Abstract
Objective:
Humpback (hpbk) mice harbor a pathogenic mutation in the Notch3 gene and can serve as a beneficial animal model for investigating human myopathy, kyphosis, and developmental disorders, including lateral meningocele syndrome. Detection of the point mutation in hpbk mice is important for maintaining strains and scrutinizing genetic rescues, especially considering that homozygous mice are infertile and indistinguishable from their littermates at a young age. This study aimed for the development of a novel, precise, and time-saving genotyping method to identify the mutation in hpbk mice.
Materials and Methods:
In order to study the hpbk mouse line, we describe how we applied several tools, including quantitative polymerase chain reaction (qPCR), multiplex tetra-primer amplification-refractory mutation system (ARMS-PCR) and Sanger sequencing, toward the recognition of heterozygous and homozygous mice.
Results:
The Notch3 mutation was clearly identified using qPCR and ARMS assays, but the latter was a more precise and cost-effective approach. The lengths of the ARMS-PCR amplicons are 210 bp and 164 bp for the wild-type and hpbk alleles, respectively. Moreover, the genotyping results for each mouse were corroborated by Sanger DNA sequencing.
Conclusion:
Our newly developed PCR-based ARMS system affords a swift and precise way to genotype the hpbk mice. ARMS-PCR does not rely on any advanced equipment and is useful as a genotyping method for other model organisms that harbor a pathogenic variant.
Keywords: Lateral meningocele syndrome, Notch3 mutation, ARMS, genotyping, skeletal disease
Introduction
Humpback (hpbk) mice can be used as precious animal models for investigating developmental disorders related to myopathy and kyphosis. They carry a single nucleotide mutation (G/A) in the Notch3 gene, which may be of interest for studying the role of Notch signaling dysregulation in human diseases [1,2]. The Notch3 protein belongs to the evolutionarily conserved Notch family (Notch1–Notch4), and is a transmembrane receptor that controls cell fate decisions and maintains homeostasis for almost all organs, such as the skeleton and muscle [3,4]. Gain-of-function mutations in the human Notch3 gene are associated with lateral meningocele syndrome (LMS), also known as Lehman syndrome [5,6] and infantile myofibromatosis [7]. Mice homozygous (hpbk/hpbk) for the mutation are comparable in appearance to wild-type mice (+/+) when born, but develop musculoskeletal phenotypes such as a kyphotic curved spine soon after weaning [8]. They become easily identifiable at approximately 5 weeks of age and are infertile. Heterozygote mice (hpbk/+) are indistinguishable from their wild-type littermates. Because homozygous mice are infertile, these mutant mice’s maintenance requires either homozygous ovarian reproduction or reproduction between heterozygous pairs. Therefore, to detect the mutant allele in young mice early and preserve and study this model, it is essential to develop a swift and precise genotyping method.
In this study, we established three methods for recognizing +/+, hpbk/+, and hpbk/hpbk mice, including the use of quantitative polymerase chain reaction (qPCR), the multiplex tetra-primer amplification-refractory mutation system (ARMS-PCR), and Sanger sequencing, respectively. Among them, the original ARMS-PCR system amplified wild-type and mutant alleles in two separate PCR reactions [9]. Here, we applied a simple multiplex ARMS-PCR in a single PCR reaction. Complex equipment is not required [9–11]. Moreover, our data demonstrate that ARMS-PCR analysis is a precise and time-saving method to identify mutation alleles in hpbk mice.
Materials and Methods
Ethical approval
Animals were used in accordance with the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals. Our animal protocol (No. 150-08-21C) was approved by the Sanford Research Institutional Animal Care and Use Committee.
Animal radiographic assessment
Heterozygote hpbk/+ mice (C57BL/6J-Notch3hpbk/GrsrJ) were obtained from Jackson Laboratories (JAX, stock #005330). Whole-body x-ray for mutant mice and their littermate’s musculoskeletal phenotypes were analyzed using a Bruker Xtreme imaging system (Bruker, Billerica, MA).
