Abstract
Objectives
The error rate in the total testing process (TTP) of point-of-care (POC) glucose measurement remains high although a total quality management system has been applied. Quality indicators (QIs) in the TTP of glucose meter were established via risk assessment. Their two-year Six Sigma values were reviewed for quality improvement.
Design
The TTP of POC glucose measurement was mapped to identify risks in key steps. The risks were assessed for their frequency and severity of impact on patient safety. Whenever possible, measurable data from the data management system and other sources was collected to establish QIs for risk monitoring. Average Six Sigma value of each QI in the last two years was calculated for acceptance and for determining corrective action.
Results
29 risks were identified in eight key steps of the TTP. Eight QIs were established for monitoring six risks and three QIs for two accepted risks were established for improving operator testing skill. The QIs had a good coverage to key steps. Two, five and four QIs showed Six Sigma values <3, 3-4 and >4 respectively. Six Sigma values of two QIs related to quality control (QC) testing were improved by using meters with accurate QC sample loading.
Conclusions
The establishment of QIs for glucose measurement by risk assessment with measurable data from the data management system and on Six sigma scale was effective, efficient, and manageable. Most of QIs’ Six Sigma values were between 3 and 5, which could be improved by using upgraded meters.
Keywords: Point-of-care testing, Glucose meter, Quality indicator, Risk assessment, Six sigma
Highlights
-
•
The total testing process of POC glucose measurement was assessed to identify all risks that might impact patient safety.
-
•
QIs that established from data management system monitored the risks related to all of the meters and operators.
-
•
Six Sigma values of QIs provided a straightforward acceptance in their performance evaluation.
-
•
Most of the Six Sigma values of QIs for glucose meters were between 3 and 5 under current total quality management system.
1. Introduction
Glucose meters have been used widely as Point-of-Care Testing (POCT) for monitoring patient blood glucose concentration due to many advantages such as small sample volume, quick turnaround time, and convenience. To ensure the quality of glucose testing, a total quality management system is required for appropriately implementing, managing, and monitoring the measurement. However, the occurrence of analytical and extra analytical errors in the TTP still remains high that certainly affects the quality of results on glucose meters [[1], [2], [3], [4]].
In order to prevent and reduce the errors in testing process, International Organization for Standardization (ISO) 14971:2019 specifies a process of risk management for manufacturers to identify the risks associated with medical devices in the entire life cycle, including in vitro diagnostic (IVD) medical devices, to estimate and evaluate the risks, to control the risks, and to monitor the effectiveness of the controls [5]. The local accreditation requirements for risk management are also increased with the Institute for Quality Management of Healthcare (IQMH) [6]. In risk management, risk assessment is firstly conducted to identify the risks in vulnerable steps of the TTP and to determine the hierarchy of risks that need to be corrected according to their impact on patient safety. Then corrective actions such as procedure change, training or education, and effectiveness monitoring using quality indicators (QIs) are built in specific to the risks. The risk management principles should be applied to identify the risks in the TTP of POCT for quality improvement as well [1,2,4].
Different QIs have been developed to monitor the errors in critical processes of laboratory testing based on particular characteristics of organization such as health care contexts, laboratory’s services, patient population, and staff training [7].The Working Group “Laboratory Errors and Patient Safety” (WG-LEPS) of the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) recommended the harmonization of QIs in laboratory medicine. The Working Group currently is focused on the implementation of an efficient tool for obtaining meaningful data on the errors occurring throughout the TTP and for establishing reliable information about error frequencies and distribution [[7], [8], [9]]. There are a variety of methods in data collection for QI monitoring such as audits, manual recording processes, incident reporting mechanisms, and reports from the laboratory information system (LIS). The use of LIS or middleware is recommended as it provides the easiest and most standardized mechanism of data capture [10].
The poor outcomes in the process can be measured as defects per million and expressed on the Six Sigma scale (0-6). The Six Sigma was initially developed in industry to evaluate the quality of products. It measures the degree of deviation from the goal in any process. Six Sigma scale 4, regardless their complexity, means the average quality. Six Sigma scale 6 represents “world class quality” and contains 3.4 defects in a million events [11]. Therefore, Six Sigma scale has been recognized as a useful tool in quality management systems for determining the quality and continuous process improvement. In laboratory medicine, Six Sigma scale is frequently applied as a benchmark for evaluating the performance of pre-analytical, analytical, and post-analytical processes in Core Laboratory testing [[11], [12], [13]]. Usually, the QIs are presented as the percentage of defects. The percentages sometimes seem low because of a small number of defects occurring in a large number of total events. This may mislead as unimportant if the acceptance criteria are not clearly defined. When QIs are presented on Six Sigma scale, they are straightforward for acceptance and directly correlated to the quality level and customer satisfaction [11]. The QIs with poor Six Sigma values need corrective action for improvement.
