Abstract
There has been a recent surge of interest in the unique low molecular weight dietary thiol/thione, ergothioneine. This compound can accumulate at high levels in the body from diet and may play important physiological roles in human health and development, and possibly in prevention and treatment of disease. Blood levels of ergothioneine decline with age and onset of various diseases. Here we highlight recent advances in our knowledge of ergothioneine.
Keywords: Ergothioneine, OCTN1, Antioxidant, Thiol/thione, Mushroom
Abbreviations: 7KC, 7-ketocholesterol; AD, Alzheimer disease; AMD, age-related macular degeneration; ARE, antioxidant response element; CD, Crohn's disease; CDNB, 1-chloro-2,4-dinitrobenzene; COX, cyclooxygenase; DMN, dimethylnitrosamine; EFSA, European Food Safety Authority; ET, ergothioneine; ETT, ergothioneine transporter; GSH, reduced glutathione; GST, glutathione-S-transferases; HMEC, human mammary epithelial cells; IBD, inflammatory bowel disease; ICAM, intercellular adhesion molecule; IL, interleukin; LDL, low-density lipoprotein; MCI, mild cognitive impairment; NF-κB, nuclear factor kappa B; NOX, NADPH oxidase; Nrf2, nuclear factor erythroid 2–related factor 2; OCTN1, organic cation transporter novel type-1; PD, Parkinson disease; ROS, reactive oxygen species; SNPs, single nucleotide polymorphisms; SOD, superoxide dismutase; TNFα, tumor necrosis factor alpha; VCAM, vascular cell adhesion molecule
1. Introduction
The unique sulphur-based chemistry of thiols plays an important role in cellular function and protection of cellular components (reviewed in Ref. [1]). The body has evolved to rely on highly abundant low molecular weight thiols such as glutathione (GSH), to maintain redox homeostasis but also play other important roles including xenobiotic detoxification and signalling [1,2]. Some of these thiols may also be derived from diet, such as the trimethyl-betaine derivative of histidine, ergothioneine (ET). Discovered more than a century ago, there has been a resurgence in interest in this low molecular weight thiol/thione, owing in part to its unique chemistry (e.g. its unusual stability, reviewed in Refs. [3,4]) and also the identification of a transporter, the organic cation transporter novel-type 1 (OCTN1), responsible for ET uptake and accumulation in human tissues from our diet and also the selective distribution to tissues owing to differential transporter expression [[5], [6], [7]]. Consistent with the recent attention on ET is the exponential growth in publications mentioning this compound (Fig. 1). These include a wide range of studies uncovering association of blood levels of ET with health and disease, mechanisms of cellular and tissue protection, potential therapeutic applications of ET, transporter expression and tissue distribution, biosynthetic mechanisms, and many others. This review highlights new insights and recent findings on this extraordinary compound.
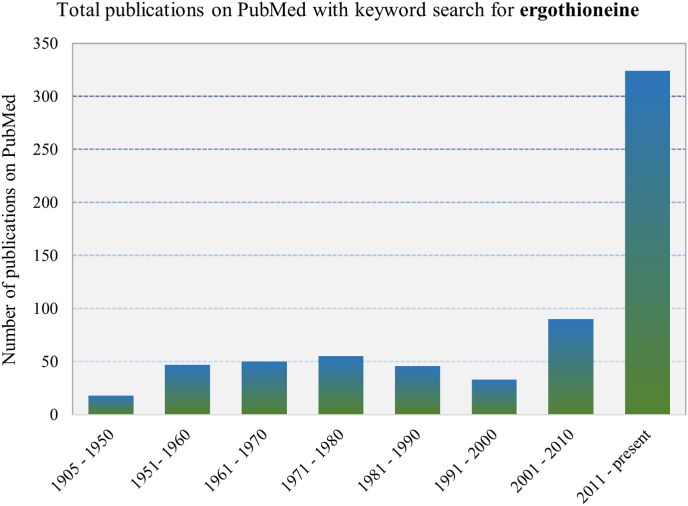
Fig. 1.
Since its first isolation in 1905, there was initially significant interest in ET, however the interest waned until the discovery of its transporter (ETT/OCTN1) in 2005 and has since been the focus of intense research. The graph shows the number of publications on PubMed (https://pubmed.ncbi.nlm.nih.gov/) by time period for the keyword ‘ergothioneine’.
2. The ergothioneine transporter
2.1. The organic cation transporter novel-type 1, an ergothioneine transporter?
It was discovered in 2005 that OCTN1 (encoded by the gene slc22a4) is primarily a transporter for ET, with higher transport efficiency than many other related metabolites [5], and without it (as seen in knockout animal models) there is an absence of ET in cells and tissues [8,9]. From the initial notion that OCTN1 primarily serves as an ET transporter, numerous other studies surfaced declaring that OCTN1 was involved in the transport of compounds such as nucleosides [10], acetylcholine [11], tetraethylammonium, spermine, l-carnitine, sulpiride, homostachydrine [12], cytarabine, gemcitabine, gabapentin, oxaliplatin, and metformin. However, many of these claims used unconventional models that were far from physiological. Recently the selective preference of ET as a substrate for OCTN1 was reaffirmed using both natively expressed [7] and over-expressed [6] OCTN1 in human cells, endorsing a suggestion to rename OCTN1 as the ergothioneine transporter (ETT) [13]. However, as most groups still use the term OCTN1, we will follow that for this review.
The transporter is responsible for the avid uptake of ET from diet, with a wide distribution in many tissues (Fig. 2) [5,14]. ET is found in most, if not all, tissues of laboratory rats and mice despite there only being trace levels of ET in their diet [4,15], indicating efficient uptake and retention of this nutrient. OCTN1 expression is also seen on the rat proximal renal tubules [16], presumably important for renal reabsorption of ET, which may facilitate ET accumulation in the body. Indeed, OCTN1 knockout mice administered with ET intravenously revealed higher urinary excretion rates (>50% of administered dose) compared with wildtype animals (<10%) [17]. This impressive ability for ET to amass in the body and the selective distribution in tissues (through differential expression of the transporter), suggests that ET plays important physiological roles. However, OCTN1 knockout models (lacking tissue ET), have not yet revealed any substantial overt phenotypes or deficiency syndromes [8,9,18]. This could be due to compensatory defence pathways to maintain homeostasis in the absence of ET in healthy animals, although when these knockout models are stressed the phenotype is more pronounced [8,9]. Consequences of deficiency are hard to demonstrate in humans due to the abundant presence of ET in diet and its avid retention by the body. However, lower blood levels of ET have been associated with incidence of several disorders including Parkinson disease (PD) [19], mild cognitive impairment (MCI) [20], Crohn's disease (CD) [21] and frailty [22], while correspondingly higher levels of blood ET are correlated with lower risk of cardiometabolic disorders and associated mortality [23] and lower prevalence of peripheral neuropathy [24]. This is consistent with the notion that a deficiency in ET may elevate the risk of disease. Whether this decline in ET is a cause or consequence of disease remains to be established by in-depth, longitudinal studies.
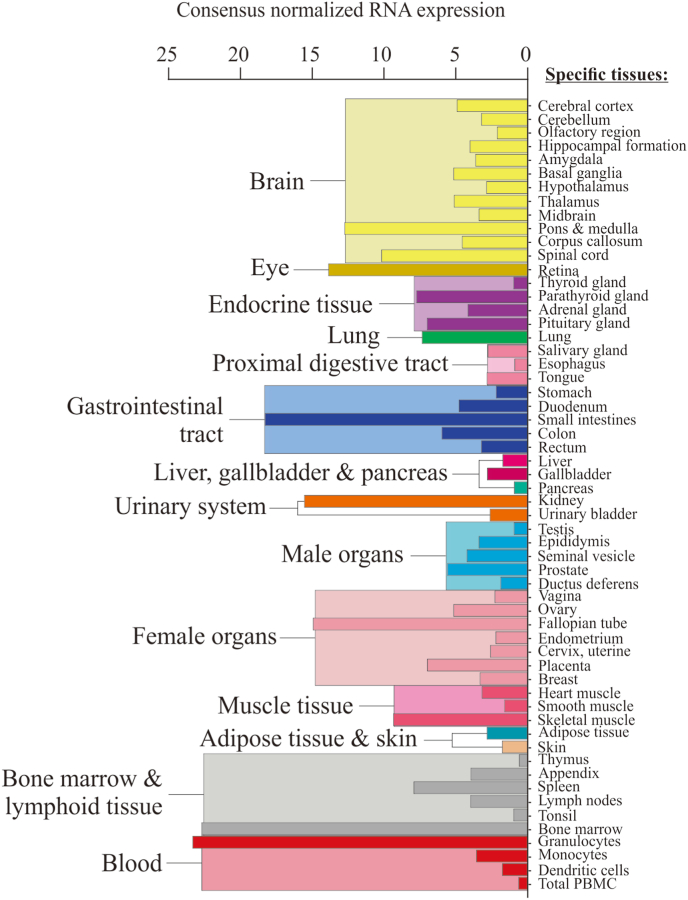
Fig. 2.
RNA expression of slc22a4 (encoding OCTN1) in the human body using normalized consensus RNA expression data from 3 data sets (genotype-tissue expression project, human protein atlas dataset, and the FANTOM5 project). Graph and data adapted from the Human Protein Atlas (https://www.proteinatlas.org/) [177].
2.2. Factors affecting ergothioneine transport
ET can be found in most (if not all) tissues, with differential rates of accumulation [4,15], owing to differing expression of the transporter [5]. High expression of the transporter, and hence high levels of ET, is observed in certain cells (e.g. blood cells, bone marrow, ocular tissues, brain etc.; Fig. 2) that are likely predisposed to oxidative stress, although other tissues can accumulate high levels of ET with sustained administration [4,15]. In certain animal disease models e.g. fatty liver disease [25], CD [14] and chronic kidney disease [26], an elevation in the OCTN1 transporter (via transcriptional activators including RUNXI, Sp1, and NFκB [27]) was seen in response to tissue injury. This has been suggested to be an adaptive physiological response to elevate ET in the damaged tissue and thereby limit further injury [28].
Although ET is obtained from diet [4,29,30], studies suggest that tissue levels of ET are closely associated with OCTN1 expression or polymorphisms of the transporter [14,31]; populations possess a number of single-nucleotide polymorphic variants (SNPs) of OCTN1 which affect ET transport kinetics [31]. Supporting this notion, subjects with higher basal blood levels of ET also had a greater uptake and accumulation of ET following oral administration, possibly due to polymorphisms of the transporter [32]. However, it is not yet known whether polymorphisms of OCTN1 (possibly accounting for lower blood levels of ET) may predispose certain individuals to disease. Although earlier studies associated the L503F variant of OCTN1, and the elevated ET in these individuals [33], with an increased risk of inflammatory bowel diseases (IBD), CD and ulcerative colitis [34,35], this was found to be a case of genetic hitch-hiking due to the association of this OCTN1 variant with the IBD5 haplotype, a known risk factor for CD [36]. The authors proposed that this variant, which results in up to 50% greater transport efficiency and 3-fold greater affinity for ET [33], was an adaptive mutation resulting from lower ET in the diets of these historic populations or positive selection due to high UV exposure (see section 3.2.6) of ancient farmers from this region [36]. The claimed associations between OCTN1 polymorphisms and IBD or rheumatoid arthritis remain controversial [36,37]. Longitudinal studies into SNPs of OCTN1 in populations, their blood levels of ET, and association with disease, should give further insights.
2.3. Cellular location
Early studies [[38], [39], [40]] suggested (but did not rigorously establish) that OCTN1 is found in the mitochondria, allowing ET accumulation in this organelle [41]. Indeed, ET could protect mitochondrial DNA from oxidative damage due to hydrogen peroxide [39] or UV exposure [42]. Since then, few studies have investigated the intracellular localization of OCTN1 and ET. Mitochondrial dysfunction is implicated in a wide range of disorders and the therapeutic benefits of ET have been suggested to be mediated through protection of this organelle [43]. We recently demonstrated that ET protects mitochondrial morphology in transgenic Drosophila models of PD (unpublished data). Definitive evidence of ET uptake and accumulation in the mitochondria is required.
2.4. An alternative transporter?
A recent study of an orphaned solute carrier (the superfamily to which OCTN1 belongs) SLC22A15, demonstrated its capability to transport ET into transfected HEK293 cells, although with much lower transport efficiency than OCTN1 [44]. The authors suggest that SLC22A15, which has a higher expression than OCTN1 in the brain, may be important for deposition of ET in the brain, or perhaps translocation across the BBB [44] where the level of OCTN1 expression remains uncertain [45,46]. However, SLC22A15 does not appear to be involved in uptake into the body from the diet since OCTN1 knockout mice do not have appreciable levels of ET in blood and tissues following oral administration (ref. [8] and our unpublished data). Indeed, SLC22A15 expression is low in the gastrointestinal tract [44]. However, when ET was intravenously administered levels appear to be comparable, albeit low, in the brain between OCTN1 knockout and wildtype mice [17]. Further studies are needed to establish if SLC22A15 is indeed involved in brain uptake and accumulation of ET.
