Abstract
Background
End-stage kidney disease patients on dialysis are particularly susceptible to COVID-19 infection due to comorbidities, age, and logistic constraints of dialysis making social distancing difficult. We describe our experience with hospitalized dialysis patients with COVID-19 and factors associated with mortality.
Methods
From March 1, 2020, to May 31, 2020, all dialysis patients admitted to 4 Emory Hospitals and tested for COVID-19 were identified. Sociodemographic information and clinical and laboratory data were obtained from the medical record. Death was defined as an in-hospital death or transfer to hospice for end-of-life care. Patients were followed until discharge or death.
Results
Sixty-four dialysis patients with COVID-19 were identified. Eighty-four percent were African-American. The median age was 64 years, and 59% were males. Four patients were on peritoneal dialysis, and 60 were on hemodialysis for a median time of 3.8 years, while 31% were obese. Fever (72%), cough (61%), and diarrhea (22%) were the most common symptoms at presentation. Thirty-three percent required admission to intensive care unit, and 23% required mechanical ventilation. The median length of stay was 10 days, while 11 patients (17%) died during hospitalization and 17% were discharged to a temporary rehabilitation facility. Age >65 years (RR 13.7, CI: 1.9–100.7), C-reactive protein >100 mg/dL (RR 8.3, CI: 1.1–60.4), peak D-dimer >3,000 ng/mL (RR 4.3, CI: 1.03–18.2), bilirubin >1 mg/dL (RR 3.9, CI: 1.5–10.4), and history of peripheral vascular disease (RR 3.2, CI: 1.2–9.1) were associated with mortality. Dialysis COVID-19-infected patients were more likely to develop thromboembolic complications than those without COVID-19 (RR 3.7, CI: 1.3–10.1).
Conclusion
In a predominantly African-American population, the mortality of end-stage kidney disease patients admitted with COVID-19 infection was 17%. Age, C-reactive protein, D-dimer, bilirubin, and history of peripheral vascular disease were associated with worse survival.
Keywords: COVID-19, Dialysis, Outcomes
Introduction
Dialysis patients carry a higher risk of infectious complications, and from early reports, this risk includes complications from the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) which causes COVID-19. In fact, the first US reported death from COVID-19 occurred in a hemodialysis patient [1]. Since then, there have been several reports describing the effect of COVID-19 in the dialysis population [2, 3, 4, 5, 6, 7, 8, 9].
A report of hospitalized COVID-19 patients from Wuhan, China, showed that hemodialysis patients had worse outcomes compared to controls without kidney failure [5]. In addition, the hemodialysis COVID-19 patients developed serious complications such as shock, acute respiratory distress syndrome (ARDS), arrhythmias, and acute cardiac injury at a higher rate. The authors concluded that patients on hemodialysis with COVID-19 were at higher risk of death (14 vs. 4%) [5]. In a population of hospitalized dialysis patients in New York City, there was a 31% mortality rate and 75% of those who required mechanical ventilation died [2]. This dialysis patient population had similar mortality outcomes to those reported from Europe [7, 8, 9].
Dialysis patients are a high-risk population for transmission of COVID-19 due to some of their underlying comorbidities and logistical difficulties of keeping adequate social distancing as recommended by CDC guidelines [6, 10, 11, 12]. However, it is unclear whether their outcomes are different than the nondialysis population. Additionally, new information suggests that the rate of asymptomatic COVID-19 infection in dialysis patients is substantial [13]. In the USA, African Americans progress to end-stage kidney disease (ESKD) at higher rates [14, 15] and make up nearly 30% of the ESKD population [16]. Furthermore, African Americans have been disproportionally affected by the COVID-19 pandemic due to a higher prevalence of comorbid conditions such as obesity, diabetes, and hypertension [17, 18, 19, 20] and potentially socioeconomic disparities such as institutional racism, poor housing, front-line jobs that limit social distancing, and unemployment and loss of health insurance exacerbated by the pandemic [21, 22, 23, 24].
Given the vulnerability of the ESKD and African-American populations to COVID-19 exposure and its complications, we sought to examine the risk factors, clinical characteristics, and outcomes in hospitals serving this patient population. Here we present our early experience with COVID-19 in a predominantly African-American ESKD population admitted to our large academic health system with special emphasis on factors associated with mortality.
Materials and Methods
Data Selection and Extraction
This study was approved by the Emory University Institutional Review Board. The Emory nephrology consult database and Emory Clinical Data Warehouse (CDW) containing electronical medical records from Emory University Hospital, Emory University Hospital Midtown, Emory Saint Joseph's Hospital, and Emory Johns Creek Hospital were searched to identify all ESKD patients who were admitted between March 1, 2020, and May 31, 2020. Patients undergoing emergency room visits or <24-h observation visits were excluded. ESKD was defined as patients receiving maintenance dialysis prior to admission. ICD10 codes, hemodialysis, and peritoneal dialysis orders were identified. Patients tested for COVID-19 by a polymerase chain reaction (PCR) were selected, and those with positive results were identified. Our institutions used nasopharyngeal samples for COVID-19 testing. Patients were followed until discharge from the hospital or death.
