Abstract
Catheter‐based renal sympathetic denervation (RSD) is a novel technique that is being investigated as treatment for resistant hypertension. To systematically evaluate the existing literature on the safety and efficacy of RSD in persons with resistant hypertension, online searches of Medline and the Cochrane Library Database (up to June 2012) were performed. Randomized controlled trials, observational studies, and conference proceedings published in English language were included. Nineteen studies (N=683 persons) were included. Follow‐up duration ranged from 1 to 24 months. All studies reported significant reductions in systolic and diastolic pressures. Maximal reduction of blood pressure ranged from 18 mm Hg to 36 mm Hg (systolic) and 9 mm Hg to 15 mm Hg (diastolic). Sustained benefit of blood pressure reduction at 12 months was seen in 5 studies. No worsening of renal function was reported and there were few procedure‐related adverse events such as pseudoaneurysm formation, hypotension, and bradycardia. Data from short‐term studies suggest that RSD is a safe and effective therapeutic option in carefully selected patients with resistant hypertension. Long‐term studies with large patient populations are needed to study whether this benefit is sustained with a demonstrable difference in cardiovascular disease event rates.
Resistant hypertension is defined as the failure to achieve a goal blood pressure (BP) in persons adhering to full doses of a 3‐drug antihypertensive regimen that includes a diuretic. 1 The prevalence of resistant hypertension is difficult to estimate due to nonadherence and/or an inadequate treatment regimen but is reported to vary from 8% to 20%. 1 , 2 , 3 , 4 , 5 A retrospective cohort study of 205,750 incident hypertensive patients from two health plans within the Cardiovascular Research Network Hypertension Registry estimated a 2% incidence of resistant hypertension after 18 months of office BP measurements. 6 Historically, nonselective surgical sympathectomy (splanchnicectomy) was performed in patients with malignant hypertension. This reduced sympathetic outflow to the kidneys, increased natriuresis and diuresis, and decreased renin release, without adversely affecting other functions of the kidney. 7 However, this was associated with significant postsurgical morbidity. 8 This concept is now being revisited using catheter‐based endovascular renal artery sympathetic denervation. We performed a systematic review to analyze the available data and determine the safety and efficacy of this procedure in the treatment of resistant hypertension.
Methods
Data Sources and Searches
We searched MEDLINE (2000 to June 2012) and the Cochrane Library (inception to June 2012) using keywords and/or medical subject headings (MeSH) for “resistant hypertension” and “renal sympathetic denervation.” A sample search strategy is shown in Table I. The American College of Cardiology, the American Society of Nephrology, and Google Scholar databases were also searched for conference proceedings and presentations. Screening of titles and abstracts of the identified references was followed by full‐text review by two independent reviewers (PG and RA).
Table I.
Sample Search Strategy in OVID MEDLINE
| 1. Hypertension/or hypertension.mp |
| 2. (resistant adj hypertension).mp |
| 3. (blood adj pressure).mp |
| 4. or/1–3 |
| 5. (renal adj sympathetic adj denervation).mp |
| 6. Sympathectomy/ |
| 7. Kidney/ |
| 8. 6 and 7 |
| 9. 5 or 8 |
| 10. 4 and 9 |
| 11. Limit 10 to English language |
Study Selection
Randomized controlled trials (RCTs), observational studies, case series, and conference presentations published in the English language were included for review. Studies with <5 patients were excluded.
Data Extraction and Synthesis
Data from the primary studies were extracted by one reviewer (PG) and verified by another reviewer (PSG) for accuracy. Baseline characteristics of the patients, including age, sex, body mass index, number of preprocedural antihypertensive drugs, and use of diuretics were recorded. Our primary outcome was change in office recordings of systolic and diastolic BP. Secondary outcomes were maintenance of reduction in BP during the study period, reduction in the need for antihypertensive medications, and changes in renal function defined by estimated glomerular filtration rate (eGFR), serum creatinine, and/or urinary protein excretion. Periprocedural complications were also reviewed. Meta‐analyses could not be performed because of the heterogeneity in study design and inconsistent reporting of the standard deviation in the BP readings.
Results
Our initial literature search yielded 133 articles. Screening of titles and abstracts followed by full‐text screening yielded 19 studies that met inclusion criteria (Figure).
Figure.
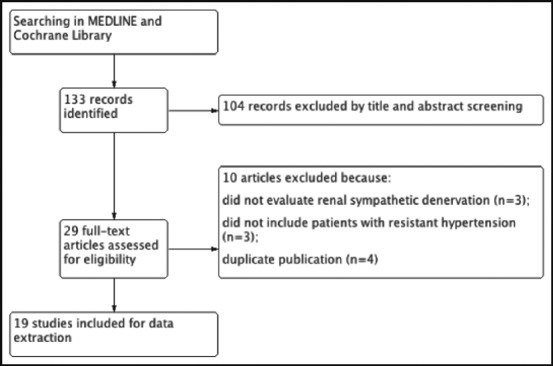
Flow diagram for selection of included studies.
