Abstract
Background
Histopathological analysis can provide additional clues in COVID-19 understanding. During the last year, autopsy reports have revealed that diffuse alveolar damage (DAD) is the most significant observed finding. The aim of this study is to review cases in the literature about COVID-19 autopsies that reported microthrombi in different organs.
Methods
We performed a systematic literature review in PubMed, Virtual Health Library (VHL), and Google Scholar.
Results
In total, 151 autopsies were included, and 91 cases presented microthrombi in the lung (73%), heart (11.2%), kidney (24%), and liver (16.3%). The age range was between 27 and 96 years. Males were 64.8%. The patients with microthrombi had more comorbidities such as arterial hypertension (62%), obesity or overweight (64%), diabetes mellitus type 2 (51%), and heart disease (53%). The most common histopathological changes found in patients with lung microthrombosis were DAD in exudative phase (78%), pulmonary embolism (59%), and lung infarct (81%). Presence of microthrombi was associated with arterial hypertension (p < 0.0001) and DAD in exudative and proliferative phases (p = 0.02).
Discussion
The analysis of these results shows that microthrombi in COVID-19 autopsies may be found in different organs and are more frequent in patients with comorbidities, pulmonary embolism, and lung infarct.
Keywords: Autopsy, COVID-19, Microthrombi
Introduction
On January 30, 2020, the WHO declared COVID-19 a global pandemic. COVID-19 is caused by SARS-CoV-2, a type of coronavirus that can affect humans. SARS-CoV-2 shares 79.6% genome sequence with SARS-Cov and infects the same human lung alveolar epithelial cells through receptor-mediated endocytosis using the ACE2 receptor [1].
The symptoms of COVID-19 infection frequently appear after an incubation period of 1–14 days, commonly lasting 3–7 days but can extend up to 24 days. The most common symptoms that have been reported are fever (88.7%), cough (67.8%), fatigue (38.1%), sputum production (33.7%), shortness of breath (18.7%), sore throat (13.9%), and headache (13.6%). Some COVID-19 patients presented gastrointestinal symptoms such as diarrhea (3.8%) and vomiting (5.0%). In severe cases, patients may develop pneumonia, acute respiratory distress syndrome, acute cardiac problems, and multiorgan failure [2].
Many scientific studies have focused in understanding the viral infection mechanism; however, a histopathological analysis could uncover additional clues about COVID-19 [3]. Throughout history, autopsies have played a very important role in identifying emerging and re-emerging infectious diseases [4]. In the last months, several changes associated with the virus infection or as a secondary effect of the treatment have been reported in the respiratory, cardiovascular, urinary, gastrointestinal, reproductive, and immune systems, among others [5, 6]. Polak et al. [3] published a systematic review about the observed changes in COVID-19 autopsies. Their recount was mainly focused on pulmonary pathological findings, proposing to categorize changes as epithelial, vascular, or fibrotic. They also found that an epithelial pattern of lung injury occurs early in the course of the disease, even before the onset of symptoms in some cases, and persisting throughout the clinical course, but gradually declining by 28 days after the onset of symptoms.
During the COVID-19 pandemic, autopsy findings have contributed to a better comprehension of the disease and to develop therapeutic options considering the presence of thrombotic events that might be an indication of a serious derangement of the clotting balance. We have performed a systematic review to discuss the location and findings associated with evidence of microthrombi in COVID-19 autopsies.
Materials and Methods
Search Strategy
A systematic review of PubMed and Virtual Health Library (VHL), from inception through March 30, 2020, was performed to find articles providing information about autopsy findings in patients infected with SARS-CoV-2. The following strings were used to do the search: (SARS-CoV-2 or COVID-19) and autopsy. Manual search was performed in Google (Google LLC, Mountain View, CA, USA). Included studies were available in English and Spanish. Preprint manuscripts in databases such as bioRxiv and medRxiv were also considered. Articles published until September 30, 2020, were manually added to acquire more data.
Study Selection and Data Extraction
Case reports or series cases with histopathological findings in autopsies of patients infected with COVID-19 and presence of microthrombi were included. Three authors (R.P.M., S.H., and J.M.) independently screened the articles and extracted relevant information. Disagreements about source relevance were resolved by consensus. The following data were extracted from the autopsies that presented microthrombi: author, number of cases, total number of cases, comorbidities, and histopathological findings. Articles without this information on each case were removed.
Due to heterogeneity of terms used in the studies, we grouped those terms into two diffuse alveolar damage (DAD) phases. Terms such as hyaline membranes, interstitial and intra-alveolar edema, collapsed alveoli, and necrosis of endothelial cells were grouped as the exudative phase. Terms such as organizing or remnants of the hyaline membrane, interstitial and intra-alveolar proliferation of fibroblasts or myofibroblasts, and proliferation of type II pneumocyte were grouped as the proliferative phase.
