Abstract
Urology is one of the fields that are always at the frontline of bringing scientific advancements into clinical practice, including 3D printing (3DP). This study aims to discuss and presents the current role of 3D-printed phantoms and devices for organ-specified applications in urology. The discussion started with a literature search regarding the two mentioned topics within PubMed, Embase, Scopus, and EBSCOhost databases. 3D-printed urological organ phantoms are reported for providing residents new insight regarding anatomical characteristics of organs, either normal or diseased, in a tangible manner. Furthermore, 3D-printed organ phantoms also helped urologists to prepare a pre-surgical planning strategy with detailed anatomical models of the diseased organs. In some centers, 3DP technology also contributed to developing specified devices for disease management. To date, urologists have been benefitted by 3D-printed phantoms and devices in the education and disease management of organs of in the genitourinary system, including kidney, bladder, prostate, ureter, urethra, penis, and adrenal. It is safe to say that 3DP technology can bring remarkable changes to daily urological practices.
Keywords: 3D printing, Phantoms, Devices, Urology
1. Introduction
Improvements and innovations in 3D printing (3DP) technology have influenced wide applications, including aerospace[1], automotive[2], and medicine[3]. In aerospace, 3DP is used to optimize a material structural component topologically and reduces the element stiffness, which could reduce its weight or volume and benefitted the aerospace design process, respectively[1]. Meanwhile, 3DP in automotive industries is applied for tooling up a stamping process for producing body panels. This approach enables the manufacturing of stamping inserts using similar high performance alloy steel as in conventional tooling without losing tool mechanical properties[2].
In medicine, 3DP has been utilized to fabricate prosthetics[4], implants[5,6], medical instruments[7], and pharmaceutical products[8]. 3DP technology also offers significant advantages and potentials, which may facilitate a patient-specific treatment planning[9]. Even further, this technology branched into 3D bioprinting, which aims to pattern and assemble living and non-living material transfer processes to produce bioengineered structures[10]. In practice, 3D bioprinting commonly combines living cells, extracellular matrices (ECM), and polymeric scaffolds as artificial organs for tissue engineering and regenerative medicine purposes[10,11].
In surgeries, a thorough anatomical understanding of the targeted organ must be obtained preoperatively to achieve a suitable surgical strategy. Such essential step was often obtained from the preceding interpretation of the conventional two-dimensional radiology imaging[12]. Although 3D visualization of these images might be available, 3D-printed models were shown to be superior to 3D images in pre-operative planning. This was due to the ability of 3D models to provide more details and tactile representations of organ anatomical aspects for the operators[13-15].
The anatomical models may also be used for mock surgeries and pre-surgical adjustment of instrumentation, thus reducing the operation time, ensuring better approach, and instrument compatibility. Several surgeries have received advantages from this approach, including vascular surgery for endovascular aneurysm repair, cardiac surgery for pre-surgical tumor resection planning and congenital defect repair, neurosurgery for navigation training, and in orthopedic surgery for tumor resection planning and trauma injury treatment[15]. In addition to its utilization as an anatomical model and surgical guide, 3DP technology is also commonly used in implant manufacturing. Patient-specific implant (PSI) is a perfect-fit implant used to restore the anatomy, relationship, and function of a patient’s organ. These implants have been reported in orthopedics, thoracic surgery, ophthalmology, and neurosurgery[15].
The field of urology has also been at the frontline of bringing scientific advancement into clinical practice and surely 3DP technology is no exception. Many reviews have discussed the involvement of 3DP technology in urology. Unfortunately, the existing reviews are mostly focused on the technological aspects and general clinical impacts of 3D-printed products in urological practices without explicitly categorizing the importance of 3DP technology per genitourinary organ[16-20]. This study aims to present and discuss the recent advancement of 3D printed phantoms and devices for organ-specified appliances in the field of urology.
2. Methods
We conducted a comprehensive search in the literature discussing 3D printed phantoms and devices for urological organ appliances within PubMed, Embase, Scopus, and EBSCOhost databases. To identify relevant studies, we used the search terms “3D Printing” and “Urology.” Initially, from the four databases, 56 potentially relevant publications were listed. A total of 35 journals have been included for analysis after exclusions.
3. 3D Printing
There are three manufacturing techniques: Formative, subtractive, and additive manufacturing (AM). Compared to two other techniques, AM, or the so-called 3DP, is the most suitable for prototyping and low volume production of complex designs as it may produce parts in almost any geometry[21].
To create a model, 3DP integrates two simultaneous subprocesses: The physical formation and the sequential attachment of each layer. Each layer is two-dimensionally contoured in an xy-plane before finally, the layer stack forms the third (z) dimension. In 2015, the ISO/ASTM 52900 Standard was set and established seven categories in the 3DP processes. These categories include material extrusion (ME), vat polymerization (VP), powder bed fusion (PBD), material jetting (MJ), binder jetting (BJ), direct energy deposition (DEP), and sheet lamination (SL). Each process has its supporting technology as well as the materials used[21,22].
Each category of 3DP processes in the ISO/ASTM 52900 Standard has also been used for medical purposes[23]. ME and VP have been used for casing fabrication of medical instruments[7,24], scaffolds[6], prostheses[25,26], and phantoms of organs[27]. Meanwhile, PBD, MJ, BJ, DEP, and SL are reported to be used for bone reconstructions and porous implants made of metals (i.e., Fe, Mg, and Ti)[28,29]. Metal-based scaffolds can also be fabricated using MJ and BJ[29], which are mainly applied for bone fracture repairment.
Printing a 3D printed model requires several steps (Figure 1). Printing an original design started with a 3D modeling using computer-aided design (CAD) software. Subsequently, the CAD file was converted into a standard tessellation language (.stl) file so that it can be processed in computer-aided manufacturing (CAM) software. In the field of 3D printing, the CAM software is commonly known as 3D slicer software. In a 3D slicer software, a user can set up and adjust printing parameters according to the user’s specification. Some 3D slicers are open source, but some others are exclusively provided by the 3D printer manufacturers.
Figure 1.
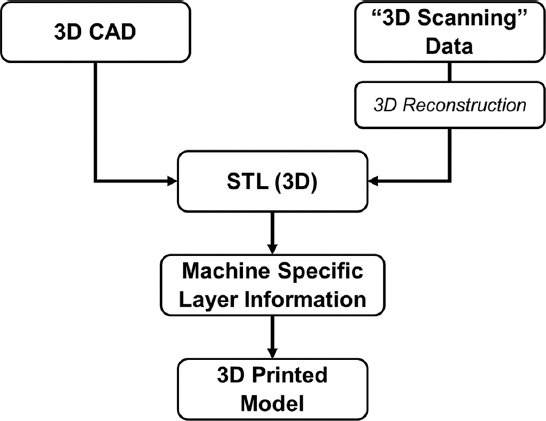
3D printing sequences from design to product.
The design for 3D printing can also be obtained without 3D modeling. This can be performed using 3D scanning. The widely used “3D scanning” equipment in the medical field includes computed tomography (CT) and magnetic resonance imaging (MRI) scanners, which commonly results in a digital imaging and communications in medicine (.dicom) file. A DICOM file is filled with a two-dimensional (2D) image of a scanned body part. The compilation of DICOM files for a body part can be reconstructed into a 3D model[30], which is subsequently converted into a standard tessellation (.stl) file for 3D printing[31]. However, before conversion, the 3D model is refined or segmented to isolate a specific to-print tissue. The refinement is required since the 3D reconstructed model is sometimes incomplete due to the limitations in the 3D reconstruction software. In this case, transfer learning, one of neural network techniques, and generative adversarial network (GAN) can be implemented to fine-tune the incomplete 3D reconstructed model[32,33]. Similar to the refinement process, the segmentation process requires neural networks to accurately classify the desired tissues. Nonetheless, the accuracy of segmentation depends on the dataset used during the training of neural networks. To date, transfer learning can be used to develop a segment classifier for a limited DICOM data[32].
