Abstract
Morphological diversity and several new distinct pathologic subtypes of hepatocellular carcinoma (HCC) are now well-recognized. Recent advances in tumor genomics and transcriptomics have identified several recurrent somatic/genetic alterations that are closely related with histomorphological subtypes and have therefore, greatly improved our understanding of HCC pathogenesis. Pathologic subtyping allows for a diagnosis which is clinically helpful and can have important implication in patient prognostication as some of these subtypes are extremely aggressive with vascular invasion, early recurrence, and worst outcomes. Several targeted treatments are now being considered in HCC, and the reporting of subtypes may be quite useful for personalized therapeutic purpose. This manuscript reviews the recently identified histomorphological subtypes and molecular alterations in HCC.
Keywords: Pathology, Hepatocellular carcinoma subtypes, Macrotrabecular massive, Steatohepatitic, Fibrolamellar, Molecular alterations
Core Tip: We summarize several new distinct histologic subtypes of hepatocellular carcinoma (HCC) and recurrent molecular alterations in HCC. Major histologic subtypes like macrotrabecular massive, fibrolamellar HCC, steatohepatitic HCC, scirrhous HCC, lymphoepithelioma-like HCC, and combined hepatocellaular-cholangiocarcinoma are discussed in detail. Rare and provisional histological variants are also discussed.
INTRODUCTION
The incidence of hepatocellular carcinoma (HCC) has been increasing steadily over the past two decades and currently ranks as the fifth most common cancer in men and seventh in women[1,2]. HCC is now the fourth-most common cause of cancer-related deaths and the most frequent primary liver neoplasia, causing more than 80%-85% of liver cancer cases globally[3]. Major risk factors associated with HCC are chronic infection with hepatitis B virus and hepatitis C virus, chronic alcohol consumption, and non-alcoholic fatty liver disease associated with metabolic syndrome, diabetes and obesity. Prognosis of patients with HCC remains poor with 5-year survival rate of 18%, as the majority of these tumors are detected at a clinically advanced stage[3]. Hepatocarcinogenesis is a multistep process of malignant transformation of hepatocytes through the sequential accumulation of multiple genomic and epigenomic alterations. HCC is a histologically and genetically diverse cancer[4]. Indeed, several new pathologic subtypes of HCC have been reported recently and new underlying genetic alterations have been described. HCC histological growth patterns are closely related to molecular alterations and oncogenic pathways.
PATHOLOGY OF PRECANCEROUS LESIONS AND CONVENTIONAL HCC
It is now well-established that HCC evolves from precancerous lesions (dysplastic foci/dysplastic nodules). By consensus, the sequence of hepatocarcinogenesis includes low-grade dysplastic nodule (LGDN), high-grade dysplastic nodule (HGDN), early HCC, and small progressed HCC[5,6] (Figure 1). This classification is also supported by molecular studies on increasing accumulation of clonal molecular alterations[7]. Dysplastic foci (< 1 mm in size) are identified incidentally in chronic liver disease (CLD) and are microscopic lesions composed of dysplastic hepatocytes (Figure 2A). The nature of dysplasia is similar to that observed in dysplastic nodules: Large cell change, small cell change, or focal iron free area. Large cell change is characterized by cellular enlargement with enlarged pleomorphic nuclei, abundant cytoplasm, and frequent multinucleation of hepatocytes. The nuclear-cytoplasmic ratio is preserved in large cell change. Small cell change is characterized by decreased cell volume, increased nuclear-cytoplasmic ratio, cytoplasmic basophilia, mild nuclear pleomorphism, and hyperchromasia. Iron-free foci in patients with marked hepatic iron overload show immunohistochemical evidence of proliferative activity and are associated with a high incidence of HCC. Dysplastic nodules are usually identified in livers with cirrhosis but are also occasionally found along with CLD without cirrhosis. These are around 5-15 mm in diameter and can be single or multiple lesions. A LGDN is a distinctly nodular lesion displaying a monotonous cell population with a mild increase in cellular density, a clear trabecular arrangement, and no architectural atypia in comparison to the neighbouring cirrhotic liver. HGDNs are characterized by hepatocyte proliferation with atypical cytological and/or architectural features that are not sufficient for a diagnosis of HCC. HGDN show higher cellular density and frequently demonstrates small cell change.
Figure 1.
International consensus group for hepatocellular neoplasia classification of small hepatocellular lesions. HCC: Hepatocellular carcinoma.
Figure 2.
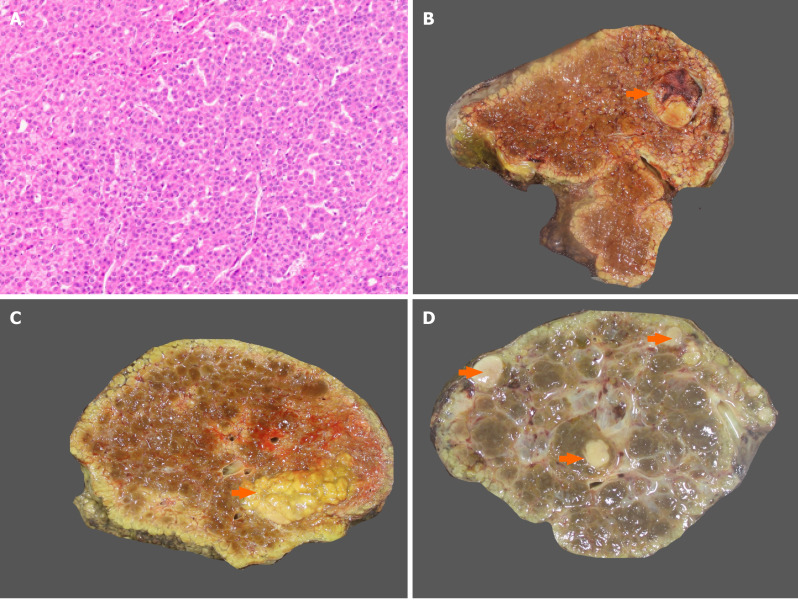
Dysplasia and gross morphology of hepatocellular carcinoma. A: Dysplastic foci with small cell change (hematoxylin and eosin); B: Nodular hepatocellular carcinoma (HCC) in a cirrhotic liver (arrow); C: Multinodular HCC in a cirrhotic liver (arrow); D: Multicentric HCC (arrow).
Macroscopically, lesions with foci of malignant transformation may demonstrate variable features like vaguely nodular, expansile nodular, multinodular, multicentric, cirrhotomimetic, nodular with perinodular extension, and infiltrative types (Figure 2B-D). Small HCCs, ≤ 2 cm, are divided into two groups. Early HCC are vaguely nodular with indistinct margins and usually show higher cellular density than the surrounding cirrhotic tissue with increased nuclear to cytoplasmic ratio, irregular trabeculae, pseudoacini formation, and unpaired arterioles (Figure 3A). Stromal invasion is one of the most important characteristics to differentiate early HCC from HGDN, but is however difficult to identify. Progressed HCC are distinctly nodular with distinguishable margins, frequently capsulated, and show infiltrative or expansile growth pattern. Morphologically, conventional HCC show 4 major architectural growth patterns: Trabecular, solid, pseudoglandular/acinar, and macrotrabecular (Figure 3B-D) and several cytological features (clear cell, steatosis, pleomorphism, multinucleation, foamy cells, oncocytic cells, spindle cells), with frequent co-existence of several features (Figure 4)[8,9]. Various intra-hepatocytic inclusions may be seen like hyaline globules (Figure 5A), Mallory-Denk bodies, bile, and pale bodies. Two histological grading systems for HCC are available. The WHO three-tiered grading system is based on a combination of cytological features and differentiation, and further grades the tumor into well, moderately, and poorly differentiated types[10]. Primary hepatic undifferentiated carcinoma is not included in the WHO grading system as it shows no evidence of either hepatic or biliary differentiation. It is the system most commonly used by pathologists[10,11]. Edmondson and Steiner grading system divides HCC into four grades based on histological differentiation with grade 1 being very well differentiated[12]. A correlation between the histological grade and patient prognosis has been reported[13]. Poorly differentiated HCC are associated with higher recurrence after surgery[14].
Figure 3.
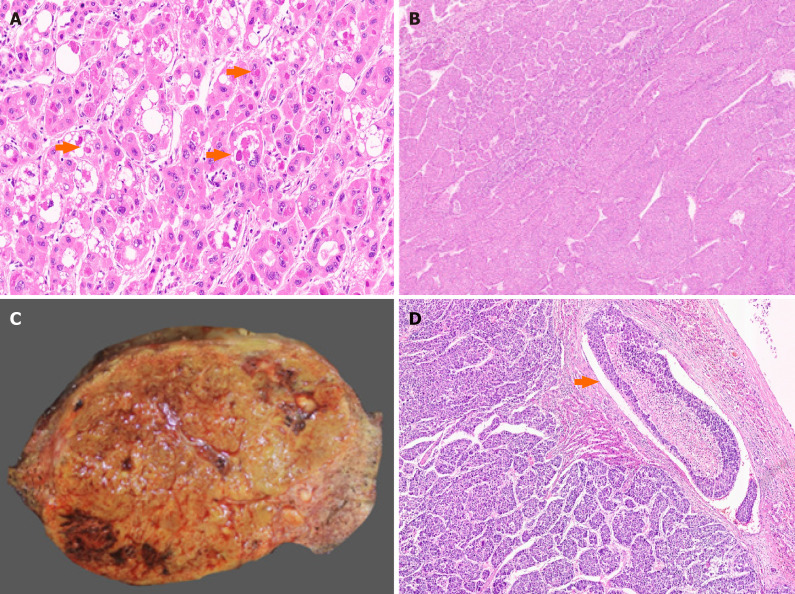
Well differentiated hepatocellular carcinoma. A: Early hepatocellular carcinoma (HCC) with pseudoacinar pattern [hematoxylin and eosin (H&E)]; B: Well differentiated HCC with thin trabeculae (H&E); C: Well differentiated HCC with pseudoacini (H&E); D: HCC with solid sheet growth pattern (H&E).
