Abstract
Introduction
Tyrosine kinase (TKI) and checkpoint inhibitors (CI) prolonged overall survival in metastatic renal cell carcinoma (mRCC). Early prediction of treatment response is highly desirable for the individualization of patient management and improvement of therapeutic outcome; however, serum biochemistry is unable to predict therapeutic efficacy. Therefore, we compared 18F-PSMA-1007 PET imaging for response assessment in mRCC patients undergoing TKI or CI therapy compared to CT-based response assessment as the current imaging reference standard.
Methods
18F-PSMA-1007 PET/CT was performed in mRCC patients prior to initiation of systemic treatment and 8 weeks after therapy initiation. Treatment response was evaluated separately on 18F-PSMA-PET and CT. Changes on PSMA-PET (SUVmean) were assessed on a per patient basis using a modified PERCIST scoring system. Complete response (CRPET) was defined as absence of any uptake in all target lesions on posttreatment PET. Partial response (PRPET) was defined as decrease in summed SUVmean of > 30%. The appearance of new, PET-positive lesions or an increase in summed SUVmean of > 30% was defined as progressive disease (PDPET). A change in summed SUVmean of ± 30% defined stable disease (SDPET). RECIST 1.1 criteria were used for response assessment on CT. Results of radiographic response assessment on PSMA-PET and CT were compared.
Results
Overall, 11 mRCC patients undergoing systemic treatment were included. At baseline PSMA-PET1, all mRCC patients showed at least one PSMA-avid lesion. On follow-up PET2, 3 patients showed CRPET, 3 PRPET, 4 SDPET, and 1 PDPET. According to RECIST 1.1, 1 patient showed PRCT, 9 SDCT, and 1 PDCT. Overall, concordant classifications were found in only 2 cases (2 SDCT + PET). Patients with CRPET on PET were classified as 3 SDCT on CT using RECIST 1.1. By contrast, the patient classified as PRCT on CT showed PSMA uptake without major changes during therapy (SDPET). However, among 9 patients with SDCT on CT, 3 were classified as CRPET, 3 as PRPET, 1 as PDPET, and only 2 as SDPET on PSMA-PET.
Conclusion
On PSMA-PET, heterogeneous courses were observed during systemic treatment in mRCC patients with highly diverging results compared to RECIST 1.1. In the light of missing biomarkers for early response assessment, PSMA-PET might allow more precise response assessment to systemic treatment, especially in patients classified as SD on CT.
Keywords: Metastatic renal cell carcinoma, 18F-PSMA-1007 PET, CT, Response assessment, Tyrosine kinase therapy, Checkpoint inhibitor therapy
Introduction
Tyrosine kinase inhibitors (TKIs) and checkpoint inhibitors (CIs) significantly prolong survival in mRCC patients [1–3]. Early prediction of treatment response is highly desirable for individualization of patient management and improvement of outcome. However, established predictive biomarkers for response assessment are lacking [4, 5]. Currently, criteria-based reporting for response assessment relies on morphological imaging criteria such as RECIST 1.1. Unlike most other malignancies, the application of 18F-FDG PET/CT in RCC is limited by its low FDG-avidity [6]. Although preliminary data have indicated a potential role of 18F-FDG PET/CT for treatment monitoring of nivolumab in RCC patients [7], discordant published data lead to a missing recommendation in current guidelines [8]. PSMA is increasingly recognized in prostate cancer imaging [9]. Moreover, PSMA is highly expressed on the cell surface of the tumor microvasculature of several solid tumors [10, 11]. Initial data showed promising results for PSMA-targeted PET imaging in mRCC and might improve diagnostic accuracy [10, 12–15].
We hypothesized that PSMA expression as a tumoral feature of RCC changes under TKI or CIs therapy and that 18F-PSMA-1007 PET provides pathophysiological information beyond morphological extent on CT. We therefore compared 18F-PSMA-1007 PET using modified PERCIST criteria to CT response based on RECIST 1.1 in mRCC patients undergoing TKI or CI therapy.
