Abstract
Primary central nervous system Hodgkin's lymphoma (CNS-HL) is extremely rare. This current case report describes a 60-year-old male patient that presented with numbness of the left lower extremity and worsening headache. After a full range of investigations and a partial resection of the right cerebellum, external ventricular drainage reservoir placement and cranioplasty, he was diagnosed with primary CNS-HL. The patient was treated with 3 g/m2 methotrexate (intravenous [i.v.], once a day, day 1) and 1 g/m2 cytarabine (i.v., every 12 h, days 2 + 3), followed by anti-programmed cell death protein 1 antibodies (200 mg sintilimab, i.v., once a day, day 1, every 3 weeks). After six courses of treatment with intrathecal injections of 50 mg cytarabine (once a day, day 1) and 5 mg dexamethasone (once a day, day 1), there was no residual lesion on cranial magnetic resonance imaging. No significant drug-related adverse events were observed. The patient has been followed up every 3 months and no relapse has occurred.
Keywords: Primary central nervous system, Hodgkin's lymphoma, programmed cell death-1, treatment, prognosis
Introduction
Primary central nervous system Hodgkin's lymphoma (CNS-HL) is rare, with only two of 14 868 cases with CNS involvement reported in the recent literature.1 The incidence of CNS involvement was as low as 0.02%.2,3 Primary CNS-HL is extremely rare and only 22 cases have been reported in the literature.4 To date, the longest survival time reported was 9 years.5 There is no consensus on diagnosis, treatment and prognosis of primary CNS-HL because the incidence is so low that large-scale clinical trials cannot be carried out.
Case report
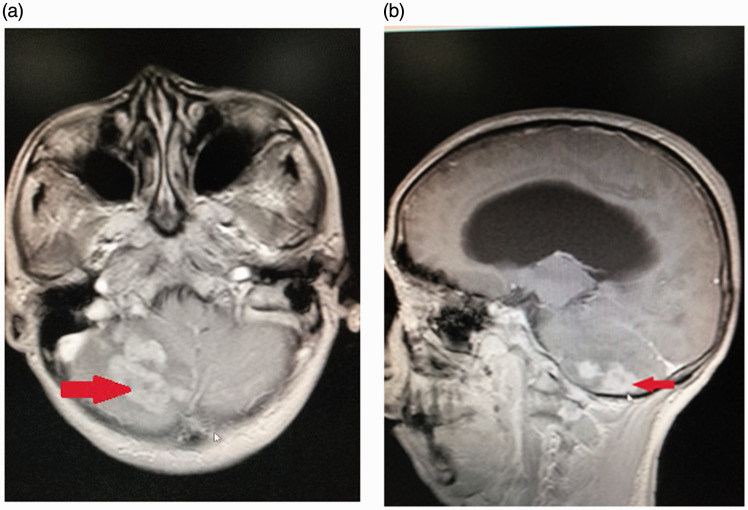
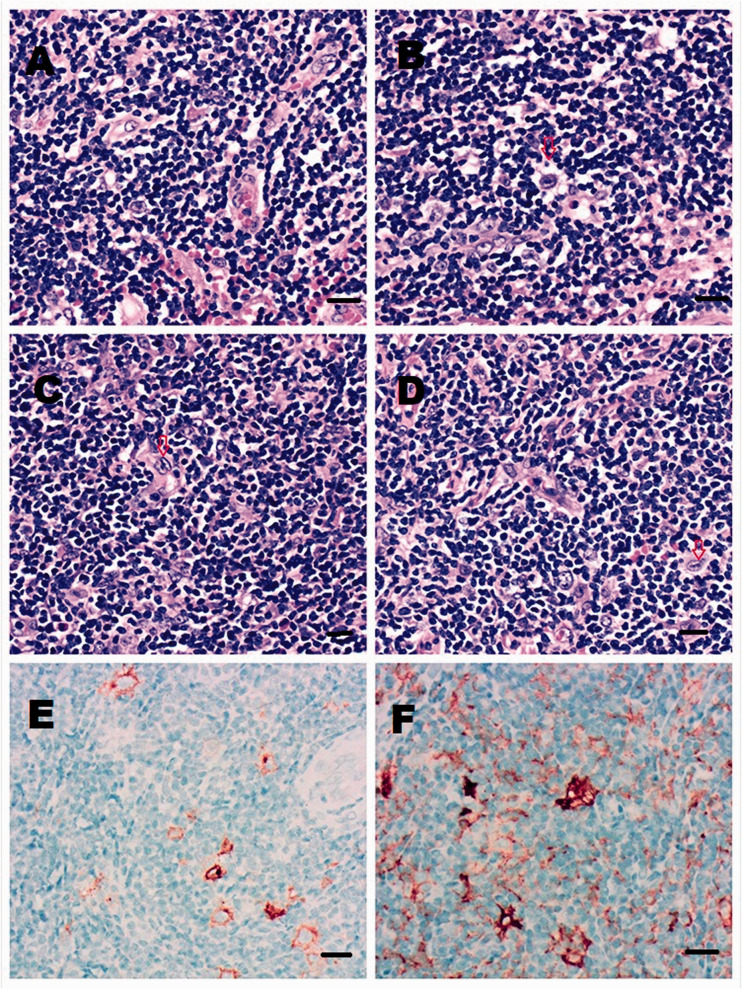
In February 2015, a 60-year-old male developed symptoms of a headache and presented to the Department of Neurosurgery, Fujian Medical University Union Hospital, Fuzhou, Fujian Province, China. Head computed tomography (CT) imaging showed obstructive hydrocephalus of unknown cause. Prior to the headache, the patient had no history of other diseases. His symptoms improved after he received mannitol (125 ml intravenous [i.v.], every 12 h, days 1–7) treatment to reduce the intracranial pressure. Subsequently, the patient continued