Abstract
Background
The novel severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is responsible for the worldwide coronavirus disease-19 (COVID-19) pandemic, starting in late 2019. The standard diagnostic methods to detect SARS-CoV-2 are PCR-based genetic assays. Antigen-antibody-based immunochromatographic assays are alternative methods of detecting this virus. Rapid diagnosis kits to detect SARS-CoV-2 are urgently needed.
Study design
Three monoclonal antibodies against SARS-CoV-2 nucleocapsid (N) protein were used to develop an antigen-antibody-based immunochromatographic kit to detect SARS-CoV-2. These assays were evaluated using nasopharyngeal swab specimens collected from patients suspected of having COVID-19.
Results
These assays detected recombinant SARS-CoV-2 N protein at concentrations >0.2 ng/mL within 10 min after protein loading, but did not detect the N proteins of Middle East respiratory syndrome coronavirus (MERS-CoV), human coronaviruses OC43 (HCoV-OC43) and 299E (HCoV-229E) and other pathogens causing respiratory infections. Nasopharyngeal swab specimens obtained 1~3, 4~9, and ≥ 10 days after symptom onset from COVID-19 patients diagnosed by RT-PCR showed positivity rates of 100 %, >80 %, and <30 %, respectively.
Conclusions
Kits using this immunochromatographic assay may be a rapid and useful tool for point-of-care diagnosis of COVID-19 when samples are obtained from patients 1~9 days after symptom onset.
Keywords: Immunochromatographic kit, Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Coronavirus disease-19 (COVID-19)
1. Background
The novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is responsible for the worldwide coronavirus disease (COVID-19) pandemic, starting in late 2019 (WHO, 2021a Coronavirus disease 2019 (COVID-19) pandemic). The World Health Organization (WHO) has recommended the use of nucleic acid amplification tests based on real-time reverse-transcription polymerase chain reaction (rRT-PCR) to detect SARS-CoV-2 (WHO, 2021b Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases). Other genetic methods have also been described, including nested RT-PCR (Shirato et al., 2020), single-tube reverse transcription loop-mediated isothermal amplification (RT-LAMP) (Baek et al., 2020) and modified rRT-PCR methods (Loeffelholz and Tang, 2020)
Antigen-antibody-based immunochromatographic assays are alternative methods to detect SARS-CoV-2. Immunochromatographic assays have become point-of-care diagnostic methods to detect seasonal influenza (Sturenburg and Junker, 2009), as well as outbreaks of emerging infectious diseases, including severe acute respiratory syndrome (Kogaki et al., 2005), Middle East respiratory syndrome (MERS; Song et al., 2015) and pandemic H1N1 (2009) influenza virus (Miyoshi-Akiyama et al., 2010; Kawachi et al., 2011). Rapid diagnosis kits to detect SARS-CoV-2, perhaps using immunochromatographic assays, are urgently needed.
2. Objectives
This study describes the development of a novel immunochromatographic assay using three monoclonal antibodies against recombinant SARS-CoV-2 nucleocapsid (N) protein to detect SARS-CoV-2.
3. Study design
Nasopharyngeal swab samples were collected from patients suspected of having COVID-19 who had symptoms such as fever, dry cough, fatigue, loss of taste/smell, nasal congestion, conjunctivitis, sore throat, headache, muscle/joint pain, skin rash, nausea/vomiting, diarrhea and/or chills/dizziness. Nasopharyngeal swab samples were collected during 18 March 2021 to 20 August 2021 at a university hospital in Tokyo. A total number of samples were 98. All samples were subjected to RT-PCR for the detection of SARS-CoV-2 according to a protocol published by the National Institute of Infectious Disease, Japan (Manual for the detection of pathogen 2019-nCoV Ver2.6). Of them, 45 were RT-PCR positive. Of the 45 samples, 11 were collected 1–3 days after symptom onset, 16 were 4–6 days after symptom onset, 8 were 7–9 days after symptom onset and 7 were 10 days after symptom onset, respectively. Regarding the remaining 3 samples, the collection days after symptom onset were not available. In addition, nasopharyngeal swab samples were collected from 21 healthy medical staffs without the above symptoms during 28–30 January 2021 at a hospital in Iwate prefecture, Japan.
