Abstract
Marine resources have tremendous potential for developing high-value biomaterials. The last decade has seen an increasing number of biomaterials that originate from marine organisms. This field is rapidly evolving. Marine biomaterials experience several periods of discovery and development ranging from coralline bone graft to polysaccharide-based biomaterials. The latter are represented by chitin and chitosan, marine-derived collagen, and composites of different organisms of marine origin. The diversity of marine natural products, their properties and applications are discussed thoroughly in the present review. These materials are easily available and possess excellent biocompatibility, biodegradability and potent bioactive characteristics. Important applications of marine biomaterials include medical applications, antimicrobial agents, drug delivery agents, anticoagulants, rehabilitation of diseases such as cardiovascular diseases, bone diseases and diabetes, as well as comestible, cosmetic and industrial applications.
Keywords: Biological properties, Biomedical applications, Marine biomaterials, Marine biopolymers, Marine organisms
Graphical abstract

Highlights
-
•
Ocean provides such an abundant of materials that humans have been using marine biomaterials since ancient times.
-
•
The efficacy in utilizing these renewable natural resources has been greatly improved because of technological revolution.
-
•
Marine organisms possess a plethora of excellent bioactive properties.
-
•
The last century has witnessed rapid development in the interdisciplinary utilization of marine biomaterials.
1. Introduction
Oceans and seas account for more than 70% of the earth's surface area [1]. Ocean ecosystem is the largest one on Earth with approximately 2.2 million different species reside in. Close to 91% of these species still await identification and exploration [2]. The ocean provides such an abundant source of materials that humans have been using marine-derived biomaterials since ancient times. Marine-derived biomaterials are, in general, devoid of human pathogens [3].
Contemporary research on marine biomaterials is currently undergoing a renaissance. The efficacy in utilizing these renewable natural resources has been greatly improved because of technological revolution [4]. Marine organisms possess a plethora of excellent bioactive properties. Many of these organisms have been utilized in biomedical applications, either directly, or in modified forms, as biomimetic templates. The first use of marine biomaterials dates back to the last century, when a substance known as holothurin was extracted from marine organisms in 1967. Holothurin is the earliest biologically-active substance of marine origin that was used experimentally as an anti-tumor agent in in vivo cancer models [5]. This discovery spurred intensive researches on marine-derived biomaterials.
Review of prior literature indicates that marine biomaterials have undergone four major stages of discovery and development. In the first stage, studies were focused on coral bone graft substitutes. Dr. Chiroff first extracted calcium carbonate exoskeleton from coral (coralline hydroxyapatite) via the replamineform process. This biomaterial resembles natural bone and possesses considerable osteoconductivity, excellent biocompatibility and osteogenic potential [[6], [7], [8], [9]]. In vivo application of this biomaterial paves the way for future investigations on ocean-derived biomaterials [10].
The focus of marine biomaterials shifted to polysaccharide-based polymers during the second stage of development. Chitin, the most abundant polysaccharide of marine origin, was identified as early as 1972 [11]. However, chitin did not receive its deserved attention until the early 21th century, when studies revealed its important biological characteristics as well as those of its derivatives. In 2007, chitin was discovered as a significant component of skeletal fibers in certain classes of marine sponges [12,13]. Subsequent identification of chitin-silicon composite by Professor Hermann Ehrlich further led to intense investigations of novel biomaterials based on chitin and its deacetylated form, chitosan [13]. These investigations identified that chitin has low-toxicity, biocompatible, biodegradable and possesses hemostatic activity and antimicrobial capability [14,15].
In the third stage, rapid advances in tissue regeneration during the last decade redirected the focus of marine biomaterials research to marine-derived collagen. The increasing risks of pathogen exposure generated by terricolous animal-derived collagen greatly stimulated exploration of new collagen alternatives. A novel porous jellyfish collagen scaffold was discovered in 2006. Further research validated the low immunogenicity, excellent biocompatibility and biodegradability of marine-derived collagens [16]. Sophisticated approaches have since been used to generate highly valuable marine collagen-based materials [17]. These inspiring advances revolutionized tissue regeneration and other industrial fields [18,19]. For instance, echinoderm-derived collagen membranes were identified as a promising material for guided tissue regeneration. These echinoderm-derived collagen membranes are thinner but stiffer compared with commercial collagen membranes [20]. Likewise, collagen derived from squids in Antarctic and sub-Antarctic areas were recognized as inspiring hybrid scaffolds for tissue regeneration [21].
The fourth developmental stage of marine-derived biomaterials involves the combination of marine-derived composites with other materials via advanced approaches such as nanotechnology and three-dimensional (3D) printing. Hybrid scaffolds that combine collagen with other modified materials possess improved mechanical and biological properties. Lyophilized scaffolds, for example, were fabricated with eel skin-derived collagen and alginate hydrogel through extrusion-based 3D printing technology. Compared with pristine alginate hydrogels that are devoid of collagen, the hybrid scaffolds exhibited enhanced metabolic and cell proliferation activities [18]. Likewise, hybrid sponge-like scaffolds have been prepared using ulvan and gelatin. The sponge-like scaffold possessed both porous and interconnected structures, with excellent bioactivity, making it a valuable biomaterial in bone tissue engineering [22]. Taken together, these studies indicate that marine-derived materials are capable of enhancing the properties of these raw materials and hold great promise for future applications.
The last century has witnessed rapid development in the interdisciplinary utilization of marine biomaterials such as regenerative medicine, antimicrobial materials and drug delivery systems. Marine biomaterials are still abundant despite industrial pollution, overfishing and unanticipated climate change. Hence, the present review aims to provide a detailed coverage of advanced marine biomaterials. The structures, characteristics, origin, extraction, processing and applications of maribe-based biomaterials will be discussed in subsequent sections. The present review should be of interest to researchers in marine biology, bioengineering, biochemistry, biomineralization and biomimetics.
2. Biomaterials from marine organisms
The marine environment encompasses a wide range of habitats with colossal microbial biodiversity. More than 25,000 biologically-active compounds have been identified from these habitats [23]. Fungi, fungi-like protists and bacteria exhibit the highest potential of producing bioactive materials. Mycosporines and mycosporine-like amino acids, carotenoids, exopolysaccharides, fatty acids and chitosan derived from these marine microorganisms represent sustainable, low-cost and fast-production alternatives to the use of other natural molecules in photo-protective, anti-aging and skin-whitening products for facial, body and hair care [24]. Extraction of polyketides, peptides and alkaloids from marine bacteria and fungi has been investigated extensively [25]. The excellent properties of these marine biomaterials are elucidated in this section.
2.1. Polysaccharides and glycans from marine organisms
Marine organisms such as shellfish, macro-algae, fungi, micro-algae and coral produce polysaccharides with variable structures and diverse biological activities that terrestrial organisms lack. This is because the marine environment is featured by high pressure, high salinity, low temperature and oxygen deficiency. The polysaccharides and glycans have been investigated extensively because of their anticoagulant [26], antimicrobial, antioxidant, antithrombotic and antitumor properties [27].
Recently, researchers have proposed that polysaccharides extracted from marine organisms are potential antiviral therapeutic agents against SARS-CoV-2, the coronavirus accounting for the ongoing COVID-19 pandemic [28]. In addition, sulfated polysaccharides such as ulvan, carrageenan and fucoidan exhibit exceptional properties that include hydrogel formation, scaffold formation, extracellular matrix-mimicking, augmentation of alkaline phosphatase activity, reinforcement of stem cell differentiation and biomineralization ability [29]. Six kinds of representative polysaccharides or glycans with excellent properties are listed below.
2.1.1. Chitin and chitosan
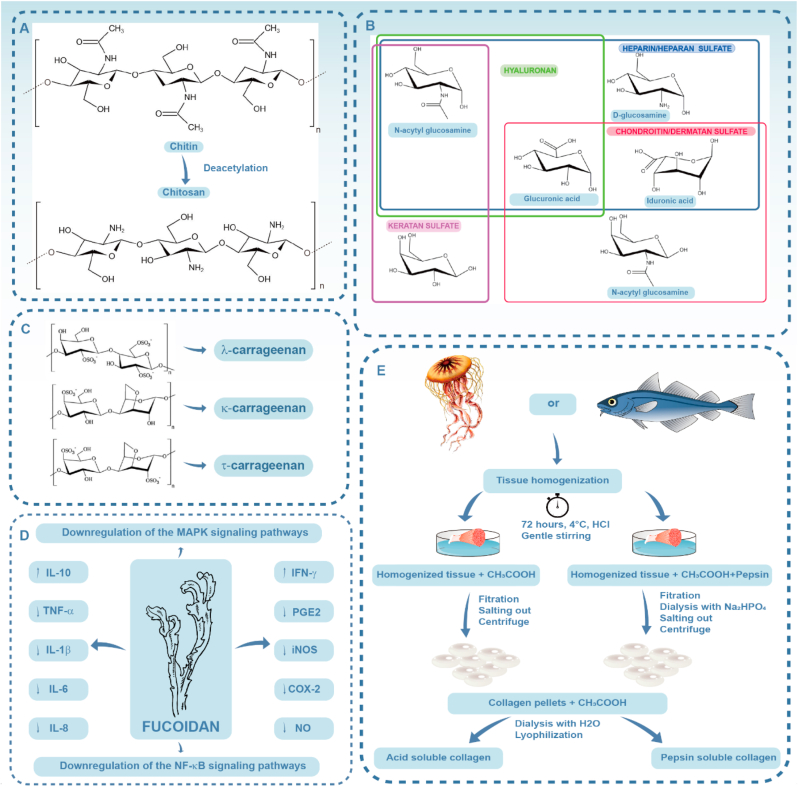
Chitin is the second most common polysaccharide on earth, after cellulose. Chitin originates from the shells of crustaceans like crabs and shrimps, and is also present in cytoderm of fungi and green algae. Chitosan is a natural amino-polysaccharide composed of (1–4)-linked 2-acetamido-2-deoxy-d-glucosamine, which is obtained through the deacetylation of chitin (Fig. 1A). Chitin and chitosan exist in nature as pure chitin, nanoorganized chitin-proteins, chitin-pigments and chitin-mineral composite biomaterials, among which pure chitin exhibits the strongest resistance to alkaline condition [30]. The deacetylation and molecular weight of chitin and chitosan have great impact on their physicochemical and biological properties including solubility, hydrophilicity and cell response [31]. Chitin-based scaffolds are also involved in calcium biomineralization and biosilica formation. These scaffolds provide the inspiration for biomimetic synthesis [32].
Fig. 1.
A) Structural formula of chitin and chitosan; B) Monosaccharides in glycosaminoglycans; C) Chemical structures of different forms of carrageenan; D) Putative anti-inflammatory mechanisms of fucoidan; E) Collagen extraction process for producing acid-soluble and pepsin-soluble marine collagen. Tissue homogenization is conducted with the aim of size reduction via physical methods and mild chemical pretreatment. Ethylenediamine tetra-acetic acid or HCl is then employed to demineralize the raw materials to facilitate extraction. Acidic solutions (e.g. CH3COOH) are subsequently used for dissolving the demineralized collagen to complete the extraction process. Pepsin is co-cultured with the mixture for eradication of non‐collagenous components. Finally, the soluble collagen is processed via lyophilization by mixing the collagen pellets, CH3COOH and H2O. (A is reproduced from Ref. [33] with permission from the publisher; B is reproduced from Ref. [34] with permission from the publisher; C is reproduced from Ref. [35] with permission from the publisher; D is reproduced from Ref. [36] with permission from the publisher; E is reproduced from Ref. [37] with permission from publisher).
Intensive research have been conducted on the antibacterial properties of chitosan and its derivatives. Compared with chitin, chitosan has more potent antibacterial ability because of its positively-charged amines. The latter interact electrostatically with the negatively-charged macromolecules on the surface of bacteria to form a thick layer around the cell wall, thereby preventing the exchange of pivotal solutes [33]. There is robust evidence that chitosan is capable of combating Streptococcus mutans, Actinomyces actinomycetemcomitans, and Porphyromonas gingivalis [38]. These bacteria are commonly identified in teeth with caries and periodontal diseases. Glass ionomer cement modified with chitosan exhibits apparent antibacterial effect against dental caries and periodontal diseases [39]. Chitin and chitosan are potential substitutes for antibiotics against drug-resistant pathogens. Another remarkable property of sponge chitin is that it can be used as a thermostable template for the preparation of inorganic-organic nanostructured composites in extreme conditions due to its excellent thermal stability up to 200 °C [40]. The electrochemical deposition of copper and copper (I) oxide nanoparticles on chitin-based scaffold provides novel insight in Extreme Biomimetics [41].
Nevertheless, the applications of chitin and chitosan are restricted by their poor solubility. The firm inter-molecular and intra-molecular hydrogen bonds in their sturdy structure is responsible for the low solubility in common solvents such as water and organic solvent [31]. Extensive efforts have been devoted to enhancing their water solubility and antimicrobial properties. For example, chitosan was functionalized via the addition of cationic and anionic groups. The introduction of cationic groups, including quaternary ammonium functionalities [42] and metal/metal oxide nanoparticles [43], apparently increased the antibacterial capability of chitosan. Reducing the positive charges of the protonated amino groups on the surface of chitosan is another method for improving its water solubility. Chitosan modified by sulfonate groups produced anionic chitosan with enhanced water solubility and selective antimicrobial activity [44].
2.1.2. Glycosaminoglycans
Glycosaminoglycans (GAGs) are sulfated polysaccharides. They are found extensively in the extracellular matrix and cell surface of animals. The basic structure of GAGs comprises a linear sequence of repetitive disaccharide units. The latter is composed of an amino sugar and uronic acid or galactose. Glycosaminoglycans derived from marine animals are different from those present in terrestrial organisms in both molecular weight and sulfation characteristics. The GAGs include chondroitin sulfate derived from shark cartilage, heparan sulfate dervied from scallops, dermatan sulfate derived from shark skin and heparin derived from shrimp heads (Fig. 1B).
Glycosaminoglycans are involved in inflammation, thrombosis, cancer, coagulation and tissue development. The sulfation patterns of GAGs, even when they are of the same class, can vary among marine animals, which are closely related to their physiological functions. Glycosaminoglycans exert their influence through interaction with other proteins indirectly (including regulating protein folding through steric hindrance, elimination) or directly (via electrostatic interaction). These interactions are greatly affected by their degree of sulfation and negative charges [45]. Holothurian fucosylated chondroitin sulfate extracted from sea cucumbers is a typical example of marine GAG [46]. Exotic disaccharide units such as CS-K are unique to marine organisms. The GAGs in some marine animals feature a special pattern of saccharide arrangement, such as a specific sequence of consecutive saccharides [34]. Marine organisms also produce GAGs mimetics with structures that are different from those derived from terrestrial sources, such as the presence of sulfated glycans. These marine organisms include macro-algae (brown, red and green algae) and invertebrates (ascidians, mollusks, crustaceans and echinoderms).
2.1.3. Alginate
Alginates are salts of alginic acid, a linear anionic polysaccharide copolymer consisting of (1–4)-linked β-mannuronic acid and α-guluronic acid monomers. Alginates form the cell walls of brown algae. They possess gelling, viscosity modification and stabilizing properties, and are used extensively as thickeners and emulsification stabilizers.
Recently, the anti-obesity capability of alginate has been investigated extensively. Alginate has been shown to inhibit digestive enzymes. These properties are not influenced by the cooking process (even when cooking at temperature up to 200 °C) and digestion. This renders alginate a potential drug for obesity management [47]. The anti-obesity effect of alginate is influenced by the conformational structure of the polysaccharide; studies have reported that the anti-obesity of enzyme-digested alginate is stronger than acid-hydrolyzed alginate [48]. In addition, blood cholesterol levels may be reduced after the administration of calcium alginate [49].
A significant feature of alginates is gelation. Hence, they may be used to fabricate highly-biocompatible hydrogels, the physical properties of which may be customized to guide 3D cell growth and differentiation. The pore size (5–200 mm) of alginate accelerates the spread of large molecules. Consequently, alginate is frequently used as bioink in 3D bioprinting for tissue regeneration. The stiffness of scaffolds synthesized by alginate as bioink in 3D bioprinting may be controlled by altering the ratio of alginate to ionic crosslinker. Such a property enables spatial regulation of mesenchymal stem cells (MSCs) in a printed scaffold [50]. The stability of alginate hydrogels may be enhanced by post-printing treatment with BaCl2 [51]. A bioactive peptide may also be covalently linked to the alginate to endow it with cell interaction properties. For example, highly efficient coupling of bioactive peptides to alginates has been achieved using periodate oxidation followed by reductive amination with non-toxic 2-picoline-borane as the reducing agent [52].
2.1.4. Carrageenan
Carrageenan, a sulfated polysaccharide with mean molecular weight over 100 kDa, contains 15–40% ester sulfate. It is a reproducible and sustainable phycocolloid obtained from red seaweeds. The basic unit of carrageenan is d-galactose and 3,6-anhydrogalactose that are linked by α-1,3- and β-1,4-glycosidic bonds. Carrageenan is classified into three categories (λ, κ, and ι) based on the ester sulfate group cotent [53] (Fig. 1C).
Carrageenan shows great commercialization potential because it is inexpensive, biocompatible and biodegradable. The hydrophilicity and high ester sulfate content make carrageenan films useful in edible food packaging, pharmacology, biomedicine and electrial applications. The overall tensile strength of carrageenan films is better than those of other bio-based films. Films produced from refined κ- or κ/ι-hybrid carrageenan possess excellent properties, including high tensile strength and low water vapor permeability [54].
The biological activities of carrageenan include anti-inflammatory, antitumor, antimicrobial, antioxidant, anti-hyperlipemic, anticoagulant and immunomodulatory properties. These bioactivities are dependent on the molecular weight, content and site of sulfate groups [55]. Carrageenan may be functionalized via chemical modifications to achieve improved properties. The antiviral activity of carrageenan against herpes simplex virus (HSV-1 and HSV-2) is enhanced by partial oxidation [56]. Fully-oxidized or partially-oxidized carrageenan at C-6 of the β-d-galactopyranose units demonstrates better anticoagulant effect [57]. The anti-proliferative and antitumor activities of carrageenan are novel and promising fields. κ-carrageenan possesses the highest cytotoxic effects. The 50% inhibitory concentration of κ-carrageenan is close to those antineoplastic with alkylating properties with official approval [55].
The limitation for the application of carrageenan films is their poor water vapor permeability and water-resistance. Different approaches have been used to improve carrageenan film properties, such as blending, plasticizing, and nanoparticle incorporation. By producing a concentrated and stable structure in blended matrix, polymerides such as starch and protein blending with carrageenan greatly improves the mechanical properties of the film. Plasticizers are alternative agents to promote the flexibility of carrageenan films. The inclusion of organic acids and inorganic materials may also improve the antimicrobial activity of these films [54,58]. Reinforcement of carrageenan polymers with homogeneously-dispersed chitin nanofibrils renders the film more smooth and flexible, along with increased tensile stress and Young's modulus [59]. Although carrageenan is considered as safe for human use, there has been concern regarding the long-term response of carrageenan in vivo. The cumulative effects of carrageenan are similar to chronic low-level inflammation in obesity and other metabolic syndromes. The controversy on the safety of carrageenan should be resolved prior to its practical applications [60].
2.1.5. Ulvan
Ulvan is a polyanionic sulfated polysaccharide consisting of glucuronic acid, iduronic acid, rhamnose-3-sulfate and a small amount of xylose. Ulvan has been used extensively in chemical, pharmaceutical, agricultural and comestible fields because of its anti-coagulation, anti-oxidation, anti-tumor, blood lipid level reduction activities as well as the capability in eliciting plant defenses. Currently, there are two chemical ulvan saccharification methods: acid hydrolysis and enzymatic hydrolysis. Enzymatic hydrolysis of polysaccharides, which is conducted under mild reaction condition, higher saccharification rate, low energy consumption and low content of aggressive chemicals, is deemed superior to the former [61].
Compared with those with high-molecular weight, low-molecule weight ulvans have better solubility, lower viscosity, easier absorption and better exposure of reactive groups [62,63]. Degradation of ulvan generates monosaccharides and oligo saccharides with unexpected biological properties, including antiviral and anticoagulant capability, as well as protein localization at the cell surface. Hence, it is promising to utilize depolymerization of monosaccharides as fertilizer to produce bioethanol and high-value food additives [61].
Ulvan shows a broad range of bioactivities. It possesses excellent antibacterial activities against pathogens such as Pasteurella multocida, Mannheimia haemolytica and Erysipelothrix rhusiopathiae. After administration, ulvan stimulates the human immune system by secreting cytokines that are involved in cell-mediated immunity via the PI3K/Akt signaling pathway [64,65]. As sulfated polysaccharides, ulvan also exhibits potent antiviral activity as well as immunomodulatory activity [63,66]. Ulvan is capable of activating RAW264.7 macrophages to produce pro-inflammatory mediators such as TNF-α, IL-6 and PGE2. These mediators are responsible for initiating immune responses. Because of the heterogeneous composition and stuctures of the polysaccharides present in ulvan, the relationship between their immunomodulating effects and structural features is obscure and requires further investigation [67]. Ulvan also demonstrates anti-tumor activity against human breast cancer, hepatocellular carcinoma and cervical cancer [68]. The antioxidant and anti-hyperlipidemic activities of ulvan has also been reported [69]. Ulvan also shows affinity to heavy metal ions. Hydrogels prepared with gelatin and ulvan are potential adsorbents of heavy metals and the dye methylene blue. These hydrogels are also promising alternatives as anti-pollution agents [70].
2.1.6. Fucoidan
Fucoidans are obtained from the cell walls of brown algae, seagrasses and other marine invertebrates. They are composed of sulfated polysaccharides, including repeating α-(1 → 3)-linked l-fucopyranose residues or alternating α-(1 → 3)- and α-(1 → 4)-linked l-fucopyranoses. Other monosaccharides such as xylose, galactose and uronic acids may also combine with the fucosyl main chain to form fucoidan. The specific structure of fucoidans is complicated and variable, depending on the source and extraction process [36,71,72].
Fucoidans possess biomedically-significant pharmacological properties including anti-inflammatory, antimicrobial, immunomodulatory and antitumor properties. For anti-inflammatory activity, fucoidans are able to block lymphocyte adhesion and invasion, inhibit multiple enzymes, induce apoptosis and regulate excretion of pro-inflammatory cytokines (Fig. 1D) [36]. Moreover, fucoidan initiates a variety of immunoregulatory effects. It activates dendritic cells, natural killer cells and T cells to enhance antiviral and anti-tumor responses [73]. For example, fucoidan derived from the algae Nizamuddinia zanardinii exhibits well-defined immune-enhancing activity on murine macrophage cell and human natural killer cells. In this in vitro model, fucoidan contributes to the secretion of cytokines, including nitrous oxide, TNF-α, IL-1β, IL-6, NF-κB and stimulates the MAPK signaling pathways [74].
The immunomodulatory activity of fucoidans is regulated by different parameters. The content of fucose and sulfate groups in fucoidans is considered as one of the most significant parameters. For example, fucoidan extracted from Macrocystis pyrifera has higher uronic acid content and is the most effective in activating natural killer cells, dendritic cells and T cells, producing the strongest adjuvant effect [73]. Molecular weight is another essential factor that determines the biological activity of fucoidans. High molecular-weight fucoidan (130 kDa) increases the survival rate of cells and secretion of IFN-γ and nitrous oxide, while low-molecular-weight fucoidan (30 kDa) with similar composition has lower activity [75,76].
Fucoidan are also considered effective agents for combating cancer. This is achieved via the regulation of cancer cell apoptosis. The ratio of monosaccharides, the content of sulfate groups and binding of sugar residues all account for the anti-tumor activity of fucoidans [77]. Fucoidan is a reactive oxygen species (ROS) scavenger. It reduces ROS accumulation by amyloid-β-reduction [78]. The antioxidant ability of fucoidans is determined by their concentration, molecular weight, sulfate content, extent of polymerization, presence of substitution groups, the position and type of sugar and glycosylation branching [71,79]. Fucoidan enhances angiogenesis in a dose-dependent manner by inflencing tube formation and movement of human microvascular endothelial cells [80]. The angiogenesis potential of fucoidan is attributed to the activation of p38 and C-Jun N-terminal kinase via affecting the AKT/MMP-2 signaling axis [81].
2.2. Proteins and peptides from marine organisms
2.2.1. Collagen
Collagen is the most abundant extracellular matrix protein [82]. It is commonly found in tendons, skin, bones and blood vessel walls. To date, researchers have recognized and categorized 29 types of collagen, among which type I collagen makes up 90% of the collagen present in the animal body [83]. Collagen possesses complex supramolecular structure. A collagen molecule is around 2–6 nm thick, 30–50 nm wide and 60–100 nm long [84]. The collagen molecule consists of three characteristic α‐chains, which form a triple‐helical α‐domain(s) known as “tropocollagen” [85]. Repetition of the Gly-X-Y triplet constitutes the α chain, in which proline and hydroxyproline constitute the X and Y domains [86]. Self-assemblly of the tropocollagen molecules is responsible for maintaining the structural and biological integrity of a collagen fibril on a molecular scale. The collagen fibril contains well-defined protein binding sites for cell interactions [89].
The collagen content in the human body decreases due to aging [87], infection [88] and exposure to ultraviolet light [89]. The loss of collagen results in disorders such as skin mismanagement and stiffened joints. Consequently, afforable and easily extractable collagen sources are highly sought after especially in the fields of regenerative medicine.
Type I collagen originates mainly from animals [90]. Animal-derived collagen, which serves as the gold standard for collagen application, is obtained from donated human tissues or large mammals such as cattle, horses, pigs or sheep [91]. However, the availability of land-derived materials is hampered by the outbreak and pathophoresis of zoonotic infectious diseases. Extraction of collagen from marine organisms is now regarded as the safest approach for large-scale collagen acquisition [92]. Marine collagen is usually extracted from sponges [[93], [94], [95]], fishes [96,97], squids [21] and echinoderms [33]. These collagen sources may be categorized into those that are derived from invertebrate animals and those that are extracted from vertebrate sources [37] (Table 1). Marine vertebrate collagen has always been identified as alternative biomaterials. However, similar to land-derived collagen, the potential risks arising from bovine spongiform encephalopathy and the swine influenza crisis greatly hampered its further application [98].
Table 1.
Collagen derived from marine organisms.
| Genre | Source | Scientific Name | Collagen Type | Activity | Refs |
|---|---|---|---|---|---|
| Invertebrate organisms | Jellyfish | Catostylus tagi | Collagen V/XI | Healthy food. | [113] |
| Jellyfish | Rhopilema esculentum | Collagen I | Enhance cell migration, heal wound, hemostatic properties, modulate chondrogenic differentiation of human mesenchymal stem cells and cell culture substrate. | [114] [115] [116] [117] |
|
| Jellyfish | Rhizostoma pulmo | Collagen I | Trigger similar responses in cell adhesion, proliferation or migration | [118] | |
| Jellyfish | Catostylus mosaicus | Collagen I | Support cell attachment and proliferation | [119] | |
| Sea urchin | Paracentrotus lividus | Collagen I | Skin regenerative medicine and collagenous extracellular matrix biomaterials for tissue engineering | [120] [121] |
|
| Squid | Peru squid | Collagen II | Degenerative osteoarthritis-relieving effects | [122] | |
| Persian Gulf squid skin | Uroteuthis duvauceli | Collagen I | Anti-cytotoxic properties | [123] | |
| Starfish | Asterias amurensis | Collagen I | Production of two-dimensional membranes | [124] | |
| Vertebrate organisms | Cat Fish | Ictalurus punctatus | Collagen I | Food, medical and cosmetic industries, produce high-value-added product | [125] [126] |
| Cat Fish | Pangasius sp. | Collagen I | Substitutes for commercial collagen | [127] | |
| Sponges | Chondrosia reniformis | Collagen IV | ROS scavenging capability; photo-protective activivy as well as wound-healing characteristic | [128] | |
| Blue Shark | Prionace glauca | Collagen I | Promote osteoblast proliferation rates | [129] | |
| Blue Shark | Prionace glauca | Collagen II | Antioxidant activity and increase apoptotic gene expression | [130] | |
| Salmon |
Salmo salar Tilapia nilotica |
Collagen I | Accelerate wound healing by altering cutaneous microbiome colonization | [131] | |
| Yellowfin tuna | Thunnus albacares | Collagen I | Antioxidant activities | [132] | |
| Sardine | Sardinella longiceps | Collagen I | Mosquito larvae against Aedes aegypti | [133] | |
| Salmon | N/A | Collagen I | Protect DNA against oxidation-induced damage | [134] | |
| Medusa fish | Centrolophus niger | Collagen I | Antibacterial active food and packaging film | [135] |
Marine-derived collagen is a remarkable source of raw biomaterials for multidisciplinary applications [99]. This is because collagen extracted from marine sources has an amino acid composition that is similar to human type I collagen [100]. In addition, owing to their low molecular weight and small particle size, marine-derived collagen circulates rapidly in the blood stream and is absorbed into human body up to 1.5 times faster than conventional collagen derived from terricolous sources [91,101]. Most importantly, marine-derived collagen is available in high yield and at low cost [102]. The principal procedures involved in the isolation of marine-derived collagen are illustrated in Fig. 1E [101]. Vertebrate marine animal-derived collagen, such as fish collagen, is one of the most important sources of marine-derived collagen. These extracted pepsin-soluble and acid-soluble collagen moieties are capable of accelerating wound healing in vivo [103]. The principal procedures involved in the isolation of fish-derived collagen include preparation, extraction and recovery [101]. The preparation process includes cleaning and size reduction via physical methods and mild chemical pretreatment for the eradication of noncollagenous components. Ethylenediamine tetra-acetic acid or HCl is then employed to demineralize the raw materials to facilitate extraction. Acidic solutions are subsequently used for dissolving the demineralized collagen to complete the extraction process [104]. Collagen extracted from marine invertebrate, especially those extracted from glass sponges, are excellent organic templates for in vitro silicification [105]. Marine glass sponge-derived collagen accounts for the most abundant naturally-silicified biomaterials on earth. Heavy interfibrillar silicification and structural hierarchy were detected when silica precursors were mixed with these collagen moieties. The silicified collagen possesses considerable toughness, stiffness as well as resilience. Owing to its superior biocompatible and mechanical properties, silicified marine-derived collagen has generated intense interest in hard tissue regeneration research [106].
Among the invertebrate-derived collagen, there is a special sponge-derived collagen known as spongin. Spogin displays distinct morphological traits and chemical constitution. Spongin fibers are large microstructures which are identified as halogenated scleroproteins or neurokeratin-like proteins [107]. There are two categories of spongin, spongin A and spongin B. Spongin A is the fine intercellular collagen fibrils which can be detected only with electron microscopy. Spongin B corresponds to the typical macroscopical and rigid fibrils present keratosan demosponges [108]. Spongin had a long scientific research record dated as far back as 1705 [109]. Spongin chemistry is considerably complex. Halogens (I, Br) and other unknown biological constituents account for the water-insoluble characteristic of spongin [110]. In addition, spongin possesses excellent resistence to harsh chemical substances and extremely thermal conditions. What is even more inspiring is that spongin can be fabricated as nanoscale biomaterials which maintain their excellent properties at extreme conditions [111]. Because of the aforementioned advantages, spongin is considered an attractive macroscopical biomaterials in contemporary bioinspired materials science and regenerative medicine [112].
There are problems associated with the application of marine-derived collagen. For example, mackerel collagen is an important pan-allergen in the Japanese population [136]. Marine-derived collagen denatures easily, which limits its biomedical applications. Collagen gels derived from chum salmon (Oncorhynchus keta), for example, denatures at 19 °C [37]. Religious constraints are another factor that hampers the marketing of marine collagen-based products.
