Abstract
Endoscopic procedures are the front-runner of the management of bleeding duodenal ulcer. Rarely, surgical intervention is sought for acute bleeding, not amenable to endoscopic procedures. Oversewing of the gastroduodenal artery at ulcer crater by transduodenal approach is the most acceptable and recommended method of treatment. We describe a case of an intraoperative duodenal injury that occurred during an attempt to oversew the gastroduodenal artery after a duodenotomy, leading to an unsatisfactory and meagre duodenal stump. This case will highlight the intraoperative turmoil, postoperative complications and management of a series of anticipated but unfortunate events that have rendered us wiser in terms of surgical management of a bleeding duodenal ulcer.
Keywords: ulcer, stomach and duodenum, adult intensive care, gastrointestinal surgery, general surgery
Background
Upper gastrointestinal (UGI) haemorrhage due to a bleeding duodenal ulcer is a common presentation in the emergency room. The majority of these cases are managed conservatively, by resuscitation with volume replacement by transfusion of blood products and the correction of coagulopathy. UGI endoscopy is both diagnostic and therapeutic in most of these cases. Surgical intervention is rarely needed. Surgical options include transduodenal ligation of bleeding vessel, ligation of periduodenal vessels, ulcer excision combined with vagotomy and gastric resection. The most common procedure is a duodenotomy, with direct control of the bleeding site. The gastroduodenal artery, the most common culprit, is located on the posterior wall of the first part of the duodenum. The ulcers situated at the posterior wall of the duodenum can erode this vessel to cause torrential haemorrhage. The bleeding vessel is usually approached through a longitudinal incision over the pylorus and first part of duodenum, followed by identification and ligation of bleeding vessel at the ulcer crater. Periduodenal ligation of vessels may be attempted in case of failure to control bleeding by the transduodenal approach. Since this surgical procedure is not performed routinely, many young surgeons and residents may find this procedure formidable. Although the surgical procedure sounds simple, the intraoperative journey can be extremely arduous in the presence of active bleeding. Visualisation of the bleeding vessel and oversewing it through a small incision over the duodenum is not a cakewalk in an inexperienced hand. Even periduodenal ligation of the vessels is onerous in the presence of scarring and active bleeding. This article aimed to highlight the challenges faced by our operating team during the surgical management of a bleeding duodenal ulcer. We would also like to emphasise the errors committed during the operation and the management of these complications.
Case presentation
A 65-year-old man presented to the emergency with massive haematemesis, tachycardia and hypotension. After initial resuscitation, an urgent UGI endoscopy revealed active bleeding in the posterior wall of the first part of the duodenum. With the application of all available methods, bleeding could not be controlled by the endoscopic approach. Because of active bleeding and haemodynamic instability, the patient was wheeled in for surgical exploration.
Investigations
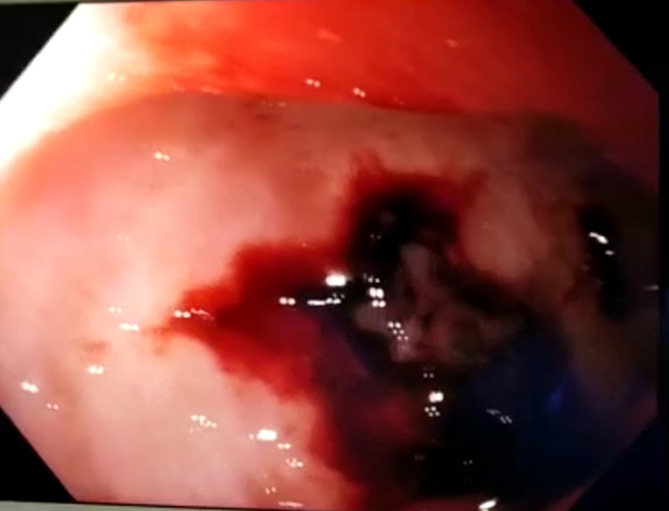
UGI endoscopy revealed a bleeding posterior duodenal ulcer, located at D1–D2 junction-Forrest 1A (figure 1).
Figure 1.
Bleeding duodenal ulcer.
