Abstract
With the rapid development of research on coronavirus disease 2019 (COVID-19), more and more attention has been drawn to its damage to extrapulmonary organs. There are increasing lines of evidence showing that liver injury is closely related to the severity of COVID-19, which may have an adverse impact on the progression and prognosis of the patients. What is more, severe acute respiratory syndrome coronavirus-2 infection, cytokine storm, ischemia/hypoxia reperfusion injury, aggravation of the primary liver disease and drug-induced liver injury may all contribute to the hepatic damage in COVID-19 patients; although, the drug-induced liver injury, especially idiosyncratic drug-induced liver injury, requires further causality confirmation by the updated Roussel Uclaf Causality Assessment Method published in 2016. Up to now, there is no specific regimen for COVID-19, and COVID-19-related liver injury is mainly controlled by symptomatic and supportive treatment. Here, we review the clinical features of abnormal liver enzymes in COVID-19 and pathogenesis of COVID-19-related liver injury based on the current evidence, which may provide help for clinicians and researchers in exploring the pathogenesis and developing treatment strategies.
Keywords: Coronavirus disease 2019, Abnormal liver enzymes, Liver injury, Idiosyncratic drug-induced liver injury, Roussel Uclaf Causality Assessment Method, Clinical characteristics
Introduction
Coronavirus disease 2019 (COVID-19) is an acute infectious respiratory disease caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), which is currently in the global pandemic stage. According to statistics, the average duration from onset of symptoms to hospital discharge of COVID-19 patients is about 24.7 days, with a crude case fatality ratio of 3.67%, which poses a severe threat to human public health.1 Although COVID-19 mainly causes respiratory failure, an increasing number of extrapulmonary organ dysfunction cases have been reported, especially in critically ill patients.2 About 14–53% of patients have presented elevated levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) at varying degrees during COVID-19,3 suggesting that liver injury may be a common extrapulmonary manifestation of COVID-19. In this work, we reviewed the current research progress of abnormal liver enzymes in COVID-19, which may deepen our understanding of its characteristics and pathogenesis, ultimately providing help for future studies.
Definitions of COVID-19-related liver injury
At present, there is no unified standard for the definition of COVID-19-related liver injury. The China Digestion Association and the Chinese Society of Hepatology published a standard depending on the upper limit of normal (ULN) of liver enzymes. In this protocol, COVID-19-related liver injury is defined as a significant abnormality in liver biochemical test during the occurrence, development and treatment of COVID-19, namely ALT or AST >3 ULN, or total bilirubin (TBIL) >2 ULN, regardless of whether there has been any underlying liver disease in the past.4 The patterns of liver injury in COVID-19 were classified as three types:5 hepatocellular, if ALT and/or AST >3 ULN; cholestatic, if alkaline phosphatase (ALP) or gamma-glutamyl transferase (GGT) >2 ULN; and mixed type, if patients have both laboratory abnormalities. Some studies have used other standard definitions of ALT cut-offs. In these studies, liver injury was categorized into the following three grades based on ALT values: mild, if the ALT value was between 1 and 2 ULN; moderate, if between 2 and 5 ULN; and severe, if >5 ULN.6
ALT is a more specific marker for liver injury than AST, due to the more predominant extra-hepatic sources of AST, rendering it less liver-specific.6,7 However, the serum activity of AST may be a more sensitive indicator of liver injury in conditions such as acute hepatic ischemia, alcohol-related liver disease, and some cases of autoimmune hepatitis.7–10 It is still unknown how SARS-CoV-2 impacts the liver. Liver biochemical abnormalities in COVID-19 can be caused by acute hepatic ischemia, hypoxia, aggravation of the primary liver disease, and drug-induced liver injury (DILI). Thus, the definition of liver injury by ALT cut-offs is incomplete or rigorous due to the variable patterns of liver enzyme abnormalities. Actually, it is recommended that the best discriminant values for recognizing acute liver injury are 200 U/L for AST and 300 U/L for ALT.11 Nevertheless, due to the multifactorial reasons for abnormal liver enzymes in the clinical settings, the definition of COVID-19-related liver injury needs to be further explored.
Demographic and clinical characteristics of COVID-19 with abnormal liver enzymes
Demographic characteristics
For demographic characteristics, recent research studies have shown that male sex and increased age are associated with abnormal liver enzymes in COVID-19.
In the initial studies characterizing COVID-19 patients, male sex was one of the common factors associated with abnormal liver enzymes.5,12,13 In a study, the ratio of males to females was 4:1 for patients with liver injury.14 Li et al.15 found that male sex correlated with elevated ALT and AST levels (p=0.027 and 0.036, respectively). Notably, male sex has been reported to associate with hospital admission.16 Previous research found an increased male susceptibility to severe acute respiratory syndrome coronavirus (SARS-CoV) mediated by differences in estrogen receptor signalling,17 so it is worth further exploring whether sex-specific differences also exist in SARS-CoV-2 infection. Furthermore, sex has been shown to influence ALT activity,18 as men tend to have a higher serum ALT activity than women.7 This may explain the higher percentage of males in COVID-19-related liver injury.
Further studies showed that age, male sex and body mass index (commonly known as BMI) were predictors of peak hospitalization in patients with abnormal liver enzymes >5 ULN.5,19 However, in another study, although advanced age was associated with poor prognosis, multivariate analysis found that younger age was one of the most effective predictors of severe liver injury (ALT >5 ULN).6 This contradiction may be explained by the overactive immune and inflammatory response to COVID-19 in young patients.
Additionally, it has been reported that the prevalence of abnormal liver enzymes in the USA-based cohorts is considerably higher than that in Chinese cohorts.20,21 The prevalence of ALT and AST elevations in Chinese cohorts ranged between 4–33% and 4–53% respectively, while the elevated admission ALT and AST were up to 39.0% and 58.4% in a large New York, USA cohort.20–22 Higher BMI and metabolic-associated fatty liver disease (MAFLD) prevalence in the USA may explain some of the discrepancies with Chinese cohorts.20,21,23
Laboratory examination characteristics
Increased ALT and AST levels were the most common abnormality found among the liver enzymes in COVID-19. Most patients presented mild to moderate elevation, rarely with more than 10 ULN abnormalities. The increase of GGT and TBIL were relatively common, while ALP level was usually not increased. In addition, inflammatory markers, such as C-reactive protein (CRP), procalcitonin (PCT), serum ferritin (SF) and interleukin (IL)-6, have tended to show an increase that is more obvious in COVID-19-related liver injury patients.5,12,14,20,24–26
A study that included 417 COVID-19 cases showed that a total of 318 patients had abnormal liver enzymes, of which more than 90% showed mild (<2 ULN) at admission; patients with elevated ALT, AST, GGT, and TBIL levels exceeding 3 ULN during hospitalization accounted for 10.4%, 5.7%, 11.6%, and 2.8%, while ALP level did not increase significantly.5 Further studies determined the dynamic changes of the patterns of liver enzymes in COVID-19-related liver injury.12,14,20 The study by Lei et al.12 showed that in severe patients, AST was significantly elevated first at admission, followed by ALT elevation, and the fluctuation of TBIL and ALP levels was relatively slight. Further analysis found that the above elevated biochemical indexes were significantly associated with adverse outcomes of COVID-19, among which AST was associated with the highest mortality risk.12 Similar results were shown by other studies,14,20,26 and correlation analysis found that AST highly correlated with ALT throughout the illness course.
