CLINICAL HISTORY
A 39‐year‐old man presented to a small local hospital in northern Saskatchewan following new‐onset seizure activity. A single seizure began focally in the left arm and subsequently became secondarily generalized. He was observed in hospital for 2 days, during which there were no further seizures. He was released after arrangements had been made for him to follow‐up with a neurologist, an appointment which he did not keep. Two months later he presented to our institution after having had three similar seizures in short succession. On examination, he was alert and oriented, but drowsy. There was a mild dysphasia, and a partial right‐sided visual field defect. The patient occasionally smoked cigarettes, had previously abused alcohol, and had previously been exposed to tuberculosis.
A CT scan of the head revealed a 3 × 2.7 × 2.2 cm mass in the left temporoparietal region, with a slightly thickened, rim‐enhancing wall following administration of contrast media (Figure 1). The mass was resected.
Figure 1.

PATHOLOGY
Microscopic examination revealed prominent central necrosis surrounded by a comparatively thin hypercellular zone of inflammation containing numerous small granulomata, which were well visualized with Masson trichrome staining (Figure 2a). Prominent but variable collagen and reticulin deposition was present within both the central necrotic zone and its surrounding rim of granulomatous tissue, where it was present between, but generally not within, granulomata. A variably thick band of karyorrhectic debris was often found at the transition between the central necrotic zone and the surrounding granulomatous tissue (Figure 2b). The granulomata consisted of a central collection of epithelioid macrophages surrounded by a prominent, but somewhat variable, inflammatory infiltrate (Figure 2c) consisting predominantly of both CD4 and CD8 immunopositive T lymphocytes, along with lesser numbers of plasma cells and macrophages. Both neutrophils and eosinophils were inconspicuous. Many granulomata contained centrally situated giant cells, some of the Langhans type (Figure 3), or small areas of caseous necrosis (Figure 2d). Many blood vessels within the central necrotic zone showed luminal fibrous occlusion. Occasional blood vessels in the granulomatous tissue and surrounding brain parenchyma showed prominent perivascular and intramural collections of lymphocytes and plasma cells. Brain parenchyma surrounding the main lesion showed prominent astrocytic gliosis, but only occasional astrocytes and their processes were seen within the lesion itself with GFAP immunohistochemistry. Microorganisms were not identified with special stains, including those for acid‐fast bacilli and fungi.
Figure 2.
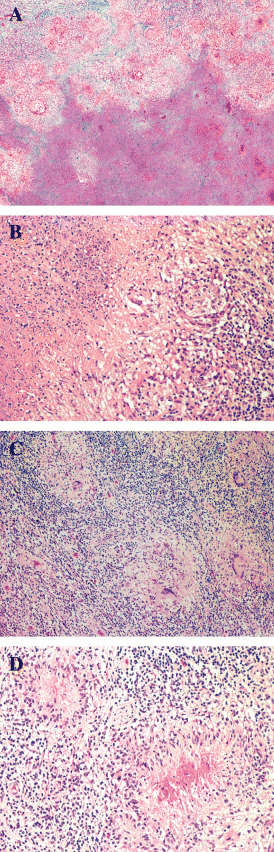
Figure 3.

DIAGNOSIS
Intracerebral tuberculoma.
DISCUSSION
This 39‐year‐old man presented with new‐onset seizures, and a CT scan of the head demonstrated a ring‐enhancing left temporoparietal mass with associated edema and mass effect. A brain tumor was suspected on the basis of the clinical and neuro‐imaging findings, and was considered to be most consistent with a high‐grade glioma. Microscopic examination revealed the characteristic features of a tuberculoma, however. Despite the absence of acid‐fast bacilli microscopically, this diagnosis was confirmed by isolation of Mycobacterium tuberculosis complex in cultures of tissue from the mass, which were obtained for microbiological studies at the time of operation. He was released from the hospital on anti‐tuberculosis medications and Dilantin. When seen at 3 months follow‐up, he had remained seizure free, and his dysphasia had improved. Repeat CT scans of the head performed 3 and 9 months post‐surgery showed no evidence of residual/recurrent tuberculoma.
Although intracranial tuberculomas are now rare in industrialized countries, they remain relatively common in developing nations 3, 4. Encountered in all age groups, tuberculomas occur most frequently in the first three decades of life. Known extracranial disease or history of tuberculosis exposure is common but inconstant (5). Tuberculomas may occur in a diversity of brain regions, including the cerebrum, cerebellum, brainstem, hypothalamus and pituitary gland, as well as within the spinal cord, cavernous sinus, and meninges 2, 7, 8, 9, 11, 13. As a result, the presenting signs and symptoms may be diverse, and include seizures, headache, papilledema, cerebellar dysfunction and hemiplegia. A tuberculoma of the midbrain or cerebellum may manifest as obstructive hydrocephalus. Fever may also be a feature. The neuro‐imaging features of tuberculomas are not specific 4, 14, but together with characteristic clinical and CSF findings, when present, they may enable early diagnosis and treatment in many patients, a situation in which response to anti‐tuberculosis medications is typically very good (1). Nonetheless, in the absence of extracranial tuberculosis the diagnosis may be difficult, and in these cases brain biopsy may be indicated. In this situation, a definitive diagnosis can be made upon demonstration of acid‐fast microorganisms by stain, culture, immunohistochemistry, or polymerase chain reaction (12).
