Abstract
Transwomen may elect to pursue fertility preservation prior beginning hormonal treatment or proceeding with gender-affirming surgery. To date, there has been little research specifically investigating factors influencing fertility and preservation thereof among transwomen. Here, we review the case of a transwoman who engaged in genital tucking behavior presenting with severe oligospermia, and we review the literature regarding transgender fertility preservation and the role of the heat stress hypothesis with regards to this common behavior.
Keywords: Transgender, Male infertility, Cryopreservation, Gender-affirming surgery
Transwomen face many challenges related to gender-affirming treatment and surgery. During the decision-making process, these patients are faced with the possibility of permanent infertility when electing to undergo either hormonal modulation, orchiectomy, or both. For patients desiring fertility preservation, it is important to obtain sperm for cryopreservation prior to initiation of hormone treatment due to the potential for irreversible infertility in response to hormone modulation.1
Other factors, including thermal influences of sperm production have not been investigated in transwomen. Furthermore, the impact of specific gender-affirming behaviors on fertility in transwomen have also not been investigated. Herein, we report the case of a transwoman desiring fertility preservation who presented with cryptozoospermia in the setting of genital tucking behavior without prior hormone therapy. Cryptozoospermia is defined as severe oligospermia with sperm identified on microscopy only after centrifugation of the semen sample and examination of the concentrated debris pellet.
Case Report
A 27-year-old transwoman was referred to our clinic for evaluation of abnormal semen analysis. Initial semen analysis was obtained by her primary care physician (PCP) in preparation for sperm cryopreservation in advance of beginning hormonal therapy for gender transition. This initial semen analysis revealed severe cryptozoospermia, with minimal sperm identified only after centrifugation. After receiving the abnormal semen analysis results, her initial hormonal evaluation was within normal limits, and she was referred to our clinic (Table 1). Her past medical history was notable for depression and generalized anxiety disorder managed with bupropion, 450 mg daily, as well as attention-deficit/hyperactivity disorder (ADHD) managed with lisdexamfetamine, 40 mg daily, for 7 years. She consumed marijuana daily and felt she was unable to discontinue this when counseled on the possible impact of marijuana use on sperm concentration and motility.2 Furthermore, she regularly engaged in tucking, a behavior in which the external genitalia are concealed (Figure 1). The patient was therefore counseled to trial cessation of tucking and to obtain repeat baseline semen analysis for confirmation.
TABLE 1.
Initial Hormonal Evaluation by the Patient's Primary Care Physician; All Parameters Within Normal Limits
| FSH (mIU/mL) | 4.8 |
| LH (mIU/mL) | 4.4 |
| TSH (mIU/L) | 0.49 |
| T (ng/dL) | 408 |
| Free T (pg/mL) | 73.4 |
FSH, follicle-stimulating hormone; LH, luteinizing hormone; T, testosterone; TSH, thyroid stimulating hormone.
Figure 1.
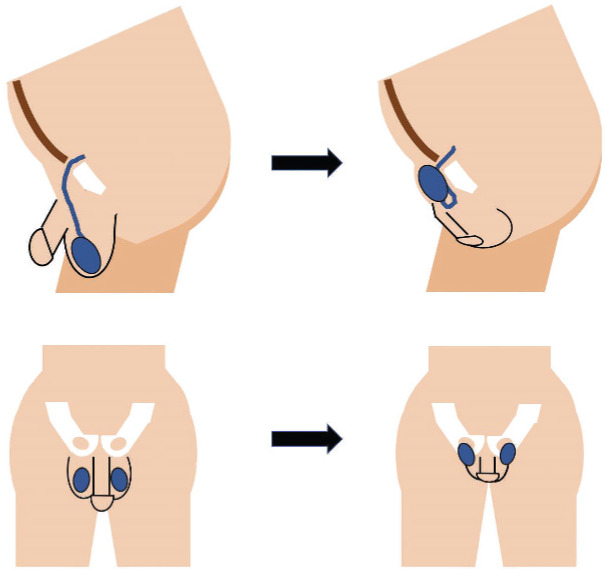
Illustration of genital tucking. Images on the left show the genitalia in the normal configuration, whereas images on the right show the position of the testis and penis during tucking.
