Abstract
Controlling gut inflammation is important in managing gut disorders in the piglet after weaning. Establishing patterns of inflammation markers in the time subsequent to weaning is important for future research to determine whether interventions are effective in controlling gut inflammation. The objective of this study was to evaluate the intestinal inflammatory response during the postweaning period in piglets. A 45-d study included 108 piglets (weaned at 22 d, body weight 5.53 ± 1.19 kg), distributed in 12 pens with nine pigs per pen. Histomorphometry, gene expression of pro- and anti-inflammatory cytokines, and the quantity of immunoglobulin (Ig) A producing cells were measured in jejunum, ileum, and colon on days 0, 15, 30, and 45 postweaning. Cytokine gene expression in peripheral blood mononuclear cells and Ig quantities were analyzed in blood from piglets on days 0, 15, 30, and 45 postweaning. Histomorphometrical results showed a lower villus length directly after weaning. Results demonstrated a postweaning intestinal inflammation response for at least 15 d postweaning by upregulation of IgA producing cells and IFN-γ, IL-1α, IL-8, IL-10, IL-12α, and TGF-β in jejunum, ileum, and colon. IgM and IgA were upregulated at day 30 postweaning. IgG was downregulated at day 15 postweaning. The results indicate that weaning in piglets is associated with a prolonged and transient response in gene expression of pro- and anti-inflammatory cytokines and IgA producing cells in the intestine.
Keywords: cytokines, immunoglobulin, inflammation, intestine, piglets, weaning
Introduction
Research for new strategies that promote gastrointestinal health in pigs is important because of an urgent need to reduce antibiotic usage and minimize risk of bacterial resistance against antibiotics, as part of the One Health strategy currently demanded by consumers (Council of the EU, 2016). Gastrointestinal health, or gut health, can be described as a generalized condition of homeostasis in the gastrointestinal tract (GIT), with respect to its overall structure and function (Pluske et al., 2018b). The GIT is essential in the maintenance of health in animals. It contains a large mucosal immune system and forms a physical barrier that prevents toxic compounds and pathogens from entering the intestinal mucosa and systemic circulation, while the GIT simultaneously has a function for the uptake of nutrients. The intestinal epithelium plays an active role in organ integrity and body defense locally and systemically (Eckmann, 1995; Pitman and Blumberg, 2000).
Weaning is one of the most stressful events pigs will face during their lifetime. Maternal separation is a major stressor to the weaned piglet, and additional psychosocial and immunological stressors increase the stress load during this time (Pluske et al., 1997; Moeser et al., 2007). The timing of commercial weaning (at ~21 d of age) also coincides with a period of declining passive immunity from sow milk contributing an additional challenge to the pig. Results of several studies have demonstrated that weaning in pigs induces a breakdown in intestinal barrier function characterized by a significant decline in intestinal trans-epithelial electrical resistance and increased permeability (Moeser et al., 2007a; Hu et al., 2013). Compared with similar age, nonweaned littermate pigs, weaned pigs exhibited increased intestinal permeability that was most pronounced at 24 hr postweaning and then gradually declined over the first 2 wk postweaning (Moeser et al., 2007b). At the same time that epithelial barrier function is disrupted, an upregulation of pro-inflammatory cytokines has been reported indicating a robust activation of the GI immune system following the process of weaning in piglets (McCracken et al., 1999; Pié et al., 2004; Hu et al., 2013). Furthermore, studies report the changes in the expression of inflammatory cytokines in the intestine of humans and animals during enteric infection and intestinal inflammatory diseases (Murtaugh et al., 1996; McClane and Rombeau, 1999; Oswald et al., 2001). Both in vitro and in vivo investigations show that uncontrolled production of pro-inflammatory cytokines can negatively influence gut integrity and epithelial functions, including permeability to macromolecules and transport of nutrients and ions (McKay and Baird, 1999). Along with disturbances in intestinal permeability, there is also accumulating evidence for lasting functional changes in the GI immune barrier function and disease susceptibility to bacterial challenges such as Escherichia coli (McLamb et al., 2013) and Streptococcus suis, which might use the disturbance in paracellular tight junctions to get to the blood stream, resulting in a septicemia (Swildens et al., 2004).
The gut mucosal immune system alone contains most of lymphocytes in the body and has a greater concentration of antibodies than other tissues in the body (Burkey et al., 2009). One of the strategies that the host utilizes to avoid an inflammatory response against the microbiota is to use the intestinal barrier, including the mucous layer and immunoglobulin (Ig) A, an antibody isotype specialized in mucosal protection (Cerutti and Rescigno, 2008; Gutzeit et al., 2014) and produced locally by plasma cells present in the mucosal wall. The naïve B-cell precursor of an IgA-secreting plasma cell is activated in the Peyer’s patches and mesenteric lymph nodes. Production of IgA is controlled by cytokine-producing T-cells within the GALT and by cytokine released from the mucosa. Within the GALT, the Th1 cytokines, interferon γ (IFN-γ) and tumor necrosis factor-β (TNF-β), downregulate IgA production, whereas the Th2 cytokines, interleukin (IL)-4, IL-5, IL-6, and IL-10, upregulate IgA production (Kramer et al., 1995; Ramsay, 1995; Stokes, 2017). A balance between Th1 and Th2 response is necessary for maintaining normal IgA immune responses, with no response in favor of the other. Peyer’s patches in the GALT are rich in cytokines with IgA-inducing functions, including TGF-β (Gonnella et al., 1998). IgA secreted into the gut lumen binds to the layer of mucus coating the epithelial surface, and it prevents the adherence of micro-organisms, as well as neutralizing their toxins or enzymes.
The gastrointestinal immune system is geared toward tolerance, in contrary to the systemic immune system, and responds to intestinal content (microbiota and feed components), and this reaction can lead to tolerance (e.g., for commensal bacteria), or to a defense reaction (Niewold, 2015). Controlling gut inflammation is important in managing gut disorders in the piglet after weaning. Establishing patterns of inflammation markers in the time subsequent to weaning are important for future research to determine whether interventions are effective in controlling gut inflammation. In the GIT, use of nutritional strategies to prevent intestinal inflammation caused by pathogenic enteric organisms is an important pillar in health maintenance and can prevent the requirement for antimicrobial intervention, as controlling postweaning intestinal inflammation is important in managing postweaning gut disorders in piglet. The objective of this study was to evaluate the intestinal inflammatory response to weaning in piglets.
Material and Methods
All the experimental procedures described in this research were carried out under the welfare rules stated in R.D. 1135/2002 for the protection of pigs and the trial was approved by the Bioethical Committee of the University of Murcia (CEEA-OH 465/2018).
Animals, diet, and experimental design
This study was performed in the Swine Production Unit at the Veterinary Teaching Farm of the University of Murcia (Spain). A total of 108 piglets (Large White) were weaned at 22 ± 3 d of age with an average body weight (BW) of 5.53 ± 1.19 kg and male:female 1:1. Piglets were obtained from 12 different litters from sows with average parity of 3.43. After weaning, all pigs were randomly allotted to 12 pens, with 9 animals per pen (pen size: 0.61 × 1.2 m). Pigs were housed in an environmentally controlled unit with natural daylight for 45 d. The room temperature was 25 to 27 °C. Pens were fully slated (plastic slat) and were equipped with one nipple drinker (ad libitum) and one 4 spaced-feeder. Piglets were routinely vaccinated against porcine circovirus-2 (Porcilis PCV-2, MSD) at day 4 postweaning and offered unmedicated feed or water.
A standard 2-phase experimental diet was produced at a local feed mill (Pigalomar, Spain; Table 1). All diets were pelleted (4 mm) and formulated to meet the current estimates for nutrient growth requirements for nursery pigs (NRC, 2012). Spray-dried plasma, antibiotics, and pharmaceutical levels of zinc oxide were not included in the diets.
Table 1.
Composition of the experimental diets
| Item | Phase 1 days 0 to 14 | Phase 2 days 15 to 45 |
|---|---|---|
| Ingredients, % | ||
| Barley | 29.98 | 25.00 |
| Wheat | 24.00 | 26.89 |
| Corn | 17.17 | 19.50 |
| Soybean Meal 47 crude protein | 6.00 | 16.83 |
| Ca carbonate | 0.45 | 0.61 |
| Monocalcium phosphate | 0.75 | 0.78 |
| Soybean oil | 3.50 | 3.67 |
| Intestinal swine mucose hydrolyzate | 2.50 | 0.00 |
| Milkpowder | 5.00 | 0.00 |
| Fysal MP1 | 0.30 | 0.30 |
| Salt | 0.30 | 0.44 |
| l-Valine (96.5%) | 0.050 | 0.025 |
| dl-Methionine (99%) | 0.175 | 0.175 |
| l-Lysine HCl (98%) | 0.542 | 0.525 |
| l-Threonine (98%) | 0.258 | 0.250 |
| l-Tryptophan (98%) | 0.033 | 0.008 |
| Protein concentrate2 | 6.00 | 2.00 |
| TN Trouwmix 30 premix3 | 3.00 | 3.00 |
| Calculated content4, % | ||
| Dry matter | 89.60 | 89.17 |
| Net energy, kcal | 2,559 | 2,549 |
| SID lysine | 1.15 | 1.17 |
| SID methionine | 0.405 | 0.406 |
| SID methionine + cysteine | 0.686 | 0.705 |
| SID tryptophan | 0.241 | 0.230 |
| SID threonine | 0.750 | 0.761 |
| Crude protein | 16.67 | 17.72 |
| Crude fiber | 2.98 | 3.14 |
| Ash | 4.72 | 4.74 |
| Neutral detergent fiber | 11.3 | 11.7 |
| Sodium | 0.24 | 0.22 |
| Chloride | 0.45 | 0.44 |
| Calcium | 0.55 | 0.59 |
| Phosphorus | 0.55 | 0.55 |
| Copper, mg/kg (as SO4) | 150 | 150 |
| Manganese, mg/kg (as MnO) | 50 | 50 |
| Zinc, mg/kg (as ZnO) | 105 | 105 |
1Blend of free and buffered organic acids.
