Abstract
Although multifunctional inorganic nanoparticles have been extensively explored for effective cancer diagnosis and therapy, their clinical translation has been greatly impeded because of significant uptake in the reticuloendothelial system and concerns about potential toxicity. In this study, we uncovered the thermosensitive biodegradability of CuS nanoparticles, which have classically been considered as stable in bulk state. Polyethylene glycol (PEG)-coated CuS nanoparticles (CuS-PEG) were well preserved at 4 ºC but were rapidly degraded at 37 ºC within 1 week in both in vitro and in vivo tests. Furthermore, real-time multispectral optoacoustic tomography, which is more convenient and accurate than traditional ex vivo analysis, was successfully employed to noninvasively demonstrate the biodegradability of CuS-PEG nanoparticles and dynamically monitor their tumor imaging capacity. The temperature-dependent controllable degradation profile and excellent tumor retention of CuS-PEG nanoparticles endows them with great potential for clinical applications since it ensures that the nanoparticles remain intact during production, transportation, and storage but degrade and clear from the body at physiological temperature after accomplishing sufficient diagnosis and therapeutic operations.
Keywords: copper sulfide, nanoparticles, biodegradable, thermosensitive, multispectral optoacoustic tomography
Graphical Abstract
INTRODUCTION
Nanoparticles have been extensively explored and applied to effective cancer diagnosis and therapy,1, 2 inspired by the concept of “magic bullet” that selectively attacks pathogens and diseased tissue but leaves healthy cells untouched.3–5 Multifunctional inorganic nanoparticles based on gold,6, 7 silicon,8–11 carbon,12, 13 and metal oxide and sulfide14, 15 are some of the most promising candidates due to their unique physical, chemical, and biological properties. However, most inorganic nanoparticles are not degradable and are retained by the reticuloendothelial system (RES), especially the liver, for months to years, or are slowly excreted into bile and feces through the hepatic route over several weeks to months.16–18 The retained inorganic nanoparticles may trigger oxidative stress and inflammation in the RES,19–21 leading to concerns about potential long-term toxicity,22 which significantly impedes the clinical translation of multifunctional inorganic nanoparticles regardless of their tremendous promise and investment in preclinical studies.
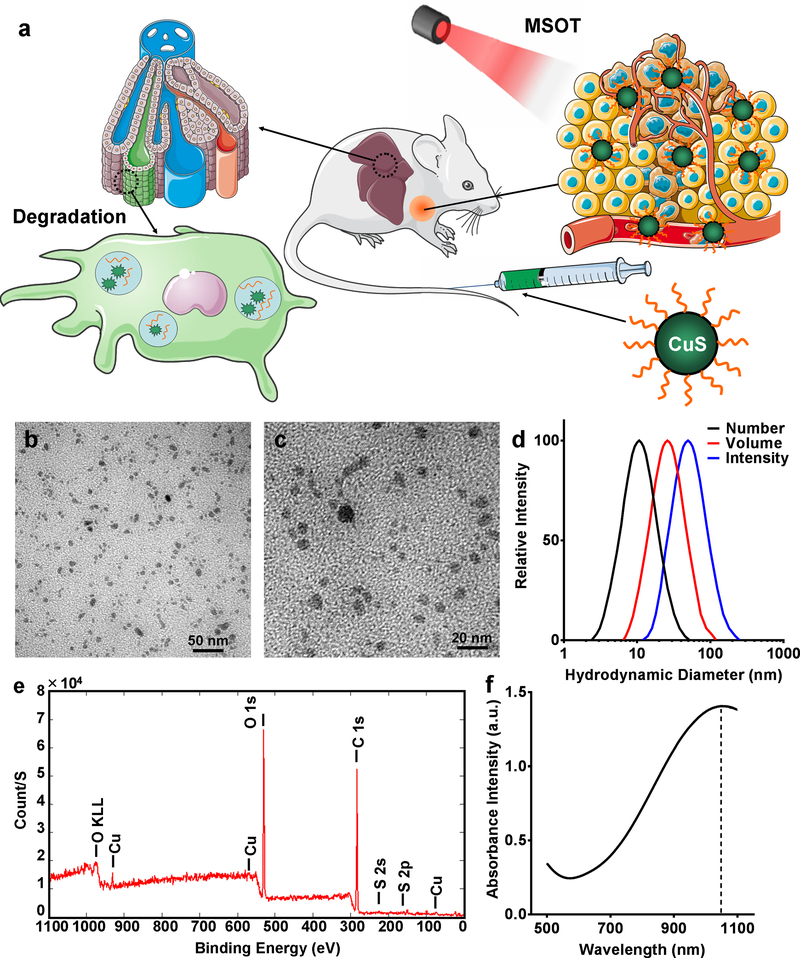
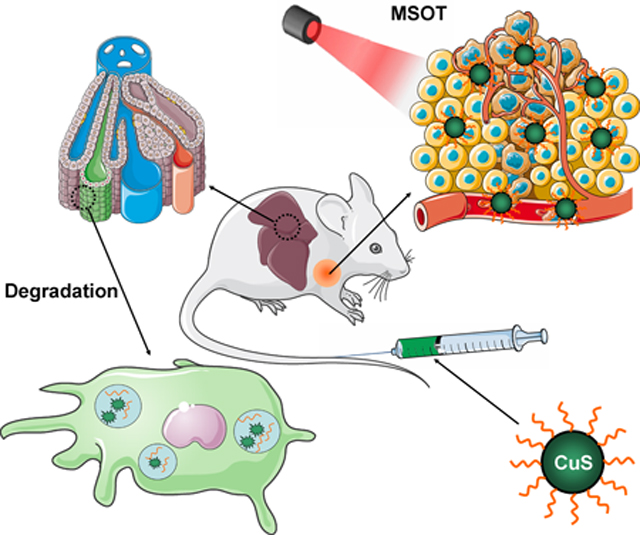
Copper sulfide (CuS) nanoparticles have been considered as one of the most promising theranostic nanoplatforms in the past decade.23, 24 Because of the strong near infrared (NIR) absorption that is derived from the d–d transition of Cu2+ ions, CuS nanoparticles can be employed for photothermal ablation of tumors.25–27 In addition, the radionuclide copper-64 (64Cu) can be intrinsically incorporated into the nanoparticles during CuS synthesis to permit in vivo positron emission tomography (PET) quantification of their retention in tumors.28, 29 Moreover, CuS nanoparticles can be conjugated with targeting ligands for enhanced tumor retention.30 In previous studies, CuS was generally considered stable due to its extremely small equilibrium constant (6 × 10−37) and relatively stable chemical structure that requires high temperature for decomposition.31, 32 However, the thermodynamic properties change dramatically when the dimensions of CuS decrease to the nanoscale.32–35 In the study reported herein, we found that CuS nanoparticles degraded almost completely in the mouse liver within 7 days after injection (Figure 1a), which alleviated concerns about potential long-term toxic effects and increase the prospect for their eventual clinical translation.