Currently the experience of establishing QIs for the testing process of POCT is not as much as Core Laboratory testing. There are very limited studies that can be used for the generation and harmonization of QIs for POC glucose meters. Most of the QIs for POCT were established by a brainstorming approach, using inadequate data and without clearly defined standards. In this study, we applied the approach of risk assessment to identify the risks and to determine their frequency and impact on patient safety in the TTP of POC glucose measurement that was specific to the meters implemented in our POCT Program. The QIs were generated by using measurable data from the data management system or other sources for monitoring the rate of risks and the effectiveness of mitigation actions. The Six Sigma values of these QIs in the last two years were reviewed retrospectively, and were used to evaluate the performance of QIs in the risk control and to identify the priority risks to be focused on immediately.
2. Method and design
2.1. POC glucose meters
A total of 158 hospital glucose meters (Nova StatStrip, Nova Biomedical, Walthum MA. USA) with electrochemistry method were implemented in Kingston Health Sciences Centre (KHSC) under the quality management of the POCT Program. It is a new generation glucose meter with advanced measurement using a very small amount (1.2 μl) of whole blood sample and with a quick reaction time (6 seconds). It is also designed with multiple management functions for better error prevention and regulatory compliance, such as QC lockout, operator lockout, and barcode scanning for identification and reagent lot. In addition, the meter displays error messages in the testing process to indicate inadequate sample volume, removal of strip inappropriately, and impaired strip with automatic test cancelation. It displays an alert on the meter to extreme temperature and if the data transfer is incomplete.
2.2. Data management system and data collection
All glucose meters were connected to the data management system (Telcor QML, Telcor Inc. Lincoln NE. USA) for data recording, transferring, and result reporting in a bidirectional manner. The results of patient and QC samples, and other information relating to patient, patient sample, operator, reagent, and meter are transferred from the meters to Telcor QML when the meters are docked. This links the information of patient, operator, reagent, and meter function status to each testing result for overall reviewing and monitoring by the POCT Program. The features of Telcor QML related to this study include automatically monitoring devices and interfaces, sending alerts, managing configuration, results and interface exceptions, automated operator and patient lists to devices, operator certification tracking and documentation, unlimited data mining, patient and QC results management, and reporting for statistics and metrics.
2.3. Risk assessment
Risk assessment principles and strategies described in CLSI guidelines EP23-A were applied in this study [14]. The process mapping from test ordering to result interpretation and reaction was performed by a group of POCT experts including the Senior Technologist, Manager, and Clinical Biochemist of the POCT Program. The risks were identified following regulatory and accreditation requirements (IQMH), measurement system information from the manufacturer (Nova Biomedical), clinical significance of glucose meter, hospital policies and procedure of POC glucose measurements, publications, and previous issues that occurred in our quality assurance and troubleshooting. The frequency (frequent, probable, occasional, remote and improbable) of risks, the severity of their impact (negligible, minor, serious, critical and catastrophic) on patient safety, and risk acceptance were determined following the guidelines [5,14].
2.4. Measurements of QIs
After the risks were identified and assessed, QIs were established following the frequency, severity of impact of risks, and the availability of measurable data as Table 1. If neither QI nor internal audit was able to be established, the risk monitoring was not available.
Table 1.
Establishment of QIs by the frequency of risk, severity of impact on patient safety and availability of measurable data.
| Frequency of risk or severity of impact on patient safety | Availability of measurable dataa | QI establishment |
|---|---|---|
| Improbable or negligible | Not available | Risk was accepted, no QI needed for monitoring |
| Improbable or negligible | Available | Risk was accepted, QI established for improving operator’s testing skill |
| >Improbable or > negligible | Not available | Risk was not accepted, internal audit established for risk monitoring if possible (not included in this study) |
| >Improbable or > negligible | Available | Risk was not accepted, QI established for risk monitoring |
Measurable data was available from glucose meter data management system or other sources (e.g. laboratory information system, documents maintained in the POCT Program).
2.5. Performance of QIs
The QI’s performance was calculated as percent of defect and also on DPMO (defects per million opportunities) derived Six Sigma scale (minimal 0) by Westgard calculator [15] using the measurable data from the last two years (October 2017 to September 2019). If the defect number was 0, Six Sigma value was expressed as >6. In this study, according to the Six Sigma principle, QI performance was good and acceptable if Six Sigma value was >4. QI performance should be improved if Six Sigma value was 3-4, corrective action should be taken immediately if Six Sigma value was <3.
