Abstract
Background
Suspicion and clinical criteria continue to serve as the foundation for ventilator-associated pneumonia (VAP) diagnosis, however the criteria used to diagnose VAP vary widely. Data from head-to-head comparisons of clinical diagnostic algorithms is lacking, thus a prospective observational study was performed to determine the performance characteristics of the Johanson criteria, Clinical Pulmonary Infection Score (CPIS), and Centers for Disease Control and Prevention’s National Healthcare Safety Network (CDC/NHSN) criteria as compared to Hospital in Europe Link for Infection Control through Surveillance (HELICS) reference standard.
Methods
A prospective observational cohort study was performed in three mixed medical-surgical ICUs from one academic medical center from 1 October 2016 to 30 April 2018. VAP diagnostic criteria were applied to each patient including CDC/NHSN, CPIS, HELICS and Johanson criteria. Tracheal aspirate cultures (TAC) and serum procalcitonin values were obtained for each patient.
Results
Eighty-five patients were enrolled (VAP 45, controls 40). Using HELICS as the reference standard, the sensitivity and specificity for each of the assessed diagnostic algorithms were: CDC/NHSN (Sensitivity 54.2%; Specificity 100%), CPIS (Sensitivity 68.75%; Specificity 95.23%), Johanson (Sensitivity 67.69%; Specificity 95%). The positive TAC rate was 81.2%. The sensitivity for positive TAC with the serum procalcitonin level > 0.5 ng/ml was 51.8%.
Conclusion
VAP remains a considerable source of morbidity and mortality in modern intensive care units. The optimal diagnostic method remains unclear. Using HELICS criteria as the reference standard, CPIS had the greatest comparative diagnostic accuracy, whereas the sensitivity of the CDC/NHSN was only marginally better than a positive TAC plus serum procalcitonin > 0.5 ng/ml. Algorithm accuracy was improved by adding serum procalcitonin > 0.5 ng/ml, but not positive quantitative TAC.
Trial Registration: Not indicated for this study type.
Keywords: Ventilator-associated pneumonia, Cross infection, Artificial respiration, Critical care
Background
The incidence of nosocomial infections (NI) amongst intensive care unit (ICU) patients is 2–5 times that of general admissions [1]. Amongst the most prevalent and threatening ICU NIs is ventilator-associated pneumonia (VAP), which may develop in patients receiving invasive mechanically ventilated (MV) for ≥ 48 h [2–6]. VAP has a cumulative incidence of 10–45%, and an attributable risk of 5–27% [7–12]. VAP-associated comorbidities include prolonged duration of MV, delayed MV weaning, increased antibiotic consumption, prolonged ICU and hospital length-of-stay (LOS), increased treatment-related expenditures, and increased crude and attributed mortality with recent studies reappraising the impact of VAP on mortality to be 10% [2–6, 13–17]. Accordingly, VAP prevention has emerged as a high priority. As such, one component of the Institute for Healthcare Improvement’s recommended ventilator bundle is the accurate diagnosis and determination of VAP incidence [18–20]. However, the optimal VAP diagnostic strategy remains contentious. Research in this field is limited by the lack of a consensus ‘gold standard’ definition against which to test the diagnostic accuracy of new diagnostic algorithms or methods of detection. VAP diagnosis remains challenging as clinical signs and symptoms may be non-specific, with clinical diagnosis being overly sensitive (leading to increased antibiotic use), and histopathology (ante- or post-mortem within 96 h of death) being limited in availability, consistency, standardization and reliability [21–23]. Moreover, quantitative respiratory cultures have been found to correlate poorly with histopathology [22, 24].
As none of the available diagnostic tests, performed alone, can provide an accurate diagnosis of VAP, a diagnostic strategy incorporating several criteria has been viewed by many to be a good compromise. To this end, great effort has been expended to generate standardized diagnostic algorithms that incorporate clinical, radiographic and microbiological data. Some examples (Table 1) include: Centers for Disease Control and Prevention’s National Healthcare Safety Network (CDC/NHSN) [25], Clinical Pulmonary Infection Score (CPIS) [26], Hospital in Europe Link for Infection Control through Surveillance (HELICS) [27], Johanson criteria [28], and others [29, 30]. As compared to immediate post-mortem lung biopsies, clinical criteria have reasonable diagnostic performance but may be highly impacted by the diagnostic thresholds used, and the lack of a uniform reference diagnostic standard has contributed to variable diagnostic performance (Table 2) and made inter-study comparisons difficult [31]. A highly performing VAP diagnostic method is greatly needed, but international guidelines disagree on the use of clinical algorithms for risk stratification to determine treatment [32, 33]. Data comparing algorithm performance head-to-head is lacking, and as most such data stems from high-income countries. Great need exists for head-to-head comparisons, as well as data from low-to-middle income countries to supplement the international data pool. To this end, a prospective non-randomized study was conducted to determine if in patients with VAP, does application of the CDC/NHSN, CPIS, or Johanson criteria provide the greatest diagnostic performance characteristics as compared to HELICS as the reference standard.
Table 1.
Ventilator-associated pneumonia diagnostic algorithms utilized in this study
| Published Criteria (citation) | Systemic Criteria | Chest Criteria | Chest Radiography Criteria | Microbiologic Criteria |
|---|---|---|---|---|
| CDC/NHSN (25) |
- Inflammatory response Temperature > 38 °C WBC > 12,000/mm3 or < 4,000/mm3 - OR new antimicrobial agent is started for ≥ 4 days → Infection-related ventilator-associated complication |
After a period of stability or improvement on the ventilator (≥ 2 calendar days of stable or ↓ FiO2 or PEEP): - Minimum daily FiO2 ↑ ≥ 0.20 lasting 2 days - Or minimum daily PEEP values ↑ ≥ 3 cm H2O lasting 2 days → Ventilator-associated condition |
– |
Microbiologic quantitative ( +), OR histologic ( +), OR ( +) for legionella, influenza, RSV, adenovirus, or parainfluenza virus AND Gram-stain evidence ≥ 25 neutrophils/lpf and ≤ 10 epithelial cells/lpf → Probable VAP |
|
CPISa (26) |
Fever: - 38.5–38.9 (1 point) - ≥ 39 or < 36.5 (2 points) WBC: - < 4,000/mm3 or > 11,000/mm3 (2 points) |
- Secretions but not purulent (1 point) - Purulent secretions (2 points) - PaO2/ FiO2 < 240 without ARDS (2 points) |
Diffuse infiltrate (1 point) Localized infiltrate (2 points) Progressive infiltrate (without cardiac disease or ARDS) (+ 2) |
Positive (1 point) |
|
HELICSb (27) |
At least 1 criterion: - Temperature > 38 °C (with no other cause) - WBC > 12,000/mm3 or < 4,000/mm3 - If age > 70 years: AMS without other cause |
At least 1 of following criteria (2 clinical pneumonia only = PN4 and PN5): New onset purulent sputum or change in sputum character (color, odor, quantity, consistency) Cough or dyspnea or tachypnea Suggestive auscultation (rales or bronchial breath sounds, rhonchi, wheezing) Worsening gas exchange (O2 desaturation, increasing FiO2 requirements or ventilation demands) |
Image suggestive of pneumonia. (≥ 2 serial chest X-rays or CT scans with suggestive imaging for patients with underlying cardiac or pulmonary disease) |
PN1 – ( +) quantitative Cx from minimally contaminated LRT specimen c PN2 – ( +) quantitative Cx from possibly contaminated LRT specimen d PN3 – Alternative methods: e ( +) blood or pleural Cx, pleural or pulmonary abscess, histology, or pathogen antigen or antibody testing PN4 – ( +) sputum Cx or non-quantitative LRT specimen Cx PN5 – No positive results |
|
Johanson (28) |
Temperature > 38.5 °C WBC > 12,000/mm3 |
Purulent secretions | New or progressive consolidation | – |
AMS: altered mental status; ARDS: acute respiratory distress syndrome; CDC/NHSN: centers for disease control and prevention national health safety network; CPIS: clinical pulmonary infection score; Cx: culture; FiO2: fraction of inspired oxygen; HELICS: hospital in Europe link for infection control through surveilance; LRT: lower respiratory tract; PaO2: partial pressure of oxygen in arterial blood; RSV: respiratory syncytial virus; VAP: ventilator associated pneumonia; WBC: white blood cell
aScore > 6 is suggestive of VAP
bVAP diagnosis if criteria met and invasive respiratory device (even intermittently) in the 48 h preceding the onset of infection
cEither: (1) Broncho-alveolar lavage (BAL) with a threshold of ≥ 104 cfu/mL or ≥ 5% of BAL obtained cells contains intracellular bacteria on direct microscopic exam; (2) Protected brush (PB Wimberley) with a threshold of ≥ 103 cfu/mL; (3) Distal protected aspirate (DPA) with a threshold of ≥ 103 cfu/mL
dQuantitative culture of LRT specimen (e.g., endotracheal aspirate) with a threshold of 106 cfu/mL
eEither: (1) positive blood culture not related to another source of infection; (2) positive growth in culture of pleural fluid; (3) pleural or pulmonary exam shows evidence of pneumonia; (4) positive exams for pneumonia with virus or particular germs (Legionella, Aspergillus, mycobacteria, Mycoplasma, Pneumocystis carinii). The latter may include: (A) positive detection of viral antigen or antibody from respiratory secretions (e.g., EIA, FAMA, shell vial assay, PCR); (B) positive direct exam or positive culture from bronchial secretions or tissue; (C) seroconversion (ex: influenza viruses, Legionella, Chlamydia); or (D) detection of antigens in urine (Legionella)
Table 2.
