31824d8f40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hersh AL, Stefanick ML, Stafford RS, 2004. National use of postmenopausal hormone therapy: annual trends and reponse to recent evidence. JAMA 291, 47–53. [DOI] [PubMed] [Google Scholar]
- Hiroi R, Weyrich G, Koebele SV, Mennenga SE, Talboom JS, Hewitt LT, Lavery CN, Mendoza P, Jordan A, Bimonte-Nelson HA, 2016. Benefits of hormone therapy estrogens depend on estrogen type: 17beta-estradiol and conjugated equine estrogens have differential effects on cognitive, anxiety-like, and depressive-like behaviors and increase tryptophan hydroxylase-2 mRNA levels in dorsal. Front. Neurosci 10, 1–20. 10.3389/fnins.2016.00517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman BL, Schorge JO, Schaffer JI, Halvorson KD, Bradshaw FG, Cunningham LE, 2012. Williams Gynecology, 2nd ed. McGraw Hill Education, New York. [Google Scholar]
- Hoyer PB, Devine PJ, Hu X, Thompson KE, Sipes IG, 2001. Ovarian toxicity of 4-vinylcyclohexene diepoxide: a mechanistic model. Toxicol. Pathol 29, 91–99. [DOI] [PubMed] [Google Scholar]
- Hsueh AJW, Billig H, Tsafriri A, 1994. Ovarian follicle atresia: a hormonally controlled apoptotic process. Endocr. Rev 15, 707–724. [DOI] [PubMed] [Google Scholar]
- Hsueh AJW, Kawamura K, Cheng Y, Fauser BCJM, 2015. Intraovarian control of early folliculogenesis. Endocr. Rev 36, 1–24. 10.1210/er.2014-1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X, Christian PJ, Thompson KE, Sipes IG, Hoyer PB, 2001. Apoptosis induced in rats by 4-vinylcyclohexene diepoxide is associated with activation of the caspase cascades. Biol. Reprod 65, 87–93. 10.1095/biolreprod65.1.87. [DOI] [PubMed] [Google Scholar]
- Kantarci K, Lowe VJ, Lesnick TG, Tosakulwong N, Bailey KR, Fields JA, Shuster LT, Zuk SM, Senjem ML, Mielke MM, Gleason C, Jack CR, Rocca WA, Miller VM, Tierney M, 2016. Early postmenopausal transdermal 17β-estradiol therapy and amyloid-β deposition. J. Alzheimers Dis 53, 547–556. 10.3233/JAD-160258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kezele P, Skinner MK, 2003. Regulation of ovarian primordial follicle assembly and development by estrogen and progesterone: endocrine model of follicle assembly. Endocrinology 144, 3329–3337. 10.1210/en.2002-0131. [DOI] [PubMed] [Google Scholar]
- Kirshner ZZ, Gibbs RB, 2018. Use of the REVERT® total protein stain as a loading control demonstrates significant benefits over the use of housekeeping proteins when analyzing brain homogenates by Western blot: an analysis of samples representing different gonadal hormone states. Mol. Cell. Endocrinol 473, 156–165. 10.1016/j.mce.2018.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirshner ZZ, Yao JK, Li J, Long T, Nelson D, Gibbs RB, 2020. Impact of estrogen receptor agonists and model of menopause on enzymes involved in brain metabolism, acetyl-CoA production and cholinergic function. Life Sci. 256. 10.1016/j.lfs.2020.117975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kling JM, Dowling NM, Bimonte-Nelson HA, Gleason CE, Kantarci K, Manson JE, Taylor HS, Brinton EA, Lobo RA, Cedars MI, Pal L, Neal-Perry G, Naftolin F, Harman SM, Miller VM, 2019. Impact of menopausal hormone formulations on pituitary-ovarian regulatory feedback. Am J Physiol Regul Integr Comp Physiol 317, R912–R920. 10.1152/ajpregu.00234.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Bimonte-Nelson HA, 2015. Trajectories and phenotypes with estrogen exposures across the lifespan: what does goldilocks have to do with it? Horm. Behav 74, 86–104. 10.1016/j.yhbeh.2015.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Bimonte-Nelson HA, 2016. Modeling menopause: the utility of rodents in translational behavioral endocrinology research. Maturitas 87, 5–17. 10.1016/j.maturitas.2016.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Bimonte-Nelson HA, 2017. The endocrine-brain-aging triad where many paths meet: female reproductive hormone changes at midlife and their influence on circuits important for learning and memory. Exp. Gerontol 94, 14–23. 