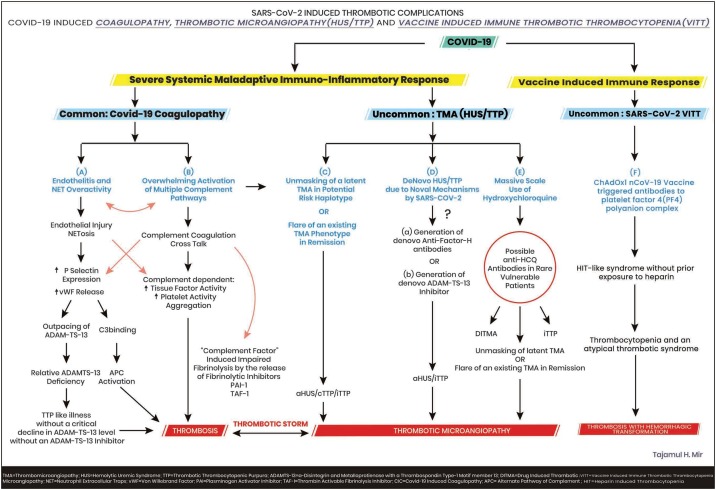
Since the emergence of the novel virus SARS CoV-2 from a Chinese province in ending 2019, its rapid global dissemination has resulted in more than a million deaths worldwide so far (Anon., 2021). Without a doubt the medical fraternity has been overstretched globally by Covid -19 pandemic both in terms of its magnitude and because of the varied presentations of the disease in some, posing a grueling challenge. With time, diligent indagation and probing has unraveled some surprising facts about the novel virus and its pathogenesis. The thrombogenic potential of Covid 19 is becoming more tangible and the so called “Covid-19 Coagulopathy” is being recognized as a conglomerate of mechanisms whipped up by the novel virus, resulting in severe endotheliitis, a maladaptive hyper-immuno-inflammatory response along-with the overwhelming activation of multiple complement pathways and a complement coagulation cross talk altogether culminating into a hypercoagulable state and its dangerous consequences adversely affecting the outcome (Martin-Rojas et al., 2020; Java et al., 2020). (Fig. 1 A and B).
Fig. 1.
The Spectrum of Covid-19 Associated Coagulopathy and Thrombotic microangiopathies.
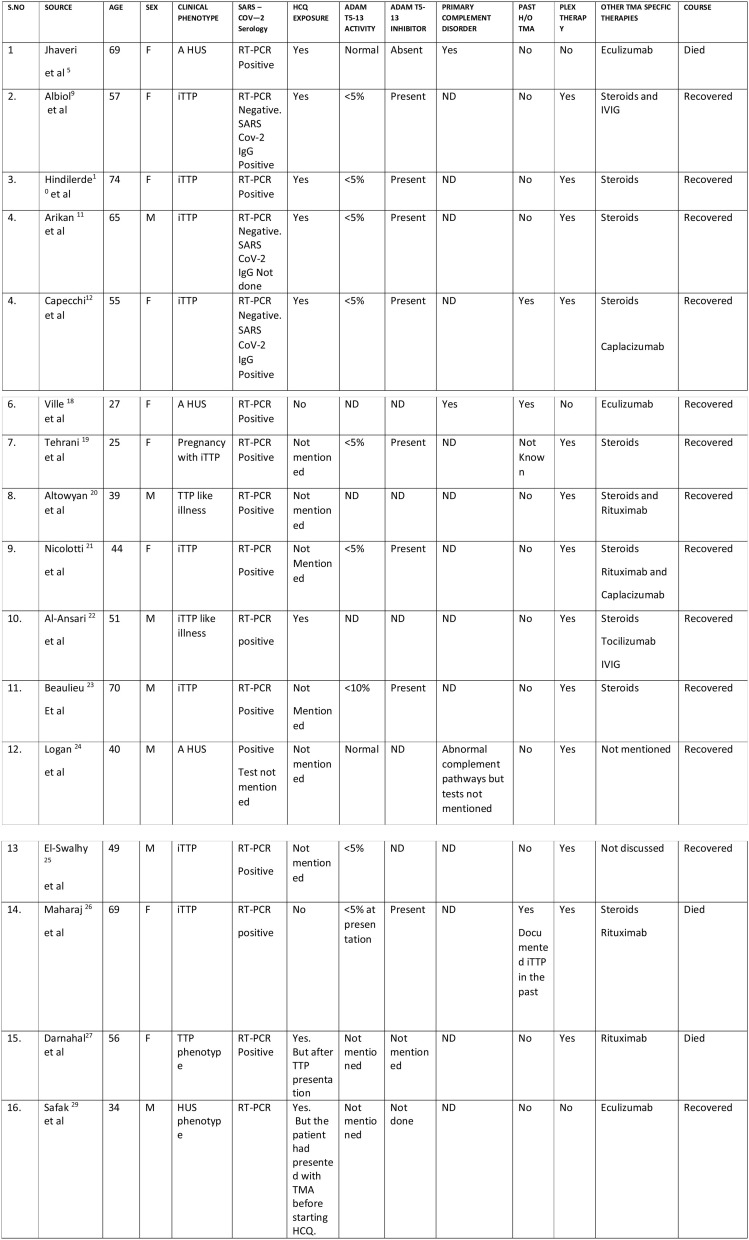
On the other hand, the COVID-19 thrombotic microangiopathy (TMA) is a complex admixture encompassing a “TTP”-like illness on the one end of the spectrum and on the other the rare but baneful, classic thrombotic microangiopathy including thrombotic thrombocytopenic purpura (TTP) and the hemolytic uremic syndrome (a HUS) which are a constellation of thrombocytopenia, microangiopathic hemolytic anemia and extensive clot formation in the microvasculature. TTP results