Performance characteristics of ventilator-associated pneumonia diagnostic algorithms
| Criteria Studied | Year, (citation) | Population | Comparator | Sample Size | Sensitivity | Specificity | PPV | NPV | ( +) LR | (-) LR | ROC AUC | Kappa (ĸ) index, agreement level a |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CDC/NHSN | 2015, (60) | Mixedb | CPIS | 38 | 0.37 | 1.0 | 1.0 | 0.84 | ĸ = 0.47, moderate | |||
| CPIS | 1999, (43) | Mixedb | Pathology | 23 | 0.77 | 0.42 | ||||||
| CPISc | 2004, (66) | Mixedb | Quantitative Cultures | 69 | 0.41 | 0.77 | 0.8 | 0.36 | 0.64 | |||
| CPIS | 2004, (67) | Mixedb | Quantitative Cultures | 88 | 0.89 | 0.47 | 0.57 | 0.84 | ĸ = 0.33, fair | |||
| CPIS | 2007, (68) | Burn | Quantitative Cultures | 28 | 0.30 | 0.80 | 0.70 | 0.50 | ||||
| CPIS | 2010, (40) | Mixedb | Pathology | 142 | 0.46 | 0.60 | 1.13 | 0.96 | ||||
| CPIS | 2015, (69) | Surgical (mixed) | Quantitative Cultures | 497 | 0.633 | 0.644 | 0.61 | 0.674 | 0.60 | |||
| CPIS | 2018, (70) | Surgical (acute care) | Quantitative Cultures | 198 | 0.611 | 0.781 | 0.64 | 0.759 | ||||
| HELICS | 2013, (36) | Mixedb | Not clearly specified | 57d | 0.86 | 0.99 | 0.77 | 0.995 | ĸ = 0.80, substantial | |||
| Johansene | 1999, (43) | Mixedb | Pathology | 23 | 0.69 | 0.75 | ||||||
| Johansen | 2018, (70) | Surgical (acute care) | Quantitative Cultures | 198 | 0.828 | 0.59 | 0.564 | 0.843 | ||||
| NTDB/NTR | 2015, (71) | Trauma | CDC/NHSN | 279 | 0.864 | 0.578 | 0.74 | 0.74 | ĸ = 0.47, moderate |
CDC/NHSN: centers for disease control and prevention national health safety network; ICU: intensive care unit; MV: mechanical ventilation; NR: not reported; NTDB/NTR: national trauma data bank / national trauma registry; NPV: negative predictive value; PPV: positive predictive value; ROC AUC: receiver operating curve area under curve; ( +) LR: positive likelihood ratio; (-) LR: negative likelihood ratio
aAgreement based on score: ≤ 0 (no agreement); 0.01–0.20 (slight); 0.21–0.40 (fair); 0.41– 0.60 (moderate); 0.61–0.80 (substantial); and 0.81–1.00 (almost perfect agreement)
bA mixed population containing both medical and surgical patients. Studies that did not specify ICU type were by default classified as mixed
cFor CPIS threshold of > 7, rather than current standard of > 6. The AUC using threshold CPIS > 6 was 0.54, other values not reported
dData from sub-population of a larger study assessing various types of ICU-acquired infections
eThe presence of all three criteria increased the specificity to 92% at the cost of a high beta error (sensitivity 23%)
Methods
A prospective observational cohort study was performed in three mixed medical-surgical ICUs from one academic medical center from 1 October 2016 to 30 April 2018. The study was approved by the Investigational Review Board at Hamadan University of Medical Sciences, Hamadan, Iran (IR.UMSAHA.REC.1395.23). All study parts were reviewed according to the Strengthening the Reporting of Observational Studies in Epidemiology ‘STROBE’ guideline [34]. Written consent was required and covered both study participation and publication of de-identified aggregate findings. Surrogate consent from the patient’s legal guardian or designated health proxy was permitted in cases where the subject lacked decision-making capacity. All patients that survived and regained their faculties were informed of the project. All data generated or analyzed during this study are included in this article. De-identified individual subject data may be available from the corresponding author on reasonable request.
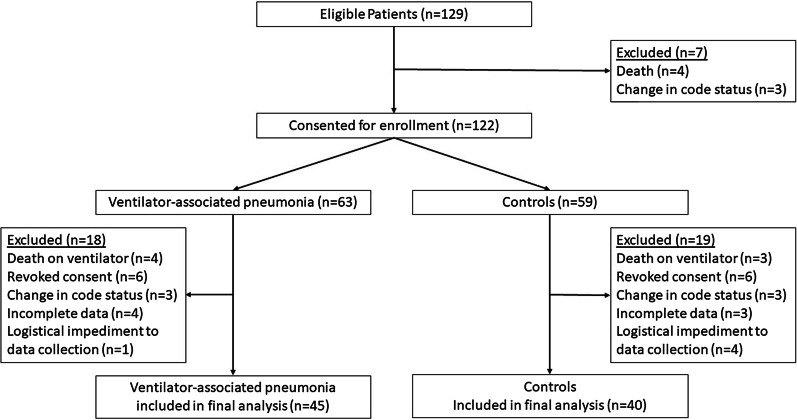
Patients were eligible for study participation if: (1) age ≥ 18 years, (2) admitted to the ICU > 48 h, (3) receiving invasive MV > 48 h (any mode except high frequency percussive ventilation or high frequency oscillatory ventilation), (4) full-code status, and (5) informed consent obtained from the patient, legal guardian or healthcare surrogate upon ICU admission (prior to intubation). Patients with any limitation of code status including (but not limited to) No Code, Do Not Resuscitate, or Do Not Intubate were excluded (Fig. 1). Patients with known pregnancy were excluded.