To prepare nasopharyngeal swab samples with various threshold cycle values (Ct values) for SARS-CoV-2 RT-PCR, various doses of SARS-CoV-2 2019-nCoV/JPN/TY/WK-521 culture, supplied by the National Institute of Infectious Disease, Tokyo, Japan, were added to RT-PCR-negative swab samples. These samples were subjected to RT-PCR to confirm Ct values for SARS-CoV-2, and then used to evaluate the positive rate of immunochromatographic assay.
A nucleotide fragment of 1260 bp encoding the entire SARS-CoV-2 N protein was synthesized based on the sequence of the complete genome, 2019-nCoV WHU01 (accession no. MN988668). The gene encoding full-length SARS-CoV-2 N protein (amino acids [aa] 1–419) and fragments encoding partial sequences of SARS-CoV-2 N, corresponding to aa 1–120, 111–220 and 210–419, were cloned into the pET8a expression vector (Novagen, Inc., Madison, WI, USA) using the primers listed in Table S1. The constructed plasmids were used to transform Escherichia coli BL21-CodonPlus(DE3)-RIP (Agilent Technologies, Santa Clara, CA, USA). All recombinant proteins were purified using Ni-NTA agarose, according to the manufacturer’s instructions (Qiagen, Hilden, Germany).
Nucleotides encoding the N proteins of SARS-CoV, MERS-coronavirus (MERS-CoV), human coronavirus OC43 (HCoV-OC43) and human coronavirus 299E (HCoV-229E) were synthesized based on the complete genome sequences of SARS-CoV Tor2 (accession no. NC_004718), the MERS-CoV isolate HCoV-EMC/2012 (accession no. NC_019843), the HCoV-OC43 strain ATCC VR-759 (accession no. NC_006213) and HCoV-229E (accession no. NC_002645), respectively. The genes encoding the recombinant N proteins of these coronaviruses were cloned into an expression vector using the primers listed in Table S1; following their expression in E. coli, the proteins were purified as described above. The gene encoding the recombinant N protein derived from the SARS-CoV-2 501Y variant 2 (also called B.1.1.7, 20B/501Y.V1 and VOC-202012/01) was amplified using a pair of primers listed in Table S1; relative to the original N protein, the recombinant N protein had two amino acid substitutions, D3L and S235 F (Leung et al., 2021).
Wistar rats were immunized with full-length recombinant SARS-CoV-2 N protein. Hybridomas were screened with an enzyme-linked immunosorbent assay (ELISA) using full-length recombinant SARS-CoV-2 N protein. Briefly, 100 μL aliquots of recombinant SARS-CoV-2 N protein in 0.05 M carbonate buffer (pH 9.0) were placed in the wells of 96-well plates. The plates were incubated for 1 h at room temperature, washed three times with phosphate-buffered saline (PBS)-0.1 % Tween 20, and incubated with SuperBlock™ Blocking Buffer in PBS (Thermo Scientific., Swedesboro USA) (150 μL/well) for 30 min at room temperature. To each well was added 100 μL of the culture supernatant of a hybridoma, followed by incubation for 1 h at room temperature. A 100 μL aliquot of peroxidase-conjugated anti-rat IgG (1:10,000 dilution) (SouthernBiotech; Birmingham, AL, USA) was added to each well, and the plates were incubated for 30 min at room temperature. A 50 μL aliquot of TMB Peroxidase ELA Substrate Kit (Bio-Rad, Hercules, CA, USA) was added to each well, followed by incubation at room temperature for 10 min and the addition of 50 μL 1 M sulfuric acid to stop the peroxidase reaction. The absorbance of each well at 450 nm, and at 620 nm as reference, was measured using an Infinite F50 microplate reader (Tecan, Mannedorf, Switzerland).
Monoclonal antibodies (mAbs) were purified using Spin Column Based Antibody Purification kits (Cosmo Bio USA; Carlsbad, CA, USA). To determine the binding regions of the mAbs, direct ELISA was performed using full-length (aa 1–419) and three partial fragments (aa 1–120, 111–220 and 210–419) of SARS-CoV-2 N protein. Sandwich ELISAs were performed using these mAbs as first or capture antibodies and horseradish peroxidase-conjugated mAbs as second or detection antibodies. The mAbs had been labeled with peroxidase using Ab-10 Rapid Peroxidase Labeling Kit (Cosmo Bio USA).