Figure 1.
Scheme and characterization of biodegradable CuS-PEG nanoparticles. (a) Schematic illustration of CuS-PEG nanoparticles that allow MSOT-based tumor detection and degradation in the liver within 7 days. The scheme was modified from Servier Medical Art (http://smart.servier.com/), licensed under a Creative Commons Attribution 3.0 Generic License. Nanoparticle size was measured by TEM at magnification of 300k (b) and 500k (c) and dynamic light scattering (DLS) based on number, volume, and intensity distributions (d). (e) Chemical composition of CuS-PEG nanoparticles was characterized by XPS. (f) Absorbance profile was characterized by UV-VIS-NIR spectrometry.
To date, the assessment of nanoparticle biodegradation has primarily relied on tedious ex vivo analyses that entail tissue harvesting after euthanizing animals at different time intervals after injection. However, these ex vivo analyses are neither convenient nor accurate since degradation can also occur during the post-harvest processing steps. In this study, we successfully utilized real-time multispectral optoacoustic tomography (MSOT) to noninvasively investigate the biodegradation of CuS nanoparticles in the liver and their concomitant accumulation in tumors. In MSOT, the imaging signal is generated by photoacoustic phenomenon in response to the fast absorption transients from CuS-accumulated tissues irradiated by short laser pulses. MSOT is more suitable than PET for exploring in vivo degradation since MSOT detects only the signal from intact CuS nanoparticles, rather than degraded Cu ions, whereas PET detects radioactive 64Cu regardless of whether it is present in intact or degraded forms.13, 36 Thus, MSOT provides an attractive avenue for tracking the degradation kinetics and in vivo behavior of amenable nanoparticles in real time and thereby elucidating crucial information for their design, fabrication, and application. In addition, MSOT is multispectral, quantitative and non-radioactive, serving as a promising imaging approach in future clinical setting.37–41
RESULTS AND DISCUSSION
CuS nanoparticles were synthesized by a one-step reaction (Figure S1a). In brief, Na2S was introduced into an aqueous solution of CuCl2 in the presence of PEG-SH and vigorously stirred at 95 ºC for 15 min. The final product, CuS-PEG nanoparticles, were well dispersed with a mean hydrodynamic diameter of 11.1 ± 1.0 nm (number), 25.4 ± 0.9 nm (volume), or 50.8 ± 2.0 nm (intensity) (Figure 1b–d, Figure S1b; n = 3). Successful PEGylation was demonstrated by carbon and oxygen peaks on X-ray photoelectron spectroscopy (XPS) (Figure 1e); PEGylation provided CuS with excellent solubility and biocompatibility for in vivo applications. As-synthesized CuS-PEG nanoparticles demonstrated absorbance in the NIR window, peaking at 1050 nm (Figure 1f), which closely matches the emission wavelength of the Nd-YAG laser that has been commonly deployed in medical applications,42 making it a desirable nanoplatform for simultaneous photoacoustic conversion for MSOT and photothermal conversion for cancer phototherapy.
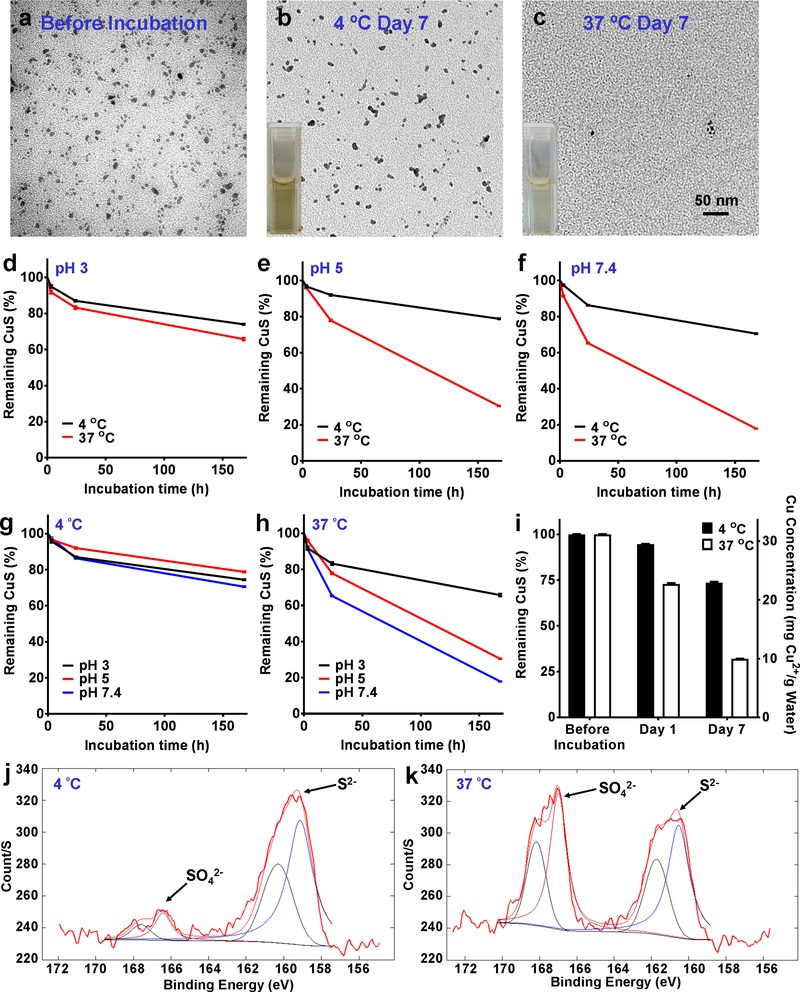
The degradation of CuS-PEG nanoparticles is temperature dependent. After 7 days, CuS-PEG nanoparticles stored in water at 4 ºC preserved their morphology well with only slight reduction in concentration, whereas those stored at 37 ºC decomposed and were hardly visible on transmission electron microscopy (TEM) (Figure 2a–c). The degradation was further investigated by comparing absorbance intensities at 1050 nm at different temperatures (4 ºC and 37 ºC) and pH values (pH 3, laboratory condition; pH 5, water solution or lysosomal environment; and pH 7.4, physiological environment in blood circulation, extracellular fluid, or cytoplasm in normal tissues). Approximately 95.8 ± 0.8% (pH 3), 96.7 ± 0.6% (pH 5) and 97.5 ± 0.4% (pH 7.4) of CuS-PEG nanoparticles remained intact after 3 h incubation at 4 ºC, and majority (74.5 ± 0.3% at pH 3, 78.8 ± 0.3% at pH 5, and 70.6 ± 0.2% at pH 7.4) remained stable even after incubation for 7 days at 4 ºC (Figure 2d–g, Figure S2, and Figure S3; n = 3). When the temperature increased to 37 ºC, although the majority of CuS-PEG nanoparticles remained stable after incubation for 3 h (91.8 ± 0.9% at pH 3, 96.1 ± 0.4% at pH 5, and 91.4 ± 0.4% at pH 7.4), their stabilities significantly decreased after incubation for 7 days (65.8 ± 0.7% at pH 3, 30.5 ± 0.1% at pH 5, and 17.9 ± 0.1% at pH 7.; Figure 2d–f, and 2h, Figure S2, Figure S3 and Figure S4; n = 3), demonstrating the thermosensitive degradation of CuS-PEG nanoparticles. This degradation profile was additionally confirmed by inductively coupled plasma-optical emission spectrometry (ICP-OES) based on the elemental concentration of Cu (Figure 2i; n = 3). Of note, lower pH exhibited relatively strong protection against the degradation of CuS-PEG nanoparticles (Figure 2h), suggesting that reducing pH can be an alternative way to preserve the nanoparticles when reducing temperature is not applicable.
