Abstract
Background:
Wastewater testing offers a cost-effective strategy for measuring population disease prevalence and health behaviors. For COVID-19, wastewater surveillance addresses testing gaps and provides an early warning for outbreaks. As U.S. federal agencies build a National Wastewater Surveillance System around the pandemic, thinking through ways to develop flexible frameworks for wastewater sampling, testing, and reporting can avoid unnecessary system overhauls for future infectious disease, chronic disease, and drug epidemics.
Objectives:
We discuss ways to transform a historically academic exercise into a tool for epidemic response. We generalize lessons learned by a global network of wastewater researchers around validation and implementation for COVID-19 and opioids while also drawing on our experience with wastewater-based epidemiology in the United States.
Discussion:
Sustainable wastewater surveillance requires coordination between health and safety officials, utilities, labs, and researchers. Adapting sampling frequency, type, and location to threat level, community vulnerability, biomarker properties, and decisions that wastewater data will inform can increase the practical value of the data. Marketplace instabilities, coupled with a fragmented testing landscape due to specialization, may require officials to engage multiple labs to test for known and unknown threats. Government funding can stabilize the market, balancing commercial pressures with public good, and incentivize data sharing. When reporting results, standardizing metrics and contextualizing wastewater data with health resource data can provide insights into a community’s vulnerability and identify strategies to prevent health care systems from being overwhelmed. If wastewater data will inform policy decisions for an entire community, comparing characteristics of the wastewater treatment plant’s service population to those of the larger community can help determine whether the wastewater data are generalizable. Ethical protocols may be needed to protect privacy and avoid stigmatization. With data-driven approaches to sample collection, analysis, and interpretation, officials can use wastewater surveillance for adaptive resource allocation, pandemic management, and program evaluation. https://doi.org/10.1289/EHP8572
Introduction
In the United States, the COVID-19 pandemic has been exacerbated by an inability to track infections in a timely and comprehensive manner (Parodi and Liu 2020). Traditional testing has proven insufficient to monitor disease transmission, not only because of a lack of test kits (American Society for Microbiology 2020), but also because of a lack of symptoms in up to 70% of infected people (Byambasuren et al. 2020; Oran and Topol, 2020; Poletti et al. 2020). To address this gap, some states have turned to wastewater testing, which provides a scalable, cost-effective way to anonymously track population-level biomarkers of drug use, diseases, and infections like COVID-19. (Keshaviah 2017; Daughton 2020). Wastewater surveillance offers three unique strengths: a) broad population coverage that extends to nearly 80% of U.S. households (CDC 2020e); b) detection of infections among asymptomatic carriers who shed virus in their stools; and c) a lead time of 1–2 wks for changes in community-level infections compared with individual case data (Peccia et al. 2020; Randazzo et al. 2020).
When COVID-19 hit, several countries—including the Netherlands (Kitajima et al. 2020), Sweden (Hellmér et al. 2014; Saguti et al. 2021), Israel (Bogler et al. 2020), India (Kumar et al. 2020a, 2020b), Pakistan (Asghar et al. 2014), and Australia (Ahmed et al. 2020)—quickly pivoted existing wastewater surveillance systems designed to monitor illicit drugs and other viral pathogens to monitor for the novel coronavirus (Daughton 2020). Based on other countries’ early successes in detecting and quantifying the SARS-CoV-2 virus that causes COVID-19, hundreds of U.S. wastewater treatment plants across at least 42 states began testing their wastewater for SARS-CoV-2 (COVID-19 WBE Collaborative 2020b; NORMAN SCORE Database 2020). However, coordination across these treatment plants, standards for sample analysis, and harmonization of reporting metrics is needed, lest we end up with hundreds of individual pilot studies instead of a national public health surveillance system (Daughton 2021).
In recognition of this need, the Centers for Disease Control and Prevention (CDC) and the U.S. Department of Health and Human Services have begun developing a National Wastewater Surveillance System (NWSS), with cross-agency collaboration from the U.S. Environmental Protection Agency, U.S. Department of Homeland Security, and other partners (CDC 2020e). As agencies coordinate to build the NWSS from the ground up, thinking through the needs of not only COVID-19, but also of future infectious disease, chronic disease, and drug epidemics, can help ensure that the NWSS is not overly optimized for the current threat. Although COVID-19 has catalyzed the use of wastewater surveillance in the United States, the next epidemic threat may differ in important ways from the novel coronavirus. We seek to generalize lessons learned by a global network of wastewater researchers with respect to validation and implementation of wastewater surveillance for COVID-19 while also drawing on examples closer to home because the needs of a U.S. NWSS may differ from those of systems developed abroad. This is in part because the United States has a much larger and more geographically dispersed population, resulting in substantially more wastewater treatment plants across which to coordinate [e.g., the United States has roughly 15,000 plants (U.S. EPA 2016), compared with roughly 1,200 in Australia (Hill et al. 2012)]. Specifically, we leverage our experience translating wastewater data for opioid epidemic management (Margetts et al. 2020) to provide a roadmap for adapting wastewater testing for pandemic preparedness and response.
To transform what has historically been an academic exercise into an operationalized policy tool, that is, from retrospective analysis geared to a peer-reviewed publication to prospective monitoring that informs policy and program decisions, we see a need to consider a broad range of factors beyond optimizing laboratory assays, including how to translate wastewater surveillance methods and metrics to address the practical needs and timelines of policymakers. Key considerations include the following: a) How quickly can test results be returned? b) How reliable are the wastewater testing results, given fluctuations in temperature, rainfall, and flow? c) What thresholds should trigger policy actions such as expanding individual testing or tightening restrictions? And d) what agencies, institutions, and funding must come together to deploy surveillance in a sustainable way? Keeping these questions in mind during NWSS development can enhance the system so that it serves multiple uses, including providing an early warning for new health threats, assessing how health biomarker levels fluctuate over time, identifying hotspots of activity, and evaluating the effectiveness of policies, programs, or community actions.
Discussion
In this paper, we draw on our experience conducting wastewater-based epidemiology to discuss frameworks for adapting wastewater surveillance so that it can flexibly guide a data-driven epidemic management strategy. Ongoing coordination between local public health officials (and, for a drug epidemic, public safety officials), wastewater plant operators, testing labs, and the research community can better ensure that the data generated from wastewater surveillance meet community needs (Figure 1). Routine and rapid communication across these partners can inform where and when to collect samples, how to prioritize method validation to align with officials’ informational needs, and the types of data synthesis required to yield policy insights. Collaborative groups composed of experts from different disciplines and sectors could be useful for method harmonization. Existing organizations focused on water quality and water regulation, such as the Water Research Foundation (WRF) and the National Association of Clean Water Agencies, could help scale implementation nationwide. Below, we discuss strategies to optimize sampling, testing, and reporting to enable officials to identify and respond to broad public health threats.
Figure 1.