Fig. 1.
Patient flow diagram
Patient selection was performed by an enrollment team of two physicians (1 critical care, 1 infectious disease) not directly involved in the study. All consecutive patients identified at the participating ICUs with VAP according to the HELICS criteria were eligible. Each case patient was matched by the enrollment team, which was blinded to the outcome, with another ICU patient that did not have VAP. Matching was based on: (1) admission indication; (2) ICU LOS ≥ 48 h; (3) receiving invasive MV > 48 h (any mode except high frequency percussive ventilation or high frequency oscillatory ventilation as these preclude proper calculation of the CBC/NHSN criteria); (4) severity of illness at ICU admission as quantified by the Acute Physiology and Chronic Health Evaluation (APACHE) II score > 15, (5) full code status, and (6) age ≥ 18 years.
VAP diagnosis was made independently by the treating clinical team. Diagnostic criteria were according to HELICS criteria [27] in accordance with the institutional standard and other published studies [2, 35–38] as it is the definition currently used in much of Europe, Australia, and the near- and middle east (including Iran). Chest radiograph interpretation was undertaken “off-line” by a team of 3 physicians (1 radiology, 1 critical care, 1 pulmonology) who were independent of the treating team. Kendal agreement coefficient between the clinicians in chest radiograph interpretation was 0.99. Procalcitonin was measured at the time of initial VAP suspicion. A single value was used, and thresholds were in accordance with prior published studies [39].
Specimen collection and processing
Protected tracheal aspirate (TA) samples were obtained through a sterile 12 French catheter (SUPA Medical Devices, Tehran, Iran). This catheter is placed in the trachea by advancing through the endotracheal tube until resistance was encountered (level of the carina) and retracted approximately 2 cm. To obtain TA samples, 5–10 mL of sterile saline was instilled followed by aspiration into a sterile syringe. This generally yielded an aspirate of 2-3 cc. The samples were then transferred to the microbiology laboratory for processing and examination within 30 min. The materials were evaluated by gram-stain and quantitative cultures. Light microscopy was utilized to assess gram stains for bacteria and white blood cells. The samples were vortexed for one minute at 3,000 rpm, diluted with saline to 1:10 ratio, and 0.01 cc inoculated onto blood agar, chocolate agar, and MacConkey agar plates. Cultures were incubated at 35 ± 1ºC for 24, followed by quantitative bacterial evaluation. The cut-off values for bacterial colony counts were taken as ≥ 105 colony forming units (CFU)/cc. When more than one bacteria type was identified, a separate colony count was performed for each. Microbial identification and antimicrobial susceptibility testing were performed using the automated Vitek® 2 Advanced Expert System (bioMérieux, Marcy-l'Étoile, France).
The criteria for sample rejection were: (1) improperly labeled specimens, (2) specimens with transport times exceeding study standards, (3) clotted specimens, (4) specimens not submitted in an appropriate transport container, (5) insufficient volume, or (6) external contamination. If an unacceptable specimen was received, the treatment team was notified, and another specimen was requested.
Data collection
Screening, data collection and reporting was undertaken by a trained, dedicated full-time nurse. The data collection tool was a two-part checklist including demographic variables, clinical and microbiological variables. The tool was developed during two 90-min meetings by a consensus multidisciplinary panel consisting of 17 physicians representing critical care (n = 5), anesthesia (n = 3), pulmonology (n = 5), internal medicine (n = 3), and forensic medicine (n = 1), and 10 critical care nurses. The Quantitative face validity was determined using Impact Score (2.5–4.5), and quantitative content validity was determined via 27 panelists. The measured content validity ratio and content validity index were 0.51 and 0.89 respectively. The internal validity of the questionnaire was determined by Cronbach's alpha coefficient to be 0.91.
Statistics
Statistical analyses were performed using IBM® SPSS version 22.0 (IBM Corp, Armonk, USA). Data were summarized using mean ± standard deviation (SD) for quantitative variables and frequency (%) for qualitative variables. Study size was determined by a prior sample size calculation. Considering a VAP prevalence of 0.5, 95% confidence interval level, 80% power, and absolute error 10%, the necessary sample size was calculated to be 85 patients.
Normally distributed variables were compared using the Student’s t-test. Categorical variables were compared using Chi-square (χ2) test or Fisher's exact test when appropriate. Trend of change in distribution of relative frequencies between ordinal data were compared using χ2 test for trend. The Youden index (or Youden’s J Statistic) was calculated as: J = sensitivity + specificity – 1.
Results
One-hundred twenty-nine patients were screened, and 85 were included in the final analysis (Fig. 1). The mean age was 46.94 ± 18.90 years with a male predominance (72.9%). Measures of illness severity and hospital course metrics are listed in Table 3. Positive tracheal culture was seen in 81.2% with cultures yielding Acinetobacter (37.6%), Staphylococcus aureus (22.4%), Escherichia coli (14.1%), Pseudomonas (10.6%), Klebsiella (10.6%), and Proteus (3.5%). Multiple drug resistant (MDR) organisms were identified in 36.5% of isolates. The sensitivity and specificity of the tested algorithms are presented in Table 4. Of note, the sensitivity for positive TAC with the serum procalcitonin level > 0.5 ng/ml was 51.8%, lower than each of the algorithms assessed. The highest Youden index, a measure of diagnostic accuracy, was seen with CPIS (Table 4).
Table 3.
Patient demographic and clinical information
| Variable | All | VAP n = 45 |
No VAP n = 40 |
p-value |
|---|---|---|---|---|
| Age, years, mean (SD) | 46.9 (18.9) | 44.2 (20.7) | 49.9 (16.4) | 0.159a |
| Male, N (%) | 62 (72.9) | 33 (73.3) | 29 (72.5) | 0.931c |
| Admission indication, N (%) | 0.652c | |||
| Trauma | 54 (63.5) | 30 (66.7) | 24 (60) | |
| Post-operative | 31 (36.5) | 15 (33.3) | 16 (40) | |
| Comorbidities, yes, N (%) | 0.932b | |||
| ARDS | 7 (8.2) | 3 (6.7) | 4 (10) | |
| Cancer | 13 (15.3) | 6 (13.3) | 7 (17.5) | |
| COPD | 7 (8.2) | 4 (8.9) | 3 (7.5) | |
| CHF | 24 (28.2) | 13 (28.9) | 11 (27.5) | |
| ESRD | 14 (16.5) | 9 (20) | 5 (12.5) | |
| Multiple trauma | 20 (23.5) | 10 (22.2) | 10 (25) | |
| Positive tracheal culture, N (%) | 69 (81.2) | 40 (88.9) | 29 (72.5) | 0.093c |
| MDR organism, yes, N (%) | 31 (36.5) | 17 (37.8) | 14 (35) | 0.825c |
| Procalcitonin, ng/mL, mean (SD) | 4.03 (4.68) | 3.53 (3.6) | 4.6 (5.6) | 0.308a |
| APACHE II, mean (SD) | 18.1 (2.84) | 17.9 (3.43) | 18.4 (1.98) | 0.399a |
| Duration of intubation, hours, mean (SD) | 177.1 (39.61) | 176.02 (38.7) | 178.32 (41.09) | 0.791a |
| Reintubation, N (%) | 32 (37.6) | 14 (31.1) | 18 (45) | 0.262c |
| MV duration prior to VAP, hours, median (IQR) | 72 (54–87.5) | 72 (52–87.5) | 72 (64.5–88.5) | 0.639a |
| ICU duration prior to developing VAP, days, median (IQR) | 7 (6–8) | 7 (6–8.5) | 7 (6–8) | 0.118a |
| VAP timing, mean (SD) | ||||
| Early (< 5 days) | – | 15 (33.3) | – | – |
| Late (≥ 5 days) | 30 (66.7) | |||
| Length-of-stay, days, mean (SD) | ||||
| ICU LOS | 9.8 (3.0) | 13.13 (3.27) | 12.72 (2.75) | 0.538a |
| Non-ICU LOS | 15.4 (3.1) | 12.67 (3.34) | 11.96 (2.99) | 0.320a |
| Mortality, N (%) | ||||
| ICU | 17 (20) | 8 (17.8) | 9 (22.5) | 0.787c |
| Hospital | 22 (25.9) | 12 (26.7) | 10 (25) | 0.861c |
VAP: ventilator-associated pneumonia; IQR: interquartile range; MDR: multiple drug resistant; APACHE: Acute Physiology and Chronic Health Evaluation; ICU: intensive care unit; VAP: ventilator-associated pneumonia; LOS: length-of-stay
aIndependent sample t-test
bFisher exact test
cChi-square
Table 4.