Immunochromatographic assays (ICAs) were developed by coating nitrocellulose membranes with capture antibodies (N1J7 and N1K1) and control goat anti-rodent IgG antibody. Detection antibody (N1K2)-conjugated colloidal golds were immersed into glass fiber. Sample pad, absorbent pad, nitrocellulose membrane with the capture antibodies, and the grass fiber containing N1K2-conjugated colloidal gold were laminated and pasted onto the polystyrene self-adhesive baseplate. The assembled on the baseplate was divided into strips. Nasopharyngeal swab specimens, recombinant proteins, and various pathogens were applied to assess the strip in the ICA. The strip was vertically immersed into sample in test tube, ensuring the sample pad of strip was fully reacted with the sample. Line formation on the strip was assessed within 10 min after immersing it.
As shown in details in Table S2, a total of 56 pathogens causing respiratory infections, except for Bordetella pertussis and Veillonella sp., were obtained from ATCC, IFO, JCM and NBRC. These pathogens were including adenovirus, coxsackievirus, echovirus, influenza A and B viruses, herpesvirus, human coronavirus 229E, parainfluenza virus, respiratory syncytial virus, rhinovirus, Acinetobacter baumannii, Bacteroides fragilis, Bordetella pertussis, Corynebacterium diphtheriae, Escherichia coli, Enterococcus faecalis, Haemophilus spp., Klebsiella pneumoniae, Lactobacillus fermentum, Legionella pneumophila, Mycobacterium fortuitum, Mycoplasma pneumoniae, Neisseria gonorrhoeae, Peptostreptococcus anaerobius, Porphyromanas gingivalis, Proteus spp., Pseudomonas spp., Serratia marcescens, Staphylococcus spp., Streptococcus spp., Veillonella sp., Yersinia enterocolitica and Candida spp. (Table S2).
A chi-square test was performed and a p-value of <0.05 was considered statistically significant.
4. Results
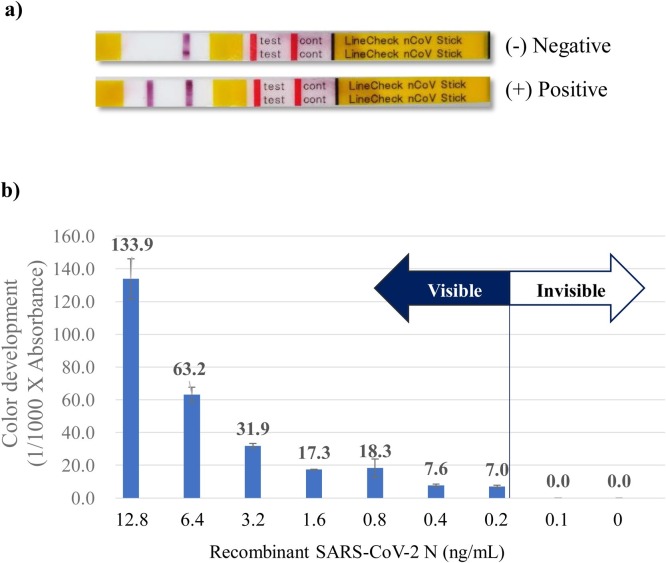
Three mAbs against recombinant SARS-CoV-2 N protein, N1J7, N1K1 and N1K2, were chosen by ELISA using full-length (aa 1–419) and fragments (aa 1–120, 111–220 and 210–419) of recombinant SARS-CoV-2 N protein (Fig. 1 ). All three mAbs showed high reactivity against full-length N protein (Fig. 1b). Two mAbs, N1K1 and N1K2, showed high reactivity against the aa 1–120 fragment, but not against the aa 110–220 and 210–419 fragments (Fig. 1b). The third mAb, N1J7, showed high reactivity against the aa 111–220 fragment, but not against the aa 1–120 and 210–419 fragments (Fig. 1b). Comparisons by sandwich ELISA showed that the highest reactivity was obtained when N1J7 and N1K1 were used as the first (capture) antibodies and horseradish peroxidase-conjugated N1K2 as the second (detection) antibody (Fig. 2 ). Therefore, an immunochromatographic assay (ICA), KBM Linecheck nCoV, was developed using N1J7 and N1K1 as capture antibodies and N1K2 as a detection antibody (Fig. 3 a). This ICA had a lower limit of detection of 0.04 ng of recombinant SARS-CoV-2 N protein, or 200 μL of a 0.2 ng/mL solution (Fig. 3b).
Fig. 1.