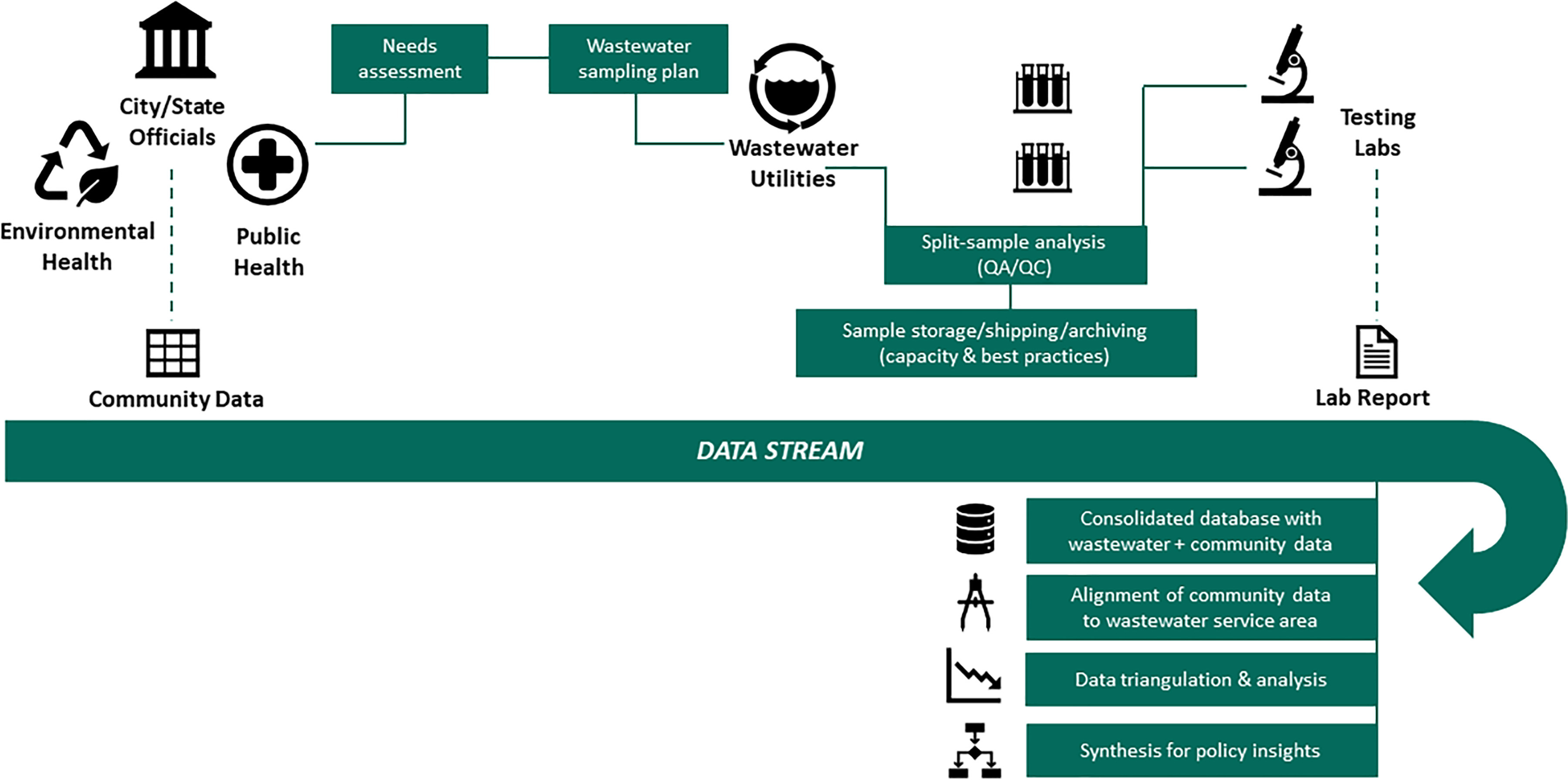
Conceptual framework for a coordinated, broad wastewater surveillance system. The figure highlights the different entities involved in surveillance for public health; the goals of coordination across these entities during planning, implementation, and reporting; and the data alignment and synthesis needs for policymaking. Note: QA, quality assurance; QC, quality control.
Adapting Sample Collection to Community Needs
A longstanding challenge of wastewater sampling has been how to collect a sample that is representative (Polo et al. 2020). In our view, representativeness should consider both the technical properties of the biomarker in the wastewater stream (e.g., how it degrades as it flows though the sewer), as well as whether the sample was collected in a manner that covers the population of interest. This concern is particularly salient when collecting point-in-time grab samples, rather than timed or flow-weighted composite samples (Polo et al. 2020). For COVID-19, collecting samples in the morning (when most people routinely use the bathroom) may help capture a larger viral load. However, some suggest that larger flows in the morning could result in a more diluted signal (WRF 2020), and morning sampling might also overlook certain underrepresented populations, such as shift workers, who may be more susceptible to COVID-19 and exacerbating chronic conditions (Lim et al. 2020). The choice of sampling day also has ramifications for population capture. When monitoring drug use, weekday sampling is more likely to capture people with habitual drug use, whereas weekend sampling could additionally reflect recreational use. Last, representativeness is inherently diminished because wastewater testing misses roughly 20% of U.S. households served by septic systems (CDC 2020e). It may be possible to partially mitigate this data bias analytically, using spatial imputation and predictive modeling (e.g., predicting wastewater biomarker concentrations for areas served by septic systems based on community demographics and other characteristics). However, such methods are imperfect and work best to characterize broad trends—not to pinpoint specific populations where resources may need to be concentrated during a public health threat.
In its regularly updated online guide, the CDC recommends considering public health data needs and the capacity for wastewater sampling and testing when developing a wastewater sampling strategy (CDC 2020d). In addition, we believe that a wastewater sampling plan should be flexibly adapted to the threat level, how vulnerable communities and health care systems are to that threat, the properties of the health biomarker of interest, and the types of health and policy decisions that wastewater data will inform. With respect to threat level and vulnerability, researchers can refer to the CDC’s Pandemic Intervals Framework, which classifies communities into one of four phases of pandemic progression: initiation, acceleration, deceleration, and preparation for future (CDC 2016). At different phases, officials might prioritize different sampling strategies, targets, and designs (Figure 2). For example, during initiation, when community transmission is low, detection at the central wastewater treatment plant can be challenging because of diluted signals. Instead, following Israel’s model of wastewater surveillance for polio pandemic management, sampling might prioritize upstream sentinel sites to provide an early warning (Manor et al. 2014), along with the top 5% of wastewater treatment plants, which together cover more than half of the U.S. population (U.S. EPA 2016). Here, the choice of composite vs. grab sampling has important tradeoffs. Composite sampling yields better representativeness and improves detectability, but it may not be feasible because of cost constraints (auto-samplers cost $5,000 to $10,000 each) or practical considerations (e.g., if auto-samplers do not fit into manholes targeting specific facilities). Instead, grab sampling can provide a cost-effective way to collect preliminary information on threat presence or absence with less delay (Betancourt et al. 2020). As transmission accelerates, testing might shift from detection to quantification, and sampling might shift to target vulnerable populations where the need for care could outweigh capacity. For COVID-19, vulnerable communities include residents of nursing homes, correctional facilities, and universities and communities with a high prevalence of chronic conditions associated with severe COVID-19 (CDC 2020d; Marvel et al. 2020). Prioritizing vulnerable communities could catch upticks in transmission early and prevent health care systems from being overwhelmed. As transmission begins to decelerate, sampling might prioritize key transmission nodes, such as international airports, shipping ports, travel hubs, and public gathering spaces (Polo et al. 2020), along with threat-specific surveillance sites, such as population centers and areas with dense living (for an infectious disease) or known trafficking routes (for a drug epidemic). Finally, when the public health threat has waned, communities can scale back sampling to once again focus on qualitative detection at sentinel sites. Because a community’s pandemic phase can change quickly, close monitoring of wastewater data is vital. However, intensive sampling may not be feasible nor realistic during a prolonged epidemic. Instead, communities might tailor sampling frequency to their general risk profile, intensifying sampling as risk increases. That risk could be determined through epidemiologic modeling, which can help determine how quickly 1 case will erupt into 10 cases or 100.
Figure 2.
