Abstract
In early studies regarding coronavirus disease 2019 (COVID-19), type 2 diabetes mellitus was considered to contribute substantially to the disease’s inflammatory response. Subsequently, even hyperglycemia, regardless of insulin resistance or diabetes mellitus, was found to be additionally harmful. Recent studies have shown inflammation of the pancreatic β cells in COVID-19, even leading to new onset diabetes mellitus. We hereby summarize core literature on glycemia and COVID-19, and present implicated pathways and mechanisms.
Keywords: COVID-19, Humans, SARS-CoV-2, Risk factors, Glucose, Inflammation, Diabetes
Core Tip: The severe acute respiratory syndrome coronavirus 2 pathogen has led to the coronavirus disease 2019 (COVID-19) pandemic. This virus exerts multi-organ actions after an initial respiratory infection. In early studies regarding COVID-19, type 2 diabetes mellitus was considered to contribute substantially to the disease’s inflammatory response. Hyperglycemia in COVID-19, irrespective of insulin resistance or history of diabetes, is a portent of worse prognosis. Further studies will help elucidate the link between glycemia and COVID-19.
INTRODUCTION
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pathogen has led to the coronavirus disease 2019 (COVID-19) pandemic. This virus exerts multi-organ actions, after an initial respiratory infection[1]. Regarding COVID-19, while age and male gender are regarded as significant risk factors, accumulating evidence suggests a strong association with an impaired cardiometabolic profile in most severely ill patients[2]. Reports from Wuhan, China were the first to indicate a higher prevalence of hypertension and diabetes mellitus (DM) among patients with severe compared to non-severe illness[3]. From the beginning of the outbreak, cardiovascular disease (CVD), obesity, type 1 DM (T1DM), type 2 DM (T2DM) and possibly hypertension have seemed to be associated with the risk of suffering or dying from COVID-19[4-6].
DIABETES AS A PREDICTOR OF THE COURSE OF COVID-19
COVID-19 patients with T2DM and/or CVD are admitted more often to intensive care units (ICUs) compared to those without T2DM or CVD[7]. Older age and T2DM are both risk factors for COVID-19, but the observation that T2DM is a disease that is frequent in advanced age, slightly confounds this association[8].The risk of developing severe COVID-19 is higher in people with DM, especially if they have other co-morbidities, thus making patients with DM an at-risk population. The worse the glycemic control, the worse the severity of infection and the greater the risk of mortality[9]. In the initial studies of COVID-19, DM appeared to be 2.26 times (95% confidence interval [CI]: 1.47-3.49) more common in patients with more severe COVID-19 compared to those with less severe infection, while at the same time the presence of DM entailed an odds ratio of 2.85 (95%CI: 1.35-6.05) of intra-hospital mortality[2]. As already mentioned, these results were not always adjusted for age, which is a major confounding factor in the prevalence of DM. In Italy, one-third of patients who died of COVID-19 had DM (median age 80.5 years) and were predominantly male (70%)[2]. Compared with the prevalence of DM in the same population segment in Italy in 2018 (20.3%), the authors reported a relative risk of diabetes of 1.75 in patients who died from COVID-19[2]. It is therefore necessary to emphasize the advanced age of patients with severe COVID-19, as well as their multiple comorbidities, defining them as a population particularly at risk.
COVID-19 AND INFLAMMATION
COVID-19 is characterized by the excessive production of inflammatory factors, leading to an “in inflammatory storm” (a combination of pro-inflammatory immunoactive molecules, such as interleukins [ILs], interferons [IFNs], chemokines and tumor necrosis factors [TNFs]) in some patients[10]. Diffuse pulmonary alveolar damage, inflammatory cell infiltration with hyaline membranes, myocardial inflammation, lymphocyte infiltration in the liver, and pancreatitis are some of the major inflammatory findings during the course of the generalized COVID-19[11]. In sharp contrast to the above, the IFN type I response is impaired in these patients[12]. For patients with severe COVID-19, this so-called cytokine storm is a potentially life- threatening event[13].
In 317 patients with laboratory-confirmed COVID-19, inflammatory responses and higher levels of IL-6 were related to disease severity[11,14]. In patients with COVID-19, inflammatory markers such as C-reactive protein, D-dimers, ferritin, and IL-6 are increased; they have a direct effect on microvascular and macrovascular structures in patients with DM[15].