Initial diagnostic evaluation in our clinic included repeat semen analysis, genetic testing, and karyotyping (Table 2). Her karyotyping was 46,XY and Y-chromosome microdeletions were not detected. The confirmatory semen analysis revealed a low sperm count, with 77 whole sperm identified, none of which demonstrated normal morphology; however, the ejaculate volume and pH were normal. At the time of her repeat semen analysis, she had stopped tucking for 5 weeks and reported a change in the appearance of her semen with increased opacification, but worsening gender dysphoria. The results of her repeat semen analysis were suspicious for an abnormality of sperm production, although the follicle-stimulating hormone (FSH) level was within normal limits.
TABLE 2.
Semen Analyses Performed in Our Clinic Before and After Intervention
| Pre-intervention | Post-intervention | |
|---|---|---|
| Semen volume (cc) | 5.7 | 4.3 |
| pH | 7.2 | 7.2 |
| Viability (%) | 15.4 | ** |
| Total Count (M) | 0.0 | 182.8 |
| Whole sperm (wet prep) | 77 | ** |
| Total motile (M) | 0.0 | 62.2 |
| Morphology (%) | 0.0 | 1 |
Data not collected or unavailable.
The ejaculate volume and pH were noted to be appropriate both before and after treatment. Before intervention, 77 sperm were identified on a wet mount slide, none of which showed normal morphology.
The patient was counseled on the potential impact of the tucking behavior on her subfertility and advised to continue cessation of the practice for 2 to 3 months prior to repeating her semen analysis. She continued marijuana use despite cessation counseling due to its positive role in treating her dysphoria and made no other known changes to medications or behaviors.
At the 3-month time point, a semen analysis was repeated (Table 2) and this revealed a return to near normal semen parameters. She was therefore able to successfully cryopreserve 8 vials of sperm. She was subsequently discharged from clinic and she elected to resume genital tucking at that time.
Discussion
The production of sperm is regulated by several concurrent mechanisms. These include both intrinsic and extrinsic factors.3 Thermal mechanisms have long been implicated as a potential regulator of this process.4,5 Most of this research arises from the study of infertility in men with varicocele. Studies in this population have shown that multiple factors may influence sperm production with thermal dysregulation as one important mediator.6
The primary thermal regulation of the testis is postulated to occur by countercurrent heat exchange between the gonadal artery and the pampiniform plexus.7 As blood at body temperature is distributed to the testicle, colder venous blood absorbs a portion of the arterial heat, thereby decreasing the temperature of the testicle. Testicular hyperthermia can be caused by either excess heat production or diminished venous cooling, which could be caused by a failure of the testis to move further away from the body during times of excess heat or venous pooling as in a varicocele.6,8,9
Increased intratesticular temperature is hypothesized to act on pathways of spermatogenesis through a variety of mechanisms. Heat-shock protein transcripts in the testes, along with other protective factors, have been demonstrated to increase in production during times of elevated temperature.10 In fact, this occurs even at body temperature, indicating that spermatogonia re quire a tightly regulated thermal environment to function optimally.10 In response to elevated temperature, hypoxia-inducible factor has also been shown to be generated.11 In conjunction with the Bax pathway, oxidative cellular stress is produced, and further leads to activation of common p53-mediated apoptotic pathways.10,11
Tucking is a behavior in which the external male genitalia are concealed, occurring among natal males wishing to diminish the outward expression of masculine phenotypic features.12 There is little data on this behavior, however one study notes a prevalence of about 70% among transwomen.13 Various approaches to this behavior exist, including the utilization of specific devices. In most methods, the testicles are secured in an elevated position, putting them closer to the abdomen and thereby theoretically increasing the intratesticular temperature. In some cases, the testicles are indeed placed adjacent to the inguinal canal. Further, the use of external devices to assist in concealment of the genitalia add a further layer of insulation, theoretically compounding the increase in intratesticular temperature. By the mechanisms addressed above, this is likely to induce cellular stress at the level of the spermatogonia and induce potential subfertility or infertility.
In our patient, the array of underlying causes for male factor infertility was considered. Obstruction was thought to be unlikely given a normal ejaculate volume and pH. The placement of the testicles upward near the inguinal canal may cause kinking or torsion of the spermatic cord and lead to ischemia. In this case we would expect to find pain or swelling associated with the act, which were not present in this patient, as well as resultant decreased androgen production and increased gonadotropin secretion. Hormonal and genetic abnormalities were not present, and it would not be reasonable for these issues to resolve spontaneously. We are therefore left with the hypothesis of abnormal sperm production. Given the chronologic relationship with heat stress of the testicles and the discontinuation thereof, the most likely explanation for our patient's subfertility is her tucking behavior leading to increased heat toxicity impairment of spermatogenesis.