2.6. Statistical analysis
Data was sorted, calculated, and analyzed in the Excel software (Microsoft, 2010).
3. Results
3.1. TTP of POC glucose measurement
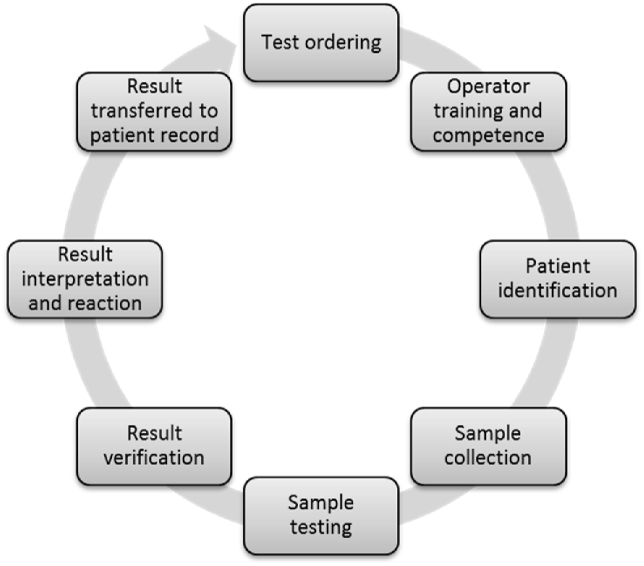
Fig. 1 describes the TTP of POC glucose measurement, which started from test ordering to result interpretation and reaction. After a POC glucose test was ordered by the physician, the certified operator checked patient identifications, scanned the barcode containing identifications or entered the identifications manually into the meter. Following the standard procedure, the operator collected the appropriate type of sample and loaded adequate volume of the sample to a test strip for testing. The glucose meters were validated before the implementation and their analytical performance was monitored by internal quality control, meter to Core Laboratory analyzer comparison and external proficiency testing. The meters required quality control testing to be performed every 24 hours of use and could only be used for patient testing when the quality control results passed the acceptance criteria. The testing strips and quality control materials were stored as per the manufacturer’s instruction. In the results verification, any critical results had to be repeated on the glucose meter and confirmed by Core Laboratory glucose assay according to our POCT policies. The glucose meters should be placed on the docks as soon as the testing was completed, so the results could be transferred to the data management system and patient electronic records simultaneously by interface. The ordering physicians should interpret the results in a timely manner and consider the factors that might affect the measurement. In addition, the discard of used strips and the disinfection of meters should follow on the standard procedure. Based on the characteristics of the glucose meters and the described TTP, eight key steps were categorized in the TTP including test ordering, patient identification, sample collection, sample testing, result verification, result interpretation and reaction, and result transferred to patient record. Unlike Core Laboratory testing, the result interpretation to the glucose measured on meters occurred right after result verification, before the glucose meters were re-docked, and before the results were transferred to patient health records since the glucose meters were used at patient bedside. The operators were involved in many steps including patient identification, sample collection, sample testing, results verification, and result transfer. All of these were included in operator’s training and competence listed in the pre-analytical phase.
Fig. 1.
Total testing process of POC glucose measurement.
3.2. Identification, assessment of risks, and generation of QIs
A total of 29 risks were identified by the risk assessment in all steps in the process mapping, each step containing 1-8 various risks. The risk frequency, severity of impact on patient safety and the rationale were listed in supplemental Table 1. Nine risks with negligible impact on patient safety or with very low frequency were accepted and did not need regular monitoring. Six risks occurring in the process of sample collection, meter operation, and inventory control needed to be monitored, but there was no measurable data for statistical analysis. Internal audits were set up for monitoring this type of risks. Eight QIs were able to be established to regularly monitor six risks on patient identification, test analytical performance, results verification, and data transfer (Table 2). Two risks in the sample/QC testing step were accepted due to the negligible impact on patient safety, so no regular risk monitoring was required. However, since the measurable data for “testing procedure with error messages” and “QC failure” was available in the data management system, three QIs to these risks were able to be generated for improving operator’s testing skill (Table 2). There were six risks with no availability to be monitored by QIs or internal audits at the moment. They were mostly related to test ordering, questioned result verification, and result interpretation.
Table 2.
