Abstract
Background
Burn injuries are the fourth most common traumatic injury, causing an estimated 180,000 deaths annually worldwide. Superficial burns can be managed with dressings alone, but deeper burns or those that fail to heal promptly are usually treated surgically. Acute burns surgery aims to debride burnt skin until healthy tissue is reached, at which point skin grafts or temporising dressings are applied. Conventional debridement is performed with an angled blade, tangentially shaving burned tissue until healthy tissue is encountered. Hydrosurgery, an alternative to conventional blade debridement, simultaneously debrides, irrigates, and removes tissue with the aim of minimising damage to uninjured tissue. Despite the increasing use of hydrosurgery, its efficacy and the risk of adverse events following surgery for burns is unclear.
Objectives
To assess the effects of hydrosurgical debridement and skin grafting versus conventional surgical debridement and skin grafting for the treatment of acute partial‐thickness burns.
Search methods
In December 2019 we searched the Cochrane Wounds Specialised Register; the Cochrane Central Register of Controlled Trials (CENTRAL); Ovid MEDLINE (including In‐Process & Other Non‐Indexed Citations); Ovid Embase and EBSCO CINAHL Plus. We also searched clinical trials registries for ongoing and unpublished studies, and scanned reference lists of relevant included studies as well as reviews, meta‐analyses and health technology reports to identify additional studies. There were no restrictions with respect to language, date of publication or study setting.
Selection criteria
We included randomised controlled trials (RCTs) that enrolled people of any age with acute partial‐thickness burn injury and assessed the use of hydrosurgery.
Data collection and analysis
Two review authors independently performed study selection, data extraction, 'Risk of bias' assessment, and GRADE assessment of the certainty of the evidence.
Main results
One RCT met the inclusion criteria of this review. The study sample size was 61 paediatric participants with acute partial‐thickness burns of 3% to 4% total burn surface area. Participants were randomised to hydrosurgery or conventional debridement. There may be little or no difference in mean time to complete healing (mean difference (MD) 0.00 days, 95% confidence interval (CI) −6.25 to 6.25) or postoperative infection risk (risk ratio 1.33, 95% CI 0.57 to 3.11). These results are based on very low‐certainty evidence, which was downgraded twice for risk of bias, once for indirectness, and once for imprecision.
There may be little or no difference in operative time between hydrosurgery and conventional debridement (MD 0.2 minutes, 95% CI −12.2 to 12.6); again, the certainty of the evidence is very low, downgraded once for risk of bias, once for indirectness, and once for imprecision. There may be little or no difference in scar outcomes at six months. Health‐related quality of life, resource use, and other adverse outcomes were not reported.
Authors' conclusions
This review contains one randomised trial of hydrosurgery versus conventional debridement in a paediatric population with low percentage of total body surface area burn injuries. Based on the available trial data, there may be little or no difference between hydrosurgery and conventional debridement in terms of time to complete healing, postoperative infection, operative time, and scar outcomes at six months. These results are based on very low‐certainty evidence. Further research evaluating these outcomes as well as health‐related quality of life, resource use, and other adverse event outcomes is required.
Plain language summary
Is surgery with a high‐pressure water jet (hydrosurgery) better than conventional surgery for early treatment of mid‐depth burns?
Background
Burns are common injuries worldwide and can cause illness, lifelong disability and even death. Deep burns often require surgery because the skin is too damaged to heal on its own. The damaged, burnt skin must therefore be cut away (debridement) and replaced with healthy skin, which is typically a very thin layer of healthy skin (graft) taken from another part of the body. Debridement is normally done with a specific surgical knife.
Recently, a high‐pressure, water‐based jet system has been developed, known as hydrosurgery. This tool removes burnt skin only, leaving behind the unburned, healthy skin. Hydrosurgery may be more accurate than a knife in terms of removing burned skin, which may lead to better healing.
All open wounds, including burns, are at risk of infection so adequate debridement is important to reduce the risk of infection. If the wound is closed quickly, it will heal better, with less scarring and less risk of infection.
What did we want to find out?
In this Cochrane Review, we wanted to know whether burns treated with hydrosurgery heal more quickly and with fewer infections than burns treated with a knife. We also wanted to see whether there were any differences in overall quality of life, how well the wound healed in terms of scarring and the amount of medical resources used (using measures like the number of dressing changes and burn clinic appointments, length of hospital stay, and whether further surgery was needed).
Our methods
We searched medical databases for randomised controlled trials that compared burn treatment using hydrosurgery with conventional debridement. Randomised controlled trials are medical studies where the treatment people receive is chosen at random. This type of study provides the most reliable evidence about whether different approaches to health care make a difference. Participants in the studies could be any age. The studies could have taken place anywhere and be reported in any language.
What are the main results of the review?
We found only one Australian study that included 61 children with small burns. The children were randomly allocated to treatment with either hydrosurgery or conventional debridement. Hydrosurgery made little or no difference in the time burns took to heal completely, infection after the operation, or scarring compared to conventional debridement. There was little or no difference in the length of time debridement took using hydrosurgery compared with conventional surgery. The study did not give any information about quality of life or resource use.
Certainty of the evidence
Our certainty (confidence) in the evidence was very limited because we found only one study. It only included children, so the results may not apply to adults or people with more severe burns. It was a randomised study, but did not report the outcomes we expected it to, so we are not sure how reliable its results are.
Conclusions
We do not know if hydrosurgery is better than conventional surgery for early treatment of mid‐depth burns. We need more studies to investigate this question.
Search date
This review includes evidence published up to December 2019.
Summary of findings
Summary of findings 1. Hydrosurgery compared with conventional debridement for partial‐thickness burns prior to skin grafting.
| Hydrosurgery compared with conventional debridement for partial‐thickness burns prior to skin grafting | ||||||
| Patient or population: acute partial‐thickness burns prior to skin grafting Setting: hospital Intervention: hydrosurgical debridement Comparison: conventional debridement | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with conventional debridement | Risk with hydrosurgery | |||||
| Time to complete healing after split‐thickness skin grafting (time to complete healing) Assessed with: clinician observation Follow‐up: 6 months |
The mean time to complete healing after split‐thickness skin grafting was 17.8 days. | MD 0 days (6.25 lower to 6.25 higher) | ‐ | 61 (1 RCT) | ⊕⊝⊝⊝ Very low1 | Time to healing classified as days to complete epithelialisation, determined independently by senior burn nurse |
| Postoperative infection (number of participants with post‐operative infection) Assessed with: clinician observation Follow‐up: 6 months |
Study population | RR 1.33 (0.57 to 3.11) | 61 (1 RCT) | ⊕⊝⊝⊝ Very low1 | Postoperative infection determined by presence of a positive wound culture with moderate–heavy bacterial growth, positive viral or fungal culture, or by clinical evidence of infection (purulent exudate or cellulitis) | |
| 226 per 1000 | 300 per 1000 (121 to 575) | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; MD: mean difference; RCT: randomised controlled trial; RR: risk ratio; TBSA: total body surface area | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
1Evidence downgraded twice for risk of bias (selection bias and other bias in outcome measurement), once for indirectness (low %TBSA burns, paediatric participants only), and once for imprecision, as study is not powered to detect differences in time to complete healing or postoperative infection, plus wide standard deviations and confidence intervals.
Background
Description of the condition
Burn injuries are the fourth most common traumatic injury, causing an estimated 180,000 deaths annually worldwide (WHO 2008). In 2004, 11 million people required medical attention for burn injuries (WHO 2018).
Most burn injuries occur in low‐ to middle‐income countries that do not have the infrastructure to reduce the incidence and severity of these injuries (CDC 2008; Peck 2011). In men, most burn injuries occur at home, outdoors, or at work with equal frequency, whereas for women and children the injury occurs most often at home during cooking and whilst children are unsupervised (Davies 1990; Hemeda 2003; Mabrouk 2003; Rossi 1998; Stewart 2016). The incidence of burn injury is significantly increased by warfare, most notably in Iraq, where the incidence has tripled since 2003 (Peck 2011; Stewart 2016).
Across all demographics, flame is the most common cause of burns in adults, accounting for 35% to 42% of hospital admissions (ABA 2005). Scalds are the most common cause of hospital admissions for children in the USA; however, in low‐ to middle‐income countries, the rate of flame burn and scalds is similar (Peck 2011). Clothing ignition is a common cause of burn injury, typically occurring in countries where open fires are frequently used in the household (Barss 1983; Demamu 1991; Kalayi 1994; Peck 2011). Occupational burns account for 20% to 25% of burn injuries, most commonly flame burns and scalds, and most often affecting people in the food industry (Peck 2011; Pruitt 2007). Other less common but important causes of burn injury include chemical, electrical (including lightning), and radiation burns (ABA 2005; Peck 2011; Stylianou 2015). In clinical practice and for the purposes of this review, burns and scalds both fall under the definition of 'burns'.
Burn injuries are a significant cause of morbidity and mortality for both adults and children in the UK (Stylianou 2015). There is a global downward trend in burn incidence, severity, hospital stay, and mortality but in the UK, approximately 250,000 people are still burnt annually (Smolle 2017). Of these, approximately 175,000 attend hospital, and in 2014, 19,000 children and adults were admitted to hospital for burn injuries (Dunn 2016).
If the burn injury extends through the epidermis only it is classified as superficial (Greenhalgh 2019; Hettiaratchy 2004). If the burn extends into the dermis it is classified as superficial dermal, mid‐dermal, and then deep dermal as it extends through the dermal layer (Greenhalgh 2019; Hettiaratchy 2004). If the burn extends through the dermis to the subcutaneous tissue, it is classified as full‐thickness (Greenhalgh 2019; Hettiaratchy 2004). The diagnosis of burn depth is made clinically on initial assessment, sometimes with adjunct tests (Jaspers 2018). In the USA and other regions, burn depth is sometimes classified as first degree (superficial), second degree (mid‐depth) and third degree (full‐thickness) (Ewbank 2020; Greenhalgh 2019). The depth of the burn is subject to change, and during the process of debridement a more accurate assessment can be made (Monstrey 2008). A clinical assessment of the amount of burnt skin is also made and quantified as a percentage of the total body surface area (%TBSA) (Monstrey 2008).
