Abstract
This experiment evaluated the impacts of administering a bovine appeasing substance (BAS) to beef calves at weaning on their performance, physiological responses, and behavior during a 42-d preconditioning program. Eighty calves (40 heifers and 40 steers; 90% British × 10% Nellore) were weaned at 233 ± 2 d of age (day 0); ranked by sex, weaning age, and body weight (BW); and assigned to receive BAS (IRSEA Group, Quartier Salignan, France; n = 40) or placebo (diethylene glycol monoethyl ether; CON; n = 40). Treatments (5 mL) were topically applied to the nuchal skin area of each animal following dam separation. Within treatment, calves were allocated to one of eight drylot pens (four pens per treatment; pen being the experimental unit) and received a free-choice total mixed ration (TMR) from day 0 to 42, intake of which was assessed daily. Live behavior observations were conducted on days 1, 2, 4, 8, 16, and 32. Temperament was assessed and blood samples were collected via jugular venipuncture on days −21, 0, 3, 7, 14, 28, and 42. Hair samples were collected from the tail switch on days 0, 14, 28, and 42. Calves were vaccinated against bovine respiratory disease viruses on days −21 and 0. Average daily gain from day 0 to 42 did not differ between treatments (P = 0.57) but was greater (P = 0.05) in BAS vs. CON calves from day 0 to 28. Intake of TMR was greater (P = 0.05) during the first week for BAS vs. CON calves (treatment × week; P = 0.08). The mean proportion of calves feeding simultaneously and performance of social and play behaviors were greater (P ≤ 0.05) for BAS vs. CON calves. Escape attempts were greater (P < 0.01) for BAS vs. CON calves on day 1 (treatment × day; P = 0.03). Exit velocity was greater (P = 0.04) for CON vs. BAS calves on day 14 and tended (P = 0.10) to be greater for CON vs. BAS calves on day 7 (treatment × day; P = 0.03). Mean plasma concentrations of haptoglobin were greater (P = 0.02) in CON vs. BAS calves. Hair cortisol concentrations were greater (P = 0.05) in CON vs. BAS calves on day 14 (treatment × day; P = 0.03). Mean serum concentrations of antibodies against bovine viral diarrhea virus were greater (P = 0.02) in BAS vs. CON calves. Collectively, BAS administration to beef calves at weaning alleviated stress-induced physiological reactions, improved temperament evaluated via chute exit velocity, enhanced humoral immunity acquired from vaccination, and appeared to have accelerated adaptation to novel management scheme and environment.
Keywords: appeasing substance, beef cattle, behavior, growth, stress, weaning
Introduction
Beef cattle are inevitably exposed to psychological, physiological, and physical stressors associated with routine management throughout their productive lives (Cooke, 2017). Weaning is a stressor inherent to beef cattle management, the process of which stimulates adrenocortical and acute-phase protein responses that have immediate and long-term impacts on calf growth and immunity (Carroll and Forsberg, 2007). The weaning process also includes other stressful procedures, such as vaccination, transport, and exposure to new environments (Cooke, 2017). Hence, strategies to alleviate the stress elicited by the weaning process are warranted to promote calf performance and welfare in cow–calf and subsequent feeding operations.
One strategy to consider is the use of appeasing pheromones, initially discovered in swine and shown to reduce the agonistic behavior of piglets (McGlone and Anderson, 2002; Archunan et al., 2014). Pheromones are species-specific chemicals that are released from one individual to induce both a behavioral and physiological response in a conspecific (Liberles, 2014). In cattle, the synthetic analog of the appeasing pheromone is based on a mixture of fatty acids, reproducing the composition of the natural substance (Pageat, 2001; Cooke et al., 2020). This synthetic analog (bovine appeasing substance [BAS]) is expected to have calming effects, improving cattle welfare and productivity upon stressful procedures. Accordingly, dairy cows administered BAS had greater milk yield and less somatic cell count during the transition from barn to pasture housing, suggesting a reduced stress response to social, management, and dietary changes (Osella et al., 2018).
Our group recently demonstrated that BAS administration to beef calves upon weaning alleviated the resultant acute-phase protein response and improved average daily gain (ADG) during a 45-d postweaning period (Cappellozza et al., 2020; Cooke et al., 2020). These studies were novel and support the use of BAS to improve calf welfare and growth during a preconditioning program. Research investigating the effects of BAS use in beef cattle, however, is still limited and warranted to further understand its biological and behavioral effects. Based on this rationale, we hypothesized that the administration of BAS to beef calves will reduce the stress caused by weaning, improving subsequent social, health, and growth responses. To test this hypothesis, this experiment evaluated the impacts of BAS administration at weaning on behavior, productive, and physiological responses of beef calves during a 42-d preconditioning period.
Materials and Methods
This experiment was conducted at the Texas A&M – Beef Cattle Systems (College Station, TX). All animals were cared for in accordance with acceptable practices, and experimental protocols reviewed and approved by the Texas A&M AgriLife Research, Agriculture Animal Care and Use Committee (#2019-019A).
Animals and treatments
Eighty calves (90% British × 10% Nellore; being 40 heifers and 40 steers) born at the Texas A&M – McGregor Research Center (McGregor, TX) were used in this experiment. On the day of weaning (day 0), calves were separated from their dams, weighed, evaluated for temperament score (chute score and exit velocity; Cooke, 2014), and loaded into a double-deck commercial livestock trailer (Legend 50′ [15 m] cattle liner; Barrett LLC., Purcell, OK) at the McGregor Research Center. Calves were transported for 170 km (2 h road transport) to Beef Cattle Systems, where they were immediately weighed upon unloading. Calves were then ranked by sex, arrival body weight (BW; 185 ± 3 kg), weaning age (233 ± 2 d), and temperament score and assigned to receive BAS (IRSEA Group, Quartier Salignan, France; n = 40) or placebo (diethylene glycol monoethyl ether; CON; n = 40). This placebo, also known as Transcutol (Sigma-Aldrich, St. Louis, MO), is the excipient used for the BAS active ingredients. The BAS active ingredient is based on a proprietary mixture of fatty acids, including palmitic, oleic acid, and linoleic acids, added at 1% of the excipient and estimated to remain in treated animals for 15 d according to the manufacturer. Calves were immediately segregated by treatment into one of two groups and processed again for treatment administration, with CON calves being processed first to avoid cross-contamination during treatment application. Treatments (5 mL) were applied topically to the nuchal skin area of each animal, according to Cooke et al. (2020) for dose and route of administration. Upon segregation, treatment groups had no physical contact, and calves within each treatment group were ranked again by the aforementioned variables and allocated to one of eight drylot pens (10 calves per pen; 5 heifers and 5 steers per pen; 4 pens per treatment) with an empty pen maintained between pens of differing treatments to preserve distance and avoid any cross-contamination among treatment groups. The dimension of each pen was 20 × 10 m, with 6 × 6 m of shaded area, and 8 m of linear bunk space. Pens were enclosed with galvanized wired panels (10 × 10 cm mesh) in addition to original metal pipe fencing. Calves from the same treatment and housed in adjacent pens had no physical contact with each other due to the fencing structure, which resulted in a 50-cm gap between all adjacent pens.
On day −21 relative to weaning, calves received vaccination against respiratory viruses (Triangle 5; Boehringer Ingelheim Animal Health USA Inc., Duluth, GA) and Clostridium (Covexin 8; Merck Animal Health, Omaha, NE). Prior to transport on day 0, calves were revaccinated against respiratory viruses (Titanium 5; Elanco Animal Health, Greenfield, IN) and Clostridium (Covexin 8; Merck Animal Health), and received a pour-on anthelmintic (Dectomax; Zoetis). Calves had free-choice access to water and a total mixed ration (TMR) from day 0 to 42 (Table 1). The TMR was offered once daily (0800 hours), in a manner to yield 10% residual orts (as-fed basis; Colombo et al., 2019).
