Abstract
177Lu-DOTATATE peptide receptor radionuclide therapy (PRRT) alone has lesser potential in the clinical setting of neuroendocrine tumor (NET) with large bulky disease and nonhomogeneous somatostatin receptors (SSTR) distribution, owing to lower energy (Eβmax 0.497 MeV) and a shorter particle penetration range (maximum 2–4 mm) of 177Lu. In large bulky NETs, 90Yttrium (90Y) has the theoretical advantages because of a longer beta particle penetration range (a maximum soft tissue penetration of 11 mm). Therefore, a combination of 177Lu and 90Y is a theoretically sound concept that can result in better response in metastatic NET with large-bulky lesion and non-homogeneous SSTR distribution. The aim of the study was to determine the feasibility of combining 90Y-DOTATATE with 177Lu-DOTATATE PRRT as sequential duo-PRRT in metastatic NET with (≥5 cm) including the post 90Y-DOTATATE-PRRT imaging and also to determine early toxicity of the duo-PRRT approach. A total of 9 patients received combination of 177Lu-DOTATATE with 90Y-DOTATATE (indigenously prepared and approved) through sequential duo-PRRT approach. These 9 NET patients were included and analyzed in this study. All 9 patients had undergone post-PRRT 90Y-DOTATATE imaging, including a whole-body planar bremsstrahlung imaging followed by regional single-photon emission computed tomography (SPECT)-computed tomography (CT) imaging and also a regional positron emission tomography–computed tomography imaging. Grading of 90Y-DOTATATE and 177Lu-DOTATATE uptake was done on post-PRRT imaging by both modalities. The size of the lesions ranged from 5.5 cm to 16 cm with average size of 10 cm before sequential duo-PRRT was decided. Sequential duo-PRRT was administered because of stable, unresponsive disease following 177Lu-DOTATATE in 5 patients (55.6%), progressive disease after 177Lu-DOTATATE in 2 patients (22.2%), and with neoadjuvant intent in 2 patients (22.2%). The total cumulative dose of 177Lu-DOTATATE before duo-pRRT ranged from 11.84 GBq to 37 GBq per patient and average administered dose of 27.21 GBq per patient in this study. Out of 9 patients, 8 patients received single cycle of 90Y-DOTATATE (ranging from 2.66 GBq to 3.4 GBq per patient with average administered dose of 3.12 GBq per patient). One patient received two cycles of 90Y-DOTATATE (total dose of 6.2 GBq). Out of 9 patients, 8 patients showed excellent tracer concentration in lesions on post-PRRT 90Y-DOTATATE imaging and the remaining 1 patient showed fairly adequate 90Y-DOTATATE tracer uptake in lesion on visual analysis. There was matched 90Y-DOTATATE uptake with 68Ga-DOTATATE and also with 177LuDOTATATE in all 9 patients. The sequential duo-PRRT was well tolerated by all patients. Two patients (22.2%) developed mild nausea, one patient (11.1%) developed transient mild-grade hemoglobin toxicity, and one patient (11.1%) developed mild-grade gastrointestinal symptoms (loose motion and abdominal pain). No nephrotoxicity, hepatotoxicity, and other hematological toxicity was observed. The combination of the indigenous 90Y-DOTATATE with 177Lu-DOTATATE PRRT in NET as sequential duo-PRRT was well tolerated, feasible and safe in stable, unresponsive/progressive disease following single isotope 177Lu-DOTATATE therapy and also in neoadjuvant PRRT setting with large bulky lesion (≥≥5cm). Post-PRRT 90Y-DOTATATE imaging showed excellent 90Y-DOTATATE uptake in nearly all NET patients. Mild-grade early adverse effects were easily manageable and controllable in this sequential duo-PRRT approach.
Keywords: 177Lu-DOTATATE, 68Ga-DOTATATE PET-CT, 90Y-DOTATATE, duo-PRRT, peptide receptor radionuclide therapy (PRRT), Tandem PRRT
INTRODUCTION
Neuroendocrine tumors (NET) are a heterogeneous group of neoplasms, arising from the endocrine cells which are present throughout body most commonly in the gastrointestinal and bronchopulmonary tracts. The unique feature of NET is overexpression of somatostatin receptors (SSTR) in primary as well as metastatic lesions. There are five subtypes of SSTR. Out of five subtypes, type 2 is commonly expressed in most of NET cases and this is a key target molecule for peptide receptor radionuclide therapy (PRRT).[1]
The treatment management of localized NET is surgery, which is possible in limited number of cases. More than 40% of NET patients present with metastatic disease at time of diagnosis and these patients require systemic therapies. The therapeutic landscape of advanced/metastatic NET has undergone a remarkable expansion in this decade specially within recent years, which includes long acting somatostatin analogs (SSAs), chemotherapy, molecular targeted treatments (everolimus and sunitinib), alpha-interferon, and PRRT.[2,3,4]
The recently published randomized phase III NETTER-1 trial[5] provided high-level evidence of efficacy and safety of 177Lu-DOTATATE PRRT in mid-gut NET. However, the result of NETTER-1 trial generated a multitude of new questions regarding timing, sequencing, and matching of patients with the most appropriate treatment protocols available for the management of metastatic/advanced NET.
The role of 177Lu-DOTATATE PRRT in NET may be doubtful in large bulky lesions (≥5 cm) with nonhomogeneous SSTR distribution and this is because of lower energy (Eβmax 0.497 MeV) and a shorter particle penetration range (maximum 2–4 mm) of 177Lu-DOTATATE. In such conditions, 90Yttrium (90Y) has theoretical advantages because of a longer beta particle penetration range (a maximum soft tissue penetration of 11 mm).[6,7]
A combination of 177Lu and 90Y therapy is a theoretically sound concept that can translate into better response in NETs with both small and large-bulky lesions. The aim of the present study was to determine the feasibility of combining 90Y-DOTATATE with 177Lu-DOTATATE PRRT as sequential duo-PRRT in NET with large bulky lesion (≥5 cm) and also to determine early toxicity of this duo-PRRT approach.
