Highlights
-
•
Effects reactions heat and mass and heat transfer on chemical bubble yield are studied.
-
•
The effect the three energetic mechanisms depend on ambient bubble radii, frequency and acoustic amplitude.
-
•
The ignorance of thermal conduction improves the bubble energy and the chemical yield.
-
•
Excluding chemical reactions heat accelerate notably the chemical bubble yield.
-
•
Excluding mass transport of water vapor lowers the chemical bubble yield.
Keywords: Sonochemistry, Chemical bubble yield, Heat transfer, Mass transport, Chemical reactions, Numerical simulation
Abstract
Numerical simulations have been performed on a range of ambient bubble radii, in order to reveal the effect of mass transport, heat exchange and chemical reactions heat on the chemical bubble yield of single acoustic bubble. The results of each of these energy mechanisms were compared to the normal model in which all these processes (mass transport, thermal conduction, and reactions heat) are taken into account. This theoretical work was carried out for various frequencies (f: 200, 355, 515 and 1000 kHz) and different acoustic amplitudes (PA: 1.5, 2 and 3 atm). The effect of thermal conduction was found to be of a great importance within the bubble internal energy balance, where the higher rates of production (for all acoustic amplitudes and wave frequencies) are observed for this model (without heat exchange). Similarly, the ignorance of the chemical reactions heat (model without reactions heat) shows the weight of this process into the bubble internal energy, where the yield of the main species (•OH, H•, O and H2) for this model was accelerated notably compared to the complete model for the acoustic amplitudes greater than 1.5 atm (for f = 500 kHz). However, the lowest production rates were registered for the model without mass transport compared to the normal model, for the acoustic amplitudes greater than 1.5 atm (f = 500 kHz). This is observed even when the temperature inside bubble for this model is greater than those retrieved for the other models. On the other hand, it has been shown that, at the acoustic amplitude of 1.5 atm, the maximal production rates of the main species (•OH, H•, O and H2) for all the adopted models appear at the same optimum ambient-bubble size (R0 ~ 3, 2.5 and 2 µm for, respectively, 355, 500 and 1000 kHz). For PA = 2 and 3 atm (f = 500 kHz), the range of the maximal yield of •OH radicals is observed at the range of R0 where the production of •OH, O and H2 is the lowest, which corresponds to the bubble temperature at around 5500 K. The maximal production rate of H•, O and H2 is shifted toward the range of ambient bubble radii corresponding to the bubble temperatures greater than 5500 K. The ambient bubble radius of the maximal response (maximal production rate) is shifted toward the smaller bubble sizes when the acoustic amplitude (wave frequency is fixed) or the ultrasound frequency (acoustic power is fixed) is increased. In addition, it is observed that the increase of wave frequency or the acoustic amplitude decrease cause the range of active bubbles to be narrowed (scenario observation for the four investigated models).
Nomenclature
- Af (Ar)
Pre-exponential factor of the forward (reverse) reaction, [(cm3 mol−1 s−1) for two body reaction and (cm6 mol−2 s−1) for three body reaction].
- bf (br)
Temperature exponent of the forward (reverse) reaction.
- c
Speed of sound in the liquid medium, (m s−1).
- Eaf (Ear)
Activation energy of the forward (reverse) reaction, (cal mol−1).
- f
Frequency of ultrasonic wave, (Hz).
- Ia
Acoustic intensity of ultrasonic irradiation, (W m−2).
- kf (kr)
Forward (reverse) reaction constant, [(cm3 mol−1 s−1) for two body reaction and (cm6 mol−2 s−1) for three body reaction].
- p
Pressure inside a bubble, (Pa).
- pmax
Maximum pressure inside a bubble (Pa).
- p∞
Ambient static pressure, (Pa).
- PA
Amplitude of the acoustic pressure, (Pa).
- Pv
Vapor pressure of water, (Pa).
- Pg0
Initial gas pressure, (Pa).
- R
Radius of the bubble, (m).
- Rmax
Maximum radius of the bubble, (m).
- R0
Ambient bubble radius, (m).
- t
Time, (s).
- T
Temperature inside a bubble, (K).
- Tmax
Maximum temperature inside a bubble, (K).
- T∞
Bulk liquid temperature, (K).
- xi
Solubility (in mole fraction) of the gas i in water.
- yH2O
Mole fraction of water vapor trapped at the collapse.
Greek letters
- γ
Specific heat ratio (cp/cv) of the gas mixture.
- σ
Surface tension of liquid water, (N m−1).
- ρ
Density of liquid water, (kg m−3).
- λ
Gas thermal conductivity (W m−2 K).
1. Introduction
Acoustic cavitation is the formation and collapse of bubbles in liquid irradiated by intense ultrasound [1]. Cavitation serves as a means of concentrating the diffuse energy of sound. It is the underlying phenomenon responsible for sonochemistry and sonoluminescence [2]. Fast collapse of the bubbles adiabatically compresses entrapped gas and vapor, which results in short and local hot spots [3], where the temperature and pressure are around 5000 K and 300 atm, respectively, or more inside the bubbles at the end of the collapse [4]. In this situation, entrapped molecules of dissolved gases, vaporized water and solutes can be brought to an excited state and dissociate [5]. As a result, water vapor and noncondensable gas present inside the hot spots leads to the formation of a variety of radicals and highly reactive species such as hydroxyl (•OH), hydrogen (H•), hydroperoxyl (HO2•) and hydrogen peroxide (H2O2) [6]. The generated substances are able to initiate other secondary chemical reactions [7], [8]. In general, three distinct regions are probable for chemical reactions to occur: within the hot bubble cavity (gaseous region), at the interface between the bubble and bulk liquid, and in the bulk media [9]. Ultrasonic irradiation (sonolysis) is one of the efficient methods of advanced oxidation processes (AOP) [10].
Now, many researches (theoretical and experimental) are focused to study and increase the rates of sonochemical reactions in order to use them in industry [11]. Hua et al. [12] investigated the effect of ultrasound frequency (from 205 to 1071 kHz), nature and ratios of saturating gases (Ar and O2) on sonoluminescence and sonochemistry, in addition, the impact of hydroxyl radical scavenger and bicarbonate ion was also examined. An optimal frequency of 358 kHz was determined for the maximal sonoluminescence and chemical reaction rates. In addition, the importance of O2 in sonoluminescence quenching was demonstrated, and a ratio of 75%Ar/25%O2 was found to be ideal for maximal reaction rate. Pétrier et al. [13], examined the rate of phenol and carbon tetrachloride degradation over a range of ultrasonic frequencies from 20 to 800 kHz, where an optimal frequency of 200 kHz was determined for the maximal degradation of phenol, conversely, the decomposition of carbon tetrachloride increases proportionally with the frequency increase. In general, the effects of acoustic amplitude [14], [15], [16], [17], [18], frequency [18], [19], [20], [21], [22], saturating gas [18], [23], [24], [25], [26], [27], solution temperature [28], [29], static pressure [30], [31] and other parameters have been widely investigated [32].
On the other hand, some of the theoretical studies focused on the effect of each of the energy parameters found in the bubble internal energy balance, such as mass transport [33], [34], chemical reactions heat [35], [36], thermal conduction [37], bulk liquid viscosity [38], and pressure force work [39]]. In general, the impact of these energy parameters was investigated for a fixed ambient bubble radius (mean value). Unfortunately, this is not the case in practice, where a range of ambient bubble radii is produced depending on the power and frequency of the acoustical wave and the other experimental conditions [17], [40], [41], [42], [43], [44]. Consequently, each bubble will be affected differently depending on its size.
To the best of our knowledge, the effect of mass transport, heat exchange and chemical reactions heat on bubble kinetics investigated on a range of ambient bubble radii is until now not examined previously. The present theoretical study states, for the first time, the effective dependence of the ignorance of transfer phenomenons and reaction heats to the initial ambient bubble size. This issue has been treated for an acoustic amplitude ranging from 1.5 to 3 atm and ultrasonic frequency of 200, 355, 515 and 1000 kHz. The numerical simulations performed in this work are based on a model of a single bubble (argon bubble).
2. Model
The employed model is based on a set of ordinary differential equations, where the effects of non-equilibrium evaporation and condensation of water vapor at the bubble wall, thermal conduction both inside and outside a bubble and chemical reactions are included. All numerical simulations have been conducted for a bubble oscillating in an argon-saturated water. Based on the above assumptions, temperature, pressure, bubble radius evolution and bubble wall velocity can be determined at any instant. The radial dynamics of the bubble is described by the modified Keller-Miksis equation (Eq. 1), developed by Yasui [37]:
| (1) |
where dots denotes the time derivative (d/dt), R is the radius of the bubble, C the speed of sound in the liquid, ρL is the density of the liquid, is the net rate of evaporation per unit area and unit time and P∞ is the ambient static pressure. PA is correlated with the acoustic intensity Ia (or power per unit area) as: PA = (2IaρLC)1/2. The liquid pressure PB(t) on the external side of the bubble wall is related to the internal pressure P(t) by the following equation [37]:
| (2) |
where σ is the surface tension, µ is the liquid viscosity and ρg is the density inside the bubble. The internal pressure P(t) is calculated using Van der Waals equation (Eq. 3):
| (3) |
where a and b are the Van de Waals constants, Rg is the universal gas constant, V is the volume of the bubble [V = 4/3(πR3)], and T is the temperature inside the bubble. The Van der Waals constants (a and b) are determined by [45]:
| (4) |
The mass flux of evaporation and condensation at the interface is expressed using Hertz-Knudsen formula derived from kinetic theory of gases [46]:
| (5) |
| (6) |
where α is the evaporation coefficient (considered constant α = 0.4), is the average velocity of molecules, ρg,H2O(R) is the density of water vapor in the bubble: ρg,H2O(R) = MH2O·nH2O10−3/[4/3(πR3)], and = 0.0173 Kg m−3 is the saturated vapor density. The temperature of the external bubble surface is Ts = Tliq.