Eleven QIs generated from risk assessment in the TTP of POC glucose measurement.
| QIs for monitoring risk | Measurements (%) |
|---|---|
| Incorrect patient identification number | Pending results with incorrect patient identification number/total number of patient testing |
| Numeric patient identification for triage used in unauthorized unitsa | Pending results with numeric patient identification for triage from unauthorized units/total number of patient testing |
| Meter analytical performance failure | |
| (1)Meter with total imprecision >10% | Number of meter with imprecision >10%/total number of meter |
| (2)EQA failure | Number of EQA failure/total number of proficiency testing |
| (3)Meter to Core Lab analyzer comparison failure | Number of testing with bias out of laboratory standards/total number of testing in comparison |
| Critical result not repeated on the meterb | Number of critical results not repeated on the same or different meter within a five-minute interval/total number of critical results |
| Critical result not confirmed by Core Laboratory testingb | Number of critical results not confirmed by Core Laboratory testing within a 30-minute interval/total number of critical results |
| Data transfer delayed due to undocking >4h | Number of test with undock time more than 4 hours/total number of patient testing |
| QIs for improving operator’s testing skill | Measurements (%) |
| QC testing failure or procedure with error messages | |
| (1)QC testing procedure with error messages | Number of QC testing flagged with a procedure error message/total number of QC testing |
| (2)QC failure | Number of QC outliers/total number of QC testing |
| Patient testing procedure with error messages | Number of patient testing flagged with a procedure error message/total number of patient testing |
The numeric number designed by the POCT Program for patients in emergency department before registration and without hospital identification number should be only used in authorized units.
All critical values (<2.5 mmol/L or >25 mmol/L) from a glucose meter must be repeated on the meter. If it is the first one of the day for a patient, it must be confirmed by Core Laboratory chemistry analyzer with the venous blood sample.
The metric calculated for QIs was included in Table 2 as well. The measurable data for eight QIs was extracted from the data management system by the Senior POCT Technologist, while for one QI it needed the data from LIS by the assistance of Laboratory IT Specialist. The data of indicators monitoring external proficiency testing and meter to Core Laboratory analyzer comparison was maintained by the POCT Program.
3.3. The performance of QIs in a two-year review
158 POCT glucose meters were implemented in 43 clinical units in the hospital to offer blood glucose monitoring for patient care. About 1500 operators were trained with demonstrated competency of using the glucose meters. During last 24 months (October 2017 to September 2019), the test volume was in the range of 15329 -18910/month with average 16808/month. In May 2019, the meters were replaced by upgraded meters from the same manufacturer with improved operational function, but the same analytical principle (the same strip). The strip port on the meters was changed from the top of the meters to the bottom. When a small drop of QC sample was applied to the end of strip, the extra QC sample (the reaction only requires 1.2 μl samples) would not flow into the meter from the strip port by the force of gravity to affect the testing. Therefore, the upgraded meters had more accurate amount of QC sample loaded on the strips to reduce QC failure and QC variation.
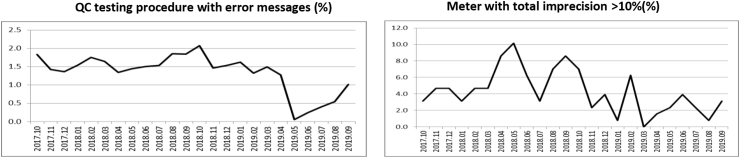
Table 3 showed the monthly or yearly performance of 11 QIs including the range and average of defect percentages, average Six Sigma values, and the possible causes of risks in the last two years. There were 2, 5 and 4 QIs showed average Six Sigma value <3, 3-4 and >4. A small number of samples were tested with incorrect patient identification number or unauthorized numerical patient identification, showing good Six Sigma value of QIs (>4). The Six Sigma values of QIs to “testing procedure with error messages” for QC material and patient samples was 3.62 and 3.16 respectively. When the meters designed with accurate QC sample loading were used, the Six Sigma value of QI to “QC testing procedure with error messages” was improved from 3.62 to 4.08 (data in four months) (Fig. 2). After the wrong level of QC material was excluded, QC failure was consistently within 0.4-1.1% (average Six Sigma value 3.85) when the manufacturer’s QC limits were used. The QC outliers were mostly due to random errors which were corrected by repeating. The critical value repeat and confirmation showed consistently poor Six Sigma values (average 0.5 and 0 respectively). An average of 84.03% of critical glucose results were not repeated on the meter for immediately verification, which did not follow the requirements in the hospital policy. An average of 95.07% of critical results on glucose meters were not confirmed by Core Laboratory testing. Around 94% of results were uploaded to the data management system within 10 minutes of the measurement, and 6% of results were uploaded after four hours because the meters were not re-docked in time. Occasionally, the meters were not put back to docking station after several days. An average of 5.2% (2.3-10.2%) meters showed total imprecision greater than 10%. When the upgraded meters were used, the Six Sigma value of QI to “meter with total imprecision >10%” was improved from 3.13 to 3.55 (Fig. 2). As for the accuracy of measurement, only one of 639 testing in 2019 was out of the accuracy limits (±1 mmol/L or 20%) of the proficiency testing program (average Six Sigma value 4.66). Also, all 70 pairs of results in meter to Core Laboratory analyzer comparison (available in 2019) were acceptable with laboratory standards (the same as the proficiency testing program). The monthly performance of the QIs is presented in Fig. 2 (two QIs) and supplemental Figure 1 (7 QIs).