The management of a burn depends on many factors beyond the scope of this review. In summary, superficial burns can be managed with dressings alone, but deeper burns, of any aetiology or those that fail to heal promptly, are usually treated surgically (Hettiaratchy 2004). Accordingly, surgery for acute burn injuries is undertaken in different situations: as an emergency (to save the life or limb of a person with burns) or at a later stage to remove the unhealthy tissue and facilitate reconstruction (Hettiaratchy 2004). For emergency burns surgery, the management depends on the depth and size of the burn, its anatomical site, and the time since the burn injury (Greenhalgh 2019). Emergency burns surgery aims to debride burnt skin until healthy tissue is reached, at which point skin grafts or temporising dressings are applied. It is important that only burnt tissue is debrided and that no healthy/viable skin or tissue is unnecessarily removed, as this may adversely affect the outcome (Gurfinkel 2010; Orgill 2009). In some cases, if there is evidence of viable dermis following debridement (superficial or mid‐dermal burns), then the wound may not require skin grafting and may heal spontaneously with dressings alone (Greenhalgh 2019). However, early debridement and skin grafting has been the standard of care for decades, and the evidence suggests that surgical debridement within 24 to 48 hours after burn injury reduces blood loss, the risk of infection, length of hospital stay, and mortality, and improves subsequent split‐thickness skin graft take (Rowan 2015).
Description of the intervention
Conventional debridement of burn wounds is performed with an angled blade by tangentially shaving burned tissue until healthy, bleeding tissue is encountered (Orgill 2009). The goal of wound debridement is to remove injured and non‐viable tissue from the area of injury to reduce the inflammatory response engendered by the burn and prevent bacterial proliferation. This reduces the risk of infection and creates the optimal wound bed for complete healing, most often by autologous split‐thickness skin grafting (SSG) (Eldad 1998; Greenhalgh 2019; Robson 1973). Commonly used instruments for conventional debridement include the Brathwaite, Cobbett or Watson knife, the Humby knife, the Goulian or Weck knife, or a simple scalpel (Jeffery 2007). These instruments are of similar type, consisting of static metallic blades used freehand by surgeons to tangentially remove layers of burnt skin. The Watson and Goulian knives are most commonly used in the UK for conventional debridement (Jeffery 2007). In the 1970s, air‐ and later electric‐powered surgical dermatomes were developed, which are similar to an electric razor, with an oscillating blade that evenly removes surface layers of the skin at a defined depth (Schwartz 1978). More recently, dermabrasion techniques have been used for the debridement of deep partial‐thickness burns (Jeffery 2007; Yontar 2017). Lastly, enzymatic debridement is a recent addition to the burn surgeon's armamentarium, although its use is limited by supply and storage issues in some countries (Rosenberg 2015). Hydrosurgery system debridement is an alternative to conventional blade debridement. The principle of hydrosurgical debridement is the emission of pressurised, sterile 0.9% sodium chloride in combination with a localised vacuum system that simultaneously debrides, irrigates, and evacuates non‐viable tissue (Yang 2007). Hydrosurgery may enable surgeons to accurately debride burned tissue whilst preserving viable dermis in the acute setting (Cubison 2006). Conversely, if the entire dermis has sustained thermal or chemical injury, such as in full‐thickness burns, then the burn requires complete excision with a blade to achieve healing, and hydrosurgery is not indicated (Greenhalgh 2019).
How the intervention might work
The hydrosurgery system uses a highly pressurised saline solution and generates a jet, emitted from the tip of the hand‐held instrument (Yang 2007). The jet travels through the nozzle of the instrument generating a Venturi effect, which increases the fluid's velocity and decreases its static pressure due to its passage through a constricted area (Venturi 1836). The high‐pressure jet clears non‐viable tissue from a wound, which is then collected through a suction catheter next to the tip of the instrument and into a collection canister (NICE 2014; Sainsbury 2009). The single‐use, 45 degree angled, hand‐held instrument attaches to a console and is activated by the surgeon using a foot‐pedal (Cubison 2006). The pressure of the fluid jet can be adjusted by the surgeon, allowing for a precise depth of debridement, and thus achieving an accurate wound debridement with complete removal of non‐viable tissue and maximal preservation of healthy tissue (Cubison 2006; Matsumura 2012).
Why it is important to do this review
Hydrosurgery may provide more accurate debridement, potentially increasing the amount of viable native tissue for healing after burns surgery and leading to a faster operating time, limiting the negative effects of general anaesthesia, blood loss, and insensible fluid loss that can be encountered with excessive blade debridement, and hypothermia from the prolonged exposure required for other debridement techniques (Gurunluoglu 2007; Hyland 2015; NICE 2014; Rees‐Lees 2008). Its proposed efficacy is based on the results of a number of clinical studies in a variety of patient populations (Cubison 2006; Kimble 2008). However, the available literature also suggests an increase in adverse events following hydrosurgery compared to control and equivalent postoperative pain (NICE 2014). Despite a number of clinical trials evaluating the use of hydrosurgery in wounds, its efficacy and risk of adverse events following surgery for burn wounds is unclear. To date there has been no formal evidence synthesis exercise to determine efficacy based on these trials, neither has there been a rigorous assessment of the quality of the evidence base. Considering its potential advantages but significant cost, an objective and thorough evaluation of its efficacy is warranted.
Objectives
To assess the effects of hydrosurgical debridement and skin grafting versus conventional surgical debridement and skin grafting for the treatment of acute partial‐thickness burns.
Methods
Criteria for considering studies for this review
Types of studies
We included all published and unpublished randomised controlled trials (RCTs), including cluster‐RCTs, irrespective of language of report. We excluded studies using quasi‐randomisation.
Types of participants
We included RCTs recruiting people of any age described in the primary report as having any acute partial‐thickness burn injury requiring surgical debridement and split‐thickness skin grafting, managed in any care setting. As the method of defining burn injury can vary, we accepted definitions as used by the study authors. We would have included studies recruiting participants with partial‐thickness burn injury alongside people with other types of wounds if the data for people with burn injury were presented separately (or were available from the study authors).
Types of interventions
The primary intervention of interest was hydrosurgical debridement. We included any RCT in which the use or type of hydrosurgery during the treatment period was the only systematic difference between treatment groups.
At the protocol stage, we anticipated that likely comparisons would include hydrosurgical debridement and split‐skin grafting compared with conventional surgical debridement and split‐skin grafting. We also sought to include trials that compared different methods or protocols for hydrosurgery with each other (i.e. hydrosurgery A versus hydrosurgery B, where the type of hydrosurgery used was the only systematic difference between the studies). We excluded studies that compared debridement methods with no debridement, as this did not capture the population of interest, that is participants with burn injuries that require acute surgical debridement.
Types of outcome measures
The primary and secondary outcomes of this review are listed below. If a study was otherwise eligible (i.e. correct study design, population, and intervention/comparator) but did not report a listed outcome, then we contacted the study authors to establish whether the outcomes of interest were measured but not reported.
We reported outcome measures at the latest time point available (assumed to be length of follow‐up if not specified) and the time point specified in the methods as being of primary interest (if different from the latest time point available). For all outcomes, we classified assessment of outcome measures from:
up to or equal to eight weeks as short term;
over eight weeks to 26 weeks as medium term;
over 26 weeks as long term.
Primary outcomes
Time to complete healing after graft (defined by the original studies and based on clinical assessment).
Postoperative infection (clinical diagnosis, as described by the original study and supported by microbiological evidence where possible).
Secondary outcomes
Operative efficiency as measured by the reported operative time (in minutes).
Scar outcome (measured using a standardised validated scar scale, e.g. Vancouver, Patient and Observer Scar Assessment Scale (POSAS)).
Resource use (including measurements of resource use such as number of dressing changes, burn clinic appointments, length of hospital stay, and reoperation/intervention).
Health‐related quality of life.
Other adverse outcomes (including blood transfusion volumes (per %TBSA) within the first seven postoperative days).
Search methods for identification of studies
Electronic searches
We searched the following electronic databases to identify reports of relevant clinical trials:
the Cochrane Wounds Specialised Register (searched 10 December 2019);
the Cochrane Central Register of Controlled Trials (CENTRAL; 2019, Issue 11) in the Cochrane Library (searched 10 December 2019);
Ovid MEDLINE (R) and Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Daily and Versions (1946 to 10 December 2019);
Ovid Embase (1974 to 10 December 2019);
EBSCO CINAHL Plus (Cumulative Index to Nursing and Allied Health Literature; 1937 to 10 December 2019).
The search strategies for the Cochrane Wounds Specialised Register, CENTRAL, Ovid MEDLINE, Ovid Embase and EBSCO CINAHL Plus can be found in Appendix 1. We combined the Ovid MEDLINE search with the Cochrane Highly Sensitive Search Strategy for identifying randomised trials in MEDLINE: sensitivity‐ and precision‐maximising version (2008 revision) (Lefebvre 2019). We combined the Embase search with the Ovid Embase filter developed by the UK Cochrane Centre (Lefebvre 2019). We combined the CINAHL Plus searches with the trial filters developed by the Scottish Intercollegiate Guidelines Network (SIGN 2019). There were no restrictions with respect to language, date of publication or study setting.
We also searched the following clinical trials registries:
US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (www.clinicaltrials.gov) (searched 10 December 2019);
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) (apps.who.int/trialsearch/) (searched 10 December 2019).
The search strategies for clinical trial registries are shown in Appendix 1.
Searching other resources
Searching reference lists of included trials and relevant reviews
We sought to identify other potentially eligible trials or ancillary publications by searching the reference lists of retrieved included trials, as well as relevant systematic reviews, meta‐analyses, and health technology assessment reports. No additional reports were found.
Searching by contacting individuals or organisations
When necessary, we contacted authors of key papers and abstracts to request further information about their trials. We did not contact the manufacturers and distributors of hydrosurgery systems as we believed the search methods implemented to be sufficiently comprehensive.
Adverse effects
We did not perform a separate search for adverse effects of interventions used, considering adverse effects described in the included studies only.
Data collection and analysis
Selection of studies
Three review authors (JCRW, RGW, JD) independently assessed the titles and abstracts of the citations retrieved by the searches for relevance. After this initial assessment, we obtained full‐text copies of all studies considered to be potentially relevant. Three review authors (JCRW, RGW, JD) independently checked the full‐text papers for eligibility, resolving any disagreements by discussion or with the input of a fourth review author (DC) when required. We contacted the study authors by email for three studies where the eligibility of the studies was unclear. We recorded all reasons for exclusion of studies for which we had obtained full copies. This process is summarised in the PRISMA flow chart (Liberati 2009).