DIABETES, OBESITY, AND INFLAMMATORY SIMILARITIES WITH COVID-19
Although T1DM is not related to obesity, the majority of patients with T2DM are overweight or obese. Resembling the inflammatory processes of COVID-19, prolonged hyperglycemia, regardless of diabetes type, can also impact immune function, whereas compromised immunological status is linked to macrovascular complications of DM[11].
Inflammation begets increased oxidative stress that can damage proteins, lipids and DNA, systemically, as well as locally, both in the liver and in muscles, the predominant organs that regulate glucose output and glucose metabolism, increasing insulin resistance[16]. In T2DM, inflammation occurs in the pancreatic β cell (insulitis)[16]. Macrophages play a key role in β cell inflammation, along with IL-1β signaling (a core inflammatory process in the locally stressed β cell). Along with the local injury of the pancreatic cells, lipotoxicity further deteriorates pancreatic function. Free fatty acids can also induce the local production of IL-1β- and IL-1-dependent pro-inflammatory cytokines, which target the pancreatic islets. This process also increases nitric oxide production, lowers mitochondrial ATP, causing additional β cell dysfunction, along with the release of reactive oxygen species by hypoxia and endothelial damage[17]. TNF-α is linked to insulin resistance, obesity and islet inflammation, while IL-6 promotes islet cell apoptosis; both lead to T2DM. Obesity and DM (which often are described as “diabesity”) favor a switch from (anti-inflammatory) M2 macrophage predominance to (pro-inflammatory) M1 macrophage predominance, further contributing to exaggerated inflammation[18]. Of note, infection with respiratory syncytial virus increases the production of IFNγ, provokes natural killer (NK) cell activation and exacerbation of inflammation in muscle and adipose tissues. Moreover, NK cell activity was found to be lower in patients with DM; glycated hemoglobin A1c (A1c) levels are associated with NK cell activity[17].
T2DM is a disease that often occurs and/or is related to obesity. Insulin resistance and related progression to overt diabetes are strongly associated with hypertrophy and hyperplasia of adipose cells[18]. According to the World Obesity Federation, obesity-related conditions seem to worsen the effects of COVID-19; indeed, the Centers for Disease Control and Prevention reported that “people with heart disease and diabetes are at higher risk of COVID-19 complications” and severe obesity (body mass index of ≥ 40) entails a higher risk for severe disease or death. As previously mentioned, COVID-19 favors an inflammatory environment that may progress to a “cytokine storm” (hypersecretion of inflammatory molecules: IL-2, IL-7, granulocyte-colony stimulating factor, IFN-γ inducible protein 10, monocyte chemo-attractant protein 1 [MCP1], macrophage inflammatory protein 1-α, and TNF-α). In an analogous fashion, obesity presents a state of low-grade inflammation, as a result of the secretion of inflammatory cytokines (TNF-α, IL-1, IL-6, IL-10), transforming growth factor-β, adipokines (leptin, resistin, adiponectin), MCP-1, C-X-C motif chemokine 5, hemostatic proteins (plasminogen activator inhibitor-1), proteins affecting blood pressure (angiotensinogen) and angiogenic molecules (vascular endothelial growth factor)[13]. Hypoxia and ischemia in adipose tissue and local endothelial damage lead to the production of reactive oxygen radicals (radical oxygen species, ROS) that affect both the microenvironment and macroenvironment of blood vessels[13].
Hyperglycemia and DM affect various target organs, including the vasculature. Obesity (and its concomitant inflammation) enables another mechanism via which COVID-19 can provoke damage., which is directly related to the microvascular complications of DM[13].
COVID-19 AND GLUCOSE METABOLISM
Hyperglycemia was observed in patients with SARS in 2003, caused by another coronavirus, closely related to COVID-19, SARS-CoV-1) possibly due to potential transient impairment of pancreatic islet cell function. Two more coronaviridae (’MERS-CoV’ and ‘HCoV-EMC’, causing Middle Eastern Respiratory Syndrome and human coronavirus;) attach to cells via dipeptidyl peptidase 4 (DPP-4, an enzyme that regulates insulin secretion)[19].