Patient data, including sociodemographic information, clinical data, medication use at time of admission, and laboratory data, were obtained from the Emory CDW. Manual chart review was performed on the COVID-19-positive patients. Symptoms on admission, comorbid conditions, and radiographic findings were extracted manually from the chart. Charlson Comorbidity Index [25] was calculated to determine the burden of comorbidities. Death was defined as an in-hospital death or transfer to hospice for end-of-life care.
Statistical Methods
Continuous variables were described as median (interquartile range [IQR]), and categorical variables as frequency rates or percentages. The Mann-Whitney-Wilcoxon test or Student's t test was used for comparison of continuous variables, whereas the χ2 test or Fisher's exact test was used for comparison of categorical variables. The Shapiro-Wilk test was used to determine distribution normality of continuous variables. Relative risk of death was calculated to explore the association of relevant clinical and laboratory findings with mortality. Data processing, analysis, and graphics were performed with R software (R Core Team [2019], R foundation for Statistical Computing, Vienna, Austria).
Results
Patients and Clinical Characteristics
From March 1, 2020, to May 31, 2020, 807 ESKD patients were admitted. 554 of these patients (69%) were tested for COVID-19 infection. 490 patients tested negative, and 64 patients receiving renal replacement therapy prior to admission had a positive test (12%) and represent the study population in this report.
These 64 ESKD patients represent 9% of COVID-19-related admissions during the study period. Sixty (94%) patients received in-center hemodialysis, and 4 were on peritoneal dialysis prior to admission. The majority of patients were African Americans (84%) with a median age of 64 years. Fifty-nine percent were males, and 62% had diabetes. The median time on dialysis was 3.8 years. These basic demographics are presented in Table 1. Gender (59 vs. 53% males), race distribution (84 vs. 75% African Americans), median age (64 vs. 61 years), and BMI (27 vs. 27 kg/m2) were similar in COVID-19-positive and COVID-19-negative patients.
Table 1.
Baseline demographics and comorbidities
| Total1 (N = 64) | Survivors1 (n = 53) | Deceased1 (n = 11) | p value2 | |
|---|---|---|---|---|
| Age, years | 64 (53–75) | 62 (52–69) | 75 (69–76) | 0.02 |
| Gender, n (%) | ||||
| Female | 26 (41) | 23 (43) | 3 (27) | 0.5 |
| Male | 38 (59) | 30 (57) | 8 (73) | |
| Race, n (%) | ||||
| African American or black* | 54 (84) | 43 (81) | 11 (100) | 0.6 |
| Non-Hispanic Caucasian or white | 6 (10) | 6 (11) | 0 (0) | |
| Hispanic white | 4 (6) | 4 (7.5) | 0 (0) | |
| Vintage | 3.8 (2.0–7.0) | 3.5 (2.0–6.2) | 6.0 (2.4–9.6) | 0.4 |
| Charlson Comorbidity Index (age adjusted) | 7 (5–9) | 6 (5–9) | 8 (7–11) | 0.06 |
| History of hypertension, n (%) | 60 (94) | 49 (92) | 11 (100) | >0.9 |
| History of diabetes, n (%) | 31 (58) | 9 (82) | 0.26 | |
| History of CV disease**, n (%) | 23 (36) | 18 (34) | 5 (45) | 0.06 |
| Congestive heart failure, n (%) | 20 (31) | 17 (32) | 3 (27) | >0.9 |
| Peripheral vascular disease, n (%) | 12 (19) | 7 (13) | 5 (45) | 0.03 |
| Chronic lung disease, n (%) | 13 (20) | 11 (21) | 2 (18) | >0.9 |
| Arrhythmias, n (%) | 11 (17) | 9 (17) | 2 (18) | >0.9 |
| Obesity (body mass index >30), n (%) | 20 (31) | 17 (32) | 3 (27) | 0.4 |
| Body mass index | 27 (23–33) | 27 (23–33) | 27 (22–30) | >0.8 |
| Dementia, n (%) | 8 (12) | 7 (13) | 1 (9.1) | >0.9 |
| History of DVT/PE***, n (%) | 6 (9.4) | 5 (9.4) | 1 (9.1) | >0.9 |
| Tobacco abuse, n (%) | 24 (38) | 17 (32) | 7 (64) | 0.08 |
| Valvular disease, n (%) | 5 (7.8) | 4 (7.5) | 1 (9.1) | >0.9 |
All African Americans were non-Hispanic.
CV disease, coronary artery disease, cerebrovascular accident, and/or valvular disease.
DVT/PE, deep venous thrombosis or pulmonary embolism.
Statistics presented: median (IQR); n (%).
Comparing survivors with deceased. Statistical tests performed: Wilcoxon rank-sum test, Fisher's exact test, and Student's t test.
The most common comorbidities recorded at the time of admission were hypertension, diabetes, congestive heart failure, coronary artery disease, peripheral vascular disease, chronic lung disease, and arrhythmias. There were 2 HIV-positive patients. Thirty-one percent of patients had a body mass index of >30 kg/m2. A detailed prevalence of comorbidities is presented in Table 1. Survivors were younger (median age 62 vs. 75 years, p = 0.01) and had a lower prevalence of peripheral vascular disease (13 vs. 45%, p = 0.025) compared to nonsurvivors. The medical burden of comorbidities was similar as judged by the Charlson Comorbidity Index. Medications prescribed in the outpatient setting and relevant inpatient medications are presented in online suppl. Table A; see www.karger.com/doi/10.1159/000514752 for all online suppl. material. Patients who expired were more likely to have used renin-angiotensin system blockers (ACEi or ARBs) than survivors (64 vs. 30%, p < 0.04).