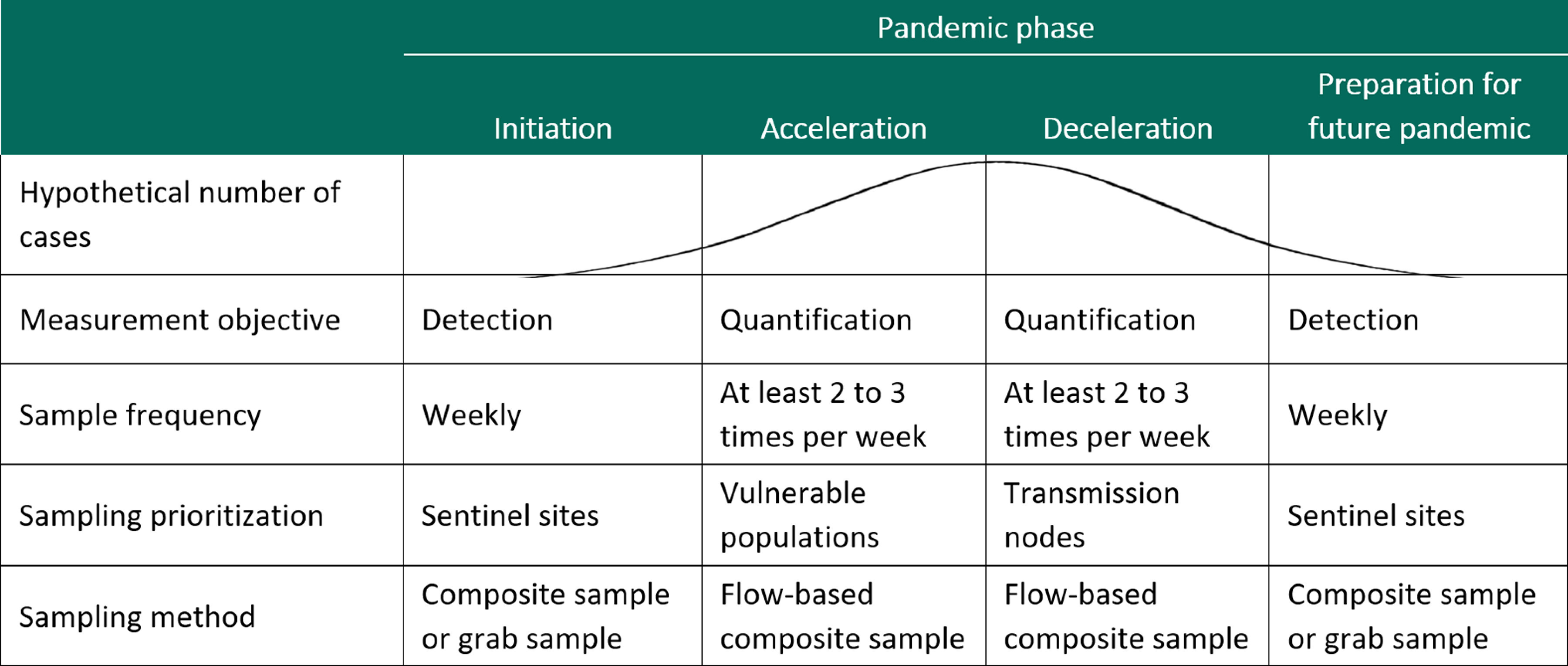
Design considerations for wastewater surveillance in communities at different stages of a pandemic. The figure provides recommendations for sampling frequency, location, and type during different phases of disease threat prevalence, defined using the Pandemic Intervals Framework from the Centers for Disease Control and Prevention.
Beyond a community’s pandemic phase, biomarker properties may also influence sample type and location (Polo et al. 2020). For biomarkers like SARS-CoV-2 that are shed in the stool rather than urine, sludge samples may yield more concentrated viral loads than wastewater samples (Peccia et al. 2020). For biomarkers that are unstable in the wastewater matrix, sampling upstream of the central plant could lead to improved detection by reducing biomarker travel time and thus degradation in the wastewater (Kocamemi et al. 2020; McCall et al. 2016). Upstream sampling may be easier in some communities than others—for example, in communities with pumping stations that collect wastewater from subbasins (Polo et al. 2020). When sampling upstream or in rural communities, researchers and officials should pay special attention to protecting the privacy of small segments of the communities surveilled and avoid stigmatizing specific groups. Although wastewater testing, as a population surveillance tool, does not yield information on specific individuals, fears of stigmatization at the municipal level have historically hampered use of this tool in the United States to tackle the opioid epidemic (Keshaviah 2017). Even in the current pandemic, some ethnic groups and those with certain health conditions have been stigmatized (Roberto et al. 2020), and targeted upstream sampling could exacerbate discrimination. Developing an ethical framework can be helpful (Keshaviah 2017), as can moving from a spotlight sampling approach, focused on a few communities, to broad surveillance in an entire region. For privacy protection, most initiatives for SARS-CoV-2 are focusing on sampling wastewater from treatment plants that serve at least 500 people (EMCDDA 2016).
Last, officials may need to tailor wastewater sampling based on how they plan to use the resulting data. If wastewater surveillance will inform adaptive resource allocation, then a sampling plan should be designed to identify local hot spots. In this case, samples could be collected less frequently over time but would need to include broad spatial coverage in a region. If wastewater data are used to evaluate the implementation or impact of a particular policy, program, or community action, such as how marijuana legalization affects marijuana use, whether vaccine uptake is high enough to reduce circulating SARS-CoV-2 viral levels (Smith et al. 2021), or whether expanded naloxone distribution reduces opioid overdose rates, then wastewater sampling would ideally include not only the target community, but also a comparison community not subject to the policy, program, or action being studied but with similar characteristics to the target community. Including a comparison community within a quasi-experimental evaluation framework can help control for external confounding and strengthen the case for a causal association between the outcome of interest and the policy, program, or action (Campbell and Stanley 2015).
Optimizing Testing to Monitor for Multiple Threats
When a new public health threat emerges, a consistent set of validation needs arise with respect to detecting and quantifying that threat in the wastewater matrix (Polo et al. 2020). There may be multiple biomarker targets that can be quantified, each with different properties. To identify the optimal target, researchers can examine the stability of each biomarker to understand degradation rates as a function of travel time in the sewer, temperature, heat, ultraviolet light, sunlight, and other environmental parameters (Chen et al. 2014; Hart and Halden 2020; Zhan et al. 2020). Detailed modeling of viral decay and wastewater travel time from its point of origin to sampling location would also help translate the viral load in a wastewater sample to an estimate of COVID-19 prevalence in the service population, which is still a work in progress (Larsen and Wigginton 2020). Validation would also require assessing the influence of different sample preparation and processing procedures, including filtering, pasteurization, and freezing, on biomarker detection (Jafferali et al. 2021).
Once a biomarker is chosen, and labs begin optimizing their analytic methods, they are likely to encounter tradeoffs between method precision and ease or speed of implementation. During this process, we recommend they consider the time and intensity required for different analytic approaches, as well as the practicalities of implementation, such as the burden placed on utility staff to follow sample preprocessing procedures like on-site pasteurization. As a leading indicator, wastewater data can buy valuable time for public health officials to make tailored decisions about how to manage the pandemic. But lab reporting delays eat into those lead-time advantages (Larsen and Wigginton 2020). To move from retrospective data analysis to near real-time decision making, officials will need lab results quickly. In our experience, many academic labs exploring cutting-edge approaches for wastewater testing for SARS-CoV-2 currently lack the capacity and rapid turnaround needed for policymaking, compared with their commercial counterparts.
