Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords: pathology competencies, disease mechanisms, immunologic mechanisms, immune dysfunction, transplantation, organ rejection
Primary Objective
Objective IM1.8: Transplantation. Discuss the consequences of tissue transplantation, including mechanisms and pathophysiology of graft versus host organ rejection, and the possible therapeutic interventions that can mitigate these effects.
Competency 1: Disease Mechanisms and Processes; Topic Immunological Mechanisms (IM); Learning Goal 1: Immune dysfunction
Patient Presentation
A 40-year-old man presents to the emergency department (ED) complaining of gradually worsening fatigue and malaise for 2 days. His medical history includes morbid obesity, hypertension, diabetes mellitus, gout, and end-stage renal disease secondary to hypertension, for which he underwent a deceased-donor kidney transplant 1 month ago. There are no other symptoms. Specifically, he denies fever, shortness of breath, myalgia, flank pain, and dysuria. He also denies any recent travel or contacts with sick individuals. The family history is positive for hypertension and diabetes but is otherwise unremarkable. He does not drink or smoke and denies any illicit drug use. Further inquiry of his post-transplant history reveals an uneventful clinical course, with a functional transplanted kidney at the time of discharge home. He also reports that he has been compliant with medications as directed by his kidney doctor, including tacrolimus and a corticosteroid.
Diagnostic Findings, Part 1
On examination, the patient’s vital signs are stable (heart rate: 74 beats per minute, temperature: 99.4 °F; respiratory rate: 16 per minute), but his blood pressure is slightly increased (145/88 mm Hg). He measures 185 cm in height and weighs 125 kg (body mass index: 36.5), appears well-nourished and in no acute distress. Review of systems and physical examination do not reveal additional relevant information. Laboratory evaluation includes a serum creatinine level of 9.9 mg/dL (reference range: <1.5 mg/dL), and the glomerular filtration rate (GFR) is markedly decreased (16 mL/min/1.73m2). The patient is hypocalcemic at 7 mg/dL (8.5-10.5 mg/dL) but electrolytes are within normal limits. The hemoglobin level is 7.1 g/dL (14-17.4 g/dL) and the white blood cell count is 8400/µL (4800-10800/µL). Qualitative urinalysis shows trace blood, protein +1, and few white blood cells. Repeated tacrolimus serum levels are all within therapeutic range.
Additional serum studies show that patient does not have circulating cytomegalovirus or BK polyomavirus in the blood.
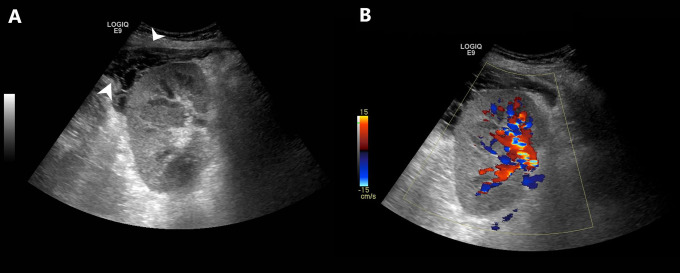
Ultrasound imaging with duplex Doppler study is performed on the allograft kidney (Figure 1) and demonstrates patent renal vasculature with a normal flow rate. There is no evidence of arterial stenosis, venous thrombosis, hydronephrosis, or renal calculi. There is a known perinephric fluid collection that has been stable in size since transplantation.
Figure 1.
Renal allograft ultrasonogram. A, The kidney is normal in appearance with no evidence of thrombosis, arterial thickening, hydronephrosis, or calculus formation. Note that there is a hypo-echoic loculated structure near the upper pole, implying fluid accumulation (arrowhead). B, Ultrasonographic enhancement by spectral imaging shows normal renal perfusion. The absence of spectral enhancement indicates the lack of fluid movement within the accumulation, which is suggestive of seroma.
Question/Discussion Points, Part 1
What Is the Differential Diagnosis Based on the Clinical History and Initial Diagnostic Findings?