To this day, stereolithography (SLA), a form of vat polymerization, is one of the common 3DP techniques for surgeries due to its high precision and great surface finishing[34]. SLA is a form of vat polymerization process where a high intensity light source is focused on to a vat of liquid polymer bath. The illuminated area of the polymer bath will thus photochemically solidify, forming the layer of the desired 3D object. The finished layer will descend and the focused light will renders the next layer[34].
Eventually, 3DP technology provides two levels of application: Rapid prototyping and rapid manufacturing. Rapid prototyping of organs may be helpful in surgical planning, resident training, and patient education, while rapid manufacturing may facilitate the creation of an on-demand patient-specific medical devices, implants, or prostheses[22].
4. Urology training and patient education
The increasingly complex urological procedures have brought more challenges toward patient education. 3D-printed organ models may provide new modes of patient education, thus may help urologists in obtaining patient consent[34]. A survey conducted by Wake et al. concluded that pre-operative 3D-printed models improved patients’ understanding of their condition and the goals of the surgery[35]. Similar findings to Wake et al., Ilie et al. demonstrated the questionnaire data that show the satisfaction of the patients regarding the use of 3D-printed model during the clinical case discussion and were satisfied with this new way of communication[36].
On the other hand, 3D-printed organ models may also help urologists and residents understand detailed anatomy of the diseased organs. Atalay et al. investigated the impact of 3D-printed renal models on residents’ perception before percutaneous nephrolithotomy (PNL) [37]. The results showed that the models provided a better comprehension of the pelvicalyceal system compared to conventional imaging. A similar investigation conducted by Lee et al. resulted in consistent results[38]. The 3D printed urological models are presented in Figure 2.
Figure 2.
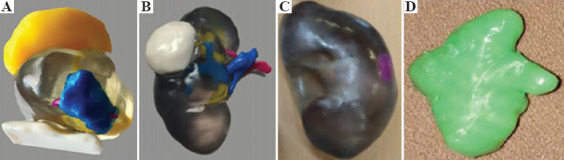
3D printed urological models for training and education: (A) prostate cancer (from ref.[35] licensed under a Creative Commons Attribution 4.0 International License), (B) kidney cancer (from ref.[35] licensed under a Creative Commons Attribution 4.0 International License) and (C) kidney cancer (from ref.[38] licensed under a Creative Commons Attribution License), and (d) kidney stone [reproduced from ref.[47] with the kind permission of Dr. Lütfi Canat (private communication)].
Since the simulation training has been utilized as a complementary training method in urology, 3D-printed models can potentially provide solutions to several drawbacks identified in the traditional cadaveric training[39]. Several studies have indicated the benefits of 3DP technology in various types of urological simulations. Unfortunately, these studies did not yet have sufficient level of evidence, thus further randomized control trials are still needed, with a particular focus on validity and educational impact[16,40-45].
5. Urological disease management
A more detailed description of the application of 3D-printed phantoms and related devices is presented below based on their use in the management of diseases in each genitourinary organ (Table 1). Some footages of 3D-printed phantoms and devices are presented in Figure 3.
Table 1.
Summary of 3D-printed products based on genitourinary organs
| Organ | Case | Application | 3DP Technique | Material | Advantage and Efficacy | Limitation | References |
|---|---|---|---|---|---|---|---|
| Kidney | Renal stones | Surgical guide for PNL | EM | PLA | •Faster access of needle during a PNL-procedure • Low-cost |
•Requiring an on-site 3D printer | Golab et al.[47] |
| Renal masses | Renal stone model for volume calculation | MJ | ABS | •Enabling stone volume calculation in renal stone treatment | •The use of ellipsoid formula in measuring the stone volume, which tends to oblate or prolate in shape | Canatet al.[39] | |
| Renal tumor model for LPN | N/A | Nylon powder | •Accurate anatomical guidance •Improved LPN performance • Low-cost |
•Small sample size •Lack of 3D printing technique information |
Fan et al.[49] | ||
| Renal transplantation | Atherosclerosis artery model for renal transplantation | MJ | •Translucent resin 28A • ABS |
•Reduced operating tie and rate of complications | •Random printing choice of calcified atheroma form with a density > 300 HU | Dezinet et al.[52] | |
| Vascular disease | Extravascular stent for PNS | PBD | Ti alloy | •No post-operative migration, collapse, of erosion of the stent •No complications, side effects, or left renal vein restenosis |
• High cost | Guo et al.[54] Wang et al.[54] |
|
| Bladder | Bladder Dysfunction | Navigation template for SNS | N/A | N/A | •Improved procedure and accuracy of puncture •Reduced intraoperative radiation e xposure |
•Small sample size for validation •Lack of 3D printing technique information |
Zhang et al.[55] |
| Implantable actuator system for bladder void dysfunction | N/A | Flexible rubber-like material | •Enabling the bladder contraction by shape memory alloy actuation | •Lack of human clinical trials •Lack of 3D printing technique information |
Hassani et al.[56] | ||
| Stress Urinary Incontinence | Pessary fitting for SUI patients | EM | • Mold: PLA •Product: Silicon elastomer |
•Anatomically compatible pessary | •Small sample size for validation •Indirect fabrication; requiring mold fabrications |
Barsky et al.[57] | |
| Bladder Cancer | In vitro bladder cancer model | Bioprinting | •Gelatin methacryloyl (GelMA)• Cells | •More accurate drug response evaluation •Opportunity of personalized medicine |
•Lacking extracellular matrix, an essential component of tumor biology | Kim et al.[61] | |
| Prostate | Prostate Biopsy | Prostate tumor model | VP | Resin | •Accurate replication of tumor construction, location, size, and morphology •Avoided missed diagnosis •Increased rate of biopsy |
•Small sample size for validation | Wang et al.[63] |
| Prostatectomy | Prostate cancer model | N/A | Resin | •Accurate concordance between the models and the histological index lesion location and extension | •Small sample size for validation •Lack of 3D printing technique information |
Shinet al.[65] | |
| Prostate cancer model | MJ | Resin | •More comprehensible model compared to MRI | •Small sample size for validation | Chandak et al.[66] | ||
| Ureter | Ureteral stent | Flow characterization of ureteral DJ stents | PBD | Nylon polyether block amide | •Comparable flow characteristics between conventional and 3D-printed stents | •Requiring access sheath for stent deployment•Lacking tapered end of the stent | Del Junco et al.[68] |
| Vesicoureteral reflux | Flap valve on the tip of anti-reflux ureteral stent | MJ | Silicon elastomer | •Stoppage of backward flow | •Slight decrease in forward flow | Parket al.[69] | |
| Ureteral stone | Upper renal calyx model for ureteral stone removal | N/A | N/A | •Safe and effective stone removal procedure | •Lack of 3D printing technique and material information | Kurodaet al.[70] | |
| Urethra | Urethral injury | Urethral model of PFUI | EM | N/A | •Understanding of complex anatomies of the posterior urethra and other organs | •Lack of material information •Downscale model to 80% of the size |
Joshi and Kulkarni[71] |
| Urethral structure | In vitro tissue engineered urethral model | Bioprinting | • PCL/PLCL •Cell-laden hydrogel |
•Multilayer of cell constructs •Good cell viability •Similarity in mechanical properties of native cells |
•Use of rabbit cells, instead of real human cells | Zhanget al.[72] | |
| Urethral dynamics | Urinary tract phantom for urodynamic investigations | EM | • Mold: PLA •Product: PVA cryogel |
•Mimicking ability in urethra’s geometric, mechanical, and hydrodynamic characteristics | •Indirect fabrication; requiring mold fabrications | Ishii et al.[27] | |
| Penis | Penile cancer | Penile surface mold brachytherapy | VP | Polycarbonate - like resin | •Non-invasive ambulatory procedure •Minimal pain during the application |
•Small sample size for validation | D’Alimonte et al.[74] |
| Penile reconstruction | Bio-scaffold for tissue engineering of penile fibrous tissue | Bioprinting | • PCL •Fibroblast cells |
•Well-defined and homogenous porous structure of the scaffolds |
•Small sample size •No investigation on the bioavailability of cell-seeded scaffold in an in vivo animal model |
Yu et al.[75] | |
| Adrenal | Adrenalectomy | Phantom for pre-surgical planning of partial adrenalectomy | VP | Translucent resin | •No intra-operative and post-operative complications •Cortisol replacement initiation right after surgery |
•Status of the study being a single case report | Srougi et al.[76] |
Figure 3.

