Abstract
This study aims to evaluate SARS-CoV-2 seroprevalence among health care workers (HCWs) and to assess self-reported risk factors for seropositivity. A total of 3255 HCWs were included and the overall seroprevalence was 7.8%. The likelihood of seropositivity was higher in participants reporting any COVID-19 symptoms within the last 4 months (OR 8.32, 95% CI 5.83-11.88, P < 0.001). Being a female HCW (OR 1.32, 95% CI 1.11–2.32, P < 0.01), having a cohabitant who was infected with SARS-CoV-2 (OR 2.55, 95% CI 1.78–3.66 P < 0.001) or a cohabitant who was a nursing home caregiver (OR 3.71, 95% CI 1.59–8.65, P = 0.002) were independently associated with an increased risk of seropositivity. Working in a COVID-19 unit (OR 1.64, 95% CI 1.21–2.23, P < 0.001) and being exposed to a SARS-CoV-2 infected co-worker (OR 1.30,95% CI 0.97–1.74, P = 0.016) resulted in higher seropositivity rate. Even if in-hospital exposure may play a significant role, increased infection risk is most likely attributable to household contact.
Key words: SARS-CoV-2, COVID-19, HCW, seroprevalence, risk factors, household contact
1. Introduction
The novel “coronavirus disease 2019” (COVID-19) has caused more than 84 million cases and 1,85 million deaths worldwide, as of December 31, 2020 (European Centre for Disease Prevention and Control). Since the onset of the pandemic in Wuhan, China, risk factors for severe and fatal forms of COVID-19 have been identified and include older age, male sex, archaic genetic variants (in Eurasians), dysregulated interferon response, and chronic underlying conditions such as hypertension, obesity, cardiovascular disease, diabetes, chronic lung, liver and kidney diseases (Bastard et al., 2020; Bennett et al., 2020; Zeberg and Paabo, 2020; Zhang et al., 2020; Zheng et al., 2020). Due to their frontline position during the COVID-19 pandemic, health care workers (HCWs) might be exposed to the virus at a greater extent than the general population. In a large Scottish register-based cohort study, Shah et al show that the risk of COVID-19 related to hospital admission during the first 3 months of the pandemic for patient-facing HCWs were three times higher than for non-patient-facing HCWs and the general population. Risk was also doubled among household members of frontline workers, in analyses adjusted for sex, age, ethnicity, socioeconomic status, and comorbidity (Shah et al., 2020). More recently in a large UK Biobank of 120,075 participants including 271 severe COVID-19, HCWs had a 7 times higher risk of severe COVID-19 in comparison to non-essential workers (Mutambudzi et al., 2020). Although this raises concerns about the safety of HCWs, the results of HCWs serological investigations vary widely, making difficult to assess the role of occupational exposure in SARS-CoV-2 infection. This study aims to evaluate the seroprevalence of antibodies against SARS-CoV-2 among hospital staff (HCW) and to assess self-reported occupational and household risk factors for seropositivity.
2. Methods
2.1. Context and study population
This study took place in the Cliniques universitaires Saint-Luc, a large teaching hospital of 1000 beds in Brussels, Belgium. During the first wave of the pandemic (March to June 2020), the hospital modified his organization to take care of COVID-19 patients and created a dedicated emergency unit, 3 intensive care units and 8 dedicated units of 20 beds. Personal protective equipment (PPE) was given to health care of those units in the early March and universal masking was mandatory inside the hospital for all HCWs, patients and visitors in April 1th. The hospital continued to manage all other emergency diseases and surgeries and only non-essential activities were cancelled.
All HCWs including administrative staff were invited to participate to this prospective study in a voluntary basis. Before serological analysis, participants were asked to answer a web-based survey in order to provide information about demographics, family environment, previous influenza vaccination, workplace assignment during the COVID-19 pandemic, possible exposure to a SARS-CoV-2 infected person, presence of any symptoms compatible with COVID-19 during the last 4 months, and the result of SARS-CoV-2 molecular test if carried out. Then, SARS-CoV-2 antibodies detection was conducted on a serum sample drawn for each participant between June 19 and June 30, 2020.
2.2. SARS-CoV-2 antibodies detection
The qualitative detection of SARS-CoV-2 antibodies in human serum was performed on the Cobas e602 analyzer using Elecsys anti-SARS-CoV-2 kit (Roche Diagnostics). This electrochemiluminescent immunoassay (ECLIA) using recombinant nucleocapsid antigens allows the detection of total anti-SARS-CoV-2 antibodies with a sensitivity of 100% and specificity of 95%. According to the manufacturer's instructions, a result was considered positive if the index was ≥ 1.00.