Sensitivity, specificity, and Youden index for assessed methods of ventilator-associated pneumonia diagnosis compared to the HELICS criteria as the reference standard
| Criteria | Ventilator-Associate Pneumonia | % Sensitivity | % Specificity | Youden index a | ||
|---|---|---|---|---|---|---|
| Positive | Negative | Total | ||||
| CDC/NHSN | ||||||
| Positive | 45 | 38 | 83 | 54.22 | 100 | 0.542 |
| Negative | 0 | 2 | 2 | |||
| Total | 45 | 40 | 85 | |||
| CPIS | ||||||
| Positive | 44 | 20 | 64 | 68.75 | 95.23 | 0.640 |
| Negative | 1 | 20 | 21 | |||
| Total | 45 | 40 | 85 | |||
| Johanson | ||||||
| Positive | 44 | 21 | 65 | 67.69 | 95 | 0.627 |
| Negative | 1 | 19 | 20 | |||
| Total | 45 | 40 | 85 | |||
CDC/NHSN = centers for disease control and prevention national health safety network; CPIS = Clinical Pulmonary Infection Score, HELICS = Hospital in Europe Link for Infection Control through Surveillance
aA measure of the maximum diagnostic accuracy, where 1 signifies a perfect test and 0 signifies no diagnostic value
The Kappa agreement coefficient results between each diagnostic algorithm and either serum procalcitonin level or positive TAC is highlighted in Table 5. The greatest correlation between positive VAP assessment and serum procalcitonin levels > 0.5 ng/ml was observed with the Johanson method and CPIS (both roughly 70%).
Table 5.
Correlation of serum procalcitonin and tracheal aspirate results with ventilator-associated pneumonia diagnostic algorithms
| Criteria | Serum Procalcitonin Level, ng/mL | Kappa (ĸ) index, agreement level a (p-Value) |
Tracheal Culture | Kappa (ĸ) index, agreement level a (p-Value) |
|||||
|---|---|---|---|---|---|---|---|---|---|
| < 0.25 | 0.25–0.5 | > 0.5 | Total | Positive | Negative | Total | |||
| Johanson, n (%) | |||||||||
| Positive | 10 (15.4) | 9 (13.8) | 46 (70.8) | 65 |
0.47, moderate (< 0.001) |
61 (93.8) | 4 (6.2) | 65 |
0.579, moderate (< 0.001) |
| Negative | 18 (90) | 0 | 2 (10) | 20 | 8 (40) | 12 (60) | 20 | ||
| Total | 28 (32.9) | 9 (10.6) | 48 (56.5) | 85 (100) | 69 (81.2) | 16 (18.8) | 85 (100) | ||
| CDC/NHSN, n (%) | |||||||||
| Positive | 26 (31.3) | 10 (12.0) | 47 (56.6) | 83 |
0.06, slight (0.58) |
67 (80.7) | 16 (19.3) | 83 |
0.04, slight (0.49) |
| Negative | 2 (100) | 0 | 0 | 2 | 2 (100) | 0 | 2 | ||
| Total | 28 (32.9) | 10 (11.8) | 47 (55.3) | 85 (100) | 69 (81.2) | 16 (18.8) | 85 (100) | ||
| CPIS, n (%) | |||||||||
| Positive | 11 (17.5) | 8 (12.7) | 44 (69.8) | 63 |
0.42, moderate (< 0.001) |
61 (96.8) | 2 (3.2) | 63 |
0.663, substantial (< 0.001) |
| Negative | 17 (77.3) | 1 (4.5) | 4 (18.2) | 22 | 8 (36.4) | 14 (63.6) | 22 | ||
| Total | 28 (32.9) | 9 (10.6) | 48 (56.5) | 85 (100) | 69 (81.2) | 16 (18.8) | 85 (100) | ||
CDC/NHSN: centers for disease control and prevention national health safety network; CPIS: Clinical Pulmonary Infection Score, HELICS: Hospital in Europe Link for Infection Control through Surveillance
aAgreement based on score: ≤ 0 (no agreement); 0.01–0.20 (slight); 0.21–0.40 (fair); 0.41– 0.60 (moderate); 0.61–0.80 (substantial); and 0.81–1.00 (almost perfect agreement)
As stated previously, CPIS correlated most closely with the HELICS standard. However, when comparing the three tested algorithms, CPIS displayed near perfect agreement with the much simpler and historical Johanson criteria, whereas CDC/NHSN showed only slight agreement with either of the other algorithms (Table 6). Moreover, CPIS correlated most closely with traditional clinical markers for pneumonia (Table 7).
Table 6.
Kappa agreement coefficient among ventilator-associated pneumonia diagnostic methods
| Criteria | Kappa (ĸ) index, agreement level a | p-Value |
|---|---|---|
| CPIS and Johanson | 0.874 | < 0.001 |
| CDC/NHSN and Johanson | 0.145 | < 0.001 |
| CDC/NHSN and CPIS | 0.129 | 0.015 |
CDC/NHSN: centers for disease control and prevention national health safety network; CPIS: Clinical Pulmonary Infection Score, HELICS: Hospital in Europe Link for Infection Control through Surveillance
aAgreement based on score: ≤ 0 (no agreement); 0.01–0.20 (slight); 0.21–0.40 (fair); 0.41– 0.60 (moderate); 0.61–0.80 (substantial); and 0.81–1.00 (almost perfect agreement)
Table 7.