MATERIALS AND METHODS
In this study, we retrospectively reviewed the medical records of 9 patients who received combination of 177Lu-DOTATATE with 90Y-DOTATATE sequential duo-PRRT, within the period from September 2019 to February 2020 and these 9 NET patients were included and analyzed in this study (this corresponds to the time period during which the approved indigenous 90Y was available to our Institute). The product was approved for the routine clinical use by the Radiopharmaceuticals Committee of the Department of Atomic Energy. The treatments were conducted after obtaining necessary approval from the Institutional Ethics Committee and were also approved by the Institutional Scientific Advisory Committee (IEC Ref: Project No P17/feb/2019).
The eligibility criteria for deciding for sequential duo-PRRT were as follows: patients with pathologically confirmed NET; unresectable and advanced large bulky (≥5 cm) disease; stable, unresponsive/progressive disease on 177Lu-DOTATATE (≥2 cycles); and SSTR positive lesions on 68Ga-DOTATATE positron emission tomography–computed tomography (PET-CT) (Krenning score ≥2, compared on maximum intensity projection (MIP), coronal and transaxial images).
The exclusion criteria for sequential duo-PRRT included low or absent SSTR expression on 68Ga-DOTATATE PET-CT, small sized disease (<5 cm), glomerular filtration rate (GFR) of <60 mL/min, hypoalbuminemia (<20 g/L), platelet count <90 × 10 9/L or pancytopenia, pregnancy, breast-feeding, severe concomitant illness including severe psychiatric disorders, Eastern Cooperative Oncology Group (ECOG) performance score 4, Karnofsky Performance Status score of <60, and expected life survival <3 months.
Treatment planning
All 9 NET patients had undergone the presequential duo-PRRT work up protocol, which included 68Ga-DOTATATE PET-CT and 18F-FDG PET-CT imaging, documentation of clinical symptoms, and measurement of serum chromogranin A (CgA) level before considering sequential duo-PRRT.
68Ga-DOTATATE positron emission tomography–computed tomography and 18F-FDG positron emission tomography–computed tomography imaging
68Ga-DOTATATE PET-CT and 18F-FDG PET-CT scans were obtained in all 9 NET patients before sequential duo-PRRT.
68Ga-DOTATATE PET-CT scan was performed approximately 60 min after intravenous injection of 74–111 MBq (2–3 mCi) of 68Ga-DOTATATE dose. The scans were performed using a time of flight PET-CT scanner (Philips Gemini TF TOF 16 PET/CT scanner) with LYSO-based PET crystal. After obtaining a scout image whole-body CT scanning was performed first in craniocaudal direction (voltage 120 kVp, slice thickness 5 mm, pitch-0.83, FOV 600 mm, rotation time −0.5 s, 250 mA, image matrix-512 × 512) without any breath hold instructions. Contrast or noncontrast CT was used for diagnostic and attenuation correction of the PET data. PET scanning was performed immediately after the CT acquisition in the three-dimensional (3D) emission mode with 3 min per bed position. Images were reconstructed iteratively using RAMLA algorithm (2 iterations, 21 subsets). The acquired studies were viewed (transaxial, coronal, and sagittal views) in multimodality workstation of Philips Gemini TF processing system. Similar imaging protocol was applied for 18F-FDG PET-CT scan, which was done 1 day after 68Ga-DOTATATE PET-CT scan. For 18F-FDG PET-CT scan, all patients were fasted for at least 6 h prior to the intravenous injection of 5MBq/kg body weight of 18F-FDG and whole-body full-ring dedicated 3D PET-CT scanning was done (50 mA, 120 kVp, noncontrast/contrast CT scan for attenuation correction and anatomical co-localization).
If PET-CT scans revealed SSTR avid large bulky lesions (≥5 cm) on 68Ga-DOTATATE imaging as shown in Figure 1a, which were stable, unresponsive/progressive disease on 177Lu-DOTATATE (≥2 cycles) then sequential duo-PRRT was planned in these patients.
Figure 1.
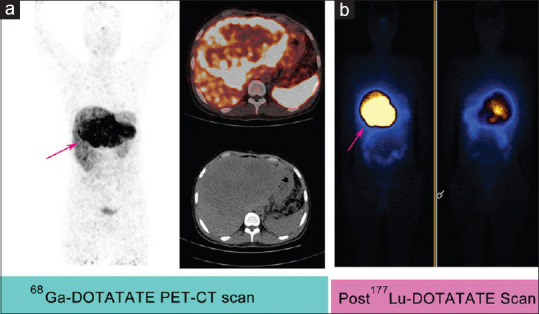
A 29 years old male patient, known case of duodenal neuroendocrine tumor (MIB Index = 8%) with bulky metastatic liver disease. Patient initially underwent surgical resection of duodenal lesion followed by 2 cycles of 177Lu-DOTATATE peptide receptor radionuclide therapy then 90Y-DOTATATE peptide receptor radionuclide therapy. 68Ga-DOTATATE PET-CT scan (a) showed somatostatin receptors avid (Krenning score = 4) large sized (16 cm × 13 cm) liver lesion (arrow) and post 177Lu-DOTATATE therapy scan (b) showed good tracer uptake (grade = 3) in liver lesion
Investigations and Health Related Quality of Life Scales
The various investigations such as hematological parameters, renal function test, GFR, liver function test, and serum CgA levels were measured and documented in all NET patients before sequential duo-PRRT. The patients were examined for health related quality of life (HRQoL) scales which included ECOG Performance Status and Karnofsky score before and following sequential duo-PRRT.