3D printed urological models and devices for disease management: (A) Cancerous kidney model for LPN (from ref.[49] licensed under a Creative Commons Attribution 4.0 International License), (B) bladder model made using a 3D printed mold (from ref.[75] Lurie K.L, Smith G.T, Khan S.A, et al. Three-dimensional, distendable bladder phantom for optical coherence tomography and white light cystoscopy, Journal of Biomedical Optics 19(3), 036009 (1 March 2014). doi.org/10.1117/1.JBO.19.3.036009, (C) navigation template for puncture of SNS (Reprinted Zhang JZ, Zhang P, Wu LY, et al., Application of an individualized and reassemblable 3D printing navigation template for accurate puncture during sacral neuromodulation, Copyright © 2018 Wiley Periodicals, Inc[55], (D) cancerous prostate model (from ref.[66] under a Creative Commons Attribution License), and (E) urethral Meatal dilator (from ref.[76] under a Creative Commons Attribution 4.0 International License).
5.1. Kidneys
(1) Renal stones
In percutaneous nephrolithotomy (PNL), needle positioning greatly influences the duration of the procedure, the duration of exposure to fluoroscopy radiation, and complications. Golab et al. reported their experience using a customized 3D-printed surgical guide made of polylactic acid (PLA) for a PNL procedure. The guide was designed with a needle insertion path and spinous process projections on its surface to facilitate a safe and precise needle insertion into the stone location. The procedure was completed without any complication[46].
The size and location of stones are crucial prognostic parameters in estimating the result of endoscopic stone removal treatment as well as the possibility of spontaneous stone passage. In a retrospective study conducted by Canat et al., renal stone volumes of 27 PNL patients were measured using a 3D-printed model. Comparison with the true stone volume showed that renal stone volume calculation using 3D-printed model provided a more accurate estimation of renal stone volume (Figure 2D)[47].
(2) Renal masses
Nephron-sparing approach is now routinely used in surgical treatment of small renal tumors. A systematic review of 27 studies conducted by Lupulescu et al. showed that the use of 3D-printed models for pre-operative planning allows for a greater amount of intraoperative tissue salvage[48].
Fan et al. conducted a retrospective analysis of 127 patients who have experienced 3DP-assisted and traditional laparoscopic partial nephrectomy (LPN) with several kidney models (Figure 3A). Complex cases in the 3DP-assisted LPN group were reported to experience significantly shorter warm ischemia time and less intraoperative blood loss compared to the traditional LPN group[49]. Training with 3DP models has also been reported to provide accurate anatomical guidance on robot-assisted partial nephrectomy procedures and laparoscopic heminephrectomy in a horseshoe kidney tumor case[50,51].
(3) Renal transplantation
Transplant patients often have numerous arterial calcifications due to their underlying diseases and risk factors. A technical note reported by Denizet et al. showed that preoperative 3D-printed models of recipients’ aortoiliac axis may aid the kidney transplant surgeons in locating the calcified plaques, thus reducing operating time and rate of complications at the anastomosis site[52].
(4) Vascular disease
The interest in 3D-printed surgical instruments for intraoperative use has also been developed. Guo et al. reported a case of posterior nutcracker syndrome treated with laparoscopic placement of a 3D-printed extravascular stent. Postoperatively, the stent was shown to be safe with no migration, collapse, or erosion[53]. Subsequently, Wang et al. conducted a similar study involving 17 nutcracker syndrome patients who had been treated with extravascular titanium stents. Follow-up examinations showed that none of the patients had complications, side effects or left renal vein restenosis[54].
5.2. Bladder
(1) Bladder dysfunction
The sacral nerve stimulation (SNS) has been a convenient therapy for lower urinary tract dysfunction that is poorly responsive to conventional treatment. Zhang et al. demonstrated the clinical application of a 3D-printed navigation template for assisting lead implantation on SNS (Figure 3C). The template was designed with puncture holes and fixation poles to facilitate secure lead implantation to the closest site possible to the sacral nerve. The utilization of this 3D-printed navigation template significantly decreased the amount of sacral foramina punctures, the puncture time, and the radiation exposure[55].
Nonetheless, patients with sacral or pudendal nerves injured or damaged, or patients with degenerated detrusor muscles will not find any benefit from neuromodulation. Hassani et al. built a flexible implantable actuator system to contract the bladder directly and physically of an anesthetized rat to achieve on demand micturition. Their findings showed that this device may has potential use in patients with voiding dysfunction[56].
(2) Stress urinary incontinence (SUI)
By compressing the urethra against the pubic symphysis and increasing the pressure of urethral closure, which simultaneously decreases urinary leakage, pessaries can effectively relieve symptoms of stress urinary incontinence (SUI). The secret to symptom enhancement is proper mechanical fit. Barsky et al. announced that a personalized 3D-printed pessary was successfully implanted into an SUI patient. 3DP permits an anatomically compatible pessary that can be generated reproducibly[57].
(3) Bladder cancer
Neobladder after radical cystectomy is the gold standard method for urinary diversion. However, various complications, such as mucus development, electrolyte imbalances and increased potential for malignant transformation, are associated with these techniques. Bladder-preserving treatments such as radio and chemotherapy are also often promoted in the interest of the quality of life of the patients. Tissue engineering has concentrated on the reconstruction of bladder tissue over the past two decades, and substantial progress has been made in this area[58,59]. In the meantime, 3D printing just found its role in the post-operative evaluation of a novel Y-pouch neobladder, as reported by Bejrananda et al. The 3D-printed neobladder model enabled evaluation for structural and functional reconstructive outcomes of the neobladder[60].
Kim et al. had developed a 3D-printed cancer cell culture environment that successfully demonstrated a similar basal action and cell-to-cell interaction with which could be seen in the in vivo environment. This 3D-cultured cancer cells exhibited higher resistance and lower sensitivity to chemotherapy than the 2D-cultured cancer cells. These findings showed that 3DP may improve bladder cancer treatment by allowing a more accurate drug response evaluation as well as prediction of specific personal responses to drugs[61].
