Abstract
The United States has faced an unprecedented opioid crisis in recent years, which has led to a rise in opioid overdose-related deaths and, consequently, an increase in the number of potential deceased donors available for transplantation. This new pool of potential organ donors is composed of younger donors with higher infectious disease transmission risk. Utilizing organs from these donors requires appropriate patient education, informed consent, and post-transplant monitoring practices. Prescription opioid use is also an important component of the evaluation of transplant and living donor candidates, as it may impact outcomes and eligibility for the procedures. In kidney transplant recipients, prescription opioid use predicts higher risk of mortality, graft loss and post-transplant complications. These effects seem to be proportional to the levels of opioid use, and to parallel patterns in other transplant populations such as liver and heart recipients. Among living kidney donors, pre-donation prescription opioid use is associated with increased risk of readmission after nephrectomy. Overall, the opioid epidemic creates educational needs for patients awaiting deceased donor transplant, and also impacts the evaluation of transplant candidates. Among transplant candidates and recipients, the identification of patients with chronic opioid use should prompt multi-disciplinary evaluation and management strategies to minimize risks.
Keywords: Kidney transplantation, Opioid analgesics, Pain, Drug misuse, Graft loss, Mortality
INTRODUCTION
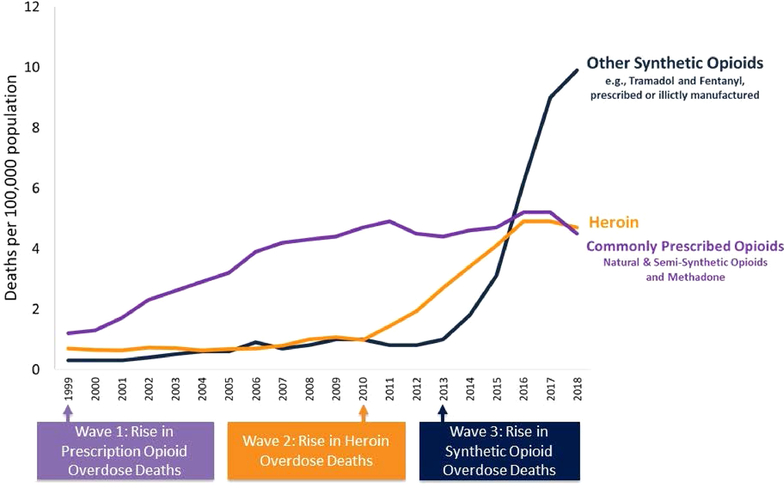
Opioid analgesic misuse has resulted in an international epidemic of opioid addiction and overdose-deaths that began to escalate in 2013, correlating with increased use of synthetic opioids [Figure 1] (1,2). The rise in opioid analgesics consumption is attributable to increased physicians’ prescriptions but also to illegal opioids like heroin and illicitly manufactured fentanyl (3). Illegally made fentanyl is sold through illicit drug markets for its heroin-like effect, and it is often mixed with heroin or other drugs, such as cocaine, or pressed into counterfeit prescription pills (4). In a study of 1064 kidney transplant recipients (2004–2008), the most commonly prescribed opioid before kidney transplantation was hydrocodone, followed by propoxyphene, oxycodone and tramadol (5). Indeed, opioid analgesics are used to manage acute but also chronic pain, and as such, became one of the most commonly prescribed classes of medications in the United States (6). Between 2013 and 2016, 6.5% of adults aged 20 years and over reported using a prescription opioid analgesic in the past 30 days (7). More than 191 million opioid prescriptions were dispensed to American patients in 2017 (8). The most common drugs involved in prescription opioid overdose deaths include methadone, oxycodone and hydrocodone (9).
Figure 1. Timeline of the three waves of opioid overdose-associated deaths.
The first wave involved an increased number of opioid prescriptions in the 1990s. The second wave was due to heroin-related overdoses. The third wave began in 2013 with the contribution of illicitly manufactured fentanyl that has caused an increase in overdose deaths involving synthetic opioids (2).
Patients with end-stage kidney disease (ESKD) can experience chronic pain from different sources, as the underlying cause of kidney disease (e.g. polycystic kidney disease, diabetic neuropathy), its complications (e.g. degenerating bones due to renal osteodystrophy, calciphylaxis), and the side effects of renal replacement therapies (e.g. muscle cramps and steal syndrome during hemodialysis) may all trigger pain (10,11). The World Health Organization (WHO) three-step analgesic ladder, which begins with non-opioid and adjunctive agents but progresses to include opioids for moderate and severe pain (12), may be used effectively in ESKD patients (13). Whether as a consequence of inadequate relief from non-opioid analgesics, concerns for toxicities of non-opioid agents (e.g., side effects of non-steroid anti-inflammatory agents or tramadol-related seizures), or failure to try the first step of the pain ladder, many ESKD patients receive prescription opioids for pain management.
Although successful transplantation might be expected to mitigate some underlying causes of pain in dialysis patients, the prevalence of pain was similar in a study comparing patients on hemodialysis (63%) with kidney transplanted patients (62%), although 63% of kidney transplant recipients reported taking analgesics compared to 85% in the hemodialysis group (14). Additionally, patients who filled opioid prescriptions for various reasons before kidney transplantation tend to continue use after transplant, well after the typical post-operative pain management period (15). With regard to outcome implications, patients with ESKD have an increased risk of opioid-related toxicity because renal impairment reduces their clearance and also increases their bioavailability (16). Medication interactions and additive medication side-effects are also a concern in transplant candidates and recipients.
This review focuses on the implications of the opioid epidemic for deceased organ donation, considerations for kidney transplant candidate selection, the impact of opioid-related deaths on the waitlist, and consequences after living donation and after transplant surgery.
DONORS
DECEASED DONORS
Over the last ten years, the number of transplants has increased by 39%, with 23,401 kidney transplants performed in the United States in 2019, compared with 16,829 in 2009. In 2019, deceased donors represented 70% of all U.S. kidney transplants (n=16,534). According to the Organ Procurement and Transplantation Network (OPTN), the number of deceased donors increased by 10.7% when compared to the previous year (17).
Impact of the opioid crisis on deceased donation
According to the Centers for Disease Control and Prevention (CDC), 47,600 opioid overdose deaths occurred in the United States in 2017. Comparatively, opioid-related deaths represent 67.8% of all drug overdose deaths (18). Between 2010 and 2016, the number of kidney donors who died from drug overdose increased by 277% (296 to 1,117 donors) (19). Deaths from drug overdoses represented 13.5% of the total adult kidney donor pool in 2016. An analysis of data from the Scientific Registry of Transplant Recipients (SRTR) in 2016 revealed that anoxic drug overdose donors accounted for 12.2% of the 2,212 kidney grafts from cardiac death (DCD) donors compared to 3.1% in 2006 (20). However, to the best of our knowledge, no previous study described the number of deaths related to opioid overdose compared to other drugs.