Table 1.
Composition and nutritional profile of the TMR offered for ad libitum consumption to calves during the experiment
| Item | Component |
|---|---|
| Composition, as-fed basis | |
| Cracked corn, % | 31.8 |
| Dried distillers grains, % | 30.0 |
| Alfalfa hay, % | 28.8 |
| Liquid molasses, % | 7.0 |
| Mineral mix1, % | 2.4 |
| Nutritional profile,2 dry matter basis | |
| Net energy for maintenance, Mcal/kg | 1.74 |
| Net energy for gain, Mcal/kg | 1.13 |
| Total digestible nutrients, % | 72.0 |
| Neutral detergent fiber, % | 30.0 |
| Acid detergent fiber, % | 21.1 |
| Crude protein, % | 16.4 |
1Containing 14% Ca, 7% P, 13% NaCl, 0.27% K, 0.4% Mg, 0.25% Cu, 0.003% Se, 0.99% Zn, 90.91 IU/kg of vitamin A, 9.09 IU/kg of vitamin D3, and 0.045 IU/kg of vitamin E (Purina Animal Nutrition, Shoreview, MN).
2Based on wet chemistry procedures by a commercial laboratory (Dairy One Forage Laboratory, Ithaca, NY). Calculations for net energy for maintenance and gain used the equations proposed by the NRC (2000).
Sampling
Samples of TMR ingredients were collected weekly, pooled across weeks, and analyzed for nutrient content (Dairy One Forage Laboratory, Ithaca, NY, USA). Calf unshrunk BW was recorded on day 0 prior to and after transport, and on days 3, 7, 14, 21, 28, 35, 42, and 43 before TMR feeding of the day. Calf initial and final BW were calculated, respectively, according to the average of two BW recorded on day 0, and the average of BW recorded on days 42 and 43. Calf ADG was calculated using initial and final BW. Moreover, the growth rate of each animal was modeled by linear regression of BW against sampling days, and each regression coefficient was used as an individual growth response. Intake of TMR from each pen was evaluated from day 0 to 42, by collecting and weighing offered and non-consumed TMR daily (0700 hours). Samples of offered and non-consumed TMR were dried for 96 h at 50 °C in forced-air ovens for dry matter calculations. Daily TMR intake of each pen was divided by the number of calves within each pen and expressed as kg/calf daily. Total BW gain and TMR intake of each pen were used for feed efficiency (G:F) calculations. Calves were observed daily for sickness and bovine respiratory disease (BRD) symptoms according to the DART system (Zoetis) as described by Sousa et al. (2019).
Blood samples were collected on days −21, 0 (prior to transport), 3, 7, 14, 21, 28, 35, and 42 via jugular venipuncture from each calf into commercial blood collection tubes (one tube per calf, with 10 mL of blood collected in each tube) containing freeze-dried sodium heparin (Vacutainer, 10 mL; Becton Dickinson, Franklin Lakes, NJ) for plasma collection. Another blood sample was also collected from each calf (one tube per calf, 10 mL of blood) on days −21, 0 (prior to transport), 14, 28, and 42 into commercial blood collection tubes with no additive (Vacutainer, 10 mL; Becton Dickinson) for serum collection. A total of 140 mL of blood was collected from each calf during the experimental period. Hair samples were collected from the tail switch for the analysis of hair cortisol concentrations (Schubach et al., 2017) on days 0 (prior to transport), 14, 28, and 42. Within each sampling, hair was collected from an area that had not been previously sampled. Hair was collected using scissors as close to the skin as possible and the hair material closest to the skin (1 cm of length and 100 mg of weight). Calves were fitted with a pedometer (HJ-321; Omron Healthcare, Inc., Bannockburn, IL, USA) upon arrival on day 0 to assess physical activity, which was placed inside a polyester patch (HeatWatch II; CowChips, LLC, Manalapan, NJ, USA) fixed behind their right shoulder. Pedometers had the capability to store daily data for seven consecutive days and remained on calves for the duration of the experiment. Pedometer results were recorded when calves were processed for sampling events as in Schubach et al. (2017), but discarding data from days of sampling to eliminate the confounding effects of gathering, handling, and processing on physical activity.
Calf temperament was assessed via chute score and exit velocity (Cooke, 2014) on day 0 (prior to transport), and then on days 3, 7, 14, 28, and 42. The chute used for this experiment was a hydraulic Silencer Chute (Commercial Pro model; Moly Manufacturing, Lorraine, KS). Chute score responses were discarded and not utilized for treatment comparison due to the physical characteristics of the squeeze chute, which completely immobilized calves and prevented proper behavior assessment during chute restraining (Cooke et al., 2019). Calf behavior in the home pen was recorded using live behavior observations on days 1, 2, 4, 8, 16, and 32. Two 2.5-m-tall observation decks were placed 10 m away from the pens in a pasture behind the pens where calves were housed. Using 10-min instantaneous scan sampling (48 scans per pen per day; Mitlöhner et al., 2001), the total number of calves standing, walking, lying, and eating within a pen was recorded (Table 2). Focal observations were used to measure the frequency of social behaviors for 10 min between scan samples (Daigle et al., 2017, 2018). Each pen was continuously observed with focal observations once every 40 min in a predetermined order resulting in a total of 120 min of continuous observation per pen for each observation day. The observation order was rotated on each observation day to ensure all pens were observed across all the time points throughout the experiment. To characterize herd synchrony, the Shannon–Wiener Diversity Index (Cronin and Ross, 2019) was calculated for each 10-min instantaneous scan observation for each pen and averaged by treatment.
Table 2.
Description of behaviors recorded from calves housed in receiving pens
| Behavior | Definition |
|---|---|
| Herd behavior | |
| Lying | Calf is recumbent, not supported by legs |
| Feeding | Calf has head in feed bunk |
| Standing | Calf is upright and supported off ground by legs |
| Walking | Movement of calf’s legs, resulting in a change in space |
| Social behavior | |
| Allogroom | Licking movements by one calf carried out on the body of another. Characterized by repetitive back-and-forth head movements performed by the actor in direct contact with the reactor. Once the actor stops grooming the reactor for more than 10 s, the bout is finished |
| Bunk displacement | Butt or push from one calf results in the complete withdrawal of the head of another calf from feed bunk |
| Escape attempt | Calf attempts to leave pen: jump, dig under, go through, or climb fence |
| Headbutt | Head of the calf connects with the body of another |
| Mounting | Calf positions body on top of another subject’s topline |
Laboratorial analyses
Feed samples
All samples were analyzed by wet chemistry procedures for concentrations of crude protein (method 984.13; AOAC, 2006), acid detergent fiber (method 973.18 modified for use in an Ankom 200 fiber analyzer, Ankom Technology Corp., Fairport, NY; AOAC, 2006), and neutral detergent fiber using a-amylase and sodium sulfite (Van Soest et al., 1991; modified for use in an Ankom 200 fiber analyzer, Ankom Technology Corp.). Calculations for net energy for maintenance and gain used the equations proposed by NRC (2000). Nutritional profile of TMR is described in Table 1.
Plasma and serum samples
After collection, all blood samples were placed immediately on ice, centrifuged (2,500 × g for 30 min; 4 °C) for plasma or serum harvest, and stored at −80 °C on the same day of collection. Plasma samples were analyzed for cortisol (radioimmunoassay kit #07221106, MP Biomedicals, Santa Ana, CA; Colombo et al., 2019), nonesterified fatty acids (NEFA; colorimetric kit HR Series NEFA-2; Wako Pure Chemical Industries Ltd. USA, Richmond, VA), β-hydroxybutyrate (BHBA; colorimetric kit #H7587; Pointe Scientific, Inc.), insulin-like growth factor I (IGF-I; human-specific enzyme-linked immunoassay kit SG100; R&D Systems Inc., Minneapolis, MN; Cooke et al., 2012a), and haptoglobin concentrations (Cooke and Arthington, 2013). The intra- and inter-assay coefficient of variation (CV) were, respectively, 8.4% and 7.9% for cortisol, 2.8% and 6.8% for haptoglobin, 4.5% and 5.8% for NEFA, 4.1% and 8.2% for BHBA, and 2.4% and 5.5% for IGF-I.