Data extraction and management
We extracted and summarised details of the eligible studies using a data extraction sheet that two review authors (JCRW and RGW) piloted independently. Two review authors (JCRW and RGW) independently extracted data and resolved disagreements by discussion, consulting a third review author (JD) where required. Where data were missing from reports, we contacted the study authors to obtain this information.
We extracted the following data where possible by treatment group for the prespecified interventions and outcomes in this review. The outcome data were collected for relevant time points as described in Types of outcome measures:
country of origin;
type of burn and surgery, including whether the burn was grafted or not;
unit of randomisation (per participant) ‐ single burn or multiple burns on the same participant;
unit of analysis;
trial design (e.g. parallel, cluster);
care setting;
number of participants randomised to each trial arm;
eligibility criteria and key baseline participant data;
details of treatment regimen received by each group;
number of operative procedures;
details of any co‐interventions, such as wound dressings;
primary and secondary outcome(s) (with definitions);
outcome data for primary and secondary outcomes (by group);
hospital stay (days);
duration of follow‐up;
number of withdrawals (by group);
publication status of study; and
source of funding for trial.
Assessment of risk of bias in included studies
Two review authors independently assessed risk of bias of the included studies using the revised Cochrane 'Risk of bias' tool (Higgins 2011a). This tool addresses six specific domains: sequence generation, allocation concealment, blinding, incomplete outcome data, selective outcome reporting, and other issues. We recorded any issues with unit of analysis, for example where a cluster trial was undertaken but analysed at the individual level in the study report. We assessed blinding and completeness of outcome data for each of the review outcomes separately. We note that, since wound healing is a subjective clinical outcome, it can be at high risk of measurement bias when outcome assessment is not blinded. We have presented our assessment of risk of bias using two 'Risk of bias' summary figures: one summarising the risk of bias for each item across all studies, and a second showing a cross‐tabulation of each trial by all the 'Risk of bias' items. We classified studies assessed as at high risk of bias for the randomisation sequence domain, allocation concealment domain, or blinded outcome assessment domain (for specified outcome, or a combination of these) as being at overall high risk of bias (for specified outcome). For trials using cluster randomisation or within‐participant randomisation, we would also have considered risk of bias in terms of recruitment bias, baseline imbalance, loss of clusters, incorrect analysis, and comparability with trials randomising participants (Higgins 2011b). None of these study designs were encountered in this review.
Measures of treatment effect
For dichotomous outcomes, we calculated the risk ratio (RR) with 95% confidence intervals (CI). For continuously distributed outcome data, we calculated the mean difference (MD) with 95% CIs, if all trials used the same or a similar assessment scale. If trials used different assessment scales, we would have used standardised mean difference (SMD) with 95% CIs. We only considered mean or median time to healing without survival analysis as a valid outcome if the study reports specified that all were wounds healed (i.e. if the trial authors regarded time to healing as a continuous measure as there is no censoring). We intended to report time‐to‐event data (e.g. time to complete wound healing) as hazard ratios (HR), where this was possible in accordance with the methods described in the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2011). In future eligible studies reporting time‐to‐event data (e.g. time to healing) but not reporting an HR, we plan to estimate the HR using other reported outcomes, such as the numbers of events, through the application of available statistical methods (Parmar 1998).
Unit of analysis issues
Had studies randomised at the participant level and measured outcomes at the wound level (e.g. wound healing), we would have treated the participant as the unit of analysis when the number of wounds assessed appeared to be equal to the number of participants (e.g. one wound per person). Particular unit of analysis issues in wound care trials can occur when studies randomise at the participant level, use the allocated treatment on multiple wounds per participant, and then analyse outcomes per wound; or when studies undertake multiple assessments of an outcome over time per participant. These approaches should be treated as cluster or within‐participant trials, alongside more standard cluster designs (e.g. delivery of interventions at an organisational level). Where a cluster trial has been conducted and correctly analysed, effect estimates and their standard errors can be meta‐analysed using the generic inverse variance method in Review Manager 5 (Review Manager 2014). We planned to record as part of the 'Risk of bias' assessment where a cluster‐RCT had been conducted but incorrectly analysed. If this was not possible, we would have approximated the correct analyses based on guidance from the Cochrane Handbookfor Systematic Reviews of Interventions (Higgins 2011b), using information on:
the number of clusters (or groups) randomised to each intervention group, or the mean size of each cluster;
the outcome data ignoring the cluster design for the total number of participants (e.g. number or proportion of participants with events, or means and standard deviations); and
an estimate of the intracluster (or intraclass) correlation coefficient (ICC).
If the study data could not be analysed correctly, we would have extracted and presented outcome data but not analysed them further. We would also have noted when randomisation had been undertaken within participant or at the wound level, that is a split‐site or split‐body design. We planned to assess whether the correct analysis had been undertaken and record any issues in the 'Risk of bias' section of this review. Had an incorrect analysis been undertaken, we would have contacted the authors to attempt to obtain the original data or try to approximate the correct analysis if the required data were available. If this was not possible, we would have extracted and presented the relevant outcome data but not further analysed or pooled them. This strategy was not required in the current iteration of the review but may be relevant in future updates.
Dealing with missing data
It is common for data to be missing from trial reports. Excluding participants postrandomisation or ignoring participants who are lost to follow‐up compromises the randomisation and potentially introduces bias. If there were missing data that we thought should be included in the analyses, we would have contacted the relevant study authors to request whether these data were available. If data had remained missing for the 'proportion of wounds healed' outcome, we would have assumed for our analysis that if randomised participants were not included in an analysis, their wound did not heal (i.e. they would have been considered in the denominator but not the numerator). In a time‐to‐healing analysis using survival analysis methods, dropouts were accounted for as censored data, so no action regarding missing data was undertaken. For continuous variables and for all secondary outcomes, we have presented available data from the study reports/study authors and have not imputed missing data. If measures of variance were missing, we would have calculated these where possible. If back‐calculation was not possible, we would have contacted the study authors. Where these measures of variance were not available, we would have excluded the study from any relevant meta‐analyses. This was not required in this iteration of the review.
Assessment of heterogeneity
Assessment of heterogeneity can be a complex, multifaceted process. We would first have considered clinical and methodological heterogeneity, that is the degree to which the included studies varied in terms of participant, intervention, outcome, and characteristics such as length of follow‐up. We would have supplemented this assessment of clinical and methodological heterogeneity with information regarding statistical heterogeneity assessed using the Chi² test (a significance level of P < 0.10 would have been considered indicative of statistically significant heterogeneity) in conjunction with the I² measure (Higgins 2003). The I² statistic examines the percentage of total variation across RCTs that is due to heterogeneity rather than to chance (Higgins 2003). In general, I² values of 25% or less may mean a low level of heterogeneity (Higgins 2003), and values of more than 75% indicate very high heterogeneity (Deeks 2011). Had there been evidence of high heterogeneity, we would have attempted to explore this further.
Assessment of reporting biases
Reporting biases arise when the dissemination of research findings is influenced by the nature and direction of results. Publication bias is one of a number of possible causes of 'small‐study effects', that is a tendency for estimates of the intervention effect to be more beneficial in smaller RCTs. Funnel plots allow a visual assessment of whether small‐study effects may be present in a meta‐analysis. A funnel plot is a simple scatter plot of the intervention effect estimates from individual RCTs against some measure of each trial's size or precision (Sterne 2011). We planned to present funnel plots for meta‐analyses comprising 10 RCTs or more using Review Manager 5 (Review Manager 2014). We also planned to cross‐reference reports with published protocols to try to assess the completeness of data reporting, omissions, and potential resultant bias. As only one study was included in this review, this strategy was not employed.
Data synthesis
Data synthesis was not possible in this iteration of the review as there was only a single included study. We had planned to combine details of included studies in a narrative review according to type of comparator and then by outcomes by time period. We would have considered clinical and methodological heterogeneity and undertaken pooling when studies appeared appropriately similar in terms of burn wound type, intervention type, duration of follow‐up, and outcome type.
In terms of meta‐analytical approach, we were unable to pre specify the amount of clinical, methodological, and statistical heterogeneity in the included studies, but it might have been extensive. We thus anticipated using a random‐effects approach for meta‐analysis. Conducting meta‐analysis with a fixed‐effect model in the presence of even minor heterogeneity may provide overly narrow confidence intervals. We only planned to use a fixed‐effect approach when clinical and methodological heterogeneity was assessed to be minimal, and the assumption that a single underlying treatment effect is being estimated held. Chi² and I² would have been used to quantify heterogeneity but would not be used to guide choice of model for meta‐analysis. We planned to exercise caution when meta‐analysed data were at risk of small‐study effects because a random‐effects model may be unsuitable. In this case, or where there are other reasons to question the selection of a fixed‐effect or random‐effects model, we planned to assess the impact of the approach using sensitivity analyses to compare results from alternate models. We intended to report any evidence suggesting that the use of a particular model might not be robust. We planned to consider meta‐analysis even when there is thought to be extensive heterogeneity, exploring the causes behind this using meta‐regression if possible (Thompson 1999).
We planned to obtain pooled estimates of treatment effect using Cochrane Review Manager 5 software (Review Manager 2014). For dichotomous outcomes, we presented the summary estimate as RR with 95% CIs. Where continuous outcomes were measured in the same way across studies, we presented the MD with 95% CI; we planned to generate an SMD when studies measured the same outcome by different methods. We have presented RR and MD as described, but they do not represent 'pooled' summary estimates as only one study is included. For time‐to‐event data, we had planned to plot (and if appropriate, pool) estimates of HRs and 95% CIs as presented in the study reports using the generic inverse variance method. Where time to healing was analysed as a continuous measure but it was not clear if all wounds had healed, we would have documented use of the outcome in the study but not summarised or used data in any meta‐analysis. Neither of these strategies was employed.