177Lu-DOTATATE therapy
Long and short-acting somatostatin was stopped for 4 weeks and 24 h respectively before 177Lu-DOTATATE PRRT.
The Bhabha Atomic Research Centre (BARC), Mumbai, India, supplied a sterile solution of 177LuCl3 in 0.01M HCl with a specific activity of >999 MBq/ug. In house labeling was carried out with a radiochemical purity of >99% for 177LuDOTATATE product.
The standard mixed amino acid preparation containing lysine and arginine was infused 30 min before PRRT and maintained over 7 h. To prevent nausea and vomiting, 4 mg of ondansetron and 4 mg of dexamethasone was administered before amino acid infusion.
177Lu-DOTATATE with dose activity of 5.5–7.4 GBq per cycle in 100 ml of normal saline was administered intravenously over 30 min. For 177Lu-DOTATATE PRRT, all NET patients were admitted in radionuclide therapy ward and monitored for 24 h after PRRT for any acute adverse effects. The 177Lu-DOTATATE PRRT was divided into cycles and cycles were repeated at intervals of 10–12 weeks.
Post-peptide receptor radionuclide therapy 177Lu-DOTATATE whole-body planar imaging
Whole-body planar imaging and single photon emission CT (SPECT) imaging were conducted 24 h after 177Lu-DOTATATE using a large field of view gamma camera with a medium-energy collimator as shown in Figure 1b.
90Y-DOTATATE therapy
Long- and short-acting somatostatins were stopped for 4 weeks and 24 h respectively before 90Y-DOTATATE PRRT therapy.
90Y-Acetate was indigenously sourced from high-level liquid waste, using separation method based on a two-stage 90Sr/ 90Y generator system based on supported liquid membrane technology, developed at Nuclear Recycle Group, BARC, and Mumbai, India. The quality control parameters of 90Y-acetate were validated and compared to the pharmacopeia standard. This 90Y-acetate was supplied as a sterile solution to Radiation Medicine Centre, Mumbai, India, and in-house labeling was carried with a radiochemical purity of >99% for 90Y-DOTATATE final product. The product was approved by the regulatory body of DAE (Department of Atomic Energy) India, the Radiopharmaceutical Committee, for routine clinical use.
The standard mixed amino acid preparation containing lysine and arginine was infused 30 min before PRRT and maintained over 7–8 h. To prevent nausea and vomiting, 4 mg of ondansetron and 4 mg of dexamethasone were administered before amino acid infusion.
90Y-DOTATATE with dose activity of 2.6–3.4 GBq per cycle in 100 ml of normal saline was administered intravenously over 30 min. For 90Y-DOTATATE PRRT, all NET patients were admitted in radionuclide therapy ward and monitored for 24 h after PRRT therapy for any acute adverse effects.
Post-peptide receptor radionuclide therapy 90Y-DOTATATE imaging
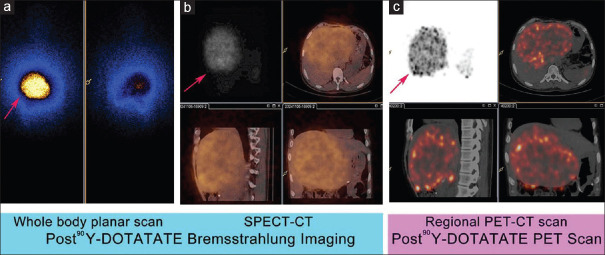
Whole-body planar bremsstrahlung imaging [Figure 2a] and SPECT-CT imaging were acquired 24 h after 90Y-DOTATATE PRRT using a large field of view gamma camera with a high-energy collimator and various windows setting with a range of 76–250 keV. A whole-body planar scan was performed at a speed of ~10 cm/min. The SPECT/SPECT-CT of regional part of body [Figure 2b] was performed after whole-body scan using a dual-head camera. The high-energy, parallel hole, general-purpose collimators were used. In the case of SPECT or SPECT/CT, acquisition parameters were: 128 × 128 matrix, zoom 1.00, 1 min/frame for 12 frames.
Figure 2.
After 2 cycles of 177Lu-DOTATATE peptide receptor radionuclide therapy, the patient received 90Y-DOTATATE therapy (single cycle, dose = 3.4 GBq). Post 90Y-DOTATATE Bremsstrahlung imaging with whole-body planar and single photon emission computed tomography-computed tomography scans (a and b) and post 90Y-DOTATATE regional positron emission tomography–computed tomography scan (c) demonstrated good tracer uptake of 90Y-DOTATATE (grade = 3) in liver lesions (arrows). No major adverse effect was seen after 90Y-DOTATATE peptide receptor radionuclide therapy in this patient
Regional PET-CT scans were conducted 24–36 h after 90Y-DOTATATE PRRT using a time of flight PET-CT scanner (Philips Gemini TF TOF 16 PET/CT scanner) with LYSO-based PET crystal. After obtaining a scout image regional CT scanning was performed first in cranio-caudal direction (voltage 120 kVp, slice thickness 5 mm, pitch-0.83, FOV 600 mm, rotation time-0.5 sec, 50 mA, image matrix-512 × 512) without any breath hold instructions. Non contrast low dose CT was used for attenuation correction of the PET data. PET scanning was performed immediately after the CT acquisition in the 3D emission mode with 20 mins per bed position for 2 beds. Images were reconstructed iteratively using RAMLA algorithm (2 iterations, 21 subsets). The acquired studies were viewed (transaxial, coronal and sagittal views) in multimodality-work-station of Philips Gemini TF processing system [Figure 2c].