Figure 2.
In vitro degradation of CuS-PEG nanoparticles. TEM images show CuS-PEG nanoparticles before incubation (a) and after 7-day incubation in water (pH 5) at 4 ºC (b) and 37 ºC (c). Thermosensitive degradation profiles were compared over 7-day incubation at pH 3 (d), pH 5 (e), and pH 7.4 (f) or at 4 ºC (g) and 37 ºC (h) (n = 3). (i) Thermosensitive degradation was confirmed by ICP-OES, which showed significantly different Cu concentrations in the retained CuS-PEG nanoparticles between the 4 ºC and 37 ºC groups, after ultracentrifugation with 30,000 kDa filter (n = 3). The degradation mechanism was validated by XPS of CuS-PEG nanoparticles after 7-day incubation in water (pH 5) at 4 ºC (j) and 37 ºC (k).
The mechanism of thermosensitive degradation of CuS-PEG nanoparticles may be primarily attributed to thermal oxidation. Thermal oxidation of larger covellite particles (CuSbulk; > 10 μm) have been performed by heating to higher than 400 ºC.31, 32 The thermal decomposition of CuSbulk involves several intermediate or final products, including different copper sulfides (Cu1.8S and/or Cu2S), oxides (Cu2O and CuO), sulfates (Cu2SO4 and CuSO4), and oxysulfates (CuO·CuSO4) under different temperatures/conditions.31, 32 It is well known that the rate of chemical reaction is highly dependent on the particle size.33–35 However, we surprisingly found that the decomposition temperature could be reduced to as low as the physiological temperature (37 ºC) when the particles were reduced to nanoscale. Given the promising imaging and phototherapeutic applications of CuS nanoparticles, the enhanced thermosensitive degradation profile offers a strong incentive to explore their potential clinical translation by greatly minimizing the concerns about their long-term toxicity. The mechanism of the degradation of CuS-PEG nanoparticles was further validated by XPS, where the majority of sulfur in the nanoparticles retained its S2− configuration under incubation at 4 ºC for 7 days (Figure 2j) but converted to SO42− form after incubation in 37 ºC for 7 days (Figure 2k).
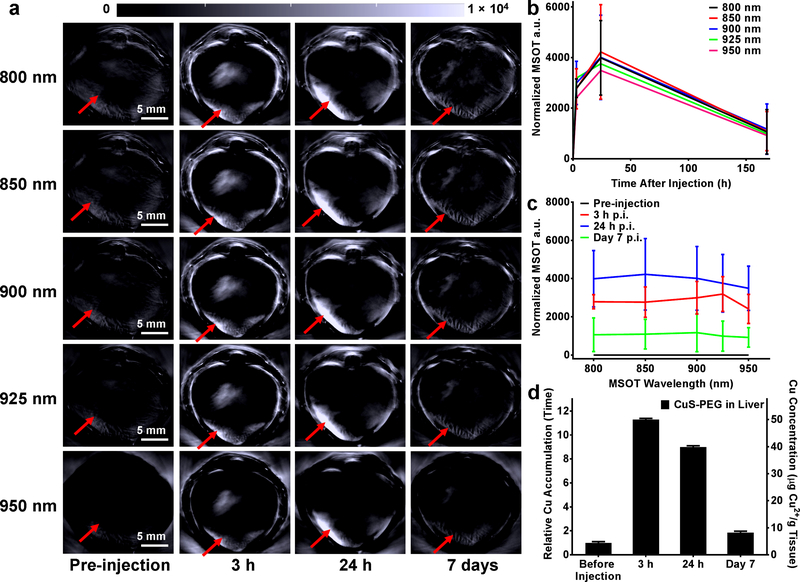
CuS-PEG nanoparticles (200 μg) were subsequently injected into healthy nude mice to investigate the in vivo degradation property. The majority of intravenously injected nanoparticles are eventually taken up by the RES, especially by the Kupffer cells in the liver,5 and enter the decomposition or clearance pathway. Therefore, the signals from the liver after injection of CuS-PEG nanoparticles were monitored by MSOT as an index of in vivo degradation. Under all the photoacoustic wavelengths acquired in the study, significant liver uptake was observed after 3 h and 24 h post injection (p.i.) while the liver uptake reduced to near background level after 7 days p.i. (Figure 3a–c; n = 3). Mice were euthanized and livers were harvested at different time points (pre-injection and 3 h, 24 h, and 7 days p.i.) and subjected to ICP-OES to validate the MSOT results (Figure 3d; n = 3). Intrinsic concentration of Cu in the liver, before injection of CuS-PEG nanoparticles, was found to be 4.4 ± 0.4 μg Cu per g of tissue, which corroborated well with the standard Cu concentration in healthy mouse liver.43 The Cu concentration dramatically increased to 50.0 ± 0.4 μg and 39.9 ± 0.1 μg Cu element in 1 g of liver tissue at 3 h p.i. and 24 h p.i, of CuS-PEG nanoparticles, and significantly decreased to almost background concentrations (8.2 ± 0.4 μg Cu elements per g of liver tissue) after 7 days p.i., confirming the in vivo degradation profile of CuS-PEG nanoparticles observed by noninvasive MSOT. Besides the intrinsic oxidation of CuS-PEG, enzymatic degradation may be another reason for the accelerated degradation rate.44, 45 Clearance of CuS-PEG nanoparticles through hepatic pathway may be also involved in addition to their in vivo degradation, as shown by reduced Cu remaining (8.3 ± 0.1 %) in mouse liver compared to in vitro results (30.5 ± 0.1 % at pH 5 and 17.9 ± 0.1 % at pH 7.4, Figure 2e–f). The degradation and clearance of CuS nanoparticles was further confirmed by the elevated Cu elemental concentration in feces at different time points after injection of CuS-PEG, as analyzed by ICP-OES (Figure S5; n = 3). In addition to MSOT, dynamic and static PET imaging was performed on [64Cu]CuS-PEG injected healthy mice to validate the biodistribution of CuS nanoparticles (Figure S6a and S6c; n = 4). The blood circulation of [64Cu]CuS-PEG was nonlinearly fitted, achieving a one-phase decay pattern (y = 24.71e-x/72.06 + 0.97; y represents the blood pool radioactivity (%ID/g); x represents the time after injection (min)) yielding a circulation half-life of 50.0 min (Figure S6b).