Methods
Inclusion criteria
This analysis was approved by the institutional ethics committee of the LMU Munich (IRB# 20-315). Criteria for inclusion were (1) histologically proven mRCC, (2) therapy with TKI or CI, (3) 18F-PSMA-1007 PET/CT prior to therapy with TKI or CI, and (4) follow-up 18F-PSMA-1007 PET/CT 8 weeks after therapy initiation.
Radiopharmaceutical and imaging protocol
A median activity of 246 MBq (range 217–268 MBq) 18F-PSMA-1007 was injected intravenously in line with previously reported radiosynthesis and administration procedures [16]. Additionally, the patients were premedicated with furosemide (20 mg) if no contraindication was given [17]. The radiopharmaceutical was used on an individual patient basis according to German Pharmaceuticals Act §13(2b). PET was performed from the skull base to the mid-thigh using a Biograph mCT scanner or a Biograph 64 PET/CT scanner (Siemens Healthineers Erlangen, Germany) 60 min after tracer injection. PET/CT included a diagnostic, contrast-enhanced CT scan in the portal–venous phase (Imeron 350; 1.5 ml/kg body weight; Bracco Imaging, Milano, Italy). PET was acquired with 2.5 min per bed position and reconstructed iteratively using TrueX (three iterations, 21 subsets) with Gaussian postreconstruction smoothing (2 mm full-width at half-maximum).
Radiographic therapy response assessment
Radiographic treatment response was separately assessed on 18F-PSMA-1007 PET and CT datasets. For 18F-PSMA-1007 PET analysis, images were analyzed independently by two experienced nuclear medicine physicians (MU, HI) on a dedicated workstation (Hermes Hybrid 3D Viewer, Hermes Medical Solutions, Stockholm, Sweden).
18F-PSMA-1007 PET
Transaxial PET slices were used for image analysis as described previously [18]. Five organ systems were included per patient comprising lymph nodes, bone, affected kidney/kidney bed, and other visceral metastatic sites. Any focal uptake of 18F-PSMA-1007 higher than the surrounding background not associated with physiological uptake was considered suspicious for malignancy. For each organ system, the two lesions with the highest 18F-PSMA-1007 uptake were analyzed on PET1 (PET1 = PET prior to therapy initiation). For quantitative analysis, the slice with the maximum 18F-PSMA-1007 was identified using an isocontour volume of interest (VOI) including all voxels above 99% of the maximum covering the whole lesion volume. In a second step, a spherical VOI with a diameter of 1.5 cm was placed over the tumor lesion centering in the slice with the maximum 18F-PSMA-1007 uptake, and the mean standardized uptake volume (SUVmean) was noted. PET2 (PET2 = PET 8 weeks after initiation) findings were compared to PET1.
Posttreatment changes were interpreted according to modified PET Response Criteria in Solid tumors (PERCIST) 1.0 [18]. The absence of any PSMA-uptake on PET2 was defined as molecular complete response (CRPET). A decrease in summed SUVmean of ≥ 30% was considered PRPET. The appearance of new PET-positive lesions on PET2 or an increase in summed SUVmean of ≥ 30% was considered progressive disease (PDPET). An intermediate change in summed SUVmean between − 30 and + 30% without new target lesions was considered stable disease (SDPET).
CT (RECIST 1.1)
For evaluation of CT datasets, response assessment was performed by two experienced radiologists (WGK, CB) according to RECIST 1.1 using a dedicated software (mint lesion™, version 3.0.1, Mint Medical GmbH, Dossenheim, Germany) [18, 19]. Target and nontarget lesions were defined and measured in baseline CT prior to therapy initiation (CT1). In the follow-up CT examination 8 weeks after initiation, target lesions were located and manually measured (CT2). Disappearance of all lesions was considered complete response (CRCT); a decrease in summed diameters of ≥ 30% was defined as partial response (PRCT). The appearance of a new target lesion on CT2 or an increase in the summed diameters of ≥ 20% with an absolute increase of at least 5 mm was defined as progressive disease (PDCT). An intermediate change in summed diameter between − 30% and + 20% without appearance of a new target lesion was considered stable disease (SDCT).
