Abstract
Immune checkpoint inhibitors are now widely used in the treatment of multiple cancers. The major toxicities of these treatments are termed immune-related adverse events and endocrine dysfunction is common. Thyroid disease, hypopituitarism and a form of diabetes resembling type 1 diabetes are now all well described, with different patterns emerging with different checkpoint inhibitors. We review the presentation and management of the common endocrine immune-related adverse events, and discuss a number of recent advances in the understanding of these important, potentially life threatening toxicities. We also discuss some remaining dilemmas in management.
Keywords: checkpoint inhibitor, cancer, thyroiditis, hypophysitis, diabetes
Introduction
Immunotherapy with immune checkpoint inhibitors (CPI’s) has been revolutionising the management of advanced malignancies with their success in improving overall patient survival (1, 2). CPI’s are antibodies that block T-cell signalling pathways, that otherwise suppress immune responses to cancer cells, thereby acting to promote an anti-tumour immune response. Current agents are monoclonal antibodies targeting either cytotoxic T lymphocyte antigen 4 (anti-CTLA4), programmed cell death-1 (anti-PD-1) or its ligand PD-L1 (anti-PD-L1) to potentiate anti-tumour immune responses. Such is their success, they have now become first-line therapy for metastatic melanoma, non-small cell lung cancer and renal cell carcinoma (3). They are now licensed for use in multiple other cancers, including Head and Neck cancers.
Unfortunately superior clinical response is often associated with treatment toxicity termed immune-related adverse effects (IRAEs) and can involve multiple organs including skin, lung, liver and bowel (4). Whilst the exact mechanism of these toxicities is not yet fully understood, it is thought to involve a reduction in tolerance, through inhibition of the immune checkpoints, resulting in autoimmunity towards normal tissues (Fig. 1). Side effects can range from mild to life threatening. The latter can include severe colitis, pneumonitis and cardiomyopathies (5). Whilst in mild cases treatment can often continue, the mainstay of moderate and severe IRAE’s involves cessation of CPI therapy and immunosuppression with glucocorticoids as first-line agents (5).
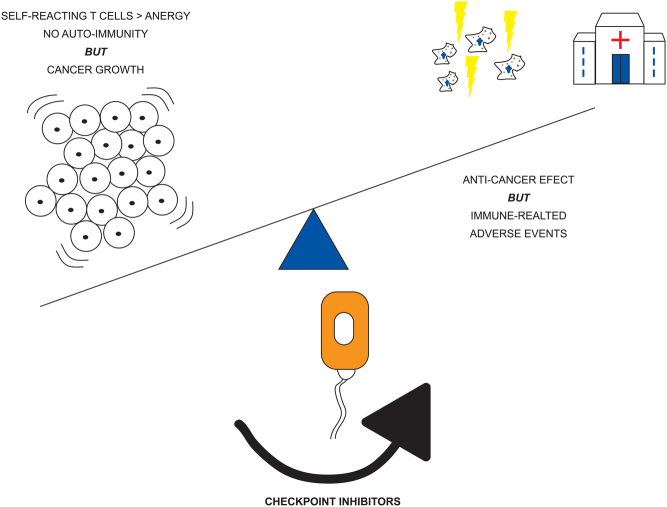
Figure 1.
Graphic illustrating checkpoint inhibitor action to shift immune response. Without treatment self and tumour reacting T-cells are driven to anergy and unchecked tumour growth. Following treatment, the anti-cancer immune response is upregulated leading to controlled tumour growth, but increased risk of self-reacting T cells contributing to immune-related adverse events.
Adverse effects have been reported more frequently with CTLA-4 inhibitors (e.g. ipilimumab) vs PD-1 inhibitors (e.g. pembrolizumab, nivolumab). Whilst it has been shown that combination therapy with both agents show superior response rates, but also carries a greater risk of such IRAE’s (6).
Endocrinopathy has emerged as an important group of IRAEs. Importantly, whilst most IRAE’s are reversible with prompt treatment cessation and glucocorticoids treatment, endocrinopathies usually persist and often require lifelong hormonal replacement. Untreated endocrinopathy can be life threatening. Whilst both CTLA-4 and PD-1 inhibitors are associated most commonly with thyroid dysfunction, hypophysitis resulting in adrenal insufficiency, and insulin-dependent diabetes also occur (7). Detection of endocrinopathy can be difficult given their subtle, non-specific symptoms such as fatigue and may be a challenge to distinguish them from pre-existing comorbidities. Therefore clinicians need to be vigilant to ensure rapid recognition and treatment, especially in those individuals with increased risk of autoimmune reactions. The major endocrine toxicities from checkpoint inhibitors and their management are summarised in Table 1.
Table 1.
Summary of checkpoint inhibitor-induced endocrinopathy.
