Summary
Background
Rotavirus vaccine effectiveness is reduced among children in low-income countries. Indirect (transmission-mediated) effects of rotavirus vaccine might contribute to the total population effect of vaccination. We aimed to examine risk factors for transmission of rotavirus to household contacts in Blantyre, Malawi, and estimated the effectiveness of rotavirus vaccine in preventing transmission of infection to household contacts.
Methods
In this prospective household cohort study, we recruited children born after Sept 17, 2012, and aged at least 6 weeks (vaccine-eligible children) with acute rotavirus gastroenteritis and their household contacts, in four government health facilities in Blantyre, Malawi. Clinical data, a bulk stool sample, and 1–2 mL of serum were collected from case children at presentation. Clinical data and stool samples were also prospectively collected from household contacts over 14 days from presentation. A single stool sample was collected from control households containing asymptomatic children who were frequency age-matched to case children. Samples were tested for rotavirus using semi-quantitative real-time PCR and for anti-rotavirus IgA using a semi-quantitative sandwich ELISA. Risk factors for household transmission of rotavirus infection and clinical disease, including disease severity and faecal shedding density, were identified using mixed effects logistic regression. Vaccine effectiveness against transmission was estimated as 1 minus the ratio of secondary attack rates in vaccinated and counterfactual unvaccinated populations, using vaccine effectiveness estimates from the associated diarrhoeal surveillance platform to estimate the counterfactual secondary attack rate without vaccination.
Findings
Between Feb 16, 2015, and Nov 11, 2016, we recruited 196 case households (705 members) and 55 control households (153 members). Household secondary attack rate for rotavirus infection was high (434 [65%] of 665 individuals) and secondary attack rate for clinical disease was much lower (37 [5%] of 698). Asymptomatic infection in control households was common (40 [28%] of 144). Increasing disease severity in an index child (as measured by Vesikari score) was associated with increased risk of transmission of infection (odds ratio 1·17 [95% CI 1·06–1·30) and disease (1·28 [1·08–1·52]) to household contacts. Estimated vaccine effectiveness against transmission was 39% (95% CI 16–57).
Interpretation
Rotavirus vaccine has the potential to substantially reduce household rotavirus transmission. This finding should be considered in clinical and health economic assessments of vaccine effectiveness.
Funding
Wellcome Trust, US National Institutes of Health, and US National Institute of Allergy and Infectious Diseases.
Introduction
Rotavirus vaccine has been introduced into over 90 countries worldwide, including 45 low-income and middle-income countries.1 However, rotavirus vaccine effectiveness is reduced in low-income countries, where disease burden is highest, compared with high-income countries.2 Thus, despite high vaccine coverage, rotavirus remains the commonest cause of hospital admissions due to diarrhoeal disease in some low-income countries.3 Because direct vaccine effectiveness is reduced in low-income and middle-income countries, additional transmission-mediated (indirect) effects of the vaccine have the potential to make important contributions to population-level vaccine effects. However, the magnitude of indirect effects that are attributable to rotavirus vaccine and the mechanisms which underpin them are poorly understood.4
Rotavirus vaccination mimics immunity induced by natural rotavirus infections, which confer incremental protection against severe rotavirus disease to a degree that varies by location.5, 6, 7 Rotavirus disease severity has been shown to correlate with density of faecal rotavirus shedding in studies in India and Malawi,8 and evidence suggests that severity of symptoms is related to the risk of transmission.9 Therefore, vaccination, although not providing complete protection against disease, might reduce the severity of gastroenteritis and diminish viral shedding after exposure to natural rotavirus infection, leading to reduced infectiousness of an index child, a reduced secondary attack rate in exposed households, and reduced rotavirus transmission.
Research in context.
Evidence before this study
We searched PubMed using the terms “rotavirus” AND (“transmission” OR “household”), “rotavirus” AND “vaccin*” AND “effectiveness”, “rotavirus” AND “indirect” and “rotavirus” AND “herd”, for all published studies up to March 20, 2020, without language restrictions. Indirect effects of rotavirus vaccine are well documented in population-based studies from high-income settings. However, data from sub-Saharan Africa are inconsistent and limited to observational studies from a few countries (Kenya, Rwanda, Malawi, Zambia, and South Africa). Household studies have been used to better understand rotavirus transmission, but most studies have been done in high-income settings. A study from New Zealand described secondary attack rates of 46% for household contacts of a rotavirus-positive index case child, and a household study from the USA reported that 33% of children and 12% of adults exposed to an index case were infected with rotavirus. We identified two studies of rotavirus transmission in middle-income countries: a study from Ecuador used quantitative real-time PCR to identify rotavirus infection and described a household secondary attack rate of 55%, and a study from India enrolled symptomatic and asymptomatic children as index cases. This study used genotyping to confirm transmission and found a much lower secondary attack rate (0·54%). We found no studies of rotavirus transmission in low-income countries or in any country in sub-Saharan Africa.
Added value of this study
To our knowledge, this is the first study of household transmission of rotavirus in a low-income country, where living conditions and contact patterns differ fundamentally from those in higher-income environments. We identified very high levels of rotavirus transmission in this context and described a strong association between disease severity in an index case and risk of transmission to household contacts. Using these data, we applied novel methods to estimate a vaccine effectiveness of approximately 40% against rotavirus transmission. Rotavirus vaccination of infants is therefore expected to considerably reduce community transmission of rotavirus in low-income countries, in addition to the direct protection afforded to vaccinated children.
Implications of all the available evidence
Understanding the total population-level effect of rotavirus vaccination is crucial to realising the public health value of rotavirus vaccines in low-income countries, where rotavirus burden remains high and individual vaccine effectiveness is reduced compared with in high-income settings. Future assessments of vaccine effectiveness cost-effectiveness should consider the substantial effect of rotavirus vaccine on community-level transmission in low-income countries.
No published data exist describing household transmission of rotavirus in people in sub-Saharan Africa. Previous studies in high-income settings have shown a high secondary attack rate within families and have highlighted the role of infants in introducing rotavirus infection into households.10, 11 A household transmission study in Ecuador showed a secondary attack rate of 55% for asymptomatic infection,9 and a study from India documented low rates of household transmission (0·54%) from symptomatic and asymptomatic children with rotavirus infection.12 However, extrapolation of data on rotavirus transmission from high-income and middle-income countries to low-income countries is not appropriate because of fundamental differences in factors that might have a major influence on the risk of transmission, such as living environments, crowding, contact patterns, access to sanitation systems, host immunity, nutritional state, and frequency of exposure to rotavirus.4
We aimed to investigate household rotavirus transmission in a semi-urban setting in Blantyre, Malawi, a low-income country in southern Africa. The monovalent, G1P[8]-containing rotavirus vaccine (RV1) was introduced into Malawi's childhood immunisation programme in 2012. Our objectives were to (1) explore risk factors for transmission of rotavirus to household contacts, including symptom severity and density of faecal rotavirus shedding, and (2) estimate the effectiveness of rotavirus vaccine in preventing transmission of rotavirus infection to household contacts.