Data Synthesis and Analysis
Univariate analysis was applied to determine distribution of clinical and pathological findings. Based on the presence or absence of microthrombi, the data were divided into groups, and the χ2 test was used to determine statistically significant differences between microthrombi and clinical and pathological findings. Statistical significance was established at p < 0.05.
Results
Systematic Review
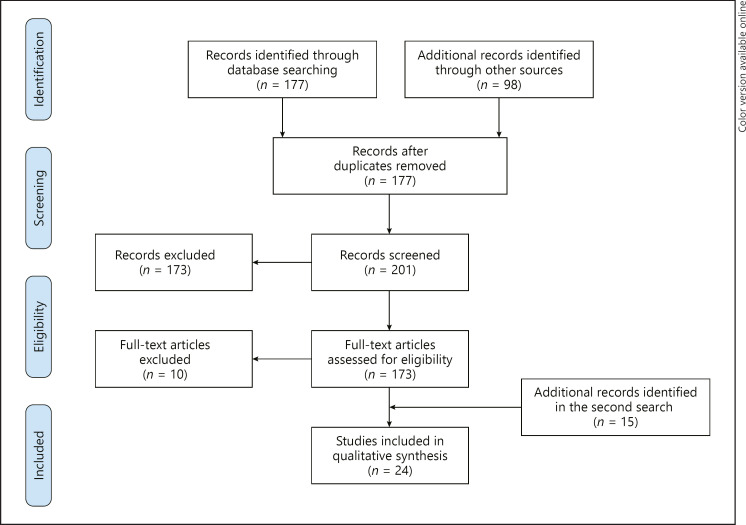
A total of 275 articles within the reviewed databases were retrieved, and 74 of those articles were recognized as duplicates. A total of 201 full-text articles were assessed for eligibility (COVID-19 autopsies). Twenty-eight full-text articles with COVID-19 autopsies were reviewed [7]. Fifteen articles were added in the second search. Finally, 24 articles that contained interpretable data and fulfilled the eligibility criteria were included (microthrombi observed in the histopathology with established characteristics in each patient were included) [8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29]. From the included articles, 18 were case reports and 6 had >10 cases. The flowchart for the systematic literature reviewed and articles included in the analysis is shown in Figure 1.
Fig. 1.
Flowchart of the systemic literature review.
Characteristics of Patients with Microthrombi
In total, 151 autopsies were included. Ninety-one autopsies detected microthrombi. Eighty-five autopsies had cases with microthrombi in the lung, 9 in the heart, 13 in the kidneys, 7 in the liver, and 2 in the trachea. The age range was between 27 and 96 years. 64.8% of the patients were male (n = 59). The following comorbidities were reported: 61.5% cases with arterial hypertension, 63.6% cases with obesity or overweight, 52.3% cases with diabetes mellitus, and 52.9% cases with heart disease. Other conditions also reported are listed in Table 1 and online suppl. Table 1 (see www.karger.com/doi/10.1159/000515104 for all online suppl. material). The statistical analysis showed association between arterial hypertension (p < 0.0001) and the presence of microthrombi.
Table 1.
Characteristics of patients included in the review
| Microthrombi (n = 91) | % | Non-microthrombi (n = 60) | % | Total (n =151) | p value | |
|---|---|---|---|---|---|---|
| Age, range, years | 27–96 | 39–94 | 34–96 | |||
| Gender* | ||||||
| Male | 59 | 57.8 | 43 | 42.2 | 102 | 0.66 |
| Female | 28 | 62.2 | 17 | 37.8 | 45 | |
| Comorbidities | ||||||
| Asthma | 5 | 83.3 | 1 | 16.7 | 6 | 0.07 |
| Cancer | 6 | 33.3 | 12 | 66.7 | 18 | 0.24 |
| Heart disease | 18 | 52.9 | 16 | 47.1 | 34 | 0.13 |
| Respiratory disease | 10 | 52.6 | 9 | 47.4 | 19 | 0.66 |
| Diabetes mellitus type 2 | 20 | 51.3 | 19 | 48.7 | 39 | 0.94 |
| Dementia | 5 | 62.5 | 3 | 37.5 | 8 | 0.24 |
| Arterial hypertension | 59 | 61.5 | 37 | 38.5 | 96 | <0.0001 |
| Obesity or overweight | 42 | 63.6 | 24 | 36.4 | 66 | 0.39 |
| Smoking | 8 | 38.1 | 13 | 61.9 | 21 | 0.02 |
| Histopathological findings | ||||||
| Lung | ||||||
| DAD, exudative | 25 | 78.2 | 7 | 21.8 | 32 | 0.46 |
| DAD, proliferative | 8 | 100 | 0 | 0 | 8 | 0.10 |
| DAD, exudative and proliferative | 32 | 62.8 | 19 | 37.2 | 51 | 0.02 |
| DAD, nonclassified | 11 | 55 | 9 | 45 | 20 | 0.04 |
| DAD, with fibrotic changes | 4 | 100 | 0 | 0 | 4 | 0.28 |
| Pulmonary embolism | 16 | 59.3 | 11 | 40.7 | 27 | 0.06 |
| Lung infarct | 13 | 81.2 | 3 | 18.8 | 16 | 0.6 |
| Emphysema | 2 | 16.7 | 10 | 83.3 | 12 | <0.0001 |
| Cytopathic viral effect | 8 | 80 | 2 | 20.0 | 10 | 0.46 |
| Bronchopneumonia (focal or diffuse) | 23 | 45.1 | 28 | 54.9 | 51 | <0.0001 |
| Vasculitis | 9 | 100 | 0 | 0 | 9 | 0.05 |
| Heart | ||||||
| Myocardial hypertrophy | 25 | 49 | 26 | 51 | 51 | 0.04 |
| Interstitial fibrosis | 11 | 40.7 | 16 | 59.3 | 27 | 0.03 |
| Myocarditis | 2 | 100 | 0 | 0 | 2 | 0.32 |