2Gluten meal, extruded soybean meal, and potato protein.
3Vitamin and mineral premix provided the following per kilogram of diet: vitamin A, 15,000 IU; vitamin D, 2,000 IU; vitamin E, 100 IU; 30 μg of vitamin B12; vitamin K, 2 mg; d-pantothenicacid 15 mg as calcium pantothenate; 30 mg of nicotinicacid; choline, 150 mg as betaine hydrchloride; Mn, 50 mg as manganese oxide; Zn, 105 mg as zinc oxide; Fe, 100 mg as iron sulphate; Cu, 120 mg as copper sulphate; I, 1.5 mg as potassium iodide; Se, 0.42 mg as sodium selenite; 6-phytase 1500 FTU.
4Nutrient levels are calculated. SID means standardized ileal digestible.
Clinical observations and sample collection
Individual BW was recorded at 18 hr postweaning (day 0), days 15, 30, and 45, and average daily gain was calculated. At day 0, 10 randomly selected piglets were euthanized using an overdose of tiobarbital IV (50 mg/kg BW, Tiobarbital Braun Medical S.A., Barcelona, Spain) to obtain blood from the vena jugular and intestinal tissue samples as a basal group. Prior to euthanasia, 10 mL blood per pig was collected into ethylenediaminetetraacetic acid tubes (Vacutainer, Becton Dickinson, UK) from each sacrificed pig. Subsequently, animals were similarly sacrified per complete pen at day 15 (n = 26), day 30 (n = 34), and day 45 (n = 34) to obtain blood and tissue samples. Peripheral blood mononuclear cells (PBMCs) were isolated by Ficoll Histopaque gradient and preserved in RNAlater (Life Technologies) at −80 °C after 24 hr of refrigeration at 8 °C. Whole blood was centrifuged to isolate serum and plasma (251 rcf, 10 min at room temperature), and preserved at −80 °C until analysis. Tissue samples were obtained from the ileum (5 cm adjacent to ileocecal valve), middle section of jejunum and apex section of the spiral colon. All samples included the full thickness of the organ. After being gently rinsed in water, each sample was divided in two subsamples: 1 (40 mg) was preserved in RNAlater (Life Technologies) and the other was fixed in 10% buffered formaldehyde.
Sample analysis
Histomorphometrical and immunohistochemical analyses
Five photomicrographs from each section of jejunum and ileum were taken with a Zeiss Axiocam 503 color (Carl Zeiss, Oberkochen, Germany) coupled to a Zeiss Axioskop 40 microscope (Carl Zeiss, Oberkochen, Germany), with 10× magnification. The height (tip to villous-crypt junction) and crypt depth (from villus-crypt junction to the base of villous) of villi were measured using the ZEISS Efficient Navigation software (Carl Zeiss). Ten randomly selected well-oriented intact villi and crypts were measured per piglet and tissue. The mean villus height and crypt depth of each intestinal section was calculated. The villus/crypt ratio was calculated by dividing villus height by crypt depth. All morphometric measurements were performed by the same researcher, who was blinded to the treatments.
The avidin–biotin–peroxidase complex technique was used for the detection of IgA producing cells in jejunum, ileum, and colon tissue. Tissue sections fixed in formaldehyde were processed routinely for histopathology. Briefly, the samples were embedded in paraffin-wax and 5 μm thick slides were obtained. The samples were dewaxed and dehydrated with graded ethanol and the endogenous peroxidase activity was quenched in 3% H2O2 in methanol for 30 min. Samples were pretreated with 10% pronase in TBS (Sigma-Aldrich) for antigen retrieval (12 min). Afterwards the samples were rinsed in TBS for (3 × 5 min) and incubated for 30 min with 100 μL of blocking solution per slide at 20 °C in a humid chamber. Subsequently the samples were incubated for 1 hr at 37 °C with the primary antibody (goat- anti-pig IgA, Bethyl) diluted 1:3,000 in TBS. The secondary antibody (biotin conjugate rabbit anti-goat, Dako), diluted 1:300 in TBS, was incubated for 30 min at 20 °C. The Vectastain Elite ABC kit (Vector) was applied for 1 hr at 20 °C. Positive labeling was detected using 3,3′-diaminobenzidine tetrahydrochloride (Dako). Sections were counterstained with Mayer’s haematoxylin, dehydrated and mounted. The number of IgA producing cells in the intestinal lamina propria was counted using a Zeiss Axioskop 40 microscope (Carl Zeiss) with a Spot Insight camera and the Spot Advanced software (Spot Imaging Solution, MI). Immunolabeled cells were counted in 10 non-overlapping consecutive high magnification fields and expressed as amount of cells/25.000 μm2.
Total serum Igs IgA, IgE, IgG, and IgM were quantified in blood samples by Enzyme Linked Immuno Sorbent Assay (Elabscience) per manufacturer instruction.
Gene expression for cytokines
Gene expression for cytokines IL-1α, IL-1β, IL-6, IL-8, IL-10, IL-12p35 (IL-12α), IL-12p40 (IL-12β), TNF-α, IFN-α, IFN-γ, and TGF-β was determined by means of relative quantification, using primers previously described by various authors (Table 2). Total RNA was isolated from 20 mg of tissue samples and PBMC by using the Micro RNeasy kit (Qiagen) and DNAc was synthetized using the Geneamp RNA PCR Core Kit (Life Technology) using oligo-dT as primers to get cDNA only from mRNA. The PCRs were performed using a 7300 ABI thermocycler (Life Technologies) and the GoTaq q-PCR Master Mix (Promega) with SYBR-Green chemistry. The specificity of the reaction was assessed by analyzing the melting curve. The samples were normalized using the average Ct for glyceraldehyde-3-phosphatedehydrogenase (GAPDH), cyclophilin and β-actin. The minimum correlation coefficient for the standard curves was 0.972 and the efficiency of q-PCR was between 91% and 105%. The expression for each sample was calculated (Pfaffl, 2001), correcting to the PCRs efficiency, which was calculated by serial decimal dilutions and using the slope offered by the thermacycler software, and used as control group the animals sampled at day 0. The minimum correlation coefficient for the serial dilutions was 0.972 and the efficiency of q-PCR was between 91% and 105%. Data were expressed as fold change, normalized to the lowest value (which was assigned a value 1).
Table 2.
Primers for cytokines IL-1β, IL-6, IL-8, IL-10, IL-12p35, IL-12p40, TNF-α, IFN-α, IFN-γ, and TGF-β and primers for glyceraldehyde-3-phosphatedehydrogenase, cyclophilin, and β-actin1
| Primer forward (5′ → 3′) | Primer reverse (5′ → 3′) | References | |
|---|---|---|---|
| IFN-α | 5′-CCCCTGTGCCTGGGAGAT-3′ | 5′-AGGTTTCTGGAGGAAGAGAAGGA-3′ | Moue et al. (2008) |
| IFN-γ | 5′-TGGTAGCTCTGGGAAACTGAATG-3′ | 5′-GGCTTTGCGCTGGATCTG-3′ | Royaee et al. (2004) |
| TNF-α | 5′-ACTCGGAACCTCATGGACAG-3′ | 5′-AGGGGTGAGTCAGTGTGACC-3′ | Gabler et al. (2008) |
| IL-12p35 | 5′-AGTTCCAGGCCATGAATGCA-3′ | 5′-TGGCACAGTCTCACTGTTGA-3′ | Moue et al. (2008) |
| IL-12p40 | 5′-TTTCAGACCCGACGAACTCT-3′ | 5′-CATTGGGGTACCAGTCCAAC-3′ | Kim et al. (2010) |
| IL-10 | 5′-TGAGAACAGCTGCATCCACTTC-3′ | 5′-TCTGGTCCTTCGTTTGAAAGAAA-3′ | Royaee et al. (2004) |
| TGF-β | 5′-CACGTGGAGCTATACCAGAA-3′ | 5′-TCCGGTGACATCAAAGGACA-3′ | Moue et al. (2008) |
| IL-8 | 5´- GCTCTCTGTGAGGCTGCAGTTC-3´ | 5′-AAGGTGTGGAATGCGTATTTATGC-3´ | Bracarense et al. (2012) |
| IL-1α | 5´- GTGCTCAAAACGAAGACGAACC-3´ | 5′-CATATTGCCATGCTTTTCCCAGAA-3´ | Verpoest et al. (2017) |
| IL-1β | 5´-AACGTGCAGTCTATGGAGT-3´ | 5′-GAACACCACTTCTCTCTTCA-3´ | Borca et al. (2008) |
| IL-6 | 5′-CTGGCAGAAAACAACCTGAACC-3´ | 5′-TGATTCTCATCAAGCAGGTCTCC-3´ | Borca et al., 2008 |
| β-actin | 5´-CTACGTCGCCCTGGACTTC-3´ | 5´-GATGCCGCAGGATTCCAT-3´ | Skovgaard et al. (2009) |
| Cyclophilin | 5´-TGCTTTCACAGAATAATTCCAGGATTTA-3´ | 5´-GACTTGCCACCAGTGCCATTA-3´ | Duvigneau et al. (2005) |
| GAPDH | 5´-ACATGGCCTCCAAGGAGTAAGA-3´ | 5´-GATCGAGTTGGGGCTGTGACT-3´ | Duvigneau et al. (2005) |
1IFN-α, interferon alpha; IFN-ɣ, interferon gamma; IL-12p35, interleukin-12, subunit p35; IL-12p40, interleukin-12, subunit p40; IL-10, interleukin-10; TGF-β, transforming growth factor beta; IL-8, interleukin-8; IL-1α, interleukin-1 alpha; IL-1β, interleukin-1 beta; IL-6, interleukin-6; GAPDH, glyceraldehyde-3 phosphate dehydrogenase.
