Abstract
Background
We previously found two distinct passenger dendritic cell (DC) subsets in the rat liver that played a central role in the liver transplant rejection. In addition, a tolerance-inducing protocol, donor-specific transfusion (DST), triggered systemic polytopical production of depleting alloantibodies to donor class I MHC (MHCI) antigen (DST-antibodies).
Methods
We examined the role of DST-antibodies in the trafficking of graft DC subsets and the alloresponses in a rat model. We also examined an anti-donor class II MHC (MHCII) antibody that recognizes donor DCs more selectively.
Results
Preoperative transfer of DST-antibodies or DST pretreatment eliminated all passenger leukocytes, including both DC subsets and depleted the sessile DCs in the graft to ~20% of control. The CD172a+CD11b/c+ immunogenic subset was almost abolished. The intrahost direct or semi-direct allorecognition pathway was successfully blocked, leading to a significant suppression of the CD8+ T-cell response in the recipient lymphoid organs and the graft with delayed graft rejection. Anti-donor MHCII antibody had similar effects without temporary graft damage. Although DST pretreatment had a priming effect on the proliferative response of recipient regulatory T cells, DST-primed sera and the anti-donor MHCII antibody did not.
Conclusion
DST-antibodies and anti-donor MHCII antibodies could suppress the CD8+ T-cell-mediated liver transplant rejection by depleting donor immunogenic DCs, blocking the direct or semi-direct pathways of allorecognition. Donor MHCII-specific antibodies may be applicable as a selective suppressant of anti-donor immunity for clinical liver transplantation without the cellular damage of donor MHCII– graft cells and recipient cells.
Keywords: CD8 T cells, donor-specific transfusion, leukocyte trafficking, multicolor immunohistochemistry, sensitization pathway
Antibody-mediated donor-DC depletion prolongs liver-graft survival
Introduction
The liver is one of the most leukocyte-rich organs and contains lymphoid cells, dendritic cells (DCs) and myeloid lineages. When liver transplantation (LTx) is performed, these cells migrate to the recipient secondary lymphoid organs (SLOs) via the blood or lymph as passenger leukocytes and elicit the recipient immune response to donor MHC antigens (1, 2). Graft-derived DCs cluster with recipient T cells and induce anti-graft CD8+ T-cell responses (3, 4). This event is known as the direct pathway of allorecognition and is considered to be a major cause of acute allograft rejection (5). Alternatively, in the semi-direct pathway, donor passenger cells may secrete extracellular vesicles containing MHC molecules, which are incorporated into recipient DCs and presented intact to recipient T cells (6). In addition, the indirect pathway, in which recipient DCs phagocytose fragments of donor passenger cells in the T-cell area of the SLOs and present them to recipient T cells in the context of self-MHCs (7), might also be involved. Thus, inhibition of passenger cell migration, especially the migration of DCs, should at least partly suppress the rejection, but a method has not yet been established and we are still uncertain which allorecognition pathway is involved.
Many studies have shown that conventional mouse, rat and human DCs are heterogeneous and comprise several subsets with distinct phenotypes and functional properties (4, 8, 9), with some DC subsets being immunogenic (4, 10) and others being tolerogenic (11, 12). Three phenotypically distinct subsets are known in the rat liver (13). We functionally identified two distinct immunogenic DC subsets following LTx (4): the class II MHC (MHCII)+CD103+CD86+CD172a (signal-regulatory protein alpha)+ CD11b/c– radiosensitive subset that underwent blood-borne migration to the recipient’s SLOs, inducing systemic CD8+ T-cell responses (3) and the highly immunogenic MHCII+CD103+CD86+CD172a+CD11b/c+ relatively radioresistant subset that underwent lymph-borne migration to the peritoneal cavity and then migrated to the parathymic lymph nodes (LNs), regional peritoneal cavity LNs, or persisted in the graft. Accordingly, the rejection may be attributable to these two immunogenic DC subsets, and their elimination may suppress the rejection. However, selective depletion of these DC subsets has not been reported.
Donor-specific transfusion (DST), which simply transfuses fresh donor blood intravenously into recipients, is one of the tolerance-inducing regimens used not only in experimental (14), but also clinical (15), transplantation. Although the production of depleting antibodies (16), blocking antibodies (17) and peripheral regulatory T cells (Tregs) (18) has been reported, the precise immunosuppressive mechanism of DST remains unclear. Recently, we found that DST induces a donor class I MHC (MHCI)-specific CD4+ T-cell-dependent alloantibody-forming cell response polytopically in the SLOs (7), and these alloantibodies (DST-antibodies) can deplete intravenously transferred donor lymphocytes or suppress the GvHD induced by donor T cells (16). This suggests that DST-antibodies may affect not only donor passenger lymphocytes, but also the migrating DC subsets and remaining DCs in the graft after LTx, inhibiting the allorecognition in recipient SLOs and the graft, respectively.
The aim of this study was to investigate the DST mechanism in a rat LTx model, regarding the role of DST-antibodies in the trafficking of passenger DC subsets and remaining DC subsets in the graft, and the CD8+ T-cell response in both recipient SLOs and the graft itself. We performed mainly an immunohistological study because for examination of kinetics and mutual relationships of involved donor and host cells in defined microstructures of the SLOs and graft liver, in situ observation is indispensable. Here, we show that the donor-MHCI-specific DST-antibodies can eliminate the immunogenic passenger DC subsets and reduce the number of DCs remaining in the graft, strongly inhibiting the allorecognition and leading to a significant suppression of the CD8+ T-cell response in the recipient SLOs and within the graft liver with delayed graft rejection. Using an mAb against donor MHCII antigens that recognizes donor DCs more selectively, we could minimize the side effects of DST-antibodies in regard to graft damage, without impeding the immunosuppression.
Methods
Animals
Inbred ACI rats (RT1.AaBa) were obtained from the National Bio Resource Project for the Rat in Japan (Kyoto, Japan). Lewis rats (RT1.AlBl) and GFP-transgenic Lewis (GFP-Lewis) rats were supplied by Charles River Japan, Inc. (Tsukuba, Japan) and by the National Research Institute for Child Health and Development, respectively. GFP-Lewis rats and ACI rats were bred and maintained in the Laboratory Animal Research Center of Dokkyo Medical University (Mibu, Japan). All rats were reared under specific pathogen-free conditions. ACI rats donated liver grafts and blood. Lewis rats were recipients. The handling and care of the animals was approved by the Dokkyo Medical University Animal Experiment Committee and in accordance of the University Dokkyo’s Regulation for Animal Experiments and Japanese Governmental Law (No. 105).
Antibodies and reagents
Monoclonal antibodies (mAbs) and labeled secondary antibodies used for immunohistology and fluorescence-activated cell sorter (FACS) analysis are listed in Supplementary Table 1. For the detection of Foxp3-positive cells, we used rabbit anti-Foxp3 polyclonal antibodies (19). Some mAbs were purified from culture supernatants and coupled to fluorescein isothiocyanate (FITC), R-phycoerythrin (Dojin, Kumamoto, Japan), PerCP/Cy5.5 (Innnova Bioscience Ltd, Cambridge, UK), Alexa-Fluor 488, 594 or 647 (Molecular Probes, Eugene, OR, USA) or biotin (Pierce, Rockford, IL, USA) in house. Brilliant Violet 421™ Streptavidin (BioLegend) was also used for staining biotinylated mAb.
Liver transplantation
Orthotopic LTx from ACI to Lewis rats was performed using a cuff method as described previously (3). At various times from 1 to 10 days after LTx, recipient rats received an intravenous injection of 5-bromo-2′-deoxyuridine (BrdU) for immunoenzyme staining or 5-ethynyl-2′-deoxyuridine (EdU) for immunofluorescence staining 1 h before sacrifice as described previously (7). Graft livers and recipient lymphoid organs, including the spleen, peripheral LNs (cervical LNs and axillary LNs) and parathymic LNs (mediastinal LNs draining the peritoneal cavity) (4) were excised and fresh-frozen for immunostaining. Sera were collected for the detection of alloantibodies and the examination of serum AST and bilirubin levels. For all groups that had received LTx, the mean survival time (MST) was estimated as reported previously (20) using four or more rats per group.
Donor-specific blood transfusion and adoptive transfer of sera or the mAb
One milliliter of freshly heparinized whole blood was aseptically obtained from the donor aorta and immediately injected intravenously into the tail vein of recipient rats 7 days prior to LTx.