Correlation of individual variables with ventilator-associated pneumonia diagnostic methods
| Parameter | Kappa agreement coefficient | ||
|---|---|---|---|
| CDC/NHSN | CPIS | Johanson | |
| PCT > 0.5 ng/ml | 0.061 | 0.423 | 0.470 |
| Infiltrate on radiograph | − 0.045 | 0.874 | 0.738 |
| Temperature | − 0.044 | 0.529 | 0.579 |
| WBC | − 0.044 | 0.739 | 0.729 |
| PaO2 | − 0.038 | 0.094 | -0.139 |
| Tracheal culture | 0.044 | 0.663 | 0.579 |
| Blood culture | − 0.011 | 0.238 | 0.165 |
CDC/NHSN: centers for disease control and prevention national health safety network; CPIS: Clinical Pulmonary Infection Score, HELICS: Hospital in Europe Link for Infection Control through Surveillance, PCT: Serum procalcitonin; WBC: White blood cell; PaO2: Partial pressure of O2 in arterial blood
Discussion
Suspicion and clinical criteria continue to serve as the foundation for VAP diagnosis, however the criteria used to diagnose VAP vary widely, impacting reports of incidence and outcomes. Historically, VAP diagnosis has been based on 2 or 3 components: (1) systemic signs of infection, (2) new or worsening infiltrates seen on chest imaging, and (3) microbiologic evidence of pulmonary parenchymal infection when available [40]. However, the false positive rate is high for clinical symptoms (e.g. fever [42%]), purulent airway secretions (67%), and chest roentenograms [41, 42]. Moreover, combining these criteria does little to improve diagnostic performance [43], and the use of histopathology and microbiology alone carries considerable limitations [21–24, 40].
Numerous diagnostic algorithms have been proposed to standardize the diagnosis, allow for easier identification, and improve inter-study comparability. Patient characteristics in our cohort were largely similar to those of other published cohorts, including age [9, 17, 44–48], male gender predominance [9, 45, 49–53], APACHE II score [45–48, 51, 52, 54], MV duration [49, 52, 54–56], re-intubation rates [9, 52, 57], ICU LOS [47–50, 52, 53, 55], and hospital LOS [47, 50, 52, 55]. In particular, the ICU LOS and mortality were similar to other published VAP cohorts in Iran [53, 58, 59]. Moreover, the array of cultured and MDR pathogens, was consistent with prior studies [51].
A direct comparison of the correlation and diagnostic performance of the VAP algorithms is important for both individual patient care and epidemiology, cross-study comparisons, and meta-analyses. If algorithms have suboptimal sensitivity, specificity, or do not correlate well, subsequent meta-analyses and epidemiologic investigations will be flawed from inception. Direct comparisons of the performance characteristics of the CDC/NHSN, CPIS, HELICS, and the historical Johanson criteria have not previously been reported. Moreover, only two studies were identified that compared VAP diagnostic algorithms [31, 60]. HELICS was chosen as the reference standard due to its wide international and regional use (Europe, Australia, Near- and Middle East [including Iran]), and as it has been used as the reference standard for numerous other studies [2, 35–38, 61]. CDC/NHSN and CPIS criteria were chosen as the other two most widely recognized and used criteria (especially in North America). The Johanson criteria was selected as the third comparator for its historical significance. The sensitivity of the CPIS and Johanson methods was moderate, whereas CDC/NHSN was poor. Moreover, the diagnostic agreement was substantial for CPIS, moderate for Johanson, and only slight for CDC/NHSN (Table 5). Algorithm accuracy was improved by adding serum procalcitonin > 0.5 ng/ml, however, similar to prior reports, the addition of microbiological data to the clinical definitions did not significantly improve the sensitivity or specificity [40].
These findings suggest that combining cohorts based on HELICS and CPIS may be reasonable for meta-analysis or population studies, but the same may not be true for studies based on CDC/NHSN criteria as the diagnostic agreement is poor. Moreover, it is recommended that studies report serum procalcitonin values to better refine their data sets to optimize data utility as diagnostic algorithms evolve to best facilitate future meta-analyses and as procalcitonin may correlate with mortality [62]. Lastly, this data highlights how little progress these complicated VAP diagnostic algorithms have made beyond that of the historical and simple Johanson criteria. These algorithms will most certainly undergo modification, and it is important that investigators clearly define their patient populations and present the data in a way that allows the data to inform future decisions as the diagnostic techniques evolve.
Limitations
The non-randomized methodology and absence of histopathology confirmation of VAP diagnosis are limitations of this study. This study was performed in a resource-limited setting in a low-to-middle income country (LMIC) and limiting the study cohort to those with ante- or post-mortem histology would have introduced selection bias and served as a barrier for subject recruitment.
The use of TAC specimens is a minor limitation as positive quantitative TAC’s have been reported to have a high degree of correlation with broncho-alveolar lavage in VAP patients and are a useful minimally invasive diagnostic tool [63–65].
Lastly, the serum procalcitonin values were not significantly elevated in the VAP vs. no-VAP group. Procalcitonin is not specific to infection location (i.e. VAP). It may rise with bacterial infections in other locations as well. The no-VAP group did not equate to “no infection anywhere.” Indeed, infections are common in ICU patients ranging from catheter-associated urinary tract infections and other device infections, to soft-tissue infections or even peritonitis from a perforated viscus. There were some patients in the no-VAP group that had non-pulmonary infections with elevated procalcitonin values that raised the mean. It would not be appropriate to remove these patients from the analysis for the following reasons: (1) it would skew remove the real-world applicability of the data, and (2) the study would fall below the necessary sample size required. Lastly, it’s worth noting that procalcitonin values were not a study endpoint and the study was not powered for this purpose.
Conclusion
Ventilator-associated pneumonia remains a considerable source of morbidity and mortality in modern ICUs. The optimal diagnostic method remains unclear. Using HELICS criteria as the reference standard, CPIS displayed substantial diagnostic agreement whereas CDC/NHSN and Johanson criteria displayed slight and moderate agreement respectively. Accuracy was improved with the addition of serum procalcitonin > 0.5 ng/ml, but not positive quantitative endotracheal aspirate culture. These findings suggest that combining cohorts based on HELICS and CPIS may be reasonable for meta-analysis or population studies, but the same may not be true for studies based on CDC/NHSN criteria.
Acknowledgements
We would like to thank the Clinical Research Development Unit of Hamadan Besat Hospital for cooperation and assistance throughout the period of study. Moreover, we give thanks for guidance and advice from the Clinical Research Development Unit of Baqiyatallah Hospital.
Abbreviations
- VAP
Ventilator-associated pneumonia
- CPIS
Clinical Pulmonary Infection Score
- CDC/NHSN
Centers for Disease Control and Prevention’s National Healthcare Safety HELICS Hospital in Europe Link for Infection Control through Surveillance
- TP
True positive
- FN
False negative
- NI
Nosocomial infection
- ICU
Intensive care unit
- MV
Mechanical ventilation
- LOS
Length-of-stay
- APACHE
Acute Physiology and Chronic Health Evaluation
- SD
Standard deviation
- χ2
Chi-square test
- J
Youden’s J Statistic, or Youden Index
- MDR
Multiple drug resistant
Authors' contributions
The original idea was conceived by FRB and AVA. Study design, implementation, and data acquisition was performed by FRB, MHY and AVA. Data analysis was performed by AVA and ACM. Manuscript writing and revision was performed by ACM and AVA. All authors read and approved the final manuscript.
Funding
No funding was received for this research.
Availability of data and materials
All data generated or analyzed during this study are included in this article. De-identified individual subject data may be available from the corresponding author on reasonable.
Declarations
Ethics approval and consent to participate
The study was approved by the Investigational Review Board at Hamadan University of Medical Sciences, Hamadan, Iran (IR.UMSAHA.REC.1395.23). Written consent was required and covered study participation. Surrogate consent from the patient’s legal guardian or designated health proxy was permitted in cases where the subject lacked decision-making capacity. All patients that survived and regained their faculties were informed of the project.
Consent to publish
Written consent was required and covered study publication of de-identified aggregate findings. Surrogate consent from the patient’s legal guardian or designated health proxy was permitted in cases where the subject lacked decision-making capacity. All patients that survived and regained their faculties were informed of the project.