10.1016/j.exger.2016.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Mennenga SE, Hiroi R, Quihuis AM, Hewitt LT, Poisson ML, George C, Mayer LP, Dyer CA, Aiken LS, Demers LM, Carson C, Bimonte-Nelson HA, 2017. Cognitive changes across the menopause transition: a longitudinal evaluation of the impact of age and ovarian status on spatial memory. Horm. Behav 87, 96–114. 10.1016/j.yhbeh.2016.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Palmer JM, Hadder B, Melikian R, Fox C, Strouse IM, DeNardo D, George C, Daunis E, Nimer A, Mayer LP, Dyer CA, Bimonte-Nelson HA, 2019. Hysterectomy uniquely impacts spatial memory in a rat model: a role for the non-pregnant uterus in cognitive processes. Endocrinology 160, 1–19. 10.1210/en.2018-00709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koebele SV, Nishimura KJ, Bimonte-Nelson HA, Kemmou S, Ortiz JB, Judd JM, Conrad CD, 2020. A long-term cyclic plus tonic regimen of 17β-estradiol improves the ability to handle a high spatial working memory load in ovariectomized middle-aged female rats. Horm. Behav 118, 104656. 10.1016/j.yhbeh.2019.104656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korol DL, Pisani SL, 2015. Estrogens and cognition: friends or foes?. An evaluation of the opposing effects of estrogens on learning and memory. Horm. Behav 74, 105–115. 10.1016/j.yhbeh.2015.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhl H, 2005. Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 8, 3–63. 10.1080/13697130500148875. [DOI] [PubMed] [Google Scholar]
- La Marca A, Broekmans FJ, Volpe A, Fauser BC, MacKlon NS, 2009. Anti-Müllerian hormone (AMH): what do we still need to know? Hum. Reprod 24, 2264–2275. 10.1093/humrep/dep210. [DOI] [PubMed] [Google Scholar]
- Lacreuse A, 2006. Effects of ovarian hormones on cognitive function in nonhuman primates. Neuroscience 138, 859–867. 10.1016/j.neuroscience.2005.09.006. [DOI] [PubMed] [Google Scholar]
- Lohff JC, Christian PJ, Marion SL, Arrandale A, Hoyer PB, 2005. Characterization of cyclicity and hormonal profile with impending ovarian failure in a novel chemical-induced mouse model of perimenopause. Comp. Med 55, 523–527. [PubMed] [Google Scholar]
- Lohff JC, Christian PJ, Marion SL, Hoyer PB, 2006. Effect of duration of dosing on onset of ovarian failure in a chemical-induced mouse model of perimenopause. Menopause 13, 482–488. 10.1097/01.gme.0000191883.59799.2e. [DOI] [PubMed] [Google Scholar]
- Long T, Yao JK, Li J, Kirshner ZZ, Nelson D, Dougherty GG, Gibbs RB, 2018. Comparison of transitional vs surgical menopause on monoamine and amino acid levels in the rat brain. Mol. Cell. Endocrinol 476, 139–147. 10.1016/j.mce.2018.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Long T, Yao JK, Li J, Kirshner ZZ, Nelson D, Dougherty GG, Gibbs RB, 2019. Estradiol and selective estrogen receptor agonists differentially affect brain monoamines and amino acids levels in transitional and surgical menopausal rat models. Mol. Cell. Endocrinol 496. 10.1016/j.mce.2019.110533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luine VN, 2014. Estradiol and cognitive function: past, present and future. Horm. Behav 66, 602–618. 10.1016/j.yhbeh.2014.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luine VN, Richards ST, Wu VY, Beck KD, 1998. Estradiol enhances learning and memory in a spatial memory task and effects levels of monoaminergic neurotransmitters. Hormones and Behavior 34 (2), 149–162. 10.1006/hbeh.1998.1473. [DOI] [PubMed] [Google Scholar]