Study Characteristics
The 19 studies included a total of 683 patients (excluding patients who were part of more than one study). 9 , 10 , 11 Of these, there were 2 RCTs, 11 , 12 4 case‐control studies, 9 , 10 , 13 , 14 and 13 case series 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 , 27 (Table II). The standard definition of resistant hypertension was used in all studies. Patients with renal artery abnormalities (eg, short main renal artery, severe renal artery stenosis, previous renal stenting or angioplasty, and multiple renal arteries) and identifiable causes of secondary hypertension were excluded. Patients with hemodynamically significant valvular disease, type I diabetes mellitus, implanted pacemakers, or implantable cardioverter‐defibrillators; pregnant patients; or those taking clonidine, moxonidine, rilmenidine, or warfarin were also excluded in most studies. In the 2 case series by Prochnau, 17 , 23 4 of 12 and 12 of 30 patients, respectively, had serum creatinine >1.4 mg/dL. Another study of 15 patients included only those with an eGFR <45 mL/min/1.73 m2. 25 The follow‐up intervals ranged from 2 weeks to 24 months.
Table II.
Overview of Studies of Renal Sympathetic Denervation for Resistant Hypertension
| Study/Location/Funding Source | Design | Patient Characteristics | Intervention Description | Follow‐Up Duration: No. |
|---|---|---|---|---|
| Ukena (2011) 9 Location: Germany Funding Source: Not declared | Randomized controlled (N=37) | Mean age: 59.1±9.4; women: 32%; BMI: 31.8±5.2; DM: 43%; eGFR: 70±24; diuretics: 89%; average number of antihypertensive medications: 5.9±1.4 | Patients recruited from Symplicity HTN‐2 trial. Methods of denervation not specified separately | 3 mo: no loss to follow‐up reported |
| Control (N=9) | Mean age: 64.9±6.4 y; women: 21%; BMI: 30.2±4.6; DM: 22%; eGFR: 64.5±16; diuretics: 78%; average number of antihypertensive medications: 5.0±1.2 | 3 mo: no loss to follow‐up reported | ||
| Symplicity HTN‐2 (2010) 10 Location: Australia Funding Source: Ardian Inc | Randomized controlled (N=52) | Mean age: 58±12 y; women: 35%; BMI: 31±5; DM: 40%; eGFR: 77±19; diuretics: 89% (aldosterone antagonist: 17%); average number of antihypertensive medications: 5.2±1.5 | 4–6 Ablations on bilateral renal arteries, duration of ≤2 min at a maximum power of 8 W. Heparin used to maintain activated clotting time of >250 s | 6 mo: 49 renal Duplex imaging (6 months): 37 MRI (6 mo): 5 CTA (6 mo): 5 |
| Control (N=54) | Mean age: 58±12 y; women: 50%; BMI: 31±5; DM: 28%; eGFR: 86±20; diuretics: 89% (aldosterone antagonist: 17%); average number of antihypertensive medications: 5.2±1.5 | 6 mo: 51 | ||
| Mahfoud (2012) 14 Location: Germany Funding source: First author supported by Deutsche Forschungsgemeinschaft (KFO 196) | Case control (N=88) | Mean age: 61.6±1.1 y; women: 39%; BMI: 29.69±0.8; DM: 17%; cystatin C GFR (mL/min): 84.6±3.6; Diuretics: 100% (aldosterone antagonists: 33%); average number of antihypertensive medications: 5.8±0.2 | ≤8 Ablations for 2 min each in each renal artery. Heparin used to maintain activated clotting time of >250 s | 3 mo; 6 mo: no loss to follow‐up reported |
| Control (N=12) | Mean age: 61.9±3.6 y; women: 42%; BMI: 28.1±1.9; DM: 33%; cystatin C GFR: 97.9±7.4; diuretics: 100% (aldosterone antagonists: 33%); average number of antihypertensive medications: 4.9±0.3 | 3 mo; 6 mo: no loss to follow‐up reported | ||
| Brandt (2012) 13 Location: Germany Funding source: Not declared | Case‐control (N=46) | Mean age: 63.1±10.2 y; women: 33%; BMI: 28.6±3.4; DM: 46%; eGFR: 83.5±27.8; diuretics: 100%; average number of antihypertensive medications: 4.7±0.5 | ≤6 Ablations at 8 W for 2 min each were performed in both renal arteries | 1 mo; 6 mo no loss to follow‐up reported |
| Controls (N=18) | Mean age: 63.0±15.3 y; women: 39%; BMI: 28.1±3.8; DM: 39%; eGFR: 80.5±29.3; diuretics: 100%; average number of antihypertensive medications: 4.8±2.5 | 1 mo; 6 mo No loss to follow‐up reported | ||
| Mahfoud (2011) 12 Location: Germany Funding Source: Ardian Inc | Case‐control (N=37) | Mean age: 58.7±1.6 y; women: 21%; BMI: 31.3± 0.9; DM: 35%; eGFR: 75.1±3.3; diuretics: 100%; average number of antihypertensive medications: 5.8±0.2 | 6 Ablations on bilateral renal arteries, with duration of 2 min at a maximum power of 8 W. Catheter tip impedance and temperature were constantly monitored. Heparin used to maintain activated clotting time of >250 s | 1 mo: 37 3 mo: 37 |
| Controls (N=13) | Mean age: 62.5±2.9 y; women: 38%; BMI: 30.7±1.7; DM: 54%; eGFR: 81±7.6; diuretics: 100%; average number of antihypertensive medications: 5.0±0.4 | 1 mo: 13 3 mo: 13 | ||
| Krum (2009) 11 Location: Australia Funding Source: Ardian Inc | Case‐control (N=45) | Mean age: 58±9 y; women: 44%; BMI: not reported; DM: 31%; eGFR: 81±23; diuretics: 96%; average number of antihypertensive medications: 4.7±1.5 | 6 Ablations on bilateral renal arteries with a maximum duration of 2 min and energy of 8 W. Heparin used to maintain activated clotting time of >250 s | 1 mo: 41 3 mo: 39 6 mo: 26 9 mo: 20 12 mo: 9 angiograms (14–30 d): 18 magnetic resonance angiography (6 mo): 14 |
| N (controls)=5 | Mean age: 51±8 y; women: 20%; BMI: not reported; DM: 40%; eGFR: 95±15; diuretics: 60%; average number of antihypertensive medications: 4.6±0.5 | 1 mo: 5 3 mo: 5 6 mo: 5 9 mo: 5 12 mo: 5 | ||