from an inherited or acquired, critical decline (<5%) in ADAMTS-13 (a disintegrin and metalloproteinase with thrombospondin type 1 motif member 13) activity, an enzyme which cleaves the platelet binding, clot promoting highly active and adhesive ultra large von willebrand factor (ULVWF) multimers released in response to injury by the damaged endothelium to smaller, lesser active and inert multimers. HUS results from inherited or acquired aberrations in the alternate complement system and its regulatory proteins leading to its perpetual hyperactivation. Recently Peyvandi and her group have elegantly shown that this Covid-19 induced TTP like illness is not a true TTP but a unique phenotype of sepsis-like TTP without a dysfunctional VWF-ADAMTS-13 axis, occurring due to excessive unleashing of vWF (Von Willebrand factor) multimers from the damaged endothelium overwhelming the cleaving protease resulting in a markedly increased VWF: Ag to ADAMTS-13 ratio causing its relative deficiency but in absence of a circulating inhibitor and without a critical decline in ADAMTS-13 activity. Thrombocytopenia and hemolytic anemia are typically absent since the platelet consuming ULVWF multimers are absent. This syndrome responds to steroids and anticoagulants and does not require plasma exchange (PEX) (Mancini et al., 2020) (Fig. 2).
Fig. 2.
COVID-19 ASSOCIATED CASES OF CLASSIC THROMBOTIC MICROANGIOPATHY (aHUS/iTTP) REPORTED SO FAR.
A HUS = Atypical Hemolytic Syndrome; iTTP = Immune Thrombotic Thrombocytopenic Purpura; RT-PCR = Reverse Transcription Polymerase Chain Reaction; HCQ = Hydroxychloroquine; ADAM-TS-13 = a Disintegrin and Metalloproteinase with Thrombospondin Type 1 Motif member 13; PLEX = Plasma exchange. ND = Not Done.
