Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords: pathology competencies, organ system pathology, skin, immune diseases of the skin, dermatitis herpetiformis, hypersensitivity, gluten hypersensitivity
Primary Objective
Objective SK3.2: Immune Diseases of the Skin. Describe the clinical features and pathologic basis for the following immunologically driven diseases with a genetic component: eczema, psoriasis, and vitiligo.
Competency 2: Organ System Pathology; Topic: Skin (SK): Learning Goal 3: Immune-Related Disorders of the Skin
Secondary Objectives
Objective IM1.4: Hypersensitivity. Compare and contrast the mechanisms of the 4 hypersensitivity reactions with respect to the situations in which each is triggered, mechanisms of injury, resulting pathologic effects on tissue, and the ultimate clinical consequences.
Competency 1: Disease Mechanisms and Processes; Topic: Immunological mechanisms (IM): Learning Goal 1: Immune Dysfunction
Objective SP1.3: Special Studies. After looking at slides of a tissue lesion or mass, the pathologist makes a diagnosis. List options for surgical and nonsurgical treatment and describe prognostic and therapy-guiding tests that may be performed on the tissue.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic: Surgical Pathology (SP): Learning Goal 1: Role in Diagnosis
Patient Presentation
A 21-year-old white man presents to his primary care provider with an itchy rash on the extensor surfaces of his arms and legs that has been present for 1 week. He has tried applying petroleum jelly to the rash with no relief. He has celiac disease, diagnosed at age 14, which he has been managing with a gluten-free diet, though admits to lately being less stringent about avoiding gluten. Additionally, he reports mild gastrointestinal distress, including bloating and pain. Of note, he had a similar rash on his knees a few years ago, which resolved without intervention. He has no notable family history and denies drug and alcohol use.
Diagnostic Findings, Part 1
On physical examination, multiple red papules and vesicles on the patient’s elbows and knees with areas of excoriation are noted (Figure 1 2).
Figure 1.
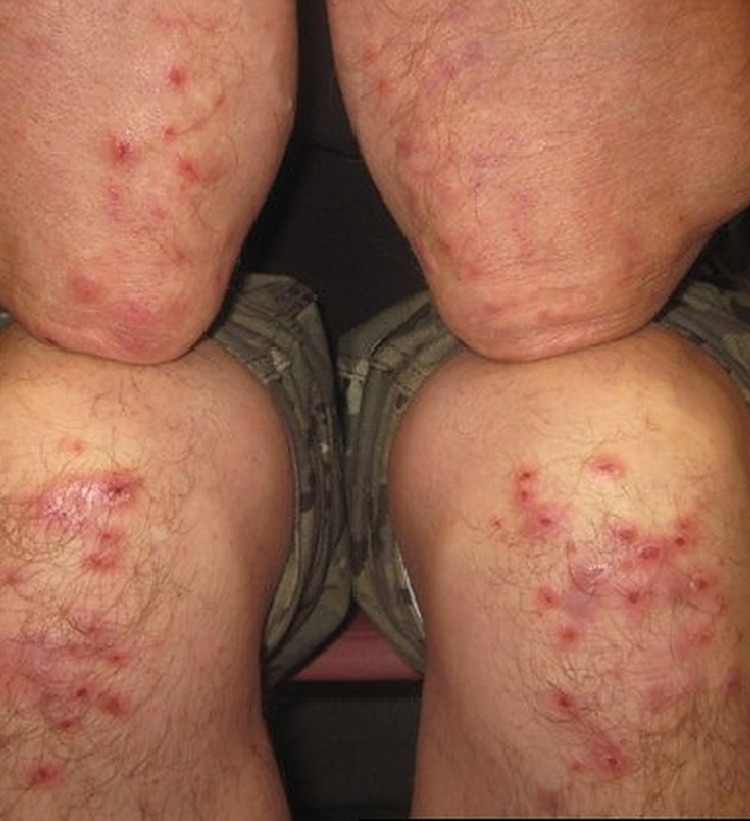
Typical clinical picture of dermatitis herpetiformis (DH) with excoriated blisters and papules on the elbows and knees. Source: Reproduced with permission from Acta Dermato-Venerealogica.2
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis at This Point?