Mouse genomic DNA isolation
One-millimeter mouse-tail tip was deposited into 0.2 ml Eppendorf tubes. Genomic DNA isolation from these samples was carried out using a modified method described previously [12]. Briefly, 50 μl of alkaline lysis reagent (25 mM NaOH) was added to the samples in the tubes. The samples were incubated on a thermocycler at 95°C for 30 min to expose genomic DNA. After being cooled to room temperature, the tissue lysate was centrifuged at 14,000 g to separate the debris. The supernatant was either immediately analyzed or stored at −20°C for later use. Toe samples from postnatal mice at less than 10 days old were analyzed following the same procedure.
ARMS-PCR, quantitative PCR, and Sanger sequencing
Standard PCR and Sanger sequencing were carried out as described previously [13,14]. For the ARMS-PCR assay, the PCR reaction (total volume, 24 μl) contained 1 μl of genomic DNA, 12 μl of AccuStart™ II GelTrack PCR SuperMix (QuantaBio, 89235-012), 1 μl of the inner mixed primers (10 μM of N3IFor and N3IRev), 1 μl of the outer mixed primers (1 μM of N3OFor1 and N3ORev1), and 9 μl of water. Amplification of genomic DNA was achieved in a T100 thermocycler (Bio-Rad) with the following program: an activation step of the DNA polymerase for 5 min at 94°C, 30 cycles of DNA amplification with each cycle including 30 sec at 94°C, 45 sec at 63°C, and 60 sec at 72°C, and a final elongation step (10 min at 72°C). The size and yield of each amplicon was resolved by standard electrophoresis on 1.5% agarose gels containing ethidium bromide. The gel pictures were taken by a ChemiDoc™ MP Imaging System (Bio-Rad). For qPCR assay, amplicons were run and analyzed by an Applied Biosystems 7,500 real-time PCR system and the dedicated software provided by the thermocycler manufacturer (Thermo Fisher). The qPCR reaction (total volume, 20 μl) contained 1 μl of genomic DNA, 10 μl 2X Power SYBR Green PCR Master Mix (Fisher, 4367659), 1 μl of the mixed primers (10 μM of N3MuP3F, and N3MuP3R), and 8 μl of water. We used the β-actin gene (primers seen in Table 1) as an internal control for qPCR assay. We used a pair of primers (seen in Table 1) to amplify genomic DNA containing the Notch3 mutation. Sanger direct DNA sequencing was carried out by Eurofins Genomics.
Table 1. Primers for identification of Notch3 gene G/A point mutation.
| Purpose | Primer name | Primer sequence (5′−3′) | Concentration | Genotyping pattern |
|---|---|---|---|---|
| PCR for sequencing | FGBC53 | Forward: ATTCTCATCAGGAACCGCTCCA | 10 uM | 277 bp |
| N3MuP3R | Reverse: CACTCTGTGGGTCCTGTTCC | |||
| ARMS-PCR primer set | N3OFor1 | Forward outer primer: CATAATGTCCTGCCATGGCTTTTGTCG | 1 uM | 319 bp (outer common) |
| N3ORev1 | Reverse outer primer: AACCAGAACACTCTGTGGGTCCTGTTCC | 1 uM | ||
| *N3IFor | Inner F primer (A allele): CCGATGTCAATGCAGTGGATGAGCTcGA | 10 uM | 164 bp (A allele-mutant allele-specific) | |
| *N3IRev | Inner R primer (G allele) : CCCTCCCCATCCCCTCCGCAGCACTaAc | 10 uM | 210 bp (G allele-wild-type allele-specific) | |
| Sanger sequencing | FGBC53 | ATTCTCATCAGGAACCGCTCCA | 10 uM | Mutant-specific forward primer |
| qPCR and ARMS | β-actinFor | Forward: GGCTGTATTCCCCTCCATCG | 10 uM | 241 bp |
| β-actinRev | Reverse: CCAGTTGGTAACAATGCCATGT | |||
| qPCR (genomic DNA) | *N3MuP3F | Notch3 F: AATGCAGTGGATGAGCTTGA | 10 uM | 148 bp (A allele-mutant allele-specific) |
| N3MuP3R | Notch3 reverse: CACTCTGTGGGTCCTGTTCC | 10 uM |
The original point mutation is indicated by a underlined letter. Specificity is increased by introducing a deliberate second mismatch at position-2 from the 3’ end of both inner forward and reverse primers, which are indicated in lowercase.