Figure 3.
In vivo degradation and MSOT of CuS-PEG nanoparticles in the liver. (a) Serial static transverse MSOT images were obtained under different wavelengths before intravenous injection and at different time points after intravenous injection of nanoparticles. The red arrows indicate the location of the liver. Region-of-interest (ROI) analyses were performed by comparing the liver signal after normalization (signal intensity at a certain time point – signal intensity before injection) at different wavelengths (b) and at different time points (c) (n = 3). (d) The MSOT results were validated by ICP-OES, which showed a decrease in Cu concentration in the liver (n = 3).
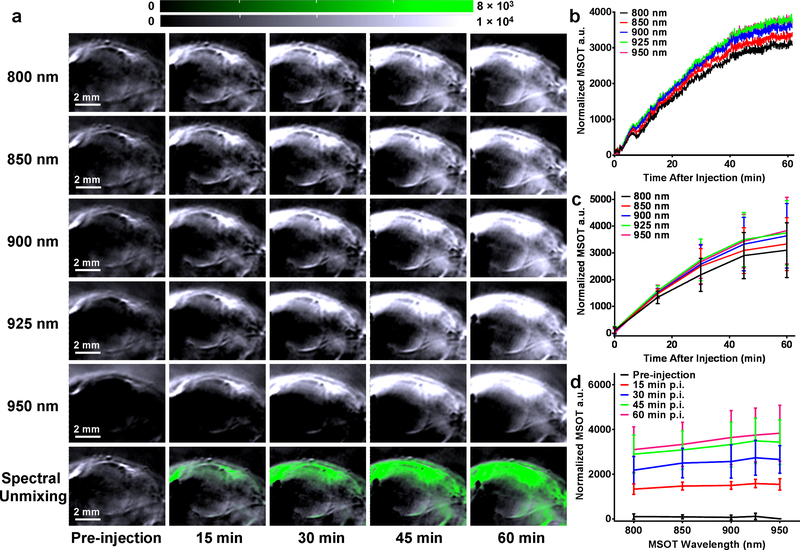
Real-time dynamic MSOT was performed after intravenous injection of CuS-PEG nanoparticles (200 μg) into SKOV-3 ovarian tumor-bearing mice to explore their in vivo cancer diagnostic capacity. Significant increase in the tumor contrast was observed within the first hour after injection under MSOT wavelengths ranging from 800 nm to 950 nm, suggesting the excellent imaging potential of CuS-PEG nanoparticles (Figure 4; n = 3). Of note, MSOT at 925 nm and 950 nm exhibited the strongest signal from the tumor, corresponding to the absorbance of CuS-PEG which peaks at 1050 nm (Figure 1f). Nonlinear fitting was performed on the basis of real-time dynamic MSOT output to understand the in vivo accumulation rate of CuS-PEG nanoparticles in tumor. One-phase exponential association was achieved (y = ymax • (1-e−x/τ); y represents normalized MSOT absorbance intensity (a.u.); x represents the time after injection (min)), showing nearly identical uptake time constant (τ) with different maximal MSOT a.u. (ymax) at different wavelengths (Figure S7). By comparing dynamic MSOT signals with the absorbance profile of CuS-PEG nanoparticles, spectral unmixing could be performed in situ to visualize the accumulation of CuS-PEG nanoparticles (Figure 4a, green channel), providing a convenient approach to pinpointing extrinsically administered probes from the intrinsic background of tissues. Taken together, these results not only demonstrate a desirable pharmacokinetic profile of CuS-PEG nanoparticles but also confirmed real-time dynamic MSOT as a convenient, efficient, and sensitive modality for cancer diagnosis.
Figure 4.
Real-time dynamic MSOT of tumor with CuS-PEG nanoparticles. (a) Serial transverse dynamic MSOT images of SKOV-3 tumor under different wavelengths at different time points after intravenous injection of CuS-PEG nanoparticles. White channel indicates the MSOT intensity. Green channel indicates the distribution of CuS-PEG nanoparticles after spectral unmixing. (b) In situ tumor MSOT intensity was acquired for the first hour after injection of CuS-PEG nanoparticles. Average tumor MSOT intensity was analyzed by comparing the tumor signals after normalization (signal intensity at a certain time point – the signal intensity before injection) at different wavelengths (c) and time points (d) (n = 3).