| Bauer (2012) 15 Location: Germany Funding source: Not declared | Case series (N=11) | Mean age: 68.9±7 y; average number of antihypertensive medications: 5.6±2.1 | Not available | 6 mo: 11 |
| Hering (2012) 25 Location: Australia Funding Source: National Health and Research Council of Australia and the Victoria Government’s Operational Infrastructure Support Program | Case series (N=15) | Mean age: 61±9 y; women: 40%; BMI: 33±8; DM: 73%; eGFR: 31.2±8.9; diuretics: 100%; average number of antihypertensive medications: 5.6±1.3 | Average of 9.9±1.5 ablation treatments using a predetermined treatment protocol as in Symplicity HTN‐1 trial | 3 months: 15 6 mo: 8 12 mo: 5 Angiography (immediately after procedure): 15 |
| Himmel (2012) 18 Location: Germany Funding Source: Not declared | Case series (N=14) | Not specified | Not described | 1 mo: 14 3 mo: 14 |
| Mabin (2012) 16 Location: South Africa Funding source: ReCor Medical | Case series (N=11) | Mean age: 55±14 y; women: 64%; BMI: not reported; DM: 27%; eGFR: not reported; diuretics: 100%; average number of antihypertensive medications: 4.5 | ≤3 Ablations on each renal artery using PARADISE catheter (ReCor Medical) with energy of up to 25–30 W for upto 50 s. Heparin used to maintain activated clotting time of >250 s | Immediate: 11 2 wk: 11 1 mo: 11 2 mo: 8 3 mo: 8 |
| Mylotte (2012) 26 Location: France Funding source: Not declared | Case series (N=35) | Mean age: 63.6±11.7 y; women: 36.5%; BMI: not reported; DM: 36.4%; eGFR <60 mL/min: 15.2%; diuretics: not reported; average number of antihypertensive medications: 4.6±1.0 | Not available | 6 mo: 35 |
| Prochnau (2012) 17 Location: Germany Funding Source: Not declared | Case series (N=30) | Mean age: 62.4±12.8 y; women: 33%; BMI: 32.4±6.8; DM: 50%; renal insufficiency (S Cr >1.4): 40%; diuretics: 97%; average number of antihypertensive medications: 6 | 6 Ablations along the length of each renal artery, with maximum of 8–13 W under continuously monitoring of impedance and temperature. Standard EP catheter (Marinr; Medtronic Inc, MN) used | 1 mo: 30 3 mo: 30 6 mo: 20 12 mo: 10 Renal ultrasound (3 and 6 mo): 20 |
| Prochnau (2012) 23 Location: Germany Funding Source: Not declared | Case series (N=12) | Mean age: 62±14.3 y; women: 25%; BMI: not reported; DM: 50%; renal insufficiency (S Cr >1.4): 33.3%; diuretics: 92%; mean number of antihypertensive medications: 5.5 | 6 Ablations on bilateral renal arteries with a maximum duration of 1 min and energy of 8–13 W. Standard EP catheter (Marinr; Medtronic Inc, MN) used | 1 mo: 12 3 mo: 12 Renal duplex scan (1 and 3 mo): 12 |
| Vase (2012) 24 Location: Denmark Funding Source: Not declared | Case series (N=9) | Mean age: 56±10 y; women: 67%; BMI: 27.5±4.7; DM: 22%; eGFR: 78±13; diuretics: 100%; mean number of antihypertensive medications: 5.4±1.4 | 4–6 Ablations on each renal artery with energy of approximately 8 W lasting 2 min each Heparin used to maintain activated clotting time of >250 s | 1 mo: 9 |
| Verloop (2012) 27 Location: The Netherlands Funding Source: Not declared | Case series (N=22) | Average number of antihypertensive medications: 3.32±1.62 | Not available | 6 mo: No loss to follow‐up reported |
| Simonetti (2011) 20 Location: Italy Funding Source: Not declared | Case series (N=5) | Mean age: 50.6 y; women: 40%; BMI: not reported; DM: not reported; eGFR: 92.5; diuretics: 100%; average number of antihypertensive medications: 5 | At least 6 ablations on bilateral renal artery, with duration of 2 min at a maximum power of 8 W | 1 mo: 5 2 mo: 5 CTA (2 mo): 5 |
| Symplicity HTN‐1 (2011) 19 Location: Australia, Europe and United States Funding Source: Ardian Inc | Case series (N=153) | Mean age: 57±11 y; women: 39%; BMI: not reported; DM: 31%; eGFR: 83±20; diuretics: 95% (aldosterone blocker: 22%); average number of antihypertensive medications: 5.1±1.4 | 4–6 Ablations on bilateral renal arteries, duration of ≤2 minutes at a maximum power of 8 W. Heparin used to maintain activated clotting time of >250 s | 1 month: 138 3 months: 135 6 months: 86 12 months: 64 18 mo: 36 24 mo: 18 CTA/MRI/Renal Duplex imaging (6 mo): 81 |
| Voskuil (2011) 21 Location: The Netherlands Funding Source: Not declared | Case series (N=11) | Mean age: 68±12 y; women: 91%; BMI: 30±8; DM: 18%; eGFR: 74±14; diuretics: 45%; Average number of antihypertensive medications: 3.1±1.5 | 4–6 Treatments of bilateral renal arteries, (distal to proximal circumferentially) with duration of 2 minutes and energy of approximately 8 W | 1 mo: 11 Angiography (immediately after procedure): 11 IVUS (immediately after procedure): 3 |
| Witkowsky (2011) 22 Location: Poland Funding Source: Ardian Inc | Case series (N=12) | Mean age: 49.5 y; women: 30%; BMI: 30.9; DM: 40%; eGFR: 81.3; diuretics: 100%; average number of antihypertensive medications: 5 | ≤6 Ablations separated both longitudinally and rotationally on bilateral renal arteries with duration lasting ≤2 min and energy of ≤8 W | 3 mo: 10 6 mo: 10 Renal duplex scan (6 mo): 10 |
Abbreviations: BMI, body mass index; CTA, computed tomographic angiography; DM, diabetes mellitus; eGFR, estimated glomerular filtration rate; IVUS, intravascular ultrasonography; MRI, magnetic resonance imaging; S Cr, serum creatinine.