Lurie et al. developed a 3D cancerous bladder phantom with multiple features, including the macroscale structure, microscale topology, subsurface microstructure, optical characteristics, optical properties, and size (Figure 3B)[62]. Using 3DP for the molds and spin-coating of polydimethylsiloxane (PDMS) for the phantom fabrication, the irregular structure of the phantom could be generated. The used techniques and materials resulted in a bladder phantom that could model realistic imaging conditions, either with optical coherence tomography (OCT) or white light cystoscopy (WLC), of bladder cancers[62].
5.3. Prostate
(1) Prostate biopsy
The primary alternative for diagnosing prostate cancer is transrectal ultrasound (TRUS) guided prostate biopsy. With existing conventional approaches, however, prostate sampling at the anterior, midline, and apex sections often leads to underdiagnosis of clinically important disease. Wang et al. utilized 3DP technology to help early prostate cancer diagnosis, which dramatically increased the rate of biopsy and avoided missed diagnosis of high-risk prostate cancer. The 3D printing technique accurately replicates the 3D image and shows the tumor’s location, size, and morphology intuitively by adding clear resin materials[62].
(2) Prostatectomy
Porpiglia et al. tested the face and content validity on the use of 3D-printed prostate cancer models before robot-assisted radical prostatectomy. The models displayed the prostatic glands, the cancer and the neurovascular bundles (NVB) configuration. Before performing live prostatectomy surgery at the 5th Techno-Urology Meeting, surgeons appeared on the screen with a live audio link to the auditorium with the 3D-printed models in their hands, enabling provokers, moderators, and attendants to actively participate in the surgical training. Surgeons and congress participants filled out the F&C Validity Questionnaire, made of open-ended questions of ten-point ranking Likert scales. Analysis of the collected 144 questionnaires showed a good result regarding both surgical planning and anatomical accuracy. It means that 3DP technology was considered to be a useful tool in surgical planning strategy[63].
Shin et al. proved the concept by building five translucent 3D-printed prostate cancer models that visualized the location, size, and extent of the index cancer lesion. The life-size reality of the models made surgeons understand the distance or proximity of the index lesion, the prostate capsule, and NVB more readily. Accurate concordance between the models and the histological index lesion location and extension resulted in negative margins[64]. This result was in accordance with the IDEAL Phase 2a study outcome conducted by Chandak et al.[65] Individuals with limited experience, such as surgical trainees, would find the prostate cancer models (Figure 3D) more comprehensible compared to MRI[66].
5.4. Ureter
(1) Ureteral stents
Stent size selection is of importance as an incorrect selection of stent length election may lead to urinary discomfort, voiding symptoms, and stent migration. Del Junco et al. characterized the flow characteristics of a novel 3D-printed ureteral stents with the conventional Double-J (DJ) stents in an ex vivo porcine model. Although several limitations were found, their results demonstrated that the mean total flow rates represented by the 3D-printed stents were comparable to the conventional stents. Continued technological and material advances will allow for a functional ureteral 3D-printed stent[67].
(2) Vesicoureteral reflux (VUR)
The most crucial side effect of stents is VUR as it can cause urinary tract infections, which can lead to pyelonephritis. An anti-reflux ureteral stent with a 3D-printed polymeric flap valve was successfully developed and manufactured by Park et al. The proposed stent consisted of a 7 French DJ stent and a polymer flap valve. Two complete DJ stents were prepared, the uncoated valve (UCV) and the parylene C coated valve (PCV). Both stents were tested and contrasted with an intact DJ stent in terms of flow path, parylene coating and side holes of the stent. The in vitro results showed that the suggested stent effectively stopped backward flow with a slight decrease in forward flow[68].
(3) Ureteral stone
Kuroda et al. made use of a 3D-printed model for selecting the best percutaneous approach on managing an allograft ureteral stone case. A model-assisted antegrade ureteroscopic lithotomy was successfully conducted. The 3D-printed model managed to aid in a safe and effective stone removal procedure[69].
5.5. Urethra
(1) Urethral injury
It is not easy even for the most experienced surgeon to find the posterior urethra when performing urethroplasty anastomosis. The available two-dimensional imaging complicates prediction in which direction the posterior urethra might be pulled. Joshi et al. created 3D-printed urethral models of ten pelvic fracture urethral injury (PFUI) patients. A survey was conducted asking whether the models were useful for them to carry out a preoperative assessment. The collected data suggested that 3DP might help understand complex anatomies of the posterior urethra, such as re-redo PFUI, bulbar necrosis, rectourethral fistula, young girls with urethrovaginal fistula, bladder neck injury, and double block. 3DP might also help the evaluation of posterior urethra’s distance from the rectum, length of the urethral gap, relation to the posterior urethra, and the direction of displacement of the urethra and whether inferior pubectomy was required or not[70].
Not only for phantoms, 3DP for urethral injuries was also used to fabricate urethra-related medical devices, meatal dilator. Meatal dilator is used to treat meatal stenosis. Meatal stenosis is a urethral abnormality in which the opening of urethra is narrowed. Although mostly caused by infection, meatal stenosis can also be caused by injuries. Chen et al. replicated a discontinued meatal urethral dilator in Australia using ME and PBD techniques (Figure 3E)[72]. The ME technique used was the fused deposition modeling (FDM), while the used PBD technique was selective laser sintering (SLS). The ME technique fabricated dilators made of polylactic acid (PLA) and acrylonitrile butadiene styrene (ABS). Meanwhile, the PBD technique fabricated dilators made of nylon. Mechanical testing of all materials showed that the nylon made by PBD was surprisingly tough and elastic, comparable with the conventionally manufactured device, whereas PLA and ABS made by ME was mechanically weak and inconsistently structured[72].
(2) Urethral structure
Zhang et al. proposed a new urethra bio-production technique using 3D bioprinting technology that combines spatially urothelial cells (UC) and smooth muscle cells (SMC) to recreate the structure and function of the native urethra. For the 3D bio-printed urethra test, where UC and SMC cells were extracted from rabbit bladder biopsy, they used rabbits as animal models. This research provided a strong basis for future 3D bioprinting studies of the urethra, where the mechanical properties and cell growth of the engineered biomimetic urethra could be optimized in vitro for urethral implantation begin with animal models and hopefully ending in future implantations in patients[71].
(3) Urethral dynamics
Ishii et al. had designed an anatomically accurate and deformable urethra phantom, capable of simulating male prostatic urethra’s geometric, mechanical, and hydrodynamic characteristics. Due to benign prostatic hyperplasia, these phantoms were created for the normal urethra and an obstructed urethra (BPH). Ultrasound imaging was then conducted over various planes to determine the morphology of the constructed phantoms. To visually observe the flow profile within the urinary tract, color Doppler imaging was also conducted. Their study showed that the fabricated phantoms could simulate urinary tract deformation, as BPH phantom managed to show reduced urethral deformation as well as higher flow velocities. These scientific advancements may eventually improve the treatment of patients with lower urinary tract symptoms (LUTS)[27].
5.6. Penis
(1) Penile cancer
Penile carcinoma is rare. For a localized tumor, the optimal treatment choice with local control rates above 90% is considered to be complete or partial penectomy with a safe 5-10 mm margin. Unfortunately, radical surgical techniques have a profound influence on the psychosexual side and quality of life of patients, so therapeutic methods such as partial penectomy, external beam radiation therapy, and interstitial brachytherapy are more commonly suggested.