Table 3.
The performance of QIs for POC glucose measurement in two years.
| QI | Defect% |
Average Six Sigma value | Possible causes of risk | |
|---|---|---|---|---|
| Range in 24 months | Average | |||
| Incorrect patient identification number (monthly) | 0.08-0.21 | 0.13 | 4.51 | Operator non-competence |
| Numeric patient identification for triage used in unauthorized units (monthly) | 0.02-0.19 | 0.08 | 4.66 | Operator non-compliance |
| Meter analytical performance failure | ||||
| (1)Meter with total imprecision >10% (monthly) | 2.3-10.2 | 5.9 | 3.13 | Operator non-competence, meter operation, meter analytical performance |
| (2)EQA failure (yearly) | 0-0.16 | 0.08 | 4.66 | Operator non-competence, meter analytical performance |
| (3)Meter to Core Lab analyzer comparison failure (yearly) (only available in 2019) | 0 | 0 | >6 | Not applicable, comparison was performed by POCT technologists |
| Critical result not repeated on the meter (monthly) | 78.3-87.7 | 84 | 0.5 | Operator non-compliance |
| Critical result not confirmed by Core Laboratory testing (monthly) | 91.1-99.0 | 95.1 | 0 | Operator non-compliance |
| Data transfer delayed due to undocking >4h (monthly) (18 month data available) | 3.0-8.0 | 4.7 | 3.17 | Operator non-compliance |
| QC test failure or procedure with error messages | ||||
| (1)QC testing procedure with error messages (monthly) | 1.3-3.1 | 1.7 | 3.62 | Operator non-competence, meter operation |
| (2)QC failure (monthly) | 0.4-1.1 | 0.95 | 3.85 | Operator non-competence, meter analytical performance |
| Patient testing procedure with error messages (monthly) | 4.2-5.6 | 4.9 | 3.15 | Operator non-competence, meter operation |
Fig. 2.
The improvement of Six Sigma value of two QIs by using upgraded glucose meters.
4. Discussion
The tools of risk management such as Failure Model and Effect Analysis (FMEA) have been used flexibly to identify and assess the risks or failure modes specific to quality needs, for example in the TTP of Core Laboratory testing, for frequently unsuccessful testing, or for procedures with a high degree of complexity [[16], [17], [18]]. In this study we mapped the TTP of POC glucose measurement in our POCT Program and conducted the risk assessment using the tools of FMEA following the process recommended in CLSI guidelines (EP23-A). Although only 11 QIs were established for monitoring risks and improving operators’ testing skill, they still covered the risks in the key steps of TTP such as patient identification, testing process, result verification/confirmation, result transfer and analytical performance monitoring. The quality requirements of POCT are similar to Core Laboratory testing. However, the detailed procedures of POCT and Core Laboratory testing are not the exact same since POCT is performed outside the laboratory by non-lab professionals and on different types of devices. Test result interpretation and reaction may occur before the results are transferred and recorded for laboratory review. Users’ competence apparently creates new challenges to the quality of POCT. Operation procedure, technology, QC setting, management function, analytical performance, and intended use are also varied between manufacturers and types of POCT instruments. In addition, procedures such as for correct patient identification, critical value verification, and users’ training are usually specific to hospitals and cannot be simply standardized. Therefore, the frequency, distribution, and impact of risks on patient safety in the TTP of POCT are different with those in the Core Laboratory, and may be different between the POCT Programs and the devices. Usually, multiple POC glucose meters are used at different units and by a large number of operators. The data collection on all meters is a challenge. The data management system for POCT is an effective tool to overcome this challenge and to improve results recording, device management, operator competency, and quality assurance [19]. The captured data related to testing from the data management system covers all meters and all operators in the institution. In addition, the comprehensive data was able to be used to generate QIs for different types of defects. In this study two risks in the sample/QC testing step were accepted with negligible impact on patient safety and no regular risk monitoring was required. However, since the measurable data related to the risks was available in the data management system, three QIs to these risks were able to be generated easily. They were perfect examples to be used for improving operator’s general testing skill and competency.