2.2.2. Gelatin
Gelatin is a heterogeneous mixture of peptides. It is extracted from fishes and sponges [100,137,138], jellyfish [139], squids [140] and snails [141] via enzymatic or acidic process [142]. Gelatin is produced by destruction of the tertiary, secondary and even primary structures of collagen [143]. Marine gelatins possess high protein values (91.5% for acid-soluble gelatin and 83.1% for pepsin-soluble gelatin). The hydroxyproline content of marine gelatin is similar to that of commercial mammalian gelatin but with higher melting temperatures [144]. Some marine gelatin such as those derived from saithe (Pollachius virens) skin has a considerable mineral content (>20%) compared to the commercially available gelatin (<0.2%) [145]. Gelatin has been used extensively in clinical platforms such as eutherapeutic plasma expanders, nanoparticles for gene delivery and drug loading [[146], [147], [148]].
2.2.3. Marine enzymes
Enzymes obtained from marine sources, including proteases, peroxidases, oxidoreductases, hydrolases, transferases, isomerases, ligases and lyases, have found comestic, industrial and medical applications. These enzymes are capable of controlling immune functions, some of which directly kill bacteria. They possess considerable immunomodulatory activities that enhance the innate immune response; they also control infection of the host effectively [149] (Table 2).
Table 2.
Marine-derived enzymes.
| Name of enzymes | Sources | Enzyme type | Activity | ref |
|---|---|---|---|---|
| l-asparaginase |
Bacterial sources E. coli Erwinia carotovora) |
Asparaginase | Convert l-asparagine into aspartic acid and ammonia Anti-carcinogenic activity Reduce carcinogenic acrylamide |
[150] [152] [157] |
| Fungal sources Aspergillus oryzae | ||||
|
Microalgae sources Spirulina maxima Chlamydomonas sp | ||||
| Carbonic anhydrase containg cadmium | Marine diatom Thalassiosira weissflogii | Metalloenzymes | Catalyze the reversible hydration of CO2 Acquire inorganic carbon for photosynthesis |
[158] [159] |
| Alkaline protease |
marine Psychrobacter Aureobasidium pullulan Bacillus mojavensis A21 Pseudoalteromonas sp |
Protease | Peptide hydrolases | [160] [161] |
| Extracellular phospholipase C |
marine streptomycete Pseudoalteromonas sp |
Lipases | Hydrolyze different oils | [162] [163] |
| Chitinase Chitosanase |
Vibrio fluvialis, Vibrio parahaemolyticus, Vibrio mimicus Vibrio alginolyticus Listonella anguillarum Aeromonas hydrophila Aspergillus griseoaurantiacus KX010988. |
Hydrolase | Degrade chitin and chitosan | [[164], [165], [166]] |
| Alginate lyases |
Algae marine invertebrates Vibrio splendidus 12B01 |
Lyases | Degrade alginate | [[167], [168], [169]] |
| Agarases |
Cytophaga Bacillus Vibrio Alteromonas Pseudoalteromonas Streptomyces Vibrio sp. F-6 |
Hydrolase | Hydrolyze agar | [170,171] |
| Carrageenases |
Pseudomonas, Cytophaga, Alteromonas atlantica, Wingyingzhuangia aestuarii OF219 Flavobacterium sp. YS-80-122 |
Hydrolase | Degrade carrageenans | [[172], [173], [174], [175]] |
| Cellulose and hemicellulose hydrolase |
Cytophaga, Cellulomonas, Vibrio, Clostridium, Streptomyces Fusarium, Chaetomium, Phoma, Sporotrichum, Glaciecola sp. strain 4H-3–7+YE-5 |
Hydrolase | Break down cellulose | [[176], [177], [178]] |
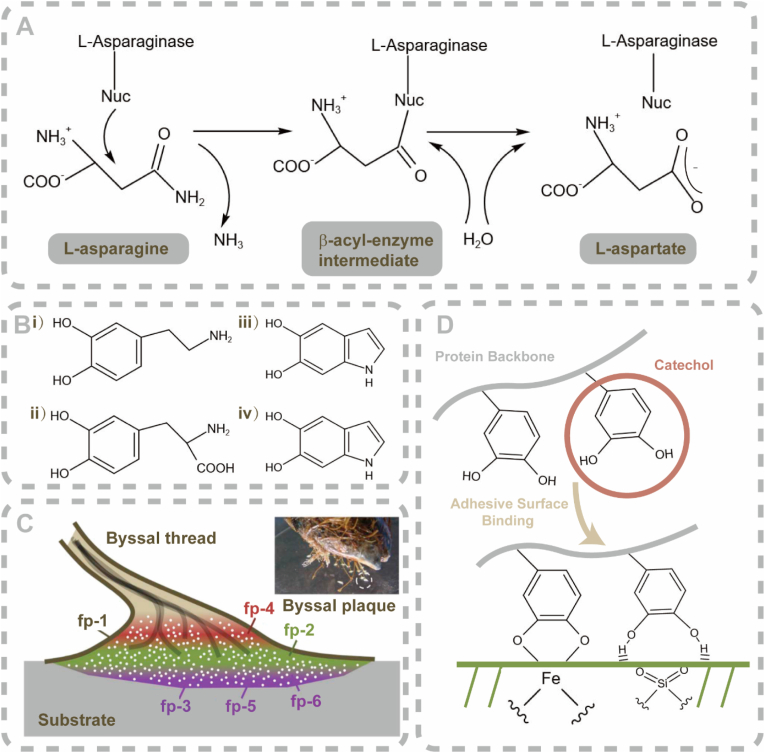
Some marine-derived enzymes show great potential in anti-cancer applications. l-asparaginase, an enzyme which catalyzes the hydrolysis of l-asparagine to l-aspartic acid, is an important enzyme of marine origin [150] (Fig. 2A). l-asparaginase is a homologous mixture of different conformations, of which the tetramer is the predominant conformation [151]. Because of its anti-neoplastic and chemotherapeutic potential, l-asparaginase formulations occupy up to one-third of the global demand on anti-leukemia/anti-lymphoma agents [152].
Fig. 2.
Marine enzymes and protein-based bioadhesives. A) Schematic of the reaction mechanism of l-asparaginase. B) Monomeric units of natural-derived eumelanin and polydopamine melanin (PDAM). i) dopamine (DA); and ii) DOPA, DHI; iii) DHICA; and iv) the porphyrin-like tetramer. C) Schematic of MAPs and their functional location in a byssal plaque. D) Schematic of the adhesive mechanism of catechols. As a DOPA analog, catechols are considered significant groups for wet adhesion because they possess strong bidentate binding ability toward mineral oxide surfaces. (A is reproduced from Ref. [153] with permission from the publisher; B is reproduced from Ref. [154] with permission from publisher; C is reproduced from Ref. [155] with permission from the publisher; D is reproduced from Ref. [156] with permission from the publisher).
Cadmium-containing carbonic anhydrase obtained from Thalassiosira weissflogii (a marine diatom) has gained recent attention. The cadmium present in this enzyme occupies the metal-binding part of the enzyme carbonic anhydrase and plays a significant role in the acquisition of inorganic carbon for photosynthesis [179]. A protein extracted from T. weissflogii with cadmium inside possesses carbonic anhydrase activity [180]. This metal-loading enzyme participates in the activities of marine organisms and facilitates trace-metal cycling in the oceans.
The last two decades have witnessed escalating attention in the identification of enzymatic hydrolysates from marine invertebrates. Enzymatic hydrolysates, also known as antioxidant peptides, are extracted and purified from mussels [181], clams [182] and oysters [183]. Enzymatic hydrolysates scavenge free radicals and chelate metal ions, suppress lipid peroxidation and protect cells against ROS-induced toxicity [184,185].
A wide range of marine-derived enzymes possess antimicrobial, anti-cancer, antioxidant, antihypertensive as well as immunomodulatory properties. The functions of these enzymes are usually intercrossed. A lot of work needs to be done before these attractive enzymes can be harnessed for clinical use.
2.2.4. Protein-based bioadhesives
The demand for bioadhesives has spurred remarkable advances in the identification of their characteristics. Many marine organisms have the ability to adhere to substrates for survival in dynamic environments with continuous mechanical stresses in the ocean. Medical grade bioadhesives are in huge demand because moisture in the human body serves as an interfacial contaminant, weakening the boundary layer at the bonding site. Stable and instant adhesion is challenging to be achieved for wet surfaces within body tissues [186].
Mussels synthesize mussel adhesive proteins (MAPs). These proteins are secreted by mussel byssuses. Byssus is a bundle of filaments that enable these bivalve mollusks to anchor their bodies efficiently to solid surfaces such as rocks and ship hulls [155] (Fig. 2C). The MAPs possess excellent adhesive capability because they bond to varous surfaces in aqueous conditions. The distinct amino acid 3,4-dihydroxyphenylalanine (DOPA) [162], which can be easily conjugated with other components [154], determines the superior adhesive capability of MAPs. To date, MAPs that have been identifed range from proteins type 1 (fp-1) to type 6 (fp-6). Each of these MAPs distributes at a specific site and plays a distinctive role in attachment. The type-3 fast variant (fp-3F) and the type-5 (fp-5) variant are significant compositions for mussel adhesive initiation. They possess primer-like activity that facilitates firm adhesion to wet marine substrata [187].
Another attractive bioadhesive protein, eumelanin, is extracted from cuttlefish (Sepia officinalis) ink (Fig. 2B). Eumelanins are DOPA-based, melanin-like heterogeneous polymers that contain aryl–aryl covalent bonds between uncycled and cycled DOPA and their analogs [171]. Eumelanins have been investigated as potential sealing and hemostatic biomaterials [188]. Catechols, derivatives of DOPA in adhesive mussel foot proteins, have recently been used to synthesize mussel-inspired adhesives. The relevant adhesive mechanism is illustrated in Fig. 2D.
Other protein-based adhesives that originate from marine invertebrates have also been envisaged as potential candidates for medical and industrial applications. The tubeworm Phragmatopoma californica assemble their tubes by gluing siliceous and calcareous particles together with a polyelectrolytic bio-cement [189]. The bio-cement is composed of several highly-repetitive and oppositely-charged proteins, DOPA, sulfated polysaccharides and metal ions such as magnesium and calcium ions [190]. Adhesive proteins derived from sandcastle worms serve as in-situ embolic coacervate agents for treating vascular malformations and defects in a rabbit renal artery model [191].
2.2.5. Oxazole-containing peptides
Oxazole-containing peptides belong to a significant category of 5-membered N,O-heterocyclic compounds. The five-membered aromatic ring binds readily to enzymes and receptors via non-covalent interactions [183]. Oxazoles extracted from marine organisms range from short linear peptides to long complex structures [192]. They can be produced via ribosomal, non-ribosomal approaches or by synthetic pathways [[193], [194], [195]]. Oxazole-containing peptides derived from marine organisms possess antibacterial [196], antiviral [197], anti-malarial [198] and anti-algal properties [199].
2.3. Marine secondary metabolites
The ocean is a promising source of structurally-novel and pharmaceutically-active marine secondary metabolites. These metabolites include alkaloids, polyketides, terpenes, lignans, steroids and cyclic peptides [200]. The bioactivity of secondary metabolites derived from marine fungi has long been recognized. However, these secondary metabolites have not yet been commercialized for pharmaceutical applications [201].
2.3.1. Mycosporines and mycosporine-like amino acids (MMAs)
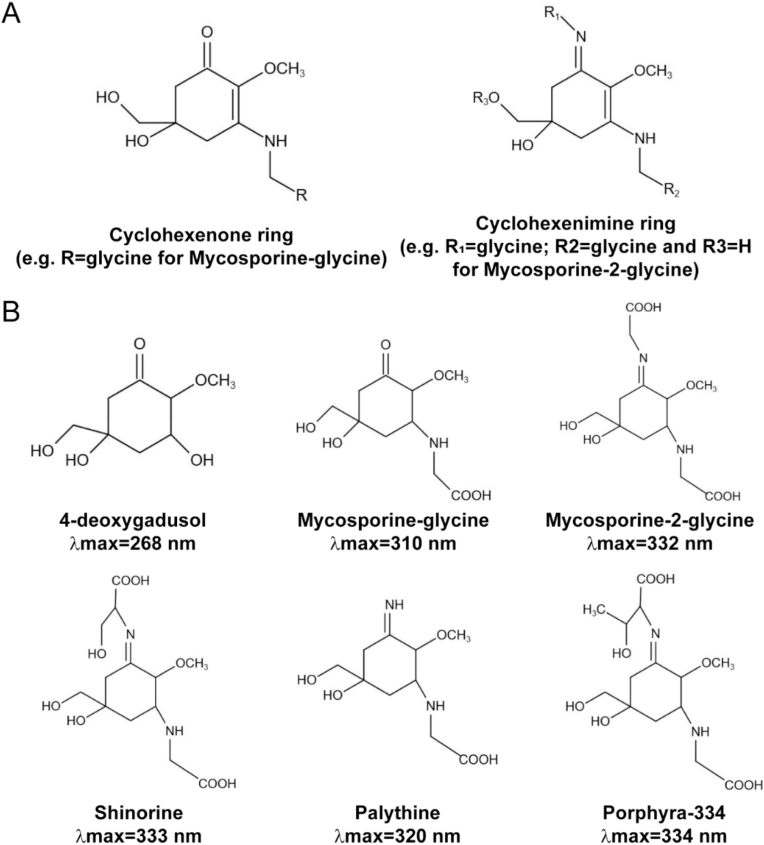
Mycosporines and MMAs are water-soluble molecules that absorb ultraviolet-A and ultraviolet-B light (Fig. 3A and B). Their absorption maxima range from 268 to 362 nm, which is determined by their molecular structure [202]. Many MMAs have been identified from red algae, sea stars, corals, dinoflagellates and cyanobacteria after the initial discovery of the mycosporines porphyra-334 and shinorine. Porphya-334 and shinorine are potent monoamine oxidase inhibitors. They are present in high concentrations in Aphanizomenon flos-aquae (a species of cyanobacteria) and Klamath blue-green microalgae [203].
Fig. 3.
Chemical structures of marine secondary metabolites. (A) The core of mycosporine-like amino acids (MAAs) is composed of a cyclohexenone, or a cyclohexenimine ring conjugated to an amino acid residue or its imino alcohol; (B) MAA precursor 4-deoxygadusol and common MAAs. The latter include mycosporine-glycine, mycosporine-2-glycine, shinorine, palythine and porphyra-334. The maximum absorbance values of these molecules are included. (A, B are reproduced from Ref. [222] with permission from the publisher).
Mycosporine-like amino acids are synthesized through the conjugation of amino acids to an intermedium via a common metabolic pathway. They are regarded as natural sunscreen molecules [204]. Hence, MAAs are promising natural alternatives for both direct (ultraviolet light absorption) and indirect (antioxidant) protection because they show strong photo-stability with absence of cytotoxicity [205]. Mycosporine-like amino acids have been incorporated into a sunscreen reagent, Helioguard®365, which contains porphyra-334 and shinorine [206].
2.3.2. Bromophenols
Bromophenols are secondary metabolites present in marine organisms. They contain at least one benzene ring with several hydroxyl and bromine groups (Fig. 5C). Bromophenols are biosynthesized via bromoperoxidases, bromase, laccase, hydrogen peroxide and bromide. The majority of bromophenols originate from marine red algae, green algae and brown algae [207,208]. Bromophenols extracted from the marine algae Leathesia nana and Rhodomela confervoides efficiently suppress the growth of human cancer cell lines ex vivo and inhibit tumor angiogenesis [209]. It has been shown that bromophenols initiate cell cycle arrest at the G0/G1 phase of adenocarcinomic human alveolar basal epithelial (A549) cells and trigger ROS-mediated apoptosis in those cells [210].
Fig. 5.
(A) Images of the scleractinian coral Acropora digitifera. (B) SEM images of ultrastructural features of the coral Balanophyllia europaea. (C) Sectional view of calcium carbonate spines derived from the sea urchin Heterocentrotus mammillatus. (D) SEM images of the inner structures of sea urchin spines (A,B are reproduced from Ref. [227] with permission from the publisher; C,D are reproduced [226] from with permission from the publisher).
Bromophenols may be used to control diabetes and obesity via the inhibition of α-glucosidase, protein tyrosine phosphatase 1B [211], α-amylase [212] and aldose reductase [213]. Bromophenols extracted from a marine red alga Symphyocladia latiuscula showed antifungal activities against Candida albicans [214].
Bromotyrosines refer to a series of bioactive secondary metabolites that originate from marine demosponges, especially from Verongida [215]. These demosponges possess two important structures: a rigid three dimensional skeleton and a broad range of secondary metabolites. These two armomentaria enable demosponges to successfully survive natural selection. Demosponges obtain nutrients by filtering water via their flexible chitinous skeletons. However, different pathogens enter the sponge body along with the nutrients simultaneously. These pathogens must be neutralized or eradicated to prevent invasive infection [216]. Secondary metabolites, mainly bromotyrosines, play significant roles in combating these pathogens. Bromotyrosines possess simple brominated tyrosine and tyramine units [217,218]. They are obtained from extraction from sponges or analog synthesis. Bromotyrosines are released when the sponges are mechanically damaged or when there are changes in the marine environment [216]. According to the latest research, the yield of bromotyrosines extracted from cultivated sponges is as high as 90% [219]. More recently, synthesis of bromotyrosines analogs has become an alternative for the extraction of naturally-occurring bromotyrosines [220]. However, many organic solvents may be involved in this multistep procedure, which will be more expensive, laborious and complex [221]. Related applications of bromotyrosines will be discussed in subsequent sections.
2.4. Hierarchical structure of marine skeletons from a macroscopical perspective
Marine skeletons are characterized by tailored architectures with distinct traits such as interconnected pores that resemble natural bone architecture. Compared with synthetic materials, marine skeletons have excellent biological and structural properties. There is an ongoing trend in tissue engineering to utilize marine skeletons with distinct structure and chemical composition as scaffolds [231] (Fig. 4).
Fig. 4.
Marine skeleton with hierarchical and porous architecture applied for tissue engineering and drug delivery system, including sea urchin, cuttlebone, coral with interconnected porous structures and seashells with dense lamellar structure (reproduced from Ref. [223] with permission from the publisher).
2.4.1. Sponges
It has been estimated that half of the marine-derived biomaterials are obtained from different species of marine sponges [223]. Sponge skeletons are soft-bodied organic and inorganic compounds with an interconnected porous architecture. The porous architecture facilitates the flow of fluids. Consequently, marine sponges are reliable sources of bone substitutes for bone tissue engineering. For example, the stony sponge Petrosia ficidormis has a highly porous (pore size 364 μm in average), interconnected 3D architecture, which is beneficial for the integration between grafting material and bone [224].
Sponge spicules are composed of amorphous silica produced under natural physiological conditions. Polyphosphate is an important component of spicules. The combination of biosilica and polyphosphate stimulates differentiation of human MSCs and increase their mineralization potential. In addition, these sponge spicules account for the intrinsic stiffness and toughness of the sponge skeleton [225]. Nanoscopical silica spheres are aligned in microscopic concentric rings and surrounded by an organic matrix to produce laminated spicules. These spicules are arranged into bundles, forming a macroscopical cylindrical lattice-like structure. The hierarchical arrangement of these spicules contributes to the mechanical strength of the sponge skeleton [223].
2.4.2. Corals
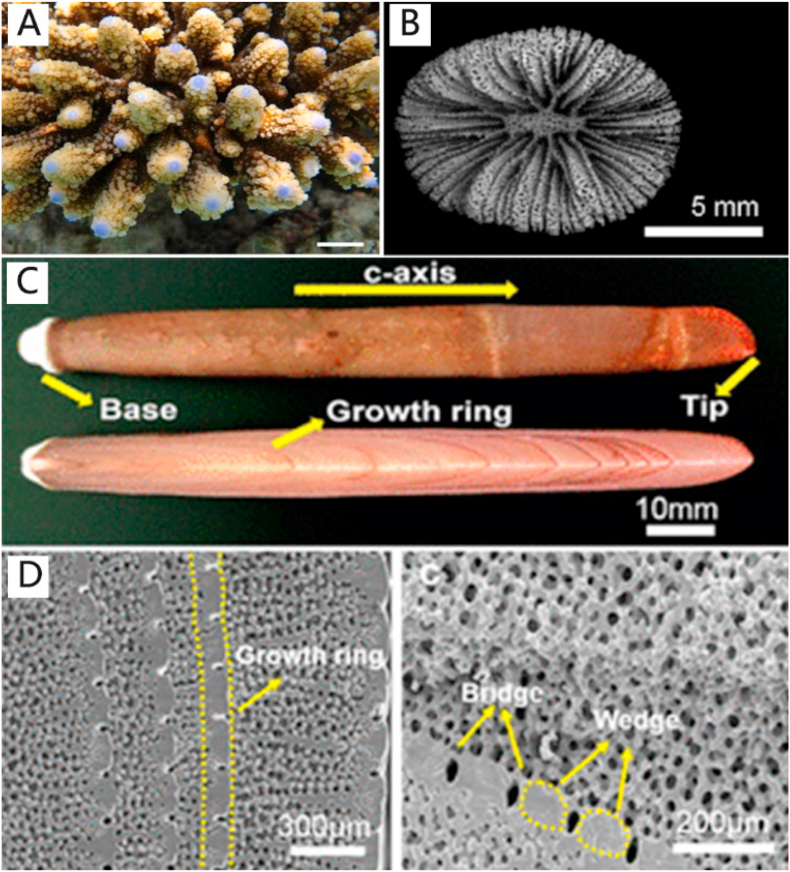
Coral skeletons are composed of a hierarchical arrangement of a calcium carbonate mineral phase known as aragonite with interconnected pores. The pores are tubular and slit-like, with a diverse range of pore size distribution that resembles the porosity and pore sizes of natural bone. The pores and channel framework are suitable for cell colonization and invasion of blood vessels. Some coral genera possess similar structure and physical properties to bone, such as Lobophylia, Acropora and Porites. Clinical applications of coral are restricted to those below 100 μm diameter pores and >20% pore volume [226]. Although coral skeletons have elegant microstructure, their mechanical properties are unfit for application as clinical bone repair scaffolds (Fig. 5A and B).
2.4.3. Sea urchins
Sea urchin skeletons have hierarchical porous structures. They have superior mechanical properties compared with corals and marine sponges. In particular, the hierarchical open-cell structure of Heterocentrotus mammillatus resembes human trabecular bone and is applicable for machinery processing because of its excellent mechanical property (compressive strength ~43 MPa) [228]. Another advantage in using sea urchin skeletons for bone tissue engineering is their interconnective porous structure, which facilitates cell penetration and vascular ingrowth [224] (Fig. 5C and D).
2.4.4. Molluscan shells
Molluscan shells are characterized by specific mineral-organic structures. Accordingly, they have been extensively used as a classic system model to study biomineralization mechanisms [229]. For example, organic scaffolds named conchixes were isolated from molluscan shells. The shells were demineralized gently by a EDTA-based method to acquire mineral-free biomaterials. Intriguingly, conchixes completely repeat the size and shapes of the shells. In the future, molluscan shells may have broad applications in biomineralization [230].
2.4.5. Siliceous skeletal structures
Silicon exists in nature as silicic acid. When concentrations are above 1–2 mM and pH is neutral, siloxane (Si-O-Si) bonds are formed between orthosilicic acid ([Si(OH)4]) through a series of polycondensation reactions, resulting in the formation of silicon. Biosilica consists of glassy amorphous silica [231]. Ehrlich et al. reviewed the diversity and potential applications of biosilicon in detail. Biosilification is an ancient and fundamental process that involves incorporation of inorganic silicon into an organic matrix. Many unicellular and multicellular marine organisms produce minerals through this biological process. A variety of biosilicon structures are formed in sponges, diatoms, mollusks, crustaceans, algae, bacteria and viruses. The siliceous skeletons produced by marine organisms, especially sponges, have attracted the attention of scientists involved in the study of biomimtics and biomedicine [232]. Biosilica skeletons and natural bone structures are both formed through biomineralization and they are quite resemblant to each other. Thus, biosilificified organic matrices for foreseeable applications in tissue regeneration. Diataom biosilica is a composite material. Apart from silica, the diatom skeleton contains other elements such as zwitterionic proteins and long‐chain polyamines. The cell walls of diatoms (diatom frustules) possess optical and highly stable properties [233]. The spicules of demosponges are silica-based with triaxonic and hexactinic symmetry. Because of these repetitive organized structures, hierarchically arranged biosilica skeletons up to 2 m have been identified in selected hexactinellid sponge species [3].
Other hierarchical structures of marine organisms have also been reported. For example, cuttlebone, the endoskeleton of the mollusk Sepia officinalis, has an interconnected porous structure with 80% porosity and pore size between 200 and 500 μm [234]. A simple enzymatic and hydrogen peroxide-based treatment has been proposed to produce nanostructurally-organized sea dollar spines [235]. Because of their porosity, microstructure and biocompatibility, marine skeletons have great potential in bone tissue engineering.
2.5. Structural coloration
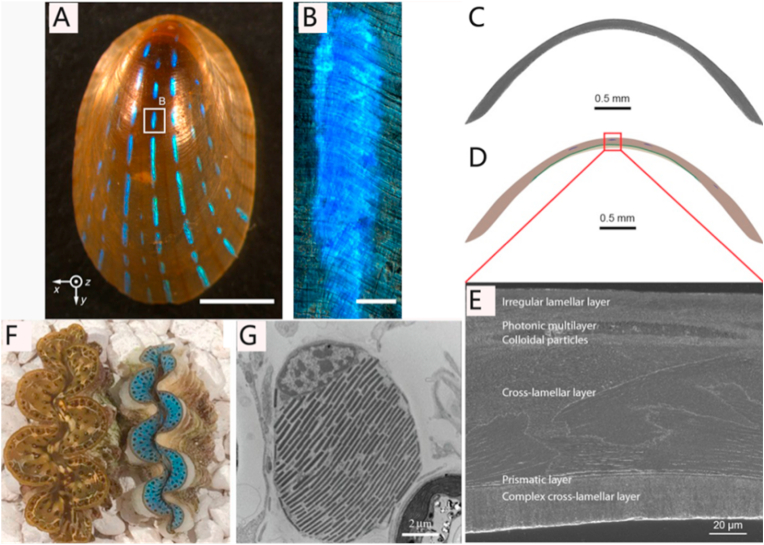
Marine organisms exhibit diverse colors because of the presence of colored molecules that absorb light (i.e. chlorophyll), luminescent compound (i.e. luciferin), as well as nano-structural and brightly-reflective cells. Marine organisms possess optical properties that are responsible for their colors. The bio-photonic structure in marine organisms consists of highly-ordered organic materials such as collagen and chitin [236]. For example, the blue iridescent stripes on the shell of the limpet Patella pellucida is produced by a microscopic mineralized multilayer with regular lamella thickness and gap spacings, which account for the light interference (Fig. 6A-E). Frustules of diatoms can also reflect ultraviolet light via their nanostructured silica [237].
Fig. 6.
(A) Optical image of a Patella pellucida shell that reflects light. (B) Optical image of reflection from a single stripe. (C,D,E) Types of microstructures observed in the shell of blue-rayed limpet. (F,G) Macroscopical and microscopical views of Tridacna maxima and T. derasa. (F) Top view of two T. maxima approximately 4–5 cm in length. (G) Transmission electron micrograph of a T. derasa iridocyte in cross section. (A,B,C,D,E are reproduced from Ref. [236] with permission from the publisher; F,G are reproduced from Ref. [238] with permission from the publisher).
Apart from their integral macrostructure, photosynthetic cells also account for the brilliant colors of many marine organisms. Iridocytes, cells with optical behavior that are found in the epithelia of giant clams, contribute to the brilliant iridescent blue, green and gold colors of these molluscs. This phenomenon is attributed to the presence of Bragg lamellae (intracellular reflective structures) inside the iridocytes. The Bragg lamellae consist of repetitive layers with high and low refractive indices. Their unique light scattering phenomenon opens new vistas in the design of solar-transductive, optical and other photo-transductive devices [238] (Fig. 6F and G). The color in the alga Cystoseira tamariscifolia is produced by opal-like photonic crystals that are present within its photosynthetic cells [239].
Multilayers of guanine crystals are the basis of highly effiective reflectors for silvery fish. They are isolated from the fish scales and exhibit anisotropic light scattering properties, which could be controlled by magnetic field [240].
3. Medical applications
3.1. Anti-cancer activity
Cancer is a global medical concern. Malignant tumors threaten 8.8 million lives every year and are responsible for 1 out of 6 deaths globally. Clinical scientists have long been looking for less toxic and more efficacious approaches to combat cancer. Natural compounds, especially marine-derived materials, have immense potential in treating cancers because of their low cost and availability. It has been almost 50 years since the approval of cytarabine (aka. cytosine arabinoside), a marine-derived chemotherapeutic medication used to treat different forms of leukemia and non-Hodgkin's lymphoma. There are currently four approved marine-derived anti-cancer drugs. In addition, eighteen compounds are in various phases of clinical trials, six of which are already in the final stage of development [241].
3.1.1. Anti-cancer mechanism of marine-derived materials
Apoptosis downregulation of cancer cells is the principal obstacle encountered in cancer suppression. Accordingly, activation of apoptosis is an important target in anti-cancer therapy. There are two pathways to initiate apoptosis: the mitochondrial (intrinsic) pathway [242] and the receptor-mediated (extrinsic) pathway [243]. The intrinsic pathway of apoptotic cell death originates from an array of intracellular damages or stress signals including oxidative stress, DNA damage, cytokine withdrawal and endoplasmic reticulum stress [244,245]. The extrinsic pathway is activated via specific receptors such as tumor necrosis factor-family receptors or Fas (CD95/TNFRSF6/Apo-1) [246]. To date, more than 60 marine sponges have demonstrated anti-cancer potential through the induction of apoptosis. However, the chemistry and anti-cancer mechanisms of pro-apoptotic marine-derived compounds have not been completely elucidated.
Marine seaweeds have been used to synthesize a wide range of bioactive substances in accordance with their maturity and ability to interact with environmental changes. Among the abundant pool of compounds derived from seaweed, terpenes, polysaccharides and polyphenols are of clinical relevance in terms of their anti-cancer activities [247]. Fucoidans refer to an array of sulfated polysaccharides that originate from the cell wall of brown seaweeds. These sulfated polysaccharides are the principal components of the sticky molecules present in brown seaweeds [248]. Compared with high molecular-weight fucoidans, the low molecular-weight versions are more biologically active [249]. The Akt signaling pathway, a significant signal transduction pathway, enhances extracellular signals on growth and survival. Studies have shown that fucoidans inhibit phosphorylation of proteins related to Akt and phosphoinositide 3-kinase (PI3K) in in vitro and in vivo models. This, in turn, inhibits tumor cell proliferation [250].
3.1.2. Approved drugs
Cytarabine is the first commercially available anti-cancer drug of marine origin [251]. It is a pyrimidine nucleoside analog that functions as a cytotoxic drug that acts selectively on the cell cycle. It functions principally in the S phase of cell division when the cells are undergoing DNA synthesis [252]. Cytarabine was approved by the US Food and Drug Administration (FDA) and commercialized under the trade name Cytosar-U®. Cytosar-U® has been recognized as a mainstay drug in the management of acute myeloid leukemia, acute lymphoblastic leukemia and chronic myelogenous leukemia [253]. Other marine-derived anti-cancer formulations have since been available. For example, CPX-351 (Vyxeos®), a liposomal formulation, consists of cytarabine and daunorubicin in a 5:1 ratio. Delivery of CPX-351 to the leukemia cells of patients diagnosed with secondary acute myeloid leukemia resulted in significant improvement in the overall survival status, compared with conventional standard of care [254].
Trabectedin (Yondelis®), an extract from the Caribbean tunicate Ecteinascidia turbinate, has also been approved by the European Medicines Agency and by the US FDA for the treatment of advanced soft tissue sarcomas. Trabectedin is a tetra-hydro isoquinoline alkaloid that can be produced synthetically [255]. This molecule acts against crucial processes in tumor cells as well as the tumor microenvironment via tissue-resident histiocytes and tumor-associated macrophages [256]. This agent demonstrated remarkable efficacy in different settings, including treatment-naïve and pre-treated patients with local advanced disease and tumor metastasis [257]. A phase III multicenter randomized clinical trial on the safety and efficacy of Trabectedin has been conducted (NCT01343277; ClinicalTrials.gov) [258]. In this clinical trial, patients with metastatic leiomyosarcoma or liposarcoma after failure of conventional chemotherapy were included. Recall after ten years of treatment demonstrated acceptable treatment outcomes without detrimental side effects.
