Abstract
Background
Due to its prognostic importance for patients with type 2 diabetes (DM2), current guidelines recommend a systolic <130 mm Hg and diastolic <80 mm Hg blood pressure target. Periodic breathing, a form of sleep-disordered breathing, acutely causes repetitive hypoxia, sympathetic nervous system activation as well as oscillations of heart rate and blood pressure. However, limited data on the association of periodic breathing and control of blood pressure (BP) in patients with DM2 are available. Thus, the aim of the present study was to assess whether there is an association between periodic breathing and increased BP above the recommended target in DM2.
Methods
Cross-sectional data of 679 patients with DM2 from the DIACORE-SDB sub-study were analysed for association of periodic breathing with BP. Sleep-disordered breathing was assessed with a 2-channel ambulatory monitoring device including validated automatic pattern recognition for periodic breathing. BP values were determined in a standardized manner with three repeated measurements at rest.
Results
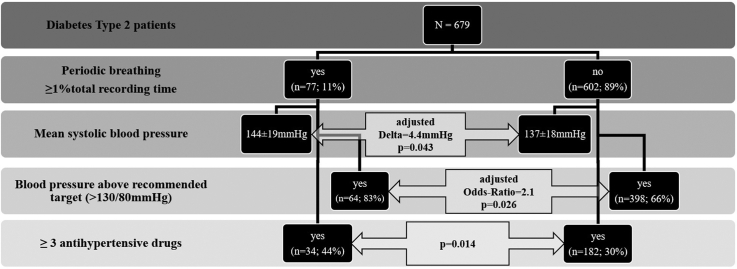
Of the 679 analysed individuals (61% male, age 66 ± 9 years, Body Mass Index [BMI] 31.0 ± 5.4 kg/m2), 11% had periodic breathing. Patients with periodic breathing had significantly higher systolic BP values (144 ± 19 mm Hg vs. 137 ± 18 mm Hg, p = 0.003). Multivariable regression analysis revealed that periodic breathing was associated with higher systolic BP (B [95% confidence interval, CI] = 4.4 [0.1; 8.7], p = 0.043) and not meeting the recommended BP target for patients with diabetes (<130/80 mmHg) (odds ratio, OR [95%CI] = 2.1 [1.1; 4.0], p = 0.026) independent of sex, age, high density lipoproteins, renal function, coronary heart disease and antihypertensive treatment.
Conclusion
Periodic breathing is associated with higher systolic BP in patients with DM2.
Keywords: Sleep-disordered breathing, Periodic breathing, Cheyne-stokes-respiration, Systolic blood pressure, Arterial hypertension, Type 2 diabetes
Graphical abstract
Highlights
-
•
periodic and sleep disordered breathing are associated with blood pressure values above blood pressure target for diabetes.
-
•
elevated blood pressure is highly prevalent in diabetes and periodic breathing despite extensive use of antihypertensives.
-
•
periodic breathing is associated with higher systolic blood pressure independently from other risk factors.
1. Introduction
Arterial hypertension is a very common disease which is strongly associated with type 2 diabetes (DM2) [1,2]. Effective blood pressure (BP) control in DM2 can significantly reduce the risk of cardiovascular events [3]. The same systolic BP reduction translates into a higher risk reduction for cardiovascular events in patients with DM2 vs. those without (adjusted relative hazard 0.31 vs. 0.74) [4]. Therefore, recommendations for BP control are stricter in patients with DM2 compared to those without [5]. Current guidelines from the European Society of Cardiology and the European Society of Hypertension recommend a BP target of systolic <130 mm Hg and diastolic <80 mm Hg in patients with DM2 [5].
Periodic breathing is an abnormal breathing pattern of central origin with waxing and waning of the airflow and the breathing effort [[6], [7], [8], [9]]. The term “Cheyne-Stokes-respiration (CSR)” has been used to describe periodic breathing with apneas in patients with heart failure or stroke [9]. In such patients, periodic breathing is a marker of disease severity and progression [9]. Periodic breathing is very common in patients with heart failure as it leads to a disturbed breathing regulation with a desynchronization of heart, lung and brain [10]: carbon dioxide (CO2) overload alters apnea thresholds with increased sensitivity for central apnea with decreasing CO2. Subsequently, rising CO2 leads to inappropriate hyperventilation caused by pulmonary congestion, enhanced central and peripheral chemoreceptor sensitivity and prolonged circulation time [10,11]. This oscillatory pattern of waxing and waning occurs in the absence of significant upper airway obstruction [11]. Periodic breathing is associated with sleep fragmentation, intermittent hypoxemia and sympathetic nervous system activation as well as oscillations of heart rate and BP [10,12,13].
The periodic fluctuation of CO2 partial pressure [14] in periodic breathing can lead to arousal-triggered [15] nocturnal BP and heart rate peaks and oscillations, which can be abolished by CO2 but not by oxygen equilibration [12]. No such studies exist in patients with DM2. While these studies evaluated the acute effects of periodic breathing on BP, data on the association of periodic breathing and BP control in a patient population with DM2 that allows accounting for potential confounders are limited. Therefore, the aim of the present study was to assess, whether there is an association between periodic breathing and increased BP above the recommended target in DM2 patients.