Symptoms and Chest-X-Ray Findings
The most common symptoms at presentation were fever (72%), cough (61%), and diarrhea (22%). Dyspnea was also reported frequently. Not a single patient reported anosmia as a symptom. The most common radiographic findings were bilateral patchy infiltrates (56%), pulmonary edema (11%), pleural effusions (6%), and atelectasis (5%). Twenty-two percent of chest radiographs were reported as normal or unremarkable at the time of admission.
Laboratory Findings
Laboratory data are presented in Table 2. Sixty-nine percent of COVID-19-positive ESKD patients had lymphopenia (defined as a lymphocyte count of <1,000 cells/mL) on admission. All but 3 patients had C-reactive protein (CRP) above the normal range for our laboratory. When tested in 33 patients, ferritin was above 800 ng/dL in 79% of patients. Troponin I was above the normal range (0.04 ng/mL) in 74% of tested patients (n = 47). Brain natriuretic peptide (BNP) level was elevated in 89% of patients. Aspartate aminotransferase (AST), bilirubin, CRP, and peak D-dimer were statistically higher in patients who expired during hospitalization. D-dimer reached their peak level after a median of 5 days (IQR 1–19). Troponin I on admission was also higher in nonsurvivors, but the difference did not reach statistical significance (Table 2). Interleukin-6 (IL-6) was measured in only 17 patients with 9 having levels above 5 pg/mL (considered the upper limit of normal in our reference laboratory), while 4 of those required ICU admission. None of the 8 patients with normal IL-6 levels required ICU admission. In comparison to our COVID-19-negative patients, ESKD COVID-19-positive patients had significantly higher AST, CRP, ferritin, hemoglobin, and troponin; meanwhile, BNP, lymphocyte count, and overall WBC count were lower (online suppl. Table B).
Table 2.
Baseline laboratory data
| Total1 (N = 64) | Survivors1 (n = 53) | Deceased1 (n = 11) | p value2 | |
|---|---|---|---|---|
| Hemoglobin, g/dL | 10.7 (9.2–11.8) | 10.7 (9.1–11.7) | 10.8 (9.8–11.9) | 0.7 |
| WBC, ×103/mL | 7.0 (5.2–11.1) | 6.0 (4.2–9.3) | 8.1 (5.1–14.2) | 0.3 |
| Platelets, ×103/mL | 210 (142–283) | 210 (152–281) | 209 (138, 295) | 0.8 |
| Lymphocyte count | 0.82 (0.56–1.14) | 0.84 (0.60–1.29) | 0.74 (0.38–0.97) | 0.14 |
| Albumin, g/dL | 3.8 (3.2–4.0) | 3.8 (3.3–4.0) | 3.6 (3.2–3.9) | 0.5 |
| Calcium, mg/dL | 8.9 (8.5–9.4) | 8.8 (8.4–9.3) | 9.2 (8.9–10.0) | 0.06 |
| AST, units/L | 28 (19–48) | 24 (19–39) | 41 (28–148) | 0.02 |
| ALT, units/L | 14 (10–26) | 15 (10–21) | 14 (11–60) | 0.2 |
| Bilirubin, mg/dL | 0.5 (0.4–0.7) | 0.5 (0.4–0.7) | 0.8 (0.5, 1.3) | 0.03 |
| Ferritin, ng/mL | 1,800 (1,081–3,633) | 1,788 (1,008–2,674) | 3,664 (1,760–11,984) | 0.2 |
| CRP (admission), mg/dL | 106 (51–182) | 81 (45–124) | 174 (138–267) | <0.01 |
| CRP (maximal), mg/mL | 162 (65–198) | 116 (49–191) | 245 (177–333) | 0.01 |
| BNP, pg/mL | 576 (291–2,046) | 575 (268–1,650) | 1,545 (205–2,115) | 0.4 |
| Troponin I, ng/mL | 0.07 (0.04–0.23) | 0.07 (0.04–0.11) | 0.25 (0.06–0.87) | 0.06 |
| D-dimer (admission), ng/mL | 1,736 (1,168–3,502) | 1,682 (1,155–2,630) | 1,844 (1,411–7,355) | 0.4 |
| D-dimer (peak), ng/mL | 3,114 (1,474–7,728) | 2,199 (1,342–4,851) | 24,145 (4,172–60,000) | <0.001 |
| Lactic acid, mmol/L | 1.10 (0.75–1.80) | 0.80 (0.70–1.40) | 1.90 (1.28–2.70 | 0.4 |
| LDH, unit/L | 371 (268–534) | 371 (268–524) | 424 (290–3,135) | 0.4 |
WBC, white blood cell count; AST, aspartate aminotransferase; ALT, alanine aminotransferase; CRP, C-reactive protein; BNP, brain natriuretic peptide; LDH, lactate dehydrogenase.
Statistics presented: median (IQR).
Comparing survivors with deceased. Statistical tests performed: Wilcoxon rank-sum test; Fisher's exact test.
