Abstract
Cell aging is the result of deteriorating competence in maintaining cellular homeostasis and quality control. Certain cell types are able to rejuvenate through asymmetric cell division by excluding aging factors, including damaged cellular compartments and extra chromosomal rDNA circles, from entering the daughter cell. Recent findings from the budding yeast S. cerevisiae have shown that gametogenesis represents another type of cellular rejuvenation. Gametes, whether produced by an old or a young mother cell, are granted a renewed replicative lifespan through the formation of a fifth nuclear compartment that sequesters the harmful senescence factors accumulated by the mother. Here, we describe the importance and mechanism of cellular remodeling at the nuclear envelope mediated by ESCRT-III and the LEM-domain proteins, with a focus on nuclear pore biogenesis and chromatin interaction during gamete rejuvenation.
Introduction
A hallmark of aging is the progressive accumulation of cellular damage. Age-induced damage is caused by a decrease in cellular homeostasis and protein quality control, affecting both the chronological age of the cell and its replicative lifespan (Longo et al., 2012). While the occurrence of self-renewal and rejuvenation have been well documented for a variety of cell types, the cellular mechanisms utilized to mitigate the effects of aging are not fully understood. Recent work in the budding yeast S. cerevisiae has begun to shine a light on these fundamental processes.
A crucial determinant in reducing the effects of cellular aging is proper trafficking across the nuclear envelope which forms a selective barrier between the cytoplasm and nucleoplasm (Ungricht and Kutay, 2015). The semipermeable barrier provided by the nuclear pore complexes (NPCs), which are embedded within the nuclear envelope, aids in maintaining nuclear integrity and nuclear envelope homeostasis (Beck and Hurt, 2017; Kim et al., 2018). In budding yeast, malformed NPCs accumulate in the Storage of Improperly Assembled Nuclear pore Complexes compartment, called the SINC (Webster et al., 2014). The SINC is not passed on to daughter cells but instead remains with the mother during mitosis, which is asymmetric in budding yeast (Colombi et al., 2013; Makio et al., 2013; Webster et al., 2014). Consequently, the daughter cell is born with a renewed replicative lifespan while the mother ages, accumulating damaged cellular compartments and protein aggregates, with each new bud until death (Sinclair et al., 1998).
In contrast to mitosis, meiosis is morphologically symmetrical and produces gametes, also referred to as spores in budding yeast (Neiman, 2011). During gametogenesis, meiotic cells reset aging such that all gametes have a renewed replicative lifespan (Unal and Amon, 2011; Unal et al., 2011). Senescence factors such as extrachromosomal rDNA circles and protein aggregates originally observed in the mother cell are not present in the newly formed gametes (Fuchs and Loidl, 2004; Unal and Amon, 2011; Unal et al., 2011; King et al., 2019; Koch et al., 2020). Recent studies of the formation of the Gametogenesis Uninherited Nuclear Compartment (GUNC) during budding yeast meiosis offer a mechanistic explanation for how aged mother cells can produce four fully renewed gametes (King et al., 2019; King and Unal, 2020; Koch et al., 2020) and the ramifications on gamete lifespan of hindering this sequestration (Koch et al., 2020). This knowledge indicates a role for nuclear envelope remodeling and nuclear pore biogenesis in rejuvenation of gametes in budding yeast, which may have implications for understanding self-renewal during animal gametogenesis.
GUNC formation during budding yeast gametogenesis
During gametogenesis in budding yeast, age-induced factors, including extrachromosomal rDNA circles, heat shock proteins, and NPCs are selectively sequestered to the GUNC (King et al., 2019; Koch et al., 2020). The wide-scale nucleoporin sequestration observed in meiosis indicates that the formation of the GUNC involves both the nuclear envelope and nucleoplasm in a coordinated fashion. GUNC initially forms via the biogenesis of the prospore membrane, projecting from the spindle pole body (King et al., 2019; King and Unal, 2020). There are two potential mechanisms by which the prospore membrane biogenesis may affect GUNC formation. First, as the prospore membrane expands, it actively pushes NPCs into the GUNC. Alternatively, the rapid growth of the nuclear membranes during meiosis II, corresponding to the formation of the prospore membrane, may passively leave behind NPCs in the GUNC. In either scenario, we envision that the prospore membrane functions as a selective filter facilitating the initial constriction of NPCs, with the Endosomal Sorting Complex Required for Transport (ESCRT)-III complex finalizing the sequestration of nucleoporins to GUNC (Koch et al., 2020).
