Abstract
Background
Absence of coronary artery calcium (CAC) identifies asymptomatic individuals at low cardiovascular disease (CVD) risk. Carotid artery plaque is a marker of increased risk, but its association with cardiovascular risk and incident CAC in people without CAC is unclear.
Methods
Multi-Ethnic Study of Atherosclerosis participants with CAC score of 0 at enrollment who also underwent carotid plaque measurement using B-mode ultrasonography were prospectively followed for incident coronary heart disease (CHD), stroke, and CVD events, and CAC (score >0 on up to 3 serial CT scans). The association of carotid plaque presence and plaque score (Ln[score+1]) at baseline with cardiovascular events and incident CAC was evaluated with Cox proportional hazards regression models adjusted for demographics, risk factors, and statin use.
Results
Among these 2,673 participants (58 years, 64% women, 34% White, 30% Black, 24% Hispanic, and 12% Chinese), carotid plaque at baseline was observed in 973 (36%) and the median plaque score (range 1–12) among those with plaque was 1. A total of 79 CHD, 80 stroke, and 151 CVD events were observed during 16.1 years of follow-up. Carotid plaque presence and plaque score were independently associated with CHD risk (HRs 1.66, 95%CI 1.04, 2.66; and 1.48, 95%CI 1.01, 2.17, respectively) but not with stroke and CVD risk. A total of 973 (36.4%) participants developed CAC over the evaluation period (median 9.3 years). Carotid plaque presence and plaque score were independently associated with incident CAC (HRs 1.34, 95%CI 1.18, 1.54; and 1.37, 95% CI 1.21, 1.54), respectively.
Conclusions
The presence and extent of carotid plaque are associated with long-term CHD risk and incident CAC among middle-aged asymptomatic individuals with an initial CAC score of 0.
Keywords: Carotid ultrasound, CAC score, atherosclerosis, cardiovascular risk, prevention
Graphical Abstract
INTRODUCTION
Cardiovascular disease (CVD) is the leading cause of death and disability in the United States and worldwide.(1, 2) Preventing CVD is a public health priority and its foundation lies in the accurate assessment of CVD risk.(3) Coronary artery calcium (CAC), a marker of subclinical coronary atherosclerosis, can be quantified with non-contrast cardiac computed tomography (CT) and the CAC score has emerged as an important tool to refine cardiovascular risk assessment.(4) The presence and extent of CAC are associated with incident cardiovascular events. On the other hand, its absence, a CAC score of zero, portends a favorable cardiovascular prognosis. Recent studies have advanced the notion that a CAC score of zero may be used to ‘de-risk’ select individuals who otherwise may be candidates for statin therapy according to risk assessment tools based on traditional cardiovascular risk factors.(5) The 2018 American Heart Association / American College of Cardiology Multisociety Cholesterol Management Guideline recommends consideration of CAC testing in primary prevention settings when the decision to initiate statin therapy is uncertain.(6) However, it is worth noting that while a CAC score of zero is associated with favorable near- and intermediate-term prognosis, there is heterogeneity in long-term CVD risk..(7) Furthermore, a significant proportion of individuals without CAC develop calcific coronary atherosclerosis and the time-course for developing incident CAC varies considerably based on underlying risk factors.(8) This observation is important since the development of CAC is associated with future CVD risk.(9, 10)
Previous reports from the Multi-Ethnic Study of Atherosclerosis (MESA) demonstrated that the presence and extent of subclinical carotid plaque (quantified as a carotid plaque score), were associated with cardiovascular risk in the entire cohort.(11, 12) In this context, measuring subclinical carotid atherosclerosis with ultrasonography may have a potential role in cardiovascular risk stratification and, in particular, identification of individuals at higher risk of developing coronary atherosclerosis and CVD events among those with an initial CAC score of zero. Therefore, we sought to evaluate the association of carotid plaque and its burden with incident cardiovascular events (coronary heart disease [CHD], stroke, and CVD) and incident CAC among MESA participants with a CAC score of zero at the time of enrollment. We hypothesized that carotid plaque is associated with the risk of cardiovascular events and incident CAC development in these individuals.
METHODS
Study Population
The details of MESA study design have been published previously.(13) Briefly, MESA is an ongoing population-based epidemiologic cohort study of 6,814 White, Black, Hispanic, and Chinese individuals aged 45–84 years.(13) Participants were recruited from six field centers (Baltimore, Maryland; Chicago, Illinois; Forsyth County, North Carolina; Los Angeles, California; New York, New York; and St. Paul, Minnesota).(13) MESA Exam 1 was conducted between 2000 and 2002 with subsequent follow-up exams held during 2002–2004 (Exam 2), 2004–2005 (Exam 3), 2005–2007 (Exam 4), 2010–2012 (Exam 5), and 2016–2018 (Exam 6). Our study population included MESA participants that were free of clinical CVD at baseline, had CAC=0 at Exam 1, and complete carotid ultrasound data. The study was approved by the institutional review boards at all MESA field centers. All participants provided informed consent. MESA data is publicly available via two mechanisms: the National Heart, Lung, and Blood Institute’s Biologic Specimen and Data Repository Information Coordinating Center repository https://biolincc.nhlbi.nih.gov/studies/mesa/ or from the MESA coordinating center https://www.mesa-nhlbi.org/ with an approved proposal. MESA participants underwent detailed assessment for traditional cardiovascular risk factors (Supplement).
Coronary artery calcium score
CAC scores at Exams 1, 2, 3, and 4 were measured using an electron-beam CT scanner (Chicago, Los Angeles, and New York) or multidetector CT scanner (Baltimore, Forsyth County, and St. Paul) as previously described.(14) Radiographic phantoms containing identical and known calcium concentrations were placed beneath each participant’s thorax and certified technologists scanned each participant two times. A radiologist or cardiologist interpreted all CT scans at a single center (Los Angeles Biomedical Research Institute at Harbor–University of California Los Angeles Medical Center, Torrance, California). The CAC score was calculated for each scan using the Agatston method and the mean of the two scans was used in analyses. Intra-observer and inter-observer agreement was excellent (kappa statistics, 0.93 and 0.90, respectively).(14) The current analysis focuses on a subset of participants with CAC score of 0 at Exam 1 (N=3,416) with complete carotid ultrasound data. Participants were followed up for adjudicated cardiovascular events (N=2,673). A subset of these participants underwent a second CT scan at either Exam 2 or 3 and the time point of this scan was randomly assigned. An additional random subset of participants was selected for a third CT scan at Exam 4. Finally, a subset of participants enrolled in the MESA AIR ancillary study underwent multidetector CT scanning at Exam 5. Thus, participants had the potential to undergo up to four CT scans (Exam 2 and 3 scans were mutually exclusive). All participants included in this study received at least one follow-up CT scan.(15)
Carotid Ultrasonography
The carotid ultrasound protocol used at Exam 1 has been previously described.(16) Briefly, B-mode ultrasound images of the right and left common, bifurcation, and internal carotid artery segments were recorded on Super-VHS videotape with a Logiq 700 ultrasound system using a M12L transducer (GE Medical Systems; 13 MHz common carotid artery frequency). These images were digitized using a medical digital recording device (PACSGEAR, Pleasanton, CA) and converted into DICOM-compatible digital records. Ultrasound images were interpreted at the MESA Carotid Ultrasound Reading Center (the University of Wisconsin Atherosclerosis Imaging Research Program, Madison, WI). Images were imported into Syngo ultrasound workplace reading stations loaded with Arterial Health Package software (Siemens Medical, Malvern, PA) for carotid intima-media thickness (IMT) measurement. Carotid IMT was measured as the mean of the left and right far wall distal common carotid artery wall thicknesses.(16) Our analysis focused on carotid plaque and carotid plaque score. Carotid plaque was defined as a discrete, focal carotid wall thickening ≥1.5 mm or focal thickening at least 50% greater than the surrounding IMT.(17) The carotid plaque score was defined as the number of carotid plaques in the internal, bifurcation, and common segments of both carotid arteries.(12) The score’s absolute value ranged from 0 to 12, and among the 12 segments analyzed in each participant, 1 point per plaque was allocated for the far and near walls of each carotid artery segment (internal, bifurcation, and common) that was evaluated.(12) The intra-reader reproducibility for carotid plaque presence and score was κ=0.83 (95% confidence interval, 0.70–0.96), and inter-reader reproducibility was κ=0.89 (95% confidence interval, 0.72–1.00).(16) Carotid IMT was not studied in this analysis because a previous MESA report demonstrated that carotid plaque is superior to IMT for predicting CVD risk.(11)
Cardiovascular Disease Events
The cardiovascular events analyzed in this study include CHD, stroke, and CVD events. CHD events are a composite of MI, resuscitated cardiac arrest, and CHD death. Stroke events include nonfatal and fatal strokes but not transient ischemic attacks. CVD events are a composite of MI, resuscitated cardiac arrest, CHD death, definite angina, probable angina (if followed by coronary revascularization), nonfatal stroke, fatal strokes, other atherosclerotic death, and other CVD death. As a sensitivity analysis, we also analyzed atherosclerotic cardiovascular disease (ASCVD) events, a composite of nonfatal MI, CHD death, and fatal or nonfatal stroke, as the guideline-recommended endpoint of interest. Details of cardiovascular event ascertainment among MESA participants is described in detail in the Supplement.