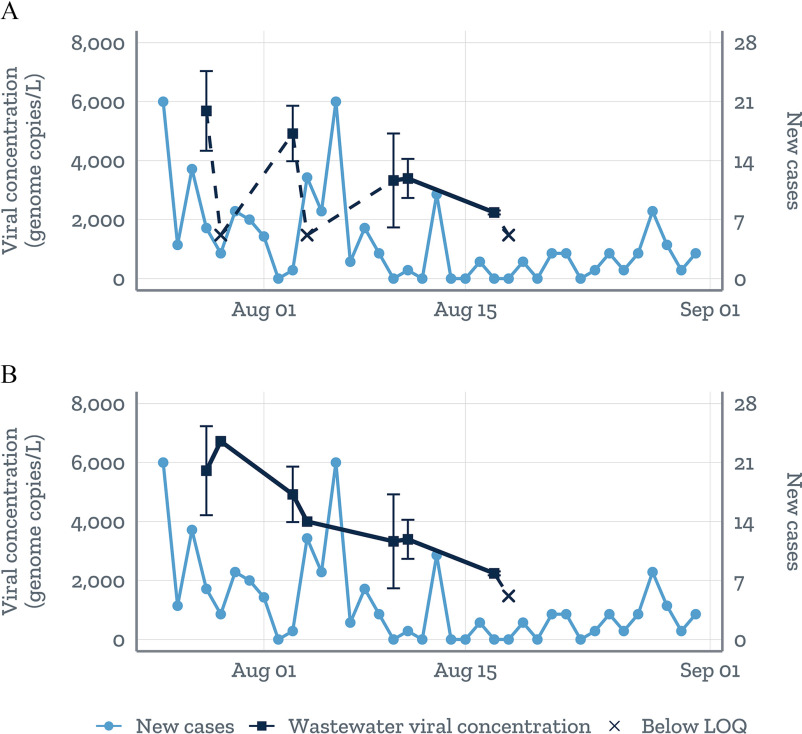
As more and more labs begin implementing new protocols for a novel public health threat, interlab comparisons are essential and can be facilitated by identifying early on during method development how best to normalize and standardize reporting metrics (Pecson et al. 2020; Pérez-Cataluña et al. 2021). A review of the literature shows that the most common unit of reporting for SARS-CoV-2 is viral RNA copies per liter of wastewater. But the specific assays used to quantify viral concentrations may differ from study to study: Across the 35 publications included in a list from the COVID-19 WBE Collaborative (2020a), more than 10 different assays were used. Using more than one target region of the SARS-CoV-2 genome can achieve better quantification of the virus (Polo et al. 2020), but it also creates difficulties comparing results across studies and complicates standardized reporting to the NWSS. Even when assay use is consistent, variations in data summarization methods could generate different results, leading to confusion for officials trying to interpret the data. For example, in a pilot study we led to assess the feasibility of wastewater testing for SARS-CoV-2 in rural western North Carolina, preliminary results suggested that wastewater trends in SARS-CoV-2 viral levels correlated with trends in COVID-19 case counts observed 8–10 d later (Keshaviah et al. 2020). Yet alignment across back-to-back days of timed 24-h composite samples, and the resulting trend line over the 4-wk sampling period, varied depending on how the data were summarized (Figure 3). The lab originally considered the viral concentration (the average of N1 and N2 viral copies per liter across four replicates) as being below the limit of quantification (LOQ; 2,200–3,530 viral copies per liter, depending on the testing procedure) if all replicates for the N1, N2, or both assays had two or fewer positive droplets (regardless of the number of viral copies per liter). With this approach to summarization, a nonspecialist might conclude that there was poor alignment between the back-to-back sampling days (Figure 3A). When we removed that threshold criterion and reported the average viral copies per liter so long as at least one N1 and N2 replicate had a value above the LOQ, alignment between the back-to-back days of sampling improved (Figure 3B). This adjustment also altered the shape of the trend line somewhat. Beyond assay and metric standardization, researchers may also need to normalize their results to account for variability in wastewater flow rates over time, the number of people contributing to the wastewater using different human biomarkers, and uncertainties around rates of SARS-CoV-2 excretion in stools, all of which could render data from different locations not directly comparable (Chen et al. 2014; He et al. 2020; Polo et al. 2020). Absent this standardization, though, wastewater data can still be used to reliably monitor temporal trends for a given wastewater treatment plant so long as the data are generated by the same lab, using the same protocols (Medema et al. 2020).
Figure 3.
Mean SARS-CoV-2 viral RNA copy numbers per liter of wastewater the standard error (Keshaviah et al. 2020). The mean is based on the average of the N1 and N2 viral copy numbers per liter across four replicates on a given day. Values below the limit of quantification (LOQ) were plotted at the midpoint between the LOQ and the limit of detection (LOD); dashes were used to denote the uncertainty in the shape of the trend line when connecting to a value below the LOQ. The panels show how the shape of the wastewater trend line varies when using two different reporting metrics. (A) Mean concentrations were considered below the LOQ if all replicates for N1, N2, or both had positive droplets, regardless of the viral copy numbers per liter. (B) No such threshold was applied, and values were reported so long as at least one N1 or N2 replicate a value above the LOD.
Besides lab-based methods, a broader set of epidemiologic and statistical approaches can validate and optimize wastewater data for a new threat (NIST 2020). Analyzing trends in wastewater data alongside clinical case surveillance data can be useful for validation and interpretation (CDC 2020c). However, because confirmed cases may represent only a fraction of total cases, missing asymptomatic patients and those not seeking care (Mor et al. 2014), other epidemiologic indicators and novel proxies should also be considered, including online symptom reports and digital thermometer data that, like wastewater data, may be leading indicators (Kogan et al. 2020). Researchers can also use meta-analysis and systematic reviews to assess how sample preparation methods and other study-level factors impact quantification, or how different normalization approaches impact measure variability. Care should be taken, however, to vet the studies analyzed. Since the beginning of the pandemic, the use of preprint servers like bioRxiv and medRxiv has skyrocketed [representing nearly 40% of English-language COVID-19 scientific work disseminated between January and April 2020 (Fraser et al. 2020)]. Although preprint platforms facilitate rapid dissemination and knowledge building, they can be challenging to navigate because they may include studies with questionable methods or lower levels of rigor. Rapid Reviews: COVID-19 (https://rapidreviewscovid19.mitpress.mit.edu/), the Novel Coronavirus Research Compendium (https://ncrc.jhsph.edu/), and similar efforts to quickly and authoritatively call out misinformation and highlight valuable research are critical to support public health action.
To scale wastewater surveillance nationally, the marketplace for testing should be considered. In our experience, the rapid spike in demand created by the COVID-19 pandemic currently outweighs capacity in many locations. To meet demand, many academic researchers have spun off their wastewater testing activities into commercial startups. This destabilization of the supply of testing services has led to harmful entrepreneurial overpromising. Some city officials who were promised near real-time information on community COVID-19 risk through wastewater testing were left waiting weeks, not days, for test results (Brasileiro 2020). Further, in recent months we have seen one academic lab decrease their testing price by up to 30% as a result of method optimization, whereas another lab—a commercial start-up—increased their prices 10-fold, prompting many public health departments to search out more cost-effective options midstream. From our recent experience, per-sample testing costs range from about $300 to $1,200 for SARS-CoV-2 and $100 to $150 for drug metabolite panels (which are less time-intensive than viral detection and quantification). Although both academic and commercial labs currently offer testing for SARS-CoV-2, few commercial labs have protocols in place to quantify drug metabolites in wastewater. As with all new technologies, costs should stabilize over time as supply ramps up to meet demand. But government investments and oversight may still be needed to ensure that commercial pressures do not undermine public health. For example, small business technology transfer grants, such as the Smart City Tools grants from the National Institute on Drug Abuse (2019), have been a useful funding mechanism because they incentivize knowledge transfer from academic to commercial labs, helping to reconcile the gap that sometimes exists between innovation and implementation.
Finally, the fragmented testing landscape created by scientific specialization presents a challenge for developing the NWSS. There is little overlap between the labs testing for disease pathogens and those testing for drug metabolites. Accordingly, officials will need to involve new sets of players for new types of threats. To flexibly monitor for a wide range of biomarkers or to study interactive effects (such as how the pandemic is exacerbating drug use), a mapped roster reflecting each lab’s testing targets, methods, capacity, and turnaround time would enable efficient coordination. Such a roster could also flag labs with the capacity for intensive, nontargeted testing to identify unknown or unexpected public health threats (O’Brien and Xagoraraki 2019). Expanding the testing protocols at existing state labs, and improving coordination across labs in the CDC’s Laboratory Response Network [composed of federal, state, and local public health laboratories that respond to biological, chemical, and other public health emergencies (CDC 2020a)], would enhance cross-region comparisons. Furthermore, strategically locating some sentinel labs near central wastewater treatment plants could help stem degradation of the target compound in the wastewater matrix during sample transport and thus avoid the need for sample preservation techniques that might damage the pathogen or weaken detection.