Treatment
Exploratory laparotomy was performed immediately with a plan to do transduodenal oversewing of the bleeding vessels. A 3 cm incision was made over the first part of the duodenum along its long axis by the operating surgeon. However, due to visual compromise, the operating surgeon extended the incision to locate and control the bleeding. Bleeding from the eroded gastroduodenal artery was under-run by using a 2-0 polypropylene suture.
Due to overzealous retraction of the duodenotomy, to control the actively bleeding ulcer, inadvertent unrepairable injury of the first part of the duodenum with almost complete transection occurred. Scarring over the posterior part of the duodenum (bleeding site) eventually made the matter atrocious. The Achilles’ heel of the operation was the management of distal duodenal stump. It was not possible to close the duodenal stump due to the cicatrisation and scarring of the posterior wall of the duodenum. The operating surgeon sought the opinion and help from senior colleagues. The vexed question was the possibility of primary closure of the duodenal stump. It was also not possible to do end-to-end anastomosis of pylorus and duodenum due to lack of duodenal stump margin posteriorly even after mobilisation. With Hobson’s choice, the team proceeded with an antrectomy and truncal vagotomy with a Bilroth II gastrojejunostomy.
The duodenal stump was attempted closure with interrupted sutures using 3-0 Polydioxanone (PDS). However, the repair was unsatisfactory, given the deficiency of margins of the posterior wall of the duodenum. Anticipating a leak in the postoperative period, we decided to put a reverse duodenostomy (RD) tube. A 12F (French) nasogastric tube was inserted into the jejunum, about 12 cm distal to the duodenojejunal junction. The tube was negotiated proximally until the second part of the duodenum. The distal end of the tube was brought out through a separate opening in the abdominal wall. The tube was fixed both at the jejunal incision site and the abdominal wall. The team decided against the feeding jejunostomy (FJ), although one member had vouched for this intraoperatively.
Outcome and follow-up
The patient improved in the postoperative period with vitals stabilised. There were no fresh episodes of gastrointestinal bleeding. He was allowed orally 2 days after the surgery, once he showed the evidence of the passage of flatus, minimal Ryle’s tube output and presence of bowel sound. The RD tube was kept on continuous drainage for the first 3 days after the surgery. It was intermittently clamped once the patient started to tolerate an oral diet. But within 48 hours of clamping the RD tube, he developed fever with rising total leucocyte count (TLC), persistent bilious vomiting and upper abdominal pain. The abdominal sonography revealed a significant echogenic collection in the right subhepatic space. A pigtail drain (10F) was inserted under ultrasound guidance and drained about 300 mL of purulent bilious fluid. The collection was most likely secondary to a duodenal stump leak. Clinical and laboratory parameters improved after the drainage of the infected collection. Hence, further evaluation with CT scan was not done. The RD tube was declamped and put on continuous drainage for a week. The patient tolerated oral feeding. In the absence of further output in the pigtail catheter, reverse duodenostomy tube was reclamped. As soon as the RD was clamped, the bilious output increased in the pigtail drain. The daily output of the bile was about 200 mL/day. In the absence of FJ, it was decided to keep the RD on continuous drainage along with the continuation of oral diet as tolerated by the patient. Fortunately, vomiting did not recur, and the patient tolerated the diet. Eventually, after 4 weeks, we could remove the pigtail catheter and clamp the RD. Removal of the RD was done after 6 weeks on ensuring no residual intra-abdominal collection by ultrasonography. At the end of 6 months, the patient is doing well without any specific complaints.
Discussion
The incidence of acute UGI bleed ranges from 50 to 150 per 1 lakh population annually and elderly people are most affected.1 The common causes are peptic ulcer (32%–36%), oesophagitis (approximately 24%), gastric erosions (18%– 22%), variceal bleeding (approximately 11%) and other causes like Mallory-Weiss tear, Dieulafoy lesions, secondary to anticoagulant and aortoenteric fistulas account for less than 5%.2 The initial presentation includes haematemesis and melaena. It may be accompanied by dizziness, syncope and sweating. Massive UGI bleed may lead to altered mentation and also carries a risk of aspiration. It is initially managed by resuscitation with intravenous fluids, infusion of proton pump inhibitor, transfusion of blood products and correction of coagulopathy. It should be followed by an urgent endoscopy to identify the source of bleeding and to do the imperative therapeutic procedure.3 Bleeding peptic ulcers are classified according to Forrest classification (table 1).4
Table 1.