To determine the role of DST-induced sera, recipient rats received 1 day prior to LTx an intra-peritoneal injection of either DST sera of Lewis rats collected 7 days after DST treatment only (DST sera group) or normal Lewis sera (control sera group). Seven milliliter of sera was injected per rat as an equivalent amount obtained from one rat with a body weight of 200 g.
To inhibit donor DC migration more selectively, an mAb to donor MHCII polymorphic antigen (MRC OX76, 0.3 mg per rat) was intra-peritoneally injected into Lewis rats 1 day prior to LTx. The minimal dose for to inhibit migration was determined in a preliminary study. Recipient rats that received the sera or the mAb were studied in the same fashion as the DST(+) group, except that samples were not collected on day 4.
Immunohistological analysis
Triple immunoenzyme staining of fresh 6-µm-thick cryosections for target cells (alkaline phosphatase-blue), BrdU (alkaline phosphatase-red) and type IV collagen (peroxidase-brown) was performed as described previously (1, 21). Type IV collagen was often immunostained to outline the T-cell area of the SLOs (3) or the liver structures (22). Donor passenger leukocytes were detected by staining of donor MHCI (RT1.Aa, MN4-91–6) or MHCII (RT1.Ba, OX76) polymorphic antigens. For the host CD8+ T-cell response, a mAb to CD8β was used, because rat natural killer cells and some activated macrophages (23) are also CD8α +.
The intrahost proliferative response and CD8+ T-cell response were estimated in the T-cell areas of the recipient spleen [inner part of the periarterial lymphocyte sheath (PALS)] and the peripheral and parathymic LNs (paracortex). Photomicrographs with a 20× objective were taken and T-cell areas in the digital images were outlined and their surface areas estimated by image analysis after subtracting other domains and blood vessels. After that, the number of BrdU+CD8β + T cells and total BrdU+ cells mm−2 of T-cell areas in printed photomicrographs were counted in a blinded fashion by different examiners. Cells with a red BrdU+ nucleus thoroughly outlined by a blue CD8β + cell membrane were registered as CD8β + proliferating T cells. BrdU+ cells with a discontinuous CD8β + cell membrane or without CD8β staining were determined as non-CD8 proliferating cells.
Concerning the intragraft response, we estimated the proportion of the sinusoidal area relative to the total surface area of stained sections, because the loss of the sinusoidal area is parallel to the loss of hepatocytes (i.e. liver function). Graft tissues were divided into three anatomical compartments: the sinusoidal, portal and hepatic vein areas (22). The portal area was defined by a presence of the bile ducts depicted by type IV collagen. Since infiltrated recipient cells densely accumulated mainly in the portal and the hepatic vein areas (24), the sinusoidal area could be easily depicted by the immunostaining for recipient MHCI (I169.1) (25) and type IV collagen. Photomicrographs with a 4× objective were taken and the proportions were estimated by image analysis. The hepatic vein area was not examined in the present study because the response was generally similar to the portal area.
To estimate the CD8+ T-cell response in the portal and the sinusoidal areas, photomicrographs of 20× and 10× objectives were taken, respectively. The number of BrdU+CD8β + T cells, total CD8β + and total BrdU+ cells mm−2 of both areas were counted in a same fashion as the SLOs by different examiners.
In addition, because the sinusoidal area is the crucial site for the liver transplant rejection, the CD8+ T-cell response was digitally quantified by image analysis. For this, whole immunostained sections of the graft livers of the DST sera, control sera, and anti-donor MHCII mAb groups on day 5 after LTx were scanned with a 20× objective by the Vectra Polaris Imaging System (Akoya Biosciences, Marlborough, MA, USA). The sinusoidal area per whole section was outlined from acquired images by InForm software (Akoya Biosciences, ver. 2.4.9), then the BrdU+CD8β + cells as well as total CD8β + cells mm−2 of the sinusoidal area were digitally calculated. The numbers of CD8+ T cells counted by this method were generally lower than the manual counting, because we omitted CD8 weakly-positive cells to improve the signal-to-noise ratio.
The phenotype of recipient infiltrating cells was examined by using GFP+ recipients with four-color immunofluorescence staining for GFP, CD antigens, type IV collagen and EdU as reported (20, 26).
For phenotype analysis of donor MHCII+ cluster-forming cells, serial fresh 2-µm-thick cryosections of both DST(–) and DST(+) graft livers on day 2 after transplant were prepared. One section was stained for donor MHCII (blue), type IV collagen (brown) and BrdU (red). A neighboring section was stained for either CD103, CD11c or CD86 (blue), BrdU (red), and IV collagen (brown). The corresponding areas of two neighboring sections were photomicrographed, and the proportion of either CD103+, CD11c+, CD86+ cluster-forming cells/total donor MHCII+ cluster-forming cells was calculated. More than 40 clusters per rat and three rats for each phenotype were examined.
To directly depict the clustering DCs, four-color immunofluorescence staining for the donor MHCII (OX76, Alexa-Fluor 488), CD103 (OX62, Alexa-Fluor 647), CD11b/c or CD172a (OX42 or ED9, Brilliant Violet 421™) and EdU (Alexa-Fluor 594) was performed with a minor modification of the previous report (27). Multichannel color fluorescence images were captured using an Axioskop 2 Plus fluorescence microscope equipped with an AxioCam MRm camera (Zeiss, Oberkochen, Germany). We assigned pseudocolors to each channel to create merged images more comprehensible by maximizing contrast using AxioVision software (Zeiss).
FACS analysis
For quantification of donor DC numbers, graft livers were digested with Liberase™ (Roche Diagnostics, Indianapolis, IN, USA), and cells were isolated, and then purified by a density gradient separation using OptiPrep (AXIS-SHIELD, Oslo, Norway) as described (3). Low-density cell suspensions were preincubated with FcγIIR-blocking mAb (clone D34-485; BD Biosciences, San Jose, CA, USA). After incubation with biotin-conjugated anti-donor MHCI mAb and streptavidin magnetic microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany), cells were positively isolated by autoMACS (Miltenyi Biotec). The isolated cells were stained for Alexa-Fluor 647-conjugated anti-donor MHCII mAb and FITC-, R-phycoerythrin- and PerCP/Cy5.5-conjugated mAbs. Cells were washed twice and acquired on a FACS Calibur (BD Biosciences). Data were analyzed using FlowJo ver.9.2 software (FlowJo LLC, Ashland, OR, USA).
DNA microarray analysis
Total RNA was prepared from graft liver and recipient spleen tissues 6 h after LTx (each group, n = 3) by QIAGEN RNA extraction kit according to the manufacturer’s instruction (QIAGEN RNeasy; QIAGEN Inc, Valencia, CA, USA), and used as template for synthesis of aminoallyl-UTP incorporated cRNA. BrdU was not injected into the recipients. Cy3 or Cy5 coupled cRNA was hybridized to Rat Oligo Microarray (Agilent Technologies, USA) for 17 h at 60°C. Images were scanned with an Agilent scanner (Agilent). Feature Extraction (Agilent) was used to assign gene names to spot densities and background values were calculated from negative control and subtracted.
Estimation of liver function
To evaluate liver function, sera from all groups were collected and examined for aspartate aminotransferase (AST) and total bilirubin levels by using automatic analyzer FUJI DRI-CHEM 3500V and FUJI DRI-CHEM SLIDE (FUJIFILM Medical Co., Ltd., Tokyo, Japan) for GOT/AST-PIII and TBIL-PIII, respectively.
Image analysis and statistics
Each parameter was measured in a blinded fashion and expressed as the mean ± SD (n = 3–4 rats). For estimation of the surface areas, image analysis was performed on a personal computer using the public domain NIH Image program (Image J1.36b). For FACS analysis, each assay was repeated three times. Statistical analysis was performed using the Student’s t-test.