ESCRT-III constriction of NPCs to GUNC during gametogenesis
ESCRT-III and its associated factors are known to function in NPC biogenesis (Webster et al., 2014; Thaller et al., 2019), and we propose many of these interactions are required for sequestration of NPCs to the GUNC. The LEM (Lap2-emerin-MAN1) family of integral inner nuclear membrane proteins, Heh1 (which is also called Src1) and Heh2 have been shown to mediate the surveillance of NPC biogenesis in conjunction with ESCRT-III components, Chm7, Snf7, and Vps4 (Webster et al., 2014; Webster et al., 2016). This surveillance mechanism involves Heh1 as the recruiting factor responsible for the focal accumulation of Chm7 at specific sites along the nuclear envelope, in turn activating ESCRT-III likely through its direct interaction between Snf7 and Chm7 (Webster et al., 2016; Thaller et al., 2019).
During meiosis, Heh1 and ESCRT-III are required for the constriction of nucleoporins to the GUNC (Koch et al., 2020). We propose that during late anaphase II, Heh1 recruits ESCRT-III and Chm7 to the junction between the expanding gamete nuclei and the GUNC. The ESCRT-III complex then likely binds Vps4 at these sites and constricts the nuclear envelope, leading to the eventual scission of the membrane leaving four gamete nuclei separated from the GUNC (Koch et al., 2020).
The formation of GUNC is analogous to nuclear division in budding yeast mitosis, in which the mother cell retains all of the nuclear senescence factors (Kaeberlein, 2010; Longo et al., 2012). Functionally, the GUNC therefore resembles the mitotic mother cell retaining the old or damaged NPCs and other aging factors. The meiotic constriction of NPCs to the GUNC is facilitated by the actions of Heh1 and ESCRT-III (Koch et al., 2020), and given that Heh1 directs ESCRT-III during mitotic NPC biogenesis through binding Chm7 following cytoplasmic exposure (Webster et al., 2016), Heh1 may be the determining factor of where and how the GUNC constriction sites are located. In support of a Heh1 dependent mechanism, recent data suggests the LEM-domain protein, LEM2, in human cells is capable of condensing on microtubules in a liquid-like phase and co-assembles with the Chm7 homolog, CHMP7, to activate ESCRT and function as an O-ring seal at the confluence between membranes (von Appen et al., 2020). The idea that budding yeast gametogenesis may utilize a similar phase separation to constrict aging factors from nascent gametes is an attractive model that requires further analysis.
Is NPC remodeling related to nuclear envelope breakdown?
We propose that ESCRT-III/Vps4 in conjunction with Heh1 and Chm7 is responsible for the membrane cleavage separating the GUNC from the newly formed gametic nuclei. One question yet to be answered is: What is the mechanism by which NPCs are remodeled at the nuclear envelope during the process of GUNC formation? Looking to other single-celled eukaryotes, the fission yeast S. pombe may provide insight into nuclear envelope dynamics and NPC remodeling. In fission yeast, the nuclear envelope is intact during meiosis I, but during anaphase II there is a virtual Nuclear Envelope Breakdown (vNEB), creating an increase in permeability which allows for diffusion of nuclear proteins into the cytoplasm (Arai et al., 2010; Asakawa et al., 2010). A localized NEB, in which the nuclear envelope breaks down at specific regions, has also been observed in fission yeast mitosis, at the microtubule bridge, giving the dividing nuclei an intermediate dumbbell shape (Dey et al., 2020) and at the centrosome (Fernandez-Alvarez et al., 2016). NPCs aggregate in the middle of the bridge in a manner dependent upon Les1 (Dey et al., 2020). The Les1 protein localizes exclusively to the inner nuclear membrane to form stalks around the mitotic spindle, constricting movement along the microtubule bridge and orienting the location of NEB (Dey et al., 2020). It has been shown that Les1 interacts with ESCRT-III proteins to ensure proper nuclear envelope sealing, and upon deletion of LES1, NPCs are no longer sequestered to the microtubule bridge (Dey et al., 2020). This new information on Les1 in fission yeast mitosis provides insight into the budding yeast homolog Msc1, which may have a similar function in the sequestration of NPCs in meiosis. Future study of Msc1 may provide insight into the connection of NEB to NPC remodeling in budding yeast meiosis.