Results and Discussion
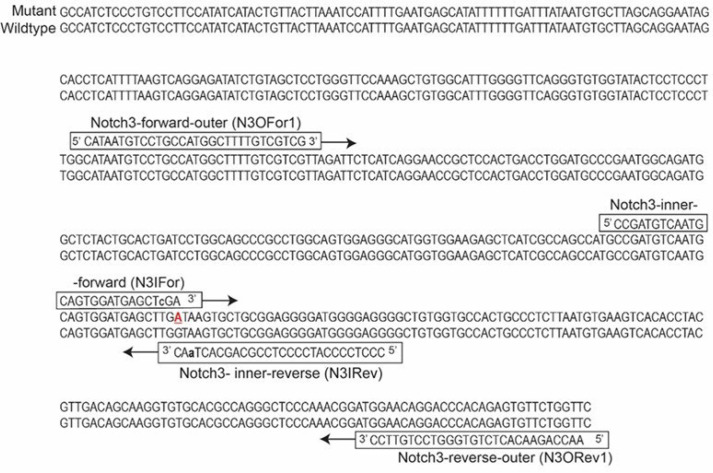
In order to design primers for genotyping hpbk mice, we first retrieved the normal genomic DNA sequence for the murine Notch3 gene from NCBI (Gene ID: 18131). The hpbk mutation is a pathogenic variant (Guanine “G” → Adenine “A”) in the splice donor site at the exon 31-intron 31 boundary, presumptively resulting in a truncated Notch3 protein. We constructed the hpbk mutant gene sequence employing a commercial software called SnapGene@ (available at snapgene.com) (Fig. 1). With this mutant DNA sequence, we exploited a web-based program called NCBI Primer-Blast tool and designed primers for ARMS-PCR, quantitative PCR, and Sanger sequencing assays (https://www.ncbi.nlm.nih.gov/tools/primer-blast/index.cgi) [15]. Generally, primer criteria included limiting the GC content to 40%–60%, primer lengths to 28 base pairs (bp), fragment sizes to the range of 150–400 bp, and paired primers with a similar melting temperature. Specifically, for ARMS-PCR primers, the forward and reverse inner primers target the sequence, including the mutation site. Additionally, we introduced a 3’ terminus mismatch (T-to-C and T-to-A for forward and reverse inner primers, respectively, at position −2 from the 3’ terminal ends of the two inner primers (lowercase in Fig. 1) (Table 1). Extra mismatch can increase PCR specificity to detect the hpbk mutation. Notably, we also attempted to design ARMS primers using another published tool but failed to obtain them [16]. For quantitative PCR primers, we designed several pairs of primers and found one pair of primers (N3MuP3F and N3MuP3R) specifically amplifying the mutant “A” allele but not the wild-type “G” allele. All primers established and used in this study are listed in Table 1.
Figure 1. Location of PCR primers and partial sequences for the Notch3 receptor mutation and wild-type alleles (NCBI, Gene ID: 18131). The primers for ARMS-PCR system were designed using NCBI Primer-Blast tool. Underline indicates the point mutation (G→A) in the splice donor site at the exon 31-intron 31 boundary of the Notch3 receptor. Specificity is increased by introducing a deliberate second mismatch at −2 base pairs (bp) from the 3’ end of both inner forward and reverse primers, which are indicated in lowercase.