3. A gerontological perspective
3.1. Infants and children - possible role of ergothioneine in human development?
Almost nothing is known about the role of ET during development in human infants, although there is evidence of its presence in blood [47], urine [48], and brain [49] of new-borns and infants. Studies have identified the expression of OCTN1 on the rat placenta [50], which may facilitate transport of ET from the mother to offspring [[4], [47]]. Our preliminary studies (unpublished data) indeed indicate a high correlation of blood ET levels between human mothers and children, suggesting that it is transported from mother to child and thus may play a role in infant development. OCTN1 is also expressed in human mammary glands [51] and a 4-fold upregulation of OCTN1 was observed in lactating human mammary epithelial cells (HMEC) compared with non-lactating HMEC [52]. Indeed, ET is present in human breast milk [47] and is also present in cow [53] and goat milk [47]. Nakamichi et al. [54] and Ishimoto et al. [55] demonstrated that ET promotes neurogenesis in mice, which may enhance memory [56], or alleviate symptoms of neuropsychiatric disorders [54]. It may therefore be plausible that ET plays a role in neuronal differentiation during growth and maturation of the brain in the developing child.
The administration of high levels of ET to pregnant female rats did not reveal any overt toxicity in either mother or offspring, however no observations were reported about possible beneficial outcomes for the offspring [57]. In part due to the absence of overt effects and the possible nutritional benefits of ET, the European Food Safety Authority (EFSA) granted approval for ET to be used as a supplement in pregnant and breastfeeding women, children and infants [58]. Further research is needed to determine if ET is beneficial or essential for human development, and if so raises the question whether supplementation in infants may be advantageous (e.g. in infant formulas, although many of the formulas are based on cow's milk and already contain some ET [47,53]). A study of healthy males in Saudi Arabia found that blood levels of ET increase from infanthood to teenage years, peaking at early adulthood, again suggesting the possible importance of ET for human development [59].
3.2. Adulthood - role of ergothioneine in disease prevention
3.2.1. Role of ergothioneine in overall adult health
We previously demonstrated the uptake and retention of ET following oral administration to young (21–29 years of age) healthy adult males [32]. The administration of ET to these young healthy individuals, did not significantly decrease oxidative damage biomarkers, although the basal oxidative damage levels were already low. The absence of a significant change may indicate that ET is not a primary defence against oxidative stress, but rather acts as a secondary antioxidant buffer whenever the redox homeostasis is perturbed and/or when primary antioxidants are depleted through chronic exposure to oxidative stress. Indeed, the unique chemistry of ET, favouring the thione tautomer at physiological pH, gives rise to its stability and resistance to autooxidation (relative to other low-molecular weight thiols such as GSH), slow turnover, and ability to accumulate in the tissues. Despite the lack of significance, ET administration still revealed a trend toward lower levels of some oxidative damage biomarkers, relative to placebo controls [32]. Hence, although ET has potent antioxidant properties in vitro [3,60], protection against oxidative stress may not be its only, or even the primary physiological role (as suggested from an evolutionary perspective [61,62]).
ET promotes neuronal stem cell differentiation [55] which could be important in development and maintenance of the central nervous system. Knockdown of OCTN1 in K562 (erythroid progenitor cells) decreased cell proliferation and differentiation, supporting the view that ET is important for erythropoiesis [4,63]. One of the highest levels of ET is found in the bone marrow [64] (Fig. 2) with a correspondingly high expression of OCTN1 [5], which may act as a reservoir for ET, which could be incorporated into erythrocytes during erythropoiesis [4]. Indeed, erythrocytes possess high levels of ET (more than 50 times higher than plasma) and in our human studies whole blood levels continued to increase for up to 4 weeks beyond the point where ET administration had ceased [32], consistent with this hypothesis. The expression of OCTN1 is also seen in monocytes and macrophages [5,14] and a recent study suggested that ET plays a role in immunoregulation through toll-like receptor signalling in macrophages [65].
3.2.2. Nutrient for reproductive health?
Expression of OCTN1 has been demonstrated in testes and seminal vesicles [8] with high levels of ET reported in seminal plasma of animals and humans [[66], [67], [68]]. Although the level of ET in seminal plasma is highly variable, the levels are typically much greater than that of GSH and cysteine [66,68]. ROS play a role in sperm function and fertilization, hence a delicate balance of ROS for physiological function versus prevention of oxidative damage is needed [69,70]. ET may be an ideal agent to maintain redox homeostasis since it does not interfere with the critical roles of ROS but may become active during times of excessive oxidative stress [[28], [47]]. The anti-cancer drug cisplatin causes testicular damage and dysfunction of the Leydig cells leading to infertility [71]. Administration of cisplatin to Wistar rats decreased sperm count and motility and increased the incidence of abnormal sperm morphologies, decreased testosterone levels and led to necrosis of testicular interstitial cells [72]. Oral supplementation with ET for 14 days before cisplatin administration abolished these detrimental effects [72]. Interestingly, oral administration of ET alone (without cisplatin) was found to increase sperm counts and motility, decrease sperm abnormalities, and significantly decrease basal levels of malondialdehyde compared with control rats [72]. Hence ET may play a role in preserving reproductive health. In recent times average sperm counts have dropped significantly, with one study in European men indicating average sperm numbers falling from 100 million/ml in 1940s to around 40 million/ml at present, increasing the risk of infertility [73]. Perhaps ET supplementation could help, and warrants this investigation.
3.2.3. Prevention of tissue injury and fibrosis
Exposure of tissues to chronic inflammation and persistent oxidative stress due to environmental (chemical or radiation) insults, allergies, persistent infections, autoimmune reactions, and so on, can lead to damage and remodelling of tissue, eventually triggering fibrosis, which may ultimately lead to organ failure [74]. Multiple studies in cell cultures and animal models have demonstrated that ET not only modulates levels of inflammatory cytokines and mitigates oxidative or nitrosative damage [[75], [76], [77]], but may also slow or prevent fibrosis [26,78,79].
For example, the hepatotoxin dimethylnitrosamine (DMN), elevated oxidative damage and smooth muscle α-actin (a marker for fibrosis) in human hepatic stellate cells, but pre-treatment of the cells with ET decreased DMN-induced elevations in 4-hydroxynonenal and fibrosis [78]. Moreover, OCTN1 expression in hepatocytes was elevated in wild-type mice following DMN treatment [78], which is consistent with an earlier study demonstrating an increase of liver OCTN1 expression and tissue ET in a guinea pig model of fatty liver disease [25]. Another study claimed that ET protected rat kidney and liver against oxidative damage caused by ferric-nitrilotriacetate [75]. Conversely, OCTN1-knockout in mice exacerbated indoxyl sulfate-induced kidney [26] and DMN-induced liver injury [78] and aggravated oxidative damage, inflammation and fibrosis in both models. The ROS-generating NADPH oxidase, NOX4, which is highly expressed in hepatocytes and may play a role in liver fibrosis, was significantly elevated in OCTN1 knockout animals, while ET supplementation of human hepatic stellate cells decreased expression of NOX4 [78].
Multiple studies also demonstrate lung protection by ET. In a mouse model of acute respiratory distress syndrome using cytokine insufflated (IL-1 and IFNγ) rats, ET protected against acute lung injury [80]. Likewise, in a model of idiopathic pulmonary fibrosis, using cigarette smoke extract to promote oxidative stress and cell migration in human alveolar epithelial cells, ET treatment prevented not only cell death [81,82], but also cell migration (fibrosis) [79]. When mice were exposed to cigarette smoke over 6 months, elevated numbers of inflammatory cells in the broncho-alveolar lavage fluid, alveolar damage and increased inflammatory cytokines were observed; these effects were more severe in OCTN1-knockout mice [82]. Collectively, these studies indicate that the distribution and accumulation of ET in organs may serve as a protectant against injury and permanent scarring. Whether these benefits of ET involve its antioxidant, or other, properties, remains to be seen.
3.2.4. Pre-eclampsia
Pre-eclampsia, a serious complication of pregnancy, is characterized by hypertension, oxidative stress, placental ischaemia, and proteinuria [83,84]. In a rodent model of pre-eclampsia, ET supplementation decreased hypertension and circulating sFlt-1(a marker of pre-eclampsia), and increased expression of placental Nrf2, superoxide dismutase 1 (SOD-1), mitochondrial enzymes (PGC-1α, UCP-1 and SOD2) and decreased mitochondrial ROS in maternal rats, leading to increased foetal weight [43,85]. Furthermore, prior studies showed that ET could also be beneficial in protecting against ischaemia-reperfusion injury [77,86,87]. These studies highlight the potential therapeutic application of ET for pre-eclampsia [84]; and EFSA has granted approval for supplementation in pregnant women [58].
3.2.5. Concomitant application in cancer therapy
While there are few studies assessing association of ET blood levels with cancers [4], some studies have explored the benefits of ET in reducing the detrimental side effects, or enhancing the effectiveness of, chemotherapies. Chronic peripheral neuropathy is a common side effect in patients undergoing chemotherapy for colorectal cancer, however Winkels et al. [24] suggest that high blood levels of ET were associated with less neuropathy. In Sprague-Dawley rats, oxaliplatin (chemotherapy for metastatic or advanced colorectal cancer) induced peripheral neuropathy, but co-administration with ET (15 mg/kg) ameliorated neuropathy, apparently by decreasing oxaliplatin accumulation in dorsal root ganglion neurons [24].
Anthracyclines such as doxorubicin, are effective and widely used chemotherapeutics for breast cancers, lymphomas, lung cancers and many others, but their clinical application is limited by the occurrence of cardiotoxicity with high doses or prolonged use [88]. Our preliminary studies in mice suggest that ET accumulates in the heart and offers protection against anthracycline-induced cardiotoxicity (manuscript in preparation).
Another study demonstrated that ET may augment the response of tumour-associated macrophages and thereby prevent an immunosuppressive microenvironment caused by cytotoxic T lymphocytes and thus enhance the efficacy of cancer vaccine immunotherapy [65,89].
3.2.6. Protection of the skin
Several studies demonstrated that ET protects skin cells from UV-irradiation [42,[90], [91], [92]]. Although this may be partially attributed to the UV-absorbing properties of ET, it was also shown to protect by raising endogenous antioxidant levels via Nrf2/ARE in human keratinocytes [91]. Silencing Nrf2 or OCTN1 expression in the keratinocytes inhibited these protective effects [91]. Another study demonstrated that ET protected against UV-induced mitochondrial DNA damage [42]. Not surprisingly, ET has found its way into various cosmetics and topical skin products.
3.3. Elderly; ET as a longevity nutrient
3.3.1. Blood levels of ET with age
A study in Saudi males found that blood ET levels increase with age until early adulthood but then gradually decline with advancing age [59]. A similar trend was seen in rats where blood ET levels increase up to 11 weeks of age then decline [41]. Plasma ET levels continue to decline in an elderly Asian population (beyond 60 years of age) [20], and an Australian epidemiological study revealed that serum ET levels were inversely correlated with age (55–85 years) [93]. This decline in blood ET with age could be due to numerous factors such as increased turnover, dietary changes or altered transport function (decreased OCTN1 expression or impaired transporter functionality). We demonstrated in a Singaporean elderly population [20] that blood ET levels were correlated to self-reported mushroom consumption, but there was no significant trend between age and mushroom consumption, indicating that the decrease in ET may not be due to altered diet in this case.
Blood (and likely tissue) levels of ET are also significantly decreased in individuals with a variety of age-related disorders [19,20,23]. This suggests that lower blood levels of ET are a risk factor for age-related disorders and supplementation could be beneficial. Indeed, Ames [94] suggested that ET should be classified as a “longevity vitamin”, i.e. it may play a role in lowering the risk, or progression, of age-related disorders, and promote healthy longevity. Consistent with this, a study investigating 131 blood metabolites and their association with frailty, identified that a declining blood ET level correlates with markers associated with frailty [22].