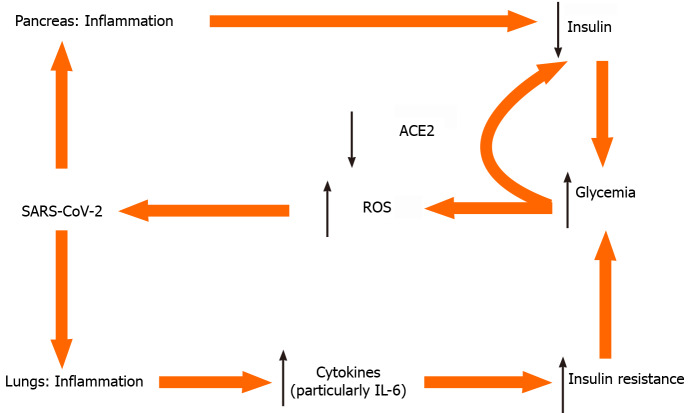
Glycemia on one hand is associated with SARS-CoV-2 replication[20]; elevated glucose levels and glycolysis increase SARS-CoV-2 replication and viral proliferation through the production of ROS (Figure 1). Notably, both T1DM and T2DM, are associated with a dysregulated immune response and increased morbidity and mortality[21]. On the other hand, in an inverse relationship, the presence of COVID-19 causes deterioration of glycemic control in already established DM. In a case series of critically ill, mostly not well-controlled patients with pre-existing T2DM (7 of 8 were on oral therapy before ICU admission), 85 to 480 units of insulin per day were needed to harness hyperglycemia[20].
Figure 1.
Selected tentative pathways for hyperglycemia in severe acute respiratory syndrome coronavirus 2 infection. ACE2: Angiotensin converting enzyme 2; IL-6: Interleukin 6; ROS: Radical oxygen species; SARS-CoV-2: Severe acute respiratory syndrome coronavirus 2.
The difference in diabetic ketoacidosis (DKA) rates in COVID-19 was four times higher in Black and two times higher in Hispanic patients with T1DM vs White patients with T1DM (but no statistical significance was documented)[22]. Potential explanations for these observations include the lower socioeconomic status of minority populations vis-à-vis that of the White population, the lack of appropriate nutrition and the lack of medical supervision in the use of insulin[22]. Although DKA is a major untoward event in T1DM, the majority of DKA cases with COVID-19, were observed in T2DM patients[22].
An initial diagnosis of DM was common in patients infected with SARS-COV-2, with neither any prior history of DM, nor using glucocorticoids. This new-onset hyperglycemia was an independent predictor for mortality[23] and was attributed to the binding of SARS-COV-2 to the angiotensin converting enzyme 2 (ACE2) receptor in pancreatic islets with concomitant local damage[23,24] (see also below). This ‘‘new-onset” hyperglycemia could be classified either as ‘‘stress-induced” hyperglycemia, as ‘‘new-onset DM” in previously unrecognized prediabetes, as hyperglycemia owing to the effects of SARS-CoV-2 to the pancreatic islets or as a result of ‘‘secondary DM”, following use of corticosteroids[23].
Quoting the definitions of the American Diabetes Association[25], new-onset hyperglycemia without DM is defined as fasting plasma glucose (FPG) between 5.6 mmol/L and 6.9 mmol/L (100-125 mg/dL) and/or A1c between 5.7% and 6.4%, in absence of such measurements in the past. New-onset DM is defined by either of FPG > 7.0 mmol/L (> 126 mg/dL) and/or an A1c > 6.5% and/or a random glucose > of 11.1 mmol/L (200 mg/dL)[25]. Thus, abnormal glucose measurements, in the absence of A1c > 6.5% could be expected, especially during this recent viral infection (that could not have affected the A1c levels yet). Several cases of hyperglycemia or new-onset DM in COVID-19 have been reported. As might be expected, COVID-19 has been associated with severe metabolic complications of already preexisting DM, including DKA and hyperosmolarity, necessitating high doses of insulin for glycemic control.
ACE2 is expressed in the respiratory system, in the intestines, kidneys, myocardium, vasculature and pancreatic islets. SARS-CoV-2 binds to ACE2, using it as a ligand for cell entry. Interestingly, ACE2-knockout mice are more vulnerable to β cell dysfunction[24], a fact that could explain why infection with SARS-CoV-2 can cause hyperglycemia in humans without preexisting DM. After endocytosis of the virus complex, ACE2 expression is downregulated, acting in a dual way. On one hand this impairs pancreatic islet cells’ function and causes β cell injury. On the other hand, downregulation of ACE2 Leads to unopposed angiotensin II action, which may further impair insulin secretion, by reducing blood flow and reducing insulin secretion while increasing oxidative stress in the pancreatic cell. Thus, coronaviruses might damage pancreatic islets, and give rise to hyperglycemia[24].