| Endocrinopathy | Incidence + timing of onset | Precipitating therapies | Symptoms | Investigations | Indications for treatment | Treatment |
|---|---|---|---|---|---|---|
| Pituitary | 11-24% (8, 9, 81) (with anti-CTLA4) | Predominantly anti-CTLA4 (8, 9, 81) | Headache, fatigue, weakness nausea, weight loss, temperature intolerance, arthralgia | Pituitary hormone profile, sodium level, visual fields | Severe headache, hyponatraemia, hypopituitarism or pituitary enlargement seen on MRI (18, 19, 20, 21, 22) | High dose glucocorticoids if pituitary enlargement (24, 25) |
| <1% (8, 9, 44, 49) (with anti-PD-1) | Rarely seen with anti-PD-1 – mostly cause ACTH deficiency (8, 9, 44, 49) | Pituitary MRI | Hormone replacement (24, 25) (hydrocortisone or prednisolone ± levothyroxine/oestrogen/testosterone) | |||
| Usually 6–14 weeks after treatment | ||||||
| Thyroid dysfunction | 5.2–8% overall (8, 9, 49, 50) | More commonly seen in anti-PD-1 or anti-PD-L1 therapies (49, 50) | May present with hyperthyroidism symptoms prior to becoming hypothyroid (lethargy, constipation, cold intolerance, etc.) | Thyroid hormone profile | Hypothyroidism | Thyroid hormone replacement |
| 5.2–5.9% (8, 9) (with anti-CTLA4) | Thyrotoxicosis | Consider beta blockers for palpitations (23) | ||||
| 5–8% (49) (with anti-PD-1) | Seen less commonly in anti-CTLA-4 therapy (8, 9) | Anti-thyroid drugs only in Graves’ disease (51, 52, 53, 54, 55) | ||||
| 14–20% (combined therapy) (49, 50) | High dose steroids if severe thyroiditis seen (49) | |||||
| Usually 1–3 months after treatment | ||||||
| Hyperglycaemia and new-onset diabetes | Approximately 1% (59) | Anti-PD-1/PD-L1 (58, 61, 62) | Polydipsia, polyuria, weight loss | Hyperglycaemia on fasting and random glucose samples, HbA1c | Persistent hyperglycaemia, diabetic ketoacidosis | Exogenous insulin replacement (usually lifelong) (62) |
| Presents usually within 3 months of therapy | Not reported with anti-CTLA therapies (58) | |||||
| Primary Adrenal insufficiency | Very rare, only a few case reports published (16, 23, 36, 40) | Anti-CTLA-4 therapy or Anti PD-1 (16, 40) | Typical Addisonian symptoms | Cortisol, ACTH, plasma renin | Treatment indicated in all cases | Hydrocortisone + Fludrocortisone (23, 27) |
This review will focus on recent advances in our knowledge of endocrinopathy following checkpoint inhibitor therapy. Current areas of clinical uncertainty, ongoing controversies and changes in the management approaches will be discussed.
Pituitary
Two distinct patterns of pituitary involvement have been described. Patients treated with the CTLA-4 inhibitor Ipilimumab can develop hypophysitis in around 10% of cases, which can present with the classical features of headache, fatigue and pituitary enlargement seen on brain imaging (8, 9, 10, 11, 12). Pituitary dysfunction is variable but ACTH deficiency seems essentially universal (13, 14). In contrast, the PD-1 and PD-L1 inhibitors result less commonly in pituitary involvement (~1%), and are reported to result in isolated ACTH deficiency, without other features of hypophysitis (15, 16). However the literature can be confusing, as hypopituitarism and hypophysitis are frequently used interchangeably (17) and there is a need for standardised case definitions (16).
Role for high dose steroids and management of hypopituitarism
By extrapolation from other inflammatory pituitary conditions, initially patients presenting with ipilimumab-induced hypophysitis were initially treated with high dose glucocorticoids ( typically 1mg/kg of methylprednisolone). In addition, early case reports frequently described pituitary enlargement on MRI associated with headache (18, 19, 20, 21, 22), so glucocorticoids may have been indicated to prevent further pituitary enlargement. When patients present with signs or symptoms of adrenal insufficiency, stress doses of glucocorticoids are also indicated (23).
However, there is now evidence that treatment with high dose glucocorticoids does not improve pituitary function (24). More concerningly, melanoma patients with ipilimumab-induced hypophysitis treated with high dose glucocorticoids had worse oncological outcomes than those receiving replacement doses. There was a shorter time to treatment failure and overall survival in those receiving high dose steroids (25). This is notable as studies in patients with non-endocrine immune-related toxicity have not shown an association with the use of high dose steroids and oncological outcomes (26).
Therefore current practice is to reserve high dose corticosteroids for those presenting with adrenal crisis or with significant pituitary enlargement. Other patients can be commenced directly on replacement doses of corticosteroids (e.g. hydrocortisone 20 mg daily in divided doses or prednisolone 3 mg once daily (27)). Indeed there are now reports of the safety of managing such patients on an out-patient basis (28).
Thyroxine is replaced by standard approaches, following glucocorticoids, and testosterone or oestrogen replacement may also be required. Notably growth hormone replacement is contra-indicated in those with active or recent history of cancer (29), so assessment of growth hormone levels is not recommended.
Prediction of hypophysitis
Given the potentially severe impact of hypopituitarism and adrenal insufficiency, early detection of hypophysitis is vital. There has therefore been interest in biomarkers to predict its onset. The summary of product characteristics for checkpoint inhibitor recommend regular testing of thyroid function during treatment. Attempting to predict the onset of hypophysitis with subtle changes to thyroid function tests has been examined. Two studies have described a fall in TSH in the cycle of treatment prior to the onset of ipilimumab-induced hypophysitis (9, 30), although a larger series showed that a fall in free T4 at cycle 3 of ipilimumab had the best predictive value for subsequent hypophysitis (13). The later study used a clinical definition of hypophysitis that required at least one other feature apart from secondary hypothyroidism, confirming that a fall in T4 levels may precede hypophysitis. However, as pituitary involvement in PD-1/PD-L1 inhibitor therapy is usually limited to ACTH deficiency, thyroid function would not be expected to be a useful predictor in this cohort.
The development of autoantibodies has been described in a number of IRAEs, including diabetes and hypophysitis (31, 32, 33). Two studies have now reported the development of auto-antibodies in hypophysitis (34, 35), although notably there was no common antibodies described, and both studies included patients with both ipilimumab hypophysitis and the more limited ACTH deficiency with nivolumab or pembrolizumab. A further study suggested that hypophysitis was more common in certain HLA types, most notably DR15 (36), although again the hypophysitis cohort was heterogeneous.
Currently, there are no reliable biomarkers for risk of hypophysitis, and given the protean presentation of hypopituitarism, adrenal insufficiency and hypophysitis, careful clinical assessment is required (9, 23, 37).
Longer term prognosis of hypophysitis
To date, most series have not reported recovery of ACTH function, although recovery of other pituitary axis is reported (9, 37, 38). As discussed previously, high dose steroids do not improve pituitary function recovery (24). Nevertheless there have been occasional reports of spontaneous recovery of pituitary function, including ACTH secretion (39, 40), so clinical and biochemical assessment over time is advised.