3.3.2. Ocular health
The cornea of the eye is constantly exposed to atmospheric levels of oxygen [1], and coupled with the fact that retina is one of the highest oxygen-consuming tissues in the human body [95], results in an organ that is exposed to high levels of oxidative stress [1] and may require high levels of antioxidant defences to maintain redox homeostasis. Earlier studies identified that high levels of ET are present in ocular tissues including the lens, retina, cornea and retinal pigmented epithelium [96,97]. Corresponding to this some of the highest levels of OCTN1 mRNA expression can be found in the eye in pigs [68], zebrafish [9] and humans (Fig. 2). In humans, ET is present in tears and aqueous humour [47]. Hence ET may help protect the eye against oxidative damage, whereas lower ET levels might predispose to ocular disorders. Indeed, ET levels in human eye lens decreased significantly with increasing severity of cataract formation [98,99]. Another ocular disease, age-related macular degeneration (AMD), the leading cause of irreversible blindness, has pathogenic mechanisms involving oxidative damage and immune dysregulation (reviewed in [1]), implying that ET could be protective for this condition. Indeed, ET protected corneal endothelial cells against hydrogen peroxide-mediated oxidative stress and the unfolded protein response [100]. However, few studies have investigated any protective role of ET in AMD and other ocular diseases.
3.3.3. Neurological disorders
The ability of ET to accumulate in the brain (although more work needs to be done to establish the distribution and cellular location of ET in the human brain, peripheral nervous system and cerebrospinal fluid) and its neuroprotective capabilities in a range of models both in vitro and in vivo, have been previously reviewed (see Refs. [3,47,101]), hence we will focus on more recent advances. Evidence suggests that ET may protect the brain against oxidative damage [102,103] and neuroinflammation [104,105], target underlying pathologies in neurodegeneration such as mitochondrial dysfunction [39,43] and toxic amyloid accumulation [106,107], and even promote neurogenesis [55,108,109]. Silencing OCTN1 prevented ET uptake in Neuro2a (mouse neuroblastoma) cells, leading to a decrease in cell proliferation and differentiation while concurrently downregulating the neurite marker GAP43 and increasing oxidative stress markers HO-1 and SOX2 [108]. Subsequently it was demonstrated that ET induced differentiation in neural progenitor cells as evidenced by a decrease in neurospheres, the upregulation of neuronal marker βIII- tubulin, and differentiation activator gene Math1, while silencing OCTN1 abolished these effects [109]. This differentiation was said to be mediated through activation of S6K1 and neurotrophin 4/5- TrkB signalling [55] and studies have shown that S6K1 overexpression promotes dopaminergic neuronal differentiation of human neural stem cells [110]. Mice administered ET for 14 days demonstrated a dose-dependent increase of ET in the hippocampus, with an enhancement of memory and learning ability, and an increase in neuronal spines in the dentate gyrus at doses greater than 20 mg/kg [56]. In cultured primary hippocampal neurons, administration of ET, elevated the expression of βIII-tubulin, synapsin I, and neurotrophins 3 and 5, suggesting that ET promotes hippocampal neuron maturation by increasing neurite and synapse formation [56]. This may also have implications for the use of ET in counteracting neurodegenerative disorders. Indeed, the ability of ET to promote neuronal differentiation may be a mechanism behind enhanced object recognition memory and prevention of neuropsychiatric disorders such as depression, as evidenced from studies in mouse models [54,56].
Chronic sleep loss and depressive symptoms are both risk factors for neurodegenerative disorders [[111], [112], [113]]. Moreover, the treatment of depressive symptoms may impede the pathological progression of AD [114]. In a mouse model of depression, oral administration of ET exhibited antidepressant-like effects, improving spleen and body weights [54]. Similarly, in a rat model of major depressive disorder, leading to sleep abnormalities and depressive social avoidance behaviours, the oral administration of ET for 7 days prior to social defeat stress ameliorated these effects [115]. The authors attributed the beneficial effects of ET on sleep and depression to a possible reduction in inflammation around the brainstem and peripheral nervous system together with management of intracerebral oxidative stress [115], and the possible involvement of ET in neurogenesis.
Another neurological condition, epilepsy, characterized by abnormal electrical-mediated activity in the brain leading to recurrent seizures, was found to be exacerbated by OCTN1 uptake of homostachydrine in the brain of mouse models [12]. However, ET administration to these mice decreased homostachydrine uptake in the brain (presumably through competing transport of ET) leading to decreased seizures and prolonged survival of the animals [12]. Glutamate receptor-mediated hyperexcitation plays a key role in seizures, but a study demonstrated that ET protected rat retinal neurons against N-methyl-d-aspartate excitotoxicity [116].
Glucose dysregulation, in part due to oxidative damage to key enzymes and dysfunction of the mitochondria, is a major component of AD [117,118]. ET restored cell viability following hyperglycaemia-induced cytotoxicity in rat pheochromocytoma cells by decreasing formation of ROS, protein carbonyls, and advanced glycation end products, and inhibiting NF-κB activation [104]. A similar rescue was observed from ET administration to human brain endothelial cells under hyperglycaemic conditions [119]. Moreover, d-galactose administration in mice triggered learning and memory deficits, but ET administration to animals not only significantly improved performance in both tasks but also decreased β-amyloid accumulation in the hippocampus and lipid peroxidation in the brain [103].
Declines in blood ET are not only seen in PD and MCI subjects but also with vascular dementia (manuscript in preparation). Indeed, the vascular endothelium plays a critical role in brain function by maintaining blood-brain barrier permeability and cerebral blood flow, and dysfunction of this system aggravates many neurodegenerative disorders [120,121]. ET administration to human brain microvascular endothelial cells prevented pyrogallol, xanthine/xanthine oxidase or high glucose-induced cell death by protecting against oxidative stress and enhancing the expression of antioxidant enzymes, while decreasing NADPH oxidase 1 (NOX1) expression [119,122]. In a diabetic rat model, these same stressors impaired acetylcholine-induced relaxation in brain basilar arteries, however ET dose-dependently decreased this impairment in relaxation [119]. The cholesterol oxidation product, 7-ketocholesterol (7KC), is elevated in the cortical tissue of AD patients [123] and is implicated in other neurodegenerative disorders [124,125]. In human brain endothelial cells 7KC induced the expression of proinflammatory genes and cyclooxygenase-2, but treatment with ET reduced the 7KC-induced expression of IL-1β, IL-6, IL-8, TNFα and COX-2 [105].
The evidence suggesting that ET is beneficial against neurodegeneration comes from cell culture and animal models. Many antioxidants and other therapeutic compounds have shown promise in animal models but failed to translate the benefits to human neurological disorders such as AD (reviewed in Ref. [1]). While ET overcomes the major hurdle of translocation across the BBB and accumulation in the brain, randomized placebo-controlled clinical studies are needed to establish any human therapeutic benefits.
3.3.4. Cardiovascular disorders
Evidence that ET may influence cardiovascular health comes from a longitudinal study associating changes in blood metabolites with cardiometabolic diseases in 3236 subjects over 20 years [23]. Out of 112 metabolites, higher blood ET had the most significant association with lower risk of cardiometabolic disease and mortality, independent of age, gender, fasting glucose, cholesterol and triglyceride levels, alcohol intake, and smoking status [23].
Dysfunction of the vascular endothelium is central to a wide range of cardiovascular disorders, including hypertension, atherosclerosis, chronic heart failure, coronary artery disease, and diabetes [126]. A possible major underlying mechanism for the benefits of ET in the cardiovascular system is protection of the vascular endothelium, as previously mentioned for brain endothelium [105,119]. Other studies revealed that ET protects endothelium from oxidative damage and cell death due to paraquat, hydrogen peroxide, high glucose [127] or oxidized-LDL [128] exposure. Moreover, ET significantly inhibited proinflammatory cytokine or oxidized LDL-induction of inflammatory cell adhesion molecules, VCAM-1, ICAM-1 and E-selectin [129].
4. Sources of ergothioneine
4.1. A culinary perspective
ET is present in a wide array of foods, but the highest dietary levels of ET are found in certain mushrooms [29,30,47,130] and Spirulina [131], which produce their own ET [132]. Several studies have investigated the putative health benefits of mushroom consumption. These include the association of increased mushroom consumption with decreased risks of dementia [133,134], cardiovascular disease, certain cancers (e.g. prostate and breast cancer) [135,136], IBD [137], metabolic syndrome [138], PD and other neurological disorders [139], viral infections [140] and many other conditions. Some of the studies attributed these perceived health benefits of mushrooms to the ET present [129,[141], [142], [143]]. However, mushrooms contain many other compounds (e.g. β-glucans, polysaccharides, etc.) which could also be beneficial [30].
4.2. Biosynthesis of ET by gut microflora?
The gut microbiota play a pivotal role in human health and disease. The genes encoded by this vast pool of microorganisms outnumber those of the host by more than 100-fold and are responsible for some essential metabolic functions including the production of certain vitamins and amino acids [144,145]. Although animals and humans are only known to obtain ET from dietary sources [4], certain bacteria can synthesise ET, including Mycobacterium spp. [[146], [147], [148]] and Burkholderia spp. [149] belonging to the phyla Actinobacteria and Proteobacteria, respectively. They produce ET from histidine, albeit through distinct mechanisms [150]. Transforming Escherichia coli (not naturally capable of ET biosynthesis) with egtBD genes, enables them to produce ET [149], although addition of egtACE genes enhanced ET production in E. coli [151,152]. Deeper in silico analysis of bacteria from the phyla Bacteroidetes, Actinobacteria, Cyanobacteria and Proteobacteria, present in normal gut microflora, reveals that many of these bacteria appear to possess the egtBD genes, or homologs of them. This therefore raises the question as to whether gut microflora can produce ET. Although evidence suggests that this does not occur (reviewed in Ref. [4]), this probably needs re-examination using more sensitive and accurate methods for ET analysis. If so, could ET production by the microbiome contribute to tissue levels of ET, and would changes in gut microflora, as seen in a wide range of diseases, influence the levels of ET in the body? This may also present another explanation for lower blood levels of ET seen in many disorders. Indeed, with the increasing evidence of an intricate link between the gut microbiota and the brain [153], an altered microbiome leading to reduced ET levels in the body could contribute to disease.
No studies to date have definitively established that gut microbiota can produce ET [4], but one study indicated that Lactobacillus reuteri may produce ET in vitro and that an elevation of L. reuteri correlated with an increase in faecal ET levels in rats [115]. However, the presence of ET in a microorganism is not definitive evidence of ET production since some bacteria can take up ET from their environment [4,149]. Studies with isotopically labelled precursors or isolation of ET biosynthetic enzymes are needed to confirm ET production. The authors did identify that elevated faecal ET correlated with an increase in L. reuteri in a rat stress model, and administration of ET in this model improved sleep abnormalities and social avoidance behaviours [115]. However, studies by our laboratory using isotopically labelled histidine as a precursor did not reveal any indication of ET biosynthesis by L. reuteri (our unpublished data), and the Lactobacilli spp. genome does not appear to contain ET biosynthetic genes. Perhaps the authors identified that Lactobacilli can accumulate ET from their environment. Nevertheless, the uptake and utilisation of ET by gut microbiota may still play a crucial role in influencing ET uptake by the host. More work is needed to study this.
Indeed, cell-free extracts of E. coli can break down ET to yield thiourocanic acid and trimethylamine, attributed to the trimethylamine-lyase, ergothionase [154]. Some other microorganisms including Burkholderia sp. [155], Treponema denticola [156], and Alcaligenes faecalis [157,158], also possess ergothionase. Further studies are needed to evaluate if other commensal bacteria can degrade ET and if this contributes to decreased absorption of ET from the gut.
4.3. Biosynthetic mechanisms
EFSA has permitted the use of ET as a food additive and supplement [159] even in pregnant women and infants [58]. Meanwhile, the US Food and Drug Administration has granted ET the status of “Generally Recognized as Safe” (GRAS) to be used as a food additive and supplement. This facilitates the broader application of ET for use in general health and wellbeing or application against a range of diseases.
Besides chemical synthesis (Tetrahedron), several groups have demonstrated the bio-production of ET in the yeast, Saccharomyces cerevisiae (not naturally capable of ET biosynthesis), through cloning of two ET biosynthetic genes from the mushroom Grifola frondosa [160] or a combination of bacterial and fungal genes from Mycobacterium smegmatis and Claviceps purpurea (the ergot fungus, where ET was first identified) [161]. Other groups have enhanced biosynthesis by recombinantly enhancing copy numbers of the ET biosynthesis genes and enhancing the histidine precursor availability (by removing histidine ammonia-lyase) in Methylobacterium [162]. Other methods have also been utilized to harness synthetic biology to produce ET and scale up its production using a range of bacterial and fungal microorganisms [151,152,161,163] and even to generate isotopically labelled variants of ET for research [164].
5. Role of ergothioneine in infectious diseases
5.1. Coronavirus infectious disease 2019 (COVID-19)
The highly contagious severe acute respiratory syndrome coronavirus 2 responsible for the devastating COVID-19 pandemic, has infected more than 100 million individuals and led to almost 2.2 million deaths across the globe at the time of writing (WHO COVID-19 situation reports). The battle to curb coronavirus infections has given rise to both repurposed and novel therapeutics to reduce the severity and duration of illness. We suggested that ET might alleviate symptoms and improve prognostic outcomes of COVID-19 patients [165], since it can protect against the underlying pathologies implicated in COVID-19, e.g. acute respiratory distress syndrome (the main cause of mortality in COVID-19) [80], excessive oxidative damage [60,166,167], inflammation [76,77,168], and endothelial dysfunction [119,127]. The presence of ET in major organs could also protect them from injury due to a hyperactive immune response and prevent permanent damage (e.g. fibrosis) [25,26,54,72,78,79,169].