STUDIES REPORTING NEW-ONSET HYPERGLYCEMIA DUE TO COVID-19
Recently, a young 37-year-old patient with COVID-19 presented with all the clinical features of hyperglycemia and DKA, this being possibly the first case of new-onset DM secondary to COVID-19[26]. Another case of DKA precipitated by COVID-19 in a 54-year-old patient with newly diagnosed DM was also reported[27]. Since DKA occurs as a result of insulin deficiency, such observations give rise to questions regarding the potential effect of COVID-19 in this dangerous condition[27].
In a study by Li et al[28], among 658 hospitalized patients with confirmed COVID-19, 42 (6.4%) out of 658 patients presented with ketosis on admission with no obvious fever or diarrhea. Patients with ketosis were younger (median age 47.0 years vs 58.0 years; P = 0.003) and had a greater prevalence of fatigue (31.0% vs 10.6%; P < 0.001), DM (35.7% vs 18.5%; P = 0.007) and digestive disorders (31.0% vs 12.0%; P < 0.001). According to their data, COVID-19 infection caused ketosis or ketoacidosis, and induced DKA for patients with DM. Ketosis increased the length of hospital stay and mortality, while DM increased the length of hospital stay for patients with ketosis but had no effect on their mortality[28].
It remains to be determined whether, after resolution of COVID-19 symptoms, glucose levels are restored to normal, thus remitting the initial diagnosis of DM. To provide answers to this conundrum, a global registry of patients with COVID-19-related diabetes has been established (COVIDIAB Project)[29].
OUTCOME IN PATIENTS WITH NEW-ONSET HYPERGLYCEMIA WITHOUT DM VS NORMOGLYCEMIC COVID-19 PATIENTS
Hyperglycemia (two or more blood glucose measurements > 10 mmol/L or 180 mg/dL within any 24-h period with an A1C < 6.5%), regardless of the presence of DM, is related to an increase in COVID-19 mortality compared to normoglycemia[30]. Hyperglycemia without DM is further related to increased need for mechanical ventilation, to need for ICU hospitalization and to mortality[30]. In the same gist, complications within the first month of hospital stay were increased in hyperglycemic patients without DM[31], resulting in a higher all-cause mortality[32]. Hyperglycemia at admission (but without confirmed DM) was related to a 71% increase in mortality in 1317 patients[33].
When hyperglycemia without the presence of DM was compared to known DM (new-onset and/or preexisting DM) in COVID-19 patients, a significant increase in mortality was observed among 271 patients with new-onset hyperglycemia without DM, compared to pre-existing DM. Nevertheless, ICU admission did not seem to differ significantly[34]. Critically and non-critically ill COVID-19 patients sometimes present with higher-than-expected glycemia, even in the absence of DM. Regarding the direct association of glycemia in already admitted patients in ICU due to COVID-19 infection, hyperglycemia was noted in 20 of 36 patients. Among those, none had a prior history of DM and the incidence of hyperglycemia proved to be higher that would be expected in an ICU due to stress-induced responses[35]. In a series of 157 patients with COVID-19, a substantial number of patients with and without DM presented with hyperglycemia upon admission, while critically ill patients showed compromised insulin secretion and/or impaired sensitivity to insulin[36].
OUTCOME IN PATIENTS WITH NEW-ONSET DM VS NORMOGLYCEMIC COVID-19
Νew-onset DM (and/or DKA) has been reported to occur in 16% to 21% of COVID-19 cases[26], but the incidence of complications, need for ICU and intubation, varies among studies (n = 413), with some showing an increase and others no difference, compared to normoglycemic patients[37,38].
OUTCOME IN PATIENTS WITH NEW-ONSET DM VS PRE-EXISTING DM
The risk of all-cause death in COVID-19 patients with new-onset DM is nearly double compared to that of patients with pre-existing DM[38]. A statistically significant association of ICU admission and/or of mortality in COVID-19 patients with new-onset DM (37%), compared to patients with pre-existing DM (20%) was noted; this association persisted after adjustment for age and gender[38].
Summing up the available literature, COVID-19 patients with new-onset hyperglycemia, even without a frank diagnosis of DM due to any cause (stress-induced/COVID-19-induced/pre-existing dysglycemia), show a worse course of the disease, higher rate of complications and all-cause mortality when compared to normoglycemic or patients with DM.