Figure 4.
Hepatocellular carcinoma cytological features. A: Hepatocellular carcinoma (HCC) with fatty change [hematoxylin and eosin (H&E)]; B: Marked pleomorphism in an HCC (H&E); C: Foamy cell cytoplasm in an HCC (H&E); D: HCC with oncocytic cells (H&E).
Figure 5.
Conventional and macrotrabecular massive hepatocellular carcinoma. A: Hyaline globules in a conventional hepatocellular carcinoma (HCC) [arrow, hematoxylin and eosin (H&E)]; B: Macrotrabecular massive HCC (H&E); C: Large macrotrabecular massive HCC with satellite nodule; D: Macrotrabecular massive HCC with vascular invasion (arrow, H&E).
ANCILLARY STUDIES FOR THE PATHOLOGIC DIAGNOSIS OF HCC
Differentiation of HCC from other malignancies can be difficult; immunostaining can be helpful to differentiate between these lesions. Arginase-1 is a binuclear manganese metalloenzyme and is the most sensitive and specific marker of hepatocytic differentiation[15]. It shows diffused nuclear and cytoplasmic staining. Carcinoma with hepatoid differentiation and rare cases of adenocarcinoma (including colorectal, pancreatic, breast, and prostatic primaries), cholangiocarcinoma, and may however, show focal or weak Arginase-1 positivity[16]. Hepatocyte paraffin 1 (Hep-Par 1) is a monoclonal antibody that reacts with the urea cycle enzyme carbamoyl phosphate synthetase 1 of liver mitochondria. It shows diffuse granular cytoplasmic staining in normal and neoplastic hepatocytes[17]. Hep-Par 1 is unfortunately frequently negative for poorly differentiated HCCs. Few cholangiocarcinoma and metastatic adenocarcinoma may show Hep-Par 1 immunopositivity. Glypican 3 is excellent marker for neoplastic hepatocytes with cytoplasmic, membranous, or golgi-zone pattern of immunopositivity[18]. Other immunostains like polyclonal carcinoembryonic antigen (pCEA), CD10 and villin shows a distinct canalicular immunostaining pattern in HCC[16]. Alpha-fetoprotein (AFP) immunohistochemistry is not very useful in diagnosis of HCC as it has low sensitivity and is often only focally positive. Albumin RNA in situ hybridization has been shown to be a highly sensitive maker for hepatocellular differentiation[19]. Its specificity is however suboptimal and it can be positive in tumors demonstrating hepatocytic differentiation, such as hepatoid carcinomas of various sites, intrahepatic cholangiocarcinoma (iCCA), gall bladder adenocarcinoma, and yolk sac tumour[20]. Well-differentiated HCCs may also be difficult to distinguish from dysplastic nodules. Loss of reticulin, stromal invasion, and neoarteriolization are particularly useful in these cases. The combination of 3 immunomarkers-glypican 3, glutamine synthetase (GS), and heat shock protein 70-can be used to differentiate early HCC from HGDN[12].
DISTINCT PATHOLOGICAL SUBTYPES WITH MOLECULAR FEATURES
Table 1 summarizes distinct pathological subtypes and their molecular features.
Table 1.
Hepatocellular carcinoma distinct subtypes with pathological and molecular features
|
Distinct subtypes
|
Pathological features
|
Molecular features
|
| Macrotrabecular massive | Macrotrabeculae > 50% of the tumor, staellite nodules, vascular invasion | TP53 mutations and FGF19 amplifications |
| Steatohepatitic | Steatohepatitis in the tumor | IL6/JAK/STAT pathway activation |
| Scirrhous | Dense fibrosis in > 50% of the tumor | Activation of (TGF-β) pathway, with overexpression of VIM, SNAIL (SNAI1), SMAD4 and TWIST |
| Fibrolamellar | Large polygonal tumor cells with abundant eosinophilic granular cytoplasm and dense bands of intratumoral fibrosis | Recurrent chimeric DNAJB1-PRKACA gene fusion |
| Lymphoepithelioma-like | Neoplastic epithelial cells with a prominent lymphoid infiltrate | Marked focal amplification of chromosome 11q13.3 |
| Progenitor | Immunohistochemical expression of biliary marker CK19 in > 5% of tumor cells | TP53 mutations |
| Combined hepatocellular-cholangiocarcinoma | Unequivocal presence of both hepatocytic and cholangiocytic differentiation | TP53, TERT, IDH mutations |
DNAJB1-PRKACA: DnaJ heat shock protein family member B1 (DNAJB1) and protein kinase 3'-5'-cyclic adenosine monophosphate-activated catalytic subunit alpha; IL6: Interleukin-6; JAK: Janus kinas; STAT: Signal transducer and activator of transcription; FGF19: Fibroblast growth factor 19; TERT: Telomerase reverse transcriptase; IDH: Isocitrate dehydrogenase; SNAIL (SNAI1): SNAIL family transcriptional repressor 1; SMAD4: SMAD family member 4; TWIST: Twist-related protein; TGF-β: Transforming growth factor beta.
MACROTRABECULAR MASSIVE HCC
The Macrotrabecular-Massive HCC (MTM-HCC) subtype represents a novel histomorphological subtype of HCC. It represents 10%–20% of all cases of HCC. Histologically, it is defined by a macrotrabecular (> 6 cells thick) architectural pattern involving > 50% of the entire tumour, regardless of the associated cytological features (Figure 5B)[4]. Most trabeculae in MTM-HCC are ≥ 10 cells thick[10]. On trucut biopsy analysis, MTM-HCC case is classified if at least 1 focus of macrotrabecular pattern is observed, and the percentage of the macrotrabecular pattern is not taken into account. Pathologists robustly identify MTM-HCC with good inter-observer agreements. MTM-HCC is also characterized by an association with tumor protein 53 (TP53) mutations and fibroblast growth factor 19 (FGF19) amplifications[21]. Being an aggressive form of HCC, it is associated with poor prognostic factors, such as higher Barcelona Clinic Liver Cancer (BCLC) stage B or C, higher AFP levels (> 100 ng/dL), larger tumor size, frequent satellite nodules, substantial necrosis, and macro or microvascular invasion; hence, there is a higher risk of early tumor recurrence and poor disease-free and overall survival rate (Figures 5C and D)[22]. These findings have been further validated by several groups. The other characteristics are its association with viral hepatitis B infection and profound activation of angiogenesis[23]. Presence of the satellite nodule on the multiphase liver magnetic resonance imaging (MRI) has been described as independent factor associated with both early and overall tumor recurrence[24]. Rhee et al[25] reported imaging findings of MTM-HCC by gadoxetic acid-enhanced MRI. With gadoxetic acid-enhanced MRI findings, including arterial phase hypovascular component, they were able to stratify the probability of MTM-HCC and obtain prognostic information[25]. The gene expression profile associated with the MTM-HCC subtype is characterized by the activation of neoangiogenesis, with overexpression of angiopoietin 2 and Vascular Endothelial Growth Factor A (VEGFA). Angiopoietin 2 is responsible for the destabilization of established vasculature and subsequent neoangiogenesis, and also disturbs interactions between endothelial and periendothelial cells, which results in an increased receptiveness to VEGFA[4]. These tumors have high expression of neoangiogenesis-related genes, which led to the discovery of Endothelial-Specific Molecule 1 as a reliable immunostaining marker[26]. Immune assessment of MTM-HCC using expression of the programmed death ligand 1 (PD-L1) and Chemokine-like factor MARVEL transmembrane domain containing 6 (CMTM6) protein coded immune-checkpoint inhibitors showed higher tumoral PD-L1 expression, higher density of inflammatory cells, and higher CMTM6 expression. Therefore, combined expression of PD-L1 and CMTM6 were associated with shorter overall and disease-free survival[27].
STEATOHEPATITIC HCC
Steatohepatitic HCC (SH-HCC) first described in 2010 by Salomao et al[28], and is a distinct histological subtype strongly associated with underlying steatosis and/or steatohepatitis and metabolic syndrome[28]. SH-HCC demonstrates morphological features similar to steatohepatitis with macrovesicular steatosis, hepatocellular ballooning with cytoplasmic clarification, Mallory-Denk bodies, pericellular fibrosis, and patchy inflammation (Figure 6A)[29]. The steatohepatitis should be a dominant part of the tumor morphology, and at least 50% of the tumor should show this pattern. Fibrosis can be best demonstrated on histochemical stain like Masson trichrome. The immunophenotyping of SH-HCC is similar to conventional HCC; however, it shows increased immunostaining with markers of inflammation like C-reactive protein due to interleukin (IL)-6/Janus kinase (JAK)-signal transducer and activator of transcription (STAT) pathway activation[21]. SH-HCC are well-differentiated to moderately differentiated tumors and are associated G4 transcriptomics subclass. In a recent transcriptomic analysis by Van Treeck et al[30] SH-HCC demonstrated a distinctive differential gene expression profile with upregulation of the sonic hedgehog signal transduction pathway based on GLI1 family zinc finger 1 (GLI1) overexpression. GLI1 gene encodes a protein that functions as a transcription factor protein and plays a role in the regulation of stem cell proliferation. There was reduced expression of carnitine palmitoyltransferase 2 (CPT2) transcripts. CPT2 is a mitochondrial enzyme with an essential role in fatty acid β-oxidation and carnitine metabolism. In a mouse model of obesity-driven and non-alcoholic steatohepatitis-driven HCC, metabolic reprogramming mediated by the downregulation of CPT2 enables protection of neoplastic hepatocytes from lipotoxicity[31]. Therefore; reduced level of CPT2 is believed to facilitate survival of malignancy in obesity-associated HCC. Lee et al[32] recently suggested that alteration of the tumor stroma might play an important role in SH-HCC development, and as compared to classical HCC, cancer-associated fibroblasts in SH-HCC and non-tumoral stellate cells were characterized by increased expression of IL-6, a key governor of the JAK/STAT pathway[32]. SH-HCC appears to have similar overall and disease-free survival, development of metastasis, or local recurrence compared with conventional HCC[29].
