Abstract
Type 2 diabetes mellitus, obesity, hypertension, and other associated metabolic complications have been demonstrated as a crucial contributor to the enhanced morbidity and mortality of patients with coronavirus disease 2019 (COVID-19). Data on the interplay between metabolic comorbidities and the outcomes in patients with COVID-19 have been emerging and rapidly increasing. This implies a mechanistic link between metabolic diseases and COVID-19 resulting in the exacerbation of the condition. Nonetheless, new evidences are emerging to support insulin-mediated aggressive glucose-lowering treatment as a possible trigger of high mortality rate in diabetic COVID-19 patients, putting the clinician in a confounding and difficult dilemma for the treatment of COVID-19 patients with metabolic comorbidities. Thus, this review discusses the pathophysiological link among severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), angiotensin-converting enzyme 2 (ACE2), metabolic complications, and severe inflammation in COVID-19 development, especially in those with multi-organ injuries. We discuss the influence of several routinely used drugs in COVID-19 patients, including anti-inflammatory and anti-coagulant drugs, antidiabetic drugs, renin-angiotensin-aldosterone system inhibitors. Especially, we provide a balanced overview on the clinical application of glucose-lowering drugs (insulin and metformin), angiotensin-converting-enzyme inhibitors, and angiotensin receptor blockers. Although there is insufficient evidence from clinical or basic research to comprehensively reveal the mechanistic link between adverse outcomes in COVID-19 and metabolic comorbidities, it is hoped that the update in the current review may help to better outline the optimal strategies for clinical management of COVID-19 patients with metabolic comorbidities.
Abbreviations: ACE, angiotensin-converting enzyme; ACE2, angiotensin-converting enzyme 2; ACEIs, angiotensin-converting enzyme inhibitors; AGT, angiotensinogen; Ang-1-7, angiotensin-(1-7); Ang II, angiotensin II; ARBs, angiotensin receptor blockers; ARDS, acute respiratory distress syndrome; AT1aR, angiotensin type 1a receptor; AT1R, angiotensin II type 1 receptor; AT2R, angiotensin II type 2 receptor; COVID-19, coronavirus disease 2019; GGT, gamma-glutamyl transferase; IL, interleukin; IFN-γ, interferon-gamma; MCP-1, monocyte chemotactic protein-1; RAAS, renin-angiotensin-aldosterone system; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; T2DM, type 2 diabetes mellitus; Th, T-helper; TMPRSS2, transmembrane protease serine 2; TNF, tumor necrosis factor
Keywords: Diabetes, Metabolic comorbidities, COVID-19, Hypertension
1. Introduction
Diabetes and its related metabolic complications have been demonstrated as the primary comorbidities potentiating severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Data from patients with confirmed coronavirus disease 2019 (COVID-19) in the U.S. [1] and China [2] consistently demonstrated that hypertension, diabetes mellitus, and cardiovascular diseases were the most common comorbidities. SARS-CoV-2 infection causes potent immune response, which results in severe systemic inflammation in COVID-19 patients [3]. Cytokine storm during COVID-19 development is considered one of the triggers to cause acute respiratory distress syndrome and multiorgan failure [3]. Given that type 2 diabetes and its related metabolic disorders are predominantly characterized by systemic, chronic, and low-grade inflammation, increasing evidence supports that inflammatory response functions as one of the key mechanisms linking COVID-19 and metabolic diseases [4], [5].
Angiotensin-converting enzyme 2 (ACE2) has been well established as a co-receptor for viral entry for SARS-CoV-2 with a growing body of evidence supporting its dominant role in the development of COVID-19 [6]. Although the significance of ACE2 expression on the development of COVID-19 and its related mortality remains largely unknown, there has been considerable evidence in animal studies in couple with some human data demonstrating the enhancement in ACE2 expression in the heart, brain, and even in urine consequent to the treatment with specific antihypertensive medications, such as angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) [7], [8], [9], [10]. Thus, there has been a raised concern that the treatment with ACEIs and ARBs affects the severity and mortality of COVID-19.
Another major concern about clinical management of metabolic comorbidities focuses on the application of glucose-lowering drugs in COVID-19 patients. Indeed, clinical and translational data regarding the interplay between metabolic comorbidities and the outcomes in patients with COVID-19 have been increasing dramatically. A large retrospective study revealed that type 2 diabetes mellitus (T2DM) with poorly controlled blood glucose was associated with higher mortality in COVID-19 patients [11]. Nonetheless, new evidences are emerging to demonstrate insulin-mediated aggressive glucose-lowering treatment as a possible trigger of high fatality in diabetic COVID-19 patients [12], putting the clinician in a confounding and difficult dilemma for the treatment of COVID-19 patients with metabolic comorbidities. Hence, the update of the new evidence is urgently needed for the exploration of optimal strategies to yield better outcomes in diabetic COVID-19 patients.
In this Review, we summarize the data showing the pathophysiological link among SARS-CoV-2, ACE2, metabolic complications, and severe inflammation in the development of COVID-19. We highlight the emerging data demonstrating the benefits and potential risks related to the use of various drugs targeting the disorders in glucose and lipid metabolism in COVID-19 patients. The overall aim of this Review is to provide a balanced overview and update on current data demonstrating the pathophysiological and therapeutic link between metabolic comorbidities and COVID-19, which might benefit for optimal strategies for the treatment of COVID-19 patients with metabolic comorbidities.
2. ACE/ACE2 imbalance aggravates metabolic diseases after SARS-CoV-2 infection
2.1. The SARS-CoV-2 virus caused COVID-19 by invading target organs containing ACE2 and TMPRSS2
As the largest positive-strand RNA virus, coronavirus has a lot of spike (S) glycoprotein on its surface, giving it the appearance of a crown under the electron microscope, hence its name [13]. Based on the genomic structure and phylogenetic analysis, the coronavirus family is currently divided into four groups: alpha-CoV (or group 1), beta-CoV (group 2), gamma-CoV (group 3), and delta-CoV (group 4) [88]. The S protein is a coronavirus structural protein that mainly mediates invasion of the virus by binding to the host cell receptor [14]. The specificity of the host depends on the type of receptor on the surface of the host cell that is bound to the coronavirus. ACE2 is considered a key receptor facilitating SARS-CoV-2 infection in various cell populations. Furthermore, the host serine protease transmembrane protease serine 2 (TMPRSS2) is essential for the activation of SARS-CoV-2. TMPRSS2 processes human coronaviruses by enzymatic cleavage of the spike protein and support cell entry [15], [16].
2.2. ACE2 as a key modulator in the renin-angiotensin system
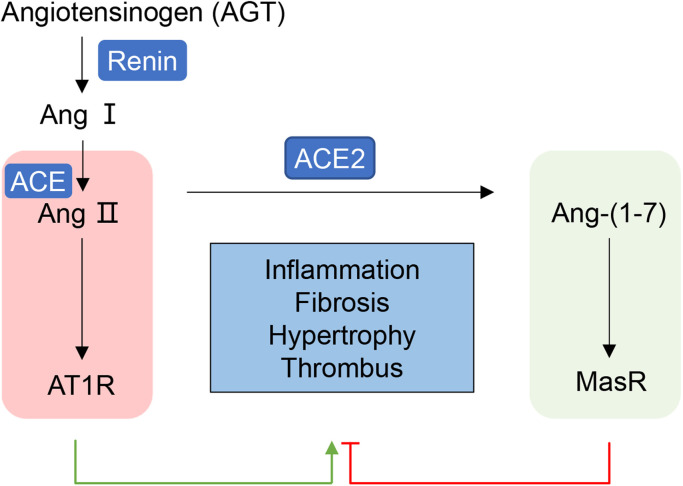
ACE2 was originally characterized and identified from the cDNA library of 2000 patients with heart failure [17], which opened a new branch in the renin–angiotensin–aldosterone system (RAAS) [7]. The RAAS functions as a key player in modulating cardiovascular physiology and homeostasis through regulating cell proliferation, apoptosis, fibrosis, oxidative stress, and inflammation [18]. It is mainly composed of two mutually antagonistic axes, the ACE/Ang II/AT1R axis and the ACE2/Ang-(1-7)/Mas receptor (MasR) axis. Angiotensinogen (AGT) is the sole precursor protein that harbors angiotensin I (Ang I) at the N-terminus and is specifically cleaved by renin [19]. ACE then catalyzes the cleavage of two amino acids from the C-terminal of Ang I to form angiotensin II (Ang II), mainly on the surface of endothelial cells, alveolar cells, and intestinal and renal proximal tubule epithelial cells [19]. Ang II increases hypertension and promotes inflammation, fibrosis, and myocardial hypertrophy by action in its two different cell-surface receptors, angiotensin II type 1 and 2 receptors (AT1R and AT2R) [18]. AT1R mediates most of the recognized detrimental effects of Ang II and is widely expressed in the cardiovascular system. Nonetheless, AT2R appears to oppose the effects of AT1R, mediating anti-inflammatory, anti-fibrosis, vasodilatory and antigrowth effects [19]. Like ACE, ACE2 belongs to the family of zinc metalloproteases with considerable homology to human ACE but quite different biological activity. ACE2 mainly degrades Ang II into the heptapeptide Ang-(1-7), and then Ang-(1-7) binds to the Mas receptor to produce an antagonistic effect on the ACE-Ang II-AT1R axis [20]. The ACE2/Ang-(1-7)/MasR axis has the opposite effect on the ACE/Ang II/AT1R axis, such as anti-inflammation, anti-fibrosis, anti-thrombus, and antihypertension and cardiovascular protection [21] ( Fig. 1).