Determination of epitopes by direct ELISA. a) The amino acid structure of SARS-CoV-2 nucleocapsid (N) protein and the regions of recombinant SARS-CoV-2 N protein. b) Competition assay using full- and partial-length recombinant SARS-CoV-2 N proteins. Assays were performed using three mAbs (N1J7, N1K1 and N1K2) as primary antibodies and horseradish peroxidase-conjugated anti-rat IgG as secondary antibody.
Fig. 2.
Determination of the optimal mABs combination by sandwich ELISA. The primary (capture) antibodies consisted of N1J7, N1K1, N1K2, and their combinations (N1J7 and N1K1, N1J7 and N1K2 and N1K1 and N1K2). The antigen was recombinant SARS-CoV-2 N protein, and the secondary (detection) antibodies consisted of horseradish peroxidase-conjugated N1J7, N1K1 and N1K2.
Fig. 3.
Details of the immunochromatographic assay (KBM Linecheck nCoV) developed using the mAbs N1J7, N1K2 and N1K2. a) Samples showing a single line at the control position were negative for N protein, whereas samples showing two lines, one each at the test and control positions, were positive for N protein. b) Antigen detection limits of KBM Linecheck nCoV. Recombinant SARS-CoV-2 N protein was firstly diluted in PBS-0.1 % Tween 20, and further diluted in Tris-based buffer (pH7.6) to 12.8, 6.4, 3.2, 1.6, 0.8, 0.4, 0.2 and 0.1 ng/mL, and 200 μL of each solution were used to determine detection limits. Color development was measured by a C10066-10 immunochromato-reader (Hamamatsu Photonics Co., Hamamatsu, Japan).
The KBM Linecheck nCoV assay was evaluated for its ability to detect SARS-CoV-2 in nasopharyngeal swab samples from 45 patients who were diagnosed with COVID-19 by RT-PCR. Of the 45 RT-PCR-positive samples, 36 (80.0 %) were ICA-positive, but nine (20.0 %) were ICA-negative; whereas, all 53 RT-PCR-negative samples were ICA-negative (chi-square test; p < 0.01) (data not shown). Of the 42 RT-PCR-positive samples, 11, 16, eight, and seven samples were obtained from patients 1~3, 4~6, 7~9, and ≥10 days, respectively after symptom onset; of these, 11 (100 %), 13 (81.3 %), seven (87.5 %), and two (28.6 %) samples, respectively, were ICA-positive (Table 1 ). The threshold cycle (Ct) values of RT-PCR correlated inversely with ICA-positive rates, with 100 % (3/3), 96.4 % (29/30), 88.5 % (23/26), and 60.0 % (18/30) samples with Ct ≤15, 15 < Ct≤20, 20 < Ct≤25, and Ct>25, respectively, being ICA-positive (Table 2 ).
Table 1.
Positive rates of KBM LineCheck nCoV in COVID-19 patients by days after symptom onset.
| Days after symptom onset * | Positive / Total samples | Positive rate |
|---|---|---|
| 1~3 days | 11 / 11 | 100 % |
| 4~6 days | 13 / 16 | 81.3 % |
| 7~9 days | 7 / 8 | 87.5 % |
| after 10 days | 2 / 7 | 28.6 % |
| Total | 33 / 42 | 78.6% |
Day of symptom onset was defined as the day when patients showed any of the following symptoms; fever, dry cough, fatigue, loss of taste/smell, nasal congestion, conjunctivitis, sore throat, headache, muscle/joint pain, skin rash, nausea/vomiting, diarrhea and chills/dizziness.
Table 2.
Positive rates of KBM Linecheck nCoV in nasopharyngeal swab samples positive for SARS-CoV-2 RT-PCR.
| Ct values * | Positive / Total samples** | Positive rate |
|---|---|---|
| Ct ≤ 15 | 3 / 3 | 100 % |
| 15 < Ct≤ 20 | 29 / 30 | 96.7% |
| 20 < Ct≤ 25 | 23 / 26 | 88.5 % |
| 25 < Ct | 18 / 30 | 60.0 % |
Ct values: threshold cycle values generated by SARS-CoV-2 RT-PCR.