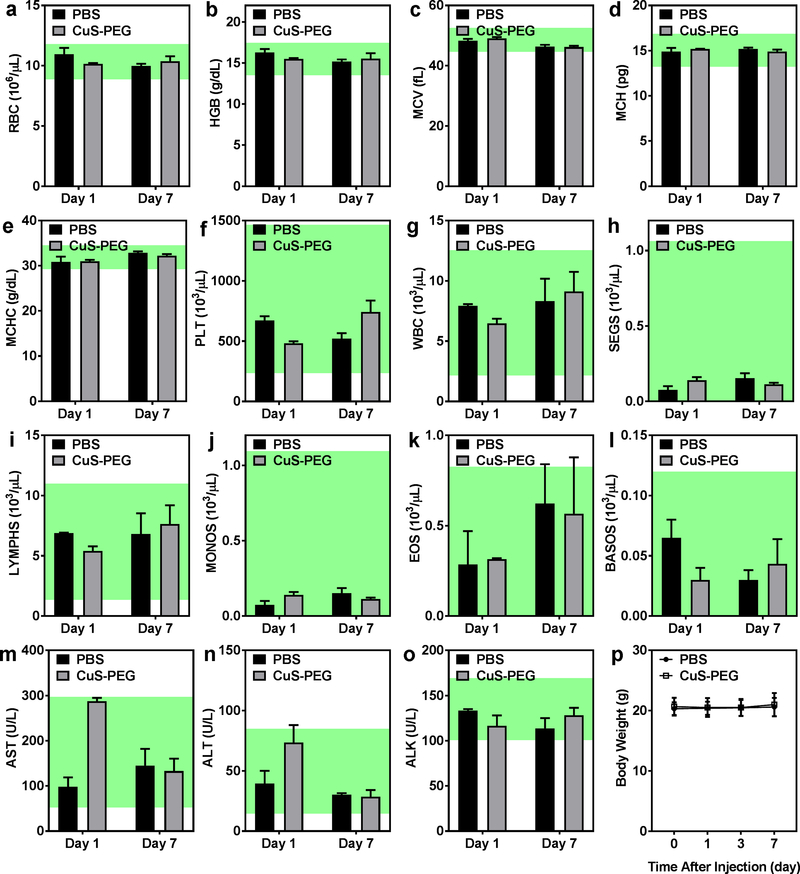
Acute toxicity of CuS-PEG nanoparticles was systematically assessed, since most nanoparticles degraded within 1 week and the degraded Cu2+ may generate free radicals including reactive oxygen species (ROS) and hydroxyl radicals (OH−) and trigger inflammatory effects.46 CuS-PEG nanoparticles at the same dose as that for in vivo tumor imaging were intravenously injected into healthy C57BL/6 mice. An acceptable elevation of liver enzymes was observed day 1 after injection of CuS-PEG nanoparticles when compared to a control group injected with PBS (aspartate aminotransferase: 2.9 ± 0.6 times; alanine aminotransferase: 1.9 ± 0.6 times; alkaline phosphatase: 0.9 ± 0.1 times; all within the normal range; Figure 5m–o; n = 3), decreasing spontaneously to background levels by day 7 (aspartate aminotransferase: 0.9 ± 0.3 times; alanine aminotransferase: 0.9 ± 0.2 times; alkaline phosphatase: 1.1 ± 0.1 times; Figure 5m–o; n = 3), suggesting negligible, short-term and tolerable hepatic toxicity. In addition, both CuS-PEG and PBS groups exhibited similar hematological results at both day 1 and day 7 and exhibited no significant decrease in body weight, further confirming the safety of CuS-PEG nanoparticles in vivo (Figure 5a–l and 5p; n = 3).
Figure 5.
Short-term toxicity of CuS-PEG nanoparticles was investigated via complete hematology assay, including red blood cell count (a), hemoglobin concentration (b), mean corpuscular volume (c), mean corpuscular hemoglobin (d), mean corpuscular hemoglobin concentration (e), platelet count (f), white blood cell count (g), segmented neutrophil count (h), lymphocyte count (i), monocyte count (j), eosinophil count (k), and basophil count (l), liver chemistry analysis, including concentrations of aspartate aminotransferase (m), alanine aminotransferase (n) and alkaline phosphatase (o), and the body weight (p) (n = 3). The shaded regions represent normal ranges of hematology and liver chemistry parameters in female C57BL/6 mice provided by Taconic Biosciences.
CONCLUSION
In conclusion, we reported a thermosensitive biodegradable CuS-PEG nanoplatform that was well preserved at 4 ºC but rapidly degraded at 37 ºC within 1 week. Noninvasive in vivo MSOT was performed, successfully visualizing the biodegradation of CuS-PEG nanoparticles and their excellent tumor imaging capacity in real time. MSOT holds great promise for cancer diagnosis and patient profiling, with its ease of operation, low cost, widespread accessibility, and patient compliance. Importantly, the controlled degradation profile of CuS-PEG nanoparticles is of great significance for their translation to clinical settings. Excellent stability at 4 °C promises convenient logistics during production, transportation, and storage, while controllable degradation at 37 °C in vivo after accomplishing sufficient diagnosis and therapeutic operations indicates all the qualities of an ideal nanotheranostic agent. Traditional inorganic nanoparticles tend to reside eternally in the liver or clear at an incredibly slow pace, which poses a major obstacle that hinder their clinical applications and bench-to-bedside transition. In contrast, our findings in this study demonstrate that tuning the particles to nanoscale level may dramatically switch many clinically relevant materials from stable bulks to biodegradable nanoparticles, alleviating the potential for long-term toxic effects and advancing closer to clinical translation.
METHODS
Reagents.
Polyethylene glycol (PEG) linkers mPEG5k-SH and Chelex 100 resin (50–100 mesh) were purchased from Sigma-Aldrich (St. Louis, MO). Water and all buffers were of Millipore grade and pre-treated with Chelex 100 resin to ensure that the aqueous solution was free of heavy metal. All other chemicals and buffers were obtained from Thermo Fisher Scientific (Fair Lawn, NJ).
Cell lines and animal models.
Human ovarian cancer cells SKOV-3 were obtained from American Type Culture Collection (Manassas, VA) and cultured according to the supplier’s instructions. When they reached ~80% confluence, the cells were harvested for tumor implantation. Six-week-old female nude mice (Taconic Biosciences, Rensselaer, NY) were each subcutaneously injected with 3 × 106 SKOV-3 cells in the upper flank to generate the ovarian cancer xenografts. Mice were used for in vivo experiments when the tumor diameters reached 6–8 mm. All animal studies were conducted under a protocol approved by The University of Texas MD Anderson Cancer Center.
Synthesis and characterization of CuS-PEG nanoparticles.
CuS-PEG nanoparticles were produced by a straightforward one-step method, modified from our previous reports.28, 29, 47 Briefly, an aqueous solution of CuCl2 (5 μmol) was added into a 10 mL aqueous solution containing 10 mg of mPEG5k-SH and incubated at room temperature under vigorous stirring for 0.5 h. An aqueous solution of Na2S (10 μmol) was subsequently added into the reaction mixture. The reaction mixture was moved to a 95 ºC oil bath and stirred for 15 min until a dark green solution was obtained, suggesting the completion of CuS-PEG synthesis. The mixture was then transferred to ice-cold water and purified by ultracentrifugation using a 30 kDa MWCO Amicon Ultra-15 centrifugal filter (EMD Millipore, Burlington, MA) for 3 times. To prevent possible aggregation, 1 mg of mPEG5k-SH was added into the mixture after each ultracentrifugation step. The final product, CuS-PEG nanoparticles, was stored at 4 °C under nitrogen.