TREATMENT FOR COVID-19 AND GLYCEMIA
In published reports, COVID-19 patients with hyperglycemia/secondary DM were usually treated effectively with insulin[33]. In early reports, patients were also treated with hydroxychloroquine[33]. The latter medication is known to increase endogenous insulin secretion[39]. Since the use of hydroxychloroquine for SARS-CoV-2 was—at least—controversial, and has been phased out, hyperglycemia may be seen more often in patients with SARS-CoV-2 (with or without DM). Possibly higher insulin dosage—than expected—may be needed. Hyperglycemia is also to be expected by the widespread use of dexamethasone in COVID-19 patients, per the newer treatment protocols[40-46].
CONCLUSION
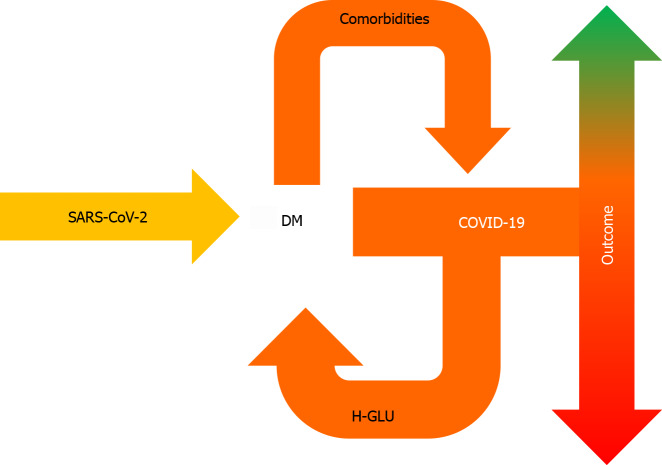
Hyperglycemia in COVID-19, irrespective of insulin resistance or history of DM, is a portent of worse prognosis (Figure 2). Further studies will help to elucidate the link between glycemia and COVID-19.
Figure 2.
Pre-existing diabetes mellitus can aggravate coronavirus disease 2019, following severe acute respiratory syndrome coronavirus 2 infection, whereas coronavirus disease 2019 can lead to hyperglycemia — even in the absence of diabetes mellitus, which is associated with worse prognosis. DM: Diabetes mellitus; COVID-19: Coronavirus disease 2019; H-GLU: Hyperglycemia; SARS-CoV-2: Severe acute respiratory syndrome coronavirus 2.
Footnotes
Conflict-of-interest statement: Nothing to declare.
Manuscript source: Invited manuscript
Peer-review started: January 19, 2021
First decision: February 12, 2021
Article in press: April 20, 2021
Specialty type: Endocrinology and metabolism
Country/Territory of origin: Greece
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Cure E, Vunnam RR S-Editor: Gao CC L-Editor: Filipodia P-Editor: Ma YJ
Contributor Information
Konstantinos Michalakis, Private Practice, Athens GR-11521, Greece.
Ioannis Ilias, Department of Endocrinology, Diabetes and Metabolism, Elena Venizelou Hospital, Athens GR-11521, Greece. iiliasmd@yahoo.com.
References
- 1.World Health Organization. Coronavirus disease (Covid-19) Pandemic. [cited 15 January 2021]. In: World Health Organization [Internet]. Available from: https://wwwwhoint/emergencies/diseases/novel-coronavirus-2019 .
- 2.Fadini GP, Morieri ML, Longato E, Avogaro A. Prevalence and impact of diabetes among people infected with SARS-CoV-2. J Endocrinol Invest. 2020;43:867–869. doi: 10.1007/s40618-020-01236-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, Wu Y, Zhang L, Yu Z, Fang M, Yu T, Wang Y, Pan S, Zou X, Yuan S, Shang Y. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8:475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, Castelli A, Cereda D, Coluccello A, Foti G, Fumagalli R, Iotti G, Latronico N, Lorini L, Merler S, Natalini G, Piatti A, Ranieri MV, Scandroglio AM, Storti E, Cecconi M, Pesenti A COVID-19 Lombardy ICU Network. Baseline Characteristics and Outcomes of 1591 Patients Infected With SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323:1574–1581. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Holman N, Knighton P, Kar P, O'Keefe J, Curley M, Weaver A, Barron E, Bakhai C, Khunti K, Wareham NJ, Sattar N, Young B, Valabhji J. Risk factors for COVID-19-related mortality in people with type 1 and type 2 diabetes in England: a population-based cohort study. Lancet Diabetes Endocrinol. 