Forrest classification based on endoscopic appearance of bleeding lesion
| Description of ulcer | Forrest classification |
| Active bleeding (type 1) | |
| Spurting haemorrhage | IA |
| Oozing haemorrhage | IB |
| Signs of recent bleed (type 2) | |
| Non-bleeding visible vessel | IIA |
| Adherent clot on lesion | IIB |
| Haematin-stained lesion | IIC |
| Without bleeding (type 3) | |
| Clean base, flat spot | III |
UGI endoscopy interventions for bleeding peptic ulcers include epinephrine injection, thermal ablation and application of endoclips. Sclerotherapy is less effective, hence not routinely used.5 6 Epinephrine injection should not be used alone.6 7 A double-lumen endoscope is preferred for simultaneous washing and removal of clots. When the bleeding occurs secondary to antiplatelet drugs, endoscopic clipping is safer than thermal ablation.6 In the case of failed endoscopic intervention, emergency surgery or angioembolisation by the interventional radiologist is warranted. The surgical intervention is required in only about 5%–10% of bleeding ulcers.6 The Blatchford score can be used to identify the patients who may require surgical intervention either to stop bleeding or decrease the chance of rebleeding (table 2).8
Table 2.
Blatchford score
| Admission risk marker | Range | Score |
| Blood urea nitrogen (mmol/L) | ≥18.2–<22.4 | 2 |
| >22.4–<28 | 3 | |
| >28–<70 | 4 | |
| >70 | 6 | |
| Haemoglobin level for men (g/dL) | >12–<13 | 1 |
| ≥10–<12 | 3 | |
| <10 | 6 | |
| Haemoglobin level for women (g/dL) | ≥10–<12 | 1 |
| <10 | 6 | |
| Systolic blood pressure (mm Hg) | ≥100–109 | 1 |
| >90–<100 | 2 | |
| <90 | 3 | |
| Other markers | Pulse rate ≥100 | 1 |
| Presentation with melaena | 1 | |
| Presentation with syncope | 2 | |
| Hepatic disease | 2 | |
| Heart failure | 2 |
The score of 6 or higher is associated with more than 50% of the risk of rebleeding. Ulcer more than 2 cm in size, located at posterior duodenum and lesser curvature, active bleeding during endoscopy, haemodynamic instability and comorbid conditions are independently associated with risk of rebleeding and can be considered as a candidate for surgical intervention.9 The surgeon should be present during the endoscopy to know the exact location of the ulcer. Surgical intervention depends on the location of the ulcer.10 Ulcers along the greater curvature are managed by wedge excision of the ulcer. However, wedge excision of the ulcer along the lesser curvature can lead to deformity of the stomach. Besides, there is a higher risk of malignancy. Hence, gastrectomy should be considered for lesions in lesser curvature. Distal gastric ulcers are managed with distal gastrectomy and reconstruction.10
Bleeding duodenal ulcer is managed with three-point suturing, which includes proximal and distal part of the gastroduodenal artery and transverse pancreatic artery (branch of the gastroduodenal artery).11 The most common approach is transduodenal. Anterior longitudinal duodenotomy is performed over the first part of the duodenum, which can be extended up to the pylorus. After ligation of vessels at ulcer crater, the duodenotomy incision should be closed transversely, as during a Heineke-Mikulicz pyloroplasty.11 This is mostly followed by vagotomy to decrease the amount of acid secretion. Antrectomy is not preferred due to cicatrisation of the duodenal end. The other option is mobilisation of the duodenum, followed by ligation of the gastroduodenal artery and periduodenal vessels. However, this procedure is more challenging and should be condemned during the emergency. In the era of proton pump inhibitors, the surgical management of peptic ulcer has fallen in favour. The young surgeons are rarely exposed to bleeding duodenal ulcers, even in high-volume centres. While in an experienced hand, surgery can be a cakewalk, the inexperienced surgeon may find the procedure challenging. The case was like an epiphany for us. It nudged us of the old saying that ‘A stitch on time saves nine’. The incision over the duodenum was made transversely instead of a vertical incision. The final result was opened up duodenum, leading to a problematic duodenal stump. We anticipated the menace of duodenal stump leak in case of primary closure of the stump. The situation was managed by antrectomy and drainage of the duodenum. The gastric end was closed by a stapler. Truncal vagotomy, followed by a gastrojejunostomy, was also performed. Duodenal stump was closed with a lot of intricacies due to cicatrisation and wide-open duodenum. RD was performed to decompress the duodenum.
