Abstract
Background
People with Chagas disease may develop progressive and lethal heart conditions. Drugs to eliminate the parasite Trypanosomacruzi (T cruzi) currently carry limited therapeutic value and are used in the early stages of the disease. Extending the use of these drugs to treat chronic chagasic cardiomyopathy (CCC) has also been proposed.
Objectives
To assess the benefits and harms of nitrofurans and trypanocidal drugs for treating late‐stage, symptomatic Chagas disease and CCC in terms of blood parasite reduction or clearance, mortality, adverse effects, and quality of life.
Search methods
We searched the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, and LILACS databases on 12 November 2019. We also searched two clinical trials registers, ClinicalTrials.gov and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP), on 3 December 2019.
Selection criteria
We included randomised controlled trials (RCTs) assessing trypanocidal drugs versus placebo or no treatment for late‐stage, symptomatic Chagas disease and CCC.
Data collection and analysis
We conducted the reporting of the review according the standard Cochrane methods. Two review authors independently retrieved articles, performed data extraction, and assessed risk of bias. Any disagreements were resolved by a third review author. We contacted study authors for additional information.
Main results
We included two studies in this review update. One RCT randomly assigned 26 participants to benznidazole 5 mg/kg/day; 27 participants to nifurtimox 5 mg/kg/day; and 24 participants to placebo for 30 days. The second RCT, newly included in this update, randomised 1431 participants to benznidazole 300 mg/day for 40 to 80 days and 1423 participants to placebo. We also identified one ongoing study.
Benznidazole compared to placebo
At five‐year follow‐up, low quality of the evidence suggests that there may be a benefit of benznidazole when compared to placebo for clearance or reduction of antibody titres (risk ratio (RR) 1.25, 95% confidence interval (CI) 1.14 to 1.37; 1 trial; 1896 participants). We are uncertain about the effects of benznidazole for the clearance of parasitaemia demonstrated by negative xenodiagnosis, blood culture, and/or molecular assays due to very limited evidence.
Low quality of the evidence suggests that when compared to placebo, benznidazole may make little to no difference in the risk of heart failure (RR 0.89, 95% CI 0.69 to 1.14; 1 trial; 2854 participants) and ventricular tachycardia (RR 0.80, 95% CI 0.51 to 1.26; 1 trial; 2854 participants).
We found moderate quality of the evidence that adverse events increase with benznidazole when compared to placebo (RR 2.52, 95% CI 2.09 to 3.03; 1 trial; 2854 participants). Adverse effects were observed in 23.9% of patients in the benznidazole group compared to 9.5% in the placebo group. The most frequent adverse effects were: cutaneous rash, gastrointestinal symptoms, and peripheral polyneuropathy.
No data were available for the outcomes of pathological demonstration of tissue parasites and quality of life.
Nifurtimox compared to placebo
Data were only available for this comparison for the outcome clearance or reduction of antibody titres, and we are uncertain about the effect due to very limited evidence.
Regarding adverse events, one RCT mentioned in a general manner that nifurtimox caused intense adverse events, without any quantification.
Authors' conclusions
There is insufficient evidence to support the efficacy of the trypanocidal drugs benznidazole and nifurtimox for late‐stage, symptomatic Chagas disease and CCC.
Plain language summary
Trypanocidal drugs for late‐stage, symptomatic Chagas disease
Review question
Is trypanocidal drug treatment effective for late‐stage, symptomatic Chagas disease and chronic chagasic cardiomyopathy (CCC)?
Background
Infection with the parasite Trypanosoma cruzi causes American trypanosomiasis, or Chagas disease. The evidence shows that trypanocidal drug treatment, using nitrofuran and imidazole compounds, can cure acute trypanosomiasis cruzi infections. However, it is unclear whether these treatments are effective for late‐stage, symptomatic Chagas disease and CCC.
Study characteristics
We searched the medical literature for randomised double‐blind controlled clinical trials with or without placebo (dummy treatment) to November 2019. Randomised controlled trials are studies in which participants are assigned to one of two or more treatment groups using a random method. In a double‐blind study, neither the participants nor the researchers know who is receiving a particular treatment.
Key results
We found two trials that looked at treatment of chronic Typanosoma cruzi infection (late‐stage, symptomatic Chagas disease and CCC) with the trypanocidal drugs benznidazole and nifurtimox. After reviewing this limited evidence, we concluded that it is insufficient to support treatment with these drugs.
Certainty of the evidence
The certainty of the evidence ranges between low to very low since a small number of trials were included and the number of participants was limited. More clinical trials are therefore required to evaluate the effect and efficacy of trypanocidal drugs in late‐stage, symptomatic Chagas disease and CCC.
Summary of findings
Background
Description of the condition
American trypanosomiasis, or Chagas disease (Chagas 1909), is caused by the flagellate protozoan parasite Trypanosoma cruzi. The geographical distribution of cases of human T cruzi infection extends from the southern United States and Mexico to the south of Argentina. Since 1991, when 18 million people were infected and an estimated 100 million were at risk of contracting the infection (WHO 1991), the prevalence of Chagas disease has significantly decreased. Today, there are between 6 to 7 million infected people, most of whom reside in countries in Latin America (WHO 2017). However, because of the growing population movement, neglected tropical diseases have spread and are becoming a major sanitary problem and an economic burden for endemic and non‐endemic countries (Rassi 2010; Sbaraglini 2016; Urbina 2015), such as Canada, the United States, Europe, and the Western Pacific (Fumadó 2014; Imai 2015; Ramos 2012; Valerio‐Sallent 2012; WHO 2015). The estimated number of infected people in Europe is between 14,000 to 181,000, with most cases being reported in Spain (Ramos 2012), Italy (Briceno 2016), and the United Kingdom (Gobbi 2014).
The World Health Organization (WHO) reports that the disease has already affected approximately 18 million people around the world, and estimates that 100 million people – 25% of the Latin American population – are at risk of contracting it in the coming years (WHO 1991).
The infection may be caught by the following routes.
Vectoral transmission. T cruzi parasites, living in the gut of triatomine insects, enter the bloodstream through the bite of the insect. Triatomine bugs live in the cracks and crevices of poor quality houses, especially in rural areas, and emerge at night to bite and extract blood from human hosts. T cruzi parasites are present in insect faeces that subsequently enter the human bloodstream through the wound when it is scratched or rubbed.
Transfusion‐linked transmission or iatrogenic transmission. Transfusion with blood infected with T cruzi or allotransplants is the second most important transmission pathway.
Vertical transmission from an infected mother to foetus, laboratory accidents, and other routes have also been described (Grant 1989; Monteón‐Padilla 1999; Nickerson 1989; Pinto 1995; Schmunis 1999).
Oral transmission. This is a more recent form of transmission, through contaminated food (Dias 2009).
The clinical course of Chagas disease is characterised by an initial 'acute phase', in which parasitaemia is easily demonstrated. During this phase, clinical symptoms may or may not be apparent; however, most people spontaneously improve within six to eight weeks. The presence of circulating parasites confirms the clinical diagnosis. After a few weeks of infection, a lifelong persisting immune response may occur. Chronic infection may result in an 'indeterminate phase' of Chagas disease, which is a long asymptomatic period that can last 30 to 40 years or more. The only proof of infection is a strong immune response with the high titre of anti‐T cruzi antibodies. During this phase, approximately 30% of chronically infected individuals develop irreversible alterations to heart muscles and an enlargement of the intestine (Barretto 1990; Schmunis 1994; Tanowitz 1992; WHO 1991). This is known as chronic Chagas disease, or late‐stage, symptomatic Chagas disease, and most patients will suffer from chronic chagasic cardiomyopathy (CCC), a specific type of cardiomyopathy characterised by heart failure with bizarre rhythm and/or conduction defects. Unfortunately, during the chronic phase is difficult to observe the presence of parasites either in peripheral blood or tissues, therefore immune serology, and recently immunohistochemistry and biomolecular techniques, are used as diagnostic assays (Appendix 1).
Description of the intervention
Although treatment of late‐stage, symptomatic Chagas disease and CCC is the focus of this review, it is important to put the impact of treatment into the broader context of the international efforts to control transmission. In 1991, the Health Ministries of Argentina, Brazil, Bolivia, Chile, Paraguay, and Uruguay created the Southern Cone Initiative, with the objective of eliminating the vector transmission of Chagas disease by the year 2010 and permanent blood donor screening to avoid transfusion‐linked transmission. To date, advances have been made. Because of this initiative, the prevalence of T cruzi infection has been significantly reduced. Uruguay was declared free of vectoral transmission in 1998, and in the other countries active transmission rates have fallen from 96% to 75%, and the infection rate in the young population (below 18 years of age) is now between 0.17% and 1% (Cecere 1999; Harry 2000; Oliveira‐Filho 1989; Rojas 1999; Schofield 1999). In 2010, the Pan American Health Organization (PAHO) certified the interruption of transmission by domestic vector and blood transfusion (WHO 2015).
It is hoped that through initiatives such as Southern Cone, transmission of T cruzi parasite will be significantly reduced or stopped in the near future. Unfortunately, Chagas disease will continue to impose a high social and economical cost (Cecere 1999; Harry 2000; Oliveira‐Filho 1989; Rojas 1999; Schofield 1999). This is because of the long latent phase of the disease (10 to 30 years), and the considerable number of infected people who are currently in the chronic (indeterminate) phase of Chagas disease. A high proportion of people who are currently symptomless will experience symptoms of Chagas disease in the future. Chagas disease burden has been estimated from 300,000 to over 800,000 disability‐adjusted life‐years (DALYs), and its global cost could be up to USD 7.19 billion per year (Robertson 2016; Schmuñis 2013). Furthermore, the costs of treatment can range between USD 200 in acute cases to more than USD 30,000 in chronic cases; however, the major cost component for lifetime was working days lost, which could range between 44% and 75%, depending on the programme scenario for timely diagnosis and treatment (Ramsey 2014). In contrast, screening strategies have strengthened since 2006, and vector control investment will cost about USD 720 million (USD 620 to 820 million) per year during 2015 to 2020; however, this may decrease to about USD 450 million (USD 390 to 520 million) per year during 2021 to 2030 as countries move forward in the attack phase (WHO 2015).
A report describing 10 years of experience in Honduras, Guatemala, and Bolivia focused on feasible protocols, safety of drug therapy, and treatment effectiveness concluded that it is possible to implement a Chagas disease diagnosis and treatment programme even in limited resource settings; however, treatment effectiveness may be related to differences in patients and parasite populations. New treatments with improved safety profiles, paediatric formulations, and a faster reliable test of cure are urgently needed (Yun 2009).
How the intervention might work
Since the eradication of Chagas disease is not a plausible solution (Salomao 2016; Schofield 2006), and control measures such as vector and transfusion control have been shown to be successful, efforts should now be directed towards the development of: 1) an effective prophylactic vaccine (Urbina 2015); 2) a molecular biomarker that could help in evaluating the efficacy of treatments; and 3) new compounds to treat chronically symptomatic patients.
Trypanocidal drugs have been used in the treatment of Chagas disease since the 1960s (Brener 1975), when nitrofurans and later imidazole drugs were introduced after a long history of therapeutic failures that lasted for 50 years. The trypanocidal drugs nifurtimox (3‐ methyl‐N‐`((5 ‐nitro‐2 furanyl)‐methylene)‐4‐morpholinamine 1,1 dioxide) and benznidazole (2‐nitro‐N‐(phenylmethyl)‐1H‐imidazole‐1‐acetamide) are thought to exert their action by generation of free radicals either in intact cells or Trypanosoma cruzi lysates. However, the mechanisms involved in such reactions are poorly defined (Stoppani 1999). Recently Kubata, who was studying intracellular trypanosomal arachidonic acid and its metabolism in these parasites, found an enzyme that reduced prostanoid metabolites as well as naphthoquinone and nitro heterocyclic drugs (Kubata 2002). This reduction results in drug derived quinone intermediates as a source of free radicals which cause damage in organelles of parasitic protozoa. These reactions clarify the mechanism of action for both nifurtimox and benznidazole, with a well‐recognised clinical activity limited to acute and early chronic Trypanosoma infections, achieving a cure rate up to 100%; however, due to the unwanted side effects, treatment is frequently discontinued.
Some promising results have been reported in the treatment of infected individuals in the chronic (indeterminate) phase of Chagas disease in trials carried out in Argentina and Brazil, where reduction of parasitaemia ranges from 58% to 95% (de Andrade 1996; Sosa 1998; Stoppani 1999). In addition, a Cochrane Review of 'Trypanocidal drugs for chronic asymptomatic Trypanosoma cruzi infection' showed that "nitro imidazole derivatives substantially and significantly modified parasite‐related outcomes compared to placebo and that other agents showed borderline or not significant effect" (Villar 2002); the review authors concluded that "Trypanocidal therapy, particularly nitro imidazole derivatives given to children or young adults with positive xenodiagnosis improve parasite‐related outcomes". That review has recently been updated: 13 studies were included (6 randomised controlled trials, 4 non‐randomised experiments, and 3 observational studies), and a total of 4229 participants received different trypanocidal drugs (benznidazole, nifurtimox, allopurinol and/or itraconazole). The review authors concluded that benznidazole and nifurtimox are still the treatments of choice for chronic asymptomatic patients. However, as most of the information about the efficacy of antitrypanocidal treatment comes from observational studies, consideration should be taken after advising the patient of the inconsistencies and uncertainties regarding the efficacy of trypanocidal therapy (Villar 2014).
Up to this point there has been no doubt that the pathophysiology of T cruzi in the acute phase has an important role in the aetiological treatment, whereas the participation of the parasite in the pathogenesis of chronic Chagas disease has been a question of debate for several decades. For the last 10 years, inhibitors of TcCYP51, an essential sterol for the survival of T cruzi, has been studied (miconazole, econazole, bis‐transole D0870, pozaconazole and E1224). Treatment efficacy in later stages of the disease remains questionable and controversial (Andrade 2011; Sales 2017). Furthermore, treatment effectiveness may be related to T cruzi strains, the susceptibility to different compounds or drugs, parasite genetic variability, geographical location, and the stage of Chagas disease when the treatment is used (Sales 2017; Urbina 2015). People with late‐stage, symptomatic Chagas disease are usually adults who are older than 25 years of age. Treatment in this phase of Chagas disease is usually limited to managing complications associated with the pathology found in the heart and digestive organs, and avoiding antiparasitics drugs because of potential adverse reactions. However, recent studies have shown that chronically ill patients treated with benznidazole had a significant reduction of blood antibodies compared with those in the placebo group. There is evidence that even if no cure was possible, fewer electrocardiographic abnormalities were found in the treatment group, which may be interpreted as due to the delay in tissue damage (Mady 2008; Moretti 1998; Segura 1994).
Control programmes have focused on vector and transfusion transmission; it is now time that treatment and validated biomarker are assumed as part of these strategies. However, Coura has proposed that until the ideal drug for the treatment of late‐stage, symptomatic Chagas disease and CCC is developed, chagasic patients be treated with a combination of currently used drugs such as nifurtimox and benznidazole, along with potential drugs like allopurinol, and first‐, second‐, and third‐generation antifungal agents, all of which have different mechanisms of action, in order to boost the action of different compounds and avoid the development of parasite resistance (Coura 2009).
The authors of a double‐blind, placebo‐controlled clinical trial conducted in 55 individuals from endemic areas of central Brazil concluded that at the used dose (900 mg/d), allopurinol was not effective to clear T cruzi from the peripheral blood of infected individuals (Rassi 2007).
Why it is important to do this review
New drugs, such as the modified candidate cancer ones which have shown picomolar activity against cultured T cruzi and have been efficacious in a mouse model of acute Chagas disease, have been tested (Kraus 2009).
Further studies need to be undertaken in both the acute and chronic phases of the disease, with strictly established cure criteria and specific diagnostic tests (e.g. polymerase chain reaction (PCR) methods) together with long‐term follow‐up of outcomes.
Given that the potential of trypanocidal therapy to modify CCC had yet to be evaluated (Urbina 2009), we performed this systematic review of the effects of treatment with nitrofuran or imidazole derivatives on clinically relevant outcomes in people with CCC.
Objectives
To assess the benefits and harms of nitrofurans and trypanocidal drugs for treating late‐stage, symptomatic Chagas disease and CCC in terms of blood parasite reduction or clearance, mortality, adverse effects, and quality of life.
Methods
Criteria for considering studies for this review
Types of studies
We included trials reported as full text, abstract, and unpublished data in which participants were randomised individually to the administration of trypanocidal drugs (nitrofurans or benznidazole) versus placebo or no treatment for late‐stage, symptomatic Chagas disease and CCC. We did not include cluster‐randomised trials since participants were included as individuals, without sharing any characteristics, and because we were not certain that they belong to the same group (Perman 2017).
Types of participants
Adults (between 25 and 65 years of age) with a definitive diagnosis of CCC defined clinically as the presence of either heart failure with or without specific dilated cardiomyopathy and/or tachyarrhythmia and/or heart block. A long 'incubation' period of usually more than 20 years prior to CCC was expected.
In addition, serum antibodies to T cruzi demonstrated by at least two different validated tests (haemagglutination, indirect immunofluorescence, enzyme‐linked immunosorbent assay (ELISA)) (Monteón 1995). We would also consider positive xenodiagnosis, positive blood culture or molecular biology assays as diagnostic tests if available, as well as pathological demonstration of tissue parasitism when biopsy or necropsy were done.