2.3. Statistical analysis
Analyses were performed using MedCalc Statistical Software version 14.8.1 (MedCalc Software bvba, Ostend, Belgium). Seropositivity rates are reported as numbers and proportions within each level of demographic, work and household characteristics. Associations between factors and seropositivity were assessed using univariable logistic regression and results are reported as odds ratio with 95% confidence interval. To identify risk factors that were independently associated with SARS-CoV-2 antibodies development, all factors with a univariate p-value <0.10 were entered into a multivariable logistic regression model. The threshold for statistical significance was set to a p value <0.05.
2.4. Ethical agreement
Ethical approval for this study (Ethical Committee N°CEHF 2020/29mai/301) was provided by the IRB (CEBH of the Université catholique de Louvain (UCLouvain), Brussels, Belgium)
3. Results
A total of 3255 HCWs were included in the study. Demographics and clinical characteristics are summarized in Table 1 . The mean age of the participants was 41.9 (SD: 12.4) years and 78% were female. Nurses accounted for 35.5% (1154/3255) of the study population, 25.6% (834/3255) of the participants were paramedics and 14.6% (474/3255) were physicians. Administrative staff accounted for 18% (585/3255) of the study population and 6.4% (208/3255) participants with other healthcare occupational assignment were also included in the study.
Table 1.
Univariate and multivariable analysis of factors associated with SARS-CoV-2 seropositivity.
| Total | Seropositive (n = 253) |
Seronegative (n = 3002) |
Univariate analysis |
Multivariate analysis |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| No. | % | No. | % | OR [95%CI] | P-value | OR [95%CI] | P-value | ||||
| Sex | 0.01 | 0.01 | |||||||||
| Female | 2513 | 77.9% | 211 | 8.4% | 2302 | 91.6% | 1.58 [1.11; 2.25] | 1.61 [1.11; 2.32] | |||
| Male | 712 | 22.1% | 39 | 5.5% | 673 | 94.5% | 1 | 1 | |||
| Missing | 30 | 3 | 10.0% | ||||||||
| Age (years) | 0.017 | 0.15 | |||||||||
| Mean ± SD | 41.9 ± 12.4 | 40.1 ± 10.6 | 42.0 ± 12.5 | ||||||||
| Median (min - max) | 41 (0 - 120) | 39 (22 - 64) | 41 (0 - 120) | ||||||||
| < 30 | 662 | 20.4% | 50 | 7.6% | 612 | 92.4% | 1.26 [0.86; 1.85] | 1.25 [0.92; 1.70] | |||
| 30 - 49 | 1539 | 47.4% | 139 | 9.0% | 1400 | 91.0% | 1.53 [1.12; 2.08] | ||||
| 50 - 65 | 1049 | 32.3% | 64 | 6.1% | 985 | 93.9% | 1 | 1 | |||
| Patient-facing occupational work | 0.08 | 0.40 | |||||||||
| No | 750 | 23.0% | 47 | 6.3% | 703 | 93.7% | 1 | 1 | |||
| Yes | 2495 | 76.7% | 205 | 8.2% | 2290 | 91.8% | 1.34 [0.96; 1.86] | 0.83 [0.55; 1.27] | |||
| Work in a COVID-19 unit | < 0.001 | < 0.001 | |||||||||
| No | 2125 | 65.3% | 126 | 5.9% | 1999 | 94.1% | 1 | 1.64 [1.21; 2.23] | |||
| Yes | 1130 | 34.7% | 127 | 11.2% | 1003 | 88.8% | 2.01 [1.55; 2.60] | 1 | |||
| Work department | 0.015 | 0.30 | |||||||||
| Nursing | 1490 | 45.8% | 145 | 9.7% | 1345 | 90.3% | 1.57 [1.09; 2.25] | 1.04 [0.66; 1.63] | |||
| Medical | 1067 | 32.8% | 60 | 5.6% | 1007 | 94.4% | 0.87 [0.57; 1.31] | 0.79 [0.51; 1.24] | |||
| Pharmacy | 77 | 2.4% | 8 | 10.4% | 69 | 89.6% | 1.68 [0.76; 3.74] | 1.49 [0.65; 3.40] | |||