Contextualizing Wastewater Data for Policy Insights
Importantly, because wastewater data cannot identify who is infected, surveillance using wastewater testing is not meant to replace traditional disease surveillance focused on individual cases and their contact histories. Instead, it should supplement data from traditional epidemiological routes, such as syndromic surveillance, to help overcome known limitations due to low population coverage (particularly of asymptomatic cases), high cost, testing and reporting delays (Polo et al. 2020; Sodré et al. 2020), and inaccuracies due to location misattribution (Calvert County Health Department 2020; Wang et al. 2020). The European Monitoring Center for Drugs and Drug Addiction (EMCDDA 2016) has compared the pros and cons of wastewater-based epidemiology with more traditional epidemiological approaches in the context of tracking the opioid epidemic. But that comparison may not hold up for COVID-19, which presents some unique challenges. For example, whereas wastewater concentrations of drug metabolites can yield reliable estimates of per capita drug use that take into account average drug dose sizes and excretion rates, COVID-19 viral shedding is much more variable, and more research is needed on the prevalence, timing, and duration of viral shedding before wastewater data can yield reliable estimates of COVID-19 prevalence (Hart and Halden 2020).
To realize the full value of wastewater data for policymaking, wastewater data should be integrated with administrative data during sample plan development, testing validation, and result interpretation. Been et al. (2015) illustrated this approach to data triangulation when testing wastewater for opioids in Switzerland. By combining wastewater data with population survey data, a registry on opioid substitution therapy, and data from a needle and syringe distribution program, they were able to parse illicit heroin use from prescription opioid use. Likewise, in our analyses of data from a multisite wastewater study in Montana, by aligning and synthesizing wastewater concentrations of opioid and illicit drug metabolites with local data on pharmacy prescriptions filled, law enforcement drug seizures, and calls placed to emergency medical services for drug overdoses, we were able to generate insights into where and when drug overdoses may occur, policing impact on community drug use, and the extent of black-market activity (Margetts et al. 2020). Although wastewater data cannot identify health outcomes among specific individuals, the risk of identifying individuals increases when wastewater data are aligned and linked with complementary data sources, particularly if the health outcome is rare. Accordingly, public health officials and wastewater researchers should implement protocols to protect individuals’ privacy and avoid community stigmatization (Polo et al. 2020).
To scale data integration for the NWSS, investments in new data pipelines, interoperable databases, and automated reporting tools may be needed. For example, interactive, dynamic data dashboards can help officials see, in real time, how wastewater results vary when they select a different sampling location, time range, or biomarker of interest. Dashboards can also be built to automatically link and align data from multiple sources and visualize the information in a manner that facilitates data triangulation and prediction. In our North Carolina pilot study, we built a dashboard that brought together information on COVID-19 testing rates, case counts, pandemic vulnerability, population mobility, and other community demographics (Figure 4). By providing a more holistic picture of COVID-19 risk in a county, including the vulnerability of the population and health care system, such a dashboard can give public health officials the confidence to act on this relatively new data source and adapt their decisions to changing profiles of community risk.
Figure 4.
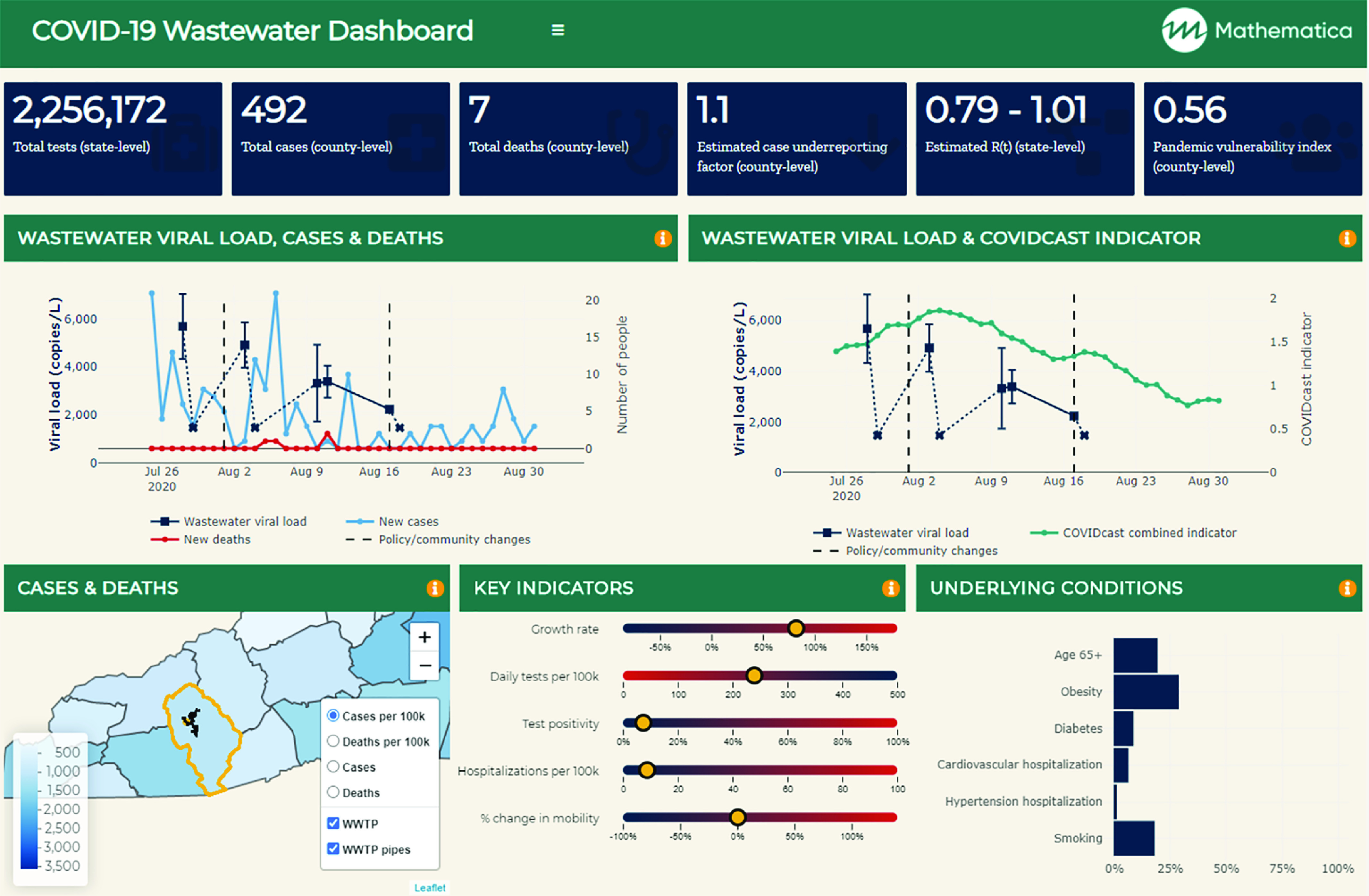
A wastewater communication dashboard to contextualize sewage concentration data for policymaking. The dynamic tool uses a series of application programming interface calls to state- and county-level data to facilitate rapid data refreshing and reporting. Two trend graphs contextualize the wastewater data with data on the 3-day rolling average of new cases and a proxy indicator of COVID-19 prevalence that combines doctor visits and Facebook symptom surveys. A series of visual displays highlight key region-specific indicators, including case growth rate, test positivity, hospitalizations, population mobility, and population vulnerability based on proportion of the community with underlying conditions associated with severe COVID-19. The dashboard synthesizes wastewater lab results with data from the USAFacts (https://usafacts.org/data/), University of Washington (https://github.com/COVID19StatePolicy/SocialDistancing), the Delphi Research Group’s COVIDCast effort (https://cmu-delphi.github.io/delphi-epidata/api/covidcast.html), the COVID Tracking Project (https://covidtracking.com/), the National Institute of Environmental Health Sciences (https://github.com/COVID19PVI/data), the Centers for Disease Control and Prevention (https://www.cdc.gov/brfss/annual_data/annual_2018.html), Google (https://www.google.com/covid19/mobility/), and the U.S. Census (https://www2.census.gov/programs-surveys/popest/datasets/2010-2019/counties/totals/co-est2019-alldata.csv). Note: WWTP, Wastewater treatment plant.