2020;8:823–833. doi: 10.1016/S2213-8587(20)30271-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, Satlin MJ, Campion TR Jr, Nahid M, Ringel JB, Hoffman KL, Alshak MN, Li HA, Wehmeyer GT, Rajan M, Reshetnyak E, Hupert N, Horn EM, Martinez FJ, Gulick RM, Safford MM. Clinical Characteristics of Covid-19 in New York City. N Engl J Med. 2020;382:2372–2374. doi: 10.1056/NEJMc2010419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Myers LC, Parodi SM, Escobar GJ, Liu VX. Characteristics of Hospitalized Adults With COVID-19 in an Integrated Health Care System in California. JAMA. 2020;323:2195–2198. doi: 10.1001/jama.2020.7202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Piva S, Filippini M, Turla F, Cattaneo S, Margola A, De Fulviis S, Nardiello I, Beretta A, Ferrari L, Trotta R, Erbici G, Focà E, Castelli F, Rasulo F, Lanspa MJ, Latronico N. Clinical presentation and initial management critically ill patients with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in Brescia, Italy. J Crit Care. 2020;58:29–33. doi: 10.1016/j.jcrc.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barron E, Bakhai C, Kar P, Weaver A, Bradley D, Ismail H, Knighton P, Holman N, Khunti K, Sattar N, Wareham NJ, Young B, Valabhji J. Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. Lancet Diabetes Endocrinol. 2020;8:813–822. doi: 10.1016/S2213-8587(20)30272-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tang Y, Liu J, Zhang D, Xu Z, Ji J, Wen C. Cytokine Storm in COVID-19: The Current Evidence and Treatment Strategies. Front Immunol. 2020;11:1708. doi: 10.3389/fimmu.2020.01708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lim S, Bae JH, Kwon HS, Nauck MA. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol. 2021;17:11–30. doi: 10.1038/s41574-020-00435-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Michalakis K, Panagiotou G, Ilias I, Pazaitou-Panayiotou K. Obesity and COVID-19: A jigsaw puzzle with still missing pieces. Clin Obes. 2021;11:e12420. doi: 10.1111/cob.12420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Michalakis K, Ilias I. SARS-CoV-2 infection and obesity: Common inflammatory and metabolic aspects. Diabetes Metab Syndr. 2020;14:469–471. doi: 10.1016/j.dsx.2020.04.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zeng Z, Yu H, Chen H, Qi W, Chen L, Chen G, Yan W, Chen T, Ning Q, Han M, Wu D. Longitudinal changes of inflammatory parameters and their correlation with disease severity and outcomes in patients with COVID-19 from Wuhan, China. Crit Care. 2020;24:525. doi: 10.1186/s13054-020-03255-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Böni-Schnetzler M, Meier DT. Islet inflammation in type 2 diabetes. Semin Immunopathol. 2019;41:501–513. doi: 10.1007/s00281-019-00745-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tsalamandris S, Antonopoulos AS, Oikonomou E, Papamikroulis GA, Vogiatzi G, Papaioannou S, Deftereos S, Tousoulis D. The Role of Inflammation in Diabetes: Current Concepts and Future Perspectives. Eur Cardiol. 2019;14:50–59. doi: 10.15420/ecr.2018.33.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schmidt MI, Duncan BB. Diabesity: an inflammatory metabolic condition. Clin Chem Lab Med. 2003;41:1120–1130. doi: 10.1515/CCLM.2003.174. [DOI] [PubMed] [Google Scholar]
- 19.Ilias I, Zabuliene L. Hyperglycemia and the novel Covid-19 infection: Possible pathophysiologic mechanisms. Med Hypotheses. 2020;139:109699. doi: 10.1016/j.mehy.2020.109699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu L, Girgis CM, Cheung NW. COVID-19 and diabetes: Insulin requirements parallel illness severity in critically unwell patients. Clin Endocrinol (Oxf) 2020;93:390–393. doi: 10.1111/cen.14288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Codo AC, Davanzo GG, Monteiro LB, de Souza GF, Muraro SP, Virgilio-da-Silva JV, Prodonoff JS, Carregari VC, de Biagi Junior CAO, Crunfli F, Jimenez Restrepo JL, Vendramini PH, Reis-de-Oliveira G, Bispo Dos Santos K, Toledo-Teixeira DA, Parise PL, Martini MC, Marques RE, Carmo HR, Borin A, Coimbra LD, Boldrini VO, Brunetti NS, Vieira AS, Mansour E, Ulaf RG, Bernardes AF, Nunes TA, Ribeiro LC, Palma AC, Agrela MV, Moretti ML, Sposito AC, Pereira FB, Velloso LA, Vinolo MAR, Damasio A, Proença-Módena JL, Carvalho RF, Mori MA, Martins-de-Souza D, Nakaya HI, Farias AS, Moraes-Vieira PM. Elevated Glucose Levels Favor SARS-CoV-2 Infection and Monocyte Response through a HIF-1α/Glycolysis-Dependent Axis. Cell Metab 2020; 32: 437-446. :e5. doi: 10.1016/j.cmet.2020.