Statistical analysis
Statistical analyses were performed with IBM SPSS® Statistics (version 25, IBM Corp., Armonk, NY). Descriptive statistics are displayed as median (range) or mean ± standard deviation (SD). Relative changes during therapy are displayed as percentage differences.
Results
Patients and treatment regimen
Eleven mRCC patients were included in this analysis (mean age 59.6 years (range 24.4–78.4 years; 8 male/3 female). Patients underwent 18F-PSMA-1007 PET/CT directly before undergoing therapy with TKI or CI and 8 weeks after therapy initiation. 7/11 (63.6%) patients underwent TKI therapy (2x cabozantinib, 3x sunitinib, 1x axitinib, and 1x levantinib + everolimus), 4/11 (36.4%) patients underwent CI therapy (2x ipilimumab + nivolumab, 1x nivolumab, and 1x pembrolizumab) using standard dosages without dose reduction during follow-up. Baseline characteristics are presented in Table 1.
Table 1.
Baseline characteristics and comparison between radiographic response on 18F-PSMA-1007 and CT
| Patient | Age | Sex | Histology | Tumor localization° | Therapy | Δ SUVmean (%) | Response on PET | Δ RECIST (%) | Response on CT |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 48.0 | F | ccRCC | LN, VO | Cabozantinib | n.e. | CR | − 13.2% | SD |
| 2 | 77.1 | F | ccRCC | K, LN, VO, B |
Ipilimumab Nivolumab |
− 12.2% | PD* | − 29.1% | SD |
| 3 | 74.8 | M | ccRCC | K, LN, VO, B |
Levantinib Everolimus |
− 28.7% | SD | − 7.2% | SD |
| 4 | 70.5 | M | pRCC | K, LN, VO, B | Sunitinib | − 44.7% | PR | − 1.5% | SD |
| 5 | 52.9 | F | ccRCC | VO | Cabozantinib | n.e. | CR | 1.8% | SD |
| 6 | 70.8 | M | ccRCC | VO, LN, B | Sunitinib | − 68.8% | PR | − 18.5% | SD |
| 7 | 44.9 | M | ccRCC | VO | Axitinib | n.e. | CR | − 26.2% | SD |
| 8 | 24.4 | M | pRCC | K, LN, VO | Nivolumab | − 9.9% | SD | 24.8% | PD |
| 9 | 42.8 | M | uRCC | K, LN, VO |
Ipilimumab Nivolumab |
− 14.2% | SD | − 35.5% | PR |
| 10 | 73.7 | M | ccRCC | K, VO, B | Pembrolizumab | − 35.9% | PR | − 18.3% | SD |
| 11 | 78.4 | M | ccRCC | K, VO | Sunitinib | − 28.1% | SD | − 18.9% | SD |
°As defined on 18F-PSMA-1007. *PD because of new lesions on PET2. f female, m male, ccRCC clear cell renal cell carcinoma, pRCC papillary renal cell carcinoma, uRCC undifferentiated renal cell carcinoma, K kidney, LN lymph nodes, VO visceral organs, B bone, n.e. not evaluable, PD progressive disease, PR partial response, SD stable disease
Response assessment
PET-based response assessment
Three of 11 (27.2%) patients showed CRPET with an absence of any PSMA uptake on PET2. Three of 11 (27.2%) showed PRPET with a decrease in summed SUVmean of ≥ 30%; in 4/11 patients (36.4%), an intermediate change in summed SUVmean between − 30% and + 30% without appearance of a new target lesion (SDPET) was seen. One of 11 patients (9.1%) presented with a new, PET-positive target lesion and was defined as PDPET (Fig. 1).