The patient’s nonspecific presentation encompasses a wide range of possible diagnoses including infection, autoimmune processes, toxic or ischemic injury, nutritional deficiencies, and even psychosomatic causes. Further evaluation is necessary to establish that diagnosis. The presence of perinephric fluid also raises the concern of possible surgical complications such as hematoma and urinoma (urine leak into perinephric tissues) as well as infection with abscess formation. Other possible diagnoses include viral infection, medication-induced reaction/toxicity, and post-transplant lymphoproliferative disease. All of these should be entertained since they can present with similar findings (Table 1 2).
Table 1.
Common Causes of Renal Allograft Dysfunction.2,*
| Common causes of renal allograft dysfunction | ||
|---|---|---|
| Immediate post-transplantation (< 7 days) | Immunological |
|
| Vascular |
|
|
| Surgical |
|
|
| Early (1 week-3 months) |
Immunological |
|
| Infectious |
|
|
| Drug toxicity |
|
|
| Late (> 3 months) |
|
|
* One of the important factors to consider during evaluation is the timing of presentation. A diagnosis can sometimes be established by physical examination and laboratory evaluation, but additional evaluation is frequently required.
The initial studies performed at the ED are helpful in ruling out various pathologic processes and revealing the underlying cause of the patient’s presentation. The absence of leukocytosis, negative findings in viral studies, and therapeutic-range tacrolimus serum level have made infection and drug toxicity less likely. Given the current findings, including the short post-transplantation time frame, organ rejection is a major concern and should be at the top of the differential diagnosis.
What Are the Different Types of Renal Allograft Rejection?
There are 2 main types of transplant rejection, one mediated by T lymphocytes, the other by circulating antibodies. They are not mutually exclusive and can at times be seen in the same biopsy.3 As outlined in Table 1, timing is usually helpful in determining the type of organ rejection, with different parts of immune system activated at different times in the post-transplant course. The classification and subcategorization of types of organ rejection continues to change as our understanding of the etiology and pathophysiology of the immune regulatory response evolves. Presently, many scholars characterize renal allograft rejection based on both temporal occurrence (hyperacute, acute, chronic) and mechanism involved (cellular- or antibody-mediated), as described in Table 2.4
Table 2.
Classification of Renal Allograft Rejection.4
| Types of renal allograft rejection | ||
|---|---|---|
| T-cell mediated (cellular) | Acute |
|
| Chronic |
|
|
| Antibody Mediated (humoral) | Hyperacute |
|
| Acute |
|
|
| Chronic |
|
|
Abbreviations: DSA, donor-specific antigen; HLA, human leukocyte antigen; MHC, major histocompatibility complex; PTC, peritubular capillary.
How Is the Pathogenesis of T Cell-Mediated Rejection Different From That of Antibody Mediated Rejection?
A tremendous amount of work has been done in the field of immunotherapy and solid organ rejection, and it is still an area of extensive research.4 Organ rejection results from a complex series of interactions between the grafted organs and the host’s immune defense.
T cell-mediated rejection, also known as acute cellular rejection, is more frequently seen during the first 6 months after transplantation. As the name suggests, the key cell type in this form of rejection is the T lymphocyte. The chain of events is initiated through the presentation and recognition of human leukocyte antigens in the donor organ that are foreign to the recipient. A special subgroup of immune cells, called the antigen presenting cell, is responsible for taking up and presenting these antigens to naïve T lymphocytes. Via interactions such as receptor binding and chemokine stimulation, a molecular signal cascade ensues, and naïve T lymphocytes undergo a maturation process to become differentiated and activated. They then migrate to and infiltrate the grafted organ and begin an inflammatory process with tissue injury.3,5
Our understanding of the regulating mechanisms and molecular pathways of antibody-mediated rejection is still evolving. As in T cell-mediated rejection, exposure of antigens from the grafted organ to the immune system is believed to be the inciting event.6 In response, allo- and auto-antibodies are expressed and released by the host’s B lymphocytes and plasma cells, leading to antibody complex formation and complement cascade activation via the classical pathway. Ultimately there is organ damage and dysfunction. As the complement cascade is activated by circulating antibodies, the breakdown product C4d is generated. It has a long half-life and covalently binds to microvascular endothelial cells and their basement membranes, allowing it to be visualized in biopsies using immunohistochemical techniques. There is a strong (although not universal) correlation between C4d staining, the presence of circulating donor-specific alloantibodies, and clinical evidence of rejection.7,8 Hence, immunohistochemical staining for C4d is a routine practice in evaluation of the renal transplant biopsy.