Results
DST pretreatment inhibited the donor cell migration and the intrahost T-cell response
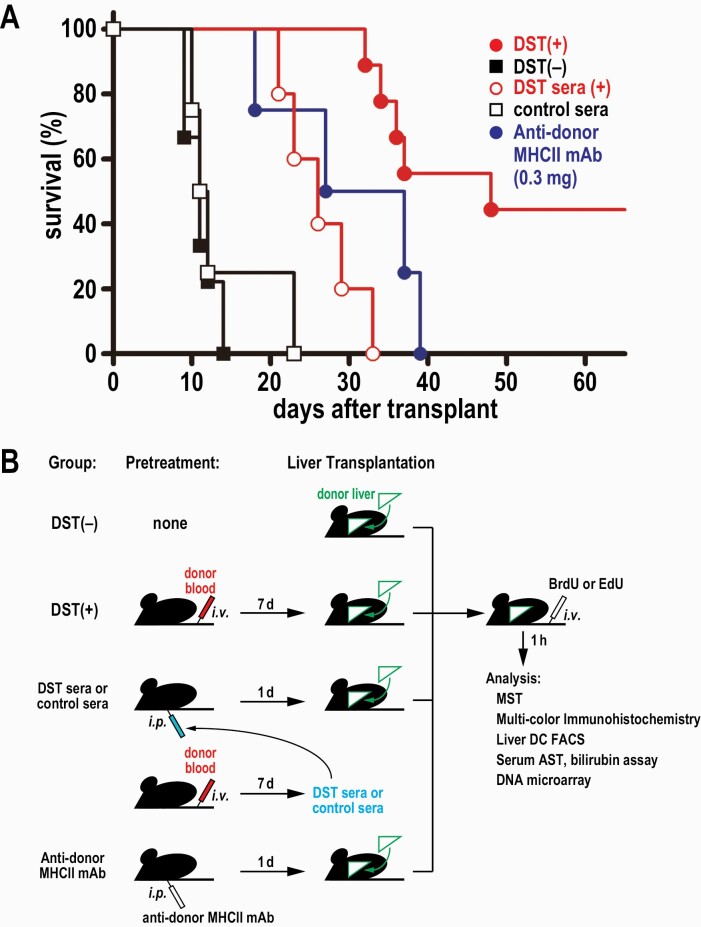
Lewis rats without DST pretreatment [DST(–) group] rejected ACI hepatic allografts with an MST of 11.1 ± 2.0 days (Fig. 1; Table 1), whereas the control Lewis to Lewis isografts survived indefinitely (not shown). DST pretreatment with donor ACI blood resulted in significant increase of MST to ~65.2 days after LTx [DST(+) group].
Fig. 1.
(A) Survival rate of liver allograft recipients after various treatments. The DST(–) or control sera transfer groups survived till 10~20 days after LTx. In contrast, the DST(+) group survived till 30 days or longer. The DST sera transfer and anti-donor MHCII mAb transfer groups survived till 20~60 days although shorter than the DST(+) group. (B) Scheme showing the experimental protocols for the DST(-), DST(+), DST sera, control sera and anti-donor MHCII mAb groups.
Table 1.
Survival time of liver graft recipients after various pretreatments
| Group | Pretreatment | Survival time (days) | MST |
|---|---|---|---|
| DST(−) | No treatment | 9, 9, 9, 11, 11, 11, 12, 14, 14 |
11.1 ± 2.0 |
| DST(+) | DST, 1 ml i.v., −7d | 32, 34, 36, 37, 48, >100 (four rats) |
65.2 ± 33.3* |
| DST sera | DST sera, 7 ml, i.p., −1 d | 23, 26, 29, 33 | 27.8 ± 4.3* |
| control sera | Normal sera, 7 ml, i.p., −1 d | 10, 11, 12, 23 | 14.0 ± 6.1NS |
| Anti-donor MHCII mAb | Anti-donor MHCII mAb, 3 mg, i.p., −1 d | 24, 26, 27, 29 | 26.5 ± 2.1* |
| Anti-donor MHCII mAb, 0.3 mg, i.p., −1 d | 18, 27, 37, 39 | 30.3 ± 9.7* |
MST, mean survival time (days ± SD); NS, not significant. Survival time of >100 days was counted as 100 days.
*P < 0.05 versus DST(−) group.
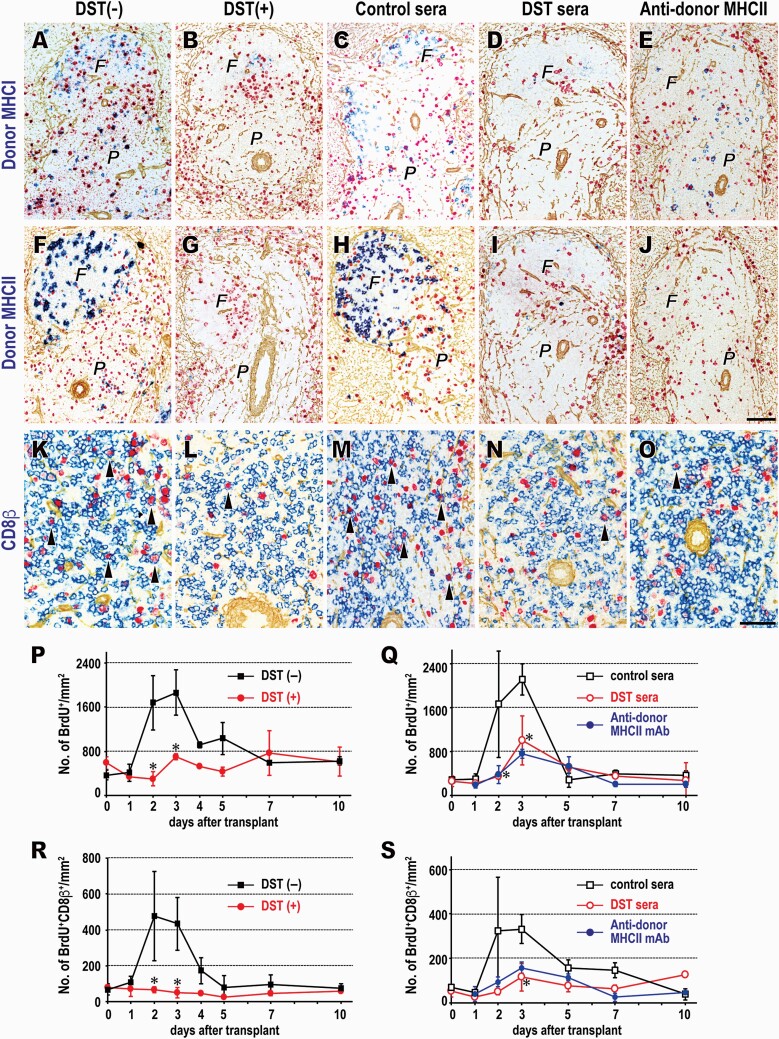
We compared the DST(+) and DST(–) groups regarding donor cell migration and the intrahost T-cell response after LTx using immunohistology. In the DST(–) group, the donor MHCI+ and MHCII+ cell migration (Fig. 2A and F; Supplementary Figure 1A and F), the whole proliferative response and CD8+ T-cell proliferative response in the T-cell area of the recipient spleen (PALS) (Fig. 2K, P and R) and the peripheral LNs (paracortex) (Supplementary Figure 1K, P and R) were readily observed on days 2–3 as reported previously (3). Donor MHCII+ cells in the T-cell area were DCs, although a small but significant number, and those in the B-cell follicle were considered to be mostly B cells (3). As expected, in the DST(+) group, these events were significantly inhibited (Fig. 2B, G, L, P and R; Supplementary Figure 1B, G, L, P and R), CD8+ T-cell proliferative responses being almost the background level (Fig. 2R; Supplementary Figure 1R).
Fig. 2.
Donor cell migration and intrahost T-cell response in the spleen in the DST(–) (A, F, K, P, R) and DST(+) (B, G, L, P, R) groups, and the control sera (C, H, M, Q, S), DST sera (D, I, N, Q, S) and anti-donor MHCII mAb (E, J, O, Q, S) groups. (A–O) Triple immunostaining of donor MHCI (A–E, blue), donor MHCII (F–J, blue), or CD8β (K–O, blue), BrdU (red), and type IV collagen (brown) 2 days after LTx. Arrowheads in K–O indicate BrdU+CD8β + proliferating T cells. F, lymph follicle; P, splenic PALS. Scale bars, 100 μm (A–J) or 50 μm (K–O). (P–S) Number of BrdU+ cells (P, Q) or BrdU+CD8β + cells (R, S) per square millimeter of splenic PALS. Mean ± SD, n = 3 rats each, *P < 0.05. * in (P) and (R) indicates significant suppression in the DST(+) versus DST(–) group and * in (Q) and (S) indicates that in the DST sera versus control sera group.