Fig. 1.
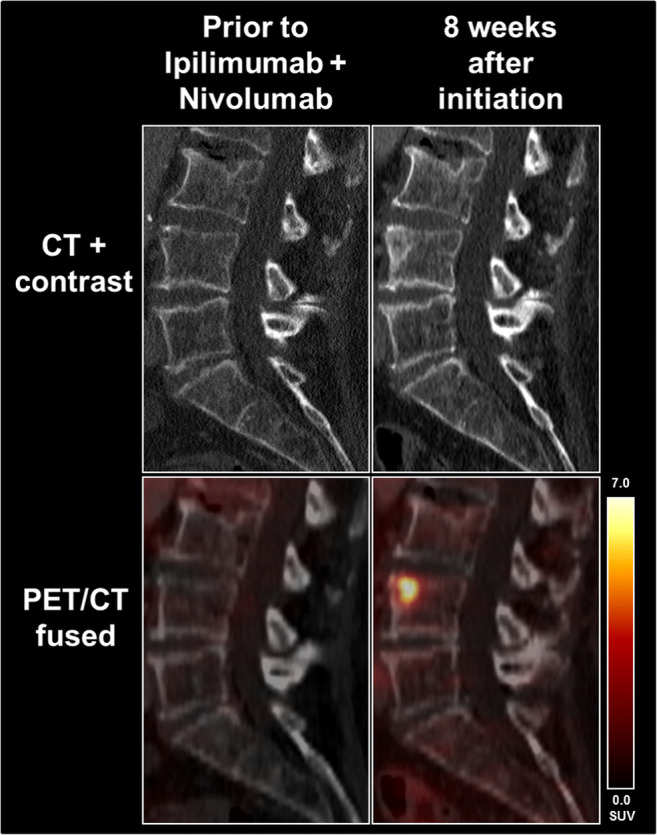
A 77-year-old female patient showed a new osteoblastic lesion on follow-up CT during therapy with Ipilimumab and Nivolumab. According to RECIST 1.1, this is not rated as PD. However, a high PSMA expression could be seen on PET indicating this lesion to be a vital metastasis rather than an avital osteoblastic reaction to therapy. Consequently, this was rated PDPET, although the other tumoral lesions showed stable uptake on PET
CT-based response assessment
When analyzing the CT-based response assessment using RECIST 1.1, 1/11 (9.1%) patient showed PRCT with a decrease in summed diameters of ≥ 30% (− 35.5%), 9/11 (81.8%) of the patients showed SDCT with an intermediate change in summed diameter between − 30% and + 20% without appearance of any new target lesion, and 1/11 (9.1%) patients had PDCT with an increase in the summed diameters of ≥ 20% with an absolute increase of at least 5 mm.
Concordance of PET- and CT-based response assessment
Overall, concordant results between PET and CT response assessments could only be obtained in 2/11 (18.2%) patients, presenting with SD both on PET and CT (2 SDCT + PET). Three patients with CRPET were classified as SDCT on CT, whereas no patient showed CRCT.
By contrast, 1 patient classified as PRCT on CT showed PSMA uptake without major changes during therapy (SDPET). However, among 9 patients with SDCT on CT, 3 were classified as CRPET, 3 as PRPET, 1 as PDPET, and only 2 as SDPET on PSMA-PET. Concordance between radiographic responses on PET and CT are presented in Table 2.
Table 2.