Heat exchange inside and outside the bubble is estimated to be [47]:
| (7) |
| (8) |
λmix, χ and Lth are the heat conductivity, thermal diffusivity of the gas mixture and the thickness of the thermal boundary layer, respectively.
The thermal conductivity (λmix) is estimated by [48]:
| (9) |
where λH2O(T) and λAr(T) are the thermal conductivity of water vapor and argon at temperature T (in K), respectively. The thermal conductivity (in W m−1 K−1) of H2O and Ar are estimated by [45]:
| (10) |
| (11) |
At the bubble wall, the thermal diffusivity (χ) is estimated by [47]:
| (12) |
where Cp is the heat capacity concentration (J m−3 K−1) for H2O and Ar mixture [47]:
| (13) |
Ci, kB, NA and fi are the molar concentration of species i (H2O and Ar) inside the bubble, Boltzmann constant, Avogadro number and the number of degrees of freedom of species i (fH2O = 6 and fAr = 3), respectively.
The temperature (T) inside bubble is calculated by solving the following equation [37]:
| (14) |
where E is the internal energy of the bubble and CV,H2O(T) (Cv,Ar(T)) is the isochoric heat capacity of vapor (Argon) at temperature T, given as a polynomial approximation (expressions are available in Ref. [49]).
The change of the internal energy of a bubble (ΔE) in time (Δt), due to the pressure work, heat of chemical reactions within the bubble, energy carried by water molecules by evaporation and condensation and heat exchange by diffusion at the interface, is expressed by [37], [38]:
| (15) |
where ΔHi and ri are the enthalpy change and the rate of the ith reaction respectively, the first term in the right-hand side of Eq. 15 is the PV work. The second term is the energy carried by evaporating vapor from the surrounding liquid into the bubble and by condensing vapor from the bubble into the liquid in Δt. The third term is the energy change due to the thermal conduction. The last term is the heat of chemical reactions taking place inside the bubble during the collapse.
In this work, a scheme of 25 reversible chemical reactions is considered (Table 1), in which K chemical species is involved. The general form of these reversible reactions is given as follows:
| (16) |
in which υki is the stoichiometric coefficient of the ith reaction and Xk is the chemical symbol for the kth species. The superscript ‘ indicates forward stoichiometric coefficients, while “ indicates reverse stoichiometric coefficients. The production rate of the kth species can be written as a summation of the rate of the variables for all reactions involving the kth species:
| (17) |
where nk is the number of moles of the kth species. The rate ri for the ith reaction is given as:
| (18) |
where [Xk] is the molar concentration of the kth species and kfi and kri are the forward and reverse rate constants of the ith reaction, respectively. The forward and reverse rate constants for the ith reactions are assumed to have the following Arrhenius temperature dependence:
| (19) |
| (20) |
where Rg is the universal gas constant, Afi (Ari) is the pre-exponential factor, bfi (bri) is the temperature exponent and Efi (Eri) is the activation energy. Arrhenius parameters of each chemical reaction are presented in Table 1.
Table 1.
Scheme of the possible chemical reactions inside a collapsing argon bubble [36], [53]. M is the third Body. Subscript “f” denotes the forward reaction and “r” denotes the reverse reaction. A is in (cm3 mol−1 s−1) for two body reaction [(cm6 mol−2 s−1) for a three body reaction], and Ea is in (cal mol−1) and ΔH in (kcal mol−1). For some of the backward reactions, the constants are not listed. Those backward reactions are neglected during calculations.
| Reaction | Af | nf | Eaf | Ar | nr | Ear | ΔH | |
|---|---|---|---|---|---|---|---|---|
| 1 | H2O + M ⇌ H•+•OH + M | 1.912 × 1023 | −1.83 | 1.185 × 105 | 2.2 × 1022 | −2.0 | 0.0 | 121.72 |
| 2 | •OH + M ⇌ O + H•+M | 9.88 × 1017 | −0.74 | 1.021 × 105 | 4.714 × 1018 | −1.0 | 0.0 | 104.36 |
| 3 | O + O + M ⇌ O2 + M | 6.165 × 1015 | −0.5 | 0.0 | 4.515 × 1017 | −0.64 | 1.189 × 105 | −120.91 |
| 4 | H•+O2 ⇌ O+•OH | 1.915 × 1014 | 0.0 | 1.644 × 104 | 5.481 × 1011 | 0.39 | −2.93 × 102 | 16.54 |
| 5 | H•+O2 + M ⇌ HO2• +M | 1.475 × 1012 | 0.6 | 0.0 | 3.09 × 1012 | 0.53 | 4.887 × 104 | − 49,0 |
| 6 | O + H2O ⇌ •OH+•OH | 2.97 × 106 | 2.02 | 1.34 × 104 | 1.465 × 105 | 2.11 | −2.904 × 103 | 17.37 |
| 7 | HO2•+H• ⇌ H2 + O2 | 1.66 × 1013 | 0.0 | 8.23 × 102 | 3.164 × 1012 | 0.35 | 5.551 × 104 | − 57.34 |
| 8 | HO2•+H• ⇌ •OH+•OH | 7.079 × 1013 | 0.0 | 2.95 × 102 | 2.027 × 1010 | 0.72 | 3.684 × 104 | − 38.82 |
| 9 | HO2•+O ⇌ •OH + O2 | 3.25 × 1013 | 0.0 | 0.0 | 3.252 × 1012 | 0.33 | 5.328 × 104 | − 55.47 |
| 10 | HO2•+•OH ⇌ H2O + O2 | 2.89 × 1013 | 0.0 | −4.97 × 102 | 5.861 × 1013 | 0.24 | 6.908 × 104 | − 72.83 |
| 11 | H2 + M ⇌ H•+H•+M | 4.577 × 1019 | −1.4 | 1.044 × 105 | 1.146 × 1020 | −1.68 | 8.2 × 102 | 106.33 |
| 12 | O + H2 ⇌ H•+•OH | 3.82 × 1012 | 0.0 | 7.948 × 103 | 2.667 × 104 | 2.65 | 4.88 × 103 | 1.97 |
| 13 | •OH + H2 ⇌ H•+H2O | 2.16 × 108 | 1.52 | 3.45 × 103 | 2.298 × 109 | 1.40 | 1.832 × 104 | − 15.4 |
| 14 | H2O2 + O2 ⇌ HO2•+HO2• | 4.634 × 1016 | −0.35 | 5.067 × 104 | 4.2 × 1014 | 0.0 | 1.198 × 104 | 41.95 |
| 15 | H2O2 + M ⇌ •OH+•OH + M | 2.951 × 1014 | 0.0 | 4.843 × 104 | 1.0 × 1014 | −0.37 | 0.0 | 52.13 |
| 16 | H2O2 + H• ⇌ H2O+•OH | 2.410 × 1013 | 0.0 | 3.97 × 103 | 1.269 × 108 | 1.31 | 7.141 × 104 | − 69.6 |
| 17 | H2O2 + H• ⇌ H2 + HO2• | 6.025 × 1013 | 0.0 | 7.95 × 103 | 1.041 × 1011 | 0.70 | 2.395 × 104 | − 15.38 |
| 18 | H2O2 + O ⇌ •OH + HO2• | 9.550 × 106 | 2.0 | 3.97 × 103 | 8.66 × 103 | 2.68 | 1.856 × 104 | − 13.42 |
| 19 | H2O2+•OH ⇌ H2O + HO2• | 1.0 × 1012 | 0.0 | 0.0 | 1.838 × 1010 | 0.59 | 3.089 × 104 | − 30.78 |
| 20 | O3 + M ⇌ O2 + O + M | 2.48 × 1020 | 0 | 2.27 × 104 | – | – | – | 26.14 |
| 21 | O3 + O ⇌ O2 + O2 | 5.2 × 1018 | 0 | 4.157 × 103 | – | – | – | − 94.77 |
| 22 | O3+•OH ⇌ O2 + HO2• | 7.8 × 1017 | 0 | 1.9 × 103 | – | – | – | − 39.46 |
| 23 | O3 + HO2• ⇌ O2 + O2+•OH | 1 × 1017 | 0 | 2.8 × 103 | – | – | – | − 29.17 |
| 24 | H•+ O3 ⇌ HO2•+O | 9 × 1018 | 0.5 | 3.99 × 103 | – | – | – | 32.45 |
| 25 | H•+ O3 ⇌ •OH + O2 | 1.6 × 1019 | 0 | 0 | – | – | – | − 23.01 |
The water vapor change with time inside the bubble, due to the evaporation and condensation at the bubble wall and due to chemical reactions is given by:
| (21) |
For other species k (except Ar):
| (22) |
The modified Keller-Miksis equation (Eq. 1), is a nonlinear second-order differential equation, which can be reduced to a system of two differential first-order equations as
| (23) |
| (24) |
Eqs. (14), (23) and (24) are solved using finite difference method. The outputs of these equations are the bubble wall velocity, evolution of the bubble radius, variation of bubble temperature and pressure all along the bubble oscillation, respectively. The variation in time of number of moles of each species housed within the bubble is given by Eqs. (21) and (22). The amount of argon remains unchanged during the bubble oscillation (i.e., chemically inert).