Notably, abnormal liver enzymes could also associate with systemic involvement of COVID-19. The study conducted by Bloom et al.20 showed that AST had mild to moderate correlation with creatine kinase (CK), lactate dehydrogenase (LDH), CRP, erythrocyte sedimentation rate (ESR), and SF. Piano et al.26 also found that patients with abnormal liver enzymes had a more severe systemic inflammation, suggesting collateral hepatic damage from systemic inflammation driven by COVID-19. Of note, some studies proposed that elevated AST in COVID-19 presented more like a cytotoxicity response rather than liver damage, which may potentially involve the muscles.27,28 Other studies found that AST was strongly correlated with ALT throughout the illness course and to a less extent with CK, reasonably suggesting that liver injury was the predominant source of aminotransferase elevation.12,14,20,26
Imaging changes
Xie et al.29 used computed tomography (CT) scores to analyze the relationship between chest CT manifestations and liver injury in patients with COVID-19, and the results showed that the CT scores in the liver injury group were significantly higher than those in the non-liver injury group (p<0.05); the incidence of liver injury in patients with CT scores <5, 5–15 and >15 were 13.3%, 36.4% and 77.8%, respectively; further analysis showed that CT score was an independent predictor of liver injury. Another study found that patients with abnormal liver enzymes had more frequent bilateral consolidation at chest X-ray than those without (44% vs. 32%; p=0.006).26 Pulmonary imaging findings could accurately reflect the progression of COVID-19. Although the causal relationship between pulmonary CT findings and liver injury was not clear, CT score, as an independent predictor of liver injury, suggested that liver injury may be associated with disease severity. Therefore, for patients with severe pulmonary imaging manifestations, liver function should also be monitored when treating respiratory diseases.
Furthermore, Bhayana et al.30 found that bowel abnormalities and cholestasis were common findings on abdominal imaging of patients with COVID-19. Among right upper quadrant ultrasounds, 87% (32 out of 37) were performed due to liver laboratory findings in intensive care unit (ICU) patients, and 54% (20 out of 37) demonstrated a dilated sludge-filled gallbladder which suggested cholestasis. Although the basic disease status of patients with cholestasis had not been analyzed, this research showed that not only the abnormal liver enzymes but also the cholestasis could be present in COVID-19 patients. Indeed, based on limited evidence, the prevalence of cholestatic type has ranged from 24% to 29.25% in COVID-19 patients with abnormal liver enzymes,5,26 and whether these patients have radiologically detectable cholestasis is worth further exploration. Notably, cholestasis could lead to severe liver damage if not effectively controlled, so it is necessary to provide timely and effective treatment for patients with cholestasis.
Pathological findings
At present, reports of the liver histopathological features of COVID-19 patients are limited. Previous studies have found that moderate microvesicular steatosis and mild periportal lymphocytic inflammation were the most frequent pathological findings in the liver of COVID-19 patients, indicating that either SARS-CoV-2 infection or DILI could cause the injury.5,31–34 Elsoukkary et al.35 observed peculiar basophilic structures of unknown origin in sinusoidal endothelium in 36% of COVID-19 patients. Notably, SARS-CoV-2 RNA has been detected in liver tissue by polymerase chain reaction.32,34 Lagana et al.36 analyzed the hepatic findings in autopsy specimens from 40 patients who died of COVID-19 complications. The results showed that macrovesicular steatosis was the most common finding (75%), followed by lobular necroinflammation (50%), portal inflammation (50%) and cholestasis (38%). Furthermore, a recent study showed that a large number of apoptotic hepatocytes and prominent binuclear hepatocytes were found in liver autopsies of two COVID-19 patients.37 Although no obvious viral inclusion bodies were found, transmission electron microscopy examination revealed that hepatocyte cytoplasm contained a large number of typical coronavirus particles with spike structure.37 In addition, obvious virus invasion manifestations, such as mitochondrial swelling, endoplasmic reticulum dilatation and impaired cell membrane, were also found, suggesting that SARS-CoV-2 can directly infect hepatocytes and lead to liver injury.37
Association between abnormal liver enzymes and clinical outcome of COVID-19
At present, a large number of studies have shown that liver biochemical abnormalities are more prevalent in severe patients. Guan et al.38 collected 1,099 COVID-19 cases from 552 hospitals in China, and found that the incidence of elevated AST, ALT, and TBIL levels in severe patients were significantly higher than those in non-severe patients (AST: 39.4% vs. 18.2%; ALT: 28.1% vs. 19.8%; TBIL: 13.3% vs. 9.9%;). Among 82 hospitalized patients who died of COVID-19 in Wuhan, 78% had liver injury, and further analysis showed that AST (p=0.002) and ALT (p=0.037) were significantly correlated with the time from initial symptom onset to death.39 Furthermore, a study involving 1,590 COVID-19 cases showed that patients with elevated AST, ALT and TBIL levels at admission had case fatality rate of 68.6%, 54.3% and 39% respectively, suggesting that abnormal liver enzymes may be related to poor prognosis.40 In another large cohort study, the clinical course of 145 patients with severe liver injury (ALT >5 ULN) was worse, including higher rates of ICU admission (69%), intubation (65%), renal replacement therapy (33%), and mortality (42%).6 Therefore, the severity of COVID-19 is closely related to liver injury, which may predict adverse clinical outcomes.
Recently, with the increase of clinical data, the relationship between liver injury and the clinical outcomes of COVID-19 is being explored further. Ponziani et al.41 found that baseline of liver enzyme abnormality was associated with increased risk of ICU admission (odds ratio: 2.19 [95% confidence interval: 1.24–3.89], p=0.007) but not with mortality (odds ratio: 0.84 [95% confidence interval: 0.49–1.41], p=0.51), and it tended to normalize over time. However, more studies have shown that liver enzyme abnormality is an independent predictor of poor prognosis for COVID-19 patients.26,42,43 Yip et al.42 found that the incidence of elevated ALT/AST levels and acute liver injury were significantly higher in COVID-19 patients who developed adverse clinical outcomes (including ICU admission, use of invasive mechanical ventilation and/or death) than in those who did not (ALT/AST elevation: 70.9% vs. 19.1%, p<0.001; acute liver injury: 14.5% vs. 0.9%, p<0.001); multivariate analysis showed that ALT/AST elevation and acute liver injury were independently associated with adverse clinical outcomes. Similarly, the study by Piano et al.26 showed that liver enzyme abnormality was an independent predictor of transfer to ICU or death. Therefore, according to current evidence, it is necessary to regularly monitor the liver function of COVID-19 patients.