Besides the relatively common TTP like illness of COVID-19 a few cases of the classic (primary) thrombotic microangiopathic syndromes(TMA), immune-thrombotic thrombocytopenic purpura and atypical hemolytic uremic syndrome (iTTP/aHUS causing TMA with thrombocytopenia microangiopathic hemolytic anemia and organ dysfunction) have surfaced as well in COVID-19 infection. It appears that these cases of classic TMA reported so far seem to have a mixed etiology evolving either as a consequence of some novel mechanisms generated by this virus in a few to the unmasking of a latent genetically mediated TMA or a relapse of a known TMA disorder (HUS or iTTP) already in remission and in a few even due to drugs especially hydroxychloroquine.
So far only sixteen cases of the classic TMA have been reported despite the enormous magnitude of this pandemic since testing for alternate complement pathway and ADAM TS 13 is being done only in specialized labs which could lead to a relative under reporting of this complication during this pandemic especially from underdeveloped countries. Most of these cases have been directly attributed to the novel virus barring three cases, in which the novel viral infection sparked a relapse of a known TMA and in one case TMA (iTTP) was directly attributed to hydroxychloroquine (HCQ) (Table-1). At least eight of these cases have emerged in the light of this novel virus and concomitant hydroxychloroquine therapy both known to bring about this complication, and at least six of these eight patients had a temporal relationship of the TMA with HCQ initiation therefore discerning the actual causative agent has been a challenge in some. Recently Jhaveri et al. reported a case of severe Covid 19 pneumonia and multiorgan dysfunction complicated by a fatal TMA secondary to the activation of the alternate complement pathway with low factor H levels causing advanced renal failure 2-weeks after admission. The TMA was attributed to Covid-19 infection by the authors, this was subsequently challenged in a letter to the editor correlating the temporal profile of TMA in this particular patient with hydroxychloroquine therapy. However, the authors refuted this query by a counter explanation ratiocinating a delayed two-week onset of TMA after stopping hydroxychloroquine thereby ruling out the cause and effect relation (Jhaveri et al., 2020; Hasbal, 2020; Wanchoo et al., 2018). Agreed that TMA could have been due to a genetic variant of TMA (a HUS) and the virus could have acted as a second hit in unmasking the TMA or even made a direct contribution via a novel mechanism and HCQ may or may not have contributed to the development of TMA in that particular patient howbeit, a long elimination half-life of 40 days after a single dose of 200 mg HCQ, questions the explanation given by the authors in acquitting HCQ as a cause of TMA in that particular patient and hence raises some eyebrows (Tett et al., 1989). Albiol, Hindilerden, Arikan, Capecchi and Al-Ansari and coworkers reported five cases of Covid-19 respectively who developed the classic iTTP syndrome with critically low ADAM-TS 13 levels in association with an ADAM-TS 13 inhibitor during the course of their illness. Surprisingly all the five cases reported were concurrently on HCQ treatment with a strong temporal relationship of the drug with the evolution of iTTP. All these cases responded to plasma exchange while in Capecchi’s study besides plasma exchange anti-vWF nanobody caplacizumab was given as well. In Arikan’s case RT-PCR was negative for SARS CoV 2 infection and IgG-antibody testing for Covid-19 was not done but a strong probability score favoring HCQ induced iTTP was established with the authors concluding that the iTTP being secondary to HCQ however, contrasted to this particular patient HCQ/quinine induced TTP is not a typical iTTP and has not been associated with a critical drop in ADAM-TS 13 levels or the presence of an antibody against the same which on the one hand calls into question the diagnosis made by these authors and on the other even attributes iTTP to HCQ as a novel complication (Albiol et al., 2020; Hindilerden et al., 2020; Arikan et al., 2020; Capecchi et al., 2020; Mir, 2021; Altowyan et al., 2021).