'Summary of findings' tables and the GRADE approach
The main results of the review are presented in a 'Summary of findings' table. This table presents key information concerning the certainty of the evidence, the magnitude of the effects of the interventions examined, and the sum of the available data for the main outcomes (Schünemann 2011a). The certainty of a body of evidence involves consideration of within‐trial risk of bias (methodological quality), directness of evidence, heterogeneity, precision of effect estimates, and risk of publication bias (Schünemann 2011b). Two review authors (JCRW and RGW) independently used the GRADE approach to assess the certainty of the evidence for each outcome to determine the level of confidence in the estimate of the observed effects (Schünemann 2013). We graded the evidence for each outcome as high, moderate, low, or very low certainty evidence where possible. Consensus on rating was achieved by involvement of a third review author if needed (JD). The results for important outcomes are presented in 'Summary of findings' tables.
We have presented the following outcomes in the 'Summary of findings' tables:
time to complete healing after graft;
postoperative infection.
Subgroup analysis and investigation of heterogeneity
We planned to assess potential heterogeneity across the following areas specifically. Where there was evidence of between‐trial heterogeneity, we envisaged subgroup analyses being conducted as follows:
adult versus paediatric populations;
anatomical site of burn (e.g. limbs versus face versus trunk);
%TBSA of burn (e.g. less than 10%, 10% to 20%, greater than 20%).
Sensitivity analysis
Where possible, we planned to perform sensitivity analyses to explore the effects of the following criteria:
blinding (blinded studies versus non‐blinded studies);
concealment of allocation (allocation adequately concealed versus not reported or inadequate);
presence of attrition bias;
type of randomisation (truly randomised with adequate method of generating the randomisation sequence versus not reported);
use of a fixed‐effect versus a random‐effects model.
We planned to perform further sensitivity analyses depending on the characteristics of included studies where appropriate.
Elements of this Methods section are based on the standard Cochrane Wounds protocol template.
Results
Description of studies
Results of the search
The search identified 475 records. The results of the search are presented in a PRISMA flow chart (Figure 1). Three review authors (JCRW, RGW, and JD) independently screened these records for potentially relevant studies. We assessed six records as being potentially relevant and retrieved the full texts for further evaluation, which were again screened in triplicate. One study was not available in English, and so a Chinese translator was involved in assessing the study for eligibility (Liu 2016). This study was found to be ineligible for inclusion. One study did not report sufficient data to allow further interpretation of the results. The author of this study was contacted and the data for participants eligible for the review were not available separately so it was excluded (Gravante 2007). This resulted in four potentially relevant reports. Following detailed evaluation of these, three appeared to be from the same study (Anniboletti 2011) and they did not report enough data to facilitate specific interpretation of partial‐thickness burns in participants. Attempts to contact the authors were unsuccessful in terms of retrieving further information about data specific to partial thickness burns, so this study remains as 'awaiting classification' (Anniboletti 2011). This left one study eligible for data extraction (Hyland 2015), two studies excluded (Gravante 2007; Liu 2016), and one study with three study reports awaiting classification (Anniboletti 2011). See PRISMA flow chart (Figure 1). An additional ongoing study was found that has not yet been reported (Legemate 2018).
1.
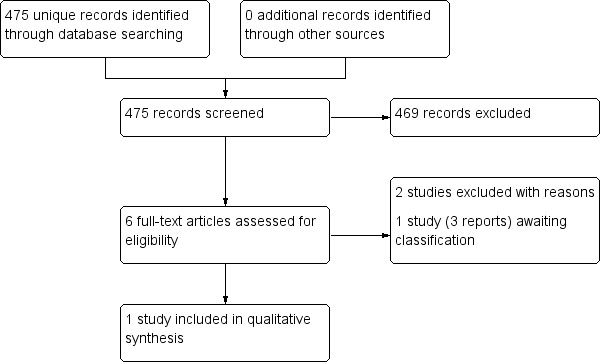
PRISMA flow chart.
Included studies
The only trial eligible for inclusion was performed by Hyland and colleagues at Sydney Children’s Hospital, an acute care hospital in Sydney, Australia, in 2015 (Hyland 2015). This study was an RCT comparing Versajet hydrosurgery and conventional debridement of partial‐thickness paediatric burns (Table 2).
1. Study details.
| First Author | Year | N | Arm | N | Demographics | Mechanism of burn (%) | Depth of burn (%) | Time from burn to surgery (days) | ||||||||||||||
| Age (median years) | Gender | Comorbidity | Other | TBSA of burn | Flame | Scald | Chemical | Contact | Friction | Electrical | Epidermal | Superficial dermal | Mid‐dermal | Deep dermal | Mixed dermal | Full thickness | ||||||
| Hyland | 2015 | 61 | Hydrosurgery | 30 | 2.2 | ‐ | ‐ | ‐ | 3 | 6.7 | 80 | ‐ | 23 | 0 | ‐ | ‐ | ‐ | 10 | 60 | 16.7 | ‐ | < 14 |
| Control | 31 | 2.9 | ‐ | ‐ | ‐ | 4 | 19.4 | 61.3 | ‐ | 12.9 | 6.5 | ‐ | ‐ | ‐ | 12.9 | 71 | 9.7 | ‐ | < 14 | |||
TBSA: total body surface area
Trialists included 61 paediatric participants who underwent conventional blade debridement (Goulian knife) and split‐thickness skin grafting (n = 31) versus hydrosurgical debridement and split‐thickness skin grafting (n = 30). The participant was the unit of randomisation and analysis. The study authors utilised a 1:1 two‐group parallel randomisation technique using 4/6 random permuted blocks. There was no statement regarding funding, but the trial protocol was approved by the local ethics committee. The primary outcome as described in the trial report was dermal preservation, assessed by 2‐millimetre punch biopsy taken immediately before and immediately after debridement and before grafting. The manuscript describes the secondary outcomes as operative time, percentage graft take at day 10 postoperatively, the time to complete healing from injury and the time to healing from grafting, postoperative infection, and scar outcome at three and six months using the modified Vancouver Scar Scale (VSS) (Baryza 1995). An a priori power calculation was performed using an effect size estimated from similar studies of hydrosurgery versus conventional debridement, although the supporting evidence and figures used for this calculation are not provided. Only participants with burn wounds of less than 10% TBSA were recruited, although there was no restriction on including larger burn %TBSA participants. There were no significant differences in baseline demographics prior to intervention allocation. There was no loss to follow‐up at three months, with nine participants lost at six months (n = 5 Versajet and n = 4 conventional). Time to healing was assessed by a blinded burns nurse and defined as days to complete epithelialisation. Postoperative infection was defined as presence of a positive wound culture or by clinical evidence of infection. Further data that were not available from the original study report were provided by Dr Hyland upon request.
Excluded studies
We excluded Liu 2016 because the study population contained participants receiving treatment one month after injury, that is delayed treatment rather than acute.
We excluded Gravante 2007 because data for participants with acute partial‐thickness burns were unavailable.
Studies awaiting classification
Three study reports were conference abstracts from the same research group and reported information from the same study. The study reports did not define the depth of burn injury in the participant groups. There was therefore insufficient data on partial thickness burn injuries to allow data extraction. We received no response from the authors on email contact. We have therefore assessed this study as awaiting classification (Anniboletti 2011).
Ongoing studies
The authors of Legemate 2018 have published a protocol for an RCT of hydrosurgery versus conventional debridement for deep partial‐thickness burns prior to skin grafting in adults and children, with a focus on long‐term (12 months) patient‐reported outcome measures. This trial is currently ongoing and in the recruitment phase.
Risk of bias in included studies
Details of the risk of bias in the included study are presented in Figure 2 and Figure 3. Overall, the one included study was at high risk of bias due to selective outcome reporting and potential inconsistencies in outcome measurement.
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Participants were randomised to the intervention or control group in a 1:1 ratio, stratified by %TBSA. Allocation was computer generated and accessed following enrolment and was concealed until intervention delivery. This was not explicit in the study report but was clarified on further information obtained from the primary author. There were no concerning baseline differences between the two groups. We judged the risk of selection bias to be low.
Blinding
Allocation blinding was not performed. A rank‐based analysis was used to estimate the effect of assignment to intervention on time to complete healing after graft (Wilcoxon test). There was no mention of the distribution of data. We judged the risk of performance bias to be unclear due to unavoidable lack of surgeon blinding. We judged the risk of detection bias to be low.
Incomplete outcome data
Full outcome data were available for all participants. However, the protocol (registered in the Australian and New Zealand Clinical Trials Registry; number ACTRN12610000956000) states that the target sample size would be 150, whilst only 60 were recruited in the study. The reason for this difference is unclear. Without absolute evidence of methodological bias, we have judged the risk of attrition bias to be low.
Selective reporting
There is a discrepancy between the study report and the aforementioned protocol. The protocol describes the primary outcome as the modified VSS at three months, whilst the study reports dermal preservation (originally listed as a secondary outcome). We assume this to represent reporting bias because the comparison regarding dermal preservation was the statistically significant finding between groups. Overall, there are concerns about reporting bias, putting it at high risk.
Other potential sources of bias
For time to healing after graft, it is unclear whether grafts/wounds were examined daily or at wider intervals (e.g. every two or three days). Equally, it is unclear whether the time interval between wound assessments was consistent for all participants. The intra‐ and inter‐rate reliability for re‐epithelialisation is unknown. These factors introduce bias in the reliability of the outcome measure.
Effects of interventions
See: Table 1
Comparison 1. Hydrosurgical debridement versus conventional surgical debridement for acute partial‐thickness burns (1 study; 61 participants)
One study with 61 participants assessed this comparison and included paediatric participants only (Table 2) (Hyland 2015).
See Table 1.
Time to healing
Hyland 2015 reported time to healing as a secondary outcome. The mean (standard deviation (SD)) time to healing in the hydrosurgery group was 17.8 (11.1) days, and the mean (SD) time to healing in the conventional group was 17.8 (13.7) days (mean difference (MD) 0.00, 95% confidence interval (CI) −6.25 to 6.25); there may be little or no difference between hydrosurgery and conventional debridement in terms of mean time to healing after graft. We assessed the evidence as of very low certainty, downgrading twice for risk of bias, once for indirectness, and once for imprecision (Analysis 1.1). All wounds had healed by the end of the study.
1.1. Analysis.

Comparison 1: Primary outcomes, Outcome 1: Time to healing postgraft
Postoperative infection
Hyland 2015 reported postoperative infection as a secondary outcome. The postoperative infection rate was 30% in the hydrosurgery group (n = 9) and 23% in the conventional debridement group (n = 7) (risk ratio 1.33, 95% CI 0.57 to 3.11); there may be little or no difference between hydrosurgery and conventional debridement in terms of postoperative infection. We assessed the evidence as of very low certainty, downgrading twice for risk of bias, once for indirectness, and once for imprecision Analysis 1.2).
