Abstract
Recognition of self-peptides in association with distinct HLA class II alleles by autoreactive CD4+ T cells is central for loss of immunological tolerance leading to autoimmune disease. However, identifying immunodominant self-peptides and characterizing autoreactive T cells is challenging. In this issue of the JCI, Falta et al. identify a disease-associated complementarity-determining region 3β motif specific for beryllium-modified C-C motif ligand 4 (CCL4) and CCL3 self-peptides in patients with chronic beryllium disease (CBD), a granulomatous lung disorder with a known HLA class II allelic association. Detection of these antigen-specific CD4+ T cells by beryllium-pulsed HLA-DP2 tetramers presenting CCL4/CCL3 confirms these autoantigens in humans and mice and enables monitoring in the progress of disease. Detection of autoreactive CD4+ T cells by peptide–MHC class II multimers allows for the detailed characterization of disease-promoting T cells. This knowledge has profound implications for the monitoring and development of targeted therapies in human autoimmune disorders.
Identifying T cell responses against innate chemokines
Etiological factors that facilitate the development of autoimmune diseases are poorly characterized. In this issue of the JCI, Falta et al. provide evidence that occupational exposure to the rare alkaline earth metal beryllium represents a risk for development of chronic beryllium disease (CBD) of the lung (1). People that carry distinct HLA-DP2 alleles are especially at risk of developing CBD (2). Both innate and adaptive immune cells, particularly beryllium-reactive CD4+ T cells, are involved in the course of disease and ultimately cause granulomatous lung inflammation. So far, knowledge on leading target antigens in CBD has been limited to plexin A and beryllium itself (3, 4). In this issue of the JCI, Falta et al. identify a CBD-associated complementarity-determining region 3β (CDR3β) motif within bronchoalveolar lavage (BAL) CD4+ T cells, which displayed beryllium-specific reactivity (1). Further, by scanning peptide libraries, positionally modifying targeted amino acids, and using sophisticated ranking approaches, the authors recognized C-C motif ligand 4 (CCL4)and CCL3 as major antigenic targets of CD4+ T cells in CBD (1). Finally, to confirm the presence of CCL4/CCL3-reactive CD4+ T cells, Falta et al. made use of beryllium-pulsed HLA-DP2-CCL4/CCL3 tetramers in CBD patients and different humanized mouse models (1). Thus, they described a so far unknown adaptive immune response of autoreactive T cells against components of the innate immune system fueling inflammation.
Algorithm for detection of autoreactive CD4+ T cells
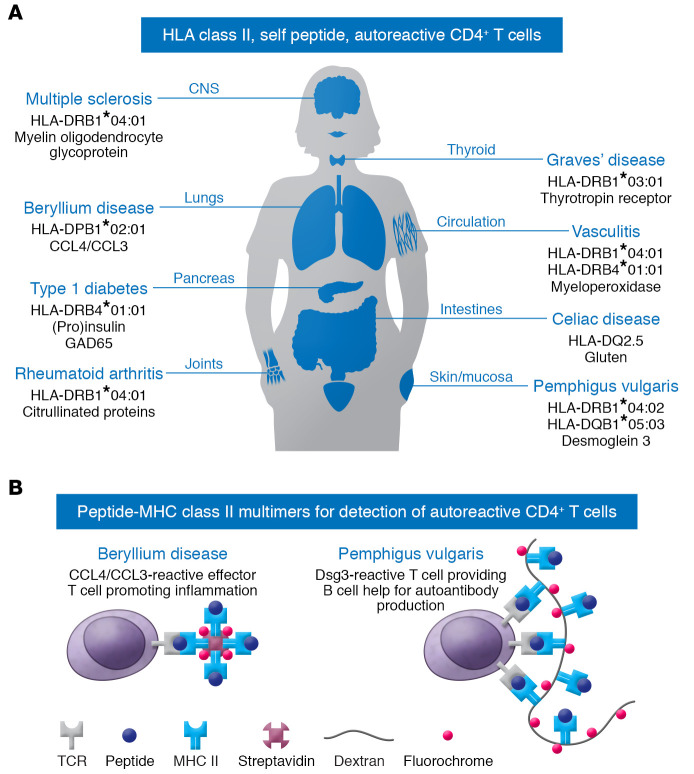
The specific interaction of T cell receptors (TCRs) with self-peptide MHC is fundamental to initiating autoimmune diseases. Autoreactive CD4+ T cells have the capacity not only to produce proinflammatory cytokines, but also to help B cells in maturation and production of high-affinity autoantibodies. Knowledge of the disease-associated HLA class II alleles and the respective disease-specific autoantigens provides the basis for detecting and monitoring disease-relevant autoreactive T cells. These factors have paved the way for the development of peptide–MHC class II (pMHC) multimers, which have been successfully applied in various autoimmune diseases, i.e., multiple sclerosis, type 1 diabetes, Graves’ disease, vasculitis, celiac disease, rheumatoid arthritis, and pemphigus vulgaris (PV), as shown in Figure 1A and refs. 5–11. Similarly to the efforts by Falta et al., previous work has assessed HLA restriction