The morphology and size of CuS-PEG nanoparticles were characterized by transmission electron microscopy (TEM; JOEL JEM-1010, JOEL USA, Inc., Peabody, MA). The hydrodynamic size distribution was measured by dynamic light scattering (DLS; Brookhaven Instruments Corporation, Holtsville, NY). Chemical composition was analyzed by X-ray photoelectron spectroscopy (XPS; PHI Quantera II scanning XPS microprobe, Physical Electronics, Chanhassen, MN). The absorbance profile was acquired with a UV-VIS-NIR spectrophotometer (DU 800, Beckman Coulter, Brea, CA). The Cu ion concentration was measured by inductively coupled plasma-optical emission spectrometry (ICP-OES; Agilent 725, Agilent Technologies, Santa Clara, CA).
In vitro degradation of CuS-PEG nanoparticles.
CuS-PEG nanoparticles (0.15 mg mL−1) were incubated in water under different temperatures (4 ºC and 37 ºC) and pH values (pH 3, laboratory condition; pH 5, water solution or lysosomal environment;48, 49 and pH 7.4, physiological environment in blood circulation, extracellular fluid, and cytoplasm in normal tissues50). The morphology, size, and density of CuS-PEG nanoparticles incubated under different test conditions were examined by TEM. Their absorbance profiles were compared based on UV-VIS-NIR analysis as the index of remaining non-degraded CuS-PEG nanoparticles. The remaining nanoparticles were purified by 30 kDa MWCO Amicon Ultra-15 centrifugal filters to remove the degraded CuS and byproducts, followed by thorough digestion in HNO3 solution (1:1 mixture of remaining CuS-PEG with 70% HNO3 solution) at 80 ºC for 24 h. The resulting solutions were then diluted with 2% HNO3 solution and sent for ICP-OES analysis to examine the Cu elemental concentration. Other batches of CuS-PEG nanoparticles after incubation under different conditions were lyophilized and sent for XPS analysis to investigate the changes in chemical composition.
In vivo degradation of CuS-PEG nanoparticles.
CuS-PEG nanoparticles (1 mg mL−1, 200 μL per mouse) were intravenously injected into healthy nude mice. Serial static multispectral optoacoustic tomography (MSOT; iThera Medical, Munich, Germany) was performed to visualize the liver uptake of CuS-PEG nanoparticles at different wavelengths (800 nm, 850 nm, 900 nm, 925 nm, and 950 nm) before injection and at different time points post-injection (p.i.) (3 h, 24 h, and 7 days). The images were reconstructed by linear regression. Region-of-interest (ROI) analyses were performed by comparing the liver signal after normalization (signal intensity at a certain time point – the signal intensity before injection; n = 3). In a separate study, nude mice were intravenously injected with CuS-PEG nanoparticles (1 mg mL−1, 200 μL per mouse) and sacrificed at different time points (before injection and 3 h, 24 h, and 7 days p.i.; n = 3) and their livers and feces were harvested for ICP-OES after complete digestion. The digestion was performed by mixing cryoground liver tissues or feces with 70% HNO3 solution (1:1 ratio) and incubating at 80 ºC for 48 h. The resulting solutions were then diluted with 2% HNO3 solution and sent for ICP-OES analysis to examine the Cu elemental concentration.
To confirm the results of MSOT, positron emission tomography (PET) was performed with [64Cu]CuS-PEG nanoparticles. [64Cu]CuS-PEG was synthesized by adding 2 mCi 64CuCl2 along with aqueous solution of CuCl2 (5 μmol) into a 10 mL aqueous solution containing 10 mg of mPEG5k-SH. The following procedures were exactly the same as the synthesis of non-radioactive CuS-PEG described above. 30 min dynamic scans were performed immediately after i.v. injection of [64Cu]CuS-PEG nanoparticles in the healthy mice, followed by static scans at 3 h p.i. and 24 p.i. (n = 4). After the last scan, the mice were sacrificed and important tissues were collected for ex vivo gamma counting studies. Nonlinear fitting was performed to calculate the blood circulation half-life, using one-phase decay (y = (y0- plateau) • e-x/τ + plateau; y represents the blood pool activity (%ID/g); y0 represents the blood uptake (%ID/g) immediately after i.v. injection of [64Cu]CuS-PEG; plateau represents the minimum blood uptake (%ID/g); x represents the time after injection (min); τ represents the uptake time constant (min)).
Real-time dynamic MSOT of tumor with CuS-PEG nanoparticles.
CuS-PEG nanoparticles (1 mg mL−1, 200 μL per mouse) were intravenously injected into SKOV-3 tumor-bearing mice. Serial transverse dynamic MSOT was performed to visualize the tumor uptake of CuS-PEG nanoparticles under different wavelengths (800 nm, 850 nm, 900 nm, 925 nm, and 950 nm) for the first hour after injection. The images were reconstructed by linear regression and spectral unmixing. ROI analyses were performed by comparing the tumor signal after normalization (signal intensity at a certain time point –signal intensity before injection; n = 3). Nonlinear fitting was performed based on tumor dynamic MSOT using one-phase exponential association (y = ymax • (1-e-x/τ); y represents normalized MSOT absorbance intensity (a.u.); x represents the time after injection (min); τ represents the uptake time constant (min)).
Toxicity assessment of CuS-PEG nanoparticles.
CuS-PEG nanoparticles (1 mg mL−1, 200 μL per mouse) were intravenously injected into 6-week-old healthy female C57BL/6 mice. Body weight and general health were monitored over 7 days. Blood was drawn at day 1 and day 7 p.i., and a complete blood panel analysis (ADVIA 120 Hematology System, Siemens Healthineers, Erlangen, Germany) and liver chemistry analysis (COBAS INTEGRA 400plus analyzer, Roche Diagnostics, Risch-Rotkreuz, Switzerland) were performed to monitor the changes in hematological and hepatological parameters in response to CuS-PEG administration.
Supplementary Material
ACKNOWLEDGMENT
We thank S. Goel and S. P. Deming for editing the manuscript. S. Shi was supported by the John S. Dunn, Sr. Distinguished Chair in Diagnostic Imaging. This work was conducted at the MD Anderson Center for Advanced Biomedical Imaging in-part with equipment support from General Electric Healthcare. The Research Animal Support Facility, High Resolution Electron Microscopy Facility, and Small Animal Imaging Facility are supported by a Cancer Center Support Grant from the National Institutes of Health (P30CA016672). X. Wen was supported in part by the China Postdoctoral Science Foundation Grant (2018M641851) and Hei Long Jiang Postdoctoral Foundation (LBH-Z18139).
Footnotes
ASSOCIATED CONTENT
Supporting Information
The Supporting Information is available free of charge on the ACS Publications website.