1.2. Analysis.

Comparison 1: Primary outcomes, Outcome 2: Postoperative infection
Operative time
Hyland 2015 reported operative time as a secondary outcome. There may be little or no difference in operative efficiency between hydrosurgery and conventional debridement as measured by operative time. The mean (SD) operative times in minutes were 42.7 (19.7) for the hydrosurgery group and 42.6 (28.8) for the conventional debridement group (MD 0.2 minutes, 95% CI −12.2 to 12.6). We assessed the evidence as of very low certainty, downgrading once for risk of bias, once for indirectness, and once for imprecision (Analysis 2.1).
2.1. Analysis.

Comparison 2: Secondary outcomes, Outcome 1: Operative time
Scar outcome
There may be little or no difference between groups in scar outcomes at six months, with a modified VSS total score of 1.7 in the hydrosurgery group and 2.1 in the conventional debridement group (P = 0.1). We have not computed an MD between‐groups given the absence of any measures of within‐group or between‐group variance, uncertainty about the mathematical methods used to generate the quoted p‐value of 0.1 (the hypothesis test used and whether it was one‐tailed or two‐tailed) and an over‐riding concern that this discrete (and typically skewed) outcome measure does not approximate the normal distribution. Photographs were not reported as used to document scar outcome.
Other outcomes
Health‐related quality of life, resource use, and other adverse outcomes were not reported. The only trial included in this review did not assess time to healing or postoperative infection as primary outcomes, and therefore may not be adequately powered to detect differences in these outcomes between the studied interventions.
In summary, according to the GRADE assessment tool, there is very low certainty evidence that there may be little or no difference between hydrosurgery and conventional debridement in terms of time to healing after skin grafting and postoperative infection. The judgement of very low certainty is due to risk of bias in the one included study, indirectness of the evidence due to solely paediatric participants, and imprecision of the outcome data (Table 1).
Discussion
Summary of main results
The aim of this review was to evaluate the evidence for hydrosurgical debridement versus conventional debridement of acute partial‐thickness burns and to summarise its effects in terms of time to complete healing. We identified one RCT undertaken in Australia with one study report consisting of 61 randomised participants. This study compared the Versajet hydrosurgery and conventional debridement in paediatric participants with acute 3% to 4% TBSA partial‐thickness burns.
There may be little or no difference in mean time to complete healing or risk of postoperative infection between hydrosurgery and conventional debridement in acute partial‐thickness burns. These results are based on very low certainty evidence, which was downgraded for risk of bias, indirectness, and imprecision.
In terms of other outcomes, there may be little or no difference in operative time or scar outcomes based on very low‐certainty evidence (1 study, 61 participants). As health‐related quality of life and resource use were not measured in the included study, we are uncertain of any differences between groups in these measures and can draw no conclusions for these outcomes. Data were insufficient to permit an assessment of adverse events other than postoperative infection, the results for which are based on very low‐certainty evidence.
Overall completeness and applicability of evidence
Overall, we found very low‐certainty evidence that there may be little or no difference between hydrosurgery and conventional debridement for acute partial‐thickness burns prior to split‐thickness skin grafting in terms of mean time to complete healing or postoperative infection. The strength of the evidence for both outcomes was downgraded four times, twice for serious risk of bias, once for serious indirectness, and once for serious imprecision. There was a high within‐trial risk of bias in the included study that was primarily due to risk of bias in the measurement of the outcome time to complete healing and concerns about selective reporting bias due to discrepancies between the trial protocol and report. Otherwise, the trial was well performed in terms of randomisation, allocation concealment, and low attrition. Hyland 2015 included only paediatric participants with small burns, therefore there is considerable concern about the applicability of the evidence to adults or individuals with larger TBSA injuries. The primary outcome of the study was reported as dermal preservation, although the primary outcome in the trial protocol was scar score at six months. The study was therefore not powered to detect differences in time to complete healing or postoperative infection, and this may account for the imprecision of the estimates observed for both outcomes. Heterogeneity and risk of publication bias were impossible to judge in this review. There is a further randomised study in progress, which may provide further information on our research question. There is currently a lack of robust experimental research evaluating hydrosurgery versus conventional debridement, especially multicentre trials in the adult population.
Quality of the evidence
There is currently no high‐quality evidence supporting hydrosurgery (over conventional debridement) in the outcomes studied in this review. The only included study was at high risk of bias due to selective outcome reporting and other bias through potentially inconsistent outcome measurement. The results may not be applicable to all populations, healthcare settings, or burn injury types, as the study considered paediatric participants in a high‐income location (Sydney, Australia), all of whom had low %TBSA burn injuries. The study was not powered to detect differences in time to complete healing or postoperative infections. There was considerable imprecision in the direction of effects in the outcome data, manifested in the wide CIs.
Potential biases in the review process
Despite a comprehensive search strategy, it is possible that relevant studies were missed by the review team. It is possible that we have not included some unpublished studies.
Agreements and disagreements with other studies or reviews
Hydrosurgery has been reported to offer potential benefits over conventional debridement that are desirable to clinicians. These include an anticipated faster operative time, greater dermal preservation, and better scar outcomes. Klein 2005 (level 4 evidence; case series) reported on 44 adults with burns receiving hydrosurgery, describing improved usability, with good but non‐specific outcomes. Cubison 2006 (level 4 evidence; case series) described outcomes for seven paediatric patients who underwent hydrosurgical debridement for burns, reporting superior dermal preservation and postoperative scar outcomes. Gurunluoglu 2007 (level 4 evidence; case series) described outcomes for 15 adult patients undergoing hydrosurgical debridement with wounds, including one with burns, and reported a faster operating time and fewer procedures. Matsumura 2012 (level 4 evidence; case series) studied outcomes of hydrosurgery in 47 adults, of whom 21 had burn wounds, and reported a high degree of tissue preservation. A large non‐randomised comparative cohort study of hydrosurgery in 2113 adult and paediatric burns found that use of hydrosurgery was associated with less blood loss, fewer procedures, and a shorter hospital stay compared with mixed conventional and hydrosurgical techniques (Legemate 2019). Younger patients and those with scald burns were more likely to have been treated with hydrosurgery. There is very little in the literature to suggest worse adverse effects of hydrosurgery compared with conventional debridement. This review focused on randomised clinical trials and did not include observational studies, which are often confounded and at high risk of bias. We have examined the only clinical trial of hydrosurgery versus conventional debridement that was eligible for inclusion in the review, the results of which detect no discernible benefits of hydrosurgery over conventional debridement (1 study, 61 participants, very low‐certainty evidence). This is in contrast with the results of the aforementioned previous studies. Three recent systematic reviews have evaluated hydrosurgery for burn wounds, which included a mixture of randomised and non‐randomised studies (Edmondson 2018; Kakagia 2018; Kwa 2019). All three reviews concluded that further RCTs are required, which is in agreement with this review.
Authors' conclusions
Implications for practice.
This review contains one randomised trial of hydrosurgery versus conventional debridement in a paediatric population with low percentage of total body surface area burn injuries. Based on the available trial data, there is very low‐certainty evidence that there may be little or no difference between hydrosurgery and conventional debridement in terms of mean time to complete healing and postoperative infection after skin graft.
Implications for research.
Further prospective research, both experimental and observational, is required to assess the effect of hydrosurgery in terms of clinically important outcomes. Large‐scale observational studies that explore trends in the use of hydrosurgery for burns, particularly in relation to global health provision, resource use, and demographics of patients receiving it, could inform future trials. Resource use and health economic outcomes are important in all future studies to determine the cost‐effectiveness of hydrosurgery compared with conventional debridement. The authors of Legemate 2018 have published a protocol for a randomised controlled trial of hydrosurgery versus conventional debridement for deep partial‐thickness burns prior to skin grafting, with a focus on long‐term (12 months) patient‐reported outcome measures. This trial is currently ongoing and in the recruitment phase. Multicentre randomised controlled trials comparing hydrosurgery with conventional debridement in both adult and paediatric populations are required to determine the effectiveness and cost‐effectiveness of hydrosurgery in acute burn injury.
History
Protocol first published: Issue 10, 2017 Review first published: Issue 9, 2020
Acknowledgements
The authors would like to thank peer reviewers Masafumi Tada, Deepika Poranki, and Roy Buffery for their helpful comments on the protocol. They would like to thank Emma Connaughton and Jason Wong for their comments on the review, and Gill Norman for her comments on both. Thanks are also due to Anne Lawson and Lisa Winer for copy editing the protocol and the review respectively, to Jocelyn Platt and Zhenmi Liu for their assistance with article translation, and to Denise Mitchell for writing the Plain Language Summary.
Appendices
Appendix 1. Search strategies
Cochrane Wounds Specialised Register
1 MESH DESCRIPTOR Burns EXPLODE ALL AND INREGISTER
2 burn or burns or burned or scald* AND INREGISTER
3 (thermal next injur*) AND INREGISTER
4 #1 OR #2 OR #3
5 MESH DESCRIPTOR Debridement EXPLODE ALL AND INREGISTER
6 MESH DESCRIPTOR Hydrotherapy EXPLODE ALL AND INREGISTER
7 hydrosurg* AND INREGISTER
8 hydro‐surg* AND INREGISTER
9 (hydroscalpel* or hydro‐scalpel*) AND INREGISTER
10 versajet* AND INREGISTER
11 water next jet* AND INREGISTER
12 waterjet* AND INREGISTER
13 water‐jet* AND INREGISTER
14 fluid next jet* AND INREGISTER
15 fluidjet* AND INREGISTER
16 fluid‐jet* AND INREGISTER
17 debrid* AND INREGISTER
18 hydro next jet* AND INREGISTER
19 hydrojet* AND INREGISTER
20 hydro‐jet* AND INREGISTER
21 #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15 OR #16 OR #17 OR #18 OR #19 OR #20
22 #4 AND #21
The Cochrane Central Register of Controlled Clinical Trials (CENTRAL)
#1 MeSH descriptor: [Burns] explode all trees
#2 burn or burns or burned or scald*:ti,ab,kw
#3 (thermal next injur*):ti,ab,kw
#4 #1 or #2 or #3
#5 MeSH descriptor: [Debridement] explode all trees
#6 MeSH descriptor: [Hydrotherapy] explode all trees
#7 hydrosurg*:ti,ab,kw
#8 hydro‐surg*:ti,ab,kw
#9 (hydroscalpel* or hydro‐scalpel*):ti,ab,kw
#10 versajet*:ti,ab,kw
#11 water next jet*:ti,ab,kw
#12 waterjet*:ti,ab,kw
#13 water‐jet*:ti,ab,kw
#14 fluid next jet*:ti,ab,kw
#15 fluidjet*:ti,ab,kw
#16 fluid‐jet*:ti,ab,kw
#17 debrid*:ti,ab,kw
#18 hydro next jet*:ti,ab,kw
#19 hydrojet*:ti,ab,kw
#20 hydro‐jet*:ti,ab,kw
#21 #5 or #6 or #7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20
#22 #4 and #21
Ovid MEDLINE
1 exp Burns/
2 (burn or burns or burned or scald*).ab,ti.
