Abstract
Proteus mirabilis is a common opportunistic pathogen causing severe illness in humans and animals. To determine the prevalence, antibiogram, biofilm-formation, screening of virulence, and antimicrobial resistance genes in P. mirabilis isolates from ducks; 240 samples were obtained from apparently healthy and diseased ducks from private farms in Port-Said Province, Egypt. The collected samples were examined bacteriologically, and then the recovered isolates were tested for atpD gene sequencing, antimicrobial susceptibility, biofilm-formation, PCR detection of virulence, and antimicrobial resistance genes. The prevalence of P. mirabilis in the examined samples was 14.6% (35/240). The identification of the recovered isolates was confirmed by the atpD gene sequencing, where the tested isolates shared a common ancestor. Besides, 94.3% of P. mirabilis isolates were biofilm producers. The recovered isolates were resistant to penicillins, sulfonamides, β-Lactam-β-lactamase-inhibitor-combinations, tetracyclines, cephalosporins, macrolides, and quinolones. Using PCR, the retrieved strains harbored atpD, ureC, rsbA, and zapA virulence genes with a prevalence of 100%, 100%, 94.3%, and 91.4%, respectively. Moreover, 31.4% (11/35) of the recovered strains were XDR to 8 antimicrobial classes that harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. Besides, 22.8% (8/35) of the tested strains were MDR to 3 antimicrobial classes and possessed blaTEM, tetA, and sul1genes. Furthermore, 17.1% (6/35) of the tested strains were MDR to 7 antimicrobial classes and harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. Alarmingly, three strains were carbapenem-resistant that exhibited PDR to all the tested 10 antimicrobial classes and shared blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. Of them, two strains harbored the blaNDM-1 gene, and one strain carried the blaKPC gene. In brief, to the best of our knowledge, this is the first study demonstrating the emergence of XDR and MDR-P.mirabilis in ducks. Norfloxacin exhibited promising antibacterial activity against the recovered XDR and MDR-P. mirabilis. The emergence of PDR, XDR, and MDR-strains constitutes a threat alarm that indicates the complicated treatment of the infections caused by these superbugs.
Subject terms: Antimicrobials, Bacteriology, Biofilms, Pathogens
Introduction
The genus Proteus includes Gram-negative, moderate-sized, non-sporulated, and motile rods. Proteus mirabilis is one of the most prevalent Proteus species. P. mirabilis normally inhabits the intestinal tract of both humans and animals as normal flora. Besides, it's a ubiquitous environmental microorganism widely distributed in nature1. P. mirabilis is known as an opportunistic bacterial pathogen that incriminated in community-acquired infections, food-borne infections, serious nosocomial infections, and urinary tract infections in humans2–5. Furthermore, several recent studies proved the existence of P. mirabilis in animals and poultry. The molecular typing of P. mirabilis from human and animal origins revealed that the animal strains could be transmitted to humans6–8.
The emergence of multidrug-resistant bacterial pathogens is reflected as a public health risk. Several investigations reported the occurrence of MDR pathogens from different origins including humans, animals, birds, fish, and food9–18. The emergence of extended-spectrum β-lactam resistant Proteus species had been reported for the first time in 1987 that is considered a thoughtful public health concern globally19. Besides, the existence of MDR-Proteus species was reported in previous studies6,20,21.
The antimicrobial resistance genes could be transmitted to P. mirabilis from other resistant pathogens in the environment and the gastrointestinal tract, especially the extended-spectrum β-lactamase genes including; blaTEM gene: encoded for penicillins-resistance, blaCTX gene: encoded for cephalosporins-resistance, blaNDM1 gene: encoded for carbapenem-resistance, and blaOXA-1 gene: encoded for and piperacillin and cephalosporins-resistance4,22. P. mirabilis is known as a common biofilm producer. The bacterial biofilm protects the bacteria from the phagocytic cells, the environmental stresses, and different antimicrobial agents. Moreover, it is considered a frequent source of infection3,5.
PCR is a rapid and specific reliable epidemiological tool used for screening virulence and antimicrobial resistance genes in certain bacterial pathogens. The atpD gene is one of the most conserved genes in Proteus species that encodes for ATP synthase β-subunit. The pathogenicity of P. mirabilis is regulated by several virulence determinants that are controlled by multiple virulence genes such as ureC, zapA, and rsbA virulence genes. The rsbA gene is responsible for swarming modulation in Proteus species. Moreover, the ureC gene is the principal gene responsible for urease enzyme production that plays a major role in stone formation in kidneys or bladder during urinary tract infections. Furthermore, the zapA gene is encoded for protease production, especially IgA protease2,5,6,23.
This study is aimed to investigate the prevalence, atpD gene sequencing, antibiogram, PCR detection of virulence genes (ureC, zapA, and rsbA), and antimicrobial resistance genes (blaTEM, blaCTX, blaNDM-1, blaKPC, blaOXA-1, sul1, and tetA) of emerging P. mirabilis in ducks.
Material and methods
Animal ethics
The study was carried out in compliance with the ARRIVE guidelines. All methods were performed according to relevant guidelines and regulations. Handling of birds and all the experimental protocols conducted by well-trained scientists and were approved by the Animal Ethics Review Committee of Suez Canal University (AERC-SCU), Egypt.
Sampling
Approximately, 240 specimens were obtained from apparently healthy (n = 40) and diseased ducks (n = 40) from private duck commercial farms (Muscovy duck with average age 70 days) at Port-Said Province, Egypt (From May 2020 to August 2020). Tracheal and cloacal swabs were collected from live birds, while the internal organs were collected separately under complete aseptic conditions from freshly dead and slaughtered ducks as illustrated in Table1. Diseased ducks exhibited diarrhea and respiratory manifestations. Specimens were collected in peptone water (Oxoid, UK) and rapidly transmitted to the lab as soon as possible for bacteriological examination.
Table 1.
Various types of samples obtained from apparently healthy and diseased ducks.
| Types of samples | Duck condition | |||
|---|---|---|---|---|
| Apparently healthy n = 40 |
Diseased ducks n = 40 |
|||
| Live n = 20 |
Freshly slaughtered n = 20 |
Live n = 20 |
Dead n = 20 |
|
|
Tracheal swabs n = 40 |
20 | – | 20 | – |
|
Cloacal swabs n = 40 |
20 | – | 20 | – |
|
Liver n = 40 |
– | 20 | – | 20 |
|
Heart n = 40 |
– | 20 | – | 20 |
|
Lung n = 40 |
– | 20 | – | 20 |
|
Gizzard n = 40 |
– | 20 | – | 20 |
| Sub-total | 40 | 80 | 40 | 80 |
| Total | 240 | |||
Isolation and identification of P. mirabilis
The obtained samples were enriched in peptone water (Oxoid, Hampshire, UK) at 37 °C for 24 h. A loopful from the enriched broth was streaked on XLD, 5% sheep blood agar, MacConkey agar, and TSI (Oxoid, Hampshire, UK), then left incubated at 37 °C for 24–48 h. The identification of suspected colonies was performed according to their culture characters, swarming activity, hemolytic activity, morphological characteristics using Gram's-staining, and biochemical characters as described by Quinn24. Moreover, the identification of P. mirabilis was confirmed by the PCR detection of the atpD gene as described by Bi25 (Table 2), followed by gene sequencing of the PCR products.
Table 2.