There are many practical use cases that wastewater testing can inform, particularly when combined with administrative data. Comparing wastewater results with data on testing coverage and test positivity can help officials see where gaps may exist in individual testing. In other words, aggregate testing can inform when individual testing should be ramped up. Comparing wastewater data with health care resource data (such as intensive care unit bed capacity in local hospitals) can help assess the community’s pandemic vulnerability and inform whether restrictive measures like shutdowns are needed to prevent health care systems from being overwhelmed. In addition, comparing wastewater viral loads with population vaccination rates can help identify areas where vaccine hesitancy may inhibit herd immunity (Smith et al. 2021). Ultimately, if wastewater data collected on a subpopulation will inform policy decisions that affect the larger community, it will be critical to compare demographic data on the sampled population vs. the community as a whole (including those on septic systems), to assess the extent to which the information generated by wastewater surveillance can be generalized.
Looking Ahead
Epidemics present a rare mix of collective outlook, desire for data, and temporary bureaucratic abatement. Historically, such crises have catalyzed the development of new infrastructure (Asghar et al. 2014; Chatterjee et al. 2013; Roberts 2013). With sewage surveillance, we can tap into a data stream that is right beneath our feet for a continually running system to measure population health. A unique strength of wastewater testing is the efficiency with which it can measure multiple public health biomarkers and their interactive effects. Wastewater testing has been used to characterize everything from exposure to pathogenic viruses and bacteria to health behaviors (such as drug use and smoking) to chronic disease prevalence and biomarkers of stress (Choi et al. 2018). It has been used to assess polio vaccine coverage in India (Chatterjee et al. 2013), eradicate polio in Israel (Roberts 2013), and combat infectious diseases like Hepatitis A in Sweden (Hellmér et al. 2014; Roberts 2013). Further, researchers in Australia are now looking to wastewater testing to provide more accurate census estimates (Choi et al. 2020). Given the multilayered bits of information embedded in this data stream, there is high potential value for building a U.S. NWSS in a manner that is generalizable and need not be retooled for each new pathogen or public health threat.
Critically, wastewater surveillance could also help us overcome systemic psychological biases that hinder our ability to react to new threats. Humans’ innate cognitive biases prioritize inaction (status quo) over action and focus our attention on immediate known problems over future unseen—or unexperienced—threats (Johnson and Levin 2009). With routine wastewater surveillance, officials can detect unexpected health threats before they evolve into epidemics. Because it may not be feasible to deploy wastewater surveillance at all 15,000 wastewater treatment plants across the country, we need to strategically identify the most important locations for sampling while also ensuring that underserved and vulnerable populations are being monitored. If we implement the NWSS correctly, we can equip officials with objective, granular, and near real-time data on changing public health risks in a localized manner that is tailored to a community.
One hundred fifty years ago, our understanding of how diseases spread leaped forward when two British pioneers took a systematic view of raging disease epidemics. During the Crimean War, nurse Florence Nightingale’s use of data visualization to tally causes of death among British soldiers revealed that bacterial infections from poor sanitation led to 10 times more deaths than battle wounds (Hammer 2020). Around the same time, physician John Snow mapped clusters of cholera infections around the Broad Street water pump to show that a tainted water source, not bad air, was the source of London’s cholera epidemic (Snow 1855). Taking lessons from those who battled pandemics in centuries past, we see the need to take a big, bold approach to adapting and advancing our infrastructure for disease surveillance now, while the crisis window is open. If we do so strategically, with a view toward the next epidemic, we may revolutionize public health once more.
Acknowledgments
We thank the community partners, funders, and researchers who contributed to the wastewater studies we highlight here. The western North Carolina pilot study was conducted in partnership with S. Carraway at the Jackson County Department of Public Health, R. Mau and D. Manring at the Tuckaseigee Water and Sewer Authority, and S. McLellan at the University of Wisconsin-Milwaukee, with partial funding provided by the Dogwood Health Trust. The Montana wastewater study was led by D. Keil and M. Margetts from Montana State University, who worked with local officials to obtain pharmacy, law enforcement, and emergency medical services data; partial funding for the comparative analyses came from the National Institute of General Medical Sciences of the National Institutes of Health under award no. U54GM115371. Together, our partners contributed insights into policy needs and fostered cross-agency relationships that facilitated data sharing and wastewater-based epidemiology.
References
- Ahmed W, Angel N, Edson J, Bibby K, Bivins A, O’Brien JW, et al. . 2020. First confirmed detection of SARS-CoV-2 in untreated wastewater in Australia: a proof of concept for the wastewater surveillance of COVID-19 in the community. Sci Total Environ 728:138764, PMID: 32387778, 10.1016/j.scitotenv.2020.138764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Society for Microbiology. 2020. Supply shortages impacting COVID-19 and non-COVID testing. https://asm.org/Articles/2020/September/Clinical-Microbiology-Supply-Shortage-Collecti-1 [accessed 22 December 2020].
- Asghar H, Diop OM, Weldegebriel G, Malik F, Shetty S, El Bassioni L, et al. . 2014. Environmental surveillance for polioviruses in the Global Polio Eradication Initiative. J Infect Dis 210(suppl 1):S294–S303, PMID: 25316848, 10.1093/infdis/jiu384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Been F, Benaglia L, Lucia S, Gervasoni JP, Esseiva P, Delémont O. 2015. Data triangulation in the context of opioids monitoring via wastewater analyses. Drug Alcohol Depend 151:203–210, PMID: 25869541, 10.1016/j.drugalcdep.2015.03.022. [DOI] [PubMed] [Google Scholar]
- Betancourt WQ, Schmitz BW, Innes GK, Prasek SM, Brown KMP, Stark ER, et al. . 2021. COVID-19 containment on a college campus via wastewater-based epidemiology, targeted clinical testing and an intervention. Sci Total Environ 779:146408, PMID: 33743467, 10.1016/j.scitotenv.2021.146408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bogler A, Packman A, Furman A, Gross A, Kushmaro A, Ronen A, et al. . 2020. Rethinking wastewater risks and monitoring in light of the COVID-19 pandemic. Nat Sustain 3(12):981– 910, 10.1038/s41893-020-00605-2. [DOI] [Google Scholar]
- Brasileiro A. 2020. Testing poop to predict Miami’s COVID-19 trends? It’s been a messy process so far. Miami Herald, online edition. 20 November 2020. https://www.miamiherald.com/news/coronavirus/article247258864.html [accessed 13 January 2021].
- Byambasuren O, Cardona M, Bell K, Clark J, McLaws ML, Glasziou P. 2020. Estimating the extent of true asymptomatic COVID-19 and its potential for community transmission: systematic review and meta-analysis. JAMMI. 10.3138/jammi-2020-0030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calvert County Health Department. 2020. New policy to list COVID-91 deaths on our websites. https://www.calvertcountycovid19.com/post/new-policy-to-list-covid-19-deaths-on-our-websites [accessed 14 October 2020].
- Campbell DT, Stanley JC. 2015. Experimental and Quasi-Experimental Designs for Research. Houghton Mifflin Company, Boston, USA. [Google Scholar]
- CDC (Centers for Disease Control and Prevention). 2016. Figure 1. Preparedness and response framework for novel influenza A virus pandemics: CDC intervals. https://www.cdc.gov/flu/pandemic-resources/national-strategy/intervals-framework.html [accessed 30 July 2020].
- CDC. 2020a. Laboratory Response Network (LRN): partners in preparedness. https://emergency.cdc.gov/lrn/index.asp [accessed 14 January 2021].
- CDC. 2020b. People with certain medical conditions. https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/people-with-medical-conditions.html [accessed 14 January 2021].
- CDC. 2020c. Public health interpretation and use of wastewater surveillance data. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/wastewater-surveillance/public-health-interpretation.html [accessed 3 March 2021].
- CDC. 2020d. Developing a wastewater surveillance sampling strategy. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/wastewater-surveillance/developing-a-wastewater-surveillance-sampling-strategy.html#anchor_1602855374139 [accessed 14 January 2021].