Methods
Study design and participants
We recruited participants for this prospective cohort study in four government health facilities in Blantyre, Malawi. Malawi is one of the poorest countries in the world, with 70% of the population falling below the international poverty line in 2016.13 Government-provided health care is free at the point of access. Health facilities in this study were Queen Elizabeth Central Hospital (QECH), the primary referral centre for Southern Malawi; and three health centres (Zingwangwa, Gateway, and Madziabango). Details of recruitment sites are included in the appendix (p 1)). Children born after Sept 17, 2012, and aged at least 6 weeks (vaccine-eligible children) presenting with acute gastroenteritis were screened for rotavirus using a point-of-care immunochromatographic rapid test (Rota-Strip, Coris BioConcept, Gembloux, Belgium). Children testing positive for rotavirus (case children) were recruited together with their household contacts, with written informed assent for children and consent for parents or guardians. Details of screening and recruitment are included in the appendix (p 2)). Recruitment at QECH was embedded within an existing rotavirus surveillance platform, designed to assess rotavirus vaccine effectiveness using a test-negative case-control methodology as previously described (appendix p 2).14
To estimate secondary attack rates, households were followed up prospectively for up to 14 days after symptom onset in the case child. Follow-up comprised active surveillance for clinical disease and stool sample collection from household contacts to detect rotavirus infection (case cohort). Control households were recruited to define the background prevalence of rotavirus infection in households in the community. Control households each contained an asymptomatic child, frequency-matched to case children on the basis of age, and were recruited from randomly generated global positioning system locations in Blantyre district. Controls were recruited from randomly generated locations rather than being individually location-matched to maximise efficiency of the study design. Control households were excluded if any household member had symptoms of gastroenteritis within 2 weeks before recruitment.
The study was approved by the University of Liverpool Research Ethics committee (000757), and the Malawi College of Medicine Research Ethics Committee (P.09/14/1623).
Procedures
Clinical data were collected at presentation by research nurses from case children, including anthropometric measurements and assessment of disease severity using the standardised 20-point Vesikari score.15 Demographic, risk factor, and symptom questionnaires were completed for household contacts and control households on recruitment. Receipt of rotavirus vaccine and HIV status were documented from government-issued handheld health passports.
Participants were defined as having HIV infection if they tested positive on an HIV rapid test (children aged ≥12 months), or HIV DNA PCR (infants aged <12 months).16 Severe acute malnutrition was defined using WHO criteria of a weight-for-height Z score up to 3 SDs from the median or mid-upper arm circumference of 11·5 cm or less.17
At presentation, case children with acute gastroenteritis had bulk stool collected for the measurement of rotavirus viral load and 1–2 mL of serum collected for anti-rotavirus IgA titres. Stool samples were collected from household contacts at days 5–7 and 10–12 after symptom onset in the case child. These timepoints were selected to maximise detection of secondary attack episodes, given published serial intervals (time between onset of disease in primary and secondary patients) for rotavirus disease of up to 11 days.18 Households were typically visited three times with symptom questionnaires done for all household members at each visit within the 14-day follow-up period. At the initial visit as soon after presentation as possible, pre-labelled sample containers and equipment for sample collection were left for each household member with instructions for use. Whenever possible, samples were collected on the same day that they were produced, or within 24 h. Samples were transported to the study laboratory in cool bags via study vehicles. In the event that samples were not obtained, data on clinical symptoms in household members were still collected. Control household members had a single stool sample collected at the point of recruitment.
Viral RNA was extracted from 10% stool suspension using Qiagen Viral RNA Mini kits (Qiagen, Hilden, Germany). Internal controls (Primerdesign RNA internal extraction control kit, Primerdesign, Southampton, UK) were added to each sample for quality control purposes and tested with a separate quantitative real-time PCR (rt-qPCR). Viral RNA was converted to cDNA using random primers (Invitrogen, ThermoFisher, Carlsbad, CA, USA).
Rotavirus was detected by use of a VP6 rt-qPCR.19 A standard curve, consisting of a linearised plasmid containing the gene that encodes VP6 from a human group A rotavirus of VP6 genotype 1, was included in each run to allow estimation of rotavirus viral load (copy numbers). Samples with cycle threshold (Ct) values greater than 35 and less than 40 on VP6 PCR underwent confirmatory testing with a second rt-qPCR assay targeting the NSP3 gene.20 Because of poor reproducibility in samples with very low viral loads, a rotavirus-positive sample was defined as containing a viral copy number of 100 or more derived from the VP6 assay and also testing positive on the NSP3 assay. All samples from case children that were positive for rotavirus antigens and rotavirus rt-qPCR-positive samples from household members with a Ct value of 35 or less underwent G and P typing using a two-stage PCR with consensus and type-specific primers using standard methods.21 Genotyping was done on cDNA extracted from faecal samples and synthesised using random primers.
Anti-rotavirus IgA geometric mean titres were measured using a semi-quantitative sandwich ELISA22 and were calculated using a minimum of two values per sample with a coefficient of variation of less than 20%. Results were defined as zero if values were below the lower limit of detection. Rotavirus infection in household contacts was defined as a rotavirus-positive stool sample, with or without symptoms of gastroenteritis, and clinical disease in household contacts was defined as any reported vomiting or diarrhoea during the follow-up period.
Statistical analysis
Distributions of continuous variables were examined and categorical variables were tabulated to generate descriptive statistics. Missing observations were assumed to be missing completely at random. According to a predetermined analysis plan, two-sided t tests were used to compare independent means of normally distributed data, and rank sum tests were used to compare non-normally distributed data. χ2 or Fisher's exact tests (depending on number of observations) were used to compare categorical variables.
Risk factors for rotavirus transmission within case households were identified using logistic regression models with a random effect to account for household-level clustering. All individuals residing in case households were included in the analysis; however, the final regression models were run using observations with complete data for included variables. A conceptual framework (appendix p 7) was developed to account for the hierarchical relationship between predictive variables.23 Variables were divided into two initial groups: those relating to the infectiousness of the symptomatic case child and those relating to the susceptibility of household contacts. Susceptibility variables were further divided into proximal susceptibility (individual level), and distal susceptibility (household level) variables. Individual models were built for each group to identify risk factors of importance while adjusting for potential confounding. A final model was then built incorporating all three groups, beginning with distal susceptibility variables then adding proximal susceptibility variables and infectiousness variables. Separate models were built for infection and disease. All variables with a p value of less than 0·1 on univariable analysis were tested for inclusion in the final models. Nested models were compared using likelihood ratio tests. Variables were retained in the model if the p value was less than 0·1 at any stage of the procedures described.