| Administration and others | 621 | 19.1% | 40 | 6.4% | 581 | 1.00 | 1 | ||||
| Exposure to a COVID-19 patient | < 0.001 | 0.08 | |||||||||
| No | 2167 | 66.6% | 138 | 6.4% | 2029 | 93.6% | 1 | 1 | |||
| Yes | 1088 | 33.4% | 115 | 10.6% | 973 | 89.4% | 1.74 [1.34; 2.25] | 1.30 [0.97; 1.74] | |||
| Exposure to a COVID-19 colleague | < 0.001 | 0.016 | |||||||||
| No | 1964 | 60.3% | 118 | 6.0% | 1846 | 94.0% | 1 | 1 | |||
| Yes | 1291 | 39.7% | 135 | 10.5% | 1156 | 89.5% | 1.83 [1.41; 2.36] | 1.41 [1.06; 1.87] | |||
| Days of eWork | 0.014 | 0.28 | |||||||||
| 0 | 2729 | 83.8% | 226 | 8.3% | 2503 | 91.7% | 1.67 [1.11; 2.52] | 1.28 [0.81; 2.02] | |||
| ≥1 | 526 | 16.2% | 27 | 5.1% | 499 | 94.9% | 1 | 1 | |||
| Mean ± SD | 2.4 ± 1.3 | 2.3 ± 1.1 | 2.4 ± 1.3 | ||||||||
| Median (min - max) | 2 (1 - 15) | 2 (1 - 5) | 2 (1 - 15) | ||||||||
| Cohabitant working in health care | 0.013 | 0.002 | |||||||||
| No | 2529 | 81.1% | 197 | 7.8% | 2332 | 92.2% | 1 | 1 | |||
| Yes, in hospital | 561 | 18.0% | 37 | 6.6% | 524 | 93.4% | 0.84 [0.58; 1.20] | ||||
| Yes, in care and rest home | 30 | 1.0% | 8 | 26.7% | 22 | 73.3% | 4.30 [1.89; 9.79] | 3.71 [1.59; 8.65] | |||
| Cohabitant infected with SARS-CoV-2 | < 0.001 | < 0.001 | |||||||||
| No | 2727 | 83.8% | 130 | 4.8% | 2597 | 95.2% | 1 | 1 | |||
| Yes | 271 | 8.3% | 46 | 17.0% | 225 | 83.0% | 4.08 [2.84; 5.87] | 2.55 [1.78; 3.66] | |||
| Having chidren | 0.17 | ||||||||||
| No | 1274 | 39.1% | 109 | 8.6% | 1165 | 91.4% | 1 | ||||
| Yes | 1976 | 60.7% | 143 | 7.2% | 1833 | 92.8% | 0.83 [0.64; 1.08] | ||||
| >12 years | 0.07 | ||||||||||
| 0 | 960 | 48.6% | 59 | 6.1% | 901 | 93.9% | 1 | ||||
| ≥1 | 1016 | 51.4% | 84 | 8.3% | 932 | 91.7% | 1.38 [0.97; 1.94] | ||||
| Mean ± SD | 1.8 ± 0.9 | 1.9 ± 0.8 | 1.8 ± 0.9 | ||||||||
| Median (min - max) | 2 (1 - 6) | 2 (1 - 4) | 2 (1 - 6) | ||||||||
| < 6 years | 0.96 | ||||||||||
| 0 | 1372 | 69.4% | 99 | 7.2% | 1273 | 92.8% | 1 | ||||
| ≥1 | 604 | 30.6% | 44 | 7.3% | 560 | 92.7% | 1.01 [0.70; 1.46] | ||||
| Mean ± SD | 1.6 ± 0.7 | 1.7 ± 0.7 | 1.6 ± 0.7 | ||||||||
| Median (min - max) | 1 (1 - 4) | 2 (1 - 3) | 1 (1 - 4) | ||||||||
| Childcare facilities | 0.59 | ||||||||||
| Yes | 298 | 49.3% | 24 | 8.1% | 282 | 94.6% | 1.18 [0.64; 2.19] | ||||
| No | 306 | 50.7% | 20 | 6.5% | 278 | 90.8% | 1 | ||||
| Influenza vaccine | 0.70 | ||||||||||
| No | 2229 | 68.5% | 170 | 7.6% | 2059 | 92.4% | 1 | ||||
| Yes | 1023 | 31.4% | 82 | 8.0% | 941 | 92.0% | 1.06 [0.80; 1.39] | ||||
| COVID-19 symptoms | < 0.001 | ||||||||||
| No | 1801 | 55.3% | 37 | 2.1% | 1764 | 97.9% | 1 | ||||
| Yes | 1447 | 44.5% | 215 | 14.9% | 1232 | 85.1% | 8.32 [5.83; 11.88] | ||||
| SARS-CoV-2 molecular test | < 0.001 | ||||||||||
| No | 2613 | 80.3% | 108 | 4.1% | 2505 | 95.9% | 1 | ||||
| Yes | 642 | 19.7% | 145 | 22.6% | 497 | 77.4% | 6.77 [5.18; 8.83] | ||||
| Positive | 515 | 80.2% | 33 | 6.4% | 482 | 93.6% | |||||
| Negative | 127 | 19.8% | 112 | 88.2% | 15 | 11.8% | |||||
SARS-CoV-2 antibodies were detected in 253 HCWs, giving an overall seroprevalence of 7.8%. Seroprevalence according to job assignment is shown in Fig. 1 . With a seropositivity rate of 9.4% (108/1154), nurses presented the highest seroconversion rate.