Fig. 1.
Opposite effects of ACE and ACE2 in RAAS AGT is specifically cleaved by renin into Ang I, which is then catalyzed by ACE to form Ang II. Ang II exerts enhancing effects on hypertension, inflammation, fibrosis, myocardial hypertrophy, and vasoconstriction in cardiovascular system by action mainly in its AT1R. ACE2 mainly degrades Ang II into the heptapeptide Ang-(1-7) that binds to the MasR to produce an antagonistic effect on the ACE- Ang II-AT1R axis. AGT, Angiotensinogen; ACE, angiotensin-converting enzyme; ACE2, angiotensin-converting enzyme 2; Ang, angiotensin; AT1R, angiotensin II type 1 receptor; AT2R, angiotensin type 2 receptor; MasR, Mas receptor; RAAS, renin–angiotensin–aldosterone system.
2.3. ACE2 downregulation consequent to SARS-CoV-2 infection enhances metabolic disorders
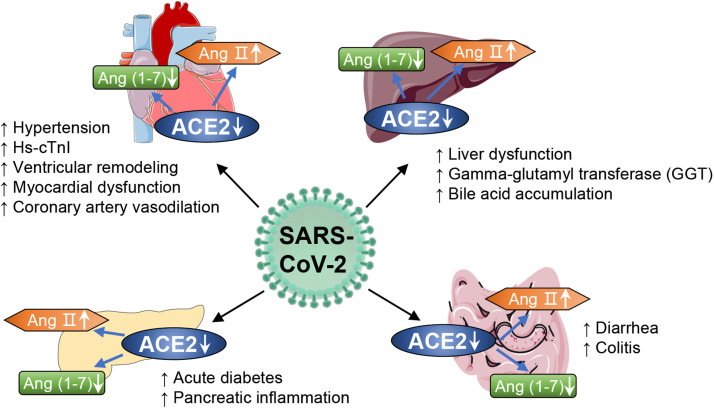
Functioning as key mediators for the entry of SARS-CoV-2 into cells, ACE2 is widely expressed in multiple organs including the kidneys, testes, adipose, and brain tissue, vascular smooth muscle cells, and the gastrointestinal tract and lungs [22], [23]. Thus, SARS-CoV-2 damages multiple organs, as reviewed elsewhere [24]. In regard that the homeostasis of glucose and lipid metabolism is predominantly orchestrated by metabolic organs including liver, pancreas, heart, and gut [25], [26], we focus on recent advances in the effects of SARS-CoV-2 infection on ACE/ACE2 imbalance, which significantly contributes to the dysfunction in various metabolic organs. The data may help to understand mechanisms underlying worsen outcomes in COVID-19 patients with metabolic morbidities ( Fig. 2).
Fig. 2.
ACE2 as a key contributor to SARS-CoV-2 infection in metabolic organs. ACE2 is widely expressed in metabolic organs including liver, pancreas, heart, and gut. SARS-CoV-2 infection evokes notable downregulation of ACE2 and consequent imbalance of ACE/ACE2, thereby causing virus-induced damage and dysfunction in metabolic organs and consequent worsen metabolic complications in patients with COVID-19. SARS-CoV-2, syndrome coronavirus 2; ACE, angiotensin-converting enzyme; ACE2, angiotensin-converting enzyme 2.
2.3.1. Liver
The liver is vital for the regulation of glucose and lipid metabolism. Damage to the liver consequent to coronavirus invasion may explain the disordered glucose and lipid metabolism in patients with COVID-19. Some retrospective studies have shown that patients with COVID-19 have abnormal liver biochemistry [27], [28], although recent reports indicate that ACE2 and TMPRSS2 are mainly expressed in human cholangiocytes, not hepatocytes [29]. Another study revealed that gamma-glutamyl transferase (GGT), a diagnostic biomarker of cholangiocyte injury, was elevated in 30 (54%) of 56 patients hospitalized with COVID-19 [27]. These results suggest that SARS-CoV-2 might directly bind to ACE2- and TMPRSS2-positive cholangiocytes. SARS-CoV-2 infection impairs the barrier and bile acid transporting functions of cholangiocytes in human liver ductal organoids and contributes to the bile acid accumulation and consequent liver damage. Of note, liver dysfunction in severely ill COVID-19 patients may also be due to drug hepatotoxicity and immune-mediated inflammation. To date, however, little is known about the direct viral infection of the liver by SARS-CoV-2.
2.3.2. Pancreas
Recent research suggests that ACE2 and TMPRSS2 are expressed in acinar, ductal, beta, alpha, mesenchymal and endothelial cells in the pancreas [30], [89]. This suggests that SARS-CoV-2 may infect the exocrine and endocrine pancreas and damage islet cells to cause acute diabetes [31]. In a retrospective analysis of COVID-19 patients, 17% of severely ill patients had pancreatic injuries [89]. Another study of 52 patients with COVID-19 pneumonia revealed that 17% had pancreatic injury. When compared with those without pancreatic injury, COVID-19 patients with pancreatic injury had a lower level of CD3+ T cells and CD4+ T cells, and a higher level of aspartate aminotransferase (AST), GGT, creatinine, lactate dehydrogenase, and erythrocyte sedimentation rate (ESR) [33]. Meanwhile, ACE2 is involved in the control of insulin secretion to the extent by regulating the levels of Ang II and/or Ang-(1-7) in pancreatic islets [34]. These findings suggest that pancreatic injury in COVID-19 patients may be directly due to the cytopathic effect mediated by ACE/ACE2 imbalance and virus-evoked immune response. Given that the pancreas is a key organ in the regulation of glucose homeostasis, any damage may adversely impact glucose metabolism. This may represent the key mechanism corresponding to abnormal blood glucose levels in patients with COVID-19.
2.3.3. Heart
ACE2 and TMPRSS2 are highly expressed in the heart [86]. Clinical studies demonstrated that 40% of patients with SARS-CoV-2 infection had cardiac-cerebrovascular disease [36]. Likewise, virus-related heart injury occurred in 12% of COVID-19 patients [3]. The decreased expression of ACE2 after coronavirus invasion increases Ang II and decreases Ang-(1-7) [37]. Ang II causes vascular remodeling of the aorta, evidenced by a significant increase in medial wall thickening and perivascular fibrosis [38]. Besides, the reduced Ang-(1-7) will weaken or extinguish the cardiovascular protection [38]. Therefore, SARS-CoV-2 may mediate myocardial inflammation and down-regulate myocardial ACE2 expression, causing an increase in Ang II and decreased Ang-(1-7).
2.3.4. Gut
Absorptive enterocytes in the ileum and colon show ACE2 expression in patients with COVID-19 [39], [40]. Furthermore, ACE2 and TMPRSS2 were detected in the ileal epithelium and lymphocytes [35]. A case in the United States demonstrated that SARS-CoV-2 was detected in stool samples of a patient who developed diarrhea and abdominal discomfort in addition to persistent fever and cough [41]. In contrast, although gastrointestinal symptoms have been infrequently reported in China [39], 17% of patients with COVID-19 in Singapore reported diarrhea [42]. Interestingly, SARS-CoV-2 RNA was detected in 50% of patient stool samples [42]. ACE2 has a RAAS-independent function, regulating dietary amino acid homeostasis, innate immunity, gut microbial ecology, and transmissible susceptibility to colitis [43]. Enhanced ACE2 expression has been observed in patients with inflammatory bowel disease [43]. These findings suggest that patients with colitis may be more vulnerable to SARS-CoV-2, and that the digestive system is a potential route for SARS-CoV-2 infection.