To assess samples with various Ct values generated by of SARS-CoV-2 RT-PCR, 45 RT-PCR positive samples and 44 swab samples to which SARS-CoV-2 culture supernatants were added at various concentrations. The 44 swab samples were prepared by adding various doses of SARS-CoV-2 2019-nCoV/JPN/TY/WK-521 culture to RT-PCR-negative swab samples. These samples were subjected to RT-PCR to confirm Ct values for SARS-CoV-2.
Twenty-one nasopharyngeal swab samples from 21 healthy medical staffs without any symptoms were tested for the ICA. All the samples were negative for both RT-PCR and ICA (data not shown).
As summarized in Table 3 by the results of the ICA and RT-PCR, the positive and negative predictive values of ICA compared with those of RT-PCR were 82.0 % (73/89) and 100 % (74/74), respectively.
Table 3.
Detection of SARS-CoV-2 in suspected COVID-19 patients and healthy medical staffs by KMB Linecheck nCoV.
| RT-PCR |
||||
|---|---|---|---|---|
| Positive | Negative | Total | ||
| KBM Line check nCoV | Positive | 73 | 0 | 73 |
| Negative | 16 | 74 | 90 | |
| Total | 89 | 74 | 163 | |
Positive and nagative predictive values were 82.0 % (73/89) and 100 % (74/74), respectively. A p-value (<0.05) in chi-square test was considered statistically significant.
Recombinant N proteins of SARS-CoV-1, SARS-CoV-2 and SARS-CoV-2 N501Y variant 2 were ICA-positive, whereas recombinant N proteins of MERS-CoV, HCoV-OC43 and HCoV-229E were ICA-negative (Table S2). All pathogens listed in Table S2 causing respiratory infections were ICA negative.
5. Discussion
The present study showed that the developed immunochromatographic kit, KBM Linecheck nCoV, will likely be a powerful tool for COVID-19 diagnosis during the early phase of the disease. All 11 samples obtained within 3 days after symptom onset were positive on these assays, as were 31 (88.6 %) of 35 samples obtained within 9 days after symptom onset. However, only 60 % of the SARS-CoV-2 positive samples with Ct>25 were positive values. Although RT-PCR methods have been reported able to detect 25 copies of SARS-CoV-2 RNA (Shirato et al., 2020), RT-PCR targeting RNA and ICA targeting N protein cannot be directly compared, with the sensitivity of the ICA kit being lower than that of RT-PCR. These findings suggest that these kits, when combined with RT-PCR methods, will be effective in diagnosing COVID-19.
The amino acid sequences of N proteins of SARS-CoV-1 and SARS-CoV-2 are well conserved (Kang et al., 2020). Nevertheless, SARS-CoV-2 variants have emerged and are spreading worldwide (Leung et al., 2021), with numerous mutations resulting in amino acid substitutions in spike proteins (Guruprasad, 2021). In contrast, few mutations have been observed in SARS-CoV-2 N proteins, with only two mutations leading to amino acid substitutions (D3L and S235 F) reported to date in SARS-CoV-2 N501Y variant 2 (also called B.1.1.7, 20B/501Y.V1 and VOC-202012/01), which was first identified in the United Kingdom in December 2020 (Leung et al., 2021). The KBM Linecheck nCoV kit was able to detect the recombinant N protein of this variant.
In conclusion, these findings indicate that the KBM Linecheck nCoV ICA kit will be useful for detection of SARS-CoV-2 in point-of-care diagnosis of COVID-19 with high specificity. The kit can rapidly detect SARS-CoV-2 within 10 min and is easily handled without any special equipment.
Authorship contributions
Please indicate the specific contributions made by each author (list the authors’ initials followed by their surnames, e.g., Y.L. Cheung). The name of each author must appear at least once in each of the three categories below.
Category 1
Conception and design of study: Teuro Kirikae, Takashi Miida, Yoko Tabe acquisition of data: May 2020, May 2020, May 2020 analysis and/or interpretation of data: Sep 2020, Sep 2020, Sep 2020
Category 2
Drafting the manuscript: Satoshi Oshiro, Teuro Kirikae, Jun-ichiro Sekiguchi, Keiji Funatogawa revising the manuscript critically for important intellectual content: Satoshi Oshiro, Teuro Kirikae, Jun-ichiro Sekiguchi, Keiji Funatogawa, Tatsuya Tada
Category 3
Approval of the version of the manuscript to be published (the name of all authors must be listed): Satoshi Oshiro,Yoko Tabe, Keiji Funatogawa, Kaori Saito, Tatsuya Tada, Tomomi Hishinuma, Naeko Mizutani, Makoto Akiwa, Jun-ichiro Sekiguchi, Takashi Miida, Teuro Kirikae,
Finding sources
This study was supported by a grant from the Japan Agency for Medical Research and Development (grant number 20he0622015h0001) and a joint research fund from Kohjin Bio Co., Ltd.