5.2. Opportunistic pathogens
A darker side of ET is that certain pathogens can synthesise ET including Burkholderia and Mycobacteria tuberculosis, as a means of protecting against host defences, or enhancing antibiotic resistance [149,170]. Indeed, in M. tuberculosis, which causes tuberculosis, ET maintains bioenergetic homeostasis and protects against oxidative stress, enhancing virulence in animal models [170]. The opportunistic fungal pathogen A. fumigatus is also capable of ET biosynthesis, which may enhance its resistance to environmental heavy metals and host-generated ROS [171].
As mentioned earlier, while most microorganisms are incapable of ET biosynthesis, some can accumulate and utilize ET from their surrounding environment [4], especially some opportunistic pathogens e.g. E. coli [154] and the oral pathogen Treponema denticola [156]. Drugs targeting the uptake mechanisms may thus be useful in limiting their pathogenicity. A clearer understanding of the role of ET in pathogenic microorganisms could reveal mechanisms to reduce virulence and/or drug resistance, for example by targeting ET biosynthesis.
6. Alternative applications of ergothioneine
With the distinctive chemistry of ET, it is not surprising that there has been a myriad of alternative applications for it. S-methyl ergothioneine and hercynine have been identified as likely metabolites of ET in the body. Indeed their levels are highly correlated with ET in blood [32] and most tissues (although there are some exceptions e.g. the brain which typically has higher levels of hercynine) [15]. However, their function remains unknown. Glutathione-S-transferases (GST) catalyse the detoxification of various xenobiotics utilizing GSH and may facilitate the methylation of GSH. Whether another such enzyme exists for ET, or if GST can also utilize ET, is unknown. One study described ET as a xenobiotic amino acid, facilitating nucleophilic substitution-type reactions to counteract toxins in vitro in a similar manner to GSH, as demonstrated by reaction with 1-chloro-2,4-dinitrobenzene (CDNB) to form an ET-CDNB product in human lung epithelial cells, improving cell viability [172]. Although the enzymes involved remain unknown, the ET-CDNB product is suggested to efflux from the cells using multi-drug resistance transporters [172]. Further studies are needed to validate this and elucidate the enzymes involved.
ET has also been used as a food preservative [30]. Extracts of edible Enoki mushroom were shown to prevent melanosis or blackspot formation in harvested shrimps, which were attributed to the anti-melanosic and antioxidant properties of ET [173]. Likewise, studies demonstrated that ET prevented oxidation of lipids and myoglobin in beef and fish meat, preventing discoloration for more than a week on ice [174]. As such, patents (US20130035361A1 and CA2680223A1) were filed in the US and Canada for the use of ET as a preservative in foods, medicines and beverages.
Beyond this, the unique chemistry of ET may enable robust photostabilization of various fluorophores (personal communication), and recent studies have also shown that ET has an intense surface-enhanced Raman scattering spectrum, which the authors suggest may enable non-invasive analysis of ET in biofluids as a prognostic tool [175]. Interestingly, ET has also been suggested as a metabolite to estimate the age of bloodstains at crime scenes, due to its stability and slow time-dependent degradation [176].
7. Final thoughts
The unique chemistry of ET gives rise to its high stability and ability to accumulate in the body, enabling a wide range of cytoprotective and disease mitigating properties. While associations have been made between low blood levels of ET and various human disorders, there is much still unknown. Nevertheless, the uptake in the foetus and infant, the near, if not fully, ubiquitous presence in tissues and the avid retention by the body are strong evidence that ET plays a critical role in human development and health. Numerous animal studies have also demonstrated its benefits, whereas preventing uptake through silencing OCTN1 appears to worsen pathology in these disease models. It remains to be seen whether supplementation in humans, especially the elderly or the subset of individuals with lower blood levels of ET (due to polymorphic variants of the transporter or diet), could reduce the risk of age-related and other diseases, or act as a therapeutic by slowing or halting disease progression. The safety profile of ET and regulatory approvals facilitate such studies. With the rapid surge in interest (Fig. 1), it is undoubtedly exciting times for this unique low molecular weight compound.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
The authors thank Tetrahedron (14 avenue de l’Opera, Paris, France) for the provision of l-ergothioneine used in our studies. The authors also thank the Tan Chin Tuan Centennial Foundation, the National Medical Research Council (Individual Research Grant NMRC/1264/2010/082/12) and the Ministry of Health Singapore, Healthy Longevity Catalyst Award (HLCA20Jan-0057) for financial support.
References
- 1.Halliwell B., Gutteridge J.M. fifth ed. Clarendon Press; Oxford, UK: 2015. Free Radicals in Biology and Medicine. [Google Scholar]
- 2.Wang M., Zhao Q., Liu W. The versatile low-molecular-weight thiols: beyond cell protection. Bioessays. 2015;37(12):1262–1267. doi: 10.1002/bies.201500067. [DOI] [PubMed] [Google Scholar]
- 3.Cheah I.K., Halliwell B. Ergothioneine; antioxidant potential, physiological function and role in disease. Biochim. Biophys. Acta. 2012;1822(5):784–793. doi: 10.1016/j.bbadis.2011.09.017. [DOI] [PubMed] [Google Scholar]
- 4.Melville D.B. Ergothioneine. Vitam Horm. 1959;17:155–204. [Google Scholar]
- 5.Grundemann D., Harlfinger S., Golz S., Geerts A., Lazar A., Berkels R., Jung N., Rubbert A., Schomig E. Discovery of the ergothioneine transporter. Proc. Natl. Acad. Sci. U. S. A. 2005;102(14):5256–5261. doi: 10.1073/pnas.0408624102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tschirka J., Kreisor M., Betz J., Grundemann D. Substrate selectivity check of the ergothioneine transporter. Drug Metab. Dispos. 2018;46(6):779–785. doi: 10.1124/dmd.118.080440. [DOI] [PubMed] [Google Scholar]
- 7.Tucker R.A.J., Cheah I.K., Halliwell B. Specificity of the ergothioneine transporter natively expressed in HeLa cells. Biochem. Biophys. Res. Commun. 2019;513(1):22–27. doi: 10.1016/j.bbrc.2019.02.122. [DOI] [PubMed] [Google Scholar]
- 8.Kato Y., Kubo Y., Iwata D., Kato S., Sudo T., Sugiura T., Kagaya T., Wakayama T., Hirayama A., Sugimoto M., Sugihara K., Kaneko S., Soga T., Asano M., Tomita M., Matsui T., Wada M., Tsuji A. Gene knockout and metabolome analysis of carnitine/organic cation transporter OCTN1. Pharm. Res. (N. Y.) 2010;27(5):832–840. doi: 10.1007/s11095-010-0076-z. [DOI] [PubMed] [Google Scholar]
- 9.Pfeiffer C., Bach M., Bauer T., Campos da Ponte J., Schomig E., Grundemann D. Knockout of the ergothioneine transporter ETT in zebrafish results in increased 8-oxoguanine levels. Free Radic. Biol. Med. 2015;83:178–185. doi: 10.1016/j.freeradbiomed.2015.02.026. [DOI] [PubMed] [Google Scholar]
- 10.Drenberg C.D., Gibson A.A., Pounds S.B., Shi L., Rhinehart D.P., Li L., Hu S., Du G., Nies A.T., Schwab M., Pabla N., Blum W., Gruber T.A., Baker S.D., Sparreboom A. OCTN1 is a high-affinity carrier of nucleoside analogues. Canc. Res. 2017;77(8):2102–2111. doi: 10.1158/0008-5472.CAN-16-2548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pochini L., Scalise M., Di Silvestre S., Belviso S., Pandolfi A., Arduini A., Bonomini M., Indiveri C. Acetylcholine and acetylcarnitine transport in peritoneum: role of the SLC22A4 (OCTN1) transporter. Biochim. Biophys. Acta. 2016;1858(4):653–660. doi: 10.1016/j.bbamem.2015.12.026. [DOI] [PubMed] [Google Scholar]
- 12.Nishiyama M., Nakamichi N., Yoshimura T., Masuo Y., Komori T., Ishimoto T., Matsuo J.I., Kato Y. Homostachydrine is a xenobiotic substrate of OCTN1/SLC22A4 and potentially sensitizes pentylenetetrazole-induced seizures in mice. Neurochem. Res. 2020;45(11):2664–2678. doi: 10.1007/s11064-020-03118-8. [DOI] [PubMed] [Google Scholar]
- 13.Grundemann D. The ergothioneine transporter controls and indicates ergothioneine activity--a review. Prev. Med. 2012;54(Suppl):S71–S74. doi: 10.1016/j.ypmed.2011.12.001. [DOI] [PubMed] [Google Scholar]
- 14.Taubert D., Jung N., Goeser T., Schomig E. Increased ergothioneine tissue concentrations in carriers of the Crohn's disease risk-associated 503F variant of the organic cation transporter OCTN1. Gut. 2009;58(2):312–314. doi: 10.1136/gut.2008.164418. [DOI] [PubMed] [Google Scholar]
- 15.Tang R.M.Y., Cheah I.K., Yew T.S.K., Halliwell B. Distribution and accumulation of dietary ergothioneine and its metabolites in mouse tissues. Sci. Rep. 2018;8(1) doi: 10.1038/s41598-018-20021-z. Article 1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nakanishi T., Fukushi A., Sato M., Yoshifuji M., Gose T., Shirasaka Y., Ohe K., Kobayashi M., Kawai K., Tamai I. Functional characterization of apical transporters expressed in rat proximal tubular cells (PTCs) in primary culture. Mol. Pharm. 2011;8(6):2142–2150. doi: 10.1021/mp200096r. [DOI] [PubMed] [Google Scholar]
- 17.Sugiura T., Kato S., Shimizu T., Wakayama T., Nakamichi N., Kubo Y., Iwata D., Suzuki K., Soga T., Asano M., Iseki S., Tamai I., Tsuji A., Kato Y. Functional expression of carnitine/organic cation transporter OCTN1/SLC22A4 in mouse small intestine and liver. Drug Metabol. Dispos. 2010;38(10):1665–1672. doi: 10.1124/dmd.110.032763. [DOI] [PubMed] [Google Scholar]
- 18.Cheah I.K., Ong R.L., Gruber J., Yew T.S., Ng L.F., Chen C.B., Halliwell B. Knockout of a putative ergothioneine transporter in Caenorhabditis elegans decreases lifespan and increases susceptibility to oxidative damage. Free Radic. Res. 2013;47(12):1036–1045. doi: 10.3109/10715762.2013.848354. [DOI] [PubMed] [Google Scholar]
- 19.Hatano T., Saiki S., Okuzumi A., Mohney R.P., Hattori N. Identification of novel biomarkers for Parkinson's disease by metabolomic technologies. J. Neurol. Neurosurg. Psychiatry. 2016;87(3):295–301. doi: 10.1136/jnnp-2014-309676. [DOI] [PubMed] [Google Scholar]
- 20.Cheah I., Feng L., Tang R.M.Y., Lim K.H.M., Halliwell B. Ergothioneine levels in an elderly population decrease with age and incidence of cognitive decline; a risk factor for neurodegeneration? Biochem. Biophys. Res. Commun. 2016;478(1):162–167. doi: 10.1016/j.bbrc.2016.07.074. [DOI] [PubMed] [Google Scholar]
