Abstract
Heads-up three-dimensional (3D) surgical visualization systems allow ophthalmic surgeons to replace surgical microscope eyepieces with high-resolution stereoscopic cameras transmitting an image to a screen. We investigated the effectiveness and safety of the heads-up NGENUITY 3D Visualization System in a retrospective evaluation of 241 consecutive vitreoretinal surgeries performed by the same surgeon using conventional microscopy (CM group) over a 1-year period versus the NGENUITY System (3D group) over a consecutive 1-year period. We included for study vitreoretinal surgeries for treatment of retinal detachment (RD) (98 surgeries), macular hole (MH) (48 surgeries), or epiretinal membrane (ERM) (95 surgeries). A total of 138 and 103 eyes were divided into 3D and CM groups, respectively. We found no differences in 3-month postoperative rates of recurrence of RD (10% versus 18%, p = 0.42), MH closure (82% versus 88%, p = 0.69), or decrease in central macular thickness of ERMs (134 ± 188 µm versus 115 ± 105 µm, p = 0.57) between the 3D and CM groups, respectively. Surgery durations and visual prognosis were also similar between both groups. We consolidate that the NGENUITY System is comparable in terms of visual and anatomical outcomes, giving it perspectives for integration into future robotized intervention.
Subject terms: Medical research, Outcomes research, Eye diseases
Introduction
The first fixed surgical microscopes arrived in the 1920s with Nylen, a Swedish ear, nose, and throat specialist, but it was not until 1946 that Perritt from Chicago made use of one for ophthalmology surgery1–6. Barraquer4 testified in 1980 that microscopes had made it possible to not only visualize previously inaccessible eye structures, but also to develop more precise surgical techniques, smaller instruments, and finer suture materials. Indeed, the advent of phacoemulsification by Kelman7, pars plana vitrectomy by Machemer et al.8, and non-perforating filtration surgery by Krasnov9 was during the 1960s and 1970s. The contribution of these techniques was considerable to the treatment of many diseases. Today vitrectomy is considered the reference technique for the surgical treatment of macular hole (MH) and epiretinal membrane (ERM), and it also harbors a wide range of indications for retinal detachment (RD)10,11.
Heads-up three-dimensional (3D) surgical visualization systems allow ophthalmic surgeons to free themselves of the eyepieces of conventional surgical microscopes, and to replace them by high-resolution dual-camera systems that retransmit an image on a screen in front of the surgeon. This switch to all-digital technology represents a major breakthrough in the conception of surgical microscopes in ophthalmology. There are now three main commercial models: the Alcon NGENUITY 3D Visualization System (Alcon Laboratories, Fort Worth, TX), the TrueVision 3D Visualization System (Leica, Wetzlar, Germany), and more recently the ARTEVO 800 system (Zeiss, Oberkochen, Germany).
Published studies have already reported on the use of these technologies in vitreoretinal surgery12–18, but only a few studies have included large series of patients19,20. The aim of this study was therefore to evaluate the effectiveness and safety of the NGENUITY System after 1 year of continuous use in our current practice by the same surgeon. We performed a retrospective evaluation of 241 consecutive vitreoretinal surgeries performed using conventional microscopy over a 1-year time period versus the NGENUITY System over a consecutive 1-year time period. We also describe the benefits and drawbacks of using the NGENUITY System according to both our experience and reports in the literature.
Materials and methods
Study design
We conducted a retrospective, descriptive, comparative study in our ophthalmology department in Toulouse University Hospital (Occitanie, France) over a 2-year time period. We compared two different patient series consecutively operated on between the 29th May 2017 and 28th May 2018 or the 29th May 2018–27th May 2019 using either a conventional microscope (the CM group) or the NGENUITY System (the 3D group), respectively. Patients were thus separated into these two groups solely based on consecutive inclusion without prior comparison of demographic or anatomical characteristics. An official waiver of ethical approval was granted from the IRB of Toulouse University Hospital given the retrospective nature of the study as asserted by French Jardé law. All the procedures performed were part of routine care, and both in accordance with institutional guidelines and with the principles and regulations of the Declaration of Helsinki. Informed patient consent was obtained from participants or their relatives accordingly. The authors affirm that healthcare staff present in the images of Figs. 1 and 2 have given informed consent for publication.
Figure 1.
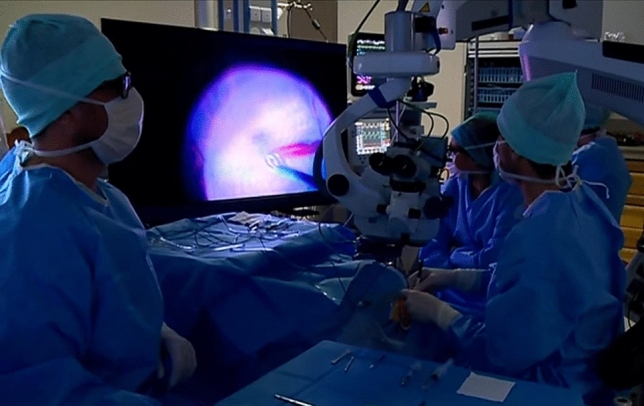
Our operating room configuration with the heads-up NGENUITY 3D Visualization System. The microscope eyepieces were left in place for the conventional microscopy (CM) group and replaced by the three dimensional (3D) camera system for the 3D group with the NGENUITY v1.2.9 software version.
Figure 2.
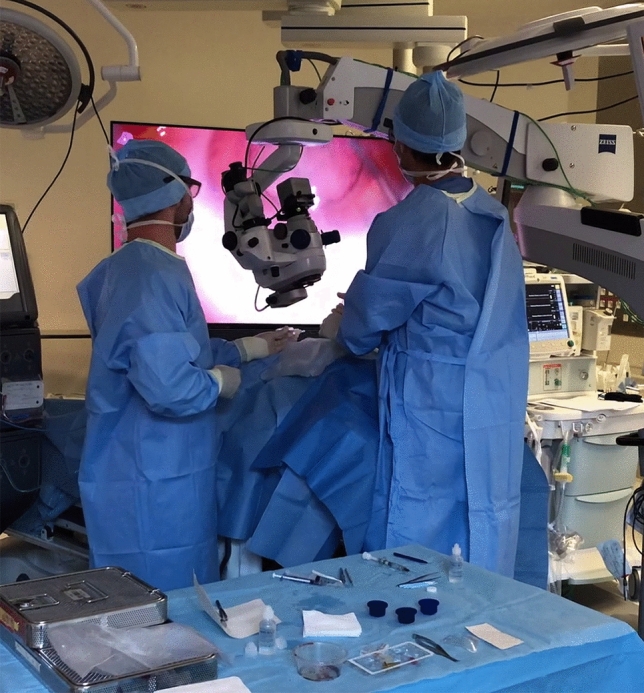
Use of the heads-up NGENUITY 3D Visualization System with a patient in semi-sitting position in our center. Patients who can only be operated on in a semi-sitting position can be operated on by the surgeon standing.
