Abstract
Purpose
To establish the feasibility of embedding a flexible, exercise-based rehabilitation program into a cancer treatment unit to allow cancer survivors early exercise support.
Method
A pre-post study was conducted using Bowen’s Framework to describe key domains of feasibility: demand (referrals), acceptability (uptake, attendance, satisfaction), implementation (resources), practicality (adverse events, costs) and limited-efficacy (function, quality of life, self-efficacy). Participants were medically stable, adult cancer survivors receiving curative or palliative treatment for cancer at the health service. Participants completed an 8-week home or hospital-based exercise program. Data were analysed descriptively. Standardised mean differences (Hedge’s g) and mean differences were calculated to determine effect size and clinical significance.
Results
The exercise-based rehabilitation service received 155 referrals over 6 months. Of those eligible, 73/119 (61%) commenced. Participants opting for twice-weekly, hospital-based exercise attended 9/16 (56%) sessions. Participants reported high satisfaction and there were no major adverse events. The program utilised existing resources, with the predominant cost being staff. The average health service cost per participant was AUD $1,104. Participants made clinically significant gains in function (6-min walk distance; + 73 m, 95% confidence interval 49 to 96) and quality of life (EORTC QLQ-C30 Global quality of life; + 8 units, 95% confidence interval 3 to 13).
Conclusion
Implementation of exercise-based rehabilitation in a co-located cancer unit was safe and feasible. Access, patient and staff education and establishing funding streams are important implementation considerations.
Implications for cancer survivors
Access to exercise in a cancer unit provides opportunity for early intervention to optimise function during treatment.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00520-021-06261-2.
Keywords: Rehabilitation, Exercise, Referrals, Survivorship
Introduction
Exercise-based rehabilitation is recommended as best practice across the cancer continuum [1–3]. Exercise improves well-being of cancer survivors, with high-level evidence demonstrating exercise reduces symptoms such as fatigue, improves physical function, mood and quality of life [2, 4]. Engaging in exercise after a cancer diagnosis is associated with reduced cancer-specific and all-cause mortality, and disease recurrence [5, 6]. Preliminary evidence suggests a possible mechanism is patients having an increased ability to tolerate systemic therapies such as chemotherapy [7]. Exercise has demonstrated efficacy across all stages of the cancer continuum including before, during and after cancer treatment [2, 8]. Therefore, recommendations suggest cancer survivors commence exercise as early as their respective conditions allow.
Despite compelling evidence for people with cancer to exercise, cancer survivors struggle to achieve exercise recommendations. Approximately 10% of people meet aerobic [9] and 13% meet resistance training recommendations [10]. Several barriers contribute to this evidence-practice gap. At a health service level, access to exercise-based rehabilitation which could support people with cancer to exercise is poor and is not part of standard care [11, 12]. This is compounded by difficult referral pathways, lack of funding and often limited clinician awareness and knowledge about the benefits of exercise and how to find suitable services [11, 13, 14]. Patient-related barriers include side effects, competing medical demands, sedentary lifestyles and practical issues like location, cost and parking [14–16]. Exercise-based rehabilitation is often not considered until after treatment is completed, when people are deconditioned [17]. Intervening with exercise-based rehabilitation within a cancer treatment unit represents an opportunity for early support, including behaviour change strategies during treatment to prevent deterioration in health status.
Efforts are required to close the evidence-practice gap and facilitate routine integration of exercise early into cancer care. A critical first step is to establish feasibility of embedding exercise therapy within pragmatic health service settings, using implementation science methodology [18]. Feasibility of exercise programs for cancer survivors has been established in controlled trial settings. However, the definition of feasibility is usually limited to safety or adherence and is commonly focussed on specific populations [19–23]. It is important to consider feasibility more broadly to understand how evidence-based interventions can be replicated in ‘real life’ settings such as hospital-based cancer treatment units to improve early access to exercise therapy.
Co-location of exercise therapy and cancer services is one strategy that has been proposed to improve access. Co-location of exercise services within cancer treatment units may offer a more flexible, convenient rehabilitation model to facilitate exercise therapy early in the treatment continuum when patients are often overwhelmed and unwell [15]. Two recent studies of co-located exercise programs evaluated several years after implementation have been documented [24, 25]. One was located in a private radiotherapy clinic [25], the other a public hospital cancer treatment centre using a home-based exercise model [24]. In these retrospective studies, positive patient outcomes were reported; however, participants presented largely with early-stage cancer and had adequate baseline levels of physical activity indicating possible selection bias [24, 25]. Feasibility of setting up a new supervised, exercise-based cancer rehabilitation program pragmatically in a public cancer treatment unit remains largely unknown. Therefore, the aim of this study is to determine the feasibility of implementing a flexible, exercise-based cancer rehabilitation program in a hospital-based cancer unit.
Method
Study design
A prospective, pre-post study was completed following Bowen’s Framework [26] to assess the feasibility of implementing an exercise-based rehabilitation program in a hospital-based cancer treatment unit. Bowen’s Framework describes eight general areas of focus addressed by feasibility studies. This study focussed on five key domains: demand, acceptability, implementation, practicality and limited-efficacy testing [22]. The study was reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement [27] and Template for Intervention Description and Replication (TIDieR) [28] checklist. Ethical approval was obtained from the hospital Human Research and Ethics Committee before participant recruitment commenced (LR19-032).
Setting
The study was set in a large, publicly funded tertiary hospital in metropolitan Melbourne, Australia. The cancer unit embedded in the hospital services approximately 3000 patients annually. It comprises an inpatient oncology ward and outpatient day oncology centre providing chemotherapy. A new exercise-based cancer rehabilitation program was developed to run within the unit and commenced in October 2019.