What Additional Testing Should Be Performed?
There are no laboratory or imaging studies that will specifically point to a diagnosis of T cell-mediated rejection or cellular rejection. To evaluate for antibody-mediated rejection, an assessment for donor-specific antibodies (DSA) is important. Although there are exceptions, DSA are present in most cases of antibody-mediated rejection. In rare instances, recurrent or de novo native renal diseases can occur in the early post-transplant course, and they are important in the evaluation. Specialized laboratory studies, however, should always be selected with care and in appropriate clinical scenarios to ensure effective and cost-efficient laboratory utilization.
Usually, when renal allograft rejection is suspected, biopsy is warranted to confirm or rule out the diagnosis. Although there are limitations, such as representative and adequate sampling, renal biopsy remains the gold standard for assessing the mechanism and the severity of allograft injury. In general, a minimum of two 1-cm cores should be obtained for accurate assessment. While most of the specimen is processed and embedded in paraffin, small portions of the core biopsy will often also be sent for immunofluorescence microscopy and electron microscopy,9 particularly for transplants in place for over 6 months.
Diagnostic Findings, Part 2
A biopsy is performed and an adequate sample containing 14 glomeruli is obtained. The findings were considered sufficient to establish the diagnosis of active T cell-mediated rejection. In the absence of clinical or laboratory evidence of other types of renal disease, it was elected to defer immunofluorescence and electron microscopy.
Question/Discussion Points, Part 2
What Are the Specific Findings in the Renal Biopsy?
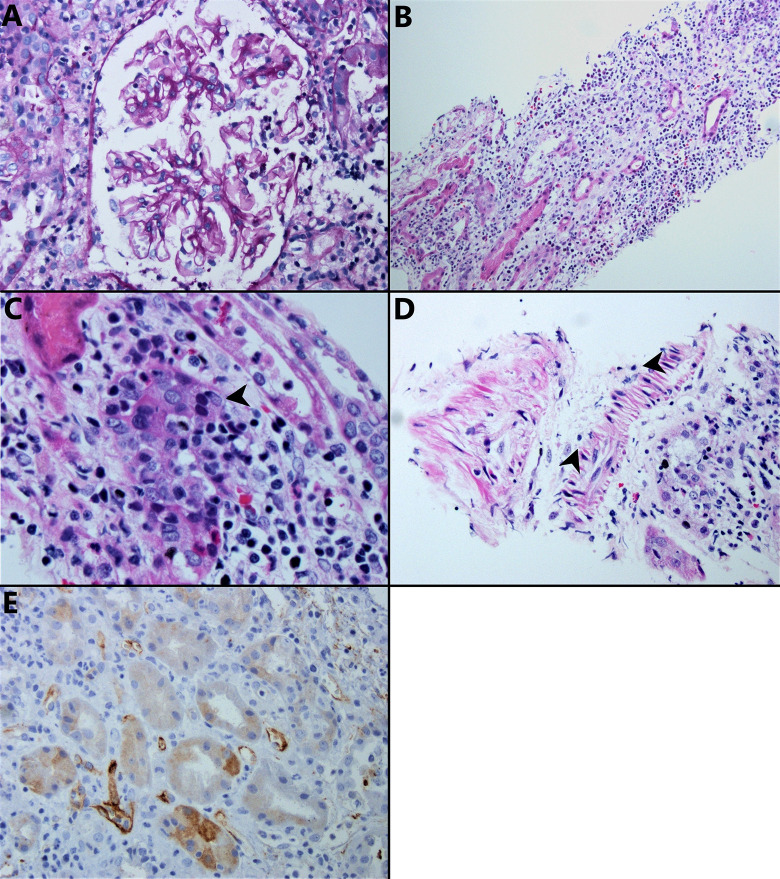
Routine histologic findings are illustrated in Figure 2A-D, with the C4d immunohistochemical stain in Figure 2E. All of the glomeruli show no histopathologic abnormality (Figure 2A). The tubulointerstitium is remarkable for the loss of tubules and a marked inflammatory infiltrate consisting predominantly of lymphocytes with few plasma cells and eosinophils (Figure 2B). Lymphocytes infiltrate into proximal tubular epithelium, in some places exceeding 10 lymphocytes per tubular cross section (Figure 2C). A few arteries contain lymphocytes within the intima (intimal arteritis, Figure 2D), but there is no transmural infiltration or frank necrosis. An immunohistochemical stain for complement component C4d shows moderate staining in less than 10% of peritubular capillaries with nonspecific staining of tubular epithelium (Figure 2E).
Figure 2.
Allograft kidney biopsy. A, Glomeruli are normal (periodic acid-Schiff stain, ×200). B, There is interstitial inflammation (hematoxylin and eosin stain, ×100). C, Tubulitis is present (arrowhead) (hematoxylin and eosin stain, ×400). D, Intimal arteritis is also present (arterial intima lymphocytic infiltration, arrowheads) (hematoxylin and eosin stain, ×200). E, There is focal peritubular capillary C4d staining (immunohistochemical stain, ×200).