Patient selection
Patients over the age of 18 years old and having undergone vitrectomy or scleral buckling for the surgical treatment of rhegmatogenous retinal detachment, full-thickness MH, or ERM by the same experienced vitreoretinal surgeon (Vincent Soler MD, PhD) were included for study. Patients with exudative retinal detachment, tractional retinal detachments, and retinal detachment secondary to an open globe injury or secondary to MH were excluded.
Surgical procedure
The choice of anesthesia type was left to the anesthetist’s discretion between general or locoregional peribulbar anesthesia with 10-min balloon compression. Three-port 25- or 27-gauge pars plana vitrectomy was performed with a CONSTELLATION Vision System (Alcon Laboratories, Fort Worth, TX, USA) using the manufacturer’s recommended aperture diaphragm of approximately 1/3 in order to optimize visualization while limiting retinal light exposure. Endoillumination levels were set at the beginning of the surgeries to 25% and 40% of maximum output for patients in the 3D group and CM group, respectively. These levels were adjusted when necessary to optimize retinal visualization. In the event of combined surgery, a classical phacoemulsification was performed in the first surgical step and a hydrophobic acrylic monofocal implant (ARTIS PL Cristalens, Lannion, France) was placed in the capsular bag. The peripheral retina was checked before trocar removal and sclerotomies were sutured if necessary after checking for leaks. All procedures were performed using an OPMI LUMERA 700 surgical microscope and a non-contact wide-angle RESIGHT Viewing System (Zeiss, Oberkochen, Germany). The microscope eyepieces were left in place for the CM group and replaced by the NGENUITY 3D camera system for the 3D group (refer to Fig. 1) with the NGENUITY v1.2.9 software version. The system had already been used for surgery in a test phase by the vitreoretinal surgeon on 60 patients before patient inclusion for study. We were thus able to include patients for study without the risk of learning bias as soon as the equipment was acquired. We did not use intraoperative OCT nor color filters.
Data collection
The computerized surgical records obtained from our operating theatre management software (Centricity Opera, GE Healthcare, Chicago, USA) made it possible to recover all patient identities, as well as the surgery durations recorded by the different operating room staff present during the different surgeries. All staff received the same instructions; start the time at the start of surgery, stop the timer after dressing finalization. Details on the surgery performed, preoperative characteristics, and follow-up were collected from these computerized reports. All data has been anonymized for publication purposes.
Evaluation of effectiveness and safety
The primary endpoints used for the analysis and comparison of effectiveness and safety were based on anatomical outcomes. Accordingly, we assessed the surgeries using conventional microscopy versus the NGENUITY System by comparing the rates of recurrence of RD, MH closure, and reduction in central macular thickness in the case of ERMs at 3 months after surgery. The secondary endpoints analyzed were surgery durations and 3-month postoperative best-corrected visual acuity (BCVA) measured in LogMAR (logarithm of the minimum angle of resolution). We analyzed patient demographic characteristics and ophthalmology history, as well as the main prognostic factors recognized in the literature for each eye disease, MH diameter, and the primary or secondary nature of ERMs in order to ensure CM-group and 3D-group comparability. Surgical techniques were also compared in order to avoid analytical biases.
Statistical analyzes
Data were analyzed by univariate analysis by comparing the CM and 3D groups for each parameter described in the former paragraph. Analyzes were performed on all diseases combined, but also independently for each MH, RD, and ERM disease subgroup (subgroup analysis). Qualitative variables were compared using the Chi-squared test or the Fisher’s exact test when applicable. Quantitative variables were compared using the Welch’s t-test and Student’s t-test. When group numbers were too small, the non-parametric Mann–Whitney U test was used. The significance level retained was the classic 5% threshold (p < 0.05). All calculations were performed using Excel 2018.
Results
Description of the patient series
A total of 224 patients were included for study, divided into patients who underwent surgery with the NGENUITY System (n = 131) or with conventional microscopy (n = 96). Note that three (0.45%) patients were operated on via conventional microscopy in their first eye and then via the NGENUITY System on their contralateral eye 1 year later. A total of 138 (57%) eyes and 103 (43%) eyes in the 3D and CM groups were included for study, respectively. Patient demographic data showed no difference between 3D and CM groups in terms of age (p = 0.69), sex (p = 0.17), preoperative refraction (p = 0.35), and ophthalmology history (p = 0.084). Table 1 summarizes the demographic data and the ophthalmology history of patients included for study.
Table 1.
Demographic data and ophthalmology history of patients included for study.
| 3D group | CM group | p value | |
|---|---|---|---|
| Number of patients, n | 131 | 96 | |
| Number of eyes, n (% total) | 138 (57%) | 103 (43%) | |
| Age in years, mean (± SD) | 65.3 (± 12.7) | 65.9 (± 12.8) | 0.69d |
| Sex, n (% total eyes) | 0.17e | ||
| Male | 67 (49%) | 60 (58%) | |
| Female | 71 (51%) | 43 (42%) | |
| Side, n (% total eyes) | 0.9f. | ||
| Left | 80 (58%) | 58 (56%) | |
| Right | 58 (42%) | 45 (44%) | |
| Preoperative refraction, n (% total eyes) | 0.35f | ||
| Low myopiaa | 66 (48%) | 42 (41%) | |
| Low hyperopiab | 56 (41%) | 43 (42%) | |
| High myopiac | 16 (12%) | 18 (17%) | |
| Lens status, n (% total eyes) | 0.30e | ||
| Phakia | 86 (63%) | 57 (55%) | |
| Pseudophakia | 51 (36%) | 46 (45%) | |
| Aphakia | 1 (0.73%) | 0 (0%) | |
| Ophthalmology history, n (% total eyes) | 0.084f | ||
| No | 70 (50.72%) | 65 (63.14%) | |
| Yes | |||
| Anterior segment surgery | 5 (3.6%) | 6 (5.82%) | |
| RD | 16 (11.6%) | 11 (10.68%) | |
| OHT/Glaucoma | 10 (7.24%) | 6 (5.85%) | |
| Macular surgery | 12 (8.69%) | 3 (2.91%) | |
| Eye diseases (RVO, ARMD, ME) | 18 (13.04%) | 10 (9.71) | |
| Posterior uveitis | 9 (6.52) | 2 (1.94%) | |
| Retinopexy | 6 (4.34%) | 4 (3.88%) | |
| Non-perforating contusion | 2 (1.45%) | 2 (1.94%) |
3D group three-dimensional group, ARMD age-related macular degeneration, CM group conventional microscopy group, ME macular edema, OHT ocular hypertension, RD retinal detachment, RVO retinal vein occlusion, SD standard deviation.
aLow myopia: spherical equivalent of 0–5.5 D.
bLow hyperopia: spherical equivalent of 0–4.5 D.
cHigh myopia: spherical equivalent of < − 6 D or axial length > 26 mm.
dWelch’s t-test.
eFisher’s exact test.
f: Chi-squared test.