Post-PRRT 177Lu-DOTATATE uptake in the lesions (on gamma camera imaging) and the post-PRRT 90Y-DOTATATE uptake in lesions on gamma camera imaging and on PET/CT scanner were divided into 1, 2 and 3 grade based on visual comparison of tracer uptake in the lesions with physiological liver/kidney uptake, where tracer uptake (177Lu-DOTATATE or 90Y-DOTATATE uptake) in lesion was less than liver/kidney uptake consider as grade 1, tracer uptake in lesion equal to liver/kidney uptake as grade 2, and tracer uptake in lesion more than liver/kidney uptake as grade 3.
Follow-up
Clinical examinations with vital signs were undertaken both before and after sequential duo-PRRT. The patients were evaluated for symptomatic, ECOG and Karnofsky scores, and biochemical parameters following sequential duo-PRRT. Blood biochemistry and hematological variables, including renal function tests, complete blood counts, serum hemoglobin levels, and liver function test, were measured at biweekly interval after sequential duo-PRRT. Any observed toxicity was recorded continuously. Adverse events were graded according to the Common Terminology Criteria for Adverse Events version 5.0 of the National Cancer Institute with special attention to nausea, vomiting, leukopenia, thrombocytopenia, anemia, and liver and renal adverse events.
RESULTS
A total of 9 patients (3 women, 6 men; median age 55 years, range 46–62 years) received sequential duo-PRRT were included and analyzed retrospectively in this study. The detail of patient characteristics in the study population is shown in Tables 1 and 2.
Table 1.
Patient characteristics
| Patients characteristics | Number of patients |
|---|---|
| Total number of patients received sequential duo-PRRT | 9 |
| Sex (male:female) | 6:3 |
| Symptomatic patients before sequential duo-PRRT | 3 |
| Metastatic disease before sequential duo-PRRT | 8 |
| Stable, unresponsive disease following 177Lu-DOTATATE | 5 |
| Progressive disease after 177Lu-DOTATATE | 2 |
| Sequential duo-PRRT used as neoadjuvant intent | 2 |
| FDG positive disease before sequential duo-PRRT | 6 |
| Site of primary disease | |
| Pancreatic NET | 4 |
| Mediastinal NET | 1 |
| Rectal NET | 1 |
| Duodenal NET | 2 |
| Unknown primary site | 1 |
| WHO Grade | |
| Grade 1 | 3 |
| Grade 2 | 5 |
| Grade 3 | 1 |
| Prior therapies | |
| Surgical resection of lesion | 3 |
| Somatostatin analog therapy | 5 |
| External beam radiotherapy | 1 |
PRRT: Peptide receptor radionuclide therapy; NET: Neuroendocrine tumor; FDG: 2-deoxy-2-[fluorine-18]fluoro-D-glucose
Table 2.
Patient details
| Case number | Primary site, MIB-1 index | Prior therapies | Size of lesions (largest diameter) | Indication for sequential duo-PRRT | Cumulative dose and cycles of 177Lu-DOTATATE PRRT | Dose of 90Y-DOTATATE PRRT | Adverse effects | Follow-up period after sequential duo-PRRT |
|---|---|---|---|---|---|---|---|---|
| I | Mediastinal NET, 30% | Surgical resection of primary lesion, Somatostatin analog therapy and external beam radiotherapy | 12 cm | Stable, unresponsive disease following 177Lu-DOTATATE | 33.3 GBq, 5 cycles | 2.7 GBq - First cycle | No | 105 days |
| II | Pancreatic NET, 2% | Somatostatin analog therapy | 8 cm | Progressive disease after 177Lu-DOTATATE | 25.23 GBq, 4 cycles | 2.8GBq-First cycle, 3.3 GBqsecond cycle | No | 135 days |
| III | Duodenal NET, 10% | No prior therapies | 10 cm | Stable, unresponsive disease following 177Lu-DOTATATE | 31.59 GBq, 5 cycles | 2.66 GBq - First cycle | No | 135 days |
| IV | Pancreatic NET, 10 % | Surgical resection of primary lesion, somatostatin analog therapy | 5.5 cm | Stable, unresponsive disease following 177Lu-DOTATATE | 37 GBq, 6 cycles | 3.14 GBq - First cycle | Transient mild-grade hemoglobin toxicity | 105 days |
| V | Rectal NET, 12% | Somatostatin analog therapy | 13 cm | Progressive disease after 177Lu-DOTATATE | 22.2 GBq, 4 cycles | 3.4 GBq- First cycle | Mild-grade of nausea | 75 days |
| VI | Pancreatic NET, 1% | No prior therapies | 5.5cm | Neoadjuvant intent | 14.8 GBq, 2 cycles | 3.25 GBq - First cycle | Mild-grade of nausea and gastrointestinal symptoms | 75 days |
| VII | Pancreatic NET, 1% | Somatostatin analog therapy | 7 cm | Stable, unresponsive disease following 177Lu-DOTATATE | 32 GBq, 5 cycles | 3.4 GBq - First cycle | No | 60 days |
| VIII | Unknown primary site, 8% | No | 13 cm | Stable, unresponsive disease following 177Lu-DOTATATE | 37 GBq, 6 cycles | 3.4 GBq - First cycle | No | 30 days |
| IX | Duodenal NET, 8% | Surgical resection of primary lesion | 16 cm | Neoadjuvant intent | 11.84 GBq, 2 cycles | 3.4GBq- First cycle | No | 15 days |
PRRT: Peptide receptor radionuclide therapy; NET: Neuroendocrine tumor; MIB: antibody directed at the protein Ki-67
The Ki-67/MiB-1 index ranged from 1% to 30% with an average of 9% in this study population. The WHO grade 1 (Ki-67 index <3%) of tumor differentiation was noted in 3 patients, grade 2 (Ki-67 index 3%–20%) in 5 patients, and grade 3 (Ki-67 index >20%) in 1 patient. Out of the 9 patients, 4 patients (44.4%) had pancreatic NET and the remaining 5 patients (55.6%) included mediastinal NET in 1 patient, rectal NET in 1 patient, duodenal NET in 2 patients, and metastases with unknown primary site in 1 patient. Three patients (33.3%) were symptomatic before sequential duo-PRRT with the most common complaints being abdominal pain, weakness, and weight loss. Out of the 9 patients, 3 patients underwent surgical resection of the primary disease (resection of pancreatic lesion in 1 patient, resection of mediastinal lesion in 1 patient, and resection of duodenal lesion in 1 patient), 5 patients received somatostatin analog therapy, and 1 patient received external beam radiation therapy before sequential duo-PRRT. Out of 9 NET patients, 8 patients (88.8%) had metastatic disease before sequential duo-PRRT and most common site for metastatic disease was liver (n = 7 patients), followed by lymph nodes (n = 3 patients), and bone (n = 2 patients). One patient had localized pancreatic disease in this study. The size of lesions ranged from 5.5 cm to 16 cm with an average size of 10 cm before sequential duo-PRRT was noted in this study. Out of 9 NET patients, 6 patients (66.6%) had FDG avid disease. The total cumulative dose of 177Lu-DOTATATE before the adoption of duo-PRRT approach ranged from 11.84 GBq to 37 GBq with average administered dose of 27.21 GBq per patient and 177Lu-DOTATATE cycles ranging from 2 to 6 cycles and an average of 4 cycles per patient was given in this study.