Thromboembolic Complications
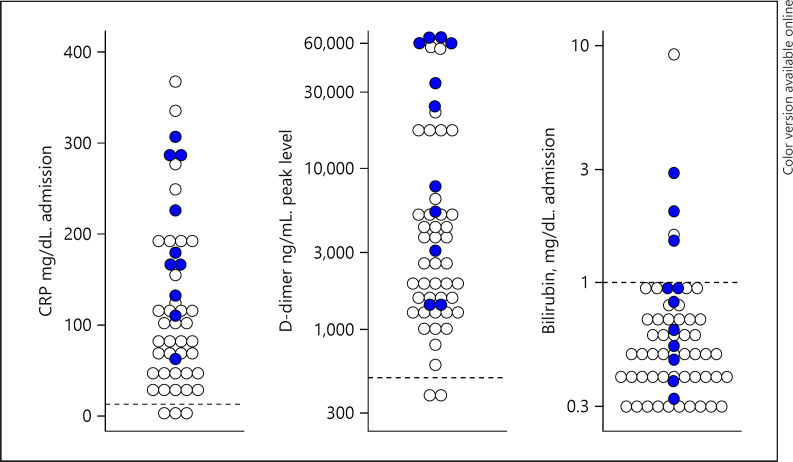
As part of our institution treatment protocol, patients were categorized based on D-dimer levels and assigned to different levels of prophylactic anticoagulation or treatment if a thromboembolic event was documented. In ESKD COVID-19-positive patients, the median peak D-dimer was 3,114 ng/mL (normal upper limit 574 ng/mL) with 42% of patients with D-dimer >3,000 ng/mL. Nonsurvivors had median peak D-dimer levels higher than survivors (24,145 vs. 2,199 ng/mL, p = 0.004) (Table 2; Fig. 1). Interestingly, ESKD COVID-19-negative patients admitted during the same period also had elevated D-dimer, with a median of 3,124 ng/mL.
Fig. 1.
Comparative serum concentration levels of admission CRP, peak D-dimer, and admission bilirubin in COVID-19-positive (blue circles) and COVID-19-negative (white circles) ESKD patients. CRP, C-reactive protein.
Five patients (8%) developed thromboembolic complications (2 pulmonary embolisms [PE] and 3 leg deep venous thromboses [DVT]). As a reference, during the same period, only 2% of ESKD COVID-19-negative patients developed thromboembolic complications, 4 PE and 8 DVT (relative risk [RR] 3.7, CI: 1.3–10.1). The median D-dimer in patients with thromboembolic complications was 11,533 ng/mL (IQR: 4,406–28,382). We did not include vascular access thrombosis as a thrombotic complication.
Outcomes and Factors Associated with Mortality
Table 3 describes the most relevant outcomes recorded during hospitalization. Ten patients died, and 1 additional patient was discharged to hospice for end-of-life care and was counted as a death. Twenty-one patients (33%) required admission to ICU. Fifteen patients required mechanical ventilation (23%). Sixty-six percent of ESKD COVID-19-positive patients were discharged home, and 17% required temporary admission to a physical rehabilitation facility.
Table 3.
Outcomes
| Total (n = 64) | Survivors1 (n = 53) | Deceased1 (n = 11) | p value2 | |
|---|---|---|---|---|
| ICU admission, n (%) | 21 (33) | 12 (23) | 9 (82) | <0.001 |
| Ventilator, n (%) | 15 (23) | 6 (11) | 9 (82) | <0.001 |
| CRRT use, n (%) | 8 (38) | 2 (4) | 6 (55) | <0.001 |
| Hospital days | 10 (5–14) | 10 (5–13) | 11 (6–22) | 0.4 |
CRRT, continuous renal replacement therapy.
Statistics presented: n (%); median (IQR).
Statistical tests performed: Fisher's exact test; Wilcoxon rank-sum test.
The mortality rate of ESKD patients with COVID-19 infection was 17%. As a reference, during the same time period, there were 490 ESKD patients admitted without COVID-19. Thirty-eight of them died during their hospitalization (8%), and 13 were discharged to terminal hospice care (3%) for a mortality rate of 11%. The mortality RR for ESKD patients with COVID-19 was 2.0 (CI: 1.1–3.8) compared to ESKD patients without COVID-19 admitted during the same period.
Age >65 years (RR 13.7, CI: 1.9–100.7), CRP >100 mg/dL (RR 8.3, CI: 1.1–60.4), peak D-dimer >3,000 ng/mL (RR 4.3, CI: 1.03–18.2), bilirubin >1 mg/dL (RR 3.9, CI: 1.5–10.4), and history of peripheral vascular disease (RR 3.2, CI: 1.2–9.1) were associated with mortality. Figure 1 displays values of D-dimer, CRP, and bilirubin on admission and their relationship with hospital mortality.
Discussion
Dialysis patients are particularly vulnerable to poor outcomes related to COVID-19 due to their underlying comorbid illnesses and immunosuppression. Also, dialysis patients are a high-risk population for transmission of COVID-19 due to difficulties to maintain adequate social distancing due to attending dialysis sessions thrice weekly. We report our outcomes of ESKD patients admitted to our academic health system with COVID-19-positive tests in a predominantly African-American population.
Our study highlighted specific risk factors for COVID-19 complications and death that have not been previously reported in the ESKD population. Older age, history of peripheral vascular disease, and liver function abnormalities as well as elevated D-dimer and CRP levels were independent risk factors for death.