Surgical procedures
A total of 241 vitreoretinal surgeries were performed without incident: 98 vitrectomies and scleral buckling surgeries were performed for the treatment of RD, 48 for the treatment of MH, 95 for the treatment of ERM, and 44 patients underwent combined surgery with phacoemulsification and intracapsular implantation. The 25-gauge was used for the majority of surgeries in both patient groups. Outpatient surgery was also favored in more than 80% of cases in both groups. Regarding surgery for RD and ERM, the surgical techniques did not differ between the 3D and CM groups. Internal limiting membrane peeling during surgery on patients with MH was slightly more frequent in the 3D group but was not statistically significant (p = 1). The indications for surgery and the surgical techniques performed in our study series are summarized in Table 2. We report no incidents requiring intraoperative re-installation of conventional microscope eyepieces when the NGENUITY System was being used. The vitreoretinal surgeon described a fast learning curve on his behalf for use of the NGENUITY System during the test phase described in materials and methods.
Table 2.
Indications for surgery and the surgical techniques performed in the three-dimensional (3D) and conventional microscopy (CM) patient groups.
| 3D group | CM group | p value | |
|---|---|---|---|
| Indication for surgery, n (% total eyes) | 0.25a | ||
| RD | |||
| Vitrectomy | 56 (41%) | 35 (48%) | |
| Scleral buckling alone | 3 (2.2%) | 4 (3.9%) | |
| ERM | 49 (36%) | 46 (45%) | |
| MH | 30 (22%) | 18 (17%) | |
| Combined surgery, n (% total eyes) | 0.81b | ||
| No | 114 (83%) | 83 (81%) | |
| Yes | 24 (17%) | 20 (19%) | |
| Vitrectomy gauge, n (% total eyes) | 0.065a | ||
| 25-Gauge | 117 (85%) | 75 (73%) | |
| 27-Gauge | 17 (12%) | 24 (23%) | |
| Scleral buckling | 3 (2.9%) | 4 (4.9%) | |
| Hospitalization type, n (% total eyes) | 0.36b | ||
| Outpatient | 112 (81%) | 89 (86%) | |
| Inpatient | 26 (19%) | 14 (14%) | |
| RD | |||
| Retinopexy, n (% total RD) | 0.261a | ||
| Cryoapplication | 37 (63%) | 20 (51%) | |
| Endolaser | 18 (31%) | 14 (36%) | |
| Cryoapplication + endolaser | 2 (3.4%) | 0 (0%) | |
| Scleral buckling, n (% total RD) | 0.165a | ||
| No | 55 (93.2%) | 32 (82%) | |
| > 2 quadrants | 2 (1.7%) | 4 (10%) | |
| 2 quadrants | 1 (1.7%) | 3 (7.7%) | |
| 1 quadrant | 1 (1.7%) | 0 (0%) | |
| Internal tamponade agent, n (% total RD) | 0.720a | ||
| C2F6 | 45 (76%) | 30 (86%) | |
| Silicone oil 1000 | 4 (6.8%) | 4 (10%) | |
| Oxane HD | 3 (5.1%) | 1 (2.6%) | |
| SF6 | 2 (3.4%) | 0 (0%) | |
| C3F8 | 1 (1.7%) | 0 (0%) | |
| Silicone oil 5000 | 1 (1.7%) | 0 (0%) | |
| MH | |||
| Flap , n (% total MH) | 0.061a | ||
| Yes | 17 (57%) | 8 (44%) | |
| No | 8 (27%) | 10 (56%) | |
| Free flap | 5 (17%) | 0 (0%) | |
| ILM peeling, n (% total MH) | 0.09a | ||
| Yes | 30 (100%) | 15 (78%) | |
| No | 0 (0%) | 3 (17%) | |
| Internal tamponade agent, n (% total MH) | 0.28b | ||
| SF6 | 26 (81%) | 18 (86%) | |
| C2F6 | 4 (13%) | 0 (0%) | |
| ERM | |||
| ILM peeling, n (% total ERM) | 1b | ||
| Yes | 37 (77%) | 35 (76%) | |
| No | 11 (23%) | 11 (24%) | |
ERM epiretinal membrane, ILM internal limiting membrane, MH macular hole, RD retinal detachment.
aFisher’s exact test.
bChi-squared test.
Initial anatomical characteristics
The initial anatomical characteristics of the different eye diseases studied did not significantly differ between the patients in the 3D and CM groups (data summarized in Table 3). Concerning patients who underwent surgery for RD, 54% (n = 32) and 51% (n = 20) of patients presented with macular detachment in the 3D and CM groups, respectively (p = 0.58). A history of ipsilateral RD was found in 20% (n = 12) and 10% (n = 4) of patients in the 3D and CM groups, respectively (p = 0.3). The initial MH diameter was 360 (± 137) µm for patients in the 3D group and 384 (± 160) µm for patients in the CM group (p = 0.20). The initial central macular thickness of ERMs was 510 (± 141) µm and 486 (± 92.3) µm in the 3D and CM patient groups, respectively (p = 0.33), and the distribution between primary (39%) and secondary (39%) ERM was identical in both groups (p = 1). The other prognostic factors were the same in both groups (data summarized in Table 3).
Table 3.