COVID-19-related liver injury in special populations
Pediatric patients with COVID-19
Severe COVID-19 is not common among children. The Chinese Center for Disease Control and Prevention survey of 72,314 cases of COVID-19 confirmed that the proportion of children under 10 years of age was less than 1%, most children presented with mild symptoms or asymptomatic infection.44 A study of 171 children with SARS-CoV-2 infection admitted to Wuhan Children’s Hospital showed that the incidence of elevated ALT and AST were 12.1% and 14.6%.45 Wang et al.46 analyzed 31 cases of SARS-CoV-2 infection in children from six provinces in northern China and found that 22.2% of them had elevated transaminases, with the peak values of ALT and AST levels being 68 U/L and 67 U/L, respectively. Due to the rare occurrence of liver enzyme abnormalities in children with COVID-19, the American Association for the Study of Liver Diseases warns that COVID-19 children with elevated AST or ALT should be fully evaluated for potential liver disease and other infections.47 Notably, Cui et al.48 reported a female infant case presented with pneumonia, liver injury and heart damage after infection with SARS-CoV-2, suggesting that children with COVID-19 may also suffer from multiple organ damage and rapid disease changes.
COVID-19 patients with chronic liver disease (CLD)
CLD poses a major public health burden. Patients with CLD, especially those with advanced liver diseases, such as cirrhosis and liver cancer, may be more susceptible to SARS-CoV-2 infection due to dysregulated innate and acquired immunity.49,50 Thus, the effects of different basic liver conditions on COVID-19-related liver injury need to be meticulously evaluated.3 According to existing reports, the proportion of CLD patients was low in COVID-19 cases. A large-scale meta-analysis showed that the pooled prevalence of underlying CLD among COVID-19 patients and critically ill patients were 3.6% and 3.9%, respectively.51 Similar to the clinical features of ordinary COVID-19 patients, the liver injury in COVID-19 patients with CLD is also correlated with the severity of disease and poor prognosis. A study involving 105 SARS-CoV-2 and chronic hepatitis B virus (HBV) co-infected patients showed that abnormal liver enzymes of those patients were relatively common at admission and significantly elevated during hospitalization, of which 14 patients (13.33%) developed liver injury, being more common in men (p=0.001); patients with liver injury had a higher proportion of severe cases, complications rate, and mortality (all p<0.05).52
Notably, the interaction between COVID-19 and pre-existing CLD could cause liver injury and aggravate the course of these two diseases.53–55 A study collected clinical data of 228 COVID-19 patients with CLD from 13 Asian countries, and found that among COVID-19 patients with CLD, 43% of CLD patients without cirrhosis developed acute liver damage, 20% of patients with compensated cirrhosis developed acute-on-chronic liver failure (ACLF) (11.6%) or acute decompensation (9.1%), 57% of patients with decompensated cirrhosis showed progressive aggravation of liver injury, and the mortality was as high as 43%.53 The cases of ACLF secondary to SARS-CoV-2 infection have also been reported.53,54,56,57 In addition, Iavarone et al.54 found that mortality was significantly higher in COVID-19 patients with cirrhosis than in COVID-19 patients without cirrhosis (34% vs. 18%; p=0.035) and cirrhotic patients hospitalized for bacterial infections (34% vs. 17%; p=0.03). Similarly, a study including 2,780 COVID-19 patients found that patients with CLD were at an increased risk for mortality (relative risk: 2.8; 95% confidence interval: 1.9–4.0; p<0.001) compared to patients without CLD, and the relative risk was markedly higher in patients with cirrhosis (relative risk: 4.6; 95% confidence interval: 2.6–8.3; p<0.001).55 The above studies suggest that COVID-19 can increase the liver burden of CLD patients and worsen the prognosis for patients with poor liver reserve capacity, and vice versa. Therefore, in order to avoid adverse clinical outcomes due to impaired liver reserve capacity, it is necessary to carry out risk stratification and personalization of the management for COVID-19 patients with CLD.
Pathogenesis of COVID-19-related liver injury
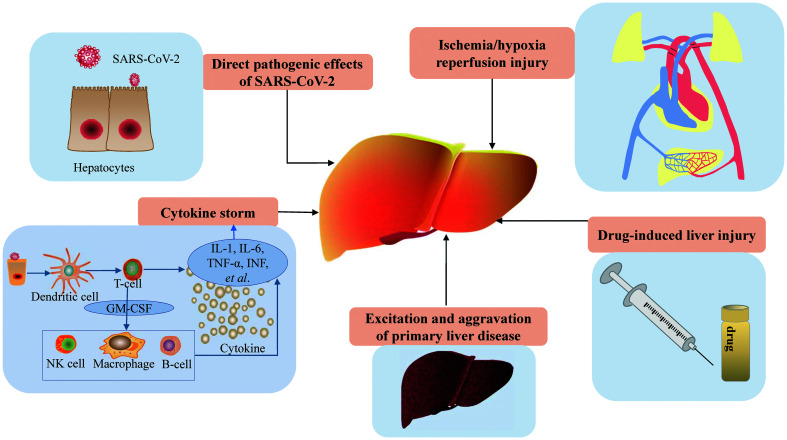
Although the mechanisms of liver injury in COVID-19 are not yet clear, multiple factors have been considered as the potential causes, including direct pathogenic effects of SARS-CoV-2, cytokine storm, hepatic ischemia and hypoxia, DILI, and aggravation of primary liver disease (Fig.1).
Fig. 1. Pathogenesis of COVID-19-related liver injury.
GM-CSF, granulocyte-macrophage colony stimulating factor; NK, natural killer.
Direct pathogenic effects of SARS-CoV-2
Wander et al.58 first reported a COVID-19 case with acute hepatitis as the initial manifestation, suggesting that liver could also be the target organ of SARS-CoV-2 infection. With the increasing studies of hepatic pathology in COVID-19, recently published data has indicated SARS-CoV-2 RNA and typical coronavirus particles in liver tissue,36,37 which suggests that SARS-CoV-2 could directly infect hepatocytes. As the cellular receptor of SARS-CoV-2, angiotensin-converting enzyme 2 (ACE2) is mainly expressed in bile duct epithelial cells rather than liver tissue; thus, the route of SARS-CoV-2 infection to hepatocytes is not clear. In a mouse model of acute liver injury with partial hepatectomy established by Guan et al.,59 the up-regulated expression of ACE2 mRNA after liver injury corresponds to the elevation of AST and ALT, suggesting that ACE2 expression in bile duct epithelial cells might be involved in the liver repair process, while those newborn hepatocytes might still maintain the characteristics of expressing ACE2, making them susceptible to SARS-CoV-2. Attention should also be paid to the non-ACE2-dependent manner of SARS-CoV-2 infection. The antibody-dependent enhancement (referred to as ADE) of virus infection is a phenomenon that virus-specific antibodies can enhance the replication and infection ability of the virus to enter monocytes/macrophages and granulocytes by interacting with Fc receptors and/or complement receptors.60 A study showed that anti-SARS-CoV spike protein antibody can trigger ADE and mediate SARS-CoV entrance into immune cells that do not express ACE2.61 Therefore, it is worth considering whether ADE can also mediate SARS-CoV-2’s infection in hepatocytes in a non-ACE2-dependent manner and then cause liver damage. In addition, a study showed that hypoxia can induce an increase of ACE2 expression in human hepatocytes.62 Since patients with COVID-19 often suffer from different degrees of hypoxia and systemic inflammatory reactions, hepatocytes may increase their susceptibility to SARS-CoV-2 through up-regulation of ACE2 expression under hypoxia conditions.