The opioid crisis correlated with a surge in deaths among young adults, which also affected the profile of deceased donors. Among all deceased donors, the proportion of young adults between 18–34 years old has increased by 43% from 2010 to 2016 (2,073 to 2,974) (19). Although donors with opioid-related death comprise a younger population with otherwise less comorbidities, physicians and patients still vary in their willingness to accept organs from these donors because of concerns related to risk of infection transmission and lack of appropriate education about the potential benefit-to-risk ratio.
The increase in number of opioid-related deaths contributed to more organs available for transplantation, but of course the rise in opioid overdose deaths requires emergent mobilization of policies and actions to halt this crisis. Other means to increase transplantation are needed, such as improved organ procurement practices, expanded use of non-standard organs including from older donors, and increased organ donation (22).
Quality of kidney grafts
This new pool of donors dying from an opioid overdose have unique donor characteristics that impact favorably the quality of the kidney grafts. According to a study by Durand et al. [Table 1], persons aged 21 to 40 years old comprised 66.3% of donors with overdose-related deaths, compared with 19.8% of donors with “medical-deaths” (e.g., deaths from intracranial hemorrhage, stroke, myocardial infarction, or natural causes) (23). Compared with “medical-death donors”, donors with opioid deaths were less likely to have hypertension (18% vs 49.1%), diabetes (4.6% vs 15.8%) or previous myocardial infarction (1.6% vs 5.7%). In 2016, the majority of drug overdose deceased donors had a kidney donor profile index (KDPI) lower than 35% (where KDPI is defined on a scale of 0 to 100%, with a lower score indicating better quality in terms of predicted graft survival) [Table 1] (19). Overall, this indicates that kidneys derived from opioid overdose donors are younger and have less comorbidities.
Table 1.
Summary of recent studies describing the frequency and outcomes related to opioid use in deceased donors and living donors.
| Deceased Donor Studies: Overdose Deaths, Organ Utilization and Recipient Outcomes | |||||
|---|---|---|---|---|---|
| Author Journal | Data source/design | Opioids use measure | Outcome measure | Findings | Limitations |
| (Year) | frequency | ||||
| Durand et al. Ann Intern Med (2018)23 | • SRTR • 138,565 deceased donors; 337,934 transplantrecipients at 297 centers (2000–2017) • Solid organ transplants • CDC: cause of death • Retrospective cohort |
• Donor mechanism of death: ODD, TDD or MDD • ODD = 7,313 with at least 1 organ recovered during the study • Organs from ODD were used in 19,897 solid organ transplants (10,347 kidneys) |
• 5-year patient survival • Death-censored 5-year graft survival • Organ discard |
• In 2017: - ODD = 13.4% of donors all organs - Prevalence for HCV among ODD = 30% - PHS IRD among ODD = 71.8% • Standardized 5-year patient survival rate = 83.1% for ODD kidney recipients • ODD kidney recipients had 3.1% lower patient survival compared with TDD kidney recipients • Standardized 5-year death-censored graft survival was similar between ODD and TDD kidneys (risk difference = 0.2%) • Kidney discard was higher for ODDs than TDDs (20% vs 14.9%); standardization for HCV and IRD status attenuated this difference (16.5% vs 16.1%) |
• Registry-based study: unmeasured confounders • No distinction between opioid vs nonopioid overdose • Did not determine the specific behavior for which ODDs were labelled as IRDs |
| Chute et al. Am J Nephrol (2018)19 | • OPTN registry • 48,891 deceased donors (2010–2016) • Retrospective cohort |
• Donor mechanism of death as “drug intoxication” • Drug overdose death = 1,117 donors (2016) 277% increase from 2010 |
• Changes in characteristics of KTx donors related to the opioid crisis | In 2016: • Drug overdose death = 13.5% of the total kidney donor pool • HCV infection = 19% of drug overdose deceased donors • HCV “infected” donors = 69% increase from 2010 • Age range 18–34 = 62% of drug overdose deceased donors • KDPI score < 35% = >50% of all drug overdose deceased donors |
• Registry-based study: unmeasured confounders • No distinction between opioid vs nonopioid overdose • No distinction between HCV exposure and HCV viremia |
| Richards et al. Ann Transplant (2018)29 | • Single center study: University of Washington, Seattle • Retrospective case series review: 82 PHS IRD kidney offers (2015–2017) |
• PHS IRD category "intravenous drug use" • 20 patients declined PHS IRD kidney offers |
• Outcomes of patients who declined vs. accepted PHS IRD offers • Effect of education on these patients’ willingness to consider another PHS IRD |
• 20 patients “declined PHS IRD offer” waited on average 9 months for another transplant with a higher-KDPI • 17/20 declined offers = PHS IRD due to intravenous drug • 92% of patients who received education on PHS IRD after declining an offer stated that they would consider another PHS IRD offer in the future |
• Retrospective review at a single institution • Effect of the education or the higher anticipation for transplant due to waiting longer |
| Living Kidney Donor Studies: Nephrectomy Outcomes | |||||
| Lentine et al. Am J Transplant (2017)45 | • Linkage of SRTR data for 14,959 living donors with pharmacy fill records and administrative records from an academic hospital consortium (2008–2012) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before donation • ME ranked by tertiles: - <130 mg/yr - 130 to 305 mg/yr - >305 mg/yr • 11.3% had at least 1 opioid prescription in the year before donation |
• Primary outcome = hospital readmission within 90 d after nephrectomy • Associations of pre-donation opioid use and other baseline clinical, procedural, and center factors with readmission within 90 d post-donation |
• Overall readmission rate: 2.9% • Donors with the highest level pre-donation opioid use were more than twice as likely as were nonusers to be readmitted (aOR 2.49) • Readmission risk was also significantly higher for: - Women (aOR, 1.25) - African Americans (aOR,1.45) - Spouses (aOR, 1.42) - Exchange participants (aOR, 1.46) - Uninsured donors (aOR, 1.40) - Donors with pre-donation eGFR < 60mL/min per1.73m2 (aOR, 2.68) - Donors with pre-donation pulmonary conditions (aOR, 1.54) - After robotic nephrectomy (aOR, 1.68) • Leading readmission diagnosis included infection and gastro-intestinal complaints |
• Registry-based study: unmeasured confounders (including patient behaviors) • Sample may not be representative of all U.S. donors • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies • Readmission was limited to transplant centers |
ACGF, All cause graft failure; aHR, adjusted hazard ratio; aOR, adjusted odds ratio; CDC, Centers for Disease Control and Prevention; eGFR, estimated Glomerular Filtration Rate; ERAS, enhanced recovery after surgery pathway; HCV, hepatitis C virus; IRD, Increased Risk Donors; KDPI, Kidney Donor Profile index; KTx, kidney transplantation;; MDD, medical-death donor; ME, morphine equivalents; ODD, overdose-death donor; OPTN, Organ Procurement and Transplantation Network; PHS, Public Health Service; SHS, Symphony Health Solutions; SOC, Standard of care; SRTR, Scientific Registry of Transplant Recipients; TDD, trauma-death donor
However, these kidneys have a higher risk of infectious transmissions. The prevalence of hepatitis C virus (HCV) positive donors was 4.5-fold higher for overdose-death donors compared to donors with “medical-deaths” (18.3% vs 4.1%) (20). In the era of highly effective direct-acting antiviral agents (DAA), transplantation of HCV-infected kidneys to uninfected recipients followed by early treatment can have successful outcomes, with cure of HCV infection (25,26). In a single-center study of kidney transplantation from HCV-infected donors, all of the 20 participants achieved HCV clearance with excellent renal function as measured by serum creatinine at 6 months (1.0 to 1.3 mg/dL or 90 to 118 μmol/L) and 12 months (1.0 to 1.3 mg/dL or 84 to 111 μmol/L) after transplant (27). Moreover, the percentage of Increased Risk Donors (IRD) among overdose-death donors was 71.8% in the study by Durand et al. (23). This is not surprising, as IRD as defined by U.S. Public Health Services (PHS) criteria was associated with increased risk of HCV, Hepatitis B and HIV infection, which includes intravenous drug use (24). In sum, it is critical that risk is recognized so that the recipient can provide informed consent, be appropriately monitored for viremia after transplant, and have access to DAA treatment (28).