Figure 6.
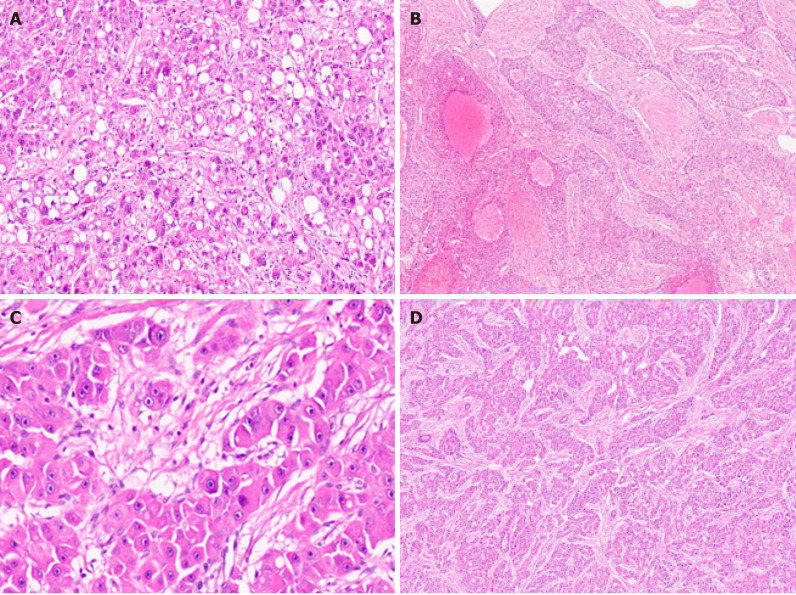
Hepatocellular carcinoma subtypes. A: Hepatocellular carcinoma (HCC) with steatohepatitic pattern [hematoxylin and eosin (H&E)]; B: Sclerotic HCC (H&E); C: Fibrolamellar HCC with large cells and prominent nucleoli (H&E); D: Fibrolamellar HCC with lamellar fibrosis (H&E).
SCIRRHOUS HCC
Scirrhous HCC represent approximately 5% of all cases[33]. Radiologic findings are atypical and often show arterial phase peripheral enhancement and venous phase persistent enhancement[34]. Scirrhous HCC is characterized by tumor cell clusters surrounded by abundant fibrous stroma which should constitute at least 50% of the tumor (Figure 6B)[11]. The presence of marked intratumoural fibrosis may lead to a faulty impression of intrahepatic CCA on radiology and macroscopic examination. Scirrhous HCC are mostly well to moderately differentiated HCC. Steatosis, clear cell change, pale bodies, and hyaline bodies have also been reported. Immunohistochemically, there is lack of positive staining for primary hepatocellular stains like HepPar-1 and pCEA in more than 60% of scirrhous HCC, with arginase and glypican 3 positivity in around 80% of cases[35]. Immunostains used for adenocarcinoma, like cytokeratin (CK) 7, CK19, and epithelial cell adhesion molecule, are positive in more than 60% of cases and can lead to erroneous diagnosis of adenocarcinoma[36]. Scirrhous HCC may resemble fibrolamellar HCC histologically, and molecular testing for DNAJ heat shock protein family member B1 (DNAJB1) and protein kinase 3'-5'-cyclic adenosine monophosphate (cAMP)-activated catalytic subunit alpha (PRKACA) fusion can be performed in histologically difficult cases[37]. There is no significant difference in prognosis in Scirrhous HCC compared with conventional HCC[38]. Expression of various cholangiocarcinoma-like and stem-cell-like genomic traits, including CK7 (KRT7), CK19 (KRT19), THY1, and CD133/Prominin-1, have been reported in scirrhous-HCC, and it has therefore been suggested that scirrhous HCC harbour intermediate molecular features, between HCC and cholangiocarcinoma[4,39]. Scirrhous HCC genomic profile also shows activation of transforming growth factor beta pathway/epithelial-to-mesenchymal transition related genes, with overexpression of Vimentin, SNAIL family transcriptional repressor 1, SMAD family member 4, and Twist-related protein[21].
FIBROLAMELLAR HCC
Fibrolamellar HCC (FL-HCC) is a rare and unique histologic subtype of liver cancer with a predilection for adolescent and young adults (male:female, 1:1) without underlying liver disease, a characteristic morphological pattern with large neoplastic cells, distinct immunostaining, and recurrent genomic abnormalities typically involving PRKACA[40]. FL-HCC comprises approximately only 1% of primary liver cancer[41]. FL-HCC commonly presents as an abdominal mass with enlargement of liver, pain in abdomen, and features of biliary obstruction secondary to external compression by the mass lesion[42]. Rarely FL-HCC can present with paraneoplastic manifestations. These tumors are mostly solitary, large, and well circumscribed grossly with a yellow tan colored cut surface, and areas of central scarring are identified in almost 70% of cases[43,44]. Importantly, FL-HCC are much more likely to invade regional lymph nodes. Histologically, the tumor cells are large, polygonal with abundant eosinophilic granular cytoplasm (because of numerous mitochondria), centrally located nuclei with vesicular chromatin, and prominent nucleoli (Figure 6C). Focal bi-or multi-nucleation are also reported. Dense bands of intratumoural fibrosis arranged in lamellar (parallel arrangement) pattern separates the trabeculae and clusters of tumor cells (Figure 6D). FL-HCC also show presence of pale or hyaline bodies; however, these are not specific and may be observed in conventional HCC. Immunophenotyping shows neoplastic cells are positive of CD68 and CK-7 (biliary lineage) apart from markers of hepatic differentiation (Arginase 1, Hep-Par 1 and albumin mRNA as detected by in situ hybridization). Honeyman et al[37] first reported a specific 400-kilobase deletion on chromosome 19 in FL-HCC leading to recurrent chimeric DNAJB1-PRKACA gene fusion, genetic footprint of FL-HCC. DNAJB1 encodes a member of heat shock protein 40 which is involved in protein folding within cells, while PRKACA codes for the cAMP-dependent protein kinase catalytic subunit alpha; the molecular alteration results in upregulation of PRKACA activity by a promoter switch mechanism[45,46]. Both fluorescence in situ hybridization (FISH) or reverse transcription polymerase chain reaction are available now to detect DNAJB1-PRKACA fusion for confirming the diagnosis of FL-HCC. Recently, the genetic alteration (DNAJB1-PRKACA gene fusion) has also been identified in a set of oncocytic pancreaticobiliary neoplasm; however, DNAJB1-PRKACA fusion is still the most accurate test when the diagnosis of FL-HCC is doubtful[47,48]. FL-HCC has a unique gene expression profile, with Erb-b2 receptor tyrosine kinase (ERBB) 2 overexpression and glycolysis upregulation leading to compensatory mitochondrial hyperplasia, and various neuroendocrine genes, including Proprotein Convertase Subtilisin/Kexin Type 1, Neurotensin, Delta/Notch Like EGF Repeat Containing and Calcitonin Related Polypeptide Alpha[49].
LYMPHOEPITHELIOMA-LIKE HCC
Lymphoepithelioma-like HCC (LEL-HCC) also known as lymphocyte-rich-HCC is an uncommon variant of HCC and comprises < 1% of primary liver cancer[11]. LEL-HCC are associated with lower rates of recurrence after surgery and has an overall favorable survival rate when compared with conventional HCC[50]. LEL-HCC morphologically resembles lymphoepithelioma-like carcinomas, a poorly differentiated epithelial tumor first described in nasopharynx, characterized by a prominent immune stroma/ microenvironment[4]. Subsequently it has been diagnosed in various organs such as stomach, colon, salivary glands, lungs, thymus, uterus, and ovaries[51]. These liver tumors are composed of poorly or undifferentiated neoplastic epithelial cells with a prominent lymphoid infiltrate[52]. A study of 11 cases of LEL-HCC by Wada et al[53] proposed quantitative criteria > 100 tumor infiltrating lymphocytes in 10 high power microscopic filed to define significant lymphocytic infiltration[53]. WHO defines LEL-HCC subtype as the condition in which lymphocytes outnumber pleomorphic neoplastic cells in most microscopic fields, but no clear cutoffs for lymphocyte number has been provided[10]. In contrast to LEL cholangiocarcinoma, which are frequently associated with EBV infection and are well described in literature, LEL-HCC are not associated with EBV infection and are not well characterized in literature[52,54,55]. Grossly, these are well circumscribed tumors with variable encapsulation. Histologically, the tumors are composed of atypical cells with syncytial cytoplasm and nuclei with prominent nucleoli and infiltrated by abundant lymphocytes (Figure 7A). Tumor cells show positivity for markers like Hep-Par 1 and Glypican 3 indicating hepatocellular origin. Immunohistochemical profile of the infiltrating immune cells shows a predominance of cytotoxic CD8+ lymphocytes[52]. Rare molecular studies are available on LEL-HCC. A recent study by Chan et al[56] showed marked focal amplification of chromosome 11q13.3 in LEL-HCC. Calderaro et al[57] showed high level of PD-L1 and programmed cell death 1 expression in intratumoural inflammatory cells in LEL-HCC. These findings indicate LEL-HCC might be sensitive to drugs targeting immune checkpoint inhibitors. No association of LEL-HCC with a transcriptomic subclass has been identified. Immune class of HCC reported by Sia et al[58] characterized by markers of an adaptive T-cell response or exhausted immune response was also not associated with increased number of somatic mutations[58].