Statistical analysis
All data were processed using SAS (version 9.4, SAS Institute; Cary). The gene expression data were transformed as Log 2 of the value for normalization. A MIXED procedure was used to find statistical differences between time points for data with the individual pig as the experimental unit. The Spearman correlation coefficient was used to measure the relation between IgA producing cells in tissues and cytokine expression. Multivariate analyses were performed using SAS, and graphical presentation methods that were used to explore associations in the set of multiple variables were heatmaps, Principal component analysis (PCA; multidimensional scaling analysis) which were carried out based on the expression of the set of 11 normalized genes included in the 3 intestinal tissues, and Canonical discriminant analysis (CDA) was used comparing the data from the different time points. Significantly different means were separated using Tukey adjust. Statistical significance and tendency were considered at P ≤ 0.05 and 0.05 ≤ P ≤ 0.10, respectively.
Results
Performance and histomorphometrical results
The animals reached an average BW of 20.49 kg on day 45 of the experiment, with an average daily gain of 334 ± 90 g/d, with an average pen-based feed intake of 18.52 ± 2.3 kg. Throughout the study, no piglets developed diarrhea or other disease clinical signs. Two animals died over the experiment, showing no digestive or respiratory related lesions at necropsy.
Data on small intestine villi characteristics are presented in Table 3. The villous length and crypt depth in jejunal tissue is significantly lower on day 0 (day of weaning) compared with days 30 and 45 postweaning, and day 45 is increased compared with day 15. The ratio between villous length and crypt depth in the jejunum is significantly lower on days 30 and 45 compared with day 0, and day 45 is significantly lower as day 15. In ileal tissue, the villous length is lower on day 0 compared with days 15, 30, and 45, with no significant differences between those timepoint, the crypt depth is lower on day 0 compared with days 15, 30, and 45. Crypt depth is lower on day 15 compared with days 30 and 45. The ratio between villous length and crypt depth in the ileum is significantly lower on days 15, 30, and 45 compared with day 0.
Table 3.
Histomorphometrical intestinal measurements in piglets in piglets on the day of weaning (day 0) and on days 15, 30, and 45 postweaning
| Day 0 | Day 15 | Day 30 | Day 45 | SEM | P-value | ||
|---|---|---|---|---|---|---|---|
| Jejunum | Villous length, μm | 430a | 475ab | 506bc | 522c | 14 | 0.0005 |
| Crypt depth, μm | 223a | 296ab | 358bc | 333c | 12 | <0.0001 | |
| V/C ratio | 1.9507a | 1.6454ab | 1.4500b | 1.5547b | 0.073 | 0.0010 | |
| Ileum | Villous length, μm | 357a | 451b | 459b | 490b | 15 | <0.0001 |
| Crypt depth, μm | 172a | 274b | 335c | 316c | 13 | <0.0001 | |
| V/C ratio | 2.1758a | 1.7026b | 1.4190c | 1.5773bc | 0.090 | <0.0001 |
a–cDifferent superscripts within a row indicate a significant difference (P < 0.05).
Ig analysis in blood and tissue
Average count of IgA producing cells (per 25.000 μm2) in jejunum (P < 0.001) was increased on days 30 and 45 compared with days 0 and 15, with day 45 being increased compared with day 30. In the ileum and colon tissue, the average count of IgA producing cells increased on days 30 and 45 (P < 0.001) compared with days 0 and 15 (Table 4).
Table 4.
Pattern of average count of IgA producing cells in jejunum, ileum, and colon in piglets on the day of weaning (day 0) and on days 15, 30, and 45 postweaning
| Day 0 | Day 15 | Day 30 | Day 45 | SEM | P-value | |
|---|---|---|---|---|---|---|
| Jejunum | 11.80a | 11.00a | 19.61b | 24.60c | 1.101 | <0.0001 |
| Ileum | 9.58a | 9.75a | 16.62b | 17.44b | 0.901 | <0.0001 |
| Colon | 9.72a | 10.20a | 18.12b | 20.27b | 1.181 | <0.0001 |
a–bDifferent superscripts within a row indicate a significant difference (P < 0.05).
A correlation between IgA producing cells in different tissues considering all time points (days 0, 15, 30, and 45) was found, between jejunum and ileum (r = 0.672, P < 0.001) and between IgA producing cells in ileum and colon (r = 0.679, P < 0.001). A moderate correlation was found between IgA producing cells in jejunum and colon (r = 0.562, P < 0.001). Furthermore, a correlation existed between the IgA quantities in serum and the IgA producing cells in ileum (r = 0.2992, P = 0.037) and in colon (r = 0.32264, P = 0.024). No significant correlation was found between IgA quantities in serum and the IgA producing cells in jejunum. Table 5 presents the Ig quantities in serum of piglets over time. Amount of IgG was increased on day 45 compared with day 15 (P = 0.043), with days 0 and 30 having intermediary levels. A correlation between IgG in blood and PCV-2 titer existed (r = 0.3825, P = 0.013). The PCV-2 titer also showed a linear increase over time (data not shown). IgA quantities were increased on days 30 and 45 compared to day 0 (P = 0.015). Quantities of IgM in blood were increased on day 30 compared with day 0 (P < 0.001). IgE quantity was increased on day 30 compared with day 15 (P = 0.013).
Table 5.
IgG, IgE, IgA, and IgM quantities in blood of piglets on the day of weaning (day 0) and on days 15, 30, and 45 postweaning
| Day 0 | Day 15 | Day 30 | Day 45 | SEM | P-value | |
|---|---|---|---|---|---|---|
| IgG, mg/mL | 8.47ab | 6.82b | 7.32ab | 8.76a | 0.544 | 0.0428 |
| IgE, ng/mL | 26.50ab | 23.52b | 30.93a | 23.98b | 1.888 | 0.0129 |
| IgA, ug/mL | 65.59a | 98.313ab | 118.23b | 116.91b | 11.286 | 0.0145 |
| IgM, ug/mL | 78.04a | 100.63ab | 123.15bc | 140.00c | 9.598 | 0.0004 |
a–c Different superscripts within a row indicate a significant difference (P < 0.05).
Cytokine gene expression in tissue
Jejunum
Gene expression of IFN-γ showed a tendency for a decrease on day 45 compared with day 15 (P = 0.094). IL-1α gene expression was increased at day 30 compared with day 0 in jejunum (P < 0.001). Gene expression of IL-1α was reduced on day 45 compared with day 0 in jejunum. Gene expression for IL-1β was reduced on day 30 compared with all other time points (P < 0.001). IL-8 gene expression in jejunum (P < 0.001) decreased on day 30 compared with all other time points. A reduction in IL-10 gene expression in the jejunum was seen on day 45 compared with days 0 and 15 (P < 0.001). Il-12α gene expression showed a tendency for a reduction on day 45 compared with day 0 in the jejunum (P = 0.080), and a tendency for a reduction on day 30 compared with day 0 (P = 0.069). A reduction in IL-12β gene expression in jejunum was observed on day 45 compared with day 0 (P = 0.039). In jejunum, gene expression for TNF-α was reduced (P < 0.001) on days 15 and 45 compared with day 0. Gene expression of TGF-β was reduced in jejunum on day 30 compared with day 0 (P = 0.027; Table 6).
Table 6.