Serum samples collected on days −21, 0, 14, 28, and 42 were analyzed for antibodies against BRD viruses (Gonda et al., 2012) using enzyme-linked immunoassay: bovine respiratory syncytial virus (BRSV; BRSV Ab #P00651-2; IDEXX Switzerland AG, Liebefeld-Bern, Switzerland), bovine herpesvirus-1 (BHV-1; BHV-1 Ab #99-41459; IDEXX), bovine viral diarrhea viruses types I and II (BVDV; BVDV Ab #99-44000; IDEXX), and parainfluenza-3 virus (PI3; PI3 Ab #P0652-2; IDEXX). The intra- and inter-assay CV were, respectively, 2.0% and 2.2% for BRSV, 4.8% and 7.6% for BHV-1, 1.8% and 2.3% for BVDV, and 4.8% and 2.4% for PI3.
Hair samples
Cortisol was extracted from hair samples as in Moya et al. (2013). Briefly, hair samples were cleaned with lukewarm water (37 °C) for 30 min and dried at room temperature for 24 h. Hair samples were then washed twice with isopropanol, dried at room temperature for 120 h, and ground in a 10-mL stainless steel milling cup with a 12-mm stainless steel ball (Retsch Mixer Mill MM400 ball mill; Retsch, Hannover, Germany) for 3 min at a frequency of 25 repetitions/s; 20 mg of ground hair and 1 mL of methanol were combined into a 7-mL glass scintillation vial, sonicated for 30 min, and incubated for 18 h at 50 °C and 100 rpm for steroid extraction. Following incubation, 0.8 mL of methanol was transferred to a 2-mL microcentrifuge tube and evaporated to dryness under a stream of compressed air. Samples were reconstituted in 100 μL of the phosphate-buffered saline supplied with an enzyme-linked immunoassay cortisol kit (Salimetrics Expanded Range, High Sensitivity 1-E3002, State College, PA) and stored at −80 °C. Samples were analyzed for cortisol concentrations using the aforementioned enzyme-linked immunoassay kit, whereas the intra- and inter-assay CV were 6.8% and 7.2%, respectively.
Statistical analysis
All data were analyzed using pen as an experimental unit and Satterthwaite approximation to determine the denominator degrees of freedom for tests of fixed effects. Data were tested for normality and homogeneity of variance using the Shapiro–Wilk and Levene tests, respectively. Pedometer results and live behavioral observations required log-transformation to achieve normality (W > 0.90) and homogeneity (P ≥ 0.10), and back-transformed to original units for description. Performance, physiological, temperament, and pedometer responses were analyzed with the MIXED procedure of SAS (SAS Inst. Inc., Cary, NC) using pen(treatment) and calf(pen × sex) as random variables, but for TMR intake and G:F that used pen(treatment) as the random variable. Model statements contained the fixed effects of treatment, day, and the treatment × day interaction, with calf sex (not for TMR and G:F data) as an independent variable. Plasma, temperament, and hair variables were analyzed using results from day 0 as an independent covariate in each respective analysis. Serum results from days −21 and 0 were averaged and used as an independent covariate in each respective analysis. The specified term for all repeated statements was day, with pen(treatment) as the subject for TMR intake and calf(pen × sex) as subject for all other analyses. The covariance structure utilized was autoregressive which provided the best fit for these analyzes according to the Akaike information criterion. All behavioral observation data were analyzed using the GLIMMIX procedure of SAS (SAS Inst. Inc.) using similar models as TMR intake. All results are reported as least square means, or covariately adjusted least square means when the model contained independent variables, and separated using least square differences. Significance was set at P ≤ 0.05, and tendencies were determined if P > 0.05 and ≤ 0.10. Results are reported according to the effect of treatment if no interactions were significant or according to the highest-order interaction detected.
Results
Performance responses
As designed, calf weaning age and initial BW were similar (P ≥ 0.81) between treatments (Table 3). ADG and final BW did not differ (P ≥ 0.57) between BAS and CON calves (Table 3). No treatment effects were also detected (P ≥ 0.41) when calf BW and growth rate were analyzed according to individual samplings (Figure 1). Nonetheless, growth rate modeled from day 0 to 28 was greater (P = 0.05) in BAS vs. CON calves (1.004 vs. 0.850 kg/d, respectively; SEM = 0.057) and did not differ (P = 0.23) from day 28 to 42 between treatments (1.81 vs. 1.90 kg/d, respectively; SEM = 0.08).
Table 3.
Performance responses during a 42-d preconditioning program of beef calves receiving BAS (n = 40) or CON (n= 40) on the day of weaning (day 0)1
| Item | CON | BAS | SEM | P-value |
|---|---|---|---|---|
| Weaning age | 233 | 234 | 2 | 0.81 |
| Growth parameters2 | ||||
| Initial BW (day 0), kg | 184.8 | 185.1 | 3.6 | 0.95 |
| Final BW (day 42), kg | 228.4 | 230.6 | 4.2 | 0.71 |
| ADG, kg/d | 1.04 | 1.08 | 0.05 | 0.57 |
| TMR intake (dry matter),3 kg/d | 6.43 | 6.45 | 0.10 | 0.94 |
| G:F,4 g/kg | 157 | 165 | 6.7 | 0.45 |
1Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) on day 0. Treatments (5 mL) were applied topically to the nuchal skin area of each animal.
2Calf initial and final BW were calculated, respectively, according to the average of two BW recorded on day 0, and the average of BW recorded on days 42 and 43. ADG was calculated using the initial and final BW.
3Calves received a TMR for ad libitum consumption from day 0 to 42. Feed intake was recorded daily from day 0 to 42 by measuring offer and refusals from each pen, divided by the number of calves within each pen, and expressed as kg per calf/d.
4G:F was calculated using total BW gain (in grams) and total TMR intake (kg of dry matter) of each pen during the experimental period.
Figure 1.
BW during a 42-d preconditioning program of beef calves receiving BAS (n = 40) or CON (n = 40) on the day of weaning (day 0). Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) topically to their nuchal skin area. The growth rate of each animal was modeled by linear regression of BW against sampling days, and each regression coefficient was used as an individual response. No treatment differences (P ≥ 0.41) in growth rate (1.15 vs. 1.21 kg/d for CON and BAS, respectively; SEM = 0.05) or BW were noted.