We excluded studies in which participants had other diseases not related to late‐stage, symptomatic Chagas disease (diabetes, nephropathies, tuberculosis, HIV, uric acid, other infectious diseases), other cardiovascular diseases not related to CCC, or heart transplant.
Participants who dropped out due to adverse effects, such as severe cutaneous reactions or peripheral polyneuropathy, or both, and the incidence of adverse events were reported.
We included studies in which there was only a subset of interest if most participants met our inclusion criteria. We then carried out a sensitivity analysis that excluded studies with mixed populations.
Types of interventions
Since only the predominant effects of the active drug were included, we analysed the following two comparisons:
benznidazole versus placebo or no treatment; and
nifurtimox versus placebo or no treatment.
Both drugs were orally administered at any dose specified for licensed trypanocidal drugs for at least 30 days.
Types of outcome measures
Reporting one or more of the outcomes listed above was not an inclusion criterion for the review. When a published report did not appear to report one of the above outcomes, we accessed the trial protocol and contacted the trial authors to ascertain whether the outcomes were measured but not reported. Relevant trials that measured these outcomes but did not report the data at all, or not in a useable format, were included in the review as part of the narrative.
Primary outcomes
-
Lowering or disappearance of parasitaemia or anti‐T cruzi antibodies, which was considered to have been achieved if one or more of the following were fulfilled.
Clearance or reduction of the mean antibody titres to T cruzi after treatment in comparison to the pretreatment results after a follow‐up period of at least 18 months. Mean anti‐T cruzi antibodies demonstrated by any of the following: haemagglutination, indirect immunofluorescence, and/or ELISA.
Clearance of parasitaemia demonstrated by a negative xenodiagnosis, blood culture, and/or molecular biology assays 12 months after treatment.
Pathological demonstration of tissue parasites (if a biopsy or necropsy was available).
Secondary outcomes
Clinical improvement, defined as a reduction or disappearance of clinical signs of heart failure, rhythm and conduction defects on electrocardiogram (ECG).
Quality of life, measured by a validated instrument, such as the 36‐item Short‐Form Health Survey (SF‐36) or the 12‐item Short‐Form Health Survey (SF‐12).
Development of adverse effects such as severe cutaneous reactions, digestive intolerance, and/or peripheral polyneuropathy.
Search methods for identification of studies
Electronic searches
We identified trials through a systematic search of the following bibliographic databases on 12 November 2019:
Cochrane Central Register of Controlled Trials (CENTRAL) (via CRS Web);
Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, MEDLINE Daily and MEDLINE (Ovid, 1946 to 11 November 2019);
Embase Classic and Embase (Ovid, 1947 to 11 November 2019);
LILACS (in English) (Latin American and Caribbean Health Science Information database) (BIREME, 1982 to 12 November 2019).
We searched the databases from their inception and imposed no restrictions on language of publication or publication status. We applied the sensitivity‐maximising randomised controlled trial (RCT) filter to MEDLINE and adaptations of it to Embase and LILACS (Lefebvre 2011). We did not perform a separate search for adverse effects of the interventions.
The electronic searches were first run in 2004 (Appendix 2), and then updated in March 2008 (Appendix 3), March 2010 (Appendix 4), and November 2019 (Appendix 5).
Searching other resources
We also searched the following clinical trials registers on 3 December 2019:
US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (www.clinicaltrials.gov);
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) (apps.who.int/trialsearch/).
Data collection and analysis
Selection of studies
Two review authors (MV and MMG) independently screened the titles and abstracts of citations retrieved from the searches and identified potentially relevant papers. Lists of these papers were compared, with any disagreements resolved through discussion or by consulting a third review author (AGG) if required. We retrieved the full texts of all papers considered potentially relevant and examined them for inclusion or exclusion. The selection process is recorded in detail in Figure 1, and Characteristics of included studies and Characteristics of excluded studies were tabulated.
1.
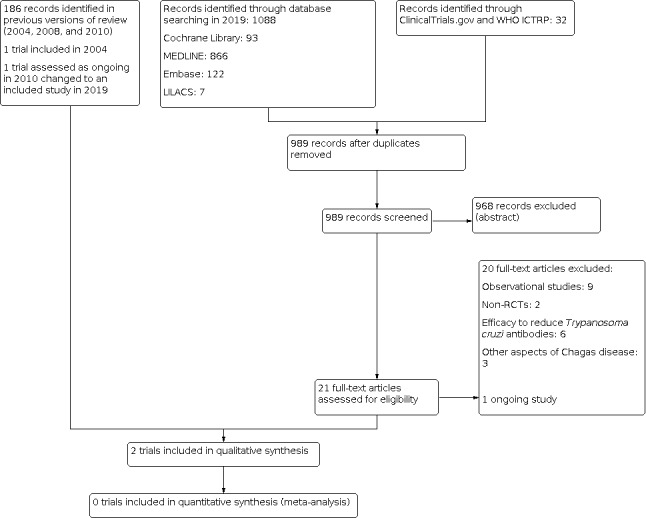
Study flow diagram.
All articles were reviewed using a checklist in which the following information was identified.
Method of random allocation of participants to the treatment group, and adequacy of concealment.
Blinding of participants and physicians to the treatment group.
Dose of trypanocidal drug, route of administration, and duration of treatment.
Duration of follow‐up after treatment.
Number and type of laboratory tests used in the diagnosis of participants and in the follow‐up.
Cure criteria, clearance or disappearance of parasites, and reduction of parasite burden.
Clinical data on cardiomyopathy.
Sample size.
Country in which the trial took place.
Proportion of loss during follow‐up either as a result of withdrawals or dropouts in the treatment/placebo groups.
Authors’ names, article title, publication type, source, language, year, and journal.
The articles that did not fulfil all the relevant inclusion criteria were removed. In order to ensure thoroughness of the study selection process, we systematised the process as follows. We first applied terms related to the systematic review objectives and subsequently reviewed the titles and abstracts of the articles identified by the search. Articles were selected if the following MeSH terms were identified:
those related to cardiomyopathy, dilated myocardiopathy, Chagas cardiomyopathy, and chronic Chagas disease;
those related to nitroimidazole, nitrofurans, imidazole, nifurtimox, benznidazole, trypanocidal, itraconazole, and allopurinol;
those related to study design, allocation, randomisation, and blinding process.
The second step was to identify amongst those articles previously selected studies for inclusion and exclusion. For this purpose, a new set of MeSH terms were defined and classified as follows.
Excluded:
Other neglected tropical diseases such as African trypanosomiasis, leishmanial, malaria, Trypanosoma evansi, or Trypanosoma brucei.
Other stages of Chagas disease (acute or acute indeterminate or gestational).
Other diseases not related to Chagas disease (diabetes, nephropathies, tuberculosis, HIV, uric acid, other infectious diseases, cardiomyopathies not Chagas‐related, heart transplant).
Other medicaments not imidazole or nitrofuran.
Studies of a different nature (bibliometric, healthcare systems, experimental, reviews, case series, and brief reports).
Included:
Randomised clinical trials
Note: The reviewed articles and experimental pharmacological studies (in vivo or in vitro) were used to update the Background.
Those studies pre‐identified as included were further examined in full text. A checklist was developed for this purpose which was based on GRADE Working Group criteria (Schunemann 2013).
Data extraction and management
Two review authors (MV and MMG) extracted the following study characteristics from the included studies.
ID article information: author’s name, title of study, journal of publication, language and country in which the study took place.
Methods: study design, total duration, number of study centres and locations, study settings, and date of study.
Participants: adults meeting the eligibility criteria, N randomised and proportion of loss to follow‐up either as a result of withdrawals or dropouts in the treatment and placebo groups.
Interventions: dose of trypanocidal drug, route of administration, and durations of treatment and follow‐up after treatment. Number and type of laboratory tests used in the diagnosis of participants and in the follow‐up.
Outcomes: cure criteria, clearance or disappearance of parasites and reduction of parasite burden, and clinical data on cardiomyopathy.
Notes: funding for trial, and notable conflicts of interest of trial authors.
Data were entered into the Review Manager 5 software package file and double‐checked against the data presented in the systematic review (Review Manager 2014).
Regarding the non‐RCTs, a further intentional search was undertaken in order to identify other studies that could have been published on the same topic, using the criteria described in the Cochrane Handbook for Systematic Reviews of Interventions. We determined the purpose of the study, author name, location and settings, details of interventions (dose, type of medicaments, follow‐up period, etc.), the number of participants, baseline data, and date of publication (Higgins 2011b).
Assessment of risk of bias in included studies
Two review authors (MV and MMG) independently assessed the risk of bias for each study using the criteria described in the Cochrane Handbook for Systematic Review of Interventions (Higgins 2011b). Any disagreements were resolved by discussion or by involving a third review author (AGG) if necessary. We also contacted study authors for additional information or in order to obtain missing data.
We assessed risk of bias according to the following domains.
Random sequence generation.
Allocation concealment.
Blinding of participants and personnel.
Blinding of outcome assessment.
Incomplete outcome data.
Selective outcome reporting.
Other bias.
Measures of treatment effect
We planned to express dichotomous data as risk ratios (RR) with 95% confidence intervals (CIs), or Peto odds ratios (OR) with 95% CIs if the sample size was very small (50 or fewer participants). Where continuous outcomes were reported using the same scale, we planned to calculate the mean difference (MD) with a 95% CI. For outcomes where data were reported using different scales, such as a different quality of life tools, we would use the standardised mean difference (SMD) with 95% CI. However, neither of the included studies reported continuous outcomes such as quality of life.
Unit of analysis issues
We considered whether any unit of analysis issues could affect our data due to level of randomisation, multiple time points, or having more than a single pair‐wise comparison.
For trials that had more than one intervention arm, we planned to combine groups to create a single pair‐wise comparison. Alternatively, we planned to reduce the sample size to control for multiple contributions from the same participants (splitting the 'shared' group into two or more groups). In the event, we included one study with three arms (benznidazole versus nifurtimox versus placebo). Since the two active treatments belonged to different comparisons, the placebo arm could be included for both groups without there being a unit of analysis problem.
Dealing with missing data
We analysed sources of missing data (randomly assigned participants, intention‐to‐treat and as‐treated and per‐protocol populations); additionally, we evaluated attrition rates (e.g. dropouts, losses to follow‐up, withdrawals) and used imputation methods such as the last observation carried forward.
If a trial did not report the mean and standard deviation (SD) for the outcomes, and the authors of the trial did not share this information, we would impute values by estimating the mean and SD from the median, the range, and the sample size of the trial (Hozo 2005). In order to calculate a missing SD from other reported data in the trials, we would use the 95% CI in the Review Manager 5. However, this was not required.
We planned to carry out sensitivity analysis to evaluate the imputation impact on meta‐analysis, but this was not required.
Assessment of heterogeneity
In the case of substantial clinical or methodological heterogeneity, we would not combine results as a pooled effect. We planned to visually inspect forest plots and standard Chi2 test with a level of significance of P = 0.1 to help identify heterogeneity or inconsistencies (Higgins 2011b). Since this test has low power, we also planned to use the I2 statistic to quantify inconsistencies across trials, thereby assessing the impact of heterogeneity on the meta‐analysis (Higgins 2002; Higgins 2003). We planned to use subgroup characteristics as well as an individual trial assessment to determine the possible reasons for heterogeneity. In the event, we were unable to pool more than one study for any outcome.
We planned to use a fixed‐effect model, unless substantial heterogeneity existed based on I2 > 50%.
Assessment of reporting biases
Had we been able to include several studies investigating the same outcome (10 or more), we would have used funnel plots to assess small‐study effects. Any identified asymmetry could have been due to the heterogeneity of effects with respect to 1) trial size or 2) poor methodological design (and therefore bias of small trials) and 3) publication bias (Sterne 2000). We would have interpreted results carefully (Sterne 2001).
Data synthesis
Had we identified sufficient studies for inclusion in the review with outcomes suitable for meta‐analysis, we would have analysed them as follows.
We planned to express dichotomous data as RR with 95% CIs, or Peto OR with 95% CIs if the sample size was very small (50 or fewer participants). The percentage of events in the placebo group was less than 10%, and the number of included studies was small. We also planned to express continuous data as the mean and SD difference from baseline to follow‐up for each comparison group. Where SD differences were not reported in source papers, we would make allowance for within‐participant correlation from baseline to follow‐up measurements by using the correlation coefficient between the two (Cochrane Heart Group; Follman 1992). We would have calculated the difference in means and 95% CI for each study.
Subgroup analysis and investigation of heterogeneity
We planned to conduct the following subgroup analyses to investigate heterogeneity.
Dose of treatment.
Treatment compliance.
Sensitivity analysis
We planned to carry out the following sensitivity analyses to explore effect sizes by restricting the analysis to the following factors.
Effect of risk of bias in included studies.
Time of interventions administration.
We also planned to test the robustness of the results by repeating the analysis using different statistical models (fixed‐effect and random‐effects models) to identify any inconsistencies in the results.
As we only included two studies, it was not possible to carry out any sensitivity or subgroup analyses.
Summary of findings and assessment of the certainty of the evidence
We used the GRADE approach to assess the quality of the evidence. The following criteria were used: limitations in the design and implementation, indirectness of evidence, unexplained heterogeneity or inconsistency of results, imprecision of results, and high probability of publication bias. We used the methods and recommendations described in Section 8.5 and Chapter 12 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011a), employing GRADEpro GDT software package (GRADEpro GDT). A separate summary table of the outcomes is presented for each comparison. Two review authors (MV and MMG) independently assessed the level of the evidence. Any disagreements were resolved by discussion or by consulting a third review author (AGG).
Results
Description of studies
For a detailed description of the trials, see Characteristics of included studies, Characteristics of excluded studies, and Characteristics of ongoing studies.
Results of the search
In the 2010 review update, we identified the ongoing study (BENEFIT) (Morillo 2015), a multicentre, randomised, double‐blind, placebo‐controlled clinical trial in which 1431 participants with CCC were randomly assigned to benznidazole 300 mg/day per 40 to 80 days and 1423 participants to placebo; for this update, the trial had finished, and we included its findings in the review.
In the current update, 989 new records were identified after de‐duplication. Of 21 articles retrieved in full text, we excluded 20 (9 observational studies; 2 non‐RCTs; 6 studies probing the efficacy of reducing T cruzi antibodies; and 3 studies concerned with other aspects of Chagas disease), and identified 1 new RCT as ongoing NCT03191162. For further details, see Figure 1.
Included studies
For details of the included studies, see Characteristics of included studies.
Source of data
We obtained data from the published literature and attempted to contact study authors for additional information or in order to obtain missing data (Appendix 6).
Comparisons
In the two included studies, we identified only two types of intervention: benznidazole or nifurtimox versus placebo (Coura 1997; Morillo 2015). Morillo 2015 compared benznidazole with placebo, and Coura 1997 compared benznidazole and nifurtimox with placebo.
Overview of trial populations
The included studies involved a total of 2940 participants. Morillo 2015 randomly assigned each participant to one of two study groups: 1431 in the benznidazole group (300 mg per day, and a variable duration of therapy based on participant weight) and 1423 in the placebo group for 60 days, with follow‐up at one, two, and five years (see Characteristics of included studies).
Coura 1997 randomly assigned 77 participants to one of three study groups: 26 in the benznidazole group (5 mg/kg/day every 12 hours); 27 in the nifurtimox group (5 mg/kg/day every 12 hours); and 24 in the placebo group, for 30 days of treatment for all groups. Outcomes were assessed at 1, 2, 3, and 12 months after the end of treatment. For details, see Characteristics of included studies.
Description of interventions related frequency and total dose/day are detailed in Appendix 7; As well as, duration of intervention, and other baseline characteristics are showed in Appendix 8 and Appendix 9.
Trial design
The two included studies were parallel comparisons with individual randomisation (Coura 1997; Morillo 2015).
Settings
In both trials, the interventions were carried out in outpatient settings (Coura 1997; Morillo 2015).
Participants
Both trials included participants from low‐ to middle‐income countries (Brazil, Argentina, Bolivia, Colombia, and El Salvador) (see Characteristics of included studies).
Diagnosis
In the case of Morillo’s trial (BENEFIT) (Morillo 2015), at least two positive serological tests (indirect immunofluorescence, indirect haemagglutination, or ELISA) for chronic Chagas disease in any combination were recorded. In Coura 1997, all participants had immunofluorescence and complement fixation reactions positive for T cruzi antibodies, and at least two xenodiagnoses were positive of the three performed before treatment.
Outcome
In Morillo 2015, the primary outcome was the composite of death; resuscitated cardiac arrest; sustained ventricular tachycardia; insertion of a pacemaker or cardiac defibrillator; cardiac transplantation; and development of new heart failure, stroke, or systemic or pulmonary thromboembolic events. The secondary outcome was the response to treatment on the basis of the results on PCR assay to detect circulating T cruzi kinetoplast DNA (kDNA) by means of an internationally validated method (Schijman 2011).
In Coura 1997, the primary outcome was the clearance of the blood parasite by xenodiagnosis and adverse effects (digestive intolerance and neuropsychiatric manifestations, cutaneous rash). See Appendix 10 for more detail of trials endpoint and Appendix 11 for the definitions of endpoint measurement.