This study was powered to detect a 25% reduction in secondary attack rates in households exposed to an index case with mild or moderate rotavirus disease compared with households exposed to severe rotavirus disease (Vesikari score ≥11), with 80% power and a significance level of 5%. Further details can be found in the appendix (p 6)).
Estimating vaccine effectiveness against transmission
We hypothesised that by reducing disease severity in symptomatic children, rotavirus vaccine has the potential to reduce transmission to close contacts. Given the high vaccine coverage in Blantyre, we could not directly investigate the effect of vaccine status of the index case child on transmission. Vaccine effectiveness against transmission (VET) of rotavirus infection was therefore calculated using vaccine effectiveness estimates from the associated diarrhoeal surveillance platform to infer the distribution of disease severity in an unvaccinated population, combined with primary data on the relationship between disease severity and transmission from the household transmission study (vaccinated population).
First, children were divided into three severity groups: very severe disease (Vesikari score ≥15), less severe disease (Vesikari score <15), and asymptomatic infection. Secondary attack rate for infection among household contacts of index cases with very severe and less severe disease and of asymptomatic children was estimated using data from the household transmission study. The prevalence of asymptomatic infection in household members of control households was used as an estimate of secondary attack rate for asymptomatic children, accepting that this value is likely to be an overestimate. Receiver operating characteristic analysis with Youden's index24 was used to verify that a cutoff of a severity score of 15 had good discriminatory power in differentiating risk of transmission (appendix p 8).
To estimate the effect of vaccination on transmission, we assumed that receipt of rotavirus vaccination would result in children with very severe disease in a counterfactual unvaccinated population moving into the less severe category and children from the less severe category moving into an asymptomatically infected category at a proportion determined by the respective vaccine effectiveness estimates (figure 1). Vaccine effectiveness against very severe (VES) and less severe rotavirus disease (VEM) were calculated as 1 minus the odds ratio (OR) of two versus no doses of vaccine in vaccine-eligible children presenting with acute gastroenteritis, as previously described.25 ORs were estimated using logistic regression models fit to data from the rotavirus vaccine surveillance platform in Blantyre from Oct 29, 2012, to June 30, 2016, consistent with the most recently published vaccine effectiveness data.25 Under idealised test-negative case-control designs, the OR provides an unbiased estimate of the relative risk.26
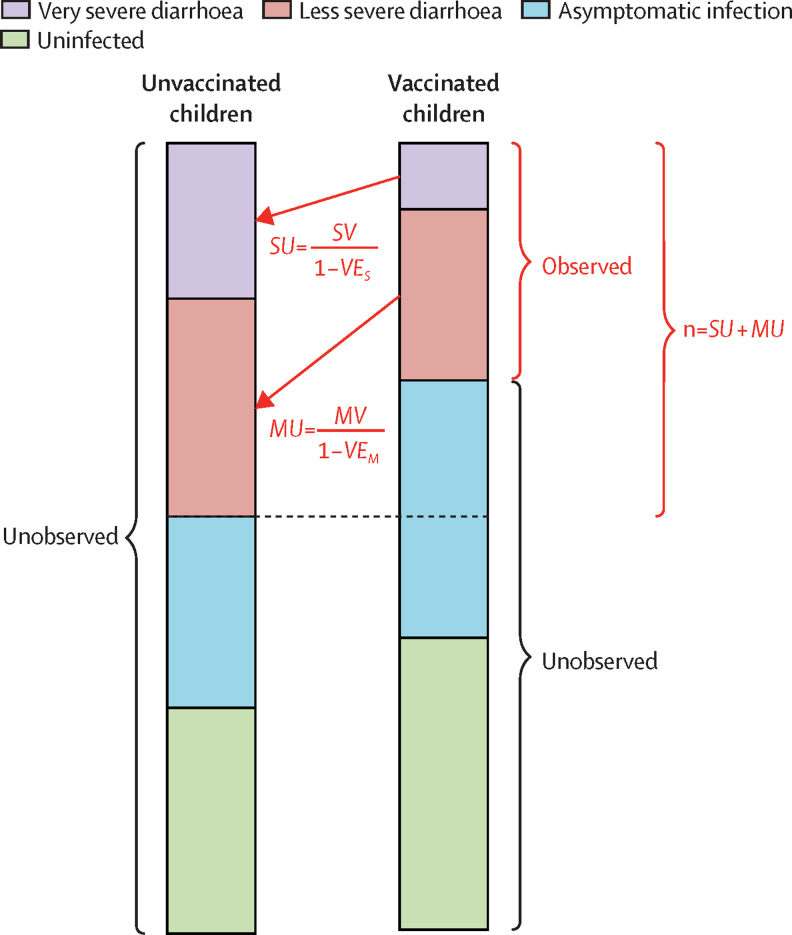
Figure 1.
Conceptual diagram of the effect of vaccination on the proportion of children with rotavirus infection of different degrees of disease severity
We found cases of very severe (SV) and less severe (MV) rotavirus diarrhoea among vaccinated children. We used the estimated vaccine effectiveness against very severe (VES) and less severe rotavirus diarrhoea (VEM) to infer the number of very severe (SU) and less severe (MU) rotavirus cases in an unvaccinated population. The dashed line represents the size of the potentially observable population (n). We explored the sensitivity of our results to different assumptions about the size of the asymptomatic population in the appendix (p 4)).
The number of vaccinated children with very severe (SV) and less severe (MV) disease was observed in the household transmission study. We then estimated the number of children with very severe rotavirus disease (SU) and less severe rotavirus disease (MU) in a hypothetical (counterfactual) unvaccinated population (figure 1). The total number of children with very severe or less severe rotavirus disease in an unvaccinated population was estimated as:
Vaccine effectiveness against transmission (indicated by VET) was estimated as 1 minus the ratio of the secondary attack rates (SARs) in vaccinated and counterfactual unvaccinated populations using the following equation:
SARx is the household secondary attack rate of infection for a household with an index child with infection severity x, where x can be either very severe (S), less severe (M), or asymptomatic (A); and Pxy indicates the proportion of index children with rotavirus infection of severity x and vaccination status y, where y can be either vaccinated (V) or unvaccinated (U). Proportions were estimated as PSV = SV/n, PMV = MV/n, PAV = (n-SV-MV)/n), PSU = SU/n, PMU = MU/n, where SV and MV are observed and SU, MU, and n are calculated as previously described (figure 1).
Bootstrapped 95% CIs for VET were generated by sampling 10 000 times from the distributions of the corresponding parameters. Bootstrap samples of vaccine effectiveness (1–OR) for very severe and less severe disease were generated by sampling from log-normal distributions using the mean and SD derived from logistic regression models. Our estimates assume that the proportion of children with asymptomatic infection in an unvaccinated population (PAU) is similar to or lower than that in a vaccinated population. We did sensitivity analyses to assess the effect of different values of PAU and SARA on estimates of VET (appendix p 4).