Scheme of the synthesis of CuS-PEG nanoparticles, average hydrodynamic diameters with number, volume, and intensity size distribution, detailed comparison of absorbance profiles when incubated at 4 ºC and 37 ºC under different conditions, detailed comparison of absorbance profiles when incubated for 0, 3, 24, and 168 h under different conditions, photographs of CuS-PEG nanoparticles under different incubation conditions, ICP-OES analysis of mouse feces, in vivo PET images, ROI analysis of the blood uptake with nonlinear fitting and ex vivo biodistribution studies of intrinsically radiolabeled CuS-PEG nanoparticles, in situ tumor MSOT intensity profiles and their nonlinear fitting.
The authors declare no competing financial interest.
REFERENCES
- (1).Cho K; Wang X; Nie S; Chen ZG; Shin DM Therapeutic nanoparticles for drug delivery in cancer. Clin. Cancer Res 2008, 14, 1310–6. [DOI] [PubMed] [Google Scholar]
- (2).Peer D; Karp JM; Hong S; Farokhzad OC; Margalit R; Langer R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol 2007, 2, 751–60. [DOI] [PubMed] [Google Scholar]
- (3).Strebhardt K; Ullrich A. Paul Ehrlich’s magic bullet concept: 100 years of progress. Nat. Rev. Cancer 2008, 8, 473–80. [DOI] [PubMed] [Google Scholar]
- (4).Schwartz RS Paul Ehrlich’s magic bullets. N. Engl. J. Med 2004, 350, 1079–80. [DOI] [PubMed] [Google Scholar]
- (5).Tsoi KM; MacParland SA; Ma XZ; Spetzler VN; Echeverri J; Ouyang B; Fadel SM; Sykes EA; Goldaracena N; Kaths JM; Conneely JB; Alman BA; Selzner M; Ostrowski MA; Adeyi OA; Zilman A; McGilvray ID; Chan WC Mechanism of hard-nanomaterial clearance by the liver. Nat. Mater 2016, 15, 1212–1221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Dykman L; Khlebtsov N. Gold nanoparticles in biomedical applications: recent advances and perspectives. Chem. Soc. Rev 2012, 41, 2256–82. [DOI] [PubMed] [Google Scholar]
- (7).Boisselier E; Astruc D. Gold nanoparticles in nanomedicine: preparations, imaging, diagnostics, therapies and toxicity. Chem. Soc. Rev 2009, 38, 1759–82. [DOI] [PubMed] [Google Scholar]
- (8).Tang F; Li L; Chen D. Mesoporous silica nanoparticles: synthesis, biocompatibility and drug delivery. Adv. Mater 2012, 24, 1504–34. [DOI] [PubMed] [Google Scholar]
- (9).Chen Y; Chen H; Shi J. In vivo bio-safety evaluations and diagnostic/therapeutic applications of chemically designed mesoporous silica nanoparticles. Adv. Mater 2013, 25, 3144–76. [DOI] [PubMed] [Google Scholar]
- (10).Shi S; Chen F; Cai W. Biomedical applications of functionalized hollow mesoporous silica nanoparticles: focusing on molecular imaging. Nanomedicine (Lond) 2013, 8, 2027–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Zhang Y; Hsu BY; Ren C; Li X; Wang J. Silica-based nanocapsules: synthesis, structure control and biomedical applications. Chem. Soc. Rev 2015, 44, 315–35. [DOI] [PubMed] [Google Scholar]
- (12).Yang K; Feng L; Shi X; Liu Z. Nano-graphene in biomedicine: theranostic applications. Chem. Soc. Rev 2013, 42, 530–47. [DOI] [PubMed] [Google Scholar]
- (13).Shi S; Xu C; Yang K; Goel S; Valdovinos HF; Luo H; Ehlerding EB; England CG; Cheng L; Chen F; Nickles RJ; Liu Z; Cai W. Chelator-Free Radiolabeling of Nanographene: Breaking the Stereotype of Chelation. Angew. Chem. Int. Ed. Engl 2017, 56, 2889–2892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).Liong M; Lu J; Kovochich M; Xia T; Ruehm SG; Nel AE; Tamanoi F; Zink JI Multifunctional inorganic nanoparticles for imaging, targeting, and drug delivery. ACS Nano 2008, 2, 889–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (15).Chandra S; Barick KC; Bahadur D. Oxide and hybrid nanostructures for therapeutic applications. Adv. Drug Deliv. Rev 2011, 63, 1267–81. [DOI] [PubMed] [Google Scholar]
- (16).Yu M; Zheng J. Clearance Pathways and Tumor Targeting of Imaging Nanoparticles. ACS Nano 2015, 9, 6655–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (17).Zhang YN; Poon W; Tavares AJ; McGilvray ID; Chan WCW Nanoparticle-liver interactions: Cellular uptake and hepatobiliary elimination. J. Control Release 2016, 240, 332–348. [DOI] [PubMed] [Google Scholar]
- (18).Longmire M; Choyke PL; Kobayashi H. Clearance properties of nano-sized particles and molecules as imaging agents: considerations and caveats. Nanomedicine (Lond) 2008, 3, 703–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Nel A; Xia T; Madler L; Li N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–7. [DOI] [PubMed] [Google Scholar]
- (20).Khanna P; Ong C; Bay BH; Baeg GH Nanotoxicity: An Interplay of Oxidative Stress, Inflammation and Cell Death. Nanomaterials (Basel) 2015, 5, 1163–1180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Xia T; Kovochich M; Brant J; Hotze M; Sempf J; Oberley T; Sioutas C; Yeh JI; Wiesner MR; Nel AE Comparison of the abilities of ambient and manufactured nanoparticles to induce cellular toxicity according to an oxidative stress paradigm. Nano Lett. 2006, 6, 1794–807. [DOI] [PubMed] [Google Scholar]
- (22).Sharifi S; Behzadi S; Laurent S; Forrest ML; Stroeve P; Mahmoudi M. Toxicity of nanomaterials. Chem. Soc. Rev 2012, 41, 2323–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (23).Goel S; Chen F; Cai W. Synthesis and biomedical applications of copper sulfide nanoparticles: from sensors to theranostics. Small 2014, 10, 631–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (24).Zhou M; Tian M; Li C. Copper-Based Nanomaterials for Cancer Imaging and Therapy. Bioconjug. Chem 2016, 27, 1188–99. [DOI] [PubMed] [Google Scholar]
- (25).Dong K; Liu Z; Li Z; Ren J; Qu X. Hydrophobic anticancer drug delivery by a 980 nm laser-driven photothermal vehicle for efficient synergistic therapy of cancer cells in vivo. Adv. Mater 2013, 25, 4452–8. [DOI] [PubMed] [Google Scholar]