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ebekozien O, Agarwal S, Noor N, Albanese-O'Neill A, Wong JC, Seeherunvong T, Sanchez J, DeSalvo D, Lyons SK, Majidi S, Wood JR, Acharya R, Aleppo G, Sumpter KM, Cymbaluk A, Shah NA, Van Name M, Cruz-Aviles L, Alonso GT, Gallagher MP, Sanda S, Feuer AJ, Cossen K, Rioles N, Jones NY, Kamboj MK, Hirsch IB. Inequities in Diabetic Ketoacidosis Among Patients With Type 1 Diabetes and COVID-19: Data From 52 US Clinical Centers. J Clin Endocrinol Metab. 2021;106:e1755–e1762. doi: 10.1210/clinem/dgaa920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Singh AK, Singh R. Hyperglycemia without diabetes and new-onset diabetes are both associated with poorer outcomes in COVID-19. Diabetes Res Clin Pract. 2020;167:108382. doi: 10.1016/j.diabres.2020.108382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Underwood PC, Adler GK. The renin angiotensin aldosterone system and insulin resistance in humans. Curr Hypertens Rep. 2013;15:59–70. doi: 10.1007/s11906-012-0323-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2020. Diabetes Care. 2020;43:S14–S31. doi: 10.2337/dc20-S002. [DOI] [PubMed] [Google Scholar]
- 26.Chee YJ, Ng SJH, Yeoh E. Diabetic ketoacidosis precipitated by Covid-19 in a patient with newly diagnosed diabetes mellitus. Diabetes Res Clin Pract. 2020;164:108166. doi: 10.1016/j.diabres.2020.108166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Heaney AI, Griffin GD, Simon EL. Newly diagnosed diabetes and diabetic ketoacidosis precipitated by COVID-19 infection. Am J Emerg Med 2020; 38: 2491.e3-2491. :e4. doi: 10.1016/j.ajem.2020.05.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li J, Wang X, Chen J, Zuo X, Zhang H, Deng A. COVID-19 infection may cause ketosis and ketoacidosis. Diabetes Obes Metab. 2020;22:1935–1941. doi: 10.1111/dom.14057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rubino F, Amiel SA, Zimmet P, Alberti G, Bornstein S, Eckel RH, Mingrone G, Boehm B, Cooper ME, Chai Z, Del Prato S, Ji L, Hopkins D, Herman WH, Khunti K, Mbanya JC, Renard E. New-Onset Diabetes in Covid-19. N Engl J Med. 2020;383:789–790. doi: 10.1056/NEJMc2018688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang Y, Li H, Zhang J, Cao Y, Zhao X, Yu N, Gao Y, Ma J, Zhang H, Guo X, Liu X. The clinical characteristics and outcomes of patients with diabetes and secondary hyperglycaemia with coronavirus disease 2019: A single-centre, retrospective, observational study in Wuhan. Diabetes Obes Metab. 2020;22:1443–1454. doi: 10.1111/dom.14086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang S, Ma P, Zhang S, Song S, Wang Z, Ma Y, Xu J, Wu F, Duan L, Yin Z, Luo H, Xiong N, Xu M, Zeng T, Jin Y. Fasting blood glucose at admission is an independent predictor for 28-day mortality in patients with COVID-19 without previous diagnosis of diabetes: a multi-centre retrospective study. Diabetologia. 2020;63:2102–2111. doi: 10.1007/s00125-020-05209-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li H, Tian S, Chen T, Cui Z, Shi N, Zhong X, Qiu K, Zhang J, Zeng T, Chen L, Zheng J. Newly diagnosed diabetes is associated with a higher risk of mortality than known diabetes in hospitalized patients with COVID-19. Diabetes Obes Metab. 2020;22:1897–1906. doi: 10.1111/dom.14099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sardu C, D'Onofrio N, Balestrieri ML, Barbieri M, Rizzo MR, Messina V, Maggi P, Coppola N, Paolisso G, Marfella R. Hyperglycaemia on admission to hospital and COVID-19. Diabetologia. 2020;63:2486–2487. doi: 10.1007/s00125-020-05216-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Coppelli A, Giannarelli R, Aragona M, Penno G, Falcone M, Tiseo G, Ghiadoni L, Barbieri G, Monzani F, Virdis A, Menichetti F, Del Prato S Pisa COVID-19 Study Group. Hyperglycemia at Hospital Admission Is Associated With Severity of the Prognosis in Patients Hospitalized for COVID-19: The Pisa COVID-19 Study. Diabetes Care. 2020;43:2345–2348. doi: 10.2337/dc20-1380. [DOI] [PubMed] [Google Scholar]