Figure 7.
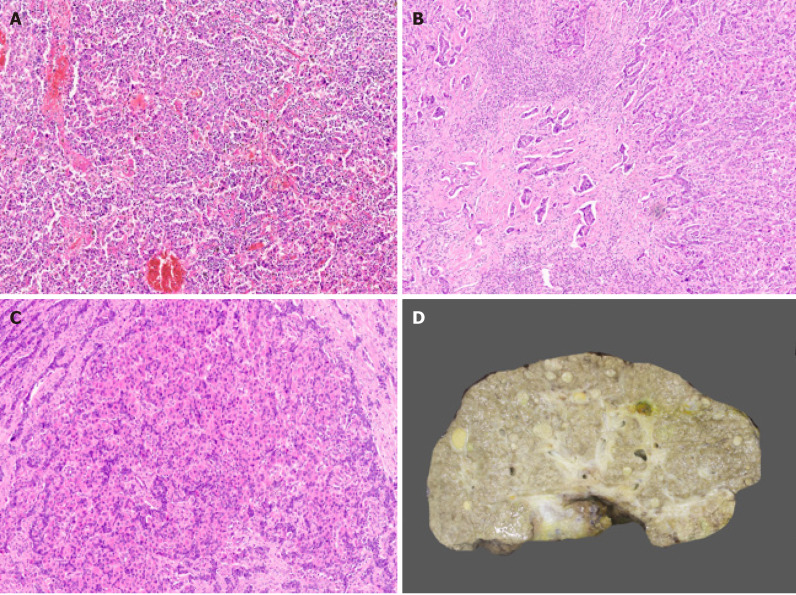
Hepatocellular carcinoma subtypes. A: Lymphoepithelioma like hepatocellular carcinoma (HCC) [hematoxylin and eosin H&E)]; B: Combined hepatocellular-cholangiocarcinoma (cHCC-CCA) with hepatocytic and cholangiocytic component (H&E); C: cHCC-CCA with stem/progenitor cell features (H&E); D: Cirrhotomimetic HCC with numerous tumor nodules.
PROGENITOR HCC
The progenitor subtype of HCC is defined by the immunohistochemical expression of biliary marker CK19, in more than 5% of neoplastic cells[59,60]. Dedifferentiation of malignant hepatocytes or malignant transformation of hepatic progenitor/stem cells may give rise to this histological subtype[4]. There is growing evidence that progenitor cells, activated during acute and CLD, can directly give rise to HCC. This phenotype is associated with mutation in TP53 and particular genomic subclasses (GI-G3, S2) of HCC[21]. CK19 expression is also reported in HCC after transarterial chemoembolization[61].
COMBINED HEPATOCELLULAR-CHOLANGIOCARCINOMA
Combined hepatocellular-cholangiocarcinoma (cHCC-CCA) is a rare primary liver cancer. Diagnosis of cHCC-CCA is challenging because of its pathological heterogeneity, unique molecular alterations, poorly defined radiological features, and non-specific clinical features. The WHO 2010 Classification defined a classical type of cHCC-CCA (tumor containing unequivocal, intimately mixed elements of both HCC and intrahepatic CCA), and 3 subtypes of cHCC-CCA with stem/progenitor cell features: Typical, intermediate cell, and cholangiocellular[62]. The WHO consensus classification published in 2019 removed the 3 different stem/progenitor cell subtypes and defined cHCC-CCA as a primary liver carcinoma with unequivocal presence of both hepatocytic and cholangiocytic differentiation (Figure 7B) within the same tumor[63]. This change was implemented because “stem/progenitor cells” identified as small cells with scant cytoplasm, a high nuclear/cytoplasmic ratio, and hyperchromatic nuclei may potentially be seen in all forms of cHCC-CCA; cholangiocellular carcinoma is not always associated with hepatocellular component and subtyping has no prognostic or clinical relevance.
The hepatocellular and cholangiocarcinoma components in cHCC-CCA may be intimately mixed or lie in separate regions of a tumor. Collision of HCC and iCCA arising separately in the same liver should not be included under cHCC-CCA. The diagnosis of cHCC-CCA should be based on hematoxylin and eosin staining only and immunophenotyping can be performed to confirm histologic components. However, IHC alone should not define the diagnosis of cHCC-CCA[64]. Stem/progenitor cell features (Figure 7C) can be mentioned in the comment section of the histology report. Intermediate cell carcinoma is a unique form of cHCC-CCA comprising of monomorphic tumor cells, smaller than hepatocytes but larger than stem/progenitor cells, and has features intermediate between hepatocytes and cholangiocytes. These malignant cells are arranged in strands or trabeculae in an abundant fibrous stroma. Molecular studies of cHCC-CCA are limited and the earlier reported literature suggested that these tumors have a distinct mutational profile with isocitrate dehydrogenase (IDH) mutations usually observed in intrahepatic CCA[4,65]. However this remains debated as a recent study performed by Joseph et al[66] demonstrated that the genetics of cHCC-CCA classical type, are distinct from intrahepatic CCA but similar to conventional HCC with alteration in telomerase reverse transcriptase (TERT), p53, and cell cycle genes[66]. Few studies have also reported enrichment in stem/progenitor-like signatures, supporting the concept of a stem/progenitor cell origin of cHCC-CCA[67]. cHCC-CCA has a dismal prognosis, worse than that of either HCC or iCCA, and currently, there are no accepted international management guidelines for cHCC-CCA.
RARE AND PROVISIONAL PATHOLOGICAL SUBTYPES OF HCC
These pathological subtypes are rare and provisional because limited published literature is available.
FIBRONODULAR HCC
Fibronodular HCC (FN-HCC) is a recently described candidate variant[68]. FN-HCC histology is characterized by extensive fibrosis dividing a single tumor into multiple well circumscribed distinct nodules with no significant intranodular fibrosis between single or clusters of neoplastic cells[54]. These tumors show well to moderate differentiation with trabecular or solid growth pattern. Scattered pseudoacini are also described. FN-HCC are reported to be more likely to arise in liver with lower fibrosis stage and lower advanced BCLC stage. They have lower rates of tumor progression. Imaging analysis of FN-HCCs revealed higher rates of non-peripheral washout and a new distinct pattern of enhancement which is characterized by the presence of multiple rounded nodules within a lesion embedded in fibrotic-appearing parenchyma, called as ‘popcorn’ appearance of the lesion[68].
CHROMOPHOBE HCC WITH ABRUPT ANAPLASIA
This histological subtype is characterized by a unique set of morphological features: smooth chromophobic cytoplasm which can be either slightly eosinophilc or basophilic, abrupt focal nuclear anaplasia (small tumor cell clusters with marked nuclear anaplasia in a background of tumor cells with bland round nuclei and inconspicuous nucleoli), and scattered microscopic pseudocysts[9,69]. This subtype is associated with distinct molecular features with respect to telomere maintenance resulting in alternative lengthening of telomeres (ALT), which can be detected by telomere FISH. ALT is a telomerase-independent mechanism of telomere maintenance and is found in > 90% of chromophobe HCC with abrupt anaplasia and < 10% of unselected HCCs. Wood et al[69] also investigated somatic mutations of alpha-thalassemia/mental retardation, X-linked, Histone H3, and Death Domain Associated Protein identified in various ALT positive tumors reported at other sites in two cases of chromophobe HCC with abrupt anaplasia; however, no mutations were identified[69-71].
GRANULOCYTE COLONY-STIMULATING FACTOR PRODUCING HCC/NEUTROPHIL-RICH HCC
This rare subtype is characterized by production of granulocyte colony-stimulating factor (G-CSF), leading to diffuse infiltrates by neutrophils[72-74]. There is no clear histological definition for this variant. Morphologically, these tumors are poorly differentiated HCC, usually with areas of sarcomatous differentiation and numerous neutrophils. These generally occur in older individuals, grow rapidly, have a high probability of distant metastases, and the overall prognosis seems to be poor as compared with conventional HCC. The mechanism of the production of G-CSF in HCC remains unclear; a close relationship between G-CSF production in malignant cells and their dedifferentiation has been reported[74].
LIPID-RICH HCC
Lipid-rich HCCs have a foamy cytoplasm resulting from lipid accumulation, with numerous very tiny droplets of fat[75-77]. These can be associated with few larger fat droplets. The differential includes lipid-rich variants of metastatic carcinoma. Immunostaining with Hep-Par 1 and Arginase is helpful in doubtful cases.
CIRRHOTOMIMETIC OR DIFFUSE CIRRHOSIS LIKE-HCC
Cirrhotomimetic (CM) or diffuse cirrhosis like-HCC is a rare variant of liver cancer characterized by small cirrhosis-like tumor nodules that are intimately admixed within the cirrhotic liver parenchyma[78-81]. This tumor pattern is often diagnosed incidentally on the native liver explanted at the time of transplantation or autopsy liver specimen, as most of the times, it is clinically and radiologically undetectable (Figure 7D). These tumors are well to moderately differentiated and majority of patients show no significant elevation in serum AFP values[79]. Pseudoacinar architectural growth pattern with bile production and numerous Mallory-Denk bodies have been demonstrated in these tumors. Few studies have investigated tumor nodules in CM-HCC and suggested that these are synchronous multiclonal HCCs[82,83]. One recent study evaluated the liver explants post transcatheter arterial chemoembolization in CM-HCC and non-CM-HCC and reported lower rates of complete pathologic necrosis and poorer overall survival in CM-HCC after liver transplantation as compared with non-CM-HCCs[84].