The oligonucleotides sequences and thermal-cycling conditions of PCR assay.
| Genes | Oligonucleotides sequences | Amplified product (bp) | PCR conditions (35 cycles) | References | ||
|---|---|---|---|---|---|---|
| Denaturation | Annealing | Extension | ||||
| atpD |
GTATCATGAACGTTCTGGGTAC TGAAGTGATACGCTCTTGCAG |
595 |
94 °C 30 s |
58 °C 40 s |
72 °C 45 s |
25 |
| ureC |
GTTATTCGTGATGGTATGGG ATAAAGGTGGTTACGCCAGA |
317 |
94 °C 30 s |
56 °C 40 s |
72 °C 40 s |
30 |
| rsbA | TTGAAGGACGCGATCAGACC ACTCTGCTGTCCTGTGGGTA | 467 |
94 °C 30 s |
58 °C 40 s |
72 °C 45 s |
30 |
| zabA |
ACCGCAGGAAAACATATAGCCC GCGACTATCTTCCGCATAATCA |
540 |
94 °C 30 s |
59 °C 40 s |
72 °C 45 s |
30 |
| tetA |
GGTTCACTCGAACGACGTCA CTGTCCGACAAGTTGCATGA |
576 |
94 °C 30 s |
50 °C 40 s |
72 °C 45 s |
31 |
| sul1 | CGGCGTGGGCTACCTGAACG GCCGATCGCGTGAAGTTCCG | 433 |
94 °C 30 s |
54 °C 40 s |
72 °C 45 s |
32 |
| blaKPC |
ATGTCACTGTATCGCCGTCT TTACTGCCCGTTGACGCCC |
892 |
94 °C 1 min |
55 °C 1 min |
72 °C 1 min |
33 |
| blaNDM-1 |
GGCGGAATGGCTCATCACGA CGCAACACAGCCTGACTTTC |
287 |
94 °C 30 s |
55 °C 30 s |
72 °C 30 s |
33 |
| blaCTX-M |
ATG TGC AGY ACC AGT AAR GTK ATG GC TGG GTR AAR TAR GTS ACC AGA AYC AGC GG |
593 |
94 °C 30 s |
54 °C 40 s |
72 °C 45 s |
34 |
| blaOXA-1 |
ATATCTCTACTGTTGCATCTCC AAACCCTTCAAACCATCC |
619 |
94 °C 30 s |
54 °C 40 s |
72 °C 45 s |
35 |
| blaTEM |
ATCAGCAATAAACCAGC CCCCGAAGAACGTTTTC |
516 |
94 °C 30 s |
54 °C 40 s |
72 °C 45 s |
35 |
The atpD gene sequencing and phylogenetic analyses
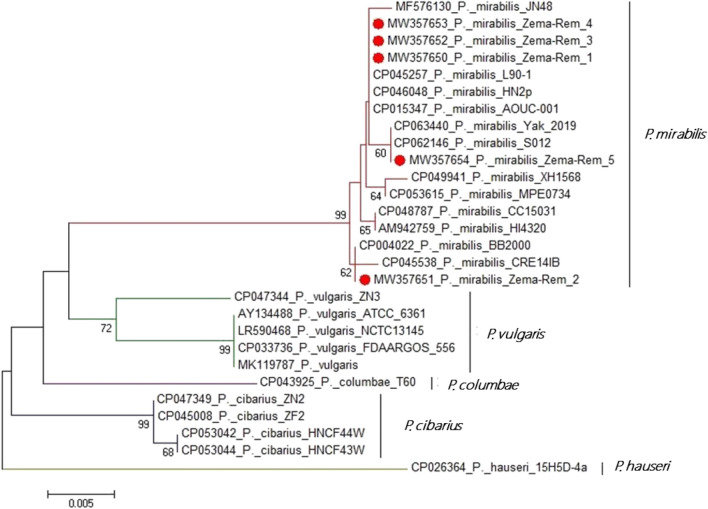
Since the retrieved isolates exhibited harmony in their phenotypic and biochemical characteristics: the PCR products of 5 randomly selected isolates were purified with a QIAquick PCR-Product extraction kit (QIAGEN Sciences Inc., Germantown, MD, USA) and sent for direct sequencing using the same set of primers. The sequencing was carried out using the Bigdye Terminator V3.1 cycle sequencing kit (Thermo Fisher Scientific, Waltham, MA, USA). The sequencing was performed using the Applied Biosystems 3130 genetic analyzer (HITACHI, Japan), and the retrieved sequences were deposited in the GenBank with accession numbers: MW357650, MW357651, MW357652, MW357653, and MW357654. To detect the sequence identity to GenBank accessions, the BLAST analysis (Basic Local Alignment Search Tool) was done. The phylogenetic tree was generated by the MegAlign module of LasergeneDNAStar version 12.1 using maximum likelihood, neighbor-joining, and maximum parsimony in MEGA626.
Antimicrobial susceptibility testing of P. mirabilis
The disc diffusion method was carried out to investigate the antibiogram of the obtained P. mirabilis isolates using Mueller-Hinton agar (Oxoid, Hampshire, UK). Fifteen antimicrobial agents were involved; colistin sulfate (CT) (10 μg), ceftazidime (CAZ) (30 μg), amoxicillin (AMX) (10 μg), norfloxacin (NOR) (10 μg), piperacillin (PRL) (10 μg), amoxicillin-clavulanic acid (AMC) (30 μg), imipenem (IPM) (10 μg), nalidixic acid (ND) (30 μg), ampicillin (AMP) (10 μg ), cefotaxime (30 μg) (CTX), erythromycin (E) (15 μg), ampicillin-sulbactam(SAM) (30 μg), meropenem (MEM) (10 μg), trimethoprim-sulfamethoxazole (SXT) (19:1 μg), and doxycycline (DOX) (10 μg) (Oxoid, UK). The test was performed using E. coli-ATCC 35218 as a control strain. The diameter of the inhibition zone was estimated as described by CLSI27. The phenotypic resistance patterns are categorized into PDR, XDR, and MDR according to Magiorakos28.
Estimation of the biofilm formation in the isolated P. mirabilis
Estimation of biofilm formation was carried out in glass test tubes as previously described by Kadam29. Briefly, each P. mirabilis strain was inoculated in tryptic soy broth (Oxoid, Hampshire, UK), and left incubated overnight at 28 °C without shaking. Negative control experiments were carried out with sterile broth. After discarding the broth, the incubated tubes were stained with 1% crystal violet (to observe cells attached to the test tube) and were incubated for 15 min. Then, the tubes were washed with sterile distilled water. The test was repeated three times for each strain. Positive results indicated by the formation of purple biofilms.
PCR detection of virulence and antimicrobial resistance genes in the retrieved P. mirabilis
The PCR-based detection of ureC, zapA, and rsbA virulence genes and blaTEM, blaCTX, blaNDM-1, blaKPC, blaOXA-1, sul1, and tetA antimicrobial resistance genes was performed. Extraction of bacterial DNA was carried out according to the descriptions of the QIAamp DNA Mini Kit (QIAGEN Sciences Inc., Germantown, MD, USA/ Cat. No. ID 51326). Accordingly, the reaction volume was 50 μl as follows: 5 μl of 10 × PCR reaction buffer, 1 μl 200 μM (of each dNTP) of dNTP mix (10 mM), 4 μl of bacterial-DNA, 0.4 μl 2 U of Taq DNA Polymerase (5 U/μl), 30 pmol of each used primer (0.1–0.6 μM), and then Sterile ddH2O was added up to 50 μl. Positive control strains (kindly supported by the Biotechnology Center of AHRI, Egypt) were involved in all PCR reactions. Besides, a reaction without a DNA-template was used as a negative control. Oligonucleotides-sequences (Thermo Fisher Scientific, Waltham, MA, USA) and the thermal-cycling protocols are described in Table 2. The agar gel electrophoresis was carried out for the separation of the obtained PCR-products using 1.5% agarose stained with ethidium bromide 0.5 μg/ml and followed by photographing the gel.
Statistical analyses
The obtained findings were analysed using the Chi-square test (SAS software, version 9.4, SAS Institute, Cary, NC, USA) (Significance-level; P < 0.05). Besides, the correlation coefficient and the non-parametric Wilcox signed-rank test were performed using R-software (version 4.0.2; https//www.r-project.org/).
Results
Phenotypic characteristics and prevalence of P. mirabilis in the examined samples
The recovered colonies are red with black center on XLD, pale colonies (non-lactose fermenter) on MacConkey agar, black colonies on TSI (H2S producer), hemolytic on blood agar, and undergo the characteristic swarming activity. Biochemically, the retrieved isolates were positive for catalase, H2S production, urease, methyl red, and citrate utilization tests, while are negative for oxidase, lactose fermentation, indole, and Voges-Proskauer tests. The prevalence of P. mirabilis among the examined birds was 25% (20/80); the prevalence was 15% (6/40) in the examined apparently healthy ducks, while it was 35% (14/40) in the examined diseased ducks (Table 3). Concerning the distribution of P. mirabilis in the examined samples, the total prevalence of P. mirabilis was 14.6% (35/240); the prevalence of P. mirabilis was 10.8% (13/120) in the examined samples of apparently healthy ducks, while the prevalence was 18.3% (22/120) in the examined samples of diseased ducks. The most predominant infected organ was the liver, followed by the heart and lung. Statistically, there is a significant difference in the prevalence of P. mirabilis between the examined apparently healthy and diseased ducks (P < 0.05), whereas there is no significant difference (P > 0.05) among different examined samples (Table 4 and Fig. 1).
Table 3.