The differential diagnosis for pruritic papules and excoriations includes dermatitis herpetiformis (DH) and common dermatologic disorders such as atopic dermatitis, scabies, and arthropod bite reactions.3
What Are the Next Steps?
The next step would be to obtain punch biopsies and direct immunofluorescence microscopy (DIF) of the lesion.
Diagnostic Findings, Part 2
Two 4 mm punch biopsies are obtained and sent for laboratory studies, including tissue pathology and DIF. Results are depicted in Figures 2, 3, and 4.
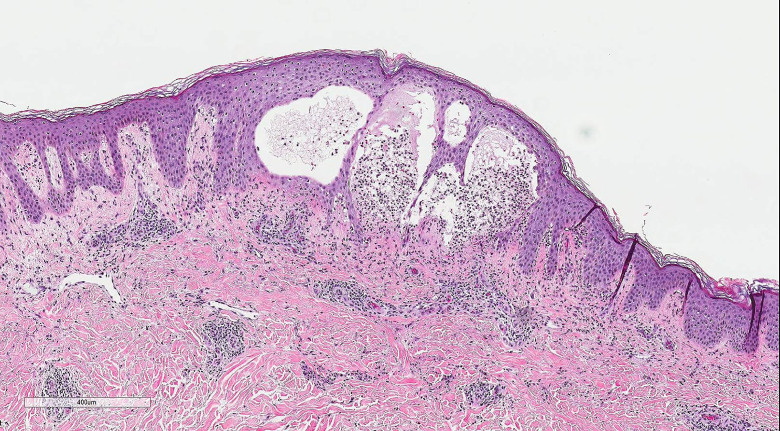
Figure 2.
Low-power field (×400) of hematoxylin and eosin (H&E) stain of the lesional biopsy. Subepidermal vesiculation and blistering with fibrin is seen as well as perivascular lymphocytes, histiocytes, and eosinophils.
Figure 3.
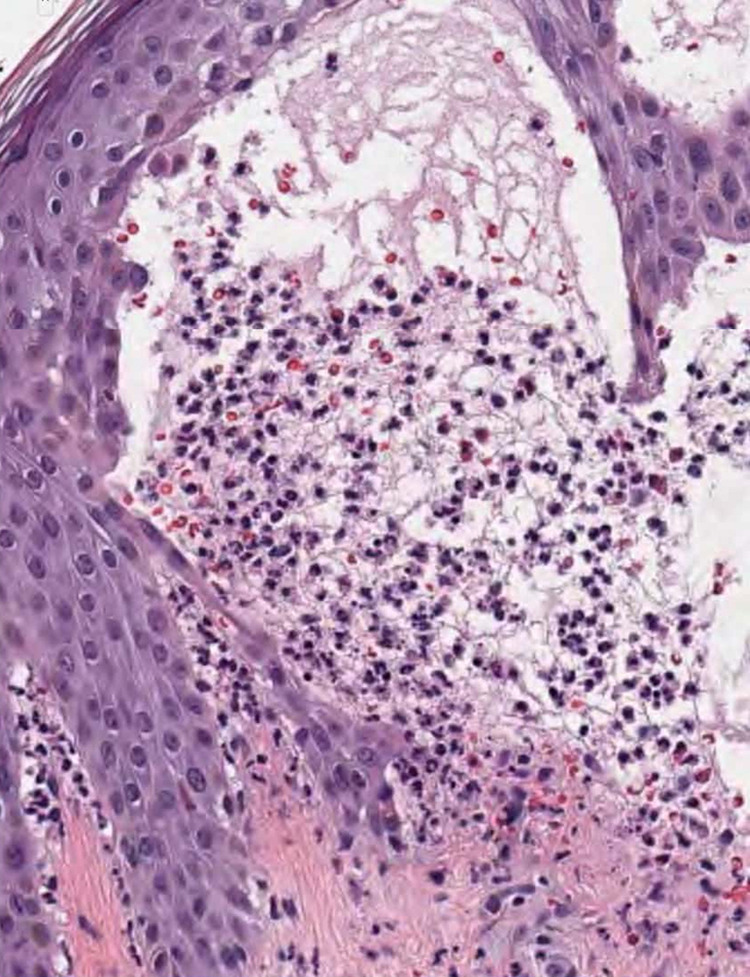
High-power field (×800) of H&E stain of the lesional biopsy. This view shows presence of numerous neutrophils and some eosinophils.