Role of the funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. AB had full access to all the data in the study and had final responsibility for the decision to submit for publication
Results
Between Feb 16, 2015, and Nov 11, 2016, we recruited 196 case households containing a symptomatic child who had tested positive for rotavirus, with 705 household members, and 55 control households containing an asymptomatic child who was frequency age-matched, with 153 household members. The median age of case and control children was 11·5 months (table 1). Rotavirus vaccine coverage was high (≥99·0% among case and control children). 89 (45%) of 196 case households had electricity at home, 68 (35%) of 195 case households (one household had missing data for this variable) did not have any member with a regular salary, and 60 (30%) of 196 case households reported problems obtaining food in the past month (appendix p 10). Anti-rotavirus IgA titres in case children at presentation were low (median 4 IU/ml [IQR 0–30]). Viral loads in case children were high, with a median Ct value of 19·1 (IQR 17·2 to 22·2), corresponding to a median viral copy number of 1·67 × 107 (1·63 × 106 to 6·37 × 107). Viral loads in household contacts were markedly lower; median Ct value 34·8 (31·8 to 36·6), corresponding to a median copy number of 712 (256 to 3704). Viral loads in control children and their household contacts were also low (median Ct value 37·9 [35·7–38·6], corresponding to a median copy number of 77 [34–211] for children; and median Ct value 37·7 [36·5–38·8], corresponding to a median copy number of 113 [57–297] for household contacts).
Table 1.
Characteristics of case and control children
| Case children (n=196) | Controls (n=55) | p value* | |||
|---|---|---|---|---|---|
| Age, months | 11·5 (8·8–15·2) | 11·5 (8·2–15·4) | 0·75† | ||
| Completed rotavirus vaccination | |||||
| Vaccinated (two doses) | 194 (99%) | 55 (100%) | 1·0 | ||
| Unvaccinated (no dose) | 2 (1%) | 0 (0%) | 1·0 | ||
| Sex | |||||
| Male | 108 (55%) | 26 (47%) | 0·30 | ||
| Female | 88 (45%) | 39 (53%) | .. | ||
| Diarrhoea | 196 (100%) | 0 (0%) | <0·0001 | ||
| Frequency, maximum per day | |||||
| 1–3 | 173 (88%) | .. | .. | ||
| 5 | 12 (6%) | .. | .. | ||
| ≥6 | 11 (6%) | .. | .. | ||
| Episodes‡ | |||||
| 1–4 | 24 (12%) | .. | .. | ||
| 5 | 86 (44%) | .. | .. | ||
| ≥6 | 86 (44%) | .. | .. | ||
| Vomiting | 182 (93%) | 0 (0%) | <0·0001 | ||
| Duration, days | |||||
| 1 | 23/182 (13%) | .. | .. | ||
| 2 | 59/182 (32%) | .. | .. | ||
| ≥3 | 100/182 (55%) | .. | .. | ||
| Frequency, maximum per day | |||||
| <5 | 123/182 (68%) | .. | .. | ||
| ≥5 | 59/182 (32%) | .. | .. | ||
| HIV | |||||
| Exposed | 25/196 (13%) | 6/54 (11·1%) | 0·75 | ||
| Infected | 2/89 (2%) | 0/11 (0%) | 1·0 | ||
| Admitted to hospital | 111 (57%) | .. | .. | ||
| Vesikari score | 14 (12–16) | .. | .. | ||
| Rectal temperature,°C | |||||
| 37·1–38·4 | 92/193 (48%) | .. | .. | ||
| 38·5–38·9 | 48/193 (25%) | .. | .. | ||
| ≥39·0 | 53/193 (28%) | .. | .. | ||
| Thirst | |||||
| No thirst | 32 (16%) | .. | .. | ||
| Thirsty | 141 (72%) | .. | .. | ||
| Drinks poorly | 23 (12%) | .. | .. | ||
| Skin pinch | |||||
| Normal | 56 (29%) | .. | .. | ||
| Goes back slowly | 104 (53%) | .. | .. | ||
| Goes back very slowly | 36 (18%) | .. | .. | ||
| General appearance | |||||
| Well and alert | 94 (48%) | .. | .. | ||
| Restless | 83 (42%) | .. | .. | ||
| Unconscious | 19 (10%) | .. | .. | ||
| Dehydration | |||||
| None | 26 (13%) | .. | .. | ||
| Some (5%) | 124 (63%) | .. | .. | ||
| Severe (10%) | 46 (23%) | .. | .. | ||
| Intravenous fluid use | 58 (30%) | .. | .. | ||
| Oral fluid use | 185 (94%) | .. | .. | ||
| Outcome | |||||
| Discharged | 194 (99%) | .. | .. | ||
| Died | 2 (1%) | .. | .. | ||
| Anthropometry | |||||
| Adjusted WHZ | −0·59 (1·61) | .. | .. | ||
| Adjusted WAZ | −0·46 (1·16) | .. | .. | ||
| Adjusted HAZ | −0·04 (2·46) | .. | .. | ||
| MUAC | 13·48 (1·28) | .. | .. | ||
| SAM | 23/194 (12%) | .. | .. | ||
| Previous diarrhoeal admission | 15 (8%) | 3 (6%) | 0·77 | ||
| Previous diarrhoeal presentation | 91 (46%) | 26 (47%) | 0·91 | ||
| Premature birth | 7 (4%) | 2/53 (4%) | 0·69 | ||
| Birthweight, kg¶ | 2·96 (0·63) | 2·95 (0·55) | 0·89§ | ||
| Ever breastfed | 195 (99%) | 53 (96%) | 0·12 | ||
| Diet includes food other than breastmilk | 190 (97%) | 48 (87%) | 0·0043 | ||
| IgA titres at presentation, GMC | 4 (0–30) | .. | .. | ||
Data are median (IQR), n/N (%), or mean (SD), unless otherwise indicated. Denominators that do not total 196 (cases) or 55 (controls) result from missing data for individual variables, unless otherwise indicated. WHZ=weight-for-height Z score. WAZ=weight-for-age Z score. HAZ=height-for-age Z score. MUAC=mid-upper-arm circumference. SAM=severe acute malnutrition. GMC=geometric mean concentration.
χ2 p values for differences in proportions between case children and control children, unless otherwise specified.
Rank-sum test.
Maximum frequency per day.
Two-sided independent t test.
Premature birth was defined as being born more than 1 month early.