The hospital mortality rate seen in our cohort (17%) is similar to that seen in a predominantly ethnic Chinese ESKD cohort in Wuhan (14%) [5]. The Wuhan cohort had a similar age, ICU admission rate, and ventilator usage to ours, but their patients had significantly less diabetes and fewer cardiovascular comorbidities. Our cohort's mortality rate was lower than the 31% seen in a predominantly Hispanic cohort from New York. Average age and comorbidities were similar between the 2 cohorts, but the New York cohort had higher ICU and ventilator use. Predominantly White, European cohorts from Italy, France, and Spain had either similar or older ages (61–72 years), similar comorbidities, and higher mortalities (27–31%) than our cohort [7, 8, 9].
There are several explanations for our mortality being lower than the European cohorts and similar to a cohort with fewer comorbidities in China. First, our average age was lower than the European cohorts, even though comorbidities were similar. Second, our institutions' ICU, ventilator, and dialysis capacity were not overwhelmed at any time during the study period. In support of this explanation, our ICU survival was also significantly higher than other institutions [26]. A third explanation is that our population is predominantly African American as historically African-American ESKD patients have had lower overall mortality rates than whites [27, 28].
One reason often given for improved survival of African Americans on dialysis is greater obesity which has shown a paradoxical relationship with mortality in the dialysis population [27, 28, 29]. Consistent with this paradox, our current report is in contradiction to the general population affected by COVID-19 by not implicating obesity as a risk factor for worse outcomes in COVID-19 [20, 30]. However, the New York, Spanish, and French ESKD COVID-19 cohorts had similar BMI to our patients and also showed no significant relationship of BMI with mortality [2, 7, 8, 9]. All of these results are consistent with the observation that in dialysis patients, racial background does not modify the relationship of BMI with mortality [31]. Thus, obesity does not explain the better survival seen in our cohort.
We experienced a lower prevalence of COVID-19 infection among ESKD patients admitted to the hospital (12%) compared to the Wuhan and Italian cohorts [5, 8]. We had time to implement rigorous infection protocols in most of our dialysis centers early on in the pandemic and may have identified patients earlier in the course of the disease resulting in a reduced risk of more severe complications. Nevertheless, ESKD patients with COVID-19 infection had a RR of death of 2.01 (CI: 1.06–3.84) compared to hospitalized ESKD patients that tested negative for COVID-19.
Activation of the coagulation cascade as measured by D-dimer has emerged as a poor prognostic indicator in patients with COVID-19 [32, 33, 34]. We are the first to report this finding in an ESKD cohort with 10-fold higher peak D-dimer in COVID-19-positive patients who died compared to those who survived. The smaller French and Spanish cohorts [7, 8] showed a trend for higher D-dimer in ESKD patients who died compared to survivors, but the difference did not reach statistical significance. We also found elevated D-dimer levels in both ESKD COVID-19-positive and ESKD COVID-19-negative patients (online suppl. Table B), consistent with the observation that ESKD itself is associated with higher D-dimer levels [35, 36]. Despite not observing any differences in D-dimer, we report a higher risk of thromboembolic complications in COVID-19-positive patients compared to ESKD COVID-19-negative patients. The elevated D-dimer levels documented in ESKD patients [36, 37] have not previously been associated with increased mortality. However, elevated D-dimer levels correlate with the severity of atherosclerotic burden and history of peripheral vascular disease [38, 39, 40, 41, 42]. Studies related to uncovering mechanisms for COVID-19 thrombotic complications are of extreme interest and ongoing [29, 30]. Intensified anticoagulation strategies based exclusively on elevation of D-dimer have not been thoroughly tested in dialysis patients with COVID-19 infection, and concerns about the risk of bleeding have also been raised [43]. It is worth mentioning that the highest D-dimer levels occurred after a median of 5 days of hospitalization. Given the finding of significantly elevated D-dimer levels in our ESKD COVID-19-negative cohort, dedicated studies are needed to answer the issue of appropriate and safe anticoagulation beyond standard care in this population.
Data from French and Spanish cohorts agree with our finding that higher CRP is a powerful marker of mortality. The French cohort found that CRP significantly correlated with death in multivariate analysis along with lactic dehydrogenase and thrombocytopenia [9]. In the Spanish cohort with 36 patients, laboratory values were similar to ours with CRP and bilirubin being significant in both univariate analyses. Age was the dominant effect on mortality in the limited multivariate analysis [7].
In the respiratory tract and vascular endothelium, ACEIs and ARBs upregulate cell surface expression of angiotensin-converting enzyme 2 (ACE2), the entry point for SARS-CoV-2 [44, 45]. However, higher ACE2 expression is associated with better outcomes in most viral pneumonia. Our data provide a signal for potentially worse outcomes in dialysis patients who were on ACEIs and ARBs, but this could be confounding by indication as patients on ACEIs and ARBs likely have a higher burden of cardiovascular disease.
Lower lymphocyte counts are a hallmark of COVID-19 disease and are associated with poor outcomes, although it is unclear if the low counts are pathogenic [46]. One study found worse survival in dialysis patients affected by COVID-19 [7]. Similarly, a lower lymphocyte count has been reported to occur in 50% of the ESKD population [47]. In our study, the absolute lymphocyte count was indeed lower in the ESKD COVID-19 cohort compared to non-COVID-19 patients (online suppl. Table B); however, we did not find an association between lymphopenia and increased risk of mortality in the COVID-19 cohort, perhaps due to our low number of patient deaths.