Excluded studies
We reviewed 21 full‐text articles and excluded 20 for the following reasons: observational studies (9 studies), non‐RCTs (2 studies), efficacy to reduce T cruzi antibodies (6 studies), and other aspects of Chagas disease (3 studies) (see Characteristics of excluded studies).
Risk of bias in included studies
In Coura 1997, the participants were recruited from two places in Minais Gerais, Brazil. All of them were chronic chagasic patients with an independently established diagnosis which included seropositivity, xenodiagnosis, and clinical assessments including ECG and esophagram. The participants were randomly allocated to three groups: placebo, benznidazole, and nifurtimox. The method of randomisation was not reported. Participants and physicians were blind to the treatment group. Sixty‐four participants completed the study and were evaluated at 1, 2, 3, and 12 months after treatment, with 3 xenodiagnostic tests at 15 days’ interval. Additionally, participants underwent two serological assays. Of the 77 participants included in the study, 83% completed the treatment. Confounding variables such as sex and age were evenly distributed between groups. The sample size was clearly too small to provide adequate power to detect clinically important differences in the outcomes (Coura 1997).
The BENEFIT study is a multicentric (Argentina, Bolivia, Brazil, Colombia, and El Salvador), randomised, double‐blind controlled trial involving participants with Chagas cardiomyopathy. Individuals were randomly assigned to either placebo or benznidazole with a randomisation ratio of 1:1 using a random‐block system. The blinding method was not reported. Participants were evaluated at 11 and 21 days and 2 months after initiation of treatment, and were followed up at the 6‐month mark, and then annually until a minimum of 4 years and a maximum of 7 years. All participants completed the full course of treatment and were available at the 1‐year follow‐up; at 2 years, 99% were available, and at 7 years, 99.5% were available (Morillo 2015).
For details, see Characteristics of included studies. For judgement of the risk of bias for each study and for studies overall, see Figure 2 and Figure 3.
2.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Only Morillo 2015 detailed the method used for random sequence generation and allocation concealment, therefore we considered Coura 1997 to be at unclear risk of selection bias.
Blinding
We assessed Coura 1997 as at unclear risk of bias as minimal information is provided regarding the blinding of physicians in charge of examining the participants. Morillo 2015 mentions that allocation, evaluation, and statistical analysis will be blinded; however, as there is no comprehensive explanation of the process, we judged the trial to be at unclear risk of bias.
Incomplete outcome data
Coura 1997 reported clearance of parasitaemia, and 96% of participants in the placebo group remained positive after treatment. Participants in the nifurtimox group exhibited a higher rate of withdrawals (29.6%), followed by the benznidazole group (11.5%). Most participants in the placebo group continued until the end of the study (attrition rate 8.3%). However, information regarding exclusion or attrition is limited, therefore we assessed this trial as at unclear risk of attrition bias.
Morillo 2015 reported a reduction of anti‐T cruzi antibodies in 66% of participants; of these, the PCR conversion rate was 66.2% in the benznidazole group and 33.5% in the placebo group. At the end of treatment, a higher rate of withdrawal was observed in the benznidazole group than in the placebo group (18% versus 12%); at two years’ follow‐up, this was higher in the placebo group than in the benznidazole group (7% versus 5%). We judged this trial as at unclear risk of attrition bias due to insufficient information on the reduction in PCR tests.
Selective reporting
Both included trials reported outcomes in insufficient detail, therefore they are assessed as at unclear risk of reporting bias.
Other potential sources of bias
We detected no other sources of bias in the two included trials.
Effects of interventions
Summary of findings 1. Benznidazole compared to placebo for treating late‐stage, symptomatic Chagas disease and CCC.
| Benznidazole compared to placebo for treating late‐stage, symptomatic Chagas disease and CCC | |||||
| Patient or population: adults with both late‐stage, symptomatic Chagas disease and CCC Setting: outpatients Intervention: benznidazole Comparison: placebo | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No. of participants (studies) | Quality of the evidence (GRADE) | |
| Assumed risk | Corresponding risk | ||||
| Placebo | Benznidazole | ||||
|
Clearance or reduction of antibody titres (N) (a) at 12 months (b) at 2 years (c) at 5 years Follow‐up: 5 years |
(a) 207 per 1000 (b) 351 per 1000 (c) 439 per 1000 |
(a) 395 per 1000 (339 to 457) (b) 506 per 1000 (453 to 562) (c) 548 per 1000 (500 to 601) |
(a) RR 1.91 (1.64 to 2.21) (b) RR 1.44 (1.29 to 1.60) (c) RR 1.25 (1.14 to 1.37) |
1896 (1 RCT) | ⊕⊕⊝⊝1 Low |
|
Clearance of parasitaemia demonstrated by negative xenodiagnosis, blood culture, and/or molecular assays (N) Follow‐up: 12 months |
42 per 1000 |
579 per 1000 (315 to 805) |
OR 31.66 (10.56 to 94.97) |
50 (1 RCT) | ⊕⊝⊝⊝2 Very low |
| Pathological demonstration of tissue parasites (N) | Not reported | ||||
|
Clinical improvement (N) (a) Heart failure (b) Sustained ventricular tachycardia Follow‐up: 5 years |
(a) 86 per 1000 (b) 29 per 1000 |
(a) 76 per 1000 (59 to 98) (b) 23 per 1000 (15 to 36) |
(a) RR 0.89 (0.69 to 1.14) (b) RR 0.80 (0.51 to 1.26) |
2854 (1 RCT) | ⊕⊕⊝⊝3 Low |
| Quality of life | Not reported | ||||
|
Adverse events (N) Follow‐up: 5 years |
95 per 1000 |
239 per 1000 (198 to 287) |
RR 2.52 (2.09 to 3.03) |
2854 (1 RCT) | ⊕⊕⊕⊝1 Moderate |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; OR: Peto odds ratio; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High quality: We are very confident that the true effect lies close to that of the estimate of the effect. Moderate quality: We are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low quality: Our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low quality: We have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | |||||
1Downgraded one level for methodological limitations due to incomplete outcome reporting and one level due to imprecision (small sample size). 2Downgraded two levels due to selection bias, performance bias, and incomplete outcome reporting, and one level due to imprecision (small sample size). 3Downgraded one level for methodological limitations due to incomplete outcome reporting and one level due to imprecision (CI consistent with benefit and harm).
Summary of findings 2. Nifurtimox compared to placebo for treating late‐stage, symptomatic Chagas disease and CCC.
| Nifurtimox compared to placebo for treating late‐stage, symptomatic Chagas disease and CCC | |||||
|
Patient or population: adults with both late‐stage, symptomatic Chagas disease and CCC Setting: outpatients Intervention: nifurtimox Comparison: placebo | |||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect (95% CI) | No. of participants (studies) | Quality of the evidence (GRADE) | |
| Assumed risk | Corresponding risk | ||||
| Placebo | Nifurtimox | ||||
|
Clearance or reduction of antibody titres (N) Follow‐up: 12 months |
42 per 1000 |
704 per 1000 (102 to 1000) |
RR 16.89 (2.44 to 116.85) |
51 (1 RCT) | ⊕⊝⊝⊝1 Very low |
| Clearance of parasitaemia demonstrated by negative xenodiagnosis, blood culture, and/or molecular assays (N) | Not reported | ||||
| Pathological demonstration of tissue parasites (N) | Not reported | ||||
| Clinical improvement (N) | Not reported | ||||
| Quality of life | Not reported | ||||
| Adverse events (N) | Not reported | ||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | |||||
| GRADE Working Group grades of evidence High quality: Further research is very unlikely to change our confidence in the estimate of effect. Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate. Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate. Very low quality: We are very uncertain about the estimate. | |||||
1Downgraded two levels due to selection bias, performance bias, and incomplete outcome reporting, and one level due to imprecision (small sample size).
Benznidazole versus placebo
See Table 1.
Primary outcomes
Lowering or disappearance of parasitaemia
Clearance or reduction of anti‐T cruzi antibodies
Only Morillo 2015 reported antibody clearance after five years of follow‐up:
for benznidazole at 12 months, the risk ratio (RR) was 1.91 (95% confidence interval (CI) 1.64 to 2.21; P < 0.001; 1 trial; 1896 participants; low quality of the evidence in favour of intervention; Analysis 1.1);
for benznidazole at 2 years, the RR was 1.44 (95% CI 1.29 to 1.60; P < 0.001; 1 trial; 1896 participants; low quality of the evidence in favour of intervention; Analysis 1.1);
for benznidazole at 5 years, the RR was 1.25 (95% CI 1.14 to 1.37; P < 0.001; 1 trial; 1896 participants; low quality of the evidence in favour of intervention; Analysis 1.1).
1.1. Analysis.

Comparison 1: Benznidazole versus placebo, Outcome 1: Clearance of antibody titres
Clearance of parasitaemia, demonstrated by negative xenodiagnosis, blood culture, and/or molecular biology assays
One trial reported applying xenodiagnosis for this comparison (Coura 1997). The Peto odds ratio (OR) was 31.66 (95% CI 10.56 to 94.97; P < 0.001; 1 trial; 50 participants; very low quality of the evidence in favour of benznidazole; Analysis 1.2).
1.2. Analysis.

Comparison 1: Benznidazole versus placebo, Outcome 2: Clearance of parasitaemia, demonstrated by negative xenodiagnosis, blood culture, and/or molecular biology assays
Lowering or disappearance of parasitaemia or anti‐T cruzi antibodies.
None of the included studies reported the disappearance of parasitaemia by pathological demonstration.
Secondary outcomes
Clinical improvement
Only Morillo 2015 reported cardiovascular outcomes:
for the risk of heart failure, the RR was 0.89 (95% CI 0.69 to 1.14; P = 0.35; 1 trial; 2854 participants; low quality of the evidence indicates that compared to placebo, the used of benznidazole may make little to no differences; Analysis 1.3);
for sustained ventricular tachycardia, the RR was 0.80 (95% CI 0.51 to 1.26; P = 0.33; 1 trial; 2854 participants; low quality of the evidence in favour of benznidazole; Analysis 1.3).
1.3. Analysis.

Comparison 1: Benznidazole versus placebo, Outcome 3: Clinical improvement
Quality of life
Neither trial evaluated this outcome.
Development of adverse effects
The most frequent adverse effects were cutaneous rashes, gastrointestinal symptoms, and peripheral polyneuropathy. In Morillo 2015, the RR to develop any adverse events was 2.52 (95% CI 2.09 to 3.03; P < 0.01; 1 trial; 2854 participants; moderate quality of the evidence in favour of placebo; Analysis 1.4).
1.4. Analysis.

Comparison 1: Benznidazole versus placebo, Outcome 4: Adverse effects
Subgroup analyses
Due to an insufficient number of studies we did not perform subgroup analyses.
Sensitivity analyses
Due to an insufficient number of studies we did not perform sensitivity analyses.
Nifurtimox versus placebo
See Table 2.
Primary outcomes
Lowering or disappearance of parasitaemia
Clearance or reduction of anti‐T cruzi antibodies
Only Coura 1997 reported antibody clearance with the administration of nifurtimox compared to placebo for 30 days, with an RR of 16.89 (95% CI 2.44 to 116.85; P = 0.004; 1 trial; 51 participants; very low quality of the evidence in favour of nifurtimox; Analysis 2.1).
2.1. Analysis.

Comparison 2: Nifurtimox versus placebo, Outcome 1: Clearance of antibody titres
Clearance of parasitaemia, demonstrated by negative xenodiagnosis, blood culture, and/or molecular biology assays
Coura 1997 did not evaluate this outcome.
Disappearance of parasites in the tissues
Coura 1997 did not evaluate the disappearance of parasitaemia by pathological demonstration.
Secondary outcomes
Clinical improvement
Coura 1997 did not evaluate this outcome.
Quality of life
Coura 1997 did not evaluate this outcome.
Development of adverse effects
The development of adverse effects such as gastrointestinal intolerance, paraesthesia, and other nervous system issues was more frequent in the intervention group. Details on adverse effects can be found in the Appendix 12; Appendix 13; Appendix 14.
Subgroup analyses
Due to an insufficient number of studies we did not perform subgroup analyses.
Sensitivity analyses
Due to an insufficient number of studies we did not perform sensitivity analyses.
Assessment of reporting bias
Due to the limited number of trials for any given outcome we did not draw funnel plots.
Discussion
Summary of main results
We included two studies in this update. Coura 1997 compared treatment with nifurtimox or benznidazole with placebo for the clearance of parasitaemia. Infection was assessed using indirect immunofluorescence in a screening assay followed by positive complement fixation as well as confirmatory parasitologic assay (at least two out of the three positive xenodiagnosis results). Twenty‐six participants were randomly assigned to benznidazole 5 mg/kg/day, 27 to nifurtimox 5 mg/kg/day, and 24 to placebo for 30 days. Morillo 2015 included 2854 participants with late‐stage, symptomatic Chagas disease and CCC: 1431 participants were randomly assigned to benznidazole 300 mg/day for 40 to 80 days, and 1423 to placebo. We also identified one ongoing study (NCT03191162).
Benznidazole compared to placebo
Low quality of the evidence from the BENEFIT study trial, Morillo 2015, suggests there may be a benefit of benznidazole when compared to placebo for clearance or reduction of antibody titres at 12 months (RR 1.91, 95% CI 1.64 to 2.21), 2 years (RR 1.44, 95% CI 1.29 to 1.60), and 5 years (RR 1.25, 95% CI 1.14 to 1.37). This shows that the effect of the intervention declines over time as a result of the natural reduction of the antibodies titres as an individual immune response rather than as the effect of the imidazolic treatment.
Due to very limited evidence, we are uncertain about the effects of benznidazole on the clearance of parasitaemia demonstrated by negative xenodiagnosis, blood culture, and/or molecular assays.
Low quality of the evidence suggests that benznidazole may make little to no difference for the risk of heart failure and ventricular tachycardia when compared to placebo.
Moderate quality of the evidence shows that adverse events increase with benznidazole when compared to placebo. Adverse effects were observed in 23.9% of patients in the Benznidazole group compared to 9.5% in the placebo group. The most frequent adverse effects were: cutaneous rash, gastrointestinal symptoms, and peripheral polyneuropathy.
No data were available for the outcomes pathological demonstration of tissue parasites and quality of life.
Nifurtimox compared to placebo
The only outcome with data available for this comparison was clearance or reduction of antibody titres in Coura 1997, with an RR of 16.89 (95% CI 2.44 to 116.85; P = 0.004; 1 trial; 51 participants). Even though the estimated RR is large in favour of nifurtimox efficacy, we are uncertain about the effect due to very limited evidence to establish the effectiveness of nifurtimox for the treatment of late‐stage, symptomatic Chagas disease and CCC.
Regarding adverse events, Coura 1997 mentioned in a general manner that nifurtimox caused more intense adverse events than benznidazole, without any quantification.
Overall completeness and applicability of evidence
This review included only two randomised clinical trials. The purpose of both studies was to clarify the therapeutic role of the nitrofuran and imidazole drugs in late‐stage, symptomatic Chagas disease and CCC; however, it was not possible to conduct a meta‐analysis due to the small number of trials identified. Furthermore, due to the low level of certainty, especially in relation to the effect of nifurtimox compared to placebo, since only 51 participants were included, only one study was reviewed, and there is a high risk of bias. Consequently, for this outcome, it remains uncertain whether the use of this type of drugs in the chronic phase has no effect once Chagas disease has provoked organ damage, and is pending further corroboration that may come from the currently ongoing study.
Quality of the evidence
It is important to note that there is a gap of almost 20 years between the two RCTs included in this review. This is significant because the burden and costs of the chronic diseases were finally accepted by health authorities in most Latin American countries. With this in mind, the quality of evidence of the first RCT may be questionable, since sources of financing were scarce, and most studies were conducted with limited resources. Consequently, selection bias may have been introduced; random allocation and concealment are not explained; attrition bias regarding adverse events was introduced; and outcome data are incomplete (Coura 1997). The second study is a multicentric RCT with a sample of 2854 participants with late‐stage, symptomatic Chagas disease and CCC, from five endemic countries, with a long follow‐up (up to seven years). Random allocation and concealment were detailed; adverse events were fully informed; and attrition bias may have occurred in relation to the PCR test since only 66% of participants were tested (Morillo 2015). We judged that this study has a low or unclear risk of bias.
Potential biases in the review process
We followed standard Cochrane methodology in order to avoid duplication and publication bias; there were no restrictions regarding the language or date of publication. The review was undertaken by three review authors working independently. We attempted to contact study authors in order to obtain missing data; however, one of the trials was published almost 20 years ago. Since Chagas disease is a neglected tropical disease, with little interest from the authorities, limited studies are conducted in relation to the efficacy of parasitological cure in the late‐stage of the disease (Tarleton 2014).
Agreements and disagreements with other studies or reviews
Human infection with T cruzi induces an immune response in most human hosts, but a chronic indolent infection is common. A proportion of the infected people develop late‐stage, symptomatic Chagas disease with two main clinical patterns: heart disease or dilated gastrointestinal 'mega' syndromes. Chronic chagasic cardiomyopathy may produce a progressive heart disease characterised by rhythm and conduction disorders with or without dilated cardiomyopathy. Both high morbidity and mortality of people with these manifestations of Chagas disease is common.