Ironically HCQ is well known for some of its anti-thrombotic properties especially in patients of auto immune connective tissue diseases and antiphospholipid antibody syndrome. HCQ induced TMA is a rare but nevertheless a defined entity, being an idiosyncratic reaction rather than dose related which can develop anytime within hours, days or months of drug exposure usually associated with antibodies against platelets and endothelium. The typical drug induced TMA (DITMA)including quinine induced TMA is a clinical syndrome of TMA without developing critically low ADAM-TS 13 levels or a circulating inhibitor. However, the potential of HCQ to spark a TMA has been extrapolated against a literature background of quinine associated TMA since only a few cases of HCQ associated TMA have been reported so far without a detailed evaluation (Fromm, 2018; Mar and Mendoza Ladd, 2011). Notwithstanding some cases of the classic iTTP with critically low ADAM-TS 13 levels and presence of an inhibitor have been reported in literature in association with the thienopyridine group of anti- platelet drugs (Zakarija et al., 2021). Whether HCQ can induce the production of an antibody against ADAM-TS 13 in certain vulnerable patients is a matter of debate and needs to be confirmed. It is worth a mention that hydroxychloroquine is not effectively removed by any extracorporeal therapy so even if the drug had contributed to the development of iTTP in some of these cases which is ambiguous though, the therapeutic effect of plasma exchange could have been mainly due to the removal of the circulating antibody i.e. ADAM TS -13 inhibitor present in all four cases, rather than the drug removal, and steroids could have attenuated the further production of antibodies even if the drug persisted in circulation for a while (Berling et al., 2020).
Ten more cases of Covid-19 associated TMA have been reported one by Ville et al. a COVID-19 induced relapse of genetic HUS in a lady which responded to eculizumab (Villi et al., 2021). In the other two cases TMA occurred in the light of Covid-19 but specific phenotype could not be established in one due to lack of testing, both responded to plasma exchange (PEX) steroids and rituximab (Altowyan et al., 2021; Nicolotti et al., 2021). Beaulieu et al. reported a case of COVID-19 associated cases of iTTP which responded to PEX and caplacizumab (Beaulieu et al., 2021). Two more cases of aHUS and iTTP complicating COVID-19 infection were very recently reported however, exposure to HCQ was not mentioned both responded to PEX (Logan et al., 2020; El-Sawalhy et al., 2021). Maharaj et al. recently reported a challenging scenario of a case of recalcitrant iTTP with a past history of frequent relapses and thrombotic complications, who was recovering from a recent relapse of TMA. She got readmitted with a COVID-19 pneumonia complicated by an atypical iTTP relapse which was characterized by an overwhelming coagulopathy but without a microangiopathic hemolytic anemia and thrombocytopenia amidst a very low ADAM TS-13 activity and a detectable inhibitor despite a markedly increased VWF: Ag to ADAMTS-13 ratio indicating a complex interaction of COVID coagulopathy and an underlying iTTP evolving into a unique disease phenotype. This challenging case is a mixed bag of COVID-19 induced TTP like syndrome and a true iTTP. It seems difficult to explain the pathophysiology of this unique phenotype since, in a known case of iTTP the novel viral infection should have aggravated the basic phenotype because iTTP can flare amidst an infection known to induce endothelial stress or injury and COVID-19 is known to cause endotheliitis and injury via multiple mechanisms.The most plausible explanation for this unique phenotype reported by Maharaj et al. could be that during this patient’s recent iTTP flare an exposure to rituximab which was not tolerated well precluding its further dosing (the dose of rituximab received by this patient is not mentioned 100 mg or 500 mg or 1 g), could have elseways masked some features of her iTTP and worsened her COVID disease and its coagulopathy and negatively influenced the outcome. Since rituximab can markedly influence the course of iTTP by significantly reducing the flares and sometimes its therapeutic effect could be delayed even by a month. So her iTTP didn’t worsen in the face of this novel viral infection possibly due to a delayed rituximab effect even if her ADAMTS-13 levels were low in presence of an inhibitor at the point of COVID-19 infection, the tradeoff being a fulminant COVID-19 and its “pseudo TTP syndrome” dominating with its thrombotic complications”. Densitometric studies of vWF multimers in this patient could have helped to decipher the predominant phenotype with absence of the hyperactive and adherent ultra large vWF (ULVWF) multimers favoring the “COVID-19 induced pseudo TTP” (Maharaj et al., 2021). The relative safety of rituximab in a critically ill Covid-19 patient is quite concerning or even disapproving given higher probabilities of an unfavorable outcome which was also shown in Darnahal et al. case (Darnahal et al., 2021). In refractory cases of iTTP in the light of this pandemic anti VWF nanobody caplacizumab is a suitable option if readily available which could effectively antagonize the ULVWF multimers without influencing the antibody response to SARS CoV- 2 infection.
