Abstract
A 75-year-old female with no history of lung disease developed severe pulmonary fibrosis within 1 month of acute severe COVID-19 pneumonia. She developed dry basal crackles, hypoxia needing home oxygen, and computed tomography changes which dramatically evolved from acute ground-glass opacities to honeycombing and traction bronchiectasis. Interestingly, these changes occurred despite her being on steroids through most of her hospital stay. She is being commenced on pirfenidone and her responses are carefully monitored, but the role of antifibrotic drugs are unclear and will only be established from large clinical trials.
KEY WORDS: Antifibrotics, COVID-19 sequelae, pulmonary fibrosis (interstitial lung disease), SARS-Co-V-2
INTRODUCTION
About 24 million people have already been affected by SARS-CoV-2 in the course of this pandemic. While the vast majority have mild infections, about 15% will develop a severe COVID-19 pneumonia, and 5% will develop ARDS, leaving almost 4.8 million with significant pulmonary involvement. While most of these will resolve without lung damage, it is likely that a significant number of patients will be left with residual sequelae.[1]
This case describes a patient with acute COVID pneumonia who, within thirty days, was left with severe fibrotic sequelae. Interestingly, this transformation from ground-glass opacities (GGO) to well-established fibrosis occurred despite her being on steroids.
CASE REPORT
A 75-year-old diabetic female was admitted with a 5-day history of fever, cough, and worsening dyspnea. There was no background of pulmonary problems and her effort tolerance was so good that she could walk 5 km daily in the week prior to admission. Her real-time polymerase chain reaction (RT-PCR) for SARS-Co-V-2 was positive and her initial computed tomography (CT) on admission showed a pattern of bilateral, diffuse, peripheral GGOs, typical of acute COVID-19 pneumonia [Figure 1].
Figure 1.
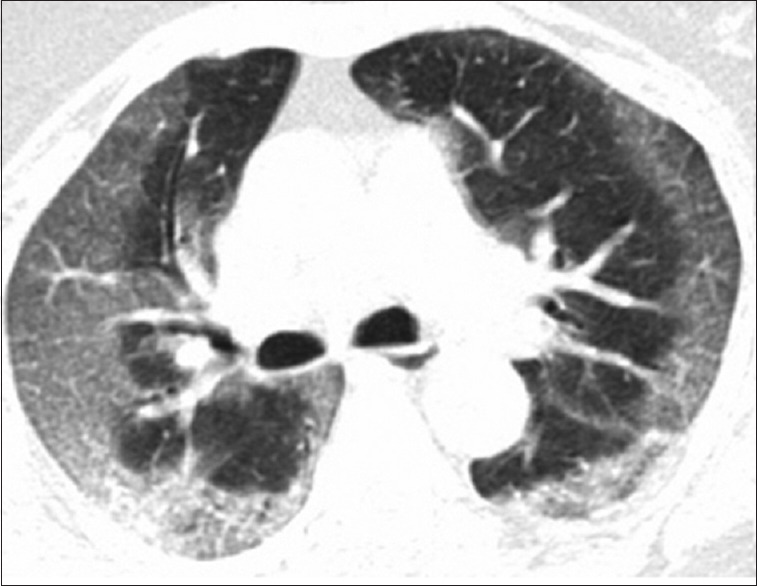
Initial chest computed tomography scan on admission reveals diffuse subpleural ground-glass opacities, typical of COVID-19 pneumonia
She remained extremely tachypnoeic and hypoxic despite high-flow nasal oxygen (PaO260 mmHg despite 15 L/min of oxygen) and on the 5th day of her illness, needed intubation and ventilation with a lung protective ventilator strategy, her FiO2 never exceeding 50%. She could be weaned and extubated within a week. During the 3rd week of her intensive care unit stay, she was diagnosed to have a cytokine storm, with a peak interleukin-6 of 216 pg/ml and a ferritin level of 1422 ng/ml. She was administered one dose of 400 mg of tocilizumab, and intravenous methyl prednisolone at a dose of 120 mg daily tapered gradually, seemed to initially respond.
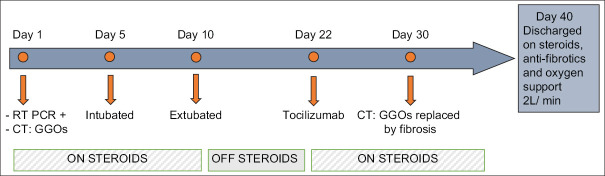
Despite negative RT-PCRs, she continued to have persistent unexplained hypoxia, requiring supplemental nasal oxygen of 4–5 L/min. This was initially felt to be secondary to pulmonary thromboembolism, but a normal CT pulmonary angiography ruled this out. On the 30th day of her illness, a repeat high-resolution CT scan showed that she had developed well-marked fibrotic interstitial lung disease (ILD), with extensive areas of subpleural and peribronchovascular septal thickening, traction bronchiectasis, and honeycombing in both lung fields [Figure 2]. These were fresh changes, not seen in her earlier scan, and represented evolution of her earlier GGOs to fibrotic ILD. Several attempts at bedside pulmonary function test (PFT) were made, but she was too breathless to perform a forced vital capacity maneuver. Steroids were continued, pirfenidone added, and she was eventually discharged after 40 days, needing 2 L/min oxygen via a concentrator. The course of her disease is summarized in Figure 3.
Figure 2.