A third of patients receiving a CPI will require high dose glucocorticoids for management of non-endocrine IRAEs, at doses associated with adrenal suppression (26, 41). Therefore, caution is also required when stopping glucocorticoids in these patients in case of the development of ACTH deficiency.
Thyroid
The thyroid is the most common endocrine gland to be affected by checkpoint inhibitors, with PD-1 and PD-L1 inhibitors showing a higher rate of clinically overt disease than ipilimumab (42, 43, 44). Hypothyroidism is common, as is transient thyrotoxicosis, usually, but not always, followed by subsequent hypothyroidism. Hypothyroidism is managed with levothyroxine replacement as per standard practice. Hyperthyroidism can usually be managed symptomatically with beta blockers, although steroids are occasionally required, with close monitoring for progression to hypothyroidism (23).
Although symptoms should prompt assessment, thyroid dysfunction is usually detected on routine treatment monitoring blood tests. Both a higher patient BMI or a higher baseline TSH may be associated with an increased risk of thyroid dysfunction (43, 45). Prior use of a tyrosine kinase inhibitor also increases the risk of thyroid dysfunction (46). Raised levels of cytokines including IL-1β and IL-2 pre-treatment, as well as an early rise in thyroglobulin have been reported to predict thyroiditis (47), although the clinical utility of these markers is unclear. FDG-PET uptake in the thyroid is also associated with subsequent hypothyroidism (48).
Notably, whilst onset can occur as early as 3 weeks post therapy initiation, most cases present 1–2 months following but it has been shown to present as late as 3 years (49, 50).
Thyroiditis vs Graves’
The main clinical challenge in thyroid practice is detecting the rare patient with Graves’ disease and distinguishing this from the more common thyroiditis (51, 52, 53), which has been described with both PD-1 and CTLA-4 inhibitors. Those with thyrotoxicosis should therefore be investigated with TSH Receptor antibodies and clinicians should consider an uptake scan to distinguish Graves’ from destructive thyroiditis, taking into account that many patients will have received recent intravenous iodine contrast. However, as the thyrotoxicosis in thyroiditis is usually short lived, these investigations may only be required in prolonged thyrotoxicosis (23). The presence of thyroid eye disease may also alert the clinician to the possibility of Graves’ as a cause of thyrotoxicosis (54, 55). First-line management is with anti-thyroid drugs such as carbimazole.
Diabetes
A form of insulin-requiring diabetes is described in around 1–2% of patients receiving PD-1 or PD-L1 inhibitors, alone or in combination with ipilimumab (33, 56, 57, 58, 59). Features closely resemble Type 1 diabetes, with low c-peptide levels, whilst GAD antibodies are present in around 50% of cases, a notably lower proportion than in spontaneous type 1 diabetes (59, 60).
Several case studies demonstrate that patients given anti-PD-1 therapy present with either severe hyperglycaemia or more commonly, diabetic ketoacidosis (DKA) (58, 61). A large scale review of 71 case reports found that 76% presented with DKA with a mean capillary blood glucose of 33.4 ± 11.5 mmol/L and HbA1c of 62 ± 0.3 mmol/mol (62). 71% of cases developed within 3 months of first exposure to anti-PD-1 or anti-PD-L1 therapy. However, this review found no correlation between HbA1c and time of diabetes diagnosis.
Notably the onset is frequently rapid, with some showing features compatible with fulminant type 1 diabetes, progressing from euglycaemia to DKA in a matter of days (63, 64, 65, 66, 67, 68, 69, 70, 71). Whilst those with very rapid onset are less likely to have classical diabetes autoantibodies, and frequently show elevations of pancreatic enzymes (60), other case series have suggested that autoantibodies were in fact associated with a shorter onset of diabetes (62). Therefore the clinical utility of autoantibodies is unclear, and close attention to any red flag symptoms such as fatigue, thirst or polyuria is required, with prompt assessment of glucose and ketones to try to avoid missing a diagnosis of DKA. Pre-existing type 2 diabetes may be a risk factor for CPI-induced diabetes, with 7–10% of those with type 2 diabetes showing deterioration in control, often requiring insulin (56).
Checkpoint inhibitors (CPIs) are now widely used in oncology treatment however, their use is often associated with treatment toxicity termed ‘immune related adverse events’ (IRAE).
Endocrinopathies are amongst the more common IRAEs and usually result in long term hormone deficiencies.
Detection of endocrinopathy may be challenging due to subtle, non specific symptoms (see Table 1).
Pituitary dysfunction can either be seen as an isolated ACTH deficiency or a more classical hypophysitis (hypopituitarism, headaches, ± pituitary enlargement) with PD-L1 and CTLA4 inhibitors respectively.
High dose glucocorticoids do not improve immunotherapy-induced hypophysitis and may result in worse oncological outcomes versus those receiving only physiological replacement.
Hypothyroidism and transient thyrotoxicosis, followed by hypothyroidism, is the most common endocrinopathy caused by CPIs.
Distinguishing between thyroiditis associated with CPIs and Grave’s disease can be challenging and may require TSH receptor antibody levels and uptake scans to determine diagnosis.
Insulin-requiring diabetes has been described with anti-PD1 therapy and may first present with rapid onset diabetic ketoacidosis (DKA).
A third of patients receiving CPIs require high dose steroids for non-endocrine IRAEs and therefore deteriorating hyperglycaemia needs careful evaluation to determine whether it is steroid-induced or CPI-induced.
Development of endocrinopathy is associated with longer overall survival versus those patients without and therefore, lifelong hormonal treatment with specialist care from both endocrinologists and oncologists is required.
Corticosteroids do not prevent progression to insulin deficiency (72), although insulin deficiency is not universal with one case describing preservation of insulin secretion after cessation of the checkpoint inhibitor (73), and one patient with prior IA2 antibodies able to come off insulin (32).
Steroids and CPI diabetes
A potential confounder is that around 30% of patients receiving CPIs will require high dose steroids for non-endocrine IRAEs (26, 41), with studies suggest that 6–8% of CPI treated patients go on to develop steroid-induced hyperglycaemia (41, 74). As such, patients with new-onset or deteriorating hyperglycaemia after CPI treatment need careful evaluation, including a history of steroid use and assessment for ketosis to ensure that CPI-induced diabetes is not missed, placing patients at particular risk of DKA.