We report that there were no differences in treatment either in regard to anticoagulation or in steroid use among survivors and nonsurvivors. Anticoagulation was given to patients with higher D-dimer per hospital protocol which could influence this finding. Steroids are recognized now as a potential beneficial therapeutic agent [48] but were not used during the time period of our report. No dialysis patient was offered remdesivir, and only 6 patients received hydroxychloroquine while hospitalized.
The low number of deaths likely limited our ability to detect more statistically significant differences in other factors typically associated with worse clinical outcomes in dialysis patients. The lack of racial diversity in this cohort also limits the determination of racial differences in risk factors and outcomes. However, our predominantly African-American population is a strength because it is compatible with the overrepresentation of African Americans in the non-ESKD population affected by COVID-19 and allows identification of potential risk factors that may be particularly informative for future mitigation strategies in this high-risk minority population. Our patient population was a 94% hemodialysis modality patient population which was similar to the ESRD Network 6 with a prevalence of 89% [49]. Lastly, the availability of detailed demographic and clinical variables allowed us to provide a more comprehensive assessment of putative risk factors for mortality not previously reported.
Conclusion
We report our experience caring for predominantly African-American ESKD patients hospitalized with COVID-19. Mortality rates are lower than other cohorts, and older age, history of peripheral vascular disease, and elevated CRP, bilirubin, and D-dimer emerged as important risk factors for death. Better understanding of risk stratification could potentially lead to more precise targeting of therapies for COVID-19 in ESKD. The finding of increased D-dimer and lymphopenia in the COVID-19-negative dialysis patients raises questions about the role of these abnormalities in the general ESKD population. In summary, our findings provide guidance on risk stratification for the vulnerable African-American ESKD population and reinforce the need for dialysis centers to remain vigilant in following appropriate infection control measures to prevent the spread of the coronavirus.
Statement of Ethics
This study was approved by our institutional review board.
Conflict of Interest Statement
The authors have no conflicts of interest to declare and have no financial disclosures.
Funding Sources
No funding was received for this work.
Author Contributions
Jose E. Navarrete: conception, design, data collection, analysis, interpretation of data, manuscript drafting, manuscript revision, and lead author. David C. Tong: conception, design, data collection, analysis, interpretation of data, and manuscript drafting. Jason Cobb: conception, design, data collection, interpretation of data, manuscript drafting, manuscript revision, and corresponding author. Frederic F. Rahbari-Oskoui: conception, design, interpretation of data, manuscript drafting, and manuscript revision. Darya Hosein: design, analysis, data collection, interpretation of data, and manuscript drafting. Sheryl C. Caberto: data collection, interpretation of data, and manuscript drafting. Janice P. Lea: conception, design, interpretation of data, and manuscript drafting. Harold A. Franch: conception, design, analysis, interpretation of data, manuscript drafting, and manuscript revision.
References
- 1.Watnick S, McNamara E. On the frontline of the COVID-19 outbreak: keeping patients on long-term dialysis safe. Clin J Am Soc Nephrol. 2020;15((5)):710–3. doi: 10.2215/CJN.03540320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Valeri AM, Robbins-Juarez SY, Stevens JS, Ahn W, Rao MK, Radhakrishnan J, et al. Presentation and outcomes of patients with ESKD and COVID-19. J Am Soc Nephrol. 2020;31((7)):1409–15. doi: 10.1681/ASN.2020040470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ma Y, Diao B, Lv X, Zhu J, Chen C, Liu L, et al. Epidemiological, clinical, and immunological features of a cluster of COVID-19-contracted hemodialysis patients. Kidney Int Rep. 2020;5((8)):1333–41. doi: 10.1016/j.ekir.2020.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang R, Liao C, He H, Hu C, Wei Z, Hong Z, et al. COVID-19 in hemodialysis patients: a report of 5 cases. Am J Kidney Dis. 2020;76((1)):141–3. doi: 10.1053/j.ajkd.2020.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu J, Li J, Zhu G, Zhang Y, Bi Z, Yu Y, et al. Clinical features of maintenance hemodialysis patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. Clin J Am Soc Nephrol. 2020;15((8)):1139–45. doi: 10.2215/CJN.04160320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rombolà G, Heidempergher M, Pedrini L, Farina M, Aucella F, Messa P, et al. Practical indications for the prevention and management of SARS-CoV-2 in ambulatory dialysis patients: lessons from the first phase of the epidemics in Lombardy. J Nephrol. 2020;33((2)):193–6. doi: 10.1007/s40620-020-00727-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Goicoechea M, Sánchez Cámara LA, Macías N, Muñoz de Morales A, Rojas ÁG, Bascuñana A, et al. COVID-19: clinical course and outcomes of 36 hemodialysis patients in Spain. Kidney Int. 2020;98((1)):27–34. doi: 10.1016/j.kint.