Participants
Patients were referred to exercise-based rehabilitation in the cancer unit between October 2019 and March 2020. To be eligible for the rehabilitation program, participants were to be adult cancer survivors currently receiving or preparing for cancer treatment (curative or palliative intent) admitted as an inpatient or outpatient, who had medical approval to exercise from their specialist or general practitioner. Participants with a cognitive impairment or receiving end of life care were excluded. All participants provided written informed consent prior to participation.
Intervention
The exercise-based cancer rehabilitation program was an 8-week supervised, rolling, exercise-based rehabilitation program, with a maximum of 8 participants per group. Participants completed a 1-h comprehensive assessment with a physiotherapist and were invited to attend an individually tailored, supervised group-based circuit exercise class once or twice weekly or be provided with a tailored home exercise program for 8 weeks. Participants opting for home-based training received an initial face-to-face assessment only and an 8-week follow-up in-person or via telephone. Exercise was prescribed in accordance with guidelines [1, 2] (Table 1).
Table 1.
Intervention description using the template for description and replication checklist (TIDier)
| Intervention | |
|---|---|
| Brief name | Cancer rehabilitation |
| Why | Exercise during cancer treatment may mitigate side effects and improve treatment efficacy |
| What: materials |
• Participants were offered access the hospital gymnasium: - Free weights - Resistance exercise bands - Pin-loaded machines (lateral pull down, leg press, chest press) - Aerobic exercise equipment (exercise bike × 2, treadmill × 1, stairs) - Balance equipment • Participants received a written, individualised home exercise program • Participants were offered a referral to a community exercise program |
| What procedures | |
| Provider | • Two physiotherapists with oncology experience (5.5 years combined) provided by the hospital |
| How | • Face to face sessions (centre-based) or unsupervised sessions (home-based) |
| Where | • Hospital gymnasium or home |
|
When/how much Type |
Aerobic: continuous training prescribed based on 6-min walk test results. Included treadmill, walking, stationary cycle, arm ergometer, exercise pedals Resistance: Prescription based on 10 repetition maximum testing. Pin-loaded machines (leg press, chest press, lat pull down), exercise bands, free weights, body weight exercise (e.g. pushups, stepups, squats, sit to stand) |
| Intensity |
Aerobic: Moderate (BORG 3–4) Resistance: 2–3 sets 10–12 RM Progression: Resistance training based on attainment of 2–3 sets of 12, RPE 3–4 Aerobic training aim to work 2–3 for first 2 weeks of the program, working up to 4 on BORG scale by week 8 Hospital-based group opted for 1 or 2 × weekly supervised training |
| Frequency |
2 × weekly strength training (all) 3X weekly aerobic training (all) |
| Session time |
Hospital: 60 min (5-min warm-up and cool-down, approx 20 min aerobic, 20 min resistance) Home: 30 min aerobic, resistance not time based |
| Overall duration | 8 weeks |
| Tailoring | • Individualised exercise program based on initial consultation and goals |
| Trial fidelity |
• Staff with a background in oncology physiotherapy who had prior formal training were employed by the hospital to provide the intervention. Staff also attended three, 1-h education sessions on exercise and cancer • Exercise log-books were completed for centre-based sessions • Records of the number and duration of completed sessions • Clinical supervision as per standard hospital policy |
Outcome measures
Participant characteristics were recorded including age, gender, cancer diagnosis and treatment details. Measures of demand, acceptability, implementation, practicality and limited efficacy are presented in Table 2.
Table 2.
Outcome measures of feasibility as described by Bowen’s Framework
| Construct | Measure | Source |
|---|---|---|
|
Demand To what extent is a new program likely to be used? |
• Overall number of referrals • Number of newly diagnosed versus long-term cancer survivors |
Routinely collected data |
|
Acceptability To what extent is a new program, judged as suitable, satisfying or attractive to program deliverers or recipients? |
• Number of individual referrers | Routinely collected data |
| • Number of accepted referrals | Exercise records | |
|
• Number of sessions attended • Participant satisfaction |
Survey | |
|
Implementation To what extent can a new program be successfully delivered to intended participants in some defined, but not fully controlled, context? |
• Description of staff training • Stakeholder engagement • Workflow • Equipment • Resources |
Project documentation |
|
Practicality To what extent can a program be carried out with intended participants using existing means, resources, and circumstances and without outside intervention? |
• Number of adverse events • Waiting time • Session duration • Ongoing referrals |
Routinely collected data |
| • Cost | Log of costs (Supplementary file 2) | |
|
Limited Efficacy Does the new program process show promise of being successful with the intended population, even in a highly controlled setting? |
• 6-min walk test • 5-times STS • Exercise self-efficacy scale • EORTC QLQ-30 |
Patient assessment |
EORTC QLQ-30 European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30, STS sit-to-stand
Patient acceptability survey was an instrument designed by the research team (Supplementary File 1)
Data analysis
A convenience sample was employed as one of the primary outcomes was to determine service demand. We aimed to recruit 35 participants over a 6-month period to detect clinically significant changes (Cohen’s d = 0.5) [29] in patient-related outcomes for limited-efficacy testing, assuming a power of 0.80 and 2-tailed alpha level of 0.05. This would also allow comparison to published data on a sub-acute exercise-based program (n = 24) within the same health service [30].