There is a paucity of scientific literature regarding the impact of specific gender-affirming behaviors, such as tucking. Although this case represents a single incident, further research into the impact of these behaviors among transwomen desiring fertility preservation is warranted. In this case, abnormal parameters that prevented this patient from undergoing cryopreservation were reversed simply and without the need for invasive procedures, although it may come at the cost of temporarily worsening gender dysphoria. In common practice, transwomen desiring fertility preservation prior to gender-affirming surgery often undergo routine semen analysis in preparation for possible cryopreservation. Prior to semen analysis, and certainly in the event of discovering abnormal parameters, transwomen should be counseled on the potential for tucking to impact the normal regulatory mechanisms underlying sperm production and the option to discontinue this behavior prior to attempting cryopreservation.
Main Points.
Genital tucking is a common behavior among transwomen.
Elevation of the testes because of tucking may contribute to heat stress and consequent impairment of spermatogenesis.
Among transwomen desiring fertility preservation, counseling on the potential impact of genital tucking on spermatogenesis may be warranted, and discontinuation of the behavior has the potential to improve subfertility if identified.
References
- 1.Jiang DD, Swenson E, Mason M, et al. Effects of estrogen on spermatogenesis in transgender women. Urology. 2019;132:117–122. doi: 10.1016/j.urology.2019.06.034. [DOI] [PubMed] [Google Scholar]
- 2.Payne KS, Mazur DJ, Hotaling JM, Pastuszak AW. Cannabis and male fertility: a systematic review. J Urol. 2019;202:674–681. doi: 10.1097/JU.0000000000000248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Neto FT, Bach PV, Najari BB, et al. Spermatogenesis in humans and its affecting factors. Semin Cell Dev Biol. 2016;59:10–26. doi: 10.1016/j.semcdb.2016.04.009. [DOI] [PubMed] [Google Scholar]
- 4.Kandeel FR, Swerdloff RS. Role of temperature in regulation of spermatogenesis and the use of heating as a method for contraception. Fertil Steril. 1988;49:1–23. doi: 10.1016/s0015-0282(16)59640-x. [DOI] [PubMed] [Google Scholar]
- 5.Shiraishi K, Takihara H, Matsuyama H. Elevated scrotal temperature, but not varicocele grade, reflects testicular oxidative stress-mediated apoptosis. World J Urol. 2010;28:359–364. doi: 10.1007/s00345-009-0462-5. [DOI] [PubMed] [Google Scholar]
- 6.Shiraishi K, Matsuyama H, Takihara H. Pathophysiology of varicocele in male infertility in the era of assisted reproductive technology. Int J Urol. 2012;19:538–550. doi: 10.1111/j.1442-2042.2012.02982.x. [DOI] [PubMed] [Google Scholar]
- 7.Mieusset R, Bujan L. Testicular heating and its possible contributions to male infertility: a review. Int J Androl. 1995;18:169–184. doi: 10.1111/j.1365-2605.1995.tb00408.x. [DOI] [PubMed] [Google Scholar]
- 8.Lazarus BA, Zorgniotti AW. Thermoregulation of the human testis. Fertil Steril. 1975;26:757–759. [PubMed] [Google Scholar]
- 9.Dada R, Gupta NP, Kucheria K. Spermatogenic arrest in men with testicular hyperthermia. Teratog Carcinog Mutagen. 2003;(Suppl 1):235–243. doi: 10.1002/tcm.10050. [DOI] [PubMed] [Google Scholar]
- 10.Kim B, Park K, Rhee K. Heat stress response of male germ cells. Cell Mol Life Sci. 2013;70:2623–2636. doi: 10.1007/s00018-012-1165-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Velickovic LJ, Stefanovic V. Hypoxia and spermatogenesis. Int Urol Nephrol. 2014;46:887–894. doi: 10.1007/s11255-013-0601-1. [DOI] [PubMed] [Google Scholar]
- 12.Deutsch MB. Binding, packing, and tucking. UCSF Transgender Care website. https://transcare.ucsf.edu/guidelines/binding-packing-and-tucking. Published June 17, 2016. Accessed March 3, 2021.
- 13.Poteat T, Malik M, Cooney E. 2148 Understanding the health effects of binding and tucking for gender affirmation. J Clin Transl Sci. 2018;2(suppl 1):76. [Google Scholar]