- 35.Ilias I, Jahaj E, Kokkoris S, Zervakis D, Temperikidis P, Magira E, Pratikaki M, Vassiliou AG, Routsi C, Kotanidou A, Dimopoulou I. Clinical Study of Hyperglycemia and SARS-CoV-2 Infection in Intensive Care Unit Patients. In Vivo. 2020;34:3029–3032. doi: 10.21873/invivo.12136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ilias I, Diamantopoulos A, Pratikaki M, Botoula E, Jahaj E, Athanasiou N, Tsipilis S, Zacharis A, Vassiliou AG, Vassiliadi DA, Kotanidou A, Tsagarakis S, Dimopoulou I. Glycemia, Beta-Cell Function and Sensitivity to Insulin in Mildly to Critically Ill Covid-19 Patients. Medicina (Kaunas) 2021;57 doi: 10.3390/medicina57010068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yang JK, Jin JM, Liu S, Bai P, He W, Wu F, Liu XF, Chai ZL, Han DM. New onset COVID-19–related diabetes: an indicator of mortality. 2020 Preprint. Available from: medRxiv:2020.2004.2008.20058040.
- 38.Fadini GP, Morieri ML, Boscari F, Fioretto P, Maran A, Busetto L, Bonora BM, Selmin E, Arcidiacono G, Pinelli S, Farnia F, Falaguasta D, Russo L, Voltan G, Mazzocut S, Costantini G, Ghirardini F, Tresso S, Cattelan AM, Vianello A, Avogaro A, Vettor R. Newly-diagnosed diabetes and admission hyperglycemia predict COVID-19 severity by aggravating respiratory deterioration. Diabetes Res Clin Pract. 2020;168:108374. doi: 10.1016/j.diabres.2020.108374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ilias I, Koukkou E. Hyperglycémie, hydroxychloroquine et SARS-CoV-2. La Presse Medicale Formation. 2021:In press. [Google Scholar]
- 40.Abubakar AR, Sani IH, Godman B, Kumar S, Islam S, Jahan I, Haque M. Systematic Review on the Therapeutic Options for COVID-19: Clinical Evidence of Drug Efficacy and Implications. Infect Drug Resist. 2020;13:4673–4695. doi: 10.2147/IDR.S289037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ahmed MH, Hassan A. Dexamethasone for the Treatment of Coronavirus Disease (COVID-19): a Review. SN Compr Clin Med. 2020:1–10. doi: 10.1007/s42399-020-00610-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ismaila MS, Bande F, Ishaka A, Sani AA, Georges K. Therapeutic options for COVID-19: a quick review. J Chemother. 2021;33:67–84. doi: 10.1080/1120009X.2020.1868237. [DOI] [PubMed] [Google Scholar]
- 43.Wahab S, Ahmad I, Usmani S, Ahmad MP. Epidemiological Situation and Efficacy of Dexamethasone for the treatment planning of COVID-19: A perspective review. Curr Drug Deliv. 2020 doi: 10.2174/1567201817666201006144008. [DOI] [PubMed] [Google Scholar]
- 44.Tortajada C, Colomer E, Andreu-Ballester JC, Esparcia A, Oltra C, Flores J. Corticosteroids for COVID-19 patients requiring oxygen support? J Med Virol. 2021;93:1817–1823. doi: 10.1002/jmv.26635. [DOI] [PubMed] [Google Scholar]
- 45.Miklowski M, Jansen B, Auron M, Whinney C. The hospitalized patient with COVID-19 on the medical ward: Cleveland Clinic approach to management. Cleve Clin J Med. 2020 doi: 10.3949/ccjm.87a.ccc064. [DOI] [PubMed] [Google Scholar]
- 46.Vallianou NG, Evangelopoulos A, Kounatidis D, Stratigou T, Christodoulatos GS, Karampela I, Dalamaga M. Diabetes mellitus and SARS-CoV-2 infection: pathophysiologic mechanisms and implications in management. Curr Diabetes Rev. 2020 doi: 10.2174/1573399817666210101110253. [DOI] [PubMed] [Google Scholar]