- Maffucci JA, Noel ML, Gillette R, Wu D, Gore AC, 2009. Age- and hormone-regulation of N-methyl-D-aspartate receptor subunit NR2b in the anteroventral periventricular nucleus of the female rat: implications for reproductive senescence. J. Neuroendocrinol 21, 506–517. 10.1111/j.1365-2826.2009.01860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maki PM, Dennerstein L, Clark M, Guthrie J, Lamontagne P, Fornelli D, Little D, Henderson VW, Resnick SM, 2011. Perimenopausal use of hormone therapy is associated with enhanced memory and hippocampal function later in life. Brain Res. 1379, 232–243. 10.1016/j.brainres.2010.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowska AL, 1999. Sex dimorphisms in the rate of age-related decline in spatial memory: relevance to alterations in the estrous cycle. J. Neurosci 19, 8122–8133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markowska AL, Savonenko AV, 2002. Effectiveness of estrogen replacement in restoration of cognitive function after long-term estrogen withdrawal in aging rats. J. Neurosci 22, 10985–10995 doi:22/24/10985 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayer LP, Pearsall NA, Christian PJ, Devine PJ, Payne CM, McCuskey MK, Marion SL, Sipes IG, Hoyer PB, 2002. Long-term effects of ovarian follicular depletion in rats by 4-vinylcyclohexene diepoxide. Reprod. Toxicol 16, 775–781. 10.1016/S0890-6238(02)00048-5. [DOI] [PubMed] [Google Scholar]
- Mayer LP, Devine PJ, Dyer CA, Hoyer PB, 2004. The follicle-deplete mouse ovary produces androgen. Biol. Reprod 71, 130–138. 10.1095/biolreprod.103.016113. [DOI] [PubMed] [Google Scholar]
- McDonald JH, 2014. Handbook of Biological Statistics, 3rd ed. Sparky House Publishing, Baltimore, MD. [Google Scholar]
- Mennenga SE, Bimonte-Nelson HA, 2015. The importance of incorporating both sexes and embracing hormonal diversity when conducting rodent behavioral assays. In: Bimonte-Nelson HA(Ed.), The Maze Book: Theories, Practice, and Protocols for Testing Rodent Cognition. Springer US, New York, pp. 299–321. 10.1007/978-1-4939-2159-1_11. [DOI] [Google Scholar]
- Mennenga SE, Gerson JE, Dunckley T, Bimonte-Nelson HA, 2015a. Harmine treatment enhances short-term memory in old rats: dissociation of cognition and the ability to perform the procedural requirements of maze testing. Physiol. Behav 138, 260–265. 10.1016/j.physbeh.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mennenga SE, Gerson JE, Koebele SV, Kingston ML, Tsang CWS, Engler-Chiurazzi EB, Baxter LC, Bimonte-Nelson HA, 2015b. Understanding the cognitive impact of the contraceptive estrogen ethinyl estradiol: tonic and cyclic administration impairs memory, and performance correlates with basal forebrain cholinergic system integrity. Psychoneuroendocrinology 54, 1–13. 10.1016/j.psyneuen.2015.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mennenga SE, Koebele SV, Mousa AA, Alderete TJ, Tsang CWS, Acosta JI, Camp BW, Demers LM, Bimonte-Nelson HA, 2015c. Pharmacological blockade of the aromatase enzyme, but not the androgen receptor, reverses androstenedione-induced cognitive impairments in young surgically menopausal rats. Steroids 99, 16–25. 10.1016/j.steroids.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mishra GD, Kuh D, 2012. Health symptoms during midlife in relation to menopausal transition: British prospective cohort study. BMJ 344, e402. 10.1136/bmj.e402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell ES, Woods NF, 2001. Midlife women’s attributions about perceived memory changes: observations from the Seattle Midlife Women’s Health Study. J. Womens. Health Gend. Based. Med 10, 351–362. 10.1089/152460901750269670. [DOI] [PubMed] [Google Scholar]
- Morris R, 2015. The watermaze. In: Bimonte-Nelson HA (Ed.), The Maze Book: Theories, Practice, and Protocols for Testing Rodent Cognition. Springer Publishing Humana Press, New York, pp. 73–92. [Google Scholar]
- Morris R, Garrud P, Rawlins JN, O’Keefe J, 1982. Place navigation impaired in rats with hippocampal lesions. Nature 10.1038/297681a0. [DOI] [PubMed] [Google Scholar]
- NAMS, 2014. Menopause Practice: A Clinician’s Guide, 5th ed. North American Menopause Society, Mayfield Heights, OH. [Google Scholar]
- Nappi RE, Sinforiani E, Mauri M, Bono G, Polatti F, Nappi G, 1999. Memory functioning at menopause: impact of age in ovariectomized women. Gynecol. Obstet. Investig 47, 29–36. 10.1159/000010058. [DOI] [PubMed] [Google Scholar]