In the present study, all simulations were conducted for a single bubble oscillating in an argon-saturated water at 20 °C, so, the initial bubble composition is a mixture of water vapor and argon. The complete bubble-dynamics model includes the liquid compressibility and viscosity, heat transfer (i.e., thermal conduction through bubble wall), mass transfer (i.e., nonequilibrium and condensation evaporation of the liquid vapor across the bubble/liquid interface), and reaction kinetics occurring inside the bubble (i.e., reactions heats are incorporated). In order to appreciate the impacts of different parameters injected into the energy equation, the results of excluding of each of these parameters on both the dynamics of the single bubble and chemical species generated inside the bubble were investigated:
-
1.
Complete model with including all parameters (i.e., denoted as normal),
-
2.
Model without heat transfer (i.e., denoted as ),
-
3.
Model without mass transfer (i.e., denoted as ),
-
4.
Model without reaction heats (i.e., denoted as ΔH = 0).
Finally, it is important be noted that our study gives a good modification of Yasui’s model, where some modifications are done. These changes are given as follows:
A more simplified equation of Hertz-Knudsen formula is used (Eq. 5), compared to that used by Yasui [37]. In addition, in our work, we consider the temperature on the external bubble wall as constant, conversely to Yasui’s studies.
For the heat exchange mechanism, a good combination is introduced by the adoption of Toegel’s heat model (for heat conduction, Eq. 7) [50], which is not the case for the Yasui’s model.
3. Results and discussion
In this section, the effect of the different models on the production rates of the main species is discussed. As it is known, the main species created inside an argon bubble are O, H•, •OH and H2 [26], [51], [52], [53], [54]. Based on this observation we limit our work to these principal species. The simulations were performed at frequencies of 200, 355, 500 and 1000 kHz. The selection of this range of ultrasound frequency (200, 355, 500 and 1000 kHz) is based on many experimental works, which found that the best frequency for sonochemistry is ranged from 200 to 1000 kHz [12], [13], [14], [18], [20], [55], [56], [57], [58], [59]. These work confirmed that lower frequencies (i.e., usually 20 kHz) is not suitable for producing efficient sonochemical effects.
3.1. Models effects on production rates
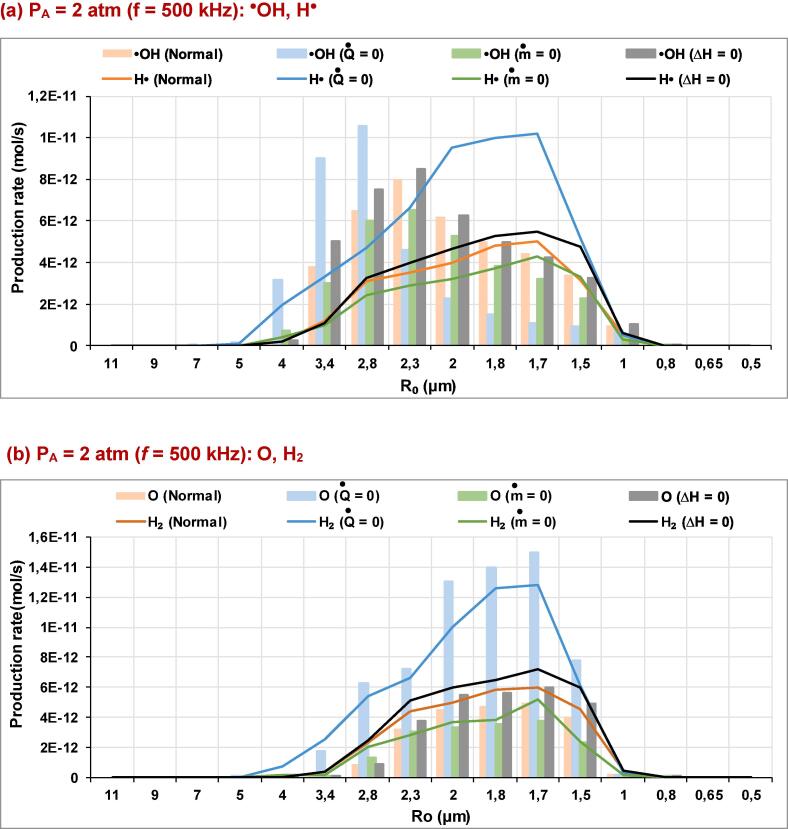
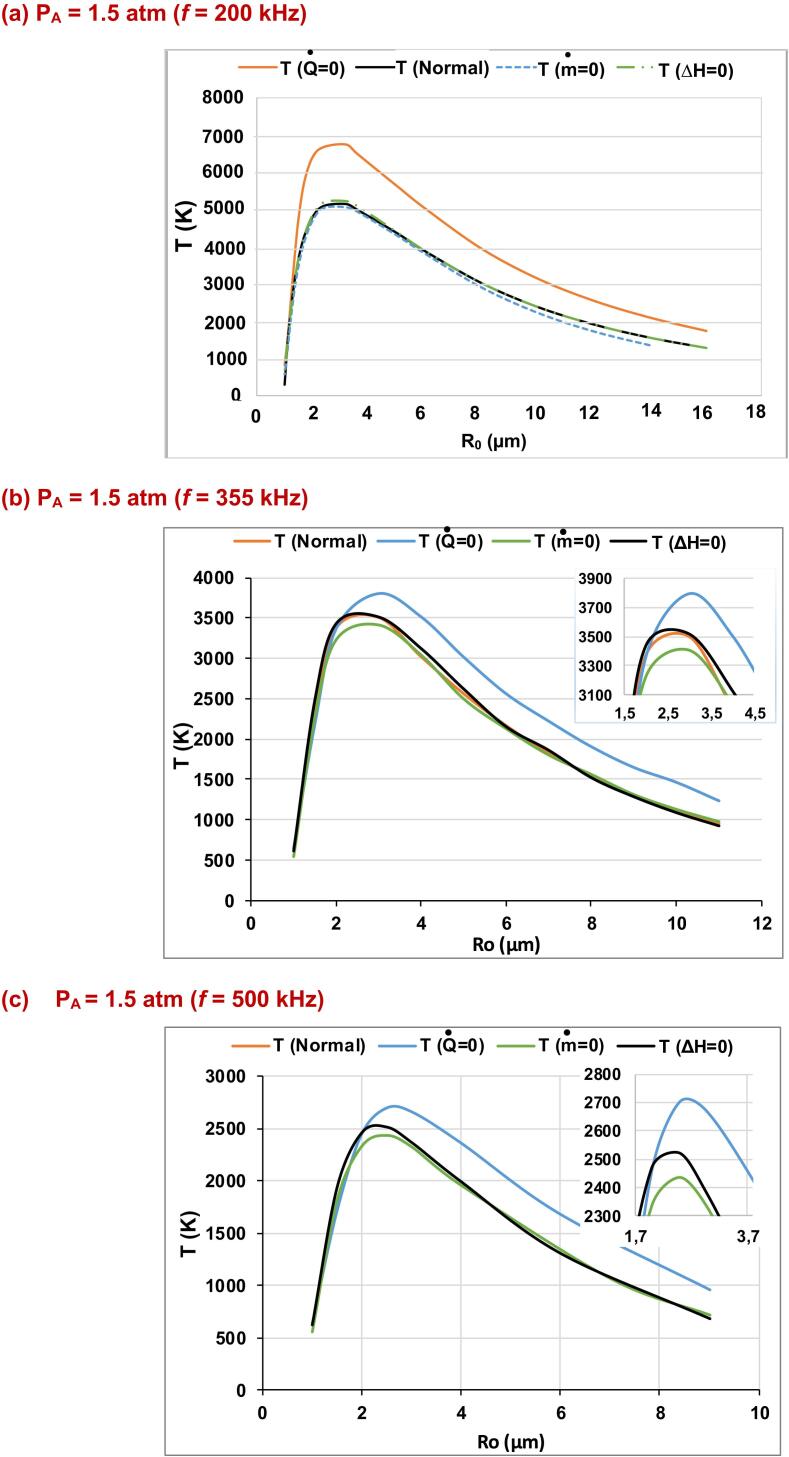
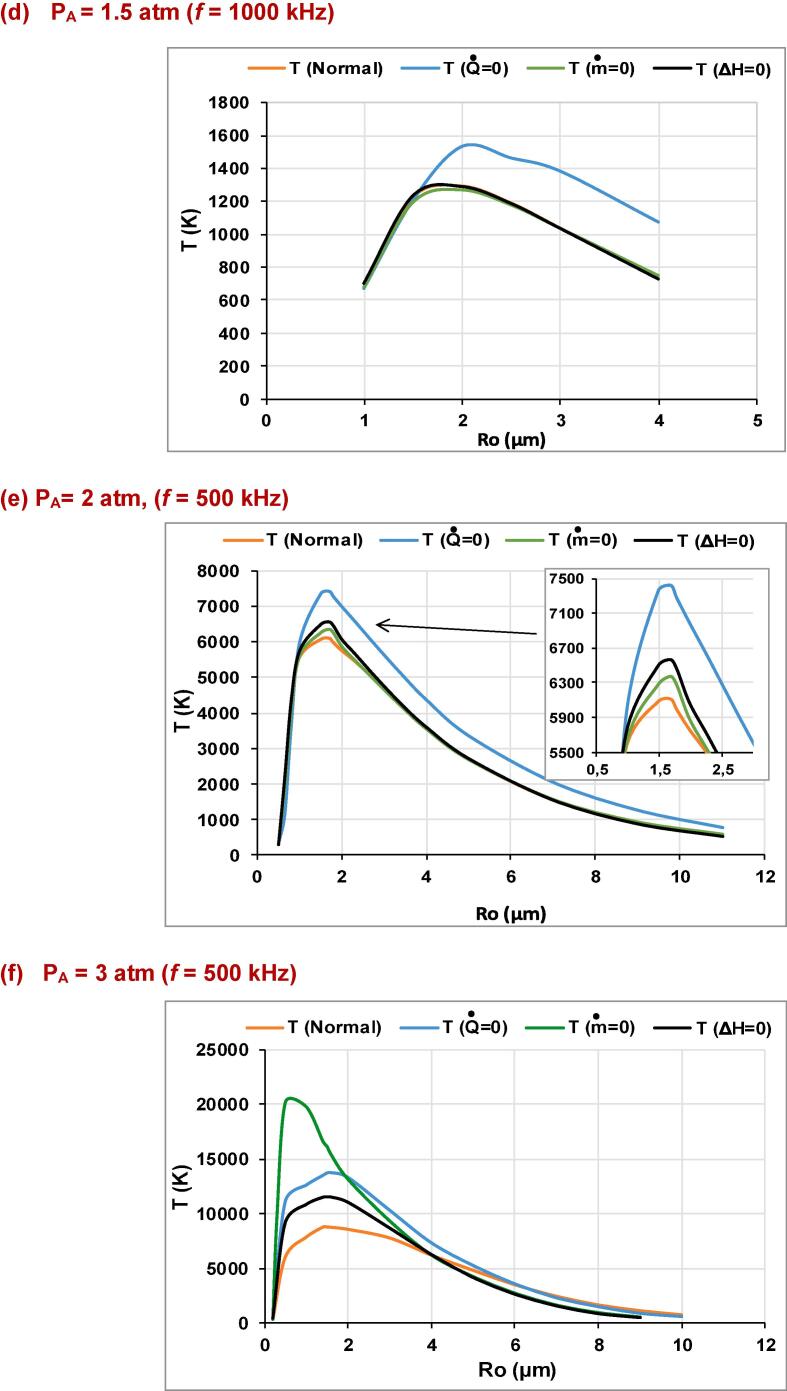
In Fig. 1(a)-(f), the production rates for the essential species (•OH, H•, O and H2) produced inside an argon saturated water bubble are shown for the ultrasound frequencies of f = 200, 355, 500 and 1000 kHz over a wide range of ambient bubble radius (R0), where the acoustic amplitude and the ambient pressure are maintained at 1.5 and 1 atm, respectively. The liquid ambient temperature is 20 °C. These production rates are given for models without mass transport, without thermal conduction and without reactions heat, compared to the compete model (i.e., denoted as normal). In Fig. 2(a)-(d), the production rates of these species (•OH, H•, O and H2) are shown for the acoustic amplitudes of 2 and 3 atm under the ultrasound frequency of 500 kHz. The rest of conditions are the same as in Fig. 1. The production rate in all these figures is defined as the amount of each indicated species created inside the bubble at the end of the bubble collapse multiplied by the driving frequency [54], [60], [61]. In Fig. 3(a)-(f), temperature profiles are given for the acoustic amplitude of 1.5 atm (f = 200, 355, 500 and 1000 kHz) and for PA = 2 and 3 atm under the ultrasound frequency of 500 kHz. Temperatures of these profiles are the maximum values achieved at the end of the first bubble collapse (at the maximum compression of the bubble) during the bubble oscillation.
Fig. 1.