Clinical presentation
Importance of fatigue as red flag
Fatigue is a common symptom in cancer, also being frequent with conventional treatments such as cytotoxic chemotherapy. Close to 40% of patients treated with PD-1/PD-L1 inhibitors have been reported to have fatigue (75), most of whom will not have endocrinopathy. However, fatigue can also be a presentation of adrenal insufficiency (either primary or due to pituitary involvement), thyroid dysfunction and diabetes. As adrenal insufficiency and diabetes leading to DKA can be life threatening, it is vital that onset of fatigue in patients who have received a checkpoint inhibitor leads to prompt investigation for endocrinopathy.
Equally though, it is important to recognise that fatigue can occur, as a result of cancer or its treatment, without endocrine dysfunction, so a thorough evaluation is required before starting hormone replacement.
Long term implications
Unlike most other IRAEs, endocrinopathy is usually non-reversible and is expected to require long term hormone replacement. Hypopituitarism is associated with reduced quality of life, although careful avoidance of over-replacement with glucocorticoids improves this (76). Type 1 diabetes is also well known to be associated with reduced quality of life (77). Both conditions need life long specialist care.
This is particularly important as it is now emerging that the development of endocrinopathy, specifically hypophysitis or thyroid dysfunction, is associated with better cancer outcomes and longer overall survival than controls without endocrinopathy (78, 79, 80). Therefore the cohort of long term survivors is likely to include many with endocrinopathy, with the associated need for hormone replacement, impact on quality of life and need for specialist follow up.
Conclusions
Endocrine dysfunction is among the more frequent immune-related adverse events following checkpoint inhibitor treatment, usually resulting in life long hormone deficiencies. The presentation can be subtle, and a careful endocrine assessment of patients presenting with red-flag symptoms including fatigue is required. Those with confirmed endocrinopathy, especially adrenal insufficiency and diabetes, need ongoing endocrine input to optimise treatment.
Declaration of interest
B A reports no conflict of interest. D M has received speaker and advisory fees from BMS, MSD and Roche.
Funding
This work did not receive any specific grant from any funding agency in the public, commercial or not-for-profit sector.
References
- 1.Drake CG, Lipson EJ, Brahmer JR.Breathing new life into immunotherapy: review of melanoma, lung and kidney cancer. Nature Reviews: Clinical Oncology 2014. 11 24–37. ( 10.1038/nrclinonc.2013.208) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Farkona S, Diamandis EP, Blasutig IM.Cancer immunotherapy: the beginning of the end of cancer? BMC Medicine 2016. 14 73. ( 10.1186/s12916-016-0623-5) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Force J, Salama AK.First-line treatment of metastatic melanoma: role of nivolumab. ImmunoTargets and Therapy 2017. 6 1–10. ( 10.2147/ITT.S110479) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Spiers L, Coupe N, Payne M.Toxicities associated with checkpoint inhibitors – an overview. Rheumatology 2019. 58 vii7–vii16. ( 10.1093/rheumatology/kez418) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haanen JBAG, Carbonnel F, Robert C, Kerr KM, Peters S, Larkin J, Jordan K.ESMO Guidelines Committee. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2017. 28 iv119–iv142. ( 10.1093/annonc/mdx225) [DOI] [PubMed] [Google Scholar]
- 6.Hodi FS, Chiarion-Sileni V, Gonzalez R, Grob J-J, Rutkowski P, Cowey CL, Lao CD, Schadendorf D, Wagstaff J, Dummer Ret al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncology 2018. 19 1480–1492. ( 10.1016/S1470-2045(1830700-9) [DOI] [PubMed] [Google Scholar]
- 7.Bai X, Lin X, Zheng K, Chen X, Wu X, Huang Y, Zhuang Y.Mapping endocrine toxicity spectrum of immune checkpoint inhibitors: a disproportionality analysis using the WHO Adverse Drug Reaction Database, VigiBase. Endocrine 2020. 69 670–681. ( 10.1007/s12020-020-02355-9) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ryder M, Callahan M, Postow MA, Wolchok J, Fagin JA.Endocrine-related adverse events following ipilimumab in patients with advanced melanoma: a comprehensive retrospective review from a single institution. Endocrine-Related Cancer 2014. 21 371–381. ( 10.1530/ERC-13-0499) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Faje AT, Sullivan R, Lawrence D, Tritos NA, Fadden R, Klibanski A, Nachtigall L.Ipilimumab-induced hypophysitis: a detailed longitudinal analysis in a large cohort of patients with metastatic melanoma. Journal of Clinical Endocrinology and Metabolism 2014. 99 4078–4085. ( 10.1210/jc.2014-2306) [DOI] [PubMed] [Google Scholar]
- 10.Chodakiewitz Y, Brown S, Boxerman JL, Brody JM, Rogg JM.Ipilimumab treatment associated pituitary hypophysitis: clinical presentation and imaging diagnosis. Clinical Neurology and Neurosurgery 2014. 125 125–130. ( 10.1016/j.clineuro.2014.06.011) [DOI] [PubMed] [Google Scholar]
- 11.De Sousa SM, Long GV, Tonks KT.Ipilimumab-induced hypophysitis: early Australian experience. Medical Journal of Australia 2014. 201 198–199. ( 10.5694/mja14.00803) [DOI] [PubMed] [Google Scholar]