Figure 4.
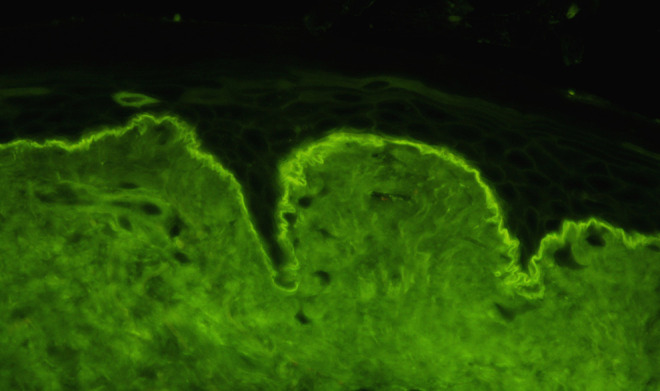
Immunoglobulin A (IgA) DIF showing granular deposits along the basement membrane that are more prominent at the tips of the dermal papillae (×400). DIF indicates direct immunofluorescence microscopy.
Questions/Discussion Points, Part 2
What Histological Findings Do You See in Figures 2, 3, and 4?
Under light microscopy (Figures 2 and 3), the biopsy shows subepidermal blisters with neutrophils, eosinophils, and fibrin. The deeper dermis shows a mixed chronic inflammatory infiltrate with perivascular lymphocytes, histiocytes, and eosinophils.
Direct immunofluorescence microscopy (Figure 4) shows granular deposits of immunoglobulin A (IgA) along the basement membrane. These deposits are more intensely fluorescent at the tips of the dermal papillae.
Now What Is the Differential Diagnosis?
The differential diagnosis of histology showing subepidermal blistering with an inflammatory infiltrate includes DH, bullous pemphigoid, linear IgA dermatitis, and bullous systemic lupus erythematosus (SLE).
Bullous pemphigoid
Bullous pemphigoid presents as large grouped and tense blisters, with distribution on the proximal limbs and inferior abdomen. The subepidermal blistering tends to show a more eosinophilic inflammatory infiltrate, and immunofluorescence shows linear IgG deposition at the basement membrane.
Linear immunoglobulin A dermatitis
Linear IgA dermatitis is usually distributed in the periorificial regions, and similarly to DH has IgA deposition on immunofluorescence. However, this IgA deposition appears in a linear pattern in the case of linear IgA dermatitis, rather than the granular pattern of DH.
Bullous systemic lupus erythematosus
Bullous SLE may look similar to DH histologically, and DIF may show linear or granular IgG, IgM, IgA, or C3 deposition at the basement membrane, which has some overlap with the granular IgA pattern seen with DH. In this case, it may be necessary to look at the broader clinical picture and additional serological testing in order to help distinguish the two.4
All of these conditions tend to include blistering more prominently than DH, with the exception of the prodromal phase of bullous pemphigoid, in which blistering may be minimal or absent.3
A thorough assessment of patient history, as well as the clinical, histopathologic, immunopathologic, and serologic findings can generally successfully distinguish these conditions from DH.
With All of This Information, What Do You Think the Diagnosis in This Case Is?
Based on the clinical presentation, microscopy findings, and with confirmatory DIF, a diagnosis of DH is made.
How Is Dermatitis Herpetiformis Diagnosed?