At least one faecal sample was collected from 665 (94%) of 705 individuals from 188 (96%) of 196 case households, with a total of 1212 samples collected. For control households a faecal sample was collected from 144 (94%) of 153 individuals from 54 (98%) of 55 control households. Eight households from the case cohort and one from the community controls either refused consent for faecal sample collection or were unable to provide samples. The secondary attack rate for infection among household contacts of case children was high, with 434 (65%) of 665 individuals positive for rotavirus (table 2). Clinical disease was much less common, with 48 (7%) of 699 household contacts reporting symptoms of gastroenteritis. Of these, 47 (98%) had samples available for testing and 37 (77%) were positive for rotavirus, resulting in a secondary attack rate for clinical rotavirus disease of 5% (table 2). Six individuals had unknown diarrhoea history for at least one visit and were excluded from this analysis. Rates of clinical disease were significantly higher among children younger than 5 years compared with those from older age groups (table 2). The prevalence of rotavirus infection in the control households was 28% (40 of 144 household contacts), which was significantly lower than in case households (p<0·0001; table 2). The secondary attack rate for infection and disease were higher in households where index children had very severe disease compared with less severe disease (appendix p 12).
Table 2.
Secondary attack rates for rotavirus infection and clinical disease
|
Infection |
Clinical rotavirus disease |
||||||
|---|---|---|---|---|---|---|---|
| Case households | Control households | p value | Case households | Control households | p value | ||
| Overall | 434/665 (65%) | 40/144 (28%) | <0·0001* | 37/698 (5%) | 0/153 (0%) | 0·0036* | |
| Age, years | |||||||
| <5 | 57/88 (65%) | 2/10 (20%) | 0·0061* | 12/91 (13%) | 0/11 (0%) | 0·20* | |
| 5–<15 | 127/193 (66%) | 14/48 (29%) | <0·0001* | 4/197 (2%) | 0/53 (0%) | 0·30* | |
| 15–45 | 240/367 (65%) | 23/84 (27%) | <0·0001* | 20/390 (5%) | 0/87 (0%) | 0·031* | |
| >45 | 9/16 (56%) | 1/2 (50%) | 0·87* | 1/18 (6%) | 0/2 (0%) | 0·73* | |
| χ2 p value† | 0·93 | 0·57 | .. | <0·0015 | NA | .. | |
One value for age was missing in household members with rotavirus infection and two values were missing for age in household members with rotavirus disease. NA=not applicable.
χ2 p value comparing difference in proportion between case children and controls.
χ2 p value for difference in proportion between children younger than 5 years and those aged 5 years and older.
195 of 196 case children with remaining faecal samples were successfully genotyped. Of these, 60 (31%) were genotype G2P[4], 48 (25%) were G1P[8], 28 (14%) were G2[P6], and 14 (8%) were G12P[6] (appendix p 9). 297 rotavirus samples from samples obtained from household contacts of case children had a Ct value of 35 or less and were genotyped; in 94 (32%) of these samples, both G and P types were the same as those identified in the case child (appendix p 12). Only 15 rotavirus-positive samples from community controls had viral loads sufficiently high to enable genotyping, prohibiting meaningful comparison with the distribution of genotypes from case households. These data are shown in the appendix (p 12)).
Increasing disease severity (per Vesikari score unit) in case children was strongly associated with transmission of infection to household contacts (OR 1·17 [95% CI 1·06–1·30]; table 3). Mid-upper arm circumference in a case child was also positively associated with risk of transmission of infection (table 3). At the household level (distal susceptibility factors), having at least one household member with a regular salary was associated with reduced susceptibility to infection (table 3). Conversely, difficulty obtaining sufficient food for the household in the past month was also associated with reduced susceptibility to infection. At the individual level (proximal susceptibility factors), we found strong evidence that the proximity of relationship with a case child was associated with risk of transmission of infection, with mothers significantly more likely to become infected with rotavirus than other adult relatives or child household contacts (table 3). Univariate analysis of infectiousness risk factors identified several clinical features associated with risk of transmission of infection, including vomiting and need for admission to hospital; however, none of these findings were significant when Vesikari score was included in the multivariable model. Details of the univariable analysis can be found in the appendix (p 13)).
Table 3.
Risk factors for transmission of rotavirus infection
| Odds ratio (95% CI) | p value | ||
|---|---|---|---|
| Infectiousness risk factors | |||
| Vesikari score in case child | 1·17 (1·06–1·30) | 0·0029 | |
| MUAC in case child | 1·32 (1·06–1·66) | 0·015 | |
| Distal susceptibility factors | |||
| Number of adults with salary in household | |||
| None | Ref | .. | |
| ≥1 | 0·44 (0·24–0·80) | 0·0069 | |
| Problems getting food in the past month (%) | |||
| No | Ref | .. | |
| Sometimes or often | 0·58 (0·31–1·06) | 0·077 | |
| Proximal susceptibility factors | |||
| Relationship with child | |||
| Mother | Ref | .. | |
| Other adult relative | 0·29 (0·16–0·50) | <0·0001 | |
| Child contact | 0·44 (0·26–0·74) | 0·0020 | |
n=645 observations with complete data included in the model. Odds ratios for Vesikari score and MUAC are per variable unit. MUAC=mid-upper-arm circumference.
For transmission of clinical disease, the primary infectiousness risk factor was disease severity in the case child, with a positive association between increasing disease severity and risk of clinical rotavirus disease in household contacts (OR 1·28 [95% CI 1·08–1·52]; table 4). We found a weak association between genotype of rotavirus in the case child and risk of disease transmission, with an increased risk of transmission with G1P[8] genotype compared with G2P[4], G2P[6], and G12P[6]. At the household level (distal susceptibility factors), use of a pit or water toilet was associated with reduced odds of rotavirus disease compared with having no toilet (appendix p 17), although this association was no longer significant in the final model (table 4). At the individual level (proximal susceptibility factors), age of the household contact was significantly associated with risk of disease; children younger than 5 years had the greatest risk of disease, compared with children aged 5–15 years and older adults (table 4).
Table 4.
Risk factors for transmission of rotavirus disease
| Odds ratio (95% CI) | p value | ||
|---|---|---|---|
| Infectiousness risk factors | |||
| Vesikari score in index child | 1·28 (1·08–1·52) | 0·0048 | |
| Rotavirus genotype in index child | |||
| G1P[8] | Ref | .. | |
| G2P[4] | 0·36 (0·12–1·03) | 0·059 | |
| G2P[6] | 0·43 (0·11–1·64) | 0·22 | |
| G12P[6] | 0·82 (0·23–2·95) | 0·76 | |
| Other | 1·10 (0·44–2·71) | 0·84 | |
| Distal susceptibility factors | |||
| Toilet type | |||
| None | Ref | .. | |
| Simple pit or VIP | 0·56 (0·11–2·83) | 0·48 | |
| Water toilet | 0·42 (0·03–5·96) | 0·52 | |
| Proximal susceptibility factors | |||
| Household member age, years | |||
| <5 | Ref | .. | |
| 5–<15 | 0·13 (0·04–0·46) | 0·0014 | |
| 15–45 | 0·38 (0·17–0·92) | 0·031 | |
| >45 | 0·35 (0·04–3·22) | 0·35 | |
n=681 observations with complete data included in the model. Odds ratio for Vesikari score is per variable unit. VIP=ventilated improved pit latrine.