Eribulin (Halaven®) is the third market-authorized formulation for combating cancer. Eribulin mesylate (eribulin) is the analog of halichondrin B, a natural product from marine-sponge [259]. Eribulin is now used as a third-line (United States) or second-line (Europe) formulation to address pretreated metastatic breast cancer [260]. Approved for use in more than 50 countries in the last decade, eribulin is the only anti-cancer drug showing improved overall survival and acceptable toxicity profile, especially for serious pre-treated patients with metastatic breast cancer [261]. A phase II non-randomized clinical trial demonstrated the tolerability and effectiveness of eribulin for partients with advanced or metastatic soft-tissue sarcoma (NCT03810976; ClinicalTrials.gov) [262]. Apart from its remarkable efficacy in treating breast cancer and sarcoma, eribulin also showed anti-cancer activity against other tumor types, such as non-small cell lung cancer [263], tumor metastases [264] and gastric cancer [265].
The CD30-targeting antibody-drug conjugate (ADC) brentuximab vedotin (Adcetris®) is derived from a sea hare known as Dolabella auricularia. It is the first commercially available medication to treat the principal cells in Hodgkin's lymphoma (HL) such as malignant Hodgkin and Reed/Sternberg cells via affecting the tumor microenvironment. Brentuximab vedotin was rapidly approved by the US FDA in 2011 to treat systemic anaplastic large cell lymphoma (sALCL) and relapsed classical HL. The CD30 membrane receptor, one of the tumor necrosis factor receptors, is expressed extensively on specific tumor cells, especially in classical HL and sALCL. For normal cells, CD30 shows limited expression. Taking all these advantages into account, the CD30 membrane receptor is currently used as a potential target for ADC-related therapy [266,267]. Brentuximab vedotin, an ADC, binds to the CD30 antibody and conjugates to the monomethyl auristatin E, a dolastatin 10 synthetic analog, via a protease cleavable linker [268]. Brentuximab vedotin provides patients with sustained, progression-free survival (PFS), instead of best supportive care alone following autologous hematopoietic stem cell transplant. In addition, peripheral neuropathy, the most common and unsatisfactory side effect of chemotherapy, was alleviated in 90% of the patients [269].
3.1.3. Agents in clinical trials
Eighteen candidates based on marine-derived metabolites are currently enrolled in the oncological pipeline (Table 3). These trials reflect preliminary interests in the marine-derived anti-cancer market.
Table 3.
Marine-derived chemotherapeutics in clinic trials.
| Stage | Compound Name | Source | Molecular Target | Interventions | Cancer Types | Status | NCT number | Refs |
|---|---|---|---|---|---|---|---|---|
| Phase III | Plitidepsin (Aplidin®) | Aplidium albicans | Rac1 and JNK activation | Plitidepsin; Dexamethasone | Relapsed/refractory myeloma | Completed; Has Results | NCT01102426 | [270] [271] |
| Phase III | Plinabulin | Aspergillus ustus | Inhibit tubulin polymerization | Plinabulin; Nivolumab | Recurrent or metastatic non-small cell lung cancer in Stage IIIB-IV, | Terminated; Has Results | NCT02846792 | [272] [273] |
| Phase III | Depatuxizumab mafodotin ABT-414 | N/A | Noncleavable maleimidocaproyl linker, epidermal growth factor receptor and microtubule cytotoxin | ABT-414; Temozolomide; Whole Brain Radiation |
Glioblastoma multiforme | Completed | NCT01800695 | [274] [275] |
| Phase III | Salinosporamide A | Salinospora tropica | Suppressing the MAPK pathway | Marizomib; Temozolomide; Radiotherapy | Newly diagnosed glioblastoma | Active, not recruiting | NCT03345095 | N/A |
| Phase III | Lurbinectedin (Zepsyre®) | Trabectedin | Phosphorylated RNA polymerase II | Lurbinectedin; Pembrolizumab | Small cell lung carcinoma | Recruiting | NCT04358237 | [276] |
| Phase II | Polatuzumab vedotin DCDS-4501A |
Dolabella auricularia | CD79b and microtubules | Obinutuzumab;Pinatuzumab Vedotin;Polatuzumab Vedotin;Rituximab | Relapsed or refractory B-Cell non-Hodgkin's lymphoma | Completed Has Results |
NCT01691898 | [277] |
| Phase II | Glembatumumab vedotin CDX-011 |
Dolabella auricularia | Inhibit microtubule and monomethyl auristatin E | Capecitabine;CDX-011 | Recurrent osteosarcoma | Completed Has Results |
NCT02487979 | [278] |
| Phase II | Glembatumumab vedotin CDX-011 |
Dolabella auricularia | Inhibit microtubule and monomethyl auristatin E | Glembatumumab vedotin; CDX-301 | Advanced melanoma | Terminated Has Results |
NCT02302339 | [279] |
| Phase II | Glembatumumab vedotin CDX-011 |
Dolabella auricularia | Inhibit microtubule and monomethyl auristatin E | Glembatumumab Vedotin | Metastatic or locally-recurrent uveal melanoma | Completed Has Results |
NCT02363283 | [280] |
| Phase II | Tisotumab vedotin (HuMax®-TF-ADC) | Dolastatin 10 | Inhibit microtubules | Tisotumab Vedotin (HuMax-TF-ADC) | Bladder cancer, ovary cancer, prostate cancer, cervix cancer, endometrium cancer, lung cancer, esophagus cancer and squamous cell carcinoma of the head and neck | Completed | NCT02001623 | [281] |
| Phase II | Telisotuzumab vedotin ABBV-399 |
Dolastatin 10 | c-Met | ABBV-399 | Recurrent squamous cell lung carcinoma and stage IV squamous cell lung carcinoma AJCC V7 | Active, not recruiting | NCT03574753 | [282] |
| Phase II | Telisotuzumab vedotin ABBV-399 |
Dolastatin 10 | c-Met | Telisotuzumab vedotin | Non-small cell lung cancer | Recruiting | NCT03539536 | N/A |
| Phase II | AGS-16C3F | Dolastatin 10 | ENPP3 and Inhibit Microtubules | AGS-16C3F | Metastatic renal cell carcinoma | Active, not recruiting Has Results |
NCT02639182 | N/A |
| Phase II | GSK2857916 | Dolastatin 10 | B-cell maturation antigen | GSK2857916 and Pembrolizumab | Multiple myeloma | Recruiting | NCT03848845 | [283] |
| Phase II | PM060184 Plocabulin |
Lithoplocamia lithistoides | Microtubules | PM060184 | Advanced colorectal cancer | Unknown | NCT03427268 | [284] |
| Phase II | Ladiratuzumab vedotin SGN-LIV1A |
Dolastatin 10 | LIV-1 and microtubules | ladiratuzumab vedotin | Small cell lung cancer, esophageal squamous cell carcinoma, gastroesophageal junction adenocarcinoma, squamous, non-small cell lung cancer, non-squamous head and neck squamous cell carcinoma, gastric adenocarcinoma, prostate cancer and melanoma | Recruiting | NCT04032704 | [285] |
| Phase II | Enfortumab vedotin ASG-22ME |
Dolastatin 10 | Nectin-4 and microtubules | Enfortumab vedotin, pembrolizumab, cisplatin, carboplatin, gemcitabine | Urothelial cancer | Recruiting | NCT03288545 | [286] |
| Phase III | Enfortumab vedotin ASG-22ME |
Dolastatin 10 | Nectin-4 and microtubules | Pembrolizumab, Surgery (radical cystectomy (RC) plus Pelvic Lymph Node Dissection, Enfortumab Vedotin | Urinary bladder cancer, muscle-invasive | Recruiting | NCT03924895 | N/A |
| Phase I | Enfortumab vedotin ASG-22ME |
Dolastatin 10 | Nectin-4 and microtubules | Enfortumab vedotin | Metastatic urothelial cancer | Completed | NCT03070990 | [287] |
| Phase I | Enfortumab vedotin ASG-22ME |
Dolastatin 10 | Nectin-4 and microtubules | ASG-22CE, AGS-22M6E | Neoplasms | Completed | NCT01409135 | N/A |
| Phase I | ABBV-085 | Dolastatin 10 from Symploca sp. | Direct against LRRC15 on CAFs Cytotoxic drug through antimitotic activity |
PF-06647020 | Advanced solid tumors, squamous cell carcinoma of the head and neck carcinoma of the breast, undifferentiated pleomorphic sarcoma | Completed | NCT02565758 | [288] [289] |
| Phase I | PF-06647020 | Marine polyphenols from seaweeds | PTK7-targeted; Cytotoxic anti-cancer agent |
ASG-15ME | Breast cancer | Recruiting | NCT03243331 | [290] [291] [292] |
| Phase I | ASG-15ME | Dolastatin 10 from Symploca sp. | Target SLITRK6 Microtubule-disrupting agent. |
AGS-67E | Metastatic urothelial cancer | Completed | NCT01963052 | [293] [294] [295] |
| Phase I | AGS-67E | Dolastatin 10 from Symploca sp. | Target CD37 Anti-tubulin activities |
AGS67E | Relapsed lymphoid malignancy Refractory lymphoid malignancy |
Completed | NCT02175433 | [296] |
3.2. Antimicrobial agents
Multidrug resistance is a major healthcare threat caused by the abuse of antibiotics. Finding a substitute for antibiotics has attracted much attention. Marine organism-derived potential antimicrobial agents include peptides, terpenes, alkaloids and proteins. In this section, typical antimicrobial agents derived from marine organisms will be thoroughly discussed.
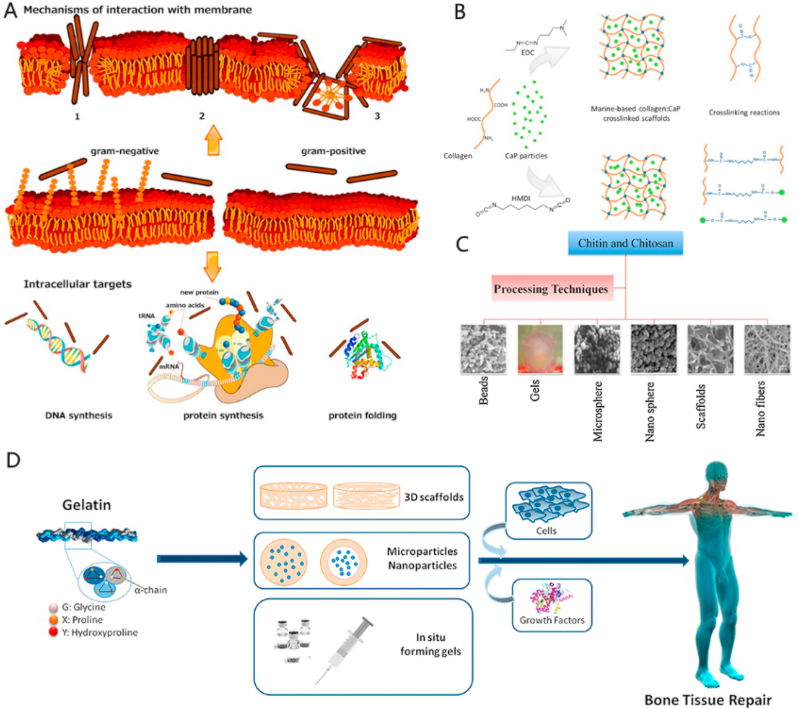
Antimicrobial peptides (AMPs) originate from marine organisms are structurally diverse. These AMPs exhibit a broad range of antibacterial activities. The AMPs are classified into four groups depending on their stuctures: amphipathic α-helical, β-sheet, β-hairpin or loops, as well as extended AMPs [297] (Fig. 7A). These AMPs target specifically to pathogens and overcome antimicrobial resistance efficiently. Marine-derived AMPs discovered in the last 5 years are discussed in this section.
Fig. 7.
(A) Antimicrobial peptides (AMPs) kill bacteria by inducing membrane damage and/or internalization. An alternative antibacterial mechanism of AMPs is intracellular targeting. Some AMPs act on intracellular targets by inhibiting cell wall synthesis, nucleic acid binding and synthesis, protein production and enzyme activity. (B) Schematic of crosslinking reactions between 1-[3-(dimethylamino)propyl]-3-ethylcarbodiimide hydrochloride (EDC) and hexamethylene diisocyanate (HMDI) crosslinking agents. (C) Various forms in which chitin and chitosan constructs can be fabricated. (D) Gelatin used for bone tissue repair. (A is reproduced from Ref. [298] with permission from publisher; B is reproduced from Ref. [299] with permission from publisher; C is reproduced from Ref. [300] with permission from publisher; D is reproduced from Ref. [301] with permission from publisher).
Tilapia piscidin 4 obtained from Nile tilapia is capable of killing Helicobacter pylori and halting the progression of gastric ulcer [302]. Venerupis philippinarum defensin (VpDef) is isolated from the clam V. philippinarum. Recombinant VpDef shows broad-spectrum antimicrobial activity. The minimum inhibitory concentration is as low as 50 μg/mL against Escherichia coli O157. Recombinant VpDef is resistant to proteinase digestion and stable under temperature and pH fluctuation. Hence, VpDef is suitable for large-scale production [303]. Cathelicidins derived from sea snakes are also regarded as potent candidates for peptide-based antimicrobial agents [304]. A study reporetd that cathelicidins kill bacteria within 1 h in zebrafish [305]. A peptidic compound obtained from Aneurinibacillus sp. YR247, has excellent antifungal ability [306]. Theonellamides are dodecapeptides originated from the marine sponge Theonella sp. These dodecapeptides show attractive antifungal and cytotoxic bicyclic activities. Theonellamides bind to 3β-hydroxysterols on the yeast membrane surface and cause membrane damage [307]. Aeroplysinin-1 from Aplysina aerophoba demosponge acts as an antibacterial agent against clinically-relevant mircoorganisms [308]. In addition, chitin-based skeletal scaffolds from marine sponges covered with silver nanoparticles and silver-bromide exhibited excellent antibacterial ability. These scaffolds may be used in water filtration systems [309]. Apart from antimicrobial ability, hepcidins derived from the spotted scat Scatophagus argus also possess antiviral activities. Hepcidins effectively eliminate Siniperca chuatsi (Chinese perch) rhabdovirus and largemouth bass Micropterus salmoides reovirus [310].
Recent reports suggest that chitin-bromotyrosine hybrid scaffolds possess superior antimicrobial activities against clinical strains of Staphylococcus aureus, Enterococcus faecalis and Propionibacterium acnes. Marine-derived chitinous skeletons have long been identified as excellent three-dimensional biomaterials-based matrices [311]. Bromotyrosines possess multitarget activities including antimicrobial, anti-cancer, antiviral as well as cytotoxic traits [312]. In the chitin-bromotyrosine hybrid scaffolds, chitin is used as drug delivery vehicles because of its mesh-like structure and capillary network [313]. A recent study reported that simultaneous extraction of both chitin and bromotyrosine is feasible [219]. Typical chitin-bromotyrosine hybrid scaffolds such as aeroplysinin-1 demonstrated effective antibacterial activity in vitro [219]. Inhibition of microbial chitinases via these halogenated compounds are highly relevant [314]. From an economical perspective, demosponge-derived chitin-bromotyrosine hybrid scaffolds are easily available. For example, A. aerophoba, a kind of demosponge with abundant bromotyrosine, grows 10 cm annually and are located at 2–5 m beneath the sea level. This easily reachable ocean depth ensues a easily obtainable and sufficient supply of bromotyrosine. It is estimated that 99 g of bromotyrosine can be harvested from every 10 m2 of A. aerophoba colony. Such an amount of extracted bromotyrosine value up to US$14,850,000 [219]. Although these naturally-occurring biomaterials possess immense potential, the gap between the ocean and clinical application remains abysmal. There is an urgent need for in-depth research in this exciting field.
Regrettably, the majority of AMPs have not undergone clinical trials. Several challenges have to be taken into consideration prior to untaking clinical trials. These challenges include how to achieve potent antimicrobial activities under physiological conditions such as pH, serum conditions, salinity, rapid degradation of AMPs by proteases as well as the high cost of production [298].
3.3. Tissue engineering and regenerative medicine
Tissue engineering refers to the promotion of tissue regeneration or replacement within the confines of engineering and life sciences. Tissue engineering utilizes 3D scaffolds for temporary support to provide a suitable environment and structure for tissue regeneration. Three dimensional scaffolds with suitable pore sizes are critical for cell migration, proliferation and diffusion of oxygen and nutrients. Qualified 3D scaffolds should possess appropriate biodegrading rate and excellent mechanical properties. In addition, low immunogenicity and low toxicity are highly-esteemed traits [225,315,316]. Many natural materials (e.g. gelatin, chitosan) and synthetic materials (e.g. bioactive ceramics and synthetic polymers) have been used for construction of scaffolds. Natural marine materials with unique and attractive architectures are promising candidates for the preparation of tissue engineering scaffolds.
3.3.1. Biomaterials extracted from marine organisms
Natural polymers are important components of the extracellular matrix. They exhibit superior biocompatibility and low immunogenic potential. Collagen obtained from marine organisms may be used for preparation of 3D composites with excellent osteogenesis potential. Scaffolds prepared by combining marine bioapatite particles with marine collagen demonstrate superior mechanical properties, bioactivity and ability to promote osteogenic differentiation (Fig. 7B) [299]. Gelatin has been extensively investigated as 3D scaffolds for bone tissue repair and regeneration (Fig. 7D). Several collagen/gelatin-based products are commercially available for bone tissue engineering. These products include DBX® and RegenOss® [317] (Table 4).
Table 4.
Biomaterials from marine organisms applied in tissue engineering.
| Biomaterials | Marine Sources | Tested cell types | Cell viability or improved properties | Application | Advantages(A) and disadvantage (DA) | Refs |
|---|---|---|---|---|---|---|
| Gelatin methacryloyl | Cold water fish | NIH3T3 cells | 90% | Tissue engineering and drug delivery | A: High degradation rate, appropriate melting and gelling points (~37 °C) DA: low compressive moduli |
[332] |
| Collagen | Salmon skin | Mesenchymal stem cells | Improved proliferative and differential ability of mesenchymal stem cells | Bone tissue engineering | A: Excellent mechanical properties, increased degradation rate and the compressive strength | [333] |
| Shark skin (Prionace glauca) | Osteoblast-like cell | Majority of the cells are viable | Bone tissue engineering | A: Bioactive stable composite structures, feasibility, low cost | [299] | |
| Fish | T-Lymphocyte leukemia cell lines and osteoblast | Decreased (T-cells), Improved osteogenic ability (osteoblast) |
Bone tissue engineering | A: Biocompatibility, high stiffness, low biodegradation rate, and osteogenesis ability | [334] | |
| Sea urchin | Fibroblast | Increased proliferation rate | Tissue regenerative medicine | A: Biocompatiblity, mechanically stable in wet conditions, resemblance the dermal layer in structure and function. | [334] | |
| Alginate | Brown sea algae | Mesenchymal stem cells | Increased cell number | Bone tissue engineering | A: Excellent mechanical properties, suitability for cell attachment and spread | [335] |
| Human osteoblasts | Improved osteoblasts proliferation | Bone tissue engineering, drug delivery | A: Suitable physical performance, bioactivity | [336] | ||
| Chitin | Demosponge Ianthella basta | Mesenchymal stromal cells | High percentage of viability, improved differential ability | Tissue engineering and regeneration, such as artificial skin | A: Ability to improve adipogenic differentiation of cells, simplicity of the isolation, interconnected porosity and excellent biocompatibility | [337] |
| Demosponge Aplysina aerophoba | Human mesenchymal stromal cells | Increased metabolic activity, cell numbers, and differential ability | Tissue engineering | A: Excellent cytocompatibility | [338] | |
| Collagen-chitosan-fucoidan cryogels | Jellyfish (collagen), brown algae(fucoidan), giant squid(chitosan) | L929 cells | 70% | Tissue engineering | A: Mechanical stability, adequate porosity for cell proliferation | [339] |
Chitosan has been extensively investigated for bone regeneration applications. It is positively-charged with excellent biodegradability, biocompatibility, muco-adhesive and hemostasis properties (Fig. 7C). A chitosan-polycaprolactone copolymer improves the ability of pre-osteoblastic cells to form bone tissue [318]. Combining chitosan with other polymers or biomaterials helps to control its porosity and water retention capability, reduce its biodegradation rate, increase its mechanical properties and enhance its bioactivity [319]. The use of a chitosan scaffold with stromal vascular fraction aliquots demonstrated excellent bone regeneration ability [320]. In vitro experiments showed that chitin-based scaffold supported chondrocyte attachment and extracellular matrix synthesis [321]. In vivo experiment showed that whitlockite/chitosan scaffold significantly improves bone repair in skull defect [322]. The chemotaxis and regenerative potential of human dental pulp cells could also be enhanced by porous chitosan-calcium aluminate scaffolds [323]. Incorporation of silver-doped bioactive glass in chitosan enhances the odontogenic differentiation potential of dental pulp stem cells [324]. The mechanical properties, bioactivity and osteogenic potential of chitosan-based scaffolds are improved when chitosan is combined with other polymers and biomaterials such as hydroxyapatite, bioglass ceramic and silicon dioxide [300]. The production of 3D chitinous scaffolds using microwave-assisted extraction is efficient and environmentally friendly. Such a fabrication method facilitates the application of 3D chitinous scaffolds in the biomedical and technological arena [325].
Three-dimensional bioprinting is rapidly developing in tissue engineering and regeneration. Bioprinting is accomplished through layer-by-layer deposition of cell-containing biomaterials in a predetermined structural architecture. High-resolution printed cell-containing scaffolds are biocompatible, biodegradable, bioactive and possess optimal mechanical properties (Fig. 8A). Compared with synthetic materials, 3D bioprinting using marine-derived biopolymers have additional advantages such as resemblance to extracellular matrix composition or structure and self-assembling capability [326] (Table 5)
Fig. 8.
A) Properties of bioink in 3D bioprinting. B) The structure of polyunsaturated fatty acids and originated oils. C) Interrelations between polysaccharides of marine origin and drug delivery systems for advanced therapeutic applications. (A is reproduced from Ref. [326] with permission from the publisher; B is reproduced from Ref. [327] with permission from the publisher; C is reproduced from Ref. [328] with permission from the publisher).
Table 5.
Biomaterials from marine organisms applied in 3D bioprinting.
| Biomaterials | Marine Sources | Tested cell types | Cell density and viability | Advantages(A) and disadvantages (DA) | Application | 3D bioprinting method | Refs |
|---|---|---|---|---|---|---|---|
| Alginate | Brown algae | Mesenchymal stem cells | 85.0 ± 5.9% | A: Spatially varying mechanical microenvironment extrusion printing | Bone tissue engineering | Extrusion printing | [340] |
| Alginate | Brown algae | Human mesenchymal stem cells | 84% ± 0.7% | A: Excellent scaffold fidelity and mechanical properties (higher alginate concentration); Excellent cell viability and cell spreading morphology (lower alginate concentration) |
Bone tissue engineering | Extrusion printing | [332] |
| Silk-like protein (aneroin) | Sea anemone | Mouse pre-osteoblasts, rat myoblasts, mouse fibroblasts, and rat-derived mesenchymal stem cells | 99% | A: Accurate printability, structural integrity, and biocompatibility | Cartilage or skeletal tissue regeneration | Extrusion printing | [341] |
| Gelatin | Cold water fish | NIH3T3 cells | Over 80% | A: Inexpensive, high emulsion stability DA: low viscosity and rapid polymerization |
Drug delivery | Extrusion printing | [342,343] |
| κ-carrageenan-gelatin hydrogel | Carrageen | C2C12 cells | Over 90% | A: Excellent structural stability and cell viability. | Biological binder for tissue scaffolds | Extrusion printing | [344] |
| Chitosan | Crab shell | Mouse pre-osteoblast cells | Over 90% | A: Good viscoelastic properties, stable under physiological conditions, proper viscosity values. | Bone tissue engineering | Extrusion printing | [345] |
| Carrageenan | Red algae | Mesenchymal stem cells | Dead cells were negligible | A: Excellent structural strength, cyto-compatible and non-toxic. | Tissue engineering and regeneration | Extrusion printing | [346] |
Researchers have combined agarose with collagen to produce a hydrogel blend for long-term cell encapsulation [347]. Tailored hydrogel blends formed by agarose-based bio-ink incorporating collagen or fibrinogen via 3D bioprinting support endothelial and fibroblast cell growth. These hydrogel blends enable the formation of a vascularized network to ensure adequate nutrition supply [348]. Gelatin is an attractive candidate for 3D bioprinting. To improve its mechanical modulus and degradation rate, gelatin is modified by methacrylation. The methacrylated gelatin supports cell adhesion and proliferation [349]. Interpenetrating hydrogels composed of alginate and gelatin exhibit excellent mechanical strength, swelling ratio, degradation and cell behavior, which meets the requirements for tissue engineering [350].
3.3.2. Marine skeletons used in tissue engineering
Marine skeletons have tailored structures. For example, seashells have dense lamellar structure. Cuttlebone and coral have an interconnected porous structure that resemble native bone structure. The porosity, microstructure, and crystallography of these structural marine skeletons is highly desirable for tissue engineering [223]. The architectures of marine skeletons have been described in Section 2.5 (Table 6).
Table 6.
Marine skeletons and other hard tissue applied in tissue engineering.
| Marine skeletons | Effective constituents | Porosity and pore size | Properties | Application | Refs |
|---|---|---|---|---|---|
| Shark teeth | Apatitic (hydroxyapatite and apatite-(CaF)), non-apatitic (whitlockite), and an apatitic phase (fluorapatite), | Pore size: ~50 μm and ~0.5–1 μm) | Enrichments of specific trace elements (Mg, F); Improved proliferation and osteogenic activity of cells |
Bone tissue regeneration | [352] |
| Mussels and oysters | Calcium carbonate | Particle size: 1.5 μm | Enhanced ECM mineralization and osteoblastic differentiation of MSC | Bone tissue regeneration | [353] |
| Coral | Hydroxyapatite | Porosity: 70–75% | Excellent osseous tissue formation, carrier for growth factors | Tissue regeneration | [354] |
| Acropora coral | Pores size: 412 ± 212 μm, porosity: 12 ± 4% | Interconnected large pores | Tissue regeneration | [355] | |
| Cuttlebone | Porosity: 90.4 ± 3.5% | Interconnective porous structure; High protein adsorption rate; promotes osteogenic differentiation of MSC |
Bone tissue regeneration | [356] | |
| Cuttlebone | Biphasic Calcium Phosphate | N/A | Excellent degradability and bioactivity | Bone tissue engineering | [357] |
| Marine sponge Aplysin aarcheri | Chitinous scaffolds | N/A | 3D chitinous scaffold for calcium carbonate deposition | Developed mineralized scaffolds | [358] |
| Coral microparticles | Calcium carbonate | Porosity: 99.05% | Enhanced compressive properties; promotes more robust osteogenic differentiation of mesenchymal stromal cells, | Bone regeneration. | [359] |
| Deep-sea bamboo coral | Calcareous structures alternated with smaller proteinaceous nodes of gorgonin | N/A | Excellent potential for colonization with human osteoblasts and osteoclasts | Tissue regeneration | [360] |
Calcium phosphate hydrothermally-converted from coral resembles chemical composition of natural bone tissue. Skeletons of coral such as Pocillopora verrucosa and Acropora formosa meet the requirement of 3D scaffolds for bone tissue engineering [329]. Two reef-building coral skeletons derived from Goniopora coral and Porites coral have been used commercially as bone graft materials [351]. Five effects of coral skeletons applied in this field include: (i) increase bioactivity via altering composition and crystal structure; ii) carry osteoinductive factors; (iii) enhance resorption properties; (iv) strengthen against compression forces in bone via converting to hydroxyapatite and fusing with hydroxyapatite nano-coatings; (v) increase bioactivity via interactions with surfaces [329] (Fig. 9A).
Fig. 9.
A) New biomedical uses of coral skeletons. B) Purported cardioprotective mechanisms associated with fish and fish oil-derived n-3 PUFAs. C) Effect of marine-derived materials on bone metabolism via up-regulation of osteoblastogenesis and downregulation of osteoclastogenesis. Some are directly involved in osteoblastogenesis by increasing the expression of Runx2, BMP and other transcription factors. Others are dependent on the regulation of cytokine production (IL-6 and TNF). Marine-derived compounds suppress osteoclastogenesis through downregulating the expression of RANKL, NFATc1 and TRAP) (A is reproduced from Ref. [329] with permission from the publisher; B is reproduced from Ref. [330] with permission from the publisher; C is reproduced from Ref. [331] with permission from the publisher).
Incorporation of bioactive molecules into coralline graft enhances its tissue regeneration properties. For example, the combination of coralline graft with vascular endothelial growth factor results in improved neovascularization and mineralization [361].
The contribution of marine sponges in bone tissue engineering is promising due to their excellent properties. For example, the interconnecting porous architecture of sponges promote the circulation of fluids. They also possess osteogenic properties, improve cell growth, facilitate bone formation and mineralization [225]. Chitinous scaffolds extracted from marine sponge Ianthella labyrinthus are suitable for cardiomyocyte attachment and growth [362]. Marine sponges combined with growth factors are effective for bone regeneration [363]. Bone regeneration results of surface-modified hydroxyapatite sponge scaffolds are comparable with the use of demineralized freeze-dried bone allografts [364]. In addition, 3D chitinous scaffolds from Aplysin aarcheri demosponges induce calcium carbonate precipitation. When used for bone tissue engineering, these scaffolds are biocompatible, biodegradable and osteoinductive [365].
Cuttlefish bone is also used as a bone graft material. Compared with commercial synthetic hydroxyapatite, bioactive hydroxyapatite-based bioceramics prepared from cuttlefish bone powder exhibit improved apatite deposition, cell proliferation and alkaline phosphatase activity [366]. The combination of inorganic components with biopolymers is a feasible method to improve the functional properties of bone graft materials. Silicon-substituted cuttlefish bone-derived hydroxyapatite has been reported to enhance osteoblast differentiation [367].
Teeth from shark are employed in experimental bone tissue engineering. Shark teeth contain not only apatite, but also comprise additional elements that are beneficial for bone regeneration, such as magnesium. Bioapatite obtained from thermal processing of shark enameloid and dentin is biocompatible and possesses osteogenic activity [352]. It is presumed that shark teeth may be used as a novel source of bioapatite for bone repair in the future [368].
Molecules obtained from marine organisms also play an important role in cartilage regeneration. Chondroitin sulfate present in natural cartilage is crucial for chondrogenesis. The mechanical performance of a chondroitin sulfate-containing double network gel is comparable with native cartilage tissue. In addition, this synthetic gel also induces chondrocytes differentiation [369]. Likewise, an elastic microporous gelatin/chondoitin-6-sulfate/hyaluronan-chitosan cryo-gel scaffold designed for cartilage tissue engineering demonstrated improved mechanical properties and chondrocyte differentiation response [370].
3.4. Drug delivery
The development of personalized, less invasive, sustained release therapeutic agents of drug delivery systems is an ideal target for patients. In the present section, approaches to construct advanced drug delivery systems via marine organisms as raw materials will be discussed.
3.4.1. Collagen-mediated drug delivery
Collagen molecules possess the amazing capability to self-assemble into higher-order fibrillar structures. Hence, collagen serve sas the pivotal component in drug delivery applications. Sophisticated approaches have developed as collagen-based drug delivery systems, such as capsules prepared from gelatin (denatured collagen), injectable hydrogels loaded with growth factor, corneal shields, collagen sponges used in chronic wounds as well as antibiotic dressings [371,372]. Moreover, collagen also serves as an attractive drug targeting site in several disease, such as lung and liver fibrosis, psoriasis and scleroderma [373]. In the models of these diseases, the use of collagen-binding proteins realizes the targeting and recognition via the signal molecule collagen. However, collagen has poor mechanical properties and inadequate thermal stability compared with synthetic polymers. Thus, the physicochemical properties of marine-derived collagens require modification by incorporation of other biomaterials to make them suitable for drug delivery.