Grossly, tuberculomas may be round, oval, or lobulated in shape, the latter arising on the basis of fusion of smaller nodules. They may vary in size, in part dependent upon their anatomical location and time to manifestation of clinical signs and symptoms. Their firm consistency and relatively well‐demarcated margins relative to surrounding edematous brain parenchyma often enable a gross total resection at surgery. Cut sections reveal a central zone of friable but firm necrotic material surrounded by a variably thick rim of firm grayish fibrous and glial tissue. Microscopically, the center of a tuberculoma consists of amorphous eosinophilic caseous necrotic material surrounded by a band of basophilic karyorrhectic debris. This latter region is where Mycobacterium tuberculosis are often most numerous, as may be demonstrated with appropriate acid‐fast bacilli stains such as the Ziehl‐Neelsen stain. Surrounding this is a firm layer of granulomatous inflammation, within which are found granulomata, some of which may contain a central area of caseous necrosis or multinucleated giant cells, as well as lymphocytes, plasma cells and variably dense collagen deposits. Neutrophils may be prominent in some lesions. Surrounding brain parenchyma exhibits gliosis and variable edema. Tuberculomas may eventually become quiescent, with a decreased or absent content of bacilli and inflammatory cells, and may become cystic or calcified. Recently, a tuberculoma “variant” has been reported in immunosuppressed patients, where it is composed of a mass of round or spindle‐shaped histiocytes containing mycobacteria of the Mycobacterium avium complex 6, 10.
REFERENCES
- 1. Al‐Deeb SM, Yaqub BA, Sharif HS, Motaery KR (1992) Neurotuberculosis: a review. Clin Neurol Neurosurg 94(Suppl.):S30–S33. [DOI] [PubMed] [Google Scholar]
- 2. Al Soub H, Al Alousi FS, Al‐Khal AL (2001) Tuberculoma of the cavernous sinus. Scand J Infect Dis 33:868–870. [DOI] [PubMed] [Google Scholar]
- 3. Artico M, De Caro GM, Carloia S, Salvati M, D’Ambrosio M, Delfini R (1999) Advances in diagnosis, treatment and prognosis of intracerebral tuberculomas in the last 50 years. Report of 21 cases. Neurochirurgie 45:129–133. [PubMed] [Google Scholar]
- 4. Dastur HM (1983) Diagnosis and neurosurgical treatment of tuberculous disease of the CNS. Neurosurg Rev 6:111–117. [DOI] [PubMed] [Google Scholar]
- 5. DeAngelis LM (1981) Intracranial tuberculoma: case report and review of the literature. Neurology 31:1133–1136. [DOI] [PubMed] [Google Scholar]
- 6. Di Patre PL, Radziszewski W, Martin NA, Brooks A, Vinters HV (2000) A meningioma‐mimicking tumor caused by Mycobacterium avium complex in an immunocompromised patient. Am J Surg Pathol 24:136–139. [DOI] [PubMed] [Google Scholar]
- 7. Indira B, Panigrahi MK, Vajramani G, Shankar SK, Santosh V, Das BS (1996) Tuberculoma of the hypothalamic region as a rare case of hypopituitarism: a case report. Surg Neurol 45:347–350. [DOI] [PubMed] [Google Scholar]
- 8. Kemaloglu S, Gur A, Nas K, Cevik R, Buyukbayram H, Sarac AJ (2001) Intramedullary tuberculoma of the conus medullaris: case report and review of the literature. Spinal Cord 39:498–501. [DOI] [PubMed] [Google Scholar]
- 9. Lindner A, Schneider C, Hofmann E, Soerensen N, Toyka KV (1995) Isolated meningeal tuberculoma mimicking meningioma: case report. Surg Neurol 43:81–84. [DOI] [PubMed] [Google Scholar]
- 10. Morrison A, Gyure KA, Stone J, Wong K, McEvoy P, Koeller K, Mena H (1999) Mycobacterial spindle cell pseudotumor of the brain: a case report and review of the literature. Am J Surg Pathol 23:1294–1299. [DOI] [PubMed] [Google Scholar]
- 11. Paramo C, De L, Nodar A, Miramontes S, Quintela JL, Garcia‐Mayor RV (2002) Intrasellar tuberculoma—a difficult diagnosis. Infection 30:35–37. [DOI] [PubMed] [Google Scholar]
- 12. Sumi MG, Mathai R, Sheela R, Radhakrishnan NS, Radhakrishnan VV, Indhulekshmy R, Mundayoor S (2001) Diagnostic utility of polymerase chain reaction and immunohistochemical techniques for the laboratory diagnosis of intracranial tuberculoma. Clin Neuropath 20:176–180. [PubMed] [Google Scholar]
- 13. Talamas O, Del Brutto OH, Garcia‐Ramos G (1989) Brain‐stem tuberculoma. An analysis of 11 patients. Arch Neurol 46:529–535. [DOI] [PubMed] [Google Scholar]
- 14. Wasay M, Kheleani BA, Moolani MK, Zaheer J, Pui M, Hasan S, Muzaffar S, Bakshi R, Sarawari AR (2003) Brain CT and MRI findings in 100 consecutive patients with intracranial tuberculoma. J Neuroimaging 13:240–247. [PubMed] [Google Scholar]