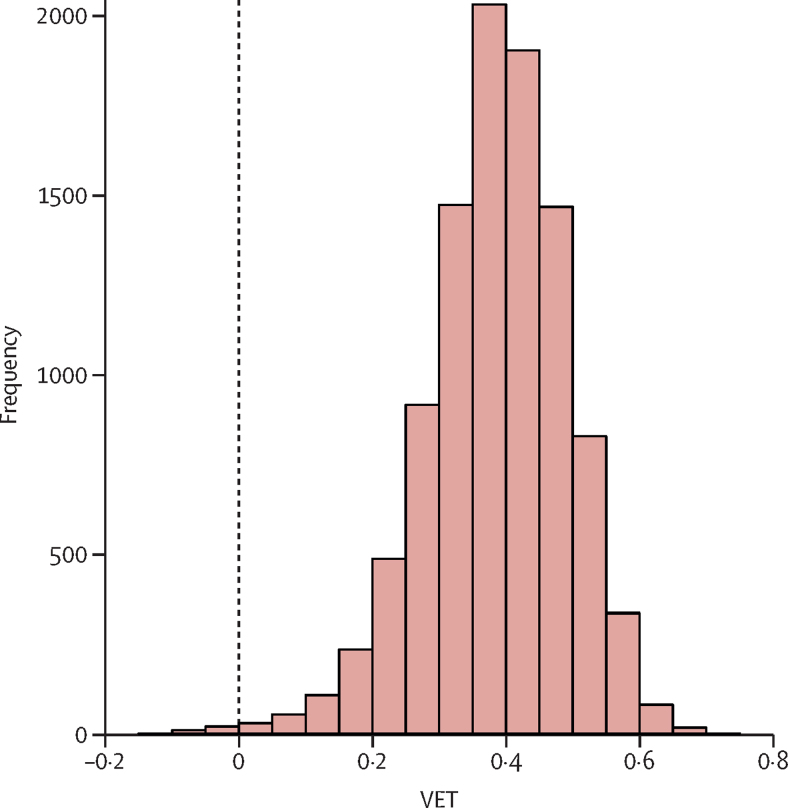
Vaccine effectiveness was estimated to be 69% (95% CI –10 to 91) against very severe disease and 56% (3 to 79) against less severe rotavirus disease on the basis of the diarrhoeal surveillance dataset.25 As a result, we estimated that the proportion of unvaccinated children with very severe disease would be 0·44 (0·15 to 0·78) and the proportion of unvaccinated children with less severe disease would be 0·55 (0·22 to 0·85). By comparison, we estimated the proportion of vaccinated children with very severe disease to be 0·13 (0·06 to 0·27) and less severe disease to be 0·23 (0·11 to 0·47). We also estimated the proportion of vaccinated children who became asymptomatic to be 0·63 (0·27 to 0·83). We found the secondary attack rate to be 72% (64 to 79) for severe disease, 64% (57 to 69) for less severe disease, and 25% (16 to 35) for asymptomatic infection. Combining this information in equation (2), we estimated a VET of 39% (16 to 57; figure 2). Sensitivity analysis showed that VET estimates were sensitive to the proportion of asymptomatic individuals in an unvaccinated population, with VET decreasing as the size of the asymptomatic unvaccinated population increased, but VET estimates remained significantly greater than zero in all scenarios (appendix p 4).
Figure 2.
Distribution of estimates for vaccine effectiveness against transmission
The histogram of 10 000 bootstrap samples of the estimated vaccine effectiveness against transmission is plotted. VET=vaccine effectiveness against transmission.
Discussion
In Malawi, high secondary attack rates for rotavirus infection were observed in households following contact with a child with rotavirus gastroenteritis, despite high rotavirus vaccine coverage. However, the secondary attack rate for rotavirus disease was much lower. Disease severity in the case child was an important predictor of transmission of infection and disease to household contacts.
Most cases in children in our study represent failures of the rotavirus vaccine; high viral shedding density and low anti-rotavirus IgA titres at the time of presentation probably explained the high secondary attack rate. Our estimate of the secondary attack rate for rotavirus infection is consistent with findings from studies from New Zealand and Ecuador, which reported secondary attack rates for rotavirus infection of 48% and 55%, respectively.9, 10 By contrast, we observed much lower attack rates for clinical disease, compared with 15% reported for Ecuador and 38% for New Zealand. This difference could be explained by high population levels of rotavirus transmission in Malawi resulting in frequent boosting of immunity against clinical disease, particularly among older children and adults. Notably, in our study the clinical secondary attack rate was highest in children aged 0–4 years.27 We also observed a high frequency of rotavirus infection in control households. Although this finding is substantially higher than that observed in higher-income settings, such as the UK and Ecuador,9, 28 it is consistent with findings from published studies from sub-Saharan Africa29, 30 and is plausible given the high force of rotavirus infection in Malawi27 and high levels of poverty, crowding, and poor access to water and sanitation. The inconsistencies between rotavirus genotypes detected in case children and their household contacts might also reflect the high frequency of asymptomatic shedding identified in the community.9
Increasing disease severity was associated with increased odds of rotavirus transmission for infection and disease. By reducing disease severity, rotavirus vaccine has the potential to reduce the infectiousness of a symptomatic child even in the event of clinical vaccine failure. This effect has been described with other pathogens such as Bordetella pertussis, but not yet with rotavirus.31 In this study, we estimated that in a semi-urban population in Malawi, with high rates of rotavirus transmission, rotavirus vaccine substantially reduces household rotavirus transmission, which could have a considerable effect on the burden of rotavirus disease in the community, and is consistent with previous estimates of indirect effectiveness of rotavirus vaccination from Malawi from hospital-based studies and mathematical models.25, 27 By contrast, horizontal transmission of vaccine-derived virus is unlikely to make a meaningful contribution to indirect effects in this setting because such transmission within households is rare.32
No regular income in the household increased the risk of transmission of infection. This finding probably reflects relative poverty compared with other study participants, which could increase rotavirus transmission for reasons including crowding, sanitation, carer education levels, or other unmeasured factors.33 Close contact and proximity of relationship to the index child also increased the risk of transmission of infection, emphasising that improved personal and hand hygiene and behavioural measures might be important for preventing rotavirus transmission within households.34 Rotavirus genotype G1P[8] was weakly associated with an increased risk of transmission, consistent with the global predominance of G1P[8] before the widespread introduction of vaccination.35
Our study has important limitations. The direction of infection cannot be defined with certainty using this study design because households were recruited only when an index child presented with rotavirus gastroenteritis; however, the pattern of transmission supports the notion of young infants bringing rotavirus into the household. Our analysis of the secondary attack rate by age did not fully account for the clustered nature of the data, and results should be interpreted with this in mind. Nevertheless, the findings are consistent with the regression analysis of risk factors for transmission, which did account for household clustering. We did not differentiate vaccine-type G1P[8] rotavirus strains from wild-type virus in this study, but we have previously shown that household transmission of rotavirus vaccine virus occurs infrequently in this population,32 and therefore vaccine-derived virus is unlikely to have contributed to rotavirus detection in stool. Our estimate of vaccine effectiveness against transmission assumed that asymptomatic infections contribute a small amount to the overall secondary attack rate, and that the prevalence of asymptomatic infection is similar or lower in an unvaccinated population. The prevalence of asymptomatic infections among unvaccinated infants and vaccine effectiveness against asymptomatic infection are unknown. Sensitivity analysis showed that vaccine effectiveness against transmission decreased as the size of the asymptomatic unvaccinated population increased, but remained significantly greater than zero in all scenarios tested. Vaccine effectiveness against transmission is dependent on locally specific parameters and our estimate might not be generalisable to other settings. Finally, in our population we could not directly measure the effect of vaccine status on household transmission of rotavirus because of high vaccine coverage. Our methods address this limitation by providing a quantitative estimate of the effect of vaccination on transmission, informed by primary transmission data from a vaccinated population and vaccine effectiveness estimates from the same recruitment platform.