Fig. 1.
Seroprevalence according to occupational job. The dotted line represents the overall seroprevalence (7.8 %).
3.1. Univariate analysis
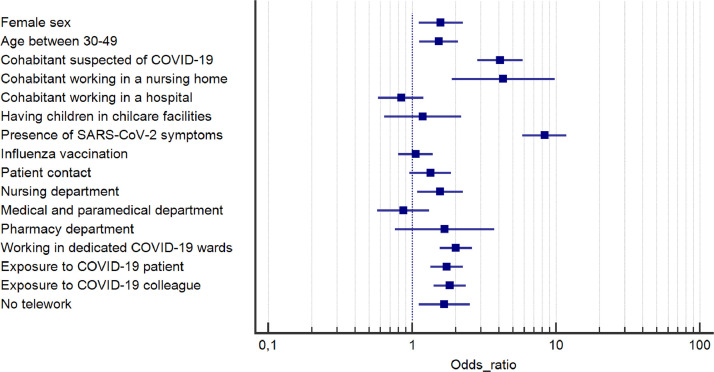
According to univariate analysis (Table 1 and Fig. 2 ), demographic factors such as female sex (OR 1.58, 95% CI 1.11–2.25, P = 0.010) and age between 30 and 49 years old (OR 1.53, 95% CI 1.12–2.08, P < 0.017) were associated with an increased risk of developing SARS-CoV-2 antibodies. Regarding the occupational risk, working in a COVID-19 dedicated unit (OR 2.01, 95% CI 1.55–2.60, P < 0.001) was associated with a higher seroconversion rate. 11,5% of nurses working in a COVID-19 dedicated unit had detectable anti-SARS-CoV-2 antibodies whereas only 6.7% of those who did not work in a COVID-19 dedicated unit had seroconverted. In the same way, having been exposed to a SARS-CoV-2 infected patient (OR 1.74, 95% CI 1.34–2.25, P < 0.001) or infected co-worker (OR 1.83, 95% CI 1.41-2.36, P < 0.001) demonstrated higher risk of seroconversion. Univariate analysis also demonstrated that HCWs who didn't have the opportunity to telework were at higher risk to develop SARS-CoV-2 antibodies (OR 1.67, 95% CI 1.11–2.52, P = 0.014). Working in the nursing department (OR 1.57, 95% CI 1.09–2.25, P = 0.015) or in the pharmacy (OR 1.68, 95% CI 0.76-3.74, P = 0.20) was also associated with higher seroconversion rate. Daily contact with patients and prior influenza vaccination did not show any statistically significant association with the presence of SARS-CoV-2 antibodies. With regard to the household contact, the odds of being seropositive were higher among HCWs who reported having a cohabitant working in a nursing home (OR 4.30, 95% CI 1.89-9.79, P = 0.013) and among HCWs who had a SARS-CoV-2 infected cohabitant (OR 4.08, 95% CI 2.84–5.87, P < 0.001). Among HCWs who had children, leaving children in childcare facilities during the pandemic was not significantly associated with a higher seroconversion (OR 1.18, 95% CI 0.64-2.19, P = 0.59).
Fig. 2.
Odds ratio with 95% confidence interval for univariate association between each characteristic and SARS-CoV-2 seropositivity.
The likelihood of being seropositive was higher in participants who reported any COVID-19 symptoms within the last 4 months (OR 8.32, 95% CI 5.83–11.88, P < 0.001) with anosmia and agueusia reported in 62.1% of seropositive people. By the end of June, real-time reverse transcription polymerase chain reaction (RT-qPCR) had been performed for 642 HCW and 127 of them were positive (127/642, 19.8%). The median time between positive RT-qPCR and the SARS-CoV-2 serology was 93 days (min:52-max:106). In the 127 HCWs with positive RT-qPCR for SARS-CoV-2, 112 (88,2%) had positive antibodies, only 15 HCWs with a previous positive RT-qPCR were seronegative (15/127, 11.8%). In these seronegative HCWs with proven SARS-CoV-2 infection by RT-qPCR, the median time between positive RT-qPCR and the serology was 89 days (min:52-max:110) and all but one presented with COVID-19 compatible symptoms. Among the 515 HCWs with a negative SARS-CoV-2 RT-qPCR result, 33 had detectable antibodies (6.4%) of whom 27 had experienced COVID-19 symptoms.
