Abstract
Background
Comparing the signal intensity (SI) of an ocular mass to that of the vitreous body has been suggested. Most ocular lesions show a hyper-intense signal compared to the vitreous body on T1-weighted (T1w) images, and malignant melanomas have been almost always determined as ‘cannot be excluded’ in reports.
Purpose
This study aimed to determine the accuracy of magnetic resonance imaging (MRI) in the diagnosis of uveal melanoma by using normal white matter as reference tissue for SI evaluation on T1w images and vitreous body on T2w compared to the conventional method using the vitreous body as a reference on both T1w and T2w images.
Methods
The MRIs of 43 patients (between August 2006 and July 2018) sent to rule out uveal melanoma were blindly reviewed by two radiologists. By using white matter as a reference for SI evaluation on T1w images and vitreous body as a reference on T2w images, uveal melanomas were suggested by hyper-intense signal on T1w and hypo-intense signal on T2w with homogeneous enhancement. The accuracy of diagnosis of uveal melanoma using white matter as a reference on T1w was compared to the conventional method using the vitreous body as a reference on both T1w and T2w images.
Results
The diagnosis of uveal melanoma using white matter as a reference gave a sensitivity of 92.31% (95% confidence interval (CI) 63.97–99.81) and specificity of 100.0% (95% CI 88.43–100.0). By using the vitreous body as a reference, sensitivity as high as 100.0% (95% CI 100.0–100.0) was obtained, but specificity was low at 53.33% (95% CI 34.33–71.66).
Conclusions
White matter is a good reference for the diagnosis of uveal melanoma, with high sensitivity and much higher specificity than conventional methods using the vitreous body as a reference.
Keywords: Uveal melanoma, magnetic resonance imaging (MRI), white matter, reference
Introduction
Uveal melanoma is the second most frequent ocular malignancy after metastasis and the most common primary ocular malignant neoplasm in adults,1 most commonly involving the choroid (90%), ciliary body (7%) and iris (2%).2 The diagnosis of uveal melanoma is usually possible by ophthalmoscopy, fluorescein angiography or sonography. However, clinical diagnosis may sometimes be difficult, especially in patients with cataracts, opaque vitreous media or lesions concealed by retinal detachment. Magnetic resonance imaging (MRI) may provide additional useful information in the diagnosis or exclusion of uveal melanoma.
On MRI, melanoma is typically hyper-intense on T1-weighted (T1w) images and hypo-intense on T2w images, and shows diffuse moderate enhancement after injection of gadolinium-based contrast material. This characteristic signal intensity (SI) of melanoma is believed to be due to the T1 and T2 shortening effect of paramagnetic stable free radicals with unpaired electrons in the melanin pigment.3,4 Studies also have shown that the degree of melanomatous pigmentation correlates with quantitative T1 signal,5,6 and this finding may also be of value in predicting patient prognosis, as strong pigmentation confers a less favourable prognosis.5
Most qualitative evaluations of the SI of the ocular mass have been suggested to be compared to the SI of the vitreous body7–11 which is the clear gel between the lens and the retina. According to Lemke et al.,11 the vitreous body was chosen as a reference medium for two reasons. First, it is the only structure in the eye with a chemical composition that remains constant unless it is affected by disturbing factors such as bleeding or diffuse vitreous hyper-intensity, which rarely occur. Diffuse vitreous hyper-intensity may be due to discrete vitritis or damage to the blood–retinal barrier, with leakage of protein.12 Second, the use of the vitreous body as a reference medium guarantees comparability to other studies.9
Since 98–99% of the vitreous body’s volume is water, the SI of the vitreous body is similar to fluid elsewhere in the body, giving a hypo-intense signal on T1w images and a hyper-intense signal on T2w images. Therefore, most ocular lesions show a hyper-intense signal compared to the SI of the vitreous body on T1w images and in only minority of cases show an iso-intense or hypo-intense SI. In our clinical practice, except for the calcification found in retinoblastoma, we almost have never found ocular neoplasms that exhibit a hypo-intense signal compared to the vitreous body on T1w images. By using the vitreous body as a reference on T1w image, most ocular lesions appear as a hyper-intense signal, and malignant melanoma have been almost always determined as ‘cannot be excluded’ in our reports. Therefore, we tried to search for other tissue within the orbit or its nearby structure which can be used as a reference for the better differentiation between ocular melanoma and other ocular masses.