Pattern of Cytokine (Ck) mRNA expression in jejunum, ileum, colon, and PBMC in piglets on the day of weaning (day 0) and on days 15, 30, and 45 postweaning1
| Tissue | Ck | DAY 0 | DAY 15 | DAY 30 | DAY 45 | SEM | P-Value |
|---|---|---|---|---|---|---|---|
| Jejunum | IFN-α | 3.137 | 2.299 | 2.381 | 2.846 | 0.338 | 0.2559 |
| IFN-γ | 3.585xy | 4.090x | 3.645xy | 3.063y | 0.290 | 0.0939 | |
| IL-1α | 4.513b | 5.272b | 7.497a | 3.436c | 0.284 | <0.0001 | |
| IL-1β | 5.833a | 5.391a | 1.998b | 5.353a | 0.299 | <0.0001 | |
| IL-6 | 2.257 | 2.184 | 2.767 | 2.338 | 0.218 | 0.2664 | |
| IL-8 | 1.961a | 2.274a | 0.992b | 2.183a | 0.138 | <0.0001 | |
| IL-10 | 3.239a | 3.316a | 2.817ab | 2.142b | 0.198 | <0.0001 | |
| IL-12α | 8.103x | 7.201xy | 6.218y | 6.320y | 0.525 | 0.0804 | |
| IL-12β | 5.322a | 4.900ab | 4.788ab | 4.530b | 0.198 | 0.0391 | |
| TNF-α | 12.505a | 9.578b | 12.134a | 9.213b | 0.681 | 0.0005 | |
| TGF-β | 12.951a | 12.865ab | 12.038b | 12.429ab | 0.231 | 0.0267 | |
| Ileum | IFN-α | 3.004 | 3.051 | 2.354 | 2.990 | 0.367 | 0.5200 |
| IFN-γ | 6.107b | 7.742a | 6.955ab | 6.393b | 0.295 | 0.0009 | |
| IL-1α | 6.939b | 7.799bc | 9.791ab | 6.407a | 0.265 | <0.0001 | |
| IL-1β | 4.974a | 5.462a | 1.478b | 5.323a | 0.348 | <0.0001 | |
| IL-6 | 4.285a | 3.775ab | 3.179b | 3.093b | 0.217 | 0.0003 | |
| IL-8 | 2.599a | 2.787a | 1.783b | 2.845a | 0.163 | <0.0001 | |
| IL-10 | 3.217a | 3.229a | 3.149b | 3.120b | 0.018 | <0.0001 | |
| IL-12α | 4.096x | 3.591xy | 2.403y | 2.871xy | 0.479 | 0.0688 | |
| IL-12β | 2.460 | 2.795 | 2.406 | 2.441 | 0.175 | 0.3850 | |
| TNF-α | 11.122 | 11.044 | 9.375 | 9.396 | 0.682 | 0.1071 | |
| TGF-β | 10.775a | 11.123a | 9.991b | 10.303b | 0.129 | <0.0001 | |
| Colon | IFN-α | 2.426a | 1.858ab | 1.309b | 2.146a | 0.186 | 0.0005 |
| IFN-γ | 2.730a | 3.185b | 2.890ab | 2.914ab | 0.081 | 0.0017 | |
| IL-1α | 5.033c | 9.008a | 6.897b | 3.224d | 0.381 | <0.0001 | |
| IL-1β | 3.124bc | 4.653a | 2.352c | 3.877ab | 0.331 | <0.0001 | |
| IL-6 | 3.013 | 3.461 | 2.907 | 3.150 | 0.205 | 0.2857 | |
| IL-8 | 2.786b | 5.085a | 2.680b | 3.471b | 0.353 | <0.0001 | |
| IL-10 | 1.687b | 3.341a | 1.920b | 1.986b | 0.171 | <0.0001 | |
| IL-12α | 4.842ab | 6.284b | 3.670a | 4.010a | 0.633 | 0.0261 | |
| IL-12β | 3.569a | 2.989b | 3.663a | 3.872a | 0.173 | 0.0041 | |
| TNF-α | 22.089a | 20.344ab | 20.379ab | 18.589b | 0.606 | 0.0008 | |
| TGF-β | 11.370a | 11.611a | 10.444b | 10.842b | 0.128 | <0.0001 | |
| PBMC | IFN-α | 11.773b | 15.977ab | 13.088ab | 17.106a | 1.335 | 0.0157 |
| IFN-γ | 12.214b | 16.006ab | 16.326ab | 18.282a | 1.334 | 0.0113 | |
| IL-1α | 12.041b | 10.811b | 11.615b | 17.537a | 1.257 | 0.0005 | |
| IL-1β | 15.483 | 17.135 | 12.756 | 15.509 | 1.218 | 0.1104 | |
| IL-6 | 13.053b | 16.629ab | 13.560ab | 17.614a | 1.323 | 0.0340 | |
| IL-8 | 21.288 | 22.925 | 23.555 | 25.829 | 1.430 | 0.1378 | |
| IL-10 | 10.090b | 14.488a | 11.859ab | 13.808ab | 1.092 | 0.0208 | |
| IL-12α | 9.317b | 17.997a | 14.135ab | 17.319a | 1.436 | <0.0001 | |
| IL-12β | 14.784ab | 19.528a | 13.506b | 17.447ab | 1.482 | 0.0276 | |
| TNF-α | 10.269b | 18.246a | 11.860ab | 17.104a | 1.471 | 0.0002 | |
| TGF-β | 10.090b | 13.634ab | 10.227b | 15.354a | 1.050 | 0.0005 |
1Values are means and results are expressed in log 2 of normalized values.
a–cDifferent superscripts within a row indicate a significant difference (P < 0.05), x,ydifferent superscripts within a row indicate a tendency difference (P < 0.1).
Ileum
Gene expression of IFN-γ was increased in ileum on day 15 compared with day 0 (P < 0.001). IL-1α gene expression was increased at day 30 compared with day 0 (P < 0.001). Gene expression for IL-1β reduced significantly on day 30 compared with all other time points (P < 0.001). A decrease in IL-6 gene expression was observed in the ileum on days 30 and 45 compared with day 0 (P < 0.001). IL-8 gene expression in ileum decreased on day 30 compared with all other time points (P < 0.001). IL-10 gene expression in the ileum was reduced on days 30 and 45 compared with days 0 and 15 (P < 0.001). IL-12α gene expression showed a tendency for a reduction on day 30 compared with day 0 in the ileum (P = 0.069). In the ileum, TGF-β gene expression was reduced on days 30 and 45 compared with days 0 and 15 (P < 0.001; Table 6).
Colon
IFN-α gene expression on day 30 was reduced compared with days 0 and 45 (P = 0.005). Expression of IFN-γ was increased in colon on day 15 compared with day 0 (P = 0.002). IL-1α gene expression was increased at day 15 compared with all time points in colonic tissue, with day 30 being higher than days 0 and 45 (P < 0.001). Gene expression of IL-1α was reduced on day 45 compared with day 0. IL-8 gene expression in colon increased on day 15 compared with all other time points (P < 0.001). In colonic tissue, IL-1β gene expression increased on day 15 compared with day 0), and increased on day 30 compared with day 15 (P < 0.001). In the colon, the expression of IL-10 was increased on day 15 compared with all other time points (P < 0.001). A reduction in IL-12β gene expression in colon tissue on day 15 compared with all other time points (P = 0.004). TNF-α gene expression reduced on day 45 compared with day 0 (P = 0.008). In the colon, TGF-β gene expression was reduced on days 30 and 45 compared with days 0 and 15 (P < 0.001; Table 6).
PBMC
Gene expression of IFN-γ in PBMC was increased on day 45 compared with day 0 (P = 0.011). Expression of IFN-α was increased on day 45 compared with day 0 (P = 0.016). In PBMC, expression of IL-1α was increased at day 45 compared with all previous time points (P < 0.001). IL-6 gene expression was increased on day 45 compared with day 0 (P = 0.034). An increase was observed in IL-10 expression on day 15 compared with day 0 (P = 0.021). The expression of IL-12α was increased on day 15 compared with day 0 (P < 0.001), where IL-12β gene expression was reduced on day 30 compared with day 15 (P = 0.028). An increase was observed in expression of TNF-α on days 15 and day 45 compared with day 0 (P < 0.001). In PBMC, the expression of TGF-β increased on day 45 compared with day 0 (P < 0. 001; Table 6).
Cytokine correlations
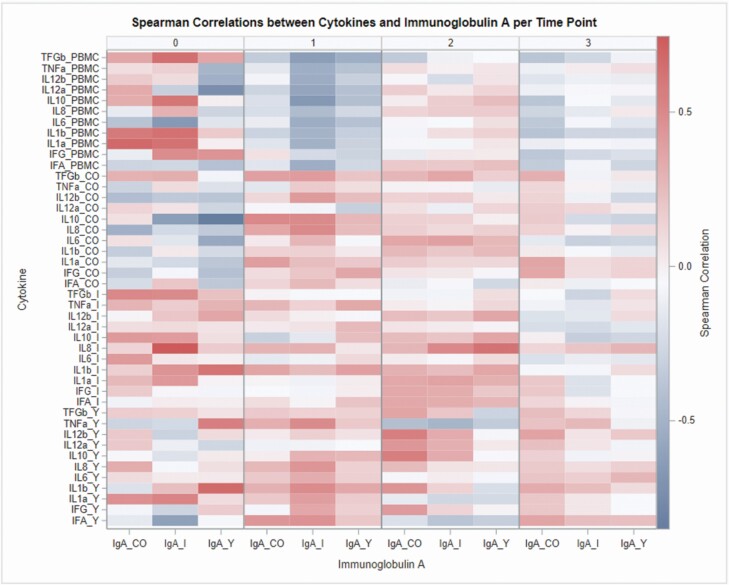
Spearman’s correlation analysis between cytokine gene expression and amount of IgA producing cells in the different tissues did not show strong existing correlations (r > 0.50). The strongest correlations were found on day 0 postweaning, where IL-1α and IL-1β gene expression in jejunum, ileum, and PBMC was correlated to IgA producing cells in intestinal tissue (Figure 1).
Figure 1.
Heatmap of Spearman correlation between IgA production and cytokine gene expression in intestinal tissues jejunum (Y), ileum (I), colon (CO), and in PBMC on different time points, where 0 = day 0, 1 = day 15, 2 = day 30 anday 3 = day 45. Cytokines refer to IL-1α, IL-1β, IL-6, IL-8, IL-10, IL-12α, IL-12β, TNF-α, IFN-α, IFN-γ, and TGF-β.