Interventions to treat Chagas disease were initiated shortly after its description early in the 20th century. However, they were unsuccessful for 50 years. In 1962, the first useful drug treatment was found using animal models – a nitrofuran derivative marketed as Nifurtimox – and was shortly followed by the imidazole drug benznidazole, both of which have become the cornerstone of treatment of American trypanosomiasis. Good results have been found in acute infections, with the absence of parasitaemia in most treated patients and remission of serologic response. These drugs are now used in recently infected children, in cases of laboratory needle stick injuries resulting in acute infection, as well as in iatrogenic infection through contaminated blood products (Rodriguez‐Coura 2002; Stoppani 1999; Urbina 1999).
In questioning the efficacy of nifurtimox and benznidazole in the treatment of late‐stage, symptomatic Chagas disease and CCC, we found only two controlled clinical trials. We did not find strong evidence that standard treatment with either nifurtimox or benznidazole in individuals with late‐stage T cruzi infection is beneficial.
A major problem in conducting trials and interpreting long‐term prognostic studies is the issue of criteria to define cure. For example, some studies have shown a significant reduction of blood antibodies and fewer ECG abnormalities in chronically infected patients treated with benznidazole compared to a placebo group, and interpreted this as an indication of a delay in tissue damage (Moretti 1998; Segura 1994; Viotti 1994). A clinical criteria for cure requires the disappearance of parasitaemia, but this is not easy to demonstrate in chronic cases, with the sensitivity of haemoculture or xenodiagnosis being no more than 50% at best. Inclusion of this criterion is clearly insufficient to determine the efficacy of drug treatment, and, perhaps not surprisingly, most papers have failed to demonstrate any reduction in parasitaemia (Contijo 1999; Urbina 1999).
The diagnosis of Chagas disease has limitations, mainly due to the great complexity of the factors involved in it, as well as the low sensitivity of parasitological techniques and the low specificity of immunological tests, such as those used in the two studies included in this review. Circulating antibodies against T cruzi is a diagnostic hallmark of chronic infection. The disappearance of antibodies has been used as a clinical cure criterion in acute disease, but does not occur in the chronic Chagas disease. The current established criteria to define cure in CCC may be insufficient and outdated. New developments in molecular biology have further challenged the existing clinical cure criteria. Parasite DNA can now be clearly demonstrated in several tissues, thereby reinforcing the notion of parasite persistence (despite apparent clinical cure) and its possible role as an inducer of an immune response. Changes in Trypanosoma cruzi‐specific immune response after treatment with benznidazole have been proven by Laucella and colleagues (Laucella 2009), who found a significant decline of peripheral interferon (IFN) gamma‐producing T cells specific for T cruzi as early as 12 months after benznidazole treatment and which became undetectable in a substantial proportion of treated participants. Finally, further RCTs are needed that consider the application of immunomics, which combines serology with proteomics. This would help to discover genes and molecules related to the susceptibility and immunity of T cruzi infection, allowing the creation of an adequate diagnosis of the disease, elucidating new targets and therapies, and, why not, making possible the creation of a vaccine against Chagas disease (Lopez‐Monteon 2019).
Both nifurtimox and benznidazole are linked to adverse reactions (peripheral neuropathy, skin disorders, and other health problems) in a relatively high proportion of users. This indicates the need for safer and effective drugs for late‐stage, symptomatic Chagas disease and CCC, where treatment is likely to be long term. On balance, the limited findings of this review cast doubt on the efficacy of treatment for CCC with either nifurtimox or benznidazole, but do provide modest evidence of potential harm from adverse effects.
We also identified non‐RCTs as follows.
A study was conducted in Brazil with 17 patients who had completed full treatment with antitrypanosomal drugs (nitrofurans or nitroimidazole 10 mg/kg/day) and were age‐ and sex‐matched with patients from a cohort study of chronic chagasic cardiomyopathy patients, all of whom showed immunological and/or serological assay evidence of T cruzi. The aim was to determine whether serological or immunological cures correlate with molecular methods of diagnosis. Although the study was not randomised, and patients and physicians were not blinded to treatment allocation, the investigators made the useful observation that despite apparent cure through immunological/serological outcome measures, the molecular markers persisted. This indicated that parasites were not eliminated after treatment with nitrofuran derivatives (Braga 2000). A further paper using the same cohort of patients reported the findings of a 10‐year follow‐up of 91 people with chronic chagasic cardiomyopathy and 41 uninfected controls. Using molecular methods of competitive PCR, they found that both treated and untreated patients showed evidence of active infection (Lauria‐Pires 2000).
Dr Viotti and colleagues have been screening patients with Chagas disease in the Chagas disease section at Eva Peron Hospital in Buenos Aires, Argentina since 1984, and have recruited almost 2000 patients during this period. The treatment of Chagas disease has been a major concern, therefore in 1994 a published study aiming to compare the clinical and serological progress of cardiomyopathy in patients treated with benznidazole and untreated individuals was undertaken. Seventy untreated patients and 131 benznidazole‐treated patients (5 mg/kg/day for 30 days) were recruited. The follow‐up period averaged eight years. The proportion of patients who were serologically negative at the end of follow‐up was higher in the treated group than in the untreated group (19.1% versus 6%; P = 0.05). The investigators concluded that treatment with benznidazole decreases antibody titres if treatment is used early in the course of chronic chagasic cardiomyopathy (Viotti 1994).
Due to the chronic implications of Chagas disease, the effectiveness of treatment has been widely studied. In a 2006 study of 566 undetermined patients 30 to 50 years old with three positive serologies and without heart failure, 283 were treated with benznidazole (5 mg/kg/day for 30 days) and the same number remained untreated. The authors concluded that treatment of Chagas disease in the early stage of the illness prevents progression towards cardiac manifestations and increases negative seroconversion (Viotti 2006).
In a further study of patients who became seronegative, either spontaneously or by parasitological treatment, clinical and epidemiological progression features of Chagas disease were analysed. The authors concluded that the parasitological cure is a favourable clinical course (Bertocchi 2013). Although the effectiveness of benznidazole has been widely proven, its adverse reactions have been a major cause of concern. Consequently, developing new drugs and new schemes of treatment is integral.
In 2016, Viotti's research team conducted a pilot study to assess the efficiency of a new treatment scheme with benznidazole for individuals with chronic Chagas disease. Twenty patients were included; however, 17 were tested by PCR. All patients received 5 mg/kg/day in 2 daily doses every 5 days for 60 days. Parasitological cure was achieved in 65% of patients, whilst 50% had adverse reactions, and only one patient suspended treatment. The authors concluded that the low rate of treatment suspension was a major accomplishment of this study. However, further studies are required to confirm these results in longitudinal assessments (Alvarez 2016).
A further study from Argentina followed 198 people drawn from a larger cohort of 492 patients that had suffered attrition due to losses to follow‐up, with the aim of examining the prognosis of chronic Chagasic cardiomyopathy in treated and untreated groups undergoing prolonged follow‐ups. There were three groups: a) 32 patients treated with nifurtimox (5 to 8 mg/kg/day for 60 days); b) 36 patients treated with benznidazole (5 mg/kg/day for 30 days); and c) 130 untreated patients. All patients underwent the following tests at the beginning or during the follow‐up of the cohort study: a) three serological Chagas‐specific tests (Direct agglutination with 2‐mercaptoethanol (DA‐2ME), Indirect Haemoagglutination (IHA), or Indirect Immunofluorescence (IIF)) with titers equal to or higher than 1/32 for at least two out of the three reactions; and b) electrocardiogram and chest X‐ray.
The patients were followed for an average of 14 years (8 to 32 years). At the outset, only 9.6% of the infected patients had cardiomyopathy; however, when comparing treated and untreated patients, the proportion of cardiomyopathy was higher in the former group (7% versus 3.2%) and reflects the use of drug treatment in those with symptomatic disease. The clinical prognosis was worse in the untreated group, with less likelihood of reductions in antibody titres (≤ 1/64 in 86% versus 38% of treated and untreated patients, respectively). These long‐term findings suggest that drug treatment is superior to no treatment (Fabbro 2000).
A further long‐term follow‐up (13 to 21 years) was reported in which the prognosis of acute and chronic Chagas disease was compared, with all patients receiving benznidazole. The cure criteria were established by means of quantitative serological tests that included: to complement fixation, indirect immunofluorescence, indirect haemagglutination, and ELISA. However, given the duration of follow‐up, not all of these tests were used in all of the patients. The cure rate was higher in patients with acute disease (76%) than in those with chronic disease (8% to 9%). It was concluded that the cure criteria should include parasitological as well as immunological criteria, and that new immunological tests have opened a door to a different way of studying the natural history of Chagas disease. It is challenging to make any inferences about the effects of treatment in this study since there was no randomised control group, neither patients nor physicians were blind to the treatment group, and the criteria for cure changed over the follow‐up (Cancado 2002; Schijman 2011).
Authors' conclusions
Implications for practice.
Due to the low quality of the evidence, the efficacy of imidazolic drugs for the treatment of late‐stage, symptomatic Chagas disease remains uncertain. Further studies are needed.
Implications for research.
Further RCTs are needed to confirm the efficacy of benznidazole treatment in late‐stage, symptomatic Chagas disease and CCC. The effects of new drugs such as those derived from molecules designed to modify parasite metabolism, but without adverse effects on human physiology – a sort of 'chemical bullet' that would aim to eliminate both blood and tissue parasitism – need to be tested.
There also needs to be consideration of the development of better and more precise cure criteria that can be used as outcome measures in such trials, as well as more sensitive and specific diagnostic methods such as those driven from molecular biology in combination with genomic and proteomic techniques.
The pathogenesis of chronic complications of T cruzi infection also requires research because the host’s contribution through the immune response could be a cornerstone for the development of late‐stage clinical disease, thereby reducing the relevance of antiparasite treatment as the main therapeutic strategy in advanced disease.
Preventive measures such as those applied in Latin America through the Southern Cone, Andean and Central America Initiatives offer the most effective cost‐ and benefits‐based approaches to sustainable control of Chagas disease by reducing acute infection, preventing reinfection and progression to chronic stages with target organ damage.
What's new
| Date | Event | Description |
|---|---|---|
| 12 November 2019 | New search has been performed | We updated the searches in November 2019. |
| 12 November 2019 | New citation required but conclusions have not changed | We included two studies in the review and identified one ongoing study. There is insufficient evidence to support the efficacy of nitrofurans or imidazolic drugs for the treatment of chronic chagasic cardiomyopathy. |
History
Protocol first published: Issue 1, 2003 Review first published: Issue 4, 2005
| Date | Event | Description |
|---|---|---|
| 13 September 2010 | New search has been performed | The searches were updated in March 2010 and the new references were checked. No new studies were identified. One ongoing study was identified and added to this update. The conclusions remain unchanged. |
| 7 May 2008 | Amended | Converted to new review format. |
| 6 May 2008 | New search has been performed | Search updated, no new trials added, conclusions remain unchanged. |
Acknowledgements
Dr G Faba, Centro Mexicano of Colaboracion Cochrane, for assistance with database searching and paper retrieval.
Appendices
Appendix 1. Glossary
Acute phase
This is the phase immediately following infection. Acute symptoms only occur in approximately 1% of cases. Most people infected do not seek medical attention. The most recognised symptom of acute Chagas infection is the Romaña sign, or swelling of the eye on one side of the face, usually at the site of the bite, or by direct infection of the conjunctiva. Other symptoms are usually not specific to Chagas infection. These may include fatigue, fever, an enlarged liver or spleen, and swollen lymph glands. A rash, loss of appetite, diarrhoea, and vomiting may occur. Infants and very young children with acute Chagas disease can develop meningoencephalitis, which can cause death. In general, symptoms last for four to eight weeks in the acute phase and then disappear even if no treatment is provided.
Indeterminate phase
During this stage, people do not exhibit any symptoms. These chronically infected individuals become seropositive and remain asymptomatic for the next 10 to 30 years or for the rest of their lives when, if the electrocardiogram is normal and no evidence of cardiomegaly or digestive mega viscera is observed in X‐rays, they are defined as being in the indeterminate phase.
Chronic chagasic cardiomyopathy (CCC)
A specific cardiomyopathy characterised by heart failure with bizarre rhythm or conduction defects, or both.
Chronic phase
The most serious symptoms of Chagas disease may develop 10 to 30 years after infection. Cardiac problems, including an enlarged heart, altered heart conduction or rhythm, heart failure, or cardiac arrest are symptoms of chronic disease. Chagas disease can also lead to an enlargement of parts of the digestive tract, which results in severe constipation or problems with swallowing. In people who are immune compromised, including those with HIV/AIDS, Chagas disease can be severe. Not everyone will develop chronic symptoms of Chagas disease.
Parasite
An animal or plant that lives in or upon another organism (technically called its host) and draws its nutriment directly from it.
PCR
Polymerase chain reaction (PCR) is a molecular biological technique for amplifying (creating multiple copies of) DNA without using a living organism, such as Escherichiacoli or yeast. PCR is commonly used in medical and biological research labs for a variety of tasks, such as the detection of hereditary diseases, identification of genetic fingerprints, diagnosis of infectious diseases, cloning of genes, and paternity testing.
Parasitaemia
The presence of parasites in the blood.
Vector
An agent, often an insect, that transfers a disease caused by a parasite or micro‐organism from one host to another. In Chagas disease, the vectors are species of triatoma bugs. They have many common names, such as 'vinchuca', 'barbeiro', 'chipo', 'the kissing bug', or 'assassin bug'. Triatominae are insects of the order Hemiptera, family Reduviidae, and subfamily Triatominae.