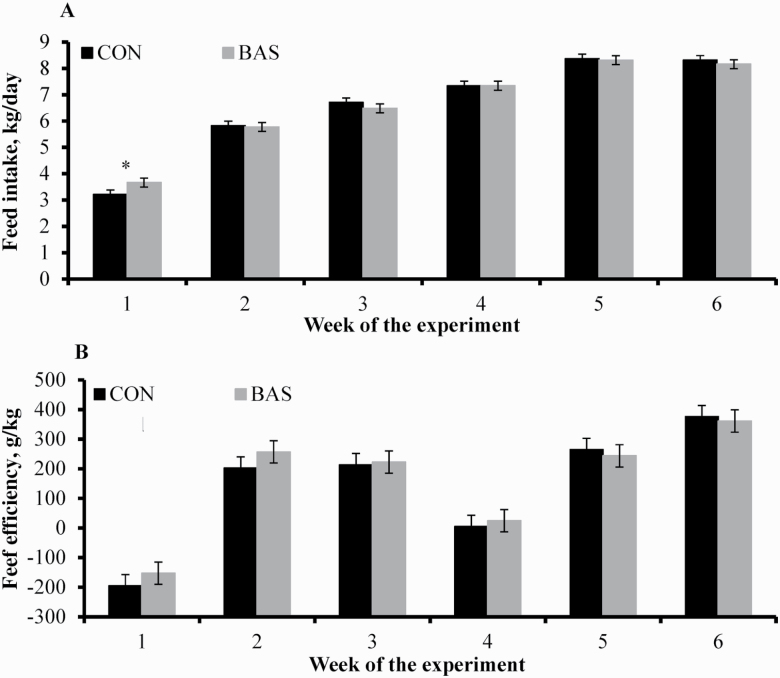
Mean daily TMR intake did not differ (P = 0.94) between treatments (Table 3), and no treatment × day interaction was noted (P = 0.29) for this variable. When evaluated on a weekly basis, TMR intake was greater (P = 0.05) during the first week for BAS vs. CON calves and similar (P ≥ 0.44) from week 2 to 6 (Figure 2; treatment × week interaction, P = 0.08). No treatment effects were detected (P ≥ 0.39) for the overall G:F (Table 3) or G:F evaluated on a weekly basis (Figure 2). No incidence of morbidity or mortality was observed during the experiment.
Figure 2.
Feed intake (A) and efficiency (B) during a 42-d preconditioning program of beef calves receiving BAS (n = 40) or CON (n = 40) on the day of weaning (day 0). Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) topically to their nuchal skin area. Calves received a TMR for ad libitum consumption. Intake was recorded daily by measuring offer and refusals from each pen, divided by the number of calves within each pen, and expressed as kg per calf/d (dry matter basis). G:F was calculated using total BW change divided by the total feed intake of each pen weekly. A tendency for treatment × week interaction was detected (P = 0.08) for feed intake, whereas no treatment nor treatment × week interaction was detected (P ≥ 0.39) for G:F. Within days: *P = 0.05.
Physiological responses
No treatment effects were detected (P ≥ 0.27) for plasma concentrations of NEFA, BHBA, cortisol, and IGF-I (Table 4). Mean plasma concentrations of haptoglobin were greater (P = 0.02) in CON vs. BAS calves (0.339 and 0.248 ng/mL, respectively; SEM = 0.028), although differences were mostly noted during the initial 14 d of the experiment (treatment × day interaction, P = 0.49; Figure 3). A treatment × day interaction (P = 0.03) was detected for hair cortisol concentrations, which were greater (P = 0.05) in CON vs. BAS on day 14, but did not differ (P ≥ 0.36) on days 28 and 42 (Figure 3). Day effects were detected (P < 0.01) for all plasma variables reported herein (Table 5).
Table 4.
Physiological responses during a 42-d preconditioning program of beef calves receiving BAS (n = 40) or CON (n = 40) on the day of weaning (day 0)1
| Item | CON | BAS | SEM | P-value |
|---|---|---|---|---|
| Plasma hormones and metabolites2 | ||||
| Cortisol, ng/mL | 27.0 | 24.6 | 1.4 | 0.27 |
| BHBA, mmol/L | 0.435 | 0.428 | 0.013 | 0.70 |
| IGF I, ng/mL | 70.3 | 77.1 | 6.3 | 0.47 |
| NEFA, μEq/L | 0.175 | 0.183 | 0.011 | 0.62 |
| Serum antibodies against respiratory viruses3 | ||||
| PI-3 virus | 0.598 | 0.666 | 0.032 | 0.09 |
| BRSV | 1.41 | 1.49 | 0.04 | 0.08 |
| BVDV types I and II | 1.15 | 1.31 | 0.05 | 0.02 |
| BHV-1 | 2.57 | 2.63 | 0.05 | 0.37 |
1Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) on day 0. Treatments (5 mL) were applied topically to the nuchal skin area of each animal.
2Blood samples were collected on days 0, 3, 7, 14, 28, and 42 for plasma extraction. Results from day 0 were used as a covariate in each respective analysis.
3Calves received vaccination against respiratory viruses on day −21 (Triangle 5; Boehringer Ingelheim Animal Health USA Inc., Duluth, GA) and day 0 (Titanium 5; Elanco Animal Health, Greenfield, IN). Blood samples were collected on days −21, 0, 14, 28, and 42 for serum extraction. Serum samples were analyzed and results were expressed as sample:positive control ratio as in Gonda et al. (2012). Results obtained on days −21 and 0 were averaged and used as a covariate in each respective analysis.
Figure 3.
Concentrations of plasma haptoglobin (A) and cortisol in hair from the tail switch (B) from beef calves receiving BAS (n = 40) or CON (n = 40) on the day of weaning (day 0). Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) topically to their nuchal skin area. Blood samples were collected on days 0, 3, 7, 14, 28, and 42. Hair samples were collected on days 0, 14, 28, and 42 as in Schubach et al. (2017). Results from day 0 were used as a covariate in each respective analysis. Mean plasma concentrations of haptoglobin were greater (P = 0.02) in CON vs. BAS calves. A treatment × day interaction was detected (P = 0.03) for hair cortisol concentrations. Within day, *P = 0.05.
Table 5.
Serum concentrations of antibodies against PI-3 virus, BRSV, BVDV types I and II, and BHV-1 and plasma concentrations of cortisol (ng/mL), BHBA (mmol/L), IGF-I (ng/mL), haptoglobin (mg/dL), and NEFA (μEq/L) in beef calves during a 42-d preconditioning program1,2
| Serum antibodies against respiratory viruses | Plasma hormones and metabolites | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Day | PI3 | BRSV | BVDV | BHV | Cortisol | BHBA | IGF-I | Haptoglobin | NEFA |
| −21 | 0.082e | 0.526d | 0.034e | 0.286d | — | — | — | — | — |
| 0 | 0.380d | 1.52a | 0.581d | 2.33c | 33.9a | 0.301d | 26.8f | 0.098c | 0.361a |
| 3 | — | — | — | — | 24.7cd | 0.409c | 37.5e | 0.441a | 0.384a |
| 7 | — | — | — | — | 26.9bc | 0.457b | 46.8d | 0.484a | 0.155b |
| 14 | 0.691a | 1.53a | 1.309a | 2.67a | 26.8bc | 0.404c | 75.2c | 0.331b | 0.116c |
| 28 | 0.645b | 1.44b | 1.241b | 2.59b | 26.2cd | 0.390c | 98.1b | 0.119c | 0.124bc |
| 42 | 0.559c | 1.38c | 1.149c | 2.52b | 23.9d | 0.491a | 110a | 0.079c | 0.143b |
| SEM | 0.022 | 0.036 | 0.035 | 0.041 | 1.4 | 0.012 | 4.8 | 0.035 | 0.016 |
| P-value | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 | <0.01 |
1Within columns, values with different superscripts differ (P ≤ 0.05). Serum antibodies were reported as in Gonda et al. (2012).
2Blood samples were collected on days −21, 0, 3, 7, 14, 28, and 42 relative to weaning (day 0). Calves received vaccination against respiratory viruses on days −21 (Triangle 5; Boehringer Ingelheim Animal Health USA Inc., Duluth, GA) and 0 (Titanium 5; Elanco Animal Health, Greenfield, IN).
Calves that received BAS had greater (P ≤ 0.05) mean serum concentrations of antibodies against BVDV and tended (P ≤ 0.09) to have greater serum antibody concentrations against BRSV and PI3 during the experiment compared with CON calves (Table 4). No treatment effects were noted (P = 0.37; Table 4) for serum antibody concentrations against BHV1, whereas day effects were detected (P < 0.01) for all of these serum variables (Table 5).