to experience occasional painful headaches, but he did not seek further medical advice for a diagnosis. In October 2018, the patient presented to the Department of Neurosurgery, Fujian Medical University Union Hospital with numbness of the left lower extremity and worsening headache. Head CT imaging revealed an abnormal density shadow with supratentorial ventricular dilatation hydrops in the bilateral cerebellar hemispheres. Magnetic resonance imaging (MRI) revealed multiple nodules, masses and abnormal signals of Gyri in the bilateral cerebellar hemispheres, bilateral cerebellar oedema and hydrocephalus (Figure 1). Partial resection of the right cerebellum, external ventricular drainage reservoir placement and cranioplasty were performed. Pathological examination of the right cerebellar mass showed that there was lymphocyte infiltration and multiple foci of necrosis (Figure 2). There were large cells with small lymphocytes around and some of the cells had binucleate nucleoli. Reed–Sternberg (RS) cells were detected. Based on immunohistochemistry, in situ hybridization and T cell receptor gene rearrangement findings, the diagnosis of CNS-HL was made. The HL was mixed cellularity HL (MCHL) with a lot of monoclonal T cell infiltration. Immunohistochemical staining showed that the large cells were stained as follows: CD30+, cyclinD1+, MUM-1+, Pax5±, CD15±, Bob-1±, KI67+ (40%), CD20–, CD21–, CD10–, Oct-2– and ALK–. Small lymphoma cells were stained as follows: CD2+, CD3+, CD5+, CD43+, BCL-2+, TIA-1+, CD7±, BCL-6±, KI67+ (20–30%), CD20–, CD21–, CD56–, CD10–, GRB–, perforin–, TDT–, MPO– and SOX11–. The rate of positive immunostaining of programmed death-ligand 1 (PD-L1) was 15%. The peripheral blood Epstein-Barr virus (EBV)-DNA level was 1.26 x 104 copies/ml. EBV-encoded small RNAs (EBERs) staining was positive. The karyotype was 46,XY. No gene mutations were found for lymphoma hotspot genes by next generation sequencing.
Figure 1.
Magnetic resonance imaging of a 60-year-old male patient that presented with numbness of the left lower extremity and worsening headache revealed the following: multiple nodules, masses and abnormal signals of Gyri in the bilateral cerebellar hemispheres (a), bilateral cerebellar oedema (arrow) (a) and hydrocephalus (arrow) (b).
Figure 2.