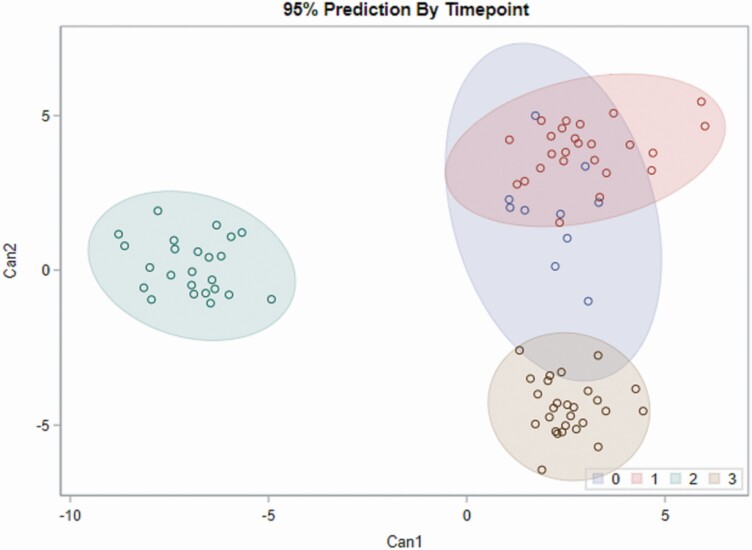
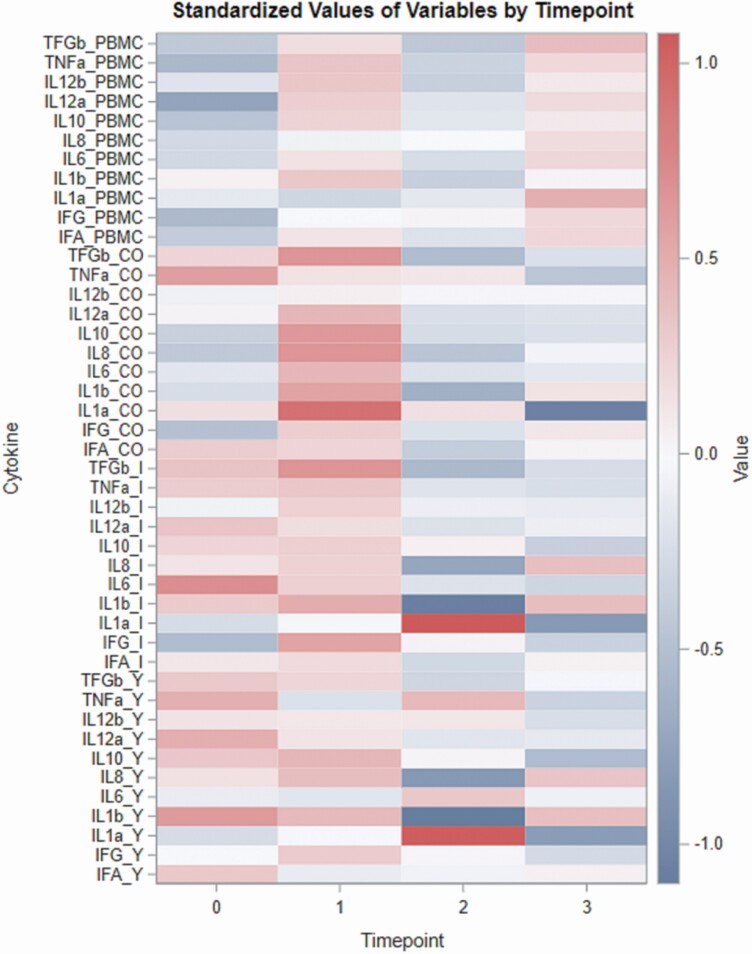
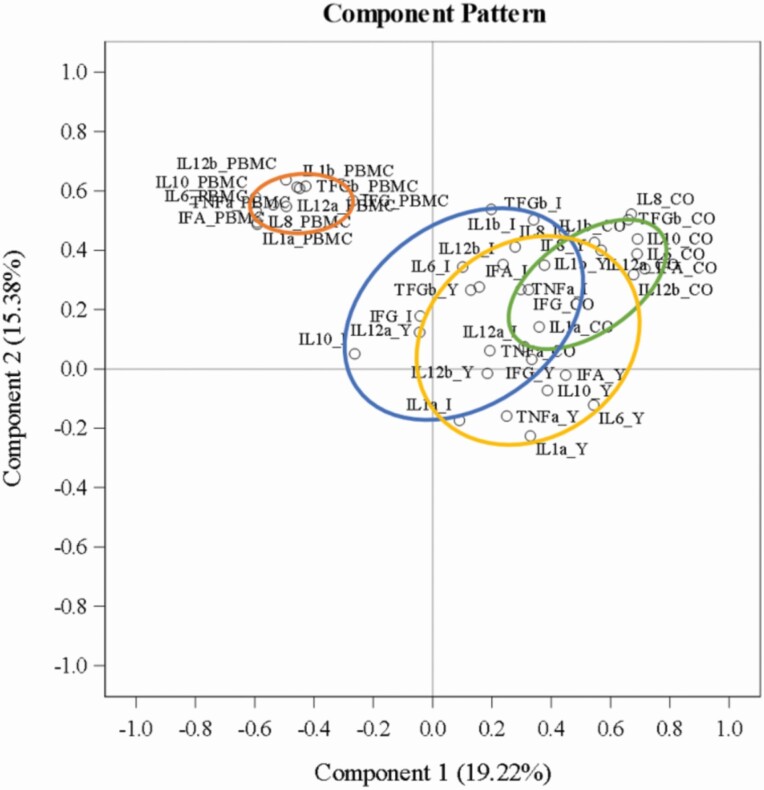
The CDA of different time points showed a separation of days 30 and 45 (Figure 2), with canonical 1 mainly explained by IL-1α in jejunum and ileum and canonical 2 by IL-1β and TGF-β in colon. Heatmap analysis underlines the difference in expression of all the tested cytokines observed in the jejunum and ileum tissue, with IL-1α being the main differentiator (Figure 3). PCA analysis of all cytokines in jejunum, ileum, colon, and PBMC showed a clear separation of cytokines in PBMC compared with the cytokines in the intestinal tissues, with the 2 components explaining 32% of the variation (Figure 4).
Figure 2.
CDA of cytokine (Ck) IL-1α, IL-1β, IL-6, IL-8, IL-10, IL-12α, IL-12β, TNF-α, IFN-α, IFN-γ, and TGF-β gene expression in intestinal tissues jejunum (Y), ileum (I), colon (CO), and in PBMC on different time points, where 0 = day 0, 1 = day 15, 2 = day 30 anday 3 = day 45. 95% prediction analysis per timepoint (A) and definition of canonical 1 and canonical 2 (B) as pooled within class standardized canonical coefficients.
Figure 3.
Heatmap of cytokine gene expression in intestinal tissues jejunum (Y), ileum (I), colon (CO), and in PBMC on different time points, where 0 = day 0, 1 = day 15, 2 = day 30 and day 3 = day 45.
Figure 4.
Principal component analysis of cytokine IL-1β, IL-6, IL-8, IL-10, IL-12α, IL-12β, TNF-α, IFN-α, IFN-γ, and TGF-β gene expression in intestinal tissues jejunum (yellow), ileum (blue), colon (green), and in PBMC (orange).
Discussion
Commercially, piglets are weaned at an age when their mucosal immune system is not fully mature, as evidenced by high morbidity and mortality resulting from postweaning diarrhea (McCracken et al., 1999; Lallès et al., 2004; Fairbrother et al., 2005). Of particular interest is the role of cytokines as major epithelial cell growth and development regulators, including intestinal inflammation and epithelial restitution following mucosal damage. Weaning is known to change the intestinal morphology, caused by the changes in feed intake pattern, for example changing from liquid to solid feed and a decrease in intake postweaning, and/or physiological or environmental stressors (Pluske et al., 1997). This study showed that the height of villi, the depth of the crypts. and the villus/crypt ratio differed significantly over time. The values of the morphometric measurements (villus height and crypt depth) were comparable to the data obtained by other authors, such as Manzanilla et al. (2006) with piglets 19 and 21 d postweaning and with Pluske et al. (1996) on 5 d postweaning. Also, the observed decrease in villus length on day of weaning followed by partial recovery on 15 d postweaning is in agreement with the results of others (van Beers-Schreurs et al., 1998; Marion et al., 2002; Vente-Spreeuwenberg et al., 2003). The animals in this study were not experiencing any clinical disease symptoms and had a growth performance during the study period normal for the genetic line. The lack of individual performance data such as daily gain and feed intake limits the possibility to correlate those parameters to intestinal immune markers such as cytokine gene expression and IgA producing cells.
Results demonstrated an increase in IgA producing cells in the GIT 30 d postweaning, confirming previous results showing an increase in IgA concentration in intestinal content 20 d postweaning (García et al., 2016). Furthermore, the results confirmed the existence of different distribution patterns of immune cells in the jejunum, ileum and colon, as previously reported (Allen and Porter, 1973, 1977; Pié et al., 2004; Bianco et al., 2014). Results indicated that the IgA production in piglets can be insufficient up to day 30 postweaning. In addition to the changes on an intestinal level with regards to IgA producing cells, IgA concentrations in plasma are increased over time, where the main increase occurred on days30 and 45 postweaning. Previous studies have shown a reduction in plasma IgA concentration directly after weaning, with the removal of IgA derived from the sow’s milk (Heo et al., 2013). This study could not confirm that, as the concentrations have only been measured postweaning with a 15-d interval, starting 18 hr postweaning. The current study shows a reduction in IgG concentration after weaning, in agreement with previous studies showing a significant decrease in piglet’s plasma IgG concentrations immediately after weaning, which is in concurrence with the depletion of maternal immunity (García et al., 2016; Moeser et al., 2017). A positive correlation between IgG in blood and PCV-2 titer existed. It is widely believed an effective vaccination of young piglets is challenged by a lower and less effective antibody responses in the presence of higher maternal antibodies (Salmon et al., 2009; Hodgins and Shewen, 2012). However, this study design is not adequate to elaborate more on this topic.