By using fat and muscle as reference tissues, Marx et al.13 found that only 7/13 ocular melanomas exhibited typical imaging appearance, and concluded that although T1 and T2 shortening signal patterns are typical of melanoma, the absence of these findings does not exclude the diagnosis. We tried to use the normal uveal tract, such as the ciliary body, in the opposite globe as a reference, but most uveal tracts were too small to be reliably used as a reference. We also had tried to use extra-ocular muscles and nasal turbinate as a reference, but the results were also not satisfactory. We did not try the intensity of the cerebrospinal fluid (CSF) on either T1w or T2w images, since the SI of the CSF space is almost similar to that of the vitreous body. We finally had the idea that normal white matter of the partly included brain may be a good reference due to the slightly hyper-intense SI. Xian et al.14 suggested that ‘The brain as a reference of the signal intensity of the uveal tumour is better than the vitreous body or the orbital fat’ in their study describing the MRI features of uveal schwannoma. However, they did not mention exactly which part of brain they recommend to be used as a reference. To our knowledge, no research exists using white matter as a reference tissue in the SI evaluation of ocular mass. Our hypothesis is that uveal melanoma should exhibit a hyper-intense SI on T1w compared to normal white matter and a hypo-intense SI on T2w images compared to the vitreous body and diffuse enhancement after contrast media injection while other lesions do not.
The objective of our study was to determine the accuracy of MRI in the diagnosis of uveal melanoma by using normal white matter, including in the standard orbital study, as a reference tissue on the T1w image compared to the convention method using the vitreous body.
Methods
This retrospective study was approved by our Institutional Review Board. The medical records and MRIs from the Picture Archive and Communication System (Synapse PACS, Fujifilm v3.2.0) of 43 patients who underwent MRI due to intraocular lesions in order to rule out melanoma between August 2006 and July 2018 were reviewed. Final proven diagnoses were evaluated. Children younger than three years old were excluded from our study due to the possibility of immature myelination.15
We considered a total of 43 patients (18 men and 25 women; mean age 50.95 years; age range 5–80 years), including 19 patients with histopathologically proven diagnoses by enucleation (Table 1) and 24 patients with presumed clinical diagnoses (Table 2). All clinically presumed benign lesions such as retinal/choroidal detachment and haemorrhage need at least two years of follow-up to confirm their benignity.
Table 1.
Clinical data from the patients with histopathological proven diagnoses.
| Histopathology | No. of patients | Sex (M:F) | Average age (range) |
|---|---|---|---|
| Melanoma | 12 | 4:8 | 61 (49–80) |
| Metastasis | 3 | 1:2 | 44 (37–43) |
| Retinoblastoma | 2 | 1:1 | 10 (5–15) |
| Medulloepithelioma | 1 | 1:0 | 19 |
| Retinal detachment with haemorrhage | 1 | 1:0 | 46 |
Table 2.
Clinical data from the patients with clinically presumed diagnoses.
| Clinical diagnosis | No. of patients | Sex (M:F) | Average age (age range) |
|---|---|---|---|
| Melanoma | 1 | 1:0 | 17 |
| Metastases | 7 | 0:7 | 48 (35–70) |
| Choroidal/retinal detachment haemorrhage/haemangioma | 16 | (7:6) | 56 (24–79) |
All patients underwent MRI studies on a 3 Tesla clinical scanner (Achieva; Philips Medical Imaging System, Best, The Netherlands) equipped with an eight-channel head coil with SENSE factor or on a 1.5 Tesla scanner (Magnetom Vision; Siemens Medical System, Erlangen, Germany) with a circular polarised array head coil. Thin slices (2–3 mm) high-resolution SE T1w images, axial and coronal T2w images and post-contrast-enhanced T1w with fat suppression in axial, sagittal and coronal planes were included in all studies.
Two neuroradiologists (S.H. and N.S.) blinded to the diagnosis analysed the preoperative MRI studies from our routine PACS workstation separately. The SI of the lesions on T1w images were visually inspected and classified as hyper-intense, iso-intense or hypo-intense compared to normal white matter in the partly included brain and also with the vitreous body. The diagnoses of uveal malignant melanomas in our study were based primarily on the SI and pattern of enhancement. The diagnosis was suggested when the ocular mass revealed a hyper-intense signal on T1w image compared to the normal white matter, a hypo-intense signal on T2w images compared to the vitreous body and homogeneous enhancement after contrast media injection. The lesions were then categorised into suspicious melanoma, non-melanoma neoplasm and benign non-neoplastic lesions. Other imaging appearances such as the site, size and shape of the lesion did not affect our decision. In the event of disagreement, the final decision was made by consensus. The sensitivity and specificity of the diagnosis of malignant melanoma using normal white matter as a reference were calculated, analysed and compared to the results using vitreous body as a reference. Inter-observer correlation was evaluated by Kappa statistic using Stata MP v15 (64-bit; StataCorp, College Station, TX).