Participant characteristics, satisfaction, demand, uptake and adherence are reported descriptively. Feasibility thresholds for demand, uptake and adherence were based on comparison to routinely collected data from the existing sub-acute cancer rehabilitation program within the health service in the 2019 calendar year. Fisher’s exact test was used to determine differences between referrer type and uptake. Means and standard deviations were calculated for normally distributed data and median and ranges for non-normally distributed data. Content of open-ended survey comments was grouped into themes independently by two researchers and discussed until consensus was met on the main themes using an inductive approach. Standardised mean differences and 95% confidence intervals were calculated using pre- and post-intervention data to determine effect sizes. Cohen’s d was used to describe the magnitude of the change with 0.2 representing a small effect; 0.5 a moderate effect and 0.8 a large effect [31]. Mean differences and 95% confidence intervals were calculated using paired t tests to determine the clinical significance of any changes. To explore differences between program type (hospital-based or home exercise program), analysis of covariance (ANCOVA) was used with baseline measures and time since diagnosis as covariates. Data were analysed using SPSS version 26.
Results
Demand
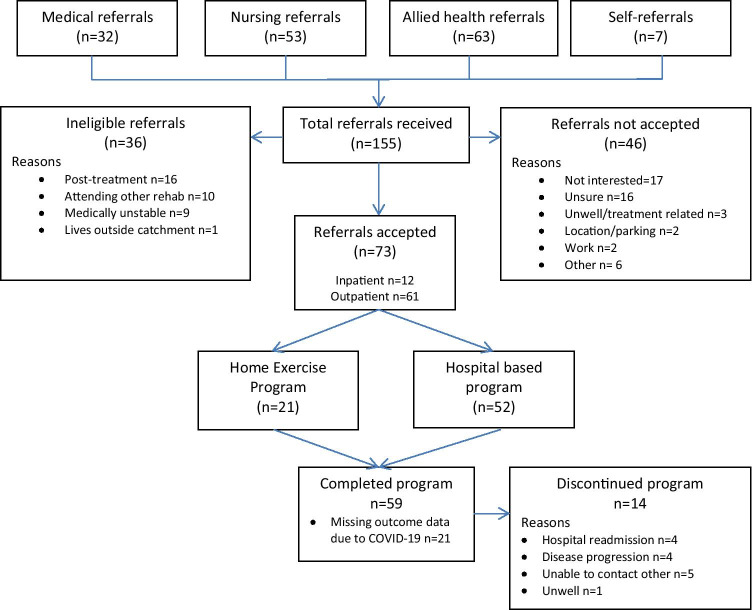
Over the 6-month recruitment period, 155 referrals were received. Most referrals were received from allied health professionals (n = 63, 41%). As demand exceeded expectations, the protocol was amended to recruit all eligible participants within the 6-month period. Of 155 referrals, 119 were eligible (77%) and 73 patients agreed to participate (61% uptake) (Fig. 1). There was no difference in uptake regardless of who referred the patient (p = 0.142).
Fig. 1.
Flow of referrals
Most accepted referrals were outpatients (n = 61, 84%) at program admission. Thirty-nine (53%) patients were newly diagnosed with cancer and 34 (47%) had pre-existing cancer. On average, participants were 63 years old (SD 11) and 9-months post-diagnosis (range 0 to 298). Most participants were diagnosed with breast cancer (n = 15, 21%) or haematological malignancy (n = 28, 38%) and over half with known disease stage were diagnosed with stage 3 or 4 cancers (n = 40, 55%) (Table 3). Most participants were receiving chemotherapy when admitted to the program (n = 41, 64%). Two referrals were made for prehabilitation prior to stem cell transplant and three prior to breast cancer surgery.
Table 3.
Participant demographics
| Characteristic | |
|---|---|
| Age (years), mean (SD) | 63 (11) |
| Gender (male), n (%) | 41 (56) |
| Type of cancer, n (%) | |
| Breast | 15 (21) |
| Lower GI | 8 (11) |
| Upper GI | 2 (3) |
| Gynaecological | 3 (4) |
| Multiple myeloma | 11 (15) |
| Lymphoma | 9 (12) |
| Leukaemia | 8 (11) |
| Lung | 5 (7) |
| Renal | 5 (7) |
| Other | 7 (10) |
| Cancer stage, n (%)* | |
| I | 1 (2) |
| II | 9 (12) |
| III | 21 (29) |
| IV | 19 (26) |
| Unknown | 23 (32) |
| Time since diagnosis, median (range) months | 9 (0 to 298) |
| Current treatment received, n (%) | |
| Chemotherapy only | 41 (46) |
| Radiotherapy only | 3 (4) |
| Immunotherapy only | 15 (21) |
| Targeted therapy only | 2 (3) |
| Combination of any of the above | 6 (8) |
| Stem cell transplant | 6 (8) |
| Treatment intent, n (%) | |
| Curative | 36 (49) |
| Palliative/maintenance | 37 (51) |
*Stage data missing for most haematological malignancies
Acceptability
Referrals were received from 53 individual referrers. Most participants (n = 67, 92%) attended their first appointment. Two participants did not attend beyond the first appointment (n = 1 readmission, n = 1 refused). The preferred rehabilitation pathway was hospital-based rehabilitation (n = 52, 71%), with the majority opting for twice-weekly sessions (n = 38, 52%). On average, participants attended 7 of 16 available sessions. Participants who opted for twice-weekly exercise attended 9/16 sessions (56%), and once-weekly attended 3/8 sessions (40%). Primary reasons for sessions missed were refusal (25% of missed sessions) or unwell due to treatment (23% of missed sessions). Ten participants were unable to participate in group exercise sessions due to the introduction of COVID-19 restrictions. These participants completed home exercise for the remainder of their program. Fourteen participants (19%) did not complete the program (4/21 (19%) home-based, 10/52 (19%) hospital-based) with the most common reasons being hospital readmission (n = 4) or disease progression (n = 4) (Fig. 1). Half of participants were referred to ongoing community-based rehabilitation after program completion, with most being referred to the health service’s existing sub-acute, multi-disciplinary cancer rehabilitation program (n = 20, 27%). Remaining participants accepted an ongoing unsupervised home exercise program. One other multi-disciplinary referral was made to an occupational therapist.