Pathological examination of the right cerebellar mass of a 60-year-old male patient that presented with numbness of the left lower extremity and worsening headache revealed the following: (a) histological photomicrograph of the biopsy specimen stained with haematoxylin and eosin showing diffuse lymphocytic infiltration with necrosis; (b) scattered large cells with large nucleoli (arrow) within a background of diffuse small lymphocytes; (c) binucleate cells (arrow) within a background of diffuse small lymphocytes; (d) scattered large cells (arrow) within a background of diffuse small lymphocytes; (e) immunohistochemical staining showed the large cells stained for CD30; (f) immunohistochemistry showed that the rate of positive staining for programmed death-ligand 1 was 15%. Scale bar 50 µm. The colour version of this figure is available at: http://imr.sagepub.com.
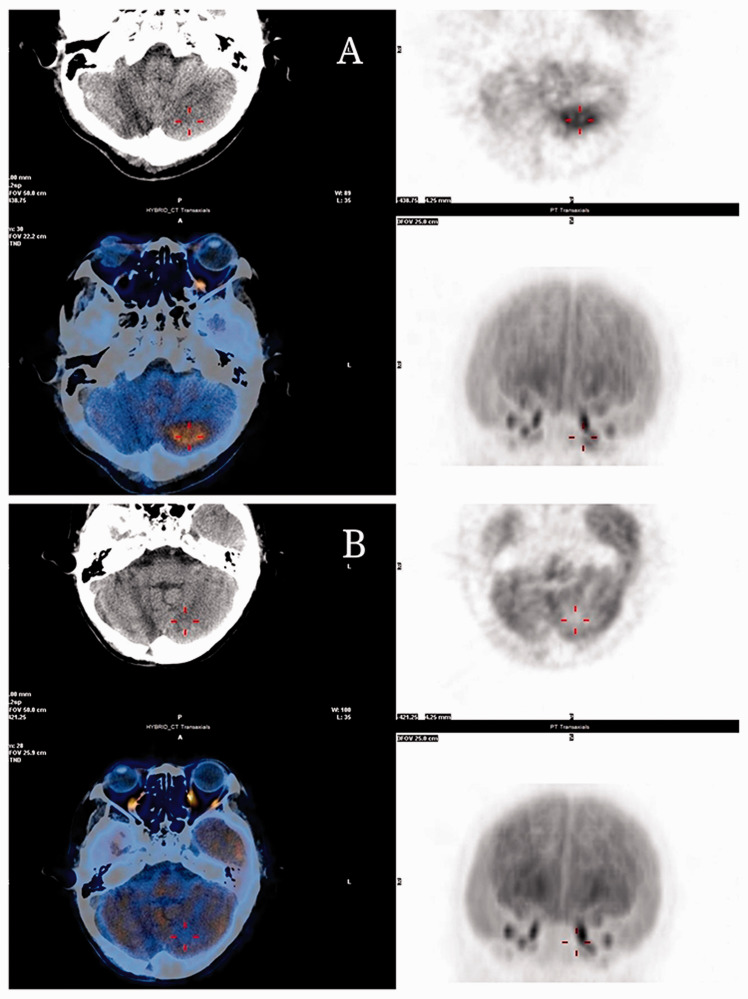
Postoperative positron emission tomography-CT (PET-CT) imaging revealed no hypermetabolic lesion caused by residual lymphoma. There was a hypermetabolic lesion at the left cerebellar tonsil after surgery, which was considered to be lymphoma infiltration (Deauville Score 5) (Figure 3a). There were no abnormal findings in the rest of the body (suvs 2.4 in liver background and Max 1.3 in mediastinal large blood pool). Cerebrospinal fluid (CSF) examination showed there were 7.2 × 107 nucleated cells: 97% were mononuclear cells and 3% were multinucleated cells. Pan's test was positive and the total protein level in the CSF was 866 mg/l. No tumour cells were found in the CSF.
Figure 3.
Positron emission tomography-computed tomography imaging findings of a 60-year-old male patient that presented with numbness of the left lower extremity and worsening headache revealed the following: (a) postoperative changes were seen in the right cerebellum. There was no hypermetabolic lesion caused by residual lymphoma in the operation area. There was a left cerebellar tonsillar hypermetabolic lesion, which was considered to be lymphoma infiltration (Deauville Score 5) (images taken on 28 February 2019); (b) compared with previous images taken on 28 February 2019, the right cerebellum showed post-operative changes. The left cerebellar tonsillar infiltrating lesion had receded (Deauville Score 1) (images taken on 19 July 2019). The colour version of this figure is available at: http://imr.sagepub.com.