3.2. Multivariate analysis
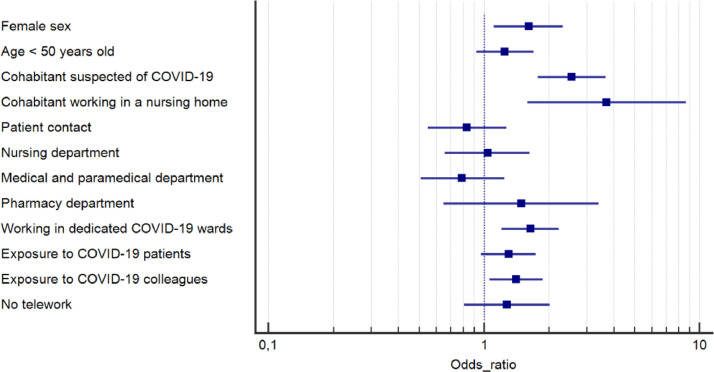
In the multivariate analysis (Fig. 3 ), being a female HCW (OR 1.32, 95% CI 1.11–2.32, P = 0.010), having a cohabitant who was infected with SARS-CoV-2 (OR 2.55, 95% CI 1.78–3.66, P < 0.001) or a cohabitant who was a nursing home caregiver (OR 3.71, 95% CI 1.59-8.65, P = 0.002) were independently associated with an increased seroconversion rate. In terms of occupational risk, working in a COVID-19 dedicated unit (OR 1.64, 95% CI 1.21–2.23, P < 0.001) and being exposed to a co-worker infected with SARS-CoV-2 (OR 1.30, 95% CI 0.97–1.74, P = 0.016) resulted in a higher rate of seropositivity.
Fig. 3.
Odds ratio with 95% confidence interval for independent contribution of each characteristic to the likelihood of SARS-CoV-2 seropositivity using a multivariable logistic regression model with 10 covariates.
4. Discussion
Providing relevant information about the proportion of people who got infected by SARS-CoV-2, seroprevalence studies conducted in the community but also in high-risk subgroups are useful to assess the level of exposure and identify risk factors of infection. Even if little is known about the neutralizing capacity of antibodies, the titer at which antibodies should confer protection and the duration of the protective immunity, there are growing evidence on the humoral immunological responses against SARS-CoV-2 and HCWs who developed SARS-CoV-2 antibodies were found to have lower risk of reinfection in the ensuing 6 months than those who were seronegative, suggesting a certain degree of protection of these antibodies (Lumley et al., 2020).
The most important findings of this study were that HCWs SARS-CoV-2 seroprevalence was 7.8% in our tertiary hospital in Brussels, a high burden region of SARS-CoV-2 infection in Belgium, and that the seropositivity rate was higher in those working in a COVID-19 dedicated unit or being exposed to a SARS-CoV-2 infected co-worker. Being a woman, having a cohabitant who was infected with SARS-CoV-2 or a cohabitant who was a nursing home caregiver also increased the risk of seroconversion according to the multivariate analysis.
Seroprevalence in the general population and in HCWs vary across regions, countries and periods of pandemic. Some variations also appeared to be related to the serological assay used (Galanis et al., 2021; Rostami et al., 2021). A recent meta-analysis demonstrated that the estimated overall seroprevalence of SARS-CoV-2 antibodies among HCWs was 8.7% (95% CI: 6.7-10.9%). Seroprevalence was higher in studies that were conducted in North America (12.7%) compared to those in Europe (8.5%), Africa (8.2%), and Asia (4%) (Galanis et al., 2021). Herzog et al performed a prospective serial cross-sectional nationwide seroprevalence study, stratified by age, sex and region, in whom 3000 to 4000 residual samples were collected during and after national lockdown between 30 March and 5 July 2020 in Belgium. They found that overall seroprevalence initially increased from 2.9% (95% CI 2.3–3.6) to 6.0% (95% CI 5.1–7.1), implying a seroincidence of 3.1% (95% CI 1.9– 4.3) between the 1st and 2nd period. Thereafter, seroprevalence stabilized and decreased from the 3rd to 5th period from 6.9% (95% CI 5.9-8.0) to 4.5% (95% CI 3.7–5.4) (Herzog et al., 2020). At the same period, SARS-CoV-2 serological screening of blood donors reported a seroprevalence of 4.3% (Sciensano, 2020). However, in this study there were significant discrepancies between regions, and the seroprevalence of blood donors in Brussels was significantly higher and reached approximately 8%. This would suggest than the SARS-CoV-2 seroprevalence among HCWs included in the present study is almost the same as the seroprevalence of the general population living in Brussels. Our results are in line with two Belgian studies reporting a seroprevalence of 6,4% and 7,7% (Steensels et al., 2020) (Mortgat et al., 2020). In contrast to our results, HCWs seroprevalence found in other Belgian studies was higher, ranging from 9.5% to 14.6% (Blairon et al., 2021; Duysburgh et al., 2021; Martin et al., 2020). Divergencies in prevention and infection control precautions may explain these discrepancies as well as virus circulation rates in the region surrounding the hospitals. Differences in seroprevalence might also come from differences in the formats and/or antigens used in the immunoassays currently available and clearly lacking of standardization.