- 21.Lai Y., Xue J., Liu C.W., Gao B., Chi L., Tu P., Lu K., Ru H. Serum metabolomics identifies altered bioenergetics, signaling cascades in parallel with exposome markers in Crohn’s disease. Molecules. 2019;24(3):449. doi: 10.3390/molecules24030449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kameda M., Teruya T., Yanagida M., Kondoh H. Frailty markers comprise blood metabolites involved in antioxidation, cognition, and mobility. Proc. Natl. Acad. Sci. U. S. A. 2020;117(17):9483–9489. doi: 10.1073/pnas.1920795117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Smith E., Ottosson F., Hellstrand S., Ericson U., Orho-Melander M., Fernandez C., Melander O. Ergothioneine is associated with reduced mortality and decreased risk of cardiovascular disease. Heart. 2020;106(9):691–697. doi: 10.1136/heartjnl-2019-315485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Winkels R.M., van Brakel L., van Baar H., Beelman R.B., van Duijnhoven F.J.B., Geijsen A., van Halteren H.K., Hansson B.M.E., Richie J.P., Sun D., Wesselink E., van Zutphen M., Kampman E., Kok D.E. Are ergothioneine levels in blood associated with chronic peripheral neuropathy in colorectal cancer patients who underwent chemotherapy? Nutr. Canc. 2020;72(3):451–459. doi: 10.1080/01635581.2019.1637005. [DOI] [PubMed] [Google Scholar]
- 25.Cheah I.K., Tang R., Ye P., Yew T.S., Lim K.H., Halliwell B. Liver ergothioneine accumulation in a Guinea pig model of non-alcoholic fatty liver disease. A possible mechanism of defence? Free Radic. Res. 2016;50(1):14–25. doi: 10.3109/10715762.2015.1099642. [DOI] [PubMed] [Google Scholar]
- 26.Shinozaki Y., Furuichi K., Toyama T., Kitajima S., Hara A., Iwata Y., Sakai N., Shimizu M., Kaneko S., Isozumi N., Nagamori S., Kanai Y., Sugiura T., Kato Y., Wada T. Impairment of the carnitine/organic cation transporter 1-ergothioneine axis is mediated by intestinal transporter dysfunction in chronic kidney disease. Kidney Int. 2017;92(6):1356–1369. doi: 10.1016/j.kint.2017.04.032. [DOI] [PubMed] [Google Scholar]
- 27.Maeda T., Hirayama M., Kobayashi D., Miyazawa K., Tamai I. Mechanism of the regulation of organic cation/carnitine transporter 1 (SLC22A4) by rheumatoid arthritis-associated transcriptional factor RUNX1 and inflammatory cytokines. Drug Metab. Dispos. 2007;35(3):394–401. doi: 10.1124/dmd.106.012112. [DOI] [PubMed] [Google Scholar]
- 28.Halliwell B., Cheah I.K., Drum C.L. Ergothioneine, an adaptive antioxidant for the protection of injured tissues? A hypothesis. Biochem. Biophys. Res. Commun. 2016;470(2):245–250. doi: 10.1016/j.bbrc.2015.12.124. [DOI] [PubMed] [Google Scholar]
- 29.Beelman R.B., Kalaras M.D., Phillips A.T., Richie J.P. Is ergothioneine a ‘longevity vitamin’ limited in the American diet? J. Nutr. Sci. 2020;9:e52. doi: 10.1017/jns.2020.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Martinez-Medina G.A., Chávez-González M.L., Verma D.K., Prado-Barragán L.A., Martínez-Hernández J.L., Flores-Gallegos A.C., Thakur M., Srivastav P.P., Aguilar C.N. Bio-functional components in mushrooms, a health opportunity: ergothionine and huitlacohe as recent trends. Journal of Functional Foods. 2021;77:104326. [Google Scholar]
- 31.Toh D.S., Cheung F.S., Murray M., Pern T.K., Lee E.J., Zhou F. Functional analysis of novel variants in the organic cation/ergothioneine transporter 1 identified in Singapore populations. Mol. Pharm. 2013;10(7):2509–2516. doi: 10.1021/mp400193r. [DOI] [PubMed] [Google Scholar]
- 32.Cheah I.K., Tang R.M.Y., Yew T.S., Lim K.C., Halliwell B. Administration of pure ergothioneine to healthy human subjects; Uptake, metabolism and effects on biomarkers of oxidative damage and inflammation. Antioxidants Redox Signal. 2017;26(5):193–206. doi: 10.1089/ars.2016.6778. [DOI] [PubMed] [Google Scholar]
- 33.Taubert D., Grimberg G., Jung N., Rubbert A., Schomig E. Functional role of the 503F variant of the organic cation transporter OCTN1 in Crohn's disease. Gut. 2005;54(10):1505–1506. doi: 10.1136/gut.2005.076083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Peltekova V.D., Wintle R.F., Rubin L.A., Amos C.I., Huang Q., Gu X., Newman B., Van Oene M., Cescon D., Greenberg G., Griffiths A.M., St George-Hyslop P.H., Siminovitch K.A. Functional variants of OCTN cation transporter genes are associated with Crohn disease. Nat. Genet. 2004;36(5):471–475. doi: 10.1038/ng1339. [DOI] [PubMed] [Google Scholar]
- 35.Lin Z., Nelson L., Franke A., Poritz L., Li T.Y., Wu R., Wang Y., MacNeill C., Thomas N.J., Schreiber S., Koltun W.A. OCTN1 variant L503F is associated with familial and sporadic inflammatory bowel disease. J Crohns Colitis. 2010;4(2):132–138. doi: 10.1016/j.crohns.2009.09.003. [DOI] [PubMed] [Google Scholar]
- 36.Huff C.D., Witherspoon D.J., Zhang Y., Gatenbee C., Denson L.A., Kugathasan S., Hakonarson H., Whiting A., Davis C.T., Wu W., Xing J., Watkins W.S., Bamshad M.J., Bradfield J.P., Bulayeva K., Simonson T.S., Jorde L.B., Guthery S.L. Crohn's disease and genetic hitchhiking at IBD5. Mol. Biol. Evol. 2012;29(1):101–111. doi: 10.1093/molbev/msr151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Newman B., Wintle R.F., van Oene M., Yazdanpanah M., Owen J., Johnson B., Gu X., Amos C.I., Keystone E., Rubin L.A., Siminovitch K.A. SLC22A4 polymorphisms implicated in rheumatoid arthritis and Crohn's disease are not associated with rheumatoid arthritis in a Canadian Caucasian population. Arthritis Rheum. 2005;52(2):425–429. doi: 10.1002/art.20854. [DOI] [PubMed] [Google Scholar]
- 38.Lamhonwah A.M., Tein I. Novel localization of OCTN1, an organic cation/carnitine transporter, to mammalian mitochondria. Biochem. Biophys. Res. Commun. 2006;345(4):1315–1325. doi: 10.1016/j.bbrc.2006.05.026. [DOI] [PubMed] [Google Scholar]
- 39.Paul B.D., Snyder S.H. The unusual amino acid L-ergothioneine is a physiologic cytoprotectant. Cell Death Differ. 2010;17(7):1134–1140. doi: 10.1038/cdd.2009.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shitara Y., Nakamichi N., Norioka M., Shima H., Kato Y., Horie T. Role of organic cation/carnitine transporter 1 in uptake of phenformin and inhibitory effect on complex I respiration in mitochondria. Toxicol. Sci. 2013;132(1):32–42. doi: 10.1093/toxsci/kfs330. [DOI] [PubMed] [Google Scholar]
- 41.Kawano H., Otani M., Takeyama K., Kawai Y., Mayumi T., Hama T. Studies on ergothioneine. VI. Distribution and fluctuations of ergothioneine in rats. Chem. Pharm. Bull. (Tokyo) 1982;30(5):1760–1765. doi: 10.1248/cpb.30.1760. [DOI] [PubMed] [Google Scholar]
- 42.Bazela K., Solyga-Zurek A., Debowska R., Rogiewicz K., Bartnik E., Eris I. l-Ergothioneine protects skin cells against UV-induced damage—a preliminary study. Cosmetics. 2014;1(1):51–60. [Google Scholar]
- 43.Williamson R.D., McCarthy F.P., Manna S., Groarke E., Kell D.B., Kenny L.C., McCarthy C.M. L-(+)-Ergothioneine significantly improves the clinical characteristics of preeclampsia in the reduced uterine perfusion pressure rat model. Hypertension. 2020;75(2):561–568. doi: 10.1161/HYPERTENSIONAHA.119.13929. [DOI] [PubMed] [Google Scholar]
- 44.Yee S.W., Buitrago D., Stecula A., Ngo H.X., Chien H.C., Zou L., Koleske M.L., Giacomini K.M. Deorphaning a solute carrier 22 family member, SLC22A15, through functional genomic studies. FASEB J. 2020;34(12):15734–15752. doi: 10.1096/fj.202001497R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Al-Majdoub Z.M., Al Feteisi H., Achour B., Warwood S., Neuhoff S., Rostami-Hodjegan A., Barber J. Proteomic quantification of human blood-brain barrier SLC and ABC transporters in healthy individuals and dementia patients. Mol. Pharm. 2019;16(3):1220–1233. doi: 10.1021/acs.molpharmaceut.8b01189. [DOI] [PubMed] [Google Scholar]
- 46.Tsuji, A., Influx transporters and drug targeting: application of peptide and cation transporters. Int. Congr., 2005. 1277: p. 75-84.
- 47.Halliwell B., Cheah I.K., Tang R.M.Y. Ergothioneine - a diet-derived antioxidant with therapeutic potential. FEBS Lett. 2018;592(20):3357–3366. doi: 10.1002/1873-3468.13123. [DOI] [PubMed] [Google Scholar]
- 48.Vavich J.M., Howell R.R. Ultraviolet-absorbing compounds in urine of normal newborns and young children. Clin. Chem. 1970;16(8):702–706. [PubMed] [Google Scholar]
- 49.Graham S.F., Chevallier O.P., Kumar P., Türko Gcaron Lu O., Bahado-Singh R.O. Metabolomic profiling of brain from infants who died from Sudden Infant Death Syndrome reveals novel predictive biomarkers. J. Perinatol. 2017;37(1):91–97. doi: 10.1038/jp.2016.139. [DOI] [PubMed] [Google Scholar]
- 50.Wu X., George R.L., Huang W., Wang H., Conway S.J., Leibach F.H., Ganapathy V. Structural and functional characteristics and tissue distribution pattern of rat OCTN1, an organic cation transporter, cloned from placenta. Biochim. Biophys. Acta. 2000;1466(1–2):315–327. doi: 10.1016/s0005-2736(00)00189-9. [DOI] [PubMed] [Google Scholar]
- 51.Kwok B., Yamauchi A., Rajesan R., Chan L., Dhillon U., Gao W., Xu H., Wang B., Takahashi S., Semple J., Tamai I., Nezu J., Tsuji A., Harper P., Ito S. Carnitine/xenobiotics transporters in the human mammary gland epithelia, MCF12A. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006;290(3):R793–R802. doi: 10.1152/ajpregu.00087.2005. [DOI] [PubMed] [Google Scholar]
- 52.Alcorn J., Lu X., Moscow J.A., McNamara P.J. Transporter gene expression in lactating and nonlactating human mammary epithelial cells using real-time reverse transcription-polymerase chain reaction. J. Pharmacol. Exp. Therapeut. 2002;303(2):487–496. doi: 10.1124/jpet.102.038315. [DOI] [PubMed] [Google Scholar]
- 53.Sotgia S., Pisanu E., Cambedda D., Pintus G., Carru C., Zinellu A. Ultra-performance liquid chromatographic determination of L-ergothioneine in commercially available classes of cow milk. J. Food Sci. 2014;79(9):C1683–C1687. doi: 10.1111/1750-3841.12564. [DOI] [PubMed] [Google Scholar]
- 54.Nakamichi N., Nakayama K., Ishimoto T., Masuo Y., Wakayama T., Sekiguchi H., Sutoh K., Usumi K., Iseki S., Kato Y. Food-derived hydrophilic antioxidant ergothioneine is distributed to the brain and exerts antidepressant effect in mice. Brain Behav. 2016 doi: 10.1002/brb3.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ishimoto T., Masuo Y., Kato Y., Nakamichi N. Ergothioneine-induced neuronal differentiation is mediated through activation of S6K1 and neurotrophin 4/5-TrkB signaling in murine neural stem cells. Cell. Signal. 2019;53:269–280. doi: 10.1016/j.cellsig.2018.10.012. [DOI] [PubMed] [Google Scholar]
- 56.Nakamichi N., Nakao S., Nishiyama M., Takeda Y., Ishimoto T., Masuo Y., Matsumoto S., Suzuki M., Kato Y. Oral administration of the food derived hydrophilic antioxidant ergothioneine enhances object recognition memory in mice. Curr. Mol. Pharmacol. 2021;14(2):220–233. doi: 10.2174/1874467213666200212102710. [DOI] [PubMed] [Google Scholar]
- 57.Forster R., Spezia F., Papineau D., Sabadie C., Erdelmeier I., Moutet M., Yadan J.C. Reproductive safety evaluation of L-Ergothioneine. Food Chem. Toxicol. 2015;80:85–91. doi: 10.1016/j.fct.2015.02.019. [DOI] [PubMed] [Google Scholar]