Initial anatomical characteristics of patients in the three-dimensional (3D) and conventional microscopy (CM) patient groups.
| 3D group | CM group | p value | |
|---|---|---|---|
| Initial BCVA in LogMAR, mean (± SD) | |||
| Total | 0.837 (± 0.741) | 0.713 (± 0.606) | 0.17a |
| RD | 1.19 (± 0.887) | 0.930 (± 0.564) | 0.143a |
| MH | 0.637 (± 0.312) | 0.832 (± 0.393) | 0.062a |
| ERM | 0.531 (± 0.530) | 0.487 (± 0.365) | 0.64a |
| RD | |||
| Macular status, n (% total RD) | 0.58b | ||
| Detached | 32 (54%) | 20 (51%) | |
| Flat | 22 (37%) | 13 (33%) | |
| Pucker | 5 (8.5%) | 6 (15%) | |
| Lesion type, n (% total RD) | 0.23b | ||
| Tear | 45 (75%) | 27 (69%) | |
| Hole | 9 (15%) | 11 (28%) | |
| PVR | 3 (5%) | 0 (0%) | |
| Not visible | 3 (5%) | 1 (2.6%) | |
| Ear size in quadrant lengths, n (% total RD) | 1b | ||
| < 1 | 44 (91.7%) | 34 (91.9%) | |
| > 1 | 3 (6.1%) | 3 (8.1%) | |
| PVR, n (% total RD) | 0.56b | ||
| Grade A | 2 (3.4%) | 2 (5.1%) | |
| Grade B | 12 (20%) | 8 (21%) | |
| Grade C1 | 4 (6.8%) | 6 (15%) | |
| Grade > C1 | 3 (5.1%) | 3 (7.7%) | |
| No | 38 (66%) | 24 (51%) | |
| History of ipsilateral RD, n (% total RD) | 0.3c | ||
| Yes | 12 (20%) | 4 (10%) | |
| No | 47 (80%) | 35 (90%) | |
| Duration of evolution, n (% total RD) | 0.782c | ||
| 1–3 days | 11 (19%) | 6 (15%) | |
| 4–7 days | 16 (27%) | 9 (23%) | |
| ≥ 8 days | 22 (37%) | 18 (46%) | |
| Unknown | 10 (17%) | 6 (11%) | |
| MH | |||
| CMT in µM, mean (± SD) | 423 (± 63.4) | 439 (± 78.8) | 0.48d |
| MH diameter in µM, mean (± SD) | 360 (± 137) | 384 (± 160) | 0.20a |
| Duration of evolution, n (% total MH) | 0.29b | ||
| 8 days–3 months | 12 (0%) | 10 (53%) | |
| 3–6 months | 8 (27%) | 1 (5.6%) | |
| 6 months–1 year | 4 (13%) | 2 (11%) | |
| Unknown | 5 (17%) | 6 (33%) | |
| ERM | |||
| Etiology, n (% total ERM) | 1b | ||
| Primary | 29 (61%) | 28 (61%) | |
| Secondary | 19 (39%) | 18 (39%) | |
| Initial macular thickness in µM, mean (± SD) | 510 (± 141) | 486 (± 92.3) | 0.33e |
BCVA best corrected visual acuity, CMT central macular thickness, ERM epiretinal membrane, LogMAR logarithm of the minimum angle of resolution, MH macular hole, PVR proliferative vitreoretinopathy, RD retinal detachment, SD standard deviation.
aStudent’s t-test.
bFisher’s exact test.
cChi-squared test.
dMann–Whitney U test.
eWelch’s t-test.
Disease subgroup analysis of primary and secondary outcomes
Subgroup analysis did not identify any statistically significant differences in primary or secondary outcomes analyzed during surgery follow-up between 3D and CM groups (data summarized in Table 4). The overall rate of recurrence of RD in our study series was 12.6%, with a recurrence rate of 10% (6 eyes) in the 3D group and 18% (7 eyes) in the CM group (p = 0.42). The overall rate of MH closure at 3 months was 85%: 82% (n = 23) in the 3D group and 88% (n = 16) in the CM group (p = 0.69). ERM removal was successful in both groups. The overall reduction in central macular thickness of ERMs at 3 months after surgery was 134 µm: 154 (± 159) µm in the 3D group and 115 (± 105) µm in the CM group (p = 0.23).
Table 4.
Anatomical outcomes according to disease subgroup in the three-dimensional (3D) and conventional microscopy (CM) patient groups.
| 3D group | CM group | p value | |
|---|---|---|---|
| 1-month postoperative BCVA in LogMAR, mean (± SD) | |||
| Total | 0.494 (± 0.545) | 0.487 (± 0.539) | 0.096a |
| RD | 0.706 (± 0.750) | 0.516 (± 0.591) | 0.18a |
| ERM | 0.272 (± 0.200) | 0.379 (± 0.358) | 0.079a |
| MH | 0.458 (± 0.243) | 0.406 (± 0.337) | 0.31b |
| 3-month postoperative BCVA in LogMAR mean (± SD) | |||
| Total | 0.428 (± 0.531) | 0.388 (± 0.461) | 0.34a |
| RD | 0.576 (± 0.691) | 0.415 (± 0.508) | 0.23a |
| ERM | 0.241 (± 0.235) | 0.283 (± 0.294) | 0.49a |
| MH | 0.431 (± 0.425) | 0.443 (± 0.348) | 0.086b |
| 1-month postoperative BCVA gain in LogMAR, mean (± SD) | |||
| Total | − 0.331 (± 0.640) | − 0.221 (± 0.461) | 0.15a |
| RD | − 0.471 (± 0.823) | − 0.249 (± 0.706) | 0.46a |
| ERM | − 0.268 (± 0.510) | − 0.115 (± 0.284) | 0.082a |
| MH | − 0.179 (± 0.345) | − 0.426 (± 0.369) | 0.048b |
| 3-month postoperative BCVA gain in LogMAR, mean (± SD) | |||
| Total | − 0.371 (± 0.707) | − 0.347 (± 0.522) | 0.79a |
| RD | − 0.533 (± 0.721) | − 0.474 (± 0.716) | 0.90a |
| ERM | − 0.286 (± 0.452) | − 0.208 (± 0.335) | 0.38a |
| MH | − 0.199 (± 0.532) | − 0.398 (± 0.380) | 0.19b |
| 3-month postoperative outcomes, n (% total eyes) | 0.322c | ||
| Cataract | 21 (8.7%) | 14 (5.8%) | |
| RD | 7 (2.9%) | 8 (3.31%) | |
| Secondary ERM | 4 (1.65%) | 3 (1.25) | |
| Post-operative macular edema | 22 (9.1) | 19 (7.92) | |
| PCO | 1 (0.4%) | 5 (2.1%) | |
| Other | 8 (3.32%) | 2 (0.83%) | |
| None | 77 (32%) | 55 (22.8%) | |
| RD | |||
| Homolateral recurrence, n (% total RD) | 0.42d | ||
| Not within 3-postoperative months | 53 (90%) | 32 (82%) | |
| Within 3-postoperative months | 6 (10%) | 7 (18%) | |
| MH | |||
| 3-month postoperative closure, n (% total MH) | 0.69c | ||
| Yes | 23 (82%) | 16 (89%) | |
| No | 5 (18%) | 2 (11%) | |
| ERM | |||
| 3-month postoperative CMT in µM, mean (± SD) | 364 (± 77.6) | 379 (± 81.2) | 0.46a |
| 3-month postoperative CMT decrease in µM, mean (± SD) | − 154 (± 159) | − 115 (± 105) | 0.23a |
CMT central macular thickness, ERM epiretinal membrane, LogMAR logarithm of the minimum angle of resolution, MH macular hole, PCO posterior capsular opacification, RD retinal detachment, SD standard deviation.
aWelch’s t-test.
bMann–Whitney U test.
cFisher’s exact test.
dChi-squared test.