Prevalence of P. mirabilis among the examined birds.
| Bird condition | No of examined birds | Positive for P. mirabilis | |
|---|---|---|---|
| No | % | ||
| Apparently healthy | Alive (n = 20) | 2 | 10 |
| Freshly dead (n = 20) | 4 | 20 | |
| Subtotal | 6 | 15 (6/40) | |
| Diseased | Alive (n = 20) | 5 | 25 |
| Freshly dead (n = 20) | 9 | 45 | |
| Subtotal | 14 | 35(14/40) | |
| Total | 80 | 20 | 25 (20/80) |
Table 4.
Prevalence of P. mirabilis in different examined samples of apparently healthy and diseased ducks.
| Type of sample n = 40 (for each type of samples) |
Proteus mirabilis | Total (total number of samples = 240) | ||||
|---|---|---|---|---|---|---|
| Apparently healthy ducks (No. of samples = 120) | Diseased ducks (No. of samples = 120) | |||||
| N | % | N | % | N | % | |
| Tracheal swabs | 2 | 1.6 | 3 | 2.5 | 5 | 2.08 |
| Cloacal swabs | 2 | 1.6 | 2 | 1.6 | 4 | 1.66 |
| Heart | 2 | 1.6 | 5 | 4.16 | 7 | 2.9 |
| Liver | 4 | 3.3 | 5 | 4.16 | 9 | 3.75 |
| Lung | 2 | 1.6 | 4 | 3.3 | 6 | 2.5 |
| Gizzard | 1 | 0.8 | 3 | 2.5 | 4 | 1.66 |
| Total | 13 | 10.8 | 22 | 18.3 | 35 | 14.6 |
|
Chi square P-value |
2.2308 0.8164 |
2 0.8491 |
3.2286 0.6648 |
|||
| Wilcoxon test p-value | 0.04123 | |||||
Figure 1.
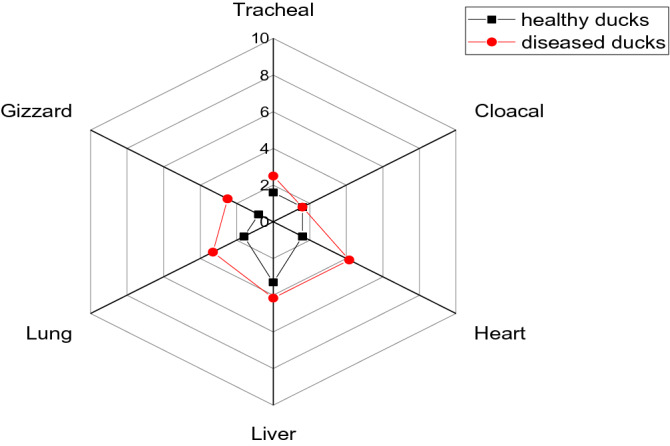
The radar illustrates the prevalence of P. mirabilis in different examined samples of apparently healthy and diseased ducks.
Sequence analysis of the atpD gene
The atpD gene sequencing and the phylogenetic analysis proved that the tested P. mirabilis isolates (n = 5) shared a common ancestor. Moreover, the tested isolates showed high genetic identity to other strains of P. mirabilis such as P. mirabilis strain HI4320 of United Kingdom (Accession No. AM942759), P. mirabilis strain BB2000 of China (Accession No. MF576130), P. mirabilis strain BB2000 (Accession No. CP045538) and strain AOUC-001 (Accession No. CP015347) of Italy, and P. mirabilis strain BB2000 of USA (Accession No. CP004022) as illustrated in Fig. 2.
Figure 2.
The phylogenetic analyses were based on the atpD gene sequencing. The phylogenetic tree illustrates the genetic relatedness of the retrieved P. mirabilis isolates and other relevant isolates deposited in the GenBank database. The tree topology was assessed by bootstrap analysis of 1000 replicates. The bacteria recovered in the present study are marked with solid red circles.
Antibiogram and the phenotypic resistance patterns of the isolated P. mirabilis
The recovered P. mirabilis isolates exhibited remarkable resistance-patterns to various antimicrobial classes including; Penicillins: amoxicillin and penicillin (100%), and piperacillin (77.1%), β-Lactam-β-lactamase-inhibitor combination: ampicillin-sulbactam and amoxicillin-clavulanic acid (71.7%), Sulfonamides: trimethoprim-sulfamethoxazole (100%); Tetracyclines: doxycycline (100%), Quinolones: nalidixic acid (62.8%), Macrolides: erythromycin (62.8%), and Cephalosporins: ceftazidime and cefotaxime (57.1%). Moreover, the tested isolates displayed intermediate resistance to Polymyxins: colistin sulfate (51.4%). Besides, the retrieved isolates were sensitive to Fluoroquinolones: norfloxacin (85.7%), and Carbapenems: meropenem (77.1%), and imipenem (74.3%). Furthermore, 8.3% of the tested P. mirabilis isolates (n = 3) were found to be carbapenem-resistant strains (Table 5, Supplementary Table S1, and Fig. 3). Statistically, there is a significant difference (P < 0.05) in the susceptibility of the obtained P. mirabilis isolates to different tested antimicrobial agents. Moreover, the correlation-coefficient between various involved antimicrobial agents was estimated.
Table 5.
Antibiogram of the isolated P. mirabilis strains (n = 35).
| Antimicrobial classes | Antimicrobial agents | P. mirabilis (n = 35) | |||||
|---|---|---|---|---|---|---|---|
| S | I | R | |||||
| N | % | N | % | N | % | ||
| Fluoroquinolones | Norfloxacin | 30 | 85.7 | 2 | 5.7 | 3 | 8.6 |
| Penicillins | Amoxicillin | 0 | 0 | 0 | 0 | 35 | 100 |
| Penicillin | 0 | 0 | 0 | 0 | 35 | 100 | |
| Piperacillin | 3 | 8.6 | 5 | 14.3 | 27 | 77.1 | |
| β -Lactam- β-lactamase-inhibitor combinations | Ampicillin-Sulbactam | 3 | 8.6 | 5 | 14.3 | 27 | 77.1 |
| Amoxicillin-clavulanic acid | 4 | 11.4 | 4 | 11.4 | 27 | 77.1 | |
| Carbapenems | Meropenem | 27 | 77.1 | 5 | 14.3 | 3 | 8.6 |
| Imipenem | 26 | 74.3 | 6 | 17.1 | 3 | 8.6 | |
| Cephalosporins | Cefotaxime | 5 | 14.3 | 10 | 28.6 | 20 | 57.1 |
| Ceftazidime | 6 | 17.1 | 9 | 25.7 | 20 | 57.1 | |
| Sulfonamides | Trimethoprim-Sulfamethoxazole | 0 | 0 | 0 | 0 | 35 | 100 |
| Quinolones | Nalidixic acid | 0 | 0 | 13 | 37.1 | 22 | 62.8 |
| Tetracyclines | Doxycycline | 0 | 0 | 0 | 0 | 35 | 100 |
| Macrolides | Erythromycin | 3 | 8.6 | 10 | 28.6 | 22 | 62.8 |
| Polymyxins | Colistin sulfate | 3 | 8.6 | 18 | 51.4 | 14 | 40 |
| Chi square | 219.73 | 69.034 | 87.152 | ||||
| P value | < 0.0001 | < 0.0001 | < 0.0001 | ||||
Figure 3.
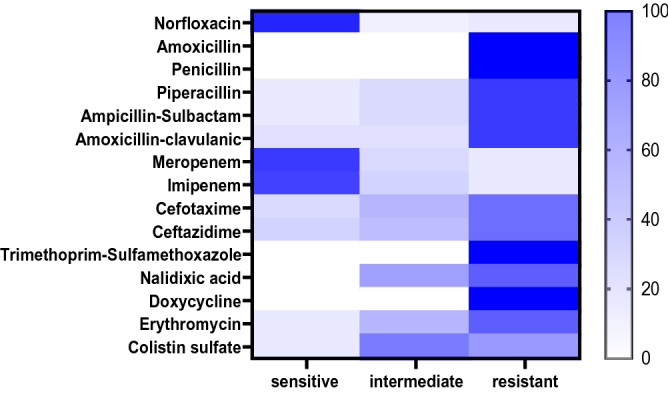
The heat-map illustrates the different degrees of susceptibility (sensitive, intermediate, and resistant) of the retrieved P. mirabilis to different tested antimicrobial agents.