Production of mucosal IgA is controlled by cytokine-producing T-cells within the GALT and by cytokines released from the mucosa. Within the GALT, the Th1 cytokines, IFN-γ and TNF-β, downregulate IgA production, whereas the Th2 cytokines, IL-4, IL-5, IL-6, and IL-10, upregulate IgA production (Kramer et al., 1995; Ramsay, 1995). The current study demonstrated that mRNA expression of pro- and anti-inflammatory cytokines varied according to type tissue analyzed and a difference in expression of cytokines was seen during the life of the piglet, with increasing levels of pro-inflammatory cytokines postweaning. These results are confirming previous studies, where a different distribution pattern of cytokines was found in the small intestine, where bacterial colonization patterns in the gut can impact cytokine expression patterns in the intestine, and cytokines could be indicators of bacterial population dynamics (Shirkey et al., 2006; Zhao et al., 2008). No correlation has been found between IgA producing cells in the different intestinal tissues and the cytokine gene expression in the tissue and/or in PBMCs. A potential reason for the lack of correlation can be the large interval (15 d) in tissue sampling, therefor the correlation between the production of cytokines and responding increase in IgA producing cells could be missed.
PCA indicated a separation between cytokine gene expression in different GIT tissues and in PBMC, with a clustering of cytokine gene expression in the small intestine (jejunum and ileum). Furthermore, a separation existed between cytokines IL-1α and TGF-β vs. IL-1β and IL-8 within all tissue locations but mainly in jejunum and ileum. This study shows that early (days 0 and 15) upregulation is mainly seen in cytokines IFN-γ, IL-10, TGF-β, upregulation on day 30 is seen in IL-1α. A pronounced similar pattern in expression of IL-1β and IL-8 is observed, decreasing the expression on day 30 postweaning in jejunum and ileum, which is the opposite pattern for IL-1α, where an increase is occurring on day 30 in jejunum and ileum. This increased levels of anti-inflammatory cytokine expression, such as IL-10 and TGF-β, can indicate that on days 15 and 30 the negative feedback loop has been started, to possibly protect against intestinal inflammation by maintaining the barrier function (Madsen et al., 1997; Howe et al., 2005). The increase in cytokine gene expression postweaning results found in current study builds on existing evidence (Pié et al., 2004; Moeser et al., 2007b) that demonstrated the weaning process in piglets is associated with an inflammation in the intestine. According to these authors, an intestinal upregulation of IL-1β, IL-6, and TNF-α mRNA occurred during the first 2 d postweaning. After 2 d postweaning, the mRNA level of these cytokines returned to preweaning levels, with the exception of the TNF-α mRNA level in the distal intestine, which remained high. Hu et al. (2013) reported in 21 d weaned piglets alleviated levels of TNF-α and IL-6 at 3 and 7 d postweaning, and the levels returned to the preweaning levels at day 14 postweaning. In accordance with our results, McCracken et al., (1995) also found a transient increase in plasma IL-1 (unspecified) concentrations over the first 2 d postweaning period in piglets. However, these authors did not show any changes in plasma TNF-α and IL-6 concentrations. Additional to previous mentioned studies, the current study found changes in gene expression of cytokines and Igs on an intestinal level as well as in blood after 2 wk still to be present. This is also reflected by the canonical discrimination analyses that indicated a clear separation of days 30 and 45 postweaning compared with days 0 and day 15 postweaning. The sample interval of 15 d in the current study limits the possibility to conclude a short-term effect of the weaning process on the inflammation response. Furthermore, it limits the possibility of observing intestinal and immunological maturation to be causing the changes in Ig and cytokine expression over time.
The changes in cytokine expression in time in both intestinal tissue as in blood found in this study indicate that the pigs seem to recover from intestinal inflammation, observed by an increase in pro-inflammatory cytokines as previously discussed, followed by an increase anti-inflammatory cytokine expression as a negative feedback. However, at the first 15 d postweaning, the upregulation of pro-inflammatory cytokines, such as TNF-α, IFN-γ, and IL-6, may increase intestinal epithelial permeability in, favoring the trans-epithelial passage of micro-organisms and leading to bacterial infections and diarrhea (Al-Sadi and Ma, 2007, Al-Sadi et al., 2009). Many studies reported that enteric infection and intestinal inflammatory diseases were associated with the alternations in the expression of pro-inflammatory cytokines in the intestine of humans and animals (Murtaugh et al., 1996; McClane and Rombeau, 1999). An example is an upregulation of TNF-α, IL-6, IL-8, and IL-1β in jejunal mucosa of weaned piglets after an LPS challenge (Upadhaya et al., 2015; Xiao et al., 2016; Cao et al., 2018). Local expression of IL-1, IL-6, and TNF-α mRNA has been largely documented after bacterial or viral infection in pigs (Murtaugh et al., 1996; Fossum, 1998) and in humans during gut inflammatory diseases such as Crohn’s disease and ulcerative colitis (Mahida et al., 1989; Stevens et al., 1992).
The present study found an increase in gene expression of intestinal cytokines and IgA producing cells from day 15 postweaning onward. While there is a lot of data available on the short-term immunological response postweaning, these data can give new insights on the potential prolonged immune response postweaning and help to design strategies to control postweaning inflammatory responses. A lot of previous research has proven the potential negative effect of increased expression of inflammation markers on intestinal integrity (Spreeuwenberg et al., 2001; Moeser et al., 2017), morphology of intestinal structures such as villous length and crypt depth (McCracken et al., 1995; Pié et al., 2004) and disruption of the microbiota (Schachtschneider et al., 2013; Gresse et al., 2017), favoring a delay in intestinal maturation and a predisposal to diseases (Jayaraman and Nyachoti, 2017; Pluske et al., 2018b). Furthermore, studies have demonstrated that an induction of an immune response requires the mobilization of energy and resources from other activities (i.e., nutrition and growth), with estimations of a 20% to 25% increase in protein and amino acid usage (Kurpad, 2006). Recent research by Pluske et al. (2018a) has shown the potential of different management measures such as reducing physical and physiological stressors, implementing nutritional strategies reducing intestinal microbial load, increasing digestion and preventing production and activity of pro-inflammatory cytokines, all with the main goal to manipulate the immune system of pigs for improving performance, aiming to have an appropriate immune response for each specific circumstance, preventing to maximize the immune response (Pluske et al., 2018a). Results from this study could have implications for nutritional recommendations for nursery pigs, especially in the current era with recommendations for lower use of antimicrobial ingredients such as preventive antibiotics and pharmacological levels of zinc oxide.
Conclusion
Results have shown that the process of weaning in piglets was associated with a prolonged and transient response in gene expression of intestinal inflammatory cytokines and an increased amount of IgA producing cells in the intestinal tissue. These increased responses were still present at days 15 and 30 postweaning.
Glossary
Abbreviations
- BW
body weight
- CDA
canonical discriminant analysis
- GALT
gut associated lymphoid tissue
- GIT
gastrointestinal tract
- Ig
immunoglobulin
- IV
intravenous
- PBMC
peripheral blood mononuclear cell
- PCA
principal component analysis
- PCR
polymerase chain reaction
- PCV-2
porcine circovirus-2
- TBS
tris-buffered saline
Conflict of Interest Statement
The authors declare no real or perceived conflicts of interest.