Statistical Analyses
The baseline characteristics of study participants were described as count (proportion) for categorical variables and continuous variables were presented as mean (standard deviation) or median [interquartile range] depending on variable distribution. These characteristics were compared among those with and without carotid plaque at Exam 1 using Fisher’s exact test, or the Mann-Whitney U test, as appropriate. The association of carotid plaque (presence/absence) and plaque score (Ln[score+1]) with cardiovascular events was evaluated with Kaplan-Meier survival curves and Cox proportional hazards regression models. The outcome variables for cardiovascular events included time to first CHD event, time to first stroke event, time to first CVD event, and time to first ASCVD event. Time of censoring was defined as the last follow-up completed prior to December 31, 2017. The absolute annualized rate of cardiovascular events among participants with and without carotid plaque was studied. The difference in event rate between the two groups was calculated and used to compute the number needed to screen. The proportion of participants with incident CAC among those with and without carotid plaque at baseline was studied at each MESA exam. The association of carotid plaque (presence/absence) and plaque score (Ln[score+1]) with incident CAC was studied using Cox models where the outcome variable was time to first non-zero CAC measurement and the time of censoring was defined as the last follow-up CT scan. For each outcome of interest (CHD, stroke, CVD, ASCVD, and non-zero CAC score), three Cox models were used to study the relationship between carotid plaque or plaque score and time from Exam 1 to the minimum of observed outcome time or time of censoring. The three Cox models included a) an unadjusted model, b) model 1 adjusted for age, sex, and race/ethnicity, and b) model 2 additionally adjusted for diabetes, ever smoking, systolic blood pressure [SBP], antihypertensive use, TC, HDL-C, and statin use. Notably, follow-up CAC scores for each participant could be measured at Exams 2 or 3, 4, and/or 5, and thus incident CAC can be viewed as an interval censored outcome. Thus, a sensitivity analysis was performed in which incident CAC was treated as an interval-censored outcome in Cox regression models.(18) In an exploratory analysis, the multiplicative interaction of carotid plaque presence and plaque score with median participant age for incident cardiovascular events and CAC was studied. The assumption of proportional hazards in Cox models was verified using Schoenfeld residuals for right censored outcomes,(19, 20) and plots of the transformed survival function for interval censored outcomes.(21) Last, the impact of carotid plaque on ASCVD risk discrimination and reclassification beyond risk predicted by traditional risk factors was studied using change in model C-statistic,(22) and continuous net reclassification index (NRI), respectively. All statistical analyses were performed in R version 3.6.1 (R Foundation for Statistical Computing, Vienna, Austria). A two-sided p-value <0.05 was considered statistically significant.
RESULTS
Baseline Characteristics
Among 6,814 MESA participants, 3,416 had baseline CAC=0 and among these 2,673 had carotid ultrasound and follow-up data available. The median participant age was 57 years. The baseline characteristics of these participants are described in Table 1. Carotid plaque (plaque score ≥1) was observed in 36% (n=973) of the cohort and the distribution of carotid plaque score is depicted in Online Figure 1. The median plaque score for participants with carotid plaque was 1 [IQR: 1–2], and they were older, more frequently White, and had a higher prevalence of diabetes mellitus, smoking status, antihypertensive use, statin use, family history of MI or stroke, and lower educational attainment (Table 1). Additionally, participants with carotid plaque, on average, had higher SBP, total cholesterol, LDL-C, and lower eGFR as compared with those without carotid plaque (Table 1).
Table 1:
Baseline characteristics of study participants
| Overall Cohort | Plaque absent | Plaque present | P-value | |
|---|---|---|---|---|
| n=2673 | n=1700 | n=973 | ||
| Age (years) | 57.8 (±9.0) | 56.4 (±8.6) | 60.3 (±9.0) | <0.001 |
| Race/ethnicity | ||||
| White | 902 (33.7%) | 544 (32.0%) | 358 (36.8%) | <0.001 |
| Chinese | 318 (11.9%) | 246 (14.5%) | 72 (7.4%) | |
| Black | 808 (30.2%) | 481 (28.3%) | 327 (33.6%) | |
| Hispanic | 645 (24.1%) | 429 (25.2%) | 216 (22.2%) | |
| Gender | ||||
| Female | 1,699 (63.6%) | 1,062 (62.5%) | 637 (65.5%) | 0.120 |
| Male | 974 (36.4%) | 638 (37.5%) | 336 (34.5%) | |
| Diabetes | 232 (8.7%) | 108 (6.4%) | 124 (12.7%) | <0.001 |
| SBP (mmHg) | 122.2 (±20.3) | 119.6 (±18.9) | 126.8 (±21.8) | <0.001 |
| DBP (mmHg) | 71.2 (±10.1) | 71.0 (±10.0) | 71.5 (±10.4) | 0.260 |
| Antihypertensive use | 772 (28.9%) | 415 (24.4%) | 357 (36.7%) | <0.001 |
| Smoking | 347 (13.0%) | 196 (11.5%) | 151 (15.5%) | 0.003 |
| TC (mg/dL) | 193.8 (±34.8) | 191.8 (±34.0) | 197.3 (±36.0) | <0.001 |
| HDL-C (mg/dL) | 52.4 (±15.2) | 51.9 (±14.6) | 53.2 (±16.2) | 0.140 |
| Triglycerides (mg/dL) | 106.0 [74.2 – 156.0] |
105.0 [73.0 – 154.0] |
108.0 [77.0 – 159.0] |
0.074 |
| LDL-C (mg/dL) | 116.0 (±30.4) | 114.9 (±30.0) | 117.9 (±31.1) | 0.025 |
| Statin use | 266 (10.0%) | 123 (7.2%) | 143 (14.7%) | <0.001 |
| eGFR (ml/min/1.73 m2) | 76.1 (±14.7) | 76.9 (±14.3) | 74.8 (±15.4) | <0.001 |
| BMI (kg/m2) | 28.3 (±5.6) | 28.1 (±5.6) | 28.5 (±5.5) | 0.027 |
| Family history of MI | 951 (35.6%) | 572 (33.6%) | 379 (39.0%) | 0.005 |
| Family history of stroke | 824 (30.8%) | 472 (27.8%) | 352 (36.2%) | <0.001 |
| Education | ||||
| High school or lower | 1,691 (63.3%) | 1,042 (61.3%) | 649 (66.7%) | 0.005 |
| College or higher | 972 (36.4%) | 652 (38.4%) | 320 (32.9%) |
Categorical variables presented as count (proportion) and continuous variables presented as mean (standard deviation) or median [25th – 75th percentile] depending on variable distribution. Abbreviations: SBP=systolic blood pressure, DBP=diastolic blood pressure, TC=total cholesterol, HDL-C=high-density lipoprotein cholesterol, LDL-C=low-density lipoprotein cholesterol, eGFR=estimated glomerular filtration rate, BMI=body mass index, MI=myocardial infarction
Association of Carotid Plaque with Cardiovascular Disease Events
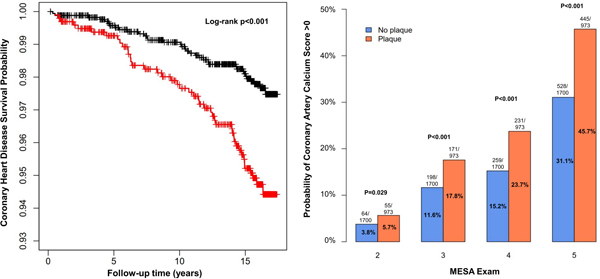
The 2,673 participants included in this analysis were followed for cardiovascular events for a median duration of 16.1 [15.4–16.6] years. Baseline carotid ultrasound revealed 973 participants with and 1700 participants without carotid plaque. A total of 79 CHD, 80 stroke, 151 CVD, and 149 ASCVD events were observed, among which 44 CHD, 29 stroke, 69 CVD, and 69 ASCVD events occurred in individuals with carotid plaque. Participants with carotid plaque identified at exam 1 were at a higher risk of CHD, CVD, and ASCVD events, but not stroke events in Kaplan-Meier survival analyses (Figures 1A–C and Online Figure 2). The absolute annualized rate of cardiovascular events was low (<0.6%) among study participants (Table 2). Participants with carotid plaque had a slightly higher annualized rate of events as compared with those without plaque and the annual number needed to screen with carotid ultrasonography in this low-risk population was high (>550 for all events, Table 2).