All participants reported they were satisfied with the program and would recommend it to others (Supplementary file 2). They described physical and psychosocial benefits and highly valued staff.
‘I found that the ‘treadmill’ of cancer was mitigated considerably by the improvement of my physical health and fitness. My concern of adding another set of activities to the rounds of blood tests, MRIs, CAT scans, doctor’s appointments, treatment was in hindsight unfounded’.
(participant 10, age 74, metastatic melanoma and lymphoma)
‘I found the staff really supportive… they tailored the program to suit my needs… were really flexible and attentive…’
(participant 52, age 53, leukaemia)
Participants had high confidence to continue exercising after the program finished according to the self-efficacy item on the survey (mean 9 units, SD 1).
‘It gave me motivation. I wish I had started earlier’.
(participant 60, age 71, leukaemia)
Six participants (8%) reported difficulties with program access. The main issue reported was parking.
‘I would still be exercising but I had to stop due to lack of parking’.
(participant 1, age 67, metastatic lung cancer)
Implementation strategies
A senior physiotherapist (20 h per week) and mid-level physiotherapist (19.5 h per week) with combined 5.5 years oncology-specific experience were employed to implement and administer the program. They were supported by a senior research physiotherapist in the initial setup to promote and coordinate the program and provide leave cover for staff absence. The physiotherapists had prior cancer-specific training in acute and community cancer settings.
To raise awareness and knowledge of cancer rehabilitation, three 1-h education sessions about cancer and rehabilitation were completed with program staff and other hospital physiotherapists. Additionally, three presentations were made to cancer services medical, nursing and allied health staff to provide updates throughout program implementation. A flyer, poster and fortnightly newsletter was developed, and the organisation’s communications officer was engaged to promote the program within and outside the health service. Implementation was guided by a steering committee including a consumer, clinical directors, physiotherapy manager and community partner.
The referral process was made simple so all oncology clinicians could readily refer. Referrals could be received verbally (in-person or via phone) directly to the physiotherapist by any clinician, or via a centralised email address by including patient name and contact details. Patients could also self-refer. All clinicians were encouraged to have a conversation with patients about exercise and cancer prior to referral. However, in practice, referrals may have been received without a conversation being had.
Assessments were conducted in a dedicated room in the cancer unit. Exercise sessions were conducted in the existing physiotherapy gym with pre-existing equipment. A flexible program was deliberately offered in an attempt to improve uptake. A timetable was created which allowed exercise sessions to be run 4 days per week (Monday to Thursday). Participants were scheduled to attend a one-hour exercise session once or twice a week or receive only a home-based program. A clinician-to-patient ratio of 1:4 was employed for the group classes. Existing forms and electronic medical records were used for program documentation.
Initial funding for the pilot was provided by an external service improvement grant. Philanthropic funds were sought to sustain the program beyond the pilot period to fund ongoing clinical staff salaries. There was no cost to patients to participate in the program. Participants were offered subsidised on-site parking through the day oncology unit but reported not using it due to limited availability.
Practicality
Three minor adverse events related to the intervention were reported in the hospital-based participants which resolved with rest and exercise modification (shoulder pain, muscle soreness, dizziness). No major adverse events were recorded.
The average wait time to the first appointment was 20 days (range 0–99). Variation in wait time was due to patients not feeling ready to participate at first contact. The average time taken for first appointment was 51 min (SD 7). Exercise sessions were 1-h duration.
No additional expenses were incurred for equipment or venue. A direct mobile phone line for the program was initiated as there was no administration support available. The primary resource cost was the funding of staff resources (Supplementary file 3). The average overall cost to the health service per patient was AUD $1,104.
Limited efficacy
Participants demonstrated significant changes in function. Objectively, 6MWT improved by a mean difference of 72.8 m (95% CI 49.4 to 96.2 m) and timed sit-to-stand improved by 1.3 s (95% CI 2.6 to 2.0 s). Significant improvements were also made on all functional subscales of the EORTC QLQ-C30 (Table 4).
Table 4.