- Neves-E-Castro M, Birkhauser M, Samsioe G, Lambrinoudaki I, Palacios S, Borrego RS, Llaneza P, Ceausu I, Depypere H, Erel CT, Pérez-López FR, Schenck-Gustafsson K, Van Der Schouw YT, Simoncini T, Tremollieres F, Rees M, 2015. EMAS position statement: the ten point guide to the integral management of menopausal health. Maturitas 81, 88–92. 10.1016/j.maturitas.2015.02.003. [DOI] [PubMed] [Google Scholar]
- Olejnik S, Algina J, 2003. Generalized eta and omega squared statistics: measures of effect size for some common research designs. Psychol. Methods 8, 434–447. 10.1037/1082-989X.8.4.434. [DOI] [PubMed] [Google Scholar]
- Orr PT, Lewis MC, Frick KM, 2009. Dorsal hippocampal progesterone infusions enhance object recognition in young female mice. Pharmacol. Biochem. Behav 93, 177–182. 10.1016/j.pbb.2009.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinkerton JV, Sánchez Aguirre F, Blake J, Cosman F, Hodis H, Hoffstetter S, Kaunitz AM, Kingsberg SA, Maki PM, Manson JE, Marchbanks P, McClung MR, Nachtigall LE, Nelson LM, Todd Pace D, Reid RL, Sarrel PM, Shifren JL, Stuenkel CA, Utian WH, 2017. The 2017 hormone therapy position statement of The North American Menopause Society. Menopause 24, 1–26. 10.1097/GME.0000000000000921. [DOI] [PubMed] [Google Scholar]
- Prakapenka AV, Hiroi R, Quihuis AM, Carson C, Patel S, Berns-Leone C, Fox C, Sirianni RW, Bimonte-Nelson HA, 2018. Contrasting effects of individual versus combined estrogen and progestogen regimens as working memory load increases in middle-aged ovariectomized rats: one plus one does not equal two. Neurobiol. Aging 64, 1–14. 10.1016/j.neurobiolaging.2017.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapp PR, Morrison JH, Roberts JA, 2003. Cyclic estrogen replacement improves cognitive function in aged ovariectomized rhesus monkeys. J. Neurosci 23, 5708–5714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rocca WA, Bower JH, Maraganore DM, Ahlskog JE, Grossardt BR, de Andrade M, Melton LJI, 2007. Increased risk of cognitive impairment or dementia in women who underwent oophorectomy before menopause. Neurology 69, 1074–1083. [DOI] [PubMed] [Google Scholar]
- Rodgers SP, Bohacek J, Daniel JM, 2010. Transient estradiol exposure during middle age in ovariectomized rats exerts lasting effects on cognitive function and the hippocampus. Endocrinology 151, 1194–1203. 10.1210/en.2009-1245. [DOI] [PubMed] [Google Scholar]
- Savonenko AV, Markowska AL, 2003. The cognitive effects of ovariectomy and estrogen replacement are modulated by aging. Neuroscience 119, 821–830. 10.1016/S0306-4522(03)00213-6. [DOI] [PubMed] [Google Scholar]
- Sherwin BB, 2003. Estrogen and cognitive functioning in women. Endocr. Rev 24, 133–151. 10.1210/er.2001-0016. [DOI] [PubMed] [Google Scholar]
- Shumaker SA, Legault C, Rapp SR, Thal L, Wallace RB, Ockene JK, Hendrix SL, Jones III BN, Assaf AR, Jackson RD, Morley Kotchen J, Wassertheil-Smoller S, Wactawski-Wende J, 2003. Estrogen plus progestin and the incidence of dementia and mild cognitive impairment in postmenopausal women. JAMA 289, 2651–2662. 10.1001/jama.289.20.2651. [DOI] [PubMed] [Google Scholar]
- Shumaker SA, Legault C, Rapp SR, Thal L, Lane DS, Fillit H, Stefanick ML, Hendrix SL, Lewis CE, Coker LH, 2004. Conjugated equine estrogens and incidence of probable dementia and mild cognitive impairment in postmenopausal women. JAMA 291, 2947–2958. [DOI] [PubMed] [Google Scholar]
- Sprague BL, Trentham-Dietz A, Cronin KA, 2012. A sustained decline in post-menopausal hormone use: results from the national health and nutrition examination survey, 1999–2010. Obstet. Gynecol 120, 595–603. 10.1097/AOG.0b013e318265df42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Springer LN, Flaws J. a, Sipes IG, Hoyer PB, 1996a. Follicular mechanisms associated with 4-vinylcyclohexene diepoxide-induced ovotoxicity in rats. Reprod. Toxicol 10, 137–143. 10.1016/0890-6238(95)02056-X. [DOI] [PubMed] [Google Scholar]
- Springer LN, McAsey ME, Flaws J. a, Tilly JL, Sipes IG, Hoyer PB, 1996b. Involvement of apoptosis in 4-vinylcyclohexene diepoxide-induced ovotoxicity in rats. Toxicol. Appl. Pharmacol 139, 394–401. 10.1006/taap.1996.0180. [DOI] [PubMed] [Google Scholar]