Figure 1: Kaplan Meier survival curves for the association of carotid plaque presence with risk of coronary heart disease events (1A), stroke events (1B), and cardiovascular disease events (1C).
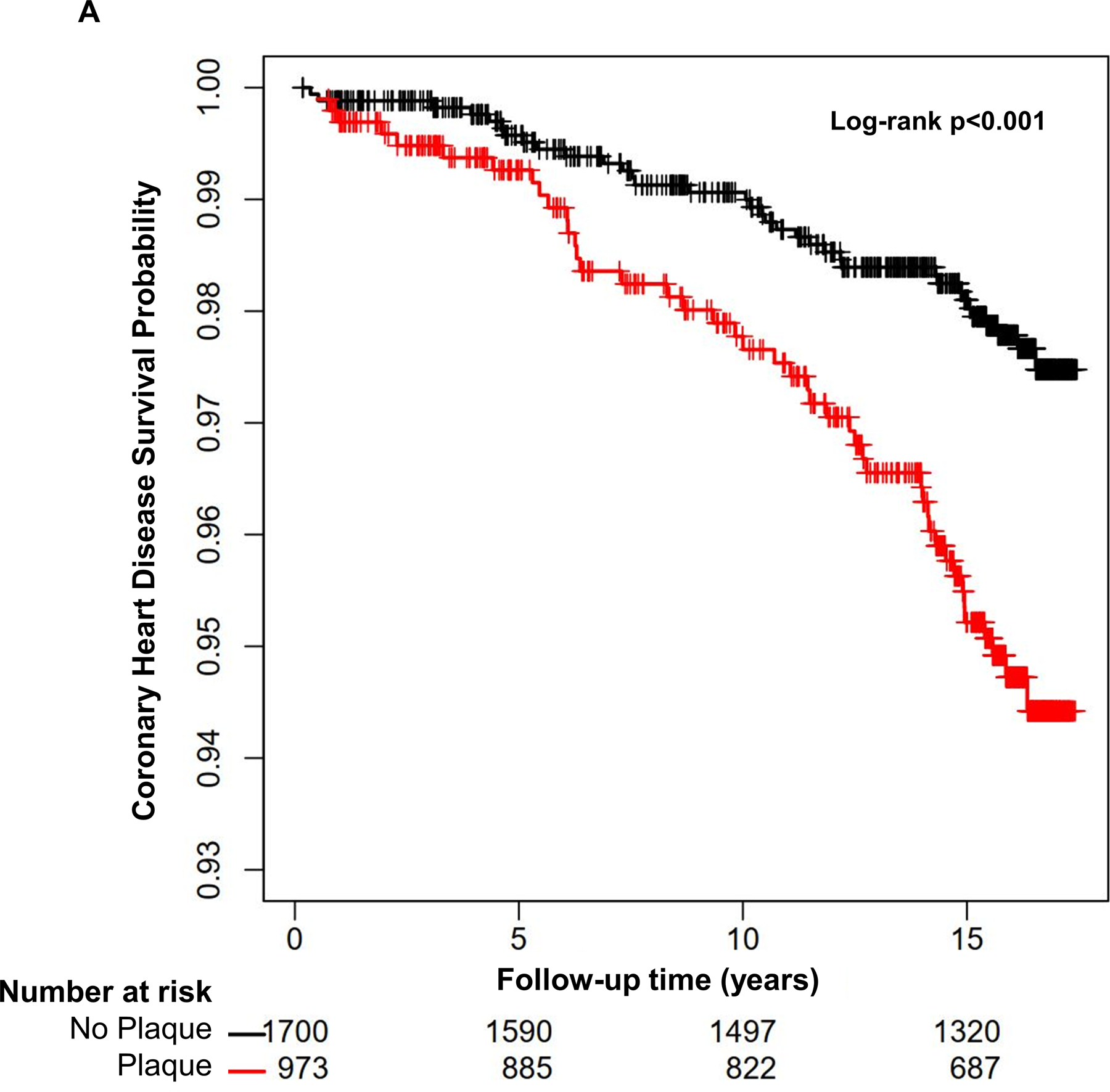
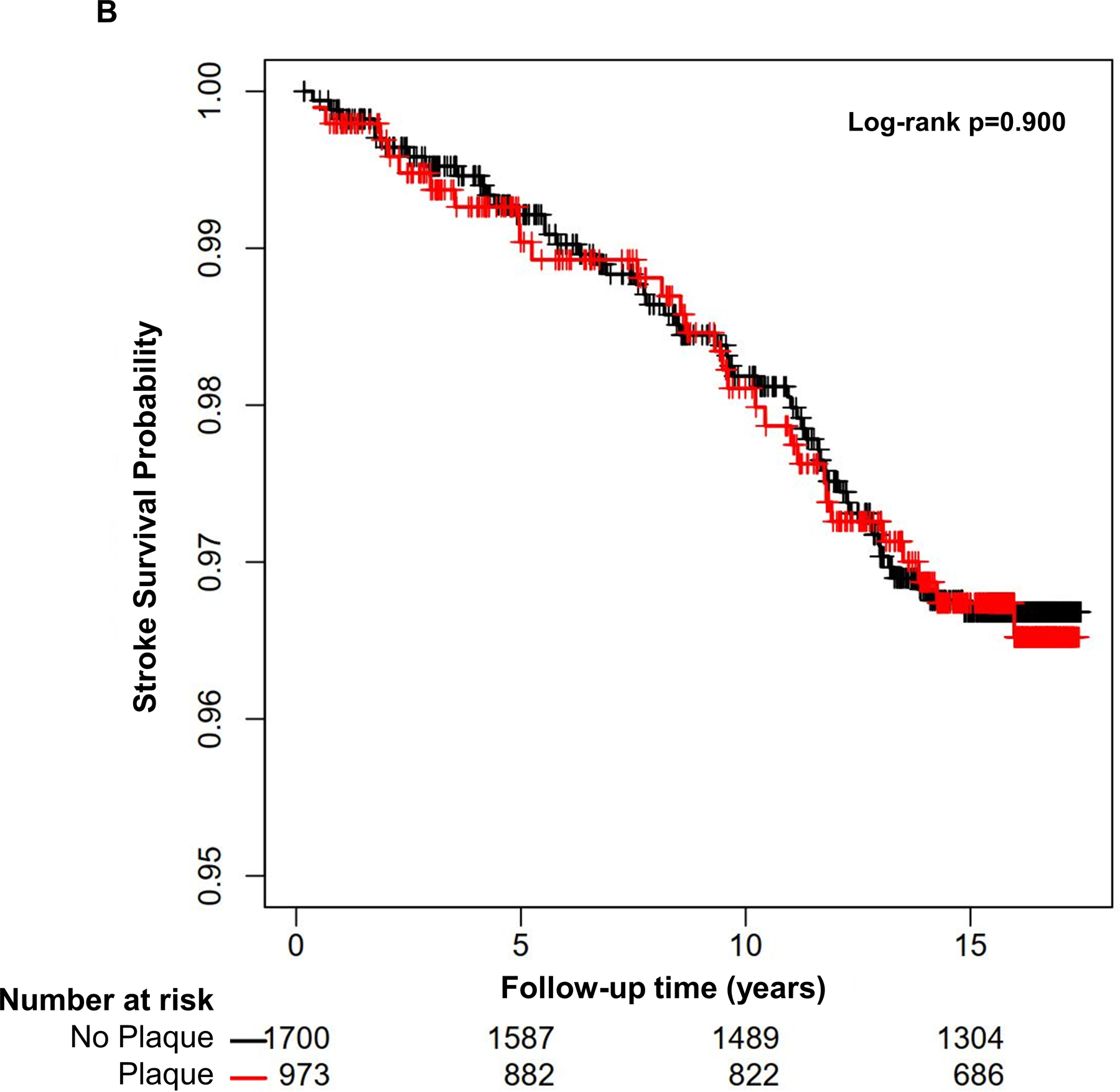
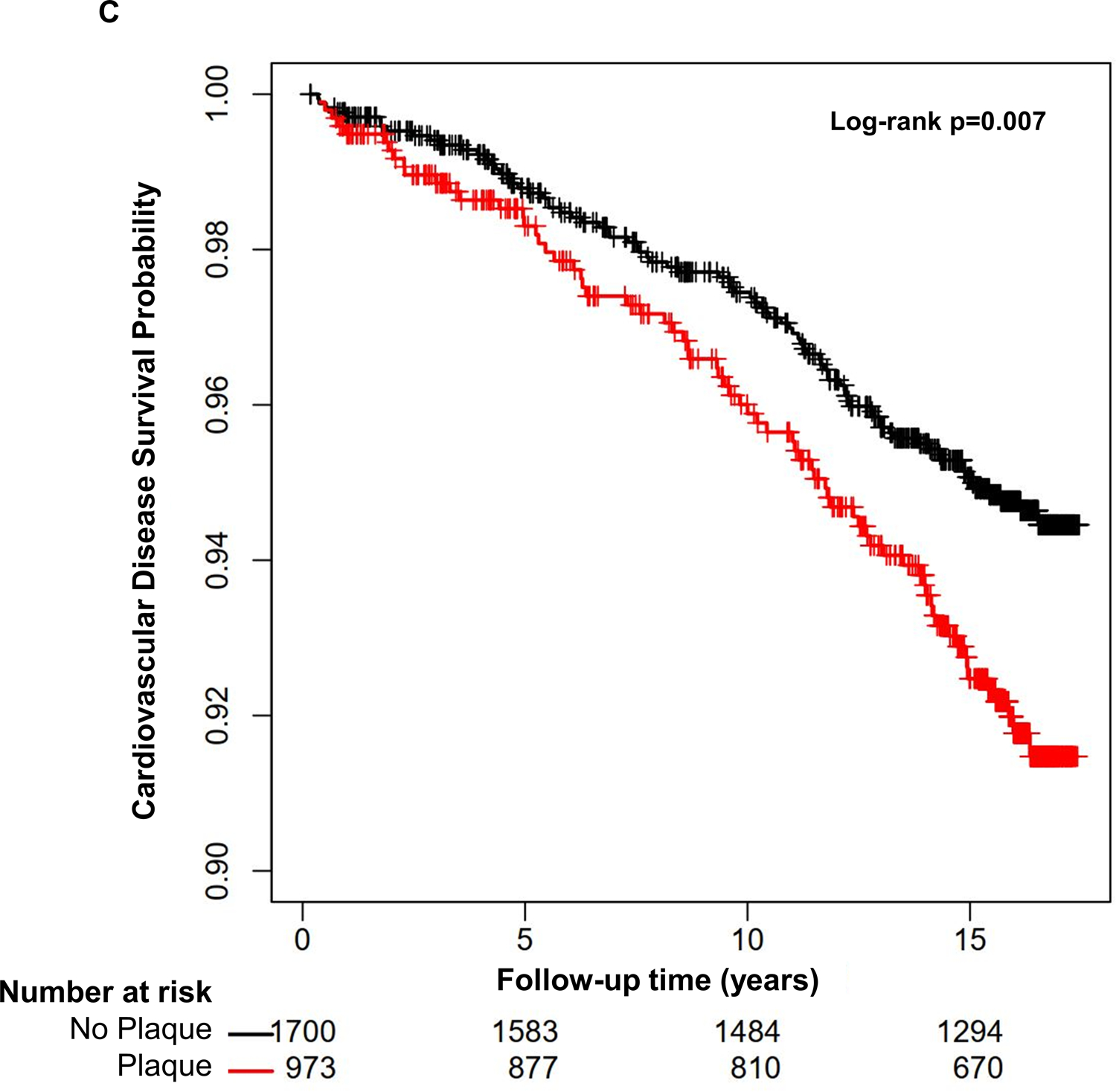
The presence of carotid plaque is associated with an increased risk of coronary heart disease and cardiovascular disease events, but not stroke events (N=2,673).
Table 2:
Absolute annualized rate of cardiovascular events, risk difference, and number needed to screen
| Event | Plaque present | Plaque absent | Difference | Number Needed to Screen |
|---|---|---|---|---|
| CHD | 0.321% | 0.141% | 0.180% | 556 |
| Stroke | 0.212% | 0.206% | 0.006% | 16667 |
| CVD | 0.510% | 0.333% | 0.167% | 599 |
| ASCVD | 0.483% | 0.311% | 0.172% | 581 |
Abbreviations: CHD=coronary heart disease, CVD=cardiovascular disease, ASCVD=atherosclerotic cardiovascular disease.
The presence of carotid plaque at baseline was associated with a 2.3-fold increased risk of CHD events in an unadjusted Cox regression model (Table 3A). This association remained significant after adjusting for age, sex, and race/ethnicity (Table 3A, Model 1); as well as after further adjusting for diabetes mellitus, smoking status, SBP, antihypertensive use, TC, HDL-C, and statin use (Table 3A, Model 2). Thus, the presence of carotid plaque was independently associated with a 66% increase in CHD risk (HR 1.66, 95%CI 1.04, 2.66). Increasing age and smoking were also independently associated with CHD events (Table 3A, Model 2). Furthermore, increasing carotid plaque score was independently associated with CHD risk as well (Online Table 1A). A multiplicative interaction between median age (57 years) and carotid plaque (p=0.30) or plaque score (p=0.61) for CHD risk was not observed.