Results
The MRI appearances of the ocular masses in all patients are summarised in Table 3. The typical MRI appearance of uveal melanoma using the white matter as a reference was seen in 92.30% (12/13) of cases (Figure 1), giving a sensitivity of 92.31% (95% confidence interval (CI) 63.97–99.81), a specificity of 100.0% (95% CI 88.43–100.0), a positive predictive value (PPV) of 100.0% (95% CI 100.0–100.0) and a negative predictive value (NPV) of 96.77% (95% CI 82.03–99.50). By using the vitreous body as a reference, sensitivity as high as 100.0% (95% CI 100.0–100.0) was obtained, but specificity was low at 53.33% (95% CI 34.33–71.66), PPV was 48.15% (95% CI 38.78–57.65) and NPV was 100.0% (95% CI 100.0–100.0). The inter-observer agreement showed almost perfect agreement between both radiologists (κ=0.81; 95% CI 0.875–0.985, p<0.001).
Table 3.
Magnetic resonance imaging appearances of the ocular masses in all patients.
| Patients | Age (years)/sex | T1w WM | T2w VB | T1w VB | Enhancement | Final diagnosis | Diagnosis by |
|---|---|---|---|---|---|---|---|
| 1 | 49/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 2 | 52/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 3 | 80/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 4 | 69/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 5 | 56/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 6 | 60/M | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 7 | 53/M | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 8 | 51/F | Iso | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 9 | 72/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 10 | 65/M | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 11 | 65/M | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 12 | 17/M | Hyper | Hypo | Hyper | Enhanced | Melanoma | Clinical |
| 13 | 57/F | Hyper | Hypo | Hyper | Enhanced | Melanoma | Histopathology |
| 14 | 35/F | Hypo | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Clinical |
| 15 | 42/F | Iso | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Histopathology |
| 16 | 45/F | Iso | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Clinical |
| 17 | 45/F | Iso | Hypo | Hyper | Enhanced | Hepatocellular carcinoma with choroidal metastasis | Clinical |
| 18 | 43/F | Iso | Hypo | Hyper | Enhanced | Cervical cancer with multiple metastases | Histopathology |
| 19 | 45/F | Hypo | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Histopathology |
| 20 | 37/M | Hypo | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Histopathology |
| 21 | 54/F | Hypo | Hypo | Hyper | Enhanced | Nasopharyngeal cancer with choroidal metastasis | Histopathology |
| 22 | 49/M | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 23 | 67/F | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 24 | 54/F | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 25 | 49/F | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 26 | 64/F | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 27 | 38/M | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 28 | 72/M | Hypo | Hypo | Iso | Not enhanced | Haemorrhage | Clinical |
| 29 | 67/M | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 30 | 51/F | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 31 | 49/M | Hyper | Hypo | Hyper | Not enhanced | Haemorrhage | Clinical |
| 32 | 35/M | Hyper | Hyper | Hyper | Not enhanced | Hematoma mimic mass with retinal detachment | Clinical |
| 33 | 79/F | Hyper | Hyper | Hyper | Not enhanced | Retinal detachment with haemorrhage | Clinical |
| 34 | 79/M | Iso | Hyper | Hyper | Not enhanced | Choroidal detachment | Clinical |
| 35 | 19/M | Hypo | Hypo | Hyper | Enhanced | Medulloepithelioma | Histopathology |
| 36 | 15/F | Iso | Hypo | Hyper | Enhanced | Retinoblastoma | Histopathology |
| 37 | 5/M | Hypo | Hypo | Hyper | Enhanced | Retinoblastoma | Histopathology |
| 38 | 60/F | Iso | Hyper | Hyper | Enhanced | Choroidal haemangioma | Clinical |
| 39 | 24/M | Iso | Hypo | Hyper | Enhanced | Benign lesion could be infection/inflammation | Clinical |
| 40 | 60/M | Hyper | Hyper | Hyper | Not enhanced | Old retinal haemorrhage | Clinical |
| 41 | 70/F | Iso | Hypo | Hyper | Enhanced | Lung cancer with choroidal metastasis | Clinical |
| 42 | 46/M | Hyper | Hypo | Hyper | Not enhanced | Retinal detachment with haemorrhage | Histopathology |
| 43 | 47/F | Iso | Hypo | Hyper | Enhanced | Breast cancer with choroidal metastasis | Clinical |
T1w WM: T1-weighted image compared to normal white matter; T1w VB: T1-weighted image compared to the vitreous body; T2w VB: T2-weighted image compared to the vitreous body.