| Pericarditis | 1 | 50 | 1 | 50 | 2 | 0.67 |
| Kidney | ||||||
| Acute tubular damage | 19 | 38 | 31 | 62 | 50 | 0.17 |
| Diabetic nephropathy | 1 | 25 | 3 | 75 | 4 | 0.18 |
| Arteriosclerosis | 19 | 36.5 | 33 | 63.5 | 52 | 0.06 |
| Liver | ||||||
| Steatosis | 31 | 68.9 | 14 | 31.1 | 45 | 0.25 |
| Shock necrosis | 4 | 80 | 1 | 20 | 5 | 0.51 |
| Lymphocytic inflammatory infiltrate | 5 | 71.4 | 2 | 28.6 | 7 | 0.59 |
| Centrilobular congestion | 6 | 37.5 | 10 | 62.5 | 16 | 0.004 |
| Spleen | ||||||
| Reduce white pulp | 2 | 40 | 3 | 60 | 5 | 0.32 |
| Red pulp hemorrhages | 3 | 100 | 0 | 0.00 | 3 | 0.20 |
DAD, diffuse alveolar damage. Values in bold indicate statistical significance.
Data not available for Fox et al. [18].
Lung
All autopsies evaluated lung tissue [8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29] and showed DAD in different stages. DAD in the exudative phase was reported in 32 patients and DAD in the proliferative phase in 8 patients. Fifty-one cases showed changes in both exudative and proliferative phases. In 20 cases, the DAD could not be further classified. Damiani et al. [16] classified 4 patients with features of a more advanced proliferative phase of DAD (late fibrotic disease). Bronchopneumonia was observed in 32 cases. Cases with coinfections such as Candida glabrata, Pseudomonas aeruginosa, and Staphylococcus epidermidis, among others, were reported [16].
Microthrombosis was reported in 73% (85/116 cases), and 78% had DAD in the exudative phase and 63% had DAD in the exudative and proliferative phase (p = 0.02). Fifty-nine percent had pulmonary embolism and 81% had lung infarct, and in 100% of the cases, vasculitis was reported.
Heart
Heart tissue was evaluated in 10 articles (59 patients) [10, 11, 12, 13, 21, 23, 25, 27, 28, 30]. Fifty-one cases showed changes associated with pre-existing pathologies such as myocardial hypertrophy and myocardial interstitial fibrosis. In 9 patients (11.2%), microthrombi in the heart were reported [14, 25, 30]. The range of the age in these patients was between 27 and 64 years. Five were male, and 6 had arterial hypertension and 4 obesity. They presented nonspecific symptoms like fever, chills, dyspnea, cough, and depression.
Kidney
Kidney tissue was evaluated in 7 articles (n = 62 patients) [9, 11, 12, 23, 27, 30, 31]. Acute tubular damage was the most common finding. Arteriosclerosis was observed in 49 cases, and microthrombi were detected in the glomeruli of 13 cases (24%). In several cases, viral particles in some glomerular endothelial cells were identified.
Liver
Liver tissue was evaluated in 7 articles (n = 66) [11, 12, 23, 25, 27, 30, 32]. Steatosis was reported in 45 cases, centrilobular congestion in 16 cases, and necrosis in 5 cases. Rapkiewicz et al. [26] found 7 cases with microthrombi (16.3%), one with cirrhosis and the other case with zone 3 necrosis with a hepatic vein thrombus.
Spleen
Three articles reported spleen findings (n = 23) [11, 12, 25]. Five cases showed reduced white pulp, and 3 cases had red pulp hemorrhages.
Discussion
Autopsy is a valuable medical tool in understanding the progression of disease which can eventually lead to the investigation of new therapeutic options. In the present review, we found the presence of microthrombi in 60% of autopsies. Microthrombi were detected in the lung, trachea, heart, kidneys, and liver along with other variable findings that may be related with the virus infection or secondary to treatment. We found that the patients with microthrombi had more comorbidities, namely, arterial hypertension (62%), obesity or overweight (64%), diabetes mellitus type 2 (51%), and heart disease (53%). In the statistical analysis, the presence of microthrombi was significantly associated with a clinical history of arterial hypertension (p < 0.0001) (Table 1).