Collagen molecules possess conservative sequences that permit specific collagen-protein interactions. Such interactions can either interfere with collagen-related diseases or supply collagen at the sites where normal collagen function is impaired [374]. A type IV collagen triple helical peptide has been investigated to bind tightly to target of some tumor cells such as CD44, a chondroitin sulfate proteoglycan receptor that is overexpressed in several tumor cells [375]. Liposomes loaded with doxorubicin and a triple-helical peptide-amphiphile were capable of targeting the over-expressed CD44 receptors in tumor sites, reducing tumor size to 60% compared with the control group [376].
3.4.2. Polysaccharide-mediated drug delivery
Marine polysaccharides have currently been applied in food, cosmetics and pharmaceutical industries (Fig. 8C). Algae are rich in marine polysaccharides. Commercial alginates originate principally from alginic acid in brown marine algae (Phaeophyceae). Alginate may also be obtained from marine bacterial sources such as Laminaria digitata, Laminaria hyperborea, Ascophyllum nodosum, Laminaria japonica and Macrocystis pyrifera via aqueous alkali solutions (NaOH) treatment [377,378].
Alginate hydrogels have been employed as drug delivery systems for a long time because of their biocompatibility, non-toxic nature and bio-availability. It is used as a stabilizer agent in pharmaceutical formulations to control drug release. For example, sodium alginate has long been processed as a tablet binding material. Alginic acid, nevertheless, has been currently used as a tablet disintegrating material with the aim of immediate drug release [377]. Alginate may also be used for pH-sensitive drug release in the colon region for treating patients with inflammatory bowel diseases [379].
Carrageenans are typical sulfated polysaccharides originated from the cell walls of red seaweeds. Carrageenans may be categorized accroding to their level of sulfation into kappa (κ), iota (ι), and lambda (λ) carrageenans, which contain one, two or three sulfate groups, respectively [380]. Because of their organized, ordered and stable networks as well as wide water-pockets, carrageenans are used as vehicles to deliver drug cargoes [381]. Carrageenan-based formulations used as drug delivery systems include hydrogels, hydrogel beads, microspheres and microparticles [[382], [383], [384]]. Carrageenan is an ideal excipient for controlled release in drug delivery systems. The dissolution rate and mechanical properties of carrageenan are greatly affected by its sulfation extent. As a result, the drug loading capacity and the release efficiency of the loaded cargo are also altered [385]. κ-carrageenans resemble native glycosaminoglycans and form fragile hydrogels by ionic crosslinking. They can protect cells from strong shear forces and are excellent devices for cell encapsulation. Human mesenchymal stem cells remain predominatly viable and maintain their circular morphology after encapsulation with κ-carrageenan. Shear-thinning carrageenan-based hydrogels may be used as cell-delivering vehicles in cartilage tissue regeneration and 3D bioprinting [386].
Fucoidans are sulfated polysaccharides extracted from brown algae. Chitosan/fucoidan nanoparticles have been used experimentally for pulmonary delivery of gentamicin, a commonly used aminoglycoside antibiotic. These nanoparticles demonstrated a zero-order release of gentamicin for the first 10 h. This was followed by stable release of up to 72 h [387]. Moreover, chitosan/fucoidan nanoparticles were capable of sustained antibiotic release in simulated gastric fluid and effectively suppressed the growth of clinically-relevant pathogens such as S. aureus and E. coli [388].
3.5. Anticoagulant
Fucoidan participates in the blood coagulation cascade by interacting with the natural thrombin inhibitors including antithrombin, heparin cofactor II, activated factor II (thrombin) and activated factor X [389,390]. Fucoidan-modified surfaces possessed potent anticoagulation activity. The thrombin time threshold was prolonged when contacting with the fucoidan-modified surfaces. It is presumed that these fucoidan-modified surfaces may serve as the alternatives as anti-clotting devices such as blood storage bags [391]. Chitosan-fucoidan nanoparticles possess better in vitro anticoagulant activity than pristine fucoidan. Solid evidences showed that this hybrid nanoparticles could reduce thrombus formation even in a deep vein thrombosis model [392]. The positive surface charge present in chitosan-fucoidan nanoparticles contributes to the increase in activated partial thromboplastin time and is concentration dependent. It is inspiring that fucoidans also exhibit antithrombotic activity when administered orally. Low molecular weight fucoidans are better absorbed orally and have superior potent anti-thrombotic activity compared to medium molecular weight fucoidans [393].
3.6. Applications in other diseases
3.6.1. Cardiovascular diseases
Cardiovascular diseases (CVD) result in 17.3 million deaths per year and are the major cause of global deaths [394]. Long-chain omega-3 polyunsaturated fatty acids (PUFAs), such as eicosapentaenoic acid and docosahexaenoic acid, are produced by fish and other marine organisms. These PUFAs have already been used extensively to combat CVD (Fig. 8B). The mechanisms responsible for the use of PUFAs against CVD may be summarized as follows: i) modulation on the function of cardiac ion channels, such as Na and L-type Ca channels; ii) prevention of the transformation from arachidonic acid into pro-inflammatory eicosanoid; iii) anti-inflammatory mediators including enzymatically-oxygenated metabolites (Fig. 9B) [395]. The anti-inflammation properties of these fatty acids are prominent. The PUFAs are involved in the inhibition of leukocyte chemotaxis, interactions between leukocyte and endothelial cells, and secretion of inflammatory cytokines [396]. In murine models, long-chain monounsaturated fatty acids obtained from fish oil exhibited anti-atherogenic effects [397]. Clinical trials demonstrated that patients with acute myocardial infarction who took omega-3 fatty acids exhibited less adverse left ventricular remodeling and non-infarct myocardial fibrosis [398].
3.6.2. Bone diseases
Natural products derived from marine cyanobacteria, dinoflagellates, algae, sponges, soft corals, mollusks, fishes and mangroves significantly affect bone metabolism by inhibiting osteoclastogenesis and up-regulating osteoblastogenesis through regulation of the RANK/RANKL/osteoprotegerin pathway [331] (Fig. 9C).
Osteoporosis, the main cause of bone fracture among the elderly, is a systemic skeletal disorder characterized by low bone mass, micro architectural deterioration of bone tissue. Bisphosphonates refer to one of the most commonly used drugs for prevention and treatment of osteoporosis currently. Bisphosphonates induce apoptosis of osteoclasts, slowing bone loss and inhibiting the digestion of bone. However, prescription of even mild versions of bisphosphonates such as alendronic acid (Forsamax) may result in a medical reaction known as medication-related osteonecrosis of the jaw, a condition manifested by progressive bone destruction in the maxillofacial region of patients. Scientists have resorted to the marine ecosystem to search alternatives for combating osteoporosis-related bone resorption by modulating immune mediators such as transcription factors, immune receptors and key factors like RANKL/osteoprotegerin.
Marine-derived organisms also show considerable effects in remineralization of bone defects in disease models. The gastrointestinal digests obtained from the abalone Haliotis discus hannai have been used with fish collagen and the polymer poly(ε-caprolactone) to create experimental scaffolds via 3D printing for bone tissue engineering. Marine mesenchymal stem cells were seeded onto the fabricated scaffolds and the resultant constructs were implanted into rabbit tibia. The seeded scaffolds that contained osteogenic abalone intestine gastrointestinal digests demonstrated strong osteoinduction capability in the rabbit tibia model, with augmented mineralization levels and alkaline phosphatase activity [399].
Osteoarthritis causes chronic pain of joint involving the entire joint. In the presence of the nacre extract, which was able to induce the early calcium precipitation in cells (after 7 days), the expression levels of osteogenic markers, including osteocalcin, osteopontin and Runt-related transcription factor, were higher than in normal with a dose-dependent manner. Nacre extract is likely to stimulate MC3T3-E1 and osteoarthritic osteoblasts in vitro through containing ethanol-soluble diffusible factors. Thus, nacre extract was able to restore a physiological phenomenon and induce mineralization of osteoarthritis osteoblasts [400]. However, no in vivo experiments have been conducted owing possibly to the immunogenic properties of nacre.
Biselyngbyasides are macrolides derived from Lyngbya sp, a marine cyanobacterium. They display potent cytotoxicity against a great variety of human cancer cells. A study showed that biselyngbyasides were potent inhibitors of the sarco/endoplasmic reticulum calcium-ATPases, the Ca2+-pumps responsible for establishing calcium ion concentration gradient across the sacrco/endoplasmic reticulum membrane. By binding specifically to cancer cells and inhibition of the aforementioned calcium pumps, biselyngbyasides activates the endoplasmic reticulum stress response in the cancer cells and results in apoptotic cell death [401]. Furthermore, biselyngbyasides can inhibit RANKL-induced osteoclastogenesis and induce apoptosis of mature osteoclasts in murine RAW 264.7 cells at a low concentration [402].
Histone deacetylases (HDACs) play significant roles in regulating gene expression in cells epigenetically. They are emerging as therapeutic targets for treating an array of solid tumors and hematological malignancies. Largazole, extracted from the marine cyanobacterium Symploca sp, is a potent inhibitor of HDACs. It has already been approved by the FDA for cancer treatment [403]. Histone deacetylase inhibitors have also been used to treat non-malignant diseases involving pathological bone loss. Inhibition of HDACs by largazole results in increased Runx2 activation. This, in turn, suppresses bone resorption and increases bone formation, repair and regeneration [404]. Soft corals such as Sinularia maxima and Lobophytum crassum show potent antioxidant activity (peroxyl radical-scavenging and reducing activity) on pre-osteoblastic MC3T3-E1 cells and anti-osteoporosis capacity on pre-osteoclastic RAW 264.7 cells [405].
3.6.3. Diabetes
Polyphenol obtained from marine algae has the ability to control diabetes mellitus. Experiments in vitro and in vivo have testified that polyphenol extracted from the brown macroalga Lessonia trabeculate significantly attenuated hyperglycemia and hyperlipidemia in high-fat diet and streptozotocin-induced diabetic rats. Purified polyphenol extracts exhibited superior activities such as antioxidant, α-glucosidase and lipase inhibition. Compared with control diabetic rats, fasting blood glucose levels and insulin levels are lower in diabetic rats treated with polyphenol extracts [406]. A dieckol-rich extract derived from the brown alga Ecklonia cava was used for pre-diabetic human subjects in a randomized, double-blind clinical trial. The results demonstrated that the levels of postprandial hyperglycemia, insulin and C-peptide were significantly reduced after 12 weeks [407].
Bioactive marine peptides are efficient in management of type 2 diabetes mellitus. The proposed mechanisms include protection of pancreatic β-cells, enhancing secretion of insulin stimulated by glucose, influence on the insulin sensitivity and its signaling pathways, and inhibiting bioactive peptides to dipeptidyl peptidase IV, α-amylase and α-glucosidase activities [408].
4. Other applications
4.1. Cosmeceutical applications
Marine-derived biomaterials also have great cosmeceutical potential. Recently, the combination of cosmeceuticals with bioactive ingredients obtained from marine organisms has become the hallmark of cosmetic industries [409].
Biologically-active proteins and peptides from marine sources possess a broad range of biological activities, including antioxidant, anti-aging, anti-photoaging and antimicrobial activities. Thus, they have attracted great attention in the pharmaceutical and cosmeceutical industries. Collagen is the major component of skin and it may decrease with age. This is the main reason why collagen is utilized extensive in cosmeceuticals like skin and hair care. Collagen obtained from fish, jellyfish and cuttlefish has been investigated for its moisturizing, regenerating and film-forming properties. Collagen fillers are also used as hydrogels to produce cosmetic face masks [410,411]. Collagen is administered via subcutaneous injection for repairing dermatological defects, such as the reduction of wrinkles and healing of acne scars [412]. Bioactive peptides are obtained from organisms and seafood waste products through enzymatic hydrolysis. These bioactive peptides are employed for anti-aging, light protection, whitening, anti-wrinkle and skin soothing [409].
Chitosan is another promising cosmeceutical agent because of its biocompatibility, high charge density and muco-adhesive property. Blending chitosan with hyaluronic acid and collagen promotes hair health such as increase in thickness and mechanical properties [413]. Chitosan oligosaccharide inhibits undesirable changes in hairless mouse skin caused by 10 weeks of ultraviolet light radiation [414]. Chitosan also exhibits excellent properties to vehiculate active ingredients for cosmeceuticals [415]. Sponge chitin-based membranes exhibit excellent mechanical stability and electrochemical properties due to the specific structure of the chitinous skeleton derived from demosponges [416]. Other polysaccharides used as cosmetics include fucoidans, alginate and laminarin, carrageenan and ulvan [417]. For example, pre-processed laminarin protects skin from ultraviolet radiation. The proposed mechanisms for this protection include decreased oxidative stress and increased antioxidant enzyme production [418].
Hyaluronic acid is still the most important ingredient in cosmetics because of its ability to replenish skin moisture and make skin softer, smoother and more radiant [410]. Hyaluronic acid may be cross-linked with different materials, such as 1,4-butanediol diglycidyl ether, to render it more resistant to enzymatic degradation [419]. Hyaluronic acid-based formulations have been used for skin rejuvenation. These formulations, which include gels, creams, intra-dermal filler injections, dermal fillers, facial fillers, autologous fat gels, lotion, serum and implants, exhibit remarkable anti-wrinkle, anti-nasolabial fold formation, anti-aging and space-filling properties [410]. Fabrication of hyaluronic acid into different types of porous and 3D structures widens its application in cosmeceuticals [420].
4.2. Food applications
Marine-derived products have been utilized extensively in the food industry as ingredients, additives, preservatives and packaging materials. The last decade has witnessed an upsurge of interest by consumers toward health promotion food products. The health promotion effects of marine-derived ingredients are supported by growing evidence from recent studies. Collagen undergoes degradation with age. Hence, a lot of interest has been devoted to the use of marine-derived collagen for anti-aging applications. Marine-derived collagen is added to health-promoting diets as functional and nutritional ingredients. In addition, collagen derived from marine sources are used as food additives. To reduce food deterioration and spoilage, marine collagen can also function as antioxidants in films or coatings [421].
Marine-derived chitosan is used increasingly as food preservatives and fruit coatings because of its antimicrobial properties [420]. Chitosan has also been employed in food packaging to replace plastic packaging materials because of its biocompatibility, edibility, bioadhesivity and biodegradability [422].
Marine-derived polysaccharides have also been used in food industry. For example, alginate has potential to be employed as an eco-friendly food packaging material by reducing the dependence on fossil resources as well as cutting down carbon output [423]. Alginate has also been used as coatings for fruits and vegetables and as a gelling, thickening, stabilizing or emulsifying agent in food industry [424].
A novel polyvinyl alcohol biocomposite film containing cellulose nanocrystals extracted from seaweed has been used as food packaging because of its antimicrobial and antioxidant properties [425]. Seafood wastes contain important bioactive polysaccharides that are good candidates for food additives. For example, cellulose nanofibers isolated from the waste of brown algae have been used as milk thickeners because of their high viscosity and shear-thinning rheological property [426].
5. Perspectives and future directions
Environment pollution and energy shortage are the driving forces in human's quest for renewable and envionmental-friendly resources. The ocean appears to be a perfect choice. More than 70% of the earth's surface is covered by marine environment, the majority of which remain unexplored. Continuous research in this exciting field has unveiled a wide spectrum of biomaterials with superior properties including biocompatibility, biodegradability and bioactivity. Marine-derived materials have generated considerable inspirations as raw materials for bioengineering or building blocks for biomimetics [219,312]. The use of marine-derived materials has undergone a long journey from ancient times. Today, utilization of marine biomaterials has progressed to the industrial level. This is attributed to the advances in research and technology on processing of these biomaterials.
The safety of marine-derived biomaterials has become the principal concerns among users. Marine-derived materials are obtained from heterogeneous sources and most of them contain active biological macromolecules that have unknown immunogenicity. Residual proteins from marine-based biomaterials may lead to hypersensitivity reaction once a certain threshold is exceeded in human body. In addition, the microstructure and physiochemical properties of marine biomaterials are difficult to be standardized owing to the high batch-to-batch variations. Therefore, it is not easy for these biomaterials to be used in the biomedical arena because of stringent quality control requirements. Because the evaluation criteria for these biomaterials has not been well established, there still exists safety concerns associated with the use of marine-derived biomaterials in medicine. Thus, further study and refinement are necessary.
Despite the immense potential of marine-derive biomaterials, the conversion of raw ingredients into ready-to-use market products require further research. A multidisciplinary and multi-scale approach that covers the structures of biomaterials and the mechanisms of thier conversion into useable products is required. Further research and design of new products are impossible if the mechanisms of their formation and their structural characteristics are unknown [109]. Recent research principally follows the property-application pattern, which explores a bioactive material and identifies its market potential. This pattern narrows the gap between marine-derived biomaterials and their potential markets. Nevetheless, by following this pattern, researchers merely answer the question ‘how’ but do not explain ‘why’. Deeper research that digs into fundamental understand of these biomaterials is required to exploit their full market potential. This will involve financial support from the government as well as collaboration among scientists from interdisciplinary fields.
Declaration of competing interest
The authors declare no conflict of interests.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81722015 and 81870805), the Shaanxi Key Scientific and Technological Innovation Team(2020TD-033), the Innovative research team of high-level local universities in Shanghai and the Oral and maxillofacial regeneration and functional restoration.
Footnotes
Peer review under responsibility of KeAi Communications Co., Ltd.
References
- 1.Ehrlich H. first ed. Springer Nature Switzerland AG; Switzerland: 2019. Marine Biological Materials of Invertebrate Origin. [Google Scholar]
- 2.Mora C., Tittensor D.P., Adl S., Simpson A.G.B., Worm B. How many species are there on earth and in the ocean? PLoS Biol. 2011;9 doi: 10.1371/journal.pbio.1001127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Russo P., Kisialiou A., Lamonaca P., Moroni R., Prinzi G., Fini M. New drugs from marine organisms in alzheimer's disease. Mar. Drugs. 2016;14:5–21. doi: 10.3390/md14010005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Khrunyk Y., Lach S., Petrenko I., Ehrlich H. Progress in modern marine biomaterials research. Mar. Drugs. 2020;18:589–635. doi: 10.3390/md18120589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nigrelli R.F., F Stempien M., Jr., Ruggieri G.D., Liguori V.R., Cecil J.T. Substances of potential biomedical importance from marine organisms, fed. Proc. 1967;26:1197–1205. [PubMed] [Google Scholar]
- 6.West T.L., Brustein D.D. Freeze-dried bone and coralline implants compared in the dog. J. Periodontol. 1985;56:348–351. doi: 10.1902/jop.1985.56.6.348. [DOI] [PubMed] [Google Scholar]
- 7.White E., Shors E.C. Biomaterial aspects of interpore-200 porous hydroxyapatite, dent. Clin. North Am. 1986;30:49–67. [PubMed] [Google Scholar]
- 8.Lopez E., Gill G.A., Camprasse G., Camprasse S., Lallier F. Soldering without transition (osteoassimilation) between the human maxillary bone and a compact dental implant in natural calcite from marine invertebrate. C. R. Acad. Sci. III. 1989;309:203–210. [PubMed] [Google Scholar]
- 9.Demers C., Reggie Hamdy C., Corsi K., Chellat F., Tabrizian M., Yahia L. Natural coral exoskeleton as a bone graft substitute: a review. Bio Med. Mater. Eng. 2002;12:15–35. [PubMed] [Google Scholar]
- 10.Chiroff R.T., White E.W., Weber K.N., Roy D.M. Tissue ingrowth of replamineform implants. J. Biomed. Mater. Res. 1975;9:29–45. doi: 10.1002/jbm.820090407. [DOI] [PubMed] [Google Scholar]
- 11.Peters W. Occurrence of chitin in Mollusca. Comp. Biochem. Physiol. Part B Comp. Biochem. 1972;41:541–550. [Google Scholar]
- 12.Ehrlich H., Maldonado M., Spindler K.D., Eckert C., Born T., Hanke R., Goebel C., Simon P., Heinemann S., Worch H. First evidence of chitin as a component of the skeletal fibers of marine sponges. Part I. Verongidae (demospongia: Porifera) J. Exp. Zool. B Mol. Dev. Evol. 2007;308:347–356. doi: 10.1002/jez.b.21156. [DOI] [PubMed] [Google Scholar]
- 13.Ehrlich H., Krautter M., Hanke T., Simon P., Knieb C., Heinemann S., Worch H. First evidence of the presence of chitin in skeletons of marine sponges. Part II. Glass sponges (hexactinellida: Porifera) J. Exp. Zool. B Mol. Dev. Evol. 2007;308:473–483. doi: 10.1002/jez.b.21174. [DOI] [PubMed] [Google Scholar]
- 14.Lahiji A., Sohrabi A., Hungerford D.S., Frondoza C.G. Chitosan supports the expression of extracellular matrix proteins in human osteoblasts and chondrocytes. J. Biomed. Mater. Res. 2000;51:586–595. doi: 10.1002/1097-4636(20000915)51:4<586::aid-jbm6>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- 15.Synowiecki J., Al-Khateeb N.A. Production, properties, and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nutr. 2003;43:145–171. doi: 10.1080/10408690390826473. [DOI] [PubMed] [Google Scholar]
- 16.Song E., Yeon Kim S., Chun T., Byun H.-J., Lee Y.M. Collagen scaffolds derived from a marine source and their biocompatibility. Biomaterials. 2006;27:2951–2961. doi: 10.1016/j.biomaterials.2006.01.015. [DOI] [PubMed] [Google Scholar]
- 17.Salvatore L., Gallo N., Natali M.L., Campa L., Lunetti P., Madaghiele M., Blasi F.S., Corallo A., Capobianco L., Sannino A. Marine collagen and its derivatives: versatile and sustainable bio-resources for healthcare. Mater. Sci. Eng. C Mater. Biol. Appl. 2020;113:110963–110980. doi: 10.1016/j.msec.2020.110963. [DOI] [PubMed] [Google Scholar]
- 18.Govindharaj M., Roopavath U.K., Rath S.N. Valorization of discarded marine eel fish skin for collagen extraction as a 3D printable blue biomaterial for tissue engineering. J. Clean. Prod. 2019;230:412–419. [Google Scholar]
- 19.Sousa R.O., Alves A.L., Carvalho D.N., Martins E., Oliveira C., Silva T.H., Reis R.L. Acid and enzymatic extraction of collagen from atlantic cod (Gadus morhua) swim bladders envisaging health-related applications. J. Biomater. Sci. Polym. Ed. 2020;31:20–37. doi: 10.1080/09205063.2019.1669313. [DOI] [PubMed] [Google Scholar]
- 20.Ferrario C., Leggio L., Leone R., Di Benedetto C., Guidetti L., Cocce V., Ascagni M., Bonasoro F., La Porta C.A.M., Carnevali M.D.C. Marine-derived collagen biomaterials from echinoderm connective tissues. Mar. Environ. Res. 2017;128:46–57. doi: 10.1016/j.marenvres.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 21.Coelho R.C.G., Marques A.L.P., Oliveira S.M., Diogo G.S., Pirraco R.P., Moreira-Silva J., Xavier J.C., Reis R.L., Silva T.H., Mano J.F. Extraction and characterization of collagen from antarctic and sub-antarctic squid and its potential application in hybrid scaffolds for tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;78:787–795. doi: 10.1016/j.msec.2017.04.122. [DOI] [PubMed] [Google Scholar]
- 22.Tziveleka L.A., Sapalidis A., Kikionis S., Aggelidou E., Demiri E., Kritis A., Ioannou E., Roussis V. Hybrid sponge-like scaffolds based on ulvan and gelatin: design, characterization and evaluation of their potential use in bone tissue engineering. Materials (Basel) 2020;13:1763–1782. doi: 10.3390/ma13071763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blunt J.W., Copp B.R., Keyzers R.A., Munro M.H.G., Prinsep M.R. Marine natural products. Nat. Prod. Rep. 2016;33:382–431. doi: 10.1039/c5np00156k. [DOI] [PubMed] [Google Scholar]
- 24.Corinaldesi C., Barone G., Marcellini F., Dell'Anno A., Danovaro R. Marine microbial-derived molecules and their potential use in cosmeceutical and cosmetic products. Mar. Drugs. 2017;15:1–21. doi: 10.3390/md15040118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Huang H., Song Y., Li X., Wang X., Ling C., Qin X., Zhou Z., Li Q., Wei X., Ju J. Abyssomicin monomers and dimers from the marine-derived streptomyces koyangensis SCSIO 5802. J. Nat. Prod. 2018;81:1892–1898. doi: 10.1021/acs.jnatprod.8b00448. [DOI] [PubMed] [Google Scholar]
- 26.Cui J., Li Y., Wang S., Chi Y., Hwang H., Wang P. Directional preparation of anticoagulant-active sulfated polysaccharides from enteromorpha prolifera using artificial neural networks. Sci. Rep. 2018;8:3062–3070. doi: 10.1038/s41598-018-21556-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gao Y., Zhang L., Jiao W. Chapter seven - marine glycan-derived therapeutics in China. In: Zhang L., editor. Glycans and Glycosaminoglycans as Clinical Biomarkers and Therapeutics - Part B. Elsevier.; Amsterdam: 2019. pp. 113–134. [Google Scholar]
- 28.Pereira L., Critchley A.T. The COVID 19 novel coronavirus pandemic 2020: seaweeds to the rescue? Why does substantial, supporting research about the antiviral properties of seaweed polysaccharides seem to go unrecognized by the pharmaceutical community in these desperate times? J. Appl. Phycol. 2020;32:1875–1877. doi: 10.1007/s10811-020-02143-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Venkatesan J., Anil S., Rao S., Bhatnagar I., Kim S. Sulfated polysaccharides from macroalgae for bone tissue regeneration. Curr. Pharmaceut. Des. 2019;25:1200–1209. doi: 10.2174/1381612825666190425161630. [DOI] [PubMed] [Google Scholar]
- 30.Tsurkan M.V., Voronkina A., Khrunyk Y., Wysokowski M., Petrenko I., Ehrlich H. Progress in chitin analytics. Carbohydr. Polym. 2021;252:117204. doi: 10.1016/j.carbpol.2020.117204. [DOI] [PubMed] [Google Scholar]
- 31.El Knidri H., Belaabed R., Addaou A., Laajeb A., Lahsini A. Extraction, chemical modification and characterization of chitin and chitosan. Int. J. Biol. Macromol. 2018;120:1181–1189. doi: 10.1016/j.ijbiomac.2018.08.139. [DOI] [PubMed] [Google Scholar]
- 32.Brunner E., Richthammer P., Ehrlich H., Paasch S., Simon P., Ueberlein S., van Pée K.H. Chitin-based organic networks: an integral part of cell wall biosilica in the diatom Thalassiosira pseudonana. Angew. Chem. Int. Ed. 2009;48:9724–9727. doi: 10.1002/anie.200905028. [DOI] [PubMed] [Google Scholar]