In addition to clinical assessment, laboratory studies such as tissue pathology, DIF, and serology can be helpful in diagnosis. Direct immunofluorescence microscopy is generally regarded as the most specific test for diagnosing DH.5
The first step toward confirmatory diagnosis of suspected DH is to obtain a lesional skin biopsy—ideally from an intact vesicle—for routine hematoxylin and eosin (H&E) staining, and a perilesional skin biopsy for DIF. Usually, 4 mm punch biopsies are sufficient for both specimens. It is important that the perilesional biopsy for DIF not be placed in formalin to maintain the integrity of the complexes.
When viewed microscopically with H&E staining, early lesions may show collections of neutrophils and fibrin at the tips of the dermal papillae, while lesions older than 48 hours generally show subepidermal vesiculation at the papillary tips, which may eventually coalesce to form larger subepidermal blisters.6
Because the histologic findings can resemble other subepithelial blistering disorders, DIF is an important adjunct test.
Characteristically, DIF testing will show granular deposits of IgA, and sometimes IgM and C3, within the dermal papillae. Less frequently, the IgA deposits will be fibrillar, rather than granular.7-9
When the diagnosis is still unclear, serologic testing can be useful for confirming DH. In serologic studies, DH often results in elevated levels of IgA tissue transglutaminase antibodies, IgA epidermal transglutaminase antibodies, and IgA endomysial antibodies.10,11
Additional testing that may be considered in an individual with DH would be thyroid function tests and diabetes screening, due to the association of DH with other autoimmune conditions including thyroid disease and type I diabetes.12
What Is the Pathogenesis of Dermatitis Herpetiformis and What Conditions Predispose Toward It?
The pathogenesis of DH is multifactorial. Primarily, DH is considered a result of gluten-sensitivity that is most likely to be seen in those individuals who are genetically predisposed, such as those with celiac disease.5 In fact, one study found that more than 90% of patients with DH exhibit small bowel biopsy findings which are consistent with gluten-sensitive enteropathy.13
The link between gluten sensitivity and DH is believed to be reliant on gliadin peptides and tissue transglutaminase. When a patient ingests gluten-containing food, gliadin gets absorbed into the intestinal mucosa, and deaminated by tissue transglutaminase. Deaminated gliadin peptides bind to HLA DQ2 and HLA DQ8 molecules on antigen presenting cells, where they are recognized by helper T cells, which produce pro-inflammatory cytokines and matrix metalloproteinases that promote damage to the gut mucosa and the production of antibodies against tissue transglutaminase by B cells.14
These IgA anti-epidermal transglutaminase antibodies travel throughout the bloodstream and can complex and cross-react with epidermal transglutaminase in the dermis of the skin, stimulating neutrophil chemotaxis and proteolytic cleavage within the lamina lucida, leading to blistering and formation of the characteristic dermal infiltrates seen on pathology.13
The human leukocyte antigen (HLA) genes that appear to predispose individuals to DH are the HLA DQ2 and HLA DQ8 haplotypes, which are present in virtually all DH patients.15 The significance of these 2 haplotypes is likely related to their role in evoking an immune response against gliadin peptides.15
Epidemiological risk factors which are believed to predispose individuals to DH include northern European heritage, being male, and being in the third to fifth decade of life.16
What Other Common Immunologically Driven Skin Diseases Contain a Genetic Component?
Three commonly seen immune diseases of the skin are eczema, psoriasis, and vitiligo.
Eczema, also known as atopic eczema, can occur in patients as young as 3 months of age. Eczema most commonly affects extremities, and often resolves by adulthood. Eczema is associated with other allergic conditions such as asthma, allergic rhinitis, and food allergies, as well as elevated IgE.
Pathologically, eczema is aggravated by disruption of the skin barrier, the primary defense of the innate immune system. Patients have a genetic propensity to be more reactive to antigens, causing an immune response and pruritis when their skin gets disrupted.
Psoriasis, seen in 1% to 3% of the population, appears as papulosquamous plaques that show sharply demarcated thick silvery scale, most commonly on the scalp, trunk, buttocks, elbows, and knees. Psoriasis has a strong genetic component, and is associated with increased inflammation.