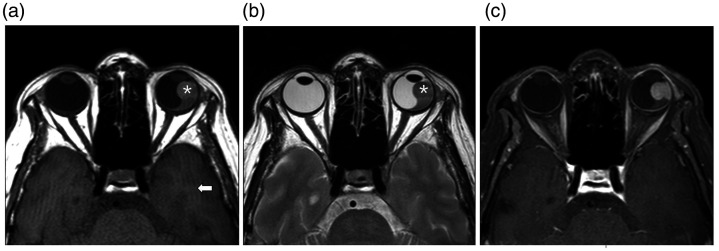
Figure 1.
The typical magnetic resonance imaging (MRI) appearance of melanoma. A 67-year-old woman with blurred vision in the left eye for 10 days. (a) Axial T1-weighted spin-echo MRI (400/10.10) shows an ocular mass (*) with a hyper-intense signal compared to normal white matter (arrow). (b) Axial T2-weighted spin-echo MRI (3000/90) shows a hypo-intense mass (*) compared to the vitreous body. (c) Contrast-enhanced T1-weighted image (400/12) shows homogeneous enhancement.
Discussion
Our results indicated a low specificity (58.33%) in spite of a very high sensitivity (100%) in the diagnosis of uveal melanoma by using the vitreous body as a reference tissue in the evaluation on both T1w and T2w images. Therefore, this conventional method cannot reliably distinguish melanoma from other ocular pathologies, and it has been suggested that the role of MRI should be limited mainly to the detection of extra-ocular extension and to exclude any other disease rather than to establish a diagnosis.6
By using normal white matter as a reference tissue on T1w and vitreous body on T2w images, our results indicate high sensitivity (92.31%) and specificity (100%) in the diagnosis or exclusion of uveal melanoma. The recently published study by Sun et al.16 used grey matter as a reference in their study. The paper reported:
The diagnostic accuracy of low signal on T2w and high signal on T1w (compared with gray matter) was significantly higher than those of vitreous (Delong test, p<0.001). The mass with hypointense on T2w (compared with grey matter) was the best MRI feature in the differential diagnosis of uveal melanoma and other intraocular masses, with the accuracy of 86.2%.
White matter is a good reference tissue due to its cholesterol and glycolipids content15 which normally give a slightly brighter SI on T1w than most other normal soft tissues. However, its signal is still usually less intense than melanin-containing lesions, such as melanomas, which was confirmed by the results of our study. Tumours with a higher content of melanin pigment have also been reported to appear more hyper-intense on T1w images.11 Concerns about the possibility of the brain being affected by various pathological conditions as well as aging caused no problems for interpretations in our study.
Melanomas have a characteristic orange pigment that can be seen at the fundus examination. However, the colour of melanoma varies from darkly pigmented to colourless (amelanotic) during the clinical examination. Approximately 20–25% of melanomas have been reported to be amelanotic in the Western literature, thereby lacking characteristic T1 and T2 shortening effects on MRI.11,17 Reports of amelanotic melanoma in Asian patients seem to be less common than in Caucasian patients. Shields et al.18 reported a rate of 14% non-pigmented melanoma in their Asian group of patients, and Pal et al.19 reported a rate of 8.25% amelanotic melanoma in the Indian population. We did not find a single case of amelanotic melanoma in our study, which is the same result as in the large study conducted by Liu et al. in China.20 Therefore, we can be less concerned about amelanotic melanoma in our population.
The reason for the lower incidence of amelanotic melanoma in Asian patients is unclear, but it may be related to genetic differences between Asian and Western populations, for example the dark eye colour and more severe exposure to sunlight in the Asian population compared to the light eye colour and less severe exposure to sunlight in the Western population.