CLEAR CELL HCC
Clear cell HCC is an uncommon histological variant of HCC. WHO defines this tumor as the condition when > 80% of the neoplastic cells show clear cell morphology[10]. Glycogen accumulation leads to clearing of the cytoplasm; admixed minor steatosis is also acceptable. These are well to moderately differentiated tumors with similar or better prognosis than conventional HCC[85-87]. There is, however, no distinct definition of this subtype and clear cells may be observed in other subtypes.
HEPATIC CARCINOSARCOMA
Hepatic carcinosarcomas are composed of both malignant epithelial component and mesenchymal components[9]. These neoplasms are extremely rare. The carcinomatous component is moderately to poorly differentiated HCC. The sarcomatous component shows morphologic or immunohistochemical evidence of mesenchymal differentiation, such as leiomyosarcoma, rhabdomyosarcoma, chondrosarcoma, fibrosarcoma, or rarely osteosarcoma. There is scant data on molecular alterations[88,89]. One earlier study revealed mutation in TP53, Phosphatidylinositol-4,5-Bisphosphate 3-Kinase Catalytic Subunit Alpha and FGFR3 genes[88]. One recent study using targeted next-generation sequencing with a panel of 329 cancer-related genes identified TP53, Neurofibrin 1/2 mutations, and VEGFA amplification in both carcinomatous and sarcomatous components[89]. Amplifications involving MET and platelet-derived growth factor receptor A were identified only in the sarcomatous components, whereas mutation affecting ERBB4 and amplifications of Cyclin D1 and FGF 3/4/19 were present only in the carcinomatous components.
MYXOID HCC
This rare morphological subtype of HCC shows well to moderately differentiated neoplastic cells with a trabecular growth pattern, separated by abundant extracellular myxoid/mucin material[9,90]. The neoplastic cells stain strongly with HepPar1 and Arginase-1, and are negative for biliary marker CK19. These tumors typically show loss of liver fatty acid binding protein and also immunostaining with strong and diffuse positivity for GS.
CONCLUSION
Pathology of HCC has evolved significantly in the last two decades. We are now well versed with various dysplastic liver lesions and multiple distinct pathologic subtypes of HCC. There is also remarkable improvement in our understanding of HCC pathogenesis as tumor genome sequencing has identified recurrent molecular alterations and oncogenic pathways and how this correlates with various morphological findings. Identification of genetic alterations also gives us an opportunity to develop targeted therapies that can prevent recurrence and improve patient survival.
Footnotes
Conflict-of-interest statement: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Manuscript source: Invited manuscript
Peer-review started: December 20, 2020
First decision: January 7, 2021
Article in press: March 31, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Niu ZS, Wang C S-Editor: Fan JR L-Editor: A P-Editor: Xing YX
Contributor Information
Mukul Vij, Department ofPathology, Dr Rela Institute and Medical Center, Chennai 600044, Tamil Nadu, India. mukul.vij.path@gmail.com.
Julien Calderaro, Department of Pathology, Groupe Hospitalier Henri Mondor, Creteil F-94010, France.
References
- 1.GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392:1736–1788. doi: 10.1016/S0140-6736(18)32203-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Villanueva A. Hepatocellular Carcinoma. N Engl J Med. 2019;380:1450–1462. doi: 10.1056/NEJMra1713263. [DOI] [PubMed] [Google Scholar]
- 4.Calderaro J, Ziol M, Paradis V, Zucman-Rossi J. Molecular and histological correlations in liver cancer. J Hepatol. 2019;71:616–630. doi: 10.1016/j.jhep.2019.06.001. [DOI] [PubMed] [Google Scholar]
- 5.International Consensus Group for Hepatocellular NeoplasiaThe International Consensus Group for Hepatocellular Neoplasia. Pathologic diagnosis of early hepatocellular carcinoma: a report of the international consensus group for hepatocellular neoplasia. Hepatology. 2009;49:658–664. doi: 10.1002/hep.22709. [DOI] [PubMed] [Google Scholar]
- 6.Park YN. Update on precursor and early lesions of hepatocellular carcinomas. Arch Pathol Lab Med. 2011;135:704–715. doi: 10.5858/2010-0524-RA.1. [DOI] [PubMed] [Google Scholar]
- 7.Niu ZS, Niu XJ, Wang WH, Zhao J. Latest developments in precancerous lesions of hepatocellular carcinoma. World J Gastroenterol. 2016;22:3305–3314. doi: 10.3748/wjg.v22.i12.3305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nzeako UC, Goodman ZD, Ishak KG. Comparison of tumor pathology with duration of survival of North American patients with hepatocellular carcinoma. Cancer. 1995;76:579–588. doi: 10.1002/1097-0142(19950815)76:4<579::aid-cncr2820760407>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 9.Torbenson MS. Morphologic Subtypes of Hepatocellular Carcinoma. Gastroenterol Clin North Am. 2017;46:365–391. doi: 10.1016/j.gtc.2017.01.009. [DOI] [PubMed] [Google Scholar]
- 10.Torbenson MS, Ng IOL, Park YN, Roncalli M, Sakamoto M. Hepatocellular carcinoma. In: WHO Classification of Tumors Editorial Board. Digestive system tumours. WHO classification of tumours series. 5th ed. Lyon: International Agency for Research on Cancer, 2019: 229-239. [Google Scholar]
- 11.Ferrell LD, Kakar S, Terracciano LM, Wee A. Tumors and tumor like lesions of the liver. In: Burt AD, Ferrell LD, Hubscher SG. MacSween’s Pathology of the Liver. 7th ed. Philadelphia: Elsevier, 2018: 780-879. [Google Scholar]
- 12.Lauwers GY, Terris B, Balis UJ, Batts KP, Regimbeau JM, Chang Y, Graeme-Cook F, Yamabe H, Ikai I, Cleary KR, Fujita S, Flejou JF, Zukerberg LR, Nagorney DM, Belghiti J, Yamaoka Y, Vauthey JN International Cooperative Study Group on Hepatocellular Carcinoma. Prognostic histologic indicators of curatively resected hepatocellular carcinomas: a multi-institutional analysis of 425 patients with definition of a histologic prognostic index. Am J Surg Pathol. 2002;26:25–34. doi: 10.1097/00000478-200201000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Duffy JP, Vardanian A, Benjamin E, Watson M, Farmer DG, Ghobrial RM, Lipshutz G, Yersiz H, Lu DS, Lassman C, Tong MJ, Hiatt JR, Busuttil RW. Liver transplantation criteria for hepatocellular carcinoma should be expanded: a 22-year experience with 467 patients at UCLA. Ann Surg. 2007;246:502–9; discussion 509. doi: 10.1097/SLA.0b013e318148c704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.DuBay D, Sandroussi C, Sandhu L, Cleary S, Guba M, Cattral MS, McGilvray I, Ghanekar A, Selzner M, Greig PD, Grant DR. Liver transplantation for advanced hepatocellular carcinoma using poor tumor differentiation on biopsy as an exclusion criterion. Ann Surg. 2011;253:166–172. doi: 10.1097/sla.0b013e31820508f1. [DOI] [PubMed] [Google Scholar]
- 15.Yan BC, Gong C, Song J, Krausz T, Tretiakova M, Hyjek E, Al-Ahmadie H, Alves V, Xiao SY, Anders RA, Hart JA. Arginase-1: a new immunohistochemical marker of hepatocytes and hepatocellular neoplasms. Am J Surg Pathol. 2010;34:1147–1154. doi: 10.1097/PAS.0b013e3181e5dffa. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Choi WT, Kakar S. Immunohistochemistry in the Diagnosis of Hepatocellular Carcinoma. Gastroenterol Clin North Am. 2017;46:311–325. doi: 10.1016/j.gtc.2017.01.006. [DOI] [PubMed] [Google Scholar]
- 17.Nguyen T, Phillips D, Jain D, Torbenson M, Wu TT, Yeh MM, Kakar S. Comparison of 5 Immunohistochemical Markers of Hepatocellular Differentiation for the Diagnosis of Hepatocellular Carcinoma. Arch Pathol Lab Med. 2015;139:1028–1034. doi: 10.5858/arpa.2014-0479-OA. [DOI] [PubMed] [Google Scholar]