Our findings proved a remarkable positive correlations (r = 0.5–1) between: NOR, IPM, and MEM (r = 0.99); E and CAZ (r = 0.99); CTX and CAZ (r = 0.99); SAM, PRL, and CAZ (r = 0.99); AMP, AMX, SXT, AMC, DOX, and CTX (r = 0.94); NA and CT (r = 0.99); AMP, AMX, SXT, AMC, DOX, and CAZ (r = 0.98); CTX and SAM (r = 0.98); SAM and CTX (r = 0.97); PRL and CTX (r = 0.96); E and SAM (r = 0.96); E and PRL (r = 0.94); AMP, AMX, SXT, AMC, DOX, and E (r = 0.93); E and CT (r = 0.89); CTX and CT (r = 0.87); as described in the heat-map (Fig. 4).
Figure 4.

The heat-map illustrates the correlation-coefficient (r) among various antimicrobial agents. Blue and red color points to the positive and negative correlations, respectively.
The prevalence of biofilm formation among the recovered P. mirabilis strains
Approximately 94.3% (33/35) of the isolated P. mirabilis strains were biofilm producers, while 5.7% (2/35) of the tested strains are non-biofilm producers. Of the biofilm producers (n = 33), 25 strains (75.8%) were strong biofilm producers, 5 strains (15.1%) were moderate biofilm producers, and 3 strains were weak biofilm producers (9.1%) as described in Fig. 5.
Figure 5.
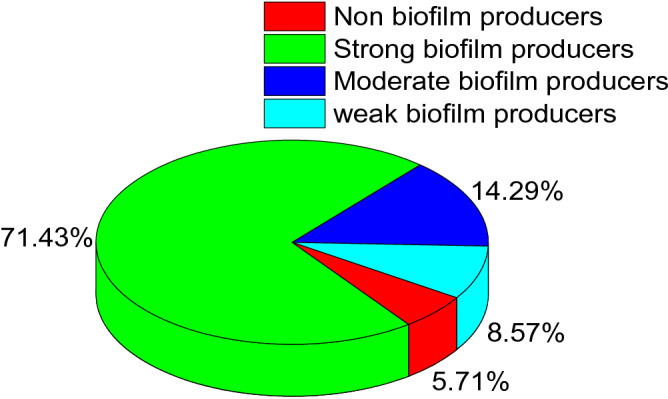
Illustrates the prevalence of biofilm-formation among the retrieved P. mirabilis strains. The percentage was calculated according to the total number of the retrieved isolates (n = 35).
Virulence and antimicrobial resistance genes of the recovered P. mirabilis strains
The PCR revealed that the isolated P. mirabilis strains harbored atpD, ureC, rsbA, and zapA virulence genes with a prevalence of 100%, 100%, 94.3%, and 91.4%, respectively. Furthermore, the tested strains harbored blaTEM, sul1, tetA, blaOXA-1, blaCTX-M, blaNDM-1, and blaKPC antimicrobial-resistance genes with a prevalence of 100%, 100%,100%, 80%, 57.1%, 5.7%, and 2.9%, respectively, as illustrated in Table 6 and Fig. 6. Statistically, there is no significant difference (P > 0.05) in the occurrence of virulence genes among the retrieved P. mirabilis strains, whereas there is a significant difference (P < 0.05) in the frequency of the antimicrobial resistance genes between the tested strains.
Table 6.
Virulence and antimicrobial resistance genes that associated with the retrieved P. mirabilis (n = 35).
| Types of genes | N | % | Chi square P-value |
|
|---|---|---|---|---|
| Virulence-determinant genes | atpD | 35 | 100 |
0.2 0.9776 |
| ureC | 35 | 100 | ||
| rsbA | 33 | 94.3 | ||
| zapA | 32 | 91.4 | ||
| Antimicrobial resistance genes | blaTEM | 35 | 100 |
62.256 < 0.0001 |
| tetA | 35 | 100 | ||
| sul1 | 35 | 100 | ||
| blaOXA-1 | 28 | 80 | ||
| blaCTX-M | 20 | 57.1 | ||
| bla-NDM-1 | 2 | 5.7 | ||
| blaKPC | 1 | 2.9 |
Figure 6.
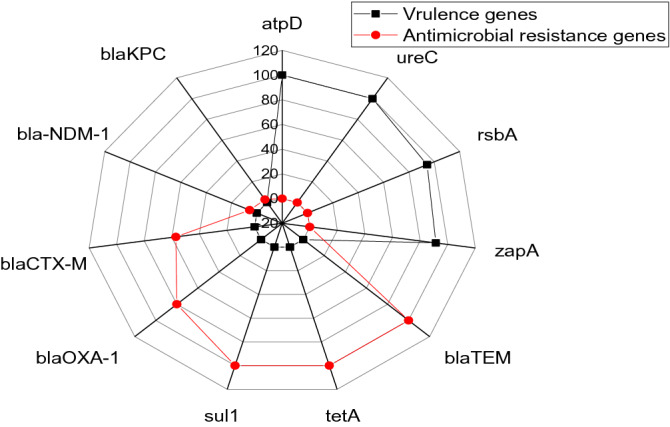
The radar reveals the frequency of virulence genes and antimicrobial resistant genes between the retrieved P. mirabilis strains.
The correlation between the phenotypic and genotypic multidrug-resistance patterns in P. mirabilis
Our findings revealed that 31.4% (11/35) of the retrieved P. mirabilis strains are extensively drug-resistant (XDR: resistant to ≥ one agent in all but ≤ two antimicrobial classes) to 8 antimicrobial classes (Penicillins: amoxicillin, ampicillin, and piperacillin, β -Lactam- β-lactamase inhibitor combination: ampicillin-sulbactam, and amoxicillin-clavulanic acid, Cephalosporins: cefotaxime, and ceftazidime, Sulfonamides: trimethoprim-Sulfamethoxazole, Tetracyclines: doxycycline, Quinolones: nalidixic acid, Macrolides: erythromycin, and Polymyxins: colistin sulfate) and harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 resistance genes. Furthermore, 22.8% (8/35) of the tested strains revealed multidrug resistance (MDR: resistant to ≥ one agent in ≥ 3 antimicrobial classes) to 3 antimicrobial classes (Tetracyclines: doxycycline, Penicillins: amoxicillin, and ampicillin, and Sulfonamides: trimethoprim-sulfamethoxazole) and possessed tetA, blaTEM, and sul1 resistance genes.
Besides, 17.1% (6/35) of the tested strains are MDR to 7 antimicrobial classes (Penicillins: amoxicillin, ampicillin, and piperacillin, β -Lactam- β-lactamase inhibitor combinations, Cephalosporins: cefotaxime, and ceftazidime, Sulfonamides: trimethoprim-Sulfamethoxazole, Tetracyclines: doxycycline, Quinolones: nalidixic acid, and Macrolides: erythromycin), and carried blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 resistance genes. Moreover, 14.2% (5/35) of the tested strains are MDR to 4 antimicrobial classes (Penicillins: amoxicillin, ampicillin, and piperacillin, β -Lactam- β-lactamase inhibitor combinations, Sulfonamides: trimethoprim-Sulfamethoxazole, and Tetracyclines: doxycycline) and harbored blaTEM, blaOXA-1, tetA, and sul1 resistance genes.
Unfortunately, 3 strains are Pan-drug resistant (PDR) to all the tested 10 antimicrobial classes (Carbapenems: imipenem and meropenem, Fluoroquinolones: norfloxacin, Penicillins: amoxicillin, ampicillin, and piperacillin, β -Lactam- β-lactamase inhibitor combinations, Sulfonamides: trimethoprim-Sulfamethoxazole, Cephalosporins: cefotaxime, and ceftazidime, Tetracyclines: doxycycline, Quinolones: nalidixic acid, Macrolides: erythromycin, and Polymyxins: colistin sulfate); two strains harbored blaTEM, blaOXA-1, blaCTX-M, blaNDM-1, tetA and, sul1 genes, while one strain harbored blaTEM, blaOXA-1, blaCTX-M, blaKPC, tetA, and sul1 resistant genes as described in Table 7 and Fig. 7.
Table 7.