In a national analysis of transplants from 2000 to 2017, the 5-year patient survival rate for recipients of kidneys from overdose-death donors was 83.1% compared to 86.2% and 81% for recipients from trauma-death donors and medical-death donors, respectively (23). Regarding death-censored graft survival, at 5-years, 87.1% of recipients from overdose-death donors still had functioning allografts, versus 86.9% and 82.2% of recipients from trauma-death and medical-death donors, respectively (23).
The standardized risk of discard for overdose-death organs was 5.2-times that of trauma-death organs (20% vs 14.9%, respectively). However, this risk disappeared after adjustment for HCV and IRD status. No difference was observed between overdose-death kidneys and medical-death kidneys for the risk of organ discard (23). In a single center retrospective case series of 82 IRD kidney offers [Table 1], 20 patients declined the IRD offer and chose to wait for another offer (29). Among them, 16 patients underwent kidney transplantation after an average wait of 9 months, and received an organ with a higher KDPI compared to the declined IRD offer; 6 patients received a transplant from another IRD. In the same study, the authors assessed the influence of education on attitudes toward IRD transplantation. Ninety two percent of patients who declined an IRD kidney offer would consider a new offer after discussion, reinforcing the importance of educating patients on the waiting list accurately about the risks and benefits of IRD kidneys.
From an organ quality perspective, donors with opioid-related deaths seem to be good sources for transplantation that may be used safely with appropriate patient consent, monitoring and management. Benefits are greatest when IRD organ acceptance substantially reduces waiting time on dialysis (30,31).
LIVING DONORS
Living kidney donation can provide the ESKD patient with the best chance of long-term dialysis-free survival (32). Living donation is an act of profound human generosity, but it is not without risks. Therefore, living donor candidates must undergo a careful physical and psychological assessment to determine suitability to donate from the perspective of their own health and well-being (33).
Assessment of opioid use in the donor candidate evaluation
The goals of living donor candidate evaluation are to protect donor safety but also avoid unnecessary exclusions, based on careful assessment of medical and psychosocial suitability. In the United States, OPTN Policy requires that the psychosocial evaluation include a review of the living donor’s history of smoking, alcohol, and drug use, abuse, and dependency (34). Details of candidacy determination are generally left to the policies and judgement of the individual transplant center, although OPTN Policy mandates exclusion of donors with uncontrolled diagnosable psychiatric conditions requiring treatment before donation, which can include significant substance use disorders. Such conditions could impact the ability to make an informed donation decision and to participate in self-care, health-promoting lifestyle habits, and follow-up after donation (35). The 2017 Kidney Disease: Improving Global Outcomes (KDIGO) guideline on the evaluation and care of living kidney donors recommends review of current or past history of illicit/prescribed drug abuse or dependence in the psychosocial evaluation of donor candidates (33).
With regard to opioid use patterns in the general population, some of whom may present for living donor evaluation, a Danish study used a national health survey to analyze a sample representative of their general population, and found an increased risk of addictive behaviors in opioid users with pain compared to non-opioid users with pain (36). Interestingly, they did not find any association between problematic use of alcohol and use of opioids in patients with chronic pain (odds ratio [OR] 1.10; 95% CI 0.73–1.65 for long-term opioid users; OR 1.03; 95% CI 0.79–1.34 for short-term opioid users). However, individuals with chronic pain and long-term opioid use showed a 2.6-fold higher likelihood of cannabis use and also a 12-times higher odds of long-term use of benzodiazepines (OR 12.47; 95% CI 6.92–22.45) than individuals with chronic pain not using opioid medications (36). Another study provided data on the association of psychiatric comorbidity with the prescription of regular opioid medications (37). The presence of a common mental disorder (depression, dysthymia, generalized anxiety disorder or panic disorder) was a significant predictor of opioid prescription amongst individuals with low pain interference (OR 3.15; p<0.001).
Duerincks et al. summarized surveys and tools used for psychometric testing in the donor candidate evaluation from prior guidelines, consensus statements and transplant program protocols such as drug abuse questionnaires (38). The only test they identified to assess substance abuse amongst liver and kidney living donor candidates is the Drug Abuse Screening Test (DAST). The DAST is a 28-item self-report measure of problematic substance use other than alcohol that is utilized for clinical screening. A review of the psychometric properties of the DAST has shown moderate to high levels of validity, sensitivity (from 80.9% to 96% depending on 4 different studies), and specificity (ranged from 71% to 93.9%) (39–42); therefore DAST could have the appropriate reliability and validity needed for a clinical screening test (43). Of course, as a self-report test, DAST may be impacted by respondent bias regarding their actual substance use.
Specific instruments exist to monitor opioid efficacy, safety and misuse for patients with chronic pain (44), but data assessing these tests amongst living kidney donors are lacking at this time. There is a need to develop evidence-based guidance regarding how to measure opioid use in living donor candidates, which candidates warrant screening for opioid dependency and further evaluation, and how the information should be applied to candidacy determination.