and peptide immunogenicity as well as TCR clonality in PV (12). PV is a chronic autoimmune bullous disease of mucous membranes and the skin in which IgG autoantibodies interfere with desmosomal cell-cell adhesion of epidermal keratinocytes (12). The immunogenetic restriction of the autoimmune response in PV is well understood, as the HLA class II alleles HLA-DRβ1*0402 and/or HLA-DQβ1*0503 are prevalent in PV. Both alleles shape the CD4+ T cellular immune response against a restricted set of immunodominant epitopes of the major autoantigen desmoglein 3 (Dsg3) (12). Dsg3-reactive T cells are required to induce and perpetuate, via B cell help, pathogenic IgG antibodies, which eventually leads to the loss of epidermal cell-cell adhesion and the clinical outcome of blisters and erosions of the mucous membranes and the skin. This intimate T cell–B cell interaction is critical, as indicated by therapeutic B cell depletion experiments in which the anti-CD20 monoclonal antibody rituximab eliminates both peripheral B cells and Dsg3-specific T cells. This elimination is probably due to the fact that these T cells require B cells as antigen-presenting cells (13). Moreover, immunization of HLA-DRβ1*04:02 humanized mice with T cell epitopes of human Dsg3 induces pathogenic anti-Dsg3 IgG antibodies via the generation of Dsg3-specific T cells (14). This IgG response to Dsg3 is abrogated by anti-CD40L monoclonal antibodies, which interfere with T cell–B cell interaction (14). Using a similar approach, Falta et al. showed that exposure of HLA-DP2 transgenic mice to beryllium leads to the induction of CCL4/CCL3-reactive BAL CD4+ T cells, as seen in human CBD (1).
Figure 1. pMHC multimer technology can reveal HLA class II–restricted, autoreactive CD4+ T cells in human autoimmune disorders.
(A) Autoimmune diseases with known HLA association and identified immunogenic self-peptides enable the study of autoreactive CD4+ T cells by pMHC multimers. (B) Available multimeric pMHC complexes can detect autoreactive CD4+ T cells in CBD and PV. In CBD (left), CCL4/CCL3-reactive effector T cells promote inflammation. In PV (right), Dsg3-reactive T cells help B cells with autoantibody production.
Tetramer/dextramer technology
The advent of MHC class II–peptide technology has facilitated a better understanding of the immune pathogenesis of several HLA class II–linked autoimmune disorders. Since autoreactive CD4+ T cells are critical inducers and drivers of these disorders, their detection and monitoring have now become possible by multimeric pMHC complexes (Figure 1B). Technically, distinct synthetic HLA class II alleles are loaded (open multimers) or covalently linked (closed multimers) with/to immunodominant autoantigenic peptides. Multimerization is achieved via biotinylation to fluorochrome-conjugated streptavidin or via binding to a fluorochrome-conjugated dextran backbone. While this multimerization results in increased avidity by crosslinking various TCRs and increased fluorescence intensity due to numerous bound fluorochromes, pMHC monomers also imply some limitations and pitfalls. First, the number and frequency of autoreactive CD4+ T cells in peripheral blood are low, as demonstrated in various studies (6, 8, 15, 16). However, as shown in other reports, including Falta et al., autoreactive T cells may have a higher frequency in targeted organs (1, 10). Alternatively, autoreactive T cells can be expanded ex vivo prior to labeling with pMHC multimers (10, 17). Second, the TCR of autoreactive T cells must initially display a low affinity to escape negative selection in the thymus, which may result in poor pMHC multimer binding.