A major limitation of our study is its small sample size. There were only 13 patients with melanoma from the total of 43 patients in our study. Had we seen more patients, we may have had some cases of amelanotic melanoma, and may also have detected other kinds of mimicking melanoma lesions. However, this was the best we could do within the 12-year-period in our tertiary hospital, which is the largest referral centre in Southern Thailand. Melanoma is far more common in Caucasians, with 98% of cases occurring in this group.2 It is rarely found among Africans or African Americans. The risk is also low among Asian populations21 and American populations of Asian descent.22
Other common causes of high SI on T1w image and low signal on T2w are vitreous haemorrhage and proteinaceous fluid. However, the differential diagnosis from melanoma should not be a problem in most cases, since there is no enhancement in vitreous haemorrhage and proteinaceous fluid. Most haemorrhages also show a more hypo-intense signal on T2w than melanoma due to the intracellular methemoglobin or hemosiderin. The commonly associated retinal or choroidal detachment also have the pathognomonic V-shaped appearance in retinal detachments6 or ‘tennis ball sign’ in choroidal detachment.
The main differential diagnosis of uveal melanoma is choroidal metastases (from lung, breast, hyper-vascular and haematological malignancies). Choroidal metastases are usually depicted as broad-based flat lesions and iso- to hyper-intense on T1w images and hypo-intense on T2w images. The high levels of proteins or haemoglobin degradation products in the choroidal metastases from a mucin-producing adenocarcinoma and haemorrhagic metastases can mimic sign of melanoma at MRI (T1 hyper-intense, T2 hypo-intense). Obtaining the clinical history of the patient is therefore mandatory.6 However, by using white matter as a reference tissue on T1w evaluation, no choroidal metastasis revealed a hyper-intense SI in our study. Similar to the literature,5 lung cancers are the most common primary to metastases to globes in our study (67%, 6/9 cases; Figure 2). We missed the diagnosis of malignant melanoma in only one case interpreted as iso-SI on T1w images which may be explained by the evidence of associated internal haemorrhage (Figure 3).
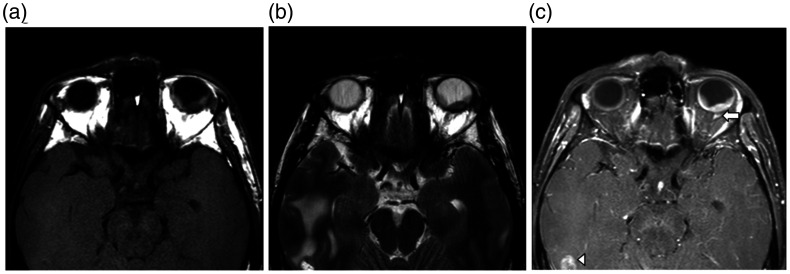
Figure 2.
MRI of a 36-year-old man with known adenocarcinoma of the lungs who developed blurred vision in the left eye. (a) Axial T1-weighted spin-echo MRI (471/12) shows a hypo-intense mass. (b) Axial T2-weighted spin-echo MRI (3000/90) shows a hypo-intense mass. (c) Axial contrast-enhanced T1-weighted spin-echo MRI (551/12) shows enhancement intraocular mass and adjacent retrobulbar fat as evidence of extra-ocular invasion (arrow). Small metastasis with surrounding brain oedema in the right temporal lobe are noted (arrowhead).
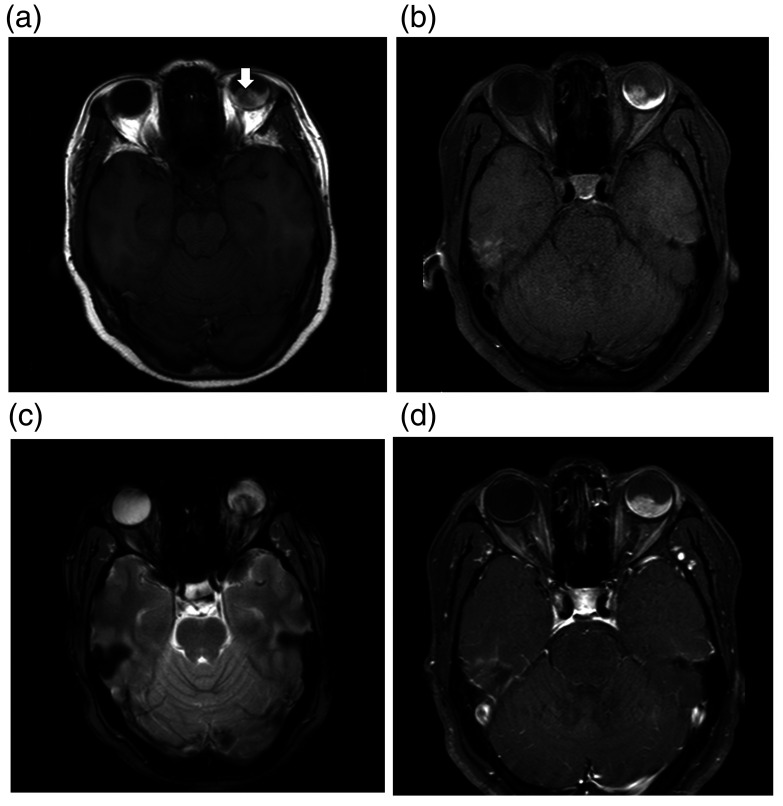
Figure 3.