Nuclear envelope remodeling and chromosome segregation
During GUNC formation, the core subunits of the NPC are sequestered, whereas basket nucleoporins appear distributed along the periphery of the gamete nuclei (King et al., 2019; Koch et al., 2020). The significance of this partial disassembly of the NPC remains unclear, but it is consistent with the idea of dynamic NPC modularity, which is tightly regulated (Knockenhauer and Schwartz, 2016). The dissociation of basket nucleoporins from the core and their subsequent localization to the gamete nuclei late in meiosis may allow the amphipathic helices of basket nucleoporins to dock onto and curve the nuclear membrane (Meszaros et al., 2015). Also in meiosis II, chromosomes initially localize to the midzone, then migrate to the nascent gamete nuclei upon GUNC formation (King et al., 2019; Koch et al., 2020). Chromosome movement during this process is concomitant with that of the basket nucleoporins (King et al., 2019; Koch et al., 2020). Given that basket nucleoporins associate with chromatin during cell divisions (Dultz et al., 2008; Markossian et al., 2015; Suresh et al., 2017), we speculate that separation of basket nucleoporins from the core may be crucial for disrupting the interaction between the NPC and chromatin that would otherwise inhibit NPC constriction to the GUNC. LEM proteins tether chromatin to the nuclear periphery (Grund et al., 2008; Mekhail et al., 2008), we therefore hypothesize that the interaction between NPCs and chromatin is perturbed in the absence of Heh1, Heh2 or Vps4. Along with the lack of sequestration to the GUNC in the absence of these proteins, we observed a mutant phenotype in which the NPCs coalesced around a single gamete nucleus (Figure 1A; Koch et al., 2020), indicating that the NPC and chromatin interaction somehow remains intact. This phenotype was observed in approximately one quarter of heh1 and vps4 mutants, but in over two thirds of heh2 mutants (Koch et al., 2020). Another chromosomal defect we observed was NPCs encapsulating a fifth chromatin mass in the GUNC-like compartment in a little under one quarter of heh1 and vps4 mutants (Figure 1A; Koch et al., 2020). The five nuclear bodies produced from these meiotic cycles were inviable, indicating a severe chromosome segregation defect (our unpublished data). We hypothesize that ESCRT-III and the LEM-domain proteins play dual roles during yeast gametogenesis (Figure 1B). First, they participate in NPC constriction and GUNC formation (Koch et al., 2020). Second, they function in meiotic chromosome segregation, by linking chromosomes to NPCs either directly or indirectly. The LEM-domain protein to chromatin interaction at the NPC potentially can be severed by ESCRT-III/Vps4, as shown in fission yeast (Pieper et al., 2020). In contrast to Heh1, which is highly abundant in late meiosis II, the expression level of HEH2 is dramatically downregulated (Chu et al., 1998), suggesting another layer of regulation of LEM protein and chromatin interaction. Disconnecting chromatin from the NPC may trigger the release of the chromosomes and basket nucleoporins into the daughter nuclei, while the NPC core subunits remain constricted to GUNC.
Figure 1.