The correlation between phenotypic and genotypic resistance patterns among the retrieved P. mirabilis (n = 35).
| No. of strains | % | Type of resistance | In-vitro phenotypic resistance | The antimicrobial resistance genes |
|---|---|---|---|---|
| 11 | 31.4 | XDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 |
| β-Lactam-β-lactamase inhibitor combination: | ||||
| ampicillin-sulbactam, and amoxicillin-clavulanic acid | ||||
| Cephalosporins: cefotaxime, and ceftazidime, | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| Quinolones: nalidixic acid | ||||
| Macrolides: erythromycin | ||||
| Polymyxins: colistin sulfate | ||||
| 8 | 22.8 | MDR | Penicillins: amoxicillin, and ampicillin | blaTEM, tetA, and sul1 |
| Tetracyclines: doxycycline | ||||
| Sulfonamides: trimethoprim-sulfamethoxazole | ||||
| 6 | 17.1 | MDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 |
| β -Lactam- β-lactamase inhibitor combinations | ||||
| Cephalosporins: cefotaxime, and ceftazidime | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| Quinolones: nalidixic acid | ||||
| Macrolides: erythromycin | ||||
| 5 | 14.2 | MDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, tetA, and sul1 |
| β -Lactam- β-lactamase inhibitor combinations | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| 2 | 5.7 | PDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, blaCTX-M, blaNDM-1, tetA, and sul1 |
| β -Lactam- β-lactamase inhibitor combinations | ||||
| Cephalosporins: cefotaxime, and ceftazidime | ||||
| Fluroquinolones: Norfloxacin | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| Quinolones: nalidixic acid | ||||
| Macrolides: erythromycin | ||||
| Polymyxins: colistin sulfate | ||||
| Carbapenems: imipenem and meropenem | ||||
| 2 | 5.7 | MDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, tetA, and sul1 |
| β -Lactam- β-lactamase inhibitor combinations | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| Quinolones: nalidixic acid | ||||
| Macrolides: erythromycin | ||||
| 1 | 2.9 | PDR | Penicillins: amoxicillin, ampicillin, and piperacillin | blaTEM, blaOXA-1, blaCTX-M, blaKPC, tetA, and sul1 |
| β -Lactam- β-lactamase inhibitor combinations | ||||
| Cephalosporins: cefotaxime, and ceftazidime | ||||
| Fluroquinolones: Norfloxacin | ||||
| Sulfonamides: trimethoprim-Sulfamethoxazole | ||||
| Tetracyclines: doxycycline | ||||
| Quinolones: nalidixic acid | ||||
| Macrolides: erythromycin | ||||
| Polymyxins: colistin sulfate | ||||
| Carbapenems: imipenem and meropenem |
Figure 7.
Illustrates the distribution of PDR, XDR, and MDR among the retrieved P. mirabilis strains.
The correlation-coefficient (r) was estimated between different tested antimicrobial agents and the antimicrobial resistance genes. our findings proved a remarkable positive correlations between: (r = 0.5–1); sul1 gene and SXT (r = 1); tetA gene and DOX (r = 1), blaCTX-M gene and CTX (r = 1); blaCTX-M gene and CAZ (r = 0.99); blaKPC gene, MEM, and IPM (r = 0.99); blaNDM-1 gene, MEM, and IPM (r = 0.99); blaTEM gene, AMX, and AMP (r = 0.99); blaOXA-1 gene, AMC, AMX, and AMP (r = 0.97); blaOXA-1 gene and PRL (r = 0.96); blaOXA-1 gene and SAM (r = 0.95); blaOXA-1 gene and CAZ (r = 0.90); blaOXA-1 gene and CTX (r = 0.84); blaCTX-M gene and blaOXA-1 gene (r = 0.84); as illustrated in the heat-map (Fig. 8).
Figure 8.
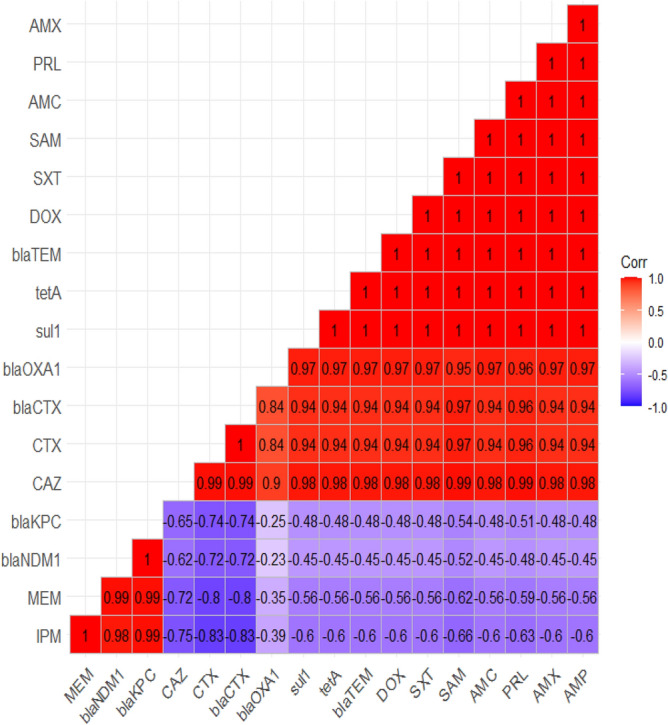
The Heat-map demonstrates the correlation-coefficient (r) between different tested antimicrobial agents and the antimicrobial resistance genes.
Discussion
Proteus mirabilis is frequently incriminated in food-borne infections and urinary tract infections in humans. Few studies are concerning the emergence of P. mirabilis in birds. The current study was directed to investigate the prevalence, atpD gene sequencing, antimicrobial-resistance profiles, PCR-based detection of virulence genes (ureC, zapA, and rsbA), and antimicrobial resistance genes (blaTEM, blaCTX, blaKPC, blaNDM-1, blaOXA-1, sul1, and tetA) of emerging P. mirabilis in ducks.
The bacteriological examination evidenced that the prevalence of P. mirabilis in the examined samples was 14.6% (35/240). Besides, there is no ambivalence in the phenotypic characteristics of the retrieved P. mirabilis strains that revealed a significant harmony between the isolates: red colonies with black center on XLD, pale colonies (non-lactose fermenter) on MacConkey agar, black colonies on TSI, and undergo a characteristic swarming activity. Biochemically: the retrieved isolates are positive for catalase, H2S, urease, methyl-red, and citrate utilization tests, whereas they are negative for oxidase, lactose fermentation, indole, and Voges-Proskauer tests. These results are in agreement with those obtained by Lei6 and Reich7. In the present study, P. mirabilis was isolated from the internal organs of the examined birds in a pure form suggesting that the retrieved isolates were the primary bacterial cause of these infections in ducks. These results were supported by the previous findings that were reported by Barbour36 and Yeh37. P. mirabilis is a ubiquitous pathogen widely distributed in the environment38. P. mirabilis is an opportunistic pathogen that is incriminated in various infections in humans, animals, and poultry. Recently, several studies reported the emergence of P. mirabilis in food-producing animals, especially poultry36,38,39.
In the present study, P. mirabilis could be isolated from the internal organs of both apparently healthy and diseased birds. P. mirabilis is an opportunistic microorganism that normally inhabits the alimentary tract of birds, animals, and humans. The microorganism could escape from the intestinal tract and reach other internal organs. Thus, it could be responsible for other illnesses associated with the spread of P. mirabilis to other internal organs, and in severe cases, it could cause sepsis. In the meantime, the development of the clinical signs depends mainly on the onset of the disease as well as the immune status of the bird40.
The atpD gene phylogenetic analysis revealed that the tested P. mirabilis isolates (n = 5) are shared a common ancestor. Besides, they exhibited high genetic identity with other P. mirabilis strains of human origin that were previously isolated in Italy41,42, China43, USA44, and United Kingdom45. Our findings conceived the epidemiological map and emphasized the zoonotic impact of P. mirabilis that is considered a public health threat.
Concerning the in-vitro antimicrobial-resistance profiles; the recovered P. mirabilis strains showed remarkable resistance-patterns to penicillins, β-Lactam β-lactamase-inhibitor combinations, cephalosporins, sulfonamides, tetracyclines, macrolides, and quinolones. The development of such resistant strains reflected as a public health alarm. Moreover, the retrieved strains were sensitive to norfloxacin (85.7%), meropenem (77.1%), and imipenem (74.3%). Our findings are consistent with those reported by Wong46 and Nahar47. The improper application of antimicrobial agents in the poultry industry and the ability of P. mirabilis to acquire the antimicrobial-resistant genes from other resistant pathogens are the major causes of the emergence of these MDR-strains. Unfortunately, P. mirabilis could resist various antimicrobial classes due to the presence of chromosomal antibiotic-resistant genes as well as the resistant-plasmids37.