A penile surface mold brachytherapy technique using a custom 3D-printed high-dose-rate (HDR) brachytherapy applicator and its clinical outcome in an early patient cohort was identified by D’Alimonte et al. This approach gave the patient a non-invasive ambulatory procedure with minimal pain during each application. This choice made the opportunity to imagine the treatment with simple applicators on the setup an alternative to current approaches to treatment[72].
(2) Penile reconstruction
Yu et al. indicated that for tunica albuginea replacement, 3D-printed polycaprolactone (PCL) scaffolds seeded with fibroblast cells would be feasible. The tensile strength of a scaffold built with an oblique pattern will be greater. The biocompatibility and strength of two kinds of PCL scaffolds built using a 3D bioprinting technique were assessed in this preliminary analysis. All the scaffolds had a well-defined architecture and a homogeneous porous structure, based on their findings. Fibroblast cells were well attached, proliferated, and differentiated in the PCL scaffolds. These findings indicate that 3D-printed PCL scaffolds may be used for tissue engineering applications in the field of sexual medicine[73].
5.7. Adrenal
(1) Adrenalectomy
Partial adrenalectomy (PA) as a key in managing adrenal gland diseases still has weaknesses. When doing a partial resection, there was a risk of tumor recurrence and the patient may also require hormone replacement. To achieve good functional results after a PA, determining the resection limit and the volume of the residual gland is therefore important.
Srougi et al. reported a successful partial adrenalectomy using a pre-operative 3D-printed model. Neither intraoperative nor post-operative complications were present. The operation time was 190 min, with bleeding estimated at < 50 ml. This single case demonstrates a new technique that could maximize the functional effects of a partial adrenalectomy[74].
6. Discussion
A new era of clinical treatments has been created by relatively recent discoveries in imaging and surgical system technology at the hands of urologists and the concurrent growth of 3DP technology has contributed to its incorporation into other areas of urological care. To the best of our knowledge, this review is the first to present and discuss the application of 3DP technology in every urinary system organ. 3DP is a rapidly evolving new technology which has shown great promise in the medical and surgical fields. This may be most important in surgical decision-making and preparation for difficult cases in urology field, undoubtedly to avoid any adverse effects[77,78].
However, the use of 3DP technology in the development of urological models, phantoms, and appliances requires thorough and comprehensive strategies. In terms of value for money, the selection of 3D printer must be considered meticulously since each type of 3D printers has its own trade-off. For instance, the purchase of SLA 3D printer may deliver smoother and more delicate products with faster production time compared to extrusion printers. However, the price for one unit of SLA 3D printer is relatively more expensive than one unit of extrusion printer, as well as their consumable goods. In this case, a urology research center should define the preference, whether the delicacy of the 3D products or cost efficiency[78].
In terms of technological aspects, urologists do not have adequate experience to segment medical images and print 3D models of patient anatomy. Therefore, it is important for the urologists to collaborate with engineers in the field of 3D modeling and 3D printing technology. Such collaboration may strengthen both clinical and technical aspects of the 3D models, phantoms, and appliances.
Material selection in 3DP is not always straightforward. For example, for truly mimicking soft tissues, especially the mechanical properties, 3DP requires flexible materials. However, flexible materials, such as thermal polyurethane (TPU) and thermoplastic elastomer (TPE), are not easy to proceed. Pre-adjustment processes are needed. Apart from this issue, some materials require post-processing to be fully biocompatible. In addition, some materials cannot withstand sterilization procedures, such as autoclave sterilization. Polylactic acid (PLA) is one of the materials that failed to preserve its integrity due to high temperature and high pressure of an autoclave.
In addition, specific regulation guidelines are still rarely enforced and limited in the usage of customized 3D-printed medical devices, which may contribute to medical misconducts[19,78].
7. Conclusion
3D printing technology may bring remarkable changes in medical fields, and particularly in for the phantom and device development of organ-specified appliances in urology. Nevertheless, further studies are needed to explore broader knowledge about the desirable functionalities and clinical outcomes of 3D-printed materials and products. As 3DP technology continues to grow the potential of personalized surgery from research institutions to regular urological practice, the years ahead will be an exciting time.
Acknowledgments
We thank the Cipto Mangunkusumo General Hospital and Universitas Indonesia for making the production of this article possible.
Conflict of interest
No conflict of interest was reported by all authors.
Author contribution
NPA drafted the manuscript and prepared the review materials for clinical aspects in the manuscript. MHN drafted the manuscript and prepared the review materials for technical aspects in the manuscript. GAI provided some clinical insights. CAM supervised the drafting and secured the funding.
References
- 1.Kamal M, Rizza G. Additive Manufacturing for the Aerospace Industry. Vol. 2019. Amsterdam, Netherlands: Elsevier; Design for metal additive manufacturing for aerospace applications; pp. 67–86. https://doi.org/10.1016/b978-0-12-814062-8.00005-4. [Google Scholar]
- 2.Leal R, Barreiros FM, Alves L, et al. Additive Manufacturing Tooling for the Automotive Industry. Int J Adv Manuf Technol. 2017;92:1671–6. https://doi.org/10.1007/s00170-017-0239-8. [Google Scholar]
- 3.Roopavath UK, Kalaskar DM. 1-Introduction to 3D Printing in Medicine. In: Kalaskar DM, editor. 3D Printing in Medicine. Woodhead Publishing; United Kingdom: 2017. pp. 1–20. https://doi.org/10.1016/b978-0-08-100717-4.00001-6. [Google Scholar]
- 4.O'Brien KW, Xu PA, Levine DJ, et al. Elastomeric Passive Transmission for Autonomous Force-Velocity Adaptation Applied to 3D-Printed Prosthetics. Sci Robot. 2018;3:eaau5543. doi: 10.1126/scirobotics.aau5543. https://doi.org/10.1126/scirobotics.aau5543. [DOI] [PubMed] [Google Scholar]