Predonation opioid use and postdonation outcomes
To date, few studies have investigated relationships of pre-donation opioid use with post-donation outcomes (45). In a study integrating U.S. living donor registry data, records from a nationwide pharmacy claims warehouse, and administrative records from an academic hospital consortium for 14,959 living donors (2008 to 2012), the authors measured the readmission rate after nephrectomy surgery as the primary outcome [Table 1] (45). Overall, 2.9% of donors were readmitted to the hospital within 90 days of donation. Eleven percent of donors filled one or more opioid prescriptions in the year before donation, and those with the highest level of pre-donation opioid use were more than twice as likely as nonusers to be readmitted within 90 days post-donation (6.8% vs 2.6%; adjusted odds ratio [aOR], 2.49; 95% CI 1.74 to 3.58). Adjusted readmission risk was also significantly higher for women (aOR 1.25; p<0.05), African-Americans (aOR 1.45), spouses (aOR 1.42), exchange participants (aOR 1.46), uninsured donors (aOR 1.40), donors with pre-donation eGFR <60 mL/min/1.73 m2 (aOR 2.68), donors with pre-donation pulmonary disease (aOR 1.54), and after robotic nephrectomy (aOR 1.68). Leading readmission diagnoses included infection and gastro-intestinal complaints (45).
Among all these risk factors for readmission to hospital within 90 days post-donation, pre-donation opioid use was the strongest risk factor. It is also notable that the risk for readmission was present starting at level 2 of opioid use (130 to 350 mg/year morphine equivalents) and that this risk increased with the level of opioid use. To improve risk assessment counseling and selection, future studies should assess if pre-donation opioid use is a risk factor for other adverse outcomes in living kidney donors, and whether interventions to modify use can mitigate risk.
RECIPIENTS
CHRONIC OPIOID USE AND TRANSPLANT CANDIDACY
Candidates for transplant also undergo careful evaluation including assessment of comorbid conditions, overall fitness for surgery, and psychosocial status including risk factors for poor adherence after transplant (46,47). Some instruments and tools exist to assess opioid misuse among patients using opioid therapy for chronic pain: the Current Opioid Misuse Measure (COMM), the Prescription Drug Use Questionnaire-patient version (PDUQ-p), the modified Pain Medication Questionnaire (mPMQ), and the Prescription Opioid Misuse Index (POMI) (44). Since patients may under-report opioid use due to fear of not being considered an acceptable candidate, it might be important to obtain collateral or corroborating information from independent sources, which may include toxicology screening, pharmacy records and/or opioid prescription registries, and other medical records (47).
With regard to evaluation and candidacy determination, the 2020 KDIGO transplant candidate guideline includes several recommendations related to substance use and transplantation (47), including a recommendation to not refer patients for transplant evaluation who have an active or ongoing substance use disorder that affects decision-making or poses a level of post-transplant risk that is higher than acceptable to the program. The guideline recommends referring candidates with a substance use disorder for pre-transplant counseling and services, but not transplanting patients with ongoing substance use disorders at higher than “acceptable risk.” While consensus on threshold of acceptable risk requires more data, and risk tolerance can vary by center, it is important to assess before transplant the likelihood that the potential recipient will be adherent to medications, especially immunosuppressants, and follow-up care to enhance the likelihood of favorable post-transplant outcomes (48). Moreover, opioid use disorder can be associated with addictive behaviors and mental disorders that can affect the decision-making process and compliance of patients after transplant (36,37,47).
Ensuring the patient with chronic pain on opioids is following with an expert pain provider, that the patient is on a stable dose of opioids, and no prescriptions are received from other providers are crucial in the preparation for kidney transplantation. Patients with a history of pre-transplant opioid use who are selected for transplant may benefit from ongoing education, support and post-transplant surveillance.
OUTCOMES AFTER TRANSPLANTATION
Death and graft failure
Recent studies have examined associations of prescription opioid use with mortality and graft failure after kidney transplantation [Table 2]. In a U.S. single-center medical record-based study (n=1,064 kidney transplant recipients between 2004 and 2008), 10.2% had chronic opioid use as ascertained by multidisciplinary evaluation at the initial candidate assessment (5). Chronic opioid use before transplantation was associated with 65% higher adjusted risk of death over 7 years post-transplantation (HR 1.65, 95% CI 1.04–2.60) but was not associated with death-censored kidney graft loss or hospital readmission [Table 2].
Table 2.
Summary of recent studies describing the frequency and outcomes related to opioid use in kidney transplant recipients.
| Author Journal (Year) | Data source/design | Opioids use measure & frequency | Outcome measure | Findings | Limitations |
|---|---|---|---|---|---|
| Barrantes et al. Kidney Int (2013)5 |
• Single center data • 1,064 KTx patients (2004-2010) • Retrospective cohort |
• Analgesics were coded as opioids and nonopioids • COU = daily regular use for at least 3 months before KTx • 108 (10.2%) of recipients reported a prior history of chronic opioid use |
• Primary outcome = composite endpoint: - Overall death - Kidney graft loss • Secondary outcomes: - Death - Death-censored kidney graft loss - Hospital readmissions - Causes of death |
• Most commonly used opioid analgesics: - Hydrocodone (43.1%) - Propoxyphene (18.1%) - Oxycodone (16.4%) - Tramadol (13.8%) • After regression, no difference in composite endpoint: overall death and kidney graft loss (HR = 1.39, p=0.19) • A history of COU was independently associated with 1.65-fold increase in the risk for death • No difference in: - Death-censored kidney graft loss (HR,0.96, p=0.910) - Hospital readmission (aOR,1.03, p=0.884) |
• Retrospective study at a single institution • Opioid use was self-reported • Continued use of opioids after KTx was not assessed |
| Lentine et al. Transplantation (2015)49 | • Linkage OPTN data for 31,197 KTx recipients with pharmacy fill records (2005-2010) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before KTx • ME ranked by quartiles: - 0.1 to 1.7 mg/kg - 1.8 to 5.4 mg/kg - 5.5 to 23.7 mg/kg - >23.8 mg/kg • 28.3% filled opioid prescriptions in the year before KTx |
• Adjusted associations of ME levels with post-KTx 3-yr: - Graft survival - Patient survival |
• Risk of post-KTx death was 1.33 higher for quartile 3 and 1.63 for quartile 4 of pre-KTx ME use • All cause graft loss was 1.25 higher for quartile 3 and 1.41 for quartile 4 of pre-KTx ME use • Patterns were similar when considered by donor type |
• Registry-based study: unmeasured confounders (including patient behaviors) • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies • Continued use of opioids after KTx was not assessed |
| Lentine et al. Am J Transplant (2018)15 | • Linkage SRTR data for 75 430 KTx recipients with pharmacy fill records (2005-2015) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before and after KTx • ME ranked by quartiles: - >0 to 300 mg/year - 301 to 600 mg/year - 601 to 1000 mg/year - >1000 mg/year • 43.1% filled opioid prescription in the year before KTx |
• Primary outcome = composite endpoint: - All-cause mortality - Graft failure • In the analysis of pre-KTx opioid use, outcomes were assessed at 1-yr post-KTx • In the analysis of post-KTx opioid use, outcomes were assessed >1 to 2 yrs post-KTx |