Appendix 2. Search strategies 2004
| CENTRAL |
| #1 CHAGAS‐DISEASE*:ME #2 CHAGA* #3 TRYPANOSOMIA* #4 ((#1 or #2) or #3) #5 IMIDAZOLES*:ME #6 TRYPANOCIDAL‐AGENTS*:ME #7 TRYPANOCIDAL* #8 IMIDAZOLE* #9 NITROFURA* #10 BENZNIDAZOL* #11 CHEMOTHERAPY #12 TREATMENT* #13 (((((((#5 or #6) or #7) or #8) or #9) or #10) or #11) or #12) #14 (#4 and #13) |
| Other databases Both English and Spanish search terms were used for searching: |
| English: Chagas disease (title or abstract word) or trypanosomiasis/trypanosomosis (title or abstract word) AND treatment (title or abstract word) or chemotherapy (title or abstract word) OR Imidazolic drugs (title or abstract word) or trypanocidal drugs (title or abstract word). |
| Spanish: Enfermedad de Chagas (title or abstract word) o trypanosomiasis (title or abstract word) AND tratamiento (title or abstract word) o antiparasitarios (title or abstract word) o imidazólicos (title or abstract word). |
Appendix 3. Search strategies 2008
| CENTRAL |
| #1 MeSH descriptor Chagas Disease explode all trees #2 chaga* in All Text #3 MeSH descriptor Trypanosomiasis this term only #4 trypanosomiasis in All Text #5 cardiomyopath* in All Text #6 (#1 or #2 or #3 or #4 or #5) #7 nifurtimox in All Text #8 allopurinol in All Text #9 benznidazole in All Text #10 itraconazole in All Text #11 trypanocid* in All Text #12 antitrypanosom* in All Text #13 anti‐trypanosom* in All Text #14 MeSH descriptor trypanocidal agents explode all trees #15 MeSH descriptor nitrofurans explode all trees #16 nitrofurans in All Text #17 bzd in All Text #18 MeSH descriptor allopurinol this term only #19 MeSH descriptor itraconazole this term only #20 bay next 2502 in All Text #21 (#7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20) #22 (#6 and #21) |
| MEDLINE |
| 1 exp chagas disease/ 2 trypanosomiasis/ 3 chaga$.tw. 4 trypanosomiasis.tw. 5 cardiomyopath$.tw. 6 or/1‐5 7 nifurtimox.tw. 8 allopurinol.tw. 9 benznidazole.tw. 10 itraconazole.tw. 11 trypanocid$.tw. 12 antitrypanosom$.tw. 13 anti‐trypanosom$.tw. 14 exp Trypanocidal Agents/ 15 exp Nitrofurans/ 16 nitrofurans.tw. 17 bzd.tw. 18 Allopurinol/ 19 Itraconazole/ 20 trypanocidal.tw. 21 or/7‐20 22 6 and 21 23 randomized controlled trial.pt. 24 controlled clinical trial.pt. 25 Randomized controlled trials/ 26 random allocation/ 27 double blind method/ 28 single‐blind method/ 29 or/23‐28 30 exp animal/ not humans/ 31 29 not 30 32 clinical trial.pt. 33 exp Clinical Trials as Topic/ 34 (clin$ adj25 trial$).ti,ab. 35 ((singl$ or doubl$ or trebl$ or tripl$) adj (blind$ or mask$)).ti,ab. 36 placebos/ 37 placebo$.ti,ab. 38 random$.ti,ab. 39 research design/ 40 or/32‐39 41 40 not 30 42 31 or 41 43 22 and 42 44 limit 43 to yr="2004 ‐ 2008" |
| EMBASE |
| 1 chagas disease/ 2 trypanosomiasis/ 3 chaga$.tw. 4 trypanosomiasis.tw. 5 cardiomyopath$.tw. 6 or/1‐5 7 exp Antitrypanosomal Agent/ 8 exp Nitrofuran Derivative/ 9 nifurtimox.tw. 10 allopurinol.tw. 11 benznidazole.tw. 12 itraconazole.tw. 13 trypanocid$.tw. 14 antitrypanosom$.tw. 15 anti‐trypanosom$.tw. 16 nitrofurans.tw. 17 bzd.tw. 18 Allopurinol/ 19 Itraconazole/ 20 trypanocidal.tw. 21 or/7‐20 22 6 and 21 23 controlled clinical trial/ 24 random$.tw. 25 randomized controlled trial/ 26 follow‐up.tw. 27 double blind procedure/ 28 placebo$.tw. 29 placebo/ 30 factorial$.ti,ab. 31 (crossover$ or cross‐over$).ti,ab. 32 (double$ adj blind$).ti,ab. 33 (singl$ adj blind$).ti,ab. 34 assign$.ti,ab. 35 allocat$.ti,ab. 36 volunteer$.ti,ab. 37 Crossover Procedure/ 38 Single Blind Procedure/ 39 or/23‐38 40 22 and 39 41 limit 40 to yr="2004 ‐ 2008" |
| LILACS |
| (random$ or trial$ or clinical stud$ or rct) and (chaga$ or trypanosomiasis or cardiomyopath$) [Words] and antitrypanocidal$ or anti‐trypanocidal$ or nitrofurans or nifurtimox or allopurinol or benznidazole or itraconazole or anti‐trypanosom$ or antitrypanosom$ [Words] and 2004 or 2005 or 2006 or 2007 or 2008 [Country, year publication] |
Appendix 4. Search strategies 2010
| CENTRAL |
| #1 MeSH descriptor Chagas Disease explode all trees #2 chaga* in All Text #3 MeSH descriptor Trypanosomiasis this term only #4 trypanosomiasis in All Text #5 cardiomyopath* in All Text #6 (#1 or #2 or #3 or #4 or #5) #7 nifurtimox in All Text #8 allopurinol in All Text #9 benznidazole in All Text #10 itraconazole in All Text #11 trypanocid* in All Text #12 antitrypanosom* in All Text #13 anti‐trypanosom* in All Text #14 MeSH descriptor trypanocidal agents explode all trees #15 MeSH descriptor nitrofurans explode all trees #16 nitrofurans in All Text #17 bzd in All Text #18 MeSH descriptor allopurinol this term only #19 MeSH descriptor itraconazole this term only #20 bay next 2502 in All Text #21 (#7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20) #22 (#6 and #21) |
| MEDLINE (OVID) |
| 1. exp Chagas Disease/ 2. Trypanosomiasis/ 3. chaga$.tw. 4. trypanosomiasis.tw. 5. cardiomyopath$.tw. 6. or/1‐5 7. nifurtimox.tw. 8. allopurinol.tw. 9. benznidazole.tw. 10. itraconazole.tw. 11. trypanocid$.tw. 12. antitrypanosom$.tw. 13. anti‐trypanosom$.tw. 14. exp Trypanocidal Agents/ 15. exp Nitrofurans/ 16. nitrofurans.tw. 17. bzd.tw. 18. Allopurinol/ 19. Itraconazole/ 20. trypanocidal.tw. 21. or/7‐20 22. 6 and 21 23. randomized controlled trial.pt. 24. controlled clinical trial.pt. 25. Randomized Controlled Trials/ 26. Random Allocation/ 27. Double‐Blind Method/ 28. Single‐Blind Method/ 29. or/23‐28 30. exp animal/ not humans/ 31. 29 not 30 32. clinical trial.pt. 33. exp Clinical Trials as Topic/ 34. (clin$ adj25 trial$).ti,ab. 35. ((singl$ or doubl$ or trebl$ or tripl$) adj (blind$ or mask$)).ti,ab. 36. Placebos/ 37. placebo$.ti,ab. 38. random$.ti,ab. 39. Research Design/ 40. or/32‐39 41. 40 not 30 42. 31 or 41 43. 22 and 42 44. 43 and (2008$ or 2009$ or 2010$).ed. |
| EMBASE (OVID) |
| 1. Chagas disease/ 2. trypanosomiasis/ 3. chaga$.tw. 4. trypanosomiasis.tw. 5. cardiomyopath$.tw. 6. or/1‐5 7. exp antitrypanosomal agent/ 8. exp nitrofuran derivative/ 9. nifurtimox.tw. 10. allopurinol.tw. 11. benznidazole.tw. 12. itraconazole.tw. 13. trypanocid$.tw. 14. antitrypanosom$.tw. 15. anti‐trypanosom$.tw. 16. nitrofurans.tw. 17. bzd.tw. 18. allopurinol/ 19. itraconazole/ 20. trypanocidal.tw. 21. or/7‐20 22. 6 and 21 23. controlled clinical trial/ 24. random$.tw. 25. randomized controlled trial/ 26. follow‐up.tw. 27. double blind procedure/ 28. placebo$.tw. 29. placebo/ 30. factorial$.ti,ab. 31. (crossover$ or cross‐over$).ti,ab. 32. (double$ adj blind$).ti,ab. 33. (singl$ adj blind$).ti,ab. 34. assign$.ti,ab. 35. allocat$.ti,ab. 36. volunteer$.ti,ab. 37. crossover procedure/ 38. single blind procedure/ 39. or/23‐38 40. 22 and 39 41. 40 and (2008$ or 2009$ or 2010$).em. |
| LILACS (in English) |
| (random$ or trial$ or clinical stud$ or rct) [Words] and (chaga$ or trypanosomiasis or cardiomyopath$) and (antitrypanocidal$ or anti‐trypanocidal$ or nitrofurans or nifurtimox or allopurinol or benznidazole or itraconazole or anti‐trypanosom$ or antitrypanosom$) [Words] and 2008 or 2009 or 2010 [Country, year publication] |
Appendix 5. Search strategies 2019
| [CENTRAL] |
| #1MeSH descriptor: [Chagas Disease] explode all trees #2chaga* #3MeSH descriptor: [Trypanosomiasis] this term only #4trypanosomiasis #5cardiomyopath* #6(#1 or #2 or #3 or #4 or #5) #7nifurtimox #8allopurinol #9benznidazole #10itraconazole #11trypanocid* #12antitrypanosom* #13anti‐trypanosom* #14MeSH descriptor: [Trypanocidal Agents] explode all trees #15MeSH descriptor: [Nitrofurans] explode all trees #16nitrofurans #17bzd #18MeSH descriptor: [Allopurinol] this term only #19MeSH descriptor: [Itraconazole] this term only #20bay next 2502 #21(#7 or #8 or #9 or #10 or #11 or #12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20) #22(#6 and #21) |
| MEDLINE (Ovid) |
| 1. exp Chagas Disease/ 2. Trypanosomiasis/ 3. chaga$.tw. 4. trypanosomiasis.tw. 5. cardiomyopath$.tw. 6. or/1‐5 7. nifurtimox.tw. 8. allopurinol.tw. 9. benznidazole.tw. 10. itraconazole.tw. 11. trypanocid$.tw. 12. antitrypanosom$.tw. 13. anti‐trypanosom$.tw. 14. exp Trypanocidal Agents/ 15. exp Nitrofurans/ 16. nitrofurans.tw. 17. bzd.tw. 18. Allopurinol/ 19. Itraconazole/ 20. trypanocidal.tw. 21. or/7‐20 22. 6 and 21 23. randomized controlled trial.pt. 24. controlled clinical trial.pt. 25. randomized.ab. 26. placebo.ab. 27. drug therapy.fs. 28. randomly.ab. 29. trial.ab. 30. groups.ab. 31. 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 32. exp animals/ not humans.sh. 33. 31 not 32 34. 22 and 33 35. (201003* or 201004* or 201005* or 201006* or 201007* or 201008* or 201009* or 20101* or 2011* or 2012* or 2013* or 2014* or 2015* or 2016* or 2017* or 2018* or 2019*).ed. 36. 34 and 35 |
| EMBASE |
| 1. Chagas disease/ 2. trypanosomiasis/ 3. chaga$.tw. 4. trypanosomiasis.tw. 5. cardiomyopath$.tw. 6. or/1‐5 7. exp antitrypanosomal agent/ 8. exp nitrofuran derivative/ 9. nifurtimox.tw. 10. allopurinol.tw. 11. benznidazole.tw. 12. itraconazole.tw. 13. trypanocid$.tw. 14. antitrypanosom$.tw. 15. anti‐trypanosom$.tw. 16. nitrofurans.tw. 17. bzd.tw. 18. allopurinol/ 19. itraconazole/ 20. trypanocidal.tw. 21. or/7‐20 22. 6 and 21 23. random$.tw. 24. factorial$.tw. 25. crossover$.tw. 26. cross over$.tw. 27. cross‐over$.tw. 28. placebo$.tw. 29. (doubl$ adj blind$).tw. 30. (singl$ adj blind$).tw. 31. assign$.tw. 32. allocat$.tw. 33. volunteer$.tw. 34. crossover procedure/ 35. double blind procedure/ 36. randomized controlled trial/ 37. single blind procedure/ 38. 23 or 24 or 25 or 26 or 27 or 28 or 29 or 30 or 31 or 32 or 33 or 34 or 35 or 36 or 37 39. (animal/ or nonhuman/) not human/ 40. 38 not 39 41. 22 and 40 42. (2010* or 2011* or 2012* or 2013* or 2014* or 2015* or 2016* or 2017* or 2018* or 2019*).dd. 43. 41 and 42 |
| LILACS |
| 2010 or 2011 or 2012 or 2013 or 2014 or 2015 or 2016 or 2017 or 2018 or 2019[Country, year publication] and (chaga$ or trypanosomiasis or cardiomyopath$) and (random$ or trial$ or clinical stud$ or rct) [Words] and antitrypanocidal$ or anti‐trypanocidal$ or nitrofurans or nifurtimox or allopurinol or benznidazole or itraconazole or anti‐trypanosom$ or antitrypanosom$ [Words] |
| ClinicalTrials.gov (Advanced search) |
|
Conditions: Chagas Cardiomyopathy OR Chagas Disease Interventions: benznidazole OR nifurtimox OR allopurinol OR itraconazole OR nitrofurans Age Group: Adult (18‐65) Study type: Interventional studies (Clinical trials) |
| International Clinical Trials Registry Plataform (ICTRP) (Advanced search) |
| chagas disease AND benznidazole OR chagas disease AND nifutimox |
Appendix 6. Survey of trial investigators providing information on included trials
| Trial ID | Date trial author contacted | Date trial author replied | Date trial author was asked for additional information (short summary) | Date trial author provided data (short summary) |
| Morillo 2015 | 25 December 2017 12 February 2018 27 February 2018 |
No answer 12 February 2018 27 February 2018 |
— Clarification of data inconsistencies with respect to positive and negative PCR and seroconversion of participants. Information about distribution of participants to treatment or placebo groups by country. Same information |
— Data were reviewed by statistician from the PHRI in Hamilton. Information will be sent by the end of the week. He did not possess the data and was waiting for statistician’s response. |
| Coura 1997 | 16 February 2018 | No answer | Type of participants: undetermined or chronic with clinical manifestations (cardiac, gastrointestinal, or both) | NA |
| "—" denotes not reported NA: not applicable; PCR: polymerase chain reaction | ||||
Appendix 7. Description of interventions
| Trial ID | Interventions(s) (route, frequency, total dose/day) | Comparator(s) (route, frequency, total dose/day) |
| Morillo 2015 | Benznidazole (oral, 300 mg per day, between 40 to 80 days) | Placebo (oral, per day, between 40 to 80 days) |
| Coura 1997 | I.1. Benznidazole (oral, 5 mg/kg/day every 12 hours for 30 days) I.2. Nifurtimox (oral, 5 mg/kg/day every 12 hours for 30 days) | Placebo group (oral, every 12 hours for 30 days) |
Appendix 8. Baseline characteristics (I)
| Trial ID | Intervention(s) and comparator(s) | Duration of intervention | Description of participants | Trial period (year to year) | Country | Setting | Ethnic groups (%) |
| Morillo 2015 | I. Benznidazole 300 mg/day C. Placebo |
40 to 80 days | Chagasic patients aged ≥ 18 years and ≤ 75 years, having any combination of at least 2 positive serologic tests for Chagas disease (indirect immunofluorescence, indirect haemagglutination, or ELISA), have evidence of cardiomyopathy | 7 years | 1. Argentina and Bolivia 2. Brazil 3. Colombia and El Salvador |
Multicentric study | 1. 20 2. 48 3. 32 |
| Coura 1997 | I.1. Benznidazole 5 mg/kg/day
I.2. Nifurtimox 5 mg/kg/day C. Placebo |
30 days | All participants with chronic Chagas disease with immunofluorescence with filter paper (IFP) and complement fixation (CP) reaction positive and at least 2 xenodiagnoses positive in 3 performed before treatment | 1 year | Brazil | Institute Oswaldo Cruz | 100 |
| ELISA: enzyme‐linked immunosorbent assay | |||||||
Appendix 9. Baseline characteristics (II)
| Trial ID | Intervention(s) and comparator(s) | Sex (female %) | Age (mean/range years SD, or as reported) | Comedications/cointerventions (% of participants) | Comorbidities (% of participants) |
| Morillo 2015 | I. Benznidazole 300 mg/day C. Placebo |
51 | ≥ 18 years and ≤ 75 years | — | — |
| Coura 1997 | I.1. Benznidazole 5 mg/kg/day
I.2. Nifurtimox 5 mg/kg/day C. Placebo |
— | — | — | — |
| "—" denotes not reported SD: standard deviation | |||||
Appendix 10. Matrix of trial endpoints (publications and trial documents)
| Trial ID | Endpoints quoted in trial document(s) (ClinicalTrials.gov, FDA/EMA document, manufacturer's website, published design paper) | Endpoints quoted in publication(s) | Endpoints quoted in abstract of publication(s) |
| Morillo 2015 | Published paper | The primary study outcome in the time‐to‐event analysis was the first occurrence of death, resuscitated cardiac arrest, insertion of a pacemaker or an implantable cardioverter–defibrillator, sustained ventricular tachycardia, cardiac transplantation, new heart failure, stroke or transient ischaemic attack, or a systemic or pulmonary thromboembolic event | The primary outcome in the time‐to‐event analysis was the first event of any of the components of the composite outcome of death, resuscitated cardiac arrest, sustained ventricular tachycardia, insertion of a pacemaker or implantable cardioverter–defibrillator, cardiac transplantation, new heart failure, stroke, or other thromboembolic event. |
| Coura 1997 | Source: NT | Primary outcome measure(s): — | Primary outcome measure(s): — |
| "—" denotes not reported EMA: European Medicines Agency; FDA: US Food and Drug Administration; NT: no trial document available | |||
Appendix 11. Definition of endpoint measurement
| Trial ID | Lowering or clearance of parasitaemia or anti‐Trypanosoma cruzi antibodies | Reduction or disappearance of clinical signs of heart failure, rhythm and conduction defects on ECG | Improvement in quality of life | Adverse effects (cutaneous reactions, digestive intolerance, and/or peripheral polyneuropathy) |
| Morillo 2015 | Blood sample for quantitative serological tests Drops of blood sample on filter papers for PCR |
The first occurrence of any component of the following cluster over the duration of follow‐up.