(a)-(h). Production rates of •OH, O, H• and H2 over a wide range of R0 for the model without thermal conduction (), model without mass transport (), or model without reactions heat (ΔH = 0), compared to the complete model (normal) (PA = 1.5 atm, frequency = 200, 355, 500 and 1000 kHz).
Fig. 2.
(a)-(d). Production rates of •OH, O, H• and H2 over a wide range of R0 for the model without thermal conduction (), model without mass transport (), or model without reactions heat (ΔH = 0), compared to the complete model (normal) (PA = 2 and 3 atm, frequency = 500 kHz).
Fig. 3.
(a)-(f): The pick temperature profiles over a wide range of R0 for the model without thermal conduction (), model without mass transport (), or model without reactions heat (ΔH = 0), compared to the complete model (normal) (PA = 1.5 atm for (a)-(d) [frequency = 200, 355, 500 and 1000 kHz] and PA = 2 and 3 atm [f = 500 kHz] for (e) and (f)).
As it is expected, the production rate of the different species (for all the adopted models) is increased by the decrease of the ultrasound frequency, Fig. 1(a)-(h), or if the acoustic power is increased Fig. 2(a)-(d). These finding are similar to those found by Sochard et al.[62], Yasui et al. [30], [60] and Merouani et al. [31], [61], [63]. This increase of production rate is clearly observed at around a determined ambient bubble size (R0 of the maximal response), where the acoustic parameters (frequency and amplitude) are suitable to get a maximal intensity of response. The predominance of the thermal conduction mechanism is confirmed through the production rates obtained when the model without heat conduction is adopted compared the other models (normal model and models without mass transport or reaction heat). This sensitivity to the heat transfer mechanism is observed for all the acoustic amplitudes and frequencies [Figs. 1(a)-(h) and 2(a)-(d)], even when the temperature obtained using the model without mass transfer is greater than that of the model without thermal conduction (Fig. 3(f)). This is because of the importance of heat transfer into bubble internal energy balance especially during the compression phase [37]. Bubble internal energy increases during the compression phase due to the work applied on bubble wall by the external liquid. However, some energy should be carried out by thermal conduction and vapor condensation. Therefore, the unawareness of heat transfer mechanism increases largely bubble temperature, thus the production rates of different species created inside bubble increases (i.e., the increase of bubble temperature increases the thermal dissociation of water vapor, consequently, the yield of the other species is surely increased). This conclusion is reinforced by the absence of the oxidant consumer. In addition, this increase of temperature inside the bubble is promoted when the temperature on the external bubble wall is considered constant, as it is done here in our work by using Toegel model [50] for heat transfer. It is also observed in Fig. 1(a)-(h) that the production rate of the different species (•OH, H•, O and H2) using the model without mass transfer is greater than that of the model without reactions heat or the normal model, which is explained by the lower acoustic amplitude (PA = 1.5 atm) used in this case (lower expansion and compression ratios). Due to this low acoustic power, we can see easily in Fig. 3(a)-(d) that the peak temperatures attained for the four frequencies (5100, 3500, 2500 and 1300 K for 200, 355, 500 and 1000 kHz, respectively) are insufficient to reveal especially the impact of the reactions heat compared to the mass transport mechanism. This in spite of the lower amount of vapor housed into bubble for the model without mass transport. In other worlds, as we know, the condition to get a violent collapse is the presence of a sufficient quantity of water vapor inside the bubble during the collapse. Thus, at 1.5 atm, for the model without mass transport we found approximately the same response (Tmax) as that of the model without reactions heat or the normal model, which means that the amount of vapor inside the bubble for the model without mass transport is sufficient in this case (1.5 atm, for all frequencies).