All surgeries combined, there was a statistically significant improvement in 1-month and 3-month postoperative BCVA in both groups studied (p < 0.001, data not shown). The analysis of BCVA gains at 1 (p = 0.15) and 3 (p = 0.79) months after surgery for all surgeries combined was similar between 3D and CM groups. There were also no differences in BCVA gains between the CM and 3D groups for RD (p = 0.46/p = 0.90) and ERM (p = 0.082/p = 0.38) disease subgroups at 1 and 3 months after surgery, respectively. BCVA gains between the CM and 3D groups for patients with MH was only slightly different at 1 month (p = 0.048) but not at 3 months (p = 0.19) after surgery.
There was no difference in surgery duration between both groups: 45.4 (± 20.1) min in the 3D group and 46 (± 19.8) min in the CM group (p = 0.81) (refer to Table 5). Analysis of the different disease subgroups showed shorter surgery duration for RD in the 3D group: 47.9 (± 24.6) min versus 58.5 (± 4.24) min in the 3D and CM groups (p = 0.037), respectively. We found a shorter surgery duration for MH in the CM group: 40.8 (± 9.24) min versus 51.9 (± 18.6) min in the CM and 3D groups (p = 0.023), respectively. Surgery duration for ERM was similar between both groups (p = 0.68).
Table 5.
Surgery duration according to disease subgroup in the three-dimensional (3D) and conventional microscopy (CM) patient groups.
| Surgery duration in min, mean (± SD) | 3D group | CM group | p value |
|---|---|---|---|
| Total | 45.4 (± 20.1) | 46.0 (± 19.8) | 0.81a |
| RD | 47.9 (± 24.6) | 58.5 (± 4.24) | 0.037a |
| ERM | 38.5 (± 11.4) | 37.5 (± 12.2) | 0.68a |
| MH | 51.9 (± 18.6) | 40.8 (± 9.24) | 0.023a |
ERM epiretinal membrane, MH macular hole, RD retinal detachment, SD standard deviation.
aWelch’s t-test.
Discussion
Heads-up 3D surgical visualization systems allow ophthalmic surgeons to replace conventional surgical microscope eyepieces with cameras retransmitting an image on a screen in front of them. Published studies have already reported on the use of these technologies in vitreoretinal surgery12–18, but only a few studies have included large series of patients19,20. Here, in this study we compared 1 year of continuous use of conventional microscopy versus 1 consecutive year of continuous use of the heads-up NGENUITY 3D Visualization System in our practice for a total of 241 consecutive vitreoretinal surgeries performed by the same surgeon. Overall, we did not find any significant differences in terms of visual outcomes, anatomical outcomes, or surgery durations between both techniques for the surgical treatment of RD, ERM, and MH. Furthermore, we summarize the benefits and drawbacks of using heads-up 3D visualization systems compared to conventional microscopy according to our experience and reports in the literature.
Firstly, we found no differences in anatomical outcomes and postoperative BCVA gains between patients operated on with the NGENUITY System versus conventional microscopy in our study series. These results are in agreement with other study series12,13,15,19,20 also demonstrating no differences in terms of safety or long-term visual prognosis when comparing the same techniques in vitrectomy surgery, and additionally for vitrectomy in the treatment of macular diseases14,16,17 and RD18. Among the aforementioned studies, Zhang et al.20, conducted the largest study on 23-gauge vitrectomy for the treatment of patients with vitreoretinal diseases in China. The authors found no differences in final BCVA, anatomical findings, or outcomes among patients operated on with conventional microscopy compared to the NGENUITY System. Similarly, a very recent French study on a series of 180 vitrectomy surgeries for treatment of patients with RD and MH also found no significant differences between both techniques19.
Regarding anatomical outcomes, the 3-month postoperative rate of recurrence of RD was 12.6% in our study series and showed no difference between the 3D and CM groups (p = 0.42). This value is similar to those described in the literature which vary between 12 and 15%21,22. In a recent meta-analysis, the rate of primary MH closure varied from 90 to 100%, depending on if internal limiting membrane peeling was performed23,24. Although we found no difference between 3D and CM groups (p = 0.69), these rates are slightly higher than those found in our study series here (85%). We can explain this difference by the fact that our study series initially included only some patients presenting with MH and high myopia (4 out of 10 non-closed MH), as well as only some patients with MH above 500 µm in diameter (3 out of 11 non-closed MH). The number of patients with MH was thus limited and made it difficult to make comparisons. In addition, the evolution of MH over time, an important prognostic factor, was unknown for a large proportion of cases (17% and 33% for the 3D and CM groups, respectively). This could be the cause of a comparison bias. Regarding ERM, Guber et al.25, have demonstrated a reduction in 91.9 µm in central macular thickness at 3 months after vitrectomy for treatment of patients with primary ERM. This decrease is in line with the decrease found in our study series (− 134 µm), showing again no difference between 3D and CM groups (p = 0.57), even if we did not separate primary and secondary ERM for data analysis. The collection of follow-up data was only carried out over a 3-month postoperative period in this current study due to loss of contact with a significant number of patients (patients referred to thus via their patient representatives). This allowed us to measure only the short-term effectiveness and safety without the possibility of drawing any long-term conclusions.
Likewise, surgery durations showed no differences between the 3D and CM patient groups in our study series as in accordance with previously published reports12,20. However, there was a difference between the MH and RD disease subgroups; surgery for RD in the 3D group was shorter, and surgery for MH in the CM group was shorter. An increase in internal limiting membrane peeling has been described by Talcott et al.14, but no significant differences were reported for this in other studies, for neither surgery for RD nor MH. Our overall surgery durations are slightly longer than those previously reported20. Moreover, our surgery durations do not include the time taken to set-up the operating room or to position the patient. On the other hand, our surgery durations were collected retrospectively by different healthcare staff present during the different surgeries. Data collection could therefore be staff-dependent, in turn contributing at least in part to the differences in our surgery durations compared to those previously reported.