• 70.5% of recipients who filled an opioid prescription in the year before KTx continued to fill opioid prescriptions in the year after • After adjustment, level 4 post-KTx opioid use was significantly associated with: - aHR = 2.24 for the risk of death - aHR = 1.35 for death censored graft failure - aHR = 1.68 for all cause graft failure • Increased risk of mortality and death-censored graft failure persisted over 5 years (aHR,1.67, p<0.0001; aHR,1.13, p=0.008 respectively) |
• Registry-based study: unmeasured confounders (including patient behaviors) • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies • Lack of information on laboratory values and KTx biopsy pathology results • Patients with drug dependency, addiction issues or noncompliance were not identified |
| Lentine et al. Am J Nephrol (2015)50 | • Linkage OPTN data for 16,322 KTx recipients with pharmacy ill records and Medicare billing claims (2006–2010) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before KTx • ME ranked by quartiles: - 0.1 to 1.7 mg/kg - 1.8 to 5.4 mg/kg - 5.5 to 23.7 mg/kg - >23.8 mg/kg • 28.3% filled opioid prescriptions in the year before KTx |
• Adjusted associations of ME levels with 3-year post-KTx clinical complications* | • After adjustment, level 4 pre-KTx morphine equivalents use compare with non-users: - aHR = 3.92 for post-KTx ventricular arrhythmias - aHR = 2.03 for mental status changes - aHR = 3.17 for drug abuse/dependence - aHR = 3.09 for accidents |
• Registry-based study: unmeasured confounders (including patient behaviors) • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies • Risk of diagnosis misclassification • Risk of ascertainment bias (recipients with a known history of chronic opioid use may be more likely to be diagnosed with related adverse events than non-users) |
aHR, adjusted hazard ratio; COU, chronic opioid use; HR, hazard ratio; KTx, kidney transplantation; ME, morphine equivalents; OPTN, Organ Procurement and Transplantation Network; SRTR, Scientific Registry of Transplant Recipients; clinical complications*, cardiac arrest, aspiration pneumonia, mental status changes, drug abuse/dependence, alcohol abuse, non-compliance
Self-reported opioid use may differ from actual use and suffer from under-reporting. Pharmacy fill records can offer a nonobtrusive measure of medication use that does not rely on self-report. Based on linkage of records from the U.S. transplant registry and a large pharmaceutical claims data clearinghouse (n=31,197 recipients between 2005 and 2010), 29% of kidney transplant recipients filled opioid prescriptions in the year before transplant [Table 2] (49). Compared with transplant recipients who did not use prescription opioids before transplant, those who used high levels of opioids were more commonly women, of white race, not working, publicly insured, and less likely to be college-educated. Patients who received high levels of opioids before transplant also had longer duration of pre-transplant ESKD, higher burden of comorbidities, and higher rates of sensitization.
After adjustment, the risk of post-transplant death and all-cause graft loss increased with higher level of pre-transplant opioid use. Compared to those without opioid use, the post-transplant risk of death was higher for those in quartiles 3 (adjusted hazard ratio [aHR] 1.33; 95% CI 1.15–1.54) and 4 (aHR 1.63; 95% CI 1.40–1.88) of pre-transplant opioid use. Similarly, the risk of all-cause graft loss rose significantly with the degree of pre-transplant prescription opioid exposure, with a 41% higher relative risk for recipients in the quartile with the highest use (aHR 1.41; 95% CI 1.26–1.59) compared to those without opioid use. The results were similar in living donor and deceased donor transplant recipients.
Patients who use prescription opioids before kidney transplant might continue to use opioids for longer than the typical post-operative pain management period. In a retrospective study, 70.5% of kidney transplant recipients who filled an opioid prescription in the year before transplant continued to fill opioid prescriptions in the year after transplant (15). In the first to second post-transplant year period, the risks of death, death-censored graft failure, and all-cause graft failure were increased for recipients with level 2 to 4 opioid use in the first post-transplant year compared with non-users (15). After adjustment for demographic, clinical, and procedure factors, level 4 post-transplant opioid use was associated with a two-fold increased risk of death over the next post-transplant year (aHR, 2.24; 95% CI 1.93–2.60), 35% increased risk of death-censored graft failure (aHR, 1.35; 95% CI 1.15–1.59), and 68% increased risk of all-cause graft failure (aHR, 1.68; 95% CI, 1.50–1.89). These risk relationships persisted when the follow-up time was extended to the 5-year post-transplant period.
Clinical complications
Limited data are available on the clinical complications that may mediate associations of opioid use with death and graft loss after kidney transplantation. One study integrating a U.S. national transplant registry (OPTN), pharmacy records from a nationwide pharmacy claims warehouse, and administrative billing claims from Medicare (n=16,322 kidney transplant recipients between 2006 and 2010) assessed whether pre-transplant prescription opioid fills predict post-transplant clinical complications (50). Twenty eight percent of recipients in the study filled an opioid prescription in the year before transplant. Patients with pre-transplant opioid fills were categorized into four levels of annual morphine equivalent use [Table 2]. The goal of this study was to examine the association of pre-transplant prescription opioid use with cardiovascular, respiratory, and neurological clinical complications, as well as substance abuse, accidents and non-compliance events at 3 years post-transplantation, as defined by medical diagnostic codes on billing claims. Compared with no use, the highest level of pre-transplant opioid use was significantly associated with post-transplant ventricular arrhythmias (aHR 3.92; 95% CI 1.74–8.84), mental status changes (aHR 2.03; 95% CI 1.46–2.83), drug abuse/dependence (aHR 3.17; 95% CI 2.35–4.28), alcohol abuse (aHR 2.41; 95% CI 1.38–4.20), and accidents (aHR 3.09; 95% CI 1.28–7.45) [Table 2] (41). These poor post-transplant outcomes are additional reasons to practice attentive post-transplant surveillance amongst recipients with a history of pre-transplant prescription opioid use, especially as such complications may mediate the pathway to allograft loss and death.
OPIOIDS AND OTHERS SOLID ORGAN TRANSPLANTS
Similar associations of prescription opioid use with poor post-transplant outcomes have been observed in other solid organ transplant populations. Among potential liver recipients with cirrhosis, opioid use is common, in part related to the lack of alternative analgesic options, especially for moderate to severe pain (51,52). Liver diseases are a risk factor for side effects related to overdose of opioids such as respiratory and central nervous system depression (53). After linking a national transplant registry with pharmacy claims data (n=29,673 liver transplant recipients between 2007–2014), Randall et al. found that 9.3% of the recipients filled opioid prescriptions while on the waiting list [Table 3] (54). Over 5 years, compared to those without opioid use on the waiting list, the risk of death and all-cause graft loss was increased by 52% for those with level 3 pre-transplant use (aHR 1.52; 95% CI 1.16–1.98) and 42% for the highest opioid use group (aHR 1.42; 95% CI 1.10–1.84). Opioid use in the year after transplantation was associated with similar risks of subsequent graft loss and mortality [Table 3].
Table 3.