• Death
• Resuscitated cardiac arrest
• Documented sustained ventricular tachycardia requiring
cardioversion
• Insertion of pacemaker or implantable cardiac defibrillator
• Thromboembolic phenomena (stroke, pulmonary or
systemic embolism)
• New development of symptomatic HF characterised by 2 of the following: a) signs and symptoms of HF (orthopnoea, paroxysmal nocturna dyspnoea, shortness of breath, oedema); b) chest X‐ray: pulmonary congestion; c) need for intravenous therapy
(diuretics, inotropics); d) hospital admission due to HF Additional data will be collected in subgroups of participants to investigate the following other hypotheses. • New development of any of the following echo changes: segmental wall motion abnormalities, ventricular aneurysm, reduction in LVEF > 5%, increase in LVDD >5.0 mm compared with baseline • New 12‐lead ECG alterations (complete bundle branch block, fascicular block, advanced atrio‐ventricular block, atrial fibrillation, etc.) • Progression |
— | — |
| Coura 1997 | Participants were evaluated parasitological with xenodiagnosis. | — | — | In each medical visit participants were evaluated in an open‐ended manner about adverse events during the treatment period. |
| "—" denotes not reported ECG: electrocardiogram; HF: heart failure; LVDD: left ventricular diastolic dysfunction; LVEF: left ventricular ejection fraction; ND: not defined | ||||
Appendix 12. Adverse events (I)
| Trial ID | Intervention(s) and comparator(s) | Participants included in analysis (N) | Deaths (N) | Deaths (% of participants) | Participants with at least 1 adverse event (N) | Participants with at least 1 adverse event (%) | Participants with at least 1 severe/serious adverse event (N) | Participants with at least 1 severe/serious adverse event (%) |
| Morillo 2015 | I. Benznidazole 300 mg C. Placebo |
I. 1431 C. 1423 |
— | — | I. 342 C. 135 |
I. 23.9 C. 9.5 |
I. 19 C. 20 |
I. 8.3 C. 1.4 |
| Coura 1997 | I.1. Benznidazole 5 mg/kg/day
I.2. Nifurtimox 5 mg/kg/day C. Placebo |
I.1. 26 I.2. 27 C. 24 |
— | — | I.1. 3 I.2. 8 C. 2 |
I.1. 11.5 I.2. 29.6 C. 8.3 |
— | — |
| "—" denotes not reported | ||||||||
Appendix 13. Adverse events (II)
| Trial ID | Intervention(s) and comparator(s) | Participants included in analysis (N) | Participants discontinuing trial due to an adverse event (N) | Participants discontinuing trial due to an adverse event (%) | Participants with at least 1 hospitalisation (N) | Participants with at least 1 hospitalisation (%) | Participants with at least 1 outpatient treatment (N) | Participants with at least 1 outpatient treatment (%) |
| Morillo 2015 | I. Benznidazole 300 mg C. Placebo |
I. 1431 C. 1423 |
I. 2 C. 1 |
I. 0.1 C. 0.1 |
— | — | I. 1431 C. 1423 |
I. 100 C. 100 |
| Coura 1997 | I.1. Benznidazole 5 mg/kg/day
I.2. Nifurtimox 5 mg/kg/day C. Placebo |
I.1. 26 I.2. 27 C. 24 |
I.1. 3 I.2. 8 C. 2 |
I.1. 11.5 I.2. 29.6 C. 8.3 |
— | — | I.1. 26 I.2. 27 C. 24 |
I.1. 100 I.2. 100 C. 100 |
| "—" denotes not reported | ||||||||
Appendix 14. Adverse events (III)
| Trial ID | Intervention(s) and comparator(s) | Participants included in analysis (N) | Participants with a specific adverse event (description) | Participants with at least 1 specific adverse event (N) | Participants with at least 1 specific adverse event (%) |
| Morillo 2015 | I. Benznidazole 300 mg | I. 1431 | 1. Cutaneous rash 2. Gastrointestinal symptoms 3. Nervous system disorders |
1. 137 2. 112 3. 52 |
1. 9.6 2. 7.8 3. 3.6 |
| C. Placebo | C. 1423 | 1. Cutaneous rash 2. Gastrointestinal symptoms 3. Nervous system disorders |
1. 18 2. 41 3. 19 |
1. 1.3 2. 2.9 3. 1.3 |
|
| Coura 1997 | I.1. Benznidazole 5 mg/kg/day | I.1. 26 | 1. Gastrointestinal symptoms 2. Nervous system disorders 3. Cutaneous rash |
— | — |
| I.2. Nifurtimox 5 mg/kg/day | I.2. 27 | 1. Gastrointestinal symptoms 2. Nervous system disorders 3. Cutaneous rash |
|||
| C. Placebo | C. 24 | 1. Aerophagia 2. Postprandial fullness 3. Anoxia 4. Headache 5. Drowsiness 6. Fatigue |
|||
| "—" denotes not reported | |||||
Data and analyses
Comparison 1. Benznidazole versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Clearance of antibody titres | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1.1 At 12 months | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1.2 At 2 years | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1.3 At 5 years | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.2 Clearance of parasitaemia, demonstrated by negative xenodiagnosis, blood culture, and/or molecular biology assays | 1 | Peto Odds Ratio (Peto, Fixed, 95% CI) | Totals not selected | |
| 1.3 Clinical improvement | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.3.1 Heart failure | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.3.2 Sustained ventricular tachycardia | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected | |
| 1.4 Adverse effects | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected |
Comparison 2. Nifurtimox versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Clearance of antibody titres | 1 | Risk Ratio (IV, Fixed, 95% CI) | Totals not selected |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Coura 1997.
| Study characteristics | ||
| Methods |
Randomised controlled trial (1:1:1) Number of study centres: 1 Country: Brazil |
|
| Participants |
Inclusion criteria: all participants with chronic Chagas disease with immunofluorescence with filter paper (IFP) and complement fixation (CP) reaction positive and at least 2 xenodiagnoses positive in 3 performed before treatment Exclusion criteria: no data available Diagnostic criteria: positive IFP for Chagas disease confirmed with CP and at least 2 positive xenodiagnoses |
|
| Interventions |
Treatment before study: no Intervention: Benznidazole group: n = 26 (5 mg/kg/day every 12 hours) Nifurtimox group: n = 27 (5 mg/kg/day every 12 hours) Comparison: Placebo group: n = 24 Cointervention: not reported Treatment duration: 30 days Placebo was identical to treatment. Groups were age and gender matched. Follow‐up: 12 months after end of treatment |
|
| Outcomes |
Parasite related: parasitologic cure did not differ between treatment groups. Serological reaction did not change over the follow‐up. Patient related: 100% positive serology Positive xenodiagnoses were 1.8% in benznidazole group, 9.6% in nifurtimox group, and 34.3% in placebo group post‐treatment. There were no clinical ECG or X‐ray changes one year after treatment. |
|
| Study details |
Run‐in period: 1 year Trial terminated early: no |
|
| Publication details |
Language of publication: Portuguese Commercial funding: non‐funding mentioned Publication status: full article |
|
| Stated aim for study | Quote from publication: "Study to evaluate the therapeutic efficacy and tolerance of nifurtimox and benznidazole." | |
| Notes | 17% losses to follow‐up It is not clear if participants had chronic Chagas disease in the undetermined phases or in the symptomatic one. All serologic reactions continued positive, and there were no clinical, ECG, or X‐ray changes 1 year after treatment. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Unclear risk |
Quote from publication: Organization and treatment groups. Three groups of patients were organized with average age, sex and clinical form similar to each other. The drug was administered to each group at random and the doctor who administered it knew only that group I should take drug A, II drug B and III drug C (page 140) Comment: insufficient information. (Quote is a translated text.) |
| Random sequence generation (selection bias) | Unclear risk |
Quote from publication: Organization and treatment groups. Three groups of patients were organized with average age, sex and clinical form similar to each other. The drug was administered to each group at random and the doctor who administered it knew only that group I should take drug A, II drug B and III drug C (page 140) Comment: insufficient information. (Quote is a translated text.) |
| Blinding of participants and personnel (performance bias) Serum antibodies | Unclear risk |
Quote from publication: Patients received the medication for weekly treatment and weekly they were reviewed by the doctor, who examined them and noted the complaints and side effects, throughout the treatment period and up to 15 days after (page 141) Comment: insufficient information. (Quote is a translated text.) |
| Blinding of participants and personnel (performance bias) Negative xenodiagnosis | Unclear risk |
Quote from publication: The patients selected for treatment had an immunofluorescence reaction in blood collected on filter paper (IFP) positive for Chagas infection, confirmed by this reaction and by the fixation of complement in the serum (IFS, RFC) and by at least two positive xenodiagnostics for T cruzi in three performed, in consecutive months before treatment. Of the 80 patients, 51 (63.7%) had the three positive xenodiagnosis and 29 (36.2%) had two positive xenodiagnosis in the three performed, therefore, they were considered to have high parasitaemia (page 140) Comment: insufficient information. (Quote is a translated text.) |
| Blinding of participants and personnel (performance bias) Signs of heart failure | Unclear risk | Outcome not evaluated. |
| Blinding of participants and personnel (performance bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Blinding of outcome assessment (detection bias) Serum antibodies | Unclear risk |
Quote from publication: Also in this last review, blood was collected by venipuncture and immunofluorescence reactions and fixation of the complement were carried out to search for anti‐T cruzi antibodies (page 141) Comment: insufficient information. (Quote is a translated text.) |
| Blinding of outcome assessment (detection bias) Negative xenodiagnosis | Unclear risk |
Quote from publication: Cure control. The cure control was carried out in the 1st, 2nd and 3rd months after treatment, when the patients were re‐examined and submitted to xenodiagnosis with the same technique previously described. In the 12th month after treatment, patients were reexamined and submitted to three xenodiagnosis at 15‐day intervals, totaling six xenodiagnosis, after treatment (page 141) Comment: insufficient information. (Quote is a translated text.) |
| Blinding of outcome assessment (detection bias) Signs of heart failure | Unclear risk | Outcome not evaluated. |
| Blinding of outcome assessment (detection bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Incomplete outcome data (attrition bias) Serum antibodies | Unclear risk |
Quote from publication: Serological reactions were still positive in all patients in all groups in the 12th month after treatment (page 141) Comment: insufficient information. (Quote is a translated text.) |
| Incomplete outcome data (attrition bias) Negative xenodiagnosis | Unclear risk |
Quote from publication: Of the 110 xenodiagnostics performed in group I after treatment, only two (1.8%) were positive, one in the 2nd and the other in the 12th month, while of the 83 xenodiagnosis performed in group II, eight (9.6%) were positive, seven of which in the first three months after treatment and one in the 12th month. In the placebo group of 67 post‐treatment xenodiagnostics performed, 23 (34.3%) were positive (page 141) Comment: insufficient information. (Quote is a translated text.) |
| Incomplete outcome data (attrition bias) Signs of heart failure | Unclear risk | Outcome not evaluated. |
| Incomplete outcome data (attrition bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Selective reporting (reporting bias) | Unclear risk | Comment: adverse events outcomes were analysed but results not reported |
| Other bias | Low risk | Comment: no other potential source of bias |
Morillo 2015.
| Study characteristics | ||
| Methods |
Randomised, double‐blind, placebo‐controlled trial Number of study centres: 49 Countries: Argentina, Bolivia, Brazil, Colombia, and El Salvador |
|
| Participants |
Inclusion criteria: chagasic patients aged ≥ 18 years and ≤ 75 years were eligible if, in addition to having any combination of at least 2 positive serologic tests for Chagas disease (indirect immunofluorescence, indirect haemagglutination, or ELISA), have evidence of cardiomyopathy Exclusion criteria:
Diagnostic criteria: at least 2 positive serologic tests for Trypanosomacruzi, and evidence of cardiomyopathy |
|
| Interventions |
Treatment before study: no Intervention: Benznidazole group: n = 1431 in (300 mg per day and a variable duration of therapy on the basis of participant weight) Placebo group: n = 1423 Co‐intervention: not reported Treatment duration: between 40 and 80 days Placebo was identical to treatment. Follow‐up: 7 years |
|
| Outcomes |
Primary outcome: in the time‐to‐event analysis was the first event of any of the components of the composite outcome of death, resuscitated cardiac arrest, sustained ventricular tachycardia, insertion of a pacemaker or implantable cardioverter‐defibrillator, cardiac transplantation, new heart failure, stroke, or other thromboembolic event Secondary outcome: included the response to treatment on the basis of results in PCR assay overall and according to geographic region corresponding to the common prevalent T cruzi discrete type units: T cruzi I in Colombia and El Salvador, T cruzi II in Brazil, T cruzi V and VI in Argentina and Bolivia, respectively |
|
| Study details |
Run‐in period: 1 year Trial terminated early: no |
|
| Publication details |
Language of publication: English Commercial funding: supported by the Population Health Research Institute Hamilton Health Sciences and McMaster University, a grant (MCT‐79704) from the Canadian Institutes of Health Research, a grant (A30755) from the United Nations Children’s Fund, United Nations Development Program, World Bank, and World Health Organization Special Program for Research and Training in Tropical Diseases, Fundação de Amparo à Pesquisa, Ensino e Assistência, Hospital das Clínicas da Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo, and Ministerio de Salud and Fundación Bunge y Born, Argentina. Dr Morillo reports receiving lecture fees from Bayer and Boehringer Ingelheim and grant support from Boston Scientific and Merck. Dr Lazdins reports receiving consulting fees from Merck. No other potential conflict of interest relevant to this article was reported. Publication status: full article |
|
| Stated aim for study | Quote from publication: "In conclusion, among patients with established Chagas’ cardiomyopathy, benznidazole treatment significantly reduced the detection of circulating parasites but did not reduce cardiac clinical progression." | |
| Notes | A total of 14 participants (0.5%) were lost to follow‐up at the end of the study. There was funding for only 1896 PCR tests. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment (selection bias) | Low risk | Quote from publication: "The patient identification (ID) and treatment numbers will be provided at .... randomisation by a centralized randomisation service ..." (page 17) |
| Random sequence generation (selection bias) | Low risk | Quote from publication: "The patient identification (ID) and treatment numbers will be provided at randomisation by a centralized randomisation service located at the Brazilian Coordinating Centre" (page 17) |
| Blinding of participants and personnel (performance bias) Serum antibodies | Unclear risk |
Quote from publication: "Detection of parasite and measures of parasite burden by PCR will be done at a core laboratory for each involved country" (page 40) Comment: insufficient information |
| Blinding of participants and personnel (performance bias) Negative xenodiagnosis | Unclear risk | Outcome not evaluated. |
| Blinding of participants and personnel (performance bias) Signs of heart failure | Low risk | Quote from publication: "A central adjudicator at the project office, blinded to treatment assignment and using essential supportive documentation to confirm the diagnosis, adjudicate study outcomes" (page 40) |
| Blinding of participants and personnel (performance bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Blinding of outcome assessment (detection bias) Serum antibodies | Unclear risk |
Quote from publication: "Detection of parasite and measures of parasite burden by PCR will be done at a core laboratory for each involved country" (page 40) Comment: insufficient information |
| Blinding of outcome assessment (detection bias) Negative xenodiagnosis | Unclear risk | Outcome not evaluated. |
| Blinding of outcome assessment (detection bias) Signs of heart failure | Low risk | Quote from publication: "Patients will have a baseline echocardiogram before treatment allocation and another one at the end of follow‐up. All analyses will be blinded and carried centralized at a core laboratory" (page 40) |
| Blinding of outcome assessment (detection bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Incomplete outcome data (attrition bias) Serum antibodies | Low risk |
Quote from publication: "A 10 ml blood sample to be used for a qualitative conventional PCR assay for the detection of circulating T cruzi kinetoplast DNA (kDNA) by means of an internationally validated method was obtained from 1896 patients at baseline ..." (page 1297) Comment: data for primary outcome available for 78% of benznidazole group and 82% of placebo group |
| Incomplete outcome data (attrition bias) Negative xenodiagnosis | Unclear risk | Outcome not evaluated. |
| Incomplete outcome data (attrition bias) Signs of heart failure | Low risk | Quote from publication: "The primary study outcome in the time‐to‐event analysis was the first occurrence of death, resuscitated cardiac arrest, insertion of a pacemaker or an implantable cardioverter–defibrillator, sustained ventricular tachycardia, cardiac transplantation, new heart failure, stroke or transient ischemic attack, or a systemic or pulmonary thromboembolic event. An event adjudication committee reviewed all cardiovascular outcomes in a blinded fashion" (page 1297) |
| Incomplete outcome data (attrition bias) Quality of life | Unclear risk | Outcome not evaluated. |
| Selective reporting (reporting bias) | Unclear risk | Comment: serum antibodies outcomes were analysed but results not reported |
| Other bias | Low risk | Comment: no other potential source of bias |
ELISA: enzyme‐linked immunosorbent assay NYHA: New York Heart Association PCR: polymerase chain reaction
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Aguiar 2012 | Purpose of study was related to diagnostic methods (to evaluate the serological and parasitological status of individuals with chronic Chagas disease after chemotherapy with benznidazole). (page 368) |
| Alvarez 2012 | Chronic Chagas disease patients were treated with benznidazole, and the efficacy of treatment was evaluated serologically and parasitologically. However, treatment was not completed because of adverse effects. The efficacy of treatment could therefore not be evaluated. (page 636) |
| Alvarez 2016 | Purpose of study was to probe other therapeutic schemes, and was only a pilot short‐term follow‐up study. (page 833) |
| Bertocchi 2013 | A longitudinal prospective cohort was used, without randomisation of patients to treatment groups. (page 372) |
| Bestetti 2011 | Case report pertaining to the efficacy of the treatment in the chronic phase of Chagas disease (page 2527) |
| Braga 2000 | Randomisation was not followed, and participants and physicians were not blind to the treatment groups. (page 158) |
| Bravo 2011 | A cross‐sectional study that aimed to determine the epidemiological, clinical, and inflammatory risk factors associated with seronegative or seropositive heart failure patients (page 691) |
| Cancado 2002 | An observational study with a long follow‐up to investigate the cure rate in acute (13 to 21 years) and chronic (6 to 18 years) patients (page 29) |
| EQUITY 2019 | The aim of the EQUITY trial is to evaluate, amongst young adults with chronic Trypanosomacruzi infection but without symptomatic chronic Chagas cardiomyopathy, the trypanocidal effect and safety of a treatment with nifurtimox compared to benznidazole or placebo. (page 2) |
| Fabbro 2000 | This longitudinal study compares the evolution of chronic undetermined chagasic patients treated with benznidazole or nifurtimox versus those untreated. Patients were not randomised to study groups. Study was neither single‐ nor double‐blinded. (page 100) |
| Lauria‐Pires 2000 | Study is not a clinical trial. (page 111) |
| Machado‐de‐Assis 2012 | The aim of this study was to evaluate conventional serology test followed by supplementary serological, parasitological, and molecular tests to determine the effectiveness of aetiologic treatment of Chagas disease. Included participants were in the undetermined phase of Chagas disease and were treated 10 years before the study. (pages 1283‐4) |
| Moreira 2013 | This study belongs to the same group as the BENEFIT one, and its main purpose was to standardise qPCR strategy to quantify Trypanosomacruzi DNA in blood. A comparative quantitative analysis was therefore performed among CCC patients from Argentina, Brazil, and Colombia, prior to benznidazole/placebo administration. (page 24) |
| Moreno 2010 | This is an experimental study that developed a methodology to quantify benznidazole susceptibility in blood culture‐derived isolates from patients in the chronic phase of Chagas disease in order to assess the therapeutic failure of benznidazole. (page 919) |
| Murcia 2010 | The purpose of this study was to assess the efficacy of treatment in individuals with chronic Chagas disease and to monitor the effect of the treatment by means of PCR. (page 1759) |
| Perez‐Ayala 2010 | A cross‐sectional study that describes the clinical‐epidemiological characteristics of the largest cohort of Latin American immigrants with chronic Trypanosomacruzi infection, as well as providing preliminary data on response to and tolerance of treatment with benznidazole. (page 1109) |
| SaMiTrop 2018 | A prospective cohort study. The goal of this study was to understand the role of benznidazole in preventing the appearance of cardiac lesions when used in less advanced diseases, which is still controversial. (page 3) |
| TRAENA 2015 | The primary objective of this study is to probe whether trypanocidal drugs have a direct effect on the reduction or even elimination of anti‐Trypanosomacruzi antibodies. Patients in different stages of the disease were included. The final results have not been published; however, preliminary findings demonstrate that benznidazole has a trypanocidal effect capable of eliminating parasites. (page 6) |
| Viotti 1994 | This is a quasi‐experimental study, and the follow‐up was not the same for all participants. (page 154) |
| Viotti 2006 | A clinical trial with unblinded and non‐random assignment of patients. (page 724) |
CCC: chronic chagasic cardiomyopathy PCR: polymerase chain reaction qPCR: quantitative polymerase chain reaction
Characteristics of ongoing studies [ordered by study ID]
NCT03191162.