- Stuenkel CA, Davis SR, Gompel A, Lumsden MA, Murad MH, Pinkerton JAV, Santen RJ, 2015. Treatment of symptoms of the menopause: an endocrine society clinical practice guideline. J. Clin. Endocrinol. Metab 100, 3975–4011. 10.1210/jc.2015-2236. [DOI] [PubMed] [Google Scholar]
- Talboom JS, Williams BJ, Baxley ER, West SG, Bimonte-Nelson HA, 2008. Higher levels of estradiol replacement correlate with better spatial memory in surgically menopausal young and middle-aged rats. Neurobiol. Learn. Mem 90, 155–163. 10.1016/j.nlm.2008.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson KE, Sipes IG, Greenstein BD, Hoyer PB, 2002. 17b-estradiol affords protection against 4-vinylcyclohexene diepoxide-induced ovarian follicle loss in Fischer-344 rats. Endocrinology 143, 1058–1065. 10.1210/en.143.3.1058. [DOI] [PubMed] [Google Scholar]
- Van Kempen TA, Milner TA, Waters EM, 2011. Accelerated ovarian failure: a novel, chemically induced animal model of menopause. Brain Res. 1379, 176–187. 10.1016/j.brainres.2010.12.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughan L, Espeland MA, Snively B, Shumaker SA, Rapp SR, Shupe J, Robinson JG, Sarto GE, Resnick SM, 2013. The rationale, design, and baseline characteristics of the Women’s Health Initiative Memory Study of Younger Women (WHIMS-Y). Brain Res. 1514, 3–11. 10.1016/j.brinres.2013.03.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace WHB, Kelsey TW, 2010. Human ovarian reserve from conception to the menopause. PLoS One 5 (e8772), 1–9. 10.1371/journal.pone.0008772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace M, Luine V, Arellanos A, Frankfurt M, 2006. Ovariectomized rats show decreased recognition memory and spine density in the hippocampus and prefrontal cortex. Brain Res. 1126, 176–182. 10.1016/j.brainres.2006.07.064. [DOI] [PubMed] [Google Scholar]
- Wang VC, Neese SL, Korol DL, Schantz SL, 2009. Chronic estradiol replacement impairs performance on an operant delayed spatial alternation task in young, middle-aged, and old rats. Horm. Behav 56, 382–390. 10.1016/j.yhbeh.2009.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber MT, Mapstone M, 2009. Memory complaints and memory performance in the menopausal transition. Menopause 16, 694–700. 10.1097/gme.0b013e318196a0c9. [DOI] [PubMed] [Google Scholar]
- Weber MT, Rubin LH, Maki PM, 2013. Cognition in perimenopause: the effect of transition stage. Menopause 10.1097/gme.0b013e31827655e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerlind KC, Gibson KJ, Malone P, Evans GL, Turner RT, 1998. Differential effects of estrogen metabolites on bone and reproductive tissues of ovariectomized rats. J. Bone Miner. Res 13, 1023–1031. 10.1359/jbmr.1998.13.6.1023. [DOI] [PubMed] [Google Scholar]
- Wharton W, Gleason CE, Miller VM, Asthana S, 2013. Rationale and design of the kronos early estrogen prevention study (KEEPS) and the KEEPS cognitive and affective sub study (KEEPS cog). Brain Res. 1514, 12–17. 10.1016/j.brainres.2013.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witty CF, Foster TC, Semple-Rowland SL, Daniel JM, 2012. Increasing hippocampal estrogen receptor alpha levels via viral vectors increases MAP kinase activation and enhances memory in aging rats in the absence of ovarian estrogens. PLoS One 7, 1–10. 10.1371/journal.pone.0051385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright LE, Frye JB, Lukefahr AL, Marion SL, Hoyer PB, Besselsen DG, Funk JL, 2011. 4-Vinylcyclohexene diepoxide (VCD) inhibits mammary epithelial differentiation and induces fibroadenoma formation in female Sprague Dawley rats. Reprod. Toxicol 32, 26–32. 10.1016/j.reprotox.2011.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Yan D, Yang Y, Ma A, Li L, Wang Z, Pan Q, Sun Z, 2016. The comparison of animal models for premature ovarian failure established by several different source of inducers. Regul. Toxicol. Pharmacol 81, 223–232. 10.1016/j.yrtph.2016.09.002. [DOI] [PubMed] [Google Scholar]