Summary of recent studies describing the frequency and outcomes related to opioid use in recipients of other solid organ transplants.
| Author Journal (Year) | Data source/design | Opioids use measure & frequency | Outcome measure | Findings | Limitations |
|---|---|---|---|---|---|
| Lentine et al. Am J Transplant (2019)56 | • Linkage SRTR data for 13,958 HTx recipients with pharmacy fill records (2007-2016) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before and after HTx • ME ranked by quartiles: - >0 to 300 mg/year - 301 to 600 mg/year - 601 to 1000 mg/year - >1000 mg/year • 5,583 (40%) filled opioid prescriptions in the year before HTx |
• Primary outcome = - All-cause mortality - Graft loss |
• After adjustment, level 4 pre-HTx opioid use was significantly associated with: - aHR = 1.33 risk of death within the 1st year post-transplant - aHR = 1.27 risk of graft loss • After adjustment, level 4 post-HTx opioid use was significantly associated with: - aHR = 1.70 risk of death from >1 to 5 years post-transplant - aHR = 1.55 risk of graft loss |
• Registry based study: unmeasured confounders (including patient behaviors) • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies • Lack of information for causes of death and graft failure |
| Randall et al. Liver Transplant (2017)54 | • Linkage SRTR data for 29,673 LTx recipients with pharmacy fill records (2007-2014) • Retrospective cohort |
• Pharmacy fill data for opioid analgesics in the year before and after LTx • ME ranked by quartiles: - 0 to 2 mg/day - >2 to 10 mg/day - >10 to 70 mg/day - >70 mg/day • 9.3% filled opioid prescriptions while on the waiting list |
• Primary outcomes = - All cause mortality - Graft failure |
• After adjustment, recipients at level 4 of pre-transplant opioid use was significantly associated with: - aHR = 1.52 for risk of death - aHR = 1.42 for all-cause graft loss • Level 2 or higher opioid use in the 1st year after liver transplant was associated with 40–50% higher risk of death and graft failure over >1–5 years: - Risk of death for level 4 (aHR=1.53) - Graft failure for level 4 (aHR=1.55) |
• Registry based study: unmeasured confounders (including patient behaviors like alcohol dependence) • Missing illicit drug use or “pharmacy shopping” behaviors or prescription fills at other pharmacies |
| Cron et al. Transplantation (2020)55 | • Truven Health MarketScan® Research Databases (commercial insurance claims) • 1,257 LTx between 2009-2015 • Retrospective study |
• Classification of preoperative opioid use: - Opioid-naïve - Chronic users - Intermittent users • 45% of patients filled ≥ 1 opioid prescription in the year before transplant: - 35% intermittent use - 10% chronic |
• Primary outcome = prevalence of opioid fills over the year before and after LTx • Secondary outcome = evaluate changes in the duration of opioid use post-LTx (persistent or increased) |
• Preoperative intermittent users = 71% remained intermittent users after LTx • Preoperative chronic users = 73% remained chronic users after LTx • Pre-LTx opioid use was independently associated with risk of persistent or increased post-LTx opioid use |
• Commercially-insured sample captures only a portion of LTx performed in the United States • Opioid dependency can be an exclusion criterion for LTx for some centers • Indications and complications related to opioid use were not assessed |
aHR, adjusted hazard ratio; HTx, heart transplantation; LTx, liver transplant; OPTN, Organ Procurement and Transplantation Network; SHS, Symphony Health Solutions; SRTS, Scientific Registry of Transplant Recipients
Using a national dataset of employer-based insurance claims, Cron et al. identified 1,257 adults who underwent liver transplantation between 2009 and 2015 (55). Patients were categorized based on their duration of opioid fills during the year before and after transplant admission as opioid-naïve/no fills, chronic opioid use (≥120 days supply), and intermittent use (all other use). In this study population, oxycodone was the most common opioid prescribed before and after liver transplant. Forty-five percent of patients filled more than one opioid prescription in the year before transplant (35% intermittent use and 10% chronic); 61% of patients filled an opioid prescription 0 to 2 months after discharge, and 19 to 25% filled between 2- and 12-months post-discharge. Pre-transplant opioid use was independently associated with increased risk of persistent or increased post-transplant opioid use [Table 3] (47).
Regarding heart transplant recipients, risk relationships for prescription opioid use are similar to observations for other solid organ transplant recipients. Forty percent of heart transplant recipients (n=13,958 between 2007–2016) in a study linking national U.S. registry and pharmacy fill records filled at least one opioid prescription in the year before the transplant (13% of doses represented the highest level of morphine equivalents >1000 mg/year) [Table 3] (56). Highest‐level use predicted 33% increased risk of death in the year after transplant (aHR 1.33; 95% CI 1.10–1.61). Of those with the highest level of pre-transplant opioid use, 71% continued to use opioids after heart transplant. Opioid use level in the first year post‐heart transplant was associated with a graded increase in subsequent mortality (20%, 30%, 50%, 60%) compared to non‐use over >1 to 5 years post‐heart transplant.
CONCLUSION
The rise in opioid-overdose related deaths has contributed to an increased number of organs available for patients in need of transplantation. Despite the risk of viral infection transmission from a donor after opioid-overdose related death, accepting these kidneys from these donors provides a survival benefit over chronic dialysis for many patients awaiting transplantation. Utilizing organs from these donors requires appropriate patient education, informed consent, and post-transplant monitoring practices.
Regarding prescription opioid use in the transplant population, what is known at this time is relevant to prognosis and risk assessment. Preoperative pharmacy fills for prescription opioids are associated with: increased risk of death, graft loss and clinical complications after kidney transplant; death after liver transplant and heart transplant; and readmission after living donation. Importantly, opioid use is also potentially modifiable. However, what remains unknown is substantial, including the implications of prescription opioid use in those who were never listed or transplanted, indication for the opioid prescription, and interaction of opioid use with other substances in determining risk. Presently, we believe appropriate practice includes offering focused assessment and support related to prescription opioids at evaluation, but also beyond as part of routine post-transplant care. This assessment may need to include more than self-report. More studies are needed to improve prognostication for the population and the individual patient, along with more studies on how to reduce prescription opioid use, and whether abstinence can mitigate risk. Overall, opioid use can have a significant impact on both the organ donor and transplant recipient. Careful individualized assessment before and after transplant is essential to ensure patient safety and the best clinical outcomes.