Table 3A:
Association of carotid plaque with coronary heart disease events (N=2673)
| Unadjusted | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Carotid Plaque | 2.31 (1.48, 3.61) |
<0.001 | 1.95 (1.23, 3.08) |
0.004 | 1.66 (1.04, 2.66) |
0.033 |
|
Age
(per 5-years) |
1.20 (1.06, 1.36) |
0.004 | 1.20 (1.05, 1.38) |
0.009 | ||
| Male | 1.13 (0.71, 1.79) |
0.612 | 1.00 (0.60, 1.65) |
0.985 | ||
| Race (ref=White) | 0.490 | 0.698 | ||||
| Chinese | 0.56 (0.22, 1.47) |
0.56 (0.21, 1.47) |
||||
| Black | 1.18 (0.70, 2.01) |
0.94 (0.54, 1.65) |
||||
| Hispanic | 1.10 (0.61, 1.98) |
0.96 (0.53, 1.75) |
||||
| Diabetes | 1.54 (0.79, 2.99) |
0.201 | ||||
| Smoking | 2.57 (1.50, 4.43) |
<0.001 | ||||
|
SBP
(per 10-mmHg) |
1.08 (0.96, 1.2) |
0.207 | ||||
| Antihypertensive use | 1.24 (0.75, 2.06) |
0.409 | ||||
|
TC
(per 40-mg/dL) |
1.00 (0.77, 1.3) |
0.984 | ||||
|
HDL-C
(per 5-mg/dL) |
0.96 (0.89, 1.05) |
0.410 | ||||
| Statin use | 1.27 (0.68, 2.38) |
0.452 | ||||
Carotid plaque (Table 3B) and plaque score (Online Table 1B) were not associated with stroke events in the study population. Increasing age, SBP, diabetes mellitus, and smoking status were independently associated with stroke risk (Table 3B). A nominal interaction between median age and carotid plaque (p=0.06), and a significant interaction between median age and plaque score (p=0.01) were observed. In younger participants (age <57 years), carotid plaque was nominally associated (HR 1.78, 95%CI 0.73, 4.35), and plaque score was significantly associated (HR 2.34, 95%CI 1.08, 5.06) with stroke risk. Plaque presence (HR 0.66, 95%CI 0.39, 1.11) and plaque score (HR 0.71, 95%CI 0.44, 1.15) were not associated with stroke risk in older participants (age ≥57 years).