2. Patients and methods
2.1. Study design
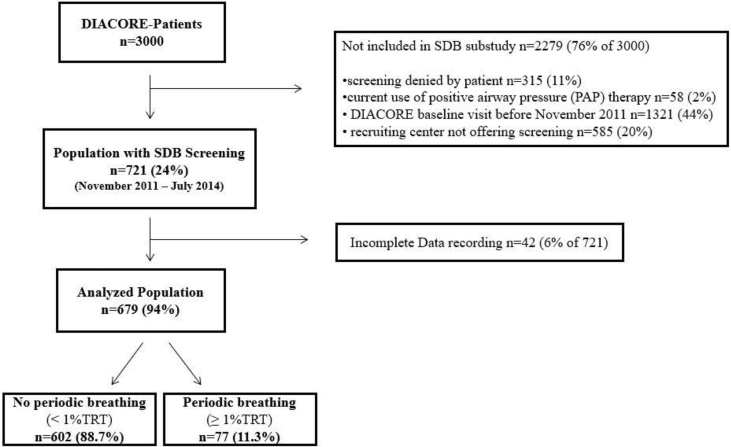
The investigated patients were participants of the DIACORE (DIAbetes COhoRtE)-SDB (sleep disordered breathing) sub-study [16]. DIACORE has been designed as a prospective two-centre study of DM2 patients of European descent. The baseline survey analysed here was conducted between 2010 and 2014 as described previously [16,17]. Briefly, written invitations were mailed to outpatients with DM2 by five medical insurance companies and by major diabetologists. Invitations were also sent to patients who had previously received inpatient treatment at the Departments of Internal Medicine of the University Medical Centre Regensburg. The diabetes status was ascertained by assessing diabetes medication intake or by validating self-report. Patients filled in a standardized online questionnaire and underwent blood sampling and a physical examination at either of the two study centres [16]. Of the 3000 DIACORE participants recruited at baseline, all who came to the Regensburg study centre between November 2011 and December 2014 were invited to participate in the DIACORE-SDB sub-study. Monitoring for SDB was not conducted in the study centre in Mannheim. Of the 1036 individuals invited to participate in the DIACORE-SDB sub-study [17,18], 721 agreed and were tested with a two-channel ambulatory SDB-monitoring device (ApneaLink, ResMed, 70%). Complete SDB parameters were recorded for 679 patients (94% of the 721 tested participants) [17,18]. The protocol, the data protection strategy, and the study procedures were approved by the Ethics Committees of the participating institutions and were in accordance with the Declaration of Helsinki. Patients participated in the DIACORE study only after providing informed written consent.
2.2. Study population
For DIACORE, all DM2 outpatients living in the cities and counties of Regensburg or Speyer were eligible [16]. Further inclusion criteria were as follows: fully understanding the study information and to give written informed consent, age ≥18 years, and self-reported Caucasian ethnicity [16]. Exclusion criteria were chronic renal replacement therapy (hemodialysis, peritoneal dialysis, or transplantation), history of active malignancy within the past five years, presence of autoimmune-disease potentially affecting kidney function, hemochromatosis, known pancreoprivic or self-reported type 1 diabetes, acute infection, fever, pregnancy, chronic viral hepatitis, or human immunodeficiency virus (HIV) [16]. For the DIACORE-SDB sub-study, patients were included if they were from the Regensburg recruitment region and consented to SDB screening; patients were excluded if they currently used continuous positive airway pressure (CPAP) therapy [[16], [17], [18]]. The participants were subjected to a standardized online survey, asked for their medical history (eg, smoking behaviour, time of onset of DM2), and former operations, interventions, or hospitalizations.
2.3. Assessment of coronary artery disease and antihypertensive treatment
Coronary heart disease (defined as coronary intervention, coronary bypass surgery or myocardial infarction) was assessed by self-report and validated from medical records and direct contact with local physicians. Drug intake was obtained via medical reports and self-report. Antihypertensive treatment was defined as intake of at least one agent out of diuretics, beta blockers, calcium antagonists, ACE inhibitors, angiotensin-receptor inhibitors or renin inhibitors.
2.4. Assessment of blood pressure and heart rate
BP was assessed using a GE Dinamap vital signs monitor, model V100 (CareScape, Germany). The BP cuff was chosen according to the patient's upper arm circumference (either 23–33 cm or 31–40 cm) at heart level on the dominant arm [16]. Patients had to rest for at least 5 min, before three measurements of heart rate and BP were conducted every 2 min. The third measurement of heart rate and the mean value of the second and the third BP measurement were used for analyses. In the case of systolic BP greater than 180 mm Hg/less than 90 mm Hg or diastolic BP greater than 110 mm Hg/less than 50 mm Hg, and heart rate greater than 90/min or less than 50/min, this was reported to the patient at the end of detection [16]. For DM2 patients, current European Society of Cardiology (ESC)/European Society of Hypertension (ESH)-Guidelines recommend a BP target of systolic <130 mm Hg and diastolic <80 mm Hg [5].
2.5. Assessment of sleep disordered breathing
Nasal flow and pulse oximetry were measured with the ApneaLink device (ResMed, Australia, Sydney) that has been validated in several studies on monitoring SDB as described previously [[19], [20], [21]]. Trained study personnel instructed participants in the use of the device in a standardized fashion [16]. Comparing ApneaLink to the gold standard polysomnography, other studies have yielded a sensitivity rate of 73–94% and a specificity rate of 85–95% using an apnea-hypopnea-index (apneas + hypopneas/hour: AHI) cut-off value of 15/h [19,22]. AHI, oxygen desaturation index, mean oxygen saturation, and minimum SpO2, time with oxygen saturation less than 90% (time SaO2<90%) were documented. The default settings of the screening device were used for defining apnea, hypopnea, and oxygen desaturation: apnea was defined as a ≥80% decrease in airflow for ≥10 s and hypopnea as a decrease in airflow by 50–80% versus baseline for ≥10 s followed by a ≥4% decrease in oxygen saturation. No or mild SDB was defined as AHI <15/h (‘no or mild SDB group’) and clinically relevant SDB as AHI ≥15/h (‘SDB group’) [17,18].