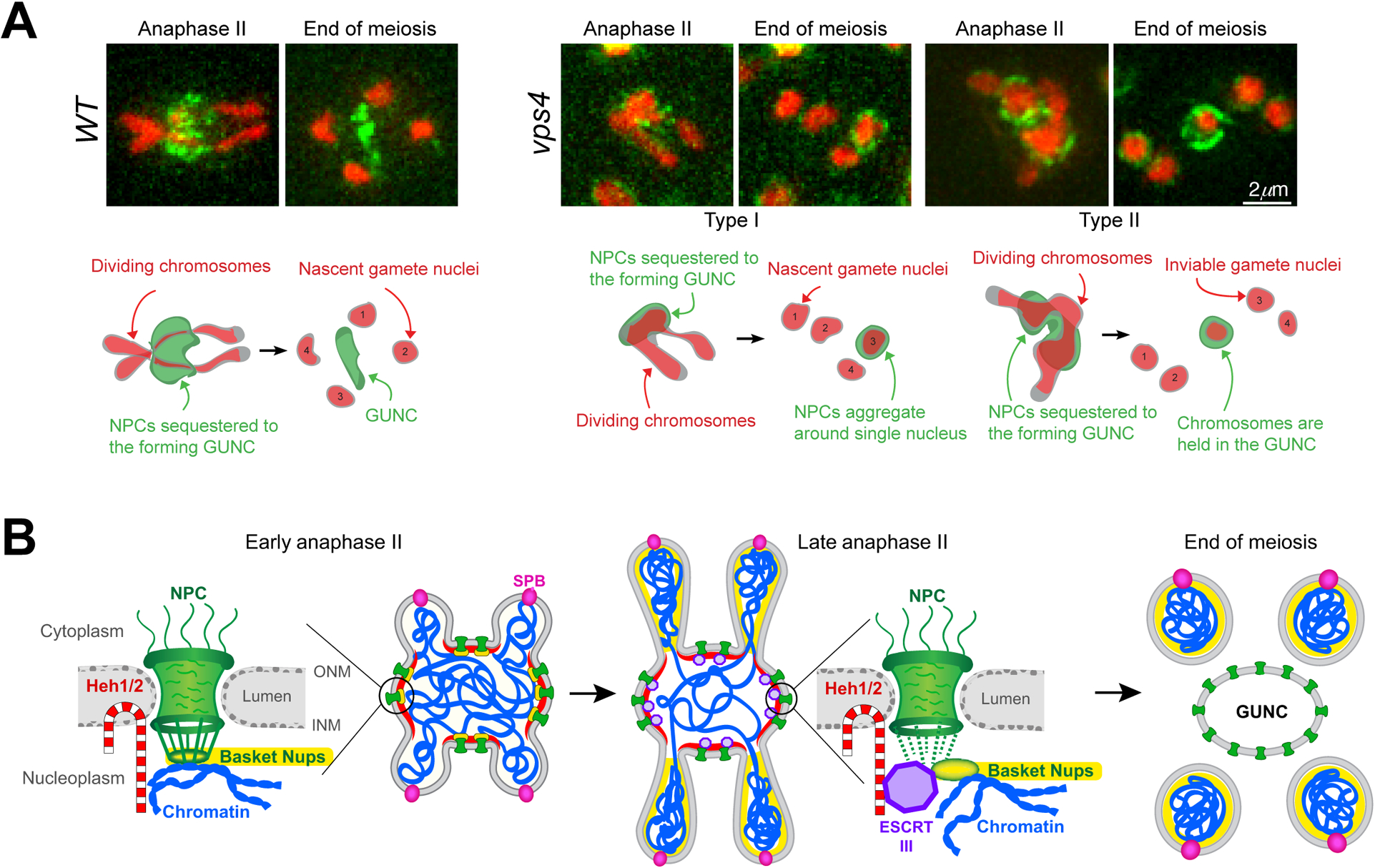
LEM-domain proteins and ESCRT-III regulate NPC sequestration and chromosome segregation during gametogenesis in budding yeast. (A) Selected images from time-lapse microscopy showing the nucleoporin Pom34-GFP (green) localization in wild-type (WT) and vps4 mutant cells. Hta1-mApple (red) marks the chromosomes. The corresponding diagrams shown below detail chromosome movement and GUNC (gametogenesis uninherited nuclear compartment) formation in WT and vps4 mutants during meiosis II. (B) Hypothetical model for LEM protein and ESCRT-III action at the nuclear envelope during gametogenesis in budding yeast. Recruitment of ESCRT-III/Vps4 (shown in purple) by the LEM-domain proteins (red) to the nuclear envelope promotes the disassociation of basket nucleoporins (yellow) from the core. Nuclear pore complexes (NPCs) are shown in green, chromosomes in blue, and the spindle pole body (SPB) in pink.