Literature Cited
- Allen, W. D., and Porter P.. . 1973. The relative distribution of IgM and IgA cells in intestinal mucosa and lymphoid tissues of the young unweaned pig and their significance in ontogenesis of secretory immunity. Immunology 24:493– 501. [PMC free article] [PubMed] [Google Scholar]
- Allen, W. D., and Porter P.. . 1977. The relative frequencies and distribution of immunoglobulin-bearing cells in the intestinal mucosa of neonatal and weaned pigs and their significance in the development of secretory immunity. Immunology 32:819–824. [PMC free article] [PubMed] [Google Scholar]
- Al-Sadi, R. M., and Ma, T. Y. (2007). IL-1β Causes an Increase in Intestinal Epithelial Tight Junction Permeability. The Journal of Immunology, 178(7), 4641–4649. doi: 10.4049/jimmunol.178.7.4641 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Sadi, R., Boivin M., and Ma T.. . 2009. Mechanism of cytokine modulation of epithelial tight junction barrier. Front. Biosci. (Landmark Ed). 14:2765–2778. doi: 10.2741/3413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Beers-Schreurs, H. M. G., Nabuurs, M. J. A., Vellenga, L., Valk, H. J. K. der, Wensing, T., and Breukink, H. J. (1998). Weaning and the weanling diet influence the villous height and crypt depth in the small intestine of pigs and alter the concentrations of short-chain fatty acids in the large intestine and blood. J. Nutr., 128(6), 947–953. doi: 10.1093/jn/128.6.947 [DOI] [PubMed] [Google Scholar]
- Bianco, C., Felice V., Panarese S., Marrocco R., Ostanello F., Brunetti B., Muscatello L. V., Leotti G., Vila T., Joisel F., . et al. 2014. Quantitative immunohistochemical assessment of IgA, IgM, IgG and antigen-specific immunoglobulin secreting plasma cells in pig small intestinal lamina propria. Vet. Immunol. Immunopathol. 160:281–287. doi: 10.1016/j.vetimm.2014.05.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borca, M. V., Gudmundsdottir I., Fernández-Sainz I. J., Holinka L. G., and Risatti G. R.. . 2008. Patterns of cellular gene expression in swine macrophages infected with highly virulent classical swine fever virus strain Brescia. Virus Res. 138:89–96. doi: 10.1016/j.virusres.2008.08.009 [DOI] [PubMed] [Google Scholar]
- Bracarense, A. P., Lucioli J., Grenier B., Drociunas Pacheco G., Moll W. D., Schatzmayr G., and Oswald I. P.. . 2012. Chronic ingestion of deoxynivalenol and fumonisin, alone or in interaction, induces morphological and immunological changes in the intestine of piglets. Br. J. Nutr. 107:1776–1786. doi: 10.1017/S0007114511004946 [DOI] [PubMed] [Google Scholar]
- Burkey, T. E., Skjolaas K. A., and Minton J. E.. . 2009. Board-invited review: porcine mucosal immunity of the gastrointestinal tract. J. Anim. Sci. 87:1493–1501. doi: 10.2527/jas.2008-1330 [DOI] [PubMed] [Google Scholar]
- Cao, S., Zhang Q., Wang C., Wu H., Jiao L., Hong Q., and Hu C.. . 2018. LPS challenge increased intestinal permeability, disrupted mitochondrial function and triggered mitophagy of piglets. Innate Immun. 24:221–230. doi: 10.1177/1753425918769372 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerutti, A., and Rescigno M.. . 2008. The biology of intestinal immunoglobulin A responses. Immunity 28:740–750. doi: 10.1016/j.immuni.2008.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Council of the EU. (2016). Council conclusions on the next steps under a One Health approach to combat antimicrobial resistance. Rome (Italy): Official Journal of the European Union; p.26–30. [Google Scholar]
- Duvigneau, J. C., Hartl R. T., Groiss S., and Gemeiner M.. . 2005. Quantitative simultaneous multiplex real-time PCR for the detection of porcine cytokines. J. Immunol. Methods 306:16–27. doi: 10.1016/j.jim.2005.06.021 [DOI] [PubMed] [Google Scholar]
- Eckmann, L., Kagnoff M. F., and Fierer J.. . 1995. Intestinal epithelial cells as watchdogs for the natural immune system. Trends Microbiol. 3:118–120. doi: 10.1016/s0966-842x(00)88894-0 [DOI] [PubMed] [Google Scholar]
- Fairbrother, J. M., Nadeau E., and Gyles C. L.. . 2005. Escherichia coli in postweaning diarrhea in pigs: an update on bacterial types, pathogenesis, and prevention strategies. Anim. Health Res. Rev. 6:17–39. doi: 10.1079/ahr2005105 [DOI] [PubMed] [Google Scholar]
- Fossum, C. 1998. Cytokines as markers for infections and their effect on growth performance and well-being in the pig. Domest. Anim. Endocrinol. 15:439–444. doi: 10.1016/s0739-7240(98)80001-5 [DOI] [PubMed] [Google Scholar]
- Gabler, N. K., Spencer J. D., Webel D. M., and Spurlock M. E.. . 2008. n-3 PUFA attenuate lipopolysaccharide-induced down-regulation of toll-like receptor 4 expression in porcine adipose tissue but does not alter the expression of other immune modulators. J. Nutr. Biochem. 19:8–15. doi: 10.1016/j.jnutbio.2006.11.014 [DOI] [PubMed] [Google Scholar]
- García, G. R., Dogi C. A., Ashworth G. E., Berardo D., Godoy G., Cavaglieri L. R., de Moreno de LeBlanc A., and Greco C. R.. . 2016. Effect of breast feeding time on physiological, immunological and microbial parameters of weaned piglets in an intensive breeding farm. Vet. Immunol. Immunopathol. 176:44–49. doi: 10.1016/j.vetimm.2016.02.009 [DOI] [PubMed] [Google Scholar]
- Gonnella, P. A., Chen Y., Inobe J., Komagata Y., Quartulli M., and Weiner H. L.. . 1998. In situ immune response in gut-associated lymphoid tissue (GALT) following oral antigen in TCR-transgenic mice. J. Immunol. 160:4708–4718. [PubMed] [Google Scholar]
- Gresse, R., Chaucheyras-Durand F., Fleury M. A., Van de Wiele T., Forano E., and Blanquet-Diot S.. . 2017. Gut Microbiota Dysbiosis in Postweaning Piglets: understanding the Keys to Health. Trends Microbiol. 25:851–873. doi: 10.1016/j.tim.2017.05.004 [DOI] [PubMed] [Google Scholar]
- Gutzeit, C., Magri G., and Cerutti A.. . 2014. Intestinal IgA production and its role in host-microbe interaction. Immunol. Rev. 260:76–85. doi: 10.1111/imr.12189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heo, J. M., Opapeju F. O., Pluske J. R., Kim J. C., Hampson D. J., and Nyachoti C. M.. . 2013. Gastrointestinal health and function in weaned pigs: a review of feeding strategies to control post-weaning diarrhoea without using in-feed antimicrobial compounds. J. Anim. Physiol. Anim. Nutr. (Berl). 97:207–237. doi: 10.1111/j.1439-0396.2012.01284.x [DOI] [PubMed] [Google Scholar]
- Hodgins, D. C., and Shewen P. E.. . 2012. Vaccination of neonates: problem and issues. Vaccine 30:1541–1559. doi: 10.1016/j.vaccine.2011.12.047 [DOI] [PubMed] [Google Scholar]
- Howe, K. L., Reardon, C., Wang, A., Nazli, A., and McKay, D. M. (2005). Transforming growth factor-β regulation of epithelial tight junction proteins enhances barrier function and blocks enterohemorrhagic Escherichia coli O157:H7-induced increased permeability. Am. J. Pathol., 167(6), 1587–1597. doi: 10.1016/S0002-9440(10)61243-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu, C. H., Xiao K., Luan Z. S., and Song J.. . 2013. Early weaning increases intestinal permeability, alters expression of cytokine and tight junction proteins, and activates mitogen-activated protein kinases in pigs. J. Anim. Sci. 91:1094–1101. doi: 10.2527/jas.2012-5796 [DOI] [PubMed] [Google Scholar]
- Jayaraman, B., and Nyachoti, C. M. (2017). Husbandry practices and gut health outcomes in weaned piglets: a review. Animal Nutrition, 3(3), 205–211. doi: 10.1016/j.aninu.2017.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim, C. J., Kovacs-Nolan J. A., Yang C., Archbold T., Fan M. Z., and Mine Y.. . 2010. l-Tryptophan exhibits therapeutic function in a porcine model of dextran sodium sulfate (DSS)-induced colitis. J. Nutr. Biochem. 21:468–475. doi: 10.1016/j.jnutbio.2009.01.019 [DOI] [PubMed] [Google Scholar]
- Kramer, D. R., Sutherland R. M., Bao S., and Husband A. J.. . 1995. Cytokine mediated effects in mucosal immunity. Immunol. Cell Biol. 73:389–396. doi: 10.1038/icb.1995.61 [DOI] [PubMed] [Google Scholar]
- Kurpad, A. V. 2006. The requirements of protein and amino acid during acute and chronic infections. Indian J. Med. Res. 124:129– 148. [PubMed] [Google Scholar]
- Lallès, J.-P., Boudry, G., Favier, C., Le Floc’h, N., Luron, I., Montagne, L., Oswald, I. P., Pié, S., Piel, C., and Sève, B. (2004). Gut function and dysfunction in young pigs: physiology. Anim. Res., 53(4), 301–316. doi: 10.1051/animres:2004018 [DOI] [Google Scholar]
- Madsen, K. L., Lewis S. A., Tavernini M. M., Hibbard J., and Fedorak R. N.. . 1997. Interleukin 10 prevents cytokine-induced disruption of T84 monolayer barrier integrity and limits chloride secretion. Gastroenterology 113:151–159. doi: 10.1016/s0016-5085(97)70090-8 [DOI] [PubMed] [Google Scholar]
- Mahida, Y. R., Wu, K., and Jewell, D. P. (1989). Enhanced production of interleukin 1-β by mononuclear cells isolated from mucosa with active ulcerative colitis of Crohn’s disease. Gut, 30(6), 835–838. doi: 10.1136/gut.30.6.835 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manzanilla, E. G., Nofrarías M., Anguita M., Castillo M., Perez J. F., Martín-Orúe S. M., Kamel C., and Gasa J.. . 2006. Effects of butyrate, avilamycin, and a plant extract combination on the intestinal equilibrium of early-weaned pigs. J. Anim. Sci. 84:2743–2751. doi: 10.2527/jas.2005-509 [DOI] [PubMed] [Google Scholar]
- Marion, J., Biernat, M., Thomas, F., Savary, G., Le Breton, Y., Zabielski, R., Le Huërou-Luron, I., and Le Dividich, J. (2002). Small intestine growth and morphometry in piglets weaned at 7 days of age effects of level of energy intake. Reprod. Nutr. Dev., 42(4), 339–354. doi: 10.1051/rnd:2002030 [DOI] [PubMed] [Google Scholar]