2.6. Assessment of periodic breathing
Periodic breathing was detected by automatic pattern recognition at a sensitivity rate of 75–99% and a specificity rate of 88–100%, compared to gold standard polysomnography [23]. Pattern recognition was based on a classifier algorithm using the features cycle length (including apnea-hypopnea- and hyperpnea-length), shape of hyperpnea (waxing/waning) and the jump feature (moderate flow increase in periodic breathing vs. sudden in obstructive sleep apnea) [23]. For a high probability score, periodic breathing was assumed in epochs of 30 min and displayed in % of total recording time (TRT) [23]. Present periodic breathing was defined as ≥1%TRT and absent as <1%TRT, as median was 0%.
2.7. Statistical analysis
We analyzed the association of periodic breathing with systolic BP/diastolic BP using a two-sided Student's t-test and with the currently recommended BP target of 130/80 mmHg for patients with diabetes using a χ2 test. To investigate whether this association would hold when adjusting for known risk factors for arterial hypertension (which were significantly different in groups of periodic breathing) multivariable linear regression analysis was applied to evaluate the association of periodic breathing with systolic BP and logistic regression analysis for the association for periodic breathing with the currently recommended BP target of 130/80 mm Hg for patients with diabetes. The regression analyses were adjusted for sex, age, high density lipoproteins, renal function, coronary heart disease and antihypertensive treatment. P-values <0.05 were considered as statistically significant. The data was analysed using the commercial statistics program IBM® SPSS® Statistics Version 23.0 (IBM Corp., Armonk, USA).
3. Results
3.1. Patient characteristics
Of the 721 patients with DM2 tested for SDB, 42 (6%) had incomplete or no records of SDB parameters due to technical problems, such as loss of the oximeter during sleep or failure to switch the device on at night. Thus, 679 patients entered the analysis (Fig. 1) as reported previously [17,18]. This patient group exhibited rather elderly patients, mostly male gender, visceral obesity and dyslipidemia. More than the half were current or former smokers and about one fourth suffered from coronary heart disease (Table 1) [17,18].
Fig. 1.
Table 1.
| Characteristic | Distribution |
|---|---|
| Age [years] | 65.6 ± 8.8 |
| Sex (male), n (%) | 412 (61) |
| Body-mass index [kg/m2] | 31.0 ± 5.4 |
| Waist-hip ratio | 0.96 ± 0.08 |
| Estimated glomerular filtration rate [ml/min/1.73 m2] | 78.1 ± 19.3 |
| Low density lipoproteins [mg/dl] | 117.6 ± 35.7 |
| High density lipoproteins [mg/dl] | 52.8 ± 14.9 |
| Haemoglobin A1c [%] | 6.6 [6.1; 7.2] |
| Diabetes duration [years] | 7.6 [3.9; 13.5] |
| Current smoker, n (%) | 68 (11) |
| Former smoker, n (%) | 310 (47) |
| Coronary heart disease, n (%) | 149 (22) |
| Periodic breathing (≥1%TRT), n (%) | 77 (11) |
| Sleep disordered breathing (AHI ≥ 15/h), n (%) | 228 (34) |
Results are provided as n (%), mean ± standard deviation or median [25th percentile; 75th percentile]. TRT: total recording time, AHI: apnea-hypopnea index.
3.2. Risk factors for systemic hypertension according to the presence of periodic breathing and sleep disordered breathing (SDB)
Of the 679 analyzed patients, 77 (11.3%) had periodic breathing. These showed an absolute time of periodic breathing ranging from 3 to 334 min (2–72 %TRT) with a median of 31 min (7%TRT). They were predominantly male, of higher age, higher waist-hip ratio (WHR) and had lower high-density lipoprotein (HDL). They had a lower estimated glomerular filtration rate (eGFR) and suffered more often from coronary heart disease (Table 2).
Table 2.
Risk factors for systemic hypertension according to periodic breathing category.
| Characteristic | Periodic breathing | No Periodic breathing | p-value |
|---|---|---|---|
| n (%) | 77 (11) | 602 (89) | |
| Age [years] | 68.9 ± 7 | 65.0 ± 9 | <0.001b |
| Sex (male), n (%) | 60 (78) | 352 (59) | 0.001a |
| Body mass index [kg/m2] | 31.0 ± 5 | 31.0 ± 5 | 0.989b |
| Waist-hip ratio | 0.98 ± 0.08 | 0.96 ± 0.08 | 0.039b |
| Estimated glomerular filtration rate [ml/min/1.73 m2] | 67.8 ± 20 | 79.2 ± 19 | <0.001b |
| Low density lipoproteins [mg/dl] | 115 ± 34 | 118 ± 36 | 0.551b |
| High density lipoproteins [mg/dl] | 49 ± 14 | 53 ± 15 | 0.006b |
| Haemoglobin A1c [%] | 6.5 [6.1; 7.2] | 6.6 [6.1; 7.2] | 0.543c |
| Diabetes duration [years] | 6.7 [4.2; 15.3] | 7.8 [3.9; 13.2] | 0.884c |
| Current smoker, n (%) | 6 (8) | 62 (10) | 0.490a |
| Former smoker, n (%) | 39 (52) | 271 (46) | 0.361a |
| Coronary heart disease, n (%) | 26 (34) | 123 (21) | 0.008a |
Results are provided as n (%), mean ± standard deviation or median [25th percentile; 75th percentile].
χ2 test.