The patient was diagnosed with a primary CNS classical Hodgkin's lymphoma (MCHL) with the help of a multidisciplinary treatment meeting. The patient was first treated with 3 g/m2 methotrexate (i.v., once a day, day 1) and 1 g/m2 cytarabine (i.v., every 12 h, days 2 + 3). Unfortunately, drug-induced acute renal injury and myelosuppression developed. Subsequently, the patient was treated with anti-programmed cell death protein 1 (PD-1) antibodies (200 mg sintilimab, i.v., once a day, day 1, every 3 weeks). After four courses of treatment, PET-CT imaging revealed no cell infiltration at the left cerebellar tonsil (Deauville Score 1) (Figure 3b). The CSF was normal and a complete response was achieved. Anti-PD-1 antibodies (200 mg sintilimab, i.v., once a day, day 1, every 3 weeks) was continued for two courses. During this period, lumbar puncture and intrathecal injections of 50 mg cytarabine (once a day, day 1) and 5 mg dexamethasone (once a day, day 1) were administrated for six courses. After six courses of treatment, there was no residual lesion on cranial MRI performed on 15 September 2019. No significant drug-related adverse events were observed. The patient has been followed up every 3 months and was last seen in December 2020. No relapse has occurred.
Discussion
Primary CNS-HL is extremely rare and there is no consensus on diagnosis, treatment and prognosis because the incidence is so low that large-scale clinical trials cannot be undertaken. EBV was found in 40–50% of the RS cells of patients, indicating that chronic immunosuppression may cause CNS-HL.4–7 The most common histological type of primary CNS-HL is MCHL (44%), followed by nodular sclerosis Hodgkin's lymphoma (31%).4,8 The clinical features of CNS-HL are similar to those of other intracranial tumours, cranial nerve palsy, headache, papillary oedema of the optic nerve, epilepsy, and sensory impairment. The current patient had a chronic course of headaches culminating in limb numbness. PET-CT examination revealed multiple intracranial lesions and no extracranial lesions. A craniotomy biopsy confirmed a classic MCHL. Histopathological staining showed many small T cells, which were positive for T cell receptor gene rearrangement but had no significant morphological abnormalities. The origin of these T cells was unknown. Studies have shown that an increase in the number of regulatory T (Treg) cells may impair antitumour immunity and promote tumour development and growth.9,10 Treg cells in HL tissue may inhibit activation of CD8 + and natural killer cells in response to tumour antigens by inhibiting the production of interleukin 2 (IL-2) and up-regulating the expression of Il-2ra-chain CD25, thus protecting RS cells from immune surveillance and clearance.11–14 It has been found that a high proportion of Treg cells in the microenvironment surrounding RS cells is associated with a significant reduction in disease-free survival time.15Whether excessive Treg cells may be have been a risk factor for CNS-HL in this current patient remains unknown.
The role of EBV infection in systemic and primary CNS-HL has also been proposed.4 The presence of EBV in RS cells is associated with an increase of Treg cells in the tumour microenvironment.5 Fortunately, the presence of EBV in patients with HL does not affect the survival of these patients.16 In this current patient, EBERs staining was positive in the biopsy. The peripheral blood EBV-DNA was 1.26 x 104 copies/ml. EBV infection may also be one of the risk factors for primary CNS-HL. Although there is no standard treatment for CNS-HL, surgical resection, local radiotherapy, whole brain radiotherapy, chemotherapy, radiotherapy in combination with chemotherapy, intrathecal injection and autotransplantation have all been used.17–21 Considering the possibility of serious side-effects from whole brain radiotherapy, the current patient received a chemotherapy regimen of high-dose methotrexate and cytarabine combined with lumbar puncture plus intrathecal chemotherapy. After a course of this treatment, the patient developed severe renal impairment and bone marrow suppression.
Anti-PD-1 antibody is now used to treat refractory and recurrent HL and it has been investigated as a first-line treatment for HL in several clinical studies.22 It has been reported that 82% of patients with classic HL expressed PD-L on the surface of tumour cells and EBV infection increased the expression of PD-L on RS cells in these patients.22,23 Engagement of PD-1 by its ligands PD-L1 or PD-L2 transduces a signal that inhibits T cell proliferation, cytokine production and cytolytic function.23 As the current case had PD-L1 immunostaining (15%) in the biopsy specimen, anti-PD-1 was used in the subsequent treatment regimen. Fortunately, this patient achieved complete remission after four treatment courses and no significant drug-related adverse events were observed.