- 33.Khattak S., Wahid F., Liu L.P., Jia S.R., Chu L.Q., Xie Y.Y., Li Z.X., Zhong C. Applications of cellulose and chitin/chitosan derivatives and composites as antibacterial materials: current state and perspectives. Appl. Microbiol. Biotechnol. 2019;103:1989–2006. doi: 10.1007/s00253-018-09602-0. [DOI] [PubMed] [Google Scholar]
- 34.Valcarcel J., Novoa-Carballal R., Pérez-Martín R.I., Reis R.L., Vázquez J.A. Glycosaminoglycans from marine sources as therapeutic agents. Biotechnol. Adv. 2017;35:711–725. doi: 10.1016/j.biotechadv.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 35.Cunha L., Grenha A. Sulfated seaweed polysaccharides as multifunctional materials in drug delivery applications. Mar. Drugs. 2016;14:42–82. doi: 10.3390/md14030042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Apostolova E., Lukova P., Baldzhieva A., Katsarov P., Nikolova M., Iliev I., Peychev L., Trica B., Oancea F., Delattre C., Kokova V. Immunomodulatory and anti-inflammatory effects of fucoidan: a review. Polymers. 2020;12:2338–2359. doi: 10.3390/polym12102338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Felician F.F., Xia C., Qi W., Xu H. Collagen from marine biological sources and medical applications. Chem. Biodivers. 2018;15 doi: 10.1002/cbdv.201700557. [DOI] [PubMed] [Google Scholar]
- 38.Cicciù M., Fiorillo L., Cervino G. Chitosan use in dentistry: a systematic review of recent clinical studies. Mar. Drugs. 2019;17:417–431. doi: 10.3390/md17070417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mishra A., Pandey R.K., Manickam N. Antibacterial effect and physical properties of chitosan and chlorhexidine-cetrimide-modified glass ionomer cements. J. Indian Soc. Pedod. Prev. Dent. 2017;35:28–33. doi: 10.4103/0970-4388.199224. [DOI] [PubMed] [Google Scholar]
- 40.Wysokowski M., Petrenko I., Stelling A L., Stawski D., Jesionowski T., Ehrlich H. Chitin as a versatile template for extreme biomimetics. Polymers. 2015;7:235–265. [Google Scholar]
- 41.Petrenko I., Bazhenov V.V., Galli R., Wysokowski M., Fromont J., Schupp P.J., Stelling A.L., Niederschlag E., Stöcker H., Kutsova V.Z., Jesionowski T., Ehrlich H. Chitin of poriferan origin and the bioelectrometallurgy of copper/copper oxide. Int. J. Biol. Macromol. 2017;104:1626–1632. doi: 10.1016/j.ijbiomac.2017.01.084. [DOI] [PubMed] [Google Scholar]
- 42.Morkaew T., Pinyakong O., Tachaboonyakiat W. Structural effect of quaternary ammonium chitin derivatives on their bactericidal activity and specificity. Int. J. Biol. Macromol. 2017;101:719–728. doi: 10.1016/j.ijbiomac.2017.03.159. [DOI] [PubMed] [Google Scholar]
- 43.Yu N., Cai T., Sun Y., Jiang C., Xiong H., Li Y., Peng H. A novel antibacterial agent based on AgNPs and Fe3O4 loaded chitin microspheres with peroxidase-like activity for synergistic antibacterial activity and wound-healing. Int. J. Pharm. 2018;552:277–287. doi: 10.1016/j.ijpharm.2018.10.002. [DOI] [PubMed] [Google Scholar]
- 44.Sun Z., Shi C., Wang X., Fang Q., Huang J. Synthesis, characterization, and antimicrobial activities of sulfonated chitosan. Carbohydr. Polym. 2017;155:321–328. doi: 10.1016/j.carbpol.2016.08.069. [DOI] [PubMed] [Google Scholar]
- 45.Soares Da Costa D., Reis R.L., Pashkuleva I. Sulfation of glycosaminoglycans and its implications in human health and disorders. Annu. Rev. Biomed. Eng. 2017;19:1–26. doi: 10.1146/annurev-bioeng-071516-044610. [DOI] [PubMed] [Google Scholar]
- 46.Vasconcelos A., Pomin V. the sea as a rich source of structurally unique glycosaminoglycans and mimetics. Microorganisms. 2017;5:51–68. doi: 10.3390/microorganisms5030051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Houghton D., Wilcox M.D., Chater P.I., Brownlee I.A., Seal C.J., Pearson J.P. Biological activity of alginate and its effect on pancreatic lipase inhibition as a potential treatment for obesity. Food Hydrocolloids. 2015;49:18–24. doi: 10.1016/j.foodhyd.2015.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nakazono S., Cho K., Isaka S., Abu R., Yokose T., Murata M., Ueno M., Tachibana K., Hirasaka D.K. Kim. Anti-obesity effects of enzymatically-digested alginate oligomer in mice model fed a high-fat-diet. Bioact. Carbohydrates Diet. Fibre. 2016;7:1–8. [Google Scholar]
- 49.Idota Y., Kogure Y., Kato T., Ogawa M., Kobayashi S., Kakinuma C., Yano K., Arakawa H., Miyajima C., Kasahara F. Cholesterol-lowering effect of calcium alginate in rats. Biol. Pharm. Bull. 2016;39:62–67. doi: 10.1248/bpb.b15-00503. [DOI] [PubMed] [Google Scholar]
- 50.Freeman F.E., Kelly D.J. Tuning alginate bioink stiffness and composition for controlled growth factor delivery and to spatially direct MSC fate within bioprinted tissues. Sci. Rep. 2017;7:17042–17053. doi: 10.1038/s41598-017-17286-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tabriz A.G., Hermida M.A., Leslie N.R., Shu W. Three-dimensional bioprinting of complex cell laden alginate hydrogel structures. Biofabrication. 2015;7 doi: 10.1088/1758-5090/7/4/045012. 045012-045022. [DOI] [PubMed] [Google Scholar]
- 52.Dalheim M., Vanacker J., Najmi M.A., Aachmann F.L., Strand B.L., Christensen B.E. Efficient functionalization of alginate biomaterials. Biomaterials. 2016;80:146–156. doi: 10.1016/j.biomaterials.2015.11.043. [DOI] [PubMed] [Google Scholar]
- 53.Zhu B., Ni F., Sun Y., Zhu X., Yin H., Yao Z., Du Y. Insight into carrageenases: major review of sources, category, property, purification method, structure, and applications. Crit. Rev. Biotechnol. 2018;38:1261–1276. doi: 10.1080/07388551.2018.1472550. [DOI] [PubMed] [Google Scholar]
- 54.Sedayu B.B., Cran M.J., Bigger S.W. A review of property enhancement techniques for carrageenan-based films and coatings. Carbohydr. Polym. 2019;216:287–302. doi: 10.1016/j.carbpol.2019.04.021. [DOI] [PubMed] [Google Scholar]
- 55.Khotimchenko M., Tiasto V., Kalitnik A., Begun M., Khotimchenko R., Leonteva E., Bryukhovetskiy I., Khotimchenko Y. Antitumor potential of carrageenans from marine red algae. Carbohydr. Polym. 2020;246:116568. doi: 10.1016/j.carbpol.2020.116568. [DOI] [PubMed] [Google Scholar]
- 56.Cosenza V.A., Navarro D.A., Pujol C.A., Damonte E.B., Stortz C.A. Partial and total C-6 oxidation of gelling carrageenans. Modulation of the antiviral activity with the anionic character. Carbohydr. Polym. 2015;128:199–206. doi: 10.1016/j.carbpol.2015.04.030. [DOI] [PubMed] [Google Scholar]
- 57.Dos Santos-Fidencio G.C., Gonçalves A.G., Noseda M.D., Duarte M.E.R., Ducatti D.R.B. Effects of carboxyl group on the anticoagulant activity of oxidized carrageenans. Carbohydr. Polym. 2019;214:286–293. doi: 10.1016/j.carbpol.2019.03.057. [DOI] [PubMed] [Google Scholar]
- 58.Hauser C., Thielmann J., Muranyi P. Chapter 46 - organic acids: usage and potential in antimicrobial packaging. In: Barros-Velázquez J., editor. Antimicrobial Food Packaging. Academic Press; San Diego: 2016. pp. 563–580. [Google Scholar]
- 59.Shankar S., Reddy J.P., Rhim J.W., Kim H.Y. Preparation, characterization, and antimicrobial activity of chitin nanofibrils reinforced carrageenan nanocomposite films. Carbohydr. Polym. 2015;117:468–475. doi: 10.1016/j.carbpol.2014.10.010. [DOI] [PubMed] [Google Scholar]
- 60.David S., Shani Levi C., Fahoum L., Ungar Y., Meyron-Holtz E.G., Shpigelman A., Lesmes U. Revisiting the carrageenan controversy: do we really understand the digestive fate and safety of carrageenan in our foods? Food Funct. 2018;9:1344–1352. doi: 10.1039/c7fo01721a. [DOI] [PubMed] [Google Scholar]
- 61.Qin H.M., Gao D., Zhu M., Li C., Zhu Z., Wang H., Liu W., Tanokura M., Lu F. Biochemical characterization and structural analysis of ulvan lyase from marine alteromonas sp. Reveals the basis for its salt tolerance. Int. J. Biol. Macromol. 2020;147:1309–1317. doi: 10.1016/j.ijbiomac.2019.10.095. [DOI] [PubMed] [Google Scholar]
- 62.Gao J., Du C., Chi Y., Zuo S., Ye H., Wang P. Cloning, expression, and characterization of a new PL25 family ulvan lyase from marine bacterium alteromonas sp. A321. Mar. Drugs. 2019;17:568–578. doi: 10.3390/md17100568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Li P., Wen S., Sun K., Zhao Y., Chen Y. Structure and bioactivity screening of a low molecular weight ulvan from the green alga ulothrix flacca. Mar. Drugs. 2018;16:281–293. doi: 10.3390/md16080281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Berri M., Olivier M., Holbert S., Dupont J., Demais H., Le Goff M., Collen P.N. Ulvan from ulva armoricana (chlorophyta) activates the PI3K/Akt signalling pathway via TLR4 to induce intestinal cytokine production. Algal Res. 2017;28:39–47. [Google Scholar]
- 65.Berri M., Slugocki C., Olivier M., Helloin E., Jacques I., Salmon H., Demais H., Le Goff M., Collen P.N. Marine-sulfated polysaccharides extract of ulva armoricana green algae exhibits an antimicrobial activity and stimulates cytokine expression by intestinal epithelial cells. J. Appl. Phycol. 2016;28:2999–3008. [Google Scholar]
- 66.Chi Y., Zhang M., Wang X., Fu X., Guan H., Wang P. Ulvan lyase assisted structural characterization of ulvan from ulva pertusa and its antiviral activity against vesicular stomatitis virus. Int. J. Biol. Macromol. 2020;157:75–82. doi: 10.1016/j.ijbiomac.2020.04.187. [DOI] [PubMed] [Google Scholar]
- 67.Tziveleka L.A., Ioannou E., Roussis V. Ulvan, a bioactive marine sulphated polysaccharide as a key constituent of hybrid biomaterials: a review. Carbohydr. Polym. 2019;218:355–370. doi: 10.1016/j.carbpol.2019.04.074. [DOI] [PubMed] [Google Scholar]
- 68.Thanh T.T.T., T Quach T.M., Nguyen T.N., Vu Luong D., Bui M.L., Tran T.T. Van. Structure and cytotoxic activity of ulvan extracted from green seaweed ulva lactuca. Int. J. Biol. Macromol. 2016;93:695–702. doi: 10.1016/j.ijbiomac.2016.09.040. [DOI] [PubMed] [Google Scholar]
- 69.Li B., Xu H., Wang X., Wan Y., Jiang N., Qi H., Liu X. Antioxidant and antihyperlipidemic activities of high sulfate content purified polysaccharide from ulva pertusa. Int. J. Biol. Macromol. 2020;146:756–762. doi: 10.1016/j.ijbiomac.2019.11.061. [DOI] [PubMed] [Google Scholar]
- 70.Wahlström N., Steinhagen S., Toth G., Pavia H., Edlund U. Ulvan dialdehyde-gelatin hydrogels for removal of heavy metals and methylene blue from aqueous solution. Carbohydr. Polym. 2020;249:116841–116853. doi: 10.1016/j.carbpol.2020.116841. [DOI] [PubMed] [Google Scholar]
- 71.Wang Y., Xing M., Cao Q., Ji A., Liang H., Song S. Biological activities of fucoidan and the factors mediating its therapeutic effects: a review of recent studies. Mar. Drugs. 2019;17:183–200. doi: 10.3390/md17030183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lin Y., Qi X., Liu H., Xue K., Xu S., Tian Z. The anti-cancer effects of fucoidan: a review of both in vivo and in vitro investigations. Canc. Cell Int. 2020;20:154–167. doi: 10.1186/s12935-020-01233-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhang W., Oda T., Yu Q., Jin J.O. Fucoidan from Macrocystis pyrifera has powerful immune-modulatory effects compared to three other fucoidans. Mar. Drugs. 2015;13:1084–1104. doi: 10.3390/md13031084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Tabarsa M., Dabaghian E.H., You S.G., Yelithao K., Cao R.A., Rezaei M., Alboofetileh M., Bita S. The activation of NF-ΚB and MAPKs signaling pathways of RAW264.7 murine macrophages and natural killer cells by fucoidan from Nizamuddinia zanardinii. Int. J. Biol. Macromol. 2020;148:56–67. doi: 10.1016/j.ijbiomac.2020.01.125. [DOI] [PubMed] [Google Scholar]
- 75.Flórez-Fernández N., Torres M.D., González-Muñoz M.J., Domínguez H. Potential of intensification techniques for the extraction and depolymerization of fucoidan. Algal Res. 2018;30:128–148. [Google Scholar]
- 76.Jang J.Y., Moon S.Y., Joo H.G. Differential effects of fucoidans with low and high molecular weight on the viability and function of spleen cells. Food Chem. Toxicol. 2014;68:234–238. doi: 10.1016/j.fct.2014.03.024. [DOI] [PubMed] [Google Scholar]
- 77.Atashrazm F., Lowenthal M.R., Woods G.M., Holloway A.F., Dickinson J.L., Fucoidan, Cancer A multifunctional molecule with anti-tumor potential. Mar. Drugs. 2015;13:2327–2346. doi: 10.3390/md13042327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wang X., Yi K., Zhao Y. Fucoidan inhibits amyloid-β-induced toxicity in transgenic: Caenorhabditis elegans by reducing the accumulation of amyloid-β and decreasing the production of reactive oxygen species. Food Funct. 2018;9:552–560. doi: 10.1039/c7fo00662d. [DOI] [PubMed] [Google Scholar]
- 79.Jin W., Ren L., Liu B., Zhang Q., Zhong W. Structural features of sulfated glucuronomannan oligosaccharides and their antioxidant activity. Mar. Drugs. 2018;16:291–302. doi: 10.3390/md16090291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cong Q., Chen H., Liao W., Xiao F., Wang P., Qin Y., Dong Q., Ding K. Structural characterization and effect on anti-angiogenic activity of a fucoidan from sargassum fusiforme. Carbohydr. Polym. 2016;136:899–907. doi: 10.1016/j.carbpol.2015.09.087. [DOI] [PubMed] [Google Scholar]
- 81.Kim B.S., Park J.-Y., Kang H.-J., Kim H.-J., Lee J. Fucoidan/FGF-2 induces angiogenesis through JNK- and P38-mediated activation of AKT/MMP-2 signalling. Biochem. Biophys. Res. Commun. 2014;450:1333–1338. doi: 10.1016/j.bbrc.2014.06.137. [DOI] [PubMed] [Google Scholar]
- 82.Goldberga I., Li R., Duer M.J. Collagen structure-function relationships from solid-state NMR spectroscopy. Acc. Chem. Res. 2018;51:1621–1629. doi: 10.1021/acs.accounts.8b00092. [DOI] [PubMed] [Google Scholar]
- 83.Bazrafshan Z., Stylios G.K. International journal of biological macromolecules spinnability of collagen as a biomimetic material: a review. Int. J. Biol. Macromol. 2019;129:693–705. doi: 10.1016/j.ijbiomac.2019.02.024. [DOI] [PubMed] [Google Scholar]
- 84.Holmes D.F., Lu Y., Starborg T., Kadler K.E. Chapter three - collagen fibril assembly and function. In: Litscher E.S., Wassarman, editors. vol. 130. Elsevier Inc.; Amsterdam: 2018. pp. 107–142. (Extracellular Matrix and Egg Coats). [Google Scholar]
- 85.Sorushanova A., Delgado L.M., Wu Z., Shologu N., Kshirsagar A., Raghunath R., Mullen A.M., Bayon Y., Pandit A., Raghunath M. The collagen suprafamily: from biosynthesis to advanced biomaterial development. Adv. Mater. 2019;31:1801651–1801689. doi: 10.1002/adma.201801651. [DOI] [PubMed] [Google Scholar]
- 86.Gu L., Shan T., Ma Y., Tay F.R., Niu L. Novel biomedical applications of crosslinked collagen. Trends Biotechnol. 2018;37:464–491. doi: 10.1016/j.tibtech.2018.10.007. [DOI] [PubMed] [Google Scholar]
- 87.Panwar P., Butler G.S., Jamroz A., Azizi P., Overall M., Br D. Aging-associated modifications of collagen affect its degradation by matrix metalloproteinases preety. Matrix Biol. 2018;65:30–44. doi: 10.1016/j.matbio.2017.06.004. [DOI] [PubMed] [Google Scholar]
- 88.Squeglia F., Ruggiero A., Berisio R. Collagen degradation in tuberculosis pathogenesis: the biochemical consequences of hosting an undesired guest. Biochem. J. 2018;475:3123–3140. doi: 10.1042/BCJ20180482. [DOI] [PubMed] [Google Scholar]
- 89.Subasinghe S.K., Ogbuehi K.C., Dias G.J. Current perspectives on corneal collagen crosslinking (CXL) Graefes Arch. Clin. Exp. Ophthalmol. 2018;256:1363–1384. doi: 10.1007/s00417-018-3966-0. [DOI] [PubMed] [Google Scholar]
- 90.Böhm S., Strauß C., Stoiber S., Kasper C., Charwat V. Impact of source and manufacturing of collagen matrices on fibroblast cell growth and platelet aggregation. Materials (Basel) 2017;10:1086–1099. doi: 10.3390/ma10091086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Salvatore L., Gallo N., Natali M.L., Campa L., Lunetti P., Madaghiele M., Blasi S.F., Corallo A., Capobianco L., Sannino A. Marine collagen and its derivatives: versatile and sustainable bio-resources for healthcare. Mater. Sci. Eng. C. 2020;113:110963. doi: 10.1016/j.msec.2020.110963. [DOI] [PubMed] [Google Scholar]
- 92.Pal G.K., Suresh P.V. Sustainable valorisation of seafood by-products: recovery of collagen and developmentof collagen-based novel functional food ingredients. Innovat. Food Sci. Emerg. Technol. 2016;37:201–215. [Google Scholar]
- 93.Zheng M.H., Hinterketiser K., Solomon K., Kunert V., Pavlos N.J., Xu J. Collagen-derived biomaterials in bone and cartilage repair. Macromol. Symp. 2007;253:179–185. [Google Scholar]
- 94.Swatschek D., Schatton W., Kellermann J., Müller W.E.G., Kreuter J. Marine sponge collagen: isolation, characterization and effects on the skin parameters surface-PH, moisture and sebum. Eur. J. Pharm. Biopharm. 2002;53:107–113. doi: 10.1016/s0939-6411(01)00192-8. [DOI] [PubMed] [Google Scholar]
- 95.Barros A.A., Aroso I.M., Silva T.H., Mano J.F., Duarte A.R.C., Reis R.L. Water and carbon dioxide: green solvents for the extraction of collagen/gelatin from marine sponges. Acs Sustain. Chem. Eng. 2015;3:254–260. [Google Scholar]
- 96.Nagai N., Yunoki S., Suzuki T., Sakata M., Tajima K., Munekata M. Application of cross-linked salmon atelocollagen to the scaffold of human periodontal ligament cells. J. Biosci. Bioeng. 2004;97:389–394. doi: 10.1016/S1389-1723(04)70224-8. [DOI] [PubMed] [Google Scholar]
- 97.Mahboob S. Isolation and characterization of collagen from fish waste material- skin, scales and fins of catla catla and cirrhinus mrigala. J. Food Sci. Technol. 2015;52:4296–4305. doi: 10.1007/s13197-014-1520-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Zhang J., Sun Y., Zhao Y., Wei B., Xu C., He L., Oliveira C.L.P., Wang H. Centrifugation-induced fibrous orientation in fish-sourced collagen matrices. Soft Matter. 2017;13:9220–9228. doi: 10.1039/c7sm01871a. [DOI] [PubMed] [Google Scholar]
- 99.Heinemann B.S., Heinemann C., Ehrlich H., Meyer M., Baltzer H., Worch H., Hanke T. A novel biomimetic hybrid material made of silicified Collagen : perspectives for bone replacement. Adv. Eng. Mater. 2007;9:1061–1068. [Google Scholar]
- 100.Silva J.C., Barros A.A., Aroso I.M., Fassini D., Silva T.H., Reis R.L., Duarte A.R.C. Extraction of collagen/gelatin from the marine demosponge chondrosia reniformis (nardo, 1847) using water acidified with carbon dioxide – process optimization. Ind. Eng. Chem. Res. 2016;55:6891–7060. [Google Scholar]
- 101.Jafari H., Lista A., Siekapen M.M., Bohlouli P.G., Nie L., Alimoradi H., Shavandi A. Fish collagen: extraction, characterization, and applications for biomaterials engineering. Polymers (Basel) 2020;12:2230–2266. doi: 10.3390/polym12102230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yang H., Wang H., Zhao Y., Zhang H. Effect of heat treatment on the enzymatic stability of grass carp skin collagen and its ability to form fibrils in vitro. J. Sci. Food Agric. 2014;95:329–336. doi: 10.1002/jsfa.6724. [DOI] [PubMed] [Google Scholar]
- 103.Chen J., Gao K., Liu S., Wang S., Elango J., Bao B., Dong J., Liu N., Wu W. Fish collagen surgical compress repairing characteristics on wound healing process in vivo. Mar. Drugs. 2019;17:1–12. doi: 10.3390/md17010033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Moreira-silva J., Diogo G.S., Marques A.L.P., Silva T.H., Reis R.L. Chapter 2-marine collagen isolation and processing envisaging biomedical applications. In: Neves N.M., Reis R.L., editors. Biomaterials from Nature for Advanced Devices and Therapies. John Wiley & Sons, Inc.; New Jersey: 2016. pp. 16–36. [Google Scholar]
- 105.Ehrlich H., Heinemann S., Heinemann C., Simon P., V Bazhenov V., Shapkin N.P., Born R., Tabachnick K.R., Hanke T., Worch H. Nanostructural organization of naturally occurring composites—Part I: silica-collagen-based biocomposites. J. Nanomater. 2008;2008:145–152. [Google Scholar]
- 106.Heinemann S., Ehrlich H., Knieb C., Hanke T. Biomimetically inspired hybrid materials based on silicified collagen. Int. J. Mater. Res. 2007;98:603–608. [Google Scholar]
- 107.Ueberlein S., Machill S., Schupp P.J., Brunner E. Determination of the halogenated skeleton constituents of the marine demosponge Ianthella basta. Mar. Drugs. 2017;15:34–48. doi: 10.3390/md15020034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ehrlich H., Wysokowski M., Zółtowska-Aksamitowska S., Petrenko I., Jesionowski T. Collagens of poriferan origin. Mar. Drugs. 2018;16:1–21. doi: 10.3390/md16030079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Khrunyk Y., Lach S., Petrenko I., Ehrlich H. Progress in modern marine biomaterials research. Mar. Drugs. 2020;18:1–46. doi: 10.3390/md18120589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Jesionowski T., Norman M., Zółtowska-Aksamitowska S., Petrenko I., Joseph Y., Ehrlich H. Marine spongin: naturally prefabricated 3D scaffold-based biomaterial. Mar. Drugs. 2018;16:1–23. doi: 10.3390/md16030088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Petrenko I., Summers A.P., Simon P., Żółtowska-Aksamitowska S., Motylenko M., Schimpf C., Rafaja D., Roth F., Kummer K., Brendler E. Extreme biomimetics: preservation of molecular detail in centimeter-scale samples of biological meshes laid down by sponges. Sci. Adv. 2019;5 doi: 10.1126/sciadv.aax2805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tsurkan D., Wysokowski M., Petrenko I., Voronkina A., Khrunyk Y., Fursov A., Ehrlich H. Modern scaffolding strategies based on naturally pre-fabricated 3D biomaterials of poriferan origin. Appl. Phys. A. 2020;126 382-340. [Google Scholar]
- 113.Morais Z., Raposo A., Gonçalves A. CEFood 2016-8th Central European Congress on Food. 2016. Healthy food innovation using natural resources, catostylus tagi jellyfish: sensory evaluation. [Google Scholar]
- 114.Felician F.F., Yu R.-H., Li M.-Z., Li C.-J., Chen H.-Q., Jiang Y., Tang T., Qi W.-Y., Xu H.-M. The wound healing potential of collagen peptides derived from the jellyfish rhopilema esculentum. Chin. J. Traumatol. 2019;22:12–20. doi: 10.1016/j.cjtee.2018.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Cheng X., Shao Z., Li C., Yu L., Raja M.A., Liu C. Isolation, characterization and evaluation of collagen from jellyfish rhopilema esculentum kishinouye for use in hemostatic applications. PLoS One. 2017;12 doi: 10.1371/journal.pone.0169731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Pustlauk W., Paul B., Brueggemeier S., Gelinsky M., Bernhardt A. Modulation of chondrogenic differentiation of human mesenchymal stem cells in jellyfish collagen scaffolds by cell density and culture medium. J. Tissue Eng. Regen. Med. 2017;11:1710–1722. doi: 10.1002/term.2065. [DOI] [PubMed] [Google Scholar]
- 117.Paradiso F., Fitzgerald J., Yao S., Barry F., Taraballi F., Gonzalez D., Conlan R.S., Francis L. Marine collagen substrates for 2D and 3D ovarian cancer cell systems. Front. Bioeng. Biotechnol. 2019;7:343–372. doi: 10.3389/fbioe.2019.00343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Addad S., Exposito J.-Y., Faye C., Ricard-Blum S., Lethias C. Isolation, characterization and biological evaluation of jellyfish collagen for use in biomedical applications. Mar. Drugs. 2011;9:967–983. doi: 10.3390/md9060967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Rastian Z., Pütz S., Wang Y., Kumar S., Fleissner F., Weidner T., Parekh S.H. Type I collagen from jellyfish catostylus mosaicus for biomaterial applications. ACS Biomater. Eng. 2018;4:2115–2125. doi: 10.1021/acsbiomaterials.7b00979. [DOI] [PubMed] [Google Scholar]
- 120.Ferrario C., Rusconi F., Pulaj A., Macchi R., Landini P., Paroni M., Colombo G., Martinello T., Melotti L., Gomiero C. From food waste to innovative biomaterial: sea Urchin-derived collagen for applications in skin regenerative medicine. Mar. Drugs. 2020;18:414–431. doi: 10.3390/md18080414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Goh K.L., Holmes D.F. Collagenous extracellular matrix biomaterials for tissue engineering: lessons from the common sea urchin tissue. Int. J. Mol. Sci. 2017;18:901–948. doi: 10.3390/ijms18050901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Dai M., Liu X., Wang N., Sun J. Squid type II collagen as a novel biomaterial: isolation, characterization, immunogenicity and relieving effect on degenerative osteoarthritis via inhibiting STAT1 signaling in pro-inflammatory macrophages. Mater. Sci. Eng. C. Mater. Biol. Appl. 2018;89:283–294. doi: 10.1016/j.msec.2018.04.021. [DOI] [PubMed] [Google Scholar]
- 123.Delphi L., Sepehri H., Motevaseli E., Khorramizadeh M.R. Collagen extracted from Persian gulf squid exhibits anti-cytotoxic properties on apple pectic treated cells: assessment in an in vitro bioassay model, Iran. J. Public Health. 2016;45:1054–1063. [PMC free article] [PubMed] [Google Scholar]
- 124.Blowes L.M., Egertová M., Liu Y., Davis G.R., Terrill N.J., Gupta H.S., Elphick M.R. Body wall structure in the starfish asterias rubens. J. Anat. 2017;231:325–341. doi: 10.1111/joa.12646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Tan Y., Chang S.K.C. Isolation and characterization of collagen extracted from channel catfish (Ictalurus punctatus) skin. Food Chem. 2018;242:147–155. doi: 10.1016/j.foodchem.2017.09.013. [DOI] [PubMed] [Google Scholar]
- 126.Zhang Q., Wang Q., Lv S., Lu J., Jiang S., Regenstein J.M., Lin L. Comparison of collagen and gelatin extracted from the skins of nile Tilapia (Oreochromis niloticus) and channel catfish (Ictalurus punctatus) Food Bioence. 2016;13:41–48. [Google Scholar]
- 127.Hukmi N.M.M., Sarbon N.M. Isolation and characterization of acid soluble collagen (ASC) and pepsin soluble collagen (PSC) extracted from silver catfish (pangasius sp.) skin. Int. Food Res. J. 2018;25:2601–2607. [Google Scholar]
- 128.Pozzolini M., Millo E., Oliveri C., Mirata S., Salis A., Damonte G., Arkel M., Scarfì S. Elicited ROS scavenging activity, photoprotective, and wound-healing properties of collagen-derived peptides from the marine sponge chondrosia reniformis. Mar. Drugs. 2018;16:465–490. doi: 10.3390/md16120465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Elango J., Lee J.W., Wang S., Henrotin Y., de Val J.E.M.S., Regenstein J.M., Lim S.Y., Bao B., Wu W. Evaluation of differentiated bone cells proliferation by blue shark skin collagen via biochemical for bone tissue engineering. Mar. Drugs. 2018;16:350–364. doi: 10.3390/md16100350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Bu Y., Elango J., Zhang J., Bao B., Guo R., Palaniyandi K., Robinson J.S., Geevaretnam J., Regenstein J.M., Wu W. Immunological effects of collagen and collagen peptide from blue shark cartilage on 6T-CEM cells. Process Biochem. 2017;57:219–227. [Google Scholar]
- 131.Mei F., Liu J., Wu J., Duan Z., Chen M., Meng K., Chen S., Shen X., Xia G., Zhao M. Collagen peptides isolated from Salmo salar and Tilapia nilotica skin accelerate wound healing by altering cutaneous microbiome colonization via upregulated NOD2 and BD14. J. Agric. Food Chem. 2020;68:1621–1633. doi: 10.1021/acs.jafc.9b08002. [DOI] [PubMed] [Google Scholar]
- 132.Nurilmala M., Hizbullah H.H., Karnia E., Kusumaningtyas E., Ochiai Y. Characterization and antioxidant activity of collagen, gelatin, and the derived peptides from yellowfin tuna (Thunnus albacares) skin. Mar. Drugs. 2020;18:98–109. doi: 10.3390/md18020098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Muthumari K., Anand M., Maruthupandy M. Collagen extract from marine finfish scales as a potential mosquito larvicide. Protein J. 2016;35:391–400. doi: 10.1007/s10930-016-9685-7. [DOI] [PubMed] [Google Scholar]
- 134.Wu R., Wu C., Liu D., Yang X., Huang J., Zhang J., Liao B., He H. Antioxidant and anti-freezing peptides from salmon collagen hydrolysate prepared by bacterial extracellular protease. Food Chem. 2018;248:346–352. doi: 10.1016/j.foodchem.2017.12.035. [DOI] [PubMed] [Google Scholar]
- 135.Bhuimbar M.V., Bhagwat P.K., Dandge P.B. Extraction and characterization of acid soluble collagen from fish waste: development of collagen-chitosan blend as food packaging film. J. Environ. Chem. Eng. 2019;7:102983–102989. [Google Scholar]
- 136.Kobayashi Y., Akiyama H., Huge J., Kubota H., Chikazawa S., Satoh T., Miyake T., Uhara H., Okuyama R., Nakagawara R. Fish collagen is an important panallergen in the Japanese population. Allergy. 2016;71:720–723. doi: 10.1111/all.12836. [DOI] [PubMed] [Google Scholar]
- 137.Barros A.A., Aroso I.M., Silva T.H., Mano J.F., Duarte A.R.C., Reis R.L. Water and carbon dioxide: green solvents for the extraction of collagen/gelatin from marine sponges. ACS Sustain. Chem. Eng. 2014;3:254–260. [Google Scholar]
- 138.Tziveleka L.-A., Ioannou E., Tsiourvas D., Berillis P., Foufa E., Roussis V. Collagen from the marine sponges axinella cannabina and Suberites carnosus: isolation and morphological, biochemical, and biophysical characterization. Mar. Drugs. 2017;15:152–168. doi: 10.3390/md15060152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chancharern P., Laohakunjit N., Kerdchoechuen O., Thumthanaruk B. Extraction of type A and type B gelatin from jellyfish (lobonema smithii) Food Res. Int. 2016;23:419–424. [Google Scholar]