Of the 19 studies, 5 were funded by Ardian Inc, the manufacturer of the SYMPLICITY catheter, while one was funded by ReCor Medical, the manufacturer of the PARADISE catheter. Another study was funded by the National Health and Research Council of Australia. Other studies did not report a source of funding.
The studies included 30% to 50% women and one study included 91% women. 21 The age of the patients ranged between 50 and 70 years across all studies.
The method of measuring BP varied across studies. In 2 studies, BP was measured noninvasively in the sitting position in triplicate and then averaged, 22 , 25 while in 4 studies it was performed according to the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure guidelines, 9 , 10 , 12 , 13 , 19 and in one study it was measured by blinded, experienced physicians. 11 Two studies performed 24‐hour dynamic arterial pressure monitoring. 20 , 23 Ten studies measured ambulatory BP at baseline and at follow‐up. 12 , 15 , 16 , 17 , 20 , 22 , 24 , 25 , 26
Baseline recordings of systolic BP ranged between 167 and 203 mm Hg and of diastolic BP 94 and 109 mm Hg.
Outcomes
Change in BP. The 2 RCTs reported a significant decrease in BP among those undergoing renal denervation as compared with controls. Ukena and colleagues showed a decrease in BP of −31/−9 mm Hg at 3 months compared with 0/−1 in the control group. 9 The investigators in the Symplicity HTN‐2 trial reported an absolute decrease of −32/−12 mm Hg at 6 months 12 (compared with +1/0 in the control group) and −28/−9 at 1‐year follow‐up. 28 The earliest reduction in BP was reported immediately after the procedure and at 2 weeks. 16 Krum and colleagues followed patients up to 1 year and showed a sustained decrease of −27/−17 mm Hg. 11 All case series reported similar reductions in BP. The longest available follow‐up was at 2 years in the Symplicity HTN‐1 trial, in which a decrease of −32/−14 mm Hg from baseline was reported (Table III).
Table III.
Change in Office Blood Pressure
| Reference, Year | N (I; C) | Baseline Office Recording of Blood Pressure, SBP/DBP | 1 Mo | 3 Mo | 6 Mo | 9 Mo | 12 Mo | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| I | C | I | C | I | C | I | C | I | C | I | C | ||
| Randomized control trials | |||||||||||||
| Ukena (2011) | 37; 9 | 172/94 | 166/90 | NR | NR | −31/−9 | 0/−1 | NR | NR | NR | NR | NR | NR |
| Symplicity HTN‐2 (2010) | 52; 54 | 178/97 | 178/98 | NR | NR | NR | NR | −32/−12 | +1/0 | NR | NR | −28.1/−9.7 | NR |
| Case control studies | |||||||||||||
| Mahfoud (2012) | 88; 12 | 174/95 | 184/97 | NR | NR | −22.7/−7.7 | −7.2/−4.1 | −26.6/−9.7 | −4.4/−3.0 | NR | NR | NR | NR |
| Brandt (2012) | 46; 18 | 180.7/95.8 | 184.5/98.2 | −22.5/−7.2 | −2.9/−0.2 | NR | NR | −27.8/−8.8 | −2.7/+1.6 | NR | NR | NR | NR |
| Mahfoud (2011) | 37; 13 | 178/96 | 177/96 | −28/−10 | ‐8/−4 | −32/−12 | −5/−3 | NR | NR | NR | NR | NR | NR |
| Krum (2009) | 45; 5 | 177/101 | 173/98 | −14/−10 | +3/−2 | −21/−10 | +2/+3 | −22/−11 | +14/+9 | −24/−11 | +26/+17 | −27/−17 | NR |
| Case series | |||||||||||||
| Hering (2012) | 15 | 174/91 | −34/−14 | −25/−11 | −32/−15 | NR | −33/−19 | ||||||
| Himmel (2012) | 14 | 170/94 | −16/−6 | −23/−8 | NR | NR | NR | ||||||
| Mabin (2012) | 11 | 180/109 | −30/−15 | −36/−17 | NR | NR | NR | ||||||
| Mylotte (2012) | 33 | 181.1/100.8 | NR | NR | −30.3/−14.6 | NR | NR | ||||||
| Verloop (2012) | 22 | 197/106 | NR | NR | −24/−13 | NR | NR | ||||||
| Symplicity HTN‐1 (2011) | 153 | 176/98 | −20/−10 | −24/−11 | −25/−11 | NR | −23/−11 | ||||||
| Voskuil (2011) | 11 | 203/109 | −25/−12 | NR | NR | NR | NR | ||||||
| Witkowsky (2011) | 12 | 173/106 12 | NR | −22 | −34/−13 | NR | NR | ||||||
Abbreviations: C, control group; DBP, diastolic blood pressure; I, intervention group; NR, not reported; SBP, systolic blood pressure.