- 5.Ho CM, Ng SH, Yoon YJ. A Review on 3D Printed Bio-Implants. Int J Precis Eng Manuf. 2015;16:1035–46. [Google Scholar]
- 6.Whulanza Y, Hidayaturrahmi P, Kurniawati T, et al. Depok City, Indonesia: 2017. Realization and Testing of Multi-Material 3D Printer for Bone Scaffold Fabrication; p. 040001. https://doi.org/10.1063/1.4976786. [Google Scholar]
- 7.Abuzairi T, Sumantri NI, Irfan A, et al. Infrared Thermometer on the Wall (iThermowall):An Open Source and 3-D Print Infrared Thermometer for Fever Screening. HardwareX. 2021;9:e00168. doi: 10.1016/j.ohx.2020.e00168. https://doi.org/10.1016/j.ohx.2020.e00168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Norman J, Madurawe RD, Moore CM, et al. A New Chapter in Pharmaceutical Manufacturing:3D-Printed Drug Products. Adv Drug Deliv Rev. 2017;108:39–50. doi: 10.1016/j.addr.2016.03.001. https://doi.org/10.1016/j.addr.2016.03.001. [DOI] [PubMed] [Google Scholar]
- 9.Noorani R. CRC Press; United States: 2017. 3D Printing:Technology, Applications, and Selection. [Google Scholar]
- 10.Ng WL, Chua CK, Shen YF. Print Me An Organ!Why We Are Not There Yet. Prog Polym Sci. 2019;97:101145. https://doi.org/10.1016/j.progpolymsci.2019.101145. [Google Scholar]
- 11.Jordan JM. MIT Press; United States: 2019. 3D Printing. [Google Scholar]
- 12.Pugliese L, Marconi S, Negrello E, et al. The Clinical use of 3D Printing in Surgery. Updates Surg. 2018;70:381–8. doi: 10.1007/s13304-018-0586-5. https://doi.org/10.1007/s13304-018-0586-5. [DOI] [PubMed] [Google Scholar]
- 13.Kim GB, Lee S, Kim H, et al. Three-Dimensional Printing:Basic Principles and Applications in Medicine and Radiology. Korean J Radiol. 2016;17:182–97. doi: 10.3348/kjr.2016.17.2.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zheng Y, Yu D, Zhao J, et al. 3D Printout Models vs. 3D-Rendered Images:Which Is Better for Preoperative Planning? J Surg Educ. 2016;73:518–23. doi: 10.1016/j.jsurg.2016.01.003. https://doi.org/10.1016/j.jsurg.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 15.Shilo D, Emodi O, Blanc O, et al. Printing the Future-Updates in 3D Printing for Surgical Applications. Rambam Maimonides Med J. 2018;9:e0020. doi: 10.5041/RMMJ.10343. https://doi.org/10.5041/rmmj.10343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Smith B, Dasgupta P. 3D printing Technology and Its Role in Urological Training. World J Urol. 2020;38:2385–91. doi: 10.1007/s00345-019-02995-1. https://doi.org/10.1007/s00345-019-02995-1. [DOI] [PubMed] [Google Scholar]
- 17.Chen MY, Skewes J, Desselle M, et al. Current Applications of Three-Dimensional Printing in Urology: 3D Printing in Urology. BJU Int. 2020;125:17–27. doi: 10.1111/bju.14928. https://doi.org/10.1111/bju.14928. [DOI] [PubMed] [Google Scholar]
- 18.Mathews DA, Baird A, Lucky M. Innovation in Urology:Three Dimensional Printing and Its Clinical Application. Front Surg. 2020;2020:7. doi: 10.3389/fsurg.2020.00029. https://doi.org/10.3389/fsurg.2020.00029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cacciamani GE, Okhunov Z, Meneses AD, et al. Impact of Three-Dimensional Printing in Urology:State of the Art and Future Perspectives. A Systematic Review by ESUT-YAUWP Group. Eur Urol. 2019;76:209–21. doi: 10.1016/j.eururo.2019.04.044. https://doi.org/10.1016/j.eururo.2019.04.044. [DOI] [PubMed] [Google Scholar]
- 20.Sun Z, Liu D. A Systematic Review of Clinical Value of Three-Dimensional Printing in Renal Disease. Quant Imaging Med Surg. 2018;8:311–25. doi: 10.21037/qims.2018.03.09. https://doi.org/10.21037/qims.2018.03.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Redwood B, Schöffer F, Garret B. 3D Hubs B.V; Netherlands: 2017. The 3D Printing Handbook:Technologies, Design and Applications. [Google Scholar]
- 22.Gebhardt A, Hötter JS. Carl Hanser Verlag. GmbH &Company KG: Germany: 2016. Additive Manufacturing:3D Printing for Prototyping and Manufacturing. https://doi.org/10.3139/9781569905838.bm. [Google Scholar]
- 23.Rybicki FJ, Grant GT. Springer International Publishing; Germany: 2017. 3D Printing in Medicine:A Practical Guide for Medical Professionals. [Google Scholar]
- 24.Culmone C, Smit G, Breedveld P. Additive Manufacturing of Medical Instruments:A State-of-the-Art Review. Addit Manuf. 2019;27:461–73. https://doi.org/10.1016/j.addma.2019.03.015. [Google Scholar]
- 25.Nyberg EL, Farris AL, Hung BP, et al. 3D-Printing Technologies for Craniofacial Rehabilitation, Reconstruction, and Regeneration. Ann Biomed Eng. 2017;45:45–57. doi: 10.1007/s10439-016-1668-5. https://doi.org/10.1007/s10439-016-1668-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li W, Mille LS, Robledo JA, et al. Recent Advances in Formulating and Processing Biomaterial Inks for Vat Polymerization-Based 3D Printing. Adv Healthc Mater. 2020;9:2000156. doi: 10.1002/adhm.202000156. https://doi.org/10.1002/adhm.202000156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ishii T, Ho CK, Nahas H, et al. Deformable Phantoms of the Prostatic Urinary Tract for Urodynamic Investigations. Med Phys. 2019;46:3034–43. doi: 10.1002/mp.13558. https://doi.org/10.1002/mp.13558. [DOI] [PubMed] [Google Scholar]
- 28.Lowther M, Louth S, Davey A, et al. Clinical, Industrial, and Research Perspectives on Powder Bed Fusion Additively Manufactured Metal Implants. Addit Manuf. 2019;28:565–84. https://doi.org/10.1016/j.addma.2019.05.033. [Google Scholar]
- 29.Putra NE, Mirzaali MJ, Apachitei I, et al. Multi-Material Additive Manufacturing Technologies for Ti-, Mg-, and Fe-Based Biomaterials for Bone Substitution. Acta Biomater. 2020;109:1–20. doi: 10.1016/j.actbio.2020.03.037. https://doi.org/10.1016/j.actbio.2020.03.037. [DOI] [PubMed] [Google Scholar]
- 30.Matsiushevich K. Quantitative Comparison of Freeware Software for Bone Mesh from DICOM Files. J Biomech. 2019;5. 84:247–51. doi: 10.1016/j.jbiomech.2018.12.031. https://doi.org/10.1016/j.jbiomech.2018.12.031. [DOI] [PubMed] [Google Scholar]
- 31.Laycock SD, Hulse M, Scrase CD, et al. Towards the Production of Radiotherapy Treatment Shells on 3D Printers using Data Derived from DICOM CT and MRI:Preclinical Feasibility Studies. J Radiother Pract. 2015;14:92–8. https://doi.org/10.1017/s1460396914000326. [Google Scholar]
- 32.Ng WL, Chan A, Ong YS, et al. Deep Learning for Fabrication and Maturation of 3D Bio-Printed Tissues and Organs. Virtual Phys Prototyp. 2020;15:340–58. [Google Scholar]
- 33.Ying X, Guo H, Ma K, et al. 2019 IEEE/CVF Conference on Computer Vision and Pattern Recognition (CVPR) Long Beach, CA, USA: 2019. X2CT-GAN:Reconstructing CT From Bi-Planar X-Rays With Generative Adversarial Networks; pp. 10611–20. https://doi.org/10.1109/cvpr.2019.01087. [Google Scholar]