Vitiligo is an autoimmune disease which causes depigmentation of the skin due to an autoimmune mediated loss of melanocytes, and is associated with autoimmune thyroid disease. Like eczema and psoriasis, vitiligo has a genetic component.17
What Type of Hypersensitivity Reaction Is Dermatitis Herpetiformis?
Dermatitis herpetiformis is a type III (immune complex) hypersensitivity reaction. It relies on the formation of immune complexes through binding of antibodies (IgG and IgM) to antigens. These complexes activate the complement cascade and induce neutrophils to secrete lysosomal enzymes. In DH, there is deposition of antigen-antibody complexes along the dermal-epidermal junction. The transglutaminase antibody and the antigen (epidermal transglutaminase) cross react and bind in the dermis. The deposition of this antigen-antibody complex stimulates inflammation and proteolysis, leading to the formation of the characteristic subepidermal blistering that is seen in DH.3
In contrast, type I hypersensitivity is immediate, and results from the antigen cross-linking with an IgE antibody that is bound to a mast cell or basophil. The binding of the antigen with the antibody results in immediate release of inflammatory cytokines, such as histamine, which leads to an allergic reaction that may be localized (allergic dermatitis) or systemic (anaphylaxis).18
Type II hypersensitivity is an antibody-mediated cytotoxic reaction in which an antibody (IgG or IgM) reacts directly with an antigen, which may be a self-antigen or an extrinsic antigen. This leads to complement activation and cell lysis. Examples of type II hypersensitivity include autoimmune diseases such as pemphigus vulgaris, bullous pemphigoid, and Goodpasture’s syndrome.18
Type IV hypersensitivity is a cell-mediated delayed reaction. This reaction is initiated by the interaction of an antigen with T-lymphocytes. T-lymphocytes which have previously been sensitized will produce cytokines. This process typically occurs over 48 to 72 hours. Examples of type IV hypersensitivity include atopic dermatitis, granulomatosis with polyangiitis, and hypersensitivity pneumonitis.18
How Is Dermatitis Herpetiformis Treated?
The standard treatment for DH includes both dapsone therapy and a strict gluten-free diet. A gluten-free diet may require several months to a few years to achieve complete remission as a monotherapy, while dapsone typically resolves active skin lesions within days.19 For this reason, the preferred approach is a combination of the 2 therapies, with dapsone being prescribed to promote rapid improvement, and a strict gluten-free diet being strongly encouraged for long-term remission.3,5 Once symptoms have subsided, dapsone therapy can be slowly tapered.
Even at optimum dosing, mild eruptions of 1 to 2 new lesions per week are common and, do not indicate a need to increase dosage but can be treated with a topical corticosteroid.3
There are several potential adverse effects associated with dapsone, most notably hemolysis. Although most patients tolerate dapsone therapy well, those with glucose-6-phosphate dehydrogenase (G6PD) deficiency are at an increased risk for severe hemolytic anemia. Additional side effects can include methemoglobinemia, agranulocytosis, or dapsone hypersensitivity reactions, which typically present with flu-like symptoms, a morbilliform cutaneous eruption, fever, lymphadenopathy, hepatitis, and eosinophilia.
While undergoing dapsone therapy, laboratory testing is advised, including a complete blood count, liver function tests, renal function tests, and a G6PD test prior to starting therapy.20
While dapsone therapy is generally effective, a strict gluten-free diet is the preferred long-term therapy.21 To aid in adherence, counseling with a dietician is recommended for patients.
What Is the Prognosis for Dermatitis Herpetiformis?
Dermatitis herpetiformis is commonly a life-long condition. A strict gluten-free diet can be observed in order to maintain remission, with dapsone therapy used as needed. Symptoms of DH can recur within 2 days after discontinuing dapsone therapy, and within 3 months of resuming the eating of gluten-containing foods. A small number, 10% to 15%, of patients may achieve complete remission that persists even after discontinuing both therapies.21
Teaching Points
Type 1 hypersensitivity is IgE mediated and involves mast cells and basophils which, when activated, lead to immediate degranulation and release of inflammatory cytokines. This release causes the characteristic immediate allergic response.