Out of the 9 patients, sequential duo-PRRT was considered because of stable, unresponsive disease following 177Lu-DOTATATE in 5 patients (55.6%), progressive disease after 177Lu-DOTATATE in 2 patients (22.2%) and in an neoadjuvant intent in 2 patients (22.2%). At the time of analysis, single cycle of 90Y-DOTATATE was given in 8 patients, administered dose ranging from 2.66 GBq to 3.4 GBq per patient and average administered dose of 3.12 GBq per patient. One patient was given two cycles of 90Y-DOTATATE with total dose of 6.2 GBq.
All 9 patients were underwent post-PRRT 90Y-DOTATATE imaging, a whole-body planar bremsstrahlung imaging followed by regional SPECT-CT and also a regional PET-CT imaging on PET/CT scanner. Out of 9 patients, 8 patients showed excellent (grade 3) 90Y-DOTATATE tracer concentration in lesions on post-PRRT 90Y-DOTATATE imaging and 1 patient (Grade 1) showed fairly adequate 90Y-DOTATATE tracer uptake on visual analysis. In all 9 patients, the lesions visualized on 68Ga-DOTATATE PET-CT, post-PRRT 177Lu-DOTATATE and post-PRRT 90Y-DOTATATE imaging (bremsstrahlung—planar, regional SPECT-CT and regional PET-CT) demonstrating congruent good tracer activity of 90Y-DOTATATE as shown in the comparison in Table 3.
Table 3.
Tracers uptake in the lesions on visual analysis
| Case number | 68Ga-DOTATATE uptake in lesions on PET/CT scanner with Krenning score | Post-PRRT 177Lu-DOTATATE uptake in lesions on gamma camera imaging with grading* | Post-PRRT 90Y-DOTATATE uptake in lesions on gamma camera imaging with grading* | Post-PRRT 90Y-DOTATATE uptake in lesions on PET/ CT scanner with grading* |
|---|---|---|---|---|
| I | 4 | 3 | 3 | 3 |
| II | 4 | 3 | 3 | 3 |
| III | 4 | 3 | 3 | 3 |
| IV | 4 | 3 | 3 | 3 |
| V | 4 | 3 | 3 | 3 |
| VI | 2 | 2 | 2 | 2 |
| VII | 4 | 3 | 3 | 3 |
| VIII | 4 | 3 | 3 | 3 |
| IX | 4 | 3 | 3 | 3 |
*Grade 1: Tracer uptake in lesion less than physiological liver/kidney uptake, Grade 2: Tracer uptake in lesion equal to physiological liver/kidney uptake, Grade 3: Tracer uptake in lesion more than physiological liver/kidney uptake. PRRT: Peptide receptor radionuclide therapy; PET/CT: Positron emission tomography/computed tomography
At the time of analysis, the follow-up period after 90Y-DOTATATE therapy ranged from 15 to 135 days (average of 82 days). During this follow-up period, two patients (22.2%) developed mild nausea (controlled well by giving another dose of 4 mg of ondansetron), one patient (11.1%) developed transient mild-grade hemoglobin toxicity (recovered within 10 days) and one patient (11.1%) developed mild-grade gastrointestinal symptoms (loose motion and abdominal pain recovering within 7 days). No nephro-toxicity, hepato-toxicity and other hematological toxicity was observed after the 90Y-DOTATATE therapy (as part of sequential duo-PRRT), as mentioned in Table 4.
Table 4.