In this study there were 2, 5 and 4 QIs that showed average Six Sigma value <3, 3-4 and >4 respectively. No standardized Six Sigma values of QIs for the POC glucose meter were available for reference, and it seems that generally the Six Sigma values of QIs were slightly lower than Core Laboratory testing. In IFCC External Quality Assurance Program (EQAP), the performance specifications for some QIs of the key processes of Core Laboratory testing were listed. Most of the Six Sigma values of high performance were greater than 5 (Six Sigma values were calculated from defect percentages by the author of this study), and Six Sigma values for low performance were between 4 and 5. The QI of unacceptable performances in EQA-PT schemes showed the lowest performance with high and low performance specifications of Six Sigma value at 3.48 and 3.27 respectively [8]. In this study the analytical performance of glucose meters including imprecision and accuracy was monitored by three QIs for “meter with total imprecision >10%”, “EQA failure”, and “meter to Core Laboratory comparison failure”. The Six Sigma value (4.45) of “EQA failure” was at high specification performance according to IFCC EQAP [8]. The current EQA program uses ±20% or 1.0 mmol/L as the acceptance limits of proficiency testing, which may be too loose for the new generation of glucose meters. In meter to Core Laboratory chemistry analyzer comparison, no meter failed with laboratory standards that were the same as the EQA limits. If the CLSI recommended targets of ±12.5% (glucose ≥5.6 mmol/L) or ±0.67 mmol/L (<5.6 mmol/L) [20] were used, the EQA failure rate was increased (1.5%) in 2018 with Six Sigma value decreasing to 3.66 but remaining the same (4.45) in 2019.
The QIs for “critical results were not repeated on the meter” and “critical results not confirmed by Core Laboratory testing” showed poor Sigma performance (0.5 and 0). The latter might be slightly over-estimated since a small amount of second and subsequent critical values of the day for a patient were not able to be excluded in data analysis. The low rate of confirming critical value was also observed in the other hospitals [21]. A variety of practices were identified in POCT glucose critical value verification or confirmation. Repeating the critical glucose results on meters or confirming the results by Core Laboratory chemistry analyzer can help in identifying the errors and reducing the impact of falsely high or low results on patient care [22]. These procedures were highly dependent on users’ awareness and behavior, which corresponded to the high error rate in POCT resulting from non-compliance with policy/procedure [1,[21], [22], [23]]. A separate project has been initiated to identify the patterns of repeating or confirming critical results and to review the analytical performance of glucose meters at low and high critical levels, followed by the education or communication with users and possible policy changes.
For five QIs with Six Sigma values between 3 and 4, operator’s non-compliance, competence, meter operation, or analytical performance might be the possible causes of the risks. The operator’s non-compliance and non-competency are the major sources of errors in POC glucose measurement as identified [1,2]. Although the risks were addressed in initial training, competency maintenance, and education reminders for unit managers and operators, the Six Sigma values of most of QIs in two years were not improved. The new generation of glucose meter has been designed to have certain management functions or internal checks to prevent the errors that commonly occur. For example, barcode scanning for entering operators’ and patients’ identifications as well as reagent lot numbers, has significantly reduced the errors that resulted from non-trained operator, wrong patient identification, and expired reagent [24,25]. Also, the connectivity of meters to the data management system allows the information of patient, operator, reagent, and meter function status to be linked to every test result for overall reviewing and monitoring by the POCT Program [19]. These improvements greatly reduce the errors in the TTP of glucose meters [25,26]. Our data showed that the Six Sigma values of two QIs reflecting the QC testing procedure and meter imprecision was increased from 3.62 to 4.08 and from 3.13 to 3.55 respectively, when the meters with accurate QC sample loading were implemented.
There were many challenges in establishing the QIs for the POC glucose measurement. 1) The risk identification and assessment should be completed by the group, including users and POCT experts who understand the process very well in order to establish the QIs monitoring the risks specific to the hospital clinical environment. 2) There is no standard way to analyze a large amount of measurable data and to generate specific reports for targeted QIs in regular review. We used Microsoft Excel for sorting and analyzing extracted data, which made the QI monitoring a manageable task. 3) The data of QI for monitoring critical value confirmation by Core Laboratory testing needed the assistance of laboratory informatics specialist to extract the data from laboratory reports. The multidisciplinary cooperation is necessary. 4) A relative high error rate occurs in test ordering, result interpretation, and follow-up [27]. Unfortunately, currently they are not able to be monitored.