How Do Pathologists Evaluate Renal Allograft Diseases?
Historically, pathologists described renal allograft abnormalities based on patterns of injury.10 While this helped clinicians to understand the etiology of renal dysfunction, the lack of standardization caused significant interobserver variability as well as difficulty in creating treatment plans. A reporting schema was proposed by a group of renal pathologists, nephrologists, and transplant surgeons at an international conference in Banff, Canada, in 1991.11 The proposed system evolved into the Banff Classification, which has been reviewed and updated every 2 years since then using evidence-based studies. It is now the gold standard for diagnosis of allograft disease in the kidney as well as other transplanted solid organs. It is widely accepted by pathologists and clinicians as it standardizes renal allograft biopsy reporting and allows meaningful comparison of clinical studies.
The Banff Classification considers several parameters, including (1) inflammation and resultant damage to any of the renal histologic compartments; (2) alterations in microscopic structure; (3) evidence of chronic injury; and (4) deposition of molecules associated with immune-mediated reactions. Numerous individual features are analyzed and assigned scores on a point-based system. The scores are then used in categorizing the overall observed lesions. The classification scheme provides a highly granular, objective method for evaluation of renal transplant biopsies.
For this patient, marked interstitial inflammation and tubulitis with mild vasculitis produces a Banff classification of active T cell-mediated rejection, Grade IIA. In addition, C4d staining may indicate additional antibody-mediated rejection. However, the staining is weak, and in the absence of microvascular injury or DSA, the finding is only suggestive.
How Is Acute Renal Allograft Rejection Treated?
Immunosuppression is crucial to prevent or mitigate damage from the recipient’s immune system. When rejection does occur, augmentation of immunosuppressive medications is the standard treatment.12 Depending on the severity of inflammation, the dosage of the drugs will be adjusted and as renal function returns, tapered. Antithymocyte globulin may be administered in severe or nonresponsive cases of T cell-mediated rejection. This has a potent effect of T-lymphocyte depletion with resulting decrease and eventual elimination of the inflammatory reaction.13
Treatment for antibody-mediated rejection, however, is not always as efficacious. While the primary goal is removal of cytotoxic donor-specific antibodies as well as the clonal B-cells that produce them, currently available treatment regimens have shown mixed results.10 In addition to steroid administration and augmentation of immunosuppression, plasmapheresis and intravenous immunoglobulin may also be given to sequester donor-specific antibodies. In cases of severe rejection, anti-CD20 medication such as rituximab may also be considered.