As expected, the patient developed a duodenal stump leak in the postoperative period. The leak was reckoned because of fever, high TLC and the subhepatic collection demonstrated by the ultrasound. The collection was drained by inserting a pigtail catheter. We subsequently noticed that the pigtail output was directly related to the clamping of RD. Clamping the RD resulted in an increase in the output from the pigtail catheter. In contrast, putting RD on continuous drainage was associated with a fall in pigtail catheter output.
We also felt the need for FJ as the patient had persistent bilious vomiting and repeated attempt to feed him orally was not successful. In the presence of FJ, we could have started feeding early.12 Many studies recommend early feeding to reduce the risk of infection and malnutrition. It also prevents complications related to parenteral nutrition.
The literature supports the use of ancillary procedures like pyloric exclusion, tube duodenostomy (either antegrade or retrograde) and FJ for managing duodenal injury.13 The same procedures may be applied to manage difficult duodenal stumps. The procedure like side-to-side duodenojejunostomy may be attempted. But we do not recommend this procedure in an emergency and by an inexperienced surgeon. The anastomosis is time-consuming and precarious due to poor blood supply and high bile load. The triple tube decompression, that is, duodenostomy, gastrostomy and FJ, is easier to perform and is safer even in inexperienced hand.13 Due to the excellent blood supply of the stomach, the prospect of an anastomotic leak with gastrojejunostomy is very minimal.14 The residents are allowed to hone the skill of anastomosis with this procedure, before graduating to the difficult anastomosis. Hence, given a choice, the gastrojejunostomy should be chosen over the duodenojejunostomy. Retrograde duodenostomy is relatively easy to perform. A stab incision is made at the antimesenteric border of proximal jejunum. A 12F Ryle’s tube should be negotiated into the descending duodenum. The enterotomy can be closed with a purse-string suture. The other steps are similar to the FJ, which is routinely performed by the surgeons. The duodenostomy tube should be connected to a closed drainage system for continuous drainage for a few days to weeks, depending on the severity of the duodenal injury and condition of the patient. After it has served its purpose, it can be clamped. The patients should be observed for at least 48–72 hours to look for any complications like abdominal distension, pain and vomiting. In the absence of any further complications, the patients can be discharged safely. The RD should be removed after 4–6 weeks, once there is fistulisation of the tract.
We performed a Bilroth II or loop gastrojejunostomy reconstruction following antrectomy. An isoperistaltic segment of jejunum was brought in through an incision in the transverse mesocolon, and side-to-side anastomosis was performed with the posterior wall of the stomach. Although theoretically, Roux-en-Y gastrojejunostomy has fewer risks of bile reflux, a loop gastrojejunostomy is the method of choice in an emergency setting as it avoids multiple anastomoses.
FJ should be done 10–15 cm distal to the enterotomy made for reverse duodenostomy. FJ serves the purpose of a saviour in the case of proximal anastomotic leak. Feeding through FJ saves the patients from complications related to the parenteral nutrition and translocation of bacteria. We regretted later that FJ was not performed, which could have enhanced the recovery of the patient. Also, since this was an emergency surgery, and the patient’s situation was critical, the operating surgeon developed undue anxiety, which leads to intraoperative complications. This should be avoided, and an experienced surgeon should always be called for help as soon as possible.
So, the matter which could have been settled with adjustment of few sutures instead took a dramatic turn. Starting with the prolonged operative time, a risky anastomosis and an insecure duodenal stump, the misery of the patient continued in the postoperative period due to duodenal stump leak. Finally, it ended with a postoperative hospital stay of 26 days. Despite all these problems, the patient could go home just because the mishap was identified in quick seconds and was aptly managed.
Learning points.
Avoiding undue anxiety and early call for help are imperative for preventing intraoperative complications.