On the other hand, it is also seen in Fig. 1(a)-(d) that the production rate of •OH, H•, O and H2 for the model without reaction heat is greater than that of the normal model. This is because at these conditions of acoustic amplitude and ultrasound frequencies (1.5 atm and 200/355 kHz), the elimination of the reactions heat starts to give its contribution into bubble internal energy balance. So that, maximal temperature is increased compared to the normal model where the endothermal nature of the most chemical reactions taking place inside bubble (Table. 1) during collapse causes a decrease of the bubble temperature and consequently the production rate [35], [37]. In Fig. 1(c)-(d), the maximal production rates of •OH, H•, O and H2 for the model without reactions heat are 4.33 × 10−14, 3.50 × 10−14, 5.6 × 10−17 and 4.26 × 10−15 mol s−1, respectively. This maximum is retrieved at around R0 = 3 µm. However, for the normal model, the production rates of •OH, H•, O and H2 are 2.86 × 10−14, 2.36 × 10−14, 2.52 × 10−17 and 2.51 × 10−15 mol s−1, respectively, at the same ambient bubble size, i.e., 3 µm. For the ultrasonic frequencies of 500 and 1000 kHz at the acoustic amplitude of 1.5 atm, Fig. 1(c)-(f), it is seen that the response (maximal production rate) of the model without reactions heat and the normal model is similar for all the main species (•OH, H•, O and H2). This is obviously explained by the lower maximal temperatures retrieved for the two models on their ranges of ambient bubble radius [Fig. 3(c)-(d)].
It should be noted here that the maximal response (maximal production rate) is shifted toward lower ambient bubble radius as the ultrasound frequency is reduced (acoustic amplitude is constant) [Fig. 1(a)-(h)]. At the ultrasound frequencies of 200, 355, 500 and 1000 kHz, the ambient bubble sizes of the maximal response are around 3.2, 3, 2.5 and 2 µm, respectively. This result is due to the effect of the ultrasound frequency. As the ultrasonic frequency is increased, bubbles are found to be enables to largely expand so that the maximal response is automatically shifted toward small bubble radii [64]. Conversely, when the ultrasonic frequency is reduced, bubbles will get more time to expand, therefore expansion and compression ratios are higher in this case, and the maximal response is moved toward larger bubble radii. This trend is observed in the work of Yasui et al. [60], where the intensity of sonoluminescence and sonochemical reactions have been studied on a range of ambient bubble radii for an air bubble in liquid water.
Until now, we have only treated the effect of the different models (without mass transport, thermal conduction and reactions heat compared to the normal model) on production rates by changing the ultrasound frequency (200, 355, 500 and 1000 kHz) and considering the acoustic amplitude as constant at 1.5 atm. However, to reveal more the impact of these mechanisms on the different productions rates, we fix the ultrasonic frequency at 500 kHz and we change gradually the acoustic amplitude (1.5, 2 and 3 atm). It should be noted here that the choice of this ultrasound frequency is arbitrary, because of the behaviors similarity observed for the four frequencies studied herein.
It is seen from Figs. 1(e)-(f) and 2(a)-(d) that the maximal production rates attained at different ambient bubble sizes are increased when the acoustic amplitude is increased or if the ultrasound frequency is decreased. The same result is shown previously when the ultrasonic frequency is changed by considering the acoustic amplitude as constant (1.5 atm). On the other hand, the ambient bubble radius of the maximal response (maximal production rate) is shifted toward small values. For the acoustic amplitudes of 1.5, 2 and 3 atm, the ambient bubble radii of the maximal production rates are 2.5 , 1.7 and 1.5 µm, respectively, under the ultrasound frequency of 500 kHz. Conversely to the previous case where the acoustic amplitude is fixed, the increase of the acoustic amplitude (from 2 atm) shows that the production rate of the model without reactions heat or the normal model is greater than the case of the model without mass transport for all the main species (i.e., •OH, H•, O and H2) [Figs. 1(e)-(f) and 2(a)-(d)]. This is even if the temperature of the model without mass transport is greater than that of the model without reactions heat or the normal model. This behavior is clearly observed when the peak temperature of collapse corresponding to its ambient bubble radius is greater or around 4500 K [Fig. 3(c), (e), and (f)]. This decrease of production rate is due to the endothermal nature of the most chemical reactions taking place inside bubble during collapse (Table 1) and especially to the elimination of evaporation mechanism for the model without mass transport. Therefore, the elimination of the reactions heat increases more bubble temperature and consequently the production rate. Even for the normal model where all the mechanisms are taken into account, the production rate of •OH, H•, O and H2 in this model is greater than that obtained by the model without mass transport. This is because of the elimination of evaporation mechanism during the expansion phase, so that during the compression phase the ignorance of the condensation mechanisms for the model without mass transport is insufficient to largely increase its production rate. The effect of thermal conduction mechanism is found to be dominant when the acoustic amplitude is increased [Fig. 2(a)-(d)], which confirms again the importance of energy carried out of the bubble by heat transfer during the compression phase.
It should be noted here that for small bubble radii (smaller than R0 of the maximal production rate), some interferences of maximal production rates and temperatures are exhibited by all the adopted models (normal model, and models without mass transport, thermal conduction or reactions heat) as shown in Figs. 1(a)-(h), 2(a)-(d) and 3(a)-(f). This is due to the impact of ambient bubble radius. Because for this range of ambient bubble radii, a small amount of vapor is expected to be housed into bubble at the end of rarefaction phase due to the small expansion ratio. Consequently, the difference of vapor amount between the normal model and the model without mass transport is not large for this range of ambient bubble radii. During the compression phase, a lower production rate is expected; therefore, bubble energy balance during this phase is not largely affected by the elimination of reactions heat, which means a similar response (production rate and maximal temperature) for the model without reactions heat compared to the normal model. On the other hand, a small increase of heat capacity (due to the small expansion ratio) for the normal model on this range of ambient bubble radii compared to the model without mass transport, so that the two models give a similar response, in spite of the elimination of condensation mechanism for the model without mass transport. Because of the short time of compression and the lower expansion ratio, the heat conduction during the compression phase for the normal model is very lowered, which implies that the maximal production rates (and temperatures) obtained by this model is relatively the same as the model without thermal conduction.
Similar interferences are also observed at larger ambient bubble radii (greater than R0 of the maximal response) for all the acoustic amplitudes and the ultrasound frequencies. Because, for this range of ambient bubble radii, lower expansion and compression ratios are expected and also a huge amount of vapor is found inside bubble at the end of rarefaction phase. The result is a milder collapse for all the treated models, which implies that the effect of each of these mechanisms is hidden in this range of ambient bubble radius.
According to Figs. 1(a)-(h) and 2(a)-(d), we can see that at lower acoustic amplitude (PA = 1.5 atm), the maximal production rates of all the main species created inside bubble (•OH, H•, O and H2) at collapse appear at the same ambient bubble radius. This is confirmed for all the ultrasonic frequencies. Under the ultrasound frequencies of 200, 355, 500 and 1000 kHz (PA = 1.5 atm), the maximal production rates of •OH, H•, O and H2 are shown at 3.2, 3, 2.5 and 2 µm, respectively [Fig. 1(a)-(h)]. However, at the acoustic amplitude of 2 atm (f = 500 kHz), the maximal production rate of •OH radicals for the normal model and models without mass transport or reactions heat is given at around R0 = 2,3 µm (Fig. 2(a)). Some exception is observed for the maximal production rate of •OH radicals for the model without thermal conduction, where R0 (of the maximal response) is shifted toward 2.8 µm [Fig. 2(a) and (b)]. For the rest of species (H•, O and H2), at this acoustic amplitude (2 atm), the maximal production rates are observed at the ambient bubble radius of 1.7 µm. At the acoustic amplitude of 3 atm (f = 500 kHz), the maximal production rate of •OH radicals (for all models) is given at R0 = 5 µm, however, the maximal production rates of H•, O and H2 are observed at R0 = 1.5 atm (Fig. 2(c)-(d)). The interpretation of all these results can be made using the temperatures profiles of the treated models and the kinetic constants of some chemical reactions which have an important effect on the production rates of the main investigated species. To address this, in Fig. 4, the kinetic constants of reactions 1, 2, 3, 6, 11 and 12 of Table 1 are represented on a temperature range from 0 to 21000 K. The choice of these chemical reactions is based on their effects on the amount of the main species created into bubble at collapse.
Fig. 4.
(a)-(f). Evolutions vs. temperature of the forward and reverse rate constant of reactions (1), (2), (3), (6), (11) and (12) of Table 1. Unit: (cm3 mol−1 s−1) for two body reaction and (cm6 mol−2 s−1) for a three body reaction.
From Fig. 3(a)-(d), it is seen that the maximal temperatures retrieved for all models (expecting that without heat conduction) under the ultrasound frequencies of 200, 355, 500 and 1000 kHz for PA = 1.5 atm are around 5100, 3500, 2500 and 1400 K, respectively. In Fig. 4(a), it is found that the thermal decomposition of water (for all models) is proportionally reduced as the ultrasound frequency is increased. Consequently, the formation of H•, •OH and O is reduced [Fig. 4(a) and (b)] as the ultrasonic frequency is decreased. The production rate of O radicals is found to be lowered according to the 3rd reaction of Table 1 and especially the 6th reaction [Fig. 4(c) and 4(d)]. The amount of H2 is increased according to 11th reaction [Fig. 4(e)], especially by the mass action of H• radicals and the lower value of the forward reaction kinetic constant. According to the 12th reaction, the formation of O and H2 is promoted, due to the relatively higher kinetic constant of the backward reaction [Fig. 4(f)]. As a result, for all the adopted models under the ultrasound frequencies of 200, 355, 500 and 1000 kHz, the formation of •OH radicals is found to be predominant compared to the other species at this acoustic amplitude of 1.5 atm. On the other hand, the production rate of H• radicals is greater than that of O and H2, where the formation of H2 for these ultrasonic frequencies is favored than that of O radicals. It should be noted that according to the previous interpretations, the maximal production rates of the main species (•OH, H•, O and H2) are observed at the same ambient bubble radius for each of the ultrasound frequencies, conversely to the next case where the production rates are largely affected by the temperature profiles.