DAD in different stages was a major finding. Of the patients with microthrombosis, 87% had DAD in the exudative phase and 67% had DAD in the exudative and proliferative phase. The lung injury may explain the ventilation-perfusion alteration that these patients have. The SARS-CoV-2 virus may induce more severe alveolar epithelial changes compared with the other respiratory viruses [33]. DAD associated with the SARS-CoV-2 virus has shown similarities with other coronaviruses (SARS-CoV-1 and Middle East respiratory syndrome) at the histopathological level [33]. In contrast to the SARS-CoV-2 virus, in H1N1 virus autopsies, more extensive necrosis and hemorrhage in the lungs were observed. Also, there were more CD8+ T cells, CD57 NK cells, and granzyme B+ cells [34] compared to the SARS-CoV-2 virus that usually shows paucity of CD8+ T cells and CD57+ NK cells [33]. We observed microthrombi in 73% of COVID-19 patients, while in the literature, it has been observed in 58% of SARS-CoV-1 and 24% of H1N1 influenza patients [35, 36].
Thrombi formation in severe COVID-19 may be due several factors such as upregulation of procoagulant mechanisms, downregulation of natural anticoagulants, resistance to fibrinolysis, and endothelial damage [37]. D-dimer is a product of the coagulation with clinical utility; this is a fibrin degradation product that is present in the blood following degradation of blood clots by fibrinolysis. D-dimer is a sensitive test to diagnose thrombotic states, including pulmonary embolism and disseminated intravascular coagulation [38, 39]. A systematic review and meta-analysis showed that serum D-dimer concentrations in patients with severe COVID-19 are significantly higher than the patients with less severe forms [38].
The endothelial damage in severe COVID-19 can be assimilated to a vasculopathy syndrome characterized by microvasculitis, endothelial degeneration, and resultant basement membrane zone disruption and reduplication [40]. Magro et al. [40, 41] found microthrombi formation in the lung, kidneys, brain, heart, skin, and liver associated with thrombotic vasculopathy and with complement system activation (C4d deposits, terminal complement components C5b-9 [membrane attack complex], and mannose-binding lectin-associated serine protease 2). The complement system can be directly activated by the virus or by the proinflammatory-associated response [42]; the endothelial injury and proinflammatory cytokines can influence the development of vasculitis [43]. Besides, the complement system is an associated factor to the hypercoagulable status in SARS-CoV-2 infection [42].
Leukocytoclastic vasculitis has been reported in COVID-19 patients; in our data, all patients with reported vasculitis had microthrombosis. The inflammatory cells may be cuffed around (periarteritis) and/or inside (panarteritis) the affected vascular wall [44]. Endothelial damage may be directly caused by virus infection [45] or by the systematic secondary inflammatory response that ensues [43]. These features along with the pre-existing endothelial dysfunction associated with chronic diseases (hypertension, diabetes mellitus, dyslipidemia, and chronic kidney disease, among others) [46] could explain the severity seen in certain COVID-19 patients [47]. However, some cases had thrombi formation without atherosclerosis [26]. Ackermann et al. [45] found SARS-CoV-2 virus particles within the endothelial cells and significant changes in the morphology of ACE-positive endothelial cells. The changes found were disruption of intercellular junctions, cell swelling, and loss of contact with the basal membrane. They also found distorted vascularity with structurally deformed capillaries, the presence of sprouting, and intussusceptive angiogenesis. The finding of intussusceptive angiogenesis was significantly correlated with increasing duration of hospitalization (p < 0.001) [45].
Megakaryocytes and platelets are also involved in thrombi formation. Rapkiewicz et al. [26] found 7 cases with high number of megakaryocytes in the cardiac microvasculature, glomeruli, and lungs associated with microthrombi in the lung, trachea, heart, kidneys, and liver. Four of these 7 patients were treated with anticoagulation (enoxaparin [n = 3] or unfractionated heparin [n = 3]) and/or antiplatelet treatment (aspirin [n = 1]). Megakaryocytes count increased in COVID-19 hearts compared to other acute respiratory distress syndromes (p = 0.02). In the bone marrow, megakaryocytes were mildly increased with morphologic features indicating active platelet production and contained virions as identified by electron microscopy.
Neutrophil extracellular traps (NETs) are another factor that interact with the coagulation activation due to the infection [48]. The presence of NETs has been observed in different diseases [49, 50] like in acute respiratory distress syndrome [50]. In COVID-19 patients, high levels of NETs in the blood have been observed [51]. Other factors contributing to coagulation have been found in different studies. Zuo et al. [52] found elevated levels of cell-free DNA, myeloperoxidase-DNA, and citrullinated histone H3. Buja et al. [12] found, in electron microscopy, precipitated fibrin and entrapped neutrophils within alveolar capillaries as well as larger deposits of fibrin in alveolar spaces.