Concordance between radiographic response on PET and CT
| Response PET | Response CT | ||||
|---|---|---|---|---|---|
| Progressive disease | Stable disease | Partial response | Complete response | Total | |
| Progressive disease | 0 | 1* | 0 | 0 | 1 |
| Stable disease | 1° | 2 | 1* | 0 | 4 |
| Partial response | 0 | 3° | 0 | 0 | 3 |
| Complete response | 0 | 3° | 0 | 0 | 3 |
| Total | 1 | 9 | 1 | 0 | 11 |
*Better response on CT. °Better response on PET
Discussion
Our data demonstrate a change of PSMA-PET expression during systemic therapy of mRCC in the majority of patients; even a complete remission of PSMA-expression was observed in 3/11 patients (27.2%) despite remaining tumor mass with SD on CT (Fig. 2). Interestingly, the evaluated PET response assessment using PERCIST criteria showed vast discrepancies to morphological response assessment using RECIST 1.1. Only 2/11 patients comprised a concordant finding on PET and CT, whereas 9/11 patients (81.8%) showed in parts highly diverging classifications on PSMA-PET and CT. 6/11 patients (54.5%) showed CR or PR on PET and SD using RECIST 1.1. This result suggests that 18F-PSMA-1007 PET may be able to assess treatment response on a molecular level earlier than morphological changes on standard imaging (Fig. 2) with potential adjustments of the treatment regimen. These findings underline current data, which could show that PSMA-PET is advantageous over standard imaging with CT alone in mRCC, particularly for the identification of small lesions such as lymph nodes [14]. This additional pathophysiological information beyond CT morphology could also lead to a decision of continuing or changing current therapy or to de-escalate therapy in order to reduce drug-related side effects [3].
Fig. 2.
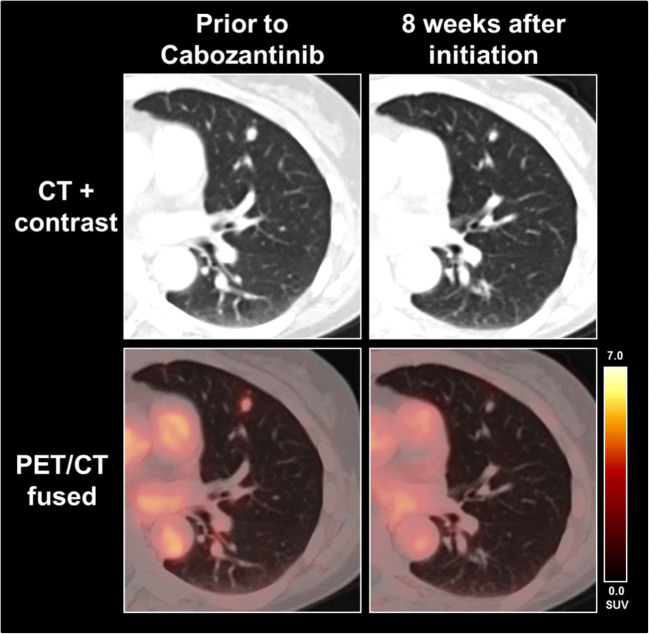
A 53-year-old female patient showed a slightly decreasing pulmonary metastasis, which, however, completely lost PSMA expression during therapy with cabozantinib
Conversely, we also observed changes towards progression on PET with one patient showing PD on PET, but SD on CT. Here, new osteoblastic lesions in vertebra T7 and L4 with focally increased PSMA uptake (Fig. 1) were identified. According to RECIST 1.1, osteoblastic metastases are non-measurable lesions, as they can be seen as a potential sign of treatment response, when changing from lytic to blastic [20]. Therefore, a distinction of vital bone metastases and bone metastases with therapy response remains highly challenging using morphological imaging with CT [21, 22]. Here, PSMA-PET could potentially add relevant clinical information with regard to the response assessment of osseous lesions (Fig. 1).
Also, the scenario of PD on CT, but SD on PET could be observed in the current cohort. It is known that pseudoprogression can occur in patients undergoing immunotherapy [23] leading to an early enlargement of tumor manifestations as part of the treatment effect during the early phases followed by a subsequent shrinkage of tumor lesions [24]. Using RECIST 1.1, this phenomenon would directly lead to the classification of PD. To overcome these limitations of RECIST 1.1, several modified response criteria were suggested. For example, using iRECIST, this phenomenon leads to the classification of immune unconfirmed progressive disease (iUPD) [25], which would lead to an additional earlier follow-up CT scan to confirm either true progression or pseudoprogression during ongoing immunotherapy. In this scenario, 18F-PSMA-1007 PET could contribute in the early identification of pseudoprogression and real progression in mRCC patients undergoing immunotherapy.