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Alberici F, Delbarba E, Manenti C, Econimo L, Valerio F, Pola A, et al. A report from the Brescia renal COVID task force on the clinical characteristics and short-term outcome of hemodialysis patients with SARS-CoV-2 infection. Kidney Int. 2020;98((1)):20–6. doi: 10.1016/j.kint.2020.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tortonese S, Scriabine I, Anjou L, Loens C, Michon A, Benabdelhak M, et al. COVID-19 in patients on maintenance dialysis in the Paris region. Kidney Int Rep. 2020 Sep;5((9)):1535–44. doi: 10.1016/j.ekir.2020.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xiong F, Tang H, Liu L, Tu C, Tian JB, Lei CT, et al. Clinical characteristics of and medical interventions for COVID-19 in hemodialysis patients in Wuhan, China. J Am Soc Nephrol. 2020;31((7)):1387–97. doi: 10.1681/ASN.2020030354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ikizler TA, Kliger AS. Minimizing the risk of COVID-19 among patients on dialysis. Nat Rev Nephrol. 2020;16((6)):311–3. doi: 10.1038/s41581-020-0280-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Corbett RW, Blakey S, Nitsch D, Loucaidou M, McLean A, Duncan N, et al. Epidemiology of COVID-19 in an Urban dialysis center. J Am Soc Nephrol. 2020;31((8)):1815–23. doi: 10.1681/ASN.2020040534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Clarke C, Prendecki M, Dhutia A, Ali MA, Sajjad H, Shivakumar O, et al. High prevalence of asymptomatic COVID-19 infection in hemodialysis patients detected using serological screening. J Am Soc Nephrol. 2020 Sep;31((9)):1969–75. doi: 10.1681/ASN.2020060827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Norris KC, Williams SF, Rhee CM, Nicholas SB, Kovesdy CP, Kalantar-Zadeh K, et al. Hemodialysis disparities in African Americans: the deeply integrated concept of race in the social fabric of our society. Semin Dial. 2017;30((3)):213–23. doi: 10.1111/sdi.12589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Umeukeje EM, Young BA. Genetics and ESKD disparities in African Americans. Am J Kidney Dis. 2019;74((6)):811–21. doi: 10.1053/j.ajkd.2019.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Saran R, Robinson B, Abbott KC, Agodoa LYC, Bragg-Gresham J, Balkrishnan R, et al. United States renal data system. 2019 USRDS annual data report: epidemiology of kidney disease in the United States. Am J Kidney Dis. 2019;73((Suppl 1)):A7–A8. doi: 10.1053/j.ajkd.2019.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gold JAW, Wong KK, Szablewski CM, Patel PR, Rossow J, da Silva J, et al. Characteristics and clinical outcomes of adult patients hospitalized with COVID-19: Georgia, March 2020. MMWR Morb Mortal Wkly Rep. 2020;69((18)):545–50. doi: 10.15585/mmwr.mm6918e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mohamed MMB, Lukitsch I, Torres-Ortiz AE, Walker JB, Varghese V, Hernandez-Arroyo CF, et al. Acute kidney injury associated with coronavirus disease 2019 in Urban New Orleans. Kidney360. 2020;1((7)):614–22. doi: 10.34067/KID.0002652020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Suleyman G, Fadel RA, Malette KM, Hammond C, Abdulla H, Entz A, et al. Clinical characteristics and morbidity associated with coronavirus disease 2019 in a series of patients in metropolitan detroit. JAMA Netw Open. 2020;3((6)):e2012270. doi: 10.1001/jamanetworkopen.2020.12270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Stefan N, BirkenfeldBirkenfeld ALAL, Schulze MB, Ludwig DS. Obesity and impaired metabolic health in patients with COVID-19. Nat Rev Endocrinol. 2020;16((7)):341–2. doi: 10.1038/s41574-020-0364-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Crews DC, Purnell TS. COVID-19, racism, and racial disparities in kidney disease: galvanizing the kidney community response. J Am Soc Nephrol. 2020;31((8)):1–3. doi: 10.1681/ASN.2020060809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kullar R, Marcellin JR, Swartz TH, Piggott DA, Macias Gil R, Mathew TA, et al. Racial disparity of coronavirus disease 2019 in African American Communities. J Infect Dis. 2020;222:890–3. doi: 10.1093/infdis/jiaa372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Centor RM, Essien UR. Disparities and COVID-19. Ann Intern Med. 2020;16:172. doi: 10.7326/A19-0032. [DOI] [PubMed] [Google Scholar]
- 24.Johnson-Mann C, Hassan M, Johnson S. COVID-19 pandemic highlights racial health inequities. Lancet Diabetes Endocrinol. 2020;8((8)):663–4. doi: 10.1016/S2213-8587(20)30225-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40((5)):373–83. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 26.Auld SC, Caridi-Scheible M, Blum M, Robichaux C, Kraft C, Jacob JT, et al. ICU and ventilator mortality among critically ill adults with coronavirus disease 2019. Crit Care Med. 2020;48:e799–e804. doi: 10.1097/CCM.0000000000004457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Held PJ, Pauly MV, Diamond L. Survival analysis of patients undergoing dialysis. JAMA. 1987;257((5)):645–50. [PubMed] [Google Scholar]