To determine whether donor cell trafficking was modulated by chemokines or cytokines that had been produced in the DST (+) group, we performed microarray analysis of both the graft liver and the recipient spleen shortly after LTx. Although a significant up-regulation of mRNA involved in T-cell trafficking, such as CXCL10, was observed in the DST(+) livers, other mRNA involved in DC trafficking were not detected (Supplementary Table 2), suggesting that chemokine-dependent migration inhibition is unlikely.
DST-antibodies alone inhibited donor cell migration and the intrahost CD8+ T-cell response
To confirm the inhibitory effects of DST-antibodies, recipient sera collected 7 days after DST were adoptively transferred to naive recipients 1 day prior to LTx (DST sera group). As reported previously (16), DST-antibodies recognize donor class I MHC, having donor passenger cell-depleting activity in vitro and in vivo and being able to suppress acute GvHD caused by donor T cells. As expected, this group had almost complete inhibition of donor cell migration to the SLOs (Fig. 2D and I; Supplementary Figure 1D and I), including the parathymic LNs (Supplementary Figure 2), whereas the group that was given normal sera (control sera group) exhibited the migration similar to the DST(–) group (Fig. 2C and H; Supplementary Figure 1C and H). DST sera also significantly suppressed the total and CD8+ T-cell proliferative responses in the splenic PALS (Fig. 2N, Q and S) and in the LN paracortex (Supplementary Figure 1N, Q and S), CD8+ T-cell proliferative responses being almost the background level (Fig. 2S; Supplementary Figure 1S). MST was significantly prolonged to ~28 days in the DST sera group, compared to ~14 days in the control sera group (Fig. 1; Table 1).
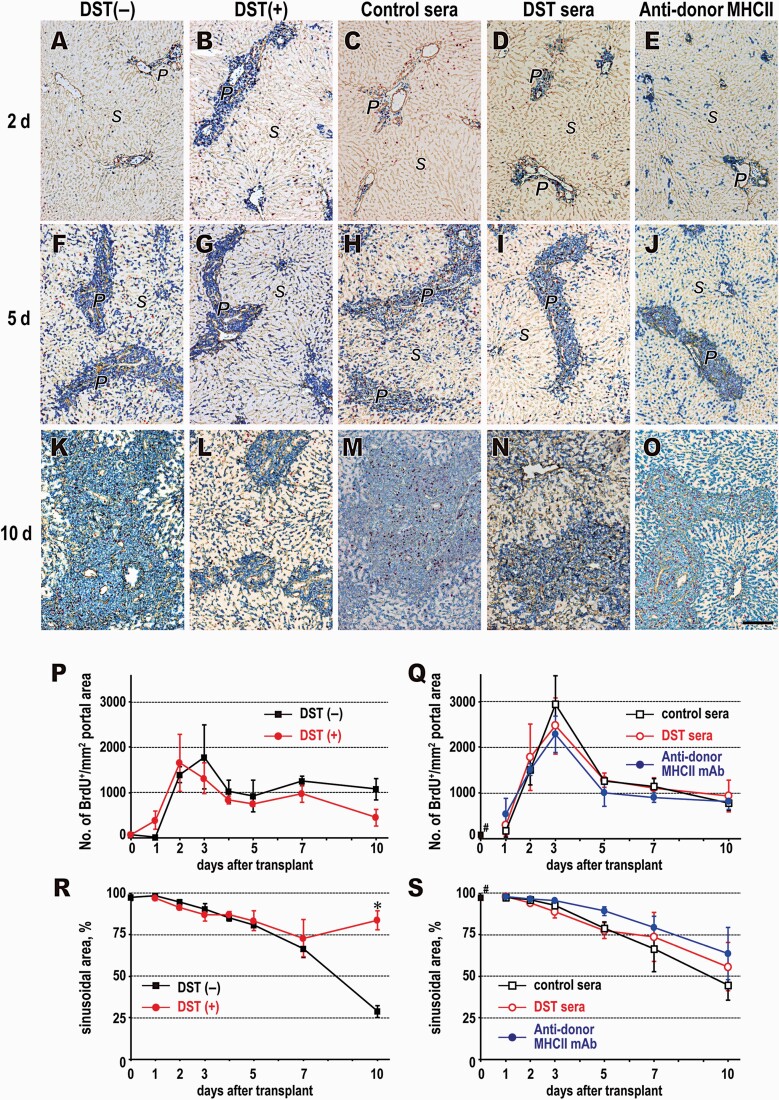
DST pretreatment suppressed the recipient cell infiltration and CD8+ T-cell response in the graft
The portal areas (22) in the DST(–) group demonstrated the recipient MHCI+ cell infiltration (Fig. 3A) and a proliferative response from 2 to 10 days (Fig. 3P), with TCRαβ + T cells as the major proliferating cells on day 3 (Supplementary Figure 3). In the sinusoidal area, the recipient MHCI+ cell infiltration started from day 2 (Fig. 3A), and piecemeal necrosis of hepatocytes progressively increased on days 7–10 (data not shown). By the time of rejection on day 10, the proportion of sinusoidal area was reduced to ~25% of the total surface area (Fig. 3R). In the DST(+) graft, the reduction in the sinusoidal area stopped on 7 days later, maintaining 75~80% of the total surface area (Fig. 3L and R).
Fig. 3.
DST pretreatment suppressed the intragraft host cell response on day 10. (A–O) Triple immunostaining of recipient MHCI (blue), BrdU (red) and type IV collagen (brown) in the graft, 2, 5 and 10 days after LTx in the DST(–) (A, F, K), DST(+) (B, G, L), control sera (C, H, M), DST sera (D, I, N) and anti-donor MHCII mAb (E, J, O) groups. P, portal area; S, sinusoidal area. Scale bars, 200 μm. (P–S) Time kinetics for the number of BrdU+ cells mm−2 in the portal area (P, Q) and the proportion of the sinusoidal area in total area (R, S). Note the conserved sinusoidal area (L, R) in DST(+) group on day 10. Mean ± SD, n = 3 rats each, *P < 0.05. #Normal donor liver before LTx.
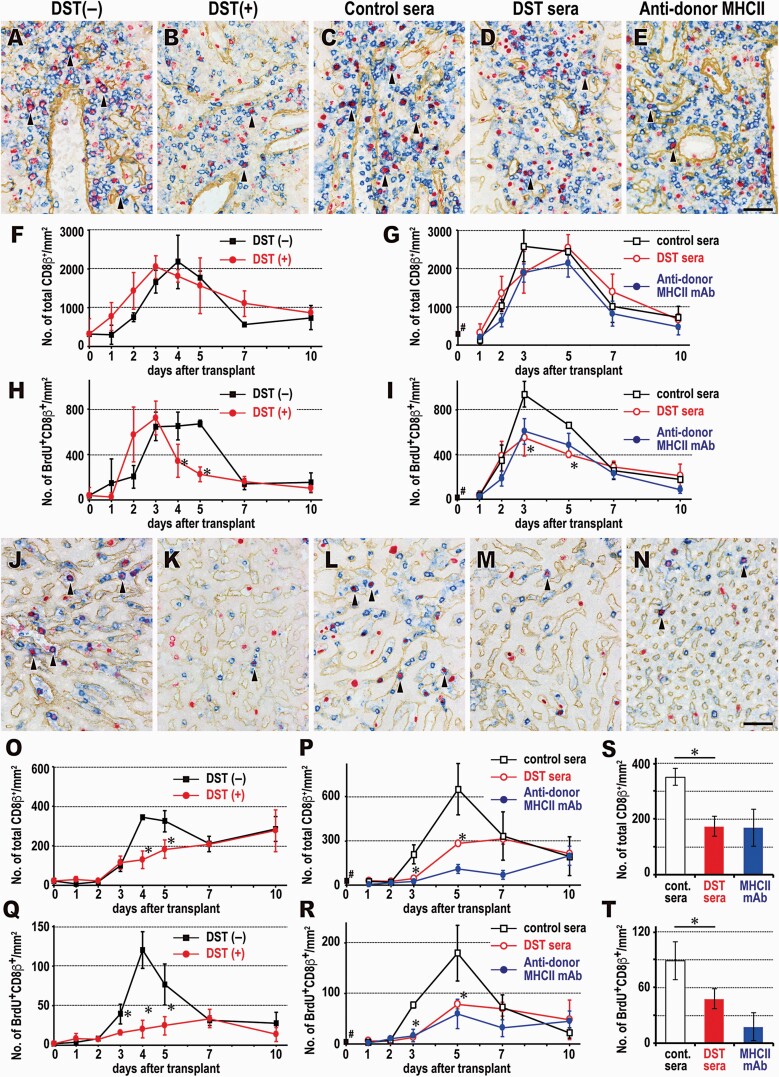
In the DST(–) graft liver, the number of CD8+ T cells increased progressively until day 4 (Fig. 4F and O) and actively proliferated from day 3 to 5 after LTx (Fig. 4A, H, J and Q). CD8+ T cells were the major proliferating cells, constituting >50% of the total BrdU+ cells on day 4 in both the portal (Supplementary Figure 4A) and sinusoidal areas (Supplementary Figure 4B).