Cytokine storm
Laboratory examination of COVID-19 patients has mainly showed a decrease of lymphocytes, an increase of infection-related markers (including PCT, ESR, SF, and CRP) and inflammatory cytokines (including tumor necrosis factor-α [TNF-α], IL-2 receptor [IL-2R], and IL-6). These biochemical indicators were significantly increased in severe cases, which can lead to aggravation of inflammatory reactions and generation of cytokine storms.63 Cytokine storm is the excessive activation of the immune system caused by infection, drugs, or certain diseases. It involves the continuous activation and proliferation of various lymphocytes and macrophages, and leads to a rapid and large secretion of various cytokines, including TNF-α, interferons (INFs, including INF-α, INF-β, INF-γ), IL-1, IL-6, IL-8, finally causing serious damage to vital organs through the inflammatory cascade reaction.64 Similar to SARS, cytokine storm is a characteristic manifestation in critically ill COVID-19 patients, which is also the main cause of disseminated intravascular coagulation, acute respiratory distress syndrome, and multiple organ failure.65 Phipps et al.6 found that in COVID-19 patients, severe liver injury was associated with markers of end-organ dysfunction, including peak levels of high-sensitivity troponin, CK, and serum creatinine, as well as inflammatory markers, including peak PCT, CRP, D-dimer, SF and IL-6 levels (all p<0.001), suggesting that liver injury may be related to the inflammatory response and cytokine storm.6 In addition, compared with ALT, elevated AST level seems to be more common in COVID-19 patients with liver injury and is closely related to poor prognosis. AST is widely distributed in various organs, such as liver, heart muscle, skeletal muscle, kidney, etc. Lei et al.12 found that AST elevation was positively correlated with lymphocyte reduction and neutrophil elevation in COVID-19 patients, and the latter was a reliable indicator of disease severity, suggesting that multi-organ damage caused by cytokine storms may contribute to AST level abnormality.
Hepatic ischemia and hypoxia
Hypoxic hepatitis (HH), which is also known as ischemic hepatitis and shock liver, refers to reversible central necrosis of the hepatic lobule caused by severe hypotension and hypoxemia. It is characterized by sharp and transient elevation of serum transaminase, which usually reaches a peak level within 24 h (>20 ULN) and returns to normal within 1–2 weeks. HH is caused by many factors, with heart failure (39–70%) being the main cause, followed by septic shock (32%), septicemia (23%) and respiratory failure (15%).66 Severe heart failure can cause liver congestion. The release of inflammatory mediators and endotoxin during septic shock may reduce the oxygen uptake ability of hepatocytes. When the above conditions are combined with oxidative stress induced by tissue ischemia-reperfusion, HH is more likely to occur.66–68 A cohort study of 191 COVID-19 cases in Wuhan showed that the most common complication of COVID-19 patients was sepsis (59%), followed by respiratory failure (54%), acute respiratory distress syndrome (31%), heart failure (23%) and septic shock (20%).69 Moreover, most COVID-19 patients have different degrees of hypoxemia and more than 40% of them need oxygen therapy.38 The above complications are all risk factors for HH, indicating that COVID-19 patients may have a high risk of HH. In addition, in liver autopsy of a patient who died of COVID-19, a few hepatocytes with slight vesicular steatosis and watery degeneration as well as inflammatory cells were observed, which was considered to have been caused by hypoxia and ischemia-reperfusion.5 Therefore, from the perspective of pathophysiology and histology, ischemia and hypoxia may be one of the pathogeneses of COVID-19-related liver injury. It should be noted that in the current reports of COVID-19-related liver injury, most cases present with mild to moderate elevation of liver enzymes, while cases of significant liver enzyme elevation (>10 ULN) are still uncommon. Therefore, further studies are needed to clarify HH as a possible mechanism to liver injury in COVID-19.
DILI
Currently, there are no specific antiviral drugs for COVID-19. Interferon-α, lopinavir/ritonavir, ribavirin, chloroquine phosphate and traditional Chinese medicine are the main drugs for antiviral treatment.70 It has been reported that more than 50% of COVID-19 patients received antibiotic treatment during hospitalization.38 In addition, it is relatively common for fever patients to take antipyretic-analgesics before admission. As the main component of antipyretic-analgesics, acetaminophen is the primary cause of acute liver failure in occident.71 Of note, the above drugs have been reported to associate with abnormal liver enzymes in COVID-19 patients.12,25,26,42,72 Piano et al.26 found that de novo abnormalities of liver enzymes were more common in COVID-19 patients receiving lopinavir/ritonavir (64% vs. 48%, p=0.045), acetaminophen (63% vs. 47%, p=0.048), piperacillin/tazobactam (72% vs. 50%, p=0.013) and tocilizumab (82% vs. 52%, p=0.009). Another study showed that the use of lopinavir/ritonavir, ribavirin, interferon-beta and corticosteroids was independently associated with ALT/AST elevation in COVID-19 patients.42 Falcão et al.72 reported a significant liver enzyme elevation (∼10 ULN) in a COVID-19 patient after using hydroxychloroquine, suggesting that hydroxychloroquine may have hepatotoxicity. Lopinavir/ritonavir is mainly metabolized by liver, and Fan et al.25 found that the proportion of patients treated with lopinavir/ritonavir was significantly higher in the group with liver injury than the group without (57.8% vs. 31.3%, p=0.01), suggesting that liver injury may be related to the use of lopinavir/ritonavir.
Remdesivir has been reported to cause a significant increase in serum transaminase after usage in COVID-19 patients.73,74 In a study, after remdesivir treatment, marked ALT/AST elevations and life-threatening elevations were observed in 6% and 2% of COVID-19 patients, respectively.75 Furthermore, a pharmacovigilance analysis performed using VigiBase to summarize hepatic impairment showed that with remdesivir, increased liver enzymes were the most frequent adverse drug reactions (114, 88%), involving ALT/AST in 79 cases (61%) and bilirubin in 4 cases (3%).76 According to Leegwater et al.,77 who reported that one severe COVID-19 patient experienced an acute increase in ALT (1,305 IU/L) and AST (1,461 U/L) levels after receiving remdesivir treatment, and the interaction between P-glycoprotein inhibitors chloroquine and remdesivir had been considered to cause this acute toxic liver injury.
Muhović et al.78 described the first case of a severe COVID-19 patient who developed DILI associated with the use of tocilizumab, marked by a 40-fold increase in transaminases levels. Notably, in this research, the diagnosis of DILI induced by tocilizumab was based on the Roussel Uclaf Causality Assessment Method (RUCAM). RUCAM is appreciated as a structured, standardized, validated, and liver-specific diagnostic approach that attributes scores to individual key items, providing final quantitative causality grading for each suspect drug/herb in a case report.79 In brief, RUCAM quantifies the strength of association between a liver injury and the medication implicated as the cause of the injury, so it is recommended for assessing DILI cases.80,81 In practice, DILI commonly stands for the idiosyncratic DILI (iDILI) which lacks clear dose dependency and is caused by unpredictable events due to immunologic or metabolic drug reactions.82 However, at present, not many studies have used RUCAM to evaluate iDILI in COVID-19. Considering the use of pharmacotherapy for COVID-19 may reflect disease severity, the diagnosis of iDILI in COVID-19 may be confounded. Thus, using the updated RUCAM to verify causality in suspected iDILI cases is necessary.