Pulmonary embolism was present in 59% of patients with microthrombi in the lungs. Pulmonary embolism occurred in hospitalized patients and in patients who died in an outpatient or domestic setting [53]. In the cohort of Edler et al. [53], there were 76 cases classified as COVID-19 deaths. On those cases, the most frequent cause of death was pneumonia followed by pulmonary artery embolism combined with pneumonia. Pulmonary embolism has been found with absence of coexisting deep venous thrombosis [28, 53]. The presence of deep venous thrombosis has been associated with a higher rate of admissions to the intensive care unit (p = 0005) and more deaths (p = 0.001) [54]. In the meta-analysis conducted by Henrina et al. [55], which included 1,237 subjects, they found that venous thromboembolism in patients hospitalized was associated with higher mortality (RR 2.48 [1.35, 4.55], p = 0.003), intensive care unit admission (RR 2.32 [1.35, 4.55], p < 0.0001), and mechanical ventilation (RR 2.73 [1.56, 4.78], p = 0.001). The presence of macrothrombi has also been reported in the cerebral venous sinus [56], prostatic venous plexus [28, 53], esophageal veins [53], liver central vein [57], arterial thrombosis in the spleen [33], abdominal aorta [58], aortoiliac, low inguinal, upper limb vessels [59], and testis [12, 33].
We realize that there are limitations in the present study. First, the clinical characteristics of the included cases could not be thoroughly retrieved. Second, almost every case report and serial cases were limited to the changes observed in the heart, liver, or kidneys. Few studies had complete autopsies; in consequence, information from other organs is rather fragmented. Third, a comprehensive statistical analysis is hampered due to the dispersed and fragmented information in the articles reviewed. Fourth, the clinical information and the histopathological characteristics of the case reports or the series of cases were limited. Consequently, the quality of each article was not assessed as it was undesirable [60]. However, valuable information has been collected which we deem useful to advance in the understanding of the disease.
In conclusion, our systematic review provides a summary of cases with fibrin microthrombi in COVID-19 autopsies. We found microthrombi in the lung, trachea, heart, kidneys, and liver. Patients with microthrombi had more comorbidities (hypertension, obesity or overweight, diabetes mellitus type 2, and heart disease) and pulmonary embolism, lung infarct, and vasculitis. The development of thrombi formation is due to the activation of megakaryocytes, platelets, complement system, endothelial injury, and changes induced by severe inflammation, which constitute a complex vasculopathy syndrome. The multiorgan thrombosis in COVID-19 patients despite the anticoagulation therapy may explain the symptoms, signs, and laboratory tests present in different stages of the disease. Therefore, it is crucial to consider adequate therapeutic strategies to prevent thrombosis formation and avoid certain side effects of the infection.
Statement of Ethics
Ethical approval was not required because this study is a systematic review.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
The authors did not receive any funding.
Author Contributions
Rafael Parra-Medina: acquisition, analysis, and interpretation of data/references and drafting and approving the manuscript. Sabrina Herrera: acquisition, analysis, and interpretation of data/references and approving the manuscript. Jaime Mejia: analysis, interpretation of data, and approving the manuscript.
Acknowledgments
In honor of Rafael Parra Puccetti. The manuscript was supported by Fundación Universitaria de Ciencias de la Salud (FUCS).
References
- 1.Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020 Mar;579((7798)):270–3. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tu H, Tu S, Gao S, Shao A, Sheng J. The epidemiological and clinical features of COVID-19 and lessons from this global infectious public health event. J Infect. 2020 Jul;81((1)):1–9. doi: 10.1016/j.jinf.2020.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Polak SB, Van Gool IC, Cohen D, von der Thüsen JH, van Paassen J. A systematic review of pathological findings in COVID-19: a pathophysiological timeline and possible mechanisms of disease progression. Mod Pathol. 2020 Nov;33((11)):2128–38. doi: 10.1038/s41379-020-0603-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Schwartz DA, Herman CJ. The importance of the autopsy in emerging and reemerging infectious diseases. Clin Infect Dis. 1996;23((2)):248–54. doi: 10.1093/clinids/23.2.248. [DOI] [PubMed] [Google Scholar]
- 5.Deshmukh V, Motwani R, Kumar A, Kumari C, Raza K. Histopathological observations in COVID-19: a systematic review. J Clin Pathol. 2021 Feb;74((2)):76–83. doi: 10.1136/jclinpath-2020-206995. [DOI] [PubMed] [Google Scholar]