- 12.Lammert A, Schneider HJ, Bergmann T, Benck U, Kramer BK, Gartner R, Metzner C, Schofl C, Berking C.Hypophysitis caused by ipilimumab in cancer patients: hormone replacement or immunosuppressive therapy. Experimental and Clinical Endocrinology and Diabetes 2013. 121 581–587. ( 10.1055/s-0033-1355337) [DOI] [PubMed] [Google Scholar]
- 13.Siddiqui MS, Lai ZM, Spain L, Greener V, Turajlic S, Larkin J, Morganstein DL.Predicting development of ipilimumab-induced hypophysitis: utility of T4 and TSH index but not TSH. Journal of Endocrinological Investigation 2021. 44 195–203. ( 10.1007/s40618-020-01297-3) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Albarel F, Gaudy C, Castinetti F, Carré T, Morange I, Conte-Devolx B, Grob JJ, Brue T.Long-term follow-up of ipilimumab-induced hypophysitis, a common adverse event of the anti-CTLA-4 antibody in melanoma. European Journal of Endocrinology 2015. 172 195–204. ( 10.1530/EJE-14-0845) [DOI] [PubMed] [Google Scholar]
- 15.Faje A, Reynolds K, Zubiri L, Lawrence D, Cohen JV, Sullivan RJ, Nachtigall LB, Tritos N.Hypophysitis secondary to nivolumab and pembrolizumab is a clinical entity distinct from ipilimumab-associated hypophysitis. European Journal of Endocrinology 2019. 181 211–219. ( 10.1530/EJE-19-0238) [DOI] [PubMed] [Google Scholar]
- 16.Percik R, Shlomai G, Tirosh A, Tirosh A, Leibowitz-Amit R, Eshet Y, Greenberg G, Merlinsky A, Barhod E, Steinberg-Silman Yet al. Isolated autoimmune adrenocorticotropic hormone deficiency: from a rare disease to the dominant cause of adrenal insufficiency related to check point inhibitors. Autoimmunity Reviews 2019. 19 102454. ( 10.1016/j.autrev.2019.102454) [DOI] [PubMed] [Google Scholar]
- 17.Garon-Czmil J, Petitpain N, Rouby F, Sassier M, Babai S, Yéléhé-Okouma M, Weryha G, Klein M, Gillet P.Immune check point inhibitors-induced hypophysitis: a retrospective analysis of the French pharmacovigilance database. Scientific Reports 2019. 9 19419. ( 10.1038/s41598-019-56026-5) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rodrigues BT, Otty Z, Sangla K, Shenoy VV.Ipilimumab-induced autoimmune hypophysitis: a differential for sellar mass lesions. Endocrinology, Diabetes and Metabolism Case Reports 2015. 2014 140098. ( 10.1530/EDM-14-0098) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dillard T, Yedinak CG, Alumkal J, Fleseriu M.Anti-CTLA-4 antibody therapy associated autoimmune hypophysitis: serious immune related adverse events across a spectrum of cancer subtypes. Pituitary 2010. 13 29–38. ( 10.1007/s11102-009-0193-z) [DOI] [PubMed] [Google Scholar]
- 20.Torino F, Barnabei A, De Vecchis L, Salvatori R, Corsello SM.Hypophysitis induced by monoclonal antibodies to cytotoxic T lymphocyte antigen 4: challenges from a new cause of a rare disease. Oncologist 2012. 17 525–535. ( 10.1634/theoncologist.2011-0404) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Faje A.Immunotherapy and hypophysitis: clinical presentation, treatment, and biologic insights. Pituitary 2016. 19 82–92. ( 10.1007/s11102-015-0671-4) [DOI] [PubMed] [Google Scholar]
- 22.Carpenter KJ, Murtagh RD, Lilienfeld H, Weber J, Murtagh FR.Ipilimumab-induced hypophysitis: MR imaging findings. American Journal of Neuroradiology 2009. 30 1751–1753. ( 10.3174/ajnr.A1623) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Higham CE, Olsson-Brown A, Carroll P, Cooksley T, Larkin J, Lorigan P, Morganstein D, Trainer PJ.Society for Endocrinology Clinical Committee. SOCIETY FOR ENDOCRINOLOGY ENDOCRINE EMERGENCY GUIDANCE: Acute management of the endocrine complications of checkpoint inhibitor therapy. Endocrine Connections 2018. 7 G1–G7. ( 10.1530/EC-18-0068) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Min L, Hodi FS, Giobbie-Hurder A, Ott PA, Luke JJ, Donahue H, Davis M, Carroll RS, Kaiser UB.Systemic high-dose corticosteroid treatment does not improve the outcome of ipilimumab-related hypophysitis: a retrospective cohort study. Clinical Cancer Research 2015. 21 749–755. ( 10.1158/1078-0432.CCR-14-2353) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Faje AT, Lawrence D, Flaherty K, Freedman C, Fadden R, Rubin K, Cohen J, Sullivan RJ.High-dose glucocorticoids for the treatment of ipilimumab-induced hypophysitis is associated with reduced survival in patients with melanoma. Cancer 2018. 124 3706–3714. ( 10.1002/cncr.31629) [DOI] [PubMed] [Google Scholar]
- 26.Horvat TZ, Adel NG, Dang TO, Momtaz P, Postow MA, Callahan MK, Carvajal RD, Dickson MA, D’Angelo SP, Woo KMet al. Immune-related adverse events, need for systemic immunosuppression, and effects on survival and time to treatment failure in patients with melanoma treated with ipilimumab at Memorial Sloan Kettering Cancer Center. Journal of Clinical Oncology 2015. 33 3193–3198. ( 10.1200/JCO.2015.60.8448) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Simpson H, Tomlinson J, Wass J, Dean J, Arlt W.Guidance for the prevention and emergency management of adult patients with adrenal insufficiency. Clinical Medicine 2020. 20 371–378. ( 10.7861/clinmed.2019-0324) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cooksley T, Knight T, Gupta A, Higham C, Lorigan P, Adam S.Emergency ambulatory outpatient management of immune-mediated hypophysitis. Supportive Care in Cancer 2020. 28 3995–3999. ( 10.1007/s00520-020-05581-z) [DOI] [PubMed] [Google Scholar]