Advantageous imaging benefits using a heads-up 3D visualization system over conventional microscopy have already been described. To begin, the field depth has been described as similar or superior in 3D versus conventional microscopes due to the better light sensitivity of the software and the HDR cameras; the aperture can be decreased and accordingly the field depth can be increased12,17,26. According to Franklin et al.27, the field depth is two-to-three times greater than that of the standard analogue surgical microscope if the aperture of the NGENUITY System camera is reduced to 30%, but this difference is not significant at higher zoom levels26. The sharpness is thus greater and the operator requires less accommodative effort, which is even more noticeable among older surgeons who no longer have a large reserve of accommodation27.
Secondly, the image is obtained by the fusion of two HDR cameras and then processed by algorithms that allow the detection and magnification of lower light levels than the human eye. Luminance is also improved27. Levels of endoillumination are therefore reduced while at the same time maintaining satisfactory visualization13. Indeed, low endoillumination values of 10%15, 3%28, and even 1%29 have been reported without loss of visual quality. This decrease therefore reduces the blinding effect of the light during surgeries with local anesthesia and reduces retinal phototoxicity, which is more frequent during posterior segment and particularly macular surgery28.
In relation to the facility of use of the heads-up NGENUITY 3D Visualization System, operators' opinions have been previously analyzed via satisfaction questionnaires to gather feedback from surgeons and by comparing fine surgical tasks in the operating room13,15. Palacios et al.15, showed that the heads-up NGENUITY 3D Visualization System was favored over conventional microscopy for the majority of the 14 surgeons who gave their feedback. Their reasoning owed to a better resolution, field depth, educational interest, and field of vision (results only met a statistically significant difference for educational interest). The same authors also demonstrated that the type of surgery performed most often with the 3D System was internal limiting membrane peeling15. These results are similar to recent reports from a team of four French surgeons who completed the same satisfactory questionnaire19. The authors found the main advantages to be better focusing under higher magnifications and that the light source can be kept at a greater distance from the retina (in order to limit macular phototoxicity).
These satisfactory questionnaires also showed a clear improvement in ergonomics, comfort, and a reduction in muscular pain for the users. Back and neck pain are frequently detected among ophthalmologists, and especially among surgeons. Indeed, 50.6% and 31.8% of ophthalmologists participating in a national study in the UK reported back and neck pain, respectively30. The consequences can be limited by correct eyepiece positioning or the use of a head-ups visualization system31–33. We also report here a fast learning curve for our main user of the NGENUITY System, which is in line with experiences in other study series17. Moreover, patient installation and surgery performance is easier with the heads-up system for patients presenting with a spinal deformity, such as kyphosis, or for patients requiring surgery in the Trendelenburg position34 or semi-sitting position. Figure 2 demonstrates use of the NGENUITY System in our center on a patient in semi-sitting position with the surgeon remaining standing.
There are additional pedagogical advantages to using a head-ups 3D visualization system. Firstly, all the operating room personnel present have access to the same and live surgical image, contrary to the classic configuration where only the surgeon can see a high definition and 3D image for the majority of the surgery12. In this light, the surgeon can teach more easily and allow trainee doctors to operate by reducing their installation time33. In the same way, the recording quality of the 2D or 3D videos provides a high-quality teaching aid for reviewing surgeries at distance or live. The pedagogical value of this system has also been studied in other surgical disciplines, particularly in digestive surgery and microsurgery33,35.
The transition from direct visualization using a surgical microscope to analogue visualization via an indirect digital system must fulfil certain technical criteria27. Two slightly different and shifted images are retransmitted by two HD cameras to a 4 K monitor and passive polarized glasses allow for the phenomenon of disparity for each eye36,37. Along with this, there has been a handful of drawbacks identified in the use of head-ups 3D visualization systems. For example, the latency time is 70–80 ms for the NGENUITY 3D Visualization System. In our practice, we did not perceive this latency time as a handicap; it has actually been reduced compared to former versions13. Furthermore, some surgical aids have been reported by surgeons as being less comfortable, resulting in asthenopia by the end of surgery. Underlying exophoria has also been put forward as a risk factor38, but our main operator here throughout this study presents with exophoria and did not encounter any problems during surgery. In addition, some anesthesia teams have conveyed a dislike to the large size of the visualization system with respect to complicated patient and monitoring access, especially during general anesthesia39. The operating room must therefore be of sufficient size and reorganized to avoid these difficulties40. Figure 1 shows the configuration of our operating room with the heads-up NGENUITY 3D Visualization System.
In addition to the significant potential in vitreoretinal surgery, heads-up 3D visualization systems are compatible with the performance of other types of ophthalmology surgery. This includes cataract surgery, with some teams showing effective results without increased risk of complications or increased surgery durations40,41. There also lies an interest in: (1) corneal transplantation, especially for lamellar keratoplasty given the high-zoom quality and field depth42, (2) strabismus surgery, given the surgeon does not require the external light as it is replaced by a HDR camera and gain adjustment43, and (3) glaucoma surgery, with the iStent implantation being simplified by the small camera sizes15. ARGUS II retinal implantation has also been carried out with this system as it renders it easy to perform sclerotomy and intracavitary placement44. Nevertheless, some surgeons have reported technical difficulties during anterior segment surgery and external eye surgeries, with difficulties in acquiring a clear image and instrument positioning. Likewise, these difficulties have also been evoked during scleral buckling15,19,44.
On the whole, changing to use of a single digital display system makes it easier to insert other multimodal imaging components and live details, such as vitrectomy settings (DATAFUSION software), and it can be used to guide incision positions, toric implantation, and rhexis size (VERION Image Guided module)45. Intraoperative OCT is also accessible on a single screen, without the surgeon having to look away or use only one of the two eyepieces46. Ultimately, this transition to a live, digital, and high-quality image is an essential step in enabling image transmission during robot-assisted remote surgery. From here, we can imagine a surgeon operating on a patient at distance without the need to travel. In the same way, due to the recent exponential development of artificial intelligence in ophthalmology, we can hypothesize the development of future applications that allow live operator assistance or even a 100% robotized intervention.
In conclusion, the heads-up NGENUITY 3D Visualization System appears comparable to conventional surgical microscopy in terms of effectiveness and safety in surgical treatment of RD, ERM, and MH. It has proven to be a valuable tool from an ergonomic and pedagogical point of view, while at the same time maintaining high image quality. In the future we can expect this system to be integrated into the framework of intraoperative multimodal imaging in ophthalmology surgery.