Fig. 4.
CD8 T-cell response in the portal (A–I) and sinusoidal (J–T) areas of the graft liver after various treatments. (A–E, J–N) Triple immunostaining 4 days after LTx for CD8β (blue), BrdU (red) and type IV collagen (brown). Many BrdU+CD8β + proliferating T cells (arrows) were found in the DST(–) (A,J) and control sera (C,L) groups but fewer in the DST(+) (B,K), DST sera (D,M) and anti-donor MHCII mAb (E,N) groups. Scale bars, 50 μm. (F–I, O–R) Number of total CD8β + and BrdU+CD8β + cells mm−2 area in the DST(–) and DST(+) groups (F, H, O, Q) or control sera, DST sera and anti-MHCII mAb groups (G, I, P, R). (S, T) Digital quantification of (P) and (R) on day 5, respectively, by image analysis. Mean ± SD, n = 3 rats each, *P < 0.05. * in (H), (O) and (Q) indicates significant suppression in the DST(+) vs DST(–) group and * in (I), (P), (R), (S) and (T) indicates that in the DST sera vs control sera group. #Normal donor liver before LTx.
In the DST(+) graft liver, infiltration of CD8+ T cells in the sinusoidal area was significantly suppressed from day 4 to 5 (Fig. 4K and O). The CD8+ T-cell proliferative response was also suppressed significantly but partially in the portal and sinusoidal areas on days 4 and 5 (Fig. 4B, H, K and Q). Notably, CD8+ T cells were not the major proliferating cells in the DST(+) group, constituting only ~20% of total BrdU+ cells on day 4 in both areas (Supplementary Figure 4A and B). Other proliferating cells were mostly recipient MHCI+ cells and partly donor cells (Supplementary Figure 5). Some T cells and a few B and NK cells but neither monocytes nor polymorphonuclear leukocytes were proliferating. Bile duct epithelia and CD163+ cells in the sinusoidal area showed active proliferation and up-regulation of donor MHCII molecules in all groups examined (Supplementary Figure 5), as reported in rats during alloresponses (28), probably via IFN-γ (29). Because donor MHCII+ cells in the sinusoidal area were mostly CD163+ Kupffer cells, other non-parenchymal cells such as sinusoidal endothelial cells and Ito cells in this rat model may not up-regulate MHCII.
DST-antibodies suppressed the CD8+ T-cell response in the graft tissues
By transfer of DST sera, the total proliferative response in the portal area and the expansion of the sinusoidal area were not suppressed compared to the control sera group (Fig. 3Q and S). However, infiltration of CD8+ T cells in the sinusoidal area (Fig. 4M and P) and the CD8+ T-cell proliferative response in the portal (Fig. 4D and I) and sinusoidal (Fig. 4M and R) areas on days 3 and 5 were suppressed significantly but partially. Digital quantification by image analysis of the sinusoidal areas on day 5 confirmed the significant suppression of CD8+ T cells and the CD8+ T-cell proliferative response in the DST sera group (Fig. 4S and T) compared to the control sera group. Concerning the proportion of BrdU+CD8+ T cells to total BrdU+ cells, the control sera group had ~40% in both areas, and this was suppressed in the DST sera group on day 3 in the portal area (Supplementary Figure 4A) and day 5 in the sinusoidal area (Supplementary Figure 4B).
Decrease in graft resident DCs with DST treatment or DST sera transfer
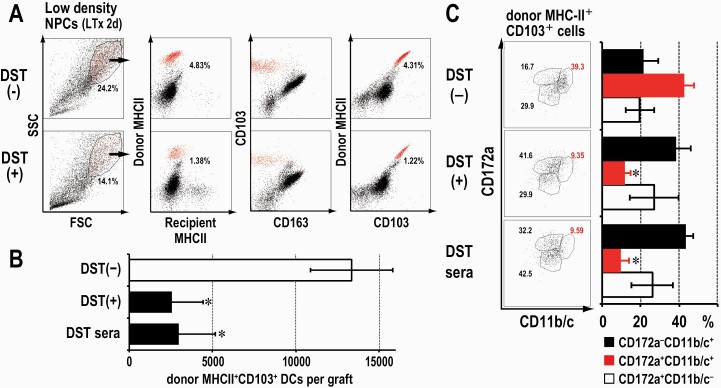
When non-parenchymal cells were isolated from graft livers on days 1 and 2 and analyzed by FACS, ~4 and ~1% of low-density cells were donor MHCII+CD103+CD163– DCs in the DST(–) and DST(+) groups, respectively (Fig. 5A). CD163 is a macrophage marker including Kupffer cells and not expressed by DCs (3). The absolute number of donor MHCII+CD103+ DCs per graft was ~1.3 × 104, ~2.5 × 103, and ~3 × 103 in the DST(–), DST(+), and DST sera groups, respectively (Fig. 5B). The proportion of the previously reported radioresistant immunogenic CD172a+CD11b/c+ DC subset (4) among total DCs was reduced from ~40% in the DST(–) group to ~10% in the DST(+) and DST sera groups (Fig. 5C). These results indicate that DST-antibodies reduced the total DCs in the graft to ~25% of the DST(–) group and, more selectively, almost depleted the CD172a+CD11b/c+ subset to ~6%.
Fig. 5.
Resident DCs were depleted by DST treatment or DST sera transfer, whereas remaining DCs still formed clusters with proliferating cells. (A–C) FACS analysis of donor DCs in the graft livers of the DST(–), DST(+) and DST sera groups. Representative data from three independent experiments. Mean ± SD, n = 3 rats each, *P < 0.05. (A) Proportions of donor MHCII+CD103+CD163– DCs in non-parenchymal cells of the whole graft. Red plots correspond to donor DC population. (B) Absolute numbers of donor MHCII+CD103+ DCs in the whole graft. *P < 0.05. (C) Proportions of three DC subsets isolated from grafts. Note that the proportion of CD172a+CD11b/c+ cells decreased significantly. *P < 0.05.
Using immunohistology, cluster formation by donor MHCII+ cells with proliferating BrdU+ cells was observed constantly in the portal area from days 2 to 3 in both the DST(–) and DST(+) groups (Supplementary Figure 6A and C), though the number of donor MHCII+ cells decreased in the DST(+) group (Fig. 5A). Among these cluster-forming cells, 53.1–88.4% in the DST(–) group and 45.6–69.6% in the DST(+) group were CD103+ (Supplementary Figure 6B, D and E), CD11c+ or CD86+ (Supplementary Figure 6E), which indicates that more than half of donor MHCII+ cells possess DC markers and suggests that the DC cluster is a site for the direct pathway. Four-color immunofluorescence staining of the DST(–) graft confirmed the presence of donor MHCII+CD103+ DCs either CD11b/c+ (Supplementary Figure 6F–I) or CD11b/c– (data not shown), clustering with proliferating EdU+ cells. Proliferating cells were mostly recipient MHCI+ cells (Supplementary Figure 5).