5. Conclusions
This study showed a systematic approach to identify and assess the risks in the TTP of POC glucose measurement and to establish appropriate QIs. Although the measurable data could not be obtained for every risk, the current data management system still provided comprehensive and accurate data for a satisfactory coverage to the key steps in TTP. The Six Sigma values facilitated with the determination of performance of QIs and the improvement of risk control. The approach is demonstrated to be effective, efficient, and manageable in the quality improvement of POC glucose meters. It may also be a useful approach in harmonizing the QIs for POCT. Currently, most of QIs’ Six Sigma values in the POC glucose measurement were between 3 and 5 under the quality management system. However, the risks should be able to be mitigated by upgrading meter functionality, improving operators’ compliance and competence, and revised policy or procedure.
Author statement
Anne Vincent: Methodology, Data curation, Writing – original draft, Writing – review & editing, Donnah Pocius: Methodology, Resources, Writing – review & editing, Yun Huang: Conceptualization, Methodology, Formal analysis, Writing – original draft, Writing – review & editing.
Declaration of competing interest
There are no known conflicts of interest associated with this publication.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.plabm.2021.e00215.
Appendix A. Supplementary data
The following is the Supplementary data to this article:
References
- 1.Nichols J.H. Blood glucose testing in the hospital: error sources and risk management. J. Diabetes. Sci. Technol. 2011;5:173–177. doi: 10.1177/193229681100500124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Plebani M. Does POCT reduce the risk of error in laboratory testing? Clin. Chim. Acta. 2009;404:59–64. doi: 10.1016/j.cca.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 3.O’Kane M.J., McManus P., McGowan N., Lynch P.L. Quality error rates in point-of-care testing. Clin. Chem. 2011;57:1267–1271. doi: 10.1373/clinchem.2011.164517. [DOI] [PubMed] [Google Scholar]
- 4.Nichols J.H. Risk management for point-of-care testing. EJIFCC. 2014;25:154–161. [PMC free article] [PubMed] [Google Scholar]
- 5.International Organization for Standardization, ISO 14971:2019 Medical Devices - Application of Risk Management to Medical Devices.
- 6.Institute for Quality Management in Healthcare . 2019. Medical Laboratory Accreditation Requirements. Version 8. [Google Scholar]
- 7.Plebani M., Sciacovelli L., Aita A. Quality indicators for the total testing process. Clin. Lab. Med. 2017;37:187–205. doi: 10.1016/j.cll.2016.09.015. [DOI] [PubMed] [Google Scholar]
- 8.Sciacovelli L., Panteghini M., Lippi G., Sumarac Z., Cadamuro J., Galoro C.A.O., Pino Castro I.G.D., Shcolnik W., Plebani M. Defining a roadmap for harmonizing quality indicators in laboratory medicine: a consensus statement on behalf of the IFCC working group “laboratory error and patient safety” and EFLM task and finish group “performance specifications for the extra-analytical phases”. Clin. Chem. Lab. Med. 2017;55:1478–1488. doi: 10.1515/cclm-2017-0412. [DOI] [PubMed] [Google Scholar]
- 9.Sciacovelli L., Aita A., Plebani M. Extra-analytical quality indicators and laboratory performances. Clin. Biochem. 2017;50(10-11):632–637. doi: 10.1016/j.clinbiochem.2017.03.020. [DOI] [PubMed] [Google Scholar]
- 10.West J., Atherton J., Costelloe S.J., Pourmahram G., Stretton A., Cornes M. Preanalytical errors in medical laboratories: a review of the available methodologies of data collection and analysis. Ann. Clin. Biochem. 2017;54:14–19. doi: 10.1177/0004563216669384. [DOI] [PubMed] [Google Scholar]
- 11.Nevalainen D., Berte L., Kraft C., Leigh E., Picaso L., Morgan T. Evaluating laboratory performance on quality indicators with the six sigma scale. Arch. Pathol. Lab. Med. 2000;124:516–519. doi: 10.5858/2000-124-0516-ELPOQI. [DOI] [PubMed] [Google Scholar]