Competing interests
The authors declare that they have no competing interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Farshid Rahimibashar, Email: Fr_rahimibashar@yahoo.com.
Andrew C. Miller, Email: Taqwa1@gmail.com
Mojtaba H. Yaghoobi, Email: Yaghobi.h@yahoo.com
Amir Vahedian-Azimi, Email: Amirvahedian63@gmail.com.
References
- 1.Li Y, Cao X, Ge H, Jiang Y, Zhou H, Zheng W. Targeted surveillance of nosocomial infection in intensive care units of 176 hospitals in Jiangsu province, China. J Hosp Infect. 2018;99(1):36–41. doi: 10.1016/j.jhin.2017.10.009. [DOI] [PubMed] [Google Scholar]
- 2.Álvarez Lerma F, Sánchez García M, Lorente L, Gordo F, Añón JM, Álvarez J, et al. Guidelines for the prevention of ventilator-associated pneumonia and their implementation. The Spanish “Zero-VAP” bundle. Med Intensiva. 2014;38(4):226–236. doi: 10.1016/j.medin.2013.12.007. [DOI] [PubMed] [Google Scholar]
- 3.Righi E, Aggazzotti G, Ferrari E, Giovanardi C, Busani S, Rinaldi L, et al. Trends in ventilator-associated pneumonia: impact of a ventilator care bundle in an Italian tertiary care hospital intensive care unit. Am J Infect Control. 2014;42(12):1312–1316. doi: 10.1016/j.ajic.2014.08.009. [DOI] [PubMed] [Google Scholar]
- 4.Tedja R, Nowacki A, Fraser T, Fatica C, Griffiths L, Gordon S, et al. The impact of multidrug resistance on outcomes in ventilator-associated pneumonia. Am J Infect Control. 2014;42(5):542–545. doi: 10.1016/j.ajic.2013.12.009. [DOI] [PubMed] [Google Scholar]
- 5.Chen JK-H, Chen T-H, Liu H-E, Kao C-C, Chen CF, Ou T-Y, et al. Bundle care for preventing ventilator-associated pneumonia at a medical center: a preliminary report. J Exp Clin Med. 2014;6(5):157–60. 10.1016/j.jecm.2014.08.003
- 6.Fihman V, Messika J, Hajage D, Tournier V, Gaudry S, Magdoud F, et al. Five-year trends for ventilator-associated pneumonia: Correlation between microbiological findings and antimicrobial drug consumption. Int J Antimicrob Agents. 2015;46(5):518–525. doi: 10.1016/j.ijantimicag.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 7.Hudson JKC, McDonald BJ, MacDonald JC, Ruel MA, Hudson CCC. Impact of subglottic suctioning on the incidence of pneumonia after cardiac surgery: a retrospective observational study. J Cardiothorac Vasc Anesth. 2015;29(1):59–63. doi: 10.1053/j.jvca.2014.04.026. [DOI] [PubMed] [Google Scholar]
- 8.Mohamed KAE. Compliance with VAP bundle implementation and its effectiveness on surgical and medical sub-population in adult ICU. Egypt J Chest Dis Tuberc. 2014;63(1):9–14. doi: 10.1016/j.ejcdt.2013.10.019. [DOI] [Google Scholar]
- 9.Dey A, Bairy I. Incidence of multidrug-resistant organisms causing ventilator-associated pneumonia in a tertiary care hospital: a nine months’ prospective study. Ann Thorac Med. 2007;2(2):52–57. doi: 10.4103/1817-1737.32230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Memish ZA, Cunningham G, Oni GA, Djazmati W. The incidence and risk factors of ventilator-associated pneumonia in a Riyadh hospital. Infect Control Hosp Epidemiol. 2000;21(4):271–273. doi: 10.1086/501758. [DOI] [PubMed] [Google Scholar]
- 11.Rosenthal VD. Device-associated nosocomial infections in limited-resources countries: findings of the International Nosocomial Infection Control Consortium (INICC). Am J Infect Control. 2008;36(10):S171.e7–12. 10.1016/j.ajic.2008.10.009 [DOI] [PubMed]
- 12.Guanche-Garcell H, Requejo-Pino O, Rosenthal VD, Morales-Pérez C, Delgado-González O, Fernández-González D. Device-associated infection rates in adult intensive care units of Cuban university hospitals: International Nosocomial Infection Control Consortium (INICC) findings. Int J Infect Dis. 2011;15(5):e357–e362. doi: 10.1016/j.ijid.2011.02.001. [DOI] [PubMed] [Google Scholar]
- 13.Melsen WG, Rovers MM, Groenwold RHH, Bergmans DCJJ, Camus C, Bauer TT, et al. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2013;13(8):665–671. doi: 10.1016/S1473-3099(13)70081-1. [DOI] [PubMed] [Google Scholar]
- 14.Sharpe JP, Magnotti LJ, Weinberg JA, Swanson JM, Wood GC, Fabian TC, et al. Impact of pathogen-directed antimicrobial therapy for ventilator-associated pneumonia in trauma patients on charges and recurrence. J Am Coll Surg. 2015;220(4):489–495. doi: 10.1016/j.jamcollsurg.2014.12.016. [DOI] [PubMed] [Google Scholar]
- 15.Kollef MH, Hamilton CW, Ernst FR. Economic impact of ventilator-associated pneumonia in a large matched cohort. Infect Control Hosp Epidemiol. 2012;33(3):250–256. doi: 10.1086/664049. [DOI] [PubMed] [Google Scholar]
- 16.Melsen WG, Rovers MM, Koeman M, Bonten MJM. Estimating the attributable mortality of ventilator-associated pneumonia from randomized prevention studies. Crit Care Med. 2011;39(12):2736–2742. doi: 10.1097/CCM.0b013e3182281f33. [DOI] [PubMed] [Google Scholar]
- 17.Nguile-Makao M, Zahar J-R, Français A, Tabah A, Garrouste-Orgeas M, Allaouchiche B, et al. Attributable mortality of ventilator-associated pneumonia: Respective impact of main characteristics at ICU admission and VAP onset using conditional logistic regression and multi-state models. Intensive Care Med. 2010;36(5):781–789. doi: 10.1007/s00134-010-1824-6. [DOI] [PubMed] [Google Scholar]
- 18.Eom JS, Lee M-S, Chun H-K, Choi HJ, Jung S-Y, Kim Y-S, et al. The impact of a ventilator bundle on preventing ventilator-associated pneumonia: a multicenter study. Am J Infect Control. 2014;42(1):34–37. doi: 10.1016/j.ajic.2013.06.023. [DOI] [PubMed] [Google Scholar]
- 19.Resar R, Pronovost P, Haraden C, Simmonds T, Rainey T, Nolan T. Using a bundle approach to improve ventilator care processes and reduce ventilator-associated pneumonia. Jt Comm J Qual patient Saf. 2005;31(5):243–248. doi: 10.1016/s1553-7250(05)31031-2. [DOI] [PubMed] [Google Scholar]
- 20.Samra SR, Sherif DM, Elokda SA. Impact of VAP bundle adherence among ventilated critically ill patients and its effectiveness in adult ICU. Egypt J Chest Dis Tuberc. 2017;66(1):81–86. doi: 10.1016/j.ejcdt.2016.08.010. [DOI] [Google Scholar]
- 21.Kirtland SH, Corley DE, Winterbauer RH, Springmeyer SC, Casey KR, Hampson NB, et al. The diagnosis of ventilator-associated pneumonia. Chest. 1997;112(2):445–457. doi: 10.1378/chest.112.2.445. [DOI] [PubMed] [Google Scholar]
- 22.Fabregas N, Torres A, El-Ebiary M, Ramirez J, Hernandez C, Gonzalez J, et al. Histopathologic and microbiologic aspects of ventilator-associated pneumonia. Anesthesiology. 1996;84(4):760–771. doi: 10.1136/thx.54.10.867. [DOI] [PubMed] [Google Scholar]
- 23.Baselski VS, El-Torky M, Coalson JJ, Griffin JP. The standardization of criteria for processing and interpreting laboratory specimens in patients with suspected ventilator-associated pneumonia. Chest. 1992;102(5):571S–579S. doi: 10.1378/chest.102.5_Supplement_1.571S. [DOI] [PubMed] [Google Scholar]
- 24.Stewart NI, Cuthbertson BH. The problems diagnosing ventilator-associated pneumonia. J Intens Care Soc. 2009;10(4):266–272. doi: 10.1177/175114370901000410. [DOI] [Google Scholar]
- 25.National Healthcare Safety Network. Ventilator Associated Event (VAE). United States Center for Disease Control and Prevention; 2020:1–49. https://www.cdc.gov/nhsn/pdfs/pscmanual/10-vae_final.pdf. (Accessed 12/21/2019).