- 140.Abdelmalek B.E., Gómez-estaca J., Sila A., Martinez O., Gómez-guillén M.C., Chaabouni-ellouz S., Ayadi M.A. Characteristics and functional properties of gelatin extracted from squid (Loligo vulgaris) skin. LWT - Food Sci. Technol. 2015;65:924–931. [Google Scholar]
- 141.Enrique D., Angulo L., José P. International journal of biological macromolecules characterization of gelatin/chitosan scaffold blended with aloe vera and snail mucus for biomedical purpose. Int. J. Biol. Macromol. 2016;92:645–653. doi: 10.1016/j.ijbiomac.2016.07.029. [DOI] [PubMed] [Google Scholar]
- 142.Mureithi A.W., Onyari J.M., Chisutia Wanyonyi W., Mulaa F.J. Amino acid composition of gelatin extracted from the scales of different marine fish species in Kenya. Int. J. Sci. Res. Sci. Eng. Technol. 2017;3:558–563. [Google Scholar]
- 143.Liu D., Nikoo M., Boran G., Zhou P., Regenstein J.M. Collagen and gelatin. Annu. Rev. Food. Sci. Technol. 2015;6:527–557. doi: 10.1146/annurev-food-031414-111800. [DOI] [PubMed] [Google Scholar]
- 144.Gaspar-pintiliescu A., Stefan L.M., Anton E.D., Berger D., Matei C., Negreanu-pirjol T., Moldovan L. Physicochemical and biological properties of gelatin extracted from marine snail rapana venosa. Mar. Drugs. 2019;17:589–602. doi: 10.3390/md17100589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Casanova F., Mohammadifar M.A., Jahromi M., Petersen H.O., Sloth J.J., Eybye K.L., Kobbelgaard S., Jakobsen G., Jessen F. Physico-chemical, structural and techno-functional properties of gelatin from saithe (pollachius virens) skin. Int. J. Biol. Macromol. 2020;156:918–927. doi: 10.1016/j.ijbiomac.2020.04.047. [DOI] [PubMed] [Google Scholar]
- 146.Lopetuso L., Graziani C., Guarino A., Lamborghini A., Masi S., Stanghellini V. Gelatin tannate and tyndallized probiotics: a novel approach for treatment of diarrhea. Eur. Rev. Med. Pharmacol. Sci. 2017;21:873–883. [PubMed] [Google Scholar]
- 147.Madkhali O., Mekhail G., Wettig S.D. Modified gelatin nanoparticles for gene delivery. Int. J. Pharm. 2019;554:224–234. doi: 10.1016/j.ijpharm.2018.11.001. [DOI] [PubMed] [Google Scholar]
- 148.Moeller C., Fleischmann C., Thomas-Rueddel D., Vlasakov V., Rochwerg B., Theurer P., Gattinoni L., Reinhart K., Hartog C.S. How safe is gelatin? A systematic review and meta-analysis of gelatin-containing plasma expanders vs crystalloids and albumin. J. Crit. Care. 2016;35:75–83. doi: 10.1016/j.jcrc.2016.04.011. [DOI] [PubMed] [Google Scholar]
- 149.Kang H.K., Lee H.H., Seo C.H., Park Y. Antimicrobial and immunomodulatory properties and applications of marine-derived proteins and peptides. Mar. Drugs. 2019;17:350–374. doi: 10.3390/md17060350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Prihanto A.A., Wakayama M. Marine microorganism: an underexplored source of l-asparaginase. Adv. Food Nutr. Res. 2016;79:1–25. doi: 10.1016/bs.afnr.2016.07.005. [DOI] [PubMed] [Google Scholar]
- 151.Long S., Zhang X., Rao Z., Chen K., Xu M., Yang T., Yang S. Amino acid residues adjacent to the catalytic cavity of tetramer L-asparaginase II contribute significantly to its catalytic efficiency and thermostability. Enzym. Microb. Technol. 2016;82:15–22. doi: 10.1016/j.enzmictec.2015.08.009. [DOI] [PubMed] [Google Scholar]
- 152.Qeshmi F.I., Homaei A., Fernandes P., Javadpour S. Marine microbial L-asparaginase: biochemistry, molecular approaches and applications in tumor therapy and in food industry. Microbiol. Res. 2018;208:99–112. doi: 10.1016/j.micres.2018.01.011. [DOI] [PubMed] [Google Scholar]
- 153.Batool T., Makky E.A., Jalal M., Yusoff M.M. A comprehensive review on L-asparaginase and its applications. Appl. Biochem. Biotechnol. 2016;178:900–923. doi: 10.1007/s12010-015-1917-3. [DOI] [PubMed] [Google Scholar]
- 154.Solano F. Melanin and melanin-related polymers as materials with biomedical and biotechnological applications-cuttlefish ink and mussel foot proteins as inspired biomolecules. Int. J. Mol. Sci. 2017;18:1561–1578. doi: 10.3390/ijms18071561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kim H.J., Yang B., Park T.Y., Lim S., Cha H.J. Complex coacervates based on recombinant mussel adhesive proteins: their characterization and applications. Soft Matter. 2017;13:7704–7716. doi: 10.1039/c7sm01735a. [DOI] [PubMed] [Google Scholar]
- 156.Park K.H., Seong K.Y., Yang S.Y., Seo S. Advances in medical adhesives inspired by aquatic organisms' adhesion. Biomater. Res. 2017;21:16–25. doi: 10.1186/s40824-017-0101-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Muneer F., Siddique M.H., Azeem F., Rasul I., Muzammil S., Zubair M., Afzal M., Nadeem H. Microbial L-asparaginase: purification, characterization and applications. Arch. Microbiol. 2020;202:967–981. doi: 10.1007/s00203-020-01814-1. [DOI] [PubMed] [Google Scholar]
- 158.Lane T.W., Saito M.A., George G.N., Pickering I.J., Prince R.C., Morel F.M. Biochemistry: a cadmium enzyme from a marine diatom. Nature. 2005;435 doi: 10.1038/435042a. 42-42. [DOI] [PubMed] [Google Scholar]
- 159.Alterio V., Langella E., De Simone G., Monti S.M. Cadmium-containing carbonic anhydrase CDCA1 in marine diatom Thalassiosira weissflogii. Mar. Drugs. 2015;13:1688–1697. doi: 10.3390/md13041688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Barzkar N. Marine microbial alkaline protease: an efficient and essential tool for various industrial applications. Int. J. Biol. Macromol. 2020;161:1216–1229. doi: 10.1016/j.ijbiomac.2020.06.072. [DOI] [PubMed] [Google Scholar]
- 161.Wu S., Liu G., Zhang D., Li C., Sun C. Purification and biochemical characterization of an alkaline protease from marine bacteria pseudoalteromonas sp. 129-1. J. Basic Microbiol. 2015;55:1427–1434. doi: 10.1002/jobm.201500327. [DOI] [PubMed] [Google Scholar]
- 162.Mo S., Kim J.H., Cho K.W. A novel extracellular phospholipase C purified from a marine bacterium, pseudoalteromonas sp. J937. Biotechnol. Lett. 2009;31:89–94. doi: 10.1007/s10529-008-9833-z. [DOI] [PubMed] [Google Scholar]
- 163.Mo S., Kim J.H., Cho K.W. Enzymatic properties of an extracellular phospholipase C purified from a marine streptomycete. Biosci. Biotechnol. Biochem. 2009;73:2136–2137. doi: 10.1271/bbb.90323. [DOI] [PubMed] [Google Scholar]
- 164.Shehata A.N., Abd El Aty A.A., Darwish D.A., Abdel Wahab W.A., Mostafa F.A. Purification, physicochemical and thermodynamic studies of antifungal chitinase with production of bioactive chitosan-oligosaccharide from newly isolated Aspergillus griseoaurantiacus KX010988. Int. J. Biol. Macromol. 2018;107:990–999. doi: 10.1016/j.ijbiomac.2017.09.071. [DOI] [PubMed] [Google Scholar]
- 165.Itoh T., Kimoto H. Bacterial chitinase system as a model of chitin biodegradation. Adv. Exp. Med. Biol. 2019;1142:131–151. doi: 10.1007/978-981-13-7318-3_7. [DOI] [PubMed] [Google Scholar]
- 166.Li Y., Lei X., Zhu H., Zhang H., Guan C., Chen Z., Zheng W., Fu L., Zheng T. Chitinase producing bacteria with direct algicidal activity on marine diatoms. Sci. Rep. 2016;6:21984–21996. doi: 10.1038/srep21984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Xu F., Wang P., Zhang Y.Z., Chen X.L. Diversity of three-dimensional structures and catalytic mechanisms of alginate lyases. Appl. Environ. Microbiol. 2018;84 doi: 10.1128/AEM.02040-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Inoue A. Characterization of PL-7 family alginate lyases from marine organisms and their applications. Methods Enzymol. 2018;605:499–524. doi: 10.1016/bs.mie.2018.01.030. [DOI] [PubMed] [Google Scholar]
- 169.Badur A.H., Jagtap S.S., Yalamanchili G., Lee J.K., Zhao H., Rao C.V. Alginate lyases from alginate-degrading Vibrio splendidus 12B01 are endolytic. Appl. Environ. Microbiol. 2015;81:1865–1873. doi: 10.1128/AEM.03460-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Jahromi S.T., Barzkar N. Future direction in marine bacterial agarases for industrial applications. Appl. Microbiol. Biotechnol. 2018;102:6847–6863. doi: 10.1007/s00253-018-9156-5. [DOI] [PubMed] [Google Scholar]
- 171.Fu W., Han B., Duan D., Liu W., Wang C. Purification and characterization of agarases from a marine bacterium Vibrio sp. F-6. J. Ind. Microbiol. Biotechnol. 2008;35:915–922. doi: 10.1007/s10295-008-0365-2. [DOI] [PubMed] [Google Scholar]
- 172.Matard-Mann M., Bernard T., Leroux C., Barbeyron T., Larocque R., Préchoux A., Jeudy A., Jam M., Nyvall Collén P., Michel G. Structural insights into marine carbohydrate degradation by family GH16 κ-carrageenases. J. Biol. Chem. 2017;922:19919–19934. doi: 10.1074/jbc.M117.808279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Chauhan P.S., Saxena A. Bacterial carrageenases: an overview of production and biotechnological applications. 3 Biotech. 2016;6:146–163. doi: 10.1007/s13205-016-0461-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Shen J., Chang Y., Chen F., Dong S. Expression and characterization of a κ-carrageenase from marine bacterium wenyingzhuangia aestuarii OF219: a biotechnological tool for the depolymerization of κ-carrageenan. Int. J. Biol. Macromol. 2018;112:93–100. doi: 10.1016/j.ijbiomac.2018.01.075. [DOI] [PubMed] [Google Scholar]
- 175.Li S., Hao J., Sun M. Cloning and characterization of a new cold-adapted and thermo-tolerant ι-carrageenase from marine bacterium flavobacterium sp YS-80-122. Int. J. Biol. Macromol. 2017;102:1059–1065. doi: 10.1016/j.ijbiomac.2017.04.070. [DOI] [PubMed] [Google Scholar]
- 176.Trivedi N., Reddy C.R.K., Lali A.M. Chapter two - marine microbes as a potential source of cellulolytic enzymes. In: Kim S.-K., Toldrá F., editors. vol. 79. Academic Press.; Amsterdam: 2016. pp. 27–41. (Marine Enzymes Biotechnology: Production and Industrial Applications, Part II - Marine Organisms Producing Enzymes). [Google Scholar]
- 177.Klippel B., Lochner A., Bruce D.C., Davenport K.W., Detter C., Goodwin L.A., Han J., Han S., Land M.L., Mikhailova N. Complete genome sequence of the marine cellulose- and xylan-degrading bacterium glaciecolasp. strain 4H-3-7+YE-5. J. Bacteriol. 2011;193:4547–4548. doi: 10.1128/JB.05468-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Qeshmi F.I., Homaei A., Fernandes P., Hemmati R., Dijkstra B.W., Khajeh K. Xylanases from marine microorganisms: a brief overview on scope, sources, features and potential applications. Biochim. Biophys. Acta Protein Proteonomics. 2020;1868:140312–140325. doi: 10.1016/j.bbapap.2019.140312. [DOI] [PubMed] [Google Scholar]
- 179.Li W., Sunda W.G., Lin W., Hong H., Shi D. The effect of cell size on cellular Zn and Cd and Zn-Cd-CO2 colimitation of growth rate in marine diatoms. Limnol. Oceanogr. 2020;65:2896–2911. [Google Scholar]
- 180.Lane T.W., Saito M.A., George G.N., Pickering I.J., Prince R.C., Morel F.M. A cadmium enzyme from a marine diatom. Nature. 2005;435 doi: 10.1038/435042a. 42-42. [DOI] [PubMed] [Google Scholar]
- 181.Park S.Y., Kim Y.S., Ahn C.B., Je J.Y. Partial purification and identification of three antioxidant peptides with hepatoprotective effects from blue mussel ( Mytilus edulis ) hydrolysate by peptic hydrolysis. J. Funct. Foods. 2016;20:88–95. [Google Scholar]
- 182.Chi C.F., Hu F.Y., Wang B., Li T., Ding G.F. Antioxidant and anticancer peptides from the protein hydrolysate of blood clam (tegillarca granosa) muscle. J. Funct. Foods. 2015;15:301–313. [Google Scholar]
- 183.Zhang Z., Zhou F., Liu X., Zhao M. Particulate nanocomposite from oyster (Crassostrea Rivularis) hydrolysates via zinc chelation improves zinc solubility and peptide activity. Food Chem. 2018;258:269–277. doi: 10.1016/j.foodchem.2018.03.030. [DOI] [PubMed] [Google Scholar]
- 184.Vijaykrishnaraj M., Prabhasankar P. Marine protein hydrolysates: their present and future perspectives in food chemistry – a review. RSC Adv. 2015;5:34864–34877. [Google Scholar]
- 185.Miao J., Liao W., Kang M., Jia Y., Wang Q. Anti-fatigue and anti-oxidant activities of oyster (Ostrea rivularis) hydrolysate prepared by compound protease. Food Funct. 2018;9:6577–6585. doi: 10.1039/c8fo01879k. [DOI] [PubMed] [Google Scholar]
- 186.Yuk H., Varela C.E., Nabzdyk C.S., Mao X., Padera R.F. Dry double-sided tape for adhesion of wet tissues and devices. Nature. 2019;575:169–174. doi: 10.1038/s41586-019-1710-5. [DOI] [PubMed] [Google Scholar]
- 187.Yang B., Jin S., Park Y., Jung Y.M., Cha H.J. Coacervation of interfacial adhesive proteins for initial mussel adhesion to a wet surface. Small. 2018;14 doi: 10.1002/smll.201803377. [DOI] [PubMed] [Google Scholar]
- 188.Kaushik N.K., Kaushik N., Pardeshi S., Sharma J.G., Lee S.H., Choi E.H. Biomedical and clinical importance of mussel-inspired polymers and materials. Mar. Drugs. 2015;13:6792–6817. doi: 10.3390/md13116792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Buffet J.P., Corre E., Duvernois-Berthet E., Fournier J., Lopez P.J. Adhesive gland transcriptomics uncovers a diversity of genes involved in glue formation in marine tube-building polychaetes. Acta Biomater. 2018;72:316–328. doi: 10.1016/j.actbio.2018.03.037. [DOI] [PubMed] [Google Scholar]
- 190.Almeida M., Reis R.L., Silva T.H. Marine invertebrates are a source of bioadhesives with biomimetic interest. Mater. Sci. Eng. C. 2020;108:110467–110476. doi: 10.1016/j.msec.2019.110467. [DOI] [PubMed] [Google Scholar]
- 191.Jones J.P., Sima M., O'Hara R.G., Stewart R.J. Water-borne endovascular embolics inspired by the undersea adhesive of marine sandcastle worms. Adv. Healthc. Mater. 2016;5:795–801. doi: 10.1002/adhm.201500825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Mhlongo J.T., Brasil E., de la Torre B.G., Albericio F. Naturally occurring oxazole-containing peptides. Mar. Drugs. 2020;18:203–227. doi: 10.3390/md18040203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Manfrin A., Borduas-Dedekind N., Lau K., McNeill K. Singlet oxygen photooxidation of peptidic oxazoles and thiazoles. J. Org. Chem. 2019;84:2439–2447. doi: 10.1021/acs.joc.8b02684. [DOI] [PubMed] [Google Scholar]
- 194.Chen S., Ji X., Gao M., Dedkova L.M., Hecht S.M. In cellulo synthesis of proteins containing a fluorescent oxazole amino acid. J. Am. Chem. Soc. 2019;141:5597–5601. doi: 10.1021/jacs.8b12767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Joshi S., Bisht A.S., Juyal D. Systematic scientific study of 1 , 3-oxazole derivatives as a useful lead for Pharmaceuticals : a review. Pharma Innov. 2017;6:109–117. [Google Scholar]
- 196.Chiacchio M.A., Lanza G., Chiacchio U., Giofrè S.V., Romeo R., Iannazzo D., Legnani L. Oxazole-based compounds as anticancer agents. Curr. Med. Chem. 2019;26:7337–7371. doi: 10.2174/0929867326666181203130402. [DOI] [PubMed] [Google Scholar]
- 197.Vishvakarma V.K., Shukla N., Reetu, Kumari K., Patel R., Singh P. A model to study the inhibition of NsP2B-NsP3 protease of dengue virus with imidazole, oxazole, triazole thiadiazole, and thiazolidine based scaffolds. Heliyon. 2019;5 doi: 10.1016/j.heliyon.2019.e02124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Yamamuro D., Uchida R., Ohtawa M., Arima S., Futamura Y., Katane M., Homma H., Nagamitsu T., Osada H., Tomoda H. Synthesis and biological activity of 5-(4-methoxyphenyl)-oxazole derivatives. Bioorg. Med. Chem. Lett. 2015;25:313–316. doi: 10.1016/j.bmcl.2014.11.042. [DOI] [PubMed] [Google Scholar]
- 199.Apostol T.V., Barbuceanu S.F., Socea L.I., Draghici C., Saramet G., Iscrulescu L., Olaru O.T. Synthesis, characterization and cytotoxicity evaluation of new heterocyclic compounds with oxazole ring containing 4- (phenylsulfonyl) phenyl moiety. Rev. Chim. 2019;70:3793–3801. [Google Scholar]
- 200.Imhoff J.F. Natural products from marine fungi — still an underrepresented resource. Mar. Drugs. 2016;14:19–37. doi: 10.3390/md14010019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Khalifa S.A.M., Elias N., Farag M.A., Chen L., Saeed A., Hegazy M.F., Moustafa M.S., Abd El-Wahed A., Al-Mousawi S.M., Musharraf S.G. Marine natural products: a source of novel anticancer drugs. Mar. Drugs. 2019;17:491–521. doi: 10.3390/md17090491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.Naoki W., Sakamoto T., Matsugo S. Mycosporine-like amino acids and their derivatives as natural antioxidants. Antioxidants (Basel) 2015;4:603–646. doi: 10.3390/antiox4030603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Righi V., Parenti F., Schenetti L., Mucci A. Mycosporine-like amino acids and other phytochemicals directly detected by high-resolution NMR on Klamath (Aphanizomenon flos-aquae) blue-green algae. J. Agric. Food Chem. 2016;64:6708–6715. doi: 10.1021/acs.jafc.6b02615. [DOI] [PubMed] [Google Scholar]
- 204.Gao X. Scytonemin plays a potential role in stabilizing the exopolysaccharidic matrix in terrestrial cyanobacteria. Microb. Ecol. 2017;73:255–258. doi: 10.1007/s00248-016-0851-4. [DOI] [PubMed] [Google Scholar]
- 205.Colabella F., Moline M., Libkind D. UV sunscreens of microbial origin: mycosporines and mycosporine- like aminoacids. Recent Pat. Biotechnol. 2014;8:179–193. doi: 10.2174/1872208309666150102104520. [DOI] [PubMed] [Google Scholar]
- 206.Chrapusta E., Kaminski A., Duchnik K., Bober B., Adamski M., Bialczyk J. Mycosporine-like amino acids: potential health and beauty ingredients. Mar. Drugs. 2017;15:326–354. doi: 10.3390/md15100326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Öztaskın N., Taslimi P., Maraş A., Gülcin İ., Göksu S. Novel antioxidant bromophenols with acetylcholinesterase, butyrylcholinesterase and carbonic anhydrase inhibitory actions. Bioorg. Chem. 2017;74:104–114. doi: 10.1016/j.bioorg.2017.07.010. [DOI] [PubMed] [Google Scholar]
- 208.Choi Y.K., Ye B.R., Kim E.A., Kim J., Kim M.S., Lee W.W., Ahn G.N., Kang N., Jung W.K., Heo S.J. Bis (3-bromo-4,5-dihydroxybenzyl) ether. A novel bromophenol from the marine red alga polysiphonia morrowii that suppresses LPS-induced inflammatory response by inhibiting ROS-mediated ERK signaling pathway in RAW 264.7 macrophages. Biomed. Pharmacother. 2018;103:1170–1177. doi: 10.1016/j.biopha.2018.04.121. [DOI] [PubMed] [Google Scholar]
- 209.Amr A.E.E., El-Naggar M., Al-Omar M.A., Elsayed E.A., Abdalla M.M. In vitro and in vivo anti-breast cancer activities of some synthesized pyrazolinyl-estran-17-one candidates. Molecules. 2018;23:1572–1582. doi: 10.3390/molecules23071572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Wang L.J., Guo C.L., Li X.Q., Wang S.Y., Jiang B., Zhao Y., Luo J., Xu K., Liu H., Guo S.J. Discovery of novel bromophenol hybrids as potential anticancer agents through the ros-mediated apoptotic pathway: design, synthesis and biological evaluation. Mar. Drugs. 2017;15:343–360. doi: 10.3390/md15110343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Li X., Xu Q., Li C., Luo J., Li X., Wang L., Jiang B., Shi D. Toward a treatment of diabesity: in vitro and in vivo evaluation of uncharged bromophenol derivatives as a new series of PTP1B inhibitors. Eur. J. Med. Chem. 2019;166:178–185. doi: 10.1016/j.ejmech.2019.01.057. [DOI] [PubMed] [Google Scholar]
- 212.Taslimi P., Aslan H.E., Demir Y., Oztaskin N., Maraş A., Gulçin İ., Beydemir S., Goksu S. Diarylmethanon, bromophenol and diarylmethane compounds: discovery of potent aldose reductase, α-amylase and α-glycosidase inhibitors as new therapeutic approach in diabetes and functional hyperglycemia. Int. J. Biol. Macromol. 2018;119:857–863. doi: 10.1016/j.ijbiomac.2018.08.004. [DOI] [PubMed] [Google Scholar]
- 213.Demir Y., Taslimi P., Ozaslan M.S., Oztaskin N., Çetinkaya Y., Gulçin İ., Beydemir Ş., Goksu S. Antidiabetic potential: in vitro inhibition effects of bromophenol and diarylmethanones derivatives on metabolic enzymes. Arch. Pharm. (Weinheim) 2018;351 doi: 10.1002/ardp.201800263. [DOI] [PubMed] [Google Scholar]
- 214.Xu X., Yin L., Gao J., Gao L., Song F. Antifungal bromophenols from marine red alga Symphyocladia latiuscula. Chem. Biodivers. 2014;11:807–811. doi: 10.1002/cbdv.201300239. [DOI] [PubMed] [Google Scholar]
- 215.Klinger C., ółtowska-Aksamitowska S., Wysokowski M., Tsurkan M.V., Galli R., Petrenko I., Machałowski T., Ereskovsky A., Martinović R., Muzychka L. Express method for isolation of ready-to-use 3D chitin scaffolds from aplysina archeri (aplysineidae: verongiida) demosponge. Mar. Drugs. 2019;17:131–155. doi: 10.3390/md17020131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Muzychka L., Voronkina A., Kovalchuk V., Smolii O.B., Wysokowski M., Petrenko I., Youssef D.T.A., Ehrlich I., Ehrlich H. Marine biomimetics: bromotyrosines loaded chitinous skeleton as source of antibacterial agents. Appl. Phys. Mater. Sci. Process. 2021;127:15–25. doi: 10.1007/s00339-020-04167-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Niemann H., Marmann A., Lin W., Proksch P. Sponge derived bromotyrosines: structural diversity through natural combinatorial chemistry. Nat. Prod. Commun. 2015;10:219–231. [PubMed] [Google Scholar]
- 218.Wang Q., Tang X.L., Luo X.C., de Voog N.J., Li P.L., Li G.Q. Aplysinopsin-type and bromotyrosine-derived alkaloids from the south China sea sponge fascaplysinopsis reticulata. Sci. Rep. 2019;9:2248–2257. doi: 10.1038/s41598-019-38696-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Binnewerg B., Schubert M., Voronkina A., Muzychka L., Wysokowski M., Petrenko I., Djurović M., Kovalchuk V., Tsurkan M., Martinovic R., Bechmann N., Fursov A., Ivanenko V.N., Tabachnick K.R., Smolii O.B., Joseph Y., Giovine M., Bornstein S.R., Stelling A.L., Tunger A., Schmitz M., Taniya O.S., Kovalev I.S., Zyryanov G.V., Guan K., Ehrlich H. Marine biomaterials: biomimetic and pharmacological potential of cultivated Aplysina aerophoba marine demosponge. Mater. Sci. Eng. C. Mater. Biol. Appl. 2020;109:110566–110577. doi: 10.1016/j.msec.2019.110566. [DOI] [PubMed] [Google Scholar]
- 220.Moreels L., Bhat C., Voráčová M., Peigneur S., Goovaerts H., Mäki-Lohiluoma E., Zahed F., Pardo L.A., Yli-Kauhaluoma J., Kiuru P. Synthesis of novel purpurealidin analogs and evaluation of their effect on the cancer-relevant potassium channel KV10.1. PLoS One. 2017;12 doi: 10.1371/journal.pone.0188811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Bhat C., Ilina P., Tilli I., Voráčová M., Bruun T., Barba V., Hribernik N., Lillsunde K.E., Mäki-Lohiluoma E., Rüffer T. Synthesis and antiproliferative activity of marine bromotyrosine purpurealidin I and its derivatives. Mar. Drugs. 2018;16:481–506. doi: 10.3390/md16120481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Rosic N.N. Mycosporine-like amino acids: making the foundation for organic personalised sunscreens. Mar. Drugs. 2019;17:638–654. doi: 10.3390/md17110638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Macha I.J., Ben-Nissan B. Marine skeletons: towards hard tissue repair and regeneration. Mar. Drugs. 2018;16:225–235. doi: 10.3390/md16070225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Barros A.A., Aroso I.M., Silva T.H., Mano J.F., Duarte A.R.C., Reis R.L. Surface modification of silica-based marine sponge bioceramics induce hydroxyapatite formation. Cryst. Growth Des. 2014;14:4545–4552. [Google Scholar]
- 225.Granito R.N., Custódio M.R., Rennó A.C.M. Natural marine sponges for bone tissue engineering: the state of art and future perspectives. J. Biomed. Mater. Res. B Appl. Biomater. 2017;105:1717–1727. doi: 10.1002/jbm.b.33706. [DOI] [PubMed] [Google Scholar]
- 226.Green D.W., Ben-Nissan B., Yoon K.S., Milthorpe B., Jung H.S. Natural and synthetic coral biomineralization for human bone revitalization. Trends Biotechnol. 2017;35:43–54. doi: 10.1016/j.tibtech.2016.10.003. [DOI] [PubMed] [Google Scholar]
- 227.Falini G., Fermani S., Goffredo S. Coral biomineralization: a focus on intra-skeletal organic matrix and calcification. Semin. Cell Dev. Biol. 2015;46:17–26. doi: 10.1016/j.semcdb.2015.09.005. [DOI] [PubMed] [Google Scholar]
- 228.Cao L., Li X., Zhou X., Li Y., Vecchio K.S., Yang L., Cui W., Yang R., Zhu Y., Guo Z., Zhang X. Lightweight open-cell scaffolds from sea urchin spines with superior material properties for bone defect repair. ACS Appl. Mater. Interfaces. 2017;9:9862–9870. doi: 10.1021/acsami.7b01645. [DOI] [PubMed] [Google Scholar]
- 229.Schoeppler V., Lemanis R., Reich E., Pusztai T., Gránásy L., Zlotnikov I. Crystal growth kinetics as an architectural constraint on the evolution of molluscan shells. Proc. Natl. Acad. Sci. U. S. A. 2019;116:20388–20397. doi: 10.1073/pnas.1907229116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Ehrlich H., Martinović R., Joksimović D., Petrenko I., Schiaparelli S., Wysokowski M., Tsurkan D., Stelling A.L., Springer A., Gelinsky M., Joksimović A. Conchixes: organic scaffolds which resemble the size and shapes of mollusks shells, their isolation and potential multifunctional applications. Appl. Phys. A. 2020;126:562–574. [Google Scholar]
- 231.Schröder H.C., Wang X., Tremel W., Ushijima H., Müller W.E. Biofabrication of biosilica-glass by living organisms. Nat. Prod. Rep. 2008;25:455–474. doi: 10.1039/b612515h. [DOI] [PubMed] [Google Scholar]
- 232.Wysokowski M., Jesionowski T., Ehrlich H. Biosilica as a source for inspiration in biological materials science. Am. Mineral. 2018;103:665–691. [Google Scholar]
- 233.Sumper M., Brunner E. Learning from diatoms: nature's tools for the production of nanostructured silica. Adv. Funct. Mater. 2006;16:17–26. [Google Scholar]
- 234.Palaveniene A., Harkavenko V., Kharchenko V., Daugela P., Pranskunas M., Juodzbalys G., Babenko N., Liesiene J. Cuttlebone as a marine-derived material for preparing bone grafts. Mar. Biotechnol. (NY) 2018;20:363–374. doi: 10.1007/s10126-018-9816-6. [DOI] [PubMed] [Google Scholar]
- 235.Ehrlich H., Elkin Y.N., Artyoukov A.A., Kozlovskaya E., Stonik V.A., Safronov P.P., Kurek D.V., Bazhenov V.V., Kummer K., Vyalykh D., Molodtsov S., Sivkov V., Born R., Ruhnow M., Meissner H., Richter G. Тhe spines of sand dollar Scaphechinus mirabilis (Agassiz 1863): analytical and structural study. J. Adv. Microsc. Res. 2010;5:100–109. [Google Scholar]
- 236.Li L., Kolle S., Weaver J.C., Ortiz C., Aizenberg J., Kolle M. A highly conspicuous mineralized composite photonic architecture in the translucent shell of the blue-rayed limpet. Nat. Commun. 2015;6:6322–6332. doi: 10.1038/ncomms7322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Aguirre L.E., Ouyang L., Elfwing A., Hedblom M., Wulff A., Inganäs O. Diatom frustules protect DNA from ultraviolet light. Sci. Rep. 2018;8:5138–5143. doi: 10.1038/s41598-018-21810-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Ghoshal A., Eck E., Gordon M., Morse D.E. Wavelength-specific forward scattering of light by bragg-reflective iridocytes in giant clams. J. R. Soc. Interface. 2016;13:20160285–20160292. doi: 10.1098/rsif.2016.0285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Lopez-Garcia M., Masters N., O'Brien H.E., Lennon J., Atkinson G., Cryan M.J., Oulton R., Whitney H.M. Light-induced dynamic structural color by intracellular 3D photonic crystals in Brown algae. Sci. Adv. 2018;4 doi: 10.1126/sciadv.aan8917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Iwasaka M., Mizukawa Y., Roberts N.W. Magnetic control of the light reflection anisotropy in a biogenic guanine microcrystal platelet. Langmuir. 2016;32:180–187. doi: 10.1021/acs.langmuir.5b03522. [DOI] [PubMed] [Google Scholar]
- 241.Pereira R.B., Evdokimov N.M., Lefranc F., Valentão P., Kornienko A., Pereira D.M., Andrade P.B., Gomes N.G.M. Marine-derived anticancer agents: clinical benefits, innovative mechanisms, and new targets. Mar. Drugs. 2019;17:329–349. doi: 10.3390/md17060329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Burke P.J. Mitochondria, bioenergetics and apoptosis in cancer. Trends Canc. 2017;3:857–870. doi: 10.1016/j.trecan.2017.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Jackson N.M., Ceresa B.P. EGFR-mediated apoptosis via STAT3. Exp. Cell Res. 2017;356:93–103. doi: 10.1016/j.yexcr.2017.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Kim H.J., Maiti P., Barrientos A. Mitochondrial ribosomes in cancer. Semin. Canc. Biol. 2017;47:67–81. doi: 10.1016/j.semcancer.2017.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Zhang P., Zhang Y., Liu K., Liu B., Xu W., Gao J., Ding L., Tao L. Ivermectin induces cell cycle arrest and apoptosis of HeLa cells via mitochondrial pathway. Cell Prolif. 2019;52:e12543. doi: 10.1111/cpr.12543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Yi F., Frazzette N., Cruz A.C., Klebanoff C.A., Siegel R.M. Beyond cell death: new functions for TNF family cytokines in autoimmunity and tumor immunotherapy. Trends Mol. Med. 2018;24:642–653. doi: 10.1016/j.molmed.2018.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Gutiérrez-Rodríguez A.G., Juárez-Portilla C., Olivares-Bañuelos T., Zepeda R.C. Anticancer activity of seaweeds. Drug Discov. Today. 2018;23:434–447. doi: 10.1016/j.drudis.2017.10.019. [DOI] [PubMed] [Google Scholar]