- 29.Yuen KCJ, Heaney AP, Popovic V.Considering GH replacement for GH-deficient adults with a previous history of cancer: a conundrum for the clinician. Endocrine 2016. 52 194–205. ( 10.1007/s12020-015-0840-2) [DOI] [PubMed] [Google Scholar]
- 30.De Sousa SMC, Sheriff N, Tran CH, Menzies AM, Tsang VHM, Long GV, Tonks KTT.Fall in thyroid stimulating hormone (TSH) may be an early marker of ipilimumab-induced hypophysitis. Pituitary 2018. 21 274–282. ( 10.1007/s11102-018-0866-6) [DOI] [PubMed] [Google Scholar]
- 31.Way J, Drakaki A, Drexler A, Freeby M.Anti-PD-L1 therapy and the onset of diabetes mellitus with positive pancreatic autoantibodies. BMJ Case Reports 2017. 2017 bcr2017220415. ( 10.1136/bcr-2017-220415) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ohara N, Kobayashi M, Ikeda Y, Hoshi T, Morita S, Kanefuji T, Yagi K, Suda T, Takada T, Hasegawa Get al. Non-insulin-dependent diabetes mellitus induced by immune checkpoint inhibitor therapy in an insulinoma-associated antigen-2 autoantibody-positive patient with advanced gastric cancer. Internal Medicine 2019. 59 551–556. ( 10.2169/internalmedicine.3208-19) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Galligan A, Xu W, Fourlanos S, Nankervis A, Chiang C, Mant AM, Parente P, Rischin D, Krishnamurthy B, Sandhu Set al. Diabetes associated with immune checkpoint inhibition: presentation and management challenges. Diabetic Medicine 2018. 35 1283–1290. ( 10.1111/dme.13762) [DOI] [PubMed] [Google Scholar]
- 34.Tahir SA, Gao J, Miura Y, Blando J, Tidwell RSS, Zhao H, Subudhi SK, Tawbi H, Keung E, Wargo Jet al. Autoimmune antibodies correlate with immune checkpoint therapy-induced toxicities. PNAS 2019. 116 22246–22251. ( 10.1073/pnas.1908079116) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Leiter A, Gnjatic S, Fowkes M, Kim-Schulze S, Laface I, Galsky MD, Gallagher EJ.A common pituitary autoantibody in two patients with immune checkpoint inhibitor-mediated hypophysitis: ZCCHC8. AACE Clinical Case Reports 2020. 6 e151–e160. ( 10.4158/ACCR-2019-0585) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yano S, Ashida K, Sakamoto R, Sakaguchi C, Ogata M, Maruyama K, Sakamoto S, Ikeda M, Ohe K, Akasu Set al. Human leucocyte antigen DR15, a possible predictive marker for immune checkpoint inhibitor-induced secondary adrenal insufficiency. European Journal of Cancer 2020. 130 198–203. ( 10.1016/j.ejca.2020.02.049) [DOI] [PubMed] [Google Scholar]
- 37.Snyders T, Chakos D, Swami U, Latour E, Chen Y, Fleseriu M, Milhem M, Zakharia Y, Zahr R.Ipilimumab-induced hypophysitis, a single academic center experience. Pituitary 2019. 22 488–496. ( 10.1007/s11102-019-00978-4) [DOI] [PubMed] [Google Scholar]
- 38.Lin CH, Chen KH, Chen KY, Shih SR, Lu JY.Immune checkpoint inhibitor therapy-induced hypophysitis approximately a case series of Taiwanese patients. Journal of the Formosan Medical Association 2019. 118 524–529. ( 10.1016/j.jfma.2018.07.014) [DOI] [PubMed] [Google Scholar]
- 39.Erra A, Pannu BS, Patel S, Qureshi F, Soliman M.A rare case of ipilimumab-induced reversible hypophysitis and permanent primary hypothyroidism. Cureus 2019. 2019 e5001. ( 10.7759/cureus.5001) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Thapi S, Leiter A, Galsky M, Gallagher EJ.Recovery from secondary adrenal insufficiency in a patient with immune checkpoint inhibitor therapy induced hypophysitis. Journal for ImmunoTherapy of Cancer 2019. 7 248. ( 10.1186/s40425-019-0729-3) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Agarwal K, Yousaf N, Morganstein D.Glucocorticoid use and complications following immune checkpoint inhibitor use in melanoma. Clinical Medicine 2020. 20 163–168. ( 10.7861/clinmed.2018-0440) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bai X, Chen X, Wu X, Huang Y, Zhuang Y, Lin X.Immune checkpoint inhibitor-associated thyroid dysfunction: a disproportionality analysis using the WHO Adverse Drug Reaction Database, VigiBase. European Journal of Endocrinology 2020. 182 1–9. ( 10.1530/EJE-19-0535) [DOI] [PubMed] [Google Scholar]
- 43.Morganstein DL, Lai Z, Spain L, Diem S, Levine D, Mace C, Gore M, Larkin J.Thyroid abnormalities following the use of cytotoxic T-lymphocyte antigen-4 and programmed death receptor protein-1 inhibitors in the treatment of melanoma. Clinical Endocrinology 2017. 86 614–620. ( 10.1111/cen.13297) [DOI] [PubMed] [Google Scholar]
- 44.Barroso-Sousa R, Barry WT, Garrido-Castro AC, Hodi FS, Min L, Krop IE, Tolaney SM.Incidence of endocrine dysfunction following the use of different immune checkpoint inhibitor regimens: a systematic review and meta-analysis. JAMA Oncology 2018. 4 173–182. ( 10.1001/jamaoncol.2017.3064) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pollack R, Ashash A, Cahn A, Rottenberg Y, Stern H, Dresner-Pollak R.Immune checkpoint inhibitor-induced thyroid dysfunction is associated with higher body mass index. Journal of Clinical Endocrinology and Metabolism 2020. 105 dgaa458. ( 10.1210/clinem/dgaa458) [DOI] [PubMed] [Google Scholar]