3. Metabolic inflammation links cytokine storm and multiorgan failure during COVID-19 progression
3.1. Cytokine storm is involved in disease progression of severe COVID-19
Cytokine storm is considered one of the major causes of acute respiratory distress syndrome (ARDS) and multiorgan failure [44]. Serum levels of many pro-inflammatory cytokines (IFN-γ, IL-1β, IL-6, IL-12, and TGF-β) and chemokines (CCL2, CXCL10, CXCL9, and IL-8) are markedly elevated in patients with COVID-19 [3], [45]. Moreover, IL-6 is a potential biomarker for severe illness, evidenced by serum IL-6 levels that were increased 10-fold in COVID-19 patients in ICU [5], [45]. Further study has also demonstrated a high concentration of IL-1β, interferon-gamma (IFN-γ), interferon-induced protein 10 (IP-10), and monocyte chemotactic protein-1 in COVID-19 patients that might lead to activated T-helper-1 (Th1) cell responses [3], [45].
Further study of immune mechanisms in animal models revealed that innate immunity was activated immediately and adaptive immunity quickly after SARS-CoV-2 infection. Following initial infection, lung-resident respiratory dendritic cells caught the pathogens or antigens from infected lung epithelial cells, processed the antigen, then migrated and presented the processed antigens to naive T cells [46], [47]. The T cells were then activated and could proliferate and migrate to the site of infection. After reaching the site of infection, activated virus-specific effector T cells produced antiviral cytokines (IFN-γ, TNF-α, IL-2) and chemokines (CXCL-9, 10, and 11) that recruited more innate and adaptive cells [46], [47]. The accumulated and activated mononuclear macrophages will produce more chemoattractants (such as CCL2, CCL7, and CCL12), resulting in further accumulation of macrophages and enhanced production of pro-inflammatory cytokines (TNF, IL-6, IL-1β) [48]. Meanwhile, virus-specific CD4 and CD8 T cells also clear the virus by eliminating virus-infected cells [47]. The chemokines produced by activated T cells boost the recruitment of innate and adaptive cells. Thus, following coronavirus infection, an overactive innate immunity may induce a cytokine storm and cause pathophysiological changes in multiple organs, especially the lungs.
3.2. Metabolic diseases are strongly related to the sustained production of pro-inflammatory cytokines
Metabolic diseases, such as obesity, T2DM, hypertension, and nonalcoholic fatty liver disease, are typically associated with an atypical immune system and several active cytokines. Tumor necrosis factor α (TNF-α) induces insulin resistance in rodents and animal models. A raised level of TNF-α induces insulin resistance in adipocytes and peripheral tissues by impairing insulin signaling through serine phosphorylation with consequent development of T2DM [49]. Similarly, hypertension is considered a low-grade inflammatory condition characterized by the presence of various pro-inflammatory cytokines: TNF-α is associated with salt-sensitive hypertension and related renal injury [50]. Interestingly, IL-1β is involved in pancreatic β-cell damage, whereas TNF-α is a key molecule involved in peripheral insulin resistance [51]. Meanwhile, the inhibition of IL-1β or the blockade of IL-1 receptor exerts protective effects on hypertension and hyperglycemia [52]. Moreover, IL-6 can either increase or decrease insulin resistance and enhance insulin secretion. In healthy individuals, IL-6 is a beneficial regulator of glucose disposal. On the contrary, in obese patients, IL-6 enhances the prevailing inflammation and precipitates insulin resistance and β-cell secretory dysfunction [51]. Expression of IL-6 has also been shown to be increased in hypertension and a decrease affords protection against Ang II-induced thrombosis, avoiding further damage due to hypertension [53].
The presence of T2DM is correlated significantly with increased production of interferon-gamma (IFN-γ), as well as alterations in the proliferation of macrophages and impaired function of natural killer cells [54], [55]. When CD4+ T cells are encountered by IL-6 and TGF-β, they tend to differentiate to Th17 cells that play a pathogenic role in various inflammatory disorders. CD4+ T cells polarize more easily to pro-inflammatory Th1 cells and Th17 cells in T2DM patients, with the polarization of anti-inflammatory Th2 cells simultaneously decreased [56]. Innate immunity also participates in the process of hypertension. Th1 and Th17 effectors participate in inflammation that causes increased blood pressure. Beyond T2DM and hypertension, cytokines may play a role in other long-term complications, including non-alcoholic steatohepatitis and cardiovascular complications [57].
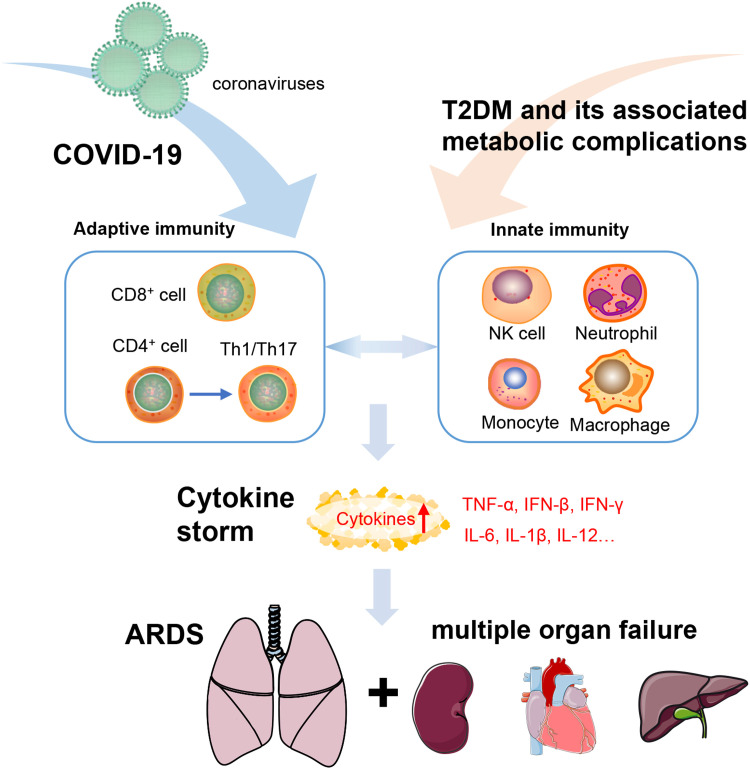
Moreover, high glucose directly promotes SARS-CoV-2 replication in close association with enhanced production of reactive oxygen species and proinflammatory cytokines (TNF-α, IL-1β, and IL-6) [58]. Hence, T2DM and its associated metabolic complications have been supported by a growing body of evidence as crucial contributors to the enhanced morbidity and mortality in COVID-19 patients. This impaired ability to heal due to metabolic inflammation results in prolonged recovery with the consequent risk of severe SARS-CoV-2 infection and ultimately a cytokine storm that may accelerate organ failure, even cause death ( Fig. 3).
Fig. 3.
Inflammatory mechanisms underlying the aggravation of COVID-19 patients with pre-existing metabolic disorders. The infection of SARS-CoV-2 evokes potent adaptive immunity, which is predominantly modulated by T-cell responses. In patients with pre-existing metabolic disorders, chronic and systemic inflammation largely mediated by innate immune cells (NK cells, neutrophils, monocyte, and macrophages) promotes the susceptibility to cytokine storm through the interplay with virus-elicited adaptive immune response, thereafter aggregating the development of ARDS and multiorgan failure. SARS-CoV-2, syndrome coronavirus 2; ARDS, acute respiratory distress syndrome.
4. The use of glucose-lowering drugs in COVID-19 patients
4.1. Insulin
Insulin is one of the most commonly used glucose-lowering drugs currently. It has been documented that insulin is widely used in the clinical management of COVID-19 patients with hyperglycemia, especially in severe cases [59]. Nonetheless, contradictory evidence is recently emerging to show that insulin therapy per se may promote fatality in patients with COVID-19 and diabetes [12]. The retrospective study investigating 689 patients with COVID-19 and T2DM revealed that insulin treatment for patients with COVID-19 and T2DM was associated with a significant increase in mortality (27.2% vs. 3.5%), which was accompanied by promoted systemic inflammation and enhanced injuries of vital organs, indicating that insulin treatment for patients with COVID-19 and T2DM should be used with caution. Thus, these data highlight the importance of close monitoring of blood glucose, vital signs, and organ injuries during clinical management of patients with COVID-19 and T2DM. Nonetheless, more robust data from large, randomized prospective controlled clinical trials are required to consolidate the findings regarding deleterious effects of insulin therapy in COVID-19 patients.