In all the sixteen cases of Covid-19 associated classic TMA reported so far two different phenotypes of TMA evolved in the light of an ongoing Covid-19 infection and eight of them being on hydroxychloroquine (in six additional cases exposure to HCQ was not mentioned). A past history of documented TMA was present in three patients, with the novel virus triggering a flare in the them. Picking up the real culprit unequivocally in the remaining would be a difficult task. What should be the future approach in a similar setting? In recovered patients with an exposure to HCQ, rechallenge with the drug could solve the puzzle but this re-exposure would be neither ethical nor clinically prudent since the consequence of a second exposure could be more severe if the drug was the culprit. The role of novel invitro HCQ sensitizing tests need to be determined as well (Galbiati et al., 2021). The virus seems to have directly triggered a primary thrombotic microangiopathy through some novel mechanisms in some of these patients (Fig. 1-D). However, long term follow- up of the above mentioned patients who recovered is recommended with collaborative registry data.
The classification of primary versus secondary TMA is not an absolute one since many patients with the genetic variants or even latent acquired cases of the disease could be clinically silent for years to be unmasked by a secondary event like infection, endothelial injury, drugs and pregnancy etc. which act as second hits and at times baffle even the most experienced in the field regarding the origin of the clinical entity which could have been the fons et origo in the cases of Tehrani and Safak (Tehrani et al., 2020; Safak et al., 2021). In these predisposed patients for TMA both the virus and the drug could operate in tandem or individually as second hits by triggering a flare or by unmasking a latent disease.
The history of HCQ use in medicine has been notable and on a large scale in treating countless number of patients with malaria and various connective tissue diseases annually, with HCQ induced TMA being a rare entity reported in a miniscule of patients (more with the parent drug quinine) which goes in perfect harmony with only a few cases of TMA (TTP/HUS) in Covid 19 patients on hydroxychloroquine reported in literature so far despite the massive use of the drug. As already mentioned HCQ induced TTP is not a typical iTTP albeit very few cases of HCQ associated TTP reported so far, hence shutting doors to a possible novel mechanism behind an iTTP induced by the drug would be imprudent. Given the intricate and complex interplay of multiple mechanisms operating in this “inflammatory onslaught” in critically ill COVID-19 patients, one of the less suspected culprits could easily escape clinical attention and go virtually unnoticed (Fig. 1 C, D and E) (Varga et al., 2020; Martinelli et al., 2020).
Summing up the syndrome of COVID-19 associated TMA is not a single entity but an omnium gatherum of different mechanisms and etiologies, “the pseudo TTP-like syndrome of COVID-19″, “the true-TTP/HUS flare by COVID-19″, “the true- de novo iTTP/HUS syndrome of COVID-19″, “the overlap-TTP syndrome of COVID-19″ (reported by Mahajan et al) and a possible “drug-induced-TTP/HUS syndrome occurring in COVID-19″. Therefore, in the case of classic TMA(aHUS/iTTP) in the light of Covid 19, the direct effect of the novel virus, an underlying genetic predisposition and even hydroxychloroquine therapy: “before attributing everything to the novel virus it would be worthwhile to be astute about other possibilities as well until proven otherwise”.
Author contribution
THM Conceptualized drafted, collected the data and wrote the manuscript and also created Fig. 1.
Funding
NO Funding Involved.
ETHICAL APPROVAL
This article does not contain any of the studies with human participants or animals performed by the author.
Declaration of Competing Interest
The authors report no declarations of interest.