Diagnostic Findings, Part 3
The patient is admitted to the transplant service and is promptly started on treatment with intravenous steroids and mycophenolate mofetil (an immunosuppressive agent that selectively inhibits B- and T-cell proliferation), in addition to his usual tacrolimus dose. Antithymocyte globulin is also administered when creatinine level remains markedly increased. Renal function eventually recovers over a 1-week period with creatinine levels gradually decreasing and an increasing glomerular filtration rate. The patient is discharged on a tapering dose of oral steroids, daily mycophenolate mofetil, and his previous dose of tacrolimus. At a follow-up appointment 1 week later, he feels well and both the GFR and creatinine level have returned to the normal range.
Questions/Discussion Points, Part 3
What Should Be the Long-Term Management Plan for This Patient?
The primary goal of transplant care management is to maximize the longevity of allograft organ while minimizing possible treatment-related complications.14 To prevent recurrent acute rejection episodes and onset of chronic organ rejection, maintaining immunosuppressant medication levels within therapeutic ranges is paramount. Additionally, other than routine laboratory testing (Table 3 12,14), prevention of infection in these immunosuppressed patients is crucial. In this context, there is a window for opportunistic infections, leading to morbidity and in some cases mortality. Other important factors to consider as parts of long-term management include proper patient education, social support, and access to medication. Involving the patient, the family members, and even social services is vital to optimize the complex regimen and clinical outcome. As such, building a strong rapport between the patient and the transplant nephrology specialists and primary care physicians is essential to monitor renal function as well as to maintain an overall healthy lifestyle.
Table 3.
This Table Outlines a Set of Routine Laboratory Tests/Recommendations That Specialists or Primary care Physicians Should Monitor in Transplant Recipients.12,14,*
| Routine laboratory tests | |
|---|---|
| Renal allograft functions |
|
| Management of immunosuppression |
|
| Electrolytes/endocrine balance |
|
| Hematologic disorder |
|
| Screening/prophylaxis |
|
* Although a general clinical guideline was established by the 2009 Kidney Disease: Improving Global Outcomes (KDIGO), the types and the frequency of testing should be individualized and discussed with the patients, based on their health conditions.
†Including serum level of sodium, potassium, magnesium, calcium, and phosphates.
‡Including levels of vitamin D and parathyroid hormone.
§Control modifiable risk factor such as smoking, drinking, weight control, and dietary intake.
Teaching Points
Solid organ rejection in a kidney transplant recipient, while complex and multifactorial in nature, is a result of imbalance between host immune response to the allograft and immunosuppressive therapy.
Rejection must be differentiated from a host of other inflammatory processes that may affect the kidney, including infections, drug toxicity, and recurrent or de novo nonrejection renal disease.
Transplant rejection may be T lymphocyte-mediated or antibody-mediated. Each has distinctive histopathological findings, but both types of rejection may occur together, complicating biopsy interpretation.
T lymphocyte-mediated rejection is most commonly seen in the first 6 months after transplantation and has a well-elucidated pathophysiology.
Antibody-mediated rejection more commonly occurs later in the post-transplantation course, and our understanding of its pathophysiology is still evolving.
Although clinical findings are sensitive for detecting allograft dysfunction, kidney biopsy remains the gold standard in diagnosing organ rejection.
The Banff classification is a detailed schema for characterizing the nature and extent of kidney transplant rejection in order to guide treatment.