Acknowledgements
We thank Dr Jacqueline Butterworth for professional language editing of the manuscript.
Author contributions
P.K., F.V., and V.S. contributed to study conception and design. Data collection and analysis was performed by P.K., F.V., and V.S. P.K. prepared Fig. 1 and V.S. prepared Fig. 2. The first manuscript draft was prepared by P.K. F.M., F.V., V.S., V.P.M,. P.F., and V.S. commented on the previous drafts of the manuscript. All authors have approved the final version of the manuscript.
Funding
None.
Data availability
The datasets analyzed and generated during the current study are available from the corresponding author upon reasonable request.
Competing interests
Vincent Soler has consulted for Alcon Laboratories, Allergan, Bausch & Lomb, Bayer Healthcare, Cristalens, Novartis, Laboratories Théa and received compensation. The other authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Keeler, R. The evolution of the ophthalmic surgical microscope. Hist Ophthal Intern.1, 35–66. https://depot.ceon.pl/bitstream/handle/123456789/9977/The_Evolution_of_the_Ophthalmic_Surgical_Microscope.pdf?isAllowed=y&sequence=1 (2015).
- 2.Roper-Hall MJ. Microsurgery in ophthalmology. Br. J. Ophthalmol. 1967;51:408–414. doi: 10.1136/bjo.51.6.408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schulze, F. How the humble stereomicroscope found its way into modern surgery: The Zeiss operating microscope. https://www.microscopy-uk.org.uk/mag/artjan13/fs-Operating-Microscopes-2.pdf (2012).
- 4.Barraquer JI. The history of the microscope in ocular surgery. J. Microsurg. 1980;1:288–299. doi: 10.1002/micr.1920010407. [DOI] [PubMed] [Google Scholar]
- 5.Barraquer JI, Barraquer J, Littman H. A new operating microscope for ocular surgery. Am. J. Ophthalmol. 1967;63:90–97. doi: 10.1016/0002-9394(67)90581-8. [DOI] [PubMed] [Google Scholar]
- 6.Troutman RC. The operating microscope in ophthalmic surgery. Trans. Am. Ophthalmol. Soc. 1965;63:335–348. [PMC free article] [PubMed] [Google Scholar]
- 7.Kelman CD. Phaco-emulsification and aspiration. A new technique of cataract removal. A preliminary report. Am. J. Ophthalmol. 1967;64:23–35. doi: 10.1016/0002-9394(67)93340-5. [DOI] [PubMed] [Google Scholar]
- 8.Machemer R, Buettner H, Norton EW, Parel JM. Vitrectomy: A pars plana approach. Trans. Am. Acad. Ophthalmol. Otolaryngol. 1971;75:813–820. [PubMed] [Google Scholar]
- 9.Krasnov MM. Externalization of Schlemm’s canal (sinusotomy) in glaucoma. Br. J. Ophthalmol. 1968;52:157–161. doi: 10.1136/bjo.52.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Caputo, G., Metge, F., Arndt, C. & Conrath, J. Chirurgie endoculaire in Décollements de rétine rapport SFO 2011. (ed Masson, E.) 137–236 (2011).
- 11.Bodaghi, B., Creuzot-Garcher, C., Delyfer, M. N., Kodjikian, L. & Zayani, A. Chirurgie vitréorétinienne et injections intravitréennes in Rétine et vitré rapport AAO et SFO. (ed Masson, E.) 369–394 (2018).
- 12.Romano MR, et al. Evaluation of 3D heads-up vitrectomy: outcomes of psychometric skills testing and surgeon satisfaction. Eye (Lond.) 2018;32:1093–1098. doi: 10.1038/s41433-018-0027-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eckardt C, Paulo EB. Heads-up surgery for vitreoretinal procedures: An experimental and clinical study. Retina (Philadelphia, Pa.) 2016;36:137–147. doi: 10.1097/IAE.0000000000000689. [DOI] [PubMed] [Google Scholar]
- 14.Talcott KE, et al. Comparison of a three-dimensional heads-up display surgical platform with a standard operating microscope for macular surgery. Ophthalmol. Retina. 2019;3:244–251. doi: 10.1016/j.oret.2018.10.016. [DOI] [PubMed] [Google Scholar]
- 15.Palácios RM, et al. An experimental and clinical study on the initial experiences of Brazilian vitreoretinal surgeons with heads-up surgery. Graefes Arch. Clin. Exp. Ophthalmol. 2019;257:473–483. doi: 10.1007/s00417-019-04246-w. [DOI] [PubMed] [Google Scholar]
- 16.Kumar A, et al. Comparison of clinical outcomes between ‘heads-up’ 3D viewing system and conventional microscope in macular hole surgeries: A pilot study. Indian J. Ophthalmol. 2018;66:1816–1819. doi: 10.4103/ijo.IJO_59_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Palácios RM, Maia A, Farah ME, Maia M. Learning curve of three-dimensional heads-up vitreoretinal surgery for treating macular holes: A prospective study. Int. Ophthalmol. 2019 doi: 10.1007/s10792-019-01075-y. [DOI] [PubMed] [Google Scholar]
- 18.Coppola M, La Spina C, Rabiolo A, Querques G, Bandello F. Heads-up 3D vision system for retinal detachment surgery. Int. J. Retina Vitreous. 2017;3:46. doi: 10.1186/s40942-017-0099-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Palácios RM, et al. Clinical study on the initial experiences of French vitreoretinal surgeons with heads-up surgery. Curr. Eye Res. 2020 doi: 10.1080/02713683.2020.1737136. [DOI] [PubMed] [Google Scholar]
- 20.Zhang T, Tang W, Xu G. Comparative analysis of three-dimensional heads-up vitrectomy and traditional microscopic vitrectomy for vitreoretinal diseases. Curr. Eye Res. 2019 doi: 10.1080/02713683.2019.1612443. [DOI] [PubMed] [Google Scholar]
- 21.Jackson TL, Donachie PHJ, Sallam A, Sparrow JM, Johnston RL. United Kingdom National Ophthalmology Database study of vitreoretinal surgery: Report 3, retinal detachment. Ophthalmology. 2014;121:643–648. doi: 10.1016/j.ophtha.2013.07.015. [DOI] [PubMed] [Google Scholar]
- 22.Haugstad M, Moosmayer S, Bragadόttir R. Primary rhegmatogenous retinal detachment—Surgical methods and anatomical outcome. Acta Ophthalmol. 2017;95:247–251. doi: 10.1111/aos.13295. [DOI] [PubMed] [Google Scholar]
- 23.Parravano, M. et al. Vitrectomy for idiopathic macular hole. Cochrane Database Syst. Rev. 2015(5), CD009080. 10.1002/14651858.cd009080.pub2 (2015). [DOI] [PMC free article] [PubMed]
- 24.Morescalchi F, et al. Controversies over the role of internal limiting membrane peeling during vitrectomy in macular hole surgery. Surv. Ophthalmol. 2017;62:58–69. doi: 10.1016/j.survophthal.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 25.Guber J, Pereni I, Scholl HPN, Guber I, Haynes RJ. Outcomes after epiretinal membrane surgery with or without internal limiting membrane peeling. Ophthalmol. Therapy. 2019;8:297. doi: 10.1007/s40123-019-0185-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Freeman WR, et al. resolution, depth of field, and physician satisfaction during digitally assisted vitreoretinal surgery. Retina (Philadelphia, Pa.) 2019;39:1768–1771. doi: 10.1097/IAE.0000000000002236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Franklin, A.J., Sarangapani, R., Yin, L., Tripathi, B. & Riemann, C. Digital vs analog surgical visualization for vitreoretinal surgery. Retinal Physician. https://www.retinalphysician.com/issues/2017/may-2017/digital-vs-analog-surgical-visualization-for-vitre (2017).