Of 128 patients, 48 (37%) achieved a systolic BP of <140 mm Hg at different times in the follow‐up period.
Home‐Based BP Measurement and ABPM. The investigators of Symplicity HTN‐2 reported a significant decrease in home‐based BP recording (mean decrease of 20/12 mm Hg in 32 intervention patients compared with a rise of 2/0 mm Hg in 40 control patients, P<.01). 12 Ambulatory BP measurement (ABPM) at 6 months, available for 20 patients, also showed a significant decrease from baseline (mean decrease 11/7 mm Hg, P<.01). 12 Krum and colleagues also reported a close correlation between decreased office systolic BP and ABPM (decrease of systolic BP by 11 mm Hg with ABPM and 27 mm Hg in office recordings at corresponding times, P=.01). 13 Witkowski and colleagues 22 did not find a significant decrease in the ABPM at 3 and 6 months even though significant changes were reported in clinic measurement of BP. In patients with chronic kidney disease, the ABPM readings showed a significant decrease in nighttime BP at 3‐month follow‐up; the daytime decrease in BP was not significant. 25 Four studies were designed specifically to record effects of renal denervation on 24‐hour BP measured by ABPM. 15 , 17 , 23 , 24 Prochnau and colleagues 17 showed a significant decrease in both systolic and diastolic BP. Bauer 15 showed an improvement in the maximum systolic pressure and a significant decrease in the range of standard deviation for systolic pressures. Vase 24 did not report a significant decreased in ABPM readings. This may have been due to the limited number of patients and the lack of complete ablation achieved in two patients (due to renal artery spasm) 24 (Table IV).
Table IV.
Change in 24‐Hour Ambulatory Blood Pressure
| Reference, Year | N (I; C) | Baseline Blood Pressure, SBP/DBP | 1 Mo | 3 Mo | 6 Mo | 12 Mo | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | C | I | C | I | C | I | C | I | C | ||
| Symplicity HTN‐2 (2010) | 20;25 | 178/97 20 | 178/98 25 | NR | NR | NR | NR | −11/−7 | −3/−1 | NR | NR |
| Bauer (2012) | 11 | 190 | NR | NR | −18 | NR | |||||
| Hering (2012) | 15 | 159/85 | NR | −6/−7 | −5/−6 | NR | |||||
| Mabin (2012)a | 10 | 169/101 | −20/−11 | −22/−12 | NR | NR | |||||
| Mylotte (2012) | 33 | 171.6/93.5 | NR | NR | −23.3/−10.2 | NR | |||||
| Prochnau (2012) | 30 | 166/88 | −15.5/−3.4 | −25.5/−10.9 | −24.1/−10.6 | −15/−4.3 | |||||
| Prochnau (2012) | 12 | 167/88 | −11/−7 | −24/−14 | NR | NR | |||||
| Simonetti (2011) | 5 | 171/100 | −18/−5 | NR | NR | NR | |||||
| Vase (2012) | 9 | 152/89 | −7/−15b | NR | NR | NR | |||||
| Witkowsky (2011) | 12 | 140/82 | NR | NR | −6b | NR | |||||
Abbreviations: C, control group; DBP, diastolic blood pressure; I, intervention group; NR, not reported; SBP, systolic blood pressure. aHome blood pressure readings. bNot significant.
Norepinephrine spillover is a marker of the effectiveness of efferent renal denervation. Krum and colleagues reported a 47% decrease in renal norepinephrine spillover in 10 patients in the intervention group. 13 Simonetti 20 reported no change in urine cateholamines.
Secondary Outcomes. Maintenance of BP Reduction at 12 Months. Five studies with 312 patients in total reported follow‐up of 88 patients at 12 months. 12 , 13 , 17 , 19 , 25 Each of these studies showed maintenance of BP reduction at various intervals up to 12 months. There was no significant incremental improvement in BP at this interval follow‐up.
Decrease in Number of Medications. The average number of antihypertensive medications being used by patients in most studies was 5. Nine studies reported data on change in number of antihypertensive medications after the intervention. 9 , 12 , 13 , 19 , 20 , 21 , 23 , 27 In 3 studies with a total of 236 patients, about 10% to 20% (52 patients) required reduction in number of medicines, while 10% to 25% (25 patients) required an increase in the number of medications. 12 , 13 , 19 One study with 5 participants reported that antihypertensive medications were reduced in 4 patients. 20 Three studies with 129 patients reported a decrease in medications in 15% to 25% of the intervention group. 14 , 16 , 17 Other studies with a total of 60 patients reported no change in number of medications. 9 , 21 , 23
Change in renal functionIn the Symplicity HTN‐1 study, no significant change in the eGFR (mean 83±20 mL/min/1.73 m2) was seen in the first 12 months after the procedure. In 10 of 153 patients (6.5%) for whom data were available at the end of 24 months, there was a mean reduction of 16 mL/min/1.73 m2. 19 No significant change in serum creatinine, development of CKD Stage IV or the requirement for dialysis was reported. Krum and colleagues studied eGFR changes in 25 patients and reported a decrease in GFR by 4% in one patient and an increase of ≥20% in six patients. 13 The Symplicity HTN‐2 study reported no significant change in renal function at 6 months (assessed by eGFR, serum creatinine, and cystatin C concentration) in the intervention or control groups when compared with baseline. 12 Other studies also reported no significant change in eGFR values 20 , 22 , 23 or proteinuria. 20 , 21 , 23 In one study of 15 patients with CKD and a mean baseline eGFR of 31.2±8.9 mL/min/1.73 m2, no significant change in eGFR, serum creatinine, or proteinuria was reported 25 (Table V).