| Study name | MULTIBENZ |
| Methods | Phase II clinical trial |
| Participants | 240 participants |
| Interventions | 3 different benznidazole regimens: 300 mg/day for 60 days, 150 mg/day for 60 days, and 400 mg/day for 15 days |
| Outcomes | Proportion of participants with negative parasitaemia measured by polymerase chain reaction (PCR) during the first 12 months after start of treatment |
| Starting date | 21 April 2017 |
| Contact information | Contact: Israel Molina, PhD, 0034 93 274 6251i, molina@vhebron.net Contact: Fernando Salvador, MD, 0034 93 274 6251, fmsalvad@vhebron.net |
| Study details | A phase II clinical trial to evaluate the efficacy of different benznidazole regimens (300 mg/day for 60 days, 150 mg/day for 60 days, and 400 mg/day for 15 days) for the treatment of chronic Chagas disease in adult patients. Efficacy is assessed through the proportion of participants with negative parasitaemia measured by PCR during the first 12 months after commencing treatment. The study was proposed to be performed in Spain, Brazil, Argentina, and Colombia. |
| Publication details | None |
| Stated aim for study | 21 April 2017 |
| Notes | Last update was posted on 19 June 2017. |
Differences between protocol and review
Since a long time has passed since the publication of the protocol for this review (2004), we have amended the current update to comply with the latest requirements for Cochrane Reviews.
Contributions of authors
The final draft was read and approved by all authors.
Maite Vallejo (MV): conceived and wrote the protocol, undertook retrieval and review of citations, selected studies for inclusion, performed data extraction, data interpretation, and review of drafts.
Pedro A Reyes (PAR): conceived and wrote the protocol, undertook retrieval and review of citations.
Mireya Martinez Garcia (MMG): undertook retrieval and review of citations, selected studies for inclusion, performed data extraction, data interpretation, and review of drafts.
Alejandro Gonzalez Garay (AGG): data interpretation and review of drafts.
Sources of support
Internal sources
Instituto Nacional de Cardiologia, Ignacio Chavez, Tlalpan, Mexico
External sources
-
NIHR, UK
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to the Heart Group. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health and Social Care.
Declarations of interest
There is no conflict of interest in this work.
MV: none known.
PAR: none known.
MMG: none known.
AGG: none known.
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Coura 1997 {published data only}
- Coura JR, De Abreu L, Percy H, Willcox F, Petana W. A controlled comparative study using benznidazole, nifurtimox and placebo in chronic Chagas disease patients, in a field area with interrupted transmission: I. preliminary evaluation [Estudo comparativo controlado com emprego de benznidazole, nifurtimox e placebo, na forma crônica da doença de Chagas, em uma área de campo com transmissäo interrompida: I. avaliaçäo preliminar]. Revista da Sociedade Brasileira de Medicina Tropical 1997;30:139-44. [DOI: 10.1590/s0037-86821997000200009] [DOI] [PubMed] [Google Scholar]
Morillo 2015 {published data only}
- Morillo C, Marin-Neto J, Avezum A, Sosa-Estani S, Rassi A, Rosas F, et al, BENEFIT Investigators. Randomized trial of benznidazole for chronic Chagas' cardiomyopathy. New England Journal of Medicine 2015;373:1295-306. [DOI: 10.1056/NEJMoa1507574] [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Aguiar 2012 {published data only}
- Aguiar C, Batista A, Pavan T, Almeida E, Guariento M, Wanderleyn J, et al. Serological profiles and evaluation of parasitaemia by PCR and blood culture in individuals chronically infected by Trypanosoma cruzi treated with benzonidazole. Tropical Medicine & International Health 2012;17(3):368-73. [DOI: 10.1111/j.1365-3156.2011.02936.x] [DOI] [PubMed] [Google Scholar]
Alvarez 2012 {published data only}
- Alvarez M, Vigliano C, Lococo B, Petti M, Bertocchi G, Viotti R. Seronegative conversion after incomplete benznidazole treatment in chronic Chagas disease. Transactions of the Royal Society of Tropical Medicine and Hygiene 2012;106(10):636-8. [DOI: 10.1016/j.trstmh.2012.07.010] [DOI] [PubMed] [Google Scholar]
Alvarez 2016 {published data only}
- Álvarez M, Hernández Y, Bertocchi G, Fernández M, Lococo B, Ramírez J, et al. New scheme of intermittent benznidazole administration in patients chronically infected with Trypanosoma cruzi: a pilot short-term follow-up study with adult patients. Antimicrobial Agents and Chemotherapy 2016;60:833–7. [DOI: 10.1128/AAC.00745-15] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bertocchi 2013 {published data only}
- Bertocchi GL, Vigliano C, Lococo B, Petti M, Viotti R. Clinical characteristics and outcome of 107 adult patients with chronic Chagas disease and parasitological cure criteria. Transactions of the Royal Society of Tropical Medicine and Hygiene 2013;107:372–6. [DOI: 10.1093/trstmh/trt029] [DOI] [PubMed] [Google Scholar]
Bestetti 2011 {published data only}
- Bestetti R, Cardinalli-Neto A. Antitrypanosomal therapy for chronic Chagas’ disease. New England Journal of Medicine 2011;364(26):2527-34. [DOI: 10.1056/NEJMc1108653] [DOI] [PubMed] [Google Scholar]
Braga 2000 {published data only}
- Braga M, Lauria-Pires L, Argalaraz E, Nascimento R, Teixeira A. Persistent infection in chronic Chagas disease patients treated with anti-trypanosoma cruzi nitroderivatives. Revista do Instituto de Medicina Tropical de Sao Paulo 2000;42:157-61. [DOI: 10.1590/s0036-46652000000300009] [DOI] [PubMed] [Google Scholar]
Bravo 2011 {published data only}
- Bravo I, Parra F, Nello C, Rodríguez-Bonfante C, Useche F, Bonfante-Cabarcas R. Prevalence of Trypanosoma cruzi antibodies and inflammatory markers in uncompensated heart failure. Revista da Sociedade Brasileira de Medicina Tropical 2011;44(6):691-6. [DOI: 10.1590/s0037-86822011000600008] [DOI] [PubMed] [Google Scholar]
Cancado 2002 {published data only}
- Cancado J. Long term evaluation of etiological treatment of Chagas disease with benznidazole. Revista do Instituto de Medicina Tropical de Sao Paulo 2002;44:29-37. [DOI: 10.1590/S0036-46652002000100006 ] [PubMed] [Google Scholar]
EQUITY 2019 {published data only}
- Villar J, Herrera V, Pérez J, Váquiro E, Castellanos Y, Vásquez S, et al. Nifurtimox versus benznidazole or placebo for asymptomatic Trypanosoma cruzi infection (Equivalence of Usual Interventions for Trypanosomiasis - EQUITY): study protocol for a randomised controlled trial. Trials 2019;20(1):1-10. [DOI: 10.1186/s13063-019-3423-3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Fabbro 2000 {published data only}
- Fabbro de Suasnábar D, Arias E, Streiger M, Placenza M, Ingraramo M, Del Barco M, et al. Evolutive behavior towards cardiomyopathy of treated (nifurtimox or benznidazole) and untreated Chagasic patients. Revista do Instituto de Medicina Tropical de Sao Paulo 2000;42:99-109. [DOI: 10.1590/s0036-46652000000200007] [DOI] [PubMed] [Google Scholar]
Lauria‐Pires 2000 {published data only}
- Lauria-Pires L, Braga M, Vexenat A, Nitz N, Simoes-Barbosa A, Tinoco D, et al. Progressive chronic Chagas heart disease ten years after treatment with anti-Trypanosoma cruzi nitroderivatives. American Journal of Tropical Medicine and Hygiene 2000;63(3-4):111-8. [DOI: 10.4269/ajtmh.2000.63.111] [DOI] [PubMed] [Google Scholar]
Machado‐de‐Assis 2012 {published data only}
- Machado-de-Assis G, Silva A, Do Bem V, Bahia M, Martins-Filho O, Dias J, et al. Posttherapeutic cure criteria in Chagas' disease: conventional serology followed by supplementary serological, parasitological, and molecular tests. Clinical and Vaccine Immunology 2012;19(8):1283-91. [DOI: 10.1128/CVI.00274-12] [DOI] [PMC free article] [PubMed] [Google Scholar]
Moreira 2013 {published data only}
- Moreira O, Ramírez J, Velázquez E, Melo M, Lima-Ferreira C, Guhl F, et al. Towards the establishment of a consensus real-time qPCR to monitor Trypanosoma cruzi parasitaemia in patients with chronic Chagas disease cardiomyopathy: a substudy from the BENEFIT trial. Acta Tropica 2013;125(1):23-31. [DOI: 10.1016/j.actatropica.2012.08.020] [DOI] [PubMed] [Google Scholar]
Moreno 2010 {published data only}
- Moreno M, D'ávila D, Silva M, Galvão L, Macedo A, Chiari E, et al. Trypanosoma cruzi benznidazole susceptibility in vitro does not predict the therapeutic outcome of human Chagas disease. Memorias do Instituto Oswaldo Cruz 2010;105(7):918-24. [DOI: 10.1590/s0074-02762010000700014] [DOI] [PubMed] [Google Scholar]
Murcia 2010 {published data only}
- Murcia L, Carrilero B, Muñoz M, Iborra M, Segovia M. Usefulness of PCR for monitoring benznidazole response in patients with chronic Chagas’ disease: a prospective study in a non-disease-endemic country. Journal of Antimicrobial Chemotherapy 2010;65(8):1759-64. [DOI: 10.1093/jac/dkq201] [DOI] [PubMed] [Google Scholar]
Perez‐Ayala 2010 {published data only}
- Pérez-Ayala A, Pérez-Molina J, Norman F, Navarro M, Monge-Maillo B, Díaz-Menéndez M, et al. Chagas disease in Latin American migrants: a Spanish challenge. Clinical Microbiology and Infection 2011;17(7):1108-13. [DOI: 10.1111/j.1469-0691.2010.03423.x] [DOI] [PubMed] [Google Scholar]
SaMiTrop 2018 {published data only}
- Cardoso C, Ribeiro A, Oliveira C, Oliveira L, Ferreira A, Bierrenbach A, et al. Beneficial effects of benznidazole in Chagas disease: NIH SaMi-Trop cohort study. PLOS Neglected Tropical Diseases 2018;12:1-12. [DOI: 10.1371/journal.pntd.0006814] [DOI] [PMC free article] [PubMed] [Google Scholar]
TRAENA 2015 {published data only}
- Riarte A. TRENA: Treatment with benznidazole in adult patients with chronic Chagas' disease. A phase 3 randomised clinical trial (RCT) double blinded [TRAENA: TRAtamiento con benznidazol EN pacientes Adultos con enfermedad de Chagas crónica. Un ensayo clínico aleatorizado (ECA) a doble ciego en Fase 3]. Informativo. Plataforma de investigación clínica en enfermedad de Chagas 2015;4:6-7. [Google Scholar]
Viotti 1994 {published data only}
- Viotti R, Vigliano C, Armenti H, Segura E. Treatment of chronic Chagas' disease with benznidazole: clinical and serological evolution of patients with long-term follow-up. American Heart Journal 1994;127:151-62. [DOI: 10.1016/0002-8703(94)90521-5] [DOI] [PubMed] [Google Scholar]
Viotti 2006 {published and unpublished data}
- Viotti R, Viglian C, Lococo B, Bertochi G, Petti M, Alvarez M, et al. Long-term cardiac outcomes of treating chronic Chagas disease with benznidazole versus no treatment: a non-randomised trial. Annals of Internal Medicine 2006;144:724-34. [DOI: 10.7326/0003-4819-144-10-200605160-00006] [DOI] [PubMed] [Google Scholar]
References to ongoing studies
NCT03191162 {unpublished data only}
- NCT03191162. Evaluation of different benznidazole regimens for the treatment of chronic Chagas disease (MULTIBENZ). clinicaltrials.gov/ct2/show/NCT03191162 (first received 19 June 2017).
Additional references
Andrade 2011
- Andrade J, Marin J, Paola A, Vilas-Boas F, Oliveira G, Bacal F, et al. I Latin American guidelines for the diagnosis and treatment of Chagas heart disease: executive summary. Arquivos Brasileiros de Cardiologia 2011;96:434-42. [DOI: 10.1590/s0066-782x2011000600002] [DOI] [PubMed] [Google Scholar]
Barretto 1990
- Barretto A, Azul L, Mady C, Ianni B, De Brito V, Belloti G, et al. Indeterminate form of Chagas' disease. A polymorphic disease [Forma indeterminada da doenca de chagas: Uma doenca polimorfica]. Arquivos Brasileiros de Cardiologia 1990;55:347-53. [PMID: ] [PubMed] [Google Scholar]
Brener 1975
- Brener Z. Chemotherapy of Trypanosoma cruzi infections. Advances in Pharmacology 1975;13:1-44. [DOI: 10.1016/S1054-3589(08)60229-X] [DOI] [PubMed] [Google Scholar]
Briceno 2016
- Briceno L, Mosca W. What is not searched, it is difficult to find: Chagas' disease [Quello che non si cerca difficilmente si trova: la malattia di Chagas]. Giornale Italiano di Cardiologia 2016;17(5):343-7. [DOI: 10.1714/2252.24258] [DOI] [PubMed] [Google Scholar]
Cecere 1999
- Cecere M, Castañera M, Canale D, Chuit R, Gürtler R. Trypanosoma cruzi infection in Triatoma infestans and other triatomines: long-term effects of a control program in rural northwestern Argentina. Revista Panamericana de Salud Publica [Pan American Journal of Public Health] 1999;5:392-9. [DOI: 10.1590/s1020-49891999000500003] [DOI] [PubMed] [Google Scholar]
Chagas 1909
- Chagas C. New human trypanosomia. Studies on the morphology and evolutionary cycle of the Schizotrypanum cruzi n.gen., N.sp., ethiological adjunct of a new morbid entity of man [Nova tripanozomiaze humana. Estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n.gen., n.sp., ajente etiolojico de nova entidade morbida do homem]. Memorias do Instituto Oswaldo Cruz 1909;1:159-218. [DOI: 10.1590/S0074-02761909000200008] [DOI] [Google Scholar]
Cochrane Heart Group
- Cochrane Heart Group. Preferred method for handling continuous variables. www.heart.cochrane.org/Files/Handling%20continuous%20variables.pdf (accessed June 2005).