The biofilm assay revealed that 94.3% (33/35) of the isolated P. mirabilis strains are biofilm producers. Our findings are consistent with those reported by Kwiecinska-Piróg48. The biofilm is one of the most important virulence determinants of bacteria. It preserves bacteria during adverse environmental conditions. Moreover, biofilm protects bacteria from phagocytosis, antibodies, and antibiotics. Besides, it plays a vital role in antimicrobial resistance. P. mirabilis produces biofilm in various environments includes: biological and non-biotic surfaces such as glass, silicone, and polystyrene. The formation of biofilm on the non-biotic surface is considered the main source of nosocomial infections49,50.
The PCR proved that the recovered P. mirabilis strains are virulent and harbored atpD, ureC, rsbA, and zapA virulence genes with a prevalence of 100%, 100%, 94.3%, and 91.4%, respectively. Our findings are nearly agreed with those reported by Pathirana30 and Sun51. The atpD gene is encoded for ATP synthase β-subunit for the production of ATP from ADP. The atpD gene is more conservative in Proteus species when compared with 16SrRNA25. Infections caused by P. mirabilis are controlled by several virulence-determinants that are regulated by specific virulence genes. IgA‐degrading proteases are commonly accompanied by the pathogenic strains of P. mirabilis. ZapA-protease could degrade IgG, IgA1, and IgA2. It is regulated by the zapA gene. P. mirabilis is frequently incriminated in urinary tract infections that are mediated by stone-formation due to the release of urease enzyme. Urease is a metalloenzyme that acts by increasing the pH of urine that induces crystal formation. The urease production is controlled by the ureC gene. Besides, the characteristic swarming activity of P. mirabilis is encoded by the rsbA gene. The rsbA gene expresses a membrane sensor that induces the production of extracellular polysaccharides. Also, it regulates the swarming phenomena and enhances the biofilm formation by P. mirabilis23,50,52,53.
Concerning the correlation between the phenotypic and genotypic antimicrobial resistance patterns; our findings revealed that 31.4% (11/35) of the retrieved P. mirabilis strains are XDR to 8 antimicrobial classes, and harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. Moreover, 22.8% (8/35) of the tested strains are MDR to 3 antimicrobial classes and possessed blaTEM, tetA, and sul1genes. Besides, 17.1% (6/35) of the tested strains are MDR to 7 antimicrobial classes and harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. The Extended-spectrum β-lactamases (ESBLs) were reported for the first time in 198354. ESBLs are responsible for the hydrolysis of Broad-spectrum β-lactam antibiotics including penicillins, piperacillin, and cephalosporins. EBSLs are frequently produced by Enterobacteriales. Recently, P. mirabilis strains reported harboring various acquired antimicrobial resistance genes. The high prevalence of the blaTEM gene among the recovered P. mirabilis strains enabling them to resist penicillins (amoxicillin and ampicillin). Moreover, the resistance to cephalosporins (cefotaxime, and ceftazidime) is mediated by the presence of the blaCTX-M gene. The resistance to piperacillin is mainly attributed to the blaOXA-1 gene which also promoting the resistance to cephalosporins. Besides, both blaOXA-1 and blaCTX-M genes synergistically enable P. mirabilis to resist the β-Lactam-β-lactamase-inhibitor-combinations55,56. In addition, P. mirabilis is frequently resistant to tetracyclines and sulfonamides due to the presence of tetA and sul1genes, respectively. On the other hand, P. mirabilis is usually susceptible to fluoroquinolones such as norfloxacin57. The polymyxins exert their effect by increasing the permeability of the Gram-negative bacterial cell membrane through displacing Mg2 + and Ca2 + from the lipid A content of LPS that results in leakage of the cell contents. The resistance to polymyxins is common in the mutant P. mirabilis due to the alteration of LPS that is controlled by the expression of the eptC gene and the modification of the L- Ara4N. However, several previous studies reported the sensitivity of some P. mirabilis isolates to polymyxins, especially those of animal origin as reported by Sun51. The eptC gene may be present but not expressed. Besides, the alterations of the LPS in the cell envelop occurs only in the mutant strains and varies among different strains of P. mirabilis as previously reported by McCoy58.
In the present study, three strains are carbapenem-resistant as well as PDR to all the tested ten antimicrobial classes and are sharing blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 genes. Of them, two strains harbored the blaNDM-1 gene, and one strain carried the blaKPC gene. Globally, the emergence of carbapenem-resistance in P. mirabilis is relatively low; however, it inclines to increase over time. The carbapenem-resistance is attributed to the presence of blaNDM-1 and blaKPC genes. The existence of the blaKPC gene in P. mirabilis was recorded for the first time in a diabetic Patient in the USA in 200859, followed by China in 201060, and Brazil in 201561. Moreover, the blaNDM-1 is recognized for the first time in P. mirabilis strain retrieved from urinary infection in France in 201262 and followed by China in 201563.
Concerning the correlation between the antimicrobial resistance genes and the virulence determinants, a previous study that was reported by Filipiak64 revealed an inversed correlation between the virulence factors and the presence of the resistance genes in the retrieved P. mirabilis strains. However, in the present study, the majority of the screened virulence genes were found in the recovered isolates. Besides, there is no significant difference in the distribution of the virulence genes among the retrieved isolates either the susceptible or the antimicrobial-resistant strains. These findings suggest that the P. mirabilis pathogenicity is not affected by the presence of antimicrobial resistance genes.
Study limitations
Multilocus sequence typing (MLST) should be carried out to illustrate the genetic relatedness among the recovered P. mirabilis strains.
In conclusion, to the best of our knowledge, this is the first report regarding the emergence of XDR and MDR-P. mirabilis in ducks. P. mirabilis is more prevalent in diseased birds than the apparently healthy ones, and the liver is the most prominent infected organ. P. mirabilis is a common biofilm-producing pathogen. The recovered P. mirabilis isolates commonly harbor the atpD, ureC, zapA, and rsbA virulence genes. The retrieved P. mirabilis strains are extensively drug-resistant (XDR) or multidrug-resistant (MDR) to several antimicrobial classes (penicillins, β-Lactam-β-lactamase-inhibitor-combinations, cephalosporins, sulfonamides, tetracyclines, quinolones, macrolides, and polymyxins), and commonly harbored blaTEM, blaOXA-1, blaCTX-M, tetA, and sul1 antimicrobial resistance genes. In-vitro, norfloxacin exhibited promising antibacterial activity against the recovered XDR and MDR-P. mirabilis. Furthermore, the emergence of carbapenem-resistant (harbored either blaKPC or blaNDM-1 genes) and PDR-strains constitutes a threat alarm that indicates a complicated treatment of the diseases caused by such superbugs. Accordingly, it endorses the incessant surveillance of antimicrobial susceptibility testing as well as the limited and appropriate use of antibiotics in health and veterinary practices.
Supplementary Information
Author contributions
A.M.A and R.M.E Conceptualization; A.M.A, R.M.E, H.R.H, H.F.H, H.R, K.J.A, and N.S.S conducted the experiments. A.M.A and R.M.E drafted the manuscript. A.M.A, R.M.E, H.R.H., K.J.A, H.F.H., H.R and N.S.S did the statistical analysis, investigation, data validation and accuracy, and supervision. AMA wrote and revised the manuscript. All authors have revised and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-88861-w.