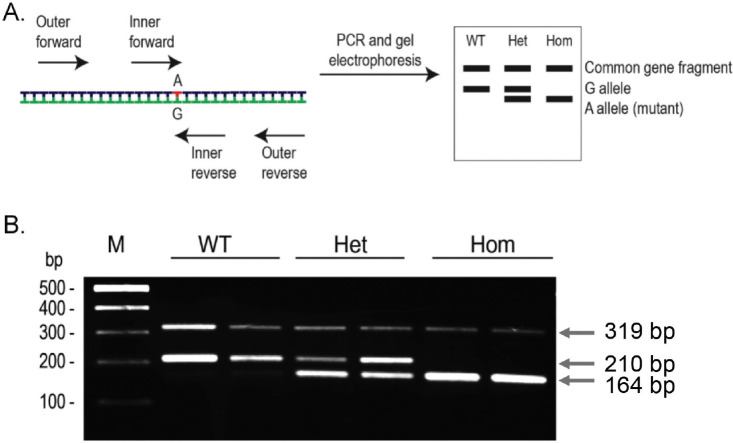
Our Notch3 ARMS-PCR pairing forward outer primer (i.e., N3OFor1) and reverse outer primer (i.e., N3ORev1) results in a 319-bp PCR product from all wild-type and mutant mice, which can be used as a quality control for the PCR reagents (Fig. 2A). Pairing the forward inner and reverse outer primers (i.e., N3IFor and N3ORev1) produces a 164-bp amplicon, which is specific for the mutant allele. Moreover, pairing N3OFor1 and the reverse inner primer (i.e., N3IRev) produces a 210-bp amplicon, which is specific for the wild-type allele. Figure 2B shows a representative PCR result from six mice bearing three genotypes [i.e., wild-type (WT, or +/+), hpbk/+ (or Het), and hpbk/hpbk (or Hom)]. We observed that all six mice (two for each genotype) have a common band of 319-bp. We also observed that wild-type mice have a specific 210-bp band, hpbk/hpbk mice have a specific 164-bp band, and hpbk/+ mice have both. Therefore, our data support that the method we developed in this study has a high degree of specificity and reproducibility.
Figure 2. Detection of the hpbk mutation by the tetra-primer ARMS-PCR analysis. (A) Schematic summary of ARMS-PCR primer design and amplicon gel patterns for the different genotypes. The two outer primers amplify a common large fragment of the Notch3 gene that contains a G→A point mutation (red and underlined). The remaining two inner primers amplify the two allelic states (i.e., in a G→A point mutation, one primer will amplify the A allele (homozygous mutant, Hom) and the other the G allele (wild-type, WT) of Notch3 gene. (B) Genotyping of hpbk mice through tetra-primer ARMS-PCR. M: 100 bp ladder.
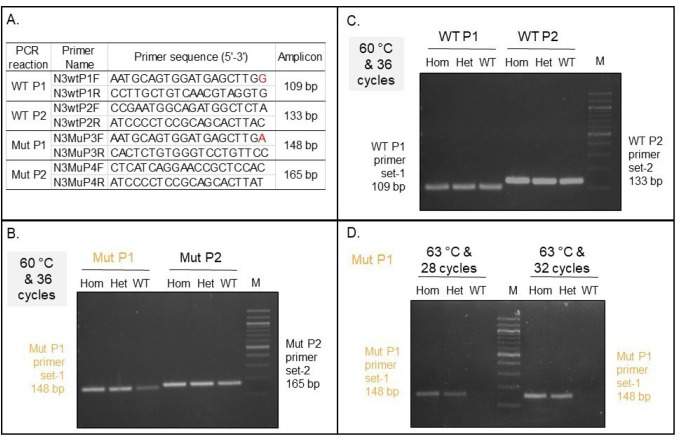
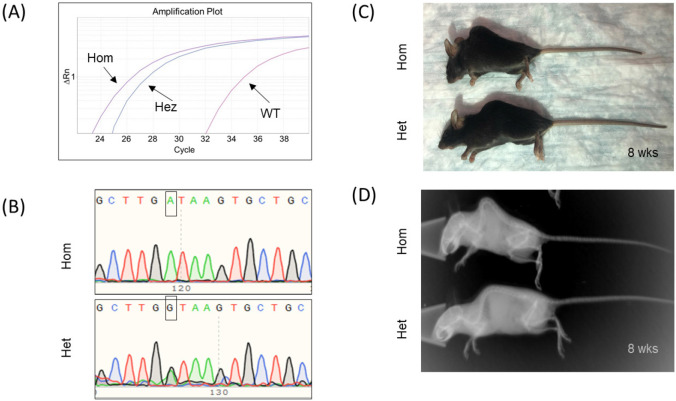
We have also developed two other methods for genotyping hpbk mice, including qPCR and Sanger sequencing, which validate our ARMS-PCR approach. Using the NCBI Primer-Blast tool, we first designed and tested four pairs of PCR primers (two for the wild-type allele and two for the hpbk allele) (Fig. 3A). We found that a pair of primers, namely Mut P1 (N3MuP-3F and -3R), but not Mut P2, detected the hpbk mutant allele. Figure 3B shows that both hpbk/+ and hpbk/hpbk mice have a specific 148-bp band, which wild-type mice do not have in the gel image. Two pairs of primers (i.e., WT P1 and P2) targeting the wild-type allele failed at distinguishing it from the hpbk allele (Fig. 3C). We further optimized PCR cycles (28 vs. 32 cycles) at annealing temperature (63°C vs. 60°C) for the Mut P1 primers and found that there are differential production of amplicons from hpbk/+ and hpbk/hpbk mice under the PCR condition 28 cycles and 63°C (Fig. 3B and 3D). After optimizing Mut P1 primers, we carried out qPCR analysis and found hpbk/+ and hpbk/hpbk mice can be quantitatively distinguished (Fig. 4A). However, because there is only one copy of the mutant DNA allelic difference between them, it is sometimes difficult to accurately analyze the genotype. To overcome this drawback, we in parallel developed our Sanger PCR sequencing assay, which is different from a genotyping protocol accessible from Jackson Laboratory. We designed and applied a pair of PCR primers (FGBC53 and N3MuP3R) to generate a 227-bp amplicon. After sequencing