Table V.
Change in Renal Function
| Reference, Year | Baseline eGFR, mL/min/1.73 m2 | Baseline Creatinine, μmol/L | Follow‐Up Interval | Change in eGFR | Change in Creatinine | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| I | C | I | C | I | C | P Value | I | C | P Value | ||
| Randomized control trials | |||||||||||
| Ukena (2011) | 70±24 | 64.5±16 | NR | NR | 3 mo | NR | NR | NR | NR | ||
| Symplicity HTN‐2 (2010) | 77±19 | 86±20 | 91±25 | 78±18 | 6 mo | 0.2 | 0.9 | .76 | 0.2 | −1.1 | .67 |
| Case‐control studies | |||||||||||
| Mahfoud (2012) | 84.6±3.6a | 97.9±7.4a | NR | NR | 3 mo | −4.2 | −9.4 | NS | NR | ||
| 6 mo | −4.0 | −15.1 | NS | NR | |||||||
| Brandt (2012) | 83.5±27.8 | 80.5±29.3 | 86.2±26.4 | 86.2±44 | 1 mo | −3.5 | +0.4 | NS | +2.6 | +0.9 | NS |
| 6 mo | +1.2 | +9.5 | NS | −4.3 | −7 | NS | |||||
| Mahfoud (2011) | 75.1±3.3 | 81.0±7.6 | NR | NR | 3 mo | NR | NR | NR | NR | ||
| Krum (2009) | 79±21 | 95±15 | NR | NR | 6 mo | 4 | NR | NR | NR | NR | |
| Case series | |||||||||||
| Hering (2012) | 31.2±8.9 | 186.7±64.4 | 6 mo | −2.16 | .22 | 30.7 | .28 | ||||
| Prochnau (2012) | NR | >130 | 6 mo | NR | No change | ||||||
| Prochnau (2012) | NR | >130 | 3 mo | NR | No change | ||||||
| Vase (2012) | 78±13 | 79±20 | 1 mo | 1 | NS | 5 | NS | ||||
| Symplicity HTN‐1 (2011) | 83±20 | NR | 12 mo | −2.9 | NR | NR | |||||
| 24 mo | −16 | ||||||||||
| Simonetti (2011) | 91.6±15 | NR | 2 mo | 0 | NR | ||||||
| Voskuil (2011) | 74±14 | 78±17 | 1 mo | NR | 0 | .92 | |||||
| Witkowsky (2011) | 81.3 | NR | 6 mo | No change | No change | ||||||
Abbreviations: C, control group; eGFR, estimated glomerular filtration rate; I, intervention group; NR, not reported. aCystatin C GFR.
Adverse Events. Periprocedural adverse events included pseudoaneurysm 9 , 12 , 13 , 19 and renal artery dissection in 2 patients. 13 , 19 Back and/or flank pain was also reported in 12 patients. 12 , 16 , 19 Intraprocedural bradycardia requiring atropine occurred in 7 patients. 12 Hypotension occurred in 6 patients. 12 , 24 There were no changes in renal artery anatomy or development of clinically significant stenosis on follow‐up computed tomographic angiography and magnetic resonance imaging studies (Table II).
Discussion
To our knowledge, this is first systematic review of currently available evidence on renal sympathetic denervation for treatment of resistant hypertension. Limited data from RCTs and observational studies demonstrate a significant and sustained decrease in BP using this technique. The incidence of periprocedural adverse events was low.
The American Heart Association guidelines recommend combinations of antihypertensive agents with different mechanisms of action for the treatment of resistant hypertension. 1 The UK’s National Institute for Health and Clinical Excellence (NICE) recently issued guidelines on renal sympathetic denervation for resistant hypertension. 29 The guidelines conclude that there is evidence supporting the efficacy of BP reduction with renal sympathetic denervation in the short‐ and medium‐term. The guidelines advise clinicians to carefully select patients using a multidisciplinary team (including a physician with expertise in hypertension and a specialist in endovascular interventions). The French Scientific Societies’ (Cardiology, Radiology, Hypertension) guidelines suggest that while this technique is an option for resistant hypertension (defined as essential hypertension uncontrolled by ≥4 antihypertensive therapies with at least one being a diuretic and spironolactone at a dose of 25 mg), patients who undergo this procedure should be enrolled in observational studies. 30 The European Society of Hypertension has released a statement recommending that renal sympathetic denervation be carried out only in centers of excellence for hypertension by experienced interventional cardiologists or radiologists specifically trained in this procedure. 31
Two types of catheters are available for renal sympathetic denervation. The SYMPLICITY catheter (Ardian Inc, Palo Alto, CA) was used in 16 of the 19 studies. After establishing access to the renal artery via the femoral artery, the catheter is advanced to the distal section of one renal artery. Low‐power (<8 W) radiofrequency energy is delivered to the endothelial layer through an electrode for an average of 2 minutes. This leads to transmural lesions and damage to the sympathetic fibers traveling in the adventitia of the arterial wall. The catheter is then pulled back and radiofrequency energy applied 5 or 6 times longitudinally and circumferentially in the artery. This is repeated at the contralateral renal artery. 32 One study used the PARADISE catheter (ReCor Medical, Ronkonkoma, NY). 16 This catheter has a balloon that enables cooled fluid to circulate during the energy delivery process and keeps the artery wall cool, minimizing damage to nontarget tissues. 16 The energy used with this catheter is higher than the Ardian catheter (25 W compared with approximately 8 W). 16 The two studies by Prochnau and colleagues 17 , 23 used the standard steerable radiofrequency ablation catheter.