DST-antibodies temporarily affected graft function at a very early stage
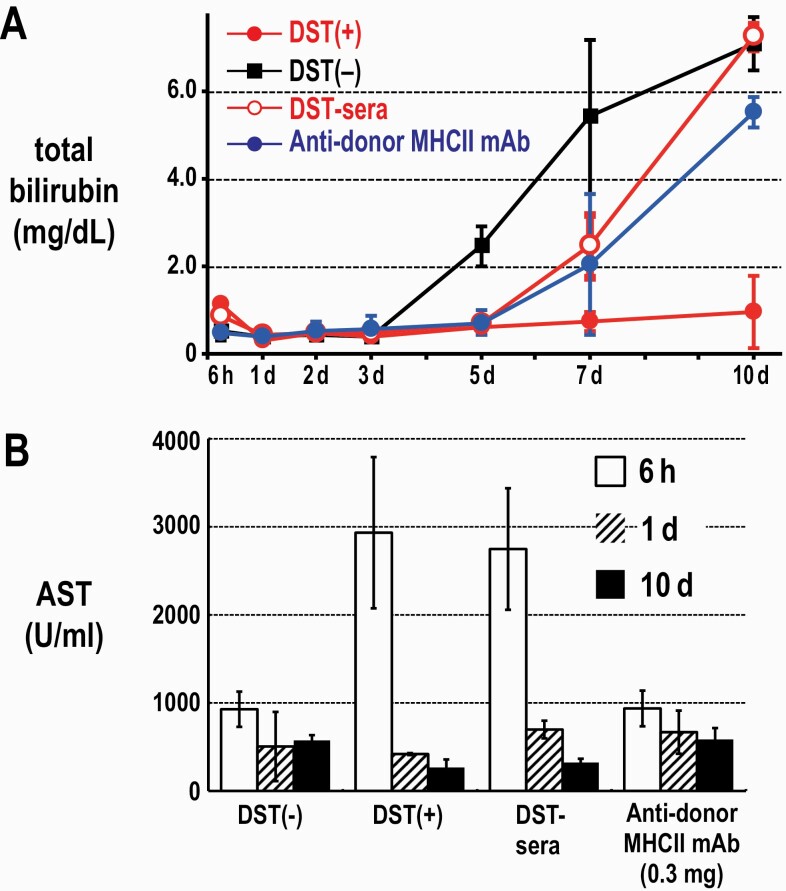
Because most nucleated cells are MHCI+, DST-antibodies may have deleterious effects on graft cells as reported for kidney (30) and heart allografts (31). In the DST(–) group, serum total bilirubin concentration progressively increased to a high level from day 5 until rejection. In contrast, the DST(+) group maintained normal bilirubin levels throughout the experimental period. In the DST sera group, the bilirubin levels were normal from day 1 to 5, but increased from day 7 and became comparable to the DST(–) group at day 10 (Fig. 6A). The serum AST levels were <1000 U ml−1 throughout the experimental period in the DST(–) group. However, both the DST(+) and DST sera groups at a very early stage, 6 h after LTx, exhibited a temporal increase in serum AST to as high as 3000 U ml−1 (Fig. 6B). This indicates that DST-antibodies also recognized graft hepatocytes and/or non-parenchymal cells that are donor MHCI+, leading to reversible temporary graft damage within 1 day following LTx.
Fig. 6.
Time kinetic change in total bilirubin (A) and serum AST (B) levels after LTx. Note the temporary increase in AST level at 6 h after LTx in the DST(+) and DST sera groups but not in the DST(–) or anti-donor MHCII mAb groups (B). Mean ± SD, n = 3 rats each.
Antibody to donor MHCII suppressed the rejection without early graft damage
In general, the liver parenchymal cells, non-parenchymal cells and T cells are MHCI+ but MHCII–, whereas DCs, B cells and some Kupffer cells in rats are MHCI+MHCII+ (32). Accordingly, we searched antibodies specific for rat polymorphic donor MHCII (RT1.Ba) antigen, and we found that intra-peritoneal injection of 0.3 mg per rat (1.5 mg kg−1 BW) of anti-RT1.Ba OX76 mAb 1 day prior to LTx almost completely inhibited the migration of donor MHCII+ cells, including DCs, to the recipient SLOs (Fig. 2E and J; Supplementary Figure 1E and J). MST was also significantly prolonged to ~30 days (Table 1). As expected, the serum AST level in the anti-donor MHCII mAb group was similar to the DST(–) group at 6 h indicating that this antibody did not induce the early temporary graft damage (Fig. 6B). The serum total bilirubin levels were similar to the DST sera group (Fig. 6A).
With this pretreatment, the total and CD8+ T-cell proliferative responses in the spleen (Fig. 2Q and S) and total proliferative response in the peripheral LNs (Supplementary Figure 1Q) on day 3 were suppressed in a similar fashion as the DST sera group. The CD8+ T-cell infiltration (Fig. 4N, P and S) and CD8+ T-cell proliferative responses (Fig. 4N, R and T) in the graft sinusoidal area were also suppressed in a similar fashion as the DST sera group on day 5. These results indicate that the suppressive effect of anti-MHCII antibody on the CD8+ T-cell response, as well as rejection, was almost similar to that of DST-antibodies directed to donor MHCI.
In contrast, donor passenger MHCI+MHCII– cells were not depleted by this mAb (Fig. 2E; Supplementary Figure 1E): they were mostly T cells (data not shown), as rat T cells are mostly MHCI+MHCII– (33). Donor T cells temporarily showed a proliferative response, indicating a presence of weak GvH reaction at the migrated sites via the direct pathway (Fig. 2E), as in the DST(–) group and in the previous study (3). The sustained CD8+ T-cell proliferative response in the LNs on day 3 may be due, at least in part, to these cells (Supplementary Figure 1S).
DST treatment enhanced the recipient Treg response
The Treg response was estimated as the number of BrdU+Foxp3+ cells mm−2 area as reported previously (16), with a significant increase in the PALS 3 days after DST treatment only. The Treg response after LTx was significantly enhanced in the splenic PALS from day 5 to 7 (Supplementary Figure 7A and C) and in the graft portal area from day 4 to 7 (Supplementary Figure 7E and G) in the DST(+) group compared to the DST(–) group. In contrast, other groups, including the DST sera, control sera and MHCII antibody groups, exhibited a similar response as the DST(–) group (Supplementary Figure 7D and H). Because recipient spleen in the DST(+) group had almost no donor cell migration (Fig. 2B), actively proliferating Tregs (Supplementary Figure 7B) are mostly of recipient origin in the spleen. In normal donor liver before LTx to DST-pretreated recipient contained very few Foxp3+ cells, whereas one day after LTx, the graft showed quick increase of Foxp3+ cells (data not shown). This indicates that most of them are of recipient origin recruited from the recipient spleen and other SLOs. These results demonstrate that only DST pretreatment, but not antibody transfer could induce a priming effect on the recipient Treg response after the LTx.
DST or antibody treatment did not enhance the donor-specific antibody response
We examined time kinetics of the donor-specific antibody production after LTx, which might become problematic at the late stage. Both IgM and IgG1 anti-donor antibody titers in the DST sera and anti-donor MHCII antibody groups as well as the DST(+) group were comparable to, or lower than the DST(–) group up to day 10 (Supplementary Figure 8). Accordingly, we consider that neither DST nor the antibody pretreatments enhance alloantibody production at the late stage. The relatively high antibody response in the MHCII antibody and DST(−) group may be due to remaining donor T cells, which can readily induce DST-antibody production via the indirect pathway by recipient DCs as reported recently (7).
Discussion
In this study, we examined the role of DST-antibodies in DST-induced immunosuppression following LTx. There are several novel findings. First, DST-antibodies inhibited the migration of donor passenger cells, including both CD172+CD11b/c+ and CD172+CD11b/c– immunogenic liver DC subsets, to recipient SLOs, and significantly suppressed the proliferative response of CD8+ T cells. Second, DST-antibodies depleted sessile DCs in the graft to ~20% of control, with a strong reduction in the CD172+CD11b/c+ subset. Within the grafts, the CD8+ T-cell response was also significantly suppressed, and MST was prolonged to ~28 days, compared to ~14 days in control. Third, an mAb to donor MHCII had similar effects as DST-antibodies, except for the absence of the temporary graft damage and the presence of a weak GvH reaction. Fourth, only the DST(+) group enhanced the expansion of Foxp3+ Tregs in the spleen and the graft portal area after LTx.
The inhibition of passenger cell migration by DST sera is due to donor cell elimination by the complement-dependent cytolytic activity of donor MHCI-specific depleting-antibodies (16). The DNA microarray analysis suggested that chemokines or cytokines play a minor role. We previously showed that recipient NK cells are crucial for rejecting donor cells in a rat GvHD model following LTx (20) and for elimination of transfused allogeneic T cells in the splenic PALS and peripheral LNs (7). These cells may be also involved in inhibition of passenger cell migration. Suppression of the CD8+ T-cell response and the proliferative response in the T-cell areas of the recipient SLOs indicates an absence of the allorecognition because of passenger cell elimination by DST-antibodies.