4.2. Metformin
Metformin is well considered as the preferred glucose-lowering drug for T2DM patients. Pleiotropic effects may contribute to its anti-diabetic benefits, such as the enhancement of glucose uptake in skeletal muscle and adipose tissues, the decrease in the amount of glucose generated in the liver, and the decrease in the absorption of glucose from the intestine [60]. A large retrospective cohort analysis recruiting 6256 patients demonstrated that metformin treatment was significantly associated with decreased mortality in female patients with COVID-19 and T2DM, but not in male patients [61]. Although the precise mechanisms underlying the gender effect of metformin treatment remains poorly understood, the benefit of clinical use of metformin in diabetic patients with COVID-19 is supported by additional retrospective studies [62], [63]. Nonetheless, the retrospective nature of these studies exploring metformin’s effects should be fully taken into consideration because of various potential confounders. In addition, metformin may have potential effects on the upregulation of ACE2, which may promote the progression of COVID-19 [64].
4.3. Other anti-diabetic drugs
Apart from insulin and metformin, several side effects have been reported in other anti-diabetic drugs, including hypoglycemia associated with sulfonylureas [65], dehydration and euglycemic diabetic ketoacidosis associated with SGLT-2 inhibitors [66]. In addition, single-use of either dipeptidyl peptidase-4 inhibitors or α-glucosidase inhibitors exhibits insufficient effectiveness to control blood glucose in diabetes patients [67]. Although these side effects are rare, an expert panel has recommended that these drugs should be discontinued for patients with severe symptoms of COVID-19 [66].
5. Antihypertensive drugs ACEIs and ARBs
Coronavirus binding to ACE2 can cause extensive degradation of the protein, resulting in decreased expression of ACE2. Thus, SARS-CoV-2 is associated with the activation of RAAS and blunted cardiovascular protection and antihypertensive effects of the ACE2/Ang-(1-7)/MasR axis [37], [87]. Given the pivotal role of the ACE2 receptor in cell entry of SARS-CoV-2, antihypertensive therapies, especially clinical use of two most common antihypertensive drugs ACEIs or ARBs, has raised considerable debate around the optimized strategy to treat COVID-19 patients with hypertension [6]. Profiling data revealed relatively enriched ACE2 expression in type II alveolar cells in the lung and vascular endothelium [36]. Meanwhile, some reports demonstrated enhanced ACE2 expression after the exposure to antihypertensive drugs [7], [8], [9], [10]. Thus, these findings have raised the concern that the treatment with ACEIs/ARBs may facilitate viral entry of SARS-CoV-2 entry thus promoting COVID-19 development. Hence, we summarize recent studies on this topic and provide a comprehensive and unbiased overview of the biological effects and clinical evidence about the use of ACEIs/ARBs and COVID-19 development.
5.1. ACEIs and ARBs may enhance ACE2 expression
ACEIs reduce the production of Ang II by inhibiting ACE, while ARBs block the Ang II receptor AT1 [68]. Early experiments have shown that ARBs and recombinant human ACE2 protein can reduce lung injury by antagonizing angiotensin receptor subtype 1a (AT1aR) and inactivating Ang II, respectively [69]. Therefore, for patients with COVID-19, ACEIs/ARBs remain important antihypertensive drugs and may reduce lung injury and prevent or reduce ARDS.
In theory, using renin-angiotensin system blockers such as ACEIs or ARBs may increase susceptibility to SARS-CoV-2. ACEIs or ARBs antihypertensive drugs may increase ACE2 while inhibiting ACE or AT1R [7]. This concern is supported by the most recent data demonstrating that human ACE2 transgenic mice infected with SARS-CoV-2 develop severe and fatal respiratory disease [70]. In an experiment to study the effect of ACEIs and ARBs on the cardiac ACE2 gene, selective blocking of Ang II synthesis increased cardiac ACE2 mRNA expression and activity [7]. Furthermore, some experiments confirmed that ARBs can increase the expression of ACE2 in the kidneys, heart, and lungs [8], [9], [10]. Although blood pressure decreased after the treatment with ACEIs and ARBs, both ACEIs and ARBs caused an increase in cardiac ACE2 mRNA [71]. Nonetheless, the other study failed to demonstrate such shifts in ACE2, although Ang-(1-7) and Ang II balance is positively shifted towards the beneficial vasopeptide Ang-(1-7) significantly [72]. Likewise, recent clinical data demonstrated that therapy with ACEIs and ARBs did not increase plasma ACE2 concentrations [32]. Therefore, the pathophysiological relevance of potential alteration in ACE2 expression consequent to the therapy with ACEIs/ARBs in the development of COVID-19 warrants further investigations.
5.2. ACEIs/ARBs did not aggravate cytokine storm or metabolic disease in patients with severe COVID-19
The recombinant human ACE2 protein (GSK2586881) has been demonstrated to protect against severe lung injury [73], [74]. In human studies, although the physiology and clinical indicators of patients with ARDS were not improved by GSK2568881, it rapidly decreased Ang-(1-7) and Ang-(1-5) levels [74]. Consistently, the mice with ACE2 deficiency were dramatically susceptible to endotoxin-induced ARDS when compared with wild-type control mice [75]. Furthermore, a systematic review and meta-analysis revealed that ACEIs exerted a putative protective role with close association with a decrease in pneumonia-related mortality, especially in patients with the previous stroke [76].
Some latest retrospective reviews of medical records from hospitalized COVID-19 patients with hypertension showed that ACEIs and ARBs improved the clinical outcomes and reduced a cytokine storm [77], [78]. Another multicenter, retrospective study that included 1128 adult patients with hypertension diagnosed with COVID-19 also showed that inpatients' use of ACEIs/ARBs were associated with a lower risk of all-cause mortality compared with ACEIs/ARBs non-users [79]. Currently, a multicenter, double-blind, placebo-controlled phase 2 randomized clinical trial investigating losartan use in patients with COVID-19 in an outpatient setting (ClinicalTrials.gov identifier: NCT04311177) and an in-patient setting (ClinicalTrials.gov identifier: NCT04312009) are being planned [80]. More clinical or scientific evidence is required from epidemiological studies and prospective trials before it can be concluded whether ACEIs/ARBs are safe to use and whether they can reduce the incidence or mortality of COVID-19–associated acute lung injury or ARDS.
6. Abnormal blood coagulation as a player contributing to the potentiating effects of metabolic disorders on the development of COVID-19
Severely ill patients with COVID-19 have also been shown to have higher levels of C-reactive protein and D-dimer [2], [40]. It was observed that elevated D-dimer levels at initial presentation predicted bleeding complications, thrombotic complications, critical illness, and death [81]. Likewise, many COVID-19 patients have been reported with clotting disorders and micro-thrombotic formation in peripheral blood vessels [82]. Anticoagulation therapy is recommended in patients with early-stage COVID-19 [81]. The level of D-dimer is a potential measurable prognostic biomarker with potential benefits for clinical management and prognosis of COVID-19 patients. A current study suggests that a D-dimer level on admission greater than 2.0 µg/mL (four-fold increase) could effectively predict in-hospital mortality in patients with COVID-19 [83]. Furthermore, hyper-fibrinolysis associated with plasmin led to elevated D-dimer in severely ill patients [84]. A plasminogen system may prove a promising therapeutic target for combating COVID-19.
7. Conclusions and future perspectives
T2DM and its associated metabolic comorbidities have been supported by a growing body of evidence as a predominant contributor to increased susceptibility and disease aggravation in patients with severe COVID-19, for which ACE2 and TMPRSS2 may play a key role. Hence, there are at least two important messages for the clinical management of COVID-19 patients with metabolic diseases. First, the optimal management of metabolic homeostasis of blood pressure, glucose, and lipids is essential to ensure better clinical outcomes and avoid exacerbating the disease. Second, in addition to symptomatic treatment, attention should be paid to the ACE/ACE2 imbalance, a cytokine storm caused by SARS-CoV-2 infection, and potential risk consequent to insulin treatment.