Two-sided Student's t-test.
Mann-Whitney U rank sum test.
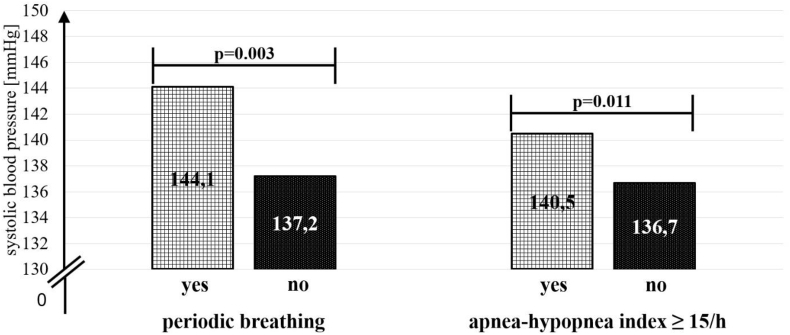
No or mild SDB (defined as AHI < 15/h) was found in 451 patients (66.4%), and 228 (33.6%) had SDB (AHI ≥ 15/h) [17,18]. Patients with SDB had higher systolic BP (140.5 ± 18.2 mmHg vs. 136.7 ± 18.1 mmHg, p = 0.011) (Fig. 2), whereas no significant differences in diastolic BP could be found (p = 0.178). Patients with SDB were predominantly more obese men and were older, had a higher BMI with higher WHR, and a longer history of DM2 (eTable 1 and eTable 2, online supplement).
Fig. 2.
Bar charts indicating the systolic blood pressure between groups with and without periodic breathing as well as with and without sleep-disordered breathing.
3.3. Association of periodic breathing and blood pressure
Of the 679 analyzed patients, 462 (68%) did not meet the currently recommended BP target of 130/80 mm Hg for patients with diabetes [5]. The 679 patients had a mean systolic BP of 138 ± 18.2 mm Hg and a mean diastolic BP of 74.6 ± 10.1 mm Hg. Patients with periodic breathing did more often not meet the currently recommended BP for patients with diabetes than those without periodic breathing (p = 0.003), although they were more frequently treated with antihypertensive drugs (Table 3). Patients with periodic breathing had higher systolic BP than those without (p = 0.002, Table 3, Fig. 2). No significant differences in diastolic BP could be found (p = 0.189, Table 3). Effective antihypertensive treatment seemed to be more challenging in patients with periodic breathing as a higher percentage used ≥3 antihypertensive drugs or BP was above the currently recommended BP target for patients with diabetes. Accordingly, they were more frequently treated with beta-blockers and had a lower heart rate (Table 3).
Table 3.
Blood pressure, heart rate and antihypertensive treatment according to periodic breathing category.
| Characteristic | Periodic breathing | No Periodic breathing | p-value |
|---|---|---|---|
| n (%) | 77 (11) | 602 (89) | |
| Blood pressure above target (systolic ≥130 or diastolic ≥80 mmHg), n (%)a | 64 (83) | 398 (66) | 0.003b |
| Systolic blood pressure [mmHg] | 144.1 ± 18.9 | 137.2 ± 18.0 | 0.002c |
| Diastolic blood pressure [mmHg] | 76.0 ± 11.8 | 74.4 ± 9.9 | 0.189c |
| Mean heart rate [/min] | 62.3 ± 10.1 | 66.5 ± 9.3 | <0.001c |
| Antihypertensive treatment, n (%) | 71 (92) | 475 (79) | 0.006b |
| Beta-blocker treatment, n (%) | 52 (68) | 268 (45) | <0.001b |
| ≥ 3 antihypertensive drugs, n (%) | 34 (44) | 182 (30) | 0.014b |
Results are provided as n (%) or mean ± standard deviation.
According to ESC/ESH-Guideline 2018 [5].
χ2 test.
Two-sided Student's t-test.
Periodic breathing was strongly associated with BP above the currently recommended BP target for patients with diabetes (p = 0.003) and higher systolic BP values (p = 0.002) (Table 3).
Since age, sex, WHR, HDL, eGFR, coronary heart disease, and antihypertensive treatment differ significantly between periodic breathing groups, we analyzed the association of periodic breathing with systolic BP with linear regression analysis and the association of periodic breathing with BP above the currently recommended BP target for patients with diabetes with logistic regression analysis. Age, sex, WHR and antihypertensive treatment were significantly associated with BP above the currently recommended BP target for patients with diabetes and with systolic BP in univariable regression analysis (eTable 3, online supplement). The association between periodic breathing with BP above the currently recommended BP target for patients with diabetes (OR [95%CI] 2.1 [1.1; 4.0], p = 0.026) and systolic BP (B [95%CI] 4.4 [0.1; 8.7] mmHg, p = 0.043) was independent of other risk factors in multivariable regression analysis models, respectively (Table 4).
Table 4.