them with Eurofins Genomics, we analyzed and verified the results using the SnapGene software. The representative sequencing chromatogram in Figure 4B shows that hpbk/hpbk mice only carry mutant alleles (point mutation “A”) (top panel), while hpbk/+ mice carry both mutant and wild-type alleles (“A” and “G”) (bottom panel). On the one hand, we established qPCR and Sanger sequencing detection assays and applied them to verify the results produced by our ARMS-PCR method, but on the other hand, we concluded that although qPCR analysis can accurately separate wild-type alleles from mutant alleles (Fig. 3D), it may result in false positives to distinguish between hpbk/+ and hpbk/hpbk mice (Fig. 4A). In addition, although Sanger sequencing is very accurate to distinguish hpbk/+ and hpbk/hpbk mice (Fig. 4B), it is time-consuming and more expensive than the ARMS-PCR method.
Figure 3. Optimization of the qPCR conditions with newly designed primers. (A) A list of primers (two pairs for the wild-type allele and two pairs for the hpbk allele), primer sequence, and size of amplicons. Red colors indicate the point mutation base pair (G–A). (B) A representative gel image for PCR products from tails of three mice (one per genotype) using two pairs of wild-type primers (WT P1 and WT P2) with PCR conditions (annealing temperature at 60°C and a 36-cycle reaction). (C) A representative gel image for PCR from three mice (one per genotype) using two pairs of primers (mutant Mut P1 and Mut P2) under the same PCR condition (annealing temperature at 60°C and a 36-cycle reaction). (D) Optimization of the PCR cycles (28 and 32) using the same annealing temperature (63°C) for the Mut P1 pair of primers.
Figure 4. Detection and verification of hpbk mutation with qPCR and Sanger sequencing methods. (A) An example of a qPCR amplification plot showing homozygous (purple), heterozygous (blue), and wild-type (pink) mice with values of Ct. (cycle threshold) at 24.07, 25.39, and 32.76, respectively. (B) Representative Sanger sequencing chromatograms showing a mutation allele (point mutation “A”) in a homozygous hpbk/hpbk mouse (top panel) and a heterozygous mouse (bottom panel) with a black arrow pointing to “G/A” mutation site and wild-type allele. (C) Photo of 8-week-old Hom and their Het littermate. (D) X-ray image of 8-week-old Hom and their Het littermate.
The genotyping methods developed in this study may further promote the research on the mechanism and treatment of diseases related to the gain-of-function mutations in Notch3, such as LMS. Lehman et al. [17] first described the vertebral and musculoskeletal abnormalities in LMS patients in 1977. Intending to understand the skeletal phenotype of hpbk mice, we carried out a whole-body x-ray scan. The representative radiographic images in Figure 4C and D shows that when compared with hpbk/+ mice, hpbk/hpbk mice develop a hunched back and curved spine by about 4 weeks of age, which is consistent with the patient’s symptoms. We also observed by both naked eyes and radiographs that, at least until hpbk/+ mice reached 4 months of age, they are no different from wild-type littermates. Future musculoskeletal and molecular studies in homozygous and heterozygous mice may substantiate whether this model resembles the human disease.