References
- 1.O’Hara CM, Brenner FW, Miller JM. Classification, identification, and clinical significance of Proteus, Providencia, and Morganella. Clin. Microbiol. Rev. 2000;13:534–546. doi: 10.1128/CMR.13.4.534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen CY, et al. Proteus mirabilis urinary tract infection and bacteremia: Risk factors, clinical presentation, and outcomes. J. Microbiol. Immunol. Infect. 2012;45:228–236. doi: 10.1016/j.jmii.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 3.Jansen AM, Lockatell CV, Johnson DE, Mobley HLT. Visualization of Proteus mirabilis morphotypes in the urinary tract: The elongated swarmer cell is rarely observed in ascending urinary tract infection. Infect. Immun. 2003;71:3607–3613. doi: 10.1128/IAI.71.6.3607-3613.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Shi X, et al. Comparative screening of digestion tract toxic genes in Proteus mirabilis. PLoS ONE. 2016;11:e0151873. doi: 10.1371/journal.pone.0151873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wang Y, et al. An outbreak of Proteus mirabilis food poisoning associated with eating stewed pork balls in brown sauce, Beijing. Food Control. 2010;21:302–305. doi: 10.1016/j.foodcont.2009.06.009. [DOI] [Google Scholar]
- 6.Lei CW, et al. Characterization of SXT/R391 integrative and conjugative elements in Proteus mirabilis isolates from food-producing animals in China. Antimicrob. Agents Chemother. 2016;60:1935–1938. doi: 10.1128/AAC.02852-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Reich F, Atanassova V, Klein G. Extended-spectrum ß-lactamase- and ampc-producing Enterobacteria in healthy broiler chickens, Germany. Emerg. Infect. Dis. 2013;19:1253–1259. doi: 10.3201/eid1908.120879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Olonitola OS, Fahrenfeld N, Pruden A. Antibiotic resistance profiles among mesophilic aerobic bacteria in Nigerian chicken litter and associated antibiotic resistance genes. Poult. Sci. 2015;94:867–874. doi: 10.3382/ps/pev069. [DOI] [PubMed] [Google Scholar]
- 9.Makharita RR, et al. Antibiogram and genetic characterization of carbapenem-resistant Gram-negative pathogens incriminated in healthcare-associated infections. Infect. Drug Resist. 2020;13:3991–4002. doi: 10.2147/IDR.S276975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Algammal AM, Wahdan A, Elhaig MM. Potential efficiency of conventional and advanced approaches used to detect Mycobacterium bovis in cattle. Microb. Pathog. 2019;134:103574. doi: 10.1016/j.micpath.2019.103574. [DOI] [PubMed] [Google Scholar]
- 11.El-Sayed ME, Algammal AM, Abouel-Atta ME, Mabrok M, Emam AM. Pathogenicity, genetic typing, and antibiotic sensitivity of Vibrio alginolyticus isolated from Oreochromis niloticus and Tilapia zillii. Rev. Med. Vet. J. 2019;170(4–6):80–86. [Google Scholar]
- 12.Algammal AM, et al. Molecular typing, antibiogram and PCR-RFLP based detection of Aeromonas hydrophila complex isolated from Oreochromis niloticus. Pathogens. 2020;9:238. doi: 10.3390/pathogens9030238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Algammal AM, et al. Prevalence, the antibiogram and the frequency of virulence genes of the most predominant bacterial pathogens incriminated in calf pneumonia. AMB Express. 2020;10:1–8. doi: 10.1186/s13568-020-01037-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Algammal AM, et al. Genes encoding the virulence and the antimicrobial resistance in enterotoxigenic and shiga-toxigenic E. coli isolated from diarrheic calves. Toxins. 2020;12:383. doi: 10.3390/toxins12060383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Abolghait SK, Fathi AG, Youssef FM, Algammal AM. Methicillin-resistant Staphylococcus aureus (MRSA) isolated from chicken meat and giblets often produces staphylococcal enterotoxin B (SEB) in non-refrigerated raw chicken livers. Int. J. Food Microbiol. 2020;328:108669. doi: 10.1016/j.ijfoodmicro.2020.108669. [DOI] [PubMed] [Google Scholar]
- 16.Enany ME, et al. The occurrence of the multidrug resistance (MDR) and the prevalence of virulence genes and QACs resistance genes in E. coli isolated from environmental and avian sources. AMB Express. 2019;9:1–9. doi: 10.1186/s13568-019-0920-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Algammal AM, Enany ME, El-Tarabili RM, Ghobashy MOI, Helmy YA. Prevalence, antimicrobial resistance profiles, virulence and enterotoxin-determinant genes of MRSA isolated from subclinical bovine mastitis samples in Egypt. Pathogens. 2020;9:1–11. doi: 10.3390/pathogens9050362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Algammal AM, et al. Emerging MDR-Pseudomonas aeruginosa in fish commonly harbor oprL and toxA virulence genes and bla TEM, bla CTX-M, and tetA antibiotic-resistance genes. Sci. Rep. 2020;10:1–12. doi: 10.1038/s41598-019-56847-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Uzunović S, Ibrahimagić A, Hodžić D, Bedenić B. Molekularna epidemiologija i antimikrobna osjetljivost kliničkih izolata Proteus spp. Koji produciraju AmpC-i/ili beta-laktamaza proširenog spektra djelovanja u Zeničko-Dobojskom kantonu, Bosna i Hercegovina. Med. Glas. 2016;13:103–112. [Google Scholar]
- 20.Li X, et al. SXT/R391 integrative and conjugative elements in Proteus species reveal abundant genetic diversity and multidrug resistance. Sci. Rep. 2016;6:1–9. doi: 10.1038/s41598-016-0001-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tonkić M, et al. High prevalence and molecular characterization of extended-spectrum β-lactamase-producing Proteus mirabilis strains in southern Croatia. J. Med. Microbiol. 2010;59:1185–1190. doi: 10.1099/jmm.0.016964-0. [DOI] [PubMed] [Google Scholar]
- 22.Naas T, Zerbib M, Girlich D, Nordmann P. Integration of a transposon Tn1-encoded inhibitor-resistant β-lactamase gene, blaTEM-67 from Proteus mirabilis, into the Escherichia coli chromosome. Antimicrob. Agents Chemother. 2003;47:19–26. doi: 10.1128/AAC.47.1.19-26.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li X, Mobley HLT. Vaccines for Proteus mirabilis in urinary tract infection. Int. J. Antimicrob. Agents. 2002;19:461–465. doi: 10.1016/S0924-8579(02)00102-4. [DOI] [PubMed] [Google Scholar]
- 24.Quinn PJ, Markey BK, Carter ME, Donnelly WJ, Leonard FC. Veterinary Microbiology and Microbial Disease. New York: Blackwell Science; 2002. Bacterial causes of bovine mastitis; pp. 465–475. [Google Scholar]
- 25.Bi SL, Tang SZ, Wu XY, Chen SY. Quantitative detection of Proteus species by real-time polymerase chain reaction using SYBR Green i. Ann. Microbiol. 2013;63:1205–1208. doi: 10.1007/s13213-012-0519-3. [DOI] [Google Scholar]
- 26.Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013;30:2725–2729. doi: 10.1093/molbev/mst197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.CLSI. Clinical and Laboratory Standards Institute : Performance standards for antimicrobial susceptibility testing : 27th ed 424 informational supplement. CLSI Doc. M100-S20 (2017).