Association between periodic breathing and blood pressure in dependence of other risk factors.
| Variable | Multivariable Analysis |
|||||
|---|---|---|---|---|---|---|
| Blood pressure above targeta |
Systolic blood pressure |
Diastolic blood pressure |
||||
| OR (95% CI) | p-value | B (95% CI) | p-value | B (95% CI) | p-value | |
| Periodic breathing (≥1%TRT) | 2.1 (1.1; 4.0) | 0.026 | 4.4 (0.1; 8.7) | 0.043 | 2.2 (−0.1; 4.5) | 0.063 |
| Age [/years] | 1.02 (1.00; 1.05) | 0.041 | 0.5 (0.3; 0.6) | <0.001 | −0.1 (−0.2; −0.03) | 0.011 |
| male Sex | 1.3 (0.9; 2.1) | 0.182 | −0.5 (−4.0; 3.0) | 0.768 | 3.9 (2.0; 5.7) | <0.001 |
| Waist-hip ratio [/0.1] | 1.4 (1; 1.8) | 0.030 | 3.6 (1.4; 5.8) | 0.001 | 2.3 (1.1; 3.5) | <0.001 |
| High density lipoproteins [/10 mg/dl] | 1.1 (0.9; 1.2) | 0.255 | 0.4 (−0.6; 1.3) | 0.481 | 0.5 (−0.04; 1.0) | 0.069 |
| Estimated glomerular filtration rate [/10 ml/min/1.73 m2] | 1.0 (0.9; 1.1) | 0.565 | 0.3 (−0.5; 1.1) | 0.509 | 0.6 (0.1; 1.0) | 0.012 |
| Coronary heart disease | 0.7 (0.5; 1.1) | 0.145 | −2.7 (−6.1; 0.7) | 0.124 | −2.7 (−4,5; −0,8) | 0.005 |
| Antihypertensive treatment | 1.9 (1.3; 3.0) | 0.003 | 5.3 (1.7; 8.8) | 0.004 | 0.7 (−1.2; 2.6) | 0.481 |
Multivariable regression models of periodic breathing and BP above currently recommended BP target for patients with diabetes (logistic)/systolic and diastolic BP (linear), respectively. B = linear regression coefficient. 95% CI = 95% confidence interval. OR = Odds-Ratio. TRT: total recording time.
Systolic ≥130 mm Hg or diastolic ≥80 mm Hg according to ESC/ESH-Guideline 2018 [5].
In the same multivariable regression analysis model using the binary exposure variable SDB instead of periodic breathing, SDB was not independently associated with BP above the currently recommended BP target for patients with diabetes (OR [95%CI] 1.2 [0.8; 1.8], p = 0.298) or systolic BP (B [95%CI] 1.7 [-1.2; 4.6], p = 0.257) or diastolic BP (B [95%CI] 0.3 [-1.3; 1.9], p = 0.698) (eTable 4, online supplement). Duration of arterial oxygen saturation <90% and self-reported sleeping time were not associated with BP above the currently recommended BP target for patients with diabetes, systolic BP or diastolic BP (eTables 5a/b, online supplement).
After stratification for coronary heart disease, there was no more significant difference of systolic BP between periodic breathing groups in those patients with coronary heart disease (p = 0.634). Patients without coronary heart disease had a significantly higher systolic BP when they had periodic breathing (146.7 ± 17.8 vs. 136.4 ± 17.7 mmHg, p < 0.001; eFig. 1, online supplement).
4. Discussion
The present cross-sectional analysis in patients with DM2 yielded the following novel findings: Periodic breathing and SDB (defined as AHI>15/h) were significantly associated with BP above the currently recommended BP target of 130/80 mmHg for patients with diabetes (ESC/ESH) and higher systolic BP. This association was independent of other risk factors such as age, sex, WHR, HDL, eGFR, coronary heart disease, and antihypertensive treatment. Despite the use of more antihypertensive drugs in those patients, periodic breathing was independently associated with BP above the target range in patients with DM2.
The association of SDB with arterial hypertension/higher systolic BP in the general population has been reported before [24,25] and was confirmed in this sample of DM2 outpatients [17,18]. There are several studies on the association between obstructive sleep apnea and BP [26]. Treatment of obstructive sleep apnea with CPAP leads to a significant reduction of BP, at least in patients with good adherence to the device [27,28].
Data on the association of periodic breathing and BP are scarce. Only acute night time effects of periodic breathing were reported [8]. BP fluctuations during periodic breathing were shown to be primarily related to oscillations in ventilation during the periodic breathing cycle and partially occur in the absence of arousals [8]. Therefore, intermittent hypoxia, and not recurrent arousals, seems to be the dominant cause of daytime increases in BP [30]. In patients with systolic heart failure (ejection fraction ≤ 45%), central sleep apnea appears to have little or slightly positive effects on stroke volume [29]. Although the bi-directional relation between arterial hypertension and chronic heart failure is well known [31,32], there are no applicable epidemiologic studies on the association between periodic breathing and daytime BP. Our study is the first to examine and to find such an association in patients with DM2.
The standardized manner of measuring of BP in DIACORE [16] ensures a sufficient comparability of values in DIACORE. Usually, measurements have been taken during morning or until earlier afternoon, which provides a higher probability to observe sleep related BP effects prolonged to daytime [33]. However, no direct association between nocturnal BP and prolonged daytime BP increases can be evaluated, as no nighttime BP measurement has been conducted in this study.
Assuming that at least part of the observed difference in systolic BP among DM2 patients with and without periodic breathing (4.4 [95% CI: 0.1; 8.7] mmHg) is reversible by treatment, findings might have prognostic significance. Positive airway pressure may contribute to reach the target BP in 0%–25% of DM2 patients with BP above the target. According to a recent meta-analysis, a sufficient control of BP in target in DM2 patients according to current ESC/ESH guidelines [5] accounts for a significant risk reduction of major cardiovascular events to 0.82 [95% CI: 0.73; 0.92] [34].
In the present analysis, patients with periodic breathing had a lower heart rate, probably due to more frequent beta-blocker intake as part of antihypertensive treatment. In patients with coronary heart disease, there was no significant difference in systolic BP between those with and those without periodic breathing.