Table 3B:
Association of carotid plaque with stroke events (N=2673)
| Unadjusted | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Carotid Plaque | 1.03 (0.65, 1.62) |
0.912 | 0.78 (0.49, 1.25) |
0.310 | 0.64 (0.39, 1.04) |
0.073 |
|
Age
(per 5-years) |
1.37 (1.22, 1.55) |
<0.001 | 1.29 (1.13, 1.47) |
<0.001 | ||
| Male | 1.22 (0.78, 1.92) |
0.387 | 1.03 (0.63, 1.69) |
0.910 | ||
| Race (ref=White) | 0.376 | 0.748 | ||||
| Chinese | 0.74 (0.32, 1.73) |
0.62 (0.26, 1.48) |
||||
| Black | 1.41 (0.82, 2.42) |
0.96 (0.54, 1.71) |
||||
| Hispanic | 1.14 (0.63, 2.09) |
0.89 (0.48, 1.67) |
||||
| Diabetes | 1.89 (1.01, 3.53) |
0.045 | ||||
| Smoking | 2.34 (1.29, 4.25) |
0.005 | ||||
|
SBP
(per 10-mmHg) |
1.31 (1.19, 1.45) |
<0.001 | ||||
| Antihypertensive use | 0.96 (0.58, 1.59) |
0.864 | ||||
|
TC
(per 40-mg/dL) |
0.86 (0.66, 1.12) |
0.268 | ||||
|
HDL-C
(per 5-mg/dL) |
0.94 (0.86, 1.03) |
0.196 | ||||
| Statin use | 0.92 (0.46, 1.83) |
0.811 | ||||
Presence of carotid plaque (Table 3C) and plaque score (Online Table 1C) were associated with CVD events, a composite of CHD and stroke events, in unadjusted models. However, this relationship was attenuated after adjusting for confounders. Increasing age, SBP, diabetes mellitus, and smoking status were independently associated with CVD risk (Table 3C). A multiplicative interaction between median age and carotid plaque (p=0.34) or plaque score (p=0.11) for CVD risk was not observed.
Table 3C:
Association of carotid plaque with cardiovascular disease events (N=2673)
| Unadjusted | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Carotid Plaque | 1.54 (1.12, 2.13) |
0.008 | 1.25 (0.90, 1.73) |
0.189 | 1.07 (0.76, 1.50) |
0.700 |
|
Age
(per 5-years) |
1.28 (1.17, 1.4) |
<0.001 | 1.26 (1.14, 1.38) |
<0.001 | ||
| Male | 1.24 (0.89, 1.73) |
0.195 | 1.05 (0.73, 1.50) |
0.805 | ||
| Race (ref=White) | 0.227 | 0.592 | ||||
| Chinese | 0.69 (0.36, 1.3) |
0.64 (0.34, 1.23) |
||||
| Black | 1.28 (0.86, 1.88) |
0.96 (0.63, 1.45) |
||||
| Hispanic | 1.16 (0.76, 1.78) |
0.97 (0.62, 1.51) |
||||
| Diabetes | 1.70 (1.06, 2.73) |
0.027 | ||||
| Smoking | 2.64 (1.76, 3.95) |
<0.001 | ||||
|
SBP
(per 10-mmHg) |
1.18 (1.09, 1.28) |
<0.001 | ||||
| Antihypertensive use | 1.05 (0.72, 1.52) |
0.796 | ||||
|
TC
(per 40-mg/dL) |
0.93 (0.77, 1.13) |
0.482 | ||||
|
HDL-C
(per 5-mg/dL) |
0.95 (0.89, 1.01) |
0.110 | ||||
| Statin use | 0.94 (0.57, 1.55) |
0.801 | ||||
In a sensitivity analysis, presence of carotid plaque (Online Table 2A) and plaque score (Online Table 2B) were associated with ASCVD events, but this association was attenuated after adjusting for risk factors. A model comprising of traditional risk factors had good ASCVD risk discrimination capability with model C-statistic 0.736. Addition of carotid plaque to this model did not result in improvement in ASCVD risk discrimination (new C-statistic 0.737, p=0.471) or risk reclassification (continuous NRI 0.098, 95% CI −0.130, 0.325, p=0.399).
Association of Carotid Plaque with Incident Coronary Artery Calcium
Study participants were followed for development of CAC for a median duration of 9.3 years. The number of participants with 1, 2, and 3 follow-up cardiac CT scans was 595, 1382, and 438, respectively, and the median number of follow-up CT scans was 2. A total of 973 participants, 36.4% of the cohort without CAC at baseline, had at least one follow-up CT scan with CAC score >0. The proportion of participants with incident CAC at MESA exams 2, 3, 4, and 5 was higher among those with carotid plaque as compared to those without carotid plaque at baseline (Figure 2). Among participants without carotid plaque (N=1700), 528 (31%) developed incident CAC. The corresponding proportion for those with carotid plaque (N=973) was 445 (45.7%). This relationship was evident in Cox regression models as well and remained significant after adjusting for relevant confounders (Table 4). Thus, carotid plaque presence was associated with a 34% higher risk of developing CAC during follow-up (Table 4, Model 2). Increasing age, male sex, white race, higher SBP, antihypertensive use, higher TC, lower HDL-C, and statin use were also independently associated with incident CAC (Table 4, Model 2). Increasing carotid plaque score was independently associated with incident CAC as well (Online Table 3). Significant interactions between median age and carotid plaque (p<0.01), as well as median age and plaque score (p<0.01) were observed. In younger participants (age <57 years), carotid plaque and plaque score were significantly associated with incident CAC (HRs 1.96, 95%CI 1.60, 2.38; and 2.06, 95%CI 1.71, 2.48, respectively). The association of plaque presence (HR 1.37, 95%CI 1.16,1.62) and plaque score (HR 1.44, 95%CI 1.25, 1.66) with incident CAC was relatively weaker in older participants (age ≥57 years). In a sensitivity analysis, the independent association of carotid plaque and plaque score with incident CAC was unchanged when follow-up CAC score was treated as an interval-censored outcome (Online Tables 4A and 4B).
Figure 2: Carotid plaque presence and incident coronary artery calcium during MESA follow-up exams.