To ensure better clinical outcomes in COVID-19 patients, new drugs with potent anti-inflammatory capacity are urgently needed, especially for those with metabolic comorbidities. Several drugs targeting inflammation, including tocilizumab, sarilumab, siltuximab (IL-6 receptor antagonist monoclonal antibodies), anakinra (IL-1 inhibitors), the Janus kinase inhibitors fedratinib and baricitinib, and the C-C chemokine receptor type 5 (CCR5) antagonist leronlimab are all being trialed in COVID-19 patients. Nonetheless, the beneficial effects of anti-inflammatory drugs must be weighed against the possible detrimental effect of impaired immunity. Other emerging treatments for cytokine release syndrome (such as TMPRSS2 inhibitor, Vitamin C, Chloroquine, and hydroxychloroquine) [85], which may serve as potential therapies for severe COVID-19 patients, warrant further investigation in future studies.
CRediT authorship contribution statement
Shuang Hua, Yong Yang, and Danqi Zou drafted the manuscript and prepared the figures. Jufei Li, Kaixuan Yan, Ying Xu, Xue Jiang, and Xianglu Rong contributed to literature search, data analysis, and summary. Dewei Ye contributed to conceptualization and manuscript writing. All authors approved the final draft and agreed to submit it for publication.
Conflict of interest statement
The authors declare that they have no competing interests.
Acknowledgment
Research work in the authors' laboratory was supported by the National Natural Science Foundation of China (NSFC) Grant (81922074, 81770849, 81570701), Major basic and applied basic research projects in Guangdong Province (2019B030302005), Key Laboratory of Model Animal Phenotyping and Basic Research in Metabolic Diseases (2018KSYS003), Key Research Program in Department of Education of Guangdong Province (2016KZDXM041).
References
- 1.Garg S., Kim L., Whitaker M., O’Halloran A., Cummings C., Holstein R., Prill M., Chai S.J., Kirley P.D., Alden N.B., Kawasaki B., Yousey-Hindes K., Niccolai L., Anderson E.J., Openo K.P., Weigel A., Monroe M.L., Ryan P., Henderson J., Kim S., Como-Sabetti K., Lynfield R., Sosin D., Torres S., Muse A., Bennett N.M., Billing L., Sutton M., West N., Schaffner W., Talbot H.K., Aquino C., George A., Budd A., Brammer L., Langley G., Hall A.J., Fry A. Hospitalization rates and characteristics of patients hospitalized with laboratory-confirmed coronavirus disease 2019 - COVID-NET, 14 states, March 1-30, 2020. MMWR Morb. Mortal. Wkly Rep. 2020;69(15):458–464. doi: 10.15585/mmwr.mm6915e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bornstein S.R., Dalan R., Hopkins D., Mingrone G., Boehm B.O. Endocrine and metabolic link to coronavirus infection. Nat. Rev. Endocrinol. 2020;16(6):297–298. doi: 10.1038/s41574-020-0353-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen X., Zhao B., Qu Y., Chen Y., Xiong J., Feng Y., Men D., Huang Q., Liu Y., Yang B., Ding J., Li F. Detectable serum severe acute respiratory syndrome coronavirus 2 viral load (RNAemia) is closely correlated with drastically elevated interleukin 6 level in critically Ill patients with coronavirus disease 2019. Clin. Infect. Dis. 2020;71(8):1937–1942. doi: 10.1093/cid/ciaa449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wang Q., Zhang Y., Wu L., Niu S., Song C., Zhang Z., Lu G., Qiao C., Hu Y., Yuen K.Y., Wang Q., Zhou H., Yan J., Qi J. Structural and functional basis of SARS-CoV-2 entry by using human ACE2. Cell. 2020;181(4) doi: 10.1016/j.cell.2020.03.045. 894-904.e899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tipnis S.R., Hooper N.M., Hyde R., Karran E., Christie G., Turner A.J. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J. Biol. Chem. 2000;275(43):33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- 8.Tunçdemir M., Demirkesen O., Öztürk M., Atukeren P., Gümüştaş M., Turan T. Antiapoptotic effect of angiotensin-II type-1 receptor blockade in renal tubular cells of hyperoxaluric rats. Urol. Res. 2010;38:71–80. doi: 10.1007/s00240-010-0255-8. [DOI] [PubMed] [Google Scholar]
- 9.Ishiyama Y., Gallagher P., Averill D., Tallant E., Brosnihan K., Ferrario C. Upregulation of angiotensin-converting enzyme 2 after myocardial infarction by blockade of angiotensin II receptors. Hypertension. 2004;43:970–976. doi: 10.1161/01.HYP.0000124667.34652.1a. [DOI] [PubMed] [Google Scholar]
- 10.Kaiqiang J., Minakawa M., Fukui K., Suzuki Y., Fukuda I. Olmesartan improves left ventricular function in pressure-overload hypertrophied rat heart by blocking angiotensin II receptor with synergic effects of upregulation of angiotensin converting enzyme 2. Ther. Adv. Cardiovasc Dis. 2009;3(2):103–111. doi: 10.1177/1753944708098691. [DOI] [PubMed] [Google Scholar]
- 11.Zhu L., She Z.-G., Cheng X., Qin J.-J., Zhang X.-J., Cai J., Lei F., Wang H., Xie J., Wang W., Li H., Zhang P., Song X., Chen X., Xiang M., Zhang C., Bai L., Xiang D., Chen M.M., Liu Y., Yan Y., Liu M., Mao W., Zou J., Liu L., Chen G., Luo P., Xiao B., Zhang C., Zhang Z., Lu Z., Wang J., Lu H., Xia X., Wang D., Liao X., Peng G., Ye P., Yang J., Yuan Y., Huang X., Guo J., Zhang B.H., Li H. Association of blood glucose control and outcomes in patients with COVID-19 and pre-existing type 2 diabetes. Cell Metab. 2020;31(6) doi: 10.1016/j.cmet.2020.04.021. 1068-1077.e1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yu B., Li C., Sun Y., Wang D.W. Insulin treatment is associated with increased mortality in patients with COVID-19 and type 2 diabetes. Cell Metab. 2021;33(1):65–77. doi: 10.1016/j.cmet.2020.11.014. 65-77.e62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tyrrell D., Sparrow P., Beare A. Relation between blood groups and resistance to infection with influenza and some picornaviruses. Nature. 1968;220:819–820. doi: 10.1038/220819a0. [DOI] [PubMed] [Google Scholar]
- 14.Gallagher T., Perlman S. Public health: broad reception for coronavirus. Nature. 2013;495(7440):176–177. doi: 10.1038/495176a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Iwata-Yoshikawa N., Okamura T., Shimizu Y., Hasegawa H., Takeda M., Nagata N. TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection. J. Virol. 2019;93(6) doi: 10.1128/JVI.01815-18. e01815-01818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280. doi: 10.1016/j.cell.2020.02.052. 271-280.e278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Donoghue M., Hsieh F., Baronas E., Godbout K., Gosselin M., Stagliano N., Donovan M., Woolf B., Robison K., Jeyaseelan R., Breitbart R.E., Acton S. A novel angiotensin-converting enzyme–related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ. Res. 2000;87(5):e1–e9. doi: 10.1161/01.res.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 18.Mendoza-Torres E., Oyarzún A., Mondaca-Ruff D., Azocar A., Castro P.F., Jalil J.E., Chiong M., Lavandero S., Ocaranza M.P. ACE2 and vasoactive peptides: novel players in cardiovascular/renal remodeling and hypertension. Ther. Adv. Cardiovasc. Dis. 2015;9(4):217–237. doi: 10.1177/1753944715597623. [DOI] [PubMed] [Google Scholar]
- 19.Xu Y., Rong J., Zhang Z. The emerging role of angiotensinogen in cardiovascular diseases. J. Cell Physiol. 2021;236(1):68–78. doi: 10.1002/jcp.29889. [DOI] [PubMed] [Google Scholar]
- 20.Medina D., Arnold A.C. Angiotensin-(1-7): translational avenues in cardiovascular control. Am. J. Hypertens. 2019;32(12):1133–1142. doi: 10.1093/ajh/hpz146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pang X., Cui Y., Zhu Y. Recombinant human ACE2: potential therapeutics of SARS-CoV-2 infection and its complication. Acta Pharmacol. Sin. 2020;41:1255–1257. doi: 10.1038/s41401-020-0430-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hikmet F., Méar L., Edvinsson Å., Micke P., Uhlén M., Lindskog C. The protein expression profile of ACE2 in human tissues. Mol. Syst. Biol. 2020;16(7):9610. doi: 10.15252/msb.20209610. e9610-e9610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Singh M., Bansal V., Feschotte C. A single-cell RNA expression map of human coronavirus entry factors. Cell Rep. 2020;32(12) doi: 10.1016/j.celrep.2020.108175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Harrison A.G., Lin T., Wang P. Mechanisms of SARS-CoV-2 transmission and pathogenesis. Trends Immunol. 2020;41(12):1100–1115. doi: 10.1016/j.it.2020.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Guilherme A., Henriques F., Bedard A.H., Czech M.P. Molecular pathways linking adipose innervation to insulin action in obesity and diabetes mellitus. Nat. Rev. Endocrinol. 2019;15(4):207–225. doi: 10.1038/s41574-019-0165-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lempradl A., Pospisilik J.A., Penninger J.M. Exploring the emerging complexity in transcriptional regulation of energy homeostasis. Nat. Rev. Genet. 2015;16(11):665–681. doi: 10.1038/nrg3941. [DOI] [PubMed] [Google Scholar]
- 27.Zhang C., Shi L., Wang F.-S. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol. Hepatol. 2020;5(5):428–430. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hung I.F., Lung K.C., Tso E.Y., Liu R., Chung T.W., Chu M.Y., Ng Y.Y., Lo J., Chan J., Tam A.R., Shum H.P., Chan V., Wu A.K., Sin K.M., Leung W.S., Law W.L., Lung D.C., Sin S., Yeung P., Yip C.C., Zhang R.R., Fung A.Y., Yan E.Y., Leung K.H., Ip J.D., Chu A.W., Chan W.M., Ng A.C., Lee R., Fung K., Yeung A., Wu T.C., Chan J.W., Yan W.W., Chan W.M., Chan J.F., Lie A.K., Tsang O.T., Cheng V.C., Que T.L., Lau C.S., Chan K.H., To K.K., Yuen K.Y. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395(10238):1695–1704. doi: 10.1016/S0140-6736(20)31042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chai X., Hu L., Zhang Y., Han W., Lu Z., Ke A., Zhou J., Shi G., Fang N., Fan J. et al.: Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019-nCoV Infection. bioRxiv 2020:2020.2002.2003.931766.