- 6.Hanley B, Naresh KN, Roufosse C, Nicholson AG, Weir J, Cooke GS, et al. Histopathological findings and viral tropism in UK patients with severe fatal COVID-19: a post-mortem study. Lancet Microbe. 2020;1((6)):e245–53. doi: 10.1016/S2666-5247(20)30115-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Parra-Medina R, Herrera S, Mejía J. Comments to: a systematic review of pathological findings in COVID-19: a pathophysiological timeline and possible mechanisms of disease progression. Mod Pathol. 2020 Jul 21;:1–2. doi: 10.1038/s41379-020-0631-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020 Jul 9;383((2)):120–8. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Adachi T, Chong JM, Nakajima N, Sano M, Yamazaki J, Miyamoto I, et al. Clinicopathologic and immunohistochemical findings from autopsy of patient with COVID-19, Japan. Emerg Infect Dis. 2020 Sep;26((9)):2157–61. doi: 10.3201/eid2609.201353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Barton LM, Duval EJ, Stroberg E, Ghosh S, Mukhopadhyay S. COVID-19 autopsies, Oklahoma, USA. Am J Clin Pathol. 2020;1–9 doi: 10.1093/ajcp/aqaa062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bradley BT, Maioli H, Johnston R, Chaudhry I, Fink SL, Xu H, et al. Histopathology and ultrastructural findings of fatal COVID-19 infections. Lancet. 2020 Aug 1;396((10247)):320–32. doi: 10.1016/S0140-6736(20)31305-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Buja LM, Wolf DA, Zhao B, Akkanti B, McDonald M, Lelenwa L, et al. The emerging spectrum of cardiopulmonary pathology of the coronavirus disease 2019 (COVID-19): report of 3 autopsies from Houston, Texas, and review of autopsy findings from other United States cities. Cardiovasc Pathol. 2020 Sep-Sep;48:107233. doi: 10.1016/j.carpath.2020.107233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cipolloni L, Sessa F, Bertozzi G, Baldari B, Cantatore S, Testi R, et al. Preliminary post-mortem COVID-19 evidence of endothelial injury and factor VIII hyperexpression. Diagnostics. 2020;10((8)):1–16. doi: 10.3390/diagnostics10080575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cîrstea A-E, Buzulică RL, Buzulică RL, Pirici D, Ceauşu MC, Iman RV, et al. Histopathological findings in the advanced natural evolution of the sars-cov-2 infection. Rom J Morphol Embryol. 2020;61((1)):209–18. doi: 10.47162/RJME.61.1.23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.COVID-19 Autopsy The first COVID-19 autopsy in Spain performed during the early stages of the pandemic. Rev Esp Patol. Jul-Sep 2020;53((3)):182–7. doi: 10.1016/j.patol.2020.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Damiani S, Fiorentino M, De Palma A, Foschini MP, Lazzarotto T, Gabrielli L, et al. Pathological post mortem findings in lungs infected with Sars‐Cov 2. J Pathol. 2020 doi: 10.1002/path.5549. [DOI] [PubMed] [Google Scholar]
- 17.Fitzek A, Sperhake J, Edler C, Schröder AS, Heinemann A, Heinrich F, et al. Evidence for systematic autopsies in COVID-19 positive deceased: case report of the first German investigated COVID-19 death. Rechtsmedizin. 2020;30((3)):184–9. doi: 10.1007/s00194-020-00401-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fox SE, Akmatbekov A, Harbert JL, Li G, Brown JQ. Pulmonary and cardiac pathology in Covid-19 : the first autopsy series from New Orleans (1) Department of Pathology, LSU Health Sciences Center, New Orleans 2) pathology and laboratory medicine service, Southeast Louisiana veterans healthcare system 3. medRxiv. 2020 [Google Scholar]
- 19.Grimes Z, Bryce C, Sordillo EM, Gordon RE, Reidy J, Paniz Mondolfi AE, et al. Fatal pulmonary thromboembolism in SARS-CoV-2-infection. Cardiovasc Pathol. 2020 Sep-Sep;48:107227. doi: 10.1016/j.carpath.2020.107227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grosse C, Grosse A, Salzer HJF, Dünser MW, Motz R, Langer R. Analysis of cardiopulmonary findings in COVID-19 fatalities: high incidence of pulmonary artery thrombi and acute suppurative bronchopneumonia. Cardiovasc Pathol. 2020 Nov-Dec;49:107263. doi: 10.1016/j.carpath.2020.107263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Heinrich F, Sperhake JP, Heinemann A, Mushumba H, Lennartz M, Nörz D, et al. Germany's first COVID-19 deceased: a 59-year-old man presenting with diffuse alveolar damage due to SARS-CoV-2 infection. Virchows Arch. 2020;477((3)):335–9. doi: 10.1007/s00428-020-02872-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Konopka KE, Wilson A, Myers JL. Postmortem lung findings in an asthmatic patient with coronavirus disease 2019. Chest. 2020 Apr;158((3)):e99–e101. doi: 10.1016/j.chest.2020.