Behavioral responses
A treatment × day interaction was detected (P = 0.03) for exit velocity, which tended (P = 0.10) to be greater for CON vs. BAS calves on day 7 and were greater (P = 0.04) for CON vs. BAS calves on day 14 (Figure 4). A treatment × day interaction was also detected (P = 0.03) for physical activity (Figure 4), which was greater (P < 0.01) for BAS vs. CON calves on day 1 and similar (P ≥ 0.34) between treatments for the remainder of the experiment.
Figure 4.
Exit velocity (A) and physical activity (B) of beef calves receiving BAS (n = 40) or CON (n= 40) on the day of weaning (day 0). Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) topically to their nuchal skin area. Exit velocity was evaluated on days 0, 3, 7, 14, 28, and 42 (Cooke, 2014), whereas values recorded on day 0 were used as a covariate. Physical activity was evaluated via pedometers from day 1 to 42 (Schubach et al., 2017). Treatment × day interactions were detected for exit velocity (P ≤ 0.03) and physical activity. Within days: †P = 0.08; *P = 0.03; **P < 0.01.
No treatment effects were detected (P ≥ 0.23) for mean Shannon–Wiener Diversity Index, bunk displacement and headbutt counts, and proportion of pen lying, standing, or walking during the experiment (Table 6). The mean proportion of calves per pen feeding was greater (P = 0.05) for BAS vs. CON calves during the experiment (Table 6). Mean mounts per pen during the experiment was also greater (P = 0.04) for BAS vs. CON calves (0.52 vs. 0.08 mounts per pen, respectively; SEM = 0.18), although treatment differences were mostly noted during the initial 14 d of the experiment (treatment × day interaction, P = 0.82; Figure 5). Moreover, calves that received BAS tended to engage in more allogrooming bouts (P = 0.09) compared with CON calves during the experiment (Table 6). A treatment × day interaction was detected (P = 0.03) for escape attempts, which were greater (P < 0.01) in BAS vs. CON calves on day 1 and did not differ (P > 0.20) between treatments for the remainder of the experiment (Figure 5). Day effects were detected (P < 0.01) for the Shannon–Wiener Diversity Index and all herd behaviors, but for mounts that yielded a tendency (P = 0.08) for a day effect (Figure 5; Supplementary Figures S1–S3).
Table 6.
Behavioral responses during a 42-d preconditioning program of beef calves receiving BAS (n = 40) or CON (n= 40) on the day of weaning (day 0)1,2
| Item | CON | BAS | SEM | P-value |
|---|---|---|---|---|
| Herd behavior | ||||
| Feeding, proportion of pen | 0.142 | 0.162 | 0.010 | 0.05 |
| Lying, proportion of pen | 0.399 | 0.382 | 0.029 | 0.52 |
| Standing, proportion of pen | 0.422 | 0.414 | 0.024 | 0.70 |
| Walking, proportion of pen | 0.031 | 0.035 | 0.005 | 0.23 |
| Shannon-Weiner Diversity Index3 | 0.645 | 0.690 | 0.025 | 0.25 |
| Social behavior | ||||
| Allogroom, bout/pen | 3.02 | 4.03 | 0.44 | 0.09 |
| Bunk displacement, count/pen | 1.35 | 1.03 | 0.29 | 0.83 |
| Headbutt, count/pen | 3.04 | 4.98 | 1.25 | 0.93 |
1Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) on day 0. Treatments (5 mL) were applied topically to the nuchal skin area of each animal.
2Live behavioral observations were conducted from 0800 to 1800 hours on days 1, 2, 4, 8, 16, and 32. Herd behaviors were assessed through scan sampling (Mitlöhner et al., 2001), whereas the frequency of social behaviors was assessed through continuous sampling (Daigle et al., 2017). Herd behavior was quantified by the number of calves observed expression the behavior divided by total calves in the pen (Daigle et al., 2017, 2018).
3The Shannon–Weiner Diversity Index (Cronin and Ross, 2019) was calculated for each 10-min instantaneous scan observation for each pen and averaged by treatment.
Figure 5.
Escape attempts (A) and mounts (B) observed from beef calves receiving BAS (n = 40) or CON (n = 40) on the day of weaning (day 0). Calves individually received 5 mL of a BAS (IRSEA Group, Quartier Salignan, France) or CON (diethylene glycol monoethyl ether) topically to their nuchal skin area. Live behavioral observations were conducted on days 1, 2, 4, 8, 16, and 32 using 10-min long focal observations (Daigle et al., 2017). A treatment × day interaction was detected (P = 0.02) for escape attempts, whereas mean mounts per pen were greater (P = 0.04) for BAS vs. CON calves (0.437 vs. 0.146, respectively; SEM = 0.099).
Discussion
Weaning leads to a complex perceived stress by the calf, including loss of the dam, change in diet, and altered social structure (Weary et al., 2008). Weaning is also typically accompanied by compounding stressors, including transportation, vaccination, novel humans, and novel environments. Accordingly, the weaning process is one of the most physically and psychologically stressful events within the beef production cycle and known to stimulate adrenocortical and inflammatory reactions that impact cattle performance (Cooke, 2017). Day effects observed herein for plasma cortisol and haptoglobin corroborate that calves experienced an adrenocortical and acute-phase protein response elicited by the combination of weaning, road transport, novel management, and vaccination against BRD pathogens (Marques et al., 2016 ,2017). Similarly, calves in this experiment responded behaviorally to the stressors associated with the weaning process based on day effects noted across all live behavior observations. These latter outcomes are novel and may provide insight into monitoring cattle behavior during the postweaning period, including the Shannon–Wiener Diversity Index to quantify behavioral synchrony of calves within a pen after weaning (Cronin and Ross, 2019).
Circulating cortisol concentrations have been widely used as a biomarker of stress in cattle (Carroll and Forsberg, 2007); however, calves receiving BAS or CON had similar plasma cortisol concentrations during this experiment. Handling cattle for blood sampling also elicits an acute stress response that rapidly increases circulating cortisol (Schubach et al., 2017), possibly confounding results. For these reasons, cortisol concentrations in hair from the tail switch were evaluated. This variable has been validated as a biomarker of chronic stress in cattle (Moya et al., 2013), as cortisol is gradually accumulated in the emerging hair and its concentration represents long-term adrenocortical activity (Schubach et al., 2017). Heightened adrenocortical function has also been positively associated with circulating haptoglobin concentrations in cattle (Cooke and Bohnert, 2011; Cooke et al., 2012b). Corroborating this rationale, BAS administration resulted in reduced cortisol concentrations in hair from the tail switch on day 14 and the overall reduced circulating haptoglobin concentrations, supporting our hypothesis that BAS administration alleviates the stress elicited by the weaning process (Cooke, 2017; Cooke et al., 2020). Treatment differences detected for hair cortisol concentrations also indicate that circulating cortisol concentrations were chronically greater in CON vs. BAS calves. Additionally, BAS appears to be active for 15 d upon administration, when treatment differences for hair cortisol and plasma haptoglobin were more evident.
Serum concentrations of antibody against BRD viruses increased during the experiment across treatments, denoting that calves effectively acquired humoral immunity against these pathogens upon vaccination (Richeson et al., 2008). However, BAS administration improved humoral immunity against BRSV, BVDV, and PI3 during the 42-d preconditioning period. Vaccine efficacy is reduced when administered to highly stressed animals, thereby decreasing protection against BRD pathogens and increasing disease susceptibility (Blecha et al., 1984; Cooke, 2017; Schumaher et al., 2019). Cortisol influences the innate immune system in multiple ways, including stimulation of immune cell proliferation and differentiation, effector cell function, and cytokine expression (Carroll and Forsberg, 2007; Cooke and Bohnert, 2011; Cooke et al., 2012b). Cortisol also suppresses the inflammatory and innate immune systems, which can lead to immunosuppression if circulating cortisol concentrations are chronically heightened (Munck et al., 1984; Biolatti et al., 2005). Accordingly, the improved humoral immunity against BRD viruses in calves administered BAS can be attributed, at least partially, to alleviated stress elicited by weaning and reduced subsequent adrenocortical and acute-phase protein responses (Cooke, 2017).