Adverse events documentation
| Adverse events↓/Grade*→ | Grade 1 (%) | Grade 2 | Grade 3 | Grade 4 |
|---|---|---|---|---|
| During/related to PRRT in number of NET patients | ||||
| Nausea | 2 (22.2%) | 0 | 0 | 0 |
| Vomiting | 0 | 0 | 0 | 0 |
| Gastrointestinal symptoms | 1 (11.1%) | 0 | 0 | 0 |
| Anemia | 1 (11.1%) | 0 | 0 | 0 |
| Thrombocytopenia | 0 | 0 | 0 | 0 |
| Neutropenia | 0 | 0 | 0 | 0 |
| Nephrotoxicty | 0 | 0 | 0 | 0 |
| Hepatotoxicity | 0 | 0 | 0 | 0 |
*Grade: Signifies grade of toxicity. PRRT: Peptide receptor radionuclide therapy; NET: Neuroendocrine tumor
DISCUSSION
For the metastatic/advanced NET, PRRT with radiolabelled somatostatin analogs has become an established method of treatment. Historically, 111Indium labeled with (diethylenetriaminepentaacetic acid [DTPA] 0) octreotide was used in some clinical trials, which had a short tissue penetration of the emitted Auger electrons (ranging between nanometers and micrometers) with best result observed in small sized and highly SSTR avid NETs.[8] In one of study, amongst the 50 NET patients treated with 111In-pentetreotide, 3 patients developed leukemia or myelodysplastic syndrome (MDS) who received >100 GBq therapeutic dose of 111In-pentetreotide.[9] Presently, 111In-pentetreotide is not commonly used in the management of NET, because of limited therapeutic result, higher therapeutic doses requirement, and also availability of better β-emitter radionuclides for PRRT in NET.
Also subsequently a modified somatostatin analog (Tyr3) octreotide with a higher affinity for the SSTR subtype-2 was developed and new chelator, 1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetraacetic acid (DOTA) instead of DTPA was used to enable a more stable binding of the intended β-emitter (90Y and 177Lutetium) radionuclide. The beta-emitter radionuclides such as 90Y (pure β-emitter) emits beta particles with a high maximum energy (Emax 2.27 MeV) and longer maximum particle range in tissues (11 mm) with physical half-life of 64 h. The use of 90Y labeled with DOTA-Tyr3-octreotide (90Y-DOTATOC) in NET had showed objective response rates of 6%–37% and occurrence of high renal toxicity in initial studies.[10,11] This higher renal toxicity of 90Y-DOTATOC therapy may be related to longer penetration particle range of 90Y that results in “cross-fire” effect to irradiate not only the targeted cell but also the surrounding cells and also higher affinity of DOTATOC to SSTR 3 and SSTR5 as compared to SSTR2. The 177Lu emits both beta particles (Emax 0.497 MeV and a shorter particle range in tissues [maximum 2–4 mm]) and gamma rays (113 keV [6.4%]; 208 keV [11%]). Because of the γ-emission, posttherapeutic gamma camera scintigraphic imaging is possible with 177Lu. This posttherapeutic imaging provided an accurate representation of the distribution of the radionuclide therapy in all the lesions, and used to monitor SSTR uptake following PRRT treatments. The 177Lu-DOTA-Tyr3-octreotate (177Lu-DOTATATE) has shorter tissue penetration range and 9-fold higher peptide receptor affinity for SSTR2 compared to DOTATOC[12] and this resulted in higher tolerability of 177Lu-DOTATATE therapy, especially with regard to the kidneys. A lower whole-body dose, especially lower bone marrow and renal toxicity following 177Lu-DOTATATE therapy was reported in various studies.
However, 177Lu-based PRRT is less effective in large sized tumors with heterogeneous distribution of SSTR over larger areas in tumor and this is because of a lower energy and smaller particle range of 177Lu. As such the energy deposition of 177Lu is 67% inside a 2-mm lesion, whereas 90Y deposited 87% of energy in large tumors (diameter up to 5 cm).[13,14] Therefore, in patients with both small sized and large bulky NET with heterogeneous distribution of SSTR, a combination of radionuclide (177Lu and 90Y) therapy might be the useful, which potentially leads to a better effectiveness than one isotope alone. This combination of radionuclide treatment was first described by De Jong at el in their animal studies which consisted of 50% 177Lu-DOTATATE and 50% 90Y-DOTATOC. They found extended duration of survival time by 3 times in rats after use of this combination and they concluded in their studies that complementary characteristics of both isotopes allow irradiation of both large and small metastases.[14,15]
The combination of radionuclide treatment in humans was first time reported by Kunikowska et al. in their clinical trial and this trial was conducted in total 50 NET patients with multiple metastatic lesions of varying sizes. Out of 50 patients, 25 patients were given 90Y-DOTATATE alone and 25 patients were given combination of 177Lu-DOTATATE and 90Y-DOTATATE with 3.7 GBq/m 2 body surface area activity dose in three to five cycles along with amino acid infusion for renal protection. They found a longer overall survival (OS) time after treatment with 90Y/ 177Lu-DOTATATE than in the group treated with 90Y-DOTATATE alone (OS not reached vs. 26.3 months, P < 0.01). No severe adverse events occurred in 90Y-DOTATATE alone and combined 90Y/ 177Lu-DOTATATE treated groups. They found WHO hematological toxicity of grades 1 and 2 with nearly the same frequency in both groups without clinical symptoms and deterioration in kidney function was observed in three patients in each group. They concluded that PRRT with tandem approach (90Y/ 177Lu-DOTATATE) provides longer OS than with a single radioisotope (90Y-DOTATATE) with comparable safety profile in both groups.[16]