MRI of melanoma in a 51-year-old woman with blurred vision in left eye for one year. (a) Axial T1-weighted spin-echo MRI (400/9) shows the iso- to slightly hypo-signal intensity intraocular mass (arrow). (b) Fat suppressed T1-weighted spin-echo image (400/12). (c) Axial T2-weighted gradient-echo MRI (511/11, 18° FA) shows evidence of associated internal haemorrhage seen as a hypo-intense signal. (d) Post-contrast-enhanced fat-suppressed T1-weighted image (551/12) reveals moderate enhancement of the lesion.
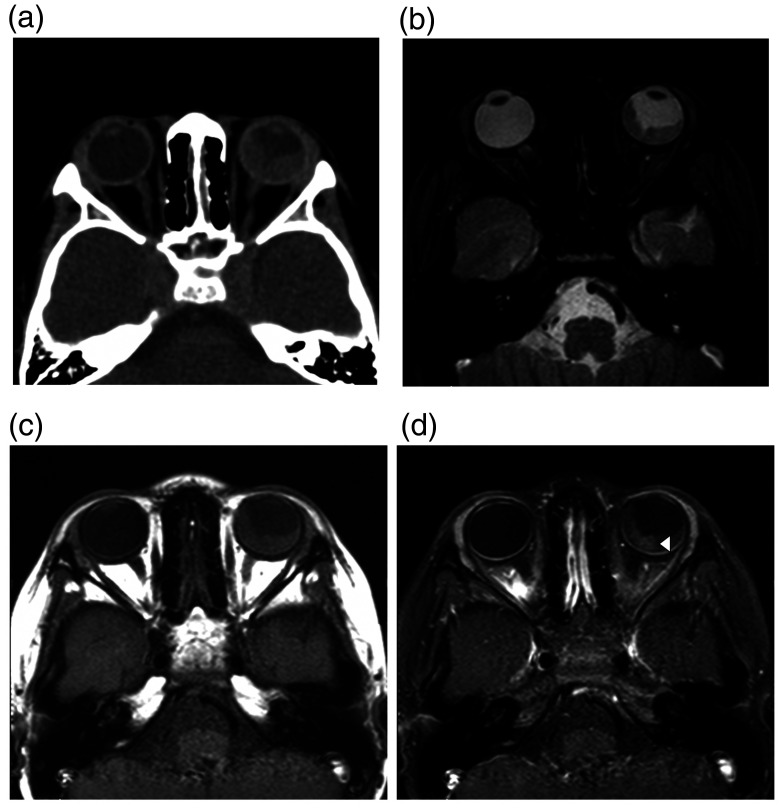
Retinoblastomas are usually seen in a younger population and commonly demonstrate typical calcifications on computed tomography (CT). However, we found a retinoblastoma in a five-year-old boy who presented with non-calcified ocular masses and revealed similar imaging appearances with metastasis on MRI (Figure 4). Young age without known primary cancer should therefore raise the possibility of retinoblastoma.
Figure 4.
(a) Axial computed tomography image of the orbit in a five-year-old boy with left eye leukocoria for one month reveals total retinal detachment in the left eye without evidence of calcification. (b) Axial T2-weighted spin-echo MRI (396.46,11.51) shows a hypo-intense mass. (c) Axial contrast-enhanced T1-weighted spin-echo MRI (400/12) and (d) axial fat-suppressed contrast-enhanced T1-weighted spin-echo MRI (400/12) show slightly homogeneous enhancement mass (arrowhead).