Regular follow-up and laboratory testing are essential for prolonging the longevity of the transplant kidney and maximizing the quality of life of transplant patients.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Kevin Kuan, MD  https://orcid.org/0000-0001-6818-2179
https://orcid.org/0000-0001-6818-2179
References
- 1. Knollmann-Ritschel BEC, Regula DP, Borowitz MJ, Conran R, Prystowsky MB. Pathology competencies for medical education and educational cases. Acad Pathol. 2017;4. doi:10.1177/2374289517715040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Goldberg RJ, Weng FL, Kandula P. Acute and chronic allograft dysfunction in kidney transplant recipients. Med Clin North Am. 2016;100:487–503. doi:10.1016/j.mcna.2016.01.002 [DOI] [PubMed] [Google Scholar]
- 3. Nadasdy T, Satoskar A, Nadasdy G. Pathology of renal transplantation. In: Zhou XJ, Laszik Z, Nadasdy T, D’Agati VD, Silva FG, eds. Silva’s Diagnostic Renal Pathology. Cambridge University Press; 2009:522–567. [Google Scholar]
- 4. Nankivell BJ, Alexander SI. Rejection of the kidney allograft. N Engl J Med. 2010;363:1451–1462. doi:10.1056/NEJMra0902927 [DOI] [PubMed] [Google Scholar]
- 5. Ingulli E. Mechanism of cellular rejection in transplantation. Pediatr Nephrol. 2010;25:61–74. doi:10.10007/s00467-008-1020-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Wang S, Zhang C, Wang J, et al. Endothelial cells in antibody-mediated rejection of kidney transplantation: pathogenesis, mechanisms, and therapeutic implications. J Immunol Res. 2017;2017:8746303. doi:10.1155/2017/8746303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lesage J, Noel R, Lapointe I, et al. Donor-specific antibodies, C4d and their relationship with the prognosis of transplant glomerulopathy. Transplantation. 2015;99:69–76. doi:10.1097/TP.0000000000000310 [DOI] [PubMed] [Google Scholar]
- 8. Djamali A, Kaufman DB, Ellis TM, Zhong W, Matas A, Samaniego M. Diagnosis and management of antibody-mediated rejection: current status and novel approaches. Am J Transplant. 2014;14:255–271. doi:10.1111/ajt.12589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Walker PD, Cavallo T, Bonsib SM; Ad Hoc Committee on Renal Biopsy Guidelines of the Renal Pathology Society. Practice guidelines for the renal biopsy. Mod Pathol. 2004;17:1555–1563. doi:10.1038/modpathol.3800239 [DOI] [PubMed] [Google Scholar]
- 10. Nickeleit V, Mengel M, Colvin RB. Renal transplant pathology. In: Jennette JC, Olson JL, Silva FG, D’Agati VD, eds. Heptinstall’s Pathology of the Kidney. Wolsters Kluwer. 2015:1321–1459. [Google Scholar]
- 11. Roufosse CR, Simmonds N, Clahsen-van Groningen M, et al. A 2018 Reference guide to the Banff classification of renal allograft pathology. Transplantation. 2018;102:1795–1814. doi:10.1097/TP.0000000000002366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kasiske BL, Zeier MG, Craig JC, et al. KDIGO clinical practice guideline for the care of kidney transplant recipients. Am J Transplant. 2009;9:1–155. doi:10.1111/j.1600-6143.2009.02834.x [DOI] [PubMed] [Google Scholar]
- 13. Thiyagarajan UM, Ponnuswamy A, Bagul A. Thymoglobulin and its use in renal transplantation: a review. Am J Nephrol. 2013;37:586–601. doi:10.1159/000351643 [DOI] [PubMed] [Google Scholar]
- 14. Chandraker A, Yeung MY. Overview of care of the adult kidney transplant recipient. UpToDate. Updated December 13, 2018. Accessed April 12, 2019. https://www.uptodate.com/contents/overview-of-care-of-the-adult-kidney-transplant-recipient.