Then, which donor leukocyte type is responsible for the host CD8+ T-cell response? We previously reported that after LTx without DST treatment, ~2 × 106 and ~2 × 103 donor cells migrated to the host spleen and Peyer’s patches, respectively. Migrated cells contained a significant number of CD11c+ and/or CD103+ DCs and other cell types were T and B cells (3). In contrast, when 1 × 108 donor thoracic duct lymphocytes were transferred, ~6 × 106 and ~5 × 103 donor cells migrated to these organs, where migrated cells were mostly T and B cells and DCs were very few. Concerning the CD8+ T-cell proliferative response, LTx induced a significantly higher response in the host spleen than the thoracic duct lymphocytes transfer group did (3). These results accord with the anti-donor MHCII mAb group in the present study, where donor passenger MHCI+MHCII– cells, mostly T cells and few DCs, readily migrated to the SLOs, while the CD8+ T-cell proliferative response in the spleen was also weak. Furthermore, by mixed leukocyte culture experiment, the DCs isolated from the donor liver with/without irradiation induced a very high proliferation of recipient T cells comparable to splenic DCs (4). These results indicate that liver DCs can induce donor-specific alloresponses and suggest that the CD8+ T-cell proliferative response is donor-specific. Although we did not examine the response of recipient cells other than CD8+ T cells to graft cells, CD4+ T cells and non-lymphoid cells may also be involved. Concerning donor T cells, we recently reported that they are rather involved in inducing helper T-cell-dependent DST-antibody response via the indirect pathway (7). Therefore, we demonstrate that donor DCs are mainly responsible for inducing the host CD8+ T-cell response and that T and B cells are less potent inducers.
For the allorecognition leading to the CD8+ T-cell response in the recipient SLOs, the direct pathway via donor DCs may be involved, as shown by the cluster formation of donor DCs with recipient proliferating cells in the T-cell areas of SLOs. However, the semi-direct pathway cannot be neglected because extracellular vesicles can be produced by migrated DCs, as exosomes or passively released from lysed donor cells (34). The cluster formation by recipient MHCII+ putative DCs was also observed in the SLOs following LTx (3), which supports a role of this pathway. Therefore, the results indicate that elimination of passenger DCs via depletion by DST-antibody is crucial for blocking the generation of an intrahost CD8+ T-cell response via either the direct or semi-direct pathway.
The CD8+ T-cell proliferative responses in the DST(+) and DST sera groups, especially in the sinusoidal area, were significantly suppressed around days 3–5. This suppression was preceded by the suppression of the CD8+ T-cell response in the recipient SLOs around days 2–3. This suggests that the intragraft CD8+ T-cell response was mainly executed by recruiting CD8+ T cells that had been preactivated in the recipient SLOs. We recently reported the transmigration of activated CD8+ T cells across portal vein endothelial cells into the portal area following LTx (24), which supports this possibility. Therefore, the result strongly suggests that a major effect of DST-antibodies on the graft tissues is suppression of the CD8+ T-cell response via inhibition of the direct or semi-direct pathway in recipient SLOs. This may lead to suppression of CD8+ T-cell-mediated early stage tissue injury in the graft liver.
The persistence of small fractions of resident DCs and the cluster formation in the graft after encountering DST-antibodies suggest that the intragraft direct or semi-direct pathway was not completely inhibited. This may explain the incomplete suppression of the intragraft CD8+ T-cell response in the DST(+) and DST sera groups. Among the remaining resident DCs, the CD172a+CD11b/c+ immunogenic DC subset, the major population in the graft of the DST(–) group, was very small in both groups. Because this subset is suggested to be responsible for the acute rejection of the irradiated graft liver (4), intragraft depletion of this subset by DST-antibodies may lead to suppression of the early stage tissue injury.
Compared to DST sera or anti-donor MHCII mAb, DST pretreatment more strongly suppressed the intragraft response and the transplant rejection. This suggests the presence of additional suppressive factors, such as Tregs (18), inhibitory costimulatory molecules (35) and other inherent factors in the liver tissue (36). In this regard, we found enhanced and sustained proliferation of Foxp3+ Tregs in the PALS and the graft of the DST(+) group but not the DST sera or anti-MHCII mAb groups. Variations of MST in the DST(+) group are difficult to explain but may reflect individual differences in generation of suppressive factors (14).
Using a depleting antibody that recognizes a polymorphic determinant of donor MHCII, we could obtain the suppression similar to that of the DST sera group, but without detectable graft damage. Concerning tolerogenic DCs, immature resident DCs have been reported in the mouse liver (12) and we had better conserve these cells. In a preliminary rat study, we also found donor MHCIIlowCD103+ DCs in the donor liver (H. Ueta et al., unpublished results) in a tolerance-inducing strain combination (37). Because they are MHCIIlow, the depleting effects of anti-MHCII antibodies on these immunosuppressive DCs may be small.
Clinically, it is known that human liver grafts possess some degree of alloantibody resistance compared to kidney or heart grafts (38). Accordingly, we consider that administration of anti-donor MHC antibodies, especially donor MHCII (HLA-DR)-specific antibodies, may ameliorate the acute phase rejection of liver transplants via selective suppression of the CD8+ T-cell response with more or less tolerable side effects. In this way, we may reduce the usage of life-threatening immunosuppressive drugs. Furthermore, we recently reported that selective T-cell depletion by ex vivo perfusion of an antibody prevents LTx-GvHD without affecting recipient immunity (20). Therefore, further selective ex vivo depletion of donor passenger DCs by mature DC-specific antibodies, such as anti-human CD83 mAbs (39), would also be applicable as another possibility.
Funding
This work was supported in part by JSPS KAKENHI grant numbers JP17K08518, JP23790235 and JP18K06838 and JST SORST3 and by official donations from Drs. Kazutaka Matsushita and Taro Kariya (no conflict of interest).
Supplementary Material
Acknowledgements
The authors are grateful to late Prof. Ralph Steinman, Dr Hiromichi Tsurui and Dr Masanori Niimi for their valuable discussions and Dr Xiao-Kang Li for donating GFP-Lewis rats. We are thankful to Dr Hiroe Kon, Ms Kyoko Ohta-Kato, Junko Sakumoto, Keiko Sasaki, Yoko Yamada, Yasuko Nonaka and Mr Shuji Abe for their excellent technical assistance. K. Matsuno and H.U. participated in research design and in writing of the paper. H.U., X.-D.X., B.Y., E.Y., Y.H. and M.M.-N. participated in performing animal experiments. Y.K., S.Z., Y.S., S.U., K.R. and T.T. participated in performing immunohistology and cell analysis. H.U., N.T., S.U. and K. Matsushima participated in data analysis.
Conflicts of interest statement: the authors declared no conflicts of interest.