As discussed above, CD4+ T cell recognition of autoantigenic peptides is mainly regulated by peptide binding to distinct motifs of the variable β chains of the disease-associated HLA-DR, -DP or -DQ alleles. In PV, the β1 chain of HLA-DRB1*04:02 exhibits a negative charge in the P4 pocket, which is critical for binding of self-peptides carrying a positive charge at their P4 anchor motifs (12, 18). Similarly, Falta et al. identified both the complete binding motif within HLA-DP2 and the epitopes of the autoantigenic targets (1). Here, beryllium exposure modifies the identified self-peptides of CCL4/CCL3 and thereby possibly enhances the binding affinity to HLA class II (1), as already shown for citrullinated peptides in rheumatoid arthritis (10). Moreover, the formation of neoantigens may lead to a break of tolerance upon recognition of T cells with high-affinity TCR. In another study, the peptide sequence was mutated at the anchoring position to stabilize HLA binding for tetramer analysis in multiple sclerosis (5).
Several studies have utilized pMHC multimers in flow cytometric analyses and identified an antigen-experienced memory phenotype of autoreactive CD4+ T cells in vasculitis, celiac disease, and rheumatoid arthritis (8–10, 17). Despite advances in the detection of autoreactive T cells by pMHC multimer technology, a rather polyclonal T cell response, which is quite common in many of these diseases, may limit identifying monoclonal or oligoclonal autoreactive T cell responses. Furthermore, possible promiscuity between different HLA class II haplotypes and TCRs of autoreactive T cells, as described for the PV-associated HLA class II alleles HLA-DRβ1*0402 and HLA-DQβ1*0503, represents a remaining uncertainty in detecting autoreactive CD4+ T cells (19).
Clinical implications and conclusions
Understanding the immune pathogenesis of distinct autoimmune diseases, including CBD and PV, has profound implications for the development of treatment options for affected patients. Falta et al. exemplify the detection of pathogenic autoreactive T cells via pMHC multimers and provide a basis to further characterize and identify therapeutic targets in T cell–driven autoimmune disorders. This study not only identifies antigenic targets in CBD, but also presents a structured approach for identifying antigenic targets in other autoimmune diseases in the future (1). Using pMHC multimers, it is possible to monitor disease-relevant T cells ex vivo, as they may reflect highly relevant parameters of disease activity and severity. For a deeper understanding, multiomics approaches will reveal more details about the role of autoreactive T cells in autoimmune diseases, such as CBD and PV, thereby contributing to the discovery of targets for future therapies. Furthermore, profiling of T cell responses by using different fluorochrome-labeled pMHC multimers may promote the development of targeted therapies (17).
Acknowledgments
This work was supported by grants from the German Research Foundation/Deutsche Forschungsgemeinschaft (DFG), Research Group Pegasus (FOR 2497). We thank Christoph Hudemann and Christian Möbs for careful reading of the manuscript.
Version 1. 05/03/2021
Electronic publication
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Copyright: © 2021, American Society for Clinical Investigation.
Reference information: J Clin Invest. 2021;131(9):e148674. https://doi.org/10.1172/JCI148674.
See the related article at Beryllium-specific CD4+ T cells induced by chemokine neoantigens perpetuate inflammation.
Contributor Information
Karolin Wieber, Email: karolin.wieber@uni-marburg.de.
Christine L. Zimmer, Email: christine.zimmer@uni-marburg.de.
Michael Hertl, Email: michael.hertl@med.uni-marburg.de.