- 30.Yang L., Han Y., Nilsson-Payant B.E., Gupta V., Wang P., Duan X., Tang X., Zhu J., Zhao Z., Jaffré F., Zhang T., Kim T.W., Harschnitz O., Redmond D., Houghton S., Liu C., Naji A., Ciceri G., Guttikonda S., Bram Y., Nguyen D.H.T., Cioffi M., Chandar V., Hoagland D.A., Huang Y., Xiang J., Wang H., Lyden D., Borczuk A., Chen H.J., Studer L., Pan F.C., Ho D.D., tenOever B.R., Evans T., Schwartz R.E., Chen S. A human pluripotent stem cell-based platform to study SARS-CoV-2 tropism and model virus infection in human cells and organoids. Cell Stem Cell. 2020;27:125–136.e7. doi: 10.1016/j.stem.2020.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kusmartseva I., Wu W., Syed F., Van Der Heide V., Jorgensen M., Joseph P., Tang X., Candelario-Jalil E., Yang C., Nick H., Harbert J.L., Posgai A.L., Paulsen J.D., Lloyd R., Cechin S., Pugliese A., Campbell-Thompson M., Vander Heide R.S., Evans-Molina C., Homann D., Atkinson M.A. Expression of SARS-CoV-2 entry factors in the pancreas of normal organ donors and individuals with COVID-19. Cell Metab. 2020;32(6):1041–1051. doi: 10.1016/j.cmet.2020.11.005. 1041-1051.e1046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sama I.E., Ravera A., Santema B.T., van Goor H., ter Maaten J.M., Cleland J.G.F., Rienstra M., Friedrich A.W., Samani N.J., Ng L.L., Dickstein K., Lang C.C., Filippatos G., Anker S.D., Ponikowski P., Metra M., van Veldhuisen D.J., Voors A.A. Circulating plasma concentrations of angiotensin-converting enzyme 2 in men and women with heart failure and effects of renin–angiotensin–aldosterone inhibitors. Eur. Heart J. 2020;41(19):1810–1817. doi: 10.1093/eurheartj/ehaa373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang F., Wang H., Fan J., Zhang Y., Wang H., Zhao Q. Pancreatic injury patterns in patients with coronavirus Disease 19 pneumonia. Gastroenterology. 2020;159(1):367–370. doi: 10.1053/j.gastro.2020.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Batlle D., Jose Soler M., Ye M. ACE2 and diabetes: ACE of ACEs? Diabetes. 2010;59(12):2994–2996. doi: 10.2337/db10-1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bertram S., Heurich A., Lavender H., Gierer S., Danisch S., Perin P., Lucas J.M., Nelson P.S., Pöhlmann S., Soilleux E.J. Influenza and SARS-coronavirus activating proteases TMPRSS2 and HAT are expressed at multiple sites in human respiratory and gastrointestinal tracts. PLoS One. 2012;7(4):35876. doi: 10.1371/journal.pone.0035876. e35876-e35876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., Xia J., Yu T., Zhang X., Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395(10223):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., Huan Y., Yang P., Zhang Y., Deng W., Bao L., Zhang B., Liu G., Wang Z., Chappell M., Liu Y., Zheng D., Leibbrandt A., Wada T., Slutsky A.S., Liu D., Qin C., Jiang C., Penninger J.M. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. 2005;11(8):875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Antoniak S., Cardenas J.C., Buczek L.J., Church F.C., Mackman N., Pawlinski R. Protease-activated receptor 1 contributes to angiotensin II-induced cardiovascular remodeling and inflammation. Cardiology. 2017;136(4):258–268. doi: 10.1159/000452269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Liang W., Feng Z., Rao S., Xiao C., Xue X., Lin Z., Zhang Q., Qi W. Diarrhoea may be underestimated: a missing link in 2019 novel coronavirus. Gut. 2020;69(6):1141–1143. doi: 10.1136/gutjnl-2020-320832. [DOI] [PubMed] [Google Scholar]
- 40.Zhang J.J., Dong X., Cao Y.Y., Yuan Y.D., Yang Y.B., Yan Y.Q., Akdis C.A., Gao Y.D. Clinical characteristics of 140 patients infected with SARS-CoV-2 in Wuhan, China. Allergy. 2020;75(7):1730–1741. doi: 10.1111/all.14238. [DOI] [PubMed] [Google Scholar]
- 41.Holshue M.L., DeBolt C., Lindquist S., Lofy K.H., Wiesman J., Bruce H., Spitters C., Ericson K., Wilkerson S., Tural A., Diaz G., Cohn A., Fox L., Patel A., Gerber S.I., Kim L., Tong S., Lu X., Lindstrom S., Pallansch M.A., Weldon W.C., Biggs H.M., Uyeki T.M., Pillai S.K., Washington State -nCoV Case Investigation T. First Case of 2019 novel coronavirus in the United States. N. Engl. J. Med. 2020;382(10):929–936. doi: 10.1056/NEJMoa2001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ong J., Young B.E., Ong S. COVID-19 in gastroenterology: a clinical perspective. Gut. 2020;69(6):1144–1145. doi: 10.1136/gutjnl-2020-321051. [DOI] [PubMed] [Google Scholar]
- 43.Khajah M.A., Fateel M.M., Ananthalakshmi K.V., Luqmani Y.A. Anti-inflammatory action of angiotensin 1-7 in experimental colitis. PLoS One. 2016;11(3) doi: 10.1371/journal.pone.0150861. e0150861-e0150861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Costela-Ruiz V.J., Illescas-Montes R., Puerta-Puerta J.M., Ruiz C., Melguizo-Rodríguez L. SARS-CoV-2 infection: the role of cytokines in COVID-19 disease. Cytokine Growth Factor Rev. 2020;54:62–75. doi: 10.1016/j.cytogfr.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Han H., Ma Q., Li C., Liu R., Zhao L., Wang W., Zhang P., Liu X., Gao G., Liu F., Jiang Y., Cheng X., Zhu C., Xia Y. Profiling serum cytokines in COVID-19 patients reveals IL-6 and IL-10 are disease severity predictors. Emerg. Microbes Infect. 2020;9(1):1123–1130. doi: 10.1080/22221751.2020.1770129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bhaskar S., Sinha A., Banach M., Mittoo S., Weissert R., Kass J.S., Rajagopal S., Pai A.R., Kutty S. Cytokine storm in COVID-19-immunopathological mechanisms, clinical considerations, and therapeutic approaches: the REPROGRAM Consortium Position Paper. Front Immunol. 2020;11:1648. doi: 10.3389/fimmu.2020.01648. 1648-1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu L., Xu L., Lin C. T cell response in patients with COVID-19. Blood Sci. 2020;2(3):76–78. doi: 10.1097/BS9.0000000000000050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang J., Jiang M., Chen X., Montaner L.J. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. J. Leukoc. Biol. 2020;108(1):17–41. doi: 10.1002/JLB.3COVR0520-272R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Akash M.S.H., Rehman K., Liaqat A. Tumor necrosis factor-alpha: role in development of insulin resistance and pathogenesis of Type 2 diabetes mellitus. J. Cell. Biochem. 2018;119(1):105–110. doi: 10.1002/jcb.26174. [DOI] [PubMed] [Google Scholar]
- 50.Mehaffey E., Majid D.S.A. Tumor necrosis factor-α, kidney function, and hypertension. Am. J. Physiol. Ren. Physiol. 2017;313(4):F1005–F1008. doi: 10.1152/ajprenal.00535.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Donath M.Y., Dinarello C.A., Mandrup-Poulsen T. Targeting innate immune mediators in type 1 and type 2 diabetes. Nat. Rev. Immunol. 2019;19(12):734–746. doi: 10.1038/s41577-019-0213-9. [DOI] [PubMed] [Google Scholar]