- 28.Abbott KC, Glanton CW, Trespalacios FC, Oliver DK, Ortiz MI, Agodoa LY, et al. Body mass index, dialysis modality, and survival: analysis of the United States renal data system dialysis morbidity and mortality Wave II Study. Kidney Int. 2004;65((2)):597–605. doi: 10.1111/j.1523-1755.2004.00385.x. [DOI] [PubMed] [Google Scholar]
- 29.Kalantar-Zadeh K, Abbott KC, Salahudeen AK, Kilpatrick RD, Horwich TB. Survival advantages of obesity in dialysis patients. Am J Clin Nutr. 2005;81((3)):543–54. doi: 10.1093/ajcn/81.3.543. [DOI] [PubMed] [Google Scholar]
- 30.Price-Haywood EG, Burton J, Fort D, Seoane L. Hospitalization and Mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382((26)):2534–43. doi: 10.1056/NEJMsa2011686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Park J, Jin DC, Molnar MZ, Dukkipati R, Kim YL, Jing J, et al. Mortality predictability of body size and muscle mass surrogates in Asian vs. white and African American hemodialysis patients. Mayo Clin Proc. 2013;88((5)):479–86. doi: 10.1016/j.mayocp.2013.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.BikdeliBikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, et al. COVID-19 and thrombotic or thromboembolic disease: implications for prevention, antithrombotic therapy, and follow-up: JACC State-of-the-Art review. J Am Coll Cardiol. 2020;75:2950–73. doi: 10.1016/j.jacc.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Levi M, Thachil J, Iba T, Levy JH. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020;7((6)):e438–40. doi: 10.1016/S2352-3026(20)30145-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guan WJ, Ni ZY, Hu Y, Guan WJ, Ni ZY, Hu Y, et al. China medical treatment expert group for Covid-19: clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–20. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miozzari M, Wahl C. D-dimers in hemodialysis patients. Nephron. 2001;88((3)):278–9. doi: 10.1159/000046004. [DOI] [PubMed] [Google Scholar]
- 36.Gordge MP, Faint RW, Rylance PB, Ireland H, Lane DA, Neild GH. Plasma D dimer: a useful marker of fibrin breakdown in renal failure. Thromb Haemost. 1989;61((3)):522–5. [PubMed] [Google Scholar]
- 37.Spring JL, Winkler A, Levy JH. The influence of various patient characteristics on D-dimer concentration in critically ill patients and its role as a prognostic indicator in the intensive care unit setting. Clin Lab Med. 2014;34((3)):675–86. doi: 10.1016/j.cll.2014.06.015. [DOI] [PubMed] [Google Scholar]
- 38.Gubensek J, Lolic M, Ponikvar R, Buturovic-Ponikvar J. D-dimer levels in maintenance hemodialysis patients: high prevalence of positive values also in the group without predisposing diseases. Hemodial Int. 2016;20((2)):198–203. doi: 10.1111/hdi.12371. [DOI] [PubMed] [Google Scholar]
- 39.Dubin R, Cushman M, Folsom AR, Fried LF, Palmas W, Peralta CA, et al. Kidney function and multiple hemostatic markers: cross sectional associations in the multi-ethnic study of atherosclerosis. BMC Nephrol. 2011;12((3)):3. doi: 10.1186/1471-2369-12-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lassila R, Peltonen S, Lepäntalo M, Saarinen O, Kauhanen P, Manninen V. Severity of peripheral atherosclerosis is associated with fibrinogen and degradation of cross-linked fibrin. Arterioscler Thromb. 1993;13((12)):1738–42. doi: 10.1161/01.atv.13.12.1738. [DOI] [PubMed] [Google Scholar]
- 41.Lee AJ, Fowkes FG, Lowe GD, Rumley A. Fibrin D-dimer, haemostatic factors and peripheral arterial disease. Thromb Haemost. 1995;74((3)):828–32. [PubMed] [Google Scholar]
- 42.Milburn JA, Ford I, Cassar K, Fluck N, Brittenden J. Platelet activation, coagulation activation and C-reactive protein in simultaneous samples from the vascular access and peripheral veins of haemodialysis patients. Int J Lab Hematol. 2012;34((1)):52–8. doi: 10.1111/j.1751-553X.2011.01356.x. [DOI] [PubMed] [Google Scholar]
- 43.Al-Samkari H, Karp Leaf RS, Dzik WH, Carlson JCT, Fogerty AE, Waheed A, et al. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136((4)):489–500. doi: 10.1182/blood.2020006520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Messerli FH, Siontis GCM, Rexhaj E. COVID-19 and renin angiotensin blockers: current evidence and recommendations. Circulation. 2020;141((25)):2042–4. doi: 10.1161/CIRCULATIONAHA.120.047022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fosbøl EL, Butt JH, Østergaard L, Andersson C, Selmer C, Kragholm K, et al. Association of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker use with COVID-19 diagnosis and mortality. JAMA. 2020;324:168–77. doi: 10.1001/jama.2020.11301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Huang I, Pranata R. Lymphopenia and severe coronavirus disease-2019 (COVID-19): systematic review and meta-analysis. J Intensive Care. 2020;8:36. doi: 10.1186/s40560-020-00453-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Agarwal R, Light RP. Patterns and prognostic value of total and differential leukocyte count in chronic kidney disease. Clin J Am Soc Nephrol. 2011;6((6)):1393–9. doi: 10.2215/CJN.10521110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, et al. Dexamethasone in hospitalized patients with covid, preliminary report. N Engl J Med. 2021 Feb 25;384((8)):693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.United States Renal Data System . 2018 USRDS annual data report: epidemiology of kidney disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; 2018. [Google Scholar]