Acknowledgments
Financial disclosure and conflict of interest statements:
KLL receives support from the Mid-America Transplant/Jane A. Beckman Endowed Chair in Transplantation.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Understanding the Epidemic | Drug Overdose | CDC Injury Center [Internet]. 2019. [cited 2020 Feb 26]. Available from: https://www.cdc.gov/drugoverdose/epidemic/index.html
- 2.2018–3-Wave-Lines-Mortality.png (960×720) [Internet]. [cited 2020 Mar 20]. Available from: https://www.cdc.gov/drugoverdose/images/epidemic/2018-3-Wave-Lines-Mortality.png
- 3.Shei A, Rice JB, Kirson NY, Bodnar K, Birnbaum HG, Holly P, et al. Sources of prescription opioids among diagnosed opioid abusers. Current Medical Research and Opinion. 2015. April 3;31(4):779–84. [DOI] [PubMed] [Google Scholar]
- 4.Commonly Used Terms | Drug Overdose | CDC Injury Center [Internet]. 2019. [cited 2020 Mar 11]. Available from: https://www.cdc.gov/drugoverdose/opioids/terms.html
- 5.Barrantes F, Luan FL, Kommareddi M, Alazem K, Yaqub T, Roth RS, et al. A history of chronic opioid usage prior to kidney transplantation may be associated with increased mortality risk. Kidney International. 2013. August;84(2):390–6. [DOI] [PubMed] [Google Scholar]
- 6.FastStats [Internet]. 2020. [cited 2020 Mar 1]. Available from: https://www.cdc.gov/nchs/fastats/drug-use-therapeutic.htm
- 7.Scholl L, Seth P, Kariisa M, Wilson N, Baldwin G. Drug and Opioid-Involved Overdose Deaths — United States, 2013–2017. MMWR Morb Mortal Wkly Rep [Internet]. 2018. December 21 [cited 2020 Feb 20];67(5152). Available from: http://www.cdc.gov/mmwr/volumes/67/wr/mm675152e1.htm?s_cid=mm675152e1_w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. 2018-cdc-drug-surveillance-report.pdf.
- 9.Prescription Opioids | Drug Overdose | CDC Injury Center [Internet]. 2019. [cited 2020 Mar 11]. Available from: https://www.cdc.gov/drugoverdose/opioids/prescribed.html
- 10.Weisbord SD, Fried LF, Arnold RM, Fine MJ, Levenson DJ, Peterson RA, et al. Prevalence, Severity, and Importance of Physical and Emotional Symptoms in Chronic Hemodialysis Patients. JASN. 2005. August;16(8):2487–94. [DOI] [PubMed] [Google Scholar]
- 11.Davison SN. The Prevalence and Management of Chronic Pain in End-Stage Renal Disease. Journal of Palliative Medicine. 2007. December;10(6):1277–87. [DOI] [PubMed] [Google Scholar]
- 12.WHO | WHO’s cancer pain ladder for adults *Internet+. WHO. World Health Organization; *cited 2020 Mar 26+. Available from: https://www.who.int/cancer/palliative/painladder/en/ [Google Scholar]
- 13.Barakzoy AS, Moss AH. Efficacy of the World Health Organization Analgesic Ladder to Treat Pain in End-Stage Renal Disease. JASN. 2006. November;17(11):3198–203. [DOI] [PubMed] [Google Scholar]
- 14.Masajtis-Zagajewska A, Pietrasik P, Krawczyk J, Krakowska M, Jarzębski T, Pietrasiewicz B, et al. Similar prevalence but different characteristics of pain in kidney transplant recipients and chronic hemodialysis patients: Pain characteristics in dialysis and kidney transplant patients. Clinical Transplantation. 2011. March;25(2):E144–51. [DOI] [PubMed] [Google Scholar]
- 15.Lentine KL, Lam NN, Naik AS, Axelrod DA, Zhang Z, Dharnidharka VR, et al. Prescription opioid use before and after kidney transplant: Implications for posttransplant outcomes. Am J Transplant. 2018. December;18(12):2987–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dean M Opioids in renal failure and dialysis patients. Journal of Pain and Symptom Management. 2004. November;28(5):497–504. [DOI] [PubMed] [Google Scholar]
- 17.National Data - OPTN [Internet]. [cited 2020 Jan 29]. Available from: https://optn.transplant.hrsa.gov/data/view-data-reports/national-data/#
- 18.Drug Overdose Deaths | Drug Overdose | CDC Injury Center [Internet]. 2019. [cited 2020 Jan 24]. Available from: https://www.cdc.gov/drugoverdose/data/statedeaths.html
- 19.Chute DF, Sise ME. Effect of the Opioid Crisis on the Donor Pool for Kidney Transplantation: An Analysis of National Kidney Deceased Donor Trends from 2010–2016. Am J Nephrol. 2018;47(2):84–93. [DOI] [PubMed] [Google Scholar]
- 20.Wanis KN, Madenci AL, Dokus MK, Tomiyama K, Al-Judaibi BM, Hernán MA, et al. The Effect of the Opioid Epidemic on Donation After Circulatory Death Transplantation Outcomes: Transplantation. 2019. May;103(5):973–9. [DOI] [PubMed] [Google Scholar]
- 21.Maghen A, Mone TD, Veale J. The Kidney-Transplant Waiting List and the Opioid Crisis. The New England Journal of Medicine. 2019;2. [DOI] [PubMed] [Google Scholar]
- 22.Advancing American Kidney Health. :38.
- 23.Durand CM, Bowring MG, Thomas AG, Kucirka LM, Massie AB, Cameron A, et al. The Drug Overdose Epidemic and Deceased-Donor Transplantation in the United States: A National Registry Study. Ann Intern Med. 2018. May 15;168(10):702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Seem DL, Lee I, Umscheid CA, Kuehnert MJ. PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis B Virus, and Hepatitis C Virus Transmission through Organ Transplantation. Public Health Rep. 2013. July;128(4):247–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sawinski D, Kaur N, Ajeti A, Trofe-Clark J, Lim M, Bleicher M, et al. Successful Treatment of Hepatitis C in Renal Transplant Recipients With Direct-Acting Antiviral Agents. Am J Transplant. 2016. May;16(5):1588–95. [DOI] [PubMed] [Google Scholar]
- 26.Levitsky J, Formica RN, Bloom RD, Charlton M, Curry M, Friedewald J, et al. The American Society of Transplantation Consensus Conference on the Use of Hepatitis C Viremic Donors in Solid Organ Transplantation. Am J Transplant. 2017. November;17(11):2790–802. [DOI] [PubMed] [Google Scholar]
- 27.Reese PP, Abt PL, Blumberg EA, Van Deerlin VM, Bloom RD, Potluri VS, et al. Twelve-Month Outcomes After Transplant of Hepatitis C–Infected Kidneys Into Uninfected Recipients: A Single-Group Trial. Ann Intern Med. 2018. September 4;169(5):273. [DOI] [PubMed] [Google Scholar]