Ancillary procedures like reverse duodenostomy and feeding jejunostomy are unchartered weapons to subdue the impact of the catastrophic duodenal leak.
Surgical management of bleeding duodenal ulcer, although sounds like a child’s play, is not a plain sailing during a desperate scenario. The mere presence of an experienced surgeon with his junior colleague can be crucial to achieve fruitful outcomes.
Acknowledgments
The authors are thankful to Dr Hemanta Kumar Nayak and the Department of Gastroenterology for providing the details of endoscopic findings and timely referral of the patient for surgery.
Footnotes
Contributors: SM, PK, PKS and TSM were involved in the care of the patient. SM has worked up the patient and done the follow-up. All authors have contributed substantially to the collection of data, revision of the article and have given necessary intellectual inputs.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Mahajan P, Chandail VS. Etiological and endoscopic profile of middle aged and elderly patients with upper gastrointestinal bleeding in a tertiary care hospital in North India: a retrospective analysis. J -Life Health 2017;8:137–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Oakland K. Changing epidemiology and etiology of upper and lower gastrointestinal bleeding. Best Pract Res Clin Gastroenterol 2019;42-43:101610. 10.1016/j.bpg.2019.04.003 [DOI] [PubMed] [Google Scholar]
- 3.Jung K, Moon W. Role of endoscopy in acute gastrointestinal bleeding in real clinical practice: an evidence-based review. World J Gastrointest Endosc 2019;11:68–83. 10.4253/wjge.v11.i2.68 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hadzibulic E, Govedarica S. Significance of Forrest classification, Rockall’s and Blatchford’s risk scoring system in prediction of rebleeding in peptic ulcer disease. Acta Medica Median 2007;46:38–43. [Google Scholar]
- 5.Hwang JH, Fisher DA, Ben-Menachem T, et al. The role of endoscopy in the management of acute non-variceal upper Gi bleeding. Gastrointest Endosc 2012;75:1132–8. 10.1016/j.gie.2012.02.033 [DOI] [PubMed] [Google Scholar]
- 6.Chen ZJ, Freeman ML. Management of upper gastrointestinal bleeding emergencies: evidence-based medicine and practical consideration. World J Emerg Med 2011;2:5–12. 10.5847/wjem.j.1920-8642.2011.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gralnek I, Dumonceau J-M, Kuipers E, et al. Diagnosis and management of nonvariceal upper gastrointestinal hemorrhage: European Society of gastrointestinal endoscopy (ESGE) guideline. Endoscopy 2015;47:a1–46. 10.1055/s-0034-1393172 [DOI] [PubMed] [Google Scholar]
- 8.Laursen SB, Hansen JM, Schaffalitzky de Muckadell OB. The Glasgow Blatchford score is the most accurate assessment of patients with upper gastrointestinal hemorrhage. Clinical Gastroenterology and Hepatology 2012;10:1130–5. 10.1016/j.cgh.2012.06.022 [DOI] [PubMed] [Google Scholar]
- 9.Roy A, Kim M, Hawes R, et al. The clinical and cost implications of failed endoscopic hemostasis in gastroduodenal ulcer bleeding. United Eur Gastroenterol J 2017;5:359–64. 10.1177/2050640616663570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee CW, Sarosi GA. Emergency ulcer surgery. Surg Clin North Am 2011:91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tarasconi A, Coccolini F, Biffl WL, et al. Perforated and bleeding peptic ulcer: WSES guidelines. World J Emerg Surg 2020;15:3. 10.1186/s13017-019-0283-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wischmeyer PE. Nutrition therapy in sepsis. Crit Care Clin 2018;34:107–25. 10.1016/j.ccc.2017.08.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fujikuni N, Tanabe K, Yamamoto H, et al. Triple-tube-ostomy: a novel technique for the surgical treatment of iatrogenic duodenal perforation. Case Rep Gastroenterol 2011;5:672–9. 10.1159/000335742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jex RK, van Heerden JONA, Wolff BG, et al. Gastrointestinal anastomoses factors affecting early complications. Ann Surg 1987;206:138–41. 10.1097/00000658-198708000-00004 [DOI] [PMC free article] [PubMed] [Google Scholar]