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Menter T, Haslbauer JD, Nienhold R, Savic S, Hopfer H, Deigendesch N, et al. Post‐mortem examination of COVID19 patients reveals diffuse alveolar damage with severe capillary congestion and variegated findings of lungs and other organs suggesting vascular dysfunction. Histopathology. 2020 Aug;77((2)):198–209. doi: 10.1111/his.14134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Navarro Conde P, Alemany Monraval P, Medina Medina C, Jiménez Sánchez A, Andrés Teruel JC, Ferrando Marco J, et al. Autopsy findings from the first known death from severe acute respiratory syndrome SARS-CoV-2 in Spain. Rev Española Patol. 2020 May; doi: 10.1016/j.patol.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Oprinca GC, Muja LA. Postmortem examination of three SARS-CoV-2-positive autopsies including histopathologic and immunohistochemical analysis. Int J Legal Med. 2021 Jan;135((1)):329–39. doi: 10.1007/s00414-020-02406-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rapkiewicz AV, Mai X, Carsons SE, Pittaluga S, Kleiner DE, Berger JS, et al. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: a case series. EClinicalMedicine. 2020 Jun;24:100434. doi: 10.1016/j.eclinm.2020.100434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Remmelink M, De Mendonça R, D'Haene N, De Clercq S, Verocq C, Lebrun L, et al. Unspecific post-mortem findings despite multiorgan viral spread in COVID-19 patients. Crit Care. 2020;24((1)):495–10. doi: 10.1186/s13054-020-03218-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wichmann D, Sperhake J-P, Lütgehetmann M, Steurer S, Edler C, Heinemann A, et al. Autopsy findings and venous thromboembolism in patients with COVID-19. Ann Intern Med. 2020 May;:M20–2003. doi: 10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yao XH, He ZC, Li TY, Zhang HR, Wang Y, Mou H, et al. Pathological evidence for residual SARS-CoV-2 in pulmonary tissues of a ready-for-discharge patient. Cell Res. 2020;30((6)):541–3. doi: 10.1038/s41422-020-0318-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rapkiewicz AV, Mai X, Carsons SE, Pittaluga S, Kleiner DE, Berger JS, et al. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: a case series. EClinicalMedicine. 2020;24:100434. doi: 10.1016/j.eclinm.2020.100434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Su H, Yang M, Wan C, Yi LX, Tang F, Zhu HY, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020 Jul;98((1)):219–27. doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Suess C, Hausmann R. Gross and histopathological pulmonary findings in a COVID-19 associated death during self-isolation. Int J Legal Med. 2020 Jul;134((4)):1285–90. doi: 10.1007/s00414-020-02319-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nunes Duarte‐Neto A, de Almeida Monteiro RA, da Silva LFF, Malheiros DMAC, de Oliveira EP, Theodoro Filho J, et al. Pulmonary and systemic involvement of COVID‐19 assessed by ultrasound‐guided minimally invasive autopsy. Histopathology. 2020 Aug;77((2)):186–197. doi: 10.1111/his.14160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mauad T, Hajjar LA, Callegari GD, da Silva LF, Schout D, Galas FR, et al. Lung pathology in fatal novel human influenza A (H1N1) infection. Am J Respir Crit Care Med. 2010 Jan;181((1)):72–9. doi: 10.1164/rccm.200909-1420OC. [DOI] [PubMed] [Google Scholar]
- 35.Thille AW, Esteban A, Fernández-Segoviano P, Rodriguez JM, Aramburu JA, Vargas-Errázuriz P, et al. Chronology of histological lesions in acute respiratory distress syndrome with diffuse alveolar damage: a prospective cohort study of clinical autopsies. Lancet Respir Med. 2013 Jul;1((5)):395–401. doi: 10.1016/S2213-2600(13)70053-5. [DOI] [PubMed] [Google Scholar]
- 36.Hariri LP, North CM, Shih AR, Israel RA, Maley JH, Villalba JA, et al. Lung histopathology in coronavirus disease 2019 as compared with severe acute respiratory sydrome and H1N1 influenza: a systematic review. Chest. 2021 Jan;159((1)):73–84. doi: 10.1016/j.chest.2020.09.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Simmons J, Pittet JF. The coagulopathy of acute sepsis. Curr Opin Anaesthesiol. 2015 Apr;28((2)):227–36. doi: 10.1097/ACO.0000000000000163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Paliogiannis P, Mangoni AA, Dettori P, Nasrallah GK, Pintus G, Zinellu A. D-dimer concentrations and covid-19 severity: a systematic review and meta-analysis. Front Public Health. 2020;8((August)):1–7. doi: 10.3389/fpubh.2020.00432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Olson JD. D-dimer: an overview of hemostasis and fibrinolysis, assays, and clinical applications. Adv Clin Chem. 2015;69:1–46. doi: 10.1016/bs.acc.2014.12.001. [DOI] [PubMed] [Google Scholar]