COVID-19 patients, especially critically ill patients, often need to take multiple drugs combined with therapy, which is more likely to increase the risk of DILI under the condition of systemic immune disorder. Therefore, the use of medicine should be rational to reduce the chance of DILI in clinical practice.
Excitation and aggravation of primary liver disease
Existing research has shown that COVID-19 patients with underlying liver disease have a higher risk of progressing to severe COVID-19 and could exacerbate original liver disease.53,54 There are multiple reports of COVID-19 patients complicated with ACLF, emphasizing the influence of several factors on basic liver diseases during the course of COVID-19. Hepatitis B patients who are receiving antiviral therapy may have hepatitis attacks if they stop using anti-HBV drugs during the period of COVID-19. For those who have not received anti-HBV therapy, receiving high-dose hormone therapy may lead to hepatitis B virus reactivation (referred to herein as HBVr).4 Therefore, as the American Gastroenterological Association recommends, antiviral prophylaxis should be provided for patients with high and moderate risk of HBVr treated with immunosuppressive drugs, while routine antiviral prophylaxis in patients with low risk of HBVr should be carefully considered.83 In addition, attention should be paid to the potential impact of MAFLD on COVID-19-related liver injury. MAFLD is associated with extra-hepatic manifestations of metabolic syndrome. As an important immune organ of the human body, the liver contains a large number of macrophages. MAFLD patients usually have different levels of elevated inflammatory cytokines and SARS-CoV-2 infection can lead to immune stress; as such, it may accelerate the progression of MAFLD and lead to liver injury.84
The pathogenesis of COVID-19-related liver injury has not been clarified at present. In addition to the possible pathogenesis mentioned above, some scholars have also proposed the hypothesis that SARS-CoV-2 directly acts on hepatocytes or activates Kupffer cells through the “intestine-liver” axis after intestinal infection, and thus leads to liver injury.85 In addition, some scholars believe that mechanical ventilation increases the positive end-expiratory pressure of the patient, causing an increase in right atrial pressure and obstructing venous return, thus leading to liver congestion, which may be one of the mechanisms of liver injury in COVID-19.86 However, no research has confirmed the above views so far. Notably, the pathological changes of COVID-19-related liver injury are complex and diverse, which may be involved in a variety of pathogenesis factors, and therefore more related research is needed to explore its mechanism.
Treatment of COVID-19-related liver injury
At present, antiviral treatment for COVID-19 remains investigational, while etiological treatment is limited. Thus, for patients with COVID-19-related liver injury, the current therapy is mainly supportive treatment based on the severity of liver injury. Mild liver biochemical abnormalities in COVID-19 patients are usually transient and can recover without special treatment. However, for patients with acute severe liver injury, the probable causes of injury should be fully considered, including a history of preexisting liver disease, exposure to hepatotoxins, hypoxia, and circulation status. Meanwhile, liver biochemical indicators should also be closely monitored to prevent the occurrence of acute liver failure. Respiratory and circulatory support should be strengthened in patients with hypoxic hepatitis. For patients with suspected DILI, discontinuation or reduction of the use of suspected drugs should be considered.4 As for patients with underlying liver disease, liver-protecting drugs should be given appropriately, and the combination of immunosuppressive drugs and antiviral therapy in those with viral hepatitis should be carefully considered. For liver transplant patients, although studies showed that reducing immunosuppressive agents did not increase the risk of mortality, the application of immunosuppressant should still be weighed.87
Conclusions
In conclusion, the overall incidence of abnormal liver enzymes is high in COVID-19 patients, especially in severe cases, and is associated with poor prognosis. Advanced age, male sex and high BMI are the predictors of abnormal liver enzymes in COVID-19. Pediatric patients with COVID-19-related liver injury usually experience minor symptoms. COVID-19 patients with CLD have a higher risk of developing liver injury and progressing to severe cases. COVID-19 may aggravate their primary liver diseases and the prognosis mainly depends on the liver reserve capacity. In clinical practice, extra attention should be paid to the potential impact of underlying diseases on COVID-19-related liver injury, although the clinical data is still insufficient.
So far, the understanding of COVID-19-related liver injury has been constantly increasing. Recent research has shown that SARS-CoV-2 can directly infect hepatocytes, providing a new vision for the pathogenesis of liver injury. In addition, close attention should be paid to the adverse effect of drugs on liver in the treatment for COVID-19, especially iDILI. In fact, there is compelling evidence that the liver injury observed in patients with COVID-19 is at least partially due to iDILI. To verify causality in suspected cases, the updated RUCAM published in 2016 should be used. Until there is verifiable evidence to support the theory, clinicians should carefully consider the risk of DILI and try to avoid the overuse of relevant drugs. At last, although COVID-19 vaccination has begun worldwide, the threat of COVID-19 and its hepatic damage should not be underestimated. Further research on effective treatment methods for COVID-19-related liver injury is essential. Clinical staff should continue to summarize the clinical characteristics and treatment experience of COVID-19-related liver injury to provide new theoretical basis for its standardized treatment and in-depth mechanism research.
Abbreviations
- ACE2
angiotensin-converting enzyme 2
- ACLF
acute-on-chronic liver failure
- ADE
antibody-dependent enhancement
- ALP
alkaline phosphatase
- ALT
alanine aminotransferase
- AST
aspartate aminotransferase
- BMI
body mass index
- CK
creatine kinase
- CLD
chronic liver disease
- COVID-19
coronavirus disease 2019
- CRP
C-reactive protein
- CT
computed tomography
- DILI
drug-induced liver injury
- ESR
erythrocyte sedimentation rate
- GGT
gamma-glutamyl transferase
- HBV
hepatitis B virus
- HBVr
hepatitis B virus reactivation
- HH
hypoxic hepatitis
- ICU
intensive care unit
- iDILI
idiosyncratic drug-induced liver injury
- INFs
interferons
- IL
interleukin
- LDH
lactate dehydrogenase
- MAFLD
metabolic-associated fatty liver disease
- PCT
procalcitonin
- RNA
ribonucleic acid
- RUCAM
Roussel Uclaf Causality Assessment Method
- SARS-CoV
severe acute respiratory syndrome coronavirus
- SARS-CoV-2
severe acute respiratory syndrome coronavirus-2
- SF
serum ferritin
- TBIL
total bilirubin
- TNF-α
tumor necrosis factor-α
- ULN
upper limit of normal
Data sharing statement
All data are available upon request.