- 248.Liu M., Liu Y., Cao M.J., Liu G.M., Chen Q., Sun L., Chen H. Antibacterial activity and mechanisms of depolymerized fucoidans isolated from Laminaria japonica. Carbohydr. Polym. 2017;172:294–305. doi: 10.1016/j.carbpol.2017.05.060. [DOI] [PubMed] [Google Scholar]
- 249.Sanjeewa K.K.A., Lee J.S., Kim W.S., Jeon Y.J. The potential of Brown-algae polysaccharides for the development of anticancer agents: an update on anticancer effects reported for fucoidan and laminaran. Carbohydr. Polym. 2017;177:451–459. doi: 10.1016/j.carbpol.2017.09.005. [DOI] [PubMed] [Google Scholar]
- 250.Choo G.S., Lee H.N., Shin S.A., Kim H.J., Jung J.Y. Anticancer effect of fucoidan on DU-145 prostate cancer cells through inhibition of PI3K/Akt and MAPK pathway expression. Mar. Drugs. 2016;14:126–137. doi: 10.3390/md14070126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Jimenez P.C., Wilke D.V., Branco P.C., Bauermeister A., Rezende-Teixeira P., Gaudêncio S.P., Costa-Lotufo L.V. Enriching cancer pharmacology with drugs of marine origin. Br. J. Pharmacol. 2020;177:3–27. doi: 10.1111/bph.14876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Murphy T., Yee K.W.L. Cytarabine and daunorubicin for the treatment of acute myeloid leukemia. Expet Opin. Pharmacother. 2017;18:1765–1780. doi: 10.1080/14656566.2017.1391216. [DOI] [PubMed] [Google Scholar]
- 253.Krauss A.C., Gao X., Li L., Manning M.L., Patel P., Fu W., Janoria K.G., Gieser G., Bateman D.A., Przepiorka D. FDA approval summary: (daunorubicin and cytarabine) liposome for injection for the treatment of adults with high-risk acute myeloid leukemia. Clin. Canc. Res. 2019;25:2685–2690. doi: 10.1158/1078-0432.CCR-18-2990. [DOI] [PubMed] [Google Scholar]
- 254.Lancet J.E., Uy G.L., Cortes J.E., Newell L.F., Lin T.L., Ritchie E.K., Stuart R.K., Strickland S.A., Hogge D., Solomon S.R. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J. Clin. Oncol. 2018;36:2684–2692. doi: 10.1200/JCO.2017.77.6112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Ratan R., Patel S.R. Trabectedin and eribulin: where do they fit in the management of soft tissue sarcoma? Curr. Treat. Options Oncol. 2017;18:34–42. doi: 10.1007/s11864-017-0477-x. [DOI] [PubMed] [Google Scholar]
- 256.De Sanctis R., Marrari A., Santoro A. Trabectedin for the treatment of soft tissue sarcomas. Expet Opin. Pharmacother. 2016;17:1569–1577. doi: 10.1080/14656566.2016.1204295. [DOI] [PubMed] [Google Scholar]
- 257.Assi T., Kattan J., El Rassy E., Honore C., Dumont S., Mir O., Le Cesne A. A comprehensive review of the current evidence for Trabectedin in advanced myxoid liposarcoma. Canc. Treat Rev. 2019;72:37–44. doi: 10.1016/j.ctrv.2018.11.003. [DOI] [PubMed] [Google Scholar]
- 258.Demetri G.D., von Mehren M., Jones R.L., Hensley M.L., Schuetze S.M., Staddon A., Milhem M., Elias A., Ganjoo K., Tawbi H. Efficacy and safety of Trabectedin or dacarbazine for metastatic liposarcoma or leiomyosarcoma after failure of conventional chemotherapy: results of a phase III randomized multicenter clinical trial. J. Clin. Oncol. 2016;34:786–793. doi: 10.1200/JCO.2015.62.4734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Cortes J., Schöffski P., Littlefield B.A. Multiple modes of action of eribulin mesylate: emerging data and clinical implications. Canc. Treat Rev. 2018;70:190–198. doi: 10.1016/j.ctrv.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 260.Perez-Garcia J.M., Cortes J. The safety of eribulin for the treatment of metastatic breast cancer. Expet Opin. Drug Saf. 2019;18:347–355. doi: 10.1080/14740338.2019.1608946. [DOI] [PubMed] [Google Scholar]
- 261.Aseyev O., Ribeiro J.M., Cardoso F. Review on the clinical use of eribulin mesylate for the treatment of breast cancer. Expet Opin. Pharmacother. 2016;17:589–600. doi: 10.1517/14656566.2016.1146683. [DOI] [PubMed] [Google Scholar]
- 262.Tawbi H.A., Burgess M., Bolejack V., Van Tine B.A., Schuetze S.M., Hu J., D'Angelo S., Attia S., Riedel R.F., Priebat D.A. Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): a multicentre, two-cohort, single-arm, open-label, phase 2 trial. Lancet Oncol. 2017;18:1493–1501. doi: 10.1016/S1470-2045(17)30624-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Swami U., Shah U., Goel S. Eribulin in non-small cell lung cancer: challenges and potential strategies. Expet Opin. Invest. Drugs. 2017;26:495–508. doi: 10.1080/13543784.2017.1292250. [DOI] [PubMed] [Google Scholar]
- 264.Shah N., Mohammad A.S., Saralkar P., Sprowls S.A., Vickers S.D., John D., Tallman R.M., Lucke-Wold B.P., Jarrell K.E., Pinti M., Nolan R.L., Lockman P.R. Investigational chemotherapy and novel pharmacokinetic mechanisms for the treatment of breast cancer brain metastases. Pharmacol. Res. 2018;132:47–68. doi: 10.1016/j.phrs.2018.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Kurata T., Fushida S., Kinoshita J., Oyama K., Yamaguchi T., Okazaki M., Miyashita T., Tajima H., Ninomiya I., Ohta T. Low-dose eribulin mesylate exerts antitumor effects in gastric cancer by inhibiting fibrosis via the suppression of epithelial-mesenchymal transition and acts synergistically with 5-fluorouracil. Canc. Manag. Res. 2018;10:2729–2742. doi: 10.2147/CMAR.S167846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Turner S.D., Inghirami G., Miranda R.N., Kadin M.E. Cell of origin and immunologic events in the pathogenesis of breast implant-associated anaplastic large-cell lymphoma. Am. J. Pathol. 2020;190:2–10. doi: 10.1016/j.ajpath.2019.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Connors J.M., Jurczak W., Straus D.J., Ansell S.M., Kim W.S., Gallamini A., Younes A., Alekseev S., Illés Á., Picardi M., Lech-Maranda E., Oki Y., Feldman T., Smolewski P., Savage K.J., Bartlett N.L., Walewski J., Chen R., Ramchandren R., Zinzani P.L., Cunningham D., Rosta A., Josephson N.C., Song E., Sachs J., Liu R., Jolin H.A., Huebner D. ECHELON-1 study group, radford J brentuximab vedotin with chemotherapy for stage III or IV Hodgkin's lymphoma. N. Engl. J. Med. 2018;378:331–344. doi: 10.1056/NEJMoa1708984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Berger G.K., McBride A., Lawson S., Royball K., Yun S., Gee K., Bin Riaz I., Saleh A.A., Puvvada S., Anwer F. Brentuximab vedotin for treatment of non-hodgkin lymphomas: a systematic review. Crit. Rev. Oncol. Hematol. 2017;109:42–50. doi: 10.1016/j.critrevonc.2016.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Moskowitz C.H., Walewski J., Nademanee A., Masszi T., Agura E., Holowiecki J., Abidi M.H., Chen A.I., Stiff P., Viviani S., Bachanova V., Sureda A., McClendon T., Lee C., Lisano J., Sweetenham J. Five-year PFS from the AETHERA trial of brentuximab vedotin for Hodgkin lymphoma at high risk of progression or relapse. Blood. 2018;132:2639–2642. doi: 10.1182/blood-2018-07-861641. [DOI] [PubMed] [Google Scholar]
- 270.Delgado-Calle J., Kurihara N., Atkinson E.G., Nelson J., Miyagawa K., Galmarini C.M., Roodman G.D., Bellido T. Aplidin (plitidepsin) is a novel anti-myeloma agent with potent anti-resorptive activity mediated by direct effects on osteoclasts. Oncotarget. 2019;10:2709–2721. doi: 10.18632/oncotarget.26831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Delgado-Calle J., Atkinson E.G., Nelson J., Kurihara N., Galmarini C., Roodman D.D., Bellido T. Aplidin (plitidepsin) regulates viability and function of myeloma cells and bone cells in combination with other anti-MM drugs. Blood. 2017;130(Supplement 1) 3065-3065. [Google Scholar]
- 272.Cimino P.J., Huang L., Du L., Wu Y., Bishop J., Dalsing-Hernandez J., Kotlarczyk K., Gonzales P., Carew J., Nawrocki S. Plinabulin, an inhibitor of tubulin polymerization, targets KRAS signaling through disruption of endosomal recycling. Biomed. Rep. 2019;10:218–224. doi: 10.3892/br.2019.1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273.Blayney D.W., Zhang Q., Feng J., Zhao Y., Bondarenko I., Vynnychenko I., Kovalenko N., Nair S., Ibrahim E., Udovista D.P. Efficacy of plinabulin vs pegfilgrastim for prevention of chemotherapy-induced neutropenia in adults with non–small cell lung cancer: a phase 2 randomized clinical trial. JAMA Oncol. 2020;6 doi: 10.1001/jamaoncol.2020.4429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274.Goss G.D., Vokes E.E., Gordon M.S., Gandhi L., Papadopoulos K.P., Rasco D.W., Fischer J.S., Chu K.L., Ames W.W., Mittapalli R.K., Lee H.J., Zeng J., Roberts-Rapp L.A., Loberg L.I., Ansell P.J., Reilly E.B., Ocampo C.J., Holen K.D., Tolcher A.W. Efficacy and safety results of depatuxizumab mafodotin (ABT-414) in patients with advanced solid tumors likely to overexpress epidermal growth factor receptor. Cancer. 2018;124:2174–2183. doi: 10.1002/cncr.31304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Lassman A.B., van den Bent M.J., Gan H.K., Reardon D.A., Kumthekar P., Butowski N., Lwin Z., Mikkelsen T., Nabors L.B., Papadopoulos K.P., Penas-Prado M., Simes J., Wheeler H., Walbert T., Scott A.M., Gomez E., Lee H.J., Roberts-Rapp L., Xiong H., Ansell P.J., Bain E., Holen K.D., Maag D., Merrell R. Safety and efficacy of depatuxizumab mafodotin + temozolomide in patients with EGFR-amplified, recurrent glioblastoma: results from an international phase I multicenter trial. Neuro Oncol. 2019;21:106–114. doi: 10.1093/neuonc/noy091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 276.Farago A.F., Drapkin B.J., Lopez-Vilarino de Ramos J.A., Galmarini C.M., Núñez R., Kahatt C., Paz-Ares L. ATLANTIS: a phase III study of lurbinectedin/doxorubicin versus topotecan or cyclophosphamide/doxorubicin/vincristine in patients with small-cell lung cancer who have failed one prior platinum-containing line. Future Oncol. 2019;15:231–239. doi: 10.2217/fon-2018-0597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Morschhauser F., Flinn I.W., Advani R., Sehn L.H., Diefenbach C., Kolibaba K., Press O.W., Salles G., Tilly H., Chen A.I., Assouline S., Cheson B.D., Dreyling M., Hagenbeek A., Zinzani P.L., Jones S., Cheng J., Lu D., Penuel E., Hirata J., Wenger M., Chu Y.W., Sharman J. Polatuzumab vedotin or pinatuzumab vedotin plus rituximab in patients with relapsed or refractory non-hodgkin lymphoma: final results from a phase 2 randomised study (ROMULUS), lancet. Haematol. 2019;6:e254–e265. doi: 10.1016/S2352-3026(19)30026-2. [DOI] [PubMed] [Google Scholar]
- 278.Kopp L.M., Malempati S., Krailo M., Gao Y., Buxton A., Weigel B.J., Hawthorne T., Crowley E., Moscow J.A., Reid J.M., Villalobos V., Randall R.L., Gorlick R., Janeway K.A. Phase II trial of the glycoprotein non-metastatic B-targeted antibody-drug conjugate, glembatumumab vedotin (CDX-011), in recurrent osteosarcoma AOST1521: a report from the children's oncology group. Eur. J. Canc. 2019;121:177–183. doi: 10.1016/j.ejca.2019.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279.Ott P.A., Pavlick A.C., Johnson D.B., Hart L.L., Infante J.R., Luke J.J., Lutzky J., Rothschild N.E., Spitler L.E., Cowey C.L., Alizadeh A.R., Salama A.K., He Y., Hawthorne T.R., Bagley R.G., Zhang J., Turner C.D., Hamid O. A phase 2 study of glembatumumab vedotin, an antibody-drug conjugate targeting glycoprotein NMB, in patients with advanced melanoma. Cancer. 2019;125:1113–1123. doi: 10.1002/cncr.31892. [DOI] [PubMed] [Google Scholar]
- 280.Hasanov M., Rioth M.J., Kendra K., Hernandez-Aya L., Joseph R.W., Williamson S., Chandra S., Shirai K., Turner C.D., Lewis K., Crowley E., Moscow J., Carter B., Patel S. A phase II study of glembatumumab vedotin for metastatic uveal melanoma. Cancers (Basel) 2020;12:2270–2281. doi: 10.3390/cancers12082270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Hong D.S., Concin N., Vergote I., de Bono J.S., Slomovitz B.M., Drew Y., Arkenau H.T., Machiels J.P., Spicer J.F., Jones R., Forster M.D., Cornez N., Gennigens C., Johnson M.L., Thistlethwaite F.C., Rangwala R.A., Ghatta S., Windfeld K., Harris J.R., Lassen U.N., Coleman R.L. Tisotumab vedotin in previously treated recurrent or metastatic cervical cancer. Clin. Canc. Res. 2020;26:1220–1228. doi: 10.1158/1078-0432.CCR-19-2962. [DOI] [PubMed] [Google Scholar]
- 282.Waqar S.N., Redman M.W., Arnold S.M., Hirsch F.R., Mack P.C., Schwartz L.H., Gandara D.R., Stinchcombe T.E., Leighl N.B., Ramalingam S.S. A phase II study of telisotuzumab vedotin in patients with c-MET-positive stage IV or recurrent squamous cell lung cancer (LUNG-MAP sub-study S1400K, NCT03574753) Clin. Lung Canc. 2020;14:S1525–S7304. doi: 10.1016/j.cllc.2020.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Trudel S., Lendvai N., Popat R., Voorhees P.M., Reeves B., Libby E.N., Richardson P.G., Anderson L.D.J., Sutherland H.J., Yong K., Hoos A., Gorczyca M.M., Lahiri S., He Z., Austin D.J., Opalinska J.B., Cohen A.D. Targeting B-cell maturation antigen with GSK2857916 antibody-drug conjugate in relapsed or refractory multiple myeloma (BMA117159): a dose escalation and expansion phase 1 trial. Lancet Oncol. 2018;19:1641–1653. doi: 10.1016/S1470-2045(18)30576-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Pereira F. Have marine natural product drug discovery efforts been productive and how can we improve their efficiency? Expet Opin. Drug Discov. 2019;14:717–722. doi: 10.1080/17460441.2019.1604675. [DOI] [PubMed] [Google Scholar]
- 285.Cortés J., Diab S., Basho R.K., Oliveira M., Pluard T., Alemany C., Brown-Glaberman U., Meisel J., Boni V., Sinha R. SGNLVA-002: single arm, open-label, phase ib/II study of ladiratuzumab vedotin (LV) in combination with pembrolizumab for first-line treatment of triple-negative breast cancer. Ann. Oncol. 2020;31 S393-S393. [Google Scholar]
- 286.Hoimes C.J., Petrylak D.P., Flaig T.W., Carret A.S., Melhem-Bertrandt A., Rosenberg J.E. EV-103 study: a phase 1b dose-escalation and dose-expansion study of enfortumab vedotin in combination with immune checkpoint inhibitor (CPI) therapy for treatment of patients with locally advanced or metastatic urothelial cancer. Eur. Urol. Suppl. 2018;2:e1151. [Google Scholar]
- 287.Balar A.V., Galsky M.D., Rosenberg J.E., Powles T., Petrylak D.P., Bellmunt J., Loriot Y., Necchi A., Hoffman-Censits J., Perez-Gracia J.L., Dawson N.A., van der Heijden M.S., Dreicer R., Srinivas S., Retz M.M., Joseph R.W., Drakaki A., Vaishampayan U.N., Sridhar S.S., Quinn D.I., Durán I., Shaffer D.R., Eigl B.J., Grivas P.D., Yu E.Y., Li S., Kadel E.E., 3rd, Boyd Z., Bourgon R., Hegde P.S., Mariathasan S., Thåström A., Abidoye O.O., Fine G.D., Bajorin D.F., IMvigor210 Study Group Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial. Lancet (London, England) 2017;389:67–76. doi: 10.1016/S0140-6736(16)32455-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Purcell J.W., Tanlimco S.G., Hickson J., Fox M., Sho M., Durkin L., Uziel T., Powers R., Foster K., McGonigal T. LRRC15 is a novel mesenchymal protein and stromal target for antibody-drug conjugates. Canc. Res. 2018;78:4059–4072. doi: 10.1158/0008-5472.CAN-18-0327. [DOI] [PubMed] [Google Scholar]
- 289.Newman D.J. The “utility ” of highly toxic marine-sourced compounds. Mar. Drugs. 2019;17:324–342. doi: 10.3390/md17060324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Damelin M., Bankovich A., Bernstein J., Lucas J., Chen L., Williams S., Park A., Aguilar J., Ernstoff E., Charati M., Dushin R., Aujay M., Lee C., Ramoth H., Milton M., Hampl J., Lazetic S., Pulito V., Rosfjord E., Sun Y., King L., Barletta F., Betts A., Guffroy M., Falahatpisheh H., O'Donnell C.J., Stull R., Pysz M., Escarpe P., Liu D., Foord O., Gerber H.P., Sapra P., Dylla S.J. A PTK7-targeted antibody-drug conjugate reduces tumor-initiating cells and induces sustained tumor regressions. Sci. Transl. Med. 2017;9:2611–2621. doi: 10.1126/scitranslmed.aag2611. [DOI] [PubMed] [Google Scholar]
- 291.Katoh M. Canonical and non-canonical WNT signaling in cancer stem cells and their niches: cellular heterogeneity, omics reprogramming, targeted therapy and tumor plasticity (review) Int. J. Oncol. 2017;51:1357–1369. doi: 10.3892/ijo.2017.4129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Aravindan S., Somasundaram D.B., Somasundaram S.T., Natarajan M., Herman T.S., Aravindan N. Targeting acquired oncogenic burden in resilient pancreatic cancer: a novel benefit from marine polyphenols. Mol. Cell. Biochem. 2019;460:175–193. doi: 10.1007/s11010-019-03579-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293.Morrison K., Challita-Eid P.M., Raitano A., An Z., Yang P., Abad J.D., Liu W., Lortie D.R., Snyder J.T., Capo L., Verlinsky A., Aviña H., Doñate F., Joseph I.B., Pereira D.S., Morrison K., Stover D.R. Development of ASG-15ME, a novel antibody-drug conjugate targeting SLITRK6, a new urothelial cancer biomarker. Mol. Canc. Therapeut. 2016;15:1301–1310. doi: 10.1158/1535-7163.MCT-15-0570. [DOI] [PubMed] [Google Scholar]
- 294.Vlachostergios P.J., Jakubowski C.D., Niaz M.J., Lee A., Thomas C., Hackett A.L., Patel P., Rashid N., Tagawa S.T. Antibody-drug conjugates in bladder cancer. Bladder Cancer. 2018;4:247–259. doi: 10.3233/BLC-180169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Newman D.J., Cragg G.M. Current status of marine-derived compounds as warheads in anti-tumor drug candidates. Mar. Drugs. 2017;15:99–117. doi: 10.3390/md15040099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Pereira D.S., Guevara C.I., Jin L., Mbong N., Verlinsky A., Hsu S.J., Aviña H., Karki S., Abad J.D., Yang P., Moon S.J., Malik F., Choi M.Y., An Z., Morrison K., Challita-Eid P.M., Doñate F., Joseph I.B., Kipps T.J., Dick J.E., Stover D.R. AGS67E, an anti-CD37 monomethyl auristatin E antibody-drug conjugate as a potential therapeutic for B/T-Cell malignancies and AML: a new role for CD37 in AML. Mol. Canc. Therapeut. 2015;14:1650–1660. doi: 10.1158/1535-7163.MCT-15-0067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Cheung R.C.F., Ng T.B., Wong J.H. Marine peptides: bioactivities and applications. Mar. Drugs. 2015;13:4006–4043. doi: 10.3390/md13074006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Falanga A., Lombardi L., Franci G., Vitiello M., Iovene M.R., Morelli G., Galdiero M., Galdiero S. Marine antimicrobial peptides: nature provides templates for the design of novel compounds against pathogenic bacteria. Int. J. Mol. Sci. 2016;17:785–802. doi: 10.3390/ijms17050785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Diogo G.S., Senra E.L., Pirraco R.P., Canadas R.F., Fernandes E.M., Serra J., Pérez-Martín R.I., Sotelo C.G., Marques A.P., González P., Moreira-Silva J., Silva T.H., Reis R.L. Marine collagen/apatite composite scaffolds envisaging hard tissue applications. Mar. Drugs. 2018;16:269–282. doi: 10.3390/md16080269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Deepthi S., Venkatesan J., Kim S.K., Bumgardner J.D., Jayakumar R. An overview of chitin or chitosan/nano ceramic composite scaffolds for bone tissue engineering. Int. J. Biol. Macromol. 2016;93:1338–1353. doi: 10.1016/j.ijbiomac.2016.03.041. [DOI] [PubMed] [Google Scholar]
- 301.Echave M.C., Sánchez P., Pedraz J.L., Orive G. Progress of gelatin-based 3D approaches for bone regeneration. J. Drug Deliv. Sci. Technol. 2017;42:63–74. [Google Scholar]
- 302.Narayana J.L., Huang H.N., Wu C.J., Chen J.Y. Efficacy of the antimicrobial peptide TP4 against Helicobacter pylori infection: In vitro membrane perturbation via micellization and in vivo suppression of host immune responses in a mouse model. Oncotarget. 2015;6:12936–12954. doi: 10.18632/oncotarget.4101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Meng D.M., Lv Y.J., Zhao J.F., Liu Q.Y., Shi L.Y., Wang J.P., Yang Y.H., Fan Z.C. Efficient production of a recombinant venerupis philippinarum defensin (VpDef) in pichia pastoris and characterization of its antibacterial activity and stability. Protein Expr. Purif. 2018;147:78–84. doi: 10.1016/j.pep.2018.03.001. [DOI] [PubMed] [Google Scholar]
- 304.Wei L., Gao J., Zhang S., Wu S., Xie Z., Ling G., Kuang Y.Q., Yang Y., Yu H., Wang Y. Identification and characterization of the first cathelicidin from sea snakes with potent antimicrobial and anti-inflammatory activity and special mechanism. J. Biol. Chem. 2015;290:16633–16652. doi: 10.1074/jbc.M115.642645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Chen C., Wang A., Zhang F., Zhang M., Yang H., Li J., Su P., Chen Y., Yu H., Wang Y. The protective effect of fish-derived cathelicidins on bacterial infections in zebrafish, Danio rerio. Fish Shellfish Immunol. 2019;92:519–527. doi: 10.1016/j.fsi.2019.06.029. [DOI] [PubMed] [Google Scholar]
- 306.Kurata A., Yamaura Y., Tanaka T., Kato C., Nakasone K., Kishimoto N. Antifungal peptidic compound from the deep-sea bacterium Aneurinibacillus sp. YR247. World J. Microbiol. Biotechnol. 2017;33:73–80. doi: 10.1007/s11274-017-2239-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Espiritu R.A., Cornelio K., Kinoshita M., Matsumori N., Murata M., Nishimura S., Kakeya H., Yoshida M., Matsunaga S. Marine sponge cyclic peptide theonellamide A disrupts lipid bilayer integrity without forming distinct membrane pores. Biochim. Biophys. Acta. 2016;1858:1373–1379. doi: 10.1016/j.bbamem.2016.03.019. [DOI] [PubMed] [Google Scholar]
- 308.Binnewerg B., Schubert M., Voronkina A., Muzychka L., Wysokowski M., Petrenko I., Djurović M., Kovalchuk V., Tsurkan M., Martinovic R., Bechmann N., Fursov A., Ivanenko V.N., Tabachnik K.R., Smolii O.B., Joseph Y., Giovine M., Bornstein S.R., Stelling A.L., Tunger A., Schmitz M., Taniya O.S., Kovalev I.S., Zyryanov G.V., Guan K., Ehrlich H. Marine biomaterials: biomimetic and pharmacological potential of cultivated Aplysina aerophoba marine demosponge. Mater. Sci. Eng. C Mater. Biol. Appl. 2020;109:110566. doi: 10.1016/j.msec.2019.110566. [DOI] [PubMed] [Google Scholar]
- 309.Machałowski T., Czajka M., Petrenko I., Meissner H., Schimpf C., Rafaja D., Ziętek J., Dzięgiel B., Adaszek Ł., Voronkina A., Kovalchuk V., Jaroszewicz J., Fursov A., Rahimi-Nasrabadi M., Stawski D., Bechmann N., Jesionowski T., Ehrlich H. Functionalization of 3D chitinous skeletal scaffolds of sponge origin using silver nanoparticles and their antibacterial properties. Mar. Drugs. 2020;18:304–319. doi: 10.3390/md18060304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Gui L., Zhang P., Zhang Q., Zhang J. Two hepcidins from spotted scat (Scatophagus argus) possess antibacterial and antiviral functions in vitro. Fish Shellfish Immunol. 2016;50:191–199. doi: 10.1016/j.fsi.2016.01.038. [DOI] [PubMed] [Google Scholar]
- 311.Wysokowski M., Machalowski T., Petrenko I., Schimpf C., Rafaja D., Galli R., Zietek J., Pantović S., Voronkina A., Kovalchuk V., Ivanenko V.N., Hoeksema B.W., Diaz C., Khrunyk Y., Stelling A.L., Giovine M., Jesionowski T., Ehrlich H. 3D chitin scaffolds of marine demosponge origin for biomimetic mollusk hemolymph-associated biomineralization ex-vivo. Mar. Drugs. 2020;18:574–590. doi: 10.3390/md18020123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Jing Q., Hu X., Ma Y., Mu J., Liu W., Xu F., Li Z., Bai J., Hua H., Li D. Marine-derived natural lead compound disulfide-linked dimer psammaplin A: biological activity and structural modification. Mar. Drugs. 2019;17:384–406. doi: 10.3390/md17070384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Kovalchuk V., Voronkina A., Binnewerg B., Schubert M., Muzychka L., Wysokowski M., Tsurkan M.V., Bechmann N., Petrenko I., Fursov A., Martinovic R., Ivanenko V.N., Fromont J., Smolii O.B., Joseph Y., Giovine M., Erpenbeck D., Gelinsky M., Springer A., Guan K., Bornstein S.R., Ehrlich H. Naturally drug-loaded chitin: isolation and applications. Mar. Drugs. 2019;17:1–17. doi: 10.3390/md17100574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 314.Tabudravu J.N., Eijsink V.G.H., Gooday G.W., Jaspars M., Komander D., Legg M., Synstad B., van Aalten D.M.F., Psammaplin A. A chitinase inhibitor isolated from the Fijian marine sponge Aplysinella rhax. Bioorg. Med. Chem. 2002;10:1123–1128. doi: 10.1016/s0968-0896(01)00372-8. [DOI] [PubMed] [Google Scholar]
- 315.Roseti L., Parisi V., Petretta M., Cavallo C., Desando G., Bartolotti I., Grigolo B. Scaffolds for bone tissue engineering: state of the art and new perspectives. Mater. Sci. Eng. C. 2017;78:1246–1262. doi: 10.1016/j.msec.2017.05.017. [DOI] [PubMed] [Google Scholar]
- 316.Neto A.S., Ferreira J.M.F. Synthetic and marine-derived porous scaffolds for bone tissue engineering. Materials (Basel) 2018;11:1702–1741. doi: 10.3390/ma11091702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Kuttappan S., Mathew D., Nair M.B. Biomimetic composite scaffolds containing bioceramics and collagen/gelatin for bone tissue engineering - a mini review. Int. J. Biol. Macromol. 2016;93:1390–1401. doi: 10.1016/j.ijbiomac.2016.06.043. [DOI] [PubMed] [Google Scholar]
- 318.Georgopoulou A., Kaliva M., Vamvakaki M., Chatzinikolaidou M. Osteogenic potential of pre-osteoblastic cells on a chitosan-graft-polycaprolactone copolymer. Materials (Basel) 2018;11:490–505. doi: 10.3390/ma11040490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Aguilar A., Zein N., Harmouch E., Hafdi B., Bornert F., Offner D., Clauss F., Fioretti F., Huck O., Benkirane-Jessel N., Hua G. Application of chitosan in bone and dental engineering. Molecules. 2019;24:3009–3025. doi: 10.3390/molecules24163009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.Mohammadi R., Amini K. Guided bone regeneration of mandibles using chitosan scaffold seeded with characterized uncultured omental adipose-derived stromal vascular fraction: an animal study. Int. J. Oral Maxillofac. Implants. 2015;30:216–222. doi: 10.11607/jomi.3542. [DOI] [PubMed] [Google Scholar]
- 321.Ehrlich H., Steck E., Ilan M., Maldonado M., Muricy G., Bavestrello G., Kljajic Z., Carballo J.L., Shiaparelli S., Ereskovsky A.V., Schupp P., Born R., Worch H., Bazhenov V.V., Kurek D., Varlamow V., Vyalikh D., Kummer K., Sivkov V.V., Molodtsov S.L., Meissner H., Richter G., Hunoldt S., Kammer M., Paasch S., Krasokhin V., Patzke G., Brunner E., Richter W. Three dimensional chitin-based scaffolds from Verongida sponges (Demospongiae: Porifera). Part II. Biomimetic potential and applications. Int. J. Biol. Macromol. 2010;47:141–145. doi: 10.1016/j.ijbiomac.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 322.Zhou D., Qi C., Chen Y.X., Zhu Y.J., Sun T.W., Chen F., Zhang C.Q. Comparative study of porous hydroxyapatite/chitosan and whitlockite/chitosan scaffolds for bone regeneration in calvarial defects. Int. J. Nanomed. 2017;12:2673–2687. doi: 10.2147/IJN.S131251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Bordini E.A.F., Cassiano F.B., Silva I.S.P., Usberti F.R., Anovazzi G., Pacheco L.E., Pansani T.N., Leite M.L., Hebling J., de Souza Costa C.A., Soares D.G. Synergistic potential of 1α,25-dihydroxyvitamin D3 and calcium–aluminate–chitosan scaffolds with dental pulp cells, Clin. Oral Invest. 2020;24:663–674. doi: 10.1007/s00784-019-02906-z. [DOI] [PubMed] [Google Scholar]
- 324.Zhu N., Chatzistavrou X., Ge L., Qin M., Papagerakis P., Wang Y. Biological properties of modified bioactive glass on dental pulp cells. J. Dent. 2019;83:18–26. doi: 10.1016/j.jdent.2019.01.017. [DOI] [PubMed] [Google Scholar]
- 325.Klinger C., Żółtowska-Aksamitowska S., Wysokowski M., Tsurkan M.V., Galli R., Petrenko P., Machałowski T., Ereskovsky A., Martinović R., Muzychka L., Smolii R.B., Bechmann N., Ivanenko V., Schupp P.J., Jesionowski T., Giovine M., Joseph Y., Bornstein S.R., Voronkina A., Ehrlich H. Express Method for isolation of ready-to-use 3d chitin scaffolds from Aplysinaarcheri (Aplysineidae: verongiida) demosponge. Mar. Drugs. 2019;17:131–154. doi: 10.3390/md17020131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 326.Gopinathan J., Noh I. Recent trends in bioinks for 3D printing. Biomater. Res. 2018;22:11–25. doi: 10.1186/s40824-018-0122-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 327.Sakamoto A., Saotome M., Iguchi K., Maekawa Y. Marine-derived omega-3 polyunsaturated fatty acids and heart failure: current understanding for basic to clinical relevance. Int. J. Mol. Sci. 2019;20:4025–4043. doi: 10.3390/ijms20164025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Cardoso M.J., Costa R.R., Mano J.F. Marine origin polysaccharides in drug delivery systems. Mar. Drugs. 2016;14:34–60. doi: 10.3390/md14020034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 329.Popov A.A., Sergeeva N.S., Britaev T.A., Komlev V.S., Sviridova I.K., Kirsanova V.A., Akhmedova S.A., Dgebuadze P.Y., Teterina A.Y., Kuvshinova E.A., Schanskii Y.D. Some physical, chemical, and biological parameters of samples of scleractinium coral aquaculture skeleton used for reconstruction/engineering of the bone tissue. Bull. Exp. Biol. Med. 2015;159:494–497. doi: 10.1007/s10517-015-3001-y. [DOI] [PubMed] [Google Scholar]