- 26.Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM. Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and nonbronchoscopic “blind” bronchoalveolar lavage fluid. Am Rev Respir Dis. 1991;143(5 Pt 1):1121–1129. doi: 10.1164/ajrccm/143.5_Pt_1.1121. [DOI] [PubMed] [Google Scholar]
- 27.Hospital in Europe Link for Infection Control through Surveillance (HELICS). Surveillance of Nosocomial Infections in Intensive Care Units. Protocol, Version 6.1. Scientific Institute of Public Health. Brussels; 2004:1–51. https://www.sicsag.scot.nhs.uk/hai/helics_protocol.pdf. (Accessed on 01/18/2020).
- 28.Johanson WG, Pierce AK, Sanford JP, Thomas GD. Nosocomial respiratory infections with gram-negative bacilli. The significance of colonization of the respiratory tract. Ann Intern Med. 1972;77(5):701–706. doi: 10.7326/0003-4819-77-5-701. [DOI] [PubMed] [Google Scholar]
- 29.Miller PR, Meredith JW, Chang MC. Optimal threshold for diagnosis of ventilator-associated pneumonia using bronchoalveolar lavage. J Trauma. 2003;55(2):263–267. doi: 10.1097/01.TA.0000075786.19301.91. [DOI] [PubMed] [Google Scholar]
- 30.Afify MH, Shaheen EA, El-Dahdouh SS, El-Feky HM. Comparison between bronchoscopic BAL and non-bronchoscopic BAL in patients with VAP. Egypt J Chest Dis Tuberc. 2016;65(1):113–119. doi: 10.1016/j.ejcdt.2015.08.001. [DOI] [Google Scholar]
- 31.Ramírez-Estrada S, Lagunes L, Peña-López Y, Vahedian-Azimi A, Nseir S, Arvaniti K, et al. Assessing predictive accuracy for outcomes of ventilator-associated events in an international cohort: The EUVAE study. Intensive Care Med. 2018;44(8):1212–1220. doi: 10.1007/s00134-018-5269-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Torres A, Niederman MS, Chastre J, Ewig S, Fernandez-Vandellos P, Hanberger H, et al. International ERS/ESICM/ESCMID/ALAT guidelines for the management of hospital-acquired pneumonia and ventilator-associated pneumonia: guidelines for the management of hospital-acquired pneumonia (HAP)/ventilator-associated pneumonia (VAP) of the European. Eur Respir J. 2017;50(3):1700582. doi: 10.1183/13993003.00582-2017. [DOI] [PubMed] [Google Scholar]
- 33.Kalil AC, Metersky ML, Klompas M, Muscedere J, Sweeney DA, Palmer LB, et al. Management of adults with hospital-acquired and ventilator-associated pneumonia: 2016 clinical practice guidelines by the Infectious Diseases Society of America and the American Thoracic Society. Clin Infect Dis. 2016;63(5):e61–111. doi: 10.1093/cid/ciw353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.White RG, Hakim AJ, Salganik MJ, Spiller MW, Johnston LG, Kerr L, et al. Strengthening the reporting of observational studies in epidemiology for respondent-driven sampling studies: “STROBE-RDS” statement. J Clin Epidemiol. 2015;68(12):1463–1471. doi: 10.1016/j.jclinepi.2015.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xie D, Xiong W, Lai R, Liu L, Gan X, Wang X, et al. Ventilator-associated pneumonia in intensive care units in Hubei Province, China: a multicentre prospective cohort survey. J Hosp Infect. 2011;78(4):284–288. doi: 10.1016/j.jhin.2011.03.009. [DOI] [PubMed] [Google Scholar]
- 36.López-Pueyo MJ, Olaechea-Astigarraga P, Palomar-Martínez M, Insausti-Ordeñana J, Alvarez-Lerma F, ENVIN–HELICS Study Group. Quality control of the surveillance programme of ICU-acquired infection (ENVIN-HELICS registry) in Spain. J Hosp Infect. 2013;84(2):126–31. 10.1016/j.jhin.2013.02.018 [DOI] [PubMed]
- 37.Duszyńska W, Barteczko B, Kübler A. Monitoring of nosocomial infections using the HELICS network. Anestezjol Intens Ter. 2008;40(1):17–21. [PubMed] [Google Scholar]
- 38.Álvarez Lerma F, Carrasco M, Otal JJ, Palomar M, Olaechea P, Peris X, et al. Invasive device-related infections after heart surgery. Med Intensiva. 2013;37(9):584–592. doi: 10.1016/j.medin.2012.12.005. [DOI] [PubMed] [Google Scholar]
- 39.Bouadma L, Luyt C-E, Tubach F, Cracco C, Alvarez A, Schwebel C, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375(9713):463–474. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 40.Tejerina E, Esteban A, Fernández-Segoviano P, Frutos-Vivar F, Aramburu J, Ballesteros D, et al. Accuracy of clinical definitions of ventilator-associated pneumonia: comparison with autopsy findings. J Crit Care. 2010;25(1):62–68. doi: 10.1016/j.jcrc.2009.05.008. [DOI] [PubMed] [Google Scholar]
- 41.Torres A, El-Ebiary M, Padró L, Gonzalez J, de la Bellacasa JP, Ramirez J, et al. Validation of different techniques for the diagnosis of ventilator-associated pneumonia. Comparison with immediate postmortem pulmonary biopsy. Am J Respir Crit Care Med. 1994;149(2 Pt 1):324–331. doi: 10.1164/ajrccm.149.2.8306025. [DOI] [PubMed] [Google Scholar]
- 42.Jiao J, Wang M, Zhang J, Shen K, Liao X, Zhou X. Procalcitonin as a diagnostic marker of ventilator-associated pneumonia in cardiac surgery patients. Exp Ther Med. 2015;9(3):1051–1057. doi: 10.3892/etm.2015.2175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Fàbregas N, Ewig S, Torres A, El-Ebiary M, Ramirez J, de La Bellacasa JP, et al. Clinical diagnosis of ventilator associated pneumonia revisited: comparative validation using immediate post-mortem lung biopsies. Thorax. 1999;54(10):867–873. doi: 10.1136/thx.54.10.867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jamaati HR, Malekmohammad M, Hashemian MR, Nayebi M, Barsharzad N. Ventilator-associated pneumonia: evaluation of etiology, microbiology and resistance patterns in a tertiary respiratory center. Tanaffos. 2010;9(1):21–27. [Google Scholar]