Limited efficacy testing, pre and post intervention means of patient outcomes
| Outcome | Baseline, mean (SD) | Post-intervention, mean (SD) | Mean difference, 95%CI | Hedges g effect size, 95% CI |
|---|---|---|---|---|
| 6MWT (m) n = 38 | 414 (92) | 486 (78) | 73 (49 to 96)* | 0.85 (0.14 to 1.46) |
| 5 times STS (s) n = 41 | 11.1 (3.7) | 9.8 (2.9) | − 1.3 (− 2.1 to − 0.6)* | 0.39 (0.05 to 0.83) |
| Self-efficacy for PA (0–90) n = 58 | 56.4 (17.6) | 60.8 (16.9) | 4.3 (− 0.90 to 9.6) | 0.25 (− 0.11 to 0.62) |
| EORTC QLQ-C30 (0–100) n = 59 | ||||
| Global quality of life | 61 (22) | 69 (16) | 8 (3 to 13)** | 0.43 (0.06 to 0.79) |
| Physical function | 77 (17) | 82 (15) | 5 (1 to 9)** | 0.32 (− 0.04 to 0.69) |
| Role function | 61 (29) | 73 (26) | 12 (4 to 21)** | 0.45 (0.08 to 0.81) |
| Emotional function | 76 (20) | 83 (17) | 7 (3 to 12)** | 0.41 (0.04 to 0.77) |
| Cognitive function | 75 (23) | 81 (20) | 5 (1 to 10)* | 0.25 (− 0.11 to 0.61) |
| Social function | 63 (26) | 74 (24) | 11 (5 to 18)** | 0.45 (0.08 to 0.82) |
| Fatigue | 42 (19) | 33 (21) | − 9 (− 15 to − 3)** | 0.43 (0.07 to 0.80) |
| Nausea | 12 (20) | 8 (16) | − 4 (− 10 to 2) | 0.22 (− 0.14 to 0.58) |
| Pain | 23 (25) | 21 (22) | − 2 (− 7 to 3) | 0.08 (− 0.28 to 0.44) |
| Dyspnoea | 29 (27) | 18 (21) | − 11 (− 18 to − 3)** | 0.44 (0.07 to 0.80) |
| Insomnia | 32 (26) | 34 (29) | 2 (− 6 to 10) | 0.08 (− 0.44 to 0.28) |
| Appetite | 16 (23) | 16 (25) | 0 (− 8 to 6) | 0.02 (− 0.34 to 0.38) |
| Constipation | 20 (28) | 14 (24) | − 7 (− 14 to 0) | 0.26 (− 0.10 to 0.62) |
| Diarrhoea | 17 (27) | 13 (22) | − 4 (− 12 to 4) | 0.16 (− 0.20 to 0.52) |
| Financial difficulty | 24 (35) | 20 (31) | − 4 (− 10 to 2) | 0.12 (− 0.24 to 0.48) |
*Significant at < 0.05 **significant at p < 0.01
Significant changes in global quality of life were achieved (MD 8.2 units, 95% CI 2.6 units to 13 units). Fatigue (MD − 8.8 units, 95% CI − 2.6 to − 15.0) and dyspnoea (MD − 10.7 units, 95% CI − 3.3 to − 18.2) also improved. There was no difference in other symptoms or exercise self-efficacy (Table 4).
When comparing program types (centre-based versus home exercise program), there was no difference in any outcome (Supplementary file 4).
Discussion
This study demonstrated exercise-based rehabilitation is safe and feasible in a hospital-based cancer unit. Participants who completed the program reported high satisfaction and made clinically significant gains in function and quality of life. Despite demand, acceptability and easy implementation of the program, there was modest uptake. This study adds to existing literature by demonstrating the feasibility of a co-located exercise-based cancer rehabilitation model including hospital and home-based exercise options with a mixed cohort of cancer survivors with moderate levels of exercise self-efficacy. It provides a practical outline of how-to setup a co-located exercise-based cancer rehabilitation program which will aid the ongoing translation of research to practice.
Implementing a co-located, exercise-based cancer rehabilitation program may help overcome barriers to cancer survivors accessing exercise. As demonstrated by this study, a broad demographic of cancer survivors participated in the program including many groups under-represented in exercise cancer trials such as males and people with advanced or haematological malignancies. Referrals were made by a variety of referrers, including doctors, indicating the program was acceptable to health professionals. Referral sources were much greater than that reported by another co-located exercise rehabilitation program where most referrals came from just two oncologists [25]. Referral rates were also higher than those previously published, with total reach of the program approximately 10% of potentially eligible patients at the health service. This rate is similar to another co-located program [25] and significantly higher than existing exercise-based cancer rehabilitation programs [11, 30]. Demand for this program is noteworthy given low numbers of referrals to cancer rehabilitation are an issue in clinical practice [11]. Success may be owed to the visibility of the program in the cancer centre, providing awareness and convenience to patients and referrers [15]. Co-location may be a particularly important consideration for developing new rehabilitation programs for people receiving treatment where additional barriers such as feeling unwell and managing multiple priorities are an issue [15]. Investment was also put into program promotion with extensive stakeholder engagement before, during and after implementation, raising the profile of cancer rehabilitation within the organisation. An advantage was that the organisation had a pre-existing sub-acute cancer rehabilitation program to help drive and promote cross-referrals. These factors may have resulted in greater readiness for implementation and culture change where exercise-rehabilitation during cancer treatment is valued [32].
Despite this program providing better access to exercise for cancer survivors receiving treatment, modest uptake of the program by potential participants was an issue. Almost half of eligible referred participants did not accept referral with the most common reasons being they were not interested or unsure of committing to a program. Uptake was lower than previous reports [33] but consistent with prior evidence people need to feel ‘ready’ to participate in cancer rehabilitation, and require multiple prompts before they act on a referral [34]. Uptake may also be reflective of the pragmatic sample from a public hospital setting where participants may have significant comorbidities or less interest in exercise. It is also possible not all participants participated in a conversation about exercise with their oncologist or haematologist prior to referral which may negatively affect uptake given an oncologist’s recommendation significantly increases participation in exercise [35]. This highlights the importance of bi-directional education and discussion with patients about why they should participate in exercise-rehabilitation during treatment. It also questions the appropriateness of routinely referring all participants to exercise-based rehabilitation as many are simply not ready to participate. Therefore, flexible models of rehabilitation with varied entry and exit points are required [36].
Many cancer rehabilitation models exist but no optimal model has been established [37]. The cardiac rehabilitation model, comprising centre-based, supervised exercise prescription and self-management, is a common model integrated into hospital settings [11] that is utilised in this study. Advantages of this model are greater efficacy of supervised training [38] and the ability to utilise pre-existing facilities and referral structures [39]. However, these programs may not be able to cater to the wide needs of cancer survivors, with issues related to program structure and access [11]. This study demonstrated the feasibility of an alternative model including home-based exercise which added greater flexibility. Cancer survivors have previously described preference for home-based exercise [40] and those participating in home-based exercise in this study achieved equivalent outcomes to those attending hospital-based exercise. Home-based exercise models could be a less resource-intensive option for implementation of cancer rehabilitation in health services. Future research could evaluate home-based models which may include telehealth, use of mobile applications and wearables for monitoring as well as assessing cost-effectiveness, uptake and exercise adherence to further aid broad implementation of exercise-based cancer rehabilitation.