A similar study was conducted by Villard et al.[17] on a larger cohort of 486 NET patients and these patients were divided into two groups, one group (n = 237 patients) received 90Y-tetraazacyclododecane-tetraacetic acid modified Tyr-octreotide (DOTATOC) alone and another group (n = 249 patients) received 90Y-DOTATOC plus 177Lu-DOTATOC PRRT. The patients received combined radionuclide therapy (OS = 5.51 years) had a significantly longer OS than patients who received 90Y-DOTATOC (OS 3.96 years) PRRT alone. The rates of adverse effects (severe hematologic and renal toxicity) were comparable in both groups. They concluded that combined radionuclide therapy associated with improved OS compared with single isotope 90Y alone PRRT in NET patients with comparable adverse effect profiles in both groups. Villard et al.[17] used DOTATOC instead of DOTATATE for PRRT in their study and found similar result as reported by Kunikowska et al.[16]
Seregni M et al. evaluated tandem 90Y-DOTATATE and 177Lu-DOTATATE PRRT in 26 metastatic NET patients in their study. These 26 patients were given four therapeutic cycles of alternating 177Lu-DOTATATE (5.55 GBq) and 90Y-DOTATATE (2.6 GBq). They calculated the absorbed doses in healthy organs using a dosimetric method after administration of 177Lu-DOTATATE. They found objective responses of 42.3% with a median progression-free survival longer than 24 months and the cumulative biologically effective doses were below the toxicity limit in the majority of patients in their study. They concluded that tandem 90Y-DOTATATE and 177Lu-DOTATATE PRRT is an effective therapeutic option in NET cases refractory to conventional therapy with absence of renal damage.[18]
Dumont et al. explored the effects of 90Y-DOTATOC (n = 30 patients) and 90Y-DOTATOC plus 177Lu-DOTATOC (n = 6 patients) on survival of metastatic gastrinoma patients. They found that longer median survival in patients who received 90Y-DOTATOC plus 177Lu-DOTATOC (median OS = 60.2 months) as compared to 90Y-DOTATOC (median OS = 27.0 months) alone received PRRT and concluded that combination of radionuclide therapy was promising tool for management of metastatic progressive gastrinoma.[19]
The size of lesions was not clearly mentioned for treatment of combined radionuclide therapy in most of the studies available in literature. Kong et al. evaluated sequential PRRT therapy (90Y-DOTATATE followed by 177Lu-DOTATATE) along radiosensitising chemotherapy in NET patients with >4 cm size lesion. They found that 42% of partial response and 21% of minor response on anatomical response evaluation after use of sequential peptide receptor chemoradionuclide therapy (PRCRT). Out of 26 patients, 8 patients and 2 patients developed grade 3/4 lymphopenia and grade 3/4 thrombocytopenia in their study respectively without significant hepatic or renal toxicity. They concluded that, PRCRT with 90Y-DOTATATE followed by 177Lu-DOTATATE achieved high clinical and morphological response in NET patients with bulky tumors.[20]
The long-term (10-year follow-up period) side effects of tandem 90Y/ 177Lu-DOTATATE therapy in 59 NET patients were evaluated by Kunikowska et al. in one of their studies. One patient (2%) developed MDS and one patient (2%) grade 3 nephrotoxicity in their study. No other grade 3 and 4 hematological or renal toxicity was observed during their 10-year follow-up period. They concluded that, tandem 90Y/ 177Lu-DOTATATE therapy was highly effective and safe considering long-term side effects of PRRT.[21]
In our study, we evaluated sequential duo-PRRT (177Lu-DOTATATE followed by 90Y-DOTATATE) in total 9 NET patients with bulky large sized lesion (≥5 cm) which was unique in the sense that, as we have started using the combined radionuclide therapy with a size criterion. In our initial observational study with the indigenous 90Y, 8 patients showed excellent (grade 3) 90Y-DOTATATE uptake in lesions on visual analysis, which were matched with 68Ga-DOTATATE uptake and 177Lu-DOTATATE uptake in both PET/CT scanner and bremsstrahlung imaging respectively as shown in Figures 1 and 2.
In our study, the follow-up period after 90Y-DOTATATE therapy ranged from 15 to 135 days with average of 82 days and during this follow-up period, two patients (22.2%) developed mild nausea, one patient (11.1%) developed transient mild-grade hemoglobin toxicity and one patient (11.1%) developed mild-grade gastrointestinal symptoms, which were easily manageable and controllable. In our study, no nephro-toxicity, hepato-toxicity and other major hematological toxicity was noted and these finding were similar to various studies available in literature that used tandem/duo-PRRT for treatment of metastatic/advanced NET. Small population size, short follow-up period, lack of dosimetry and control group, were the major limitations, though we felt it would be worthwhile to report the initial observations.
CONCLUSION
The combination of the indigenous 90Y-DOTATATE with 177Lu-DOTATATE PRRT in NET as sequential duoPRRT was well tolerated, feasible and safe in stable, unresponsive/progressive disease following single isotope 177Lu-DOTATATE therapy and also in neoadjuvant PRRT setting with large bulky lesion (≥5 cm). Post-PRRT 90Y-DOTATATE imaging showed excellent 90Y-DOTATATE uptake in nearly all NET patients. Mild-grade early adverse effects were easily manageable and controllable in this sequential duo-PRRT approach.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
The authors acknowledge the contribution of the Fuel Reprocessing Division, Nuclear Recycle Group, BARC for providing the indigenously sourced 90Y-Acetate as a Clinical Grade Radiochemical for use in Radiopharmaceutical Formulations.