- 12.Giménez-Marín A., Rivas-Ruiz F., Mdel Pérez-Hidalgo M., Molina-Mendoza P. Pre-analytical errors management in the clinical laboratory: a five-year study. Biochem. Med. 2014;24:248–257. doi: 10.11613/BM.2014.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xia Y., Wang X., Yan C., Wu J., Xue H., Li M., Lin Y., Li J., Ji L. Risk assessment of the total testing process based on quality indicators with the Sigma metrics. Clin. Chem. Lab. Med. 2020;58:1223–1231. doi: 10.1515/cclm-2019-1190. [DOI] [PubMed] [Google Scholar]
- 14.Clinical and Laboratory Standards Institute, EP23-A . 2012. Laboratory Quality Control Based on Risk Management, Approved Guideline. [Google Scholar]
- 15.Westgard J.O. 2020. Six Sigma Calculators.https://www.westgard.com/six-sigma-calculators.htm Accessed Aug. 20, 2020. [Google Scholar]
- 16.Jiang Y., Jiang H., Ding S., Liu Application of failure mode and effects analysis in a clinical chemistry laboratory. Clin. Chim. Acta. 2015;448:80–85. doi: 10.1016/j.cca.2015.06.016. [DOI] [PubMed] [Google Scholar]
- 17.Magnezi R., Hemi A., Hemi R. Using the failure mode and effects analysis model to improve parathyroid hormone and adrenocorticotropic hormone testing. Risk Manag. Healthc. Pol. 2016;9:271–274. doi: 10.2147/RMHP.S117472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Intra G., Alteri A., Corti L., Rabellotti E., Papaleo E., Restelli L., Biondo S., Garancini M.P., Candiani M., Viganò P. Application of failure mode and effect analysis in an assisted reproduction technology laboratory. Reprod. Biomed. Online. 2016;33:132–139. doi: 10.1016/j.rbmo.2016.05.008. [DOI] [PubMed] [Google Scholar]
- 19.Fung A.W.S. Utilizing connectivity and data management system for effective quality management and regulatory compliance in point of care testing. Pract. Lab. Med. 2020;22 doi: 10.1016/j.plabm.2020.e00187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Clinical and Laboratory Standards Institute . third ed. 2013. POCT12-A3: Point-of-care Blood Glucose Testing in Acute and Chronic Care Facilities. [Google Scholar]
- 21.Shaw J.L.V., McCudden C.R., Colantonio D.A., Booth R.A., Lin D.C., Blasutig I.M., Moran T., Trofimczuk D., Carriere C., Gharra A., Portelance C., Tremblay C., Dupaul D., Breton N., Angelkovski M., Jariwala C., Embleton M., Campbell C., Groulx K., Larmour K. Effective interventions to improve the quality of critically high point-of-care glucose meter results. Pract. Lab. Med. 2020;(22) doi: 10.1016/j.plabm.2020.e00184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schifman R.B., Howanitz P.J., Souers R.J., Point-of-care glucose critical values a Q-probes study involving 50 health care facilities and 2349 critical results, Arch. Pathol. Lab Med. 140 (2–16) 119–124. [DOI] [PubMed]
- 23.Meier F.A., Jones B.A. Point-of-Care testing error sources and amplifiers, taxonomy, prevention strategies, and detection monitors. Arch. Pathol. Lab. Med. 2015;129:1262–1267. doi: 10.5858/2005-129-1262-PTESAA. [DOI] [PubMed] [Google Scholar]
- 24.Nichols J.J., Bartholomew C., Brunton M., Cintron C., Elliott S., McGirr J., Morsi D., Scott S., Seipel J., Sinha D. Reducing medical errors through barcoding at the point of care. Clin. Leadersh. Manag. Rev. 2004;18:328–334. [PubMed] [Google Scholar]
- 25.Lewandrowski K., Gregory K., Macmillan D. Assuring quality in point-of-care testing: evolution of technologies, informatics, and program management. Arch. Pathol. Lab. Med. 2011;135:1405–1421. doi: 10.5858/arpa.2011-0157-RA. [DOI] [PubMed] [Google Scholar]
- 26.Lewandrowski E., Mac Millan D., Misiano D., Tochka L., Lewandrowski K. Process improvement for bedside capillary glucose testing in a large academic medical center: the impact of new technology on point-of-care testing. Clin. Chim. Acta. 2001;307(1–2):175–179. doi: 10.1016/s0009-8981(01)00462-4. [DOI] [PubMed] [Google Scholar]
- 27.Plebani M. The detection and prevention of errors in laboratory medicine. Ann. Clin. Biochem. 2010;47(Pt 2):101–110. doi: 10.1258/acb.2009.009222. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.