Implementing hospital-based rehabilitation programs is resource-intensive. Costs are a key driver of decision-making in health care. Similar to this study, recent implementation studies [24, 25] were largely reliant on philanthropic and research funding to develop and sustain cancer exercise rehabilitation programs and the volatile nature of this funding presents major barriers to ongoing sustainability [25]. The average cost per patient for this program was AUD $1,104 with the primary expense being staff. Costs were similar to other supervised exercise rehabilitation programs [41–43]. These costs may be offset by adherence to exercise recommendations being associated with lower health care expenditure in people with cancer [44]. Significant clinical improvements in patient outcomes demonstrated in this study may result in prevention of hospital readmission and reduced hospital length of stay. The average cost of a cancer-related hospital admission in Australia varies between AUD $3561 and AUD $48,659 [45]. There is emerging evidence rehabilitation programs may provide cost savings through reduction in length of stay and readmissions [46]. Cost savings related to exercise-rehabilitation are most evident in programs delivered prior or during cancer treatment [41, 42, 47, 48]. For example, a trial of supervised exercise after autologous stem cell transplant found rehabilitation was not cost-effective [42], whereas a trial of supervised exercise for women during treatment for breast cancer was likely to be cost-effective depending on willingness to pay thresholds [41]. Therefore, there is potential for large cost savings with early rehabilitation. More research is required to confirm benefits to health services to drive policy change and fund exercise-rehabilitation programs as part of standard care.
Strengths and limitations
To our knowledge, this is the first study to assess the feasibility of implementing a flexible, exercise-based cancer rehabilitation program in a public, co-located cancer unit. This study was reported in accordance with STROBE and TIDieR guidelines which will assist replication of findings in other cancer settings. A strength of this research is that it evaluates a pragmatic program utilising minimal resources which could be replicated in public hospital settings.
A limitation of this study is that it includes a small, non-randomised sample of participants. There is risk of selection bias as commonly, participants seek out interventions they value. However, the pragmatic nature of the study setting included a broad demographic of participants including those typically under-represented in exercise oncology research suggesting results may be generalizable to other settings. This study focussed on feasibility which included implementation strategies, but it is acknowledged different implementation frameworks could be used to inform future research translation. The home exercise program delivered in this study was limited to written instructions without follow-up which may reduce the effectiveness of the home-based intervention. Adherence to the home exercise program was also not recorded due to previous non-compliance with completion of exercise diaries [30]. However, the home-based model was not the focus of this study and participants still demonstrated gains. We also did not measure baseline physical activity levels. However, participants had only modest exercise confidence and a similar demographic of participants in previous studies undertaken in the same health service demonstrated very low levels of physical activity [30, 49]. Improvement in outcomes may have been influenced by natural progression over time and supportive care provided. However, the aim of this study was not to demonstrate efficacy of exercise. It is well established from randomised controlled trials that exercise-rehabilitation improves health outcomes for cancer survivors. Participants made clinically significant gains consistent with published evidence which increases confidence improvements were related to the rehabilitation intervention.
This study demonstrated implementing a co-located, exercise-based rehabilitation program in a cancer unit is safe and feasible. Clinical gains in this study reinforce exercise interventions can be readily translated pragmatically into hospital settings. Co-location may improve access to exercise for cancer survivors. However, readiness to participate in rehabilitation during treatment needs to be considered by using flexible rehabilitation models to overcome issues with uptake and adherence. More health services research is required to demonstrate the cost-effectiveness of programs so they can be integrated into standard care.
Supplementary Information
Below is the link to the electronic supplementary material.
(XLSX 62.5 kb)
Acknowledgements
We would like to thank Germaine Tan and April Chiu for their contribution to participant recruitment and data collection. We would also like to thank the staff and patients from Eastern Health who supported this project.
Author contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Amy Dennett. The first draft of the manuscript was written by Amy Dennett and Casey Peiris, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Funding
This project was funded by a service improvement grant from North Eastern Melbourne Integrated Cancer Services.
Data availability
Data is available as supplementary material.