Several studies tried to describe risk factors for SARS-CoV-2 seropositivity in HCWs with divergent results (Galanis et al., 2021; Mutambudzi et al., 2020; Rostami et al., 2021; Shah et al., 2020; Steensels et al., 2020). A recent meta-analysis found that the following factors were associated with seropositivity: male gender, Black, Asian, and Hispanic HCWs, work in a COVID-19 dedicated unit, patient-related work, frontline HCWs, health care assistants, personal protective equipment shortage, self-reported belief for previous SARS-CoV-2 infection, previous positive polymerase chain reaction test, and household contact with suspected or confirmed COVID-19 patients (Galanis et al., 2021). On the contrary, Steensels et al found that neither being directly involved in clinical care nor working in a COVID-19 unit increased the odds of being seropositive, while having a suspected COVID-19 household contact did (Steensels et al., 2020).
Our multivariate analysis suggested that in-hospital exposure may increase the risk of being seropositive but to a lesser extent than household exposure. Working in dedicated COVID-19 unit was significantly associated with seropositivity, as well as being in contact with a SARS-CoV-2 infected co-worker. However, being exposed to a COVID-19 patient was not associated with a significant higher odds of being seropositive, nor working in contact with patients. Seroprevalence was significantly higher in HCWs who declared having a COVID-19 co-worker contact which may be explained by unrespect of physical distancing, or by inappropriate facemask wearing during break times. These findings are supported by a previous study identifying working in a high-risk department, suboptimal handwashing before or after patient contact and improper use of personal protective equipment (PPE) as risk factors for SARS-CoV-2 transmission between coworkers (Ran et al., 2020). However, another large study in a tertiary hospital in Belgium, found that being involved in clinical care, having worked during the lockdown phase, being involved in care for patients with COVID-19, and exposure to COVID-19 positive co-workers were not statistically significantly associated with SARS-CoV-2 seropositivity. In contrast, this study also demonstrated that having a household contact with suspected or confirmed COVID-19 was associated with antibody positivity. It is important to take in mind that staff presenting symptoms were quarantined and therefore not tested (Steensels et al., 2020).
Our multivariate analysis revealed that household's exposure highly increased the odds of being seropositive. A recent meta-analysis by Madewell et al, showed that the estimated household secondary attack rate was 16.6% (95% CI: 14.0%–19.3%). Household secondary attack rates were increased from symptomatic index cases (18.0%; 95% CI: 14.2%–22.1%) than from asymptomatic index cases (0.7%; 95% CI: 0%–4.9%), to adult contacts (28.3%; 95% CI: 20.2%-37.1%) than to child contacts (16.8%;95%CI, 12.3%–21.7%), to spouses (37.8%; 95% CI: 25.8%–50.5%) than to other family contacts (17.8%; 95% CI: 11.7%-24.8%), and in households with 1 contact (41.5%; 95% CI: 31.7%-51.7%) than in households with 3 or more contacts (22.8%; 95% CI: 13.6%-33.5%) (Madewell et al., 2020).
Moreover, in our study, HCWs who declared having a household contact working in a nursing home were at higher risk of developing SARS-CoV-2 antibodies. This can be easily explained by the high burden of COVID-19 cases in Belgian nursing homes due partly to PPE shortage.
Interestingly, having children who attend childcare facilities did not show any statistically significant association with the presence of SARS-CoV-2 antibodies. The transmission dynamics in children have to be more clearly established but a recent modelling study demonstrated that susceptibility to SARS-CoV-2 infection in people under the age of 20 years is almost half that of adults (Davies et al., 2020). Another study found that with minimal secondary virus transmission demonstrated within families, schools and community settings, children do not appear to be major drivers of SARS-CoV-2 transmission (Williams et al., 2020).
Anosmia and agueusia were the most common symptoms reported by seropositive HCWs which is in line with previous reported data showing a strong association between anosmia, agueusia and COVID-19 (Menni et al., 2020). Makornidis et al reported a total of 590 participants enrolled via a web-based platform and responded to questions about loss of smell and taste and other COVID-19 related symptoms. A total of 77.6% of 567 participants with acute smell and/or taste loss had SARS-CoV-2 antibodies; of these, 39.8% (n = 175) had neither cough nor fever. New loss of smell was more prevalent in participants with SARS-CoV-2 antibodies, compared with those without antibodies (93.4% vs 78.7%, P < 0.001), whereas taste loss was equally prevalent (90.2% versus 89.0%, P = 0.74). Seropositivity for SARS-CoV-2 was 3 times more likely in participants with smell loss (OR 2.86; 95% CI 1.27–6.36; P < 0.001) compared with those with taste loss (Makaronidis et al., 2020).