Contijo 1999
- Contijo E, Galvão L, Eloi-Santos S. Chagas disease: criteria of cure and prognosis. Memorias do Instituto Oswaldo Cruz 1999;94(Suppl 1):357-62. [DOI: 10.1590/S0074-02761999000700069] [DOI] [PubMed] [Google Scholar]
Coura 2009
- Coura J. Present situation and new strategies for Chagas disease chemotherapy: a proposal. Memorias do Instituto Oswaldo Cruz 2009;104(4):549-54. [DOI: 10.1590/s0074-02762009000400002] [DOI] [PubMed] [Google Scholar]
de Andrade 1996
- Andrade A, Zicker F, Oliveira R, Almeida S, Luquetti A, Travassos L, et al. Randomised trial of efficacy of benznidazole in treatment of early Trypanosoma cruzi infection. Lancet 1996;348(9039):1407-13. [DOI: 10.1016/s0140-6736(96)04128-1] [DOI] [PubMed] [Google Scholar]
Dias 2009
- Dias J. Elimination of Chagas disease transmission: prospectives. Memorias do Instituto Oswaldo Cruz 2009;104(Suppl 1):41-5. [DOI: 10.1590/S0074-02762009000900007] [DOI] [PubMed] [Google Scholar]
Follman 1992
- Follman D, Elliot P, Suh I, Cutler J. Variance imputation for overviews of clinical trials with continuous response. Journal of Clinical Epidemiology 1992;45:769-73. [DOI: 10.1016/0895-4356(92)90054-q] [DOI] [PubMed] [Google Scholar]
Fumadó 2014
- Fumadó V, Juncosa T, Posada E, Fisa R, Gállego M, Gascón J. Paediatric Chagas in a non-endemic area [Chagas pediátrico en zona no endémica]. Enfermedades Infecciosas y Microbiologia Clinica 2014;32(5):293-6. [DOI: 10.1016/j.eimc.2013.04.024] [DOI] [PubMed] [Google Scholar]
Gobbi 2014
- Gobbi F, Angheben A, Anselmi M, Postiglione C, Repetto E, Buonfrate D, et al. Profile of Trypanosoma cruzi infection in a tropical medicine reference center, Northern Italy. PLOS Neglected Tropical Diseases 2014;8(12):e3361. [DOI: 10.1371/journal.pntd.0003361] [DOI] [PMC free article] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- McMaster University (developed by Evidence Prime) GRADEpro GDT. Version accessed prior to 10 December 2020. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Grant 1989
- Grant I, Gold J, Wittner M, Tanowitz H, Nathan C, Mayer K, et al. Transfusion-associated acute Chagas disease acquired in the United States. Annals of Internal Medicine 1989;111:849. [DOI: 10.7326/0003-4819-111-10-849] [DOI] [PubMed] [Google Scholar]
Harry 2000
- Harry M, Lema F, Romaña C. Chagas' disease challenge. Lancet 2000;355:236. [DOI: 10.1016/S0140-6736(05)72114-0] [DOI] [PubMed] [Google Scholar]
Higgins 2002
- Higgins J, Thompson S. Quantifying heterogeneity in a meta-analysis. Statistics in Medicine 2002;21(11):1539-58. [DOI: 10.1002/sim.1186] [DOI] [PubMed] [Google Scholar]
Higgins 2003
- Higgins J, Thompson S, Deeks J, Altman D. Measuring inconsistency in meta-analysis. BMJ 2003;327(7414):557-60. [DOI: 10.1136/bmj.327.7414.557] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Higgins 2011b
- Higgins J, Altman D, Gøtzsche P, Jüni P, Moher D, Oxman A, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 2011;18(343):d5928. [DOI: 10.1136/bmj.d5928 ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hozo 2005
- Hozo S, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Medical Research Methodology 2005;5(13):1-10. [DOI: 10.1186/1471-2288-5-13] [DOI] [PMC free article] [PubMed] [Google Scholar]
Imai 2015
- Imai K, Maeda T, Sayama Y, Osa M, Mikita K, Kurane I, et al. Chronic Chagas disease with advanced cardiac complications in Japan: case report and literature review. Parasitology International 2015;64(5):240-2. [DOI: 10.1016/j.parint.2015.02.005] [DOI] [PubMed] [Google Scholar]
Kraus 2009
- Kraus J, Verlinde C, Karimi M, Lepesheva G, Gelb M, Buckner F. Rational modification of a candidate cancer drug for use against Chagas disease. Journal of Medicinal Chemistry 2009;52(6):1639-47. [DOI: 10.1021/jm801313t] [DOI] [PMC free article] [PubMed] [Google Scholar]
Kubata 2002
- Kubata B, Kabututu Z, Nozaki T, Munday C, Fukuzumi S, Ohkubo K, et al. A key role for old yellow enzyme in the metabolism of drugs by Tripanosoma cruzi. Journal of Experimental Medicine 2002;196:1241-51. [DOI: 10.1084/jem.20020885] [DOI] [PMC free article] [PubMed] [Google Scholar]
Laucella 2009
- Laucella S, Mazliah D, Bertocchi G, Alvarez M, Cooley G, Viotti R, et al. Changes in Trypanosoma cruzi-specific immune responses after treatment: surrogate markers of treatment efficacy. Clinical Infectious Diseases 2009;49(11):1675-84. [DOI: 10.1086/648072] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lefebvre 2011
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from handbook.cochrane.org.
Lopez‐Monteon 2019
- López-Monteon A, Dumonteil E, Ramos-Ligonio A. More than a hundred years in the search for an accurate diagnosis for Chagas disease: current panorama and expectations. In: Rodriguez-Morales AJ, editors(s). Current Topics in Neglected Tropical Diseases. IntechOpen, 2019:4-14. [DOI: 10.5772/intechopen.86567] [DOI] [Google Scholar]
Mady 2008
- Mady C, Ianni B, Souza J. Benznidazole and Chagas disease: can an old drug be the answer to an old problem? Expert Opinion on Investigational Drugs 2008;17(10):1427-33. [DOI: 10.1517/13543784.17.10.1427] [DOI] [PubMed] [Google Scholar]
Monteón 1995
- Monteón V, Guzman-Bracho C, Floriani-Verdugo J, Ramos-Echavaria A, Velasco-Castrejón O, Reyes-López PA. Serological diagnosis of Chagas disease: self-sufficiency and interlaboratory concordance [Diagnóstico serológico de la enfermedad de Chagas: autosuficiencia y concordancia interlaboratorios]. Salud Publica de Mexico 1995;37:232-5. [PMID: ] [PubMed] [Google Scholar]
Monteón‐Padilla 1999
- Monteón-Padilla V, Hernández-Becerril N, Guzmán-Bracho C, Rosales-Encina J, Reyes-López PA. American Trypanosomiasis (Chagas' disease) and blood banking in Mexico City: seroprevalence and its potential transfusional transmission risk. Archives of Medical Research 1999;30:393-8. [DOI: 10.1016/S0188-4409(99)00062-4] [DOI] [PubMed] [Google Scholar]
Moretti 1998
- Moretti E, Cervetta L, Basso B, Castro I, Santamarina N. Chronic Chagas' disease: effects of treatment on the levels of antibodies to crude and partially purified Trypanosoma cruzi antigens [Enfermedad de Chagas crónica: efectos del tratamiento en los niveles de anticuerpos hacia antígenos crudos y semipurificados del Trypanosoma cruzi]. Boletin Chileno de Parasitologia 1998;53:3-9. [PMID: ] [PubMed] [Google Scholar]
Nickerson 1989
- Nickerson P, Our P, Schroeder M, Sekla L, Johnston J. Transfusion-associeted Trypanosoma cruzi infection in a non-endemic area. Annals of Internal Medicine 1989;111:851. [DOI: 10.7326/0003-4819-111-10-851] [DOI] [PubMed] [Google Scholar]
Oliveira‐Filho 1989
- Oliveira-Filho A. Cost-effectiveness analysis in Chagas' disease vector's control intervention. Memorias do Instituto Oswaldo Cruz 1989;84 (Suppl IV):409-17. [DOI: 10.1590/S0074-02761989000800074] [DOI] [Google Scholar]
Perman 2017
- Perman G. Cluster clinical trials [Ensayos clínicos por conglomerados]. EVIDENCIA Actualizacion en la Practica Ambulatoria 2017;20(1):22-5. [Google Scholar]
Pinto 1995
- Pinto Dias JC. Natural history of Chagas disease [História natural da doenca de Chagas]. Arquivos Brasileiros de Cardiologia 1995;65:359-65. [PubMed] [Google Scholar]
Ramos 2012
- Ramos J, Torrús D, Amador C, Jover F, Pérez-Chacón F, Ponce Y, et al. Multicenter epidemiological and clinical study on imported Chagas diseases in Alicante, Spain. Pathogens and Global Health 2012;106(6):340-5. [DOI: 10.1179/2047773212Y.0000000039] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ramsey 2014
- Ramsey J, Elizondo-Cano M, Sanchez-González G, Peña-Nieves A, Figueroa-Lara A. Opportunity cost for early treatment of Chagas disease in Mexico. PLOS Neglected Tropical Diseases 2014;8(4):e2776. [DOI: 10.1371/journal.pntd.0002776] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rassi 2007
- Rassi A, Luquetti A, Rassi A Jr, Rassi G, Rassi S, Da Silva IG, et al. Specific treatment for Trypanosoma cruzi: lack of efficacy of allopurinol in the human chronic phase of Chagas disease. American Journal of Tropical Medicine and Hygiene 2007;76(1):58-61. [PMID: ] [PubMed] [Google Scholar]
Rassi 2010
- Rassi A Jr, Rassi A, Marin-Neto J. Chagas disease. Lancet 2010;375(9723):1388-402. [DOI: 10.1016/S0140-6736(10)60061-X] [DOI] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Robertson 2016
- Robertson L, Devleesschauwer B, Alarcón B, Noya González O, Torgerson P. Trypanosoma cruzi: time for international recognition as a foodborne parasite. PLOS Neglected Tropical Diseases 2016;10(6):e000465. [DOI: 10.1371/journal.pntd.0004656] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rodriguez‐Coura 2002
- Rodriguez-Coura J, Castro S. A critical review on Chagas disease chemotherapy. Memorias do Instituto Oswaldo Cruz 2002;97:3-24. [DOI: 10.1590/s0074-02762002000100001] [DOI] [PubMed] [Google Scholar]
Rojas 1999
- Rojas A, Ferro E, Ferreira M, Simancas L. Chagas disease vector control through different intervention modalities in endemic localities of Paraguay. Bulletin of the World Health Organization 1999;77:331-9. [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Sales 2017
- Sales P, Molina I, Fonseca S, Sánchez-Montalvá A, Salvador F, Corrêa-Olivera R, et al. Experimental and clinical treatment of Chagas disease: a review. American Journal of Tropical Medicine and Hygiene 2017;97(5):1289-303. [DOI: 10.4269/ajtmh.16-0761] [DOI] [PMC free article] [PubMed] [Google Scholar]
Salomao 2016
- Salomao K, Menna-Barreto R, Castro S. Stairway to heaven or hell? Perspectives and limitations of Chagas disease chemotherapy. Current Topics in Medicinal Chemistry 2016;16(20):2266-89. [DOI: 10.2174/1568026616666160413125049] [DOI] [PubMed] [Google Scholar]
Sbaraglini 2016
- Sbaraglini M, Vanrell M, Bellera C, Benaim G, Carrillo C, Talevi A, et al. Neglected tropical protozoan diseases: drug repositioning as a rational option. Current Topics in Medicinal Chemistry 2016;16(19):2201-22. [DOI: 10.2174/1568026616666160216154309] [DOI] [PubMed] [Google Scholar]
Schijman 2011
- Schijman A, Bisio M, Orellana L, Sued M, Duffy T, Mejia A, et al. International study to evaluate PCR methods for detection of Trypanosoma cruzi DNA in blood samples from Chagas disease patients. PLOS Neglected Tropical Diseases 2011;5(1):e931. [DOI: 10.1371/journal.pntd.0000931] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schmunis 1994
- Schmunis G. American trypanosomiasis as a public health problem. In: Chagas' Disease and the Nervous System. Washington, DC: Pan American Health Organization, World Health Organization, 1994:3-29. [Google Scholar]
Schmunis 1999
- Schmunis G. Prevention of transfusional Trypanosoma cruzi infection in Latin America. Memorias do Instituto Oswaldo Cruz 1999;94(Suppl 1):93-101. [DOI: 10.1590/s0074-02761999000700010] [DOI] [PubMed] [Google Scholar]
Schmuñis 2013
- Schmuñis G. Status of and cost of Chagas disease worldwide. Lancet Infectious Diseases 2013;13(4):283-4. [DOI: 10.1016/S1473-3099(13)70032-X] [DOI] [PubMed] [Google Scholar]
Schofield 1999
- Schofield C, Dias J. The southern cone initiative against Chagas disease. Advances in Parasitology 1999;42:1-27. [DOI: 10.1016/s0065-308x(08)60147-5] [DOI] [PubMed] [Google Scholar]
Schofield 2006
- Schofield C, Jannin J, Salvatella R. The future of Chagas disease control. Trends in Parasitology 2006;22(12):583-8. [DOI: 10.1016/j.pt.2006.09.011] [DOI] [PubMed] [Google Scholar]
Schunemann 2013
- Schünemann H, Brożek J, Guyatt G, Oxman A. GRADE handbook for grading quality of evidence and strength of recommendations.. The GRADE Working Group. 2013. [Available from guidelinedevelopment. org/handbook.]
Segura 1994
- Segura M, Molina E, Basombrio M. Reversibility of muscle and heart lesions in chronic, Trypanosoma cruzi infected mice after late trypanomicidal treatment. Memorias do Instituto Oswaldo Cruz 1994;89:213-6. [DOI: 10.1590/s0074-02761994000200017] [DOI] [PubMed] [Google Scholar]
Sosa 1998
- Sosa S, Segura E, Ruiz A, Velazquez E, Porcel B, Yampotis C. Efficacy of chemotherapy with benznidazole in children in the indeterminate phase of Chagas' disease. American Journal of Tropical Medicine and Hygiene 1998;59:526-9. [DOI: 10.4269/ajtmh.1998.59.526] [DOI] [PubMed] [Google Scholar]
Sterne 2000
- Sterne J, Gavaghan D, Egger M. Publication and related bias in meta-analysis: power of statistical tests and prevalence in the literature. Journal of Clinical Epidemiology 2000;53:1119–29. [DOI: 10.1016/s0895-4356(00)00242-0] [DOI] [PubMed] [Google Scholar]
Sterne 2001
- Sterne J, Matthias E, Smith G. Investigating and dealing with publication and other biases in meta-analysis. BMJ 2001;323:189-208. [DOI: 10.1136/bmj.323.7304.101 ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Stoppani 1999
- Stoppani A. The chemotherapy of Chagas disease [Quimioterapia de la enfermedad de Chagas]. Medicina 1999;59(Suppl 2):147-65. [PMID: ] [PubMed] [Google Scholar]
Tanowitz 1992
- Tanowitz H, Morris S, Factor S, Weiss L, Wittner M. Parasitic disease of the heart I: acute and chronic Chagas' disease. Cardiovascular Pathology 1992;1:7-15. [DOI: 10.1016/1054-8807(92)90004-8] [DOI] [PubMed] [Google Scholar]
Tarleton 2014
- Tarleton RL, Gürtler RE, Urbina JA, Ramsey J, Viotti R. Chagas disease and the London declaration on neglected tropical diseases. PLOS Neglected Tropical Diseases 2014;10(8):e3219. [DOI: 10.1371/journal.pntd.0003219] [DOI] [PMC free article] [PubMed] [Google Scholar]
Urbina 1999
- Urbina J. Parasitological cure of Chagas disease: is it possible? Is it relevant? Memorias do Instituto Oswaldo Cruz 1999;94(Suppl 1):349-55. [DOI: 10.1590/s0074-02761999000700068] [DOI] [PubMed] [Google Scholar]
Urbina 2009
- Urbina J. Ergosterol biosynthesis and drug development for Chagas disease. Memorias do Instituto Oswaldo Cruz 2009;104(Suppl 1):311-8. [DOI: 10.1590/s0074-02762009000900041] [DOI] [PubMed] [Google Scholar]
Urbina 2015
- Urbina J. Recent clinical trials for the etiological treatment of Chronic Chagas disease: advances, challenges and perspectives. Journal of Eukaryotic Microbiology 2015;62(1):149-56. [DOI: 10.1111/jeu.12184] [DOI] [PubMed] [Google Scholar]
Valerio‐Sallent 2012
- Valerio-Sallent L, Roure S, Basile L, Ballesteros L, Sabrià M, Rodrigo C, Grupo Metropolitano de estudio del Chagas. A clinical and epidemiological study of the Trypanosoma cruzi infected population in the north metropolitan area of Barcelona [Un estudio clínico-epidemiológico de la población infectada por Trypanosoma cruzi en la zona metropolitana norte de Barcelona]. Revista Clinica Espanola 2012;212(7):329-36. [DOI: 10.1016/j.rce.2012.03.017] [DOI] [PubMed] [Google Scholar]
Villar 2002
- Villar J, Villar L, Marin-Neto J, Ebrahim S, Yusuf S. Trypanocidal drugs for chronic asymptomatic Trypanosoma cruzi infection. Cochrane Database of Systematic Reviews 2002, Issue 1. Art. No: CD003463. [DOI: 10.1002/14651858.CD003463] [DOI] [PubMed] [Google Scholar]
Villar 2014
- Villar JC, Perez JG, Cortes OL, Riarte A, Pepper M, Marin-Neto JA, et al. Trypanocidal drugs for chronic symptomatic Trypanosoma cruzi infection. Cochrane Database of Systematic Reviews 2014, Issue 5. Art. No: CD003463. [DOI: 10.1002/14651858.CD003463.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 1991
- World Health Organization. Control of Chagas' disease. Report of a WHO Expert Committee. WHO Technical Report Series No. 811. Geneva: World Health Organization, 1991. [PubMed] [Google Scholar]
WHO 2015
- WHO/Department of control of neglected tropical diseases. Investing to overcome the global impact of neglected tropical diseases: Third WHO report on neglected tropical diseases 2015. Vol. 3. World Health Organization, 2015. [Google Scholar]
WHO 2017
- WHO/Department of Control of Neglected Tropical Diseases. Fourth WHO report on neglected tropical diseases. Vol. 1. Geneva: World Health Organization, 2017. [Google Scholar]
Yun 2009
- Yun O, Lima M, Ellman T, Chambi W, Castillo S, Flevaud L, et al. Feasibility, drug safety, and effectiveness of etiological treatment programs for Chagas disease in Honduras, Guatemala, and Bolivia: 10-year experience of Medecins Sans Frontières. PLOS Neglected Tropical Diseases 2009;3(7):e488. [DOI: 10.1371/journal.pntd.0000488] [DOI] [PMC free article] [PubMed] [Google Scholar]