At the acoustic amplitude of 2 atm (f = 500 kHz), Fig. 2(a) and 2(b), it seems that the yield of the main species is highly affected by temperature profiles shown for each of the used models [Fig. 3(e)]. For H•, O and H2, the maximal production rate is observed at R0 = 1.7 µm, where the peak temperatures at this ambient bubble radius for the model without thermal conduction and the rest of models are 7400 and 6300 K, respectively [Fig. 3(e)]. On the other hand, for the model without thermal conduction, the maximal production rate of •OH radicals is given at around 2.8 µm, where the peak temperature at this ambient bubble radius is 5892 K. This maximal production rate is shifted at around R0 = 2.3 µm for the rest of models (normal model, and models without mass transport or reactions heat), where the maximal temperature attained at this ambient bubble size is 5500 K. All these results originate from the effect of bubble temperature, because at bubble temperatures at around 5500 K (all models), the thermal dissociations of water vapor is slightly promoted compared to the •OH radicals consumption [Fig. 4 (a) and (b)]. Therefore, the amount of •OH, H• and O radicals increases at this level. It is important to not here that the complete explanation of the yield of all species on a range of bubble temperature during the collapse period is a long and difficult task. However, it is interesting to try to explain these productions at around the end of collapse. Consequently, in our study, we focus on the end of the compression phase (maximum temperature), which is the most important moment for the maximal production or consumption of chemical substances.
On the other hand, the production rate of O radicals is reduced according to 6th reaction (Table. 1), especially due to the impact of mass action applied by water molecules. The effect of the backward reaction of 3rd reaction is reduced, because of the relatively larger amount of O radicals compared to O2 molecules. The consumption of H2 molecules is reduced according to the 11th reaction, due to the effect of H• radicals. In addition, the production rate of H2 and O radicals (consumption of •OH and H• radicals) is supposed to be improved according to 12th reaction, because of its higher backward kinetic constant compared to the forward one [Fig. 4(f)]. Nevertheless, at the peak temperatures of the model without thermal conduction (Tmax ~ 7400 K) or the rest of models (Tmax ~ 6300 K) [Fig. 3(e)], the decomposition of water vapor and •OH radicals is highly promoted [Fig. 4(a) and (b)]. In addition, the consumption of O radicals is reduced either by the increase of the backward kinetic constant of the 3rd reaction or by the decrease of mass action of water vapor into the 6th chemical reaction [Fig. 4(c) and 4(d)].
The production rate of H2 and O radicals is also increased by the 12th reaction, where H• and •OH radicals are consumed. The consumption of H2 molecules is reduced [Fig. 4(e)], in spite of the increase of the forward reaction kinetic constant, due to the higher dissociation of •OH radicals according to 2nd reaction which liberates a huge amount of O and H• radicals. By combining all these observations, we can conclude that at bubble temperatures of ~5500 K (PA = 2 atm, f = 500 kHz), the formation of •OH radicals is found to be promoted than that of O, H• and H2 molecules. However, at higher temperatures (greater than 5500 K), the reverse scenario is observed [Fig. 2(a)-(b)].
At PA = 3 atm [Fig. 2(c) and (d)], it is seen that the same trend found in the previous case (PA = 2 atm) is observed herein. The maximum production rate of •OH radicals is given at R0 = 5 µm, where the peak temperatures attained at this bubble size are around 5500 K (for all models). Therefore, the same interpretations made for the previous case of PA = 2 atm are applicable here. On the other hand, the maximal temperatures retrieved for the models without thermal conduction, without reactions heat, without mass transport or normal model are 13768 K (R0 = 1.5 µm), 11577 K (R0 = 1.5 µm), 19946 K (R0 = 0.5 µm) and 8800 K (R0 = 1.5 µm), respectively [Fig. 3(f)]. It should be noted that the peak temperature retrieved for the model without mass transport originate from the magnitude of acoustic power applied on bubble wall (PA = 3 atm), which promotes the increase of bubble temperature in the absence of water vapor. The elimination of vapor participates largely in the decrease of the bubble heat capacity. Consequently, during the compression phase, the bubble core temperature increases rapidly due to this decrease of heat capacity, the decrease of energy carried out by condensation and finally the amount of endothermal reactions taking place into the cavitation during the collapse phase. The same trend is found in [50], [60], [65]. For these maximal temperatures, it is clear that the thermal dissociation of water vapor and •OH radicals is highly promoted (for all models). On the other hand, the production rate of H• and O is largely accelerated [Fig. 2(c) and (d)] as it is explained previously for the case of acoustic amplitude of 2 atm. For H2 [Fig. 2(d)], some stabilization of the production rate is observed compared to its yield for the acoustic pressure PA = 2 atm [Fig. 2(b)]. This is due to the rapid increase of the forward reaction kinetic constant compared to that of the backward reaction. Consequently, H2 molecules are highly consumed at around the bubble temperature of 8000 K (for all models) [Figs. 3(e) and 2(d)]. In spite of the higher temperature found for the model without mass transport, it is seen in Fig. 2(c) and (d) that the minimal production rates of H•, O, •OH and H2 are observed only for this model. This is because of the elimination of nonequilibrium evaporation and condensation mechanism, which implies that during the compression phase no sufficient water vapor is available to increase bubble production. On the other hand, it is seen from Fig. 2(c) and (d) that the model without thermal conduction persists to be dominant compared to the other models, where all the main species created by this model are greater than those produced by the rest of models. Also, it is shown from Fig. 2(c) and (d) that the production rates of O, •OH, H• and H2 for the model without reactions heat is greater than those of the normal model, which is explained by the introduction of the reactions heat into bubble internal energy balance for the normal model. Bearing in mind all these considerations, it is clear that at lower temperatures (for all models) the formation of •OH radicals is favored compared to the other main species (O, H and H2). However, at the higher temperatures, it is found that the thermal dissociation of water vapor and •OH radicals are largely promoted, which means that in this case the yield of O, H2 and H• radicals is rapidly accelerated.
4. Conclusion
The elimination effect of thermal conduction, reactions heat and mass transport mechanism on the production rates of the main species (i.e., O, H, •OH and H2) has been studied on a range of ambient bubble radii for a wide range of frequencies and acoustic ampitudes. The results of the adopted models (without mass transport, thermal conduction and reactions heat) were compared to the normal model where all the mechanisms are taken into account. It has been shown that the thermal conduction mechanism has a great impact on bubble internal energy balance compared to the other mechanisms. This effect is increased especially when the temperature of the external bubble wall is considered constant. In addition, the important increase of the main species yield for the model without reactions heat was revealed compared to the normal model. On the other hand, the model without mass transport has given the lowest production rates compared to the other models even if the peak temperature retrieved for this model is the highest. It was found that at lower temperatures (around 5500 K), the formation of •OH radicals was promoted compared to the other species. Nevertheless, at higher temperatures (>5500 K), the yield rates of O, H2 and H• radicals is favored than that of •OH radicals. However, at the bubble temperatures higher than 8000 K, the production rate of H2 is relatively lowered conversely to the increase of the other species. It has been demonstrated that the ambient bubble radius of the maximal response (maximal production rate) is shifted toward the smaller bubble sizes, when the acoustic amplitude is increased or if the ultrasound is reduced. Finally, considering that the thermal conduction is an important factor, a comparison of simulation with different noble gases having different thermal conductivity may provide some additional information. This issue will be treated independently in further investigations.
CRediT authorship contribution statement
Aissa Dehane: Conceptualization, Methodology, Software, Formal analysis, Writing - original draft, Writing - review & editing. Slimane Merouani: Conceptualization, Project administration, Supervision, Visualization, Writing - review & editing, Methodology, Formal analysis, Writing - review & editing. Oualid Hamdaoui: Conceptualization, Supervision, Visualization, Validation, Writing - review & editing. Abdulaziz Alghyamah: Visualization, Writing - review & editing.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This study was supported by The Ministry of Higher Education and Scientific Research of Algeria (project No. A16N01UN250320180001) and the General Directorate of Scientific Research and Technological Development (GD-SRTD). The authors extend their appreciation to the Deputyship for Research & Innovation, “Ministry of Education” in Saudi Arabia for funding this research work through the project No. IFKSURG-1441-501.