- McClane, S. J., and Rombeau J. L.. . 1999. Cytokines and inflammatory bowel disease: a review. J. Parenter. Enteral Nutr. 23(5 Suppl.):S20–S24. doi: 10.1177/014860719902300506 [DOI] [PubMed] [Google Scholar]
- McCracken, B. A., Gaskins H. R., Ruwe-Kaiser P. J., Klasing K. C., and Jewell D. E.. . 1995. Diet-dependent and diet-independent metabolic responses underlie growth stasis of pigs at weaning. J. Nutr. 125:2838–2845. doi: 10.1093/jn/125.11.2838 [DOI] [PubMed] [Google Scholar]
- McCracken, B. A., Spurlock M. E., Roos M. A., Zuckermann F. A., and Gaskins H. R.. . 1999. Weaning anorexia may contribute to local inflammation in the piglet small intestine. J. Nutr. 129:613–619. doi: 10.1093/jn/129.3.613 [DOI] [PubMed] [Google Scholar]
- McKay, D. M., and Baird A. W.. . 1999. Cytokine regulation of epithelial permeability and ion transport. Gut 44:283–289. doi: 10.1136/gut.44.2.283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLamb, B. L., Gibson A. J., Overman E. L., Stahl C., and Moeser A. J.. . 2013. Early weaning stress in pigs impairs innate mucosal immune responses to enterotoxigenic E. coli challenge and exacerbates intestinal injury and clinical disease. PLoS One 8:e59838. doi: 10.1371/journal.pone.0059838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moeser, A. J., Pohl C. S., and Rajput M.. . 2017. Weaning stress and gastrointestinal barrier development: implications for lifelong gut health in pigs. Anim. Nutr. 3:313–321. doi: 10.1016/j.aninu.2017.06.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moeser, A. J., Klok C. V., Ryan K. A., Wooten J. G., Little D., Cook V. L., and Blikslager A. T.. . 2007a. Stress signaling pathways activated by weaning mediate intestinal dysfunction in the pig. Am. J. Physiol. Gastrointest. Liver Physiol. 292:G173–G181. doi: 10.1152/ajpgi.00197.2006 [DOI] [PubMed] [Google Scholar]
- Moeser, A. J., Ryan K. A., Nighot P. K., and Blikslager A. T.. . 2007b. Gastrointestinal dysfunction induced by early weaning is attenuated by delayed weaning and mast cell blockade in pigs. Am. J. Physiol. Gastrointest. Liver Physiol. 293:G413–G421. doi: 10.1152/ajpgi.00304.2006 [DOI] [PubMed] [Google Scholar]
- Moue, M., Tohno M., Shimazu T., Kido T., Aso H., Saito T., and Kitazawa H.. . 2008. Toll-like receptor 4 and cytokine expression involved in functional immune response in an originally established porcine intestinal epitheliocyte cell line. Biochim. Biophys. Acta 1780:134–144. doi: 10.1016/j.bbagen.2007.11.006 [DOI] [PubMed] [Google Scholar]
- Murtaugh, M. P., Baarsch, M. J., Zhou, Y., Scamurra, R. W., and Lin, G. (1996). Inflammatory cytokines in animal health and disease. Vet. Immunol. Immunopathol., 54(1–4), 45–55. doi: 10.1016/S0165-2427(96)05698-X [DOI] [PubMed] [Google Scholar]
- Niewold, T. A. (2015). Intestinal Health: key to maximise growth performance in livestock. Wageningen Academic Publishers; p. 1–276. doi: 10.3920/978-90-8686-792-9 [DOI] [Google Scholar]
- National Research Council. 2012. Nutrient requirements of swine, 10th rev. ed. Washington (DC): National Academic Press. [Google Scholar]
- Oswald, I. P., Dozois C. M., Barlagne R., Fournout S., Johansen M. V., and Bøgh H. O.. . 2001. Cytokine mRNA expression in pigs infected with Schistosoma japonicum. Parasitology 122(Pt 3):299–307. doi: 10.1017/s0031182001007399 [DOI] [PubMed] [Google Scholar]
- Pfaffl, M. W. 2001. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29:e45. doi: 10.1093/nar/29.9.e45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pié, S., Lallès, J. P., Blazy, F., Laffitte, J., Sève, B., and Oswald, I. P. (2004). Weaning is associated with an upregulation of expression of inflammatory cytokines in the intestine of piglets. J. Nutr., 134(3), 641–647. doi: 10.1093/jn/134.3.641 [DOI] [PubMed] [Google Scholar]
- Pitman, R. S., and Blumberg R. S.. . 2000. First line of defense: the role of the intestinal epithelium as an active component of the mucosal immune system. J. Gastroenterol. 35:805–814. doi: 10.1007/s005350070017 [DOI] [PubMed] [Google Scholar]
- Pluske, J. R., Hampson, D. J., and Williams, I. H.(1997). Factors influencing the structure and function of the small intestine in the weaned pig: a review. Livest. Prod. Sci., 51(1–3), 215–236. doi: 10.1016/S0301-6226(97)00057-2 [DOI] [Google Scholar]
- Pluske, J. R., Kim, J. C., and Black, J. L. (2018a). Manipulating the immune system for pigs to optimise performance. Anim. Prod. Sci., 58(4), 666–680. doi: 10.1071/AN17598 [DOI] [Google Scholar]
- Pluske, J. R., Turpin D. L., and Kim J. C.. (2018b). Gastrointestinal tract (gut) health in the young pig. Anim. Nutr. 4:187–196. doi: 10.1016/j.aninu.2017.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pluske, J. R., Williams, I. H., and Aherne, F. X. (1996). Villous height and crypt depth in piglets in response to increases in the intake of cows’ milk after weaning. Anim. Sci., 62(1), 145–158. doi: 10.1017/S1357729800014429 [DOI] [Google Scholar]
- Ramsay, A. J. 1995. Genetic approaches to the study of cytokine regulation of mucosal immunity. Immunol. Cell Biol. 73:484–488. doi: 10.1038/icb.1995.78 [DOI] [PubMed] [Google Scholar]
- Royaee, A. R., Husmann R. J., Dawson H. D., Calzada-Nova G., Schnitzlein W. M., Zuckermann F. A., and Lunney J. K.. . 2004. Deciphering the involvement of innate immune factors in the development of the host response to PRRSV vaccination. Vet. Immunol. Immunopathol. 102:199–216. doi: 10.1016/j.vetimm.2004.09.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salmon, H., Berri M., Gerdts V., and Meurens F.. . 2009. Humoral and cellular factors of maternal immunity in swine. Dev. Comp. Immunol. 33:384–393. doi: 10.1016/j.dci.2008.07.007 [DOI] [PubMed] [Google Scholar]
- Schachtschneider, K. M., Yeoman C. J., Isaacson R. E., White B. A., Schook L. B., and Pieters M.. . 2013. Modulation of systemic immune responses through commensal gastrointestinal microbiota. PLoS One 8:e53969. doi: 10.1371/journal.pone.0053969 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirkey, T. W., Siggers R. H., Goldade B. G., Marshall J. K., Drew M. D., Laarveld B., and Van Kessel A. G.. . 2006. Effects of commensal bacteria on intestinal morphology and expression of proinflammatory cytokines in the gnotobiotic pig. Exp. Biol. Med. (Maywood). 231:1333–1345. doi: 10.1177/153537020623100807 [DOI] [PubMed] [Google Scholar]
- Skovgaard, K., Mortensen S., Boye M., Poulsen K. T., Campbell F. M., Eckersall P. D., and Heegaard P. M.. . 2009. Rapid and widely disseminated acute phase protein response after experimental bacterial infection of pigs. Vet. Res. 40:23. doi: 10.1051/vetres/2009006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spreeuwenberg, M. A. M., Verdonk, J. M. A. J., Gaskins, H. R., and Verstegen, M. W. A. (2001). Small intestine epithelial barrier function is compromised in pigs with low feed intake at weaning. J. Nutr., 131(5), 1520–1527. doi: 10.1093/jn/131.5.1520 [DOI] [PubMed] [Google Scholar]
- Stevens, C., Walz G., Singaram C., Lipman M. L., Zanker B., Muggia A., Antonioli D., Peppercorn M. A., and Strom T. B.. . 1992. Tumor necrosis factor-alpha, interleukin-1 beta, and interleukin-6 expression in inflammatory bowel disease. Dig. Dis. Sci. 37:818–826. doi: 10.1007/BF01300378 [DOI] [PubMed] [Google Scholar]
- Stokes, C. R. 2017. The development and role of microbial-host interactions in gut mucosal immune development. J. Anim. Sci. Biotechnol. 8:12. doi: 10.1186/s40104-016-0138-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swildens, B., Stockhofe-Zurwieden N., van der Meulen J., Wisselink H. J., Nielen M., and Niewold T. A.. . 2004. Intestinal translocation of Streptococcus suis type 2 EF+ in pigs. Vet. Microbiol. 103:29–33. doi: 10.1016/j.vetmic.2004.06.010 [DOI] [PubMed] [Google Scholar]
- Upadhaya, S. D., Kim J. C., Mullan B. P., Pluske J. R., and Kim I. H.. . 2015. Vitamin E and omega-3 fatty acids independently attenuate plasma concentrations of proinflammatory cytokines and prostaglandin E3 in Escherichia coli lipopolysaccharide-challenged growing-finishing pigs. J. Anim. Sci. 93:2926–2934. doi: 10.2527/jas.2014-8330 [DOI] [PubMed] [Google Scholar]
- Vente-Spreeuwenberg, M. A. M., Verdonk, J. M. A. J., Verstegen, M. W. A., and Beynen, A. C. (2003). Villus height and gut development in weaned piglets receiving diets containing either glucose, lactose or starch. Br. J. Nutr., 90(5), 907–913. doi: 10.1079/bjn2003981 [DOI] [PubMed] [Google Scholar]
- Verpoest, S., Cay, B., Favoreel, H., and De Regge, N. (2017). Age-dependent differences in pseudorabies virus neuropathogenesis and associated cytokine expression. J. Virol., 91(2), 1–17. doi: 10.1128/jvi.02058-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao, K., Cao S. T., Jiao l. e. F., Lin F. H., Wang L., and Hu C. H.. . 2016. Anemonin improves intestinal barrier restoration and influences TGF-β1 and EGFR signaling pathways in LPS-challenged piglets. Innate Immun. 22:344–352. doi: 10.1177/1753425916648223 [DOI] [PubMed] [Google Scholar]
- Zhao, J., Harper, A. F., Webb, K. E., Kuehn, L. A., Gilbert, E., Xiao, X., and Wong, E. A. (2008). Cytokine mRNA expression in the small intestine of weanling pigs fed diets supplemented with specialized protein or peptide sources. Asian-Austral. J. Anim. Sci., 21(12), 1800–1806. doi: 10.5713/ajas.2008.80134 [DOI] [Google Scholar]