- 58.Turck D., Bresson J.L., Burlingame B., Dean T., Fairweather‐Tait S., Heinonen M., Hirsch‐Ernst K.I., Mangelsdorf I., McArdle H.J., Naska A., Neuhäuser‐Berthold M., Nowicka G., Pentieva K., Sanz Y., Siani A., Sjödin A., Stern M., Tomé D., Vinceti M., Willatts P., Engel K.H., Marchelli R., Pöting A., Poulsen M., Schlatter J.R., Ackerl R., Loveren H. Statement on the safety of synthetic l‐ergothioneine as a novel food – supplementary dietary exposure and safety assessment for infants and young children, pregnant and breastfeeding women. EFSA J. 2017;15(11) doi: 10.2903/j.efsa.2017.5060. p. report 5060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kumosani T.A. L-ergothioneine level in red blood cells of healthy human males in the Western province of Saudi Arabia. Exp. Mol. Med. 2001;33(1):20–22. doi: 10.1038/emm.2001.4. [DOI] [PubMed] [Google Scholar]
- 60.Stoffels C., Oumari M., Perrou A., Termath A., Schlundt W., Schmalz H.-G., Schäfer M., Wewer V., Metzger S., Schömig E., Gründemann D. Ergothioneine stands out from hercynine in the reaction with singlet oxygen: resistance to glutathione and TRIS in the generation of specific products indicates high reactivity. Free Radic. Biol. Med. 2017;113:385–394. doi: 10.1016/j.freeradbiomed.2017.10.372. [DOI] [PubMed] [Google Scholar]
- 61.Stampfli A.R., Blankenfeldt W., Seebeck F.P. Structural basis of ergothioneine biosynthesis. Curr. Opin. Struct. Biol. 2020;65:1–8. doi: 10.1016/j.sbi.2020.04.002. [DOI] [PubMed] [Google Scholar]
- 62.Burn R., Misson L., Meury M., Seebeck F.P. Anaerobic origin of ergothioneine. Angew Chem. Int. Ed. Engl. 2017;56(41):12508–12511. doi: 10.1002/anie.201705932. [DOI] [PubMed] [Google Scholar]
- 63.Nakamura T., Sugiura S., Kobayashi D., Yoshida K., Yabuuchi H., Aizawa S., Maeda T., Tamai I. Decreased proliferation and erythroid differentiation of K562 cells by siRNA-induced depression of OCTN1 (SLC22A4) transporter gene. Pharm. Res. (N. Y.) 2007;24(9):1628–1635. doi: 10.1007/s11095-007-9290-8. [DOI] [PubMed] [Google Scholar]
- 64.Fraser R.S. Blood ergothioneine levels in disease. J. Lab. Clin. Med. 1951;37(2):199–206. [PubMed] [Google Scholar]
- 65.Yoshida S., Shime H., Matsumoto M., Kasahara M., Seya T. Anti-oxidative amino acid L-ergothioneine modulates the tumor microenvironment to facilitate adjuvant vaccine immunotherapy. Front. Immunol. 2019;10:671. doi: 10.3389/fimmu.2019.00671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sotgia S., Taras A., Zinellu A., Cherchi R., Mangoni A.A., Carru C., Bogliolo L. Hercynine, ergothioneine and redox state in stallion’s seminal plasma. Antioxidants. 2020;9(9):855. doi: 10.3390/antiox9090855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Strzezek R., Koziorowska-Gilun M., Kowalowka M., Strzezek J. Characteristics of antioxidant system in dog semen. Pol. J. Vet. Sci. 2009;12(1):55–60. [PubMed] [Google Scholar]
- 68.Nikodemus D., Lazic D., Bach M., Bauer T., Pfeiffer C., Wiltzer L., Lain E., Schomig E., Grundemann D. Paramount levels of ergothioneine transporter SLC22A4 mRNA in boar seminal vesicles and cross-species analysis of ergothioneine and glutathione in seminal plasma. J. Physiol. Pharmacol. 2011;62(4):411–419. [PubMed] [Google Scholar]
- 69.Wagner H., Cheng J.W., Ko E.Y. Role of reactive oxygen species in male infertility: an updated review of literature. Arab J Urol. 2018;16(1):35–43. doi: 10.1016/j.aju.2017.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Halliwell B. Reflections of an aging free radical. Free Radic. Biol. Med. 2020;161:234–245. doi: 10.1016/j.freeradbiomed.2020.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Sawhney P., Giammona C.J., Meistrich M.L., Richburg J.H. Cisplatin-induced long-term failure of spermatogenesis in adult C57/Bl/6J mice. J. Androl. 2005;26(1):136–145. [PubMed] [Google Scholar]
- 72.Ayobami D., Olaniyan O., Salihu M., Illesanmi K. L-ergothioneine supplement protect testicular functions in cisplatin-treated Wistar rats. J Pharm Biol Sci. 2019;14(1):6–13. [Google Scholar]
- 73.Andersson A.M., Jorgensen N., Main K.M., Toppari J., Rajpert-De Meyts E., Leffers H., Juul A., Jensen T.K., Skakkebaek N.E. Adverse trends in male reproductive health: we may have reached a crucial 'tipping point. Int. J. Androl. 2008;31(2):74–80. doi: 10.1111/j.1365-2605.2007.00853.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wynn T.A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 2008;214(2):199–210. doi: 10.1002/path.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Deiana M., Rosa A., Casu V., Piga R., Assunta Dessi M., Aruoma O.I. L-ergothioneine modulates oxidative damage in the kidney and liver of rats in vivo: studies upon the profile of polyunsaturated fatty acids. Clin. Nutr. 2004;23(2):183–193. doi: 10.1016/S0261-5614(03)00108-0. [DOI] [PubMed] [Google Scholar]
- 76.Laurenza I., Colognato R., Migliore L., Del Prato S., Benzi L. Modulation of palmitic acid-induced cell death by ergothioneine: evidence of an anti-inflammatory action. Biofactors. 2008;33(4):237–247. doi: 10.1002/biof.5520330401. [DOI] [PubMed] [Google Scholar]
- 77.Sakrak O., Kerem M., Bedirli A., Pasaoglu H., Akyurek N., Ofluoglu E., Gultekin F.A. Ergothioneine modulates proinflammatory cytokines and heat shock protein 70 in mesenteric ischemia and reperfusion injury. J. Surg. Res. 2008;144(1):36–42. doi: 10.1016/j.jss.2007.04.020. [DOI] [PubMed] [Google Scholar]
- 78.Tang Y., Masuo Y., Sakai Y., Wakayama T., Sugiura T., Harada R., Futatsugi A., Komura T., Nakamichi N., Sekiguchi H., Sutoh K., Usumi K., Iseki S., Kaneko S., Kato Y. Localization of xenobiotic transporter OCTN1/SLC22A4 in hepatic stellate cells and its protective role in liver fibrosis. J. Pharmacol. Sci. 2016;105(5):1779–1789. doi: 10.1016/j.xphs.2016.02.023. [DOI] [PubMed] [Google Scholar]
- 79.Ehrhardt C., Koay A., Clerkin C., Selo M.A. Impact of ergothioneine on idiopathic pulmonary fibrosis markers in human lung epithelial cells in vitro. FASEB J. 2019;33(1) 127.11. [Google Scholar]
- 80.Repine J.E., Elkins N.D. Effect of ergothioneine on acute lung injury and inflammation in cytokine insufflated rats. Prev. Med. 2012;54(Suppl):S79–S82. doi: 10.1016/j.ypmed.2011.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ehrhardt C., Nickel S., Selo M.A., Clerkin C., Salomon J., Talbot B., Walsh J. OCTN1-Mediated ergothioneine uptake protects lung epithelial cells from tobacco smoke-induced damage. FASEB J. 2015;29(S1):970–977. [Google Scholar]
- 82.Ehrhardt C., Selo M.A., Clerkin C.G., Talbot B.N., Walsh J.J., Nakamichi N., Kato Y., Lewis J.B., Reynolds P.R., Nickel S. Protective role of ergothioneine from tobacco smoke-induced oxidative stress in vitro and in vivo. FASEB J. 2016;30(S1) 982.1-982.1. [Google Scholar]
- 83.Phipps E.A., Thadhani R., Benzing T., Karumanchi S.A. Pre-eclampsia: pathogenesis, novel diagnostics and therapies. Nat. Rev. Nephrol. 2019;15(5):275–289. doi: 10.1038/s41581-019-0119-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kerley R.N., McCarthy C., Kell D.B., Kenny L.C. The potential therapeutic effects of ergothioneine in pre-eclampsia. Free Radic. Biol. Med. 2018;117:145–157. doi: 10.1016/j.freeradbiomed.2017.12.030. [DOI] [PubMed] [Google Scholar]
- 85.Morillon A.C., Williamson R.D., Baker P.N., Kell D.B., Kenny L.C., English J.A., McCarthy F.P., McCarthy C. Effect of L-Ergothioneine on the metabolic plasma profile of the RUPP rat model of pre-eclampsia. PloS One. 2020;15(3) doi: 10.1371/journal.pone.0230977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Bedirli A., Sakrak O., Muhtaroglu S., Soyuer I., Guler I., Riza Erdogan A., Sozuer E.M. Ergothioneine pretreatment protects the liver from ischemia-reperfusion injury caused by increasing hepatic heat shock protein 70. J. Surg. Res. 2004;122(1):96–102. doi: 10.1016/j.jss.2004.06.016. [DOI] [PubMed] [Google Scholar]
- 87.Sakrak Ö., Kerem M., Bedirli A., Pasaoğlu H., Alper M., Ofluoğlu E., Yılmaz T.U. Ergothioneine prevents acute lung injury in mesenteric ischemia and reperfusion injury in rats. J. Crit. Care. 2008;23(2):268–269. [Google Scholar]
- 88.Swain S.M., Whaley F.S., Ewer M.S. Congestive heart failure in patients treated with doxorubicin: a retrospective analysis of three trials. Cancer. 2003;97(11):2869–2879. doi: 10.1002/cncr.11407. [DOI] [PubMed] [Google Scholar]
- 89.Yoshida S., Shime H., Funami K., Takaki H., Matsumoto M., Kasahara M., Seya T. The anti-oxidant ergothioneine augments the immunomodulatory function of TLR agonists by direct action on macrophages. PloS One. 2017;12(1) doi: 10.1371/journal.pone.0169360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Obayashi K., Kurihara K., Okano Y., Masaki H., Yarosh D.B. L-Ergothioneine scavenges superoxide and singlet oxygen and suppresses TNF-alpha and MMP-1 expression in UV-irradiated human dermal fibroblasts. J. Cosmet. Sci. 2005;56(1):17–27. [PubMed] [Google Scholar]
- 91.Hseu Y.C., Lo H.W., Korivi M., Tsai Y.C., Tang M.J., Yang H.L. Dermato-protective properties of ergothioneine through induction of Nrf2/ARE-mediated antioxidant genes in UVA-irradiated Human keratinocytes. Free Radic. Biol. Med. 2015;86:102–117. doi: 10.1016/j.freeradbiomed.2015.05.026. [DOI] [PubMed] [Google Scholar]
- 92.Damaghi N., Dong K., Smiles K., Yarosh D. The natural antioxidant L-ergothioneine and its receptor/transporter OCTN-1 participate in the skin's response to UVA-induced oxidative damage. J. Am. Acad. Dermatol. 2008;58(2) AB111-AB111. [Google Scholar]
- 93.Sotgia S., Zinellu A., Mangoni A.A., Pintus G., Attia J., Carru C., McEvoy M. Clinical and biochemical correlates of serum L-ergothioneine concentrations in community-dwelling middle-aged and older adults. PloS One. 2014;9(1) doi: 10.1371/journal.pone.0084918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ames B.N. Prolonging healthy aging: longevity vitamins and proteins. Proc. Natl. Acad. Sci. U. S. A. 2018;115(43):10836–10844. doi: 10.1073/pnas.1809045115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Schmidt M., Giessl A., Laufs T., Hankeln T., Wolfrum U., Burmester T. How does the eye breathe? Evidence for neuroglobin-mediated oxygen supply in the mammalian retina. J. Biol. Chem. 2003;278(3):1932–1935. doi: 10.1074/jbc.M209909200. [DOI] [PubMed] [Google Scholar]
- 96.Shires T.K., Brummel M.C., Pulido J.S., Stegink L.D. Ergothioneine (ert) concentrations and distribution in bovine and porcine eyes. FASEB J. 1993;7(4):A732. doi: 10.1016/s0742-8413(96)00223-x. [DOI] [PubMed] [Google Scholar]
- 97.Shires T.K., Brummel M.C., Pulido J.S., Stegink L.D. Ergothioneine distribution in bovine and porcine ocular tissues. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1997;117(1):117–120. doi: 10.1016/s0742-8413(96)00223-x. [DOI] [PubMed] [Google Scholar]
- 98.Shukla Y., Kulshrestha O.P., Khuteta K.P. Ergothioneine content in normal and senile human cataractous lenses. Indian J. Med. Res. 1981;73:472–473. [PubMed] [Google Scholar]