- 330.Lordan R., Redfern S., Tsoupras A., Zabetakis I. Inflammation and cardiovascular disease: are marine phospholipids the answer? Food Funct. 2020;11:2861–2885. doi: 10.1039/c9fo01742a. [DOI] [PubMed] [Google Scholar]
- 331.Chaugule S.R., Indap M.M., Chiplunkar S.V. Marine natural products: new avenue in treatment of osteoporosis. Front. Mar. Sci. 2017;4:384–398. [Google Scholar]
- 332.Zhang J., Wehrle E., Vetsch J.R., Paul G.R., Rubert M., Müller R. Alginate dependent changes of physical properties in 3D bioprinted cell-laden porous scaffolds affect cell viability and cell morphology. Biomed. Mater. 2019;14:65009–65042. doi: 10.1088/1748-605X/ab3c74. [DOI] [PubMed] [Google Scholar]
- 333.Raftery R.M., Woods B., Marques A.L.P., Moreira-Silva J., Silva T.H., Cryan S.A., Reis R.L., O'Brien F.J. Multifunctional biomaterials from the sea: assessing the effects of chitosan incorporation into collagen scaffolds on mechanical and biological functionality. Acta Biomater. 2016;43:160–169. doi: 10.1016/j.actbio.2016.07.009. [DOI] [PubMed] [Google Scholar]
- 334.Elango J., Zhang J., Bao B., Palaniyandi K., Wang S., Wenhui W., Robinson J.S. Rheological, biocompatibility and osteogenesis assessment of fish collagen scaffold for bone tissue engineering. Int. J. Biol. Macromol. 2016;91:51–59. doi: 10.1016/j.ijbiomac.2016.05.067. [DOI] [PubMed] [Google Scholar]
- 335.Luo Y., Lode A., Wu C., Chang J., Gelinsky M. Alginate/nanohydroxyapatite scaffolds with designed core/shell structures fabricated by 3D plotting and in situ mineralization for bone tissue engineering. ACS Appl. Mater. Interfaces. 2015;7:6541–6549. doi: 10.1021/am508469h. [DOI] [PubMed] [Google Scholar]
- 336.Yan J., Miao Y., Tan H., Zhou T., Ling Z., Chen Y., Xing X., Hu X. Injectable Alginate/hydroxyapatite gel scaffold combined with gelatin microspheres for drug delivery and bone tissue engineering. Mater. Sci. Eng. C. 2016;63:274–284. doi: 10.1016/j.msec.2016.02.071. [DOI] [PubMed] [Google Scholar]
- 337.Mutsenko V.V., Gryshkov O., Lauterboeck L., Rogulska O., Tarusin D.N., Bazhenov V.V., Schütz K., Brüggemeier S., Gossla E., Akkineni A.R., Meißner H., Lode A., Meschke S., Fromont J., Stelling A.L., Tabachnik K.R., Gelinsky M., Nikulin S., Rodin S., Tonevitsky A.G., Petrenko A.Y., Glasmacher B., Schupp P.J., Ehrlich H. Novel chitin scaffolds derived from marine sponge Ianthella basta for tissue engineering approaches based on human mesenchymal stromal cells: biocompatibility and cryopreservation. Int. J. Biol. Macromol. 2017;104:1955–1965. doi: 10.1016/j.ijbiomac.2017.03.161. [DOI] [PubMed] [Google Scholar]
- 338.Mutsenko V.V., Bazhenov V.V., Rogulska O., Tarusin D.N., Schütz K., Brüggemeier S., Gossla E., Akkineni A.R., Meißner H., Lode A., Meschke S., Ehrlich A., Petović S., Martinović R., Djurović M., Stelling A.L., Nikulin S., Rodin S., Tonevitsky A., Gelinsky M., Petrenko A.Y., Glasmacher B., Ehrlich H. 3D chitinous scaffolds derived from cultivated marine demosponge aplysina aerophoba for tissue engineering approaches based on human mesenchymal stromal cells. Int. J. Biol. Macromol. 2017;104:1966–1974. doi: 10.1016/j.ijbiomac.2017.03.116. [DOI] [PubMed] [Google Scholar]
- 339.Carvalho D.N., López-Cebral R., Sousa R.O., Alves A.L., Reys L.L., Silva S.S., Oliveira J.M., Reis R.L., Silva T.H. Marine collagen-chitosan-fucoidan cryogels as cell-laden biocomposites envisaging tissue engineering. Biomed. Mater. 2020;15:55030–55351. doi: 10.1088/1748-605X/ab9f04. [DOI] [PubMed] [Google Scholar]
- 340.Freeman F.E., Kelly D.J. Tuning alginate bioink stiffness and composition for controlled growth factor delivery and to spatially direct MSC fate within bioprinted tissues. Sci. Rep. 2017;7:17042–17053. doi: 10.1038/s41598-017-17286-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 341.Park T.Y., Yang Y.J., Ha D.H., Cho D.W., Cha H.J. Marine-derived natural polymer-based bioprinting ink for biocompatible, durable, and controllable 3D constructs. Biofabrication. 2019;11:35001–35034. doi: 10.1088/1758-5090/ab0c6f. [DOI] [PubMed] [Google Scholar]
- 342.Liu J., Tagami T., Ozeki T. Fabrication of 3D-printed fish-gelatin-based polymer hydrogel patches for local delivery of PEGylated liposomal doxorubicin. Mar. Drugs. 2020;18:325–335. doi: 10.3390/md18060325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 343.Kang M.G., Lee M.Y., Cha J.M., Lee J.K., Lee S.C., Kim J., Hwang Y.S., Bae H. Nanogels derived from fish gelatin: application to drug delivery system. Mar. Drugs. 2019;17:246–256. doi: 10.3390/md17040246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 344.Li H., Tan Y.J., Li L. A strategy for strong interface bonding by 3D bioprinting of oppositely charged κ-carrageenan and gelatin hydrogels. Carbohydr. Polym. 2018;198:261–269. doi: 10.1016/j.carbpol.2018.06.081. [DOI] [PubMed] [Google Scholar]
- 345.Demirtaş T.T., Irmak G., Gümüşderelioğlu M. A bioprintable form of chitosan hydrogel for bone tissue engineering. Biofabrication. 2017;9:35003–35033. doi: 10.1088/1758-5090/aa7b1d. [DOI] [PubMed] [Google Scholar]
- 346.Kim M.H., Lee Y.W., Jung W.K., Oh J., Nam S.Y. Enhanced rheological behaviors of alginate hydrogels with carrageenan for extrusion-based bioprinting. J. Mech. Behav. Biomed. Mater. 2019;98:187–194. doi: 10.1016/j.jmbbm.2019.06.014. [DOI] [PubMed] [Google Scholar]
- 347.Köpf M., Campos D.F.D., Blaeser A., Sen K.S., Fischer H. A tailored three-dimensionally printable Agarose–collagen blend allows encapsulation, spreading, and attachment of human umbilical artery smooth muscle cells. Biofabrication. 2016;8:25011–25025. doi: 10.1088/1758-5090/8/2/025011. [DOI] [PubMed] [Google Scholar]
- 348.Kreimendahl F., Köpf M., Thiebes A.L., Duarte Campos D.F., Blaeser A., Schmitz-Rode T., Apel C., Jockenhoevel S., Fischer H. Three-dimensional printing and angiogenesis: tailored agarose-type I collagen blends comprise three-dimensional printability and angiogenesis potential for tissue-engineered substitutes, tissue eng. Part C. Methods. 2017;23:604–615. doi: 10.1089/ten.TEC.2017.0234. [DOI] [PubMed] [Google Scholar]
- 349.Yoon H.J., Shin S.R., Cha J.M., Lee S.H., Kim J.H., Do J.T., Song H., Bae H. Cold water fish gelatin methacryloyl hydrogel for tissue engineering application. PLoS One. 2016;11 doi: 10.1371/journal.pone.0163902. 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 350.Zhang X., Kim G.J., Kang M.G., Lee J.K., Seo J.W., Do J.T., Hong K., Cha J.M., Shin S.R., Bae H. Marine biomaterial-based bioinks for generating 3D printed tissue constructs. Mar. Drugs. 2018;16:484–496. doi: 10.3390/md16120484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 351.Diaz-Rodriguez P., López-Álvarez M., Serra J., González P., Landín M. Current stage of marine ceramic grafts for 3D bone tissue regeneration. Mar. Drugs. 2019;17:1–16. doi: 10.3390/md17080471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.López-Álvarez M., Pérez-Davila S., Rodríguez-Valencia C., González P., Serra J. The improved biological response of shark tooth bioapatites in a comparative in vitro study with synthetic and bovine bone grafts. Biomed. Mater. 2016;11:35011–35023. doi: 10.1088/1748-6041/11/3/035011. [DOI] [PubMed] [Google Scholar]
- 353.Diaz-Rodriguez P., Garcia-Triñanes P., Echezarreta López M.M., Santoveña A., Landin M. Mineralized alginate hydrogels using marine carbonates for bone tissue engineering applications. Carbohydr. Polym. 2018;195:235–242. doi: 10.1016/j.carbpol.2018.04.101. [DOI] [PubMed] [Google Scholar]
- 354.Nandi S.K., Kundu B., Mukherjee J., Mahato A., Datta S., Balla V.K. Converted marine coral hydroxyapatite implants with growth factors: in vivo bone regeneration. Mater. Sci. Eng. C. 2015;49:816–823. doi: 10.1016/j.msec.2015.01.078. [DOI] [PubMed] [Google Scholar]
- 355.Decambron A., Fournet A., Bensidhoum M., Manassero M., Sailhan F., Petite H., Logeart-Avramoglou D., Viateau V. Low-dose BMP-2 and MSC dual delivery onto coral scaffold for critical-size bone defect regeneration in sheep. J. Orthop. Res. 2017;35:2637–2645. doi: 10.1002/jor.23577. [DOI] [PubMed] [Google Scholar]
- 356.Hongmin L., Wei Z., Xingrong Y., Jing W., Wenxin G., Jihong C., Xin X., Fulin C. Osteoinductive nanohydroxyapatite bone substitute prepared via in situ hydrothermal transformation of cuttlefish bone. J. Biomed. Mater. Res. B Appl. Biomater. 2015;103:816–824. doi: 10.1002/jbm.b.33261. [DOI] [PubMed] [Google Scholar]
- 357.Neto A.S., Brazete D., Ferreira J.M.F. Cuttlefish bone-derived biphasic calcium phosphate scaffolds coated with sol-gel derived bioactive glass. Mater (Basel) 2019;12:2711–2721. doi: 10.3390/ma12172711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Wysokowski M., Machalowski T., Petrenko I., Schimpf C., Rafaja D., Galli R., Zietek J., Pantovic S., Voronkina A., Kovalchuk V. 3D chitin scaffolds of marine demosponge origin for biomimetic mollusk hemolymph-associated biomineralization ex-vivo. Mar. Drugs. 2020;18:123–148. doi: 10.3390/md18020123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Sheehy E.J., Lemoine M., Clarke D., Gonzalez Vazquez A., O'Brien F.J. The incorporation of marine coral microparticles into collagen-based scaffolds promotes osteogenesis of human mesenchymal stromal cells via calcium ion signalling. Mar. Drugs. 2020;18:74–93. doi: 10.3390/md18020074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 360.Ehrlich H., Etnoyer P., Litvinov S., Olennikova M., Domaschke H., Hanke T., Born R., Worch H. Biomaterial structure in deep-sea bamboo coral (Anthozoa: gorgonacea: Isididae): perspectives for the development of bone implants and templates for tissue engineering. Mater. Werkst. 2006;37:552–557. [Google Scholar]
- 361.Du B., Gao Y., Deng Y., Zhao Y., Lai C., Guo Z., Rong M., Zhou L. Local delivery of RhVEGF165 through biocoated NHA/coral block grafts in critical-sized dog mandible defects: a histological study at the early stages of bone healing. Int. J. Clin. Exp. Med. 2015;8:4940–4953. [PMC free article] [PubMed] [Google Scholar]
- 362.Schubert M., Binnewerg B., Voronkina A., Muzychka L., Wysokowski M., Petrenko I., Kovalchuk V., Tsurkan M., Martinovic R., Bechmann N., Ivanenko V.N., Fursov A., Smolii O.B., Fromont J., Joseph Y., Bornstein S.R., Giovine M., Erpenbeck D., Guan K., Ehrlich H. Naturally prefabricated marine biomaterials: isolation and applications of flat chitinous 3D scaffolds from Ianthella labyrinthus (Demospongiae: verongiida) Int. J. Mol. Sci. 2019;20:5105. doi: 10.3390/ijms20205105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Nandi S.K., Kundu B., Mahato A., Thakur N.L., Joardar S.N., Mandal B.B. In vitro and in vivo evaluation of the marine sponge skeleton as a bone mimicking biomaterial. Integr. Biol. (Camb). 2015;7:250–262. doi: 10.1039/c4ib00289j. [DOI] [PubMed] [Google Scholar]
- 364.Carpena N.T., Min Y.K., Lee B.T. Improved in vitro biocompatibility of surface-modified hydroxyapatite sponge scaffold with gelatin and BMP-2 in comparison against a commercial bone allograft. ASAIO J. 2015;61:78–86. doi: 10.1097/MAT.0000000000000155. [DOI] [PubMed] [Google Scholar]
- 365.Wysokowski M., Machalowski T., Petrenko I., Schimpf C., Rafaja D., Galli R., Ziętek J., Pantović S., Voronkina A., Kovalchuk V., Ivanenko V.N., Hoeksma B.W., Diaz C., Khrunyk Y., Stelling A.L., Giovine M., Jesionowski T., Ehrlich H. 3D chitin scaffolds of marine demosponge origin for biomimetic mollusk hemolymph-associated biomineralization ex-vivo. Mar. Drugs. 2020;18:123–149. doi: 10.3390/md18020123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 366.Cozza N., Monte F., Bonani W., Aswath P., Motta A., Migliaresi C. Bioactivity and mineralization of natural hydroxyapatite from cuttlefish bone and Bioglass(®) Co-sintered bioceramics. J. Tissue Eng. Regen. Med. 2018;12:e1131–e1142. doi: 10.1002/term.2448. [DOI] [PubMed] [Google Scholar]
- 367.Kim B.S., Yang S.S., Yoon J.H., Lee J. Enhanced bone regeneration by silicon-substituted hydroxyapatite derived from cuttlefish bone. Clin. Oral Implants Res. 2017;28:49–56. doi: 10.1111/clr.12613. [DOI] [PubMed] [Google Scholar]
- 368.López-Álvarez M., Vigo E., Rodríguez-Valencia C., Outeiriño-Iglesias V., González P., Serra J. In vivo evaluation of shark teeth-derived bioapatites. Clin. Oral Implants Res. 2017;28:e91–e100. doi: 10.1111/clr.12934. [DOI] [PubMed] [Google Scholar]
- 369.Higa K., Kitamura N., Kurokawa T., Goto K., Wada S., Nonoyama T., Kanaya F., Sugahara K., Gong J.P., Yasuda K. Fundamental biomaterial properties of tough glycosaminoglycan-containing double network hydrogels newly developed using the molecular stent method. Acta Biomater. 2016;43:38–49. doi: 10.1016/j.actbio.2016.07.023. [DOI] [PubMed] [Google Scholar]
- 370.Kuo C.Y., Chen C.H., Hsiao C.Y., Chen J.P. Incorporation of chitosan in biomimetic gelatin/chondroitin-6-sulfate/hyaluronan cryogel for cartilage tissue engineering, carbohydr. Polym. 2015;117:722–730. doi: 10.1016/j.carbpol.2014.10.056. [DOI] [PubMed] [Google Scholar]
- 371.Luo Z., Sun W., Fang J., Lee K., Li S., Gu Z., Dokmeci M.R., Khademhosseini A. Biodegradable gelatin methacryloyl microneedles for transdermal drug delivery. Adv. Healthc. Mater. 2019;8 doi: 10.1002/adhm.201801054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 372.Tihan G.T., Rău I., Zgârian R.G., Ungureanu C., Barbaresso R.C., Kaya M.G.A., Dinu-Pîrvu C., Ghica M.V. Oxytetracycline versus doxycycline collagen sponges designed as potential carrier supports in biomedical applications. Pharmaceutics. 2019;11:363–383. doi: 10.3390/pharmaceutics11080363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 373.Biasin V., Wygrecka M., Marsh L.M., Becker-Pauly C., Brcic L., Ghanim B., Klepetko W., Olschewski A., Kwapiszewska G. Meprin β contributes to collagen deposition in lung fibrosis. Sci. Rep. 2017;7:39969. doi: 10.1038/srep39969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 374.An B., Lin Y.S., Brodsky B. Collagen interactions: drug design and delivery. Adv. Drug Deliv. Rev. 2016;97:69–84. doi: 10.1016/j.addr.2015.11.013. [DOI] [PubMed] [Google Scholar]
- 375.Morath I., Hartmann T.N., Orian-Rousseau V. CD44: more than a mere stem cell marker. Int. J. Biochem. Cell Biol. 2016;81:166–173. doi: 10.1016/j.biocel.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 376.Ndinguri M.W., Zheleznyak A., Lauer J.L., Anderson C.J., Fields G.B. Application of collagen-model triple-helical peptide-amphiphiles for CD44-targeted drug delivery systems. J. Drug Deliv. 2012;2012:592602. doi: 10.1155/2012/592602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Severino P., da Silva C.F., Andrade L.N., de Lima Oliveira D., Campos J., Souto E.B. Alginate nanoparticles for drug delivery and targeting. Curr. Pharmaceut. Des. 2019;25:1312–1334. doi: 10.2174/1381612825666190425163424. [DOI] [PubMed] [Google Scholar]
- 378.Flórez-Fernández N., Domínguez H., Torres M.D. A green approach for alginate extraction from sargassum muticum Brown seaweed using ultrasound-assisted technique. Int. J. Biol. Macromol. 2019;124:451–459. doi: 10.1016/j.ijbiomac.2018.11.232. [DOI] [PubMed] [Google Scholar]
- 379.Agüero L., Zaldivar-Silva D., Peña L., Dias M.L. Alginate microparticles as oral colon drug delivery device: a review. Carbohydr. Polym. 2017;168:32–43. doi: 10.1016/j.carbpol.2017.03.033. [DOI] [PubMed] [Google Scholar]
- 380.Prasad K., Kaneko Y., Kadokawa J. Novel gelling systems of kappa-, iota- and lambda-carrageenans and their composite gels with cellulose using ionic liquid. Macromol. Biosci. 2009;9:376–382. doi: 10.1002/mabi.200800179. [DOI] [PubMed] [Google Scholar]
- 381.Posocco B., Dreussi E., De Santa J., Toffoli G., Abrami M., Musiani F., Grassi M., Farra R., Tonon F., Grassi G. Polysaccharides for the delivery of antitumor drugs. Materials (Basel) 2015;8:2569–2615. [Google Scholar]
- 382.Obaidat R.M., Alnaief M., Mashaqbeh H. Investigation of carrageenan aerogel microparticles as a potential drug carrier. AAPS PharmSciTech. 2018;19:2226–2236. doi: 10.1208/s12249-018-1021-4. [DOI] [PubMed] [Google Scholar]
- 383.Kalsoom Khan A., Saba A.U., Nawazish S., Akhtar F., Rashid R., Mir S., Nasir B., Iqbal F., Afzal S., Pervaiz F., Murtaza G. Carrageenan based bionanocomposites as drug delivery tool with special emphasis on the influence of ferromagnetic nanoparticles. Oxid. Med. Cell. Longev. 2017;2017:8158315. doi: 10.1155/2017/8158315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 384.Yegappan R., Selvaprithiviraj V., Amirthalingam S., Jayakumar R. Carrageenan based hydrogels for drug delivery, tissue engineering and wound healing. Carbohydr. Polym. 2018;198:385–400. doi: 10.1016/j.carbpol.2018.06.086. [DOI] [PubMed] [Google Scholar]
- 385.S. Toumi, M.M. Yahoum, S. Lefnaoui, A. Hadjsadok, Synthesis, characterization and potential application of hydrophobically modified carrageenan derivatives as pharmaceutical excipients, Carbohydr. Polym. 251 (2021), 116997. [DOI] [PubMed]
- 386.Thakur A., Jaiswal M.K., Peak C.W., Carrow J.K., Gentry J., Dolatshahi-Pirouz A., Gaharwar A.K. Injectable shear-thinning nanoengineered hydrogels for stem cell delivery. Nanoscale. 2016;8:12362–12372. doi: 10.1039/c6nr02299e. [DOI] [PubMed] [Google Scholar]
- 387.Huang Y.C., Li R.Y., Chen J.Y., Chen J.K. Biphasic release of gentamicin from chitosan/fucoidan nanoparticles for pulmonary delivery. Carbohydr. Polym. 2016;138:114–122. doi: 10.1016/j.carbpol.2015.11.072. [DOI] [PubMed] [Google Scholar]
- 388.Yu S.H., Wu S.J., Wu J.Y., Wen D.Y., Mi F.L. Preparation of fucoidan-shelled and genipin-crosslinked chitosan beads for antibacterial application. Carbohydr. Polym. 2015;126:97–107. doi: 10.1016/j.carbpol.2015.02.068. [DOI] [PubMed] [Google Scholar]
- 389.Ustyuzhanina N.E., Bilan M.I., Gerbst A.G., Ushakova N.A., Tsvetkova E.A., Dmitrenok A.S., Usov A.I., Nifantiev N.E. Anticoagulant and antithrombotic activities of modified xylofucan sulfate from the Brown alga punctaria plantaginea. Carbohydr. Polym. 2016;136:826–833. doi: 10.1016/j.carbpol.2015.09.102. [DOI] [PubMed] [Google Scholar]
- 390.Kim J.M., Bae I., Lim K.S., Park J., Park D.S., Lee S., Jang E., Ji M.S., Sim D.S., Hong Y.J., Ahn Y., Park J.C., Cho J.G., Kang J.C., Kim I.S., Jeong M.H. A method for coating fucoidan onto bare metal stent and in vivo evaluation. Prog. Org. Coating. 2014;78:348–356. [Google Scholar]
- 391.Ozaltin K., Lehocky M., Humpolicek P., Pelkova J., Di Martino A., Karakurt I., Saha P. Anticoagulant polyethylene terephthalate surface by plasma-mediated fucoidan immobilization. Polymers (Basel) 2019;11:750–761. doi: 10.3390/polym11050750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.da Silva L.C.R.P., Todaro V., do Carmo F.A., Frattani F.S., de Sousa V.P., Rodrigues C.R., Sathler P.C., Cabral L.M. A promising oral fucoidan-based antithrombotic nanosystem: development, activity and safety. Nanotechnology. 2018;29:165102. doi: 10.1088/1361-6528/aaae5b. [DOI] [PubMed] [Google Scholar]
- 393.Zhao X., Guo F., Hu J., Zhang L., Xue C., Zhang Z., Li B. Antithrombotic activity of oral administered low molecular weight fucoidan from Laminaria japonica. Thromb. Res. 2016;144:46–52. doi: 10.1016/j.thromres.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 394.Yang Z.H., Emma-Okon B., Remaley A.T. Dietary marine-derived long-chain monounsaturated fatty acids and cardiovascular disease risk: a mini review. Lipids Health Dis. 2016;15:201–209. doi: 10.1186/s12944-016-0366-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Endo J., Arita M. Cardioprotective mechanism of omega-3 polyunsaturated fatty acids. J. Cardiol. 2016;67:22–27. doi: 10.1016/j.jjcc.2015.08.002. [DOI] [PubMed] [Google Scholar]
- 396.Calder P.C. Marine omega-3 fatty acids and inflammatory processes: effects, mechanisms and clinical relevance. Biochim. Biophys. Acta. 2015;1851:469–484. doi: 10.1016/j.bbalip.2014.08.010. [DOI] [PubMed] [Google Scholar]
- 397.Yang Z.H., Bando M., Sakurai T., Chen Y., Emma-Okon B., Wilhite B., Fukuda D., Vaisman B., Pryor M., Wakabayashi Y., Sampson M., Yu Z.X., Sakurai A., Zarzour A., Miyahara H., Takeo J., Sakaue H., Sata M., Remaley A.T. Long-chain monounsaturated fatty acid-rich fish oil attenuates the development of atherosclerosis in mouse models. Mol. Nutr. Food Res. 2016;60:2208–2218. doi: 10.1002/mnfr.201600142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 398.Heydari B., Abdullah S., Pottala J.V., Shah R., Abbasi S., Mandry D., Francis S.A., Lumish H., Ghoshhajra B.B., Hoffmann U., Appelbaum E., Feng J.H., Blankstein R., Steigner M., McConnell J.P., Harris W., Antman E.M., Jerosch-Herold M., Kwong R.Y. Effect of omega-3 acid ethyl esters on left ventricular remodeling after acute myocardial infarction: the OMEGA-REMODEL randomized clinical trial. Circulation. 2016;134:378–391. doi: 10.1161/CIRCULATIONAHA.115.019949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 399.Oh G.W., Nguyen V.T., Heo S.Y., Ko S.C., Kim C.S., Park W.S., Choi I.W., Jung W.K. 3D PCL/fish collagen composite scaffolds incorporating osteogenic abalone protein hydrolysates for bone regeneration application: in vitro and in vivo studies. J. Biomater. Sci. Polym. Ed. 2020;22:1–17. doi: 10.1080/09205063.2020.1834908. [DOI] [PubMed] [Google Scholar]
- 400.Brion A., Zhang G., Dossot M., Moby V., Dumas D., Hupont S., Piet M.H., Bianchi A., Mainard D., Galois L., Gillet P P. Nacre extract restores the mineralization capacity of subchondral osteoarthritis osteoblasts. J. Struct. Biol. 2015;192:500–509. doi: 10.1016/j.jsb.2015.10.012. [DOI] [PubMed] [Google Scholar]
- 401.Morita M., Ogawa H., Ohno O., Yamori T., Suenaga K., Toyoshima C. Biselyngbyasides, cytotoxic marine macrolides, are novel and potent inhibitors of the Ca2+ pumps with a unique mode of binding. FEBS Lett. 2015;589:1406–1411. doi: 10.1016/j.febslet.2015.04.056. [DOI] [PubMed] [Google Scholar]
- 402.Sato E., Sato M., Tanabe Y., Nakajima N., Ohkubo A., Suenaga K. Total synthesis of biselyngbyaside. J. Org. Chem. 2017;82:6770–6777. doi: 10.1021/acs.joc.7b00905. [DOI] [PubMed] [Google Scholar]
- 403.Decroos C., Clausen D.J., Haines B.E., Wiest O., Williams R.M., Christianson D.W. Variable active site loop conformations accommodate the binding of macrocyclic largazole analogues to HDAC8. Biochemistry. 2015;54:2126–2135. doi: 10.1021/acs.biochem.5b00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 404.Cantley M.D., Zannettino A.C.W., Bartold P.M., Fairlie D.P., Haynes D.R. Histone deacetylases (HDAC) in physiological and pathological bone remodelling. Bone. 2017;95:162–174. doi: 10.1016/j.bone.2016.11.028. [DOI] [PubMed] [Google Scholar]
- 405.Thao N.P., Luyen B.T.T., Lee S.H., Jang H.D., Kiem P.V., Minh C.V., Kim Y.H. Antiosteoporotic and antioxidant activities of diterpenoids from the Vietnamese soft corals Sinularia maxima and Lobophytum crassum. Med. Chem. Res. 2015;24:3551–3560. [Google Scholar]
- 406.Yuan Y., Zheng Y., Zhou J., Geng Y., Zou P., Li Y., Zhang C. Polyphenol-rich extracts from Brown macroalgae Lessonia trabeculate attenuate hyperglycemia and modulate gut microbiota in high-fat diet and streptozotocin-induced diabetic rats. J. Agric. Food Chem. 2019;67:12472–12480. doi: 10.1021/acs.jafc.9b05118. [DOI] [PubMed] [Google Scholar]
- 407.Lee S.H., Jeon Y.J. Efficacy and safety of a dieckol-rich extract (AG-Dieckol) of Brown algae, Ecklonia cava, in pre-diabetic individuals: a double-blind, randomized, placebo-controlled clinical trial. Food Funct. 2015;6:853–858. doi: 10.1039/c4fo00940a. [DOI] [PubMed] [Google Scholar]
- 408.Xia E.Q., Zhu S.S., He M.J., Luo F., Fu C.Z., Zou T.B. Marine peptides as potential agents for the management of type 2 diabetes mellitus-A prospect. Mar. Drugs. 2017;15:88–103. doi: 10.3390/md15040088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 409.Venkatesan J., Anil S., Kim S.K., Shim M.S. Marine fish proteins and peptides for cosmeceuticals: a review. Mar. Drugs. 2017;15:143–160. doi: 10.3390/md15050143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 410.Mitura S., Sionkowska A., Jaiswal A. Biopolymers for hydrogels in cosmetics: review. J. Mater. Sci. Mater. Med. 2020;31:50–63. doi: 10.1007/s10856-020-06390-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 411.Avila Rodríguez M.I., Rodríguez Barroso L.G., Sánchez M.L. Collagen: a review on its sources and potential cosmetic applications. J. Cosmet. Dermatol. 2018;17:20–26. doi: 10.1111/jocd.12450. [DOI] [PubMed] [Google Scholar]
- 412.Sionkowska A., Skrzyński S., Śmiechowski K., Kołodziejczak A. The review of versatile application of collagen. Polym. Adv. Technol. 2017;28:4–9. [Google Scholar]
- 413.Sionkowska A., Kaczmarek B., Michalska M., Lewandowska K., Grabska S. Preparation and characterization of collagen/chitosan/hyaluronic acid thin films for application in hair care cosmetics. Pure Appl. Chem. 2017;89:1829–1839. [Google Scholar]
- 414.Kong S.Z., Li D.D., Luo H., Li W.J., Huang Y.M., Li J.C., Hu Z., Huang N., Guo M.H., Chen Y., Li S.D. Anti-photoaging effects of chitosan oligosaccharide in ultraviolet-irradiated hairless mouse skin. Exp. Gerontol. 2018;103:27–34. doi: 10.1016/j.exger.2017.12.018. [DOI] [PubMed] [Google Scholar]
- 415.Aranaz I., Acosta N., Civera C., Elorza B., Mingo J., Castro C., Gandía M.L.L., Heras Caballero A. Cosmetics and cosmeceutical applications of chitin, chitosan and their derivatives. Polymers (Basel) 2018;10:213–237. doi: 10.3390/polym10020213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 416.Stepniak I., Galinski M., Nowacki K., Wysokowski M., Jakubowska P., Bazhenov V.V., Leisegang T., Ehrlich H., Jesionowski T. A novel chitosan/sponge chitin origin material as a membrane for supercapacitors - preparation and characterization. RSC Adv. 2016;6:4007–4013. [Google Scholar]
- 417.Ruocco N., Costantini S., Guariniello S., Costantini M. Polysaccharides from the marine environment with pharmacological, cosmeceutical and nutraceutical potential. Molecules. 2016;21:551–566. doi: 10.3390/molecules21050551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 418.Ahn J.H., Kim D.W., Park C.W., Kim B., Sim H., Kim H.S., Lee T.K., Lee J.C., Yang G.E., Her Y., Park J.H., Sim T.H., Lee H.S., Won M.H. Laminarin attenuates ultraviolet-induced skin damage by reducing superoxide anion levels and increasing endogenous antioxidants in the dorsal skin of mice. Mar. Drugs. 2020;18:345–358. doi: 10.3390/md18070345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Al-Sibani M., Al-Harrasi A., Neubert R.H.H. Effect of hyaluronic acid initial concentration on cross-linking efficiency of hyaluronic acid - based hydrogels used in biomedical and cosmetic applications. Pharmazie. 2017;72:81–86. doi: 10.1691/ph.2017.6133. [DOI] [PubMed] [Google Scholar]
- 420.Bukhari S.N.A., Roswandi N.L., Waqas M., Habib H., Hussain F., Khan S., Sohail M., Ramli N.A., Thu H.E., Hussain Z. Hyaluronic acid, a promising skin rejuvenating biomedicine: a review of recent updates and pre-clinical and clinical investigations on cosmetic and nutricosmetic effects. Int. J. Biol. Macromol. 2018;120:1682–1695. doi: 10.1016/j.ijbiomac.2018.09.188. [DOI] [PubMed] [Google Scholar]
- 421.Hu Z., Gänzle M.G. Challenges and opportunities related to the use of chitosan as a food preservative. J. Appl. Microbiol. 2019;126:1318–1331. doi: 10.1111/jam.14131. [DOI] [PubMed] [Google Scholar]
- 422.Brasselet C., Pierre G., Dubessay P., Dols-Lafargue M., Coulon J., Maupeu J., Vallet-Courbin A., de Baynast H., Doco T., Michaud P., Delattre C. Modification of chitosan for the generation of functional derivatives. Appl. Sci. 2019;9:1321–1353. [Google Scholar]
- 423.Nešić A., Cabrera-Barjas G., Dimitrijević-Branković S., Davidović S., Radovanović N., Delattre C. Prospect of polysaccharide-based materials as advanced food packaging. Molecules. 2020;25:135–169. doi: 10.3390/molecules25010135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 424.Gheorghita R.P., Lobiuc A., Dimian M., Covasa M. Alginate: from food industry to biomedical applications and management of metabolic disorders. Polymers (Basel) 2020;12:2417–2444. doi: 10.3390/polym12102417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Singh S., Gaikwad K.K., Lee Y.S. Antimicrobial and antioxidant properties of polyvinyl alcohol bio composite films containing seaweed extracted cellulose nano-crystal and basil leaves extract. Int. J. Biol. Macromol. 2018;107:1879–1887. doi: 10.1016/j.ijbiomac.2017.10.057. [DOI] [PubMed] [Google Scholar]
- 426.Gao H., Duan B., Lu A., Deng H., Du Y., Shi X., Zhang L. Fabrication of cellulose nanofibers from waste Brown algae and their potential application as milk thickeners. Food Hydrocolloids. 2018;79:473–481. [Google Scholar]