- 40.Magro CM, Mulvey J, Kubiak J, Mikhail S, Suster D, Crowson AN, et al. Severe COVID-19: a multifaceted viral vasculopathy syndrome. Ann Diagn Pathol. 2021;50:151645. doi: 10.1016/j.anndiagpath.2020.151645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020 Jun;220((January)):1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Risitano AM, Mastellos DC, Huber-Lang M, Yancopoulou D, Garlanda C, Ciceri F, et al. Complement as a target in COVID-19? Nat Rev Immunol. 2020 Jun;20((6)):343–4. doi: 10.1038/s41577-020-0320-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Buckley CD, Rainger GE, Nash GB, Raza K. Endothelial cells, fibroblasts and vasculitis. Rheumatology. 2005 Jul;44((7)):860–3. doi: 10.1093/rheumatology/keh542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Roncati L, Ligabue G, Fabbiani L, Malagoli C, Gallo G, Lusenti B, et al. Type 3 hypersensitivity in COVID-19 vasculitis. Clin Immunol. 2020 Aug;217:108487. doi: 10.1016/j.clim.2020.108487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020 Jul 9;383((2)):120–8. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Steyers CM, Miller FJ. Endothelial dysfunction in chronic inflammatory diseases. Int J Mol Sci. 2014 Jun;15((7)):11324–49. doi: 10.3390/ijms150711324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo Q, et al. Prevalence of comorbidities and its effects in patients infected with SARS-CoV-2: a systematic review and meta-analysis. Int J Infect Dis. 2020 May;94:91–5. doi: 10.1016/j.ijid.2020.03.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zucoloto AZ, Jenne CN. Platelet-neutrophil interplay: insights into neutrophil extracellular trap (NET)-driven coagulation in infection. Front Cardiovasc Med. 2019 Jun;6:85. doi: 10.3389/fcvm.2019.00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Matheus Merina LM, Parra-Medina R, Ararat Sarria M, Galvis Diaz J. Cell death. autoimmunity: from bench to bedside. Colombia: Bogotá; 2013. [Google Scholar]
- 50.Barnes BJ, Adrover JM, Baxter-Stoltzfus A, Borczuk A, Cools-Lartigue J, Crawford JM, et al. Targeting potential drivers of COVID-19: neutrophil extracellular traps. J Exp Med. 2020 Jun;217((6)):217. doi: 10.1084/jem.20200652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Veras FP, Pontelli MC, Silva CM, Toller-Kawahisa JE, de Lima M, Nascimento DC, et al. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J Exp Med. 2020 Dec;217((12)):217. doi: 10.1084/jem.20201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zuo Y, Yalavarthi S, Shi H, Gockman K, Zuo M, Madison JA, et al. Neutrophil extracellular traps in COVID-19. JCI Insight. 2020 Jun 4;5((11)):e138999. doi: 10.1172/jci.insight.138999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Edler C, Schröder AS, Aepfelbacher M, Fitzek A, Heinemann A, Heinrich F, et al. Dying with SARS-CoV-2 infection-an autopsy study of the first consecutive 80 cases in Hamburg, Germany. Int J Legal Med. 2020 Jul;134((4)):1275–84. doi: 10.1007/s00414-020-02317-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhang L, Feng X, Zhang D, Jiang C, Mei H, Wang J, et al. Deep vein thrombosis in hospitalized patients with coronavirus disease 2019 (COVID-19) in Wuhan, China: prevalence, risk factors, and outcome. Circulation. 2020 Jul 14;142((2)):114–28. doi: 10.1161/CIRCULATIONAHA.120.046702. [DOI] [PubMed] [Google Scholar]
- 55.Henrina J, Putra ICS, Cahyadi I, Gunawan HFH, Cahyadi A, Suciadi LP. Clinical characteristics and outcomes of venous thromboembolism in patients hospitalized for COVID-19: systematic review and meta-analysis. medRxiv. 2020 doi: 10.1016/j.tru.2021.100037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Poillon G, Obadia M, Perrin M, Savatovsky J, Lecler A. Cerebral venous thrombosis associated with COVID-19 infection: causality or coincidence? J Neuroradiol. 2020 May;48((2)):121–4. doi: 10.1016/j.neurad.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lax SF, Skok K, Zechner P, Kessler HH, Kaufmann N, Koelblinger C, et al. Pulmonary arterial thrombosis in covid-19 with fatal outcome: results from a prospective, single-center, clinicopathologic case series. Ann Intern Med. 2020 May;:M20–2566. doi: 10.7326/M20-2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Le Berre A, Marteau V, Emmerich J, Zins M. Concomitant acute aortic thrombosis and pulmonary embolism complicating COVID-19 pneumonia. Diagn Interv Imaging. 2020 May;101((5)):321–2. doi: 10.1016/j.diii.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bellosta R, Luzzani L, Natalini G, Pegorer MA, Attisani L, Cossu LG, et al. Acute limb ischemia in patients with COVID-19 pneumonia. J Vasc Surg. 2020 Dec;72((6)):1864–72. doi: 10.1016/j.jvs.2020.04.483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Parra-Medina R, Moreno-Lucero P, Jimenez-Moreno J, Parra-Morales AM, Romero-Rojas A. Neuroendocrine neoplasms of gastrointestinal tract and secondary primary synchronous tumors: a systematic review of case reports. Casualty or causality? PLoS One. 2019 May;14((5)):e0216647. doi: 10.1371/journal.pone.0216647. [DOI] [PMC free article] [PubMed] [Google Scholar]