The relevance of these latter outcomes to BRD incidence, however, could not be appraised as no incidence of BRD was noted during this experiment. Nonetheless, preconditioning programs prepare cattle for the feedlot through vaccination against BRD pathogens and adaptation to dry feed (Duff and Galyean, 2007). Serum antibody levels provide an indication of immune protection, disease prevention, and vaccine efficacy in cattle (Howard et al., 1989; Bolin and Ridpath, 1990; Callan, 2001). Moreover, there is a negative relationship between BRD incidence with cattle performance and carcass quality (Schneider et al., 2009; Reinhardt et al., 2012). Given the improved humoral immunity against BRD viruses from BAS administration, one can expect BAS calves to have decreased morbidity and improved performance in the feedlot, although research is warranted to validate this rationale.
Cattle temperament was also expected to be impacted by BAS due to its calming effects (Archunan et al., 2014) and has been directly associated with adrenocortical and acute-phase protein responses in growing cattle (Francisco et al., 2012, 2015; Cooke et al., 2019). Specifically, agitation or aggressive responses expressed by cattle when they are exposed to human handling can be attributed to their inability to cope with the situation, captured via temperament evaluation, and classified as a psychological stress reaction (Cooke, 2014). Reduced exit velocity of BAS calves on days 7 and 14 indicates that BAS has appeasing effects for 15 d upon administration and corroborates treatment differences noted in plasma haptoglobin and hair cortisol concentrations.
Calves physically separated from their dams exhibit an increase in physical activity associated with an increase in psychological stress (Solano et al., 2007). One can interpret the increased escape attempts and physical activity observed in BAS calves on day 1 as a state of heightened stress or agitation upon weaning (Weary et al., 2008). Previous research has also demonstrated that exploration behavior is associated with habituation, defined as a behavioral response decrement resulting from repeated stimulation (Rankin et al., 2009). Accordingly, increased activity of BAS calves on day 1 may be associated with the exploration of the pen and, therefore, an expedited familiarization of the novel environment resulting in a reduced overall stress response. This is reflected in the overall increased performance of social and play behaviors exhibited by BAS calves including allogrooming and mounting, the latter being more evident during the initial period of the experiment when BAS is suggested to be active (Cooke, 2020). Although excessive mounting (e.g., bulling) is a welfare concern in feedlot cattle, the valence of mounting is context-specific. Mounting is also a behavior associated with reproductive status, establishment of social hierarchy, as well as social play in young cattle reared in pens (Reinhardt et al., 1978; Vitale et al., 1986; Jensen and Kyhn, 2000). In turn, the BAS administration did not alter the proportion of calves lying, standing, or walking in the pen nor the incidence of headbutts and bunk displacement. One can associate the lack of treatment differences on these latter parameters to limited statistical power for pen-based live behavior observations (n = 4 per treatment), although differences were noted between BAS and CON calves for feeding proportion, mounts, allogrooming, and escape attempts. According to the G*power 3 software (Faul et al., 2007) and results reported herein and previously by our group (Daigle et al., 2017, 2018), four pens per treatment with six measurements per pen over time were sufficient to detect a 25% change between treatments in the proportion of calves lying, standing, or walking with a power ≥ 0.85 and an alpha of 0.05. Nonetheless, research with increased replications are warranted to further explore the impacts of BAS administration on behavioral responses of weaned cattle.
Overall ADG, TMR intake, and G:F during the 42-d preconditioning period were not impacted by BAS. However, the growth rate was improved in BAS calves during the initial 28 d after weaning, which can be mostly attributed to their greater feed intake during week 1 of the experiment. The growth rate from day 28 to 42 after weaning did not differ in BAS and CON calves, but numerical differences resulted in similar ADG during the experimental period. In our previous research with BAS (Cappellozza et al., 2020; Cooke et al., 2020), calves were processed for sampling at weaning, 2 wk after weaning, and at the end of the 45-d postweaning period. Perhaps the frequent sampling adopted herein was disruptive and hindered the full benefits of BAS to performance responses, resulting in similar ADG between treatments during the experimental period. Nonetheless, BAS administration likely increased initial feed intake by alleviating stress-induced physiological and acute-phase protein reactions while improving temperament and habituation to a new environment, which are all known to modulate feed intake in cattle (Cooke, 2014, 2017). Cortisol acts as a lipolytic agent during a stress reaction (Nelson and Cox, 2005) indirectly affecting voluntary feed intake, whereas plasma haptoglobin concentrations have been negatively associated with feed intake in feeder cattle (Araujo et al., 2010). Yet, increased feed intake by BAS administration was not sufficient to impact circulating concentrations of BHBA, IGF-1, or NEFA, which are metabolic markers of feed intake and nutritional status in ruminants (Hess et al., 2005). Plasma IGF-1 concentrations increased, while plasma BHBA and NEFA concentrations decreased across treatments as the experiment advanced, given the increasing nutrient intake and growth during the 42-d preconditioning period (Ellenberger et al., 1989; Hess et al., 2005; Sousa et al., 2019).
In summary, BAS administration upon weaning transiently enhanced calf initial growth rate and TMR intake and improved humoral immunity to PI3, BRSV, and BVDV compared with CON calves during a 42-d preconditioning period. These performance benefits were observed early in the experiment when treatment differences in exit velocity, behavior, hair cortisol, and plasma haptoglobin concentrations were noticeable. Research is still warranted to further examine the benefits of BAS to beef calves, including multiple BAS administrations during preconditioning (e.g., every 14 d) and potential carryover effects to feedlot growth and health responses. Such research should also investigate the duration that BAS remains present and active in cattle upon administration. Nonetheless, results from this experiment indicate that BAS administration to beef calves may be a strategy to alleviate the physiological and behavioral stress responses elicited by the weaning process.
Supplementary Material
Acknowledgment
Financial support for this research was provided by Nutricorp (Araras, SP, Brazil), which distributes BAS in the United States, Canada, and Brazil. K.M.S. is a Tom Slick Graduate Research Fellow at Texas A&M University. A.P.B. is supported by CAPES – Brazil grant (88881.128327/2016-01).
Glossary
Abbreviations
- ADG
average daily gain
- BAS
bovine appeasing substance
- BHBA
β-hydroxybutyrate
- BHV-1
bovine herpesvirus-1
- BRD
bovine respiratory disease
- BRSV
bovine respiratory syncytial virus
- BVDV
bovine viral diarrhea viruses
- BW
body weight
- G:F
feed efficiency
- IGF-I
insulin-like growth factor I
- NEFA
nonesterified fatty acids
- PI3
parainfluenza-3 virus
- TMR
total mixed ration
Conflicts of interest statement
The remaining authors declare no real or perceived conflicts of interest.