Type 2 hypersensitivity is antibody (IgG or IgM) mediated. In type 2 hypersensitivity, the antibody binds either a self or extrinsic antigen, activating the complement cascade and resulting in cell lysis.
Type 3 hypersensitivity is antibody-complex mediated. It relies on antigen-antibody complex formation and deposition. These complexes stimulate the complement cascade and inflammation by stimulating neutrophils to secrete lysosomal enzymes.
In type 4 hypersensitivity, the cell-mediated delayed reaction depends on the interaction of antigens with T-lymphocytes which have previously been sensitized to the antigen. The previously sensitized T-lymphocytes begin secreting the cytokines that produce the characteristic allergic response within 48 to 72 hours.
Dermatitis herpetiformis (DH) is a type III (immune complex) hypersensitivity reaction occurring as a result of gluten sensitivity. Dermatitis herpetiformis is commonly accompanied by celiac disease.
In addition to gluten sensitivity, genetic factors are important in the pathogenesis of DH, as essentially all patients with DH have the HLA DQ2 or HLA DQ8 haplotype.
Epidemiological risk factors which are believed to predispose individuals to DH include being of northern European heritage, male, and in the third to fifth decade of life. The primary symptom of DH is intensely pruritic papulovesicular eruption on extensor surfaces, commonly on the elbows, forearms, knees, buttocks, back, and scalp. Due to the pruritus, excoriations and erosions are often prominent.
Diagnosis of DH requires clinical assessment of symptoms, pathologic examination of biopsy specimens including tissue pathology and direct immunofluorescence microscopy, and if needed, confirmatory serology testing. The characteristic finding in tissue histology is subepidermal blistering with neutrophils in the dermal papillae, and the characteristic finding in DIF is granular deposits of IgA within the papillary dermis.
The primary treatment recommended for DH includes both dapsone therapy, for immediate resolution of symptoms, and a strict gluten-free diet (with dapsone slowly tapered) for long-term remission.
Dermatitis herpetiformis commonly affects patients throughout their lives and requires continued treatment, ideally with a gluten-free diet, to maintain remission.
Eczema, psoriasis, and vitiligo are other examples of common immune diseases of the skin with a genetic component.
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Janina Markidan  https://orcid.org/0000-0003-4564-5630
https://orcid.org/0000-0003-4564-5630
References
- 1. Knollmann-Ritschel BEC, Regula DP, Borowitz MJ, Conran R, Prystowsky MB. Pathology competencies for medical education and educational cases. Acad Pathol. 2017;4. doi:10.1177/2374289517715040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Salmi T, Hervonen K. Current concepts of dermatitis herpetiformis. Acta Derm Venereol. 2020;100:115–121. doi:10.2340/00015555-3401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Caproni M, Antiga E, Melani L, Fabbri P; Italian Group for Cutaneous Immunopathology. Guidelines for the diagnosis and treatment of dermatitis herpetiformis. J Eur Acad Dermatol Venereol. 2009;23:633–638. doi:10.1111/j.1468-3083.2009.03188.x [DOI] [PubMed] [Google Scholar]
- 4. Vodegel RM, Jonkman MF, Pas HH, de Jong MC. U-serrated immunodeposition pattern differentiates type VII collagen targeting bullous diseases from other subepidermal bullous autoimmune diseases. Br J Dermatol. 2004;151:112–118. doi:10.1111/j.1365-2133.2004.06006.x [DOI] [PubMed] [Google Scholar]