3 (thermal adj injur*).ab,ti.
4 1 or 2 or 3
5 exp Debridement/
6 exp Hydrotherapy/
7 hydrosurg*.ab,ti.
8 hydro‐surg*.ab,ti.
9 hydroscalpel*.ab,ti.
10 hydro‐scalpel*.ab,ti.
11 versajet*.ab,ti.
12 (water adj jet*).ab,ti.
13 waterjet*.ab,ti.
14 water‐jet*.ab,ti.
15 (fluid adj jet*).ab,ti.
16 fluidjet*.ab,ti.
17 fluid‐jet*.ab,ti.
18 debrid*.ab,ti.
19 (hydro adj jet*).ab,ti.
20 hydrojet*.ab,ti.
21 hydro‐jet*.ab,ti.
22 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21
23 4 and 22
24 randomized controlled trial.pt.
25 controlled clinical trial.pt.
26 randomi?ed.ab.
27 placebo.ab.
28 clinical trials as topic.sh.
29 randomly.ab.
30 trial.ti.
31 or/24‐30
32 exp animals/ not humans.sh.
33 31 not 32
34 23 and 33
Ovid Embase
1 exp burn/
2 (burn or burns or burned or scald*).ab,ti.
3 (thermal adj injur*).ab,ti.
4 1 or 2 or 3
5 exp debridement/
6 exp hydrotherapy/
7 hydrosurg*.ab,ti.
8 hydro‐surg*.ab,ti.
9 hydroscalpel*.ab,ti.
10 hydro‐scalpel*.ab,ti.
11 versajet*.ab,ti.
12 (water adj jet*).ab,ti.
13 waterjet*.ab,ti.
14 water‐jet*.ab,ti.
15 (fluid adj jet*).ab,ti.
16 fluidjet*.ab,ti.
17 fluid‐jet*.ab,ti.
18 debrid*.ab,ti.
19 (hydro adj jet*).ab,ti.
20 hydrojet*.ab,ti.
21 hydro‐jet*.ab,ti.
22 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18 or 19 or 20 or 21
23 4 and 22
24 Randomized controlled trials/
25 Single‐Blind Method/
26 Double‐Blind Method/
27 Crossover Procedure/
28 (random* or factorial* or crossover* or cross over* or cross‐over* or placebo* or assign* or allocat* or volunteer*).ti,ab.
29 (doubl* adj blind*).ti,ab.
30 (singl* adj blind*).ti,ab.
31 or/24‐30
32 exp animals/ or exp invertebrate/ or animal experiment/ or animal model/ or animal tissue/ or animal cell/ or nonhuman/
33 human/ or human cell/
34 and/32‐33
35 32 not 34
36 31 not 35
37 23 and 36
EBSCO CINAHL Plus
S36 S22 AND S35
S35 S23 OR S24 OR S25 OR S26 OR S27 OR S28 OR S29 OR S30 OR S31 OR S32 OR S33 OR S34
S34 TI allocat* random* or AB allocat* random*
S33 MH "Quantitative Studies"
S32 TI placebo* or AB placebo*
S31 MH "Placebos"
S30 TI random* allocat* or AB random* allocat*
S29 MH "Random Assignment"
S28 TI randomi?ed control* trial* or AB randomi?ed control* trial*
S27 AB ( singl* or doubl* or trebl* or tripl* ) and AB ( blind* or mask* )
S26 TI ( singl* or doubl* or trebl* or tripl* ) and TI ( blind* or mask* )
S25 TI clinic* N1 trial* or AB clinic* N1 trial*
S24 PT Clinical trial
S23 MH "Clinical Trials+"
S22 S4 AND S21
S21 S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13 OR S14 OR S15 OR S16 OR S17 OR S18 OR S19 OR S20
S20 TI hydro‐jet* OR AB hydro‐jet*
S19 TI hydrojet* OR AB hydrojet*
S18 TI hydro jet* OR AB hydro jet*
S17 TI debrid* OR AB debrid*
S16 TI fluid‐jet* OR AB fluid‐jet*
S15 TI fluidjet* OR AB fluidjet*
S14 TI fluid jet* OR AB fluid jet*
S13 TI water‐jet* OR AB water‐jet*
S12 TI waterjet* OR AB waterjet*
S11 TI water jet* OR AB water jet*
S10 TI versajet* OR AB versajet*
S9 TI ( (hydroscalpel* or hydro‐scalpel*) ) OR AB ( (hydroscalpel* or hydro‐scalpel*) )
S8 TI hydro‐surg* OR AB hydro‐surg*
S7 TI hydrosurg* OR AB hydrosurg*
S6 (MH "Hydrotherapy+")
S5 (MH "Debridement+")
S4 S1 OR S2 OR S3
S3 TI (thermal injur*) OR AB (thermal injur*)
S2 TI ( burn or burns or burned or scald* ) OR AB ( burn or burns or burned or scald* )
S1 (MH "Burns+")
US National Institutes of Health Ongoing Trials Register (ClinicalTrials.gov)
(debridement OR hydrosurgery OR waterjet OR hydrojet OR fluidjet OR versajet OR hydroscalpel) | Burns
Burn | ( debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet OR versajet OR hydroscalpel )
scald | ( debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet OR versajet OR hydroscalpel)
Thermal Injury | ( debridement OR hydrosurgery OR waterjet OR fluidjet hydrojet OR versajet OR hydroscalpel )
World Health Organization International Clinical Trials Registry Platform
Burns [Title] and debridement OR hydrosurgery OR waterjet OR hydrojet OR fluidjet OR versajet OR hydroscalpel [Interventions]
Burns [Condition] and debridement OR hydrosurgery OR waterjet OR hydrojet OR fluidjet OR versajet OR hydroscalpel [Interventions]
Burn [Title] and debridement OR hydrosurgery OR waterjet OR hydrojet OR fluidjet OR versajet OR hydroscalpel [Interventions]
Burn [Condition] and debridement OR hydrosurgery OR waterjet OR hydrojet OR fluidjet OR versajet OR hydroscalpel [Interventions]
scald [Title} and debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet OR versajet OR hydroscalpel [Intervention]
scald [Condition] and debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet OR versajet OR hydroscalpel [Intervention]
“Thermal Injury” [Title] debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet hydrojet OR versajet OR hydroscalpel [Intervention]
“Thermal Injury” [Condition] debridement OR hydrosurgery OR hydrojet OR waterjet OR fluidjet hydrojet OR versajet OR hydroscalpel [Intervention]
Data and analyses
Comparison 1. Primary outcomes.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Time to healing postgraft | 1 | 61 | Mean Difference (IV, Random, 95% CI) | 0.00 [‐6.25, 6.25] |
| 1.2 Postoperative infection | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.57, 3.11] |
Comparison 2. Secondary outcomes.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Operative time | 1 | 61 | Mean Difference (IV, Random, 95% CI) | 0.20 [‐12.15, 12.55] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Hyland 2015.
| Study characteristics | ||
| Methods | 2‐arm randomised controlled trial Conducted in Sydney, Australia Follow‐up: 6 months |
|
| Participants | 61 paediatric participants with acute partial‐thickness burns TBSA 3% to 4% Inclusion criteria: children less than 16 years of age with acute partial‐thickness burns undergoing debridement and SSG Exclusion criteria: children with full‐thickness burns, facial burns, and those requiring delayed or staged skin grafting |
|
| Interventions |
Intervention arm: Versajet II Exact (Smith+Nephew) hydrosurgical system wound debridement and SSG Control arm: conventional tangential burn wound debridement using a Goulian knife and SSG Co‐intervention: general anaesthesia, antiseptic povidone–iodine operative site preparation, sterile draping |
|
| Outcomes |
Primary review outcomes: time to complete healing, postoperative infection Secondary review outcomes: operative time, scar outcome |
|
| Notes | The primary outcome of the study was dermal preservation as measured by a 2‐millimetre punch biopsy taken pre‐ and postoperatively. All wounds had healed by the end of the study period. There was a key difference between the study protocol and report in the power calculation and primary outcome. We sought further information from the authors who clarified that the study was powered to detect a difference in the primary outcome of dermal preservation, despite this being different from the protocol (see 'Risk of bias' table). |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The randomisation process was clearly described with no group imbalances. |
| Allocation concealment (selection bias) | Low risk | Allocation was concealed until enrolment. Allocation was revealed in the operating theatre immediately prior to intervention delivery. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Surgeons were not blinded, which is unavoidable in this study. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Participants and other health professionals were blinded. There were no deviations from the assigned intervention. There was appropriate blinding, no attrition for outcomes of interest, and no cross‐over. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | There was no attrition. |
| Selective reporting (reporting bias) | High risk | There are some discrepancies between the published article and the protocol registered in the Australian and New Zealand Clinical Trials Registry (ACTRN12610000956000). The protocol states that the primary outcome would be the modified VSS at 3 months, with secondary outcomes as the % graft take, microbiology from swabs after debridement, duration of surgery, and histological assessment of the remaining dermis after debridement. However, the published report states that dermal preservation was the primary outcome. It is not clear why this change was made. Furthermore, the protocol states that the target sample size was 150, although the published report states 60 without any figures to support this power calculation. It is possible that recruitment was terminated before sufficient individuals were recruited to power the original planned primary outcome (modified VSS), so the authors selected a different outcome for the published report (dermal preservation), perhaps because it was the only comparison to yield a statistically significant difference between groups. Overall, the risk of bias is high. |
| Other bias | Unclear risk | We are told that “time to healing after‐skin grafting and burn was also determined independently by an experienced senior burn nurse as days to complete epithelialisation”, but it is unclear whether grafts/wounds were examined daily or at wider intervals (e.g. 2 or 3 days), or if this was consistent for all participants. It is not usual clinical practice to inspect burn wounds/grafts daily, and so it is likely that there was a greater time interval between wound inspections. Also, the ability of a nurse to determine re‐epithelialisation, the intra‐ and inter‐rate agreement and threshold used was not stated. Overall, there are concerns about inconsistency in the measurement of the outcome, putting this trial at unclear risk of bias for outcome measurement. |
| Overall risk of bias | High risk | Overall, there are some concerns that render the overall assessment of bias as high. |
SSG: split‐thickness skin grafting TBSA: total body surface area VSS: Vancouver Scar Scale
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Gravante 2007 | Data for target population unavailable |
| Liu 2016 | Incorrect study population |
Characteristics of studies awaiting classification [ordered by study ID]
Anniboletti 2011.