Efferent denervation decreases renin stimulation and potentiates natriuresis, while afferent denervation leads to decrease in the kidney’s contribution to central sympathetic activity. 33
Our review suggests that renal sympathetic denervation would probably be suited for patients older than 18 years with resistant hypertension without secondary causes. Patients who were pregnant, were taking anticoagulation with warfarin, or had heart failure were excluded from the studies. Intuitively, patients with chronic kidney disease and end‐stage renal disease would benefit most from this technique as these patients have high a prevalence of resistant hypertension due to increased sympathetic activity. 34 This theory has been supported by a small case series of 15 patients 25 and is being investigated in ongoing clinical trials. 34
Limitations
Our review is limited by the quality of the included studies and the limited evidence base. Only two RCTs have been conducted to date. Although all studies claimed to exclude patients with secondary hypertension, it was not explicitly stated how this was accomplished. Ambulatory BP measurements were not consistently reported in all studies across the duration of follow‐up. In the few studies that this was reported, the magnitude of reduction was smaller than the clinic BP recordings, which limits the translation of this technique to clinical benefit.
Future research should focus on RCTs with longer follow‐up to demonstrate the efficacy and safety of this technique in resistant hypertension. Symplicity HTN‐3 is an ongoing RCT that will enroll 530 patients in various centers throughout the United States. 35 Characteristics of “nonresponders” to the technique need to be studied to formulate effective exclusion criteria. Head‐to‐head trials with currently recommended pharmacologic therapy (such as spironolactone) may be needed before recommendations can be made for this as a treatment of choice for resistant hypertension. Medication combinations that work best with this technique also need to be investigated.
Conclusions
Our review suggests that renal sympathetic denervation has a role in the management of carefully selected patients with resistant hypertension. Currently ongoing and future research will provide further evidence about the efficacy and safety and should clarify unanswered questions about patient selection and the intervention itself.
References
- 1. Calhoun DA, Jones D, Textor S, et al. Resistant hypertension: diagnosis, evaluation, and treatment. A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Hypertension. 2008;51:1403–1419. [DOI] [PubMed] [Google Scholar]
- 2. Hajjar I, Kotchen TA. Trends in prevalence, awareness, treatment, and control of hypertension in the United States, 1988–2000. JAMA. 2003;290:199–206. [DOI] [PubMed] [Google Scholar]
- 3. Egan BM, Zhao Y, Axon RN, et al. Uncontrolled and apparent treatment resistant hypertension in the United States, 1988 to 2008. Circulation. 2011;124:1046–1058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Persell SD. Prevalence of resistant hypertension in the United States, 2003–2008. Hypertension. 2011;57:1076–1080. [DOI] [PubMed] [Google Scholar]
- 5. de la Sierra A, Segura J, Banegas JR, et al. Clinical features of 8295 patients with resistant hypertension classified on the basis of ambulatory blood pressure monitoring. Hypertension. 2011;57:898–902. [DOI] [PubMed] [Google Scholar]
- 6. Daugherty SL, Powers JD, Magid DJ, et al. Incidence and prognosis of resistant hypertension in hypertensive patients. Circulation. 2012;125:1635–1642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Tam GM, Yan BP, Shetty SV, Lam YY. Transcatheter renal artery sympathetic denervation for resistant hypertension: an old paradigm revisited. Int J Cardiol. 2012. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 8. Papademetriou V, Doumas M, Tsioufis K. Renal sympathetic denervation for the treatment of difficult‐to‐control or resistant hypertension. Int J Hypertens. 2011; 2011:196518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ukena C, Mahfoud F, Kindermann I, et al. Cardiorespiratory response to exercise after renal sympathetic denervation in patients with resistant hypertension. J Am Coll Cardiol. 2011;58:1176–1182. [DOI] [PubMed] [Google Scholar]
- 10. Symplicity HTN‐2 Investigators . Renal sympathetic denervation in patients with treatment‐resistant hypertension (The Symplicity HTN‐2 Trial): a randomised controlled trial. Lancet. 2010;376:1903–1909. [DOI] [PubMed] [Google Scholar]
- 11. Krum H, Schlaich M, Whitbourn R, et al. Catheter‐based renal sympathetic denervation for resistant hypertension: a multicentre safety and proof‐of‐principle cohort study. Lancet. 2009;373:1275–1281. [DOI] [PubMed] [Google Scholar]
- 12. Mahfoud F, Schlaich M, Kindermann I, et al. Effect of renal sympathetic denervation on glucose metabolism in patients with resistant hypertension. Circulation. 2011;123:1940–1946. [DOI] [PubMed] [Google Scholar]