In addition to the number of PCR cycles and annealing temperature, the hpbk multiplex ARMS-PCR system may also be affected by many other factors, including the ratio of two pairs of primers in one reaction, the method of extracting genomic DNA samples, and storage time and conditions. Our study found that 1:10 is the best ratio of the outer and inner primer sets (i.e., N3OFor1/Rev1 and N3IFor/Rev) (Figs. 2B and 5). To quickly exact genomic DNA from the tip of the mouse tail, we improved upon a method called alkaline lysis and confirmed that it is a fast, effective, and cost-efficient method for DNA isolation [18]. Considering that storage time and conditions may affect the method’s sensitivity and stability, we tested 30 long-term stored samples through ARMS-PCR analysis. These samples were stored at −20°C for several months to 2 years. As shown in Figure 5, although we observed that the 319-bp band in the gel image was weaker in some samples, we could easily identify all 30 mice’s genotypes. Of course, if necessary, we can optimize one of the above factors to ensure genotyping accuracy. Altogether, we have proved that our ARMS-PCR system is an accurate and sensitive method.
Figure 5. Sensitivity and stability test of our ARMS-PCR genotyping method. Thirty genomic DNA samples were tested using our established genotyping method. These genomic DNA samples were run after storing a few months to up to 2 years kept at −20°C after extraction. These results indicate that our genotyping method can work well even with old samples. M: 100 bp DNA ladder (the most left and right lanes). WT: 319 + 210 bp, wild-type mice; Het: 319 + 210 + 164 bp, heterozygous mice; Hom: 319 + 164 bp, mutant homozygous mice. Each lane represents one independent sample.

Conclusion
This study described and compared several methods used to detect the genotype of hpbk mice, including ARMS-PCR, qPCR, and Sanger sequencing. We have proven that ARMS-PCR is the most cost-effective and fastest genotyping method, and its use may aid in studying developmental disorders associated with Notch3 mutations.
List of abbreviations
Humpback (hpbk); quantitative polymerase chain reaction (qPCR); amplification-refractory mutation system (ARMS); National Institutes of Health (NIH).
Acknowledgments
This research was supported by the NIH COBRE grants P20 GM103620 (D.A. Pearce), P20 GM103548 (K. W. Miskimins), and R25 HD097633 (L. Lee). This work was partially supported by T. Denny Sanford Collaborative Research Award (J. Westendorf and J. Tao).
Conflict of interest
The authors declare no financial conflict of interests.
Authors’ contributions
HT and JT designed the study, interpreted the results, and arranged the publication draft. HT, TR, AV, YC, DC, and JT were involved in the collection of data and contributed to paper preparation. JT and JW took part in funding acquisition and critically editing this manuscript. All authors reviewed the manuscript individually and approved the final manuscript.
References
- [1].Mašek J, Andersson ER. The developmental biology of genetic Notch disorders. Dev Suppl. 2017;144(10):1743–63. doi: 10.1242/dev.148007. https://doi.org/10.1242/dev.148007. [DOI] [PubMed] [Google Scholar]
- [2].Fairfield H, Gilbert GJ, Barter M, Corrigan RR, Curtain M, Ding Y, et al. Mutation discovery in mice by whole exome sequencing. Genome Biol. 2011;12(9):R86. doi: 10.1186/gb-2011-12-9-r86. https://doi.org/10.1186/gb-2011-12-9-r86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Kopan R, Ilagan MX. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell. 2009;137(2):216–33. doi: 10.1016/j.cell.2009.03.045. https://doi.org/10.1016/j.cell.2009.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Tao J, Chen S, Lee B. Alteration of Notch signaling in skeletal development and disease. Ann N Y Acad Sci. 2010;1192:257–68. doi: 10.1111/j.1749-6632.2009.05307.x. https://doi.org/10.1111/j.1749-6632.2009.05307.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Gripp KW, Robbins KM, Sobreira NL, Witmer PD, Bird LM, Avela K, et al. Truncating mutations in the last exon of NOTCH3 cause lateral meningocele syndrome. Am J Med Genet Part A. 2015;167A(2):271–81. doi: 10.1002/ajmg.a.36863. https://doi.org/10.1002/ajmg.a.36863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Ejaz R, Qin W, Huang L, Blaser S, Tetreault M, Hartley T, et al. Lateral meningocele (Lehman) syndrome: a child with a novel NOTCH3 mutation. Am J Med Genet Part A. 2016;170a(4):1070–5. doi: 10.1002/ajmg.a.37541. https://doi.org/10.1002/ajmg.a.37541. [DOI] [PubMed] [Google Scholar]
- [7].Lee JW. Mutations in PDGFRB and NOTCH3 are the first genetic causes identified for autosomal dominant infantile myofibromatosis. Clin Genet. 2013;84(4):340–1. doi: 10.1111/cge.12238. https://doi.org/10.1111/cge.12238. [DOI] [PubMed] [Google Scholar]
- [8].Karst SY, Ward-Bailey PF, Kane C, Bergstrom D, Donahue LR, Davisson-Fahey MT. Humpback: a new mutation on Chromosome 17 causing kyphosis and abnormal muscle phenotypes. [Dec 15;2020 ]; MGI Direct Data Submission 2010. http://www.informatics.jax.org/reference/J:157222 .