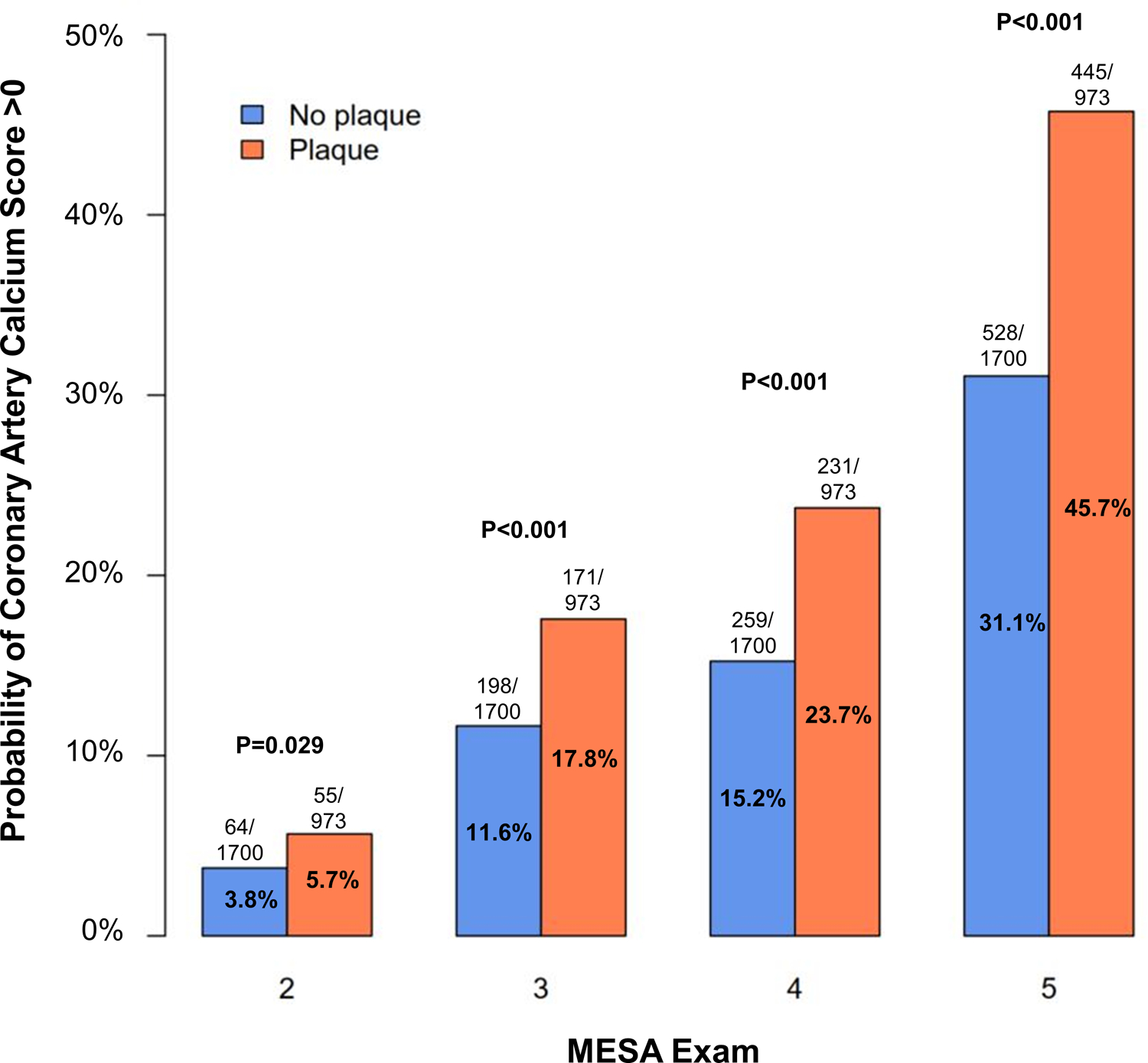
The proportion of MESA participants with incident CAC (score >0) is higher among individuals with presence of carotid plaque at baseline.
Table 4:
Association of carotid plaque with incident coronary artery calcium (N=2673)
| Unadjusted | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|
| HR (95% CI) | P-value | HR (95% CI) | P-value | HR (95% CI) | P-value | |
| Carotid Plaque | 1.77 (1.56, 2.01) |
<0.001 | 1.48 (1.30, 1.68) |
<0.001 | 1.34 (1.18, 1.54) |
<0.001 |
|
Age
(per 5-years) |
1.25 (1.21, 1.30) |
<0.001 | 1.22 (1.17, 1.27) |
<0.001 | ||
| Male | 1.53 (1.34, 1.74) |
<0.001 | 1.39 (1.20, 1.60) |
<0.001 | ||
| Race (ref=White) | <0.001 | <0.001 | ||||
| Chinese | 0.61 (0.48, 0.76) |
0.58 (0.46, 0.73) |
||||
| Black | 1.01 (0.86, 1.18) |
0.88 (0.75, 1.04) |
||||
| Hispanic | 0.93 (0.79, 1.10) |
0.83 (0.70, 0.98) |
||||
| Diabetes | 1.21 (0.97, 1.50) |
0.090 | ||||
| Smoking | 1.12 (0.93, 1.36) |
0.240 | ||||
|
SBP
(per 10-mmHg) |
1.07 (1.04, 1.11) |
<0.001 | ||||
| Antihypertensive use | 1.22 (1.05, 1.41) |
0.009 | ||||
|
TC
(per 40-mg/dL) |
1.13 (1.05, 1.21) |
0.002 | ||||
|
HDL-C
(per 5-mg/dL) |
0.94 (0.92, 0.97) |
<0.001 | ||||
| Statin use | 1.30 (1.07, 1.58) |
0.007 | ||||
DISCUSSION
It is well established that absence of calcified coronary atherosclerosis as determined by a CAC score of zero predicts a low incidence of cardiovascular events in the short- and intermediate-terms. In this analysis, we aimed to determine whether the presence of subclinical carotid atherosclerosis may impact CVD event rates and CAC development in individuals with an initial CAC of 0. We have evaluated the association of carotid plaque and its burden with incident cardiovascular events (CHD, stroke, CVD, and ASCVD) and incident CAC among MESA participants with a CAC score of zero at the time of enrollment. We report two important findings. First, we demonstrate that the presence and extent of carotid plaque is independently associated with an increased long-term risk of CHD events in asymptomatic individuals without CAC at baseline. Second, we show that the presence and extent of carotid plaque associate with incident CAC in these individuals, providing biological plausibility to the association with clinical events.
Carotid Plaque and Cardiovascular Disease Events in Absence of CAC
The absence of CAC on cardiac CT, e.g., CAC score of 0, indicates the absence of calcified coronary atherosclerosis and is a marker of low coronary atherosclerosis burden in the general population.(23) Several studies over the past decade have reported that a CAC score of 0 portends a favorable cardiovascular prognosis in diverse settings.(24–28) More recently, there has been increasing interest in the heterogeneity in risk observed among asymptomatic individuals that are deemed to be low-risk based on the absence of CAC.(7)
We studied the association of carotid plaque and plaque score with CVD events in MESA participants with a CAC score of 0 at Exam 1. Previous research demonstrated that detection of carotid plaque using ultrasound is a safe and inexpensive tool that can help predict cardiovascular risk in the general population.(29) Furthermore, assessment of carotid plaque is a more powerful predictor of ASCVD risk predictor as compared with carotid IMT.(30) Gepner et al. demonstrated that presence of carotid plaque was independently associated with incident CVD, CHD, and stroke/transient ischemic attack over 9 years in MESA participants.(11) In the same report, the authors also demonstrated that presence and extent of CAC predicted incident cardiovascular events, whereas mean common carotid IMT did not.(11) More recently, Gepner and colleagues demonstrated that the carotid plaque score was independently associated with CVD, CHD, and stroke/transient ischemic attack risk in the entire MESA cohort followed for 11 years.(12)
However, the association of carotid plaque and plaque score with long-term cardiovascular risk in individuals with baseline CAC score of 0, a group that is at low risk, has not been studied to date. Here, we show that presence and extent of carotid plaque are independently associated with long-term CHD risk even in the absence of CAC, which reaffirms that CAC of 0 defines a “low risk” group, not a “no risk” group. The only other traditional risk factors associated with CHD risk were age and smoking.
Notably, an independent association of carotid plaque with stroke, CVD, and ASCVD risk was not seen in this cohort. Furthermore, the absolute annualized rate of events among individuals with and without carotid plaque, and difference in risk between the two groups was low. This resulted in a high number needed to screen for all endpoints in this low-risk cohort. Last, addition of carotid plaque to a traditional risk factor model did not improve cardiovascular risk discrimination and reclassification. The constellation of these observations highlights the limited incremental value of screening those with CAC score of 0 with carotid ultrasonography.
Carotid Plaque and Incident CAC
Incident CAC, i.e. conversion of a CAC score of 0 to >0 score over time, remains an area of interest. Prior work established that CAC incidence and progression in asymptomatic individuals associates with future cardiovascular risk.(9, 10) Previous studies demonstrated that CAC incidence is low during the first 4–5 years after an initial cardiac CT demonstrates the absence of calcified coronary atherosclerosis.(31, 32) More recently, Dyaze and colleagues reported a 3 to 7-year window for CAC incidence in MESA participants with CAC=0, and this time period was dependent on age, sex, and baseline cardiovascular risk.(8) Nevertheless, the association of carotid plaque with CAC incidence has not been evaluated to date. Herein, we show that the presence and extent of carotid plaque are associated with a significantly increased risk of incident CAC. Importantly, this relationship was stronger in younger participants (age <57 years) and was independent of traditional cardiovascular risk factors associated with CAC incidence.