In conclusion, primary CNS-HL is extremely rare. This current case report describes a male patient with an EBV-positive primary CNS-HL. These current findings suggest that EBV infection and overexpression of Treg cells might have been risk factors for primary CNS-HL in this patient. The patient achieved complete remission with anti-PD-1 antibody treatment and is currently under close follow-up observation. A better understanding of the role of EBV infection, Treg cells and PD-L1 in the pathogenesis of CNS-HL could lead to more effective treatments (such as immunotherapy) that are less toxic than conventional radiotherapy and chemotherapy.
Footnotes
Author contributions: H.F. and T.L. were mainly responsible for the overall diagnosis and treatment of the patient. H.F. drafted the manuscript and is in charge of revising the manuscript and submitting the revisions. T.L. was the head of the treatment team. W.H. and Y.C. were the patient’s residents. S.S. was in charge of the patient's surgery. L.C. and X.W. were in charge of the patient's pathological diagnosis. B.X. was in charge of the radiation therapy. J.S. took part in the multidisciplinary treatment of the patient and put forward some suggestions for reference.
Declaration of conflicting interest: The authors declare that there are no conflicts of interest.
Funding: This study was supported in part by grants from the following: Joint Funds for the Innovation of Science and Technology, Fujian Province (no. 2018Y9010; no. 2018Y9205); Fujian Provincial Health Technology Project (no. 2016-ZQN-29); Natural Science Foundation of Fujian Province (no. 2017J05132); Construction Project of Fujian Medical Centre of Haematology (no. Min201704); and the National and Fujian Provincial Key Clinical Specialty Discipline Construction Programme, China.
ORCID iD: Haiying Fu https://orcid.org/0000-0001-8695-2640
References
- 1.Gerstner ER, Abrey LE, Schiff D, et al. CNS Hodgkin lymphoma. Blood 2008; 112: 1658–1661. DOI: 10.1182/blood2008-04-151563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hwang CY, Song YJ, Kim DC, et al. Primary cerebellar Hodgkin's lymphoma. J Korean Neurosurg Soc 2007; 42: 149–152. [Google Scholar]
- 3.Iagi J, MacKenzie RG, Berinstein N. Primary Hodgkin's disease of the CNS in an immunocompetent patient:a case study and review of the literature. Neuro Oncol 2000; 2: 239–243. DOI: 10.1215/S1522851700000284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cecyn KZ, Chaves EM, Oliveira JS. Primary central nervous system involvement in classical Hodgkin’s lymphoma: Case report and review of the literature. J Blood Lymph 2017; 8: 196. DOI: 10.4172/21657831.1000196. [Google Scholar]
- 5.Szelemej PA, Bigder MG, Krcek J, et al . Treatment and long-term follow-up of primary CNS classical Hodgkin's lymphoma – A case report and review of the literature. Interdiscip Neurosurg 2017; 9: 30–33. DOI: 10.1016/j.inat.2017.02.002 [Google Scholar]
- 6.Massarweh S, Udden MM, Shahab I, et al. HIV-related Hodgkin's disease with central nervous system involvement and association with Epstein-Barr virus. Am J Hematol 2003; 72: 216–219. DOI: 10.1002/ajh.10288. [DOI] [PubMed] [Google Scholar]
- 7.Hirmiz K, Foyle A, Wilke D, et al. Intracranial presentation of systemic Hodgkin's disease. Leuk Lymphoma 2004; 45: 1667–1671. DOI: 10.1080/10428190410001673409. [DOI] [PubMed] [Google Scholar]
- 8.Gessi M, Kuchelmeister K, Kellner U, et al . Unusual clinic – pathological features in primary Hodgkin’s lymphomas of the central nervous system. Acta Neurochir (Wien) 2013; 155: 19–24. DOI: 10.1007/s00701-012-1535-6. [DOI] [PubMed] [Google Scholar]