- 34.Colaco M, Igel DA, Atala A. The Potential of 3D Printing in Urological Research and Patient Care. Nat Rev Urol. 2018;15:213–21. doi: 10.1038/nrurol.2018.6. https://doi.org/10.1038/nrurol.2018.6. [DOI] [PubMed] [Google Scholar]
- 35.Wake N, Rosenkrantz AB, Huang R, et al. Patient-Specific 3D Printed and Augmented Reality Kidney and Prostate Cancer Models:Impact on Patient educaTion. 3D Print Med. 2019;5:4. doi: 10.1186/s41205-019-0041-3. https://doi.org/10.1186/s41205-019-0041-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ilie PC, Farquhar L, Calleja R, et al. P06-The Value of Per-sonalised 3D Printing Model of Kidney Stones for Patients Requiring Surgical Treatment. Eur Urol Suppl. 2018;17:e2011–2. https://doi.org/10.1016/s1569-9056(18)32436-9. [Google Scholar]
- 37.Atalay HA, Ülker V, Alkan İ, et al. Impact of Three-Dimensional Printed Pelvicaliceal System Models on Residents'Understanding of Pelvicaliceal System Anatomy Before Percutaneous Nephrolithotripsy Surgery:A Pilot Study. J Endourol. 2016;30:1132–7. doi: 10.1089/end.2016.0307. https://doi.org/10.1089/end.2016.0307. [DOI] [PubMed] [Google Scholar]
- 38.Lee H, Nguyen NH, Hwang SI, et al. Personalized 3D Kidney Model Produced by Rapid Prototyping Method and its Usefulness in Clinical Applications. Int Braz J Urol. 2018;44:952–7. doi: 10.1590/S1677-5538.IBJU.2018.0162. https://doi.org/10.1590/s1677-5538.ibju.2018.0162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tatar İ, Huri E, Selçuk İ, et al. Review of the Effect of 3D Medical Printing and Virtual Reality on Urology Training with “MedTRain3DModsim”Erasmus+European Union Project. Turk J Med Sci. 2019;49:1257–70. doi: 10.3906/sag-1905-73. https://doi.org/10.3906/sag-1905-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cheung CL, Looi T, Lendvay TS, et al. Use of 3-Dimensional Printing Technology and Silicone Modeling in Surgical Simulation:Development and Face Validation in Pediatric Laparoscopic Pyeloplasty. J Surg Educ. 2014;71:762–7. doi: 10.1016/j.jsurg.2014.03.001. https://doi.org/10.1016/j.jsurg.2014.03.001. [DOI] [PubMed] [Google Scholar]
- 41.Ghazi A, Stone J, Park J, et al. V8-01 Full Procedural Simulation for Transurethral Resection of Bladder Tumors (TURBT) Using 3-D Printing Technology. J Urol. 2016;195:e771. https://doi.org/10.1016/j.juro.2016.02.744. [Google Scholar]
- 42.Ghazi A, Campbell T, Melnyk R, et al. Validation of a Full-Immersion Simulation Platform for Percutaneous Nephrolithotomy Using Three-Dimensional Printing Technology. J Endourol. 2017;31:1314–20. doi: 10.1089/end.2017.0366. https://doi.org/10.1089/end.2017.0366. [DOI] [PubMed] [Google Scholar]
- 43.Sweet RM. The CREST Simulation Development Process:Training the Next Generation. J Endourol. 2017;31:S69–75. doi: 10.1089/end.2016.0613. [DOI] [PubMed] [Google Scholar]
- 44.Parkhomenko E, Yoon R, Okhunov Z, et al. Multi-Institutional Evaluation of Producing and Testing a Novel 3D-Printed Laparoscopic Trainer. Urology. 2019;124:297–301. doi: 10.1016/j.urology.2018.06.034. https://doi.org/10.1016/j.urology.2018.06.034. [DOI] [PubMed] [Google Scholar]
- 45.Uwechue R, Gogalniceanu P, Kessaris N, et al. A Novel 3D-Printed Hybrid Simulation Model for Robotic-Assisted Kidney Transplantation (RAKT) J Robot Surg. 2018;12:541–4. doi: 10.1007/s11701-018-0780-y. https://doi.org/10.26226/morressier .5a38ffa4d462b8029238b6d9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Golab A, Smektala T, Krolikowski M, et al. Percutaneous Nephrolithotomy Using an Individual 3-Dimensionally Printed Surgical Guide. Urol Int. 2018;100:485–7. doi: 10.1159/000446291. https://doi.org/10.1159/000446291. [DOI] [PubMed] [Google Scholar]
- 47.Canat L, Atalay HA, Değirmentepe RB, et al. Stone Volume Measuring Methods:Should the CT Based Three-Dimensional-Reconstructed Algorithm be Proposed as the Gold Standard?What did the Three-Dimensional Printed Models Show us? Arch Esp Urol. 2019;72:596–601. [PubMed] [Google Scholar]
- 48.Lupulescu C, Sun Z. A Systematic Review of the Clinical Value and Applications of Three-Dimensional Printing in Renal Surgery. J Clin Med. 2019;8:990. doi: 10.3390/jcm8070990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Fan G, Meng Y, Zhu S, et al. Three-Dimensional Printing for Laparoscopic Partial Nephrectomy in Patients with Renal Tumors. J Int Med Res. 2019;47:4324–32. doi: 10.1177/0300060519862058. https://doi.org/10.1177/0300060519↬58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Michiels C, Jambon E, Bernhard JC. Measurement of the Accuracy of 3D-Printed Medical Models to Be Used for Robot-Assisted Partial Nephrectomy. AJR Am J Roentgenol. 2019;213:626–31. doi: 10.2214/AJR.18.21048. https://doi.org/10.2214/ajr.18.21048. [DOI] [PubMed] [Google Scholar]
- 51.Mercader C, Vilaseca A, Moreno JL, et al. Role of the Three-Dimensional Printing Technology Incomplex Laparoscopic Renal Surgery:A Renal Tumor in a Horseshoe Kidney. Int Braz J Urol. 2019;45:1129–35. doi: 10.1590/S1677-5538.IBJU.2019.0085. https://doi.org/10.1590/s1677-5538.ibju.2019.0085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Denizet G, Calame P, Lihoreau T, et al. 3D Multi-Tissue Printing for Kidney Transplantation. Quant Imaging Med Surg. 2019;9:101–6. doi: 10.21037/qims.2018.10.16. https://doi.org/10.21037/qims.2018.10.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guo YT, Wang H, Wang JP, et al. Two-Year Follow-up on Laparoscopic Three-Dimensional Printed Extravascular Stent Placement for Posterior Nutcracker Syndrome. Chin Med J (Engl) 131:2895–6. doi: 10.4103/0366-6999.246075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wang H, Guo YT, Jiao Y, et al. A Minimally Invasive Alternative for the Treatment of Nutcracker Syndrome using Individualized Three-Dimensional Printed Extravascular Titanium Stents. Chin Med J (Engl) 2019;132:1454–60. doi: 10.1097/CM9.0000000000000255. https://doi.org/10.1097/cm9.0000000000000255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhang J, Zhang P, Wu L, et al. Application of an Individualized and Reassemblable 3D Printing Navigation Template for Accurate Puncture During Sacral Neuromodulation. Neurourol Urodyn. 2018;37:2776–81. doi: 10.1002/nau.23769. https://doi.org/10.1002/nau.23769. [DOI] [PubMed] [Google Scholar]