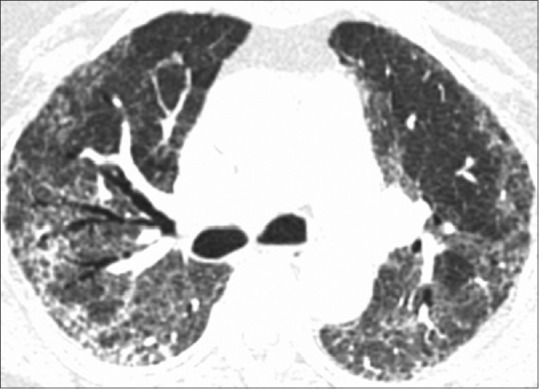
Follow-up chest computed tomography scan on discharge (day 30) reveals clearance of ground glass opacities with residual fibrosis in subpleural and peribronchovascular region with associated traction bronchiectasis and honeycombing
Figure 3.
Time course of disease in hospital along with days when steroid was used. The steroid dose varied from 20 mg to 120 mg of methylprednisolone
DISCUSSION
While most patients with viral pneumonia will heal with minimal sequelae, this case documents a patient with COVID-19 pneumonia who in a timeframe of around a month developed pulmonary fibrosis with honeycombing and traction bronchiectasis. More worryingly, she developed these changes despite being on good doses of steroid for most of her hospital stay.
Fibrosis is not common following other viral pneumonias and has almost never been reported after H1N1 pneumonia. Fibrotic changes have been reported, however, in about 8% of patients with SARS and 20% of patients with H7N9 influenza.[2] Our case report suggests that residual fibrosis is possible post-COVID-19, and significant numbers of patients are likely to recover from acute COVID-19 pneumonia only to be left with severe pulmonary fibrosis as a sequelae.
The role of antifibrotic drugs such as pirfenidone and nintedanib in the setting of post-COVID fibrosis is unclear at present. They have been shown to be useful in not just idiopathic pulmonary fibrosis (IPF),[3,4] but also in a range of progressive non-IPF fibrotic ILDs.[5] There is some therapeutic rationale for the use of antifibrotics in this setting as these drugs have been shown to be useful in acute exacerbations of IPF which are often triggered by viral infections.[6] A trial with pirfenidone is already underway in China and other trials of antifibrotics in this setting are likely to commence.[7] We feel a strong case can be made for trying them in post-COVID fibrosis and commenced our patient on pirfenidone and have planned serial CT scans and PFT.
CONCLUSION
Even though the vast majority of COVID-19 patients will resolve completely or with minimal changes, a minority, like the patient discussed, will be left with established fibrosis. If even a few of the 24 million patients affected to date globally develop fibrosis, we are likely to see significant numbers (possibly hundreds of thousands) of patients with post-COVID fibrosis. The pulmonary community will need to anticipate and prepare for this potential onslaught of patients with advanced fibrotic lung disease and potential oxygen dependence.
Steroids did not seem to prevent the development of fibrosis in this patient who developed traction bronchiectasis and honeycombing despite being on steroids for most of her hospital stay. Further trials to establish the role of anti-fibrotic drugs such as pirfenidone and nintedanib in both preventing fibrosis and controlling the progression of established fibrosis are urgently needed.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Carsana L, Sonzogni A, Nasr A, Rossi RS, Pellegrinelli A, Zerbi P, et al. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: A two-centre descriptive study? Lancet Infect Dis. 2020 doi: 10.1016/S1473-3099(20)30434-5. doi:10.1016/S1473-3099(20)30434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chang YC, Yu CJ, Chang SC, Galvin JR, Liu HM, Hsiao CH, et al. Pulmonary sequelae in convalescent patients after severe acute respiratory syndrome: Evaluation with thin-section CT. Radiology. 2005;236:1067–75. doi: 10.1148/radiol.2363040958. [DOI] [PubMed] [Google Scholar]
- 3.Richeldi L, Du Bois RM, Raghu G, Azuma A, Brown KK, Costabel U, et al. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. [Last accessed on 2020 Jun 24];Engl J Med. 2014 370:2071–82. doi: 10.1056/NEJMoa1402584. Available from: https://pubmed.ncbi.nlm.nih.gov/24836310/ [DOI] [PubMed] [Google Scholar]
- 4.King TE, Bradford WZ, Castro-Bernardini S, Fagan EA, Glaspole I, Glassberg MK, et al. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. [Last accessed on 2020 Jun 24];N Engl J Med. 2014 370:2083–92. doi: 10.1056/NEJMoa1402582. Available from: http://www.nejm.org/doi/10.1056/NEJMoa1402582 . [DOI] [PubMed] [Google Scholar]
- 5.Flaherty KR, Wells AU, Cottin V, Devaraj A, Walsh SL, Inoue Y, et al. Nintedanib in progressive fibrosing interstitial lung diseases. [Last accessed on 2020 Jun 24];N Engl J Med. 2019 381:1718–27. doi: 10.1056/NEJMoa1908681. Available from: http://www.nejm.org/doi/10.1056/NEJMoa1908681 . [DOI] [PubMed] [Google Scholar]
- 6.George PM, Wells AU, Jenkins RG. Pulmonary fibrosis and COVID-19: The potential role for antifibrotic therapy. Lancet Respir Med. 2020;8:807–15. doi: 10.1016/S2213-2600(20)30225-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.A Study to Evaluate the Efficacy and Safety of Pirfenidone with Novel Coronavirus Infection-Full Text View-ClinicalTrials.gov. [Last accessed on 2020 Jun 17]. Available from: https://www.clinicaltrials.gov/ct2/show/NCT04282902 .