Declaration of Competing Interest
The authors report no declarations of interest.
Acknowledgments
This study was approved by the ethics committees at Juntendo university (20-036) and at Kohjin Bio (02607-2001&02602-2005). The SARS-CoV-2 isolates, SARS-CoV-2 2019-nCoV/JPN/TY/WK-521 and SARS-CoV-2 TKYE6182_ 2020, were supplied by the National Institute of Infectious Disease, Tokyo, Japan, and the Tokyo Metropolitan Institute of Public Health, respectively. The authors thank Dr. Munetaka Kimura, Nansho hospital, Prof. Norio Yamamoto, Tokai University, and Dr. Mari Tohya, Juntendo university, for discussions on the research plan.
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.jviromet.2021.114183.
Appendix A. Supplementary data
The following are Supplementary data to this article:
References
- Baek Y.H., Um J., Antigua K.J.C., et al. Development of a reverse transcription-loop-mediated isothermal amplification as a rapid early-detection method for novel SARS-CoV-2. Emerg. Microbes Infect. 2020;9(1):998–1007. doi: 10.1080/22221751.2020.1756698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guruprasad L. Human SARS CoV-2 spike protein mutations. Proteins. 2021 doi: 10.1002/prot.26042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawachi S., Matsushita T., Sato T., et al. Multicenter prospective evaluation of a novel rapid immunochromatographic diagnostic kit specifically detecting influenza A H1N1 2009 virus. J. Clin. Virol. 2011;51(1):68–72. doi: 10.1016/j.jcv.2011.01.007. [DOI] [PubMed] [Google Scholar]
- Kogaki H., Uchida Y., Fujii N., et al. Novel rapid immunochromatographic test based on an enzyme immunoassay for detecting nucleocapsid antigen in SARS-associated coronavirus. J. Clin. Lab. Anal. 2005;19(4):150–159. doi: 10.1002/jcla.20070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung K., Shum M.H., Leung G.M., et al. Early transmissibility assessment of the N501Y mutant strains of SARS-CoV-2 in the United Kingdom, October to November 2020. Euro Surveill. 2021;26(1) doi: 10.2807/1560-7917.ES.2020.26.1.2002106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loeffelholz M.J., Tang Y.W. Laboratory diagnosis of emerging human coronavirus infections – the state of the art. Emerg. Microbes Infect. 2020;9(1):747–756. doi: 10.1080/22221751.2020.1745095. http://www.niid.go.jp/niid/images/epi/corona/2019-nCoVmanual20200217-en.pdf Manual for the detection of pathogen 2019-nCoV ver2.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyoshi-Akiyama T., Narahara K., Mori S., et al. Development of an immunochromatographic assay specifically detecting pandemic H1N1 (2009) influenza virus. J. Clin. Microbiol. 2010;48(3):703–708. doi: 10.1128/JCM.02262-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shirato K., Nao N., Katano H., et al. Development of genetic diagnostic methods for detection for novel coronavirus 2019 (nCOV-2019) in Japan. Jpn. J. Infect. Dis. 2020;73(4):304–307. doi: 10.7883/yoken. JJID.2020.061. [DOI] [PubMed] [Google Scholar]
- Song D., Ha G., Serhan W., et al. Development and validation of a rapid immunochromatographic assay for detection of Middle East respiratory syndrome coronavirus antigen in dromedary camels. J. Clin. Microbiol. 2015;53(4):1178–1182. doi: 10.1128/JCM.03096-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturenburg E., Junker R. Point-of-care testing in microbiology: the advantage and disadvantages of immunochromatographic test strips. Arztebl. Int. 2009;106(4):48–54. doi: 10.3238/arztebl.2009.0048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO . 2021. Coronavirus Disease 2019 (COVID-19) Pandemic Coronavirus Disease (COVID-19) (who.int) [Google Scholar]
- WHO . 2021. Laboratory Testing for 2019 Novel Coronavirus (2019-nCoV) in Suspected Human Cases. WHO-COVID-19-laboratory-2020.5-eng.pdf. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.