References
- 1.Verity R, Okell LC, Dorigatti I, Winskill P, Whittaker C, Imai N, et al. Estimates of the severity of coronavirus disease 2019: a model-based analysis. Lancet Infect Dis. 2020;20(6):669–677. doi: 10.1016/S1473-3099(20)30243-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yang X, Yu Y, Xu J, Shu H, Xia J, Liu H, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475–481. doi: 10.1016/S2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang C, Shi L, Wang FS. Liver injury in COVID-19: management and challenges. Lancet Gastroenterol Hepatol. 2020;5(5):428–430. doi: 10.1016/S2468-1253(20)30057-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.The protocol for prevention, diagnosis and treatment of liver injury in coronavirus disease 2019. Zhonghua Gan Zang Bing Za Zhi. 2020;28(3):217–221. doi: 10.3760/cma.j.cn501113-20200309-00095. [DOI] [PubMed] [Google Scholar]
- 5.Cai Q, Huang D, Yu H, Zhu Z, Xia Z, Su Y, et al. COVID-19: Abnormal liver function tests. J Hepatol. 2020;73(3):566–574. doi: 10.1016/j.jhep.2020.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Phipps MM, Barraza LH, LaSota ED, Sobieszczyk ME, Pereira MR, Zheng EX, et al. Acute liver injury in COVID-19: Prevalence and association with clinical outcomes in a large U.S. cohort. Hepatology. 2020;72(3):807–817. doi: 10.1002/hep.31404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Woreta TA, Alqahtani SA. Evaluation of abnormal liver tests. Med Clin North Am. 2014;98(1):1–16. doi: 10.1016/j.mcna.2013.09.005. [DOI] [PubMed] [Google Scholar]
- 8.Whitehead MW, Hawkes ND, Hainsworth I, Kingham JG. A prospective study of the causes of notably raised aspartate aminotransferase of liver origin. Gut. 1999;45(1):129–133. doi: 10.1136/gut.45.1.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Daniel S, Ben-Menachem T, Vasudevan G, Ma CK, Blumenkehl M. Prospective evaluation of unexplained chronic liver transaminase abnormalities in asymptomatic and symptomatic patients. Am J Gastroenterol. 1999;94(10):3010–3014. doi: 10.1111/j.1572-0241.1999.01451.x. [DOI] [PubMed] [Google Scholar]
- 10.Newsome PN, Cramb R, Davison SM, Dillon JF, Foulerton M, Godfrey EM, et al. Guidelines on the management of abnormal liver blood tests. Gut. 2018;67(1):6–19. doi: 10.1136/gutjnl-2017-314924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dufour DR, Lott JA, Nolte FS, Gretch DR, Koff RS, Seeff LB. Diagnosis and monitoring of hepatic injury. II. Recommendations for use of laboratory tests in screening, diagnosis, and monitoring. Clin Chem. 2000;46(12):2050–2068. doi: 10.1093/clinchem/46.12.2050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lei F, Liu YM, Zhou F, Qin JJ, Zhang P, Zhu L, et al. Longitudinal association between markers of liver injury and mortality in COVID-19 in China. Hepatology. 2020;72(2):389–398. doi: 10.1002/hep.31301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Qi X, Liu C, Jiang Z, Gu Y, Zhang G, Shao C, et al. Multicenter analysis of clinical characteristics and outcomes in patients with COVID-19 who develop liver injury. J Hepatol. 2020;73(2):455–458. doi: 10.1016/j.jhep.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huang H, Chen S, Li H, Zhou XL, Dai Y, Wu J, et al. The association between markers of liver injury and clinical outcomes in patients with COVID-19 in Wuhan. Aliment Pharmacol Ther. 2020;52(6):1051–1059. doi: 10.1111/apt.15962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Li T, Guo Y, Zhuang X, Huang L, Zhang X, Wei F, et al. Abnormal liver-related biomarkers in COVID-19 patients and the role of prealbumin. Saudi J Gastroenterol. 2020;26(5):272–278. doi: 10.4103/sjg.SJG_239_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Petrilli CM, Jones SA, Yang J, Rajagopalan H, O’Donnell L, Chernyak Y, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. doi: 10.1136/bmj.m1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Channappanavar R, Fett C, Mack M, Ten Eyck PP, Meyerholz DK, Perlman S. Sex-based differences in susceptibility to severe acute respiratory syndrome coronavirus infection. J Immunol. 2017;198(10):4046–4053. doi: 10.4049/jimmunol.1601896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Piton A, Poynard T, Imbert-Bismut F, Khalil L, Delattre J, Pelissier E, et al. Factors associated with serum alanine transaminase activity in healthy subjects: consequences for the definition of normal values, for selection of blood donors, and for patients with chronic hepatitis C. MULTIVIRC Group. Hepatology. 1998;27(5):1213–1219. doi: 10.1002/hep.510270505. [DOI] [PubMed] [Google Scholar]
- 19.Hundt MA, Deng Y, Ciarleglio MM, Nathanson MH, Lim JK. Abnormal liver tests in COVID-19: A retrospective observational cohort study of 1,827 patients in a major U.S. hospital network. Hepatology. 2020;72(4):1169–1176. doi: 10.1002/hep.31487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bloom PP, Meyerowitz EA, Reinus Z, Daidone M, Gustafson J, Kim AY, et al. Liver biochemistries in hospitalized patients with COVID-19. Hepatology. 2021;73(3):890–900. doi: 10.1002/hep.31326. [DOI] [PubMed] [Google Scholar]
- 21.Bertolini A, van de Peppel IP, Bodewes FAJA, Moshage H, Fantin A, Farinati F, et al. Abnormal liver function tests in patients with COVID-19: Relevance and potential pathogenesis. Hepatology. 2020;72(5):1864–1872. doi: 10.1002/hep.31480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052–2059. doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Younossi Z, Anstee QM, Marietti M, Hardy T, Henry L, Eslam M, et al. Global burden of NAFLD and NASH: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2018;15(1):11–20. doi: 10.1038/nrgastro.2017.109. [DOI] [PubMed] [Google Scholar]
- 24.Zhang Y, Zheng L, Liu L, Zhao M, Xiao J, Zhao Q. Liver impairment in COVID-19 patients: A retrospective analysis of 115 cases from a single centre in Wuhan city, China. Liver Int. 2020;40(9):2095–2103. doi: 10.1111/liv.14455. [DOI] [PubMed] [Google Scholar]