References
- 1.Pankaj, Ashokkumar M. Springer; 2011. Theoretical and Experimental Sonochemistry Involving Inorganic Systems.https://www.springer.com/gp/book/9789048138869 [Google Scholar]
- 2.Bhangu S.K., Ashokkumar M. Theory of sonochemistry. Top. Curr. Chem. 2016;374:56. doi: 10.1007/s41061-016-0054-y. [DOI] [PubMed] [Google Scholar]
- 3.Flint E.B., Suslick K.S. The temperature of cavitation. Science. 1991;253:1397–1399. doi: 10.1126/science.253.5026.1397. [DOI] [PubMed] [Google Scholar]
- 4.Ashokkumar M. The characterization of acoustic cavitation bubbles - an overview. Ultrason. Sonochem. 2011;18:864–872. doi: 10.1016/j.ultsonch.2010.11.016. [DOI] [PubMed] [Google Scholar]
- 5.Yasui K., Tuziuti T., Kozuka T., Towata A., Iida Y. Relationship between the bubble temperature and main oxidant created inside an air bubble under ultrasound. J. Chem. Phys. 2007;127 doi: 10.1063/1.2790420. [DOI] [PubMed] [Google Scholar]
- 6.Adewuyi Y.G. Sonochemistry: environmental science and engineering applications. Ind. Eng. Chem. Res. 2001;40:4681–4715. doi: 10.1021/ie010096l. [DOI] [Google Scholar]
- 7.Son Y. Advanced oxidation processes using ultrasound technology for water and wastewater treatment. In: Ashokkumar M., editor. Handbook of Ultrasonics And Sonochemistry. Springer Science+Business Media; Singapore: 2015. pp. 1–20. [DOI] [Google Scholar]
- 8.C. Pétrier, The use of power ultrasound for water treatment, in: J.A. Gallego-Juarez, K. Graff (Eds.), Power ultrasonics: Application of h high-intensity ultrasound, Elsevier, 2015: pp. 939–963. DOI:10.1016/B978-1-78242-028-6.00031-4.
- 9.K.S. Suslick, Sonoluminescence and Sonochemistry, Encycl. Phys. Sci. Technol. 3rd Ed., R. A. Meyers (Ed.); Acad. Press. Inc. San Diego, 2001.
- 10.R.A. Torres-Palma, E.A. Serna-Galvis, Sonolysis, in: Suresh C. Ameta, R. Ameta (Eds.), Advanced oxidation processes for wastewater treatment. 1st ed: Emerging of green chemical technology, Elsevier Inc., 2018: pp. 177–213, DOI:10.1016/B978-0-12-810499–6.0.
- 11.Qiu P., Park B., Choi J., Thokchom B., Pandit A.B., Khim J. A review on heterogeneous sonocatalyst for treatment of organic pollutants in aqueous phase based on catalytic mechanism. Ultrason. Sonochem. 2018;45:29–49. doi: 10.1016/j.ultsonch.2018.03.003. [DOI] [PubMed] [Google Scholar]
- 12.Beckett M.A., Hua I. Impact of ultrasonic frequency on aqueous sonoluminescence and sonochemistry. J. Phys. Chem. A. 2001;105:3796–3802. doi: 10.1021/jp003226x. [DOI] [Google Scholar]
- 13.Pétrier C., Francony A. Ultrasonic waste-water treatment: incidence of ultrasonic frequency on the rate of phenol and carbon tetrachloride degradation. Ultrason. Sonochem. 1997;4:295–300. doi: 10.1016/S1350-4177(97)00036-9. [DOI] [PubMed] [Google Scholar]
- 14.Kanthale P., Ashokkumar M., Grieser F. Sonoluminescence, sonochemistry (H2O2 yield) and bubble dynamics: frequency and power effects. Ultrason. Sonochem. 2008;15:143–150. doi: 10.1016/j.ultsonch.2007.03.003. [DOI] [PubMed] [Google Scholar]
- 15.Merouani S., Hamdaoui O., Saoudi F., Chiha M. Influence of experimental parameters on sonochemistry dosimetries: KI oxidation, Fricke reaction and H2O2 production. J. Hazard. Mater. 2010;178:1007–1014. doi: 10.1016/j.jhazmat.2010.02.039. [DOI] [PubMed] [Google Scholar]
- 16.Merouani S., Hamdaoui O., Saoudi F., Chiha M. Sonochemical degradation of Rhodamine B in aqueous phase: effects of additives. Chem. Eng. J. 2010;158:550–557. doi: 10.1016/j.cej.2010.01.048. [DOI] [PubMed] [Google Scholar]
- 17.Brotchie A., Grieser F., Ashokkumar M. Effect of power and frequency on bubble-size distributions in acoustic cavitation. Phys. Rev. Lett. 2009;102:1–4. doi: 10.1103/PhysRevLett.102.084302. [DOI] [PubMed] [Google Scholar]
- 18.Torres R.A., Pétrier C., Combet E., Carrier M., Pulgarin C. Ultrasonic cavitation applied to the treatment of bisphenol A. Effect of sonochemical parameters and analysis of BPA by-products. Ultrason. Sonochem. 2008;15:605–611. doi: 10.1016/j.ultsonch.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 19.Beckett M.A., Hua I. Elucidation of the 1,4-dioxane decomposition pathway at discrete ultrasonic frequencies. Environ. Sci. Technol. 2000;34:3944–3953. doi: 10.1021/es000928r. [DOI] [Google Scholar]
- 20.Jiang Y., Petrier C., Waite T.D. Sonolysis of 4-chlorophenol in aqueous solution: effects of substrate concentration, aqueous temperature and ultrasonic frequency. Ultrason. Sonochem. 2006;13:415–422. doi: 10.1016/j.ultsonch.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 21.Ferkous H., Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Comprehensive experimental and numerical investigations of the effect of frequency and acoustic intensity on the sonolytic degradation of naphthol blue black in water. Ultrason. Sonochem. 2015;26:30–39. doi: 10.1016/j.ultsonch.2015.02.004. [DOI] [PubMed] [Google Scholar]
- 22.Yasui K. Influence of ultrasonic frequency on multibubble sonoluminescence. J. Acoust. Soc. Am. 2002;112:1405–1413. doi: 10.1121/1.1502898. [DOI] [PubMed] [Google Scholar]
- 23.Mead E.L., Sutherland R.G., Verrall R.E. The effect of ultrasound on water in the presence of dissolved gases. Can. J. Chem. 1976;54:1114–1120. doi: 10.1139/v76-159. [DOI] [Google Scholar]
- 24.Chadi N.E., Merouani S., Hamdaoui O. Characterization and application of a 1700 kHz-acoustic cavitation field for water decontamination: a case study with toluidine blue. Appl. Water Sci. 2018;8(160):1–11. doi: 10.1007/s13201-018-0809-4. [DOI] [Google Scholar]
- 25.Hua I., Hoffmann M.R. Optimization of ultrasonic irradiation as an advanced oxidation technology. Environ. Sci. Technol. 1997;31:2237–2243. doi: 10.1021/es960717f. [DOI] [Google Scholar]
- 26.Merouani S., Ferkous H., Hamdaoui O., Rezgui Y., Guemini M. New interpretation of the effects of argon-saturating gas toward sonochemical reactions. Ultrason. Sonochem. 2015;23:37–45. doi: 10.1016/j.ultsonch.2014.09.009. [DOI] [PubMed] [Google Scholar]
- 27.Boutamine Z., Hamdaoui O., Merouani S. Sonochemical and photosonochemical degradation of endocrine disruptor 2-phenoxyethanol in aqueous media. Sep. Purif. Technol. 2018;206:356–364. doi: 10.1016/j.seppur.2018.06.010. [DOI] [Google Scholar]
- 28.Merouani S., Hamdaoui O., Boutamine Z., Rezgui Y., Guemini M. Experimental and numerical investigation of the effect of liquid temperature on the sonolytic degradation of some organic dyes in water. Ultrason. Sonochem. 2016;28:382–392. doi: 10.1016/j.ultsonch.2015.08.015. [DOI] [PubMed] [Google Scholar]
- 29.Chadi N.E., Merouani S., Hamdaoui O., Bouhelassa M. New aspect of the effect of liquid temperature on sonochemical degradation of nonvolatile organic pollutants in aqueous media. Sep. Purif. Technol. 2018;200:68–74. doi: 10.1016/j.seppur.2018.01.047. [DOI] [Google Scholar]
- 30.Yasui K., Tuziuti T., Iida Y., Mitome H. Theoretical study of the ambient-pressure dependence of sonochemical reactions. J. Chem. Phys. 2003;119:346. doi: 10.1063/1.1576375. [DOI] [Google Scholar]
- 31.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Computer simulation of chemical reactions occurring in collapsing acoustical bubble: dependence of free radicals production on operational conditions. Res. Chem. Intermed. 2015;41:881–897. doi: 10.1007/s11164-013-1240-y. [DOI] [Google Scholar]
- 32.S. Merouani, O. Hamdaoui, Sonochemical treatment of textile wastewater, in: M.P. Inamuddin, A. Asiri (Eds.), Water pollutution and remediation:Photocatalysis, Springer-Nature Switzerland, 2021. DOI:10.1007/978-3-030-54723-3_5.