- 46.Sbardella E, Tenuta M, Sirgiovanni G, Gianfrilli D, Pozza C, Venneri MA, Cortesi E, Marchetti P, Lenzi A, Gelibter AJet al. Thyroid disorders in programmed death 1 inhibitor-treated patients: is previous therapy with tyrosine kinase inhibitors a predisposing factor? Clinical Endocrinology 2020. 92 258–265. ( 10.1111/cen.14135) [DOI] [PubMed] [Google Scholar]
- 47.Kurimoto C, Inaba H, Ariyasu H, Iwakura H, Ueda Y, Uraki S, Takeshima K, Furukawa Y, Morita S, Yamamoto Yet al. Predictive and sensitive biomarkers for thyroid dysfunctions during treatment with immune-checkpoint inhibitors. Cancer Science 2020. 111 1468–1477. ( 10.1111/cas.14363) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Frelau A, Palard-Novello X, Jali E, Boussemart L, Dupuy A, James P, Devillers A, Le Jeune F, Edeline J, Lesimple Tet al. Increased thyroid uptake on 18F-FDG PET/CT is associated with the development of permanent hypothyroidism in stage IV melanoma patients treated with anti-PD-1 antibodies. Cancer Immunology, Immunotherapy 2020. [epub]. ( 10.1007/s00262-020-02712-7) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Delivanis DA, Gustafson MP, Bornschlegl S, Merten MM, Kottschade L, Withers S, Dietz AB, Ryder M.Pembrolizumab-induced thyroiditis: comprehensive clinical review and insights into underlying involved mechanisms. Journal of Clinical Endocrinology and Metabolism 2017. 102 2770–2780. ( 10.1210/jc.2017-00448) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Osorio JC, Ni A, Chaft JE, Pollina R, Kasler MK, Stephens D, Rodriguez C, Cambridge L, Rizvi H, Wolchok JDet al. Antibody-mediated thyroid dysfunction during T-cell checkpoint blockade in patients with non-small-cell lung cancer. Annals of Oncology 2017. 28 583–589. ( 10.1093/annonc/mdw640) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yamada H, Okajima F, Onda T, Fujimori S, Emoto N, Sugihara H.New-onset Graves’ disease after the initiation of nivolumab therapy for gastric cancer: a case report. BMC Endocrine Disorders 2020. 20 132. ( 10.1186/s12902-020-00613-5) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Azmat U, Liebner D, Joehlin-Price A, Agrawal A, Nabhan F.Treatment of ipilimumab induced Graves’ disease in a patient with metastatic melanoma. Case Reports in Endocrinology 2016. 2016 2087525. ( 10.1155/2016/2087525) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Borodic G, Hinkle DM, Cia Y.Drug-induced graves disease from CTLA-4 receptor suppression. Ophthalmic Plastic and Reconstructive Surgery 2011. 27 e87–e. ( 10.1097/IOP.0b013e3181ef72a1) [DOI] [PubMed] [Google Scholar]
- 54.Borodic GE, Hinkle D.Ipilimumab-induced orbital inflammation resembling Graves disease with subsequent development of systemic hyperthyroidism from CTLA-4 receptor suppression. Ophthalmic Plastic and Reconstructive Surgery 2014. 30 83. ( 10.1097/IOP.0000000000000033) [DOI] [PubMed] [Google Scholar]
- 55.Rhea L, Yoon JW, Jang S.Rapid development of graves’ ophthalmopathy after treatment with ipilimumab and recurrence with pembrolizumab in a patient with previously treated Graves’ disease. Journal of Oncology Practice 2018. 14 747–749. ( 10.1200/JOP.18.00442) [DOI] [PubMed] [Google Scholar]
- 56.Kotwal A, Haddox C, Block M, Kudva YC.Immune checkpoint inhibitors: an emerging cause of insulin-dependent diabetes. BMJ Open Diabetes Research and Care 2019. 7 e000591. ( 10.1136/bmjdrc-2018-000591) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Marchand L, Thivolet A, Dalle S, Chikh K, Reffet S, Vouillarmet J, Fabien N, Cugnet-Anceau C, Thivolet C.Diabetes mellitus induced by PD-1 and PD-L1 inhibitors: description of pancreatic endocrine and exocrine phenotype. Acta Diabetologica 2019. 56 441–448. ( 10.1007/s00592-018-1234-8) [DOI] [PubMed] [Google Scholar]
- 58.Stamatouli AM, Quandt Z, Perdigoto AL, Clark PL, Kluger H, Weiss SA, Gettinger S, Sznol M, Young A, Rushakoff Ret al. Collateral damage: insulin-dependent diabetes induced with checkpoint inhibitors. Diabetes 2018. 67 1471–1480. ( 10.2337/dbi18-0002) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Clotman K, Janssens K, Specenier P, Weets I, De Block CEM.Programmed cell death-1 (PD-1) inhibitor induced type 1 diabetes mellitus: mini-review. Journal of Clinical Endocrinology and Metabolism 2018. 103 3144–3154. ( 10.1210/jc.2018-00728) [DOI] [PubMed] [Google Scholar]
- 60.Kyriacou A, Melson E, Chen W, Kempegowda P.Is immune checkpoint inhibitor-associated diabetes the same as fulminant type 1 diabetes mellitus? Clinical Medicine 2020. 20 417–423. ( 10.7861/clinmed.2020-0054) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hughes J, Vudattu N, Sznol M, Gettinger S, Kluger H, Lupsa B, Herold KC.Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care 2015. 38 e55–e. ( 10.2337/dc14-2349) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Akturk HK, Kahramangil D, Sarwal A, Hoffecker L, Murad MH, Michels AW.Immune checkpoint inhibitor-induced Type 1 diabetes: a systematic review and meta-analysis. Diabetic Medicine 2019. 36 1075–1081. ( 10.1111/dme.14050) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gaudy C, Clevy C, Monestier S, Dubois N, Preau Y, Mallet S, Richard MA, Grob JJ, Valero R, Beliard S.Anti-PD1 pembrolizumab can induce exceptional fulminant Type 1 diabetes. Diabetes Care 2015. 2015 e182–e. ( 10.2337/dc15-1331) [DOI] [PubMed] [Google Scholar]