Atherosclerosis is a complex systemic arterial disease process that proceeds at a varying pace in different vascular beds during the lifetime.(33, 34) Our results illustrate that even in the absence of demonstrable calcified coronary atherosclerosis, subclinical carotid atherosclerosis predicts the risk of future CHD and development of calcific coronary atherosclerosis. These findings provide important insights into the heterogeneity in cardiovascular risk and the natural history of calcific coronary atherosclerosis in middle-aged asymptomatic individuals without CAC. These observations also lend credence to the notion that selective ultrasound assessment for subclinical carotid plaque in those without CAC may be useful in enhancing risk assessment and facilitating shared decision making, particularly among younger adults.(35) Importantly, the findings highlight an area that is commonly confused. Specifically, the absence of CAC is associated with low risk for ASCVD events, but this does not equate to no risk. Therefore, even in the absence of CAC, clinicians and patients must still strive to optimize cardiovascular risk control.
Strengths and Limitations
Our study has several notable strengths. We report findings from a multi-ethnic epidemiologic cohort of asymptomatic individuals with initial CAC of 0 that were followed for more than 15 years for adjudicated cardiovascular events. Our study population underwent detailed phenotyping for carotid plaque measurement using state-of-the-art ultrasonographic techniques and every participant received at least two cardiac CT scans to determine CAC incidence.
Limitations include a relatively younger cohort with a low cardiovascular event rate. MESA participants were not scanned for CAC at each exam which precluded calculation of the annual CAC incidence. Stroke events included in this study were not classified as ischemic, hemorrhagic, embolic, or cryptogenic. Longitudinal changes in risk factors and initiation of preventive cardiovascular therapies including statins and antihypertensive medications was not analyzed. Lastly, the potential for residual confounding cannot be excluded given the observational nature of this study.
Conclusion
Carotid plaque in the absence of baseline CAC is independently associated with incident CHD events and incident CAC development among asymptomatic, middle-aged participants in a multi-ethnic, population-based epidemiologic cohort. Though absence of CAC portends a low cardiovascular event rate, heterogeneity in risk tracks with the presence or absence of carotid atherosclerosis. Individuals with carotid plaque, despite absence of CAC, may benefit from more intense primary CHD prevention efforts.
Supplementary Material
CLINICAL PERSPECTIVE.
The absence of coronary artery calcium (CAC) on cardiac computed tomography (CT), a CAC score of zero, is associated with a favorable cardiovascular prognosis and can be used to guide shared decision making in primary prevention settings. However, heterogeneity in long-term cardiovascular risk is observed individuals with zero CAC score and the predictors of long-term risk in this low-risk population are incompletely understood. In particular, the role of subclinical carotid artery atherosclerosis measured as carotid plaque on ultrasonography in this group has not been previously evaluated. Thus, this study sought to determine the independent association of carotid plaque and its burden, measured as carotid plaque score, with incident cardiovascular events and development of coronary artery calcification among Multi-Ethnic Study of Atherosclerosis (MESA) participants with a CAC score of zero at enrollment. We studied 2,673 asymptomatic, middle-aged MESA participants who underwent carotid ultrasonography at enrollment and were prospectively followed for cardiovascular events and underwent serial cardiac CT scans to determine the development of CAC score greater than zero. Our results demonstrate that the presence and burden of carotid plaque are independently associated with incident coronary heart disease events and development of CAC during long-term follow-up. Our findings indicate that the heterogeneity in cardiovascular risk observed among individuals with absent CAC tracks with subclinical carotid atherosclerosis and its extent. Individuals with carotid plaque, despite the absence of CAC, might benefit from more intense primary cardiovascular prevention efforts to help decrease cardiovascular risk.
Acknowledgments
The authors thank the other investigators, the staff, and the participants of the MESA study for their valuable contributions. A full list of participating MESA investigators and institutions can be found at http://www.mesa-nhlbi.org.
Sources of Funding
The MESA study is supported by contracts HHSN268201500003I, N01-HC-95159, N01-HC-95160, N01-HC-95161, N01-HC-95162, N01-HC-95163, N01-HC-95164, N01-HC-95165, N01-HC-95166, N01-HC-95167, N01-HC-95168 and N01-HC-95169 from the National Heart, Lung, and Blood Institute, and by grants UL1-TR-000040,UL1-TR-001079, and UL1-TR-001420 from National Center for Advancing Translational Sciences. The MESA Air ancillary study was supported by a grant from the US Environmental Protection Agency’s (EPA’s) Science to Achieve Results (STAR) program - Assistance Agreement number RD831697 awarded by the EPA to the University of Washington (WA, USA).
ABBREVIATIONS
- ASCVD
Atherosclerotic cardiovascular disease
- CVD
Cardiovascular disease
- CAC
Coronary artery calcium
- CI
Confidence Interval
- HDL-C
High-density lipoprotein cholesterol
- HR
Hazard Ratio
- MDCT
Multi-Detector Computed Tomography
- MESA
Multi-Ethnic Study of Atherosclerosis
- SBP
Systolic blood pressure
- TC
Total cholesterol
Footnotes
Disclosures
None
REFERENCES
- 1.Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Delling FN, et al. Heart Disease and Stroke Statistics-2020 Update: A Report From the American Heart Association. Circulation. 2020;141(9):e139–e596. [DOI] [PubMed] [Google Scholar]
- 2.Collaborators GBDCoD. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1736–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lloyd-Jones DM, Braun LT, Ndumele CE, Smith SC Jr., Sperling LS, Virani SS, Blumenthal RS. Use of Risk Assessment Tools to Guide Decision-Making in the Primary Prevention of Atherosclerotic Cardiovascular Disease: A Special Report From the American Heart Association and American College of Cardiology. J Am Coll Cardiol. 2019;73(24):3153–67. [DOI] [PubMed] [Google Scholar]
- 4.Greenland P, Blaha MJ, Budoff MJ, Erbel R, Watson KE. Coronary Calcium Score and Cardiovascular Risk. J Am Coll Cardiol. 2018;72(4):434–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Blaha MJ, Cainzos-Achirica M, Greenland P, McEvoy JW, Blankstein R, Budoff MJ, Dardari Z, Sibley CT, Burke GL, Kronmal RA, et al. Role of Coronary Artery Calcium Score of Zero and Other Negative Risk Markers for Cardiovascular Disease: The Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2016;133(9):849–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, Braun LT, de Ferranti S, Faiella-Tommasino J, Forman DE, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019. June 25;73(24):3168–3209. [DOI] [PubMed] [Google Scholar]
- 7.Joshi PH, Blaha MJ, Budoff MJ, Miedema MD, McClelland RL, Lima JAC, Agatston AS, Blankstein R, Blumenthal RS, Nasir K. The 10-Year Prognostic Value of Zero and Minimal CAC. JACC Cardiovasc Imaging. 2017;10(8):957–58. [DOI] [PubMed] [Google Scholar]
- 8.Dzaye O, Dardari ZA, Cainzos-Achirica M, Blankstein R, Szklo M, Budoff MJ, Lima JAC, Blumenthal RS, Nasir K, Blaha MJ. Incidence of New Coronary Calcification: Time to Conversion From CAC = 0. J Am Coll Cardiol. 2020;75(13):1610–13. [DOI] [PubMed] [Google Scholar]