One major limitation is the small number of patients as well as the retrospective design of the study. According to Seitz et al., we adapted the PERCIST 1.0 criteria [18, 26] for defining the response categories on PSMA-PET. Although this modified approach has been shown to be feasible for PSMA-PET in published studies [18], a prospective validation including endpoints such as overall survival is mandatory to further investigate the use of 18F-PSMA-1007 PET for response assessment. Within this process, exact cut-off values on PSMA-PET for the accurate prediction of treatment response in terms of overall survival are yet to be defined. Additionally, new response criteria for immunotherapy monitoring such as ‘PET/CT Criteria for early prediction of Response to Immune checkpoint inhibitor Therapy’ (PECRIT) and ‘PET Response Evaluation Criteria for Immunotherapy’ (PERCIMT) that link RECIST 1.1 and PERCIST 1.0 were recently introduced [27, 28]; these particular specifications of response assessment should also be evaluated in mRCC patients undergoing PSMA-PET/CT and be correlated with the clinical outcome in order to evaluate the best predictive factors on PSMA-PET.
Nonetheless, our preliminary results provide support to the hypothesis that 18F-PSMA-1007 PET and its combination with CT provides complementary information on a molecular level for response assessment in mRCC patients undergoing systemic treatment with TKI or CI.
Conclusion
On PSMA-PET, heterogeneous courses were observed during systemic treatment in mRCC patients with highly diverging results compared to RECIST 1.1 in mRCC patients undergoing systemic treatment with TKI or CI. Hence, hybrid imaging may optimize response assessment of mRCC patients and influence patient management. In the light of missing biomarkers for early response assessment, PSMA-PET might allow more precise response assessment to systemic treatment, especially in those patients classified as stable disease on CT. Data in correlation with clinical outcome parameters are underway.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Compliance with ethical standards
Conflict of interest
M.S.: Consultant: Pfizer, GlaxoSmithKline, Novartis, Bayer, Roche, Aveo, EUSAPharm, Astellas, Ipsen, Exelixis, Pelloton, EISAI, BMS, MSD; honoraria: Pfizer, GlaxoSmithKline, AVEO, Novartis, Bayer, EUSAPharm, Astellas, Ipsen, Exelixis, Pelloton, EISAI, BMS, MSD; research funding: Pfizer, GlaxoSmithKline, AVEO, BMS, Novartis, Bayer, Roche/Genentech, Immatics, Wilex, Ipsen, Exelixis, EISAI. H.I.: Honoraria: Bayer, Sirtex Medical, Novartis. N.L.A: member of the EANM brain imaging committee. W.G.K.: member of the Imaging Group of the EORTC. All other authors declare that they have no relationships or interests that could have direct or potential influence or impart bias on the work.
Ethical approval
The analysis was authorized by the local ethics committee in accordance with the ICH Guideline for Good Clinical Practice (GCP) and the declaration of Helsinki.
Informed consent
All patients gave written consent to undergo PET/CT scans as part of the clinical routine. The radiopharmaceutical was used on an individual patient basis according to German Pharmaceuticals Act §13(2b).