References
- 1.Falta MT, et al. Beryllium-specific CD4+ T cells induced by chemokine neoantigens perpetuate inflammation. J Clin Invest. 2021;131(9):144864. doi: 10.1172/JCI144864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Greaves SA, et al. Adaptive immunity in pulmonary sarcoidosis and chronic beryllium disease. Front Immunol. 2020;11:474. doi: 10.3389/fimmu.2020.00474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Falta MT, et al. Identification of beryllium-dependent peptides recognized by CD4+ T cells in chronic beryllium disease. J Exp Med. 2013;210(7):1403–1418. doi: 10.1084/jem.20122426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fontenot AP, et al. Recombinant HLA-DP2 binds beryllium and tolerizes beryllium-specific pathogenic CD4+ T cells. J Immunol. 2006;177(6):3874–3883. doi: 10.4049/jimmunol.177.6.3874. [DOI] [PubMed] [Google Scholar]
- 5.Raddassi K, et al. Increased frequencies of myelin oligodendrocyte glycoprotein/MHC class II-binding CD4 cells in patients with multiple sclerosis. J Immunol. 2011;187(2):1039–1046. doi: 10.4049/jimmunol.1001543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.James EA, et al. DRB4*01:01 has a distinct motif and presents a proinsulin epitope that is recognized in subjects with type 1 diabetes. J Immunol. 2018;201(12):3524–3533. doi: 10.4049/jimmunol.1800723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Degen H, et al. Thyrotropin receptor-specific lymphocytes in adenovirus-TSHR-immunized native and human leukocyte antigen-DR3-transgenic mice and in graves’ disease patient blood. Thyroid. doi: 10.1089/thy.2020.0338. [published online February 8, 2021]. [DOI] [PubMed] [Google Scholar]
- 8.Free ME, et al. Restricted myeloperoxidase epitopes drive the adaptive immune response in MPO-ANCA vasculitis. J Autoimmun. 2020;106:102306. doi: 10.1016/j.jaut.2019.102306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Christophersen A, et al. Distinct phenotype of CD4+ T cells driving celiac disease identified in multiple autoimmune conditions. Nat Med. 2019;25(5):734–737. doi: 10.1038/s41591-019-0403-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pieper J, et al. Memory T cells specific to citrullinated α-enolase are enriched in the rheumatic joint. J Autoimmun. 2018;92:47–56. doi: 10.1016/j.jaut.2018.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Veldman C, et al. Detection of low avidity desmoglein 3-reactive T cells in pemphigus vulgaris using HLA-DR beta 1*0402 tetramers. Clin Immunol. 2007;122(3):330–337. doi: 10.1016/j.clim.2006.09.014. [DOI] [PubMed] [Google Scholar]
- 12.Hertl M, et al. T cell control in autoimmune bullous skin disorders. J Clin Invest. 2006;116(5):1159–1166. doi: 10.1172/JCI28547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eming R, et al. Rituximab exerts a dual effect in pemphigus vulgaris. J Invest Dermatol. 2008;128(12):2850–2858. doi: 10.1038/jid.2008.172. [DOI] [PubMed] [Google Scholar]
- 14.Eming R, et al. Pathogenic IgG antibodies against desmoglein 3 in pemphigus vulgaris are regulated by HLA-DRB1*04:02-restricted T cells. J Immunol. 2014;193(9):4391–4399. doi: 10.4049/jimmunol.1401081. [DOI] [PubMed] [Google Scholar]
- 15.Cianciotti BC, et al. CD4+ memory stem T cells recognizing citrullinated epitopes are expanded in patients with rheumatoid arthritis and sensitive to tumor necrosis factor blockade. Arthritis Rheumatol. 2020;72(4):565–575. doi: 10.1002/art.41157. [DOI] [PubMed] [Google Scholar]
- 16.Rims C, et al. Citrullinated aggrecan epitopes as targets of autoreactive CD4+ T cells in patients with rheumatoid arthritis. Arthritis Rheumatol. 2019;71(4):518–528. doi: 10.1002/art.40768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Uchtenhagen H, et al. Efficient ex vivo analysis of CD4+ T-cell responses using combinatorial HLA class II tetramer staining. Nat Commun. 2016;7:12614. doi: 10.1038/ncomms12614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tong JC, et al. Prediction of desmoglein-3 peptides reveals multiple shared T-cell epitopes in HLA DR4- and DR6-associated pemphigus vulgaris. BMC Bioinformatics. 2006;7 Suppl 5:S7. doi: 10.1186/1471-2105-7-S5-S7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hertl M, et al. Recognition of desmoglein 3 by autoreactive T cells in pemphigus vulgaris patients and normals. J Invest Dermatol. 1998;110(1):62–66. doi: 10.1046/j.1523-1747.1998.00086.x. [DOI] [PubMed] [Google Scholar]