For diastolic BP, no significant association with periodic breathing (or SDB in general) could be established in the present study. Most studies on SDB did not distinguish between isolated systolic hypertension and arterial hypertension [35]. Isolated systolic hypertension is more common in older patients (>60 years) and related to arterial stiffness, whereas arterial hypertension is more common in middle-aged patients and related to sympathetic activation [36,37]. The association of SDB and BP is stronger in middle-aged patients via the common pathway of sympathetic activation [35]. Accordingly, SDB and systolic BP were not independently associated from other risk factors in the present study in older DM2 patients; and diastolic BP levels were mostly normal.
The strengths of the present study include the large sample size of DM2 patients and high-resolution phenotyping with regard to the association between sleep parameters and BP. The following limitations warrant discussion: first, no causal relationship of the underlying mechanism can be concluded; specifically, it is unclear whether periodic breathing or systolic BP causes BP above target/higher systolic BP or vice versa, or whether there is an association without a causal relationship. Longitudinal and physiological studies are needed to establish a better understanding of development and pathophysiological mechanism of the association between periodic breathing and BP in patients with and without DM2. Second, although the technique of pattern recognition for the detection of periodic breathing was described before [23], we could not formally verify the presence of periodic breathing as there was no polysomnography performed. Third, lack of nocturnal BP measurement disables statements on the direct effects of periodic breathing on nocturnal BP values in our study as reported before [8,29]. Therefore, no direct relationship of such effects and its association with our finding of periodic breathing and daytime BP above target/systolic BP can be deducted. Forth, our data provide no statement whether the currently recommended ESC/ESH BP target of 130/80 mmHg for patients with diabetes was pursued in this study sample. Notably, guideline recommendations on BP targets are subject to a continuous change and differ even in current recommendations among the different societies (eTable 6, online supplement).
5. Conclusion
In summary, our findings in this large sample of patients with DM2 show that the presence of periodic breathing and SDB are associated with BP values above the currently recommended ESC/ESH BP target for diabetes patients and higher systolic BP levels. BP above the currently recommended target is highly prevalent in patients with DM2 and periodic breathing despite the frequent and extensive use of antihypertensive drugs. Periodic breathing is associated with BP above the currently recommended target for diabetes patients/higher systolic BP independently from other risk factors.
Further studies on periodic breathing in patients with DM2 including direct nocturnal BP measurement, longitudinal and physiological analyses are warranted to improve the understanding of the underlying pathophysiology, especially the role of periodic breathing in BP above the target. Interventional studies are needed to investigate whether CPAP or adaptive servo-ventilation might be a therapeutic option, particularly in respect of preventing cardiovascular morbidity.
Financial support
The DIACORE study has been funded by the KfH Stiftung Präventivmedizin e.V. CB received funding from the KfH Stiftung Präventivmedizin e.V., the Else Kröner-Fresenius-Stiftung, and the Dr Robert Pfleger Stiftung. The DIACORE-SDB sub-study has been funded by ResMed (Martinsried, Germany). MA received grant support from ResMed, the ResMed Foundation, and Philips Respironics as well as lecture fees from ResMed and Philips Respironics.
CRediT authorship contribution statement
Andreas W. Schreib: Conceptualization, Writing - original draft, Writing - review & editing, Funding acquisition. Michael Arzt: Conceptualization, Writing - original draft, Writing - review & editing, Funding acquisition. Iris M. Heid: Investigation. Bettina Jung: Investigation. Carsten A. Böger: Investigation. Stefan Stadler: Conceptualization, Writing - original draft, Writing - review & editing, Funding acquisition.
Acknowledgements
We thank all participating patients of the DIACORE study. We thank the physicians and health insurance companies supporting the DIACORE study: Axel Andreae, Gerhard Haas, Sabine Haas, Jochen Manz, Johann Nusser, Günther Kreisel, Gerhard Bawidamann, Frederik Mader, Susanne Kiβkalt, Johann Hartl, Thomas Segiet, Christiane Gleixner, Christian Scholz, Monika Schober (Chief of Supply Management, Allgemeine Ortskrankenkasse Bayern), Cornelia Heinrich (Communication Manager, Allgemeine Ortskrankenkasse Bayern), Thomas Bohnhoff (Disease Management, Techniker Krankenkasse), Thomas Heilmann (Head of Disease Management, Techniker Krankenkasse), Stefan Stern (Consulting Physician, Allgemeine Ortskrankenkasse Bayern), Andreas Utz (Head of Department, Allgemeine Ortskrankenkasse Bayern), Georg Zellner (Chief of Supply Management, Deutsche Angestellten Krankenkasse), Werner Ettl (Barmer-GEK), Thomas Buck (Barmer-GEK), Rainer Bleek (IKK classic), and Ulrich Blaudzun (IKK classic). We further thank the study nurses for their expert work in performing the study visits: Simone Neumeier, Särah Hufnagel, Isabel Haller, Petra Jackermeier, Sabrina Obermüller, Christiane Ried, Ulrike Hanauer, Bärbel Sendtner, Natalia Riewe-Kerow, Konstantin Dumann, and Britta Hörmann (PhD-Students) for their expert work in conducting the study visits.
Footnotes
The authors declare that they have no conflict of interest.
The ICMJE Uniform Disclosure Form for Potential Conflicts of Interest associated with this article can be viewed by clicking on the following link: https://doi.org/10.1016/j.sleepx.2020.100013.
Supplementary data to this article can be found online at https://doi.org/10.1016/j.sleepx.2020.100013.
Contributor Information
Andreas W. Schreib, Email: a-schreib@t-online.de.
Michael Arzt, Email: michael.arzt@ukr.de.
Iris M. Heid, Email: iris.heid@ukr.de.