- 5. Antiga E, Caproni M. The diagnosis and treatment of dermatitis herpetiformis. Clin Cosmet Investig Dermatol. 2015:8:257–265. doi:10.2147/CCID.S69127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Clarindo MV, Possebon AT, Soligo EM, Uyeda H, Ruaro RT, Empinotti JC. Dermatitis herpetiformis: pathophysiology, clinical presentation, diagnosis and treatment. An Bras Dermatol. 2014;89:865–877. doi:10.1590/abd1806-4841.20142966 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Salmi TT. Dermatitis herpetiformis. Clin Exp Dermatol. 2019;10:728–731. doi:10.1111/ced.13992 [DOI] [PubMed] [Google Scholar]
- 8. Patterson JW. Chapter 6: The vesiculobullous reaction pattern. In: Weedon’s Skin Pathology. 4th ed. Elsevier; 2015. [Google Scholar]
- 9. Ko CJ, Colegio OR, Moss JE, McNiff JM. Fibrillar IgA deposition in dermatitis herpetiformis—an underreported pattern with potential clinical significance. J Cutan Pathol. 2010;37:475–477. doi:10.1111/j.1600-0560.2009.01472.x [DOI] [PubMed] [Google Scholar]
- 10. Rose C, Armbruster FP, Ruppert J, Igl BW, Zillikens D, Shimanovich I. Autoantibodies against epidermal transglutaminase are a sensitive diagnostic marker in patients with dermatitis herpetiformis on a normal or gluten-free diet. J Am Acad Dermatol. 2009;61:39–43. doi:10.1016/j.jaad.2008.12.037 [DOI] [PubMed] [Google Scholar]
- 11. Dieterich W, Laag E, Bruckner-Tuderman L, et al. Antibodies to tissue transglutaminase as serologic markers in patients with dermatitis herpetiformis. J Invest Dermatol. 1999;113:133–136. doi:10.1046/j.1523-1747.1999.00627.x [DOI] [PubMed] [Google Scholar]
- 12. Bolotin D, Petronic-Rosic V. Dermatitis herpetiformis. J Am Acad Dermatol. 2011;64:1017–1033. doi:10.1016/j.jaad.2010.09.777 [DOI] [PubMed] [Google Scholar]
- 13. Bonciolini V, Bonciani D, Verdelli A, et al. Newly described clinical and immunopathological feature of dermatitis herpetiformis. Clin Dev Immunol. 2012;2012:967974. doi:10.1155/2012/967974 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Herrero-González JE. Clinical guidelines for the diagnosis and treatment of dermatitis herpetiformis [in Spanish]. Actas Dermosifiliogr. 2010;101:820–826. doi:10.1016/S1578-2190(10)70729-0 [PubMed] [Google Scholar]
- 15. Balas A, Vicario JL, Zambrano A, Acuña D, García-Novo D. Absolute linkage of celiac disease and dermatitis herpetiformis to HLA-DQ. Tissue Antigens. 1997;50:52–56. doi:10.1111/j.1399-0039.1997.tb02834.x [DOI] [PubMed] [Google Scholar]
- 16. Collin P, Salmi T, Hervonen K, Kaukinen K, Reunala T. Dermatitis herpetiformis: a cutaneous manifestation of coeliac disease. Ann Med. 2017;49:23–31. doi:10.1080/07853890.2016.1222450 [DOI] [PubMed] [Google Scholar]
- 17. Rapini R. Chapter 2: Eczematous and papulosquamous diseases. In: Practical Dermatopathology. 2nd ed. Elsevier; 2012. [Google Scholar]
- 18. Weiss RB. Hypersensitivity reactions. Semin Oncol. 1992;19:458–477. [PubMed] [Google Scholar]
- 19. Vale ECSD, Dimatos OC, Porro AM, Santi CG. Consensus on the treatment of autoimmune bullous dermatoses: dermatitis herpetiformis and linear IgA bullous dermatosis. An Bras Dermatol. 2019;94:48–55. doi:10.1590/abd1806-4841.2019940208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wozel G, Blasum C. Dapsone in dermatology and beyond. Arch Dermatol Res. 2014;306:103–124. doi:10.1007/s00403-013-1409-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Collin P, Reunala T. Recognition and management of the cutaneous manifestations of celiac disease: a guide for dermatologists. Am J Clin Dermatol. 2003;4:13–20. doi:10.2165/00128071-200304010-00002 [DOI] [PubMed] [Google Scholar]