- 33.Storey B.D., Szeri A.J., Engineering M. Water vapour, sonoluminescence and sonochemistry. Proc. R. Soc. London A Math. Phys. Eng. Sci. 2000;456:1685–1709. [Google Scholar]
- 34.Fuster D., Hauke G., Dopazo C. Influence of the accommodation coefficient on nonlinear bubble oscillations. J. Acoust. Soc. Am. 2010;128:5–10. doi: 10.1121/1.3436520. [DOI] [PubMed] [Google Scholar]
- 35.Kerboua K., Hamdaoui O. Influence of reactions heats on variation of radius, temperature, pressure and chemical species amounts within a single acoustic cavitation bubble. Ultrason. - Sonochem. 2018;41:449–457. doi: 10.1016/j.ultsonch.2017.10.001. [DOI] [PubMed] [Google Scholar]
- 36.Yasui K. Chemical reactions in a sonoluminescing bubble. J. Phys. Soc. Japan. 1997;66:2911–2920. doi: 10.1143/JPSJ.66.2911. [DOI] [Google Scholar]
- 37.Yasui K. Effects of thermal conduction on bubble dynamics near the sonoluminescence threshold. J. Acoust. Soc. Am. 1995;98:2772–2782. doi: 10.1121/1.413242. [DOI] [Google Scholar]
- 38.Shen Y., Yasui K., Zhua T., Ashokkumar M. A model for the effect of bulk liquid viscosity on cavitation bubble dynamics. Phys. Chem. Chem. Phys. 2017;19:20635–20640. doi: 10.1039/C7CP03194G. [DOI] [PubMed] [Google Scholar]
- 39.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Energy analysis during acoustic bubble oscillations: relationship between bubble energy and sonochemical parameters. Ultrasonics. 2014;54:227–232. doi: 10.1016/j.ultras.2013.04.014. [DOI] [PubMed] [Google Scholar]
- 40.Burdin F., Tsochatzidis N.A., Guiraud P., Wilhelm A.M., Delmas H. Characterisation of the acoustic cavitation cloud by two laser techniques. Ultrason. Sonochem. 1999;6:43–51. doi: 10.1016/S1350-4177(98)00035-2. [DOI] [PubMed] [Google Scholar]
- 41.Tsochatzidis N.A., Guiraud P., Wilhelm A.M., Delmas H. Determination of velocity, size and concentration of ultrasonic cavitation bubbles by the phase-Doppler technique. Chem. Eng. Sci. 2001;56:1831–1840. doi: 10.1016/S0009-2509(00)00460-7. [DOI] [Google Scholar]
- 42.Avvaru B., Pandit A.B. Oscillating bubble concentration and its size distribution using acoustic emission spectra. Ultrason. Sonochem. 2009;16:105–115. doi: 10.1016/j.ultsonch.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 43.Iida Y., Ashokkumar M., Tuziuti T., Kozuka T., Yasui K., Towata A., Lee J. Bubble population phenomena in sonochemical reactor: I Estimation of bubble size distribution and its number density with pulsed sonication - laser diffraction method. Ultrason. Sonochem. 2010;17:473–479. doi: 10.1016/j.ultsonch.2009.08.018. [DOI] [PubMed] [Google Scholar]
- 44.Labouret S., Frohly J. Bubble size distribution estimation via void rate dissipation in gas saturated liquid. Application to ultrasonic cavitation bubble field. Eur. Phys. J. Appl. Phys. 2002;19:39–54. doi: 10.1051/epjap:2002047. [DOI] [Google Scholar]
- 45.Yasui K., Tuziuti T., Kanematsu W. Extreme conditions in a dissolving air nanobubble. Phys. Rev. E. 2016;94:013106. doi: 10.1103/PhysRevE.94.013106. [DOI] [PubMed] [Google Scholar]
- 46.Holzfuss J. Unstable diffusion and chemical dissociation of a single sonoluminescing bubble. Phys. Rev. E. 2005;71:026304-1–026304-5. doi: 10.1103/PhysRevE.71.026304. [DOI] [PubMed] [Google Scholar]
- 47.Toegel R., Lohse D. Phase diagrams for sonoluminescing bubbles: a comparison between experiment and theory. J. Chem. Phys. 2003;118:1863–1875. doi: 10.1063/1.1531610. [DOI] [Google Scholar]
- 48.Yasui K. Effect of liquid temperature on sonoluminescence. Phys. Rev. E. 2001;64 doi: 10.1103/PhysRevE.64.016310. [DOI] [PubMed] [Google Scholar]
- 49.Yasui K. Waseda University; 1996. A New Formulation of Bubble Dynamics for Sonoluminescence. [Google Scholar]
- 50.Tögel R. University of Twente; UK: 2002. Reaction-Diffusion Kinetics of a Single Sonoluminescing Bubble. PhD Thesis. [Google Scholar]
- 51.Yasui K. Single-bubble sonoluminescence from noble gases. Phys. Rev. E. 2001;63 doi: 10.1103/PhysRevE.63.035301. [DOI] [PubMed] [Google Scholar]
- 52.Tuziuti T., Hatanaka S., Yasui K., Kozuka T., Mitome H. Effect of ambient-pressure reduction on multibubble sonochemiluminescence. J. Chem. Phys. 2002;116:6221. doi: 10.1063/1.1461357. [DOI] [Google Scholar]
- 53.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Mechanism of the sonochemical production of hydrogen. Int. J. Hydrogen Energy. 2015;40:4056–4064. doi: 10.1016/j.ijhydene.2015.01.150. [DOI] [Google Scholar]
- 54.Merouani S., Hamdaoui O. The size of active bubbles for the production of hydrogen in sonochemical reaction field. Ultrason. Sonochem. 2016;32:320–327. doi: 10.1016/j.ultsonch.2016.03.026. [DOI] [PubMed] [Google Scholar]
- 55.Petrier C., David B., Laguian S. Ultrasonic degradation at 20 kHz and 500 kHz of atrazine and pentachlorophenol in aqueous solution: preliminary results. Chemosphere. 1996;32:1709–1718. doi: 10.1016/0045-6535(96)00088-4. [DOI] [Google Scholar]
- 56.Merouani S., Ferkous H., Hamdaoui O., Rezgui Y., Guemini M. A method for predicting the number of active bubbles in sonochemical reactors. Ultrason. Sonochem. 2014;22:51–58. doi: 10.1016/j.ultsonch.2014.07.015. [DOI] [PubMed] [Google Scholar]
- 57.Yang L., Sostaric J.Z., Rathman J.F., Weavers L.K. Effect of ultrasound frequency on pulsed sonolytic degradation of octylbenzene sulfonic acid. J. Phys. Chem. B. 2008;112:852–858. doi: 10.1021/jp077482m. [DOI] [PubMed] [Google Scholar]
- 58.Koda S. A standard method to calibrate sonochemical efficiency of an individual reaction system. Ultrason. Sonochem. 2003;10:149–156. doi: 10.1016/S1350-4177(03)00084-1. [DOI] [PubMed] [Google Scholar]
- 59.Kang J.-W., Hung H.-M., Lin A., Hoffmann M.R. Sonolytic destruction of methyl tert-butyl ether by ultrasonic irradiation: the role of O3, H2O2, frequency, and power density. Environ. Sci. Technol. 1999;33:3199–3205. doi: 10.1021/es9810383. [DOI] [Google Scholar]
- 60.Yasui K., Tuziuti T., Lee J., Kozuka T., Towata A., Iida Y. The range of ambient radius for an active bubble in sonoluminescence and sonochemical reactions. J. Chem. Phys. 2008;128 doi: 10.1063/1.2919119. [DOI] [PubMed] [Google Scholar]
- 61.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Sensitivity of free radicals production in acoustically driven bubble to the ultrasonic frequency and nature of dissolved gases. Ultrason. Sonochem. 2014;22:41–50. doi: 10.1016/j.ultsonch.2014.07.011. [DOI] [PubMed] [Google Scholar]
- 62.Sochard S., Wilhelm A.M., Delmas H. Modelling of free radicals production in a collapsing gas-vapour bubble. Ultrason. Sonochem. 1997;4:77–84. doi: 10.1016/S1350-4177(97)00021-7. [DOI] [PubMed] [Google Scholar]
- 63.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Computational engineering study of hydrogen production via ultrasonic cavitation in water. Int. J. Hydrogen Energy. 2016;41:832–844. doi: 10.1016/j.ijhydene.2015.11.058. [DOI] [Google Scholar]
- 64.Merouani S., Hamdaoui O., Rezgui Y., Guemini M. Effects of ultrasound frequency and acoustic amplitude on the size of sonochemically active bubbles-theoretical study. Ultrason. Sonochem. 2013;20:815–819. doi: 10.1016/j.ultsonch.2012.10.015. [DOI] [PubMed] [Google Scholar]
- 65.Hilgenfeldt S., Lohse D. Predictions for upscaling sonoluminescence. Phys. Rev. Lett. 1999;82:1036–1039. doi: 10.1103/PhysRevLett.82.1036. [DOI] [Google Scholar]