- 52.Donath M.Y., Meier D.T., Böni-Schnetzler M. Inflammation in the pathophysiology and therapy of cardiometabolic disease. Endocr. Rev. 2019;40(4):1080–1091. doi: 10.1210/er.2019-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Daniele G., Guardado Mendoza R., Winnier D., Fiorentino T.V., Pengou Z., Cornell J., Andreozzi F., Jenkinson C., Cersosimo E., Federici M., Tripathy D., Folli F. The inflammatory status score including IL-6, TNF-α, osteopontin, fractalkine, MCP-1 and adiponectin underlies whole-body insulin resistance and hyperglycemia in type 2 diabetes mellitus. Acta Diabetol. 2014;51(1):123–131. doi: 10.1007/s00592-013-0543-1. [DOI] [PubMed] [Google Scholar]
- 54.Abdel-Moneim A., Semmler M., Abdel-Reheim E.S., Zanaty M.I., Addaleel W. Association of glycemic status and interferon-gamma production with leukocytes and platelet indices alterations in type2 diabetes. Diabetes Metab. Syndr. 2019;13(3):1963–1969. doi: 10.1016/j.dsx.2019.04.046. [DOI] [PubMed] [Google Scholar]
- 55.Zhou T., Hu Z., Yang S., Sun L., Yu Z., Wang G. Role of adaptive and innate immunity in Type 2 diabetes mellitus. J. Diabetes Res. 2018;2018 doi: 10.1155/2018/7457269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Channappanavar R., Zhao J., Perlman S. T cell-mediated immune response to respiratory coronaviruses. Immunol. Res. 2014;59(1–3):118–128. doi: 10.1007/s12026-014-8534-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jin C., Henao-Mejia J., Flavell Richard A. Innate immune receptors: key regulators of metabolic disease progression. Cell Metab. 2013;17(6):873–882. doi: 10.1016/j.cmet.2013.05.011. [DOI] [PubMed] [Google Scholar]
- 58.Codo A.C., Davanzo G.G., Monteiro L.B., de Souza G.F., Muraro S.P., Virgilio-da-Silva J.V., Prodonoff J.S., Carregari V.C., de Biagi Junior C.A.O., Crunfli F., Jimenez Restrepo J.L., Vendramini P.H., Reis-de-Oliveira G., Bispo dos Santos K., Toledo-Teixeira D.A., Parise P.L., Martini M.C., Marques R.E., Carmo H.R., Borin A., Coimbra L.D., Boldrini V.O., Brunetti N.S., Vieira A.S., Mansour E., Ulaf R.G., Bernardes A.F., Nunes T.A., Ribeiro L.C., Palma A.C., Agrela M.V., Moretti M.L., Sposito A.C., Pereira F.B., Velloso L.A., Vinolo M.A.R., Damasio A., Proença-Módena J.L., Carvalho R.F., Mori M.A., Martins-de-Souza D., Nakaya H.I., Farias A.S., Moraes-Vieira P.M. Elevated glucose levels favor SARS-CoV-2 infection and monocyte response through a HIF-1α/glycolysis-dependent axis. Cell Metab. 2020;32(3):498–499. doi: 10.1016/j.cmet.2020.07.015. 437-446.e435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Gianchandani R., Esfandiari N.H., Ang L., Iyengar J., Knotts S., Choksi P., Pop-Busui R. Managing hyperglycemia in the COVID-19 inflammatory storm. Diabetes. 2020;69(10):2048–2053. doi: 10.2337/dbi20-0022. [DOI] [PubMed] [Google Scholar]
- 60.Zhou J., Massey S., Story D., Li L. Metformin: an old drug with new applications. Int. J. Mol. Sci. 2018;19(10):2863. doi: 10.3390/ijms19102863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bramante C.T., Ingraham N.E., Murray T.A., Marmor S., Hovertsen S., Gronski J., McNeil C., Feng R., Guzman G., Abdelwahab N., King S., Tamariz L., Meehan T., Pendleton K.M., Benson B., Vojta D., Tignanelli C.J. Metformin and risk of mortality in patients hospitalised with COVID-19: a retrospective cohort analysis. Lancet Healthy Longev. 2021;2(1):e34–e41. doi: 10.1016/S2666-7568(20)30033-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Luo P., Qiu L., Liu Y., Liu X.L., Zheng J.L., Xue H.Y., Liu W.H., Liu D., Li J. Metformin treatment was associated with decreased mortality in COVID-19 patients with diabetes in a retrospective analysis. Am. J. Trop. Med Hyg. 2020;103(1):69–72. doi: 10.4269/ajtmh.20-0375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chen Y., Yang D., Cheng B., Chen J., Peng A., Yang C., Liu C., Xiong M., Deng A., Zhang Y., Zheng L., Huang K. Clinical characteristics and outcomes of patients with diabetes and COVID-19 in association with glucose-lowering medication. Diabetes Care. 2020;43(7):1399–1407. doi: 10.2337/dc20-0660. [DOI] [PubMed] [Google Scholar]
- 64.Zhang J., Dong J., Martin M., He M., Gongol B., Marin T.L., Chen L., Shi X., Yin Y., Shang F., Wu Y., Huang H.Y., Zhang J., Zhang Y., Kang J., Moya E.A., Huang H.D., Powell F.L., Chen Z., Thistlethwaite P.A., Yuan Z.Y., Shyy J.Y. AMP-activated protein kinase phosphorylation of angiotensin-converting enzyme 2 in endothelium mitigates pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2018;198(4):509–520. doi: 10.1164/rccm.201712-2570OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shah K., Tiwaskar M., Chawla P., Kale M., Deshmane R., Sowani A. Hypoglycemia at the time of Covid-19 pandemic. Diabetes Metab. Syndr. 2020;14(5):1143–1146. doi: 10.1016/j.dsx.2020.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bornstein S.R., Rubino F., Khunti K., Mingrone G., Hopkins D., Birkenfeld A.L., Boehm B., Amiel S., Holt R.I., Skyler J.S., DeVries J.H., Renard E., Eckel R.H., Zimmet P., Alberti K.G., Vidal J., Geloneze B., Chan J.C., Ji L., Ludwig B. Practical recommendations for the management of diabetes in patients with COVID-19. Lancet Diabetes Endocrinol. 2020;8(6):546–550. doi: 10.1016/S2213-8587(20)30152-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Donath M.Y. Glucose or insulin, which is the culprit in patients with COVID-19 and diabetes? Cell Metab. 2021;33(1):2–4. doi: 10.1016/j.cmet.2020.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Asperen R., Lutter R., Specht P., Moll G., van Woensel J., Loos C., van Goor H., Kamilic J., Florquin S., Bos A. Acute respiratory distress syndrome leads to reduced ratio of ACE/ACE2 activities and is prevented by angiotensin-(1-7) or an angiotensin II receptor antagonist. J. Pathol. 2011;225:618–627. doi: 10.1002/path.2987. [DOI] [PubMed] [Google Scholar]
- 69.Riordan J.F. Angiotensin-I-converting enzyme and its relatives. Genome Biol. 2003;4(8):225. doi: 10.1186/gb-2003-4-8-225. 225-225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Golden J.W., Cline C.R., Zeng X., Garrison A.R., Carey B.D., Mucker E.M., White L.E., Shamblin J.D., Brocato R.L., Liu J., Babka A.M., Rauch H.B., Smith J.M., Hollidge B.S., Fitzpatrick C., Badger C.V., Hooper J.W. Human angiotensin-converting enzyme 2 transgenic mice infected with SARS-CoV-2 develop severe and fatal respiratory disease. JCI Insight. 2020;5 doi: 10.1172/jci.insight.142032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ferrario C., Jessup J., Chappell M., Averill D., Brosnihan K., Tallant E., Diz D., Gallagher P. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation. 2005;111:2605–2610. doi: 10.1161/CIRCULATIONAHA.104.510461. [DOI] [PubMed] [Google Scholar]