Acknowledgement
The author would like to thank Mr. Ishrat Ul Hamid Lone (creative computers Srinagar) and Mr. Shabir Ahmed Gojri for their help in making Fig. 1).
References
- Albiol N., Awol R., Martino R. Autoimmune thrombotic thrombocytopenic purpura (TTP) associated with COVID-19. Ann. Hematol. 2020;99(7):1673–1674. doi: 10.1007/s00277-020-04097-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altowyan E., Alnujeidi O., Alhujilan A., et al. COVID-19 presenting as thrombotic thrombocytopenic purpura (TTP) BMJ Case Rep. 2021;13 doi: 10.1136/bcr-2020-238026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anon. https://www.worldmeters.info/coronavirus/coronavirus-death-toll. accessed30th October2020.
- Arikan F., Yildiz Y., Ercan T., Oruc O., Aksay S., Yilmaz F., Toptas T., Tuglular T. Hydroxychloroquine-associated thrombotic thrombocytopenic purpura. Turk. J. Haematol. 2020;(July) doi: 10.4274/tjh.galenos.2020.2020.0322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beaulieu M., Mettelus D., Masse B., et al. Thrombotic thrombocytopenic purpura as the initial presentation of COVID=19. J. Thromb. Haemost. 2021 doi: 10.1111/jth.15231. doi:10.111/JTH.15231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berling I., King J.D., Shepherd G., et al. Extracorporeal treatment of chloroquine, hydroxychloroquine and quinine poisoning: systematic review and recommendations from the EXTRIP workgroup. JASN. 2020;31(10):2475–2489. doi: 10.1681/ASN.20200505642020. Nov 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capecchi M., Cristina Mocellin C., Abbruzzese C., et al. Dramatic presentation of acquired thombotic thrombocytopenic purpura associated with COVID-19. Haematologica. 2020;105(10) doi: 10.3324/haematol.2020.262345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Darnahal M., Tehrani H., Vaezi M., et al. COVID 19 and thrombotic thrombocytopenic Purpura: a case report. IJHOSCR. 2021;15(1):72–74. doi: 10.18502/ijhoscr.v15i1.5251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Sawalhy E., Sheereef H., Manzoor H., et al. A rare presentation of COVID-19 associated thrombotic thrombocytopenic purpura: therapeutic challenges. Am. J. Clin. Case Rep. 2021;9(3):147–149. doi: 10.12691/ajmcr-9-3-3. [DOI] [Google Scholar]
- Fromm L.M. Suspected hydroxychloroquine-induced thrombotic thrombocytopenic purpura. J. Pharm. Pract Res. 2018;48(1):72–75. [Google Scholar]
- Galbiati V., Papale A., Kummer E., et al. Invitro models to evaluate drug-induced hypersensitivity: potential tests based on activation of dendritic cells. Front. Pharmacol. 2021 doi: 10.3389/fphar.2016.00204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasbal N.B. Thrombotic Microangiopathy: COVID-19 or Hydroxychloroquine? Kidney Int. 2020 doi: 10.1016/j.kint.2020.08.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hindilerden F., Yonal-Hindilerden I., Akar E., Kart-Yasar K. Covid-19 associated autoimmune thrombotic thrombocytopenic purpura: report of a case. Thromb. Res. 2020:195. doi: 10.1016/j.thromres.2020.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Java A., Apicelli A.J., Liszewski M.K., et al. The complement system in covid-19: friend and foe? JCI Insight. 2020;5(15):1–12. doi: 10.1172/jci.insight.140711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jhaveri Kd, Meir Lr, Chang Bsf, Parikh R., Wanchoo R., Barilla-La-Barca Ml, Bijol V., Hajizadeh N. Thrombotic microangiopathy in patient with COVID-19. Kidney Int. 2020;98(2):509–512. doi: 10.1016/j.kint.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logan D., Jawaid M., Anand U., et al. Plasma exchange in the treatment of thrombotic microangiopathy associated with COVID-19 infection: a case report. Chest (Ann. Meet.) 2020 doi: 10.1016/j.chest.2020.08.2123. [DOI] [Google Scholar]