| Methods | Randomised comparative study (no further information on study design) |
| Participants | 35 participants (no further information on participants) |
| Interventions | Versajet hydrosurgery system (n = 17) compared to hand‐held dermatome escharectomy (n = 18) |
| Outcomes | Primary outcomes were time for complete debridement and the efficacy of Versajet in reaching the correct dermal plane. Secondary outcomes were assessment of postoperative pain (visual analogue scale), adverse effects, and complete healing times. |
| Notes | Conference abstract only. Corresponding author emailed on two separate occasions with no response. |
Characteristics of ongoing studies [ordered by study ID]
Legemate 2018.
| Study name | Long‐term scar quality after hydrosurgical versus conventional debridement of deep dermal burns (HyCon trial) |
| Methods | A multicentre, randomised, intra‐patient, controlled trial will be conducted in the Dutch burn centres of Rotterdam, Beverwijk, and Groningen. |
| Participants | All patients with deep dermal burns that require excision and grafting |
| Interventions | Hydrosurgical debridement versus conventional debridement |
| Outcomes | Scar quality measured by the observer score of the Patient and Observer Scar Assessment Scale (POSAS) |
| Starting date | 20 January 2017 |
| Contact information | Trauma Research Unit Department of Surgery, Erasmus MC, University Medical Center, Rotterdam, the Netherlands. Email: vliesc@maasstadziekenhuis.nl |
| Notes | Due to complete recruitment at the end of 2019 |
Differences between protocol and review
The protocol for this review stated that the outcome ‘percentage of graft take post‐debridement’ would be included in the 'Summary of findings' table. However, this outcome was removed from the protocol during its development as it was felt to be a surrogate outcome for 'time to complete healing'.
Contributions of authors
Justin CR Wormald: conceived the review; designed the review; co‐ordinated the review; extracted data; checked quality of data extraction; analysed or interpreted data; undertook quality assessment; checked quality assessment; produced the first draft of the review; contributed to writing or editing the review; wrote to study authors/experts/companies; approved the final review prior to publication; is guarantor of the review.
Ryckie G Wade: designed the review; extracted data; checked quality of data extraction; analysed or interpreted data; undertook quality assessment; checked quality assessment; produced the first draft of the review; contributed to writing or editing the review; approved the final review prior to publication.
Jonathan A Dunne: designed the review; extracted data; checked quality of data extraction; analysed or interpreted data; undertook quality assessment; produced the first draft of the review; contributed to writing or editing the review; approved the final review prior to publication.
Declan P Collins: conceived the review; designed the review; analysed or interpreted data; produced the first draft of the review; contributed to writing or editing the review; advised on the review; approved the final review prior to publication.
Abhilash Jain: designed the review; analysed or interpreted data; produced the first draft of the review; contributed to writing or editing the review; advised on the review; approved the final review prior to publication.
Contributions of editorial base
Kurinchi Gurusamy (Editor): edited the protocol and the review; advised on methodology, interpretation, and content; approved the final review prior to submission.
Gill Rizzello (Managing Editor): co‐ordinated the editorial process; advised on content; edited the protocol and the review.
Naomi Shaw and Sophie Bishop (Information Specialists): designed the search strategy; ran the search; edited the search methods sections.
Tom Patterson (Editorial Assistant): edited the reference sections.
Sources of support
Internal sources
No sources of support supplied
External sources
-
The National Institute for Health Research (NIHR), UK
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Wounds. The views and opinions expressed are those of the authors and not necessarily those of the NIHR, NHS, or the Department of Health and Social Care.
Declarations of interest
Justin Wormald: none known.
Ryckie Wade: none known.
Jonathan Dunne: none known.
Declan Collins: has received two grants from charities for the purchase of equipment and service development. Neither grant application is related to this work. He has performed one lecture on an unrelated topic (autologous fibrin glue use in burns patients). Payment for this lecture was from Vivostat, a medical company who do not manufacture products that relate to this review, and the payment was not made to himself but to a hospital research fund.
Abhilash Jain: has received grants from peer review national organisations for clinical research unrelated to this review.
New
References
References to studies included in this review
Hyland 2015 {published and unpublished data}
- Hyland EJ, D’Cruz R, Menon S, Chan Q, Harvey JG, Lawrence T, et al. Prospective, randomised controlled trial comparing Versajet™ hydrosurgery and conventional debridement of partial thickness paediatric burns. Burns 2015;41(4):700-7. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Gravante 2007 {published data only (unpublished sought but not used)}
- Gravante G, Delogu D, Esposito G, Montone A. Versajet hydrosurgery versus classic escharectomy for burn debridement: a prospective randomized trial. Journal of Burn Care & Research 2007;28(5):720-4. [DOI] [PubMed] [Google Scholar]
Liu 2016 {published data only}
- Liu GC, Kan ZH, Sheng JJ, Li HH, Li L, Sun Y, et al. Efficacy of a hydrosurgery system applied in the debridement of extensive residual wounds of patients with severe burn. Chinese Journal of Burns 2016;32(9):549-54. [DOI] [PubMed] [Google Scholar]
References to studies awaiting assessment
Anniboletti 2011 {published data only (unpublished sought but not used)}
- Anniboletti T, Palombo M, Fasciani L, Santi GD, Palombo P. The use of Versajet hydrosurgery: 5 years experience. Burns 2011;37:S19. [Google Scholar]
- Delli Santi G, Palombo M, Carnì P, Cempanari M, Floris M, Schirosi M, et al. Versajet hydrosurgery: 7 years of experience in adults and pediatric patients. Annals of Burns and Fire Disasters 2015;XXVIII(Supplement EBA):196. [Google Scholar]
- Esposito G, Anniboletti T, Palombo M, Palombo P. Versajet hydrosurgery: our experience in adults and pediatric patients. Burns 2009;35:S23. [Google Scholar]
References to ongoing studies
Legemate 2018 {published data only}
- Legemate CM, Goei H, Middelkoop E, Oen IM, Nijhuis TH, Kwa KA, et al. Long-term scar quality after hydrosurgical versus conventional debridement of deep dermal burns (HyCon trial): study protocol for a randomized controlled trial. Trials 2018;19(1):239. [DOI] [PMC free article] [PubMed] [Google Scholar]
Additional references
ABA 2005
- American Burn Association (ABA). National Burn Repository 2005 Report. Dataset version 2.0. www.ameriburn.org/NBR2005.pdf.
Barss 1983
- Barss P, Wallace K. Grass-skirt burns in Papua New Guinea. Lancet 1983;1:733. [DOI] [PubMed] [Google Scholar]
Baryza 1995
- Baryza MJ, Baryza GA. The Vancouver Scar Scale: an administration tool and its interrater reliability. Journal of Burn Care & Research 1995;16:535-8. [DOI] [PubMed] [Google Scholar]
CDC 2008
- Centers for Disease Control and Prevention. Fire deaths and injuries: fact sheet overview 2008. www.cdc.gov/HomeandRecreationalSafety/Fire-Prevention/fires-factsheet.html (accessed 10 February 2017).
Cubison 2006
- Cubison TC, Pape SA, Jeffery SL. Dermal preservation using the Versajet hydrosurgery system for debridement. Burns 2006;32(6):714-20. [DOI] [PubMed] [Google Scholar]
Davies 1990
- Davies JW. The problems of burns in India. Burns 1990;16(Suppl 1):S1-24. [PubMed] [Google Scholar]
Deeks 2011
- Deeks JJ, Higgins JP, Altman DG. Chapter 9: Analysing data and undertaking meta-analyses. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Demamu 1991
- Demamu S. Community-Based Study of Childhood Injuries in Adamitulu District, Ethiopia [Doctoral thesis]. Addis Ababa (Ethiopia): Addis Ababa University, Department of Community Health, 1991. [Google Scholar]
Dunn 2016
- Dunn K, Reade C, Dudley-Southern R. International Burn Injury Database: annual report 2014/15. www.ibidb.org/downloads-mainmenu-28/doc_details/495-201415-ibid-annual-report.