- 9.Budoff MJ, Young R, Lopez VA, Kronmal RA, Nasir K, Blumenthal RS, Detrano RC, Bild DE, Guerci AD, Liu K, et al. Progression of coronary calcium and incident coronary heart disease events: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol. 2013;61(12):1231–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lehmann N, Erbel R, Mahabadi AA, Rauwolf M, Mohlenkamp S, Moebus S, Kalsch H, Budde T, Schmermund A, Stang A, et al. Value of Progression of Coronary Artery Calcification for Risk Prediction of Coronary and Cardiovascular Events: Result of the HNR Study (Heinz Nixdorf Recall). Circulation. 2018;137(7):665–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gepner AD, Young R, Delaney JA, Tattersall MC, Blaha MJ, Post WS, Gottesman RF, Kronmal R, Budoff MJ, Burke GL, et al. Comparison of coronary artery calcium presence, carotid plaque presence, and carotid intima-media thickness for cardiovascular disease prediction in the Multi-Ethnic Study of Atherosclerosis. Circ Cardiovasc Imaging. 2015;8(1) :e002262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gepner AD, Young R, Delaney JA, Budoff MJ, Polak JF, Blaha MJ, Post WS, Michos ED, Kaufman J, Stein JH. Comparison of Carotid Plaque Score and Coronary Artery Calcium Score for Predicting Cardiovascular Disease Events: The Multi-Ethnic Study of Atherosclerosis. J Am Heart Assoc. 2017;6(2):e005179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bild DE, Bluemke DA, Burke GL, Detrano R, Diez Roux AV, Folsom AR, Greenland P, Jacob DR Jr., Kronmal R, Liu K, et al. Multi-Ethnic Study of Atherosclerosis: objectives and design. Am J Epidemiol. 2002;156(9):871–81. [DOI] [PubMed] [Google Scholar]
- 14.Carr JJ, Nelson JC, Wong ND, McNitt-Gray M, Arad Y, Jacobs DR Jr., Sidney S, Bild DE, Williams OD, Detrano RC. Calcified coronary artery plaque measurement with cardiac CT in population-based studies: standardized protocol of Multi-Ethnic Study of Atherosclerosis (MESA) and Coronary Artery Risk Development in Young Adults (CARDIA) study. Radiology. 2005;234(1):35–43. [DOI] [PubMed] [Google Scholar]
- 15.Subramanya V, Zhao D, Ouyang P, Ying W, Vaidya D, Ndumele CE, Heckbert SR, Budoff MJ, Post WS, Michos ED. Association of endogenous sex hormone levels with coronary artery calcium progression among post-menopausal women in the Multi-Ethnic Study of Atherosclerosis (MESA). J Cardiovasc Comput Tomogr. 2019;13(1):41–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tattersall MC, Gassett A, Korcarz CE, Gepner AD, Kaufman JD, Liu KJ, Astor BC, Sheppard L, Kronmal RA, Stein JH. Predictors of carotid thickness and plaque progression during a decade: the Multi-Ethnic Study of Atherosclerosis. Stroke. 2014;45(11):3257–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Stein JH, Korcarz CE, Hurst RT, Lonn E, Kendall CB, Mohler ER, Najjar SS, Rembold CM, Post WS, American Society of Echocardiography Carotid Intima-Media Thickness Task F. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: a consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. Endorsed by the Society for Vascular Medicine. J Am Soc Echocardiogr. 2008;21(2):93–111. [DOI] [PubMed] [Google Scholar]
- 18.Anderson-Bergman C icenReg: Regression Models for Interval Censored Data in R. 2017. 2017;81(12):23. [Google Scholar]
- 19.Shoenfeld D Partial residuals for the proportional hazards regression model. Biometrika. 1982;69(1):239–41. [Google Scholar]
- 20.Grambsch PM, Therneau TM. Proportional hazards tests and diagnostics based on weighted residuals. Biometrika. 1994;81(3):515–26. [Google Scholar]
- 21.Anderson-Bergman C icenReg: Regression Models for Interval Censored Data in R. J Stat Softw. 2017;81(12). [Google Scholar]
- 22.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–45. [PubMed] [Google Scholar]
- 23.Budoff MJ, Achenbach S, Blumenthal RS, Carr JJ, Goldin JG, Greenland P, Guerci AD, Lima JA, Rader DJ, Rubin GD, et al. Assessment of coronary artery disease by cardiac computed tomography: a scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention, Council on Cardiovascular Radiology and Intervention, and Committee on Cardiac Imaging, Council on Clinical Cardiology. Circulation. 2006;114(16):1761–91. [DOI] [PubMed] [Google Scholar]
- 24.Sarwar A, Shaw LJ, Shapiro MD, Blankstein R, Hoffmann U, Cury RC, Abbara S, Brady TJ, Budoff MJ, Blumenthal RS, et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc Imaging. 2009;2(6):675–88. [DOI] [PubMed] [Google Scholar]
- 25.Blaha M, Budoff MJ, Shaw LJ, Khosa F, Rumberger JA, Berman D, Callister T, Raggi P, Blumenthal RS, Nasir K. Absence of coronary artery calcification and all-cause mortality. JACC Cardiovasc Imaging. 2009;2(6):692–700. [DOI] [PubMed] [Google Scholar]
- 26.Sandesara PB, Mehta A, O’Neal WT, Kelli HM, Sathiyakumar V, Martin SS, Blaha MJ, Blumenthal RS, Sperling LS. Clinical significance of zero coronary artery calcium in individuals with LDL cholesterol >/=190mg/dL: The Multi-Ethnic Study of Atherosclerosis. Atherosclerosis. 2020;292:224–29. [DOI] [PubMed] [Google Scholar]
- 27.Blaha MJ, Cainzos-Achirica M, Dardari Z, Blankstein R, Shaw LJ, Rozanski A, Rumberger JA, Dzaye O, Michos ED, Berman DS, et al. All-cause and cause-specific mortality in individuals with zero and minimal coronary artery calcium: A long-term, competing risk analysis in the Coronary Artery Calcium Consortium. Atherosclerosis. 2020;294:72–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nasir K, Bittencourt MS, Blaha MJ, Blankstein R, Agatson AS, Rivera JJ, Miedema MD, Sibley CT, Shaw LJ, Blumenthal RS, et al. Implications of Coronary Artery Calcium Testing Among Statin Candidates According to American College of Cardiology/American Heart Association Cholesterol Management Guidelines: MESA (Multi-Ethnic Study of Atherosclerosis). J Am Coll Cardiol. 2015;66(15):1657–68. [DOI] [PubMed] [Google Scholar]
- 29.Wyman RA, Mays ME, McBride PE, Stein JH. Ultrasound-detected carotid plaque as a predictor of cardiovascular events. Vasc Med. 2006;11(2):123–30. [DOI] [PubMed] [Google Scholar]
- 30.Naqvi TZ, Lee MS. Carotid intima-media thickness and plaque in cardiovascular risk assessment. JACC Cardiovasc Imaging. 2014;7(10):1025–38. [DOI] [PubMed] [Google Scholar]
- 31.Gopal A, Nasir K, Liu ST, Flores FR, Chen L, Budoff MJ. Coronary calcium progression rates with a zero initial score by electron beam tomography. Int J Cardiol. 2007;117(2):227–31. [DOI] [PubMed] [Google Scholar]
- 32.Min JK, Lin FY, Gidseg DS, Weinsaft JW, Berman DS, Shaw LJ, Rozanski A, Callister TQ. Determinants of coronary calcium conversion among patients with a normal coronary calcium scan: what is the “warranty period” for remaining normal? J Am Coll Cardiol. 2010;55(11):1110–7. [DOI] [PubMed] [Google Scholar]
- 33.Stary HC, Chandler AB, Dinsmore RE, Fuster V, Glagov S, Insull W Jr., Rosenfeld ME, Schwartz CJ, Wagner WD, Wissler RW. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation. 1995;92(5):1355–74. [DOI] [PubMed] [Google Scholar]
- 34.Faxon DP, Fuster V, Libby P, Beckman JA, Hiatt WR, Thompson RW, Topper JN, Annex BH, Rundback JH, Fabunmi RP, et al. Atherosclerotic Vascular Disease Conference: Writing Group III: pathophysiology. Circulation. 2004;109(21):2617–25. [DOI] [PubMed] [Google Scholar]
- 35.Fernandez-Friera L, Penalvo JL, Fernandez-Ortiz A, Ibanez B, Lopez-Melgar B, Laclaustra M, Oliva B, Mocoroa A, Mendiguren J, Martinez de Vega V, et al. Prevalence, Vascular Distribution, and Multiterritorial Extent of Subclinical Atherosclerosis in a Middle-Aged Cohort: The PESA (Progression of Early Subclinical Atherosclerosis) Study. Circulation. 2015;131(24):2104–13. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