- Maharaj S., Xue R., Rojan A. Thrombotic thrombocytopenic purpura response following COVID-19 infection: implications for the ADAMTS13-von Willebrand factor axis. J. Thromb. Haemost. 2021 doi: 10.1111/jth.15230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mancini I., Baronciani L., Artoni A., Colpani P., Biganzoli M., Cozzi G., Novembrino C., Boscolo Anzoletti M., De Zan V., Pagliari M.T., Gualtierotti R., Aliberti S., Panigada M., Grasselli G., Blasi F., Peyvandi F. The ADAMTS13-von Willebrand factor axis in COVID-19 patients. J. Thromb. Haemost. 2020;(November) doi: 10.1111/jth.15191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mar N., Mendoza Ladd A. Acquired thrombotic thrombocytopenic purpura: puzzles, curiosities and conundrums. J. Thromb. Thrombolysis. 2011;31(1):119–121. doi: 10.1007/s11239-010-0517-x. [DOI] [PubMed] [Google Scholar]
- Martinelli N., Montagnana M., Pizzolo F., et al. A relative ADAM TS 13 deficiency supports the presence of secondary microangiopathy in COVID 19. Thromb. Res. 2020;193:170–172. doi: 10.1016/j.thromres.2020.07.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin-Rojas R.M., Perez-Rus G., Delgado-Penos V.E., et al. Covid-19 coagulopathy: an in-depth analysis of the coagulation system. Eur. J. Haematol. 2020;00:1–10. doi: 10.1111/ejh.13501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mir T.H. 2021. Immune Thrombotic Thrombocytopenic Purpura (iTTP) in a Patient with Suspected COVID-19: Hydroxychloroquine Culprit or Just a Happenstance? [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicolotti D., Bignami E., Rossi S., et al. A case of thrombotic thrombocytopenic purpura associated with Covid-19. J. Thromb. Thrombolysis. 2021 doi: 10.1007/s11239-020-02362-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Safak S., Aksoy E., Dirim A., et al. Succesful treatment of COVID-19 patient with Thrombotic microangiopathy. Clin. Kidney J. 2021:1–2. doi: 10.1093/ckj/sfab024. doi:101093/ckj /sfab024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tehrani H., Darnahal M., Haghighi S. Covid-19 in a pregnant female with thrombotic microangiopathy a case report. Clin. Case Rep. 2020;10:1392. doi: 10.37421/jccr.2020.10.1392. [DOI] [Google Scholar]
- Tett S.E., Cutler D.J., Day R.O., Brown K.F. Bioavailability of hydroxychloroquine tablets in healthy volunteers. Br. J. Clin. Pharmacol. 1989;27:771–779. doi: 10.1111/j.1365-2125.1989.tb03439.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varga Z., Flammer A.J., Steiger P., Haberecker M., Andermatt R., Zinkernagel A.S., et al. Endothelial cell infection and endotheliitis in Covid 19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villi S., Le Bot S., Chapelet-Debout, et al. Atypical HUS relapse triggered by COVID-19. Kidney Int. 2021;99(January(1)):267–268. doi: 10.1016/j.kint.2020.10.030. Epub. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wanchoo R., Barilla-LaBarca M.L., Jhaveri K.D. Response to thrombotic microangiopathy: COVID-19 or Hydroxychloroquine? Kidney Int. 2018;48(1):72–75. [Google Scholar]
- Zakarija A., Kwaan H., Moake J., et al. Ticlopidine and clopidogrel-associated thrombotic thrombocytopenic purpura (TTP): review of clinical, laboratory, epidemiological and pharmacovigilance findings (1989-2008) Kidney Int. Suppl. (2011) 2021;2 doi: 10.1038/ki.2008.613. [DOI] [PMC free article] [PubMed] [Google Scholar]