Bettina Jung, Email: bettina.jung@ukr.de.
Carsten A. Böger, Email: carsten.boeger@ukr.de.
Stefan Stadler, Email: stefan.stadler@ukr.de.
Conflict of interest
The following is the supplementary data to this article:
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Gress T.W., Nieto F.J., Shahar E. Hypertension and antihypertensive therapy as risk factors for type 2 diabetes mellitus. Atherosclerosis Risk in Communities Study. N Engl J Med. 2000;342(13):905–912. doi: 10.1056/NEJM200003303421301. [DOI] [PubMed] [Google Scholar]
- 2.Hypertension in Diabetes Study (HDS): I. Prevalence of hypertension in newly presenting type 2 diabetic patients and the association with risk factors for cardiovascular and diabetic complications. J Hypertens. 1993;11(3):309–317. doi: 10.1097/00004872-199303000-00012. [DOI] [PubMed] [Google Scholar]
- 3.Sowers J.R., Epstein M., Frohlich E.D. Diabetes, hypertension, and cardiovascular disease: an update. Hypertension. 2001;37(4):1053–1059. doi: 10.1161/01.HYP.37.4.1053. [DOI] [PubMed] [Google Scholar]
- 4.Tuomilehto J., Rastenyte D., Birkenhager W.H. Effects of calcium-channel blockade in older patients with diabetes and systolic hypertension. Systolic Hypertension in Europe Trial Investigators. N Engl J Med. 1999;340(9):677. doi: 10.1056/NEJM199903043400902. [DOI] [PubMed] [Google Scholar]
- 5.Williams B., Mancia G., Spiering W. ESC/ESH guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the european society of cardiology and the european society of hypertension: the task force for the management of arterial hypertension of the european society of cardiology and the european society of hypertension. J Hypertens. 2018;36(10):1953–2041. doi: 10.1097/HJH.0000000000001940. [DOI] [PubMed] [Google Scholar]
- 6.Flinta I., Ponikowski P. Relationship between central sleep apnea and Cheyne-stokes respiration. Int J Cardiol. 2016;206(Suppl. S8-12) doi: 10.1016/j.ijcard.2016.02.124. [DOI] [PubMed] [Google Scholar]
- 7.Sin D.D., Fitzgerald F., Parker J.D. Relationship of systolic BP to obstructive sleep apnea in patients with heart failure. Chest. 2003;123(5):1536–1543. doi: 10.1378/chest.123.5.1536. [DOI] [PubMed] [Google Scholar]
- 8.Trinder J., Merson R., Rosenberg J.I. Pathophysiological interactions of ventilation, arousals, and blood pressure oscillations during cheyne-Stokes respiration in patients with heart failure. Am J Respir Crit Care Med. 2000;162(3 Pt 1):808–813. doi: 10.1164/ajrccm.162.3.9806080. [DOI] [PubMed] [Google Scholar]
- 9.Randerath W., Verbraecken J., Andreas S. Definition, discrimination, diagnosis and treatment of central breathing disturbances during sleep. Eur Respir J. 2017;49(1):1600959. doi: 10.1183/13993003.00959-2016. Published 2017 Jan 18. [DOI] [PubMed] [Google Scholar]
- 10.Oldenburg O. Cheyne-Stokes respiration in chronic heart failure. Circ J. 2012;76(10):2305–2317. doi: 10.1253/circj.CJ-12-0689. [DOI] [PubMed] [Google Scholar]
- 11.Drager L.F., McEvoy R.D., Barbe F. Sleep apnea and cardiovascular disease: lessons from recent trials and need for team science. Circulation. 2017;136(19):1840–1850. doi: 10.1161/CIRCULATIONAHA.117.029400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leung R.S.T., Floras J.S., Lorenzi-Filho G. Influence of Cheyne-Stokes respiration on cardiovascular oscillations in heart failure. Am J Respir Crit Care Med. 2003;167(11):1534–1539. doi: 10.1164/rccm.200208-793OC. [DOI] [PubMed] [Google Scholar]
- 13.Franklin K.A., Sandström E., Johansson G. Hemodynamics, cerebral circulation, and oxygen saturation in Cheyne-Stokes respiration. J Appl Physiol. 1997;83(4):1184–1191. doi: 10.1152/jappl.1997.83.4.1184. [DOI] [PubMed] [Google Scholar]
- 14.Berthon-Jones M., Sullivan C.E. Ventilatory and arousal responses to hypoxia in sleeping humans. Am Rev Respir Dis. 1982;125(6):632–639. doi: 10.1164/arrd.1982.125.6.632. [DOI] [PubMed] [Google Scholar]
- 15.Iftikhar I.H., Valentine C.W., Bittencourt L.R.A. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a meta-analysis. J Hypertens. 2014;32(12):2341–2350. doi: 10.1097/HJH.0000000000000372. Discussion 2350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dörhöfer L., Lammert A., Krane V. Study design of DIACORE (DIAbetes COhoRtE) - a cohort study of patients with diabetes mellitus type 2. BMC Med Genet. 2013;1425 doi: 10.1186/1471-2350-14-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stadler S., Zimmermann T., Franke F. Association of sleep-disordered breathing with diabetes-associated kidney disease. Ann Med. 2017;49(6):487–495. doi: 10.1080/07853890.2017.1306100. [DOI] [PubMed] [Google Scholar]
- 18.Stadler S., Jalili S., Schreib A. Association of sleep-disordered breathing with severe chronic vascular disease in patients with type 2 diabetes. Sleep Med. 2018:4853–4860. doi: 10.1016/j.sleep.2018.05.001. [DOI] [PubMed] [Google Scholar]