Choroidal haemangioma is typically hypo-intense on T1w and hyper-intense on T2w images with strong enhancement after contrast material injection. Differential diagnosis from melanoma is not a problem due to the different SI on T1w and T2w. Uveal melanoma was also reported to give a weaker enhancement than choroidal haemangioma.10,23 When present, a well-defined centrifugal pattern of enhancement caused by the abundant tumour vessels of choroidal haemangioma was another sign that was useful for distinguishing between the two types of neoplasm.23 Ultrasonography (US) is also a valuable and cost-effective method for differentiating both tumours, since melanoma demonstrates low to medium internal reflectivity spikes on the tumour surface, while choroidal haemangioma demonstrates high internal reflectivity at A-mode US.6 US alone had been reported to have a diagnostic accuracy of >95% in the diagnosis of choroidal melanoma.24 However, the accuracy depends on the location of the uveal lesions as well as on the experiences of the operator, and the results might be less accurate in cases of extensive ocular bleeding.23,25,26
The typical melanoma is a mushroom-shaped tumour with a broad choroidal base but can also manifest as a flat or crescentic mass. Melanoma usually protrudes into the vitreous and is associated with serous retinal detachment. At imaging, the detachment is often indistinguishable from the neoplasm without intravenous administration of contrast media.6
Apart from SI evaluation in pre-contrast enhanced studies, post-contrast enhanced studies with fat suppression are helpful and should always be included in the evaluation of the suspicious ocular lesion by MRI. Without significant enhancement, neoplasm is the unlikely diagnosis, and haemorrhage or retinal or choroidal detachments were common causes in our study. Therefore, we recommend first to look for the enhancement on the post-contrast-enhanced T1w with fat suppression image. If there is no enhancement, melanoma or other neoplasm is the unlikely diagnosis. If there is enhancement, the neoplasm is suspicious.
Choroidal osteoma, a rare benign choroidal ossifying tumour, has also been reported to show as hyper-intense on T1w images, hypo-intense on T2w images and diffuse enhancement, similar to malignant melanoma. These MRI findings are due to the presence of fatty marrow in the trabecular spaces of the bone at the level of the choroid. However, the diagnosis is not a problem practically due to the totally different clinical context, and the diagnosis of choroidal osteoma is mainly clinical. If still clinically suspicious, it can be easily proven by CT as a calcific curvilinear lesion on the posterior aspect of the globe.
Conclusion
The presence of a hyper-intense SI on T1w compared to normal white matter, a hypo-intense SI on T2w compared to the vitreous body and homogeneous enhancement appears to be a more accurate method for the diagnosis of uveal melanoma compared to the conventional method using the vitreous body as a reference of both T1w and T2w images.
Acknowledgements
Special thanks to Mr Trevor T. Pearson for proof reading and editing assistance.
Conflict of interest: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) received no financial support for the research, authorship and/or publication of this article.
ORCID iD: Pornrujee Hirunpat https://orcid.org/0000-0003-3288-8502
References
- 1.Tong KA, Osborn AG, Mamalis N, et al. Ocular melanoma. AJNR Am J Neuroradiol 1993; 14: 1359–1366. [PMC free article] [PubMed] [Google Scholar]
- 2.Shields CL, Manalac J, Das C, et al. Choroidal melanoma: clinical features, classification, and top 10 pseudomelanomas. Curr Opin Ophthalmol 2014; 25: 177–185. [DOI] [PubMed] [Google Scholar]
- 3.Smoker WR, Gentry LR, Yee NK, et al. Vascular lesions of the orbit: more than meets the eye. Radiographics 2008; 28: 185–204; quiz 325. [DOI] [PubMed] [Google Scholar]
- 4.Enochs WS, Petherick P, Bogdanova A, et al. Paramagnetic metal scavenging by melanin: MR imaging. Radiology 1997; 204: 417–423. [DOI] [PubMed] [Google Scholar]