Despite sustained high coverage of rotavirus vaccine in Malawi and other low-income African countries, the burden of rotavirus disease remains high. Indirect (herd) protection through vaccination should be considered together with direct vaccine effectiveness estimates in future clinical and health economic assessments of rotavirus vaccination, particularly in low-income, high -burden environments.
Acknowledgments
Acknowledgments
This study was supported by two Wellcome Trust Clinical PhD Fellowships (102466/Z/13/A to AB and 102464/Z/13/A to LP), a Wellcome Trust Programme Grant (091909/Z/10/Z), the Malawi-Liverpool Wellcome Trust Programme Core Grant Strategic Award (101113/Z/13/Z) and the US National Institutes of Health and National Institute of Allergy and Infectious Diseases (R01-AI112970 to VEP). KCJ is supported by an International Wellcome Trust Training Fellowship (201945/Z/16/Z). We thank all infants and their families who participated and all members of the RotaRITE study team. We are grateful for the support of the Malawi Ministry of Health and clinical staff at the recruitment sites. NAC is affiliated to the National Institute for Health Research (NIHR) Health Protection Research Unit in Gastrointestinal Infections at the University of Liverpool, Liverpool, UK, in partnership with Public Health England, in collaboration with the University of Warwick, Warwick, UK. NAC is based at the University of Liverpool. The views expressed are those of the author(s) and not necessarily those of the NIHR, the Department of Health and Social Care, or Public Health England.
Contributors
AB had overall responsibility for study design and execution; data analysis; and manuscript drafting, editing, and writing. LP and NB-Z contributed to study design, data collection, and manuscript editing and writing. KCJ contributed to laboratory data collection and supervision and manucript editing. BL contributed to study design and data analysis. JAL contributed to data analysis and manuscript editing and writing. MI-G contributed to study design, laboratory data collection and supervision, and manuscript editing. VEP contributed to study design, data analysis, and manuscript editing and writing. NAC contributed to study design, data collection, and manuscript editing and writing.
Declaration of interests
NB-Z and KCJ have received research grant support from GSK for work on rotavirus vaccines, outside of the submitted work. NB-Z also reports a grant from Takeda Pharmaceuticals, outside the submitted work. KCJ reports a grant from the Bill & Melinda Gates Foundation, outside the submitted work. BL reports personal fees for: membership of the norovirus vaccine advisory board in Takeda; travel and consultancy for the CDC Foundation; and expert testimony on infectious disease control for Hall Booth Smith, all outside of the submitted work. MI-G has received research grant support from GlaxoSmithKline Biologicals and Sanofi Pasteur/Merck Sharp & Dohme for work on rotavirus, and grants from Merck and Takeda Pharmaceuticals, all outside of the submitted work. VEP is a member of the WHO Immunization and Vaccine-related Implementation Research Advisory Committee and has received reimbursement from Merck for travel expenses to attend a Scientific Input Engagement unrelated to rotavirus vaccines. NAC has received research grant support and honoraria for participation in rotavirus vaccine advisory board meetings from GSK, outside of the submitted work. AB, LP, and JAL declare no competing interests.
Supplementary Material
References
- 1.Rotavirus vaccine support, Gavi, the Vaccine Alliance. http://www.gavi.org/support/nvs/rotavirus/
- 2.Madhi SA, Cunliffe NA, Steele D. Effect of human rotavirus vaccine on severe diarrhea in African infants. N Engl J Med. 2010;362:289–298. doi: 10.1056/NEJMoa0904797. [DOI] [PubMed] [Google Scholar]
- 3.Platts-Mills JA, Amour C, Gratz J. Impact of rotavirus vaccine introduction and postintroduction etiology of diarrhea requiring hospital admission in Haydom, Tanzania, a rural African setting. Clin Infect Dis. 2017;65:1144–1151. doi: 10.1093/cid/cix494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bennett A, Bar-Zeev N, Cunliffe NA. Measuring indirect effects of rotavirus vaccine in low income countries. Vaccine. 2016;34:4351–4353. doi: 10.1016/j.vaccine.2016.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gladstone BP, Ramani S, Mukhopadhya I. Protective effect of natural rotavirus infection in an Indian birth cohort. N Engl J Med. 2011;365:337–346. doi: 10.1056/NEJMoa1006261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fischer TK, Valentiner-Branth P, Steinsland H. Protective immunity after natural rotavirus infection: a community cohort study of newborn children in Guinea-Bissau, west Africa. J Infect Dis. 2002;186:593–597. doi: 10.1086/342294. [DOI] [PubMed] [Google Scholar]
- 7.Velázquez FR, Matson DO, Calva JJ. Rotavirus infection in infants as protection against subsequent infections. N Engl J Med. 1996;335:1022–1028. doi: 10.1056/NEJM199610033351404. [DOI] [PubMed] [Google Scholar]
- 8.Mukhopadhya I, Sarkar R, Menon VK. Rotavirus shedding in symptomatic and asymptomatic children using reverse transcription-quantitative PCR. J Med Virol. 2013;85:1661–1668. doi: 10.1002/jmv.23641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lopman B, Vicuña Y, Salazar F. Household transmission of rotavirus in a community with rotavirus vaccination in Quininde, Ecuador. PLoS One. 2013;8 doi: 10.1371/journal.pone.0067763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grimwood K, Abbott GD, Fergusson DM, Jennings LC, Allan JM. Spread of rotavirus within families: a community based study. Br Med J (Clin Res Ed) 1983;287:575–577. doi: 10.1136/bmj.287.6392.575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koopman JS, Monto AS, Longini IM., Jr The Tecumseh Study. XVI: family and community sources of rotavirus infection. Am J Epidemiol. 1989;130:760–768. doi: 10.1093/oxfordjournals.aje.a115397. [DOI] [PubMed] [Google Scholar]