Declarations
Ethics approval
This study was approved by the Eastern Health Human Research Ethics Committee (LR19-032).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
N/A
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Cormie P, et al. Clinical Oncology Society of Australia position statement on exercise in cancer care. Med J Aust. 2018;209:184–187. doi: 10.5694/mja18.00199. [DOI] [PubMed] [Google Scholar]
- 2.Campbell KL, et al. Exercise guidelines for cancer survivors: consensus statement from international multidisciplinary roundtable. Med Sci Sports Exerc. 2019;51(2375–90):3. doi: 10.1249/MSS.0000000000002116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Segal R, et al. Exercise for people with cancer: a clinical practice guideline. Curr Oncol. 2017;24:40–46. doi: 10.3747/co.24.3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dennett AM, et al. Moderate-intensity exercise reduces fatigue and improves mobility in cancer survivors: a systematic review and meta-regression. J Physiother. 2016;62:68–82. doi: 10.1016/j.jphys.2016.02.012. [DOI] [PubMed] [Google Scholar]
- 5.Cormie P, et al. The impact of exercise on cancer mortality, recurrence, and treatment-related adverse effects. Epidemiol Rev. 2017;39:1–22. doi: 10.1093/epirev/mxx007. [DOI] [PubMed] [Google Scholar]
- 6.Patel AV, et al. American College of Sports Medicine roundtable report on physical activity, sedentary behavior, and cancer prevention and control. Med Sci Sports Exerc. 2019;51(2391–2402):7. doi: 10.1249/MSS.0000000000002117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bland KA, et al. Impact of exercise on chemotherapy completion rate: a systematic review of the evidence and recommendations for future exercise oncology research. Crit Rev Oncol Hematol. 2019;136:79–85. doi: 10.1016/j.critrevonc.2019.02.005. [DOI] [PubMed] [Google Scholar]
- 8.Faithfull S, et al. Prehabilitation for adults diagnosed with cancer: a systematic review of long-term physical function, nutrition and patient-reported outcomes. Eur J Cancer Care. 2019;28(e13023):9. doi: 10.1111/ecc.13023. [DOI] [PubMed] [Google Scholar]
- 9.Thraen-Borowski KM, Gennuso KP, Cadmus-Bertram L. Accelerometer-derived physical activity and sedentary time by cancer type in the United States. PLoS ONE. 2017;12:e0182554. doi: 10.1371/journal.pone.0182554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Galvão DA, et al. Compliance to exercise-oncology guidelines in prostate cancer survivors and associations with psychological distress, unmet supportive care needs, and quality of life. Psychooncology. 2015;24:1241–1249. doi: 10.1002/pon.3882. [DOI] [PubMed] [Google Scholar]
- 11.Dennett AM, et al. Exercise therapy in oncology rehabilitation in Australia: a mixed-methods study. Asia Pac J Clin Oncol. 2017;13:e515–e527. doi: 10.1111/ajco.12642. [DOI] [PubMed] [Google Scholar]
- 12.Stout NL, et al. Towards a national initiative in cancer rehabilitation: recommendations from a subject matter expert group. Arch Phys Med Rehabil. 2016;97:2006–2015. doi: 10.1016/j.apmr.2016.05.002. [DOI] [PubMed] [Google Scholar]
- 13.Pergolotti M, et al. A health services research agenda to fully integrate cancer rehabilitation into oncology care. Cancer. 2019;125(3908–16):14. doi: 10.1002/cncr.32382. [DOI] [PubMed] [Google Scholar]
- 14.Granger CL, et al. Barriers to translation of physical activity into the lung cancer model of care. a qualitative study of clinicians' perspectives. Ann Am Thorac Soc. 2016;13:2215–2222. doi: 10.1513/AnnalsATS.201607-540OC. [DOI] [PubMed] [Google Scholar]
- 15.Dennett AM, Harding KE, Reed MS (2020) The challenge of timing: a qualitative study on clinician and patient perspectives about implementing exercise-based rehabilitation in an acute cancer treatment setting. Support Care Cancer Online ahead of print. 10.1007/s00520-020-05436-7 [DOI] [PubMed]
- 16.Henriksson A, et al. Perceived barriers to and facilitators of being physically active during adjuvant cancer treatment. Patient Educ Couns. 2016;99:1220–1226. doi: 10.1016/j.pec.2016.01.019. [DOI] [PubMed] [Google Scholar]
- 17.Courneya KS, Friedenreich CM. Physical activity and cancer control. Semin Oncol Nurs. 2007;23:242–252. doi: 10.1016/j.soncn.2007.08.002. [DOI] [PubMed] [Google Scholar]
- 18.Brownson RC, Colditz GA. Proctor EK (2018) Dissemination and implementation research in health: translating science to practice. 2. New York: Oxford University Press; 2018. [Google Scholar]
- 19.Heywood R, McCarthy AL, Skinner TL. Safety and feasibility of exercise interventions in patients with advanced cancer: a systematic review. Support Care Cancer. 2017;25:3031–3050. doi: 10.1007/s00520-017-3827-0. [DOI] [PubMed] [Google Scholar]
- 20.Finley DJ, et al. A feasibility study of an unsupervised, pre-operative exercise program for adults with lung cancer. Eur J Cancer Care. 2020;29:e13254. doi: 10.1111/ecc.13254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim Y, Park S. Feasibility and benefits of a combined programme of exercise and play for paediatric cancer survivors: a pilot study. Eur J Cancer Care. 2019;28:e13111. doi: 10.1111/ecc.13111. [DOI] [PubMed] [Google Scholar]
- 22.Peddle‐Mcintyre CJ et al (2018) The feasibility of a pragmatic distance‐based intervention to increase physical activity in lung cancer survivors. Eur J Cancer Care 2018. 27: 10.1111/ecc.12722 [DOI] [PubMed]
- 23.Schuler MK, et al. Feasibility of an exercise programme in elderly patients undergoing allogeneic stem cell transplantation – a pilot study. Eur J Cancer Care. 2016;25:839–848. doi: 10.1111/ecc.12400. [DOI] [PubMed] [Google Scholar]
- 24.Santa Mina D, et al. Development, implementation, and effects of a cancer center's exercise-oncology program. Cancer. 2019;125:3437–3447. doi: 10.1002/cncr.32297. [DOI] [PubMed] [Google Scholar]