- 18.Libbrecht L, Severi T, Cassiman D, Vander Borght S, Pirenne J, Nevens F, Verslype C, van Pelt J, Roskams T. Glypican-3 expression distinguishes small hepatocellular carcinomas from cirrhosis, dysplastic nodules, and focal nodular hyperplasia-like nodules. Am J Surg Pathol. 2006;30:1405–1411. doi: 10.1097/01.pas.0000213323.97294.9a. [DOI] [PubMed] [Google Scholar]
- 19.Kakar S, Muir T, Murphy LM, Lloyd RV, Burgart LJ. Immunoreactivity of Hep Par 1 in hepatic and extrahepatic tumors and its correlation with albumin in situ hybridization in hepatocellular carcinoma. Am J Clin Pathol. 2003;119:361–366. doi: 10.1309/8l872rphejrkf5jj. [DOI] [PubMed] [Google Scholar]
- 20.Nasir A, Lehrke HD, Mounajjed T, Said S, Zhang L, Yasir S, Shah SS, Chandan VS, Smyrk TC, Moreira RK, Boland Froemming JM, Herrera Hernandez LP, Wu TT, Graham RP. Albumin In Situ Hybridization Can Be Positive in Adenocarcinomas and Other Tumors From Diverse Sites. Am J Clin Pathol. 2019;152:190–199. doi: 10.1093/ajcp/aqz032. [DOI] [PubMed] [Google Scholar]
- 21.Calderaro J, Couchy G, Imbeaud S, Amaddeo G, Letouzé E, Blanc JF, Laurent C, Hajji Y, Azoulay D, Bioulac-Sage P, Nault JC, Zucman-Rossi J. Histological subtypes of hepatocellular carcinoma are related to gene mutations and molecular tumour classification. J Hepatol. 2017;67:727–738. doi: 10.1016/j.jhep.2017.05.014. [DOI] [PubMed] [Google Scholar]
- 22.Ziol M, Poté N, Amaddeo G, Laurent A, Nault JC, Oberti F, Costentin C, Michalak S, Bouattour M, Francoz C, Pageaux GP, Ramos J, Decaens T, Luciani A, Guiu B, Vilgrain V, Aubé C, Derman J, Charpy C, Zucman-Rossi J, Barget N, Seror O, Ganne-Carrié N, Paradis V, Calderaro J. Macrotrabecular-massive hepatocellular carcinoma: A distinctive histological subtype with clinical relevance. Hepatology. 2018;68:103–112. doi: 10.1002/hep.29762. [DOI] [PubMed] [Google Scholar]
- 23.Yoneda N, Matsui O, Kobayashi S, Kitao A, Kozaka K, Inoue D, Yoshida K, Minami T, Koda W, Gabata T. Current status of imaging biomarkers predicting the biological nature of hepatocellular carcinoma. Jpn J Radiol. 2019;37:191–208. doi: 10.1007/s11604-019-00817-3. [DOI] [PubMed] [Google Scholar]
- 24.Mulé S, Galletto Pregliasco A, Tenenhaus A, Kharrat R, Amaddeo G, Baranes L, Laurent A, Regnault H, Sommacale D, Djabbari M, Pigneur F, Tacher V, Kobeiter H, Calderaro J, Luciani A. Multiphase Liver MRI for Identifying the Macrotrabecular-Massive Subtype of Hepatocellular Carcinoma. Radiology. 2020;295:562–571. doi: 10.1148/radiol.2020192230. [DOI] [PubMed] [Google Scholar]
- 25.Rhee H, Cho ES, Nahm JH, Jang M, Chung YE, Baek SE, Lee S, Kim MJ, Park MS, Han DH, Choi JY, Park YN. Gadoxetic acid-enhanced MRI of macrotrabecular-massive hepatocellular carcinoma and its prognostic implications. J Hepatol. 2021;74:109–121. doi: 10.1016/j.jhep.2020.08.013. [DOI] [PubMed] [Google Scholar]
- 26.Liu LL, Zhang SW, Chao X, Wang CH, Yang X, Zhang XK, Wen YL, Yun JP, Luo RZ. Coexpression of CMTM6 and PD-L1 as a predictor of poor prognosis in macrotrabecular-massive hepatocellular carcinoma. Cancer Immunol Immunother. 2021;70:417–429. doi: 10.1007/s00262-020-02691-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Calderaro J, Meunier L, Nguyen CT, Boubaya M, Caruso S, Luciani A, Amaddeo G, Regnault H, Nault JC, Cohen J, Oberti F, Michalak S, Bouattour M, Vilgrain V, Pageaux GP, Ramos J, Barget N, Guiu B, Paradis V, Aubé C, Laurent A, Pawlotsky JM, Ganne-Carrié N, Zucman-Rossi J, Seror O, Ziol M. ESM1 as a Marker of Macrotrabecular-Massive Hepatocellular Carcinoma. Clin Cancer Res. 2019;25:5859–5865. doi: 10.1158/1078-0432.CCR-19-0859. [DOI] [PubMed] [Google Scholar]
- 28.Salomao M, Yu WM, Brown RS Jr, Emond JC, Lefkowitch JH. Steatohepatitic hepatocellular carcinoma (SH-HCC): a distinctive histological variant of HCC in hepatitis C virus-related cirrhosis with associated NAFLD/NASH. Am J Surg Pathol. 2010;34:1630–1636. doi: 10.1097/PAS.0b013e3181f31caa. [DOI] [PubMed] [Google Scholar]
- 29.Salomao M, Remotti H, Vaughan R, Siegel AB, Lefkowitch JH, Moreira RK. The steatohepatitic variant of hepatocellular carcinoma and its association with underlying steatohepatitis. Hum Pathol. 2012;43:737–746. doi: 10.1016/j.humpath.2011.07.005. [DOI] [PubMed] [Google Scholar]
- 30.Van Treeck BJ, Mounajjed T, Moreira RK, Orujov M, Allende DS, Bellizzi AM, Lagana SM, Davila JI, Jessen E, Graham RP. Transcriptomic and Proteomic Analysis of Steatohepatitic Hepatocellular Carcinoma Reveals Novel Distinct Biologic Features. Am J Clin Pathol. 2021;155:87–96. doi: 10.1093/ajcp/aqaa114. [DOI] [PubMed] [Google Scholar]
- 31.Fujiwara N, Nakagawa H, Enooku K, Kudo Y, Hayata Y, Nakatsuka T, Tanaka Y, Tateishi R, Hikiba Y, Misumi K, Tanaka M, Hayashi A, Shibahara J, Fukayama M, Arita J, Hasegawa K, Hirschfield H, Hoshida Y, Hirata Y, Otsuka M, Tateishi K, Koike K. CPT2 downregulation adapts HCC to lipid-rich environment and promotes carcinogenesis via acylcarnitine accumulation in obesity. Gut. 2018;67:1493–1504. doi: 10.1136/gutjnl-2017-315193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee JS, Yoo JE, Kim H, Rhee H, Koh MJ, Nahm JH, Choi JS, Lee KH, Park YN. Tumor stroma with senescence-associated secretory phenotype in steatohepatitic hepatocellular carcinoma. PLoS One. 2017;12:e0171922. doi: 10.1371/journal.pone.0171922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Theise ND, Curado MP, Franceschi S. Hepatocellular carcinoma. In: Bosman E, Carneiro F, Hruban RH, Theise ND. WHO Classification of Tumors of the Digestive System. 4th ed. Lyon, France: International Agency for Research on Cancer, 2010: 205-216. [Google Scholar]
- 34.Krings G, Ramachandran R, Jain D, Wu TT, Yeh MM, Torbenson M, Kakar S. Immunohistochemical pitfalls and the importance of glypican 3 and arginase in the diagnosis of scirrhous hepatocellular carcinoma. Mod Pathol. 2013;26:782–791. doi: 10.1038/modpathol.2012.243. [DOI] [PubMed] [Google Scholar]
- 35.Hatano M, Ojima H, Masugi Y, Tsujikawa H, Hiraoka N, Kanai Y, Shimada K, Shinoda M, Sakamoto M. Steatotic and nonsteatotic scirrhous hepatocellular carcinomas reveal distinct clinicopathological features. Hum Pathol. 2019;86:222–232. doi: 10.1016/j.humpath.2018.11.024. [DOI] [PubMed] [Google Scholar]
- 36.El Jabbour T, Lagana SM, Lee H. Update on hepatocellular carcinoma: Pathologists' review. World J Gastroenterol. 2019;25:1653–1665. doi: 10.3748/wjg.v25.i14.1653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Honeyman JN, Simon EP, Robine N, Chiaroni-Clarke R, Darcy DG, Lim II, Gleason CE, Murphy JM, Rosenberg BR, Teegan L, Takacs CN, Botero S, Belote R, Germer S, Emde AK, Vacic V, Bhanot U, LaQuaglia MP, Simon SM. Detection of a recurrent DNAJB1-PRKACA chimeric transcript in fibrolamellar hepatocellular carcinoma. Science. 2014;343:1010–1014. doi: 10.1126/science.1249484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kim SH, Lim HK, Lee WJ, Choi D, Park CK. Scirrhous hepatocellular carcinoma: comparison with usual hepatocellular carcinoma based on CT-pathologic features and long-term results after curative resection. Eur J Radiol. 2009;69:123–130. doi: 10.1016/j.ejrad.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 39.Seok JY, Na DC, Woo HG, Roncalli M, Kwon SM, Yoo JE, Ahn EY, Kim GI, Choi JS, Kim YB, Park YN. A fibrous stromal component in hepatocellular carcinoma reveals a cholangiocarcinoma-like gene expression trait and epithelial-mesenchymal transition. Hepatology. 2012;55:1776–1786. doi: 10.1002/hep.25570. [DOI] [PubMed] [Google Scholar]
- 40.Graham RP, Torbenson MS. Fibrolamellar carcinoma: A histologically unique tumor with unique molecular findings. Semin Diagn Pathol. 2017;34:146–152. doi: 10.1053/j.semdp.2016.12.010. [DOI] [PubMed] [Google Scholar]
- 41.El-Serag HB, Davila JA. Is fibrolamellar carcinoma different from hepatocellular carcinoma? Hepatology. 2004;39:798–803. doi: 10.1002/hep.20096. [DOI] [PubMed] [Google Scholar]