Lastly, our study did not show any correlation between seropositivity for SARS-Cov-2 and prior influenza vaccine. Martinez-Baz et al also showed in their study that that influenza vaccination does not significantly modify the risk of SARS-CoV-2 infection (Martinez-Baz et al., 2020).
The major strength of this study is the simultaneous collect of information about in-hospital exposure and household exposure of HCWs to SARS-CoV-2, allowing to evaluate both concurrently. This study included a large sample size of individuals representing all departments in the hospital. Furthermore, the participants were not selected on the basis of symptoms and all HCWs were invited to participate on a voluntary basis. Participants were asked to fill in the survey before doing the serological test, therefore, the result of the test did not introduce any bias in the answers. The use of a highly sensitive and specific serological test, as recently demonstrated, also strengthened the results of this study (National S-C-SAEG 2020).
The main limitation of this study is that HCW self-presented, therefore a selection bias cannot be excluded. Another limitation is the self-reported data. The majority of the reported symptoms being common and unspecific symptoms, it is not clear that they were due to SARS-CoV-2 infection rather than another respiratory virus. Moreover, we did not collect any information about the severity of the symptoms and the delay between the symptoms and the serological assessment. Finally, we cannot exclude a possible cluster had occurred among study participants regardless of occupational exposure.
In conclusion, the seroprevalence of SARS-CoV-2 antibodies in HCW was lower than expected and similar to the general population (Brussels area). Even if in-hospital exposure may play a significant role and differential occupational exposure to SARS-CoV-2 exists, our results highlighted increased infection risk most likely attributable to household contact.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Authors contributions
Anaïs Scohy: methodology, conceptualization, investigation, writing original draft
Damien Gruson: investigation, writing – review and editing
Anne Simon: investigation, writing – review and editing
Benoît Kabamba-Mukadi: investigation, writing – review and editing
Julien De Greef: methodology, conceptualization, writing – review and editing
Leïla Belkhir: methodology, conceptualization, writing – review and editing
Hector Rodriguez-Villalobos: investigation, writing – review and editing
Annie Robert: formal analysis, writing – review and editing
Jean Cyr Yombi: methodology, conceptualization, investigation, writing original draft
Declaration of competing interests
None.
References
- Bastard P, Rosen LB, Zhang Q, Michailidis E, Hoffmann HH, Zhang Y, et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020;370:eabd45. doi: 10.1126/science.abd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett S, Tafuro J, Mayer J, Darlington D, Wong CW, Muntean EA, et al. Clinical features and outcomes of adults with coronavirus disease 2019: a systematic review and pooled analysis of the literature. Int J Clin Pract. 2020:e13725. doi: 10.1111/ijcp.13725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blairon L, Mokrane S, Wilmet A, Dessilly G, Kabamba-Mukadi B, Beukinga I, et al. Large-scale, molecular and serological SARS-CoV-2 screening of healthcare workers in a 4-site public hospital in Belgium after COVID-19 outbreak. J Infect. 2021;82:159–198. doi: 10.1016/j.jinf.2020.07.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies NG, Klepac P, Liu Y, Prem K, Jit M, group CC-w. Eggo RM. Age-dependent effects in the transmission and control of COVID-19 epidemics. Nat Med. 2020;26:1205–1211. doi: 10.1038/s41591-020-0962-9. [DOI] [PubMed] [Google Scholar]
- Duysburgh E, Mortgat L, Barbezange C, Dierick K, Fischer N, Heyndrickx L, et al. Persistence of IgG response to SARS-CoV-2. Lancet Infect Dis. 2021;21:163–164. doi: 10.1016/S1473-3099(20)30943-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- European Centre for Disease Prevention and Control. [7th January 2021]. Available https://www.ecdc.europa.eu/
- Galanis P, Vraka I, Fragkou D, Bilali A, Kaitelidou D. Seroprevalence of SARS-CoV-2 antibodies and associated factors in health care workers: a systematic review and meta-analysis. J Hosp Infect. 2021;108:120–134. doi: 10.1016/j.jhin.2020.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herzog SDB; Abrams, S; Wouters, I; Ekinci, E; Patteet, L; Coppens, A; et al: Seroprevalence of IgG antibodies against SARS coronavirus 2 in Belgium – a serial prospective cross-sectional nationwide study of residual samples. 2020. [DOI] [PMC free article] [PubMed]