- 72.Hamming I., Van Goor H., Turner A.J., Rushworth C.A., Michaud A.A., Corvol P., Navis G. Differential regulation of renal angiotensin-converting enzyme (ACE) and ACE2 during ACE inhibition and ditary sodium restriction in healthy rats. Exp. Physiol. 2008;93(5):631–638. doi: 10.1113/expphysiol.2007.041855. [DOI] [PubMed] [Google Scholar]
- 73.Kleinsasser A., Pircher I., Treml B., Schwienbacher M., Schuster M., Janzek E., Loibner H., Penninger J., Löckinger A. Recombinant angiotensin-converting enzyme 2 suppresses pulmonary vasoconstriction in acute hypoxia. Wilderness Environ. Med. 2012;23:24–30. doi: 10.1016/j.wem.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 74.Khan A., Benthin C., Zeno B., Albertson T.E., Boyd J., Christie J.D., Hall R., Poirier G., Ronco J.J., Tidswell M., Hardes K., Powley W.M., Wright T.J., Siederer S.K., Fairman D.A., Lipson D.A., Bayliffe A.I., Lazaar A.L. A pilot clinical trial of recombinant human angiotensin-converting enzyme 2 in acute respiratory distress syndrome. Crit. Care. 2017;21(1):234. doi: 10.1186/s13054-017-1823-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., Yang P., Sarao R., Wada T., Leong-Poi H., Crackower M.A., Fukamizu A., Hui C.C., Hein L., Uhlig S., Slutsky A.S., Jiang C., Penninger J.M. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Caldeira D., Alarcão J., Vaz-Carneiro A., Costa J. Risk of pneumonia associated with use of angiotensin converting enzyme inhibitors and angiotensin receptor blockers: systematic review and meta-analysis. BMJ. 2012;345:4260. doi: 10.1136/bmj.e4260. e4260-e4260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Meng J., Xiao G., Zhang J., He X., Ou M., Bi J., Yang R., Di W., Wang Z., Li Z., Gao H., Liu L., Zhang G. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg. Microbes Infect. 2020;9(1):757–760. doi: 10.1080/22221751.2020.1746200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Rosenthal N., Cao Z., Gundrum J., Sianis J., Safo S. Risk factors associated with in-hospital mortality in a US national sample of patients with COVID-19. JAMA Netw. Open. 2020;3(12) doi: 10.1001/jamanetworkopen.2020.29058. e2029058-e2029058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhang P., Zhu L., Cai J., Lei F., Qin J.J., Xie J., Liu Y.M., Zhao Y.C., Huang X., Lin L., Xia M., Chen M.M., Cheng X., Zhang X., Guo D., Peng Y., Ji Y.X., Chen J., She Z.G., Wang Y., Xu Q., Tan R., Wang H., Lin J., Luo P., Fu S., Cai H., Ye P., Xiao B., Mao W., Liu L., Yan Y., Liu M., Chen M., Zhang X.J., Wang X., Touyz R.M., Xia J., Zhang B.H., Huang X., Yuan Y., Loomba R., Liu P.P., Li H. Association of inpatient use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19. Circ. Res. 2020;126(12):1671–1681. doi: 10.1161/CIRCRESAHA.120.317134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Bavishi C., Maddox T.M., Messerli F.H. Coronavirus disease 2019 (COVID-19) infection and renin angiotensin system blockers. JAMA Cardiol. 2020;5(7):745–747. doi: 10.1001/jamacardio.2020.1282. [DOI] [PubMed] [Google Scholar]
- 81.Al-Samkari H., Karp Leaf R.S., Dzik W.H., Carlson J.C.T., Fogerty A.E., Waheed A., Goodarzi K., Bendapudi P.K., Bornikova L., Gupta S., Leaf D.E., Kuter D.J., Rosovsky R.P. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136(4):489–500. doi: 10.1182/blood.2020006520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Jin Y.H., Cai L., Cheng Z.S., Cheng H., Deng T., Fan Y.P., Fang C., Huang D., Huang L.Q., Huang Q., Han Y., Hu B., Hu F., Li B.H., Li Y.R., Liang K., Lin L.K., Luo L.S., Ma J., Ma L.L., Peng Z.Y., Pan Y.B., Pan Z.Y., Ren X.Q., Sun H.M., Wang Y., Wang Y.Y., Weng H., Wei C.J., Wu D.F., Xia J., Xiong Y., Xu H.B., Yao X.M., Yuan Y.F., Ye T.S., Zhang X.C., Zhang Y.W., Zhang Y.G., Zhang H.M., Zhao Y., Zhao M.J., Zi H., Zeng X.T., Wang Y.Y., Wang X.H., for the Zhongnan Hospital of Wuhan University Novel Coronavirus Management and Research Team, Evidence-Based Medicine Chapter of China International Exchange and Promotive Association for Medical and Health Care A rapid advice guideline for the diagnosis and treatment of 2019 novel coronavirus (2019-nCoV) infected pneumonia (standard version) Mil. Med. Res. 2020;7(1):4. doi: 10.1186/s40779-020-0233-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhang L., Yan X., Fan Q., Liu H., Liu X., Liu Z., Zhang Z. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. J. Thromb. Haemost. 2020;18(6):1324–1329. doi: 10.1111/jth.14859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ji H.L., Zhao R., Matalon S., Matthay M.A. Elevated plasmin(ogen) as a common risk factor for COVID-19 susceptibility. Physiol. Rev. 2020;100(3):1065–1075. doi: 10.1152/physrev.00013.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Cancio M., Ciccocioppo R., Rocco P.R.M., Levine B.L., Bronte V., Bollard C.M., Weiss D., Boelens J.J., Hanley P.J. Emerging trends in COVID-19 treatment: learning from inflammatory conditions associated with cellular therapies. Cytotherapy. 2020;22(9):474–481. doi: 10.1016/j.jcyt.2020.04.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sakamoto A., Kawakami R., Kawai K., Gianatti A., Pellegrini D., Kutys R., Guo L., Mori M., Cornelissen A., Sato Y., Bellasi A., Faggi L., Hong C., Romero M., Guagliumi G., Virmani R., Finn A.V. ACE2 (Angiotensin-Converting Enzyme 2) and TMPRSS2 (Transmembrane Serine Protease 2) Expression and Localization of SARS-CoV-2 Infection in the Human Heart. Arterioscler Thromb Vasc Biol. 2021;41(1):542–544. doi: 10.1161/ATVBAHA.120.315229. [DOI] [PubMed] [Google Scholar]
- 87.Banu N., Panikar S.S., Leal L.R., Leal A.R. Protective role of ACE2 and its downregulation in SARS-CoV-2 infection leading to Macrophage Activation Syndrome: Therapeutic implications. Life Sci. 2020;256:117905. doi: 10.1016/j.lfs.2020.117905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Su S., Wong G., Shi W., Liu J., Lai A.C.K., Zhou J., Liu W., Bi Y., Gao G.F. Epidemiology, Genetic Recombination, and Pathogenesis of Coronaviruses. Trends Microbiol. 2016;24(6):490–502. doi: 10.1016/j.tim.2016.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Liu F., Long X., Zhang B., Zhang W., Chen X., Zhang Z. ACE2 Expression in Pancreas May Cause Pancreatic Damage After SARS-CoV-2 Infection. Clin Gastroenterol Hepatol. 2020;18(9):2128–2130 e2. doi: 10.1016/j.cgh.2020.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]