Footnotes
This article is part of the Topical Collection on Oncology - General
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
L. M. Mittlmeier, M. Unterrainer, H. Ilhan and M. Staehler contributed equally to this work.
References
- 1.Choueiri TK, Motzer RJ. Systemic therapy for metastatic renal-cell carcinoma. N Engl J Med. 2017;376:354–366. doi: 10.1056/NEJMra1601333. [DOI] [PubMed] [Google Scholar]
- 2.Reed JP, Posadas EM, Figlin RA. Developments in the use of tyrosine kinase inhibitors in the treatment of renal cell carcinoma. Expert Rev Anticancer Ther. 2019;19:259–271. doi: 10.1080/14737140.2019.1573678. [DOI] [PubMed] [Google Scholar]
- 3.Alsharedi M, Katz H. Check point inhibitors a new era in renal cell carcinoma treatment. Med Oncol. 2018;35:85. doi: 10.1007/s12032-018-1147-y. [DOI] [PubMed] [Google Scholar]
- 4.Heinemann FG, Tolkach Y, Deng M, Schmidt D, Perner S, Kristiansen G, et al. Serum miR-122-5p and miR-206 expression: non-invasive prognostic biomarkers for renal cell carcinoma. Clin Epigenetics. 2018;10:11. doi: 10.1186/s13148-018-0444-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Raimondi A, Sepe P, Claps M, Verzoni E, Procopio G. Do biomarkers play a predictive role for response to novel immunotherapeutic agents in metastatic renal cell carcinoma? : Taylor & Francis; 2019. [DOI] [PubMed]
- 6.Liu Y. The place of FDG PET/CT in renal cell carcinoma: value and limitations. Front Oncol. 2016;6:201. doi: 10.3389/fonc.2016.00201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tabei T, Nakaigawa N, Kaneta T, Ikeda I, Kondo K, Makiyama K, et al. Early assessment with 18 F-2-fluoro-2-deoxyglucose positron emission tomography/computed tomography to predict short-term outcome in clear cell renal carcinoma treated with nivolumab. BMC Cancer. 2019;19:298. doi: 10.1186/s12885-019-5510-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ljungberg B, Albiges L, Abu-Ghanem Y, Bensalah K, Dabestani S, Fernández-Pello S, et al. European association of urology guidelines on renal cell carcinoma: the 2019 update. Eur Urol. 2019;75:799–810. doi: 10.1016/j.eururo.2019.02.011. [DOI] [PubMed] [Google Scholar]
- 9.Fendler WP, Eiber M, Beheshti M, Bomanji J, Ceci F, Cho S, et al. 68 Ga-PSMA PET/CT: joint EANM and SNMMI procedure guideline for prostate cancer imaging: version 1.0. Eur J Nucl Med Mol Imaging. 2017;44:1014–1024. doi: 10.1007/s00259-017-3670-z. [DOI] [PubMed] [Google Scholar]
- 10.Siva S, Callahan J, Pryor D, Martin J, Lawrentschuk N, Hofman MS. Utility of 68Ga prostate specific membrane antigen–positron emission tomography in diagnosis and response assessment of recurrent renal cell carcinoma. Journal of medical imaging and radiation oncology. 2017;61:372–378. doi: 10.1111/1754-9485.12590. [DOI] [PubMed] [Google Scholar]
- 11.Evangelista L, Basso U, Maruzzo M, Novara G. The role of radiolabeled prostate-specific membrane antigen positron emission tomography/computed tomography for the evaluation of renal cancer. European Urology Focus. 2018. [DOI] [PubMed]
- 12.Mittlmeier L, Unterrainer M, Todica A, Cyran C, Rodler S, Bartenstein P, et al. PSMA PET/CT for tyrosine-kinase inhibitor monitoring in metastatic renal cell carcinoma. Eur J Nucl Med Mol Imaging. 2020:1–2. [DOI] [PubMed]
- 13.Meyer AR, Carducci MA, Denmeade SR, Markowski MC, Pomper MG, Pierorazio PM, et al. Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted 18 F-DCFPyL PET/CT. Ann Nucl Med. 2019;33:617–623. doi: 10.1007/s12149-019-01371-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rhee H, Blazak J, Tham CM, Ng KL, Shepherd B, Lawson M, et al. Pilot study: use of gallium-68 PSMA PET for detection of metastatic lesions in patients with renal tumour. EJNMMI Res. 2016;6:1–6. doi: 10.1186/s13550-016-0231-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marafi F, Sasikumar A, Al-Terki A, Alfeeli M. 18F-PSMA 1007 in suspected renal cell carcinoma. Clin Nucl Med. 2020. [DOI] [PubMed]