- 5.Tailor TD, Gupta D, Dalley RW, et al. Orbital neoplasms in adults: clinical, radiologic, and pathologic review. Radiographics 2013; 33: 1739–1758. [DOI] [PubMed] [Google Scholar]
- 6.Houle V, Bélair M, Allaire GS. AIRP best cases in radiologic–pathologic correlation: choroidal melanoma. Radiographics 2011; 31: 1231–1236. [DOI] [PubMed] [Google Scholar]
- 7.Lemke AJ, Hosten N, Wiegel T, et al. Intraocular metastases: differential diagnosis from uveal melanomas with high-resolution MRI using surface coil. Eur Radiol 2001; 11: 2593–2601. [DOI] [PubMed] [Google Scholar]
- 8.De Potter P, Flanders AE, Shields JA, et al. The role of fat-suppression technique and gadopentetate dimeglumine in magnetic resonance imaging evaluation of intraocular tumors and simulating lesions. Arch Ophthalmol 1994; 112: 340–348. [DOI] [PubMed] [Google Scholar]
- 9.Peyster RG, Augsburger JJ, Shields JA, et al. Intraocular tumors: evaluation with MR imaging. Radiology 1988; 168: 773–779. [DOI] [PubMed] [Google Scholar]
- 10.Mafee MF, Peyman GA, Grisolano JE, et al. Malignant uveal melanoma and simulating lesions: MR imaging evaluation. Radiology 1986; 160: 773–780. [DOI] [PubMed] [Google Scholar]
- 11.Lemke AJ, Hosten N, Bornfeld N, et al. Uveal melanoma: correlation of histopathologic and radiologic findings by using thin-section MR imaging with a surface coil. Radiology 1999; 210: 775–783. [DOI] [PubMed] [Google Scholar]
- 12.Spencer G, Lufkin R, Simons K, et al. MR of a melanoma simulating ocular neoplasm. AJNR Am J Neuroradiol 1987; 8: 921–922. [PMC free article] [PubMed] [Google Scholar]
- 13.Marx HF, Colletti PM, Raval JK, et al. Magnetic resonance imaging features in melanoma. Magn Reson Imaging 1990; 8: 223–229. [DOI] [PubMed] [Google Scholar]
- 14.Xian J, Xu X, Wang Z, et al. MR imaging findings of the uveal schwannoma. AJNR Am J Neuroradiol 2009; 30: 769–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Parazzini C, Baldoli C, Scotti G, et al. Terminal zones of myelination: MR evaluation of children aged 20–40 months. AJNR Am J Neuroradiol 2002; 23: 1669–1673. [PMC free article] [PubMed] [Google Scholar]
- 16.Sun M, Chen Q, Xu X, et al. Value of MRI in the differential diagnosis of uveal melanoma and other intraocular masses in adults. Chin J Radiol 2020; 54: 181–186. [Google Scholar]
- 17.Lee DS, Anderson SF, Perez EM, et al. Amelanotic choroidal nevus and melanoma: cytology, tumor size, and pigmentation as prognostic indicators. Optom Vis Sci 2001; 78: 483–491. [DOI] [PubMed] [Google Scholar]
- 18.Shields C, Kaliki S, Cohen M, et al. Prognosis of uveal melanoma based on race in 8100 patients: the 2015 Doyne Lecture. Eye 2015; 29: 1027–1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pal B, Garge S, Khetan V. Choroidal melanoma: a short review with an Indian perspective. Oman J Ophthalmol 2017; 10: 135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu Y, Li Y, Wei W, et al. Clinical characteristics of 582 patients with uveal melanoma in China. PLoS One 2015; 10: e0144562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kuo PK, Puliafito CA, Wang KM, et al. Uveal melanoma in China. Int Ophthalmol Clin 1982; 22: 57–71. [DOI] [PubMed] [Google Scholar]
- 22.Scotto J, Fraumeni JF, Jr, Lee JA. Melanomas of the eye and other noncutaneous sites: epidemiologic aspects. J Natl Cancer Inst 1976; 56: 489–491. [DOI] [PubMed] [Google Scholar]
- 23.Stroszczynski C, Hosten N, Bornfeld N, et al. Choroidal hemangioma: MR findings and differentiation from uveal melanoma. AJNR Am J Neuroradiol 1998; 19: 1441–1447. [PMC free article] [PubMed] [Google Scholar]
- 24.Char DH, Stone RD, Irvine AR, et al. Diagnostic modalities in choroidal melanoma. Am J Ophthalmol 1980; 89: 223–230. [DOI] [PubMed] [Google Scholar]
- 25.Verbeek AM, Koutentakis P, Deutman AF. Circumscribed choroidal hemangioma diagnosed by ultrasonography. A retrospective analysis of 40 cases. Int Ophthalmol 1995. –1996; 19: 185–189. [DOI] [PubMed] [Google Scholar]
- 26.Brab M, Adam G, Reim M, et al. [Differential intraocular tumor diagnosis in MRI using gadolinium DTPA: value in comparison with other ophthalmologic examination procedures]. Fortschr Ophthalmol 1991; 88: 53–58. [PubMed] [Google Scholar]