- 28.Magiorakos AP, et al. Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: An international expert proposal for interim standard definitions for acquired resistance. Clin. Microbiol. Infect. 2012;18:268–281. doi: 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 29.Kadam SR, et al. Diversity assessment of Listeria monocytogenes biofilm formation: Impact of growth condition, serotype and strain origin. Int. J. Food Microbiol. 2013;165:259–264. doi: 10.1016/j.ijfoodmicro.2013.05.025. [DOI] [PubMed] [Google Scholar]
- 30.Pathirana HNKS, De Silva BCJ, Wimalasena SHMP, Hossain S, Heo GJ. Comparison of virulence genes in Proteus species isolated from human and pet turtle. Iran. J. Vet. Res. 2018;19:48–52. [PMC free article] [PubMed] [Google Scholar]
- 31.Randall LP, Cooles SW, Osborn MK, Piddock LJV, Woodward MJ. Antibiotic resistance genes, integrons and multiple antibiotic resistance in thirty-five serotypes of Salmonella enterica isolated from humans and animals in the UK. J. Antimicrob. Chemother. 2004;53:208–216. doi: 10.1093/jac/dkh070. [DOI] [PubMed] [Google Scholar]
- 32.Ibekwe AM, Murinda SE, Graves AK. Genetic diversity and antimicrobial resistance of Escherichia coli from human and animal sources uncovers multiple resistances from human sources. PLoS ONE. 2011;6:e20819. doi: 10.1371/journal.pone.0020819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xia Y, Liang Z, Su X, Xiong Y. Characterization of carbapenemase genes in Enterobacteriaceae species exhibiting decreased susceptibility to carbapenems in a university hospital in Chongqing, China. Ann. Lab. Med. 2012;32:270–275. doi: 10.3343/alm.2012.32.4.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Archambault M, et al. Molecular characterization and occurrence of extended-spectrum β-lactamase resistance genes among Salmonella enterica serovar corvallis from Thailand, Bulgaria, and Denmark. Microb. Drug Resist. 2006;12:192–198. doi: 10.1089/mdr.2006.12.192. [DOI] [PubMed] [Google Scholar]
- 35.Colom K, et al. Simple and reliable multiplex PCR assay for detection of blaTEM, blaSHV and blaOXA-1 genes in Enterobacteriaceae. FEMS Microbiol. Lett. 2003;223:147–151. doi: 10.1016/S0378-1097(03)00306-9. [DOI] [PubMed] [Google Scholar]
- 36.Barbour EK, et al. Comparison of phenotypic and virulence genes characteristics in human and chicken isolates of Proteus mirabilis. Pathog. Glob. Health. 2012;106:352–357. doi: 10.1179/2047773212Y.0000000042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yeh HY, Line JE, Hinton A. Molecular analysis, biochemical characterization, antimicrobial activity, and immunological analysis of Proteus mirabilis isolated from broilers. J. Food Sci. 2018;83:770–779. doi: 10.1111/1750-3841.14056. [DOI] [PubMed] [Google Scholar]
- 38.Ram P, Rao V, Rao S, Subramanyam KV, Srinivas K. Prevalence and virulence gene profiles of Proteus mirabilis isolated from animal, human and water samples in Krishna District, Andhra Pradesh, India. Pharma Innov. J. 2019;8:19–23. doi: 10.7897/2277-4572.081120. [DOI] [Google Scholar]
- 39.Liu MC, et al. 10′(Z), 13′(E)-heptadecadienylhydroquinone inhibits swarming and virulence factors and increases polymyxin B susceptibility in Proteus mirabilis. PLoS ONE. 2012;7(9):e45563. doi: 10.1371/journal.pone.0045563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Armbruster CE, Mobley HLT, Pearson MM. Pathogenesis of Proteus mirabilisInfection. EcoSal Plus. 2018 doi: 10.1128/ecosalplus.ESP-0009-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Di Pilato V, et al. Complete genome sequence of the first KPC-type carbapenemase-positive Proteus mirabilis strain from a bloodstream infection. Genome Announc. 2016 doi: 10.1128/genomeA.00607-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bitar I, et al. Complete genome and plasmids sequences of a clinical Proteus mirabilis isolate producing plasmid mediated ndm-1 from Italy. Microorganisms. 2020;8:339. doi: 10.3390/microorganisms8030339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bie L, Fang M, Li Z, Wang M, Xu H. Identification and characterization of new resistance-conferring SGI1s (Salmonella Genomic Island 1) in Proteus mirabilis. Front. Microbiol. 2018;9:3172. doi: 10.3389/fmicb.2018.03172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sullivan NL, Septer AN, Fields AT, Wenren LM, Gibbs KA. The complete genome sequence of Proteus mirabilis Strain BB2000 reveals differences from the P. mirabilis reference strain. Genome Announc. 2013;1:5. doi: 10.1128/genomeA.00024-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pearson MM, et al. Complete genome sequence of uropathogenic Proteus mirabilis, a master of both adherence and motility. J. Bacteriol. 2008;190:4027–4037. doi: 10.1128/JB.01981-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wong MHY, Wan HY, Chen S. Characterization of multidrug-resistant Proteus mirabilis isolated from chicken carcasses. Foodborne Pathog. Dis. 2013;10:177–181. doi: 10.1089/fpd.2012.1303. [DOI] [PubMed] [Google Scholar]
- 47.Nahar A. Multidrug resistant-Proteus Mirabilis isolated from chicken droppings in commercial poultry farms: bio-security concern and emerging public health threat in Bangladesh. J. Biosaf. Heal. Educ. 2014 doi: 10.4172/2332-0893.1000120. [DOI] [Google Scholar]
- 48.Kwiecinska-Piróg J, Bogiel T, Skowron K, Wieckowska E, Gospodarek E. Proteus mirabilis biofilm: Qualitative and quantitative colorimetric methods-based evaluation. Braz. J. Microbiol. 2014;45:1415–1421. doi: 10.1590/S1517-83822014000400037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Czaczyk K, Myszka K. Mechanizmy warunkujace opornosc biofilmow bakteryjnych na czynniki antymikrobiologiczne. Biotechnologia. 2007;76:40–52. [Google Scholar]
- 50.Jacobsen SM, Shirtliff ME. Proteus mirabilis biofilms and catheter-associated urinary tract infections. Virulence. 2011;2:460–465. doi: 10.4161/viru.2.5.17783. [DOI] [PubMed] [Google Scholar]
- 51.Sun Y, et al. Association among biofilm formation, virulence gene expression, and antibiotic resistance in Proteus mirabilis isolates from diarrhetic animals in Northeast China. BMC Vet. Res. 2020;16:176. doi: 10.1186/s12917-020-02372-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Walker KE, Moghaddame-Jafari S, Lockatell CV, Johnson D, Belas R. ZapA, the IgA-degrading metalloprotease of Proteus mirabilis, is a virulence factor expressed specifically in swarmer cells. Mol. Microbiol. 1999;32:825–836. doi: 10.1046/j.1365-2958.1999.01401.x. [DOI] [PubMed] [Google Scholar]
- 53.Hussein EI, et al. Assessment of pathogenic potential, virulent genes profile, and antibiotic susceptibility of Proteus mirabilis from urinary tract infection. Int. J. Microbiol. 2020;20:20. doi: 10.1155/2020/1231807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Knothe H, Shah P, Krcmery V, Antal M, Mitsuhashi S. Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime in clinical isolates of Klebsiella pneumoniae and Serratia marcescens. Infection. 1983;11:315–317. doi: 10.1007/BF01641355. [DOI] [PubMed] [Google Scholar]
- 55.Bidet P, Verdet C, Gautier V, Bingen E, Arlet G. First description of DHA-1 ampC β-lactamase in Proteus mirabilis. Clin. Microbiol. Infect. 2005;11:591–592. doi: 10.1111/j.1469-0691.2005.01178.x. [DOI] [PubMed] [Google Scholar]
- 56.Schultz E, et al. Survey of multidrug resistance integrative mobilizable elements SGI1 and PGI1 in Proteus mirabilis in humans and dogs in France, 2010–13. J. Antimicrob. Chemother. 2015;70:2543–2546. doi: 10.1093/jac/dkv154. [DOI] [PubMed] [Google Scholar]
- 57.Yong D, et al. The first detection of CTX-M-14 extended-spectrum β-lactamase among diverse β-lactamase-producing Proteus mirabilis clinical isolates. Diagn. Microbiol. Infect. Dis. 2006;54:237–239. doi: 10.1016/j.diagmicrobio.2005.09.013. [DOI] [PubMed] [Google Scholar]
- 58.McCoy AJ, Liu H, Falla TJ, Gunn JS. Identification of Proteus mirabilis mutants with increased sensitivity to antimicrobial peptides. Antimicrob. Agents Chemother. 2001;45:2030–2037. doi: 10.1128/AAC.45.7.2030-2037.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Tibbetts R, Frye JG, Marschall J, Warren D, Dunne W. Detection of KPC-2 in a clinical isolate of Proteus mirabilis and first reported description of carbapenemase resistance caused by a KPC β-lactamase in P. mirabilis. J. Clin. Microbiol. 2008;46:3080–3083. doi: 10.1128/JCM.00979-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hu YY, et al. Emergence of Proteus mirabilis harboring bla KPC-2 and qnrD in a Chinese hospital. Antimicrob. Agents Chemother. 2012;56:2278–2282. doi: 10.1128/AAC.05519-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cabral AB, Maciel MAV, Barros JF, Antunes MM, Lopes ACS. Detection of blaKPC-2 in Proteus mirabilis in Brazil. Rev. Soc. Bras. Med. Trop. 2015;48:94–95. doi: 10.1590/0037-8682-0152-2014. [DOI] [PubMed] [Google Scholar]
- 62.Girlich D, Dortet L, Poirel L, Nordmann P. Integration of the blandm-1 carbapenemase gene into Proteus genomic island 1 (PGI1-PmPEL) in a Proteus mirabilis clinical isolate. J. Antimicrob. Chemother. 2015;70:98–102. doi: 10.1093/jac/dku371. [DOI] [PubMed] [Google Scholar]
- 63.Qin S, et al. Emergence of extensively drug-resistant Proteus mirabilis harboring a conjugative NDM-1 plasmid and a novel Salmonella genomic island 1 variant, SGI1-Z. Antimicrob. Agents Chemother. 2015;59:6601–6604. doi: 10.1128/AAC.00292-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Filipiak A, et al. Pathogenic factors correlate with antimicrobial resistance among clinical Proteus mirabilis strains. Front. Microbiol. 2020;11:579389. doi: 10.3389/fmicb.2020.579389. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.