- 16.Cardinale J, Schäfer M, Benešová M, Bauder-Wüst U, Leotta K, Eder M, et al. Preclinical evaluation of 18F-PSMA-1007, a new prostate-specific membrane antigen ligand for prostate cancer imaging. J Nucl Med. 2017;58:425–431. doi: 10.2967/jnumed.116.181768. [DOI] [PubMed] [Google Scholar]
- 17.d'Amico A, Gorczewska I, Gorczewski K, Turska-d'Amico M, Di Pietro M. Effect of furosemide administration before F-18 fluorodeoxyglucose positron emission tomography/computed tomography on urine radioactivity and detection of uterine cervical cancer. Nuclear Medicine Review. 2014;17:83–86. doi: 10.5603/NMR.2014.0022. [DOI] [PubMed] [Google Scholar]
- 18.Seitz AK, Rauscher I, Haller B, Krönke M, Luther S, Heck MM, et al. Preliminary results on response assessment using 68 Ga-HBED-CC-PSMA PET/CT in patients with metastatic prostate cancer undergoing docetaxel chemotherapy. Eur J Nucl Med Mol Imaging. 2018;45:602–612. doi: 10.1007/s00259-017-3887-x. [DOI] [PubMed] [Google Scholar]
- 19.Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1) European journal of cancer. 2009;45:228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 20.Schwartz LH, Seymour L, Litière S, Ford R, Gwyther S, Mandrekar S, et al. RECIST 1.1–standardisation and disease-specific adaptations: perspectives from the RECIST Working Group. Eur J Cancer. 2016;62:138–145. doi: 10.1016/j.ejca.2016.03.082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Schwartz LH, Litière S, de Vries E, Ford R, Gwyther S, Mandrekar S, et al. RECIST 1.1—update and clarification: from the RECIST committee. Eur J Cancer. 2016;62:132–137. doi: 10.1016/j.ejca.2016.03.081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Henze J, Maintz D, Persigehl T. RECIST 1.1, irRECIST 1.1, and mRECIST: how to do. Current Radiology Reports. 2016;4:48. doi: 10.1007/s40134-016-0178-4. [DOI] [Google Scholar]
- 23.Elias R, Kapur P, Pedrosa I, Brugarolas J. Renal cell carcinoma pseudoprogression with clinical deterioration: to hospice and back. Clinical genitourinary cancer. 2018;16:485–488. doi: 10.1016/j.clgc.2018.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Persigehl T, Lennartz S, Schwartz LH. iRECIST: how to do it. Cancer Imaging. 2020;20:1–7. doi: 10.1186/s40644-019-0281-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Seymour L, Bogaerts J, Perrone A, Ford R, Schwartz LH, Mandrekar S, et al. iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. The Lancet Oncology. 2017;18:e143–ee52. doi: 10.1016/S1470-2045(17)30074-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. Journal of nuclear medicine: official publication, Society of Nuclear Medicine. 2009;50:122S. [DOI] [PMC free article] [PubMed]
- 27.Aide N, Hicks RJ, Le Tourneau C, Lheureux S, Fanti S, Lopci E. FDG PET/CT for assessing tumour response to immunotherapy. Eur J Nucl Med Mol Imaging. 2019;46:238–250. doi: 10.1007/s00259-018-4171-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cho SY, Lipson EJ, Im H-J, Rowe SP, Gonzalez EM, Blackford A, et al. Prediction of response to immune checkpoint inhibitor therapy using early-time-point 18F-FDG PET/CT imaging in patients with advanced melanoma. J Nucl Med. 2017;58:1421–1428. doi: 10.2967/jnumed.116.188839. [DOI] [PMC free article] [PubMed] [Google Scholar]