- CDC. 2020e. National Wastewater Surveillance System (NWSS): a new public health tool to understand COVID-19 spread in a community. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/wastewater-surveillance.html [accessed 14 January 2021].
- Chatterjee A, Vidyant S, Dhole TD. 2013. Polio eradication in India: progress, but environmental surveillance and vigilance still needed. Vaccine 31(9):1268–1275, PMID: 23306357, 10.1016/j.vaccine.2012.12.060. [DOI] [PubMed] [Google Scholar]
- Chen C, Kostakis C, Gerber JP, Tscharke BJ, Irvine RJ, White JM. 2014. Towards finding a population biomarker for wastewater epidemiology studies. Sci Total Environ 487:621–628, PMID: 24300482, 10.1016/j.scitotenv.2013.11.075. [DOI] [PubMed] [Google Scholar]
- Choi PM, O’Brien JW, Tscharke BJ, Mueller JF, Thomas KV, Samanipour S. 2020. Population socioeconomics predicted using wastewater. Environ Sci Technol Lett 7(8):567–572, 10.1021/acs.estlett.0c00392. [DOI] [Google Scholar]
- Choi PM, Tscharke BJ, Donner E, O’Brien JW, Grant SC, Kaserzon SL, et al. . 2018. Wastewater-based epidemiology biomarkers: past, present and future. Trends Analyt Chem 105(August):453–469, 10.1016/j.trac.2018.06.004. [DOI] [Google Scholar]
- COVID-19 WBE Collaborative. 2020a. COVID-19 WBE Publication Map. https://www.covid19wbec.org/publication-map [accessed 14 January 2021].
- COVID-19 WBE Collaborative. 2020b. COVID19Poops Dashboard. https://www.covid19wbec.org/covidpoops19 [accessed 14 January 2021].
- Daughton CG. 2020. Wastewater surveillance for population-wide Covid-19: the present and future. Sci Total Environ 736:139631, PMID: 32474280, 10.1016/j.scitotenv.2020.139631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daughton C. 2021. Wastewater-based epidemiology: a 20-year journey may pay off for Covid-19. https://www.statnews.com/2021/01/07/wastewater-based-epidemiology-20-year-journey-pay-off-for-covid-19/ [accessed 14 January 2021].
- EMCDDA (European Monitoring Center for Drugs and Drug Addiction). 2016. Assessing Illicit Drugs in Wastewater: Advances in Wastewater-Based Drug Epidemiology. Castiglioni S, ed. Insights 22. Luxembourg: Publications Office of the European Union. http://www.emcdda.europa.eu/system/files/publications/2273/TDXD16022ENC_4.pdf [accessed 20 April 2017]. [Google Scholar]
- Fraser N, Brierley L, Dey G, Polka JK, Pálfy M, Coates JA. 2020. Preprinting a pandemic: the role of preprints in the COVID-19 pandemic. bioRxiv. Preprint posted online 23 May 2020. https://www.biorxiv.org/content/biorxiv/early/2020/05/23/2020.05.22.111294.full.pdf [accessed 1 April 2021]. [Google Scholar]
- Hammer J. 2020. The defiance of Florence Nightingale. Smithsonian Magazine. March 2020. https://www.smithsonianmag.com/history/the-worlds-most-famous-nurse-florence-nightingale-180974155/ [accessed 11 October 2020].
- Hart OE, Halden RU. 2020. Computational analysis of SARS-CoV-2/COVID-19 surveillance by wastewater-based epidemiology locally and globally: feasibility, economy, opportunities and challenges. Sci Total Environ 730:138875, PMID: 32371231, 10.1016/j.scitotenv.2020.138875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He X, Lau EHY, Wu P, Deng X, Wang J, Hao X, et al. . 2020. Temporal dynamics in viral shedding and transmissibility of COVID-19. Nat Med 26(5):672–675, PMID: 32296168, 10.1038/s41591-020-0869-5. [DOI] [PubMed] [Google Scholar]
- Hellmér M, Paxéus N, Magnius L, Enache L, Arnholm B, Johansson A, et al. . 2014. Detection of pathogenic viruses in sewage provided early warnings of hepatitis A virus and norovirus outbreaks. Appl Environ Microbiol 80(21):6771–6781, PMID: 25172863, 10.1128/AEM.01981-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hill R, Carter L, Kay R. 2012. Wastewater treatment facilities. Modified 30 August 2016. Symonston, Australian Capital Territory, Australia: Geoscience Australia. https://researchdata.edu.au/wastewater-treatment-facilities/1278436 [accessed 1 April 2021]. [Google Scholar]
- Jafferali MH, Khatami K, Atasoy M, Birgersson M, Williams C, Cetecioglu Z. 2021. Benchmarking virus concentration methods for quantification of SARS-CoV-2 in raw wastewater. Sci Total Environ 755(pt 1):142939, PMID: 33121776, 10.1016/j.scitotenv.2020.142939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson D, Levin S. 2009. The tragedy of cognition: psychological biases and environmental inaction. Curr Sci 97(11):1593–1603.https://www.jstor.org/stable/24107300 [accessed 1 April 2021]. [Google Scholar]
- Keshaviah A, ed. 2017. The Potential of Wastewater Testing for Public Health and Safety. Washington, DC: Mathematica Policy Research. https://cipre.mathematica-mpr.com/-/media/publications/pdfs/health/2017/wastewatertesting_specialreport.pdf [accessed 1 April 2021]. [Google Scholar]
- Keshaviah A, Hu C, Henry M. 2020. COVID-19 Risk in Rural North Carolina. Washington, DC: Mathematica Policy Research. https://www.mathematica.org/our-publications-and-findings/publications/covid-19-risk-in-rural-north-carolina [accessed 12 January 2021]. [Google Scholar]
- Kitajima M, Ahmed W, Bibby K, Carducci A, Gerba CP, Hamilton KA, et al. . 2020. SARS-CoV-2 in wastewater: state of the knowledge and research needs. Sci Total Environ 739:139076, PMID: 32758929, 10.1016/j.scitotenv.2020.139076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kocamemi BA, Kurt H, Hacioglu S, Yarali C, Saatci AM, Pakdemirli B. 2020. First data-set on SARS-CoV-2 detection for Istanbul wastewaters in Turkey. medRxiv. Preprint posted online 6 May 2020. https://www.medrxiv.org/content/10.1101/2020.05.03.20089417v1 [accessed 1 April 2021]. [Google Scholar]
- Kogan NE, Clemente L, Liautaud P, Kaashoek J, Link NB, Nguyen AT, et al. . 2020. An early warning approach to monitor COVID-19 activity with multiple digital traces in near real-time. arXiv:2007.00756v2. Preprint posted online 3 July 2020. https://arxiv.org/abs/2007.00756v2, PMID: 32676518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar M, Kuroda K, Dhangar K. 2020a. The most eagerly awaited summer of the Anthropocene: a perspective of SARS-CoV-2 decay and seasonal change. Groundw Sustain Dev 11:100400, PMID: 32322654, 10.1016/j.gsd.2020.100400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar M, Taki K, Gahlot R, Sharma A, Dhangar K. 2020b. A chronicle of SARS-CoV-2: part-I-epidemiology, diagnosis, prognosis, transmission and treatment. Sci Total Environ 734:139278, PMID: 32434058, 10.1016/j.scitotenv.2020.139278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsen DA, Wigginton KR. 2020. Tracking COVID-19 with wastewater. Nat Biotechnol 38(10):1151–1153, PMID: 32958959, 10.1038/s41587-020-0690-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim RK, Wambier CG, Goren A. 2020. Are night shift workers at an increased risk for COVID-19? Med Hypotheses 144:110147, PMID: 32758906, 10.1016/j.mehy.2020.110147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manor Y, Shulman L, Kaliner E, Hindiyeh M, Ram D, Sofer D, et al. . 2014. Intensified environmental surveillance supporting the response to wild poliovirus type 1 silent circulation in Israel, 2013. Euro Surveill 19(7):20708, PMID: 24576473, 10.2807/1560-7917.es2014.19.7.20708. [DOI] [PubMed] [Google Scholar]
- Margetts M, Keshaviah A, Hu C, Troeger V, Sykes J, Bishop N, et al. . 2020. Using wastewater-based epidemiology with local indicators of opioid and illicit drug use to overcome data gaps in Montana. medRxiv. Preprint posted online 28 April 2020. 10.1101/2020.04.18.20064113. [DOI] [Google Scholar]
- Marvel S, House J, Wheeler M, Song K, Zhou Y, Wright F, et al. . 2020. The COVID-19 Pandemic Vulnerability Index (PVI) Dashboard: monitoring county level vulnerability. medRxiv. Preprint posted online 13 September 2020, PMID: 32817964, 10.1101/2020.08.10.20169649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCall AK, Bade R, Kinyua J, Lai FY, Thai PK, Covaci A, et al. . 2016. Critical review on the stability of illicit drugs in sewers and wastewater samples. Water Res 88:933–947, PMID: 26618807, 10.1016/j.watres.2015.10.040. [DOI] [PubMed] [Google Scholar]
- Medema G, Been F, Heijnen L, Petterson S. 2020. Implementation of environmental surveillance for SARS-CoV-2 virus to support public health decisions: opportunities and challenges. Curr Opin Environ Sci Health 17:49–71, PMID: 33024908, 10.1016/j.coesh.2020.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mor SM, DeMaria A Jr, Naumova EN. 2014. Hospitalization records as a tool for evaluating performance of food- and water-borne disease surveillance systems: a Massachusetts case study. PLoS One 9(4):e93744, PMID: 24740304, 10.1371/journal.pone.0093744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Institute on Drug Abuse. 2019. Rapid Assessment of Drug Abuse: Smart City Tools (R43/R44 – Clinical Trial Optional). Funding Opportunity Announcement No. RFA-DA-20-021. https://grants.nih.gov/grants/guide/rfa-files/rfa-da-20-021.html [accessed 14 January 2021].