- 42.Graham RP. Fibrolamellar Carcinoma: What Is New and Why It Matters. Surg Pathol Clin. 2018;11:377–387. doi: 10.1016/j.path.2018.02.006. [DOI] [PubMed] [Google Scholar]
- 43.Ichikawa T, Federle MP, Grazioli L, Madariaga J, Nalesnik M, Marsh W. Fibrolamellar hepatocellular carcinoma: imaging and pathologic findings in 31 recent cases. Radiology. 1999;213:352–361. doi: 10.1148/radiology.213.2.r99nv31352. [DOI] [PubMed] [Google Scholar]
- 44.Fink AL. Chaperone-mediated protein folding. Physiol Rev. 1999;79:425–449. doi: 10.1152/physrev.1999.79.2.425. [DOI] [PubMed] [Google Scholar]
- 45.Hattori H, Liu YC, Tohnai I, Ueda M, Kaneda T, Kobayashi T, Tanabe K, Ohtsuka K. Intracellular localization and partial amino acid sequence of a stress-inducible 40-kDa protein in HeLa cells. Cell Struct Funct. 1992;17:77–86. doi: 10.1247/csf.17.77. [DOI] [PubMed] [Google Scholar]
- 46.Taskén K, Solberg R, Zhao Y, Hansson V, Jahnsen T, Siciliano MJ. The gene encoding the catalytic subunit C alpha of cAMP-dependent protein kinase (locus PRKACA) localizes to human chromosome region 19p13.1. Genomics. 1996;36:535–538. doi: 10.1006/geno.1996.0501. [DOI] [PubMed] [Google Scholar]
- 47.Vyas M, Hechtman JF, Zhang Y, Benayed R, Yavas A, Askan G, Shia J, Klimstra DS, Basturk O. DNAJB1-PRKACA fusions occur in oncocytic pancreatic and biliary neoplasms and are not specific for fibrolamellar hepatocellular carcinoma. Mod Pathol. 2020;33:648–656. doi: 10.1038/s41379-019-0398-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Singhi AD, Wood LD, Parks E, Torbenson MS, Felsenstein M, Hruban RH, Nikiforova MN, Wald AI, Kaya C, Nikiforov YE, Favazza L, He J, McGrath K, Fasanella KE, Brand RE, Lennon AM, Furlan A, Dasyam AK, Zureikat AH, Zeh HJ, Lee K, Bartlett DL, Slivka A. Recurrent Rearrangements in PRKACA and PRKACB in Intraductal Oncocytic Papillary Neoplasms of the Pancreas and Bile Duct. Gastroenterology 2020; 158: 573-582. :e2. doi: 10.1053/j.gastro.2019.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Malouf GG, Job S, Paradis V, Fabre M, Brugières L, Saintigny P, Vescovo L, Belghiti J, Branchereau S, Faivre S, de Reyniès A, Raymond E. Transcriptional profiling of pure fibrolamellar hepatocellular carcinoma reveals an endocrine signature. Hepatology. 2014;59:2228–2237. doi: 10.1002/hep.27018. [DOI] [PubMed] [Google Scholar]
- 50.Chan AW, Tong JH, Pan Y, Chan SL, Wong GL, Wong VW, Lai PB, To KF. Lymphoepithelioma-like hepatocellular carcinoma: an uncommon variant of hepatocellular carcinoma with favorable outcome. Am J Surg Pathol. 2015;39:304–312. doi: 10.1097/PAS.0000000000000376. [DOI] [PubMed] [Google Scholar]
- 51.Wang JK, Jin YW, Hu HJ, Regmi P, Ma WJ, Yang Q, Liu F, Ran CD, Su F, Zheng EL, Li FY. Lymphoepithelioma-like hepatocellular carcinoma: A case report and brief review of literature. Medicine (Baltimore) 2017;96:e9416. doi: 10.1097/MD.0000000000009416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Labgaa I, Stueck A, Ward SC. Lymphoepithelioma-Like Carcinoma in Liver. Am J Pathol. 2017;187:1438–1444. doi: 10.1016/j.ajpath.2017.02.022. [DOI] [PubMed] [Google Scholar]
- 53.Wada Y, Nakashima O, Kutami R, Yamamoto O, Kojiro M. Clinicopathological study on hepatocellular carcinoma with lymphocytic infiltration. Hepatology. 1998;27:407–414. doi: 10.1002/hep.510270214. [DOI] [PubMed] [Google Scholar]
- 54.Vyas M, Zhang X. Hepatocellular Carcinoma: Role of Pathology in the Era of Precision Medicine. Clin Liver Dis. 2020;24:591–610. doi: 10.1016/j.cld.2020.07.010. [DOI] [PubMed] [Google Scholar]
- 55.Zhang K, Tao C, Tao Z, Wu F, An S, Wu J, Rong W. Lymphoepithelioma-like carcinoma in liver not associated with Epstein-Barr virus: a report of 3 cases and literature review. Diagn Pathol. 2020;15:115. doi: 10.1186/s13000-020-01035-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chan AW, Zhang Z, Chong CC, Tin EK, Chow C, Wong N. Genomic landscape of lymphoepithelioma-like hepatocellular carcinoma. J Pathol. 2019;249:166–172. doi: 10.1002/path.5313. [DOI] [PubMed] [Google Scholar]
- 57.Calderaro J, Rousseau B, Amaddeo G, Mercey M, Charpy C, Costentin C, Luciani A, Zafrani ES, Laurent A, Azoulay D, Lafdil F, Pawlotsky JM. Programmed death ligand 1 expression in hepatocellular carcinoma: Relationship With clinical and pathological features. Hepatology. 2016;64:2038–2046. doi: 10.1002/hep.28710. [DOI] [PubMed] [Google Scholar]
- 58.Sia D, Jiao Y, Martinez-Quetglas I, Kuchuk O, Villacorta-Martin C, Castro de Moura M, Putra J, Camprecios G, Bassaganyas L, Akers N, Losic B, Waxman S, Thung SN, Mazzaferro V, Esteller M, Friedman SL, Schwartz M, Villanueva A, Llovet JM. Identification of an Immune-specific Class of Hepatocellular Carcinoma, Based on Molecular Features. Gastroenterology. 2017;153:812–826. doi: 10.1053/j.gastro.2017.06.007. [DOI] [PubMed] [Google Scholar]
- 59.Durnez A, Verslype C, Nevens F, Fevery J, Aerts R, Pirenne J, Lesaffre E, Libbrecht L, Desmet V, Roskams T. The clinicopathological and prognostic relevance of cytokeratin 7 and 19 expression in hepatocellular carcinoma. A possible progenitor cell origin. Histopathology. 2006;49:138–151. doi: 10.1111/j.1365-2559.2006.02468.x. [DOI] [PubMed] [Google Scholar]
- 60.Lee JS, Heo J, Libbrecht L, Chu IS, Kaposi-Novak P, Calvisi DF, Mikaelyan A, Roberts LR, Demetris AJ, Sun Z, Nevens F, Roskams T, Thorgeirsson SS. A novel prognostic subtype of human hepatocellular carcinoma derived from hepatic progenitor cells. Nat Med. 2006;12:410–416. doi: 10.1038/nm1377. [DOI] [PubMed] [Google Scholar]
- 61.Lai JP, Conley A, Knudsen BS, Guindi M. Hypoxia after transarterial chemoembolization may trigger a progenitor cell phenotype in hepatocellular carcinoma. Histopathology. 2015;67:442–450. doi: 10.1111/his.12623. [DOI] [PubMed] [Google Scholar]
- 62.Akiba J, Nakashima O, Hattori S, Tanikawa K, Takenaka M, Nakayama M, Kondo R, Nomura Y, Koura K, Ueda K, Sanada S, Naito Y, Yamaguchi R, Yano H. Clinicopathologic analysis of combined hepatocellular-cholangiocarcinoma according to the latest WHO classification. Am J Surg Pathol. 2013;37:496–505. doi: 10.1097/PAS.0b013e31827332b0. [DOI] [PubMed] [Google Scholar]
- 63.Sempoux C, Kakar S, Kondo F, Schirmacher P. Combined Hepatocellular- Cholangiocarcinoma and Undifferentiated Primary Liver Carcinoma. In: WHO Classification of Tumours Editorial Board. Digestive system tumours. WHO classification of tumours series. 5th ed. Lyon: International Agency for Research on Cancer, 2019: 260-262. [Google Scholar]
- 64.Brunt E, Aishima S, Clavien PA, Fowler K, Goodman Z, Gores G, Gouw A, Kagen A, Klimstra D, Komuta M, Kondo F, Miksad R, Nakano M, Nakanuma Y, Ng I, Paradis V, Nyun Park Y, Quaglia A, Roncalli M, Roskams T, Sakamoto M, Saxena R, Sempoux C, Sirlin C, Stueck A, Thung S, Tsui WMS, Wang XW, Wee A, Yano H, Yeh M, Zen Y, Zucman-Rossi J, Theise N. cHCC-CCA: Consensus terminology for primary liver carcinomas with both hepatocytic and cholangiocytic differentation. Hepatology. 2018;68:113–126. doi: 10.1002/hep.29789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Komuta M, Yeh MM. A Review on the Update of Combined Hepatocellular Cholangiocarcinoma. Semin Liver Dis. 2020;40:124–130. doi: 10.1055/s-0039-3402515. [DOI] [PubMed] [Google Scholar]
- 66.Joseph NM, Tsokos CG, Umetsu SE, Shain AH, Kelley RK, Onodera C, Bowman S, Talevich E, Ferrell LD, Kakar S, Krings G. Genomic profiling of combined hepatocellular-cholangiocarcinoma reveals similar genetics to hepatocellular carcinoma. J Pathol. 2019;248:164–178. doi: 10.1002/path.5243. [DOI] [PubMed] [Google Scholar]