- 25.Fan Z, Chen L, Li J, Cheng X, Yang J, Tian C, et al. Clinical features of COVID-19-related liver functional abnormality. Clin Gastroenterol Hepatol. 2020;18(7):1561–1566. doi: 10.1016/j.cgh.2020.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Piano S, Dalbeni A, Vettore E, Benfaremo D, Mattioli M, Gambino CG, et al. Abnormal liver function tests predict transfer to intensive care unit and death in COVID-19. Liver Int. 2020;40(10):2394–2406. doi: 10.1111/liv.14565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schattenberg JM, Labenz C, Wörns MA, Menge P, Weinmann A, Galle PR, et al. Patterns of liver injury in COVID-19 - a German case series. United European Gastroenterol J. 2020;8(7):814–819. doi: 10.1177/2050640620931657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Philips CA, Ahamed R, Augustine P. SARS-CoV-2 related liver impairment - perception may not be the reality. J Hepatol. 2020;73(4):991–992. doi: 10.1016/j.jhep.2020.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xie H, Zhao J, Lian N, Lin S, Xie Q, Zhuo H. Clinical characteristics of non-ICU hospitalized patients with coronavirus disease 2019 and liver injury: A retrospective study. Liver Int. 2020;40(6):1321–1326. doi: 10.1111/liv.14449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bhayana R, Som A, Li MD, Carey DE, Anderson MA, Blake MA, et al. Abdominal imaging findings in COVID-19: Preliminary observations. Radiology. 2020;297(1):E207–E215. doi: 10.1148/radiol.2020201908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bradley BT, Maioli H, Johnston R, Chaudhry I, Fink SL, Xu H, et al. Histopathology and ultrastructural findings of fatal COVID-19 infections in Washington State: a case series. Lancet. 2020;396(10247):320–332. doi: 10.1016/S0140-6736(20)31305-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Buja LM, Wolf DA, Zhao B, Akkanti B, McDonald M, Lelenwa L, et al. The emerging spectrum of cardiopulmonary pathology of the coronavirus disease 2019 (COVID-19): Report of 3 autopsies from Houston, Texas, and review of autopsy findings from other United States cities. Cardiovasc Pathol. 2020;48:107233. doi: 10.1016/j.carpath.2020.107233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tian S, Xiong Y, Liu H, Niu L, Guo J, Liao M, et al. Pathological study of the 2019 novel coronavirus disease (COVID-19) through postmortem core biopsies. Mod Pathol. 2020;33(6):1007–1014. doi: 10.1038/s41379-020-0536-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Elsoukkary SS, Mostyka M, Dillard A, Berman DR, Ma LX, Chadburn A, et al. Autopsy findings in 32 patients with COVID-19: A single-institution experience. Pathobiology. 2021;88(1):56–68. doi: 10.1159/000511325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lagana SM, Kudose S, Iuga AC, Lee MJ, Fazlollahi L, Remotti HE, et al. Hepatic pathology in patients dying of COVID-19: a series of 40 cases including clinical, histologic, and virologic data. Mod Pathol. 2020;33(11):2147–2155. doi: 10.1038/s41379-020-00649-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang Y, Liu S, Liu H, Li W, Lin F, Jiang L, et al. SARS-CoV-2 infection of the liver directly contributes to hepatic impairment in patients with COVID-19. J Hepatol. 2020;73(4):807–816. doi: 10.1016/j.jhep.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382(18):1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang B, Zhou X, Qiu Y, Song Y, Feng F, Feng J, et al. Clinical characteristics of 82 cases of death from COVID-19. PLoS One. 2020;15(7):e0235458. doi: 10.1371/journal.pone.0235458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Chen R, Liang W, Jiang M, Guan W, Zhan C, Wang T, et al. Risk factors of fatal outcome in hospitalized subjects with coronavirus disease 2019 from a nationwide analysis in China. Chest. 2020;158(1):97–105. doi: 10.1016/j.chest.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ponziani FR, Del Zompo F, Nesci A, Santopaolo F, Ianiro G, Pompili M, et al. Liver involvement is not associated with mortality: results from a large cohort of SARS-CoV-2-positive patients. Aliment Pharmacol Ther. 2020;52(6):1060–1068. doi: 10.1111/apt.15996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yip TC, Lui GC, Wong VW, Chow VC, Ho TH, Li TC, et al. Liver injury is independently associated with adverse clinical outcomes in patients with COVID-19. Gut. 2021;70(4):733–742. doi: 10.1136/gutjnl-2020-321726. [DOI] [PubMed] [Google Scholar]
- 43.Meszaros M, Meunier L, Morquin D, Klouche K, Fesler P, Malezieux E, et al. Abnormal liver tests in patients hospitalized with Coronavirus disease 2019: Should we worry? Liver Int. 2020;40(8):1860–1864. doi: 10.1111/liv.14557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 45.Lu X, Zhang L, Du H, Zhang J, Li YY, Qu J, et al. SARS-CoV-2 infection in children. N Engl J Med. 2020;382(17):1663–1665. doi: 10.1056/NEJMc2005073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wang D, Ju XL, Xie F, Lu Y, Li FY, Huang HH, et al. Clinical analysis of 31 cases of 2019 novel coronavirus infection in children from six provinces (autonomous region) of northern China. Zhonghua Er Ke Za Zhi. 2020;58(4):269–274. doi: 10.3760/cma.j.cn112140-20200225-00138. [DOI] [PubMed] [Google Scholar]
- 47.Fix OK, Hameed B, Fontana RJ, Kwok RM, McGuire BM, Mulligan DC, et al. Clinical best practice advice for hepatology and liver transplant providers during the COVID-19 pandemic: AASLD Expert Panel Consensus Statement. Hepatology. 2020;72(1):287–304. doi: 10.1002/hep.31281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cui Y, Tian M, Huang D, Wang X, Huang Y, Fan L, et al. A 55-day-old female infant infected with 2019 novel coronavirus disease: Presenting With pneumonia, liver injury, and heart damage. J Infect Dis. 2020;221(11):1775–1781. doi: 10.1093/infdis/jiaa113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Albillos A, Lario M, Álvarez-Mon M. Cirrhosis-associated immune dysfunction: distinctive features and clinical relevance. J Hepatol. 2014;61(6):1385–1396. doi: 10.1016/j.jhep.2014.08.010. [DOI] [PubMed] [Google Scholar]
- 50.Irvine KM, Ratnasekera I, Powell EE, Hume DA. Causes and consequences of innate immune dysfunction in cirrhosis. Front Immunol. 2019;10:293. doi: 10.3389/fimmu.2019.00293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kulkarni AV, Kumar P, Tevethia HV, Premkumar M, Arab JP, Candia R, et al. Systematic review with meta-analysis: liver manifestations and outcomes in COVID-19. Aliment Pharmacol Ther. 2020;52(4):584–599. doi: 10.1111/apt.15916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zou X, Fang M, Li S, Wu L, Gao B, Gao H, et al. Characteristics of liver function in patients with SARS-CoV-2 and chronic HBV coinfection. Clin Gastroenterol Hepatol. 2021;19(3):597–603. doi: 10.1016/j.cgh.2020.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sarin SK, Choudhury A, Lau GK, Zheng MH, Ji D, Abd-Elsalam S, et al. Pre-existing liver disease is associated with poor outcome in patients with SARS CoV2 infection; The APCOLIS Study (APASL COVID-19 Liver Injury Spectrum Study) Hepatol Int. 2020;14(5):690–700. doi: 10.1007/s12072-020-10072-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Iavarone M, D’Ambrosio R, Soria A, Triolo M, Pugliese N, Del Poggio P, et al. High rates of 30-day mortality in patients with cirrhosis and COVID-19. J Hepatol. 2020;73(5):1063–1071. doi: 10.1016/j.jhep.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Singh S, Khan A. Clinical characteristics and outcomes of coronavirus disease 2019 among patients with preexisting liver disease in the United States: A multicenter research network study. Gastroenterology. 2020;159(2):768–771.e3. doi: 10.1053/j.gastro.2020.04.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Qiu H, Wander P, Bernstein D, Satapathy SK. Acute on chronic liver failure from novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Liver Int. 2020;40(7):1590–1593. doi: 10.1111/liv.14506. [DOI] [PubMed] [Google Scholar]