- 12.Banerjee I, Primrose Gladstone B, Iturriza-Gomara M, Gray JJ, Brown DW, Kang G. Evidence of intrafamilial transmission of rotavirus in a birth cohort in South India. J Med Virol. 2008;80:1858–1863. doi: 10.1002/jmv.21263. [DOI] [PubMed] [Google Scholar]
- 13.Poverty & Equity Data Portal. The World Bank. http://povertydata.worldbank.org/poverty/country/MWI
- 14.Bar-Zeev N, Kapanda L, Tate JE. Impact and effectiveness of monovalent rotavirus vaccine following programmatic roll-out among infants in Malawi. Lancet Infect Dis. 2015;15:422–428. doi: 10.1016/S1473-3099(14)71060-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ruuska T, Vesikari T. Rotavirus disease in Finnish children: use of numerical scores for clinical severity of diarrhoeal episodes. Scand J Infect Dis. 1990;22:259–267. doi: 10.3109/00365549009027046. [DOI] [PubMed] [Google Scholar]
- 16.Malawi Ministry of Health Clinical management of HIV in children and adults. 2016. https://www.childrenandaids.org/sites/default/files/2017-04/Malawi_Clinical-HIV-Guidelines_2016.pdf
- 17.WHO. UN Children's Fund . World Health Organisation; Geneva: 2009. WHO child growth standards and the identification of severe acute malnutrition in infants and children. [PubMed] [Google Scholar]
- 18.Rodriguez WJ, Kim HW, Brandt CD. Longitudinal study of rotavirus infection and gastroenteritis in families served by a pediatric medical practice: clinical and epidemiologic observations. Pediatr Infect Dis J. 1987;6:170–176. doi: 10.1097/00006454-198702000-00006. [DOI] [PubMed] [Google Scholar]
- 19.Kang G, Iturriza-Gomara M, Wheeler JG. Quantitation of group A rotavirus by real-time reverse-transcription-polymerase chain reaction: correlation with clinical severity in children in South India. J Med Virol. 2004;73:118–122. doi: 10.1002/jmv.20053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Freeman MM, Kerin T, Hull J, McCaustland K, Gentsch J. Enhancement of detection and quantification of rotavirus in stool using a modified real-time RT-PCR assay. J Med Virol. 2008;80:1489–1496. doi: 10.1002/jmv.21228. [DOI] [PubMed] [Google Scholar]
- 21.Iturriza-Gómara M, Dallman T, Bányai K. Rotavirus genotypes co-circulating in Europe between 2006 and 2009 as determined by EuroRotaNet, a pan-European collaborative strain surveillance network. Epidemiol Infect. 2011;139:895–909. doi: 10.1017/S0950268810001810. [DOI] [PubMed] [Google Scholar]
- 22.Bernstein DI, Smith VE, Sherwood JR. Safety and immunogenicity of live, attenuated human rotavirus vaccine 89-12. Vaccine. 1998;16:381–387. doi: 10.1016/s0264-410x(97)00210-7. [DOI] [PubMed] [Google Scholar]
- 23.Victora CG, Huttly SR, Fuchs SC, Olinto MT. The role of conceptual frameworks in epidemiological analysis: a hierarchical approach. Int J Epidemiol. 1997;26:224–227. doi: 10.1093/ije/26.1.224. [DOI] [PubMed] [Google Scholar]
- 24.Fluss R, Faraggi D, Reiser B. Estimation of the Youden Index and its associated cutoff point. Biom J. 2005;47:458–472. doi: 10.1002/bimj.200410135. [DOI] [PubMed] [Google Scholar]
- 25.Bennett A, Pollock L, Jere KC. Direct and possible indirect effects of vaccination on rotavirus hospitalisations among children in Malawi four years after programmatic introduction. Vaccine. 2018;36:7142–7148. doi: 10.1016/j.vaccine.2018.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Foppa IM, Haber M, Ferdinands JM, Shay DK. The case test-negative design for studies of the effectiveness of influenza vaccine. Vaccine. 2013;31:3104–3109. doi: 10.1016/j.vaccine.2013.04.026. [DOI] [PubMed] [Google Scholar]
- 27.Pitzer VE, Bennett A, Bar-Zeev N. Evaluating strategies to improve rotavirus vaccine impact during the second year of life in Malawi. Sci Transl Medi. 2019;11 doi: 10.1126/scitranslmed.aav6419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Phillips G, Lopman B, Rodrigues LC, Tam CC. Asymptomatic rotavirus infections in England: prevalence, characteristics, and risk factors. Am J Epidemiol. 2010;171:1023–1030. doi: 10.1093/aje/kwq050. [DOI] [PubMed] [Google Scholar]
- 29.Ouédraogo N, Kaplon J, Bonkoungou IJO. Prevalence and genetic diversity of enteric viruses in children with diarrhea in Ouagadougou, Burkina Faso. PLoS One. 2016;11 doi: 10.1371/journal.pone.0153652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bennett A, Bar-Zeev N, Jere KC. Determination of a viral load threshold to distinguish symptomatic versus asymptomatic rotavirus infection in a high-disease-burden African population. J Clin Microbiol. 2015;53:1951–1954. doi: 10.1128/JCM.00875-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Préziosi MP, Halloran ME. Effects of pertussis vaccination on transmission: vaccine efficacy for infectiousness. Vaccine. 2003;21:1853–1861. doi: 10.1016/s0264-410x(03)00007-0. [DOI] [PubMed] [Google Scholar]
- 32.Bennett A, Pollock L, Jere KC. Infrequent transmission of monovalent human rotavirus vaccine virus to household contacts of vaccinated infants in Malawi. J Infect Dis. 2019;219:1730–1734. doi: 10.1093/infdis/jiz002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Knee J, Sumner T, Adriano Z. Risk factors for childhood enteric infection in urban Maputo, Mozambique: a cross-sectional study. PLoS Negl Trop Dis. 2018;12 doi: 10.1371/journal.pntd.0006956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zerr DM, Allpress AL, Heath J, Bornemann R, Bennett E. Decreasing hospital-associated rotavirus infection: a multidisciplinary hand hygiene campaign in a children's hospital. Pediatr Infect Dis J. 2005;24:397–403. doi: 10.1097/01.inf.0000160944.14878.2b. [DOI] [PubMed] [Google Scholar]
- 35.Bányai K, László B, Duque J. Systematic review of regional and temporal trends in global rotavirus strain diversity in the pre rotavirus vaccine era: Insights for understanding the impact of rotavirus vaccination programs. Vaccine. 2012;30(suppl 1):A122–A130. doi: 10.1016/j.vaccine.2011.09.111. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.