REFERENCES
- 1.Møller LN, Stidsen CE, Hartmann B, Holst JJ. Somatostatin receptors. Biochim Biophys Acta. 2003;1616:1–84. doi: 10.1016/s0005-2736(03)00235-9. [DOI] [PubMed] [Google Scholar]
- 2.Cives M, Soares HP, Strosberg J. Will clinical heterogeneity of neuroendocrine tumors impact their management in the future. Lessons from recent trials? Curr Opin Oncol. 2016;28:359–66. doi: 10.1097/CCO.0000000000000299. [DOI] [PubMed] [Google Scholar]
- 3.Modlin IM, Oberg K, Chung DC, Jensen RT, de Herder WW, Thakker RV, et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008;9:61–72. doi: 10.1016/S1470-2045(07)70410-2. [DOI] [PubMed] [Google Scholar]
- 4.Kim SJ, Kim JW, Han SW, Oh DY, Lee SH, Kim DW, et al. Biological characteristics and treatment outcomes of metastatic or recurrent neuroendocrine tumors: Tumor grade and metastatic site are important for treatment strategy. BMC Cancer. 2010;10:448. doi: 10.1186/1471-2407-10-448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, et al. Phase 3 trial of 177Lu-dotatate for Midgut neuroendocrine tumors. N Engl J Med. 2017;376:125–35. doi: 10.1056/NEJMoa1607427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Hörsch D, O'Dorisio MS, et al. The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2013;40:800–16. doi: 10.1007/s00259-012-2330-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bodei L, Cremonesi M, Grana CM, Chinol M, Baio SM, Severi S, et al. Yttrium-labelled peptides for therapy of NET. Eur J Nucl Med Mol Imaging. 2012;39(Suppl 1):S93–102. doi: 10.1007/s00259-011-2002-y. [DOI] [PubMed] [Google Scholar]
- 8.Kwekkeboom D, Krenning EP, de Jong M. Peptide receptor imaging and therapy. J Nucl Med. 2000;41:1704–13. [PubMed] [Google Scholar]
- 9.Valkema R, De Jong M, Bakker WH, Breeman WA, Kooij PP, Lugtenburg PJ, et al. Phase I study of peptide receptor radionuclide therapy with [In-DTPA] octreotide: The Rotterdam experience. Semin Nucl Med. 2002;32:110–22. doi: 10.1053/snuc/2002.31025. [DOI] [PubMed] [Google Scholar]
- 10.Imhof A, Brunner P, Marincek N, Briel M, Schindler C, Rasch H, et al. Response, survival, and long-term toxicity after therapy with the radiolabeled somatostatin analogue [90Y-DOTA]-TOC in metastasized neuroendocrine cancers. J Clin Oncol. 2011;29:2416–23. doi: 10.1200/JCO.2010.33.7873. [DOI] [PubMed] [Google Scholar]
- 11.Severi S, Grassi I, Nicolini S, Sansovini M, Bongiovanni A, Paganelli G. Peptide receptor radionuclide therapy in the management of gastrointestinal neuroendocrine tumors: Efficacy profile, safety, and quality of life. Onco Targets Ther. 2017;10:551–7. doi: 10.2147/OTT.S97584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Reubi JC, Schär JC, Waser B, Wenger S, Heppeler A, Schmitt JS, et al. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med. 2000;27:273–82. doi: 10.1007/s002590050034. [DOI] [PubMed] [Google Scholar]
- 13.Siegel JA, Stabin MG. Absorbed fractions for electrons and beta particles in spheres of various sizes. J Nucl Med. 1994;35:152–6. [PubMed] [Google Scholar]
- 14.de Jong M, Breeman WA, Valkema R, Bernard BF, Krenning EP. Combination radionuclide therapy using 177Lu- and 90Y-labeled somatostatin analogs. J Nucl Med. 2005;46(Suppl 1):13S–7S. [PubMed] [Google Scholar]
- 15.De Jong M, Bernard HF, Breeman WA, Bernard BF, Krenning EP. Combination of 90Y- and 177Lu-labeled somatostatin analogs is superior for radionuclide therapy compared to 90Y- or 177Lulabeled analogs only. J Nucl Med. 2002;43:123–4. [Google Scholar]
- 16.Kunikowska J, Królicki L, Hubalewska-Dydejczyk A, Mikołajczak R, Sowa-Staszczak A, Pawlak D. Clinical results of radionuclide therapy of neuroendocrine tumours with 90Y-DOTATATE and tandem 90Y/177Lu-DOTATATE: Which is a better therapy option? Eur J Nucl Med Mol Imaging. 2011;38:1788–97. doi: 10.1007/s00259-011-1833-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Villard L, Romer A, Marincek N, Brunner P, Koller MT, Schindler C, et al. Cohort study of somatostatin-based radiopeptide therapy with [(90) Y-DOTA]-TOC versus [(90) Y-DOTA]-TOC plus [(177) Lu-DOTA]-TOC in neuroendocrine cancers. J Clin Oncol. 2012;30:1100–6. doi: 10.1200/JCO.2011.37.2151. [DOI] [PubMed] [Google Scholar]
- 18.Seregni E, Maccauro M, Chiesa C, Mazzaferro V, De Braud F, Buzzoni R, et al. Treatment with tandem [90Y]DOTA-TATE and [177Lu]DOTA-TATE of neuroendocrine tumours refractory to conventional therapy. Eur J Nucl Med Mol Imaging. 2014;41:223–30. doi: 10.1007/s00259-013-2578-5. [DOI] [PubMed] [Google Scholar]
- 19.Dumont RA, Seiler D, Marincek N, Brunner P, Radojewski P, Rochlitz C, et al. Survival after somatostatin based radiopeptide therapy with 90Y-DOTATOC vs.90Y-DOTATOC plus 177Lu-DOTATOC in metastasized gastrinoma. Am J Nucl Med Mol Imaging. 2015;5:46–55. [PMC free article] [PubMed] [Google Scholar]
- 20.Kong G, Callahan J, Hofman MS, Pattison DA, Akhurst T, Michael M, et al. High clinical and morphologic response using 90Y-DOTA-octreotate sequenced with 177Lu-DOTA-octreotate induction peptide receptor chemoradionuclide therapy (PRCRT) for bulky neuroendocrine tumours. Eur J Nucl Med Mol Imaging. 2017;44:476–89. doi: 10.1007/s00259-016-3527-x. [DOI] [PubMed] [Google Scholar]
- 21.Kunikowska J, Pawlak D, Bąk MI, Kos-Kudła B, Mikołajczak R, Królicki L. Long-term results and tolerability of tandem peptide receptor radionuclide therapy with 90Y/177Lu-DOTATATE in neuroendocrine tumors with respect to the primary location: A 10-year study. Ann Nucl Med. 2017;31:347–6. doi: 10.1007/s12149-017-1163-6. [DOI] [PubMed] [Google Scholar]