- 57.Große K, Kramer M, Trautwein C, Bruns T. SARS-CoV-2 as an extrahepatic precipitator of acute-on-chronic liver failure. Liver Int. 2020;40(7):1792–1793. doi: 10.1111/liv.14540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wander P, Epstein M, Bernstein D. COVID-19 presenting as acute hepatitis. Am J Gastroenterol. 2020;115(6):941–942. doi: 10.14309/ajg.0000000000000660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Guan GW, Gao L, Wang JW, Wen XJ, Mao TH, Peng SW, et al. Exploring the mechanism of liver enzyme abnormalities in patients with novel coronavirus-infected pneumonia. Zhonghua Gan Zang Bing Za Zhi. 2020;28(2):100–106. doi: 10.3760/cma.j.issn.1007-3418.2020.02.002. [DOI] [PubMed] [Google Scholar]
- 60.Tirado SM, Yoon KJ. Antibody-dependent enhancement of virus infection and disease. Viral Immunol. 2003;16(1):69–86. doi: 10.1089/088282403763635465. [DOI] [PubMed] [Google Scholar]
- 61.Wang SF, Tseng SP, Yen CH, Yang JY, Tsao CH, Shen CW, et al. Antibody-dependent SARS coronavirus infection is mediated by antibodies against spike proteins. Biochem Biophys Res Commun. 2014;451(2):208–214. doi: 10.1016/j.bbrc.2014.07.090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Paizis G, Tikellis C, Cooper ME, Schembri JM, Lew RA, Smith AI, et al. Chronic liver injury in rats and humans upregulates the novel enzyme angiotensin converting enzyme 2. Gut. 2005;54(12):1790–1796. doi: 10.1136/gut.2004.062398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y, et al. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis. 2020;71(15):762–768. doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tisoncik JR, Korth MJ, Simmons CP, Farrar J, Martin TR, Katze MG. Into the eye of the cytokine storm. Microbiol Mol Biol Rev. 2012;76(1):16–32. doi: 10.1128/MMBR.05015-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Azkur AK, Akdis M, Azkur D, Sokolowska M, van de Veen W, Brüggen MC, et al. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy. 2020;75(7):1564–1581. doi: 10.1111/all.14364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Waseem N, Chen PH. Hypoxic hepatitis: A review and clinical update. J Clin Transl Hepatol. 2016;4(3):263–268. doi: 10.14218/JCTH.2016.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Henrion J. Hypoxic hepatitis. Liver Int. 2012;32(7):1039–1052. doi: 10.1111/j.1478-3231.2011.02655.x. [DOI] [PubMed] [Google Scholar]
- 68.Ebert EC. Hypoxic liver injury. Mayo Clin Proc. 2006;81(9):1232–1236. doi: 10.4065/81.9.1232. [DOI] [PubMed] [Google Scholar]
- 69.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Diagnosis and treatment protocol for novel coronavirus pneumonia (Trial version 7) Chin Med J (Engl) 2020;133(9):1087–1095. doi: 10.1097/CM9.0000000000000819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Yu YC, Mao YM, Chen CW, Chen JJ, Chen J, Cong WM, et al. CSH guidelines for the diagnosis and treatment of drug-induced liver injury. Hepatol Int. 2017;11(3):221–241. doi: 10.1007/s12072-017-9793-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Falcão MB, Pamplona de Góes Cavalcanti L, Filgueiras Filho NM, Antunes de Brito CA. Case report: Hepatotoxicity associated with the use of hydroxychloroquine in a patient with COVID-19. Am J Trop Med Hyg. 2020;102(6):1214–1216. doi: 10.4269/ajtmh.20-0276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Clinical and virologic characteristics of the first 12 patients with coronavirus disease 2019 (COVID-19) in the United States. Nat Med. 2020;26(6):861–868. doi: 10.1038/s41591-020-0877-5. [DOI] [PubMed] [Google Scholar]
- 74.Zampino R, Mele F, Florio LL, Bertolino L, Andini R, Galdo M, et al. Liver injury in remdesivir-treated COVID-19 patients. Hepatol Int. 2020;14(5):881–883. doi: 10.1007/s12072-020-10077-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Goldman JD, Lye DCB, Hui DS, Marks KM, Bruno R, Montejano R, et al. Remdesivir for 5 or 10 days in patients with severe Covid-19. N Engl J Med. 2020;383(19):1827–1837. doi: 10.1056/NEJMoa2015301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Montastruc F, Thuriot S, Durrieu G. Hepatic disorders with the use of remdesivir for coronavirus 2019. Clin Gastroenterol Hepatol. 2020;18(12):2835–2836. doi: 10.1016/j.cgh.2020.07.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Leegwater E, Strik A, Wilms EB, Bosma LBE, Burger DM, Ottens TH, et al. Drug-induced liver injury in a patient with coronavirus disease 2019: Potential interaction of remdesivir with P-Glycoprotein inhibitors. Clin Infect Dis. 2021;72(7):1256–1258. doi: 10.1093/cid/ciaa883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Muhović D, Bojović J, Bulatović A, Vukčević B, Ratković M, Lazović R, et al. First case of drug-induced liver injury associated with the use of tocilizumab in a patient with COVID-19. Liver Int. 2020;40(8):1901–1905. doi: 10.1111/liv.14516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Danan G, Teschke R. RUCAM in drug and herb induced liver injury: The update. Int J Mol Sci. 2015;17(1):14. doi: 10.3390/ijms17010014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Teschke R, Danan G. Drug induced liver injury with analysis of alternative causes as confounding variables. Br J Clin Pharmacol. 2018;84(7):1467–1477. doi: 10.1111/bcp.13593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.da Silva LA, Simonato LE, Ramos RR. Phylogeny and pathogenesis of SARS-CoV-2: A systematic study. Journal of Modern Medicinal Chemistry. 2020;8(1):49–55. doi: 10.12970/2308-8044.2020.08.06. [DOI] [Google Scholar]
- 82.Teschke R. Idiosyncratic DILI: Analysis of 46,266 cases assessed for causality by RUCAM and published from 2014 to early 2019. Front Pharmacol. 2019;10:730. doi: 10.3389/fphar.2019.00730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Reddy KR, Beavers KL, Hammond SP, Lim JK, Falck-Ytter YT. American Gastroenterological Association Institute guideline on the prevention and treatment of hepatitis B virus reactivation during immunosuppressive drug therapy. Gastroenterology. 2015;148(1):215–219. doi: 10.1053/j.gastro.2014.10.039. quiz e16-7. [DOI] [PubMed] [Google Scholar]
- 84.Prins GH, Olinga P. Potential implications of COVID-19 in non-alcoholic fatty liver disease. Liver Int. 2020;40(10):2568. doi: 10.1111/liv.14484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.He N, Feng G, Yao CZ, Shen NN, Qin J, Kang GL. The analysis of present researches and challenges of COVID-19 with liver injury. Chinese Journal of Gastroenterology and Hepatology. 2020;29(4):397–400. doi: 10.3969/j.issn.1006-5709.2020.04.008. [DOI] [Google Scholar]
- 86.Chen P, Zhou B. Clinical characteristics of COVID-19 patients with abnormal liver tests. J Hepatol. 2020;73(3):712–713. doi: 10.1016/j.jhep.2020.04.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rabiee A, Sadowski B, Adeniji N, Perumalswami PV, Nguyen V, Moghe A, et al. Liver injury in liver transplant recipients with coronavirus disease 2019 (COVID-19): U.S. multicenter experience. Hepatology. 2020;72(6):1900–1911. doi: 10.1002/hep.31574. [DOI] [PMC free article] [PubMed] [Google Scholar]