- NIST (National Institute of Standards and Technology). 2020. A NIST-Hosted Virtual Workshop on Measuring SARS-CoV-2 in Wastewater and Fecal Material: A Call for Standards. NIST Special Publication 1256. https://nvlpubs.nist.gov/nistpubs/SpecialPublications/NIST.SP.1256.pdf [accessed 7 October 2020].
- NORMAN SCORE Database. 2020. SARS-CoV-2 in sewage (SC2S). https://www.norman-network.com/nds/sars_cov_2/ [accessed 2 October 2020].
- O’Brien E, Xagoraraki I. 2019. A water-focused One-Health approach for early detection and prevention of viral outbreaks. One Health 7:100094, PMID: 31080867, 10.1016/j.onehlt.2019.100094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oran DP, Topol EJ. 2020. Prevalence of asymptomatic SARS-CoV-2 infection: a narrative review. [Video.] Ann Intern Med 173(5):362–367, PMID: 32491919, 10.7326/M20-3012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parodi SM, Liu VX. 2020. From containment to mitigation of COVID-19 in the US. JAMA 323(15):1441–1442, PMID: 32167525, 10.1001/jama.2020.3882. [DOI] [PubMed] [Google Scholar]
- Peccia J, Zulli A, Brackney DE, Grubaugh ND, Kaplan EH, Casanovas-Massana A, et al. . 2020. Measurement of SARS-CoV-2 RNA in wastewater tracks community infection dynamics. Nat Biotechnol 38(10):1164–1167, PMID: 32948856, 10.1038/s41587-020-0684-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pecson BM, Darby E, Haas CN, Amha Y, Bartolo M, Danielson R, et al. . 2020. Reproducibility and sensitivity of 36 methods to quantify the SARS-CoV-2 genetic signal in raw wastewater: findings from an interlaboratory methods evaluation in the U.S. medRxiv. Preprint posted online 4 November 2020. 10.1101/2020.11.02.20221622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pérez-Cataluña A, Cuevas-Ferrando E, Randazzo W, Falcó I, Allende A, Sánchez G. 2021. Comparing analytical methods to detect SARS-CoV-2 in wastewater. Sci Total Environ :143870, PMID: 33338788, 10.1016/j.scitotenv.2020.143870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poletti P, Tirani M, Cereda D, Trentini F, Guzzetta G, Sabatino G, et al. . 2020. Probability of symptoms and critical disease after SARS-CoV-2 infection. arXiv:2006.08471. Preprint posted 22 June 2020. https://arxiv.org/abs/2006.08471 [accessed 1 April 2021]. [Google Scholar]
- Polo D, Quintela-Baluja M, Corbishley A, Jones DL, Singer AC, Graham DW, et al. . 2020. Making waves: wastewater-based epidemiology for COVID-19—approaches and challenges for surveillance and prediction. Water Res 186:116404, PMID: 32942178, 10.1016/j.watres.2020.116404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Randazzo W, Truchado P, Cuevas-Ferrando E, Simón P, Allende A, Sánchez G. 2020. SARS-CoV-2 RNA in wastewater anticipated COVID-19 occurrence in a low prevalence area. Water Res 181:115942, PMID: 32425251, 10.1016/j.watres.2020.115942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberto KJ, Johnson AF, Rauhaus BM. 2020. Stigmatization and prejudice during the COVID-19 pandemic. Adm Theory Praxis 42(3):364–378, 10.1080/10841806.2020.1782128. [DOI] [Google Scholar]
- Roberts L. 2013. Infectious disease. Israel’s silent polio epidemic breaks all the rules. Science 342(6159):679–680, PMID: 24202150, 10.1126/science.342.6159.679. [DOI] [PubMed] [Google Scholar]
- Saguti F, Magnil E, Enache L, Churqui MP, Johansson A, Lumley D, et al. . 2021. Surveillance of wastewater revealed peaks of SARS-CoV-2 preceding those of hospitalized patients with COVID-19. Water Res 189:116620, PMID: 33212338, 10.1016/j.watres.2020.116620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith T, Cassell G, Bhatnagar A. 2021. Wastewater surveillance can have a second act in COVID-19 vaccine distribution. JAMA Health Forum 2(1):e201616. Published online 11 January 2021, 10.1001/jamahealthforum.2020.1616. [DOI] [PubMed] [Google Scholar]
- Snow J. 1855. On the Mode of Communication of Cholera. London, England: John Churchill. http://www.ph.ucla.edu/epi/snow/snowbook.html [accessed 1 October 2020]. [Google Scholar]
- Sodré FF, Brandão CCS, Vizzotto CS, Maldaner AO. 2020. Wastewater-based epidemiology as a strategy for community monitoring, mapping of hotspots and early warning systems of COVID-19. [In Portuguese.] Química Nova 43(4):515–519, 10.21577/0100-4042.20170545. [DOI] [Google Scholar]
- U.S. EPA (U.S. Environmental Protection Agency). 2016. Clean watersheds needs survey 2012. https://www.epa.gov/cwns [accessed 12 January 2021].
- Wang G, Gu Z, Li X, Yu S, Kim M, Wang Y, et al. . 2020. Comparing and integrating US COVID-19 daily data from multiple sources: a county-level dataset with local characteristics. arXiv arXiv:2006.01333. Preprint posted online 28 November 2020. https://arxiv.org/abs/2006.01333 [accessed 1 April 2021]. [Google Scholar]
- WRF (Water Research Foundation). 2020. Wastewater Surveillance of the Covid-19 Genetic Signal in Sewersheds: Recommendations from Global Experts. https://www.waterrf.org/sites/default/files/file/2020-06/COVID-19_SummitHandout-v3b.pdf [accessed 22 December 2020].
- Zhan J, Liu QS, Sun Z, Zhou Q, Hu L, Qu G, et al. . 2020. Environmental impacts on the transmission and evolution of COVID-19 combing the knowledge of pathogenic respiratory coronaviruses. Environ Pollut 267:115621, PMID: 33254614, 10.1016/j.envpol.2020.115621. [DOI] [PMC free article] [PubMed] [Google Scholar]