- 13. Brandt MC, Mahfoud F, Reda S, et al. Renal sympathetic denervation reduces left ventricular hypertrophy and improves cardiac function in patients with resistant hypertension. J Am Coll Cardiol. 2012;59:901–909. [DOI] [PubMed] [Google Scholar]
- 14. Mahfoud F, Cremers B, Janker J, et al. Renal hemodynamics and renal function after catheter‐based renal sympathetic denervation in patients with resistant hypertension. Hypertension. 2012;60:419–424. [DOI] [PubMed] [Google Scholar]
- 15. Bauer A, Zuern C, Rizas K, et al. Effects of renal sympathetic denervation on 24‐hour blood pressure variability. J Am Coll Cardiol. 2012;59:E1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mabin T, Sapoval M, Cabane V, et al. First experience with endovascular ultrasound renal denervation for the treatment of resistant hypertension. Eurointervention. 2012;8:57–61. [DOI] [PubMed] [Google Scholar]
- 17. Prochnau D, Figulla HR, Surber R. Efficacy of renal denervation with a standard EP catheter in the 24‐h ambulatory blood pressure monitoring‐ long‐term follow‐up. Int J Cardiol. 2012;157:447–448. [DOI] [PubMed] [Google Scholar]
- 18. Himmel F, Weil J, Reppel M, et al. Improved heart rate dynamics in patients undergoing percutaneous renal denervation. J Clin Hypertens. 2012;14:654–655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Investigators Symplicity HTN‐1 . Catheter‐based renal sympathetic denervation for resistant hypertension. Hypertension. 2011;57:911–917. [DOI] [PubMed] [Google Scholar]
- 20. Simonetti G, Spinelli A, Gandini R, et al. Endovascular radiofrequency renal denervation in treating refractory arterial hypertension: a preliminary experience. Radiol Med (Torino). 2012;117:426–444. [DOI] [PubMed] [Google Scholar]
- 21. Voskuil M, Verloop W, Blankestijn P, et al. Percutaneous renal denervation for the treatment of resistant essential hypertension: the first Dutch experience. Neth Heart J. 2011;19:319–323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Witkowski A, Prejbisz A, Florczak E, et al. Effects of renal sympathetic denervation on blood pressure, sleep apnea course, and glycemic control in patients with resistant hypertension and sleep apnea. Hypertension. 2011;58:559–565. [DOI] [PubMed] [Google Scholar]
- 23. Prochnau D. Catheter‐based renal denervation for drug‐resistant hypertension by using a standard electrophysiology catheter. Eurointervention. 2012;7:1077–1080. [DOI] [PubMed] [Google Scholar]
- 24. Vase H, Mathiassen ON, Kaltoft A, et al. Catheter‐based renal denervation for treatment of resistant hypertension. Dan Med J. 2012;59:A4439. [PubMed] [Google Scholar]
- 25. Hering D, Mahfoud F, Walton AS, et al. Renal denervation in moderate to severe CKD. J Am Soc Nephrol. 2012; 23:1250–1257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Mylotte D, Benamer H, Unterseeh T, et al. Percutaneous renal denervation for resistant hypertension: real world outcomes. J Am Coll Cardiol. 2012;59:E1708. [Google Scholar]
- 27. Verloop W, Spiering W, Agostoni P, et al. 6‐months follow up in a real life situation after renal denervation. J Am Coll Cardiol. 2012;59:E1706. [Google Scholar]
- 28. Esler MD, Krum H, Schlaich M, et al. Renal sympathetic denervation for treatment of resistant hypertension: one year results from the SYMPLICITY HTN‐2, Randomized Control Trial. J Am Coll Cardiol. 2010;59:E1705. [DOI] [PubMed] [Google Scholar]
- 29. National Institute of Health and Clinical Excellence . Percutaneous transluminal radiofrequency sympathetic denervation of the renal artery for resistant hypertension. London: National Institute of Health and Clinical Excellence; 2012. [Google Scholar]
- 30. Pathak A, Girerd X, Azizi M, et al. Expert consensus: renal denervation for the treatment of hypertension. Diagn Interv Imaging. 2012;93:386–394. [DOI] [PubMed] [Google Scholar]
- 31. Schmieder RE, Redon J, Grassi G, et al. ESH position paper: renal denervation: an interventional therapy of resistant hypertension. J Hypertens. 2012;30:837–841. [DOI] [PubMed] [Google Scholar]
- 32. Doumas M, Faselis C, Papademetriou V. Renal sympathetic denervation in hypertension. Curr Opin Nephrol Hypertens. 2011;20:647–653. [DOI] [PubMed] [Google Scholar]
- 33. Katholi RE, Rocha‐Singh KJ. The role of renal sympathetic nerves in hypertension: has percutaneous renal denervation refocused attention on their clinical significance? Prog Cardiovasc Dis. 2009;52:243–248. [DOI] [PubMed] [Google Scholar]
- 34. Schlaich M, Krum H, Sobotka P. Renal sympathetic nerve ablation: the new frontier in the treatment of hypertension. Curr Hypertens Rep. 2010;12:39–46. [DOI] [PubMed] [Google Scholar]
- 35. Kandzari DE, Bhatt DL, Sobotka PA, et al. Catheter‐based renal denervation for resistant hypertension: rationale and design of the SYMPLICITY HTN‐3 Trial. Clin Cardiol. 2012;35(9):528–528. [DOI] [PMC free article] [PubMed] [Google Scholar]