References
- 1. Saiki, T., Ezaki, T., Ogawa, M. and Matsuno, K. 2001. Trafficking of host- and donor-derived dendritic cells in rat cardiac transplantation: allosensitization in the spleen and hepatic nodes. Transplantation 71:1806. [DOI] [PubMed] [Google Scholar]
- 2. Wood, K. J. 2003. Passenger leukocytes and microchimerism: what role in tolerance induction? Transplantation 75(suppl. 9):17S. [DOI] [PubMed] [Google Scholar]
- 3. Ueta, H., Shi, C., Miyanari, N.et al. 2008. Systemic transmigration of allosensitizing donor dendritic cells to host secondary lymphoid organs after rat liver transplantation. Hepatology 47:1352. [DOI] [PubMed] [Google Scholar]
- 4. Yu, B., Ueta, H., Kitazawa, Y.et al. 2012. Two immunogenic passenger dendritic cell subsets in the rat liver have distinct trafficking patterns and radiosensitivities. Hepatology 56:1532. [DOI] [PubMed] [Google Scholar]
- 5. Lechler, R., Ng, W. F. and Steinman, R. M. 2001. Dendritic cells in transplantation—friend or foe? Immunity 14:357. [DOI] [PubMed] [Google Scholar]
- 6. Afzali, B., Lechler, R. I. and Hernandez-Fuentes, M. P. 2007. Allorecognition and the alloresponse: clinical implications. Tissue Antigens 69:545. [DOI] [PubMed] [Google Scholar]
- 7. Kitazawa, Y., Ueta, H., Sawanobori, Y.et al. 2019. Novel targeting to XCR1+ dendritic cells using allogeneic T cells for polytopical antibody responses in the lymph nodes. Front. Immunol. 10:1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Crozat, K., Guiton, R., Contreras, V.et al. 2010. The XC chemokine receptor 1 is a conserved selective marker of mammalian cells homologous to mouse CD8alpha+ dendritic cells. J. Exp. Med. 207:1283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lahoud, M. H., Ahmet, F., Kitsoulis, S.et al. 2011. Targeting antigen to mouse dendritic cells via Clec9A induces potent CD4 T cell responses biased toward a follicular helper phenotype. J. Immunol. 187:842. [DOI] [PubMed] [Google Scholar]
- 10. Nakahara, T., Uchi, H., Lesokhin, A. M.et al. 2010. Cyclophosphamide enhances immunity by modulating the balance of dendritic cell subsets in lymphoid organs. Blood 115:4384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sato, K., Yamashita, N., Yamashita, N., Baba, M. and Matsuyama, T. 2003. Regulatory dendritic cells protect mice from murine acute graft-versus-host disease and leukemia relapse. Immunity 18:367. [DOI] [PubMed] [Google Scholar]
- 12. Ochando, J., Ordikhani, F., Jordan, S., Boros, P. and Thomson, A. W. 2020. Tolerogenic dendritic cells in organ transplantation. Transpl. Int. 33:113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Yrlid, U., Cerovic, V., Milling, S., Jenkins, C. D., Klavinskis, L. S. and MacPherson, G. G. 2006. A distinct subset of intestinal dendritic cells responds selectively to oral TLR7/8 stimulation. Eur. J. Immunol. 36:2639. [DOI] [PubMed] [Google Scholar]
- 14. Josien, R., Douillard, P., Guillot, C.et al. 1998. A critical role for transforming growth factor-beta in donor transfusion-induced allograft tolerance. J. Clin. Invest. 102:1920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Jovičić, S., Ležaić, V., Simonović, R., and Djukanović, L. 2011. Beneficial effects of donor-specific transfusion on renal allograft outcome. Clin Transplant. 25:317. [DOI] [PubMed] [Google Scholar]
- 16. Ueta, H., Kitazawa, Y., Sawanobori, Y.et al. 2018. Single blood transfusion induces the production of donor-specific alloantibodies and regulatory T cells mainly in the spleen. Int. Immunol. 30:53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Jaskłowska-Englisz, M., Olszewski, W. L., Maksymowicz, M. and Ziółkowska, A. 1998. Protection of heart and rejection of lymphocyte allografts from the same donor in recipients of donor-specific transfusions. Ann. Transplant. 3:28. [PubMed] [Google Scholar]
- 18. Kitade, H., Kawai, M., Rutgeerts, O.et al. 2005. Early presence of regulatory cells in transplanted rats rendered tolerant by donor-specific blood transfusion. J. Immunol. 175:4963. [DOI] [PubMed] [Google Scholar]
- 19. Hontsu, S., Yoneyama, H., Ueha, S.et al. 2004. Visualization of naturally occurring Foxp3+ regulatory T cells in normal and tumor-bearing mice. Int. Immunopharmacol. 4:1785. [DOI] [PubMed] [Google Scholar]
- 20. Yu, E., Ueta, H., Kimura, H., Kitazawa, Y., Sawanobori, Y. and Matsuno, K. 2017. Graft-versus-host disease following liver transplantation: development of a high-incidence rat model and a selective prevention method. Am. J. Transplant. 17:979. [DOI] [PubMed] [Google Scholar]
- 21. Saiki, T., Ezaki, T., Ogawa, M., Maeda, K., Yagita, H. and Matsuno, K. 2001. In vivo roles of donor and host dendritic cells in allogeneic immune response: cluster formation with host proliferating T cells. J. Leukoc. Biol. 69:705. [PubMed] [Google Scholar]
- 22. Matsuno, K., Ezaki, T., Kudo, S. and Uehara, Y. 1996. A life stage of particle-laden rat dendritic cells in vivo: their terminal division, active phagocytosis, and translocation from the liver to the draining lymph. J. Exp. Med. 183:1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lin, T. J., Hirji, N., Stenton, G. R.et al. 2000. Activation of macrophage CD8: pharmacological studies of TNF and IL-1 beta production. J. Immunol. 164:1783. [DOI] [PubMed] [Google Scholar]
- 24. Kariya, T., Ueta, H., Xu, X. D.et al. 2016. Direct evidence for activated CD8+ T cell transmigration across portal vein endothelial cells in liver graft rejection. J. Gastroenterol. 51:985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Smilek, D. E., Boyd, H. C., Wilson, D. B., Zmijewski, C. M., Fitch, F. W. and McKearn, T. J. 1980. Monoclonal rat anti-major histocompatibility complex antibodies display specificity for rat, mouse, and human target cells. J. Exp. Med. 151:1139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yu, E., Goto, M., Ueta, H.et al. 2016. Expression of area-specific M2-macrophage phenotype by recruited rat monocytes in duct-ligation pancreatitis. Histochem. Cell Biol. 145:659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Kitazawa, Y., Ueta, H., Hünig, T., Sawanobori, Y. and Matsuno, K. 2015. A novel multicolor immunostaining method using ethynyl deoxyuridine for analysis of in situ immunoproliferative response. Histochem. Cell Biol. 144:195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Stet, R. J., Thomas, C., Koudstaal, J., Hardonk, M. J., Hulstaert, C. E. and Nieuwenhuis, P. 1986. Graft-versus-host disease in the rat: cellular changes and major histocompatibility complex antigen expression in the liver. Scand. J. Immunol. 23:81. [DOI] [PubMed] [Google Scholar]
- 29. Giroux, M., Schmidt, M. and Descoteaux, A. 2003. IFN-gamma-induced MHC class II expression: transactivation of class II transactivator promoter IV by IFN regulatory factor-1 is regulated by protein kinase C-alpha. J. Immunol. 171:4187. [DOI] [PubMed] [Google Scholar]
- 30. Tinckam, K. J. and Chandraker, A. 2006. Mechanisms and role of HLA and non-HLA alloantibodies. Clin. J. Am. Soc. Nephrol. 1:404. [DOI] [PubMed] [Google Scholar]
- 31. Furuya, T., Murase, N., Nakamura, K.et al. 1992. Preformed lymphocytotoxic antibodies: the effects of class, titer and specificity on liver vs. heart allografts. Hepatology 16:1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Matsuno, K. and Ezaki, T. 2000. Dendritic cell dynamics in the liver and hepatic lymph. Int. Rev. Cytol. 197:83. [DOI] [PubMed] [Google Scholar]
- 33. Reizis, B., Schramm, C., Cohen, I. R. and Mor, F. 1994. Expression of major histocompatibility complex class II molecules in rat T cells. Eur. J. Immunol. 24:2796. [DOI] [PubMed] [Google Scholar]
- 34. Baxter, A. A., Phan, T. K., Hanssen, E.et al. 2019. Analysis of extracellular vesicles generated from monocytes under conditions of lytic cell death. Sci. Rep. 9:7538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Yang, H. R., Chou, H. S., Gu, X.et al. 2009. Mechanistic insights into immunomodulation by hepatic stellate cells in mice: a critical role of interferon-gamma signaling. Hepatology 50:1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Morita, M., Fujino, M., Jiang, G.et al. 2010. PD-1/B7-H1 interaction contribute to the spontaneous acceptance of mouse liver allograft. Am. J. Transplant. 10:40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Dresske, B., Lin, X., Huang, D. S., Zhou, X. and Fändrich, F. 2002. Spontaneous tolerance: experience with the rat liver transplant model. Hum. Immunol. 63:853. [DOI] [PubMed] [Google Scholar]
- 38. O’Leary, J. G., Demetris, A. J., Friedman, L. S.et al. 2014. The role of donor-specific HLA alloantibodies in liver transplantation. Am. J. Transplant. 14:779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Seldon, T. A., Pryor, R., Palkova, A.et al. 2016. Immunosuppressive human anti-CD83 monoclonal antibody depletion of activated dendritic cells in transplantation. Leukemia 30:692. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.