Literature Cited
- AOAC . 2006. Official methods of analysis. 18th ed. Arlington (VA): Association of Official Analytical Chemists. [Google Scholar]
- Araujo, D B, Cooke R F, Hansen G R, Staples C R, and Arthington J D. . 2010. Effects of rumen-protected polyunsaturated fatty acid supplementation on performance and physiological responses of growing cattle following transportation and feedlot entry. J. Anim. Sci. 88:4120–4132. doi: 10.2527/jas.2009-2684 [DOI] [PubMed] [Google Scholar]
- Archunan, G, Rajanarayanan S, and Karthikeyan K. . 2014. Cattle pheromones. In: Mucignat-Caretta, C., editor. Neurobiology of chemical communication. Boca Raton (FL):CRC Press; p. 461–488. [PubMed] [Google Scholar]
- Biolatti, B, Bollo E, Cannizzo F T, Zancanaro G, Tarantola M, Dacasto M, Cantiello M, Carletti M, Biolatti P G, and Barbarino G. . 2005. Effects of low-dose dexamethasone on thymus morphology and immunological parameters in veal calves. J. Vet. Med. A. Physiol. Pathol. Clin. Med. 52:202–208. doi: 10.1111/j.1439-0442.2005.00714.x [DOI] [PubMed] [Google Scholar]
- Blecha, F, Boyles S L, and Riley J G. . 1984. Shipping suppresses lymphocyte blastogenic responses in Angus and Brahman × Angus feeder calves. J. Anim. Sci. 59:576–583. doi: 10.2527/jas1984.593576x [DOI] [PubMed] [Google Scholar]
- Bolin, S R, and Ridpath J F. . 1990. Range of viral neutralizing activity and molecular specificity of antibodies induced in cattle by inactivated bovine viral diarrhea virus vaccines. Am. J. Vet. Res. 51:703–707. [PubMed] [Google Scholar]
- Callan, R J. 2001. Fundamental considerations in developing vaccination protocols. Bovine Pract. 34:14–22. [Google Scholar]
- Cappellozza, B I, Bastos J P, and Cooke R F. . 2020. Administration of an appeasing substance to Bos indicus-influenced beef cattle improves performance after weaning and carcass pH. Livest. Sci. 238:104067. doi: 10.1016/j.livsci.2020.104067 [DOI] [Google Scholar]
- Carroll, J A, and Forsberg N E. . 2007. Influence of stress and nutrition on cattle immunity. Vet. Clin. North Am. Food Anim. Pract. 23:105–149. doi: 10.1016/j.cvfa.2007.01.003 [DOI] [PubMed] [Google Scholar]
- Colombo, E A, Cooke R F, Millican A A, Schubach K M, Scatolin G N, Rett B, and Brandão A P. . 2019. Supplementing an immunomodulatory feed ingredient to improve thermoregulation and performance of finishing beef cattle under heat stress conditions. J. Anim. Sci. 97:4085–4092. doi: 10.1093/jas/skz266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooke, R F. 2014. Bill E. Kunkle Interdisciplinary Beef Symposium: Temperament and acclimation to human handling influence growth, health, and reproductive responses in Bos taurus and Bos indicus cattle. J. Anim. Sci. 92:5325–5333. doi: 10.2527/jas.2014-8017 [DOI] [PubMed] [Google Scholar]
- Cooke, R F. 2017. Nutritional and management considerations for beef cattle experiencing stress-induced inflammation. Prof. Anim. Sci. 33:1–11. doi: 10.15232/pas.2016-01573 [DOI] [Google Scholar]
- Cooke, R F, and Arthington J D. . 2013. Concentrations of haptoglobin in bovine plasma determined by ELISA or a colorimetric method based on peroxidase activity. J. Anim. Physiol. Anim. Nutr. (Berl). 97:531–536. doi: 10.1111/j.1439-0396.2012.01298.x [DOI] [PubMed] [Google Scholar]
- Cooke, R F, and Bohnert D W. . 2011. Bovine acute-phase response after corticotrophin-release hormone challenge. J. Anim. Sci. 89:252–257. doi: 10.2527/jas.2010-3131 [DOI] [PubMed] [Google Scholar]
- Cooke, R F, Cappellozza B I, Reis M M, Bohnert D W, and Vasconcelos J L. . 2012a. Plasma progesterone concentration in beef heifers receiving exogenous glucose, insulin, or bovine somatotropin. J. Anim. Sci. 90:3266–3273. doi: 10.2527/jas.2011-4959 [DOI] [PubMed] [Google Scholar]
- Cooke, R F, Carroll J A, Dailey J, Cappellozza B I, and Bohnert D W. . 2012b. Bovine acute-phase response after different doses of corticotropin-releasing hormone challenge. J. Anim. Sci. 90:2337–2344. doi: 10.2527/jas.2011-4608 [DOI] [PubMed] [Google Scholar]
- Cooke, R F, Millican A, Brandão A P, Schumaher T F, de Sousa O A, Castro T, Farias R S, and Cappellozza B I. . 2020. Short Communication: Administering an appeasing substance to Bos indicus-influenced beef cattle at weaning and feedlot entry. Animal 14:566–569. doi: 10.1017/S1751731119002490 [DOI] [PubMed] [Google Scholar]
- Cooke, R F, Moriel P, Cappellozza B I, Miranda V F B, Batista L F D, Colombo E A, Ferreira V S M, Miranda M F, Marques R S, and Vasconcelos J L M. . 2019. Effects of temperament on growth, plasma cortisol concentrations and puberty attainment in Nelore beef heifers. Animal 13:1208–1213. doi: 10.1017/S1751731118002628 [DOI] [PubMed] [Google Scholar]
- Cronin, K A, and Ross S R. . 2019. Technical contribution: a cautionary note on the use of behavioural diversity (H-Index) in animal welfare science. Anim. Welf. 28:157–164. doi: 10.7120/09627286.28.2.157 [DOI] [Google Scholar]
- Daigle, C L, Jackson B, Gill R, Wickersham T A, and Sawyer J E. . 2017. Impact of exercise on productivity, behavior, and immune functioning of weaned Bos indicus-cross calves housed in drylots. J. Anim. Sci. 95:5230–5239. doi: 10.2527/jas2017.1788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daigle, C L, Mathias A J, Ridge E E, Gill R, Wickersham T A, and Sawyer J E. . 2018. Case Study: Effect of exercise programs during receiving in a commercial feedlot on behavior and productivity of Brahman crossbred calves: results from a commercial environment and a comparison to the research environment. Prof. Anim. Sci. 34(6): 653–663. doi: 10.15232/pas.2018-01744 [DOI] [Google Scholar]
- Duff, G C, and Galyean M L. . 2007. Board-Invited Review: Recent advances in management of highly stressed, newly received feedlot cattle. J. Anim. Sci. 85:823–840. doi: 10.2527/jas.2006-501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellenberger, M A, Johnson D E, Carstens G E, Hossner K L, Holland M D, Nett T M, and Nockels C F. . 1989. Endocrine and metabolic changes during altered growth rates in beef cattle. J. Anim. Sci. 67:1446–1454. doi: 10.2527/jas1989.6761446x [DOI] [PubMed] [Google Scholar]
- Faul, F, Erdfelder E, Lang A G, and Buchner A. . 2007. G*Power 3: a flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39:175–191. doi: 10.3758/bf03193146 [DOI] [PubMed] [Google Scholar]
- Francisco, C L, Cooke R F, Marques R S, Mills R R, and Bohnert D W. . 2012. Effects of temperament and acclimation to handling on feedlot performance of Bos taurus feeder cattle originated from a rangeland-based cow-calf system. J. Anim. Sci. 90:5067–5077. doi: 10.2527/jas.2012-5447 [DOI] [PubMed] [Google Scholar]
- Francisco, C L, Resende F D, Benatti J M, Castilhos A M, Cooke R F, and Jorge A M. . 2015. Impacts of temperament on Nellore cattle: physiological responses, feedlot performance, and carcass characteristics. J. Anim. Sci. 93:5419–5429. doi: 10.2527/jas.2015-9411 [DOI] [PubMed] [Google Scholar]