Edmondson 2018
- Edmondson SJ, Jumabhoy IA, Murray A. Time to start putting down the knife: a systematic review of burns excision tools of randomised and non-randomised trials. Burns 2018;44(7):1721-37. [DOI] [PubMed] [Google Scholar]
Eldad 1998
- Eldad A, Weinberg A, Breiterman S, Chaouat M, Palanker D, BenBassat H. Early nonsurgical removal of chemically injured tissue enhances wound healing in partial thickness burns. Burns 1998;24:166-72. [DOI] [PubMed] [Google Scholar]
Ewbank 2020
- Ewbank C, Sheckter CC, Warstadt NM, Pirrotta EA, Curtin C, Newton C, et al. Variations in access to specialty care for children with severe burns. American Journal of Emergency Medicine 2020;38(6):1146-52. [DOI] [PubMed] [Google Scholar]
Greenhalgh 2019
- Greenhalgh DG. Management of Burns. New England Journal of Medicine 2019;380:2349-59. [DOI] [PubMed] [Google Scholar]
Gurfinkel 2010
- Gurfinkel R. Histological assessment of tangentially excised burn eschars. Canadian Journal of Plastic Surgery 2010;18(3):33-6. [PMC free article] [PubMed] [Google Scholar]
Gurunluoglu 2007
- Gurunluoglu R. Experiences with waterjet hydrosurgery system in wound debridement. World Journal of Emergency Surgery 2007;2:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hemeda 2003
- Hemeda M, Maher A, Mabrouk A. Epidemiology of burns admitted to Ain Shams University Burns Unit, Cairo, Egypt. Burns 2003;29(4):353-8. [DOI] [PubMed] [Google Scholar]
Hettiaratchy 2004
- Hettiaratchy S, Dziewulski P. ABC of burns. BMJ 2004;5(328):7452. [DOI: 10.1136/bmj.328.7452.1366] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327(7414):557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JP, Altman DG, Sterne JA. Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2011b
- Higgins JP, Deeks JJ, Altman DG. Chapter 16: Special topics in statistics. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Jaspers 2018
- Jaspers ME, Van Haasterecht L, Van Zuijlen PP, Mokkink LB. A systematic review on the quality of measurement techniques for the assessment of burn wound depth or healing potential. Burns 2018;18:S0305-4179. [DOI] [PubMed] [Google Scholar]
Jeffery 2007
- Jeffery SL. Device related tangential excision in burns. Injury 2007;38:35-8. [DOI] [PubMed] [Google Scholar]
Kakagia 2018
- Kakagia DD, Karadimas EJ. The efficacy of Versajet™ Hydrosurgery system in burn surgery. A systematic review. Journal of Burn Care & Research 2018;39(2):188-200. [DOI] [PubMed] [Google Scholar]
Kalayi 1994
- Kalayi GD, Muhammad I. Clothing burns in Zaria. Burns 1994;20(4):356-9. [DOI] [PubMed] [Google Scholar]
Kimble 2008
- Kimble RM, Mott J, Joethy J. Versajet hydrosurgery system for the debridement of paediatric burns. Burns 2008;34(2):297-8. [DOI] [PubMed] [Google Scholar]
Klein 2005
- Klein MB, Hunter S, Heimbach DM, Engrav LH, Honari S, Gallery E, et al. The Versajet™ water dissector: a new tool for tangential excision. Journal of Burn Care & Rehabilitation 2005;26(6):483-7. [DOI] [PubMed] [Google Scholar]
Kwa 2019
- Kwa KA, Goei H, Breederveld RS, Middelkoop E, Van der Vlies CH, Van Baar ME. A systematic review on surgical and non-surgical debridement techniques of burn wounds. Journal of Plastic, Reconstructive & Aesthetic Surgery 2019;72(11):1752–62. [DOI] [PubMed] [Google Scholar]
Lefebvre 2019
- Lefebvre C, Glanville J, Briscoe S, Littlewood A, Marshall C, Metzendorf M-I, et al. Chapter 4: Searching for and selecting studies. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al (editors). Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated July 2019). Cochrane, 2019. Available from www.training.cochrane.org/handbook.
Legemate 2019
- Legemate CM, Goei H, Gostelie OF, Nijhuis TH, Van Baar ME, Van der Vlies CH. Application of hydrosurgery for burn wound debridement: an 8-year cohort analysis. Burns 2019;45(1):88-96. [DOI] [PubMed] [Google Scholar]
Liberati 2009
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLOS Medicine 2009;6:e1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mabrouk 2003
- Mabrouk A, Maher A, Nasser S. An epidemiologic study of elderly burn patients in Ain Shams University Burn Unit, Cairo, Egypt. Burns 2003;29(7):687-90. [DOI] [PubMed] [Google Scholar]
Matsumura 2012
- Matsumura H, Nozaki M, Watanabe K, Sakurai H, Kawakami S, Nakazawa H, et al. The estimation of tissue loss during tangential hydrosurgical debridement. Annals of Plastic Surgery 2012;69(5):521-5. [DOI] [PubMed] [Google Scholar]
Monstrey 2008
- Monstrey S, Hoeksema H, Verbelen J, Pirayesh A, Blondeel P. Assessment of burn depth and burn wound healing potential. Burns 2008;34(6):761-9. [DOI] [PubMed] [Google Scholar]
NICE 2014
- National Institute for Health and Care Excellence (NICE). The Versajet II hydrosurgery system for surgical debridement of acute and chronic wounds and burns. Medtech innovation briefing [MIB1]. February 2014. www.nice.org.uk/advice/mib1.
Orgill 2009
- Orgill DP. Excision and skin grafting of thermal burns. New England Journal of Medicine 2009;360(9):893-901. [DOI] [PubMed] [Google Scholar]
Parmar 1998
- Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analysis of the published literature for survival endpoints. Statistics in Medicine 1998;17(24):2815-34. [DOI] [PubMed] [Google Scholar]
Peck 2011
- Peck MD. Epidemiology of burns throughout the world. Part I: distribution and risk factors. Burns 2011;37(7):1087-100. [DOI: 10.1016/j.burns.2011.06.005] [DOI] [PubMed] [Google Scholar]
Pruitt 2007
- Pruitt BA, Wolf SE, Mason AD. Epidemiological, demographic, and outcome characteristics of burn injury. In: Herndon DN, editors(s). Total Burn Care. 3rd edition. Philadelphia (PA): Saunders Elsevier, 2007:14-32. [Google Scholar]
Rees‐Lees 2008
- Rees-Lees JE, Burge TS, Estela CM. The indications for Versajet® hydrosurgical debridement in burns. European Journal of Plastic Surgery 2008;31(4):165-70. [DOI: 10.1007/s00238-008-0232-4] [DOI] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Robson 1973
- Robson MC, Krizek TJ. Predicting skin graft survival. Journal of Trauma 1973;13(3):213-7. [DOI] [PubMed] [Google Scholar]
Rosenberg 2015
- Rosenberg L, Shoham Y, Krieger Y, Rubin G, Sander F, Koller J, et al. Minimally invasive burn care: a review of seven clinical studies of rapid and selective debridement using a bromelain-based debriding enzyme (Nexobrid®). Annals of Burns and Fire Disasters 2015;28(4):264-74. [PMC free article] [PubMed] [Google Scholar]
Rossi 1998
- Rossi LA, Braga EC, Barruffini RC, Carvalho EC. Childhood burn injuries: circumstances of occurrences and their prevention in Ribeirão Preto, Brazil. Burns 1998;24(5):416-9. [DOI] [PubMed] [Google Scholar]
Rowan 2015
- Rowan MP, Cancio LC, Elster EA, Burmeister DM, Rose LF, Natesan S, et al. Burn wound healing and treatment: review and advancements. Critical Care 2015;19:243. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sainsbury 2009
- Sainsbury DC. Evaluation of the quality and cost-effectiveness of Versajet hydrosurgery. International Wound Journal 2009;6(1):24-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Schwartz 1978
- Schwartz B. Dermatome apparatus and method. United States patent US 4,098,278 1978.
Schünemann 2011a
- Schünemann HJ, Oxman AD, Higgins JP, Vist GE, Glasziou P, Guyatt GH. Chapter 11: Presenting results and 'Summary of findings' tables. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Schünemann 2011b
- Schünemann HJ, Oxman AD, Higgins JP, Deeks JJ, Glasziou P, Guyatt GH. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Schünemann 2013
- Schünemann H, Brozek J, Guyatt G, Oxman A. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach (updated October 2013). GRADE Working Group, 2013. Available from gdt.guidelinedevelopment.org/app/handbook/handbook.html.
SIGN 2019
- Scottish Intercollegiate Guidelines Network (SIGN). Search filters. www.sign.ac.uk/what-we-do/methodology/search-filters/ (accessed 10 December 2019).
Smolle 2017
- Smolle C, Cambiaso-Daniel J, Forbes AA, Wurzer P, Hundeshagen G, Branski LK, et al. Recent trends in burn epidemiology worldwide: a systematic review. Burns 2017;43(2):249-57. [DOI: 10.1016/j.burns.2016.08.013] [DOI] [PMC free article] [PubMed]
Sterne 2011
- Sterne JA, Sutton AJ, Ioannidis JP, Terrin N, Jones DR, Lau J, et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomised controlled trials. BMJ 2011;22(343):d4002. [DOI] [PubMed] [Google Scholar]
Stewart 2016
- Stewart BT, Lafta R, Al Shatari SA, Cherewick M, Burnham G, Hagopian A, et al. Burns in Baghdad from 2003 to 2014: results of a randomized household cluster survey. Burns 2016;42(1):48-55. [DOI] [PMC free article] [PubMed] [Google Scholar]
Stylianou 2015
- Stylianou N, Buchan I, Dunn KW. A review of the international Burn Injury Database (iBID) for England and Wales: descriptive analysis of burn injuries 2003–2011. BMJ Open 2015;5(2):e006184. [DOI: 10.1136/bmjopen-2014-006184] [DOI] [PMC free article] [PubMed] [Google Scholar]
Thompson 1999
- Thompson SG, Sharp SJ. Explaining heterogeneity in meta-analysis: a comparison of methods. Statistics in Medicine 1999;18(20):2693-708. [DOI] [PubMed] [Google Scholar]
Venturi 1836
- Venturi GB, Nicholson W. Experimental researches concerning the principle of the lateral communication of motion in fluids, applied to the explanation of various hydraulic phenomena. In: T Thomas, editors(s). Tracts on Hydraulics. London (UK): Tredgold, 1836:123–84. [Google Scholar]
WHO 2008
- World Health Organization (WHO). The Global Burden of Disease: 2004 Update. www.who.int/healthinfo/global_burden_disease/GBD_report_2004update_full.pdf.
WHO 2018
- World Health Organization (WHO). Fact sheet: Burns. 6 March 2018. www.who.int/news-room/fact-sheets/detail/burns (accessed 2 March 2019).
Yang 2007
- Yang JY, Hwuang JY, Chuang SS. Clinical experience in using the water jet in burn wound debridement. Annals of Burns and Fire Disasters 2007;20(2):72-7. [PMC free article] [PubMed] [Google Scholar]
Yontar 2017
- Yontar Y, Coruh A, Dinc N, Kontas O. Steel wool-aided dermabrasion of deep partial-thickness burns. Journal of Burn Care & Research 2017;38(3):179-86. [DOI] [PubMed] [Google Scholar]