- 28.Axelrod DA, Schnitzler MA, Alhamad T, Gordon F, Bloom RD, Hess GP, et al. The impact of direct-acting antiviral agents on liver and kidney transplant costs and outcomes. Am J Transplant. 2018. October;18(10):2473–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Richards VL, Johnson CK, Perkins JD, Limaye AP, Sibulesky L. Willingness to Consider Increased-Risk Donors: A Single-Center Experience in Kidney Transplantation. Ann Transplant. 2018. June 5;23:387–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wolfe RA, Ashby VB, Milford EL, Ojo AO, Ettenger RE, Agodoa LYC, et al. Comparison of Mortality in All Patients on Dialysis, Patients on Dialysis Awaiting Transplantation, and Recipients of a First Cadaveric Transplant. N Engl J Med. 1999. December 2;341(23):1725–30. [DOI] [PubMed] [Google Scholar]
- 31.Meier-Kriesche H-U, Kaplan B. Waiting time on dialysis as the strongest modifiable risk factor for renal transplant outcomes: A Paired Donor Kidney Analysis1. Transplantation. 2002. November;74(10):1377–81. [DOI] [PubMed] [Google Scholar]
- 32.Axelrod DA, Schnitzler MA, Xiao H, Irish W, Tuttle-Newhall E, Chang S-H, et al. An economic assessment of contemporary kidney transplant practice. Am J Transplant. 2018;18(5):1168–76. [DOI] [PubMed] [Google Scholar]
- 33.Lentine KL, Kasiske BL, Levey AS, Adams PL, Alberú J, Bakr MA, et al. KDIGO Clinical Practice Guideline on the Evaluation and Care of Living Kidney Donors: Transplantation. 2017. August;101:S7–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Policies - OPTN [Internet]. [cited 2020 Mar 26]. Available from: https://optn.transplant.hrsa.gov/governance/policies
- 35.The Authors for the Live Organ Donor Consensus Group. Consensus Statement on the Live Organ Donor. JAMA: The Journal of the American Medical Association. 2000. December 13;284(22):2919–26. [DOI] [PubMed] [Google Scholar]
- 36.Hjsted J, Ekholm O, Kurita GP, Juel K, Sjgren P. Addictive behaviors related to opioid use for chronic pain: A population-based study: Pain. 2013. December;154(12):2677–83. [DOI] [PubMed] [Google Scholar]
- 37.Sullivan MD, Edlund MJ, Steffick D, Unützer J. Regular use of prescribed opioids: Association with common psychiatric disorders: Pain. 2005. December;119(1–3):95–103. [DOI] [PubMed] [Google Scholar]
- 38.Duerinckx N, Timmerman L, Van Gogh J, van Busschbach J, Ismail SY, Massey EK, et al. Predonation psychosocial evaluation of living kidney and liver donor candidates: a systematic literature review. Transpl Int. 2014. January;27(1):2–18. [DOI] [PubMed] [Google Scholar]
- 39.Gavin DR, Ross HE, Skinner HA. Diagnostic Validity of the Drug Abuse Screening Test in the Assessment of DSM-III Drug Disorders. Addiction. 1989. March;84(3):301–7. [DOI] [PubMed] [Google Scholar]
- 40.Staley D, El-Guebaly N. Psychometric properties of the drug abuse screening test in a psychiatric patient population. Addictive Behaviors. 1990. January;15(3):257–64. [DOI] [PubMed] [Google Scholar]
- 41.McCann S, Simpson Tracy L., Richar B Reliability and Validity of Screening Instruments for Drug and Alcohol Abuse in Adults Seeking Evaluation for Attention-Deficit/ Hyperactivity Disorder. Am J Addict. 2000. January;9(1):1–9. [DOI] [PubMed] [Google Scholar]
- 42.El-Bassel N, Schilling RF, Schinke S, Orlandi M, Sun W-H, Back S. Assessing the Utility of the Drug Abuse Screening Test in the Workplace. Research on Social Work Practice. 1997. January;7(1):99–114. [Google Scholar]
- 43.Yudko E, Lozhkina O, Fouts A. A comprehensive review of the psychometric properties of the Drug Abuse Screening Test. Journal of Substance Abuse Treatment. 2007. March;32(2):189–98. [DOI] [PubMed] [Google Scholar]
- 44.Becker WC, Fraenkel L, Edelman JE, Holt SR, Glover J, Kerns RD, et al. Instruments to assess patient-reported safety, efficacy, or misuse of current opioid therapy for chronic pain: A systematic review: Pain. 2013. June;154(6):905–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lentine KL, Lam NN, Schnitzler MA, Hess GP, Kasiske BL, Xiao H, et al. Predonation Prescription Opioid Use: A Novel Risk Factor for Readmission After Living Kidney Donation. Am J Transplant. 2017. March;17(3):744–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dew M, Switzer G, DiMartini A, Matukaitis J, Fitzgerald M, Kormos R. Psychosocial assessments and outcomes in organ transplantation. Progress in Transplantation. 2000. December;10(4):239–61. [DOI] [PubMed] [Google Scholar]
- 47. KDIGO-Txp-Candidate-GL-Public-Review-Draft-Oct-22.pdf.
- 48.Dobbels F, Hames A, Aujoulat I, Heaton N, Samyn M. Should we retransplant a patient who is non-adherent? A literature review and critical reflection: Review on retransplantation after non-adherence. Pediatric Transplantation. 2012. February;16(1):4–11. [DOI] [PubMed] [Google Scholar]
- 49.Lentine KL, Yuan H, Tuttle-Newhall JE, Xiao H, Chawa V, Axelrod D, et al. Quantifying Prognostic Impact of Prescription Opioid Use Before Kidney Transplantation Through Linked Registry and Pharmaceutical Claims Data: Transplantation. 2015. January;99(1):187–96. [DOI] [PubMed] [Google Scholar]
- 50.Lentine KL, Lam NN, Xiao H, Tuttle-Newhall JE, Axelrod D, Brennan DC, et al. Associations of Pre-Transplant Prescription Narcotic Use with Clinical Complications after Kidney Transplantation. Am J Nephrol. 2015;41(2):165–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rogal S, Dew MA, DiMartini A. High-dose opioid use and liver transplantation: An underestimated problem? Liver Transpl. 2017. March;23(3):285–7. [DOI] [PubMed] [Google Scholar]
- 52.Chandok N, Watt KDS. Pain Management in the Cirrhotic Patient: The Clinical Challenge. Mayo Clinic Proceedings. 2010. May;85(5):451–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zedler B, Xie L, Wang L, Joyce A, Vick C, Kariburyo F, et al. Risk Factors for Serious Prescription Opioid-Related Toxicity or Overdose among Veterans Health Administration Patients. Pain Med. 2014. November;15(11):1911–29. [DOI] [PubMed] [Google Scholar]
- 54.Randall HB, Alhamad T, Schnitzler MA, Zhang Z, Ford-Glanton S, Axelrod DA, et al. Survival implications of opioid use before and after liver transplantation. Liver Transpl. 2017. March;23(3):305–14. [DOI] [PubMed] [Google Scholar]
- 55.Cron DC, Tincopa MA, Lee JS, Waljee AK, Hammoud A, Brummett CM, et al. Prevalence and patterns of opioid use before and after liver transplantation: Transplantation. 2020. February;1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lentine KL, Shah KS, Kobashigawa JA, Xiao H, Zhang Z, Axelrod DA, et al. Prescription opioid use before and after heart transplant: Associations with posttransplant outcomes. Am J Transplant. 2019. December;19(12):3405–14. [DOI] [PMC free article] [PubMed] [Google Scholar]