- 28.Adam MK, et al. Minimal endoillumination levels and display luminous emittance during three-dimensional heads-up vitreoretinal surgery. Retina (Philadelphia, Pa.) 2017;37:1746–1749. doi: 10.1097/IAE.0000000000001420. [DOI] [PubMed] [Google Scholar]
- 29.Kunikata H, Abe T, Nakazawa T. Heads-up macular surgery with a 27-gauge microincision vitrectomy system and minimal illumination. Case Rep. Ophthalmol. 2016;7:265–269. doi: 10.1159/000452993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hyer JN, et al. National survey of back & neck pain amongst consultant ophthalmologists in the United Kingdom. Int. Ophthalmol. 2015;35:769–775. doi: 10.1007/s10792-015-0036-z. [DOI] [PubMed] [Google Scholar]
- 31.Shaw C, Bourkiza R, Wickham L, Mccarthy I, Mckechnie C. Mechanical exposure of ophthalmic surgeons: A quantitative ergonomic evaluation of indirect ophthalmoscopy and slit-lamp biomicroscopy. Can. J. Ophthalmol. 2017;52:302–307. doi: 10.1016/j.jcjo.2016.09.011. [DOI] [PubMed] [Google Scholar]
- 32.Honavar SG. Head up, heels down, posture perfect: Ergonomics for an ophthalmologist. Indian J. Ophthalmol. 2017;65:647–650. doi: 10.4103/ijo.IJO_711_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mendez BM, Chiodo MV, Vandevender D, Patel PA. Heads-up 3D microscopy: An ergonomic and educational approach to microsurgery. Plast. Reconstr. Surg. Glob. Open. 2016;4:e717. doi: 10.1097/GOX.0000000000000727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Skinner CC, Riemann CD. ‘heads up’ digitally assisted surgical viewing for retinal detachment repair in a patient with severe kyphosis. Retin. Cases Brief. Rep. 2018;12:257–259. doi: 10.1097/ICB.0000000000000486. [DOI] [PubMed] [Google Scholar]
- 35.Chan AC, et al. Comparison of two-dimensional vs three-dimensional camera systems in laparoscopic surgery. Surg. Endosc. 1997;11:438–440. doi: 10.1007/s004649900385. [DOI] [PubMed] [Google Scholar]
- 36.Bloch E, Uddin N, Gannon L, Rantell K, Jain S. The effects of absence of stereopsis on performance of a simulated surgical task in two-dimensional and three-dimensional viewing conditions. Br. J. Ophthalmol. 2015;99:240–245. doi: 10.1136/bjophthalmol-2013-304517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Welchman AE. The human brain in depth: How we see in 3D. Annu. Rev. Vis. Sci. 2016;2:345–376. doi: 10.1146/annurev-vision-111815-114605. [DOI] [PubMed] [Google Scholar]
- 38.Kim S-H, et al. Clinical research on the ophthalmic factors affecting 3D asthenopia. J. Pediatr. Ophthalmol. Strabismus. 2012;49:248–253. doi: 10.3928/01913913-20120207-03. [DOI] [PubMed] [Google Scholar]
- 39.Rizzo S, et al. 3D surgical viewing system in ophthalmology: Perceptions of the surgical team. Retina (Philadelphia, Pa.) 2018;38:857–861. doi: 10.1097/IAE.0000000000002018. [DOI] [PubMed] [Google Scholar]
- 40.Agranat JS, et al. The scope of three-dimensional digital visualization systems in vitreoretinal surgery. Clin. Ophthalmol. 2019;13:2093–2096. doi: 10.2147/OPTH.S213834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Weinstock RJ, Diakonis VF, Schwartz AJ, Weinstock AJ. Heads-up cataract surgery: Complication rates, surgical duration, and comparison with traditional microscopes. J. Refract. Surg. 2019;35:318–322. doi: 10.3928/1081597X-20190410-02. [DOI] [PubMed] [Google Scholar]
- 42.Galvis V, Berrospi RD, Arias JD, Tello A, Bernal JC. Heads up descemet membrane endothelial keratoplasty performed using a 3D visualization system. J. Surg. Case Rep. 2017;2017:rjx231. doi: 10.1093/jscr/rjx231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hamasaki I, et al. Lights-out surgery for strabismus using a heads-up 3D vision system. Acta Med. Okayama. 2019;73:229–233. doi: 10.18926/AMO/56865. [DOI] [PubMed] [Google Scholar]
- 44.Rachitskaya A, Lane L, Ehlers J, DeBenedictis M, Yuan A. Argus II retinal prosthesis implantation using three-dimensional visualization system. Retina (Philadelphia, Pa.) 2019;39(Suppl 1):S199–S200. doi: 10.1097/IAE.0000000000002296. [DOI] [PubMed] [Google Scholar]
- 45.Yonekawa, Y. Seeing the world through 3-D glasses. Retina Today. http://retinatoday.com/2016/10/seeing-the-world-through-3-d-glasses/ (2016).
- 46.Ehlers JP, Uchida A, Srivastava SK. The integrative surgical theater: Combining intraoperative optical coherence tomography and 3D digital visualization for vitreoretinal surgery in the DISCOVER study. Retina (Philadelphia, Pa.) 2018;38(Suppl 1):S88–S96. doi: 10.1097/IAE.0000000000001999. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets analyzed and generated during the current study are available from the corresponding author upon reasonable request.