- 25.Kennedy MA et al (2020) If you build it, will they come? Evaluation of a co-located exercise clinic and cancer treatment centre using the RE-AIM framework. Eur J Cancer Care. Online ahead of print. E13251. 10.1111/ecc.13251 [DOI] [PubMed]
- 26.Bowen DJ, et al. How we design feasibility studies. Am J Prev Med. 2009;36:452–457. doi: 10.1016/j.amepre.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Von Elm E, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12:1495–1499. doi: 10.1016/j.ijsu.2014.07.013. [DOI] [PubMed] [Google Scholar]
- 28.Hoffmann TC, et al. Better reporting of interventions: template for intervention description and replication (TiDieR) checklist and guide. BMJ. 2014;348:G1687. doi: 10.1136/bmj.g1687. [DOI] [PubMed] [Google Scholar]
- 29.Norman GR, Sloan JA, Wyrwich KW. Interpretation of changes in health-related quality of life: the remarkable universality of half a standard deviation. Med Care. 2003;41:582–592. doi: 10.1097/01.MLR.0000062554.74615.4C. [DOI] [PubMed] [Google Scholar]
- 30.Dennett AM, et al. Motivational interviewing added to oncology rehabilitation did not improve moderate-intensity physical activity in cancer survivors: a randomised trial. J Physiother. 2018;64:255–263. doi: 10.1016/j.jphys.2018.08.003. [DOI] [PubMed] [Google Scholar]
- 31.Cohen J. Statistical Power Analysis. Curr Dir Psychol Sci. 1992;1:98–101. doi: 10.1111/1467-8721.ep10768783. [DOI] [Google Scholar]
- 32.Czosnek L, et al. Implementing exercise in healthcare settings: the potential of implementation science. Sports Med. 2020;50:1–14. doi: 10.1007/s40279-019-01228-0. [DOI] [PubMed] [Google Scholar]
- 33.Kirkham AA, et al. Effectiveness of oncologist-referred exercise and healthy eating programming as a part of supportive adjuvant care for early breast cancer. Oncologist. 2018;23:105–115. doi: 10.1634/theoncologist.2017-0141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dennett AM, et al. (2020) 'A good stepping stone to normality': a qualitative study of cancer survivors' experiences of an exercise-based rehabilitation program. Support Care Cancer. 2019;27:1729–1736. doi: 10.1007/s00520-018-4429-1. [DOI] [PubMed] [Google Scholar]
- 35.Jones LW, et al. Effects of an oncologist's recommendation to exercise on self-reported exercise behavior in newly diagnosed breast cancer survivors: a single-blind, randomized controlled trial. Ann Behav Med. 2004;28:105–113. doi: 10.1207/s15324796abm2802_5. [DOI] [PubMed] [Google Scholar]
- 36.Dennett AM, Peiris CL, Shields N, Taylor NF. From cancer rehabilitation to recreation: a coordinated approach to increasing physical activity. Phys Ther. 2020;30(100):2049–2059. doi: 10.1093/ptj/pzaa135. [DOI] [PubMed] [Google Scholar]
- 37.Cheville AL, et al. Cancer rehabilitation: an overview of current need, delivery models, and levels of care. Phys Med Rehabil Clin N Am. 2017;28:1–17. doi: 10.1016/j.pmr.2016.08.001. [DOI] [PubMed] [Google Scholar]
- 38.Buffart LM, et al. Effects and moderators of exercise on quality of life and physical function in patients with cancer: an individual patient data meta-analysis of 34 RCTs. Cancer Treat Rev. 2017;52:91–104. doi: 10.1016/j.ctrv.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 39.Dittus KL, et al. Exercise-based oncology rehabilitation: leveraging the cardiac rehabilitation model. J Cardiopulm Rehabil Prev. 2015;35:130–139. doi: 10.1097/HCR.0000000000000091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wong JN, Mcauley E, Trinh L. Physical activity programming and counseling preferences among cancer survivors: a systematic review. Int J Behav Nutr Phys Act. 2018;15:48. doi: 10.1186/s12966-018-0680-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Van Waart H, et al. Cost-utility and cost-effectiveness of physical exercise during adjuvant chemotherapy. Eur J Health Econ. 2018;19:893–904. doi: 10.1007/s10198-017-0936-0. [DOI] [PubMed] [Google Scholar]
- 42.Van Dongen JM, et al. Long-term effectiveness and cost-effectiveness of an 18-week supervised exercise program in patients treated with autologous stem cell transplantation: results from the EXIST study. J Cancer Surviv. 2019;13:558–569. doi: 10.1007/s11764-019-00775-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Gordon LG, et al. A cost-effectiveness analysis of two rehabilitation support services for women with breast cancer. Breast Cancer Res Treat. 2005;94:123–133. doi: 10.1007/s10549-005-5828-9. [DOI] [PubMed] [Google Scholar]
- 44.Yan AF, Wang Y, Ng AV. Physical activity and annual medical outlay in U.S. colorectal, breast and prostate cancer survivors. Prev Med Rep. 2018;9:118–123. doi: 10.1016/j.pmedr.2018.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Independent Hospital Pricing Authority (2019) National Hospital Cost Data Collection Report, Public Sector, Round 21 (Financial year 2016–17)
- 46.Mewes JC, Steuton LM, Ijzerman MJ, van Harten WH. Effectiveness of multidimensional cancer survivor rehabilitation and cost-effectiveness of cancer rehabilitation in general: a systematic review. Oncologist. 2012;17:1581–1593. doi: 10.1634/theoncologist.2012-0151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cavalheri V, Granger CL (2017) Preoperative exercise training for patients with non-small cell lung cancer. Cochrane Database Syst Rev 6: Cd012020. [DOI] [PMC free article] [PubMed]
- 48.Van Haren I, et al. Physical exercise prior to hematopoietic stem cell transplantation: a feasibility study. Physiother Theory Pract. 2018;34:747–756. doi: 10.1080/09593985.2018.1423655. [DOI] [PubMed] [Google Scholar]
- 49.Dennett AM, et al. Cancer survivors awaiting rehabilitation rarely meet recommended physical activity levels: an observational study. Rehabil Oncol. 2018;36:214–222. doi: 10.1097/01.REO.0000000000000132. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX 62.5 kb)
Data Availability Statement
Data is available as supplementary material.