- 9.Negi N, Das BK. CNS: Not an immunoprivilaged site anymore but a virtual secondary lymphoid organ. Int Rev Immunol 2018; 37: 57–68. DOI: 10.1080/08830185.2017.1357719. [DOI] [PubMed] [Google Scholar]
- 10.Ha TY. The role of regulatory T cells in cancer. Immune Netw 2009; 9: 209–235. DOI: 10.4110/in.2009.9.6.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Piccirillo CA, Shevach EM. Cutting edge: Control of CD8+ T cell activation by CD4+CD25+ immunoregulatory cells. J Immunol 2001; 167: 1137–1140. DOI: 10.4049/jimmunol.167.3.1137. [DOI] [PubMed] [Google Scholar]
- 12.Wolf AM, Wolf D, Steurer M, et al. Increase of regulatory T cells in the peripheral blood of cancer patients. Clin Cancer Res 2003; 9: 606–612. [PubMed] [Google Scholar]
- 13.Marshall NA, Christie LE, Munro LR, et al. Immunosuppressive regulatory T cells are abundant in the reactive lymphocytes of Hodgkin lymphoma. Blood 2004; 103: 1755–1762. DOI: 10.1182/blood-2003-07-2594. [DOI] [PubMed] [Google Scholar]
- 14.Alvaro T, Lejeune M, Salvadó MT, et al. Outcome in Hodgkin’s lymphoma can be predicted from the presence of accompanying cytotoxic and regulatory T cells. Clin Cancer Res 2005; 11: 1467–1473. DOI: 10.1158/1078-0432.CCR-04-1869. [DOI] [PubMed] [Google Scholar]
- 15.Schreck S, Friebel D, Buettner M, et al. Prognostic impact of tumour-infiltrating th2 and regulatory T cells in classical Hodgkin lymphoma. Hematol Oncol 2009; 27: 31–39. DOI: 10.1002/hon.878. [DOI] [PubMed] [Google Scholar]
- 16.Assis MC, Campos AH, Oliveira JS, et al. Increased expression of CD4+CD25+ FOXP3+ regulatory T cells correlates with Epstein-Barr virus and has no impact on survival in patients with classical Hodgkin lymphoma in Brazil. Med Oncol 2012; 29: 3614–3619. DOI: 10.1007/s12032-012-0299-4. [DOI] [PubMed] [Google Scholar]
- 17.Grimm S, Chamberlain M. Hodgkin's lymphoma: a review of neurologic complications. Adv Hematol 2011; 2011: 624578. DOI:10.1155/2011/624578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Galán L, Sánchez AC, Cantos B, et al. Central nervous system involvement in Hodgkin's lymphoma. Med Oncol 2011; 28: S505–S508. DOI: 10.1007/s12032-010-9692-z. [DOI] [PubMed] [Google Scholar]
- 19.Corti M, Fioti MF, Yampolsky C, et al. Central nervous system involvement in Hodgkin's lymphoma associated with Epstein-Barr virus in a patient with AIDS: case report and review of the literature. Braz J Infect Dis 2006; 10: 403–405. DOI: 10.1590/s1413-86702006000600009. [DOI] [PubMed] [Google Scholar]
- 20.Shet AS, Saba N, Rausch D, et al. Intra-cranial lesions in a patient with Hodgkin lymphoma. Leuk Lymphoma 2004; 4: 419–422. DOI: 10.1080/1042819031000139602. [DOI] [PubMed] [Google Scholar]
- 21.Martinez DL, Gujrati M, Geoffroy F, et al. Isolated CNS Hodgkin's lymphoma: implications for tissue diagnosis. CNS Oncol 2014; 3: 383–387. DOI: 10.2217/cns.14.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Panjwani PK, Charu V, DeLisser M, et al. Programmed death-1 Ligands PD-L1 and PD-L2 Show Distinctive and Restricted Patterns of Expression in Lymphoma Subtypes. Hum Pathol 2018; 71: 91–99. DOI: 10.1016/j.humpath.2017.10.029. [DOI] [PubMed] [Google Scholar]
- 23.Younes A, Ansell SM. Novel Agents in the Treatment of Hodgkin Lymphoma: Biological Basis and Clinical Results. Semin Hematol 2016; 53: 186–189. DOI: 10.1053/j.seminhematol.2016.05.011. [DOI] [PMC free article] [PubMed] [Google Scholar]