- 99.S Y., P K.O., P K.K. Study of content of redox substances ergothioneine, glutathione and ascorbic acid in normal and senile cataractous lenses in human eyes. Indian J. Ophthalmol. 1982;30:441–443. [Google Scholar]
- 100.Kim E.C., Toyono T., Berlinicke C.A., Zack D.J., Jurkunas U., Usui T., Jun A.S. Screening and characterization of drugs that protect corneal endothelial cells against unfolded protein response and oxidative stress. Invest. Ophthalmol. Vis. Sci. 2017;58(2):892–900. doi: 10.1167/iovs.16-20147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Borodina I., Kenny L.C., McCarthy C.M., Paramasivan K., Pretorius E., Roberts T.J., van der Hoek S.A., Kell D.B. The biology of ergothioneine, an antioxidant nutraceutical. Nutr. Res. Rev. 2020:1–28. doi: 10.1017/S0954422419000301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Song T.-Y., Chen C.-L., Liao J.-W., Ou H.-C., Tsai M.-S. Ergothioneine protects against neuronal injury induced by cisplatin both in vitro and in vivo. Food Chem. Toxicol. 2010;48(12):3492–3499. doi: 10.1016/j.fct.2010.09.030. [DOI] [PubMed] [Google Scholar]
- 103.Song T.Y., Lin H.C., Chen C.L., Wu J.H., Liao J.W., Hu M.L. Ergothioneine and melatonin attenuate oxidative stress and protect against learning and memory deficits in C57BL/6J mice treated with D-galactose. Free Radic. Res. 2014;48(9):1049–1060. doi: 10.3109/10715762.2014.920954. [DOI] [PubMed] [Google Scholar]
- 104.Song T.Y., Yang N.C., Chen C.L., Thi T.L.V. Protective effects and possible mechanisms of ergothioneine and hispidin against methylglyoxal-induced injuries in rat pheochromocytoma cells. Oxid Med Cell Longev. 2017;2017:4824371. doi: 10.1155/2017/4824371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Koh S.S., Ooi S.C., Lui N.M., Qiong C., Ho L.T., Cheah I.K., Halliwell B., Herr D.R., Ong W.Y. Effect of ergothioneine on 7-ketocholesterol-induced endothelial injury. NeuroMolecular Med. 2020 doi: 10.1007/s12017-020-08620-4. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cheah I.K., Ng L.T., Ng L.F., Lam V.Y., Gruber J., Huang C.Y.W., Goh F.Q., Lim K.H.C., Halliwell B. Inhibition of amyloid-induced toxicity by ergothioneine in a transgenic Caenorhabditis elegans model. FEBS Lett. 2019;593(16):2139–2150. doi: 10.1002/1873-3468.13497. [DOI] [PubMed] [Google Scholar]
- 107.Yang N.C., Lin H.C., Wu J.H., Ou H.C., Chai Y.C., Tseng C.Y., Liao J.W., Song T.Y. Ergothioneine protects against neuronal injury induced by beta-amyloid in mice. Food Chem. Toxicol. 2012;50(11):3902–3911. doi: 10.1016/j.fct.2012.08.021. [DOI] [PubMed] [Google Scholar]
- 108.Nakamichi N., Taguchi T., Hosotani H., Wakayama T., Shimizu T., Sugiura T., Iseki S., Kato Y. Functional expression of carnitine/organic cation transporter OCTN1 in mouse brain neurons: possible involvement in neuronal differentiation. Neurochem. Int. 2012;61(7):1121–1132. doi: 10.1016/j.neuint.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 109.Ishimoto T., Nakamichi N., Hosotani H., Masuo Y., Sugiura T., Kato Y. Organic cation transporter-mediated ergothioneine uptake in mouse neural progenitor cells suppresses proliferation and promotes differentiation into neurons. PloS One. 2014;9(2) doi: 10.1371/journal.pone.0089434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Lee J.E., Lim M.S., Park J.H., Park C.H., Koh H.C. S6K promotes dopaminergic neuronal differentiation through PI3K/Akt/mTOR-Dependent signaling pathways in human neural stem cells. Mol. Neurobiol. 2016;53(6):3771–3782. doi: 10.1007/s12035-015-9325-9. [DOI] [PubMed] [Google Scholar]
- 111.Owen J.E., Veasey S.C. Impact of sleep disturbances on neurodegeneration: insight from studies in animal models. Neurobiol. Dis. 2020;139:104820. doi: 10.1016/j.nbd.2020.104820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Abbott S.M., Videnovic A. Chronic sleep disturbance and neural injury: links to neurodegenerative disease. Nat. Sci. Sleep. 2016;8:55–61. doi: 10.2147/NSS.S78947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Byers A.L., Yaffe K. Depression and risk of developing dementia. Nat. Rev. Neurol. 2011;7(6):323–331. doi: 10.1038/nrneurol.2011.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Dafsari F.S., Jessen F. Depression-an underrecognized target for prevention of dementia in Alzheimer's disease. Transl. Psychiatry. 2020;10(1):160. doi: 10.1038/s41398-020-0839-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Matsuda Y., Ozawa N., Shinozaki T., Wakabayashi K.I., Suzuki K., Kawano Y., Ohtsu I., Tatebayashi Y. Ergothioneine, a metabolite of the gut bacterium Lactobacillus reuteri, protects against stress-induced sleep disturbances. Transl. Psychiatry. 2020;10(1):170. doi: 10.1038/s41398-020-0855-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Moncaster J.A., Walsh D.T., Gentleman S.M., Jen L.S., Aruoma O.I. Ergothioneine treatment protects neurons against N-methyl-D-aspartate excitotoxicity in an in vivo rat retinal model. Neurosci. Lett. 2002;328(1):55–59. doi: 10.1016/s0304-3940(02)00427-5. [DOI] [PubMed] [Google Scholar]
- 117.An Y., Varma V.R., Varma S., Casanova R., Dammer E., Pletnikova O., Chia C.W., Egan J.M., Ferrucci L., Troncoso J., Levey A.I., Lah J., Seyfried N.T., Legido-Quigley C., O'Brien R., Thambisetty M. Evidence for brain glucose dysregulation in Alzheimer's disease. Alzheimers Dement. 2018;14(3):318–329. doi: 10.1016/j.jalz.2017.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Butterfield D.A., Halliwell B. Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat. Rev. Neurosci. 2019;20:148–160. doi: 10.1038/s41583-019-0132-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Li R.W., Yang C., Sit A.S., Kwan Y.W., Lee S.M., Hoi M.P., Chan S.W., Hausman M., Vanhoutte P.M., Leung G.P. Uptake and protective effects of ergothioneine in human endothelial cells. J. Pharmacol. Exp. Therapeut. 2014;350(3):691–700. doi: 10.1124/jpet.114.214049. [DOI] [PubMed] [Google Scholar]
- 120.Lyros E., Bakogiannis C., Liu Y., Fassbender K. Molecular links between endothelial dysfunction and neurodegeneration in Alzheimer's disease. Curr. Alzheimer Res. 2014;11(1):18–26. doi: 10.2174/1567205010666131119235254. [DOI] [PubMed] [Google Scholar]
- 121.Ahmad A., Patel V., Xiao J., Khan M.M. The role of neurovascular system in neurodegenerative diseases. Mol. Neurobiol. 2020;57(11):4373–4393. doi: 10.1007/s12035-020-02023-z. [DOI] [PubMed] [Google Scholar]
- 122.Sit A.S.M., Ho E.Y., Li R.W., Man R.Y., Kwan Y., Vanhoutte P.M., Leung G.P. Ergothioneine shows protective effect on endothelial cells in oxidative stress. FASEB J. 2011;25(S1) 630.3-630.3. [Google Scholar]
- 123.Hascalovici J.R., Vaya J., Khatib S., Holcroft C.A., Zukor H., Song W., Arvanitakis Z., Bennett D.A., Schipper H.M. Brain sterol dysregulation in sporadic AD and MCI: relationship to heme oxygenase-1. J. Neurochem. 2009;110(4):1241–1253. doi: 10.1111/j.1471-4159.2009.06213.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Poli G., Biasi F., Leonarduzzi G. Oxysterols in the pathogenesis of major chronic diseases. Redox Biol. 2013;1:125–130. doi: 10.1016/j.redox.2012.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kreilaus F., Spiro A.S., McLean C.A., Garner B., Jenner A.M. Evidence for altered cholesterol metabolism in Huntington's disease post mortem brain tissue. Neuropathol. Appl. Neurobiol. 2016;42(6):535–546. doi: 10.1111/nan.12286. [DOI] [PubMed] [Google Scholar]
- 126.Versari D., Daghini E., Virdis A., Ghiadoni L., Taddei S. Endothelial dysfunction as a target for prevention of cardiovascular disease. Diabetes Care. 2009;32(suppl 2):S314–S321. doi: 10.2337/dc09-S330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Servillo L., D'Onofrio N., Casale R., Cautela D., Giovane A., Castaldo D., Balestrieri M.L. Ergothioneine products derived by superoxide oxidation in endothelial cells exposed to high-glucose. Free Radic. Biol. Med. 2017;108:8–18. doi: 10.1016/j.freeradbiomed.2017.03.009. [DOI] [PubMed] [Google Scholar]
- 128.Saing L., Wei Y.C., Tseng C.J. Ergothioneine represses inflammation and dysfunction in human endothelial cells exposed to oxidized low-density lipoprotein. Clin. Exp. Pharmacol. Physiol. 2016;43(7):720. doi: 10.1111/1440-1681.12374. [DOI] [PubMed] [Google Scholar]
- 129.Martin K.R. The bioactive agent ergothioneine, a key component of dietary mushrooms, inhibits monocyte binding to endothelial cells characteristic of early cardiovascular disease. J. Med. Food. 2010;13(6):1340–1346. doi: 10.1089/jmf.2009.0194. [DOI] [PubMed] [Google Scholar]
- 130.Ey J., Schomig E., Taubert D. Dietary sources and antioxidant effects of ergothioneine. J. Agric. Food Chem. 2007;55(16):6466–6474. doi: 10.1021/jf071328f. [DOI] [PubMed] [Google Scholar]
- 131.Pfeiffer C., Bauer T., Surek B., Schomig E., Grundemann D. Cyanobacteria produce high levels of ergothioneine. Food Chem. 2011;129(4):1766–1769. [Google Scholar]
- 132.Kalaras M.D., Richie J.P., Calcagnotto A., Beelman R.B. Mushrooms: a rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017;233:429–433. doi: 10.1016/j.foodchem.2017.04.109. [DOI] [PubMed] [Google Scholar]
- 133.Zhang S., Tomata Y., Sugiyama K., Sugawara Y., Tsuji I. Mushroom consumption and incident dementia in elderly Japanese: the Ohsaki cohort 2006 study. J. Am. Geriatr. Soc. 2017;65(7):1462–1469. doi: 10.1111/jgs.14812. [DOI] [PubMed] [Google Scholar]
- 134.Feng L., Cheah I.K., Ng M.M., Li J., Chan S.M., Lim S.L., Mahendran R., Kua E.H., Halliwell B. The association between mushroom consumption and mild cognitive impairment: a community-based cross-sectional study in Singapore. J Alzheimers Dis. 2019;68(1):197–203. doi: 10.3233/JAD-180959. [DOI] [PubMed] [Google Scholar]
- 135.Zhang S., Sugawara Y., Chen S., Beelman R.B., Tsuduki T., Tomata Y., Matsuyama S., Tsuji I. Mushroom consumption and incident risk of prostate cancer in Japan: a pooled analysis of the Miyagi Cohort Study and the Ohsaki Cohort Study. Int. J. Canc. 2020;146(10):2712–2720. doi: 10.1002/ijc.32591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Shin A., Kim J., Lim S.Y., Kim G., Sung M.K., Lee E.S., Ro J. Dietary mushroom intake and the risk of breast cancer based on hormone receptor status. Nutr. Canc. 2010;62(4):476–483. doi: 10.1080/01635580903441212. [DOI] [PubMed] [Google Scholar]
- 137.Schwartz B., Hadar Y. Possible mechanisms of action of mushroom-derived glucans on inflammatory bowel disease and associated cancer. Ann. Transl. Med. 2014;2(2):19. doi: 10.3978/j.issn.2305-5839.2014.01.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Maheshwari G., Gessner D.K., Meyer S., Ahlborn J., Wen G., Ringseis R., Zorn H., Eder K. Characterization of the nutritional composition of a biotechnologically produced oyster mushroom and its physiological effects in obese Zucker rats. Mol. Nutr. Food Res. 2020;64 doi: 10.1002/mnfr.202000591. [DOI] [PubMed] [Google Scholar]