The idea of a dual action of ESCRT-III/Vps4 functioning with either Heh1/Chm7 or Heh2 to ensure gamete rejuvenation via restriction of senescence factors to GUNC and faithful chromosome segregation provides a testable hypothesis. Determining the role of LEM-domain proteins’ interaction with the NPC and chromatin, and how ESCRT-III helps mediate the process of NPC remodeling may be the key to understanding GUNC formation and gamete rejuvenation.
Acknowledgement
We thank Charles Badland and Jen Kennedy for their assistance. The work in authors’ lab is supported by the National Institutes of Health GM138838 and the National Science Foundation MCB1951313.
Footnotes
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
References
- Arai K, Sato M, Tanaka K, and Yamamoto M. 2010. Nuclear compartmentalization is abolished during fission yeast meiosis. Curr Biol. 20:1913–1918. [DOI] [PubMed] [Google Scholar]
- Asakawa H, Kojidani T, Mori C, Osakada H, Sato M, Ding DQ, Hiraoka Y, and Haraguchi T. 2010. Virtual breakdown of the nuclear envelope in fission yeast meiosis. Curr Biol. 20:1919–1925. [DOI] [PubMed] [Google Scholar]
- Beck M, and Hurt E. 2017. The nuclear pore complex: understanding its function through structural insight. Nat Rev Mol Cell Biol. 18:73–89. [DOI] [PubMed] [Google Scholar]
- Chu S, DeRisi J, Eisen M, Mulholland J, Botstein D, Brown PO, and Herskowitz I. 1998. The transcriptional program of sporulation in budding yeast. Science. 282:699–705. [DOI] [PubMed] [Google Scholar]
- Colombi P, Webster BM, Frohlich F, and Lusk CP. 2013. The transmission of nuclear pore complexes to daughter cells requires a cytoplasmic pool of Nsp1. J Cell Biol. 203:215–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dey G, Culley S, Curran S, Schmidt U, Henriques R, Kukulski W, and Baum B. 2020. Closed mitosis requires local disassembly of the nuclear envelope. Nature. 585:119–123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dultz E, Zanin E, Wurzenberger C, Braun M, Rabut G, Sironi L, and Ellenberg J. 2008. Systematic kinetic analysis of mitotic dis- and reassembly of the nuclear pore in living cells. J Cell Biol. 180:857–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandez-Alvarez A, Bez C, O’Toole ET, Morphew M, and Cooper JP. 2016. Mitotic Nuclear Envelope Breakdown and Spindle Nucleation Are Controlled by Interphase Contacts between Centromeres and the Nuclear Envelope. Dev Cell. 39:544–559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuchs J, and Loidl J. 2004. Behaviour of nucleolus organizing regions (NORs) and nucleoli during mitotic and meiotic divisions in budding yeast. Chromosome Res. 12:427–438. [DOI] [PubMed] [Google Scholar]
- Grund SE, Fischer T, Cabal GG, Antunez O, Perez-Ortin JE, and Hurt E. 2008. The inner nuclear membrane protein Src1 associates with subtelomeric genes and alters their regulated gene expression. J Cell Biol. 182:897–910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaeberlein M 2010. Lessons on longevity from budding yeast. Nature. 464:513–519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SJ, Fernandez-Martinez J, Nudelman I, Shi Y, Zhang W, Raveh B, Herricks T, Slaughter BD, Hogan JA, Upla P, Chemmama IE, Pellarin R, Echeverria I, Shivaraju M, Chaudhury AS, Wang J, Williams R, Unruh JR, Greenberg CH, Jacobs EY, Yu Z, de la Cruz MJ, Mironska R, Stokes DL, Aitchison JD, Jarrold MF, Gerton JL, Ludtke SJ, Akey CW, Chait BT, Sali A, and Rout MP. 2018. Integrative structure and functional anatomy of a nuclear pore complex. Nature. 555:475–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King GA, Goodman JS, Schick JG, Chetlapalli K, Jorgens DM, McDonald KL, and Unal E. 2019. Meiotic cellular rejuvenation is coupled to nuclear remodeling in budding yeast. Elife. 