- Gonda, M G, Fang X, Perry G A, and Maltecca C. . 2012. Measuring bovine viral diarrhea virus vaccine response: using a commercially available ELISA as a surrogate for serum neutralization assays. Vaccine 30:6559–6563. doi: 10.1016/j.vaccine.2012.08.047 [DOI] [PubMed] [Google Scholar]
- Hess, B W, Lake S L, Scholljegerdes E J, Weston T R, Nayigihugu V, Molle J D C, and Moss G E. . 2005. Nutritional controls of beef cow reproduction. J. Anim. Sci. 83:E90–E106. doi: 10.2527/2005.8313_supplE90x [DOI] [Google Scholar]
- Howard, C J, Clarke M C, and Brownlie J. . 1989. Protection against respiratory infection with bovine virus diarrhoea virus by passively acquired antibody. Vet. Microbiol. 19:195–203. doi: 10.1016/0378-1135(89)90066-7 [DOI] [PubMed] [Google Scholar]
- Jensen, M B, and Kyhn R. . 2000. Play behaviour in group-housed dairy calves, the effect of space allowance. Appl. Anim. Behav. Sci. 67:35–46. doi: 10.1016/s0168-1591(99)00113-6 [DOI] [PubMed] [Google Scholar]
- Liberles, S D. 2014. Mammalian pheromones. Annu. Rev. Physiol. 76:151–175. doi: 10.1146/annurev-physiol-021113-170334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marques, R S, Cooke R F, Rodrigues M C, Brandão A P, Schubach K M, Lippolis K D, Moriel P, Perry G A, Lock A, and Bohnert D W. . 2017. Effects of supplementing calcium salts of polyunsaturated fatty acids to late-gestating beef cows on performance and physiological responses of the offspring. J. Anim. Sci. 95:5347–5357. doi: 10.2527/jas2017.1606 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marques, R S, Cooke R F, Rodrigues M C, Cappellozza B I, Mills R R, Larson C K, Moriel P, and Bohnert D W. . 2016. Effects of organic or inorganic cobalt, copper, manganese, and zinc supplementation to late-gestating beef cows on productive and physiological responses of the offspring. J. Anim. Sci. 94:1215–1226. doi: 10.2527/jas.2015-0036 [DOI] [PubMed] [Google Scholar]
- McGlone, J J, and Anderson D L. . 2002. Synthetic maternal pheromone stimulates feeding behavior and weight gain in weaned pigs. J. Anim. Sci. 80:3179–3183. doi: 10.2527/2002.80123179x [DOI] [PubMed] [Google Scholar]
- Mitlöhner, F M, Morrow-Tesch J L, Wilson S C, Dailey J W, and McGlone J J. . 2001. Behavioral sampling techniques for feedlot cattle. J. Anim. Sci. 79:1189–1193. doi: 10.2527/2001.7951189x [DOI] [PubMed] [Google Scholar]
- Moya, D, Schwartzkopf-Genswein K S, and Veira D M. . 2013. Standardization of a non-invasive methodology to measure cortisol in hair of beef cattle. Livest. Sci. 158:138–144. doi: 10.1016/j.livsci.2013.10.007 [DOI] [Google Scholar]
- Munck, A, Guyre P M, and Holbrook N J. . 1984. Physiological functions of glucocorticoids in stress and their relation to pharmacological actions. Endocr. Rev. 5:25–44. doi: 10.1210/edrv-5-1-25 [DOI] [PubMed] [Google Scholar]
- Nelson, D L, and Cox M M. . 2005. Lehninger principles of biochemistry. 4th ed. New York (NY):W. H. Freeman and Company. [Google Scholar]
- NRC . 2000. Nutrient requirement of beef cattle. 7th ed. Washington (DC):National Academies Press. [Google Scholar]
- Osella, M C, Cozzi A, Spegis C, Turille G, Barmaz A, Lecuelle C L, Teruel E, Bienboire-Frosini C, Chabaud C, Bougrat L, . et al. 2018. The effects of a synthetic analogue of the Bovine Appeasing Pheromone on milk yield and composition in Valdostana dairy cows during the move from winter housing to confined lowland pastures. J. Dairy Res. 85:174–177. doi: 10.1017/S0022029918000262 [DOI] [PubMed] [Google Scholar]
- Pageat, P. 2001. Appeasing Pheromones to decrease stress, anxiety and aggressiveness. US Patent 6,054,481, 6,077,867, and 6,169,113 B1. January 2, 2001. [Google Scholar]
- Rankin, C H, Abrams T, Barry R J, Bhatnagar S, Clayton D F, Colombo J, Coppola G, Geyer M A, Glanzman D L, Marsland S, . et al. 2009. Habituation revisited: an updated and revised description of the behavioral characteristics of habituation. Neurobiol. Learn. Mem. 92:135–138. doi: 10.1016/j.nlm.2008.09.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reinhardt, C D, Hands M L, Marson T T, Wannoner J W, and Corah L R. . 2012. Relationships between feedlot health, average daily gain, and carcass traits of Angus steers. Prof. Anim. Sci. 28:11–19. doi: 10.15232/S1080-7446(15)30311-9 [DOI] [Google Scholar]
- Reinhardt, V, Mutiso F M, and Reinhardt A. . 1978. Social behaviour and social relationships between female and male prepubertal bovine calves. Appl. Anim. Ethol. 4:43–54. doi: 10.1016/0304-3762(78)90092-5 [DOI] [Google Scholar]
- Richeson, J T, Beck P A, Gadberry M S, Gunter S A, Hess T W, Hubbell D S 3rd, and Jones C. . 2008. Effects of on-arrival versus delayed modified live virus vaccination on health, performance, and serum infectious bovine rhinotracheitis titers of newly received beef calves. J. Anim. Sci. 86:999–1005. doi: 10.2527/jas.2007-0593 [DOI] [PubMed] [Google Scholar]
- Schneider, M J, Tait R G Jr, Busby W D, and Reecy J M. . 2009. An evaluation of bovine respiratory disease complex in feedlot cattle: impact on performance and carcass traits using treatment records and lung lesion scores. J. Anim. Sci. 87:1821–1827. doi: 10.2527/jas.2008-1283 [DOI] [PubMed] [Google Scholar]
- Schubach, K M, Cooke R F, Brandão A P, Lippolis K D, Silva L G T, Marques R S, and Bohnert D W. . 2017. Impacts of stocking density on development and puberty attainment of replacement beef heifers. Animal 11:2260–2267. doi: 10.1017/S1751731117001070 [DOI] [PubMed] [Google Scholar]
- Schumaher, T F, Cooke R F, Brandão A P, Schubach K M, de Sousa O A, Bohnert D W, and Marques R S. . 2019. Effects of vaccination timing against respiratory pathogens on performance, antibody response, and health in feedlot cattle. J. Anim. Sci. 97:620–630. doi: 10.1093/jas/sky466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Solano, J, Orihuela A, Galina C S, and Aguirre V. . 2007. A note on behavioral responses to brief cow-calf separation and reunion in cattle (Bos indicus). J. Vet. Behav. 2:10–14. doi: 10.1016/j.jveb.2006.12.002 [DOI] [Google Scholar]
- Sousa, O A, Cooke R F, Brandão A P, Schubach K M, Schumaher T F, Bohnert D W, and Marques R S. . 2019. Productive and physiological responses of feeder cattle supplemented with Yucca schidigera extract during feedlot receiving. J. Anim. Sci. 97:208–219. doi: 10.1093/jas/sky412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Soest, P J, Robertson J B, and Lewis B A. . 1991. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74:3583–3597. doi: 10.3168/jds.S0022-0302(91)78551-2 [DOI] [PubMed] [Google Scholar]
- Vitale, A F, Tenucci M, Papini M, and Lovari S. . 1986. Social behaviour of the calves of semi-wild Maremma cattle, Bos primigenius taurus. Appl. Anim. Behav. Sci. 16:217–231. doi: 10.1016/0168-1591(86)90115-2 [DOI] [Google Scholar]
- Weary, D M, Jasper J, and Hötzel M J. . 2008. Understanding weaning distress. Appl. Anim. Behav. Sci. 110:24–41. doi: 10.1016/j.applanim.2007.03.025 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.