- 45.Chittawatanarat K, Jaipakdee W, Chotirosniramit N, Chandacham K, Jirapongcharoenlap T. Microbiology, resistance patterns, and risk factors of mortality in ventilator-associated bacterial pneumonia in a Northern Thai tertiary-care university based general surgical intensive care unit. Infect Drug Resist. 2014;7:203–210. doi: 10.2147/IDR.S67267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bloos F, Marshall JC, Dellinger RP, Vincent J-L, Gutierrez G, Rivers E, et al. Multinational, observational study of procalcitonin in ICU patients with pneumonia requiring mechanical ventilation: a multicenter observational study. Crit Care. 2011;15(2):R88. doi: 10.1186/cc10087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Karhu J, Ala-Kokko TI, Ylipalosaari P, Ohtonen P, Laurila JJ, Syrjala H. Hospital and long-term outcomes of ICU-treated severe community- and hospital-acquired, and ventilator-associated pneumonia patients. Acta Anaesthesiol Scand. 2011;55(10):1254–1260. doi: 10.1111/j.1399-6576.2011.02535.x. [DOI] [PubMed] [Google Scholar]
- 48.Hedrick TL, Smith RL, McElearney ST, Evans HL, Smith PW, Pruett TL, et al. Differences in early- and late-onset ventilator-associated pneumonia between surgical and trauma patients in a combined surgical or trauma intensive care unit. J Trauma Inj Infect Crit Care. 2008;64(3):714–720. doi: 10.1097/TA.0b013e31811ec18e. [DOI] [PubMed] [Google Scholar]
- 49.Vallés J, Pobo A, García-Esquirol O, Mariscal D, Real J, Fernández R. Excess ICU mortality attributable to ventilator-associated pneumonia: the role of early vs late onset. Intensive Care Med. 2007;33(8):1363–1368. doi: 10.1007/s00134-007-0721-0. [DOI] [PubMed] [Google Scholar]
- 50.Giard M, Lepape A, Allaouchiche B, Guerin C, Lehot J-J, Robert M-O, et al. Early- and late-onset ventilator-associated pneumonia acquired in the intensive care unit: comparison of risk factors. J Crit Care. 2008;23(1):27–33. doi: 10.1016/j.jcrc.2007.08.005. [DOI] [PubMed] [Google Scholar]
- 51.Restrepo MI, Peterson J, Fernandez JF, Qin Z, Fisher AC, Nicholson SC. Comparison of the bacterial etiology of early-onset and late-onset ventilator-associated pneumonia in subjects enrolled in 2 large clinical studies. Respir Care. 2013;58(7):1220–1225. doi: 10.4187/respcare.02173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pawar M, Mehta Y, Khurana P, Chaudhary A, Kulkarni V, Trehan N. Ventilator-associated pneumonia: incidence, risk factors, outcome, and microbiology. J Cardiothorac Vasc Anesth. 2003;17(1):22–28. doi: 10.1053/jcan.2003.4. [DOI] [PubMed] [Google Scholar]
- 53.Japoni A, Vazin A, Davarpanah MA, Afkhami Ardakani M, Alborzi A, Japoni S, et al. Ventilator-associated pneumonia in Iranian intensive care units. J Infect Dev Ctries. 2011;5(04):286–293. doi: 10.3855/jidc.1212. [DOI] [PubMed] [Google Scholar]
- 54.Parker CM, Kutsogiannis J, Muscedere J, Cook D, Dodek P, Day AG, et al. Ventilator-associated pneumonia caused by multidrug-resistant organisms or Pseudomonas aeruginosa: prevalence, incidence, risk factors, and outcomes. J Crit Care. 2008;23(1):18–26. doi: 10.1016/j.jcrc.2008.02.001. [DOI] [PubMed] [Google Scholar]
- 55.Sundar KM, Nielsen D, Sperry P. Comparison of ventilator-associated pneumonia (VAP) rates between different ICUs: implications of a zero VAP rate. J Crit Care. 2012;27(1):26–32. doi: 10.1016/j.jcrc.2011.05.019. [DOI] [PubMed] [Google Scholar]
- 56.Giantsou E, Liratzopoulos N, Efraimidou E, Panopoulou M, Alepopoulou E, Kartali-Ktenidou S, et al. Both early-onset and late-onset ventilator-associated pneumonia are caused mainly by potentially multiresistant bacteria. Intensive Care Med. 2005;31(11):1488–1494. doi: 10.1007/s00134-005-2697-y. [DOI] [PubMed] [Google Scholar]
- 57.Ibrahim EH, Tracy L, Hill C, Fraser VJ, Kollef MH. The occurrence of ventilator-associated pneumonia in a community hospital. Chest. 2001;120(2):555–561. doi: 10.1378/chest.120.2.555. [DOI] [PubMed] [Google Scholar]
- 58.Rezai MS, Bagheri-Nesami M, Nikkhah A, Bayg AHA. Incidence, risk factors, and outcome of ventilator-associated pneumonia in 18 hospitals of Iran. Int J Adv Biotech Res. 2016;7(3):936–946. [Google Scholar]
- 59.Chouhdari A, Shokouhi S, Bashar FR, Vahedian-Azimi A, Shojaei SP, Fathi M, et al. Is a low incidence rate of ventilation associated pneumonia associated with lower mortality? A descriptive longitudinal study in Iran. Tanaffos. 2018;17(2):110–116. [PMC free article] [PubMed] [Google Scholar]
- 60.Waltrick R, Possamai DS, de Aguiar FP, Dadam M, Souza Filho VJ, de, Ramos LR,, et al. Comparison between a clinical diagnosis method and the surveillance technique of the Center for Disease Control and Prevention for identification of mechanical ventilator-associated pneumonia. Rev Bras Ter Intensiva. 2015;27(3):260–265. doi: 10.5935/0103-507X.20150047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Čiginskienė A, Dambrauskienė A, Rello J, Adukauskienė D. Ventilator-associated pneumonia due to drug-resistant Acinetobacter baumannii: risk factors and mortality relation with resistance profiles, and independent predictors of in-hospital mortality. Medicina (B Aires) 2019;55(2):49. doi: 10.3390/medicina55020049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Pham DT, Nguyen TN, Do Q. Researching the changes of serum procalcitonin levels in ventilator-associated pneumonia patients. Mycobact Dis. 2017;07(03):1000246. doi: 10.4172/2161-1068.1000246. [DOI] [Google Scholar]
- 63.Yagmurdur H, Tezcan AH, Karakurt O, Leblebici F. The efficiency of routine endotracheal aspirate cultures compared to bronchoalveolar lavage cultures in ventilator-associated pneumonia diagnosis. Niger J Clin Pract. 2016;19(1):46–51. doi: 10.4103/1119-3077.164327. [DOI] [PubMed] [Google Scholar]
- 64.Seligman R, Seligman BGS, Konkewicz L, Dos Santos RP. Accuracy of tracheal aspirate gram stain in predicting Staphylococcus aureus infection in ventilator-associated pneumonia. BMC Anesthesiol. 2015;15(1):19. doi: 10.1186/1471-2253-15-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gürgün A, Korkmaz Ekren P, Bacakoğlu F, Başoğlu OK, Dirican N, Aydemir Ş, et al. The role of endotracheal aspiration in the diagnosis of ventilator associated pneumonia. Tuberk Toraks. 2013;61(4):288–294. doi: 10.5578/tt.6610. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this article. De-identified individual subject data may be available from the corresponding author on reasonable.