- 19.Chen H., Lowe A.A., Bai Y. Evaluation of a portable recording device (ApneaLink) for case selection of obstructive sleep apnea. Sleep Breath. 2009;13(3):213–219. doi: 10.1007/s11325-008-0232-4. [DOI] [PubMed] [Google Scholar]
- 20.Erman M.K., Stewart D., Einhorn D. Validation of the ApneaLink for the screening of sleep apnea: a novel and simple single-channel recording device. J Clin Sleep Med. 2007;3(4):387–392. [PMC free article] [PubMed] [Google Scholar]
- 21.Arzt M., Woehrle H., Oldenburg O. Prevalence and predictors of sleep-disordered breathing in patients with stable chronic heart failure: the SchlaHF registry. JACC Heart Fail. 2016;4(2):116–125. doi: 10.1016/j.jchf.2015.09.014. [DOI] [PubMed] [Google Scholar]
- 22.Stehling F., Keull J., Olivier M. Validation of the screening tool ApneaLink® in comparison to polysomnography for the diagnosis of sleep-disordered breathing in children and adolescents. Sleep Med. 2017:3713–3718. doi: 10.1016/j.sleep.2017.05.018. [DOI] [PubMed] [Google Scholar]
- 23.Weinreich G., Armitstead J., Töpfer V. Validation of ApneaLink as screening device for Cheyne-Stokes respiration. Sleep. 2009;32(4):553–557. doi: 10.1093/sleep/32.4.553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nieto F.J. Association of sleep-disordered breathing, sleep apnea, and hypertension in a large community-based study. JAMA. 2000;283(14):1829. doi: 10.1001/jama.283.14.1829. [DOI] [PubMed] [Google Scholar]
- 25.Young T., Peppard P., Palta M. Population-based study of sleep-disordered breathing as a risk factor for hypertension. Arch Intern Med. 1997;157(15):1746–1752. doi: 10.1001/archinte.1997.00440360178019. [DOI] [PubMed] [Google Scholar]
- 26.Drager L.F., Polotsky V.Y., O'Donnell C.P. Translational approaches to understanding metabolic dysfunction and cardiovascular consequences of obstructive sleep apnea. Am J Physiol Heart Circ Physiol. 2015;309(7):H1101–H1111. doi: 10.1152/ajpheart.00094.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Barbé F., Durán-Cantolla J., Sánchez-de-la-Torre M. Effect of continuous positive airway pressure on the incidence of hypertension and cardiovascular events in nonsleepy patients with obstructive sleep apnea: a randomized controlled trial. JAMA. 2012;307(20):2161–2168. doi: 10.1001/jama.2012.4366. [DOI] [PubMed] [Google Scholar]
- 28.Bazzano L.A., Khan Z., Reynolds K. Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension. 2007;50(2):417–423. doi: 10.1161/HYPERTENSIONAHA.106.085175. [DOI] [PubMed] [Google Scholar]
- 29.Yumino D., Kasai T., Kimmerly D. Differing effects of obstructive and central sleep apneas on stroke volume in patients with heart failure. Am J Respir Crit Care Med. 2013;187(4):433–438. doi: 10.1164/rccm.201205-0894OC. [DOI] [PubMed] [Google Scholar]
- 30.Turnbull C.D., Sen D., Kohler M. Effect of supplemental oxygen on blood pressure in obstructive sleep apnea (SOX): a randomised, CPAP withdrawal trial. Am J Respir Crit Care Med. 2018 doi: 10.1164/rccm.201802-0240OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Messerli F.H., Williams B., Ritz E. Essential hypertension. Lancet. 2007;370(9587):591–603. doi: 10.1016/S0140-6736(07)61299-9. [DOI] [PubMed] [Google Scholar]
- 32.Aurigemma G.P., Gaasch W.H. Clinical practice. Diastolic heart failure. N Engl J Med. 2004;351(11):1097–1105. doi: 10.1056/NEJMcp022709. [DOI] [PubMed] [Google Scholar]
- 33.Becker H.F.1, Jerrentrup A., Ploch T. Effect of nasal continuous positive airway pressure treatment on blood pressure in patients with obstructive sleep apnea. Circulation. 2003 Jan 7;107(1):68–73. doi: 10.1161/01.cir.0000042706.47107.7a. PMID:12515745. [DOI] [PubMed] [Google Scholar]
- 34.Wang J., Chen Y., Xu W. Effects of intensive blood pressure lowering on mortality and cardiovascular and renal outcomes in type 2 diabetic patients: a meta-analysis. PLoS One. 2019 Apr 12;14(4):e0215362. doi: 10.1371/journal.pone.0215362. PubMed PMID: 30978254; PubMed Central PMCID: PMC6461269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Haas D.C., Foster G.L., Nieto F.J. Age-dependent associations between sleep-disordered breathing and hypertension: importance of discriminating between systolic/diastolic hypertension and isolated systolic hypertension in the Sleep Heart Health Study. Circulation. 2005;111(5):614–621. doi: 10.1161/01.CIR.0000154540.62381.CF. [DOI] [PubMed] [Google Scholar]
- 36.Esler M., Rumantir M., Kaye D. Sympathetic nerve biology in essential hypertension. Clin Exp Pharmacol Physiol. 2001;28(12):986–989. doi: 10.1046/j.1440-1681.2001.03566.x. [DOI] [PubMed] [Google Scholar]
- 37.Lakatta E.G., Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a "set up" for vascular disease. Circulation. 2003;107(1):139–146. doi: 10.1161/01.cir.0000048892.83521.58. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.