- [9].Newton CR, Graham A, Heptinstall LE, Powell SJ, Summers C, Kalsheker N, et al. Analysis of any point mutation in DNA. The amplification refractory mutation system (ARMS). Nucleic Acids Res. 1989;17(7):2503–16. doi: 10.1093/nar/17.7.2503. https://doi.org/10.1093/nar/17.7.2503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Ye S, Dhillon S, Ke X, Collins AR, Day IN. An efficient procedure for genotyping single nucleotide polymorphisms. Nucleic Acids Res. 2001;29(17):E88–e. doi: 10.1093/nar/29.17.e88. https://doi.org/10.1093/nar/29.17.e88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Jung H, Nam H, Suh JG. Rapid and efficient identification of the mouse leptin receptor mutation (C57BL/KsJ-db/db) by tetra-primer amplification refractory mutation system-polymerase chain reaction (ARMS-PCR) analysis. Lab Anim Res. 2016;32(1):70–3. doi: 10.5625/lar.2016.32.1.70. https://doi.org/10.5625/lar.2016.32.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Rudbeck L, Dissing J. Rapid, simple alkaline extraction of human genomic DNA from whole blood, buccal epithelial cells, semen and forensic stains for PCR. Biotechniques. 1998;25(4):588–90. doi: 10.2144/98254bm09. https://doi.org/10.2144/98254bm09. [DOI] [PubMed] [Google Scholar]
- [13].VanCleave A, Palmer M, Fang F, Torres H, Rodezno T, Li Q, et al. Development and characterization of the novel human osteosarcoma cell line COS-33 with sustained activation of the mTOR pathway. Oncotarget. 2020;11(27):2597–610. doi: 10.18632/oncotarget.27611. https://doi.org/10.18632/oncotarget.27611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Tao J, Chen S, Yang T, Dawson B, Munivez E, Bertin T, Lee B. Osteosclerosis owing to Notch gain of function is solely Rbpj-dependent. J Bone Miner Res. 2010;25(10):2175–83. doi: 10.1002/jbmr.115. https://doi.org/10.1002/jbmr.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden TL. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics. 2012;13:134. doi: 10.1186/1471-2105-13-134. https://doi.org/10.1186/1471-2105-13-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Collins A, Ke X. Primer1: primer design web service for tetra-primer ARMS-PCR. Open Bioinforma J. 2012;6:55–8. https://doi.org/10.2174/1875036201206010055. [Google Scholar]
- [17].Lehman RA, Stears JC, Wesenberg RL, Nusbaum ED. Familial osteosclerosis with abnormalities of the nervous system and meninges. J Ped. 1977;90(1):49–54. doi: 10.1016/s0022-3476(77)80763-4. https://doi.org/10.1016/S0022-3476(77)80763-4. [DOI] [PubMed] [Google Scholar]
- [18].Birnboim HC, Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979;7(6):1513–23. doi: 10.1093/nar/7.6.1513. https://doi.org/10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]