- 56.Hassani FA, Peh WY, Gammad GG, et al. A 3D Printed Implantable Device for Voiding the Bladder Using Shape Memory Alloy (SMA) Actuators. Adv Sci (Weinh) 2017;4:1700143. doi: 10.1002/advs.201700143. https://doi.org/10.1002/advs.201700143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Barsky M, Kelley R, Bhora FY, et al. Customized Pessary Fabrication Using Three-Dimensional Printing Technology. Obstet Gynecol. 2018;131:493–7. doi: 10.1097/AOG.0000000000002461. https://doi.org/10.1097/aog.0000000000002461. [DOI] [PubMed] [Google Scholar]
- 58.Serrano-Aroca Á, Vera-Donoso CD, Moreno-Manzano V. Bioengineering Approaches for Bladder Regeneration. Int J Mol Sci. 2018;19:1796. doi: 10.3390/ijms19061796. https://doi.org/10.3390/ijms19061796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Adamowicz J, Kuffel B, Van Breda SV, et al. Reconstructive Urology and Tissue Engineering:Converging Developmental Paths. J Tissue Eng Regen Med. 2019;13:522–33. doi: 10.1002/term.2812. https://doi.org/10.1002/term.2812. [DOI] [PubMed] [Google Scholar]
- 60.Bejrananda T, Liawrungrueang W. Successful Transitional Cell Carcinoma of Bladder Underwent Laparoscopic Radical Cystectomy with Orthotopic Intracorporeal Y Pouch Neobladder Using a 3D Digital Printing Model for Surgical Post OP Pouch Evaluation. Urol Case Rep. 2020;31:101190. doi: 10.1016/j.eucr.2020.101190. https://doi.org/10.1016/j.eucr.2020.101190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kim MJ, Chi BH, Yoo JJ, et al. Structure Establishment of Three-Dimensional (3D) Cell Culture Printing Model for Bladder Cancer. PLoS One. 2019;14:e0223689. doi: 10.1371/journal.pone.0223689. https://doi.org/10.1371/journal.pone.0223689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang Y, Gao X, Yang Q, et al. Three-Dimensional Printing Technique Assisted Cognitive Fusion in Targeted Prostate Biopsy. Asian J Urol. 2015;2:214–9. doi: 10.1016/j.ajur.2015.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Porpiglia F, Bertolo R, Checcucci E, et al. Development and Validation of 3D Printed Virtual Models for Robot-Assisted Radical Prostatectomy and Partial Nephrectomy:Urologists'and Patients'Perception. World J Urol. 2018;36:201–7. doi: 10.1007/s00345-017-2126-1. https://doi.org/10.1007/s00345-017-2126-1. [DOI] [PubMed] [Google Scholar]
- 64.Shin T, Ukimura O, Gill IS. Three-Dimensional Printed Model of Prostate Anatomy and Targeted Biopsy-Proven Index Tumor to Facilitate Nerve-Sparing Prostatectomy. Eur Urol. 2016;69:377–9. doi: 10.1016/j.eururo.2015.09.024. https://doi.org/10.1016/j.eururo.2015.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chandak P, Byrne N, Lynch H, et al. Three-Dimensional Printing in Robot-Assisted Radical Prostatectomy-An Idea, Development, Exploration, Assessment, Long-Term follow-up (IDEAL) Phase 2a study. BJU Int. 2018;122:360–1. doi: 10.1111/bju.14189. https://doi.org/10.1111/bju.14189. [DOI] [PubMed] [Google Scholar]
- 66.Ebbing J, Jäderling F, Collins JW, et al. Comparison of 3D Printed Prostate Models with Standard Radiological Information to Aid Understanding of the Precise Location of Prostate Cancer:A Construct Validation Study. PLoS One. 2018;13:e0199477. doi: 10.1371/journal.pone.0199477. https://doi.org/10.1371/journal.pone.0199477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Del Junco M, Yoon R, Okhunov Z, et al. Comparison of Flow Characteristics of Novel Three-Dimensional Printed Ureteral Stents Versus Standard Ureteral Stents in a Porcine Model. J Endourol. 2015;29:1065–9. doi: 10.1089/end.2014.0716. https://doi.org/10.1089/end.2014.0716. [DOI] [PubMed] [Google Scholar]
- 68.Park CJ, Kim HW, Jeong S, et al. Anti-Reflux Ureteral Stent with Polymeric Flap Valve Using Three-Dimensional Printing:An In Vitro Study. J Endourol. 2015;29:933–8. doi: 10.1089/end.2015.0154. https://doi.org/10.1089/end.2015.0154. [DOI] [PubMed] [Google Scholar]
- 69.Kuroda S, Kawahara T, Teranishi J, et al. A Case of Allograft Ureteral Stone Successfully Treated with Antegrade Ureteroscopic Lithotripsy:Use of a 3D-Printed Model to Determine the Ideal Approach. Urolithiasis. 2019;47:467–71. doi: 10.1007/s00240-019-01153-x. https://doi.org/10.1007/s00240-019-01153-x. [DOI] [PubMed] [Google Scholar]
- 70.Joshi PM, Kulkarni SB. 3D Printing of Pelvic Fracture Urethral Injuries-Fusion of Technology and Urethroplasty. Turk J Urol. 2020;46:76–9. doi: 10.5152/tud.2019.19165. https://doi.org/10.5152/tud.2019.19165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Zhang K, Fu Q, Yoo J, et al. 3D Bio-Printing of Urethra with PCL/PLCL Blend and Dual Autologous Cells in Fibrin Hydrogel:An In Vitro Evaluation of Biomimetic Mechanical Property and Cell Growth Environment. Acta Biomater. 2017;50:154–64. doi: 10.1016/j.actbio.2016.12.008. https://doi.org/10.1016/j.actbio.2016.12.008. [DOI] [PubMed] [Google Scholar]
- 72.D'Alimonte L, Ravi A, Helou J, et al. Optimized Penile Surface Mold Brachytherapy using Latest Stereolithography Techniques:A Single-Institution Experience. Brachytherapy. 2019;18:348–52. doi: 10.1016/j.brachy.2019.01.002. https://doi.org/10.1016/j.brachy.2019.01.002. [DOI] [PubMed] [Google Scholar]
- 73.Yu HS, Park J, Lee HS, et al. Feasibility of Polycaprolactone Scaffolds Fabricated by Three-Dimensional Printing for Tissue Engineering of Tunica Albuginea. World J Mens Health. 2018;36:66–72. doi: 10.5534/wjmh.17025. https://doi.org/10.5534/wjmh.17025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Srougi V, Rocha BA, Tanno FY, et al. The Use of Three-dimensional Printers for Partial Adrenalectomy:Estimating the Resection Limits. Urology. 2016;90:217–20. doi: 10.1016/j.urology.2015.11.043. https://doi.org/10.1016/j.urology.2015.11.043. [DOI] [PubMed] [Google Scholar]
- 75.Lurie KL, Smith GT, Khan SA, et al. Three-Dimensional, Distendable Bladder Phantom for Optical Coherence Tomography and White Light Cystoscopy. J Biomed Opt. 2014;19:36009. doi: 10.1117/1.JBO.19.3.036009. https://doi.org/10.1117/1.jbo.19.3.036009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chen MY, Skewes J, Daley R, et al. Three-Dimensional Printing Versus Conventional Machining in the Creation of a Meatal Urethral Dilator:Development and Mechanical Testing. Biomed Eng Online. 2020;19:55. doi: 10.1186/s12938-020-00799-8. https://doi.org/10.1186/s12938-020-00799-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Özgür BC, Ayyıldız A. 3D Printing in Urology:Is it Really Promising? Turk J Urol. 2018;44:6–9. doi: 10.5152/tud.2018.20856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Manning TG, O'Brien JS, Christidis D, et al. Three Dimensional Models in Uro-Oncology:A Future Built with Additive Fabrication. World J Urol. 2018;36:557–63. doi: 10.1007/s00345-018-2201-2. https://doi.org/10.1007/s00345-018-2201-2. [DOI] [PubMed] [Google Scholar]