- 64.Ishikawa K, Shono-Saito T, Yamate T, Kai Y, Sakai T, Shimizu F, Yamada Y, Mori H, Noso S, Ikegami Het al. A case of fulminant type 1 diabetes mellitus, with a precipitous decrease in pancreatic volume, induced by nivolumab for malignant melanoma: analysis of HLA and CTLA-4 polymorphisms. European Journal of Dermatology 2017. 27 184–185. ( 10.1684/ejd.2016.2923) [DOI] [PubMed] [Google Scholar]
- 65.Tsiogka A, Jansky GL, Bauer JW, Koelblinger P.Fulminant type 1 diabetes after adjuvant ipilimumab therapy in cutaneous melanoma. Melanoma Research 2017. 27 524–525. ( 10.1097/CMR.0000000000000384) [DOI] [PubMed] [Google Scholar]
- 66.Mizab Mellah C, Sanchez Perez M, Santos Rey MD, Hernandez Garcia M.Fulminant type 1 diabetes mellitus associated with pembrolizumab. Endocrinologia, Diabetes y Nutricion 2017. 64 272–273. ( 10.1016/j.endinu.2017.01.005) [DOI] [PubMed] [Google Scholar]
- 67.Takahashi A, Tsutsumida A, Namikawa K, Yamazaki N.Fulminant type 1 diabetes associated with nivolumab in a patient with metastatic melanoma. Melanoma Research 2018. 28 159–160. ( 10.1097/CMR.0000000000000418) [DOI] [PubMed] [Google Scholar]
- 68.Araujo M, Ligeiro D, Costa L, Marques F, Trindade H, Correia JM, Fonseca C.A case of fulminant Type 1 diabetes following anti-PD1 immunotherapy in a genetically susceptible patient. Immunotherapy 2017. 9 531–535. ( 10.2217/imt-2017-0020) [DOI] [PubMed] [Google Scholar]
- 69.Munakata W, Ohashi K, Yamauchi N, Tobinai K.Fulminant type I diabetes mellitus associated with nivolumab in a patient with relapsed classical Hodgkin lymphoma. International Journal of Hematology 2017. 105 383–386. ( 10.1007/s12185-016-2101-4) [DOI] [PubMed] [Google Scholar]
- 70.Okamoto M, Okamoto M, Gotoh K, Masaki T, Ozeki Y, Ando H, Anai M, Sato A, Yoshida Y, Ueda Set al. Fulminant type 1 diabetes mellitus with anti-programmed cell death-1 therapy. Journal of Diabetes Investigation 2016. 7 915–918. ( 10.1111/jdi.12531) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lowe JR, Perry DJ, Salama AK, Mathews CE, Moss LG, Hanks BA.Genetic risk analysis of a patient with fulminant autoimmune type 1 diabetes mellitus secondary to combination ipilimumab and nivolumab immunotherapy. Journal for ImmunoTherapy of Cancer 2016. 4 89. ( 10.1186/s40425-016-0196-z) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Aleksova J, Lau PK, Soldatos G, McArthur G.Glucocorticoids did not reverse type 1 diabetes mellitus secondary to pembrolizumab in a patient with metastatic melanoma. BMJ Case Reports 2016. 2016 bcr2016217454. ( 10.1136/bcr-2016-217454) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sakai G, Saito D, Nakajima R, Hatano M, Noguchi Y, Kurihara S, Katayama S, Inoue I, Noda M, Shimada A.Intrinsic insulin secretion capacity might be preserved by discontinuing anti-programmed cell death protein 1 antibody treatment in ‘anti-programmed cell death protein 1 antibody-induced’ fulminant type 1 diabetes. Journal of Diabetes Investigation 2018. 9 448–449. ( 10.1111/jdi.12662) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Williams KJ, Grauer DW, Henry DW, Rockey ML.Corticosteroids for the management of immune-related adverse events in patients receiving checkpoint inhibitors. Journal of Oncology Pharmacy Practice 2019. 25 544–550. ( 10.1177/1078155217744872) [DOI] [PubMed] [Google Scholar]
- 75.Cortellini A, Vitale MG, De Galitiis F, Di Pietro FR, Berardi R, Torniai M, De Tursi M, Grassadonia A, Di Marino P, Santini Det al. Early fatigue in cancer patients receiving PD-1/PD-L1 checkpoint inhibitors: an insight from clinical practice. Journal of Translational Medicine 2019. 17 376. ( 10.1186/s12967-019-02132-x) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ajmal A, Mckean E, Sullivan S, Barkan A.Decreased quality of life (QoL) in hypopituitary patients: involvement of glucocorticoid replacement and radiation therapy. Pituitary 2018. 21 624–630. ( 10.1007/s11102-018-0918-y) [DOI] [PubMed] [Google Scholar]
- 77.Goldney RD, Phillips PJ, Fisher LJ, Wilson DH.Diabetes, depression, and quality of life: a population study. Diabetes Care 2004. 27 1066–1070. ( 10.2337/diacare.27.5.1066) [DOI] [PubMed] [Google Scholar]
- 78.Kotwal A, Kottschade L, Ryder M.PD-L1 inhibitor-induced thyroiditis is associated with better overall survival in cancer patients. Thyroid 2020. 30 177–184. ( 10.1089/thy.2019.0250) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lima Ferreira J, Costa C, Marques B, Castro S, Victor M, Oliveira J, Santos AP, Sampaio IL, Duarte H, Marques APet al. Improved survival in patients with thyroid function test abnormalities secondary to immune-checkpoint inhibitors. Cancer Immunology, Immunotherapy 2020. [epub]. ( 10.1007/s00262-020-02664-y) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kobayashi T, Iwama S, Yasuda Y, Okada N, Okuji T, Ito M, Onoue T, Goto M, Sugiyama M, Tsunekawa Tet al. Pituitary dysfunction induced by immune checkpoint inhibitors is associated with better overall survival in both malignant melanoma and non-small cell lung carcinoma: a prospective study. Journal for ImmunoTherapy of Cancer 2020. 8 e000779. ( 10.1136/jitc-2020-000779) [DOI] [PMC free article] [PubMed] [Google Scholar]



 This work is licensed under a
This work is licensed under a