- Lumley SF, Eyre DW, McNaughton AL, Howarth A, Hoosdally S, Hatch SB, et al. SARS-CoV-2 antibody prevalence, titres and neutralising activity in an antenatal cohort, United Kingdom, 14 April to 15 June 2020. Euro Surveill. 2020;25:2001721. doi: 10.2807/1560-7917.ES.2020.25.41.2001721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madewell ZJ, Yang Y, Longini IM, Jr., Halloran ME, Dean NE. Household transmission of SARS-CoV-2: a systematic review and meta-analysis. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.31756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Makaronidis J, Mok J, Balogun N, Magee CG, Omar RZ, Carnemolla A, et al. Seroprevalence of SARS-CoV-2 antibodies in people with an acute loss in their sense of smell and/or taste in a community-based population in London, UK: an observational cohort study. PLoS Med. 2020;17 doi: 10.1371/journal.pmed.1003358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin C, Montesinos I, Dauby N, Gilles C, Dahma H, Van Den Wijngaert S, et al. Dynamics of SARS-CoV-2 RT-PCR positivity and seroprevalence among high-risk healthcare workers and hospital staff. J Hosp Infect. 2020;106:102–106. doi: 10.1016/j.jhin.2020.06.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Baz I, Trobajo-Sanmartin C, Arregui I, Navascues A, Adelantado M, Indurain J, et al. Influenza vaccination and risk of SARS-CoV-2 infection in a Cohort of health workers. Vaccines (Basel) 2020;8 doi: 10.3390/vaccines8040611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menni C, Valdes AM, Freidin MB, Sudre CH, Nguyen LH, Drew DA, et al. Real-time tracking of self-reported symptoms to predict potential COVID-19. Nat Med. 2020;26:1037–1040. doi: 10.1038/s41591-020-0916-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mortgat LB; Fischer, N; Heyndrickx, L; Hutse, V; Thomas, I; Vuylsteke, B; et al: SARS-CoV-2 prevalence and seroprevalence among healthcare workers in Belgian hospitals: baseline results of a Prospective Cohort Study. 2020. 10.1101/2020.06.08.20125179 [DOI] [PMC free article] [PubMed]
- Mutambudzi M, Niedwiedz C, Macdonald EB, Leyland A, Mair F, Anderson J. Occupation and risk of severe COVID-19: prospective cohort study of 120 075 UK Biobank participants. Occup Environ Med. 2020 doi: 10.1136/oemed-2020-106731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National S-C-SAEG Performance characteristics of five immunoassays for SARS-CoV-2: a head-to-head benchmark comparison. Lancet Infect Dis. 2020;20:1390–1400. doi: 10.1016/S1473-3099(20)30634-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ran L, Chen X, Wang Y, Wu W, Zhang L, Tan X. Risk factors of healthcare workers with Coronavirus Disease 2019: a Retrospective Cohort Study in a designated hospital of Wuhan in China. Clin Infect Dis. 2020;71:2218–2221. doi: 10.1093/cid/ciaa287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rostami A, Sepidarkish M, Leeflang MMG, Riahi SM, Nourollahpour Shiadeh M, Esfandyari S, et al. SARS-CoV-2 seroprevalence worldwide: a systematic review and meta-analysis. Clin Microbiol Infect. 2021;27:331–340. doi: 10.1016/j.cmi.2020.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciensano: COVID-19 Bulletin épidémiologique hebdomadaire du 26 juin 2020. In.; 2020.
- Shah ASV, Wood R, Gribben C, Caldwell D, Bishop J, Weir A, et al. Risk of hospital admission with coronavirus disease 2019 in healthcare workers and their households: nationwide linkage cohort study. BMJ. 2020;371:m3582. doi: 10.1136/bmj.m3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steensels D, Oris E, Coninx L, Nuyens D, Delforge ML, Vermeersch P, et al. Hospital-Wide SARS-CoV-2 antibody screening in 3056 staff in a Tertiary Center in Belgium. JAMA. 2020;324:195–197. doi: 10.1001/jama.2020.11160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams PCM, Howard-Jones AR, Hsu P, Palasanthiran P, Gray PE, McMullan BJ, et al. SARS-CoV-2 in children: spectrum of disease, transmission and immunopathological underpinnings. Pathology. 2020;52:801–808. doi: 10.1016/j.pathol.2020.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeberg H, Paabo S. The major genetic risk factor for severe COVID-19 is inherited from Neanderthals. Nature. 2020;587:610–612. doi: 10.1038/s41586-020-2818-3. [DOI] [PubMed] [Google Scholar]
- Zhang Q, Bastard P, Liu Z, Le Pen J, Moncada-Velez M, Chen J, et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science. 2020;370:eabd4570. doi: 10.1126/science.abd4570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Z, Peng F, Xu B, Zhao J, Liu H, Peng J, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020;81:e16–e25. doi: 10.1016/j.jinf.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]