- 139.Yadav S.K., Ir R., Jeewon R., Doble M., Hyde K.D., Kaliappan I., Jeyaraman R., Reddi R.N., Krishnan J., Li M., Durairajan S.S.K. A mechanistic review on medicinal mushrooms-derived bioactive compounds: potential mycotherapy candidates for alleviating neurological disorders. Planta Med. 2020;86(16):1161–1175. doi: 10.1055/a-1177-4834. [DOI] [PubMed] [Google Scholar]
- 140.Lindequist U., Niedermeyer T.H., Julich W.D. The pharmacological potential of mushrooms. Evid Based Complement Alternat Med. 2005;2(3):285–299. doi: 10.1093/ecam/neh107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Dubost N.J., Ou B., Beelman R.B. Quantification of polyphenols and ergothioneine in cultivated mushrooms and correlation to total antioxidant capacity. Food Chem. 2007;105(2):727–735. [Google Scholar]
- 142.Weigand-Heller A.J., Kris-Etherton P.M., Beelman R.B. The bioavailability of ergothioneine from mushrooms (Agaricus bisporus) and the acute effects on antioxidant capacity and biomarkers of inflammation. Prev. Med. 2012;54(Suppl):S75–S78. doi: 10.1016/j.ypmed.2011.12.028. [DOI] [PubMed] [Google Scholar]
- 143.Calvo M.S., Mehrotra A., Beelman R.B., Nadkarni G., Wang L., Cai W., Boon C.G., Kalaras M.D., Uribarri J. A retrospective study in adults with metabolic syndrome: diabetic risk factor response to daily consumption of Agaricus Bisporus (white button mushrooms) Plant Foods Hum. Nutr. 2016;71(3):245–251. doi: 10.1007/s11130-016-0552-7. [DOI] [PubMed] [Google Scholar]
- 144.Bull M.J., Plummer N.T. Part 1: the human gut microbiome in health and disease. Integr. Med. 2014;13(6):17–22. [PMC free article] [PubMed] [Google Scholar]
- 145.Valdes A.M., Walter J., Segal E., Spector T.D. Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179. doi: 10.1136/bmj.k2179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Genghof D.S., Van Damme O. Biosynthesis of ergothioneine from endogenous hercynine in Mycobacterium smegmatis. J. Bacteriol. 1968;95(2):340–344. doi: 10.1128/jb.95.2.340-344.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Sao Emani C., Williams M.J., Wiid I.J., Hiten N.F., Viljoen A.J., Pietersen R.D., van Helden P.D., Baker B. Ergothioneine is a secreted antioxidant in Mycobacterium smegmatis. Antimicrob. Agents Chemother. 2013;57(7):3202–3207. doi: 10.1128/AAC.02572-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Richard-Greenblatt M., Bach H., Adamson J., Pena-Diaz S., Li W., Steyn A.J., Av-Gay Y. Regulation of ergothioneine biosynthesis and its effect on Mycobacterium tuberculosis growth and infectivity. J. Biol. Chem. 2015;290(38):23064–23076. doi: 10.1074/jbc.M115.648642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Gamage A.K., Liao C., Cheah I.K., Chen Y., Lim D.R.X., Ku J.W.K., Chee R.S.L., Gengenbacher M., Seebeck F.P., Halliwell B., Gan Y.H. The proteobacterial species Burkholderia pseudomallei produces ergothioneine which enhances virulence in mammalian infection. FASEB J. 2018;32(12):6395–6409. doi: 10.1096/fj.201800716. [DOI] [PubMed] [Google Scholar]
- 150.Seebeck F.P. In vitro reconstitution of Mycobacterial ergothioneine biosynthesis. J. Am. Chem. Soc. 2010;132(19):6632–6633. doi: 10.1021/ja101721e. [DOI] [PubMed] [Google Scholar]
- 151.Osawa R., Kamide T., Satoh Y., Kawano Y., Ohtsu I., Dairi T. Heterologous and high production of ergothioneine in Escherichia coli. J. Agric. Food Chem. 2018;66(5):1191–1196. doi: 10.1021/acs.jafc.7b04924. [DOI] [PubMed] [Google Scholar]
- 152.Tanaka N., Kawano Y., Satoh Y., Dairi T., Ohtsu I. Gram-scale fermentative production of ergothioneine driven by overproduction of cysteine in Escherichia coli. Sci. Rep. 2019;9(1):1895. doi: 10.1038/s41598-018-38382-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Carabotti M., Scirocco A., Maselli M.A., Severi C. The gut-brain axis: interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015;28(2):203–209. [PMC free article] [PubMed] [Google Scholar]
- 154.Wolff J.B. Ergothionase from Escherichia coli. J. Biol. Chem. 1962;237:874–881. [PubMed] [Google Scholar]
- 155.Muramatsu H., Matsuo H., Okada N., Ueda M., Yamamoto H., Kato S., Nagata S. Characterization of ergothionase from Burkholderia sp. HME13 and its application to enzymatic quantification of ergothioneine. Appl. Microbiol. Biotechnol. 2013;97(12):5389–5400. doi: 10.1007/s00253-012-4442-0. [DOI] [PubMed] [Google Scholar]
- 156.Maurer A., Leisinger F., Lim D., Seebeck F.P. Structure and mechanism of ergothionase from Treponema denticola. Chemistry. 2019;25(44):10298–10303. doi: 10.1002/chem.201901866. [DOI] [PubMed] [Google Scholar]
- 157.Yanasugondha D., Appleman M.D. Degradation of ergothioneine by Alcaligenes faecalis. J. Bacteriol. 1957;74(3):381–385. doi: 10.1128/jb.74.3.381-385.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Kelly B., Appleman M.D. Degradation of ergothioneine by cell-free extracts of Alcaligenes faecalis. J. Bacteriol. 1961;81:715–720. doi: 10.1128/jb.81.5.715-720.1961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Turck D., Bresson J.L., Burlingame B., Dean T., Fairweather‐Tait S., Heinonen M., Hirsch‐Ernst K.I., Mangelsdorf I., McArdle H.J., Naska A., Neuhäuser‐Berthold M., Nowicka G., Pentieva K., Sanz Y., Siani A., Sjödin A., Stern M., Tomé D., Vinceti M., Willatts P., Engel K.H., Marchelli R., Pöting A., Poulsen M., Schlatter J., Ackerl R., Loveren H.v. Safety of synthetic l‐ergothioneine (Ergoneine®) as a novel food pursuant to Regulation (EC) No 258/97. EFSA J. 2016;14(11) p. report 4629. [Google Scholar]
- 160.Yu Y.H., Pan H.Y., Guo L.Q., Lin J.F., Liao H.L., Li H.Y. Successful biosynthesis of natural antioxidant ergothioneine in Saccharomyces cerevisiae required only two genes from Grifola frondosa. Microb. Cell Factories. 2020;19(1):164. doi: 10.1186/s12934-020-01421-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.van der Hoek S.A., Darbani B., Zugaj K.E., Prabhala B.K., Biron M.B., Randelovic M., Medina J.B., Kell D.B., Borodina I. Engineering the yeast Saccharomyces cerevisiae for the production of L-(+)-Ergothioneine. Front Bioeng Biotechnol. 2019;7:262. doi: 10.3389/fbioe.2019.00262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Fujitani Y., Alamgir K.M., Tani A. Ergothioneine production using Methylobacterium species, yeast, and fungi. J. Biosci. Bioeng. 2018;126(6):715–722. doi: 10.1016/j.jbiosc.2018.05.021. [DOI] [PubMed] [Google Scholar]
- 163.Kamide T., Takusagawa S., Tanaka N., Ogasawara Y., Kawano Y., Ohtsu I., Satoh Y., Dairi T. High production of ergothioneine in Escherichia coli using the sulfoxide synthase from Methylobacterium strains. J. Agric. Food Chem. 2020;68(23):6390–6394. doi: 10.1021/acs.jafc.0c01846. [DOI] [PubMed] [Google Scholar]
- 164.Beliaeva M.A., Burn R., Lim D., Seebeck F.P. In vitro production of ergothioneine isotopologues. Angew Chem. Int. Ed. Engl. 2020 doi: 10.1002/anie.202011096. In press. [DOI] [PubMed] [Google Scholar]
- 165.Cheah I.K., Halliwell B. Could ergothioneine aid in the treatment of coronavirus patients? Antioxidants. 2020;9(7):595. doi: 10.3390/antiox9070595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Akanmu D., Cecchini R., Aruoma O.I., Halliwell B. The antioxidant action of ergothioneine. Arch. Biochem. Biophys. 1991;288(1):10–16. doi: 10.1016/0003-9861(91)90158-f. [DOI] [PubMed] [Google Scholar]
- 167.Aruoma O.I., Whiteman M., England T.G., Halliwell B. Antioxidant action of ergothioneine: assessment of its ability to scavenge peroxynitrite. Biochem. Biophys. Res. Commun. 1997;231(2):389–391. doi: 10.1006/bbrc.1997.6109. [DOI] [PubMed] [Google Scholar]
- 168.Rahman I., Gilmour P.S., Jimenez L.A., Biswas S.K., Antonicelli F., Aruoma O.I. Ergothioneine inhibits oxidative stress- and TNF-alpha-induced NF-kappa B activation and interleukin-8 release in alveolar epithelial cells. Biochem. Biophys. Res. Commun. 2003;302(4):860–864. doi: 10.1016/s0006-291x(03)00224-9. [DOI] [PubMed] [Google Scholar]
- 169.Shimizu T., Masuo Y., Takahashi S., Nakamichi N., Kato Y. Organic cation transporter Octn1-mediated uptake of food-derived antioxidant ergothioneine into infiltrating macrophages during intestinal inflammation in mice. Drug Metabol. Pharmacokinet. 2015;30(3):231–239. doi: 10.1016/j.dmpk.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 170.Cumming B.M., Chinta K.C., Reddy V.P., Steyn A.J.C. Role of ergothioneine in microbial physiology and pathogenesis. Antioxidants Redox Signal. 2018;28(6):431–444. doi: 10.1089/ars.2017.7300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Sheridan K.J., Lechner B.E., Keeffe G.O., Keller M.A., Werner E.R., Lindner H., Jones G.W., Haas H., Doyle S. Ergothioneine biosynthesis and functionality in the opportunistic fungal pathogen, Aspergillus fumigatus. Sci. Rep. 2016;6:35306. doi: 10.1038/srep35306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Clerkin C.G., Matzinger M., Selo M.A., Talbot B.N., Walsh J.J., Ehrhardt C. Aromatic substitution reaction of ergothioneine - a novel mechanism of xenobiotic deactivation. FASEB J. 2017;31(S1) 821.1. [Google Scholar]
- 173.Encarnacion A.B., Fagutao F., Hirono I., Ushio H., Ohshima T. Effects of ergothioneine from mushrooms (Flammulina velutipes) on melanosis and lipid oxidation of kuruma shrimp (Marsupenaeus japonicus) J. Agric. Food Chem. 2010;58(4):2577–2585. doi: 10.1021/jf903944y. [DOI] [PubMed] [Google Scholar]
- 174.Bao H.N., Ushio H., Ohshima T. Antioxidative activity and antidiscoloration efficacy of ergothioneine in mushroom (Flammulina velutipes) extract added to beef and fish meats. J. Agric. Food Chem. 2008;56(21):10032–10040. doi: 10.1021/jf8017063. [DOI] [PubMed] [Google Scholar]
- 175.Fornasaro S., Gurian E., Pagarin S., Genova E., Stocco G., Decorti G., Sergo V., Bonifacio A. Ergothioneine, a dietary amino acid with a high relevance for the interpretation of label-free surface enhanced Raman scattering (SERS) spectra of many biological samples. Spectrochim. Acta Mol. Biomol. Spectrosc. 2020;246:119024. doi: 10.1016/j.saa.2020.119024. [DOI] [PubMed] [Google Scholar]
- 176.Seok A.E., Lee J., Lee Y.R., Lee Y.J., Kim H.J., Ihm C., Sung H.J., Hyun S.H., Kang H.G. Estimation of age of bloodstains by mass-spectrometry: a metabolomic approach. Anal. Chem. 2018;90(21):12431–12441. doi: 10.1021/acs.analchem.8b01367. [DOI] [PubMed] [Google Scholar]
- 177.Uhlen M., Fagerberg L., Hallstrom B.M., Lindskog C., Oksvold P., Mardinoglu A., Sivertsson A., Kampf C., Sjostedt E., Asplund A., Olsson I., Edlund K., Lundberg E., Navani S., Szigyarto C.A., Odeberg J., Djureinovic D., Takanen J.O., Hober S., Alm T., Edqvist P.H., Berling H., Tegel H., Mulder J., Rockberg J., Nilsson P., Schwenk J.M., Hamsten M., von Feilitzen K., Forsberg M., Persson L., Johansson F., Zwahlen M., von Heijne G., Nielsen J., Ponten F. Proteomics. Tissue-based map of the human proteome. Science. 2015;347(6220):1260419. doi: 10.1126/science.1260419. [DOI] [PubMed] [Google Scholar]