- (26).Goel S; Ferreira CA; Chen F; Ellison PA; Siamof CM; Barnhart TE; Cai W. Activatable Hybrid Nanotheranostics for Tetramodal Imaging and Synergistic Photothermal/Photodynamic Therapy. Adv. Mater 2018, 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Tian Q; Tang M; Sun Y; Zou R; Chen Z; Zhu M; Yang S; Wang J; Hu J. Hydrophilic flower-like CuS superstructures as an efficient 980 nm laser-driven photothermal agent for ablation of cancer cells. Adv. Mater 2011, 23, 3542–7. [DOI] [PubMed] [Google Scholar]
- (28).Zhou M; Zhang R; Huang M; Lu W; Song S; Melancon MP; Tian M; Liang D; Li C. A chelator-free multifunctional [64Cu]CuS nanoparticle platform for simultaneous micro-PET/CT imaging and photothermal ablation therapy. J. Am. Chem. Soc 2010, 132, 15351–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (29).Zhou M; Chen Y; Adachi M; Wen X; Erwin B; Mawlawi O; Lai SY; Li C. Single agent nanoparticle for radiotherapy and radio-photothermal therapy in anaplastic thyroid cancer. Biomaterials 2015, 57, 41–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (30).Zhou M; Song S; Zhao J; Tian M; Li C. Theranostic CuS Nanoparticles Targeting Folate Receptors for PET Image-Guided Photothermal Therapy. J. Mater. Chem. B 2015, 3, 8939–8948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (31).Dunn JG; Muzenda C. Thermal oxidation of covellite (CuS). Thermochim. Acta 2001, 369, 117–123. [Google Scholar]
- (32).Simonescu CM; Teodorescu VS; Carp O; Patron L; Capatina C. Thermalbehaviour of CuS (covellite) obtained from copper–thiosulfate system. J. Therm. Anal. Calorim 2007, 88, 71–76. [Google Scholar]
- (33).Li W; Xue Y; Cui Z. Size dependence of surface thermodynamic properties of nanoparticles and its determination method by reaction rate constant. Physica B Condens. Matter 2016, 495, 98–105. [Google Scholar]
- (34).Fu Q; Cui Z; Xue Y; Duan H. Research of Size- and Shape-Dependent Thermodynamic Properties of the Actual Melting Process of Nanoparticles. J. Phys. Chem. C 2018, 122, 15713–15722. [Google Scholar]
- (35).Xiong S; Qi W; Cheng Y; Huang B; Wang M; Li Y. Universal relation for size dependent thermodynamic properties of metallic nanoparticles. Phys. Chem. Chem. Phys 2011, 13, 10652–60. [DOI] [PubMed] [Google Scholar]
- (36).Zhang Y; Jeon M; Rich LJ; Hong H; Geng J; Shi S; Barnhart TE; Alexandridis P; Huizinga JD; Seshadri M; Cai W; Kim C; Lovell JF Non-invasive multimodal functional imaging of the intestine with frozen micellar naphthalocyanines. Nat. Nanotechnol 2014, 9, 631–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Wang LV; Yao J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 2016, 13, 627–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (38).Wang X; Pang Y; Ku G; Xie X; Stoica G; Wang LV Noninvasive laser-induced photoacoustic tomography for structural and functional in vivo imaging of the brain. Nat. Biotechnol 2003, 21, 803–6. [DOI] [PubMed] [Google Scholar]
- (39).Razansky D; Distel M; Vinegoni C; Ma R; Perrimon N; Köster RW; Ntziachristos V. Multispectral opto-acoustic tomography of deep-seated fluorescent proteins in vivo. Nat. Photonics 2009, 3, 412–417. [Google Scholar]
- (40).Foroutan F; Jokerst JV; Gambhir SS; Vermesh O; Kim HW; Knowles JC Sol-gel synthesis and electrospraying of biodegradable (P2O5)55-(CaO)30-(Na2O)15 glass nanospheres as a transient contrast agent for ultrasound stem cell imaging. ACS Nano 2015, 9, 1868–1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (41).Kim T; Lemaster JE; Chen F; Li J; Jokerst JV Photoacoustic Imaging of Human Mesenchymal Stem Cells Labeled with Prussian Blue-Poly(l-lysine) Nanocomplexes. ACS Nano 2017, 11, 9022–9032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (42).Beer M; Jocham D; Beer A; Staehler G. Adjuvant laser treatment of bladder cancer: 8 years’ experience with the Nd-YAG laser 1064 nm. Br. J. Urol 1989, 63, 476–8. [DOI] [PubMed] [Google Scholar]
- (43).Keen CL; Hurley LS Developmental changes in concentrations of iron, copper, and zinc in mouse tissues. Mech. Ageing. Dev 1980, 13, 161–76. [DOI] [PubMed] [Google Scholar]
- (44).Lovell JF; Jin CS; Huynh E; Jin H; Kim C; Rubinstein JL; Chan WC; Cao W; Wang LV; Zheng G. Porphysome nanovesicles generated by porphyrin bilayers for use as multimodal biophotonic contrast agents. Nat. Mater 2011, 10, 324–32. [DOI] [PubMed] [Google Scholar]
- (45).Chen M; Zeng G; Xu P; Lai C; Tang L. How Do Enzymes ‘Meet’ Nanoparticles and Nanomaterials? Trends Biochem. Sci 2017, 42, 914–930. [DOI] [PubMed] [Google Scholar]
- (46).Simpson JA; Cheeseman KH; Smith SE; Dean RT Free-radical generation by copper ions and hydrogen peroxide. Stimulation by Hepes buffer. Biochem. J 1988, 254, 519–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (47).Ku G; Zhou M; Song S; Huang Q; Hazle J; Li C. Copper sulfide nanoparticles as a new class of photoacoustic contrast agent for deep tissue imaging at 1064 nm. ACS Nano 2012, 6, 7489–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (48).Mindell JA Lysosomal acidification mechanisms. Annu. Rev. Physiol 2012, 74, 69–86. [DOI] [PubMed] [Google Scholar]
- (49).Grossi M; Morgunova M; Cheung S; Scholz D; Conroy E; Terrile M; Panarella A; Simpson JC; Gallagher WM; O’Shea DF Lysosome triggered near-infrared fluorescence imaging of cellular trafficking processes in real time. Nat. Commun 2016, 7, 10855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (50).Roos A; Boron WF Intracellular pH. Physiol. Rev 1981, 61, 296–434. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.