- 67.Wang A, Wu L, Lin J, Han L, Bian J, Wu Y, Robson SC, Xue L, Ge Y, Sang X, Wang W, Zhao H. Whole-exome sequencing reveals the origin and evolution of hepato-cholangiocarcinoma. Nat Commun. 2018;9:894. doi: 10.1038/s41467-018-03276-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tefera J, Revzin M, Chapiro J, Savic LJ, Mulligan D, Batra R, Taddei T, Jain D, Zhang X. Fibronodular hepatocellular carcinoma-a new variant of liver cancer: clinical, pathological and radiological correlation. J Clin Pathol. 2021;74:31–35. doi: 10.1136/jclinpath-2020-206574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wood LD, Heaphy CM, Daniel HD, Naini BV, Lassman CR, Arroyo MR, Kamel IR, Cosgrove DP, Boitnott JK, Meeker AK, Torbenson MS. Chromophobe hepatocellular carcinoma with abrupt anaplasia: a proposal for a new subtype of hepatocellular carcinoma with unique morphological and molecular features. Mod Pathol. 2013;26:1586–1593. doi: 10.1038/modpathol.2013.68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Heaphy CM, de Wilde RF, Jiao Y, Klein AP, Edil BH, Shi C, Bettegowda C, Rodriguez FJ, Eberhart CG, Hebbar S, Offerhaus GJ, McLendon R, Rasheed BA, He Y, Yan H, Bigner DD, Oba-Shinjo SM, Marie SK, Riggins GJ, Kinzler KW, Vogelstein B, Hruban RH, Maitra A, Papadopoulos N, Meeker AK. Altered telomeres in tumors with ATRX and DAXX mutations. Science. 2011;333:425. doi: 10.1126/science.1207313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schwartzentruber J, Korshunov A, Liu XY, Jones DT, Pfaff E, Jacob K, Sturm D, Fontebasso AM, Quang DA, Tönjes M, Hovestadt V, Albrecht S, Kool M, Nantel A, Konermann C, Lindroth A, Jäger N, Rausch T, Ryzhova M, Korbel JO, Hielscher T, Hauser P, Garami M, Klekner A, Bognar L, Ebinger M, Schuhmann MU, Scheurlen W, Pekrun A, Frühwald MC, Roggendorf W, Kramm C, Dürken M, Atkinson J, Lepage P, Montpetit A, Zakrzewska M, Zakrzewski K, Liberski PP, Dong Z, Siegel P, Kulozik AE, Zapatka M, Guha A, Malkin D, Felsberg J, Reifenberger G, von Deimling A, Ichimura K, Collins VP, Witt H, Milde T, Witt O, Zhang C, Castelo-Branco P, Lichter P, Faury D, Tabori U, Plass C, Majewski J, Pfister SM, Jabado N. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature. 2012;482:226–231. doi: 10.1038/nature10833. [DOI] [PubMed] [Google Scholar]
- 72.Araki K, Kishihara F, Takahashi K, Matsumata T, Shimura T, Suehiro T, Kuwano H. Hepatocellular carcinoma producing a granulocyte colony-stimulating factor: report of a resected case with a literature review. Liver Int. 2007;27:716–721. doi: 10.1111/j.1478-3231.2007.01468.x. [DOI] [PubMed] [Google Scholar]
- 73.Kohno M, Shirabe K, Mano Y, Muto J, Motomura T, Takeishi K, Toshima T, Yoshimatsu M, Ijichi H, Harada N, Aishima S, Uchiyama H, Yoshizumi T, Taketomi A, Maehara Y. Granulocyte colony-stimulating-factor-producing hepatocellular carcinoma with extensive sarcomatous changes: report of a case. Surg Today. 2013;43:439–445. doi: 10.1007/s00595-012-0202-0. [DOI] [PubMed] [Google Scholar]
- 74.Nomura T, Morishita A, Tani J, Takuma K, Nakahara M, Oura K, Tadokoro T, Kobayashi K, Fujita K, Mimura S, Kobara H, Tsutsui K, Sanomura T, Nishiyama Y, Ibuki E, Haba R, Sakamoto T, Yoneyama H, Himoto T, Masaki T. A case report of granulocyte colony-stimulating factor-producing hepatocellular carcinoma that recurred after long-term complete response. Clin J Gastroenterol. 2021;14:204–211. doi: 10.1007/s12328-020-01239-9. [DOI] [PubMed] [Google Scholar]
- 75.Orikasa H, Ohyama R, Tsuka N, Eyden BP, Yamazaki K. Lipid-rich clear-cell hepatocellular carcinoma arising in non-alcoholic steatohepatitis in a patient with diabetes mellitus. J Submicrosc Cytol Pathol. 2001;33:195–200. [PubMed] [Google Scholar]
- 76.Dunn R, Zhang W, Lai J, Litton T, Zhou Y, Lai JP. Foamy Histiocyte-Like Hepatocellular Carcinoma (HCC): A New Variant of HCC? Ann Hepatol. 2017;16:304–307. doi: 10.5604/16652681.1231591. [DOI] [PubMed] [Google Scholar]
- 77.Noro T, Gotohda N, Kojima M, Konishi M, Nakaghori T, Takahashi S, Hasebe T, Kinoshita T. Hepatocellular carcinoma with foamy histiocyte-like appearance: a deceptively clear cell carcinoma appearing variant. Case Rep Gastroenterol. 2010;4:286–292. doi: 10.1159/000319545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Clayton EF, Malik S, Bonnel A, Mu Y, Olthoff K, Shaked A, Abt PL, Peterman H, Rajender Reddy K, Ottmann S, Furth EE, Levine MH. Liver transplantation and cirrhotomimetic hepatocellular carcinoma: classification and outcomes. Liver Transpl. 2014;20:765–774. doi: 10.1002/lt.23876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Han YS, Choi DL, Park JB. Cirrhotomimetic type hepatocellular carcinoma diagnosed after liver transplantation--eighteen months of follow-up: a case report. Transplant Proc. 2008;40:2835–2836. doi: 10.1016/j.transproceed.2008.07.012. [DOI] [PubMed] [Google Scholar]
- 80.Jakate S, Yabes A, Giusto D, Naini B, Lassman C, Yeh MM, Ferrell LD. Diffuse cirrhosis-like hepatocellular carcinoma: a clinically and radiographically undetected variant mimicking cirrhosis. Am J Surg Pathol. 2010;34:935–941. doi: 10.1097/PAS.0b013e3181ddf52f. [DOI] [PubMed] [Google Scholar]
- 81.Agni RM. Diagnostic histopathology of hepatocellular carcinoma: A case-based review. Semin Diagn Pathol. 2017;34:126–137. doi: 10.1053/j.semdp.2016.12.008. [DOI] [PubMed] [Google Scholar]
- 82.Morimoto O, Nagano H, Sakon M, Fujiwara Y, Yamada T, Nakagawa H, Miyamoto A, Kondo M, Arai I, Yamamoto T, Ota H, Dono K, Umeshita K, Nakamori S, Sasaki Y, Ishikawa O, Imaoka S, Monden M. Diagnosis of intrahepatic metastasis and multicentric carcinogenesis by microsatellite loss of heterozygosity in patients with multiple and recurrent hepatocellular carcinomas. J Hepatol. 2003;39:215–221. doi: 10.1016/s0168-8278(03)00233-2. [DOI] [PubMed] [Google Scholar]
- 83.Ng IO, Guan XY, Poon RT, Fan ST, Lee JM. Determination of the molecular relationship between multiple tumour nodules in hepatocellular carcinoma differentiates multicentric origin from intrahepatic metastasis. J Pathol. 2003;199:345–353. doi: 10.1002/path.1287. [DOI] [PubMed] [Google Scholar]
- 84.Habibollahi P, Shamchi SP, Tondon R, Ecker BL, Gade TP, Hunt S, Soulen MC, Furth EE, Levine MH, Nadolski G. Combination of Neoadjuvant Transcatheter Arterial Chemoembolization and Orthotopic Liver Transplantation for the Treatment of Cirrhotomimetic Hepatocellular Carcinoma. J Vasc Interv Radiol. 2018;29:237–243. doi: 10.1016/j.jvir.2017.09.008. [DOI] [PubMed] [Google Scholar]
- 85.Liu Z, Ma W, Li H, Li Q. Clinicopathological and prognostic features of primary clear cell carcinoma of the liver. Hepatol Res. 2008;38:291–299. doi: 10.1111/j.1872-034X.2007.00264.x. [DOI] [PubMed] [Google Scholar]
- 86.Ji SP, Li Q, Dong H. Therapy and prognostic features of primary clear cell carcinoma of the liver. World J Gastroenterol. 2010;16:764–769. doi: 10.3748/wjg.v16.i6.764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Yang SH, Watanabe J, Nakashima O, Kojiro M. Clinicopathologic study on clear cell hepatocellular carcinoma. Pathol Int. 1996;46:503–509. doi: 10.1111/j.1440-1827.1996.tb03645.x. [DOI] [PubMed] [Google Scholar]
- 88.Luchini C, Capelli P, Fassan M, Simbolo M, Mafficini A, Pedica F, Ruzzenente A, Guglielmi A, Corbo V, Scarpa A. Next-generation histopathologic diagnosis: a lesson from a hepatic carcinosarcoma. J Clin Oncol. 2014;32:e63–e66. doi: 10.1200/JCO.2012.47.5855. [DOI] [PubMed] [Google Scholar]
- 89.Zhang X, Bai Q, Xu Y, Wang W, Chen L, Han J, Zhu H, Zhang Z, Hou Y, Zhou J, Zhou Y, Ji Y. Molecular profiling of the biphasic components of hepatic carcinosarcoma by the use of targeted next-generation sequencing. Histopathology. 2019;74:944–958. doi: 10.1111/his.13822. [DOI] [PubMed] [Google Scholar]
- 90.Salaria SN, Graham RP, Aishima S, Mounajjed T, Yeh MM, Torbenson MS. Primary hepatic tumors with myxoid change: morphologically unique hepatic adenomas and hepatocellular carcinomas. Am J Surg Pathol. 2015;39:318–324. doi: 10.1097/PAS.0000000000000382. [DOI] [PubMed] [Google Scholar]