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King GA, and Unal E. 2020. The dynamic nuclear periphery as a facilitator of gamete health and rejuvenation. Curr Genet. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knockenhauer KE, and Schwartz TU. 2016. The Nuclear Pore Complex as a Flexible and Dynamic Gate. Cell. 164:1162–1171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch BA, Staley E, Jin H, and Yu HG. 2020. The ESCRT-III complex is required for nuclear pore complex sequestration and regulates gamete replicative lifespan in budding yeast meiosis. Nucleus. 11:219–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo VD, Shadel GS, Kaeberlein M, and Kennedy B. 2012. Replicative and chronological aging in Saccharomyces cerevisiae. Cell Metab. 16:18–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makio T, Lapetina DL, and Wozniak RW. 2013. Inheritance of yeast nuclear pore complexes requires the Nsp1p subcomplex. J Cell Biol. 203:187–196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markossian S, Suresh S, Osmani AH, and Osmani SA. 2015. Nup2 requires a highly divergent partner, NupA, to fulfill functions at nuclear pore complexes and the mitotic chromatin region. Mol Biol Cell. 26:605–621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mekhail K, Seebacher J, Gygi SP, and Moazed D. 2008. Role for perinuclear chromosome tethering in maintenance of genome stability. Nature. 456:667–670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meszaros N, Cibulka J, Mendiburo MJ, Romanauska A, Schneider M, and Kohler A. 2015. Nuclear pore basket proteins are tethered to the nuclear envelope and can regulate membrane curvature. Dev Cell. 33:285–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neiman AM 2011. Sporulation in the budding yeast Saccharomyces cerevisiae. Genetics. 189:737–765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieper GH, Sprenger S, Teis D, and Oliferenko S. 2020. ESCRT-III/Vps4 Controls Heterochromatin-Nuclear Envelope Attachments. Dev Cell. 53:27–41 e26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinclair D, Mills K, and Guarente L. 1998. Aging in Saccharomyces cerevisiae. Annu Rev Microbiol. 52:533–560. [DOI] [PubMed] [Google Scholar]
- Suresh S, Markossian S, Osmani AH, and Osmani SA. 2017. Mitotic nuclear pore complex segregation involves Nup2 in Aspergillus nidulans. J Cell Biol. 216:2813–2826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thaller DJ, Allegretti M, Borah S, Ronchi P, Beck M, and Lusk CP. 2019. An ESCRT-LEM protein surveillance system is poised to directly monitor the nuclear envelope and nuclear transport system. Elife. 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unal E, and Amon A. 2011. Gamete formation resets the aging clock in yeast. Cold Spring Harb Symp Quant Biol. 76:73–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unal E, Kinde B, and Amon A. 2011. Gametogenesis eliminates age-induced cellular damage and resets life span in yeast. Science. 332:1554–1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ungricht R, and Kutay U. 2015. Establishment of NE asymmetry-targeting of membrane proteins to the inner nuclear membrane. Curr Opin Cell Biol. 34:135–141. [DOI] [PubMed] [Google Scholar]
- von Appen A, LaJoie D, Johnson IE, Trnka MJ, Pick SM, Burlingame AL, Ullman KS, and Frost A. 2020. LEM2 phase separation promotes ESCRT-mediated nuclear envelope reformation. Nature. 582:115–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster BM, Colombi P, Jager J, and Lusk CP. 2014. Surveillance of nuclear pore complex assembly by ESCRT-III/Vps4. Cell. 159:388–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Webster BM, Thaller DJ, Jager J, Ochmann SE, Borah S, and Lusk CP. 2016. Chm7 and Heh1 collaborate to link nuclear pore complex quality control with nuclear envelope sealing